Review on Zinc Isotopes in River Systems: Distribution and Application
Abstract
:1. Introduction
2. Zn Isotope Measuring
2.1. Sample Pretreatment
2.2. Chemical Separation and Purification
2.3. Mass Spectrometry
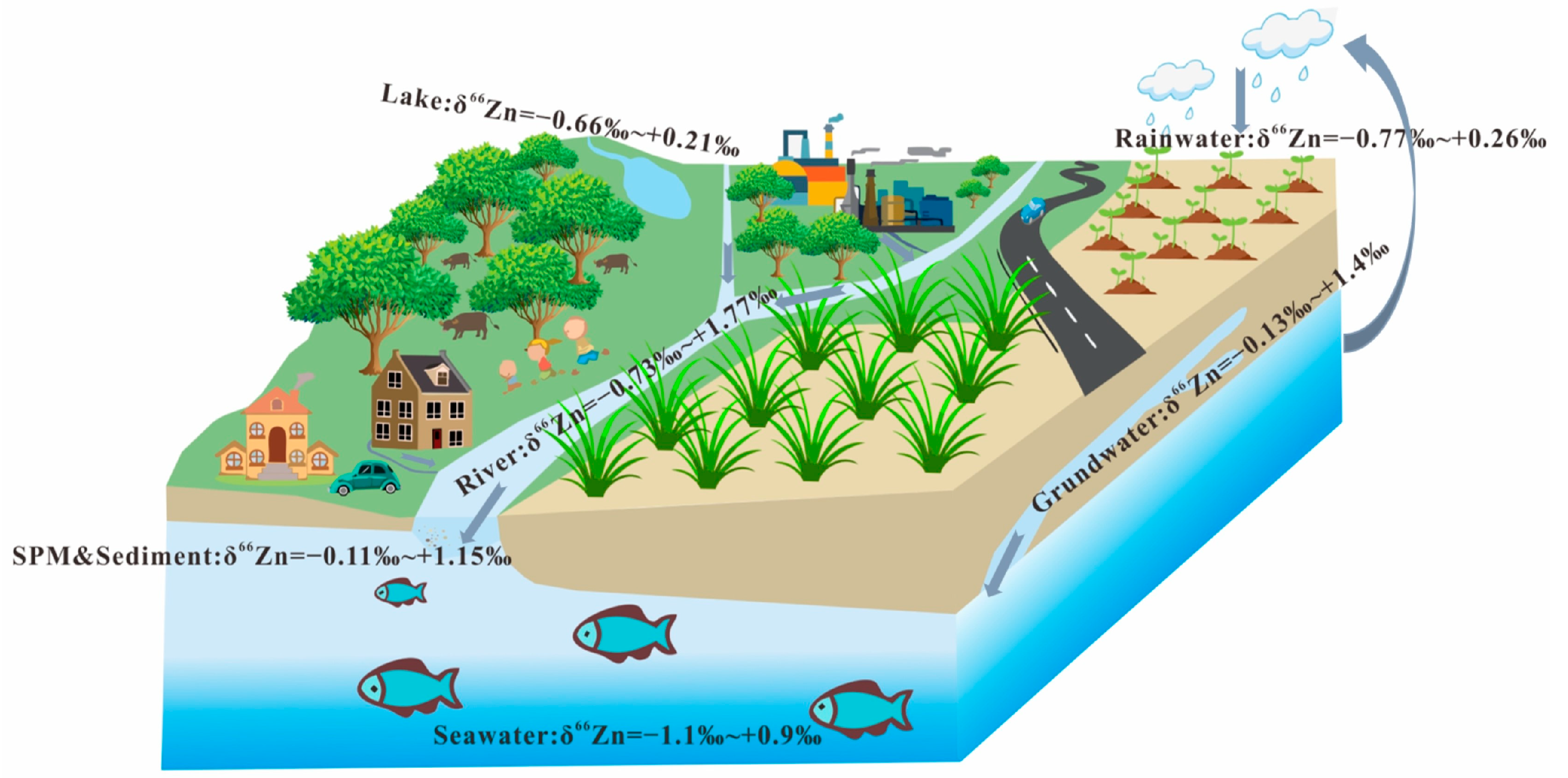
3. Distribution of Zn Isotopes and the Controlling Factors
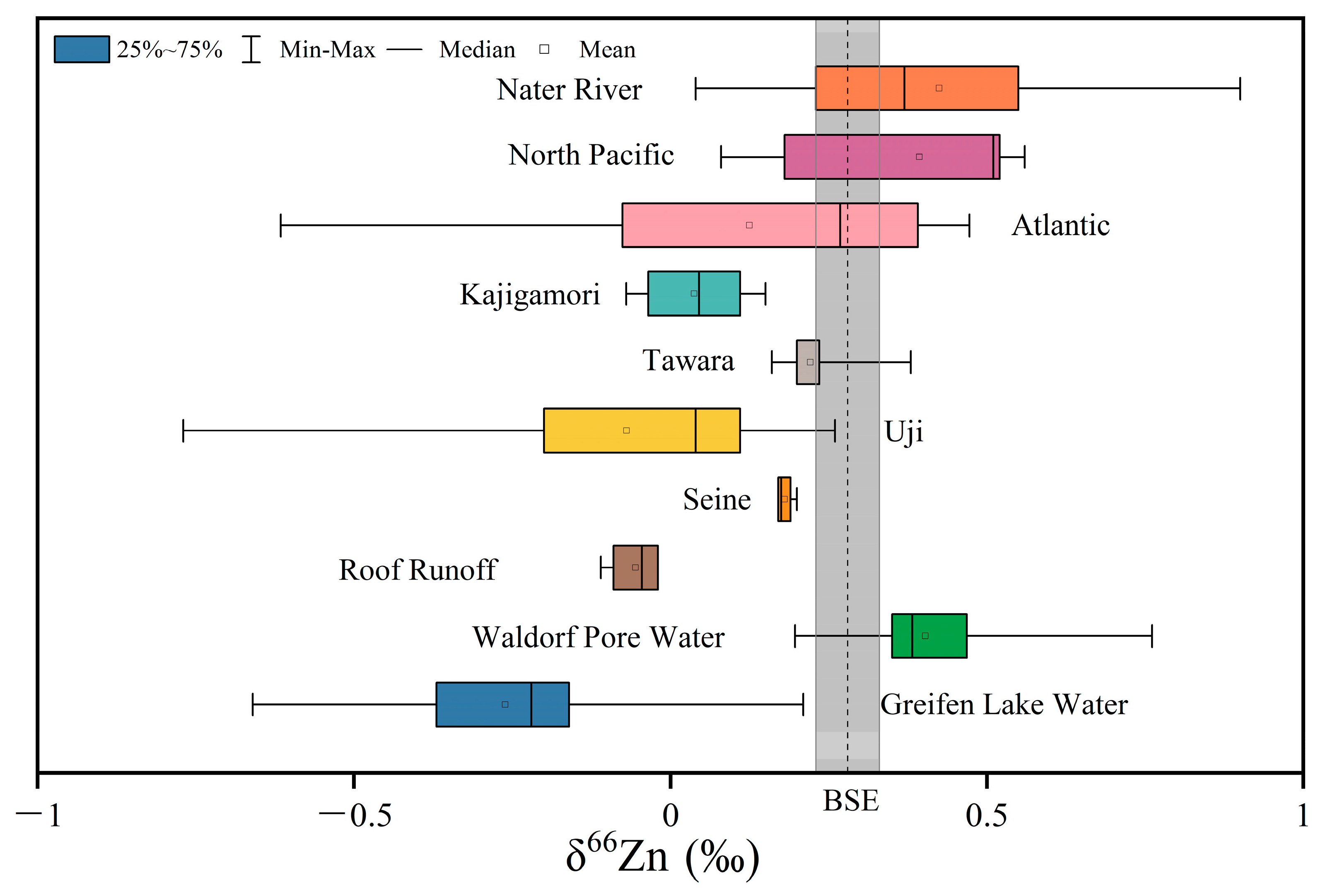
3.1. Zn Isotope Compositions in Water Systems
3.1.1. Rivers
3.1.2. Seawater
3.1.3. Rainwater, Pore Water, and Groundwater
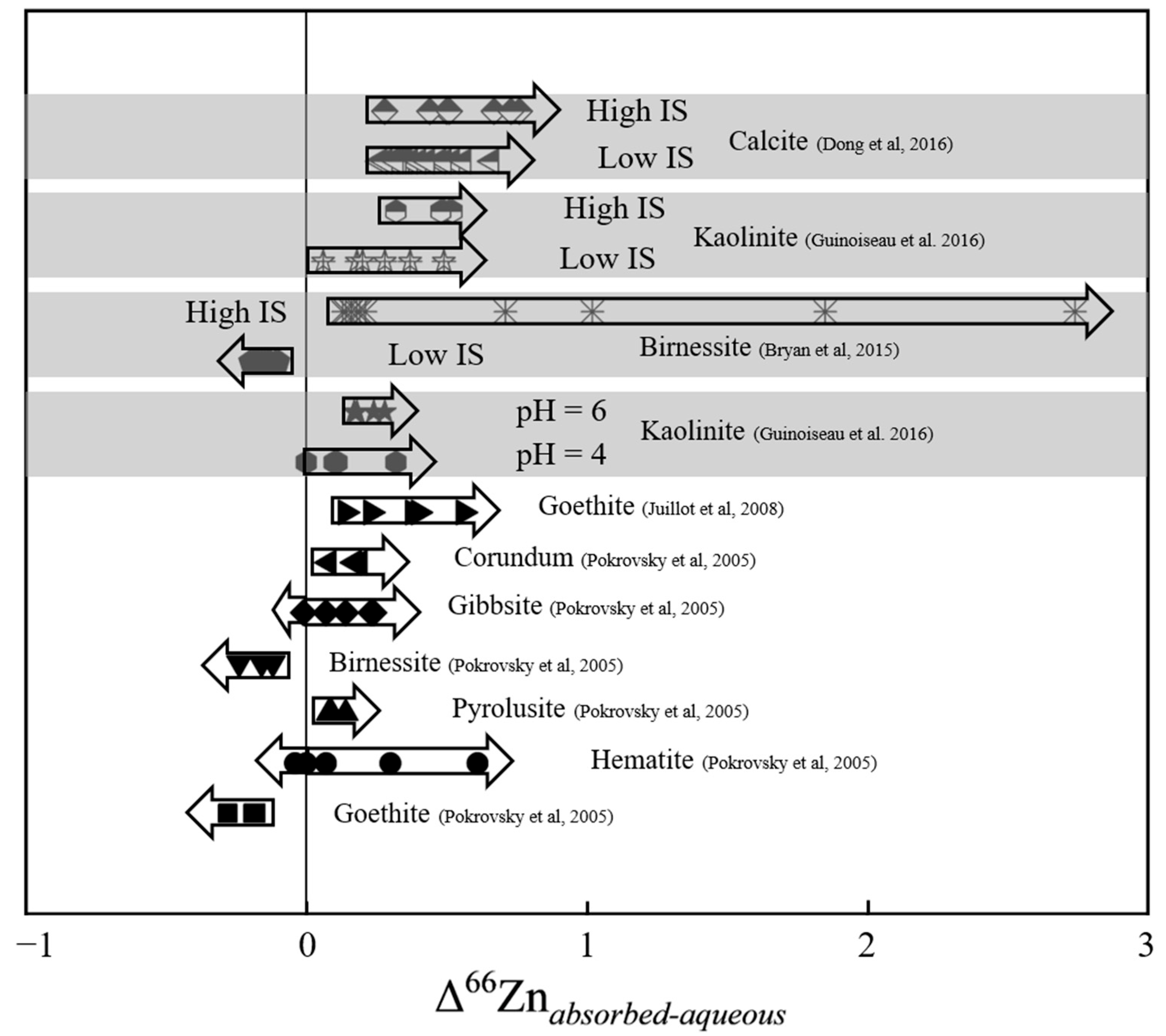
3.2. Factors Controlling Zn Isotopes’ Fractionation
3.2.1. Mineral Adsorption
3.2.2. Precipitation
3.2.3. Biological Effects
4. Application of Zn Isotopes to Source Identification in Rivers
5. Conclusions and Perspectives
- (1)
- Future research can focus on improving Zn isotope analysis methods to enhance analysis efficiency, accuracy, and stability while reducing costs and workload.
- (2)
- Single isotopes often only identify certain important sources while neglecting other potential pollution sources. Combining the isotope analysis of other elements such as lead and Cu enables multi-element isotope tracing, providing a more comprehensive understanding of pollution sources and processes.
- (3)
- Further studies on the mechanism of Zn isotopic fractionation between minerals are required. This research can provide accurate information for understanding pollution sources in rivers.
- (4)
- The current research on the seasonal variations of Zn isotopes in rivers is limited. Establishing comprehensive databases through long-term monitoring of Zn isotopes can reveal seasonal and interannual variations in river systems, particularly in the regulation of biological and mineral processes by seasonal changes in Zn isotopes, deepening our understanding of the impact of human activities on river environments.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, P.; Hu, J.; Liu, T.; Liu, J.; Ma, S.; Ma, W.; Li, J.; Zheng, H.; Lu, R. Advances in the application of metallic isotopes to the identification of contaminant sources in environmental geochemistry. J. Hazard. Mater. 2023, 458, 131913. [Google Scholar] [CrossRef] [PubMed]
- Desaulty, A.-M.; Petelet-Giraud, E. Zinc isotope composition as a tool for tracing sources and fate of metal contaminants in rivers. Sci. Total Environ. 2020, 728, 138599. [Google Scholar] [CrossRef] [PubMed]
- Cloquet, C.; Carignan, J.; Lehmann, M.F.; Vanhaecke, F. Variation in the isotopic composition of zinc in the natural environment and the use of zinc isotopes in biogeosciences: A review. Anal. Bioanal. Chem. 2008, 390, 451–463. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Gaillardet, J.; Louvat, P. Zinc Isotopes in the Seine River Waters, France: A Probe of Anthropogenic Contamination. Environ. Sci. Technol. 2008, 42, 6494–6501. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Gaillardet, J.; Louvat, P.; Huon, S. Zn isotopes in the suspended load of the Seine River, France: Isotopic variations and source determination. Geochim. Cosmochim. Acta 2009, 73, 4060–4076. [Google Scholar] [CrossRef]
- Araújo, D.F.; Boaventura, G.R.; Machado, W.; Viers, J.; Weiss, D.; Patchineelam, S.R.; Ruiz, I.; Rodrigues, A.P.C.; Babinski, M.; Dantas, E. Tracing of anthropogenic zinc sources in coastal environments using stable isotope composition. Chem. Geol. 2017, 449, 226–235. [Google Scholar] [CrossRef]
- Nriagu, J.O.; Pacyna, J.M. Quantitative assessment of worldwide contamination of air, water and soils by trace metals. Nature 1988, 333, 134–139. [Google Scholar] [CrossRef]
- Gordon, R.; Graedel, T.; Bertram, M.; Fuse, K.; Lifset, R.; Rechberger, H.; Spatari, S. The characterization of technological zinc cycles. Resour. Conserv. Recycl. 2003, 39, 107–135. [Google Scholar] [CrossRef]
- Shiel, A.E.; Weis, D.; Orians, K.J. Evaluation of zinc, cadmium and lead isotope fractionation during smelting and refining. Sci. Total Environ. 2010, 408, 2357–2368. [Google Scholar] [CrossRef]
- Caldelas, C.; Weiss, D.J. Zinc Homeostasis and isotopic fractionation in plants: A review. Plant Soil 2017, 411, 17–46. [Google Scholar] [CrossRef]
- Rosman, K.J.R.; Taylor, P.D.P. Isotopic compositions of the elements 1997 (Technical Report). Pure Appl. Chem. 1998, 70, 217–235. [Google Scholar] [CrossRef]
- Blix, R.; Ubisch, H.; Wickman, F.E. A search for variations in the relative abundance of the zinc isotopes in nature. Geochim. Cosmochim. Acta 1957, 11, 162–164. [Google Scholar] [CrossRef]
- Yang, L. Accurate and precise determination of isotopic ratios by MC-ICP-MS: A review. Mass Spectrom. Rev. 2009, 28, 990–1011. [Google Scholar] [CrossRef] [PubMed]
- Archer, C.; Vance, D. Mass discrimination correction in multiple-collector plasma source mass spectrometry: An example using Cu and Zn isotopes. J. Anal. At. Spectrom. 2004, 19, 656–665. [Google Scholar] [CrossRef]
- Maréchal, C.N.; Télouk, P.; Albarède, F. Precise analysis of copper and zinc isotopic compositions by plasma-source mass spectrometry. Chem. Geol. 1999, 156, 251–273. [Google Scholar] [CrossRef]
- Arnold, T.; Kirk, G.J.D.; Wissuwa, M.; Frei, M.; Zhao, F.-J.; Mason, T.F.D.; Weiss, D.J. Evidence for the mechanisms of zinc uptake by rice using isotope fractionation. Plant Cell Environ. 2010, 33, 370–381. [Google Scholar] [CrossRef] [PubMed]
- Aucour, A.M.; Pichat, S.; Macnair, M.R.; Oger, P. Fractionation of Stable Zinc Isotopes in the Zinc Hyperaccumulator Arabidopsis halleri and Nonaccumulator Arabidopsis petraea. Environ. Sci. Technol. 2011, 45, 9212–9217. [Google Scholar] [CrossRef]
- Couder, E.; Mattielli, N.; Drouet, T.; Smolders, E.; Delvaux, B.; Iserentant, A.; Meeus, C.; Maerschalk, C.; Opfergelt, S.; Houben, D. Transpiration flow controls Zn transport in Brassica napus and Lolium multiflorum under toxic levels as evidenced from isotopic fractionation. Comptes Rendus Geosci. 2015, 347, 386–396. [Google Scholar] [CrossRef]
- Kato, C.; Moynier, F.; Valdes, M.C.; Dhaliwal, J.K.; Day, J.M. Extensive volatile loss during formation and differentiation of the Moon. Nat. Commun. 2015, 6, 7617. [Google Scholar] [CrossRef]
- Luck, J.-M.; Ben Othman, D.; Albarède, F. Zn and Cu isotopic variations in chondrites and iron meteorites: Early solar nebula reservoirs and parent-body processes. Geochim. Cosmochim. Acta 2005, 69, 5351–5363. [Google Scholar] [CrossRef]
- Chen, J.-B.; Gaillardet, J.; Dessert, C.; Villemant, B.; Louvat, P.; Crispi, O.; Birck, J.-L.; Wang, Y.-N. Zn isotope compositions of the thermal spring waters of La Soufrière volcano, Guadeloupe Island. Geochim. Cosmochim. Acta 2014, 127, 67–82. [Google Scholar] [CrossRef]
- Larner, F.; Woodley, L.N.; Shousha, S.; Moyes, A.; Humphreys-Williams, E.; Strekopytov, S.; Halliday, A.N.; Rehkämper, M.; Coombes, R.C. Zinc isotopic compositions of breast cancer tissue†. Metallomics 2015, 7, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, K.V.; Moore, R.E.T.; Capper, M.S.; Schilling, K.; Goddard, K.; Ion, C.; Layton-Matthews, D.; Leybourne, M.I.; Coles, B.; Kreissig, K.; et al. Zinc stable isotope analysis reveals Zn dyshomeostasis in benign tumours, breast cancer, and adjacent histologically normal tissue. Metallomics 2021, 13, mfab027. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, T.; Mohamed, A.; Reese, A.; Wieser, M.; Kleeberg, U.; Pröfrock, D.; Irrgeher, J. Zinc isotopic variation of water and surface sediments from the German Elbe River. Sci. Total Environ. 2020, 707, 135219. [Google Scholar] [CrossRef] [PubMed]
- Qu, R.; Han, G. Potassium isotopes of fertilizers as potential markers of anthropogenic input in ecosystems. Environ. Chem. Lett. 2023, 21, 41–45. [Google Scholar] [CrossRef]
- Qu, R.; Han, G. Potassium Isotopes in Herbaceous Plants: A Potential New Tool for C3 and C4 Plant Research. J. Geophys. Res. Biogeosci. 2022, 127, e2021JG006682. [Google Scholar] [CrossRef]
- Liang, B.; Han, G.; Liu, M.; Li, X. Zn isotope fractionation during the development of low-humic gleysols from the Mun River Basin, northeast Thailand. Catena 2021, 206, 105565. [Google Scholar] [CrossRef]
- Tonhá, M.S.; Araújo, D.F.; Araújo, R.; Cunha, B.C.; Machado, W.; Portela, J.F.; Souza, J.P.; Carvalho, H.K.; Dantas, E.L.; Roig, H.L.; et al. Trace metal dynamics in an industrialized Brazilian river: A combined application of Zn isotopes, geochemical partitioning, and multivariate statistics. J. Environ. Sci. 2021, 101, 313–325. [Google Scholar] [CrossRef]
- Arnold, T.; Markovic, T.; Kirk, G.J.; Schönbächler, M.; Rehkämper, M.; Zhao, F.J.; Weiss, D.J. Iron and zinc isotope fractionation during uptake and translocation in rice (Oryza sativa) grown in oxic and anoxic soils. Comptes Rendus Geosci. 2015, 347, 397–404. [Google Scholar] [CrossRef]
- Jeong, H.; Ra, K.; Choi, J.Y. Copper, Zinc and Lead Isotopic Delta Values and Isotope Ratios of Various Geological and Biological Reference Materials. Geostand. Geoanal. Res. 2021, 45, 551–563. [Google Scholar] [CrossRef]
- Zhao, Y.; Vance, D.; Abouchami, W.; de Baar, H. Biogeochemical cycling of zinc and its isotopes in the Southern Ocean. Geochim. Cosmochim. Acta 2014, 125, 653–672. [Google Scholar] [CrossRef]
- Samanta, M.; Ellwood, M.J.; Mortimer, G.E. A method for determining the isotopic composition of dissolved zinc in seawater by MC-ICP-MS with a 67Zn–68Zn double spike. Microchem. J. 2016, 126, 530–537. [Google Scholar] [CrossRef]
- Bermin, J.; Vance, D.; Archer, C.; Statham, P. The determination of the isotopic composition of Cu and Zn in seawater. Chem. Geol. 2006, 226, 280–297. [Google Scholar] [CrossRef]
- Gélabert, A.; Pokrovsky, O.; Viers, J.; Schott, J.; Boudou, A.; Feurtet-Mazel, A. Interaction between zinc and freshwater and marine diatom species: Surface complexation and Zn isotope fractionation. Geochim. Cosmochim. Acta 2006, 70, 839–857. [Google Scholar] [CrossRef]
- Tu, Y.-J.; You, C.-F.; Kuo, T.-Y. Source identification of Zn in Erren River, Taiwan: An application of Zn isotopes. Chemosphere 2020, 248, 126044. [Google Scholar] [CrossRef]
- Szynkiewicz, A.; Borrok, D.M. Isotope variations of dissolved Zn in the Rio Grande watershed, USA: The role of adsorption on Zn isotope composition. Earth Planet. Sci. Lett. 2016, 433, 293–302. [Google Scholar] [CrossRef]
- Liang, B.; Han, G.; Zhao, Y. Zinc isotopic signature in tropical soils: A review. Sci. Total Environ. 2022, 820, 153303. [Google Scholar] [CrossRef]
- Zeng, J.; Han, G. Tracing zinc sources with Zn isotope of fluvial suspended particulate matter in Zhujiang River, southwest China. Ecol. Indic. 2020, 118, 106723. [Google Scholar] [CrossRef]
- Wiggenhauser, M.; Bigalke, M.; Imseng, M.; Keller, A.; Archer, C.; Wilcke, W.; Frossard, E. Zinc isotope fractionation during grain filling of wheat and a comparison of zinc and cadmium isotope ratios in identical soil–plant systems. N. Phytol. 2018, 219, 195–205. [Google Scholar] [CrossRef]
- Liang, L.; Liu, C.-Q.; Zhu, X.; Ngwenya, B.T.; Wang, Z.; Song, L.; Li, J. Zinc Isotope Characteristics in the Biogeochemical Cycle as Revealed by Analysis of Suspended Particulate Matter (SPM) in Aha Lake and Hongfeng Lake, Guizhou, China. J. Earth Sci. 2020, 31, 126–140. [Google Scholar] [CrossRef]
- Chen, J.-B.; Louvat, P.; Gaillardet, J.; Birck, J.-L. Direct separation of Zn from dilute aqueous solutions for isotope composition determination using multi-collector ICP-MS. Chem. Geol. 2009, 259, 120–130. [Google Scholar] [CrossRef]
- Wang, D.; Zheng, L.; Ren, M.; Li, C.; Dong, X.; Wei, X.; Zhou, W.; Cui, J. Zinc in soil reflecting the intensive coal mining activities: Evidence from stable zinc isotopes analysis. Ecotoxicol. Environ. Saf. 2022, 239, 113669. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-C.; Welter, L.; Kolatkar, A.; Nieva, J.; Waitman, K.R.; Huang, K.-F.; Liao, W.-H.; Takano, S.; Berelson, W.M.; West, A.J.; et al. A new anion exchange purification method for Cu stable isotopes in blood samples. Anal. Bioanal. Chem. 2019, 411, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Maréchal, C.; Albarède, F. Ion-exchange fractionation of copper and zinc isotopes. Geochim. Cosmochim. Acta 2002, 66, 1499–1509. [Google Scholar] [CrossRef]
- Gelly, R.; Fekiacova, Z.; Guihou, A.; Doelsch, E.; Deschamps, P.; Keller, C. Lead, zinc, and copper redistributions in soils along a deposition gradient from emissions of a Pb-Ag smelter decommissioned 100 years ago. Sci. Total Environ. 2019, 665, 502–512. [Google Scholar] [CrossRef] [PubMed]
- Shiel, A.E.; Barling, J.; Orians, K.J.; Weis, D. Matrix effects on the multi-collector inductively coupled plasma mass spectrometric analysis of high-precision cadmium and zinc isotope ratios. Anal. Chim. Acta 2009, 633, 29–37. [Google Scholar] [CrossRef]
- Mason, T.F.D.; Weiss, D.J.; Horstwood, M.; Parrish, R.R.; Russell, S.S.; Mullane, E.; Coles, B.J. High-precision Cu and Zn isotope analysis by plasma source mass spectrometry Part 2. Correcting for mass discrimination effects. J. Anal. At. Spectrom. 2004, 19, 218–226. [Google Scholar] [CrossRef]
- Li, W.; Gou, W.; Li, W.; Zhang, T.; Yu, B.; Liu, Q.; Shi, J. Environmental applications of metal stable isotopes: Silver, mercury and zinc. Environ. Pollut. 2019, 252, 1344–1356. [Google Scholar] [CrossRef]
- Conway, T.M.; John, S.G. The biogeochemical cycling of zinc and zinc isotopes in the North Atlantic Ocean. Glob. Biogeochem. Cycles 2014, 28, 1111–1128. [Google Scholar] [CrossRef]
- Little, S.; Vance, D.; Walker-Brown, C.; Landing, W. The oceanic mass balance of copper and zinc isotopes, investigated by analysis of their inputs, and outputs to ferromanganese oxide sediments. Geochim. Cosmochim. Acta 2014, 125, 673–693. [Google Scholar] [CrossRef]
- Takano, S.; Tsuchiya, M.; Imai, S.; Yamamoto, Y.; Fukami, Y.; Suzuki, K.; Sohrin, Y. Isotopic analysis of nickel, copper, and zinc in various freshwater samples for source identification. Geochem. J. 2021, 55, 171–183. [Google Scholar] [CrossRef]
- Peel, K.; Weiss, D.; Siggc, L. Zinc isotope composition of settling particles as a proxy for biogeochemical processes in lakes: Insights from the eutrophic Lake Greifen, Switzerland. Limnol. Oceanogr. 2009, 54, 1699–1708. [Google Scholar] [CrossRef]
- Aranda, S.; Borrok, D.M.; Wanty, R.B.; Balistrieri, L.S. Zinc isotope investigation of surface and pore waters in a mountain watershed impacted by acid rock drainage. Sci. Total Environ. 2012, 420, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Petit, J.C.; Schäfer, J.; Coynel, A.; Blanc, G.; Chiffoleau, J.-F.; Auger, D.; Bossy, C.; Derriennic, H.; Mikolaczyk, M.; Dutruch, L.; et al. The estuarine geochemical reactivity of Zn isotopes and its relevance for the biomonitoring of anthropogenic Zn and Cd contaminations from metallurgical activities: Example of the Gironde fluvial-estuarine system, France. Geochim. Cosmochim. Acta 2015, 170, 108–125. [Google Scholar] [CrossRef]
- Petit, J.C.; De Jong, J.; Chou, L.; Mattielli, N. Development of Cu and Zn Isotope MC-ICP-MS Measurements: Application to Suspended Particulate Matter and Sediments from the Scheldt Estuary. Geostand. Geoanal. Res. 2008, 32, 149–166. [Google Scholar] [CrossRef]
- Conway, T.M.; John, S.G. The cycling of iron, zinc and cadmium in the North East Pacific Ocean—Insights from stable isotopes. Geochim. Cosmochim. Acta 2015, 164, 262–283. [Google Scholar] [CrossRef]
- Sivry, Y.; Riotte, J.; Sonke, J.; Audry, S.; Schäfer, J.; Viers, J.; Blanc, G.; Freydier, R.; Dupré, B. Zn isotopes as tracers of anthropogenic pollution from Zn-ore smelters The Riou Mort–Lot River system. Chem. Geol. 2008, 255, 295–304. [Google Scholar] [CrossRef]
- Guinoiseau, D.; Gélabert, A.; Moureau, J.; Louvat, P.; Benedetti, M.F. Zn Isotope Fractionation during Sorption onto Kaolinite. Environ. Sci. Technol. 2016, 50, 1844–1852. [Google Scholar] [CrossRef]
- Li, W.; Livi, K.J.T.; Xu, W.; Siebecker, M.G.; Wang, Y.; Phillips, B.L.; Sparks, D.L. Formation of Crystalline Zn–Al Layered Double Hydroxide Precipitates on γ-Alumina: The Role of Mineral Dissolution. Environ. Sci. Technol. 2012, 46, 11670–11677. [Google Scholar] [CrossRef]
- Fujii, T.; Moynier, F.; Blichert-Toft, J.; Albarède, F. Density functional theory estimation of isotope fractionation of Fe, Ni, Cu, and Zn among species relevant to geochemical and biological environments. Geochim. Cosmochim. Acta 2014, 140, 553–576. [Google Scholar] [CrossRef]
- Wang, Z.; Peacock, C.; Kwon, K.D.; Gu, X.; Feng, X.; Li, W. Site-specific isotope fractionation during Zn adsorption onto birnessite: Insights from X-ray absorption spectroscopy, density functional theory and surface complexation modeling. Geochim. Cosmochim. Acta 2023, 348, 68–84. [Google Scholar] [CrossRef]
- Komárek, M.; Ratié, G.; Vaňková, Z.; Šípková, A.; Chrastný, V. Metal isotope complexation with environmentally relevant surfaces: Opening the isotope fractionation black box. Crit. Rev. Environ. Sci. Technol. 2022, 52, 3573–3603. [Google Scholar] [CrossRef]
- Pokrovsky, O.; Viers, J.; Freydier, R. Zinc stable isotope fractionation during its adsorption on oxides and hydroxides. J. Colloid Interface Sci. 2005, 291, 192–200. [Google Scholar] [CrossRef]
- Juillot, F.; Maréchal, C.; Ponthieu, M.; Cacaly, S.; Morin, G.; Benedetti, M.; Hazemann, J.; Proux, O.; Guyot, F. Zn isotopic fractionation caused by sorption on goethite and 2-Lines ferrihydrite. Geochim. Cosmochim. Acta 2008, 72, 4886–4900. [Google Scholar] [CrossRef]
- Bryan, A.L.; Dong, S.; Wilkes, E.B.; Wasylenki, L.E. Zinc isotope fractionation during adsorption onto Mn oxyhydroxide at low and high ionic strength. Geochim. Cosmochim. Acta 2015, 157, 182–197. [Google Scholar] [CrossRef]
- Dong, S.; Wasylenki, L.E. Zinc isotope fractionation during adsorption to calcite at high and low ionic strength. Chem. Geol. 2016, 447, 70–78. [Google Scholar] [CrossRef]
- Balistrieri, L.S.; Borrok, D.M.; Wanty, R.B.; Ridley, W.I. Fractionation of Cu and Zn isotopes during adsorption onto amorphous Fe(III) oxyhydroxide: Experimental mixing of acid rock drainage and ambient river water. Geochim. Cosmochim. Acta 2008, 72, 311–328. [Google Scholar] [CrossRef]
- Gou, W.; Li, W.; Ji, J.; Li, W. Zinc Isotope Fractionation during Sorption onto Al Oxides: Atomic Level Understanding from EXAFS. Environ. Sci. Technol. 2018, 52, 9087–9096. [Google Scholar] [CrossRef]
- Wanty, R.; Podda, F.; De Giudici, G.; Cidu, R.; Lattanzi, P. Zinc isotope and transition-element dynamics accompanying hydrozincite biomineralization in the Rio Naracauli, Sardinia, Italy. Chem. Geol. 2013, 337–338, 1–10. [Google Scholar] [CrossRef]
- Veeramani, H.; Eagling, J.; Jamieson-Hanes, J.H.; Kong, L.; Ptacek, C.J.; Blowes, D.W. Zinc Isotope Fractionation as an Indicator of Geochemical Attenuation Processes. Environ. Sci. Technol. Lett. 2015, 2, 314–319. [Google Scholar] [CrossRef]
- Jamieson-Hanes, J.H.; Shrimpton, H.K.; Veeramani, H.; Ptacek, C.J.; Lanzirotti, A.; Newville, M.; Blowes, D.W. Evaluating zinc isotope fractionation under sulfate reducing conditions using a flow-through cell and in situ XAS analysis. Geochim. Cosmochim. Acta 2017, 203, 1–14. [Google Scholar] [CrossRef]
- Mavromatis, V.; González, A.G.; Dietzel, M.; Schott, J. Zinc isotope fractionation during the inorganic precipitation of calcite—Towards a new pH proxy. Geochim. Cosmochim. Acta 2019, 244, 99–112. [Google Scholar] [CrossRef]
- Samanta, M.; Ellwood, M.J.; Sinoir, M.; Hassler, C.S. Dissolved zinc isotope cycling in the Tasman Sea, SW Pacific Ocean. Mar. Chem. 2017, 192, 1–12. [Google Scholar] [CrossRef]
- John, S.G.; Genevieve Park, J.; Zhang, Z.; Boyle, E.A. The isotopic composition of some common forms of anthropogenic zinc. Chem. Geol. 2007, 245, 61–69. [Google Scholar] [CrossRef]
- Kafantaris, F.-C.A.; Borrok, D.M. Zinc isotope fractionation during surface adsorption and intracellular incorporation by bacteria. Chem. Geol. 2014, 366, 42–51. [Google Scholar] [CrossRef]
- John, S.G.; Conway, T.M. A role for scavenging in the marine biogeochemical cycling of zinc and zinc isotopes. Earth Planet. Sci. Lett. 2014, 394, 159–167. [Google Scholar] [CrossRef]
- Vance, D.; Archer, C.; Bermin, J.; Kennaway, G.; Cox, E.; Statham, P.; Lohan, M.; Ellwood, M. Zn isotopes as a new tracer of metal micronutrient usage in the oceans. Geochim. Cosmochim. Acta 2006, 18, A666. [Google Scholar] [CrossRef]
- Wiederhold, J.G. Metal Stable Isotope Signatures as Tracers in Environmental Geochemistry. Environ. Sci. Technol. 2015, 49, 2606–2624. [Google Scholar] [CrossRef]
| Sample Types | Digestion Methods | References |
|---|---|---|
| Straw, grain, and paddy soil | 15 N HNO3, 30% H2O2,28 N HF, and 0.1 N HCl | [29] |
| Mun River Basin soil | 3:1 Concentrated HNO3 and concentrated HF and 3:1 concentrated HNO3 and HCl | [27] |
| Aquatic plants, mussels, sludge, etc. | Concentrated HNO3, HF, and HClO4 (4:5:1) | [30] |
| Silicate rocks | 6:2:1 Concentrated HF: HNO3:HClO4 | [15] |
| Sedimentary rocks | 16 N HNO3 and concentrated HF and 12N HCl | [15] |
| Diatom cells | 5% HNO3 and 5% H2O2 | [34] |
| Seine River SPM and sediment | 16 N HNO3 and 27 N HF | [5] |
| River water | Filtering, steamed dry, concentrated HNO3 | [24,35,36] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ding, Z.; Han, G.; Qu, R.; Liu, J.; Wang, P. Review on Zinc Isotopes in River Systems: Distribution and Application. Water 2024, 16, 87. https://doi.org/10.3390/w16010087
Ding Z, Han G, Qu R, Liu J, Wang P. Review on Zinc Isotopes in River Systems: Distribution and Application. Water. 2024; 16(1):87. https://doi.org/10.3390/w16010087
Chicago/Turabian StyleDing, Ziyang, Guilin Han, Rui Qu, Jinke Liu, and Peng Wang. 2024. "Review on Zinc Isotopes in River Systems: Distribution and Application" Water 16, no. 1: 87. https://doi.org/10.3390/w16010087
APA StyleDing, Z., Han, G., Qu, R., Liu, J., & Wang, P. (2024). Review on Zinc Isotopes in River Systems: Distribution and Application. Water, 16(1), 87. https://doi.org/10.3390/w16010087









