1. The Importance of the Treatment of Drinking Water
The ability to supply clean and safe drinking water has been a major concern in recent decades, given the increase in globalization, industrialization, and agricultural activities throughout the world. Adding to these causes of pollution, natural disasters and climate change have also played a role in creating favorable conditions for water eutrophication and developing pathogens in water sources. These pathogens can include viruses, bacteria, and protozoa [
1,
2,
3]. Furthermore, in areas rich in water or subjected to heavy rains, water stagnation can pose a potential problem and further leads to the development of water pathogens [
4].
The greater awareness of these ongoing problems has led to the establishment of more rigorous policies by the European Union for the control of the quality of drinking water. These policies have the aim of ensuring that safe drinking water can be distributed to everyone [
1,
2]. To guarantee safe drinking water, the water must be subjected to a treatment process that includes coagulation and flocculation, sedimentation/flotation, filtration, and disinfection prior to being stored and supplied. Such a process is shown in
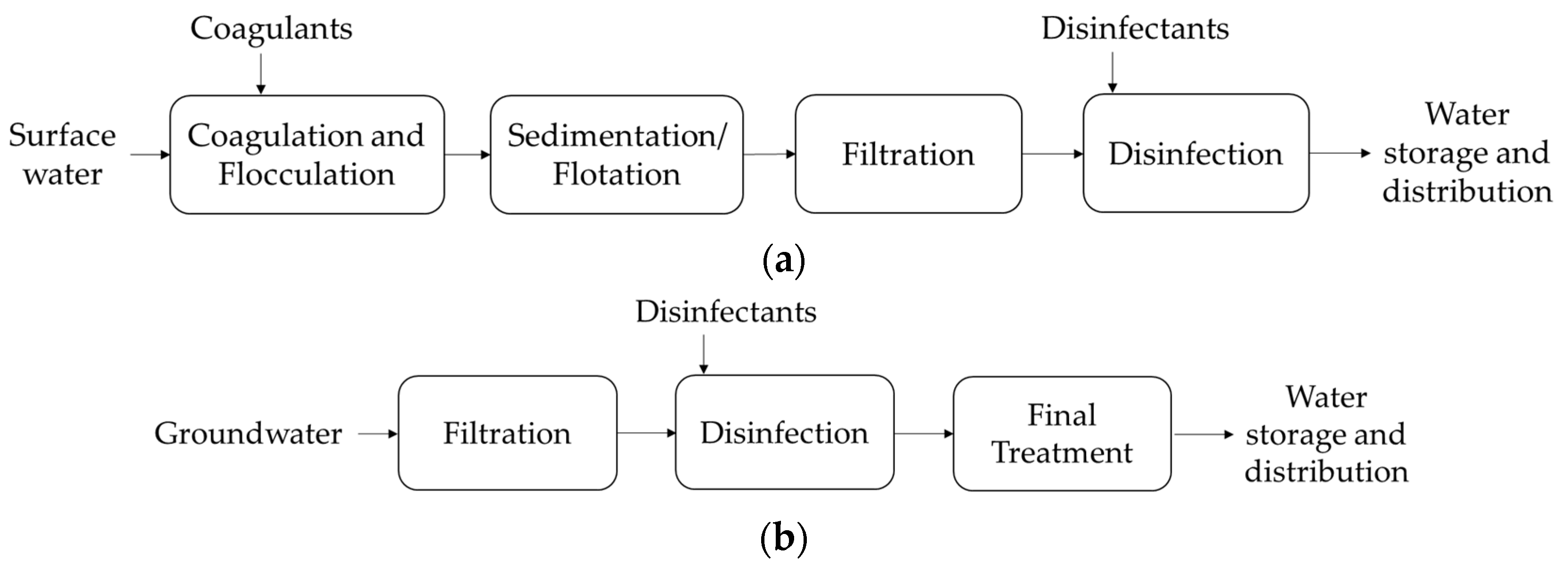
Figure 1a, illustrating a case when surface water is used as the primary water source [
1]. However, if the primary source water is groundwater, the stages for its treatment are reduced, with the filtration, disinfection, and a final treatment (e.g., chemical or UV) being applicable, followed by storage and distribution—
Figure 1b [
5].
In the coagulation and flocculation process, chemicals with a positive charge (e.g., aluminum, iron salts and polyelectrolytes) are added to the water to interact with organic matter and other dissolved particles that have a negative charge; the binding between these particles forms aggregates that are then removed by sedimentation or even flotation if the aggregates present low densities. To remove suspended and dissolved particles or several microorganisms that may be present in the water, filtration through sand, gravel and charcoal can be utilized [
1]. Next, water is subjected to disinfection (e.g., by chlorine, chloramine, chlorine dioxide, UV light or ozonolysis), which focuses on the oxidative destruction of organisms, preventing the development of waterborne diseases [
6]. In this stage, a proper disinfectant should be used to also avoid the future reappearance of these contaminants due to the presence of residual disinfectant throughout the distribution system—a process that has already been implemented in several states of the USA. Furthermore, disinfection also aims to control waterborne pathogens that are susceptible to the applied disinfection agent. However, the production of toxic compounds should be avoided once it affects certain drinking water properties, such as taste and smell, and poses risks to the human health [
3].
To implement a safe disinfection process, it is important to consider the characteristics of the water source, such as its type, the concentration of microorganisms that it may contain, and the final quality intended for the resulting water. Natural events, such as forest fires, can lead to additional run-off from burned organic matter that can enter the watershed, leading to the increased water pollution of the water supplied to the water treatment plant, which can consequently lead to an increase in the formation of DBPs [
7,
8]. Additionally, the toxicity of the disinfectant agent, the formation of DBPs, the drinking water treatment plant characteristics and the operation costs must also be considered. These costs may vary if wider and longer distribution systems are required [
7].
2. The Incidence of DBPs in Drinking Water
Water disinfection can be performed (i) via a chemical process, using chlorine, chloramine, chlorine dioxide, ozone, adsorption, a combination of ozone with hydrogen peroxide or by using ultraviolet (UV) radiation, which can be combined with either ozone, hydrogen peroxide or titanium dioxide, and (ii) via a physical process, by means of membrane recurring techniques [
3,
7].
In chlorination, the most used method for the disinfection of drinking water, the following reactions occur [
9]:
Opting for chlorination is mainly due to its oxidizing potential, residual effect, large availability, and cost effectiveness [
10]. Additionally, the efficiency of this process can be easily manipulated by changes in contact time, pH, and temperature. Moreover, the presence of residual chlorine in the distribution systems can prevent the regrowth of microorganisms in the water distribution system. In fact, with a 30 min contact time at a temperature of 20 °C and a pH lower than 8, a residual chlorine level of 0.5 mg·L
−1 or higher must be guaranteed. The presence of residual chlorine should also be maintained during storage and distribution. However, in this process, the chlorine can react with organic precursors, such as natural organic matter (NOM) (e.g., oils, sugars, humic and fulvic acids) or inorganic precursors (e.g., bromide or iodide ions), leading to the formation of disinfection byproducts (DBPs) [
1,
2,
10,
11,
12]. The formation of these DBPs is unwanted and can be a result of using either surface water or groundwater to obtain drinking water within acceptable quality levels. However, surface waters tend to present higher concentrations of these contaminants than groundwater, given their differences in quantities of organic matter. In addition, in regions affected by drought, polluted waters can contaminate aquifers, leading to an additional introduction of organic constituents and contaminants into groundwater systems, contributing to the formation of DBPs [
10,
13].
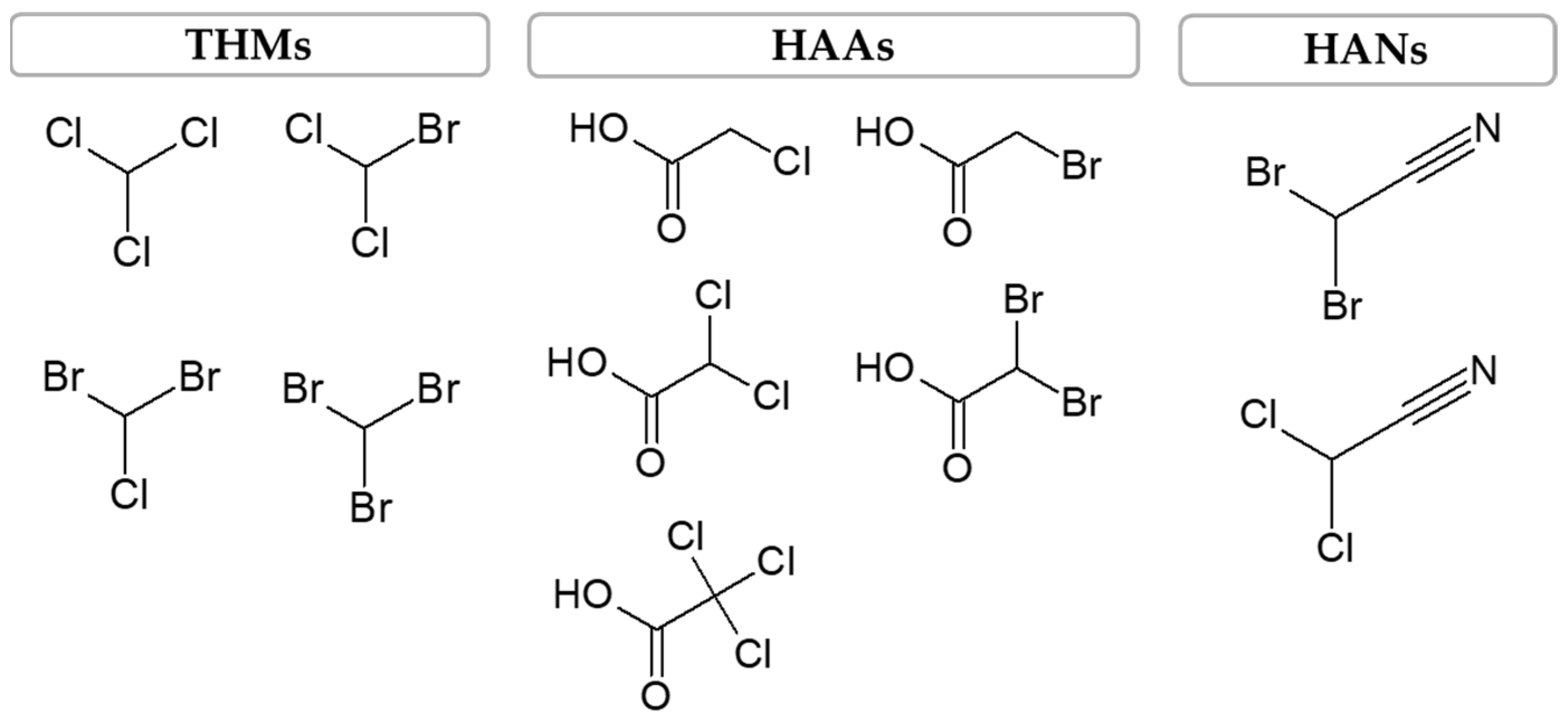
DBPs include several halogenated organic compounds, such as trihalomethanes (THMs), haloacetic acids (HAAs) and haloacetonitriles (HANs). From the aforementioned DBPs, the THMs are the most common, followed by the HAAs, which can be present in finished water at concentrations on the order of µg/L, and as for the HANs, they can be found in concentrations as low as ng/L and up to µg/L [
14]. However, regarding their toxicity, HANs are the most toxic, followed by HAAs and, finally, by THMs. The THMs group is characterized by its composition of volatile compounds, such as chloroform, bromodichloromethane (BDCM), chlorodibromomethane (CDBM) and bromoform, while HAAs include monochloroacetic acid (MCAA), dichloroacetic acid (DCAA), trichloroacetic acid (TCAA), monobromoacetic acid (MBAA) and dibromoacetic acid (DBAA) and HANs include dichloroacetonitrile (DCAN) and dibromoacetonitrile (DBAN)—
Figure 2 [
3,
7,
14].
The effects on human health resulting from contact with DBPs can occur due to the inhalation, ingestion and/or adsorption of contaminated water. The impact of DBPs at low concentrations on human health is still unknown; however, contact with higher concentrations of these compounds can lead to health problems, namely asthma, reproductive problems, carcinogenic, teratogenic and mutagenic effects [
1]. Recently, Diana et al., reviewed a potential association between the structure of DBPs formed through chlorination and the occurrence of bladder cancer [
15].
To try to reduce these problems, intensive research has been performed, not only to identify emerging DBPs but also to better understand their formation mechanisms, toxicity effects and exposure characteristics [
16]. This understanding will allow easier compliance with the regulatory values already defined for several DBPs by the United States Environmental Protection Agency (U.S. EPA), the World Health Organization (WHO) and the European Union (EU)—
Table 1. In addition, it may also lead, in the near future, to the establishment of regulatory values for DBPs that are not yet regulated. This need arises because, even though the unregulated DBPs appear in lower concentrations than the already regulated DBPs, they can be more toxic to humans as well as also being responsible for the development of diseases that regulated DBPs are already known to cause [
12]. Increasing industrialization and extreme events related to climate change are leading to a rapid decrease in the quality of water sources used for drinking water supplies. Thus, the disinfection step will require harsher conditions to reach the necessary water quality, which may lead to an increased incidence of DBPs in the supplied water in the near future. This means that further efforts must be made to understand their roles in human health.
The studies performed regarding DBPs have mostly relied on their impact on health; however, these compounds can also be harmful to the environment, so a more intensive study on this issue should be conducted in the future. Their effects on the environment could be a result of an intentional release from point sources (e.g., lakes, water ponds, springs) or of an unintentional discharge from non-point sources (e.g., farming) [
17].
Table 1.
Regulatory limits (*) or guideline values (non-regulatory limits) (**) for DBPs established by different organizations [
4,
18,
19,
20].
Table 1.
Regulatory limits (*) or guideline values (non-regulatory limits) (**) for DBPs established by different organizations [
4,
18,
19,
20].
| DBP Group | DBP | U.S. EPA (μg/L) | WHO (μg/L) | EU (μg/L) |
|---|
| THMs | Chloroform | 70 ** | 300 | |
| BDCM | 0 | 60 | |
| CDBM | 60 ** | 100 | |
| Bromoform | 0 | 100 | |
| THM4 1 | 80 * | | 100 |
| HAAs | MCAA | | 20 | |
| DCAA | | 50 | |
| TCAA | | 200 | |
| MBAA | | | |
| DBAA | | | |
| HAA5 2 | 60 * | | 60 * |
| HANs | DCAN | | 20 | |
| DBAN | | 70 | |
| Inorganic DBPs | Bromate | 10 * | 10 | 10 |
| Chlorite | 1000 * | 700 | |
| Chlorate | | 700 | |
3. Current Status and Emerging Strategies for Minimizing DBPs in Drinking Water
DBPs are mainly formed by chlorination, and they result from the interaction between chlorine and NOM. Thus, different approaches to hinder their formation can be viable. For instance, a decrease in the organic matter content in water prior to the disinfection stage would allow a decrease in DBPs’ precursors and, consequently, in chlorine use [
21]. One downfall of this is that a greater understanding of the different mechanisms that lead to the formation of DBPs is required to make this single approach viable. The recent increase in water pollution is leading to higher amounts of total organic carbon (TOC) in raw water that is collected for producing drinking water. Thus, the use of pretreatments able to reduce the number of organic compounds before the disinfection would reduce the likelihood of DBPs being produced in that stage. The types of treatments required depend upon the raw water characteristics (e.g., initial TOC, alkalinity, and electrical conductivity) as well as the targeted final TOC.
During disinfection, some modifications of the process can also be implemented, such as choosing other disinfectants for the process (e.g., chlorine dioxide, chloramine, ozone, or UV). Water disinfection by ozone effectively inactivates and prevents the reappearance of water pathogens through less time-consuming reactions; however, unwanted by-products can still result from this process, mainly bromate. Moreover, this disinfection process is complex and, when the water presents high solid content and organic matter levels, this process becomes economically unviable. The use of UV for water disinfection is largely applied, even though it is a costly process that also requires the use of residual concentrations of other disinfectants, namely chlorine. This is a consequence of the treatment with UV light not leading to the presence of residual disinfectant in the water. Furthermore, in the presence of bromide, brominated DBPs can be formed [
22,
23].
Another approach consists of the removal of DBPs formed via disinfection (without any change in the process prior to the disinfection) [
7,
21,
22,
24]. The current technologies utilized are focused mainly on the removal of DBPs after their formation and less on preventing their formation; however, the latter is the most efficient approach for their reduction. In this sense, wastewater treatment plants tend to remove organic matter in bulk, as well as other water impurities, before disinfection, but more specific treatments aimed at NOM removal before chlorination are not carried out so often [
21,
25]. Therefore, studies should be intensified, aiming at establishing preventive measures and practices to ensure drinking water quality and mitigate problems along the whole chain of distribution, thus reducing DBP formation and, subsequently, the need for intensive disinfection.
3.1. Removal of DBP Precursors
Some techniques have already been applied to reduce the quantity of NOM before disinfection. These techniques include enhanced coagulation, activated carbon adsorption, membrane techniques and advanced oxidation processes (AOPs).
Enhanced coagulation has the same operating principle as coagulation; however, the maximization of the removal of total organic carbon is the main focus of this technique. This is achieved through pH adjustments and increasing the load of coagulants used.
In adsorption, a liquid or gaseous phase comes into contact with a condensed phase (the adsorbent) and the molecules or ions present in the liquid or gaseous phase remain on the adsorbent surface. In this sense, activated carbon adsorption relies on the porous structure of this material to capture the molecules present in the liquid. The adsorption efficiency depends on the pH of the solution, the adsorbent used (e.g., its surface area, functional groups, and dosage), the adsorbate (e.g., its nature, namely its solubility, and its initial concentration), the temperature and pressure, the contact time between the two phases (liquid or gas and the condensed phase) and the mixing conditions. Granular activated carbon (GAC) adsorption has also been proven to be a good agent for the removal of NOM. However, adsorption by GAC has increased performance at the beginning of its use, whereas a longer use leads to a decrease in its adsorption capacity, given the consumption of the media [
26]. The GAC working lifetime depends on the load of DBPs present in the water. There is a cost associated with the replenishment of the media. However, in some plants, a regeneration system is available to allow for maximum adsorption.
The use of membranes takes advantage of size exclusion and diffusive mechanisms via a selective barrier to separate the molecules; however, this technique is greatly affected by the occurrence of fouling [
1,
11,
16,
27,
28,
29,
30].
On the other hand, the use of AOPs for water treatment, such as ozone-based processes, Fenton-like processes and photocatalysis, have already demonstrated their positive contributions to the minimization of water pollutants [
7,
31]. Chin and Bérubé compared the potentials of ozone, UV, and a combination of these two techniques regarding the removal of DBP precursors from water samples [
32]. That study revealed that the combination of these two techniques was able to decrease the formation potential of THMs by 80% and the formation potential of HAAs by 70% [
32]. These techniques have usually high upfront investment and operating costs; however, despite this, AOPs are integrated into drinking water facilities whenever high removal rates are required [
7,
31]. Still, their in situ application has not been reported, which means that further studies at the bench and pilot scale are required to demonstrate their efficiency in order to boost their full-scale application.
3.2. Removal of DBPs Formed after the Disinfection Process
For the removal of DBPs formed through disinfection, some techniques have already been reported, such as adsorption, aeration, air stripping and membrane techniques. The membrane techniques, as well as adsorption, can be used either for NOM removal, as seen in
Section 3.1, or for DBP removal [
10,
16].
The removal of contaminants by adsorption has been shown to be a preferred alternative, since it involves low installation costs and a simple operation design, which does not compromise its performance, as high performances are still obtained. Furthermore, adsorption does not lead to the formation of harmful substances. The application of adsorption allows the use of several adsorbents to remove unwanted materials from water. Some adsorbents already used for water treatment include activated carbon and nano adsorbents, among others [
29,
33]. Of these adsorbents, activated carbon has already been used to successfully remove organic and inorganic pollutants, dyes and heavy metals from water [
34]. This type of adsorbent can be produced from a variety of materials (e.g., coal and fruit-shells) and one of its characteristics that greatly influences its potential to be used for contaminant removal is its large surface area, which can vary from 500 to 1500 m
2/g [
29]. However, the formation of activated carbon presents a significant carbon footprint and can rarely be reused [
35]. Jiang et al., used a small column with GAC for the adsorption of aromatic halogenated DBPs, THMs and HAAs. For the former, a considerable decrease was observed, while for THMs and HAAs, the results were superior to the application of this technique prior to the disinfection via chlorination [
30].
The removal of DBPs through aeration and air stripping has proven to be an effective method to successfully decrease and mitigate the DBPs present in the water through the air/water interface inside the water storage tanks. However, its efficiency depends on the volatility of the compounds, which tends to increase with temperature. The volatility of these components can be analyzed using Henry’s constant (
KH). For the THMs, the
KH values are shown in
Table 2. Among the different THMs, chloroform presents the highest
KH value, meaning that this compound represents a lower challenge regarding its removal from water [
36].
The application of membrane techniques for DBP removal has already been performed and has been shown to be efficient for the removal of DBPs at trace levels; however, for higher concentrations of DBPs, this technique’s efficiency is decreased. Additionally, this technology involves increased energy consumption, which also poses a limitation [
16].
The adsorption through nanomaterials mainly relies on the use of aerogels, which comprise solid materials derived from gels, with a network of bonded particles with a gas phases in its pores. Aerogels are versatile, lightweight, highly porous (80–99.8%) and low-density materials, making them good candidates for the enhancement of surface-dependent processes, such as sorption. Additionally, when using silica-based aerogels, the easy manipulation and optimization of their surface chemistry can be performed due to the wide availability of silicon alkoxides [
38,
39].
In water treatment, silica aerogels have already been used for the removal of oils and other organic contaminants, such as ethanol, toluene, chlorobenzene, trichloroethylene and polycyclic aromatic hydrocarbons (e.g., pyrene, benzo(b)fluoranthene and benzo(a)pyrene) [
40,
41]. However, the application of these materials for the removal of DBPs is still residual. Mansouri et al., used a modified silica aerogel (TiO
2–SiO
2) to effectively remove humic acids, a precursor of THMs, from water samples [
42]. Thus, given the versatility of these materials and the ease of modifying their surface, in addition to the successful applications already reported for water treatment, a more intensive study on the application of aerogels in the removal of DBPs should be performed, as it represents a promising path for future research.
Even though many techniques are or can be used for DBP prevention and removal, the fate of these compounds is an important aspect to consider. The process of water treatment and of water distribution can have an impact on the fate of DBPs, since there are the necessary conditions for microbial degradation. This can occur mainly in regard to HAAs, given their biodegradability. Furthermore, the stagnation of the water during distribution has a significant impact on the quantity of the disinfecting agent required to maintain the water quality [
26].
4. Future Challenges in the Detection of DBPs in Drinking Water
The continued discovery of new DBPs suggests that the search and development of newer techniques may be a never-ending process. Furthermore, the optimization of current technologies and their monitoring throughout the entire water distribution process is a necessity to prevent the re-emergence of DBPs [
12]. Thus, there is an urgent need for the development of suitable monitoring systems in all distribution networks with the analytical capacity to detect these compounds, even at very low concentrations (μg/L and ng/L). In fact, the development of a real-time robust online monitoring system able to quickly detect potential threats to human health should be a focus in the near future. In addition, the behavior of DBPs along the drinking water distribution network and their interaction with naturally occurring materials, as well as with the pipelines, must be well understood in order to establish the placement of sensing infrastructures along the network. This would make it possible to capture the pollutants whenever and wherever they occur through the injection of disinfectants along the distribution system, allowing for the better control and maintenance of the water quality according to the legally required standards. To achieve this, carefully thought-out methodologies on the appropriate placement of the sensors, based on infrared and Raman spectroscopies, for example, should be developed. Regardless, this sensing technique, when applied, could not only be used for drinking water resources but also for surface and ground water resources, allowing a greater understanding of water status and the better use of these resources. Climate change and excessive temperatures are leading to a decrease in the quality of source water. This is an important drawback, since the lower the quality of the raw water, the harsher the disinfection conditions required, which will lead to the formation of a higher quantity of DBPs. On the other hand, the high temperatures and elevated storage tanks in distribution also contribute to DBP formation. Thus, technologies must be developed to avoid the formation of DBPs during the disinfection process by targeting NOM removal or to provide alternative disinfection technologies that avoid the formation of DBPs. Another aspect could be a downstream technology able to remove DBPs from disinfected water when their formation is unavoidable.
Another barrier to changes in drinking water management is the operator’s knowledge and understanding of DBPs. Moreover, there is always some inertia towards change and fear regarding new technologies. Thus, the involvement of society and policy makers is required to promote a smooth change.
5. Conclusions
The increasing concern regarding the supply of safe drinking water has been a focus in past few years, given the increase in activities that lead to the formation of contaminants that affect its composition. Additionally, contaminated water is a contributing factor to the development of several diseases. To overcome this, water is subjected to treatment processes that remove pathogens; these processes include a disinfection stage, where a disinfectant agent, normally chlorine, is used. However, the use of this chemical substance leads to the development of unwanted byproducts, DBPs, given the interaction of chlorine with the NOM present in the water. The negative effects of DBPs on human health and on the environment have fueled the research on their prevention (e.g., through enhanced coagulation, adsorption, membrane techniques and AOPs) or removal after formation (e.g., adsorption, aeration, air stripping and membrane techniques). The most common approach used relies on the removal of already-formed DBPs and not on their prevention, which is the most efficient methodology.
Nanomaterials have been shown to be efficient in the removal of some water contaminants (e.g., ethanol, chlorobenzene). However, their application for the removal of DBPs has not been studied in detail. Thus, these types of materials present a huge potential in this context.
Future studies should focus not only on the optimization of the current techniques for water treatment but also on monitoring water composition throughout the entire water distribution process. Moreover, the development of robust online monitoring systems able to quickly detect DBPs, even at low concentrations, is a necessity, and will have a great impact in the future. This will also be a valuable tool for better controlling the regulatory values established by different countries regarding DBPs, allowing adjustments to be made in the water during distribution, via the injection of disinfectants, if required.