Screening the Pollution-Tolerant Chlorococcum sp. (Chlorophyceae) Grown in Municipal Wastewater for Simultaneous Nutrient Removal and Biodiesel Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation and Growth Conditions of Chlorococcum sp.
2.2. Design of Nutrient Removal Experiment
2.3. Monitoring the Growth Rates of Chlorococcum sp.
2.4. Removal of Nutrients
2.5. Characterization of Fatty Acids
2.6. Characteristics of Biodiesel
2.7. Statistical Analysis
3. Results and Discussion
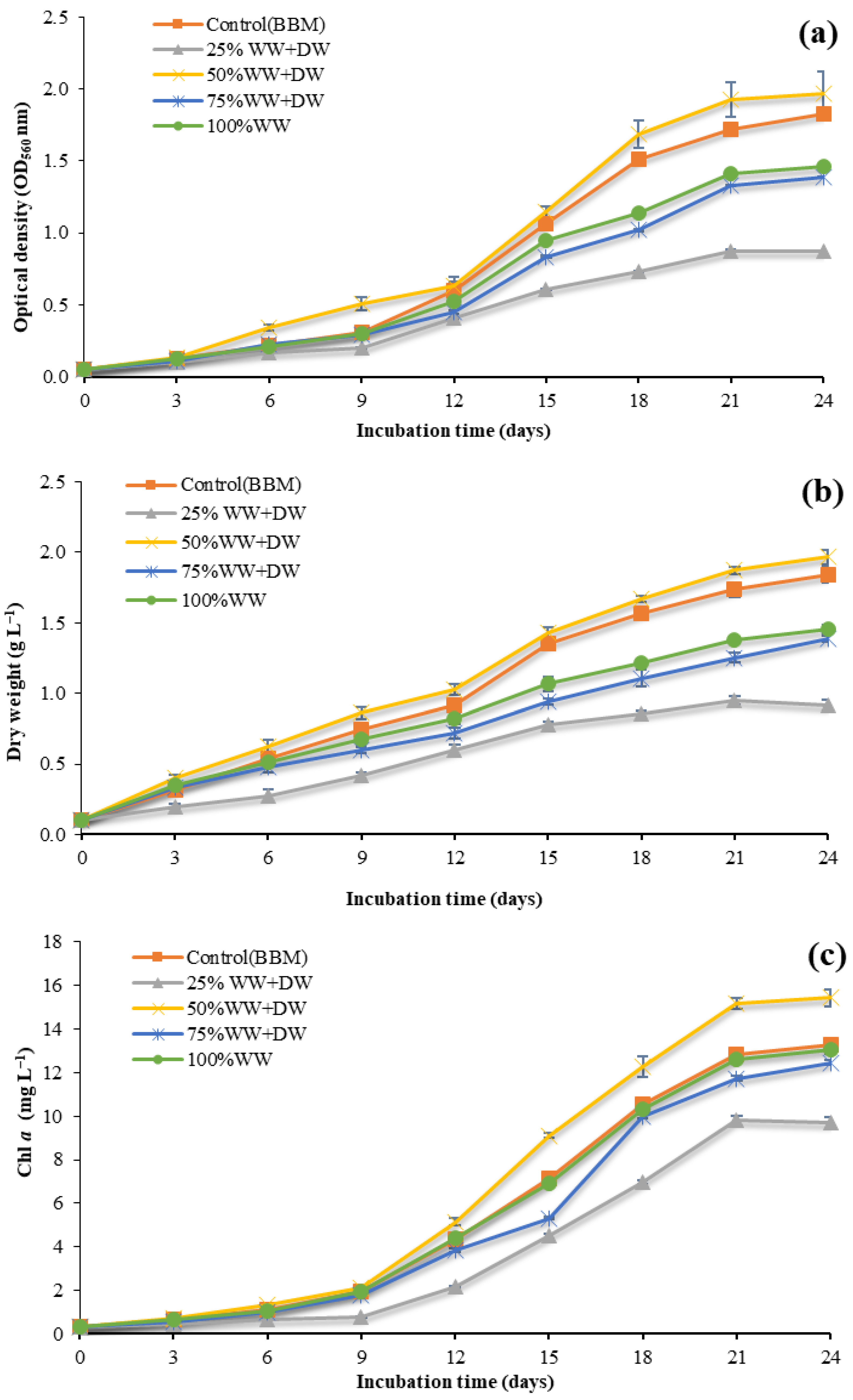
3.1. Growth Conditions
3.2. Biomass and Biochemical Composition
3.3. Removal of Nutrients
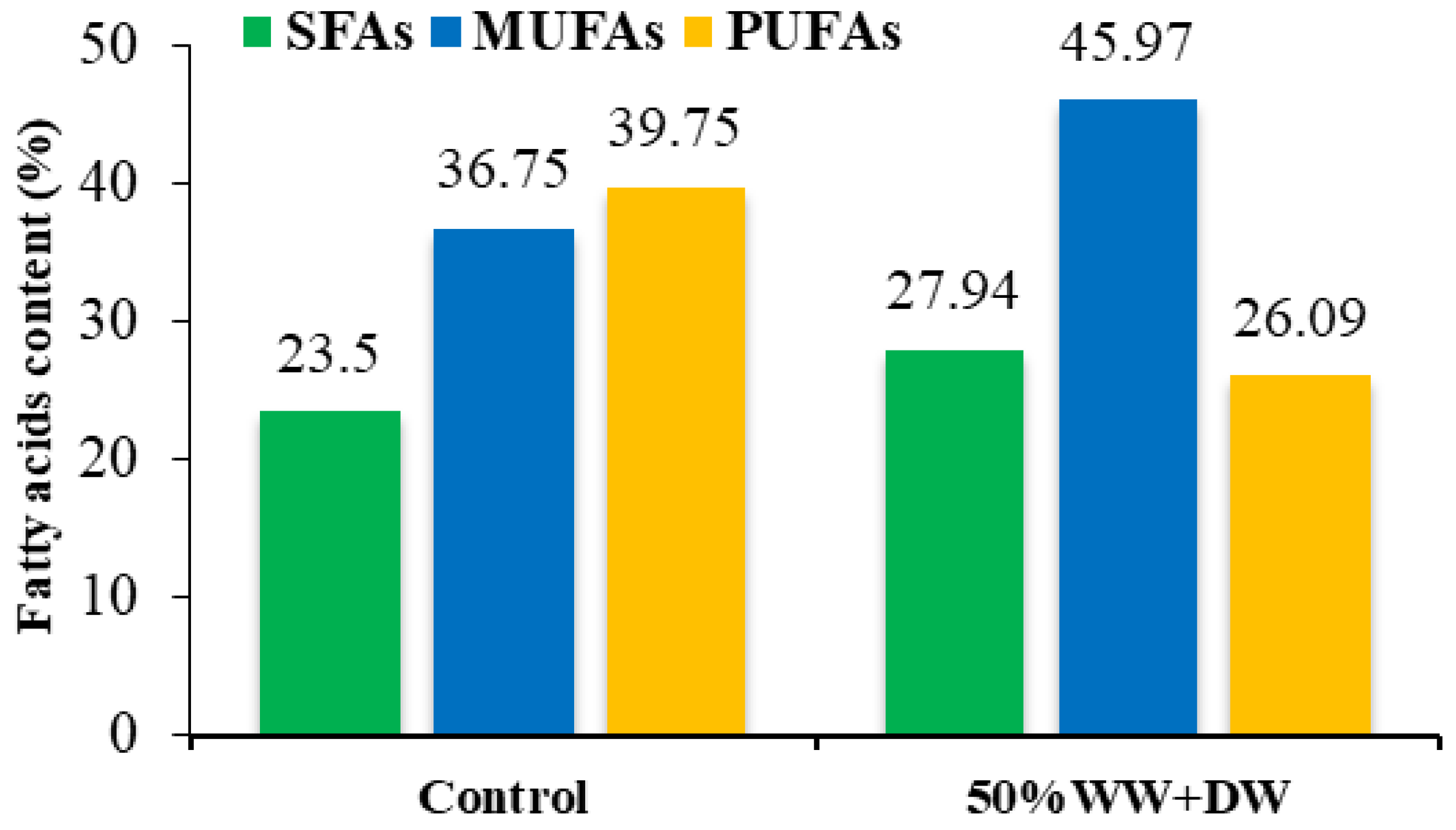
3.4. Fatty Acid Composition
3.5. Biodiesel Properties
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abubakr, H.; Vasquez, J.C.; Mahmoud, K.; Darwish, M.M.; Guerrero, J.M. Comprehensive review on renewable energy sources in Egypt–current status, grid codes and future vision. IEEE Access 2022, 10, 4081–4101. [Google Scholar] [CrossRef]
- El-Sheekh, M.M. Biodiesel from microalgae: Advantages and future prospective. Egypt. J. Bot. 2021, 61, 669–671. [Google Scholar] [CrossRef]
- Pal, P.; Chew, K.W.; Yen, H.W.; Lim, J.W.; Lam, M.K.; Show, P.L. Cultivation of oily microalgae for the production of third-generation biofuels. Sustainability 2019, 11, 5424. [Google Scholar] [CrossRef]
- Vale, M.A.; Ferreira, A.; Pires, J.C.; Gonçalves, A.L. CO2 capture using microalgae. In Advances in Carbon Capture: Methods, Technologies and Applications; Rahimpour, M.R., Farsi, M., Makarem, M.A., Eds.; Woodhead Publishing: Cambridge, UK, 2020; pp. 381–405. [Google Scholar]
- Rahpeyma, S.S.; Raheb, J. Microalgae biodiesel as a valuable alternative to fossil fuels. BioEnergy Res. 2019, 12, 958–965. [Google Scholar] [CrossRef]
- Deora, P.S.; Verma, Y.; Muhal, R.A.; Goswami, C.; Singh, T. Biofuels: An alternative to conventional fuel and energy source. Mater. Today Proc. 2022, 48, 1178–1184. [Google Scholar]
- Mathimani, T.; Pugazhendhi, A. Utilization of algae for biofuel, bio-products and bio-remediation. Biocatal. Agric. Biotechnol. 2019, 17, 326–330. [Google Scholar] [CrossRef]
- Merlo, S.; Durany, X.G.; Tonon, A.P.; Rossi, S. Marine microalgae contribution to sustainable development. Water 2021, 13, 1373. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae beats bioethanol. Trends Biotechnol. 2008, 26, 126–131. [Google Scholar] [CrossRef]
- Balat, M.; Balat, H. Progress in biodiesel processing. Appl. Energy 2010, 87, 1815–1835. [Google Scholar] [CrossRef]
- Park, J.B.K.; Craggs, R.J.; Shilton, A.N. Wastewater treatment high rate algal ponds for biofuel production. Bioresour. Technol. 2011, 102, 35–42. [Google Scholar] [CrossRef]
- Kuenz, A.; Grimm, D.; Rahmann, G. Versatility of algae exploring the potential of algae for nutrient circulation. Org. Agric. 2021, 11, 251–260. [Google Scholar] [CrossRef]
- Ansari, F.A.; Singh, P.; Guldhe, A.; Bux, F. Microalgal cultivation using aquaculture wastewater: Integrated biomass generation and nutrient remediation. Algal Res. 2017, 21, 169–177. [Google Scholar] [CrossRef]
- Al-Jabri, H.; Das, P.; Khan, S.; Thaher, M.; AbdulQuadir, M. Treatment of wastewaters by microalgae and the potential applications of the produced biomass—A review. Water 2021, 13, 27. [Google Scholar] [CrossRef]
- Eladel, H.; Abomohra, A.E.; Battah, M.; Mohmmed, S.; Radwan, A.; Abdelrahim, H. Evaluation of Chlorella sorokiniana isolated from local municipal wastewater for dual application in nutrient removal and biodiesel production. Bioprocess Biosyst. Eng. 2019, 42, 425–433. [Google Scholar] [CrossRef]
- Komárek, J.; Fott, B. Chlorophyceae (Grünalgen) Ordnung: Chlorococcales. Das Phytoplankton des Süsswassers. In Das Phytoplankton des Süsswassers (Die Binnengewässer) XVI. 7., Teil 1. Hälfte; Huber-Pestalozzi, G., Ed.; E. Schweizerbart’sche Verlangbuchhandlung (Nägele u. Obermiller): Stuttgart, Germany, 1983; pp. [1]–x, [1]–1044. [Google Scholar]
- Lichtenthaler, H.K.; Buschmann, C. Chlorophylls and carotenoids: Measurement and characterization by UV-VIS spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, 1, F4.3.1–F4.3.8. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Abomohra, A.E.F.; Wagner, M.; El-Sheekh, M.; Hanelt, D. Lipid and total fatty acid productivity in photoautotrophic fresh water microalgae: Screening studies towards biodiesel production. J. Appl. Phycol. 2013, 25, 931–936. [Google Scholar] [CrossRef]
- Lee, C.M.; Trevino, B.; Chaiyawat, M. A simple and rapid solvent extraction method for determining total lipids in fish tissue. J. AOAC Int. 1996, 79, 487–492. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.T.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Woods, J.; Mellon, M. Molybdenum blue reaction: A spectrophotometric study. Ind. Eng. Chem. Anal. Ed. 1941, 13, 760–764. [Google Scholar] [CrossRef]
- Mancy, K.H. Instrumental Analysis for Water Pollution Control; Ann Arbor Science Publishers Inc.: Ann Arbor, MI, USA, 1971; pp. 1–331. [Google Scholar]
- Arif, M.; Wang, L.; Salama, E.S.; Hussain, M.S.; Li, X.; Jalalah, M.; Liu, P. Microalgae isolation for nutrient removal assessment and biodiesel production. BioEnergy Res. 2020, 13, 1247–1259. [Google Scholar] [CrossRef]
- Talebi, A.F.; Tabatabaei, M.; Chisti, Y. BiodieselAnalyzer: A user-friendly software for predicting the properties of prospective biodiesel. Biofuel Res. J. 2014, 1, 55–57. [Google Scholar] [CrossRef]
- Pena, A.D.C.C.; Bertoldi, C.F.; Fontoura, J.T.D.; Trierweiler, L.F.; Gutterres, M. Consortium of microalgae for tannery effluent treatment. Braz. Arch. Biol. Technol. 2019, 62, e19170518. [Google Scholar] [CrossRef]
- Nwuche, C.O.; Ekpo, D.C.; Eze, C.N.; Aoyagi, H.; Ogbonna, J.C. Use of palm oil mill effluent as medium for cultivation of Chlorella sorokiniana. Biotechnol. J. Int. 2014, 4, 305–316. [Google Scholar] [CrossRef]
- Nagi, M.; He, M.; Li, D.; Gebreluel, T.; Cheng, B.; Wang, C. Utilization of tannery wastewater for biofuel production: New insights on microalgae growth and biomass production. Sci. Rep. 2020, 10, 1530. [Google Scholar] [CrossRef]
- Tan, K.A.; Lalung, J.; Wijaya, D.; Ismail, N.; Wan Omar, W.M.; Wabaidur, S.M.; Siddiqui, M.R.; Alam, M.; Rafatullah, M. Removal of nutrients by using green microalgae from lab-scale treated palm oil mill effluent. Fermentation 2022, 8, 658. [Google Scholar] [CrossRef]
- Tsavatopoulou, V.D.; Aravantinou, A.F.; Manariotis, I.D. Biofuel conversion of Chlorococcum sp. and Scenedesmus sp. biomass by one-and two-step transesterification. Biomass Convers. Biorefin. 2021, 11, 1301–1309. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Anal, A.K. Enhanced lipid and starch productivity of microalga (Chlorococcum sp. TISTR 8583) with nitrogen limitation following effective pretreatments for biofuel production. Biotechnol. Rep. 2019, 21, e00298. [Google Scholar] [CrossRef]
- Arguelles, E.D.L.R. Nitrogen starvation induced lipid accumulation by Chlorococcum infusionum (EAU-10) as potential renewable source of lipid for biodiesel production. J. Microbiol. Biotechnol. Food Sci. 2021, 11, e1931. [Google Scholar] [CrossRef]
- Tam, N.F.Y.; Wong, Y.S. Wastewater nutrient removal by Chlorella pyrenoidosa and Scenedesmus sp. Environ. Pollut. 1989, 58, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Liu, Y.; Feng, J.; Liu, Q.; Nan, F.; Xie, S. Nutrients removal from undiluted cattle farm wastewater by the two-stage process of microalgae-based wastewater treatment. Bioresour. Technol. 2018, 264, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.Y.; Zhu, J.N.; Kong, F.; Xing, D.; Zhao, L.; Ma, J.; Liu, B.F. Ultrasonic enhanced simultaneous algal lipid production and nutrients removal from non-sterile domestic wastewater. Energy Convers. Manag. 2019, 180, 680–688. [Google Scholar] [CrossRef]
- Rothermel, M.C. Coupling the Wastewater Treatment Process with an Algal Photobioreactor for Nutrient Removal and Renewable Resource Production. Ph.D. Thesis, University of Pittsburgh, Pittsburgh, PA, USA, 2011. [Google Scholar]
- Garg, S.K.; Tripathi, M. Process parameters for decolorization and biodegradation of orange II (acid orange 7) in dye-simulated minimal salt medium and subsequent textile effluent treatment by Bacillus cereus (MTCC 9777) RMLAU1. Environ. Monit. Assess. 2013, 185, 8909–8923. [Google Scholar] [CrossRef]
- Anto, S.; Pugazhendhi, A.; Mathimani, T. Lipid enhancement through nutrient starvation in Chlorella sp. and its fatty acid profiling for appropriate bioenergy feedstock. Biocatal. Agric. Biotechnol. 2019, 20, 101179. [Google Scholar] [CrossRef]
- Soares, S.; Rocha, F.R. Fast spectrophotometric determination of iodine value in biodiesel and vegetable oils. J. Braz. Chem. Soc. 2018, 29, 1701–1706. [Google Scholar] [CrossRef]
- Deshmukh, S.; Bala, K.; Kumar, R. Selection of microalgae species based on their lipid content, fatty acid profile and apparent fuel properties for biodiesel production. Environ. Sci. Pollut. Res. 2019, 26, 24462–24473. [Google Scholar] [CrossRef]
- Almarashi, J.Q.; El-Zohary, S.E.; Ellabban, M.A.; Abomohra, A.E.F. Enhancement of lipid production and energy recovery from the green microalga Chlorella vulgaris by inoculum pretreatment with low-dose cold atmospheric pressure plasma (CAPP). Energy Convers. Manag. 2020, 204, 112314. [Google Scholar] [CrossRef]
- Do, J.-M.; Jo, S.-W.; Kim, I.-S.; Na, H.; Lee, J.H.; Kim, H.S.; Yoon, H.-S. A feasibility study of wastewater treatment using domestic microalgae and analysis of biomass for potential applications. Water 2019, 11, 2294. [Google Scholar] [CrossRef]
- ASTM D6751-08; Standard Specification for Biodiesel Fuel Blend Stock (B100) for Middle Distillate Fuels. ASTM International: West Conshohocken, PA, USA, 2008.
- EN 14214:2012+A2:2019; Liquid Petroleum Products—Fatty Acid Methyl Esters (FAME) for Use in Diesel Engines and Heating Applications—Requirements and Test Methods. European Committee for Standardization: Brussels, Belgium, 2018.


| Parameters | Values |
|---|---|
| pH | 7.6 ± 0.07 |
| TDS (mg L−1) | 520.30 ± 47 |
| EC (µS cm−1) | 1106 ± 24.3 |
| Temperature (°C) | 26 ± 0.28 |
| NO3− (mg L−1) | 9.1 ± 1.24 |
| NH4+ (mg L−1) | 0.87 ± 0.06 |
| TP (mg L−1) | 1.39 ± 0.70 |
| BOD (mg L−1) | 12.17 ± 1.40 |
| COD (mg L−1) | 44.67 ± 3.20 |
| Parameters | Control (BBM) | 25% WW | 50% WW | 75% WW | 100% WW |
|---|---|---|---|---|---|
| biomass (g L−1) | 1.84 ± 0.057 b | 0.92 ± 0.041 d | 1.97 ± 0.049 c | 1.39 ± 0.019 a | 1.46 ± 0.031 b |
| biomass productivity (mg L−1 day−1) | 77.0± 1.99 b | 38.0± 2.0 d | 82.0 ± 0.987 c | 58.0 ± 2.99 a | 61.0 ± 2.99 b |
| lipid content (mg L−1) | 520.6 ± 0.754 b | 223.3 ± 2.88 d | 600.0 ± 0.865 c | 413.3 ± 0.220 a | 450.7 ± 0.650 b |
| lipid productivity (mg L−1 day−1) | 21.7 ± 0.130 a | 9.3 ± 0.868 d | 25.0 ± 0.125 c | 17.2 ± 0.158 b | 18.8 ± 0.065 ab |
| lipid yield (% CDW) | 28.3 ± 0.760 a | 24.3 ± 2.88 b | 30.5 ± 0.871 b | 29.7 ± 0.224 a | 30.9 ± 0.650 a |
| protein yield (% CDW) | 31.0 ± 0.112 a | 31.6 ± 0.782 b | 29.8 ± 0.653 b | 31.1 ± 1.47 a | 32.3 ± 1.29 a |
| carbohydrate yield (% CDW) | 20.7 ± 0.568 a | 26.7 ± 1.35 b | 18.6 ± 0.389 b | 22.5 ± 0.401 b | 22.2 ± 0.376 a |
| Parameters | Control (BBM) | 25% WW | 50% WW | 75% WW | 100% WW | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| RR | RE% | RR | RE% | RR | RE% | RR | RE% | RR | RE% | |
| NH4+ | 0.02 ± 0.0 | 90.9 ± 1.38 | 0.01 ± 0.0 | 72.88 ± 5.42 | 0.02 ± 0.0 | 98.38 ± 1.93 | 0.03 ± 0.01 | 90.20 ± 1.0 | 0.04 ± 0.0 | 93.33 ± 2.78 |
| NO3− | 0.08 ± 0.01 | 91.44 ± 1.11 | 0.10 ± 0.01 | 80.42 ± 1.09 | 0.21 ± 0.01 | 96.90 ± 1.72 | 0.21 ± 0.01 | 90.74 ± 1.2 | 0.36 ± 0.01 | 91.42 ± 1.19 |
| TP | 0.05 ± 0.0 | 79.82 ± 1.51 | 0.01 ± 0.0 | 63.07 ± 1.48 | 0.03 ± 0.0 | 90.11 ± 1.27 | 0.04 ± 0.01 | 82.23 ± 1.95 | 0.05 ± 0.0 | 83.70 ± 0.85 |
| COD | 1.45 ± 0.12 | 89.94 ± 5.31 | 0.35 ± 0.06 | 57.82 ± 7.57 | 0.98 ± 0.02 | 91.73 ± 2.93 | 1.17 ± 0.07 | 80.19 ± 1.12 | 1.73 ± 0.04 | 88.90 ± 1.56 |
| BOD | 0.29 ± 0.02 | 86.56 ± 1.38 | 0.08 ± 0.02 | 52.41 ± 5.36 | 0.26 ± 0.03 | 96.67 ± 3.09 | 0.34 ± 0.01 | 81.55 ± 1.77 | 0.46 ± 0.05 | 87.04 ± 6.17 |
| TDS | 4.12 ± 0.19 | 53.66 ± 3.12 | 2.17 ± 0.19 | 34.20 ± 3.05 | 8.55 ± 0.21 | 76.16 ± 2.94 | 8.65 ± 0.25 | 51.52 ± 1.3 | 13.33 ± 0.05 | 58.93 ± 0.59 |
| EC | 7.13 ± 0.35 | 58.52 ± 2.84 | 3.94 ± 0.40 | 28.96 ± 3.95 | 14.75 ± 0.52 | 56.80 ± 1.62 | 14.08 ± 0.42 | 35.31 ± 0.91 | 19.63 ± 0.64 | 37.64 ± 1.05 |
| Fatty Acids | Control (BBM) | 50% WW Treatment |
|---|---|---|
| Saturated fatty acids (SFAs) | ||
| Pentadecenoic acid (C15:0) | 0.42 ± 0.02 | − |
| Palmitic acid (C16:0) | 21.36 ± 1.8 | 27.38 ± 1.4 |
| Stearic acid (C18:0) | 0.93 ± 0.02 | 0.56 ± 0.04 |
| Behenic acid (C22:0) | 0.38 ± 0.01 | − |
| Octacosanoic acid (C28:0) | 0.41 ± 0.01 | − |
| Monounsaturated fatty acids (MUFAs) | ||
| Palmitoleic acid (C16:1) | − | 1.54 ± 0.1 |
| Oleic acid (C18:1) | 36.75 ± 3.0 | 44.43 ± 3.7 |
| Polyunsaturated fatty acids (PUFAs) | ||
| Hexadecadienoic acid (C16:2) | 3.49 ± 0.7 | 2.92 ± 0.5 |
| Linoleic acid (C18:2) | 23.45 ± 2.9 | 21.02 ± 2.4 |
| Hexadecatrienoic acid (C16:3) | 4.93 ± 0.6 | − |
| α-Linolenic acid (C18:3) | 0.98 ± 0.08 | − |
| Hexadecatetraenoic acid (C16:4) | 4.85 ± 0.95 | 1.50 ± 0.08 |
| Stearidonic acid (C18:4) | 2.06 ± 0.4 | 0.66 ± 0.06 |
| SFAs | 23.50 | 27.94 |
| MUFAs | 36.75 | 45.97 |
| PUFAs | 39.75 | 26.09 |
| Total | 100.00 | 100.00 |
| Predicted Biodiesel Characteristics | Chlorococcum sp. | International Standards of Biodiesel | ||
|---|---|---|---|---|
| Control | 50% WW | ASTM D6751-08 [45] | EN 14214:2012+A2:2019 [46] | |
| Degree of unsaturation (DU) | 89.73 | 89.33 | – | – |
| Saponification value (mg g−1) (SV) | 175.90 | 195.68 | – | – |
| Iodine value (gI 100 g−1) (IV) | 85.78 | 81.99 | – | less 120 |
| Cetane number (CN) | 58.03 | 55.74 | above 47 | minimum 51 |
| Long-chain saturated factor (LCSF) | 3.17 | 3.02 | – | – |
| Cold filter plugging point (°C) (CFPP) | −6.52 | −7.0 | −5 to −13 | 5 to −20 |
| Cloud point (°C) (CP) | 6.24 | 9.41 | −3 to 12 | 4 |
| Pour point (°C) (PP) | −0.04 | 3.39 | −15 to 10 | – |
| Allylic position equivalent (APE) | 89.73 | 87.79 | – | – |
| Bis-allylic position equivalent (BAPE) | 25.41 | 21.02 | – | – |
| Oxidation stability (h) (OS) | 7.42 | 8.20 | 3 | 8 |
| Higher heating value (HHV) | 34.04 | 37.69 | – | – |
| Kinematic viscosity (mm2 s−1) (υi) | 3.13 | 3.59 | 1.9–6 | 3.5–5 |
| Density (g cm−3) (ρ) | 0.76 | 0.84 | 0.88 | 0.86–0.90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morsi, H.H.; El-Sheekh, M.M.; Eladel, H.; Al-Tuwaijri, M.M.; El-Sabbagh, S.M.; Maher, A.; Saber, A.A. Screening the Pollution-Tolerant Chlorococcum sp. (Chlorophyceae) Grown in Municipal Wastewater for Simultaneous Nutrient Removal and Biodiesel Production. Water 2023, 15, 1723. https://doi.org/10.3390/w15091723
Morsi HH, El-Sheekh MM, Eladel H, Al-Tuwaijri MM, El-Sabbagh SM, Maher A, Saber AA. Screening the Pollution-Tolerant Chlorococcum sp. (Chlorophyceae) Grown in Municipal Wastewater for Simultaneous Nutrient Removal and Biodiesel Production. Water. 2023; 15(9):1723. https://doi.org/10.3390/w15091723
Chicago/Turabian StyleMorsi, Hanaa H., Mostafa M. El-Sheekh, Hamed Eladel, Majdah M. Al-Tuwaijri, Sabha M. El-Sabbagh, Ayah Maher, and Abdullah A. Saber. 2023. "Screening the Pollution-Tolerant Chlorococcum sp. (Chlorophyceae) Grown in Municipal Wastewater for Simultaneous Nutrient Removal and Biodiesel Production" Water 15, no. 9: 1723. https://doi.org/10.3390/w15091723
APA StyleMorsi, H. H., El-Sheekh, M. M., Eladel, H., Al-Tuwaijri, M. M., El-Sabbagh, S. M., Maher, A., & Saber, A. A. (2023). Screening the Pollution-Tolerant Chlorococcum sp. (Chlorophyceae) Grown in Municipal Wastewater for Simultaneous Nutrient Removal and Biodiesel Production. Water, 15(9), 1723. https://doi.org/10.3390/w15091723









