Can Zooplankton Add Value to Monitoring Water Quality? A Case Study of a Meso/Eutrophic Portuguese Reservoir
Abstract
1. Introduction
2. Materials and Methods
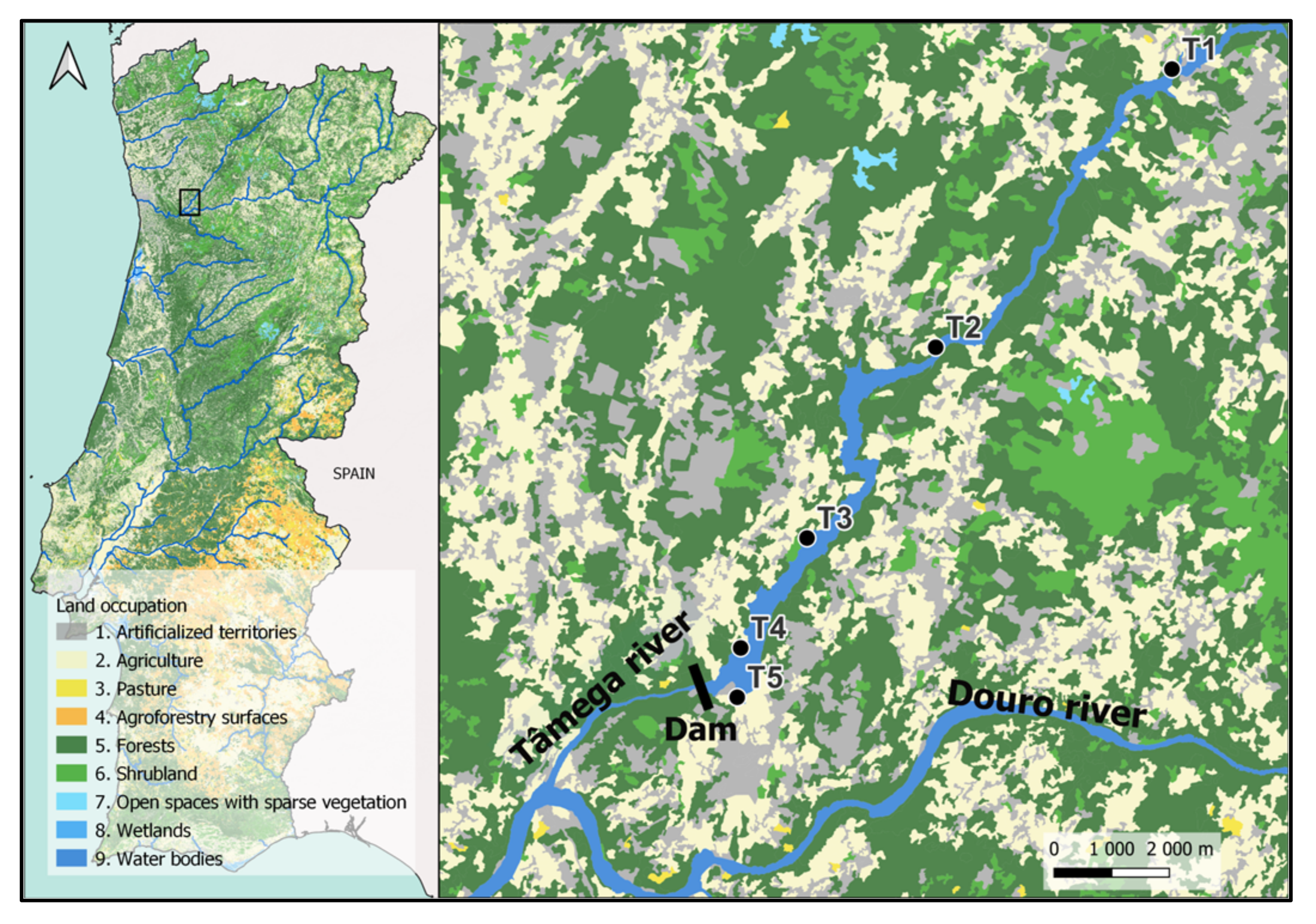
2.1. Study Area
2.2. Sampling Procedure
2.3. Physical and Chemical Analysis
2.4. Biological Analysis
2.5. Statistical Analysis
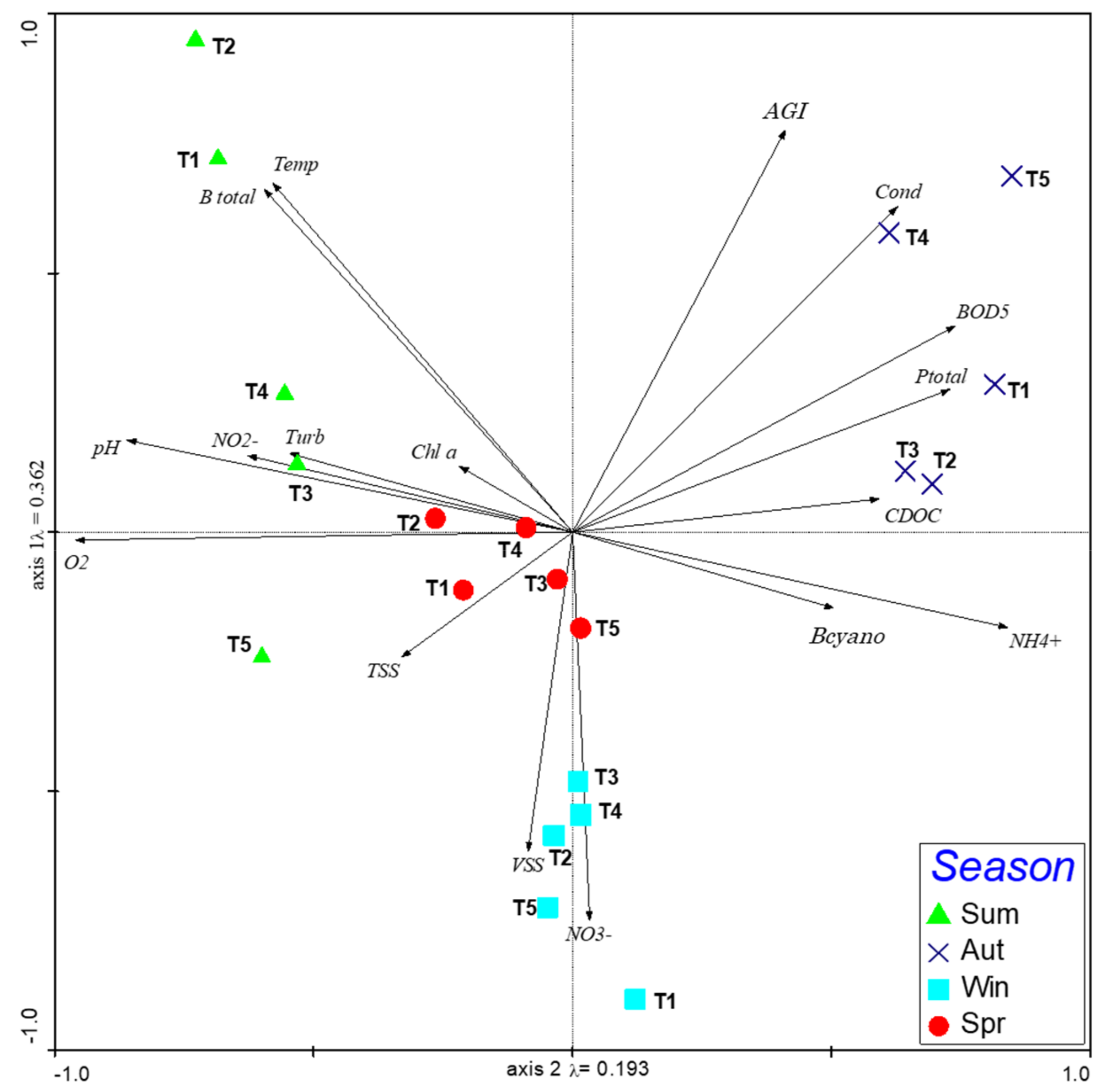
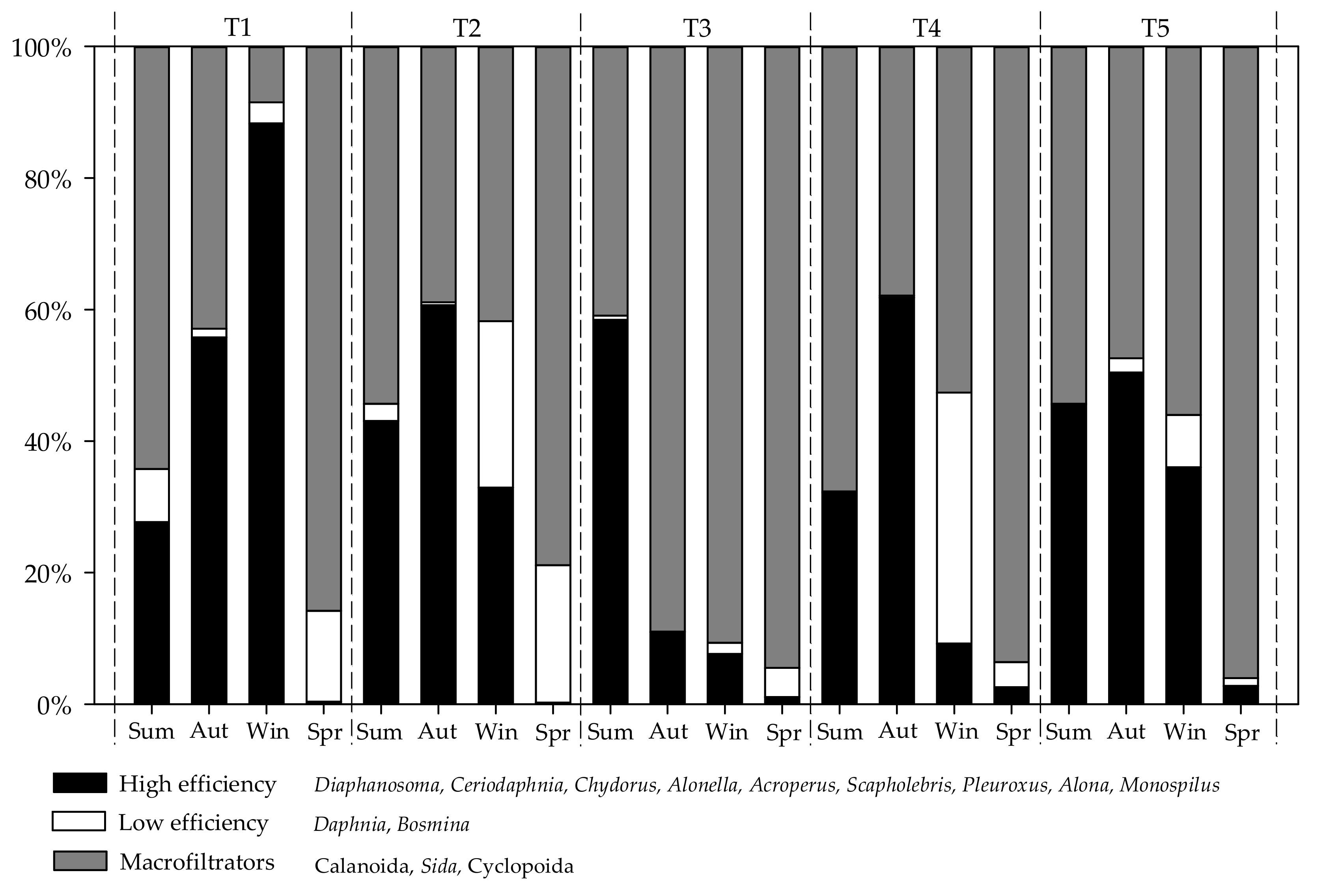
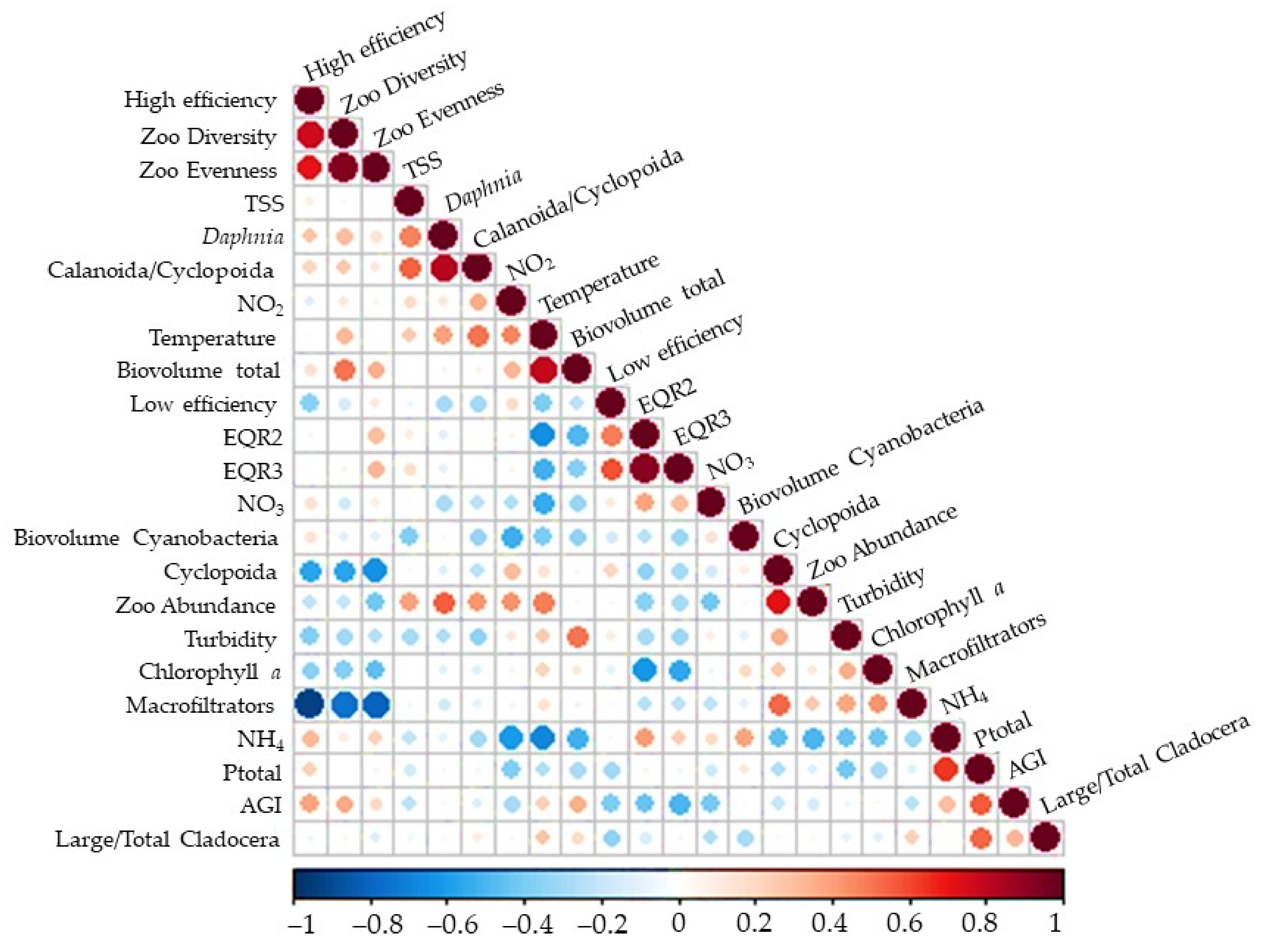
3. Results
3.1. WFD Ecological Potential
3.2. Zooplankton Community
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jackson, R.B.; Carpenter, S.R.; Dahm, C.N.; McKnight, D.M.; Naiman, R.J.; Postel, S.L.; Running, S.W. Water in a Changing World. Ecol. Appl. 2001, 11, 1027–1045. [Google Scholar] [CrossRef]
- Wada, Y.; Graaf, I.; Beek, L. High-Resolution Modeling of Human and Climate Impacts on Global Water Resources. J. Adv. Model. Earth Syst. 2016, 6, 513–526. [Google Scholar] [CrossRef]
- Rodrigues, S.; Pinto, I.; Martins, F.; Formigo, N.; Antunes, S.C. Can Biochemical Endpoints Improve the Sensitivity of the Biomonitoring Strategy Using Bioassays with Standard Species, for Water Quality Evaluation? Ecotoxicol. Environ. Saf. 2021, 215, 112151. [Google Scholar] [CrossRef] [PubMed]
- World Water Assessment Programme. The United Nations World Water Development Report 3: Water in a Changing World; UNESCO: Paris, France; Earthscan: London, UK, 2009; ISBN 978-1-84407-840-0. [Google Scholar]
- Diogo, B.S.; Rodrigues, S.; Silva, N.; Pinto, I.; Antunes, S.C. Evidence for Links between Feeding Behavior of Daphnia Magna and Water Framework Directive Elements: Case Study of Crestuma-Lever Reservoir. Water 2022, 14, 3989. [Google Scholar] [CrossRef]
- Helios-Rybicka, E.; Holda, A.; Jarosz, E. Monitoring and Quality Assessment of Selected Physical and Chemical Parameters of the Sola River System, South Poland. Inżynieria Sr. Akad. Górniczo-Hut. Im. S. Staszica W Krakowie 2005, 10, 45–58. [Google Scholar]
- INAG. Critérios Para a Classificação Do Estado Das Massas de Água Superfíciais—Rios e Albufeiras; INAG: Lisboa, Portugal, 2009. [Google Scholar]
- Simões, N.R.; Nunes, A.H.; Dias, J.D.; Lansac-Tôha, F.A.; Velho, L.F.M.; Bonecker, C.C. Impact of Reservoirs on Zooplankton Diversity and Implications for the Conservation of Natural Aquatic Environments. Hydrobiologia 2015, 758, 3–17. [Google Scholar] [CrossRef]
- Smith, V.H.; Tilman, G.D.; Nekola, J.C. Eutrophication: Impacts of Excess Nutrient Inputs on Freshwater, Marine, and Terrestrial Ecosystems. Environ. Pollut. 1999, 100, 179–196. [Google Scholar] [CrossRef]
- Bhagowati, B.; Ahamad, K. A Review on Lake Eutrophication Dynamics and Recent Developments in Lake Modeling. Ecohydrol. Hydrobiol. 2019, 19, 155–166. [Google Scholar] [CrossRef]
- Bengtsson, L.; Herschy, R.W.; Fairbridge, R.W. Encyclopedia of Lakes and Reservoirs; Springer: Dordrecht, The Netherlands, 2012. [Google Scholar]
- Pinto, I.; Rodrigues, S.; Lage, O.M.; Antunes, S.C. Assessment of Water Quality in Aguieira Reservoir: Ecotoxicological Tools in Addition to the Water Framework Directive. Ecotoxicol. Environ. Saf. 2021, 208, 111583. [Google Scholar] [CrossRef]
- European Parliament. European Union Directiva 2000/60/CE. J. Das Comunidades Eur. 2000, 1–72. [Google Scholar]
- García-Chicote, J.; Armengol, X.; Rojo, C. Zooplankton Abundance: A Neglected Key Element in the Evaluation of Reservoir Water Quality. Limnologica 2018, 69, 46–54. [Google Scholar] [CrossRef]
- Moss, B. Ecology of Freshwaters: A View for the Twenty-First Centuary, 4th ed.; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2010; ISBN 9783662068199. [Google Scholar]
- Søndergaard, M.; Jeppesen, E.; Jensen, J.P.; Amsinck, S.L. Water Framework Directive: Ecological Classification of Danish Lakes. J. Appl. Ecol. 2005, 42, 616–629. [Google Scholar] [CrossRef]
- Jeppesen, E.; Nõges, P.; Davidson, T.A.; Haberman, J.; Nõges, T.; Blank, K.; Lauridsen, T.L.; Søndergaard, M.; Sayer, C.; Laugaste, R.; et al. Zooplankton as Indicators in Lakes: A Scientific-Based Plea for Including Zooplankton in the Ecological Quality Assessment of Lakes According to the European Water Framework Directive (WFD). Hydrobiologia 2011, 676, 279–297. [Google Scholar] [CrossRef]
- Pinto, I.; Rodrigues, S.; Antunes, S.C. Assessment of the Benthic Macroinvertebrate Communities in the Evaluation of the Water Quality of Portuguese Reservoirs: An Experimental Approach. Water 2021, 13, 3391. [Google Scholar] [CrossRef]
- Brito, S.L.; Maia-Barbosa, P.M.; Pinto-Coelho, R.M. Zooplankton as an Indicator of Trophic Conditions in Two Large Reservoirs in Brazil. Lakes Reserv. 2011, 16, 253–264. [Google Scholar] [CrossRef]
- Litchman, E.; Ohman, M.D.; Kiørboe, T. Trait-Based Approaches to Zooplankton Communities. J. Plankton Res. 2013, 35, 473–484. [Google Scholar] [CrossRef]
- Barnett, A.J.; Finlay, K.; Beisner, B.E. Functional Diversity of Crustacean Zooplankton Communities: Towards a Trait-Based Classification. Freshw. Biol. 2007, 52, 796–813. [Google Scholar] [CrossRef]
- Castro, B.B.; Marques, S.M.; Gonçalves, F. Habitat Selection and Diel Distribution of the Crustacean Zooplankton from a Shallow Mediterranean Lake during the Turbid and Clear Water Phases. Freshw. Biol. 2007, 52, 421–433. [Google Scholar] [CrossRef]
- Kehayias, G.; Chalkia, E.; Chalkia, S.; Nistikakis, G.; Zacharias, I.; Zotos, A. Zooplankton Dynamics in the Upstream Part of Stratos Reservoir (Greece). Biologia 2008, 63, 699–710. [Google Scholar] [CrossRef]
- Kagalou, I.I.; Kosiori, A.; Leonardos, I.D. Assessing the Zooplankton Community and Environmental Factors in a Mediterranean Wetland. Environ. Monit. Assess 2010, 170, 445–455. [Google Scholar] [CrossRef]
- Jensen, T.C.; Dimante-Deimantovica, I.; Schartau, A.K.; Walseng, B. Cladocerans Respond to Differences in Trophic State in Deeper Nutrient Poor Lakes from Southern Norway. Hydrobiologia 2013, 715, 101–112. [Google Scholar] [CrossRef]
- Krupa, E.G.; Barinova, S.S.; Isbekov, K.B.; Assylbekova, S.Z. The Use of Zooplankton Distribution Maps for Assessment of Ecological Status of the Shardara Reservoir (Southern Kazakhstan). Ecohydrol. Hydrobiol. 2018, 18, 52–65. [Google Scholar] [CrossRef]
- Kiørboe, T. How Zooplankton Feed: Mechanisms, Traits and Trade-Offs. Biol. Rev. 2011, 86, 311–339. [Google Scholar] [CrossRef] [PubMed]
- Straile, D. Zooplankton Biomass Dynamics in Oligotrophic versus Eutrophic Conditions: A Test of the PEG Model. Freshw. Biol. 2015, 60, 174–183. [Google Scholar] [CrossRef]
- Beaver, J.R.; Miller-Lemke, A.M.; Acton, J.K. Midsummer Zooplankton Assemblages in Four Types of Wetlands in the Upper Midwest, USA. Hydrobiologia 1999, 380, 209–220. [Google Scholar] [CrossRef]
- Muñoz-Colmenares, M.E.; Soria, J.M.; Vicente, E. Can Zooplankton Species Be Used as Indicators of Trophic Status and Ecological Potential of Reservoirs? Aquat. Ecol. 2021, 55, 1143–1156. [Google Scholar] [CrossRef]
- Paes De-Carli, B.; Bressane, A.; Longo, R.M.; Manzi-Decarli, A.; Moschini-Carlos, V.; Martins Pompêo, M.L. Development of a Zooplankton Biotic Index for Trophic State Prediction in Tropical Reservoirs. Limnetica 2019, 38, 303–316. [Google Scholar] [CrossRef]
- Castro, A.; Rodrigues, V.; Vilaverde, A.; Gonçalves, F.; Silva, J.; Sopas, L.; Ferreira, M.; Sottomayor, M.; Costa, S. Plano Estratégico de Desenvolvimento Intermunicipal Do Tâmega e Sousa. Relatório Final; CEGEA: Lisboa, Portugal, 2014; p. 228. [Google Scholar]
- SNIRH: Sistema Nacional de Informação de Recursos Hídricos SNIRH: Sistema Nacional de Informação de Recursos Hídricos. Available online: https://snirh.apambiente.pt/index.php?idMain= (accessed on 24 September 2019).
- Agência Portuguesa do Ambiente Plano de Gestão Da Região Hidrográfica Do Douro RH3—Relatório Técnico; Comissão Europeia: Lisboa, Portugal, 2012.
- European Climate Adaptation Platform Climate-ADAPT Biogeophysical Characteristics. Available online: https://climate-adapt.eea.europa.eu/en/countries-regions/countries/portugal (accessed on 13 April 2023).
- Brower, J.E.; Zar, J.H.; von Ende, C.N. Field and Laboratory Methods for General Ecology, 4th ed.; WCB McGraw-Hill: Boston, MA, USA, 1997. [Google Scholar]
- Williamson, C.E.; Morris, D.P.; Pace, M.L.; Olson, O.G. Dissolved Organic Carbon and Nutrients as Regulators of Lake Ecosystems: Resurrection of a More Integrated Paradigm. Limnol. Oceanogr. 1999, 44, 795–803. [Google Scholar] [CrossRef]
- Rand, M.C.; Greenberg, A.E.; Taras, M.J. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1976; Volume 6. [Google Scholar]
- APHA. Standard Methods for the Examination of Water and Wastewater, 17th ed.; American Public Health Association: Washington, DC, USA, 1989; ISBN 978-0875532356. [Google Scholar]
- Instituto da Água. Manual Para a Avaliação Da Qualidade Biológica da Água em Lagos e Albufeiras Segundo a Diretiva Quadro da Água. Protocolo de Amostragem e Análise Para o Fitoplâncton; Ministério do Ambiente do Ordenamento do Território e do Desenvolvimento Regional: Lisboa, Portugal, 2009. [Google Scholar]
- Geller, W.; Müller, H. The Filtration Apparatus of Cladocera: Filter Mesh-Sizes and Their Implications on Food Selectivity. Oecologia 1981, 49, 316–321. [Google Scholar] [CrossRef]
- Alonso, M. Crustacea Branchiopoda. In Fauna Ibérica Vol. 7; Ramos, M.T., Ros, J., Noguera, X., Sierra, J., Mayol, A., Pierra, E., Fermin González, J., Eds.; Museo Nacional de Ciencias Naturales—CSIC: Madrid, Spain, 1996; p. 486. [Google Scholar]
- Witty, L. Practical Guide to Identifying Freshwater Crustacean Zooplankton, 2nd ed.; Cooperative Freshwater Ecology Unit: Sudbury, ON, Canada, 2004; Volume 60. [Google Scholar]
- Amoros, C. Introduction Pratique à La Systématique des Organismes des Eaux Continentales Françaises: Crustacés Cladocères; Université Claude Bernard: Lyon, France, 1984; Volume 53. [Google Scholar]
- Agência Portuguesa do Ambiente. Plano de Gestão de Região Hidrográfica—Parte 2—Caracterização e Diagnóstico—Anexos—Região Hidrográfica Do Douro (Rh3); Agência Portuguesa do Ambiente: Lisboa, Portugal, 2016. [Google Scholar]
- Agência Portuguesa do Ambiente. Critérios Para a Classificação Das Massas de Água; Agência Portuguesa do Ambiente: Lisboa, Portugal, 2021. [Google Scholar]
- Kassambara, A. Correlation Matrix: An R Function to Do All You Need. Statistical Tools for High-Throughput Data Analysis. 2022. Available online: sthda.com (accessed on 19 April 2023).
- Wei, T.; Simko, V. R Package “Corrplot”: Visualization of a Correlation Matrix (Version 0.92); R Core Team: Vienna, Austria, 2021. [Google Scholar]
- R Core Team, R. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022. [Google Scholar]
- RStudio Team. RStudio: Integrated Development Environment for R; RStudio Team: Vienna, Austria, 2022. [Google Scholar]
- Reid, J.W.; Williamson, C.E. Copepoda. Ecology and Classification of North American Freshwater Invertebrates; Elsevier: Amsterdam, The Netherlands, 2010; pp. 829–899. [Google Scholar]
- Agência Portuguesa do Ambiente, Plano de Gestão de Região Hidrográfica. 3° Ciclo 2022–2027, Douro (RH3). Parte 5, Objetivos, Anexo I—Sistematização Dos Objetivos Ambientais Por Massa de Água; Agência Portuguesa do Ambiente: Lisboa, Portugal, 2022. [Google Scholar]
- Agência Portuguesa do Ambiente, Plano de Gestão de Região Hidrográfica. Parte 5—Objetivos, Anexo II.3, Região Hidrográfica Do Douro (RH3); Agência Portuguesa do Ambiente: Lisboa, Portugal, 2016. [Google Scholar]
- Vale, S. Comunidades Planctónicas Da Albufeira Do Torrão. Eutrofização e Biodiversidade; Faculdade de Ciências da Universidade do Porto: Porto, Portugal, 2005. [Google Scholar]
- Pereira, E.; Teles, F.O.; Vasconcelos, V. Variation of Environmental Parameters and Dynamics of Phytoplankton in a Temperate Eutrophic Reservoir (Torrão, Tâmega River, Portugal). Fresenius Envrion. Bull. 2008, 17, 2193–2199. [Google Scholar]
- Bellinger, E.G.; Sigee, D.C. A Key to the more frequently occurring freshwater algae. In Freshwater Algae; John Wiley & Sons, Inc.: Hoboken, NJ, USA, UK, 2015; pp. 141–252. ISBN 9780470058145. [Google Scholar]
- Pinto, I.; Calisto, R.; Serra, C.R.; Lage, O.M.; Antunes, S.C. Bacterioplankton Community as a Biological Element for Reservoirs Water Quality Assessment. Water 2021, 13, 2836. [Google Scholar] [CrossRef]
- Rodrigues, S.; Pinto, I.; Formigo, N.; Antunes, S.C. Microalgae Growth Inhibition-Based Reservoirs Water Quality Assessment to Identify Ecotoxicological Risks. Water 2021, 13, 2605. [Google Scholar] [CrossRef]
- Teles, L.O.; Pereira, E.; Saker, M.; Vasconcelos, V. Virtual Experimentation on Cyanobacterial Bloom Dynamics and Its Application to a Temperate Reservoir. Lakes Reserv. 2008, 13, 135–143. [Google Scholar] [CrossRef]
- Regueiras, A.V.B. Comparação de Métodos de Identificação e Quantificação de Cianobactérias e Suas Toxinas Na Albufeira Do Torrão (Rio Tâmega); Faculdade de Ciências da Universidade do Porto: Porto, Portugal, 2009. [Google Scholar]
- Cabecinha, E.; Cortes, R.; Alexandre Cabral, J.; Ferreira, T.; Lourenço, M.; Pardal, M. Multi-Scale Approach Using Phytoplankton as a First Step towards the Definition of the Ecological Status of Reservoirs. Ecol. Indic. 2009, 9, 240–255. [Google Scholar] [CrossRef]
- Stamou, G.; Katsiapi, M.; Moustaka-Gouni, M.; Michaloudi, E. The Neglected Zooplankton Communities as Indicators of Ecological Water Quality of Mediterranean Lakes. Limnetica 2021, 40, 359–373. [Google Scholar] [CrossRef]
- Muñoz-Colmenares, M.E.; Sendra, M.D.; Sòria-Perpinyà, X.; Soria, J.M.; Vicente, E. The Use of Zooplankton Metrics to Determine the Trophic Status and Ecological Potential: An Approach in a Large Mediterranean Watershed. Water 2021, 13, 2382. [Google Scholar] [CrossRef]
- Abrantes, N.; Antunes, S.C.; Pereira, M.J.; Gonçalves, F. Seasonal Succession of Cladocerans and Phytoplankton and Their Interactions in a Shallow Eutrophic Lake (Lake Vela, Portugal). Acta Oecologica 2006, 29, 54–64. [Google Scholar] [CrossRef]
- Allan, J.D. Life History Patterns in Zooplankton. Am. Nat. 1976, 110, 165–180. [Google Scholar] [CrossRef]
- Hessen, D.O.; Faafeng, B.A.; Smith, V.H.; Bakkestuen, V.; Walseng, B. Extrinsic and Intrinsic Controls of Zooplankton Diversity in Lakes. Ecology 2006, 87, 433–443. [Google Scholar] [CrossRef]
- Haberman, J.; Haldna, M. Indices of Zooplankton Community as Valuable Tools in Assessing the Trophic State and Water Quality of Eutrophic Lakes: Long Term Study of Lake Võrtsjärv. J. Limnol. 2014, 73, 61–71. [Google Scholar] [CrossRef]
- Pereira Collares, M.J. Guia Dos Peixes de Água Doce e Migradores de Portugal Continental, 1st ed.; Afrontamento: Lisboa, Portugal, 2021; ISBN 978-972-36-1849-5. [Google Scholar]
- Montagud, D.; Soria, J.M.; Soria-Perpinyà, X.; Alfonso, T.; Vicente, E. A Comparative Study of Four Indexes Based on Zooplankton as Trophic State Indicators in Reservoirs. Limnetica 2019, 38, 291–302. [Google Scholar] [CrossRef]
- Almeida, R.; Sousa Pinto, I.; Antunes, S.C. Contribution of Zooplankton as a Biological Element in the Assessment of Reservoir Water Quality. Limnetica 2020, 39, 245–261. [Google Scholar] [CrossRef]
- García-Chicote, J.; Armengol, X.; Rojo, C. Zooplankton Species as Indicators of Trophic State in Reservoirs from Mediterranean River Basins. Inland Waters 2019, 9, 113–123. [Google Scholar] [CrossRef]
- Gomes, L.F.; Pereira, H.R.; Gomes, A.C.A.M.; Vieira, M.C.; Martins, P.R.; Roitman, I.; Vieira, L.C.G. Zooplankton Functional-Approach Studies in Continental Aquatic Environments: A Systematic Review. Aquat. Ecol. 2019, 53, 191–203. [Google Scholar] [CrossRef]
- Korponai, J.; Braun, M.; Forró, L.; Gyulai, I.; Kövér, C.; Nédli, J.; Urák, I.; Buczkó, K. Taxonomic, Functional and Phylogenetic Diversity: How Subfossil Cladocerans Mirror Contemporary Community for Ecosystem Functioning: A Comparative Study in Two Oxbows. Limnetica 2019, 38, 431–456. [Google Scholar] [CrossRef]
| Second Cycle of WFD | Third Cycle of WFD | ||||||
|---|---|---|---|---|---|---|---|
| Physical and Chemical Classification | Biological Classification | Final Classification | Physical and Chemical Classification | Biological Classification | Final Classification | ||
| T1 | Summer | Moderate | Moderate | Moderate | Moderate | Poor | Poor |
| Autumn | Moderate | Moderate | Moderate | Moderate | Poor | Poor | |
| Winter | Good | Good or more | Good | Moderate | Moderate | Moderate | |
| Spring | Good | Good or more | Good | Moderate | Good | Moderate | |
| T2 | Summer | Moderate | Moderate | Moderate | Moderate | Poor | Poor |
| Autumn | Moderate | Good or more | Moderate | Moderate | Moderate | Moderate | |
| Winter | Good | Good or more | Good | Moderate | Good | Moderate | |
| Spring | Good | Moderate | Moderate | Moderate | Poor | Poor | |
| T3 | Summer | Moderate | Moderate | Moderate | Moderate | Moderate | Moderate |
| Autumn | Moderate | Good or more | Moderate | Moderate | Moderate | Moderate | |
| Winter | Moderate | Good or more | Moderate | Moderate | Good | Moderate | |
| Spring | Good | Poor | Poor | Moderate | Poor | Poor | |
| T4 | Summer | Moderate | Good or more | Moderate | Moderate | Moderate | Moderate |
| Autumn | Moderate | Good or more | Moderate | Moderate | Moderate | Moderate | |
| Winter | Good | Good or more | Good | Moderate | Excellent | Moderate | |
| Spring | Good | Poor | Poor | Moderate | Poor | Poor | |
| T5 | Summer | Moderate | Good or more | Moderate | Moderate | Moderate | Moderate |
| Autumn | Moderate | Moderate | Moderate | Moderate | Moderate | Moderate | |
| Winter | Good | Good or more | Good | Moderate | Good | Moderate | |
| Spring | Good | Moderate | Moderate | Moderate | Poor | Poor | |
| Site | Season | Cyclopoida | Calanoida | Chydorus | Pleuroxus | Alonella | Alona | Acroperus | Monospilus | Bosmina | Ceriodaphnia | Scapholeberis | Daphnia | Sida | Diaphanosoma | Abundance | Diversity (Shannon—Wiener) | Evenness (Pielou) | Large Cladocera/ Total Cladocera | Calanoida/ Cyclopoida |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T1 | Summer | 3611 | 159 | 1097 | 14 | 474 | 438 | 1 | 11 | 79 | 5884 | 1.19 | 0.54 | 4.30 | 0.04403 | |||||
| Autumn | 2884 | 404 | 877 | 1 | 56 | 2946 | 10 | 47 | 125 | 601 | 7951 | 1.47 | 0.64 | 16.58 | 0.14008 | |||||
| Winter | 8 | 17 | 61 | 5 | 3 | 94 | 1.07 | 0.66 | ||||||||||||
| Spring | 7395 | 1 | 2 | 1 | 22 | 4 | 494 | 4 | 695 | 1 | 8619 | 0.53 | 0.23 | 56.91 | 0.00014 | |||||
| T2 | Summer | 373 | 167 | 287 | 1 | 32 | 66 | 127 | 175 | 1228 | 1.74 | 0.84 | 43.90 | 0.44772 | ||||||
| Autumn | 314 | 226 | 279 | 1 | 3 | 531 | 9 | 4 | 29 | 68 | 1464 | 1.59 | 0.69 | 10.93 | 0.71975 | |||||
| Winter | 33 | 2 | 23 | 1 | 20 | 79 | 1.22 | 0.76 | ||||||||||||
| Spring | 9521 | 1 | 4 | 16 | 6 | 2495 | 2 | 24 | 2 | 12,071 | 0.54 | 0.25 | 1.02 | 0.00011 | ||||||
| T3 | Summer | 194 | 346 | 1005 | 2 | 210 | 384 | 1 | 22 | 366 | 862 | 35 | 3427 | 1.84 | 0.77 | 31.07 | 1.78351 | |||
| Autumn | 53 | 69 | 46 | 2 | 1 | 1 | 54 | 3 | 1421 | 84 | 1734 | 0.78 | 0.34 | 93.36 | 1.30189 | |||||
| Winter | 107 | 5 | 4 | 2 | 118 | 0.41 | 0.29 | |||||||||||||
| Spring | 7284 | 2 | 20 | 26 | 5 | 260 | 26 | 82 | 6 | 7711 | 0.28 | 0.13 | 20.71 | 0.00027 | ||||||
| T4 | Summer | 392 | 2126 | 309 | 5 | 4 | 7 | 697 | 13 | 1139 | 3741 | 1.27 | 0.58 | 16.44 | 5.42347 | |||||
| Autumn | 90 | 109 | 4 | 1 | 2 | 1 | 10 | 2651 | 4 | 1493 | 39 | 4470 | 0.97 | 0.40 | 37.51 | 1.21111 | ||||
| Winter | 40 | 5 | 2 | 29 | 76 | 0.98 | 0.71 | |||||||||||||
| Spring | 2388 | 3 | 6 | 2 | 20 | 8 | 81 | 15 | 10 | 17 | 5 | 7 | 2560 | 0.37 | 0.15 | 15.98 | 0.00126 | |||
| T5 | Summer | 581 | 6159 | 535 | 1 | 10 | 6 | 9 | 3984 | 21 | 188 | 12,445 | 1.24 | 0.54 | 20.33 | 10.60069 | ||||
| Autumn | 88 | 87 | 1 | 7 | 291 | 7 | 136 | 105 | 656 | 1.50 | 0.72 | 37.84 | 0.98864 | |||||||
| Winter | 27 | 13 | 5 | 4 | 1 | 50 | 1.19 | 0.74 | 4.35 | |||||||||||
| Spring | 3573 | 26 | 1 | 36 | 8 | 35 | 19 | 7 | 9 | 2 | 5 | 3723 | 0.25 | 0.10 | 12.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinto, I.; Nogueira, S.; Rodrigues, S.; Formigo, N.; Antunes, S.C. Can Zooplankton Add Value to Monitoring Water Quality? A Case Study of a Meso/Eutrophic Portuguese Reservoir. Water 2023, 15, 1678. https://doi.org/10.3390/w15091678
Pinto I, Nogueira S, Rodrigues S, Formigo N, Antunes SC. Can Zooplankton Add Value to Monitoring Water Quality? A Case Study of a Meso/Eutrophic Portuguese Reservoir. Water. 2023; 15(9):1678. https://doi.org/10.3390/w15091678
Chicago/Turabian StylePinto, Ivo, Sandra Nogueira, Sara Rodrigues, Nuno Formigo, and Sara C. Antunes. 2023. "Can Zooplankton Add Value to Monitoring Water Quality? A Case Study of a Meso/Eutrophic Portuguese Reservoir" Water 15, no. 9: 1678. https://doi.org/10.3390/w15091678
APA StylePinto, I., Nogueira, S., Rodrigues, S., Formigo, N., & Antunes, S. C. (2023). Can Zooplankton Add Value to Monitoring Water Quality? A Case Study of a Meso/Eutrophic Portuguese Reservoir. Water, 15(9), 1678. https://doi.org/10.3390/w15091678








