The Assessment of Sewage Sludge Utilization in Closed-Loop Economy from an Environmental Perspective
Abstract
1. Introduction
2. Heavy Metals in the Human Environment
2.1. Sources of Heavy Metals
2.2. Impact of Wastewater Treatment Technology on the Heavy Metal Content of Sewage Sludge
2.3. Heavy Metal Speciation
2.4. Heavy Metal Accumulation Risk Indicators
2.4.1. Geoaccumulation Index of Heavy Metal in Soil (Igeo)
2.4.2. Nemerow Pollution Index (PINemerow)
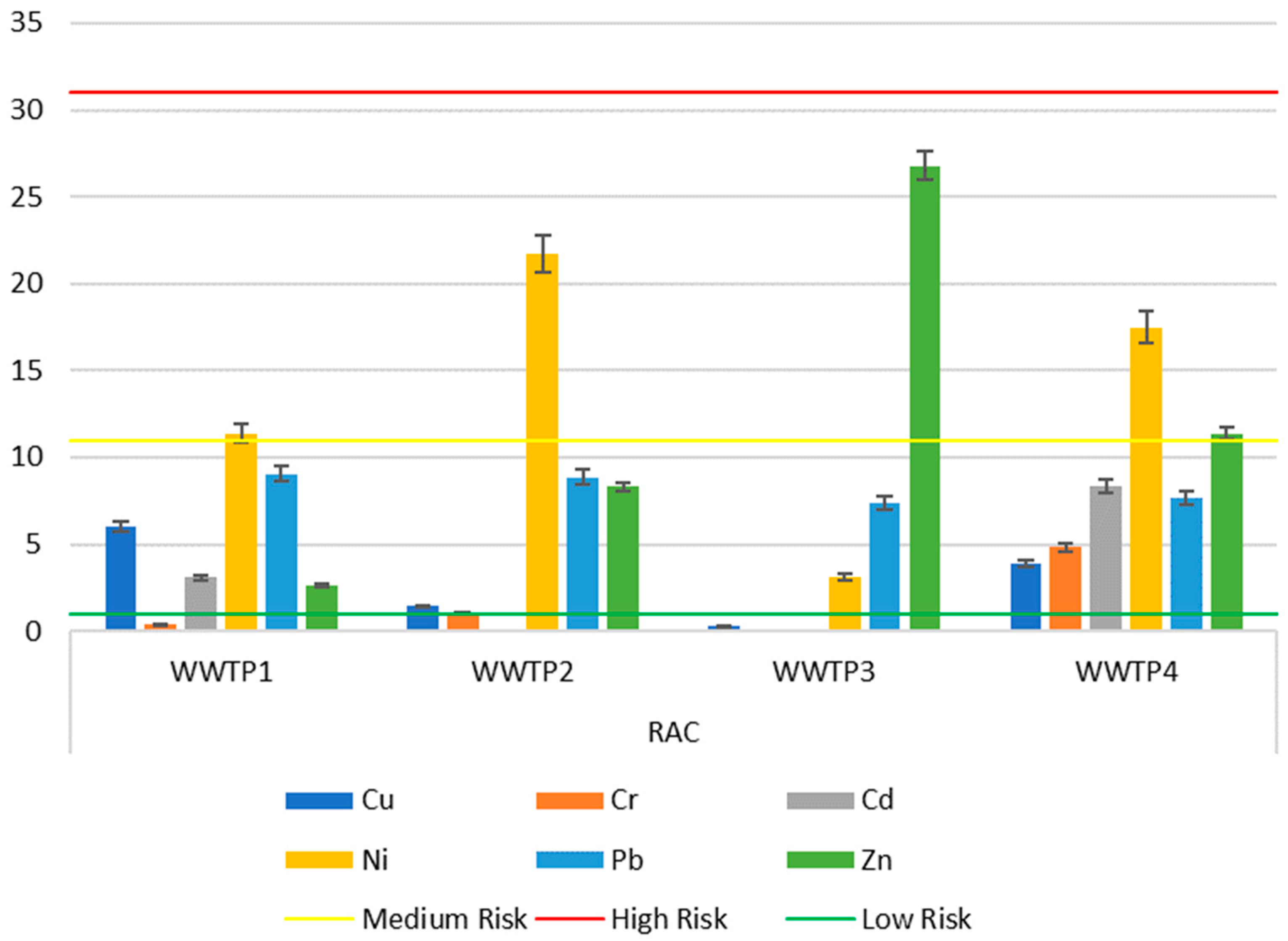
2.4.3. Risk Assessment Code (RAC)
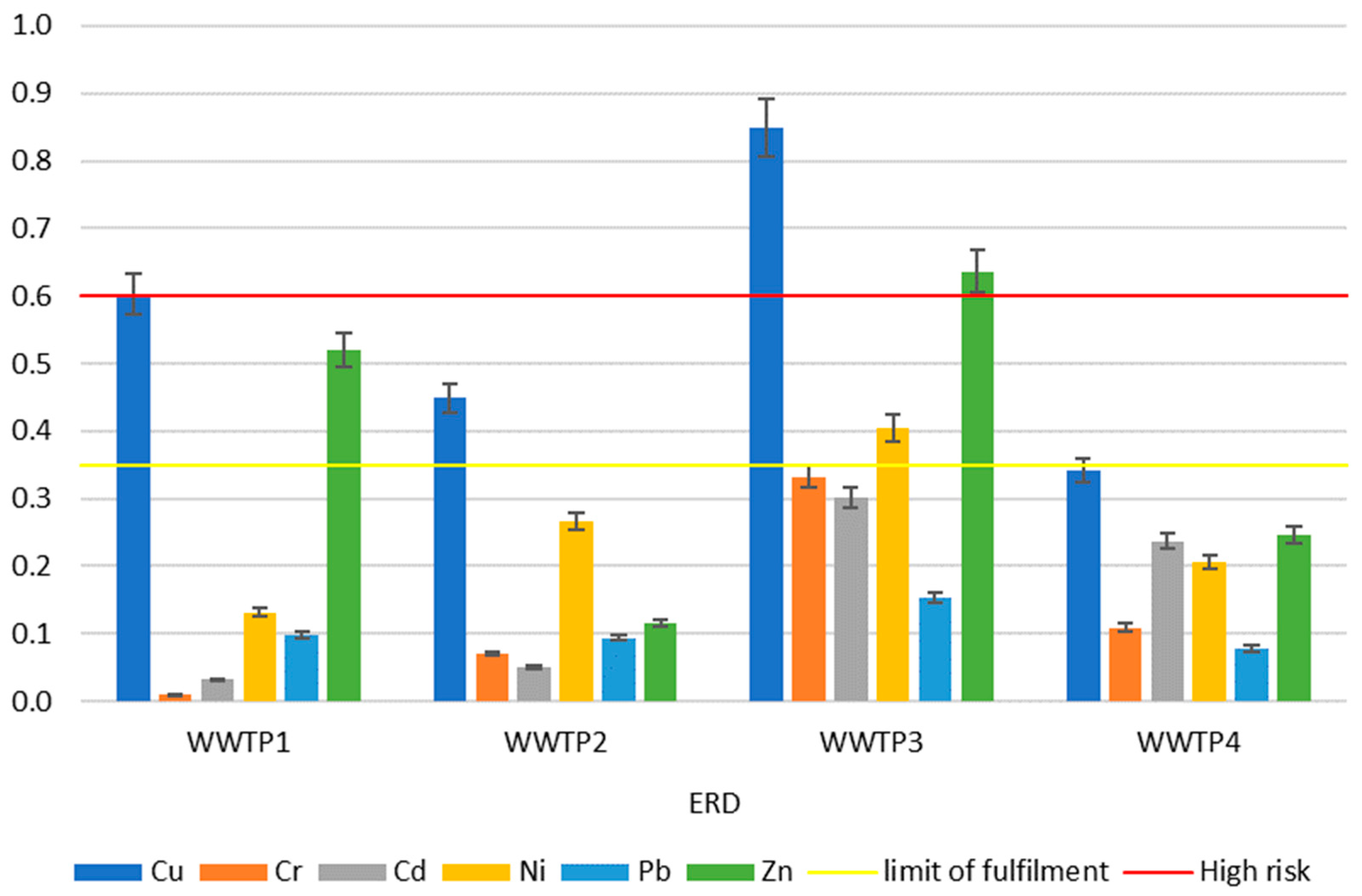
2.4.4. Environmental Risk Determinant (ERD)
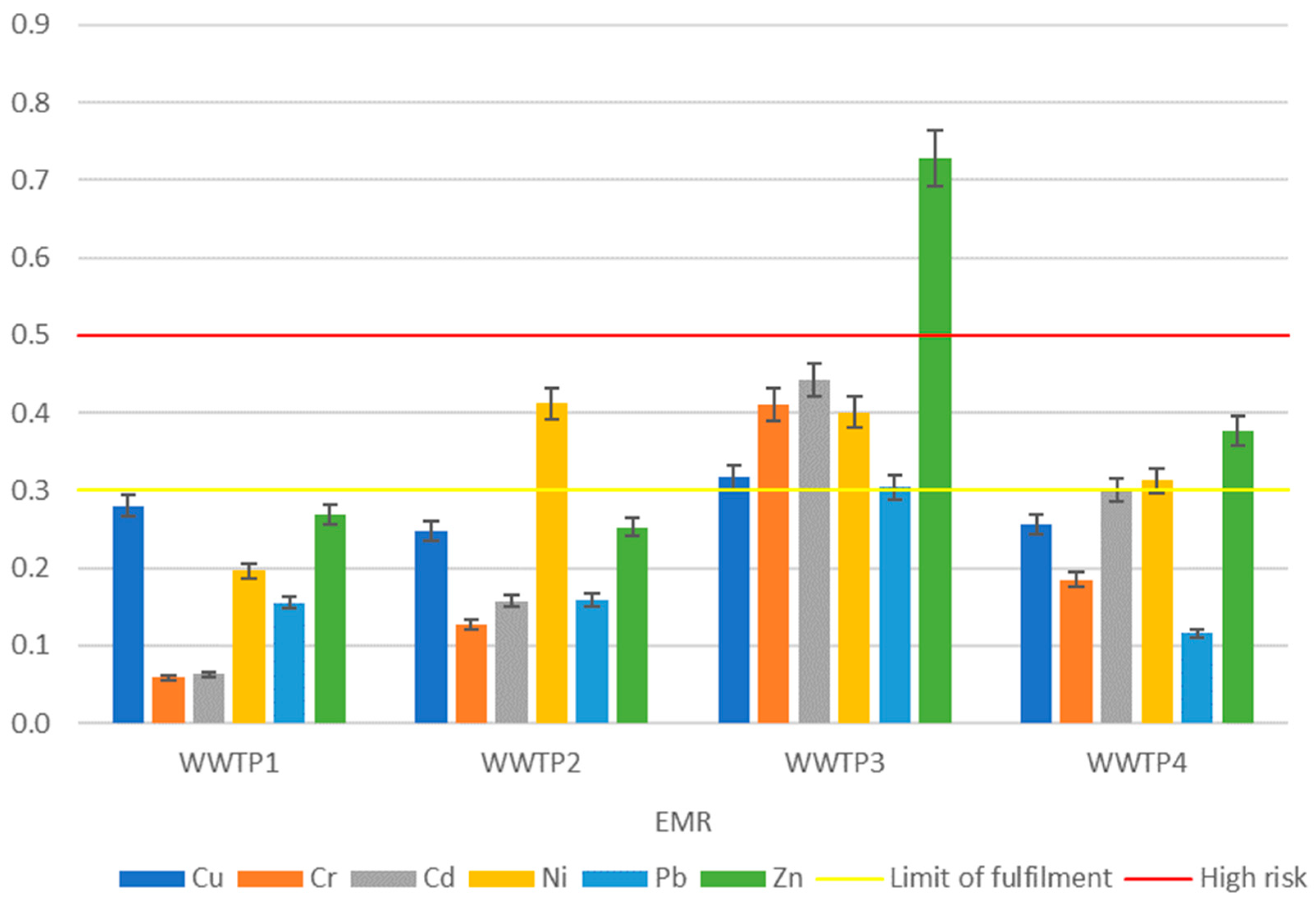
2.4.5. Indicator of Ecological Risk of Metal Mobility (EMR)
3. Results
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Miksch, K.; Sikora, J. Biotechnologia Ścieków; WN PWN: Warszawa, Poland, 2012. [Google Scholar]
- Lasaridi, K.-E.; Manios, T.; Stamatiadis, S.; Chroni, C.; Kyriacou, A. The Evaluation of Hazards to Man and the Environment during the Composting of Sewage Sludge. Sustainability 2018, 10, 2618. [Google Scholar] [CrossRef]
- Jasińska, A. The importance of heavy metal speciation in the aspect of natural management of sewage sludge. Eng. Environ. Prot. 2018, 21, 239–250. [Google Scholar] [CrossRef]
- Latosińska, J. Risk assessment of soil contamination with heavy metals from sewage sludge and ash after its incineration. De-Salin Water Treat 2020, 199, 297–306. [Google Scholar] [CrossRef]
- Ministry of Environment. Strategy for Treatment of Municipal Sewage Sludge in 2019–2022; Ministry of Environment: Warsaw, Poland, 2018; In polish: Strategia postępowania z komunalnymi osadami ściekowymi na lata 2019–2022. Minister Środowiska, 18 listopada 2018 r. Warszawa. Available online: https://www.gov.pl/attachment/2846e2b3-68c7-46eb-b36e-7643e81efd9a (accessed on 6 September 2023). (In Polish)
- Bień, J.; Neczaj, E.; Worwąg, M.; Grosser, A.; Nowak, D.; Milczarek, M.; Janik, M. Directions of sludge management in Poland after 2013. Eng. Environ. Prot. 2011, 14, 375–384. [Google Scholar]
- Sobolewska-Mikulska, K.; Cienciała, A. Legal-Surveying Documentation in the Registration of Rights to Properties of the Treasury and of Territorial Self-government Units–Case Study of Poland. Real Estate Manag. Valuat. 2020, 28, 76–88. [Google Scholar] [CrossRef]
- Minister of the Environment. Regulation of the Minister of the Environment of 6 February 2015 on the Municipal Sewage Sludge (J. L. 2015, No. 0, item. 257); Minister of the Environment: Warsaw, Poland, 2015. [Google Scholar]
- McBride, M.B. Toxic metals in sewage suldge-amended soils: Has promotion of beneficial use discounted the risks? Adv. Environ. Res. 2003, 8, 5–19. [Google Scholar] [CrossRef]
- Maddocks, G.; Lin, C.; McConchie, D. Effect of Bauxsol (TM) and biosolids on soil conditions of acid-generating mine spoil for plant growth. Environ. Pollut. 2004, 127, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Nartowska, E. The effects of potentially toxic metals (copper and zinc) on selected physical and physico-chemical properties of bentonites. Heliyon 2019, 5, e02563. [Google Scholar] [CrossRef]
- Szopińska, K.; Cienciała, A.; Bieda, A.; Kwiecień, J.; Kulesza, Ł.; Parzych, P. Verification of the Perception of the Local Community concerning Air Quality Using ADMS-Roads Modeling. Int. J. Environ. Res. Public Health 2022, 19, 10908. [Google Scholar] [CrossRef]
- Janaszek, A.; Kowalik, R. Analysis of Heavy Metal Contaminants and Mobility in Sewage sludge-soil Mixtures for Sustainable Agricultural Practices. Water 2023, 15, 3992. [Google Scholar] [CrossRef]
- Available online: https://ec.europa.eu/eurostat/databrowser/view/ten00030/default/bar?lang=en (accessed on 6 December 2022).
- European Commission. Council Directive of 12 June 1986 on the Protection of the Environment, and in Particular of the Soil, When Sewage Sludge Is Used in Agriculture (86/278/EEC); European Commission: Brussels, Belgium, 1986. [Google Scholar]
- Ministry of Environmental Protection of the People’s Republic of China. Discharge Standard of Pollutants for Municipal Wastewater Treatment Plant; Ministry of Environmental Protection of the People’s Republic of China: Beijing, China, 2002. [Google Scholar]
- Code of Federal Regulations. Protection of Environment. Chapter I—Environmental Protection Agency (Continued), Subchapter O—Sewage Sludge, Part 503—Standards for The Use Or Disposal Of Sewage Sludge, Subpart B—Land Application, Section 503.13—Pollutant limits. July 1, 2010; Office of the Federal Register (United States): Washington, DC, USA, 2010. [Google Scholar]
- Water Research Commission. Guidelines for the Utilisation and Disposal of Wastewater Sludge. Vol. 2. Requirements for the Agri-cultural Use of Wastewater Sludge; WRC Report no: TT 262/06; WRC: Pretoria, South Africa, 2009. [Google Scholar]
- Government of the Netherlands. Decree on quality and use of other organic fertilisers. In Manual of Environmental Policy; BOOM: Rijswijk, The Netherlands, 2008; Available online: https://wetten.overheid.nl/BWBR0009360/2006-01-01 (accessed on 6 September 2023). (In Dutch)
- The Sludge (Use in Agriculture) Regulations (Northern Ireland) 1990 Implement Council Directive 86/278/EEC on the Protec-tion of Public Health. Available online: https://www.daera-ni.gov.uk/articles/sewage-sludge (accessed on 5 September 2023).
- Government of Malta. Legal Notice 212 of 28 June 2002 on Sludge (Use in Agriculture) Regulations. Available online: http://www.justiceservices.gov.mt/DownloadDocument.aspx?app=lom&itemid=11496&l=1 (accessed on 8 September 2023).
- Bolan, N.; Kunhikrishan, A.; Thangarajan, R.; Kumpiene, J. Remediation of heavy metal(loid)s contaminated soils—To mobilize or to immobilize? J. Hazard. Mater. 2014, 266, 141–166. [Google Scholar] [CrossRef] [PubMed]
- Haraguchi, K.; Hisamichi, Y.; Endo, T. Accumulation and mother-to-calf transfer of anthropogenic and natural organohalo-gens in killer whales (Orcinus orca) stranded on the Pacific coast of Japan. Sci. Total Environ. 2009, 407, 2853–2859. [Google Scholar] [CrossRef] [PubMed]
- Szeląg, B.; Cienciała, A.; Sobura, S.; Studziński, J.; García, J.T. Urbanization and Management of the Catchment Retention in the Aspect of Operation of Storm Overflow: A Probabilistic Approach. Sustainability 2019, 11, 3651. [Google Scholar] [CrossRef]
- Tytła, M.; Widziewicz, K.; Zielewicz, E. Heavy metals and its chemical speciation in sewage sludge at different stages of processing. Environ. Technol. 2016, 37, 899–908. [Google Scholar] [CrossRef] [PubMed]
- Tytła, M.; Widziewicz-Rzońca, K.; Kernert, J.; Bernaś, Z.; Słaby, K. First Comprehensive Analysis of Potential Ecological Risk and Factors Influencing Heavy Metals Binding in Sewage Sludge from WWTPs Using the Ultrasonic Disintegration Process. Water 2023, 15, 666. [Google Scholar] [CrossRef]
- Witkowska, D.; Słowik, J.; Chilicka, K. Heavy Metals and Human Health: Possible Exposure Pathways and the Competition for Protein Binding Sites. Molecules 2021, 26, 6060. [Google Scholar] [CrossRef]
- Pourret, O.; Hursthouse, A. It’s time to replace the term “heavy metals” with “potentially toxic elements” when reporting environmental research. Int. J. Environ. Res. Public Health 2019, 16, 4446. [Google Scholar] [CrossRef]
- Latosińska, J.; Miłek, D.; Komór, A.; Kowalik, R. Selective Collection of Municipal Waste in a Residential District with Multi-Family Buildings—Case Study from Poland. Resources 2021, 10, 83. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, Y.J.; Seo, Y.R. An overview of carcinogenic heavy metal: Molecular toxicity mechanism and prevention. J. Cancer Prev. 2015, 20, 232–240. [Google Scholar] [CrossRef]
- Abd Elnabi, M.K.; Elkaliny, N.E.; Elyazied, M.M.; Azab, S.H.; Elkhalifa, S.A.; Elmasry, S.; Mouhamed, M.S.; Shalamesh, E.M.; Alhorieny, N.A.; Abd Elaty, A.E.; et al. Toxicity of Heavy Metals and Recent Advances in Their Removal: A Review. Toxics 2023, 11, 580. [Google Scholar] [CrossRef]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy Metal Toxicity and the Environment. Exp. Suppl. 2012, 101, 133–164. [Google Scholar]
- Shao, T.; Wang, R.; Xu, Z.; Wei, P.; Zhao, J.; Niu, J.; Song, D. Permeability and Groundwater Enrichment Characteristics of the Loess-Paleosol Sequence in the Southern Chinese Loess Plateau. Water 2020, 12, 870. [Google Scholar] [CrossRef]
- Wojnowska-Heciak, M.; Heciak, J.; Kłak, A. Concrete Paving Slabs for Comfort of Movement of Mobility-Impaired Pedestrians—A Survey. Int. J. Environ. Res. Public Health 2022, 19, 3183. [Google Scholar] [CrossRef]
- Laonamsai, J.; Pawana, V.; Chipthamlong, P.; Chomcheawchan, P.; Kamdee, K.; Kimmany, B.; Jul-phunthong, P. Groundwater Quality Variations in Multiple Aquifers: A Comprehensive Evaluation for Public Health and Agricultural Use. Geosciences 2023, 13, 195. [Google Scholar] [CrossRef]
- Arya, S.; Kumar, V.; Sharma, S. Analysis of water quality parameters of groundwater in and around Diamond Cement Industry, Jhansi, Central India. Int. J. Curr. Res. 2012, 4, 75–77. [Google Scholar]
- Rajakaruna, R.M.A.S.D.; Sewwandi, B.G.N.; Najim, M.M.M.; Baig, M.B.; Alotaibi, B.A.; Traore, A. Sus-tainable Approaches for Wastewater Treatment: An Analysis of Sludge-Based Materials for Heavy Metal Removal from Wastewater by Adsorption. Sustainability 2023, 15, 14937. [Google Scholar] [CrossRef]
- Hegazi, H.A. Removal of Heavy Metals from Wastewater Using Agricultural and Industrial Wastes as Adsorbents. HBRC J. 2013, 9, 276–282. [Google Scholar] [CrossRef]
- Rathi, B.S.; Kumar, P.S. Application of Adsorption Process for Effective Removal of Emerging Contami-nants from Water and Wastewater. Environ. Pollut. 2021, 280, 116995. [Google Scholar] [CrossRef]
- Soliman, N.K.; Moustafa, A.F. Industrial Solid Waste for Heavy Metals Adsorption Features and Chal-lenges; a Review. J. Mater. Res. Technol. 2020, 9, 10235–10253. [Google Scholar] [CrossRef]
- Ahmaruzzaman, M. Industrial Wastes as Low-Cost Potential Adsorbents for the Treatment of Wastewater Laden with Heavy Metals. Adv. Colloid Interface Sci. 2011, 166, 36–59. [Google Scholar] [CrossRef]
- Kowalik, R.; Gawdzik, J.; Bąk-Patyna, P.; Ramiączek, P.; Jurišević, N. Risk Analysis of Heavy Metals Migration from Sewage Sludge of Wastewater Treatment Plants. Int. J. Environ. Res. Public Health 2022, 19, 11829. [Google Scholar] [CrossRef]
- Kowalik, R.; Latosińska, J.; Metryka-Telka, M.; Porowski, R.; Gawdzik, J. Comparison of the Possibilities of Environmental Usage of Sewage Sludge from Treatment Plants Operating with MBR and SBR Technology. Membranes 2021, 11, 722. [Google Scholar] [CrossRef]
- Jeske, A.; Gworek, B. Review of methods for determination of bioavailability and mobility of heavy metals in soils. Environ. Nat. Resour. Prot. 2011, 49, 209–218. [Google Scholar]
- Szumska, M.; Gworek, B. Methods for the determination of heavy metal fractions in sewage sludge. Environ. Prot. Nat. Resour. 2009, 41, 42–63. [Google Scholar]
- Zimmerman, A.J.; Weindorf, D.C. Heavy metal and trace metal analysis in soil by sequential extraction: A review of procedures. Int. J. Anal. Chem. 2010, 2010, 387803. [Google Scholar] [CrossRef] [PubMed]
- Kabala, C. Comparison of single and sequential iron extraction in podzolic soils of the Sudetes. Soil Sci. Yearb. Suppl. 2001, 52, 191–197. [Google Scholar]
- Muller, G. Index of geoaccumulation in sediments of the Rhine River. GeoJournal 1969, 2, 108–118. [Google Scholar]
- Hakanson, L. An ecological risk index for aquatic pollution control: A sedimentological approach. Water Res. 1980, 14, 975–1001. [Google Scholar] [CrossRef]
- Karaouzas, I.; Kapetanaki, N.; Mentzafou, A.; Kanellopoulos, T.D.; Skoulikidis, N. Heavy metal contamination status in Greek surface waters: A review with application and evaluation of pollution indices. Chemosphere 2021, 263, 128192. [Google Scholar] [CrossRef]
- Gong, Q.; Deng, J.; Xiang, Y.; Wang, Q.; Yang, L. Calculating pollution indices by heavy metals in eco-logical geochemistry assessment and a case study in parks of Beijing. J. Earth Sci. 2008, 19, 230–241. [Google Scholar]
- Varol, M. Assessment of heavy metal contamination in sediments of the Tigris River (Turkey) using pollution indices and multivariate statistical techniques. J. Hazard. Mater. 2011, 195, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Inengite, A.K.; Abasi, C.Y.; Walter, C. Application of pollution indices for the assessment of heavy metal pollution in flood impacted soil. Int. Res. J. Pure Appl. Chem. 2015, 8, 175–189. [Google Scholar] [CrossRef]
- Perin, G.; Craboledda, L.; Lucchese, M.; Cirillo, R.; Dotta, L.; Zanetta, M.L.; Oro, A.A. Heavy metal speciation in the sediments of northern Adriatic Sea. A new approach for environmental toxicity determination. In Heavy Metals in the Environment; Lakkas, T.D., Ed.; CEP Consultants: Edinburgh, UK, 1985; Volume 2, pp. 454–456. [Google Scholar]
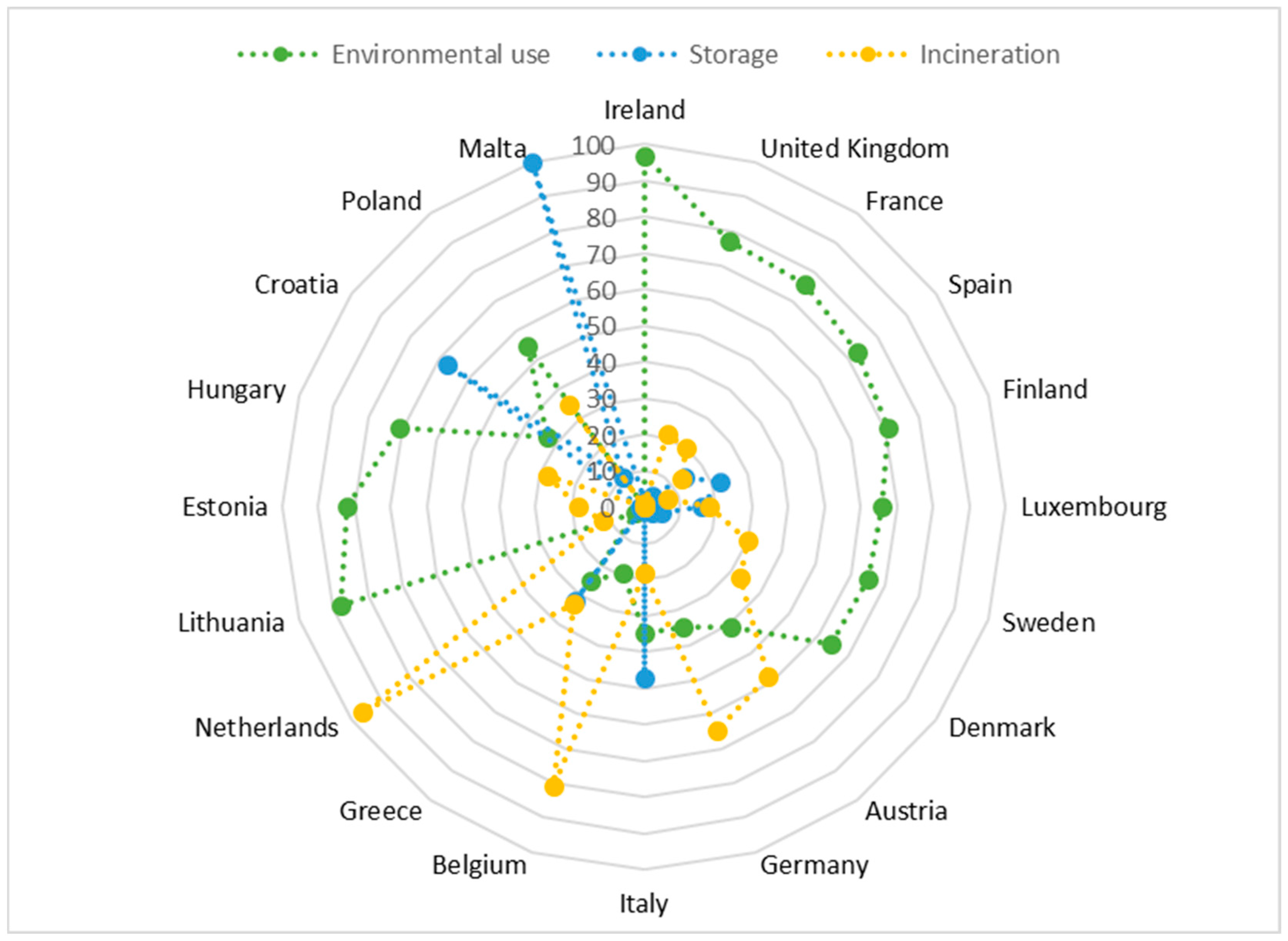
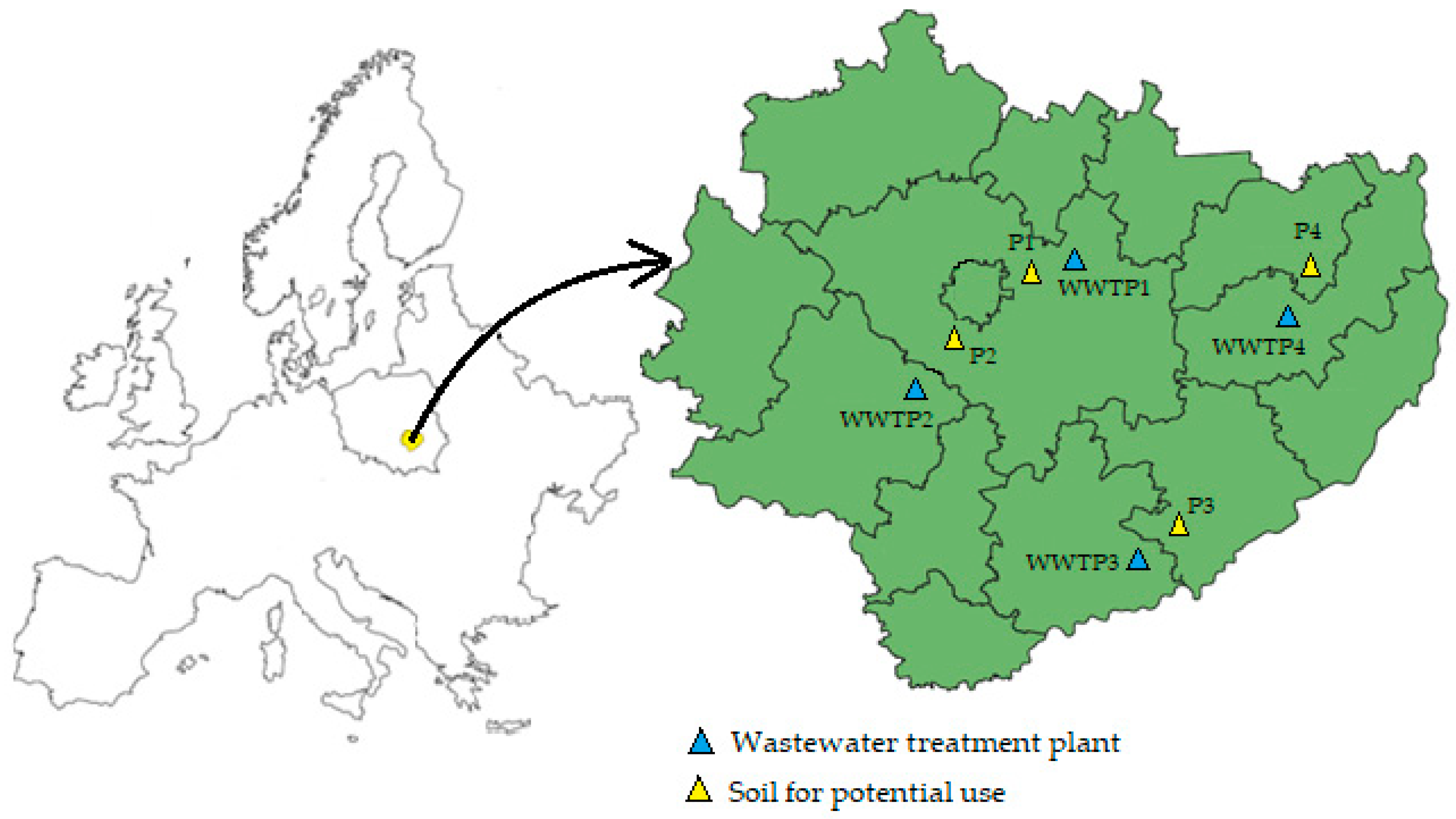

| Heavy Metal | The Permissible Levels of Heavy Metal Content in Sewage Sludge Intended for Natural Use | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Poland [8] | EU [15] | Chinese [16] | United States [17] | South African [18] | Netherlands [19] | Ireland [20] | Malta [21] | ||
| pH < 6.5 | pH > 6.5 | ||||||||
| Cd | 20 | 20–40 | 5 | 20 | 39 | 40 | 1.25 | 20 | 5 |
| Ni | 300 | 300–400 | 100 | 200 | 420 | 420 | 30 | 300 | 200 |
| Cr | 500 | - | 600 | 1000 | - | 1200 | 75 | - | 800 |
| Pb | 750 | 750–1200 | 300 | 1000 | 300 | 300 | 100 | 750 | 500 |
| Cu | 1000 | 1000–1750 | 250 | 500 | 1500 | 1500 | 75 | 1000 | 800 |
| Zn | 2500 | 2500–4000 | 500 | 1000 | 2800 | 2800 | 300 | 2500 | 2000 |
| Object | WWTP1 | WWTP2 | WWTP3 | WWTP4 |
|---|---|---|---|---|
| Location | Swieta Katarzyna | Sobkow | Pacanow | Opatow |
| Wastewater treatment plant type | MBR | SBR | EvU-Perl | Activated sludge |
| Form of sludge treatment | Oxygen stabilization of sludge | System Draimad | Oxygen stabilization of sludge | Fermentation |
| Equivalent number of inhabitants p.e. * | 2626 | 3725 | 1446 | 15,355 |
| Location | Distance from WWTP | Soil Taxonomy | pH (KCl) | Organic Matter Content | Organic Carbon | Nitrogen Total | C/N Ratio | Absorbable Phosphorus | Calcium Content | Soil Sorption Capacity |
|---|---|---|---|---|---|---|---|---|---|---|
| - | km | - | pH | % | % | % | - | mg P2O5 × 100 g−1 | Cmol × kg−1 | Cmol × kg−1 |
| Wola Kopcowa—P1 | 15.2 | Spodosols | 5.8 | 3.28 | 1.83 | 0.08 | 20.84 | 6 | 1.3 | 6.83 |
| Dyminy—P2 | 25.4 | Spodosols | 6.5 | 3.19 | 1.86 | 0.13 | 15.43 | 1.7 | 5.2 | 11.82 |
| Niedzialki—P3 | 17.3 | Inceptisols | 7.6 | 0.76 | 0.52 | 0.07 | 4.15 | 10 | 0.13 | 45.31 |
| Cmielow—P4 | 13.8 | Dystric Eutrudepts | 5.3 | 2.64 | 1.54 | 0.14 | 10.5 | 5.3 | 5.1 | 10.12 |
| Heavy Metal | Sources of Metals in Sewage Sludge |
|---|---|
| Cadmium (Cd) | Electroplating plants, production of dyes, batteries, accumulators, paints and plastics, polymer stabilizers, chemical industry, production of plant protection products, graphic arts, printing industries |
| Lead (Pb) | Production of dyes, batteries, fertilizers, automotive, energy industry, plant protection products, electrochemical |
| Chromium (Cr) | Electroplating industry, tanning industry, wood impregnation, textile, dye and plastic production, printing and graphic arts industries |
| Copper (Cu) | Metallurgical industry, dye industry, textile industry, production of plant protection products and fertilizers |
| Mercury (Hg) | Production of batteries, phosphoric acid, caustic soda, pulp mills, production of plant protection products and mercury, metallic mercury |
| Nickel (Ni) | Electroplating industry, paper industry, refineries, steel plants, fertilizer plants |
| Zinc (Zn) | Battery manufacture, paints, textile industry, plastics, polymer stabilizers, printing and graphic arts industries, printing and graphic arts |
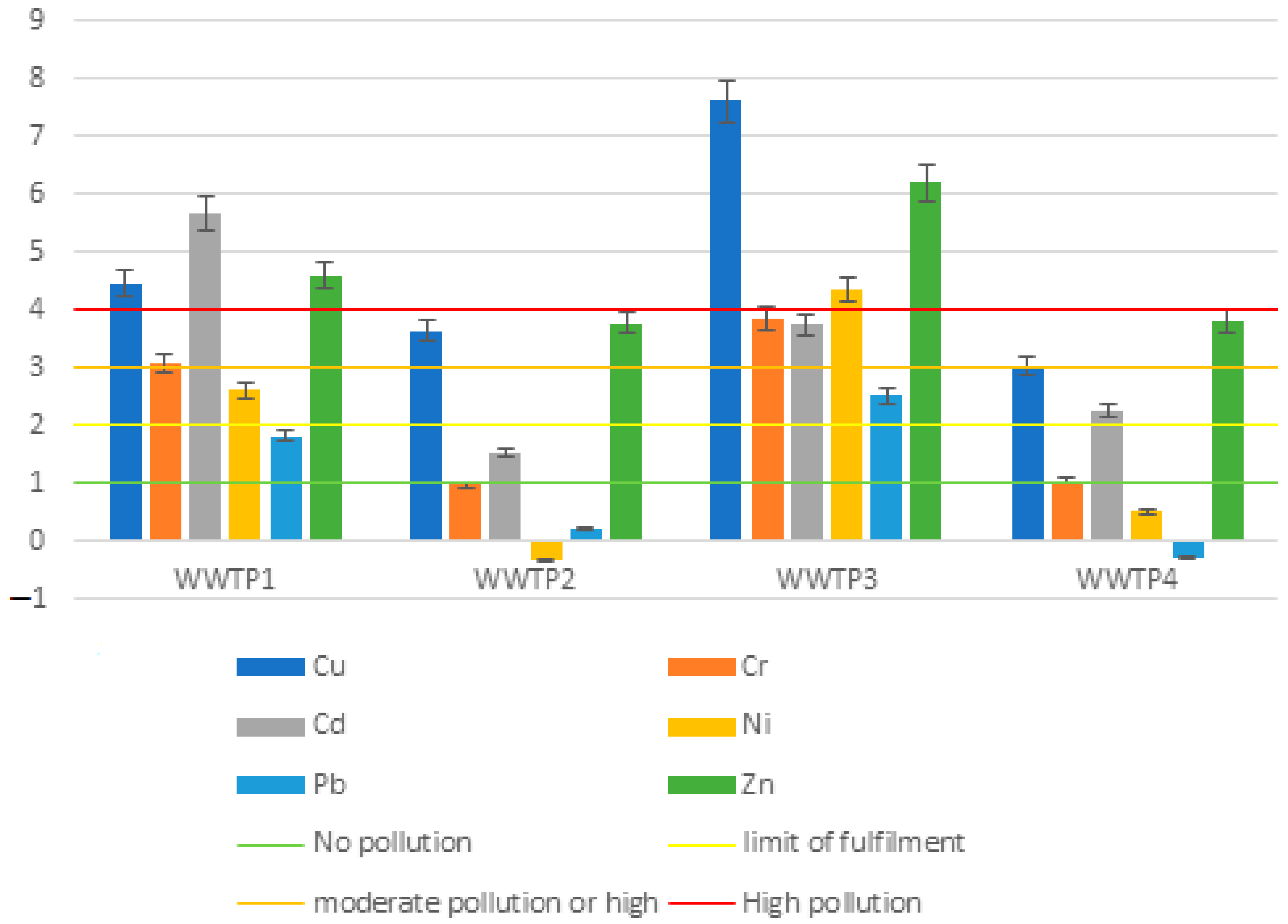
| Igeo | Contamination Value |
|---|---|
| Igeo ≤ 0 | Lack of pollution |
| 0 < Igeo ≤ 1 | Low pollution |
| 1 < Igeo ≤ 3 | Average pollution |
| 3 < Igeo ≤ 5 | High pollution |
| 5 < Igeo | Extreme pollution |
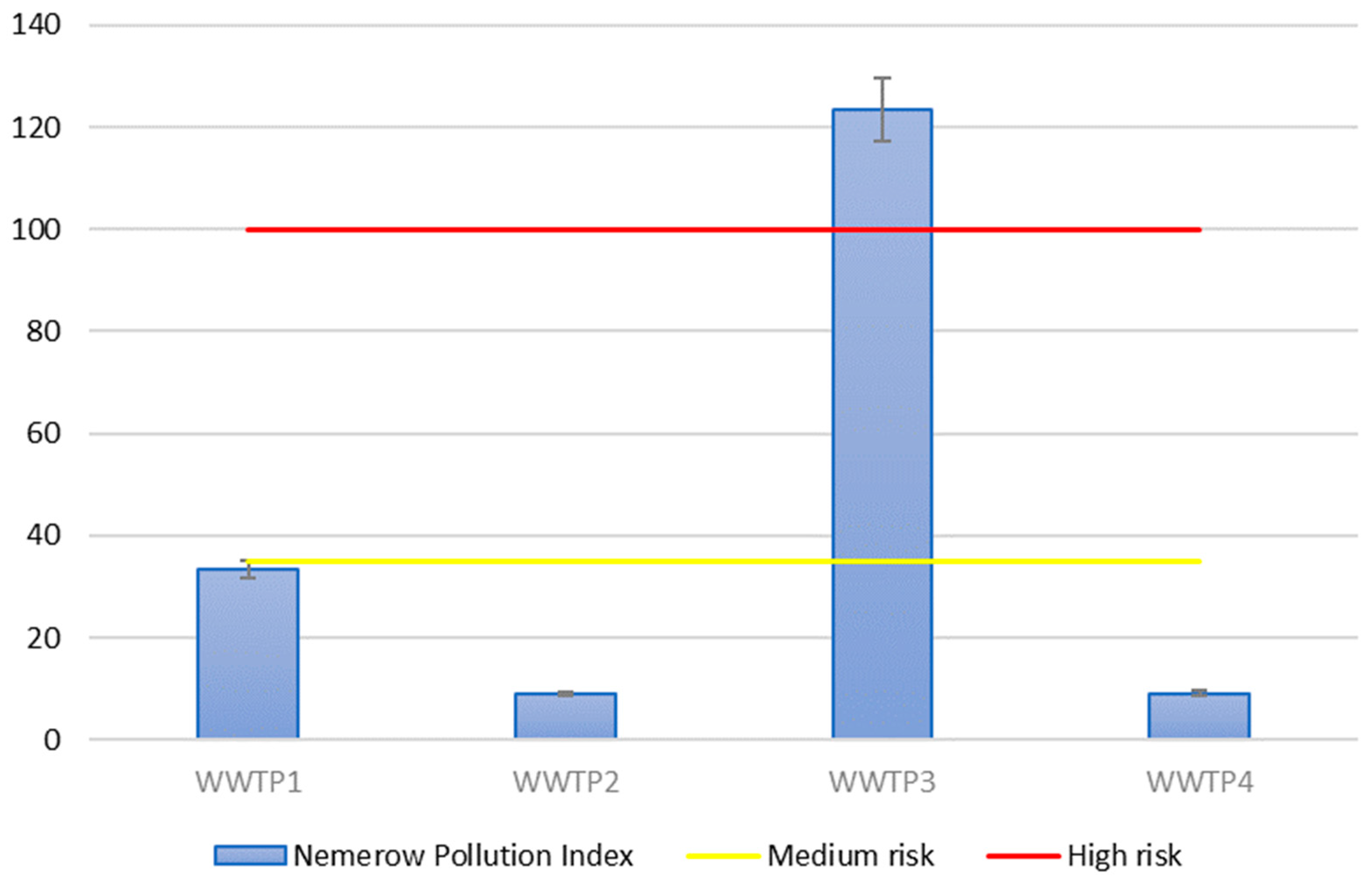
| Nemerow Pollution Index (PINemerow) | Pollution Value |
|---|---|
| 0 < PINemerow ≤ 35 | low risk |
| 35 < PINemerow ≤ 100 | medium risk |
| 100 < PINemerow | High risk |
| EMR | ERD | Risk Value |
|---|---|---|
| 0 < EMR ≤ 0.3 | 0 < ERD ≤ 0.35 | low risk |
| 0.3 < EMR ≤ 0.5 | 0.35 < ERD ≤ 0.6 | medium risk |
| 0.5 < EMR | 0.6 < ERD | High risk |
| Heavy Metal Content [mg/kg s.m.] | ||||||
|---|---|---|---|---|---|---|
| Fraction | Cu | Cr | Cd | Ni | Pb | Zn |
| Sewage sludge—Ss1 | ||||||
| F I | 7.51 ± 0.1 | 0.42 ± 0.1 | 1.22 ± 0.1 | 4.61 ± 0.1 | 8.03 ± 0.4 | 26.28 ± 0.6 |
| F II | 0.52 ± 0.1 | 0.21 ± 0.1 | 0.21 ± 0.1 | 0.33 ± 0.1 | 0.81 ± 0.1 | 8.44 ± 0.3 |
| F III | 101.95 ± 5.5 | 17.83 ± 0.1 | 3.54 ± 0.1 | 10.31 ± 0.1 | 17.02 ± 0.6 | 795.91 ± 23 |
| F IV | 15.11 ± 0.6 | 82.21 ± 0.6 | 33.56 ± 0.1 | 25.01 ± 0.2 | 62.24 ± 0.9 | 176.13 ± 8 |
| ΣFI ÷ IV | 125.03 ± 5.5 | 100.62 ± 0.6 | 38.42 ± 0.2 | 40.21 ± 0.3 | 88.02 ± 1.2 | 1006.61 ± 24.4 |
| Sewage sludge—Ss2 | ||||||
| F I | 1.52 ± 0.1 | 0.31 ± 0.2 | 0.00 ± 0.1 | 2.01 ± 0.1 | 5.71 ± 0.5 | 111.61 ± 2.0 |
| F II | 1.01 ± 0.1 | 0.01 ± 0.0 | 0.24 ± 0.1 | 1.45 ± 0.1 | 4.62 ± 0.6 | 215.24 ± 3.5 |
| F III | 79.54 ± 0.4 | 11.23 ± 0.2 | 1.06 ± 0.1 | 2.74 ± 0.1 | 4.33 ± 0.2 | 556.65 ± 4.8 |
| F IV | 23.03 ± 0.2 | 17.18 ± 0.4 | 1.54 ± 0.2 | 3.13 ± 0.1 | 49.84 ± 0.5 | 457.93 ± 4.0 |
| ΣFI ÷ IV | 105.04 ± 0.3 | 28.64 ± 0.5 | 2.81 ± 0.3 | 9.25 ± 0.2 | 64.41 ± 1.3 | 1341.22 ± 7.4 |
| Sewage sludge—Ss3 | ||||||
| F I | 1.51 ± 0.1 | 0.04 ± 0.1 | 0.01 ± 0.1 | 2.63 ± 0.1 | 3.77 ± 0.4 | 328.91 ± 0.9 |
| F II | 25.63 ± 0.2 | 24.13 ± 0.2 | 4.24 ± 0.1 | 19.62 ± 0.3 | 14.04 ± 2.3 | 743.25 ± 2.3 |
| F III | 551.44 ± 0.9 | 45.11 ± 0.3 | 5.15 ± 0.1 | 57.01 ± 0.6 | 6.01 ± 0.7 | 152.32 ± 0.9 |
| F IV | 4.72 ± 0.1 | 4.73 ± 0.1 | 0.83 ± 0.1 | 4.38 ± 0.2 | 26.74 ± 3.1 | 3.14 ± 0.1 |
| ΣFI ÷ IV | 583.31 ± 0.9 | 74.03 ± 0.6 | 10.11 ± 0.2 | 83.53 ± 0.7 | 50.23 ± 3.6 | 1228.01 ± 2.6 |
| Sewage sludge—Ss4 | ||||||
| F I | 3.36 ± 0.2 | 2.01 ± 0.3 | 0.32 ± 0.1 | 3.51 ± 0.1 | 5.21 ± 0.3 | 79.44 ± 0.9 |
| F II | 1.82 ± 0.1 | 1.15 ± 0.2 | 0.37 ± 0.1 | 1.44 ± 0.4 | 0.57 ± 0.3 | 122.82 ± 1.3 |
| F III | 57.13 ± 1.5 | 16.13 ± 0.4 | 1.92 ± 0.2 | 5.96 ± 0.2 | 7.82 ± 0.8 | 323.82 ± 3.1 |
| F IV | 22.84 ± 0.8 | 22.06 ± 0.6 | 1.13 ± 0.6 | 9.22 ± 0.2 | 54.74 ± 8.5 | 170.81 ± 3.7 |
| ΣFI ÷ IV | 85.02 ± 1.7 | 41.27± 0.8 | 3.61± 0.6 | 20.01 ± 0.3 | 68.22 ± 8.2 | 696.85 ± 4.3 |
| Soil—s1 | ||||||
| ΣHM | 3.81 ± 0.1 | 8.01 ± 0.2 | 0.5 ± 0.1 | 4.39 ± 0.1 | 16.73 ± 0.2 | 27.71 ± 0.3 |
| Soil—s2 | ||||||
| ΣHM | 5.59 ± 0.1 | 9.67 ± 0.3 | 0.64 ± 0.1 | 7.77 ± 0.2 | 36.52 ± 0.3 | 65.01 ± 0.3 |
| Soil—s3 | ||||||
| ΣHM | 2.01 ± 0.1 | 3.41 ± 0.1 | 0.55 ± 0.1 | 2.74 ± 0.1 | 5.85 ± 0.1 | 11.22 ± 0.2 |
| Soil—s4 | ||||||
| ΣHM | 6.84 ± 0.2 | 13.3 ± 0.2 | 0.5 ± 0.1 | 9.32 ± 0.1 | 54.4 ± 0.3 | 33.31 ± 0.2 |
| WWTP | Igeo | RAC | ERD | EMR |
|---|---|---|---|---|
| 1 | Cu, Zn, Cd, Cr | Ni | Cu, Zn | — |
| 2 | Cu, Zn | Ni | Cu | Ni |
| 3 | Cr, Cu, Cd, Zn, Ni | Zn | Cu, Ni, Zn | Cu, Cr, Cd, Ni, Zn |
| 4 | Cu, Zn | Ni, Zn | — | Ni, Zn |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janaszek, A.; Silva, A.F.d.; Jurišević, N.; Kanuchova, M.; Kozáková, Ľ.; Kowalik, R. The Assessment of Sewage Sludge Utilization in Closed-Loop Economy from an Environmental Perspective. Water 2024, 16, 383. https://doi.org/10.3390/w16030383
Janaszek A, Silva AFd, Jurišević N, Kanuchova M, Kozáková Ľ, Kowalik R. The Assessment of Sewage Sludge Utilization in Closed-Loop Economy from an Environmental Perspective. Water. 2024; 16(3):383. https://doi.org/10.3390/w16030383
Chicago/Turabian StyleJanaszek, Agata, Alessandra Furtado da Silva, Nebojša Jurišević, Maria Kanuchova, Ľubica Kozáková, and Robert Kowalik. 2024. "The Assessment of Sewage Sludge Utilization in Closed-Loop Economy from an Environmental Perspective" Water 16, no. 3: 383. https://doi.org/10.3390/w16030383
APA StyleJanaszek, A., Silva, A. F. d., Jurišević, N., Kanuchova, M., Kozáková, Ľ., & Kowalik, R. (2024). The Assessment of Sewage Sludge Utilization in Closed-Loop Economy from an Environmental Perspective. Water, 16(3), 383. https://doi.org/10.3390/w16030383







