Evidence for Groundwater Salinity Origin Based on Hydrogeochemical and Isotopic (2H, 18O, 37Cl, 3H, 13C, 14C) Approaches: Sousse, Eastern Tunisia
Abstract
:1. Introduction
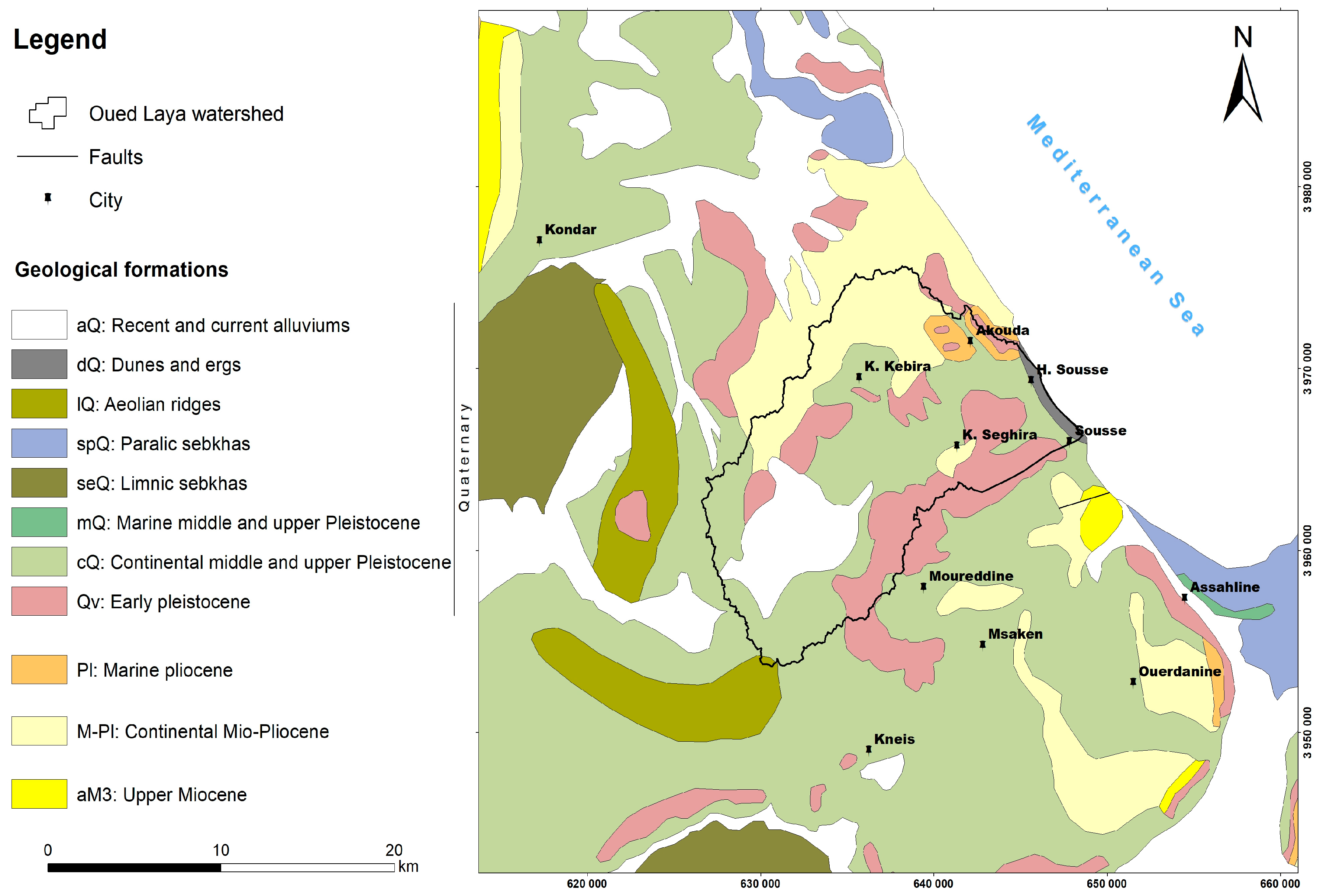
2. Main Features of the Study Area
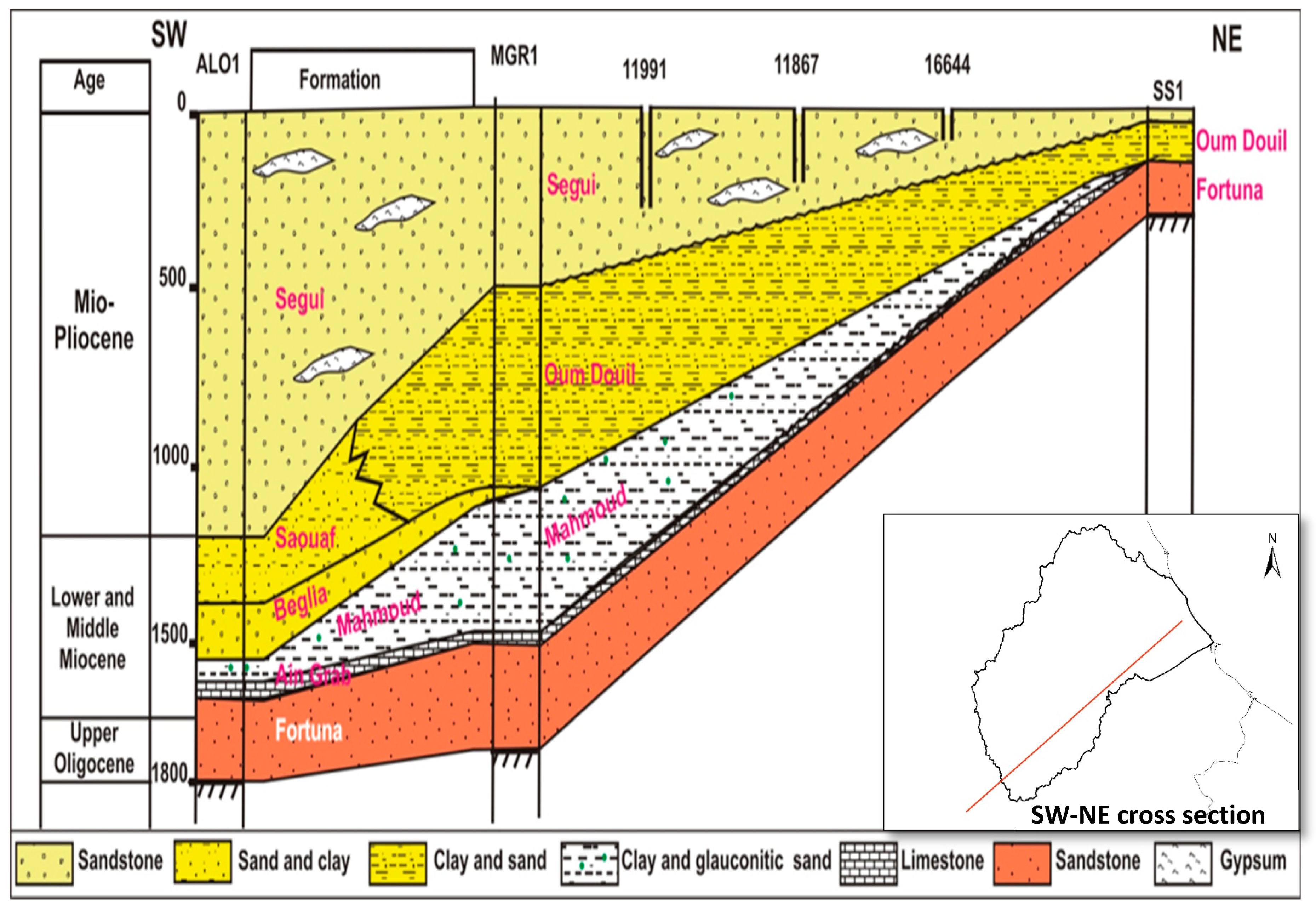
3. Geological and Hydrogeological Setting
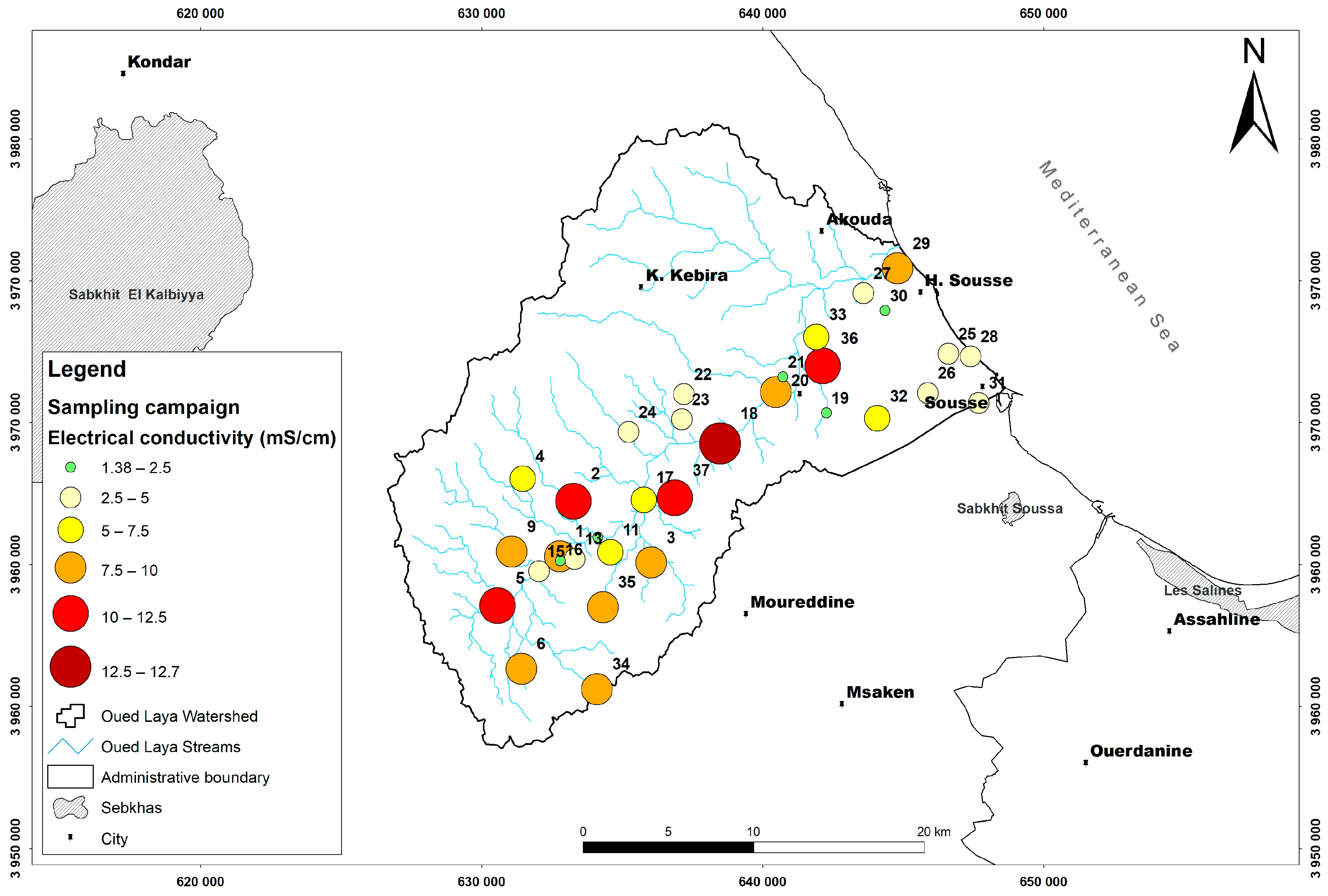
4. Material and Methods
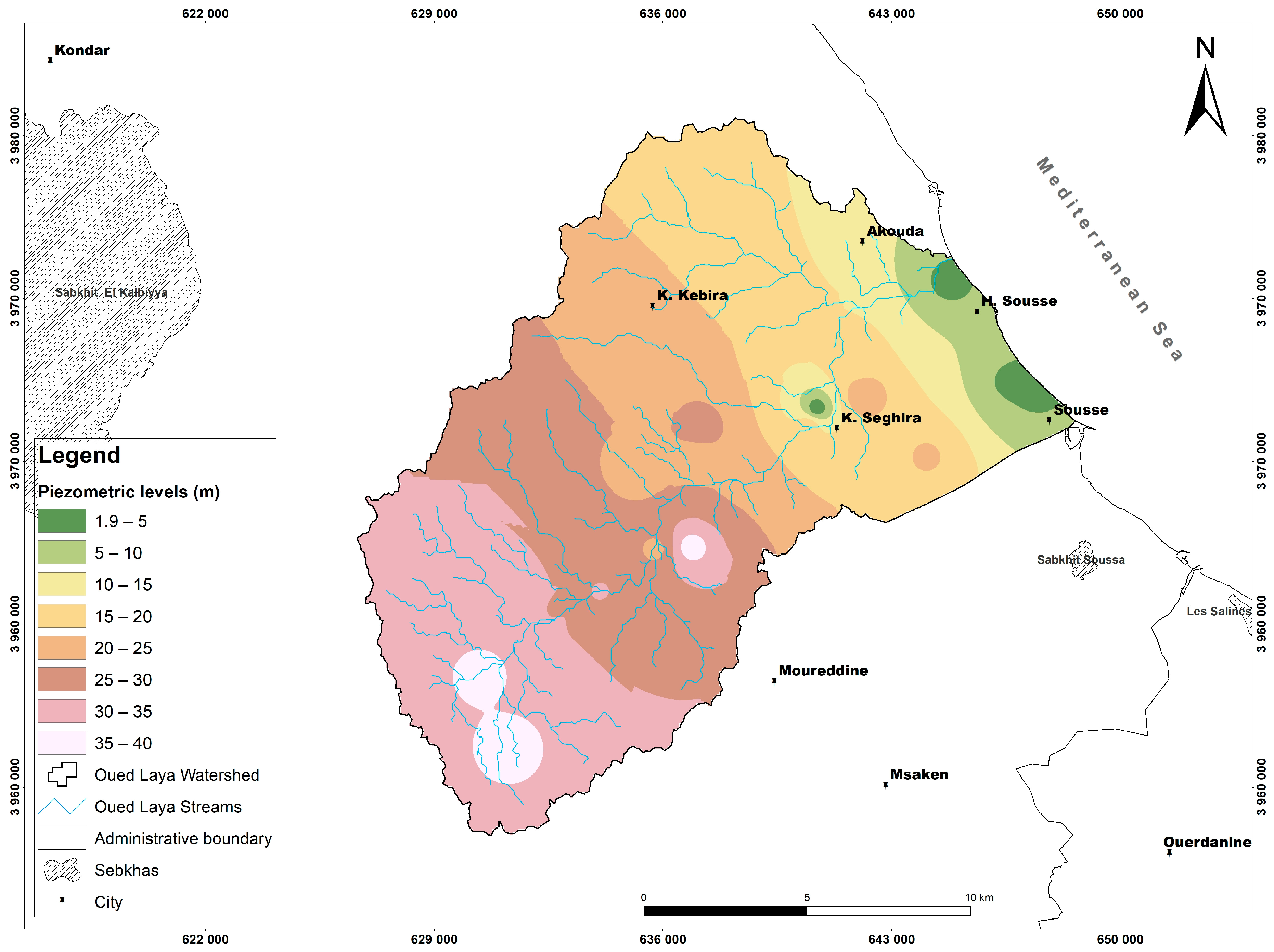
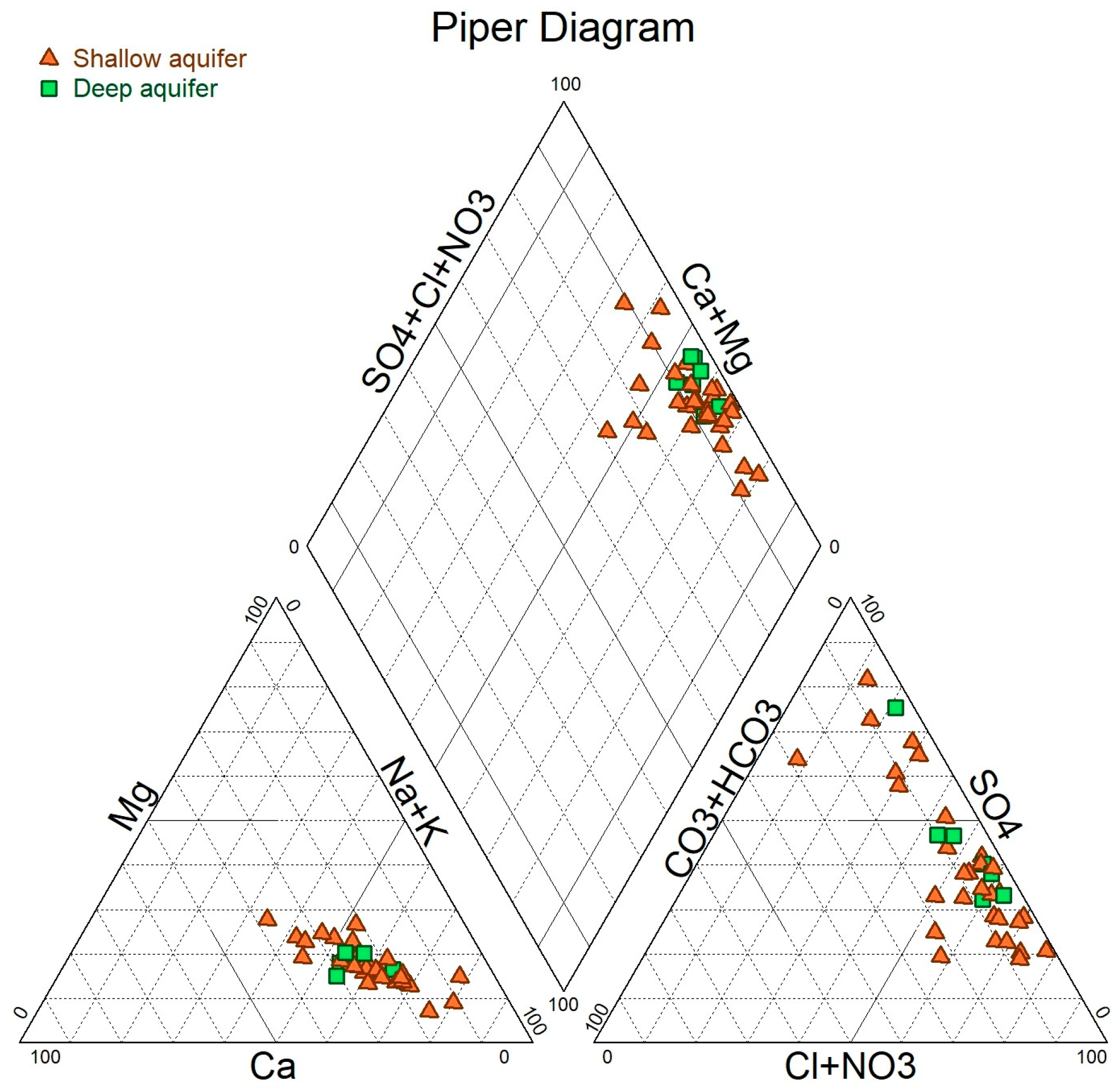
5. Results and Discussion
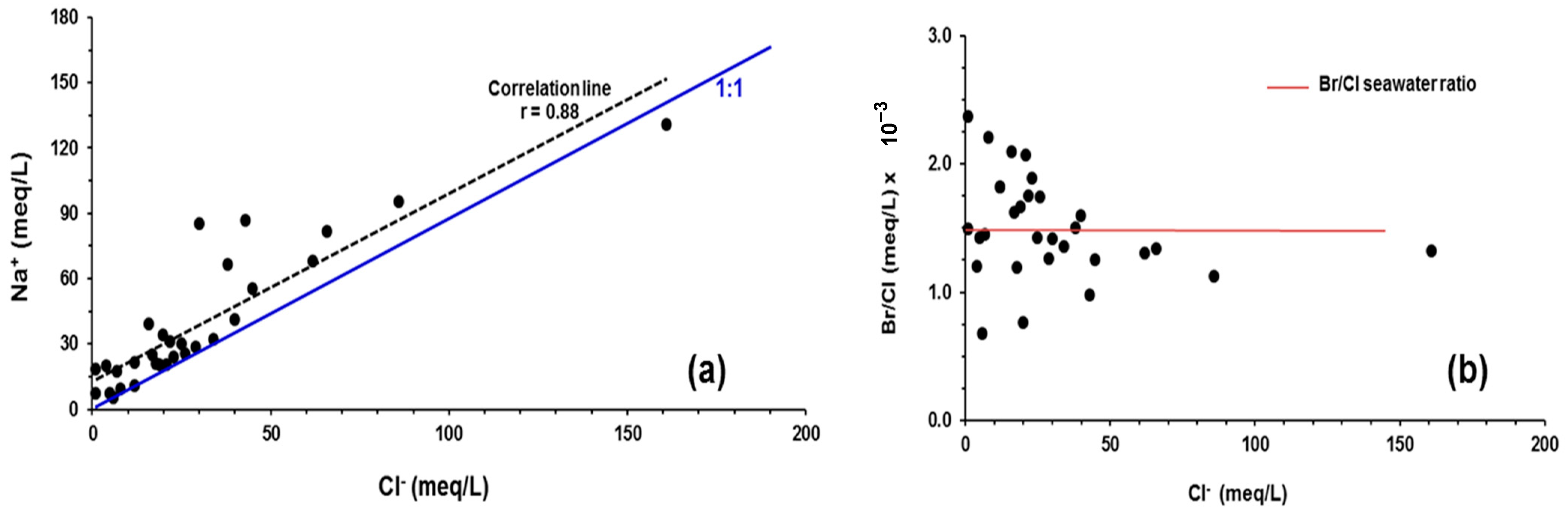
5.1. Na+/Cl− Correlation
5.2. Br−/Cl− Relationship
5.3. Ca2+/Mg2+ Relationship
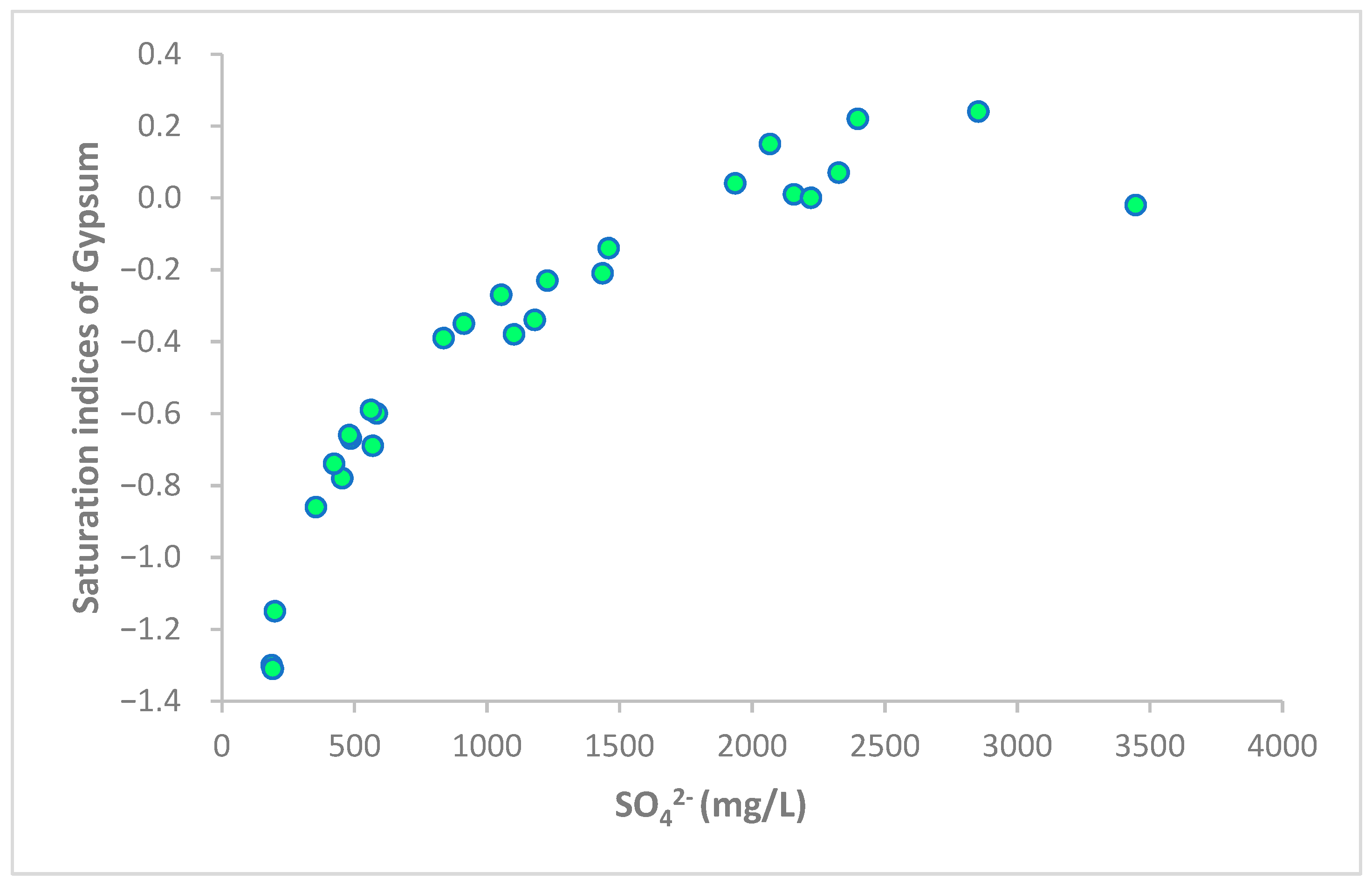
5.4. Ca2+/SO42− Relationship
5.5. Saturation Indices (SI)
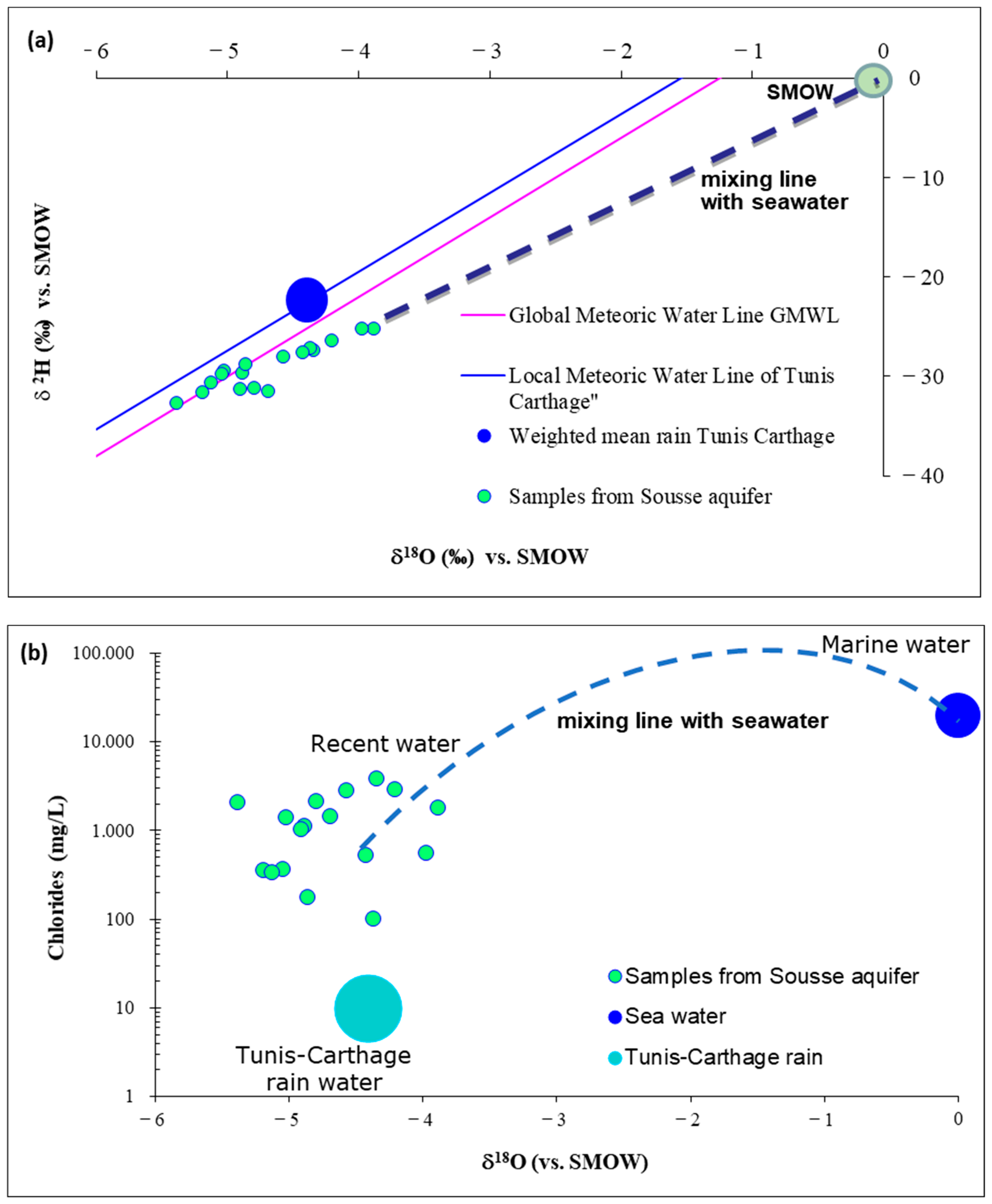
5.6. Stable Isotopes (δ2H, δ18O)
5.7. Chlorine Isotope
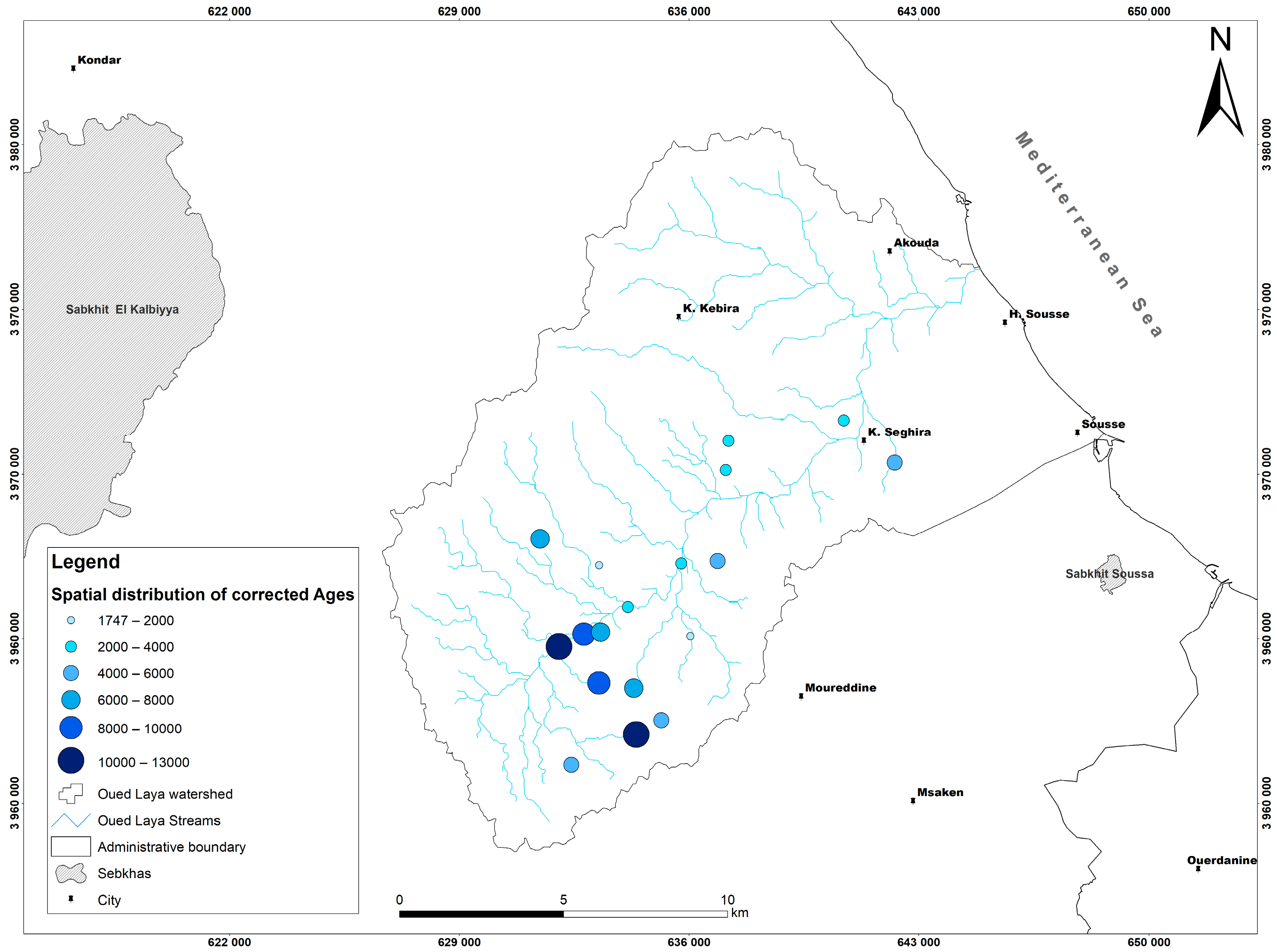
5.8. Groundwater Residence Time
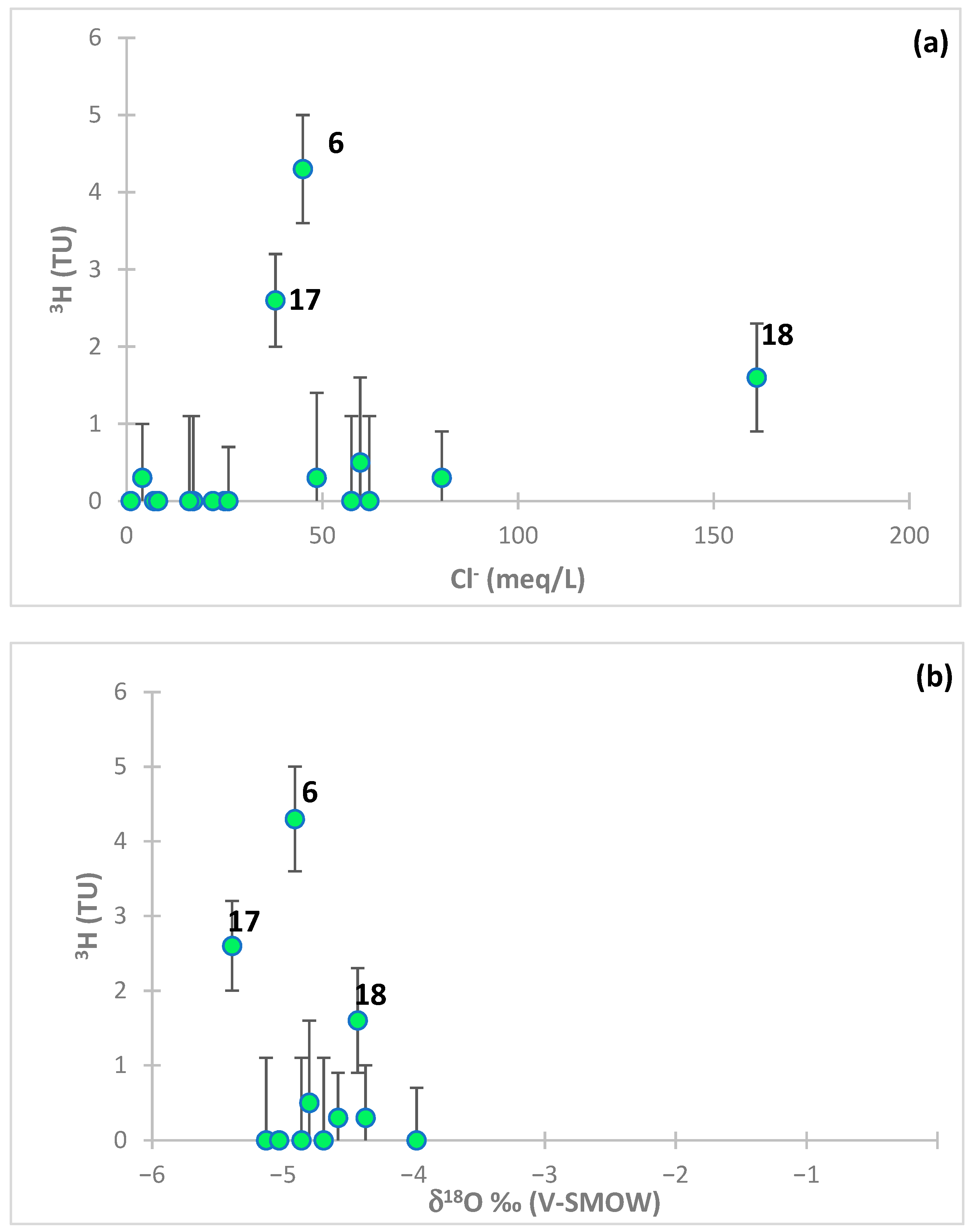
5.8.1. Tritium
5.8.2. Carbon 14 and δ13C
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kloppmann, W.; Bourhane, A.; Schomburgk, S. Groundwater Salinization in France. Procedia Earth Planet. Sci. 2013, 7, 440–443. [Google Scholar] [CrossRef] [Green Version]
- Ben Hamouda, M.F.; Tarhouni, J.; Leduc, C.; Zouari, K. Understanding the Origin of Salinization of the Plio-Quaternary Eastern Coastal Aquifer of Cap Bon (Tunisia) Using Geochemical and Isotope Investigations. Environ. Earth Sci. 2011, 63, 889–901. [Google Scholar] [CrossRef]
- Gaaloul, N.; Cheng, A. Hydrogeological and Hydrochemical Investigation of Coastal Aquifers in Tunisia-Crisis in Overex-ploitation and Salinization. In Proceedings of the 2nd International Conference on Saltwater Intrusion and Coastal Aquifers: Montoring, Modelling, and Management, Merida, Mexico, 30 March–2 April 2003; p. 13. [Google Scholar]
- Fontes, J.-C. Les Isotopes Du Milieu Dans Les Eaux Naturelles. La Houille Blanche 1976, 62, 205–221. [Google Scholar] [CrossRef] [Green Version]
- Hamouda, M.F.B.; Carreira, P.; Marques, J.M.; Egenkamp, H. Geochemical and Isotopic Investigations to Study the Origin of Mineralization of the Coastal Aquifer of Sousse, Tunisia. Procedia Earth Planet. Sci. 2013, 7, 61–64. [Google Scholar] [CrossRef] [Green Version]
- Ben Hamouda, M.F. Hydrogeological, Geochemical and Isotopic Approach of the Coastal Aquifers Systems of Cap Bon: Case of the Eastern Coastal and El Haouaria Aquifer, Tunisia. Ph.D. Thesis, National Agronomic Institute of Tunisia, Tunis, Tunisia, 2008. [Google Scholar]
- Hamouda, M.F.B.; Kondash, A.J.; Lauer, N.; Mejri, L.; Tarhouni, J.; Vengosh, A. Assessment of Groundwater Salinity Mech-anisms in the Coastal Aquifer of El Haouaria, Northern Tunisia. Procedia Earth Planet. Sci. 2015, 13, 194–198. [Google Scholar] [CrossRef] [Green Version]
- Tarhouni, J.; Hamouda, M.F.B.; Kondash, A.J.; Mejri, L.; Vengosh, A. Modeling the Recharge and the Renewal Rate Based on 3H and 14C Isotopes in the Coastal Aquifer of El Haouaria, Northern Tunisia. Procedia Earth Planet. Sci. 2015, 13, 199–202. [Google Scholar] [CrossRef] [Green Version]
- Ben Hamouda, M.F. Study of the Groundwater Salinity and Modeling the Recharge and the Renewal Rate Based on Stable and Radioactive Isotopes in El Haouaria Aquifer, Tunisia. Arab. J. Geosci. 2021, 14, 2246. [Google Scholar] [CrossRef]
- Jones, B.F.; Vengosh, A.; Rosenthal, E.; Yechieli, Y. Geochemical Investigations. In Seawater Intrusion in Coastal Aquifers—Concepts, Methods and Practices; Bear, J., Cheng, A.H.-D., Sorek, S., Ouazar, D., Herrera, I., Eds.; Theory and Applications of Transport in Porous Media; Springer: Dordrecht, The Netherlands, 1999; Volume 14, pp. 51–71. ISBN 978-90-481-5172-1. [Google Scholar]
- Vengosh, A. 11.9—Salinization and Saline Environments. In Treatise on Geochemistry, 2nd ed.; Holland, H.D., Turekian, K.K., Eds.; Elsevier: Oxford, UK, 2014; pp. 325–378. ISBN 978-0-08-098300-4. [Google Scholar]
- Stotler, R.L.; Frape, S.K.; Shouakar-Stash, O. An Isotopic Survey of Δ81Br and Δ37Cl of Dissolved Halides in the Canadian and Fennoscandian Shields. Chem. Geol. 2010, 274, 38–55. [Google Scholar] [CrossRef]
- Aggarwal, P.K.; Froehlich, K.; Gonfiantini, R. Contributions of the International Atomic Energy Agency to the Development and Practice of Isotope Hydrology. Hydrogeol. J. 2011, 19, 5–8. [Google Scholar] [CrossRef]
- Holser, W.T.; Kaplan, I.R.; Sakai, H.; Zak, I. Isotope Geochemistry of Oxygen in the Sedimentary Sulfate Cycle. Chem. Geol. 1979, 25, 1–17. [Google Scholar] [CrossRef]
- Clayton, R.N.; Friedman, I.; Graf, D.L.; Mayeda, T.K.; Meents, W.F.; Shimp, N.F. The Origin of Saline Formation Waters: 1. Isotopic Composition. J. Geophys. Res. 1966, 71, 3869–3882. [Google Scholar] [CrossRef]
- Eggenkamp, H.G.M. δ37Cl: The Geochemistry of Chlorine Isotopes. Ph.D. Thesis, Faculteit Aardwetenschappen, Utrecht, The Netherlands, 1994. [Google Scholar]
- Eggenkamp, H.G.M.; Coleman, M.L. Heterogeneity of Formation Waters within and between Oil Fields by Halogen Isotopes. In Proceedings of the 9th International Sympsium on Water-Rock Interaction, WRI 9, Taupo, New Zealand, 30 March–3 April 1998; Volume 9, pp. 309–312. [Google Scholar]
- Ferchichi, H.; Farhat, B.; Ben-Hamouda, M.F.; Ben-Mammou, A. Understanding Groundwater Chemistry in Mediterranean Semi-Arid System Using Multivariate Statistics Techniques and GIS Methods: Case of Manouba Aquifer (Northeastern Tuni-sia). Arab. J. Geosci. 2017, 10, 530. [Google Scholar] [CrossRef]
- Ferchichi, H.; Ben Hamouda, M.F.; Farhat, B.; Ben Mammou, A. Assessment of Groundwater Salinity Using GIS and Multi-variate Statistics in a Coastal Mediterranean Aquifer. Int. J. Environ. Sci. Technol. 2018, 15, 2473–2492. [Google Scholar] [CrossRef]
- Bouchaou, L.; Michelot, J.L.; Vengosh, A.; Hsissou, Y.; Qurtobi, M.; Gaye, C.B.; Bullen, T.D.; Zuppi, G.M. Application of Mul-tiple Isotopic and Geochemical Tracers for Investigation of Recharge, Salinization, and Residence Time of Water in the Souss–Massa Aquifer, Southwest of Morocco. J. Hydrol. 2008, 352, 267–287. [Google Scholar] [CrossRef]
- Vespasiano, G.; Cianflone, G.; Marini, L.; De Rosa, R.; Polemio, M.; Walraevens, K.; Vaselli, O.; Pizzino, L.; Cinti, D.; Capec-chiacci, F.; et al. Hydrogeochemical and Isotopic Characterization of the Gioia Tauro Coastal Plain (Calabria-Southern Italy): A Multidisciplinary Approach for a Focused Management of Vulnerable Strategic Systems. Sci. Total Environ. 2023, 862, 160694. [Google Scholar] [CrossRef]
- Vespasiano, G.; Cianflone, G.; Romanazzi, A.; Apollaro, C.; Dominici, R.; Polemio, M.; De Rosa, R. A Multidisciplinary Ap-proach for Sustainable Management of a Complex Coastal Plain: The Case of Sibari Plain (Southern Italy). Mar. Pet. Geol. 2019, 109, 740–759. [Google Scholar] [CrossRef]
- Ben Hamouda, M.F.; Carreira, P.M.; Marques, J.M.; Eggenkamp, H. Multi-Isotope Approach to Study the Problem of Salinity in the Coastal Aquifer of Oued Laya, Tunisia. In Advances in Sustainable and Environmental Hydrology, Hydrogeology, Hydrochemistry and Water Resources; Chaminé, H.I., Barbieri, M., Kisi, O., Chen, M., Merkel, B.J., Eds.; Advances in Science, Technology & Innovation; Springer International Publishing: Cham, Switzerland, 2019; pp. 203–206. ISBN 978-3-030-01571-8. [Google Scholar]
- Dridi, L.; Majdoub, R.; Ghorbel, F.; Ben Hlima, M. Characterization of Water and Sediment Quality of Oued Laya (Sousse/Tunisia). J. Mater Environ. Sci 2014, 5, 1500–1504. [Google Scholar]
- El Amrı, A.; Anane, M.; Drıdı, L.; Srasra, M.; Majdoub, R. A GIS Based DRASTIC, Pesticide DRASTIC and SI Methods to Assess Groundwater Vulnerability to Pollution: Case Study of Oued Laya (Central Tunisia). In Environmental Remote Sensing and GIS in Tunisia; Khebour Allouche, F., Negm, A.M., Eds.; Springer Water; Springer International Publishing: Cham, Switzerland, 2021; pp. 143–163. ISBN 978-3-030-63667-8. [Google Scholar]
- Carreira, P.M.; Bahir, M.; Salah, O.; Galego Fernandes, P.; Nunes, D. Tracing Salinization Processes in Coastal Aquifers Using an Isotopic and Geochemical Approach: Comparative Studies in Western Morocco and Southwest Portugal. Hydrogeol. J. 2018, 26, 2595–2615. [Google Scholar] [CrossRef]
- Eggenkamp, H. Theoretical and Experimental Fractionation Studies of Chloride and Bromide Isotopes. In The Geochemistry of Stable Chlorine and Bromine Isotopes; Advances in Isotope Geochemistry; Springer: Berlin/Heidelberg, Germany, 2014; pp. 75–93. ISBN 978-3-642-28505-9. [Google Scholar]
- Riou, C. A simple empiric formula for estimating potential evapotranspiration in Tunisia. Cah. ORSTOM Ser. Hydrol. 1980, 2, 129–137. [Google Scholar]
- DGRE “General Directorate of Water Resources, Tunisia”. In Directory of Groundwater Quality in Tunisia; Ministry of Agriculture, Water Resources and Fisheries: Tunis, Tunisia, 2004; p. 385. (In French)
- Khomsi, S.; Bédir, M.; Ben Jemia, M.G.; Zouari, H. Discovery of a new thrust front in the oriental Atlas of Tunisia using seis-mic reflection data. Structural context and role of salt intrusions. C. R. Geosci. 2004, 336, 1401–1408. [Google Scholar] [CrossRef]
- Khomsi, S.; Echihi, O.; Slimani, N. Structural Control on the Deep Hydrogeological and Geothermal Aquifers Related to the Fractured Campanian-Miocene Reservoirs of North-Eastern Tunisia Foreland Constrained by Subsurface Data. C. R. Geosci. 2012, 344, 247–265. [Google Scholar] [CrossRef]
- Manaa, M. Groundwater Resources in the Gouvernorate of Sousse and Hydric Pollution; Ministry of Agriculture, DGRE: Tunis, Tunisia, 1994; p. 53. [Google Scholar]
- Amri, R. Note on Implementation of a Drilling Well in Oued Laya, Sousse; Ministry of Agriculture, DGRE: Tunis, Tunisia, 1995. [Google Scholar]
- Khammasi, M. Contribution of Isotope Geochemistry to Study the Oued Laya Aquifer, Sousse. Master’s Thesis, Jendouba University, Tunis, Tunisia, 2013. [Google Scholar]
- Batti, D.; Mansour, H.; Alayet, H. Map of Water Resources in Tunisia, Region of Sousse and El Jem in 1/200,000; Ministry of Agriculture, DGRE: Tunis, Tunisia, 1976. [Google Scholar]
- IAEA Sampling Procedures for Isotope Hydrology. Available online: https://www-naweb.iaea.org/napc/ih/documents/other/Sampling%20booklet%20web.pdf (accessed on 15 November 2022).
- Epstein, S.; Mayeda, T. Variation of O18 Content of Waters from Natural Sources. Geochim. Cosmochim. Acta 1953, 4, 213–224. [Google Scholar] [CrossRef]
- Coleman, M.L.; Shepherd, T.J.; Durham, J.J.; Rouse, J.E.; Moore, G.R. Reduction of Water with Zinc for Hydrogen Isotope Analysis. Anal. Chem. 1982, 54, 993–995. [Google Scholar] [CrossRef]
- Coplen, T.B. Normalization of Oxygen and Hydrogen Isotope Data. Chem. Geol. Isot. Geosci. Sect. 1988, 72, 293–297. [Google Scholar] [CrossRef]
- Taylor, C.B. Tritium Enrichment of Environmental Waters by Electrolysis: Development of Cathodes Exhibiting High Isotopic Separation and Precise Measurement of Tritium Enrichment Factors; International Atomic Energy Agency (IAEA): Vienna, Austria, 1976; pp. 32–42. [Google Scholar]
- Taylor, C.B.; Schwarz, P. Deionization Procedure for Large Quantities of Tritium-Free Water Using an Ion-Exchange Column. Int. J. Appl. Radiat. Isot. 1977, 28, 605–606. [Google Scholar] [CrossRef]
- Fontes, J.-C. A Unit Intended for the Measurement of the Activity of Natural Radiocarbon by Liquid Scintillation. Rev. Geogr. Phys. Geol. Dyn. 1971, 13, 67–86. [Google Scholar]
- Godon, A.; Jendrzejewski, N.; Eggenkamp, H.G.M.; Banks, D.A.; Ader, M.; Coleman, M.L.; Pineau, F. A Cross-Calibration of Chlorine Isotopic Measurements and Suitability of Seawater as the International Reference Material. Chem. Geol. 2004, 207, 1–12. [Google Scholar] [CrossRef]
- Piper, A.M. A Graphic Procedure in the Geochemical Interpretation of Water-Analyses. Trans. AGU 1944, 25, 914. [Google Scholar] [CrossRef]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater and Pollution; Appelo, C.A.J., Postma, D., Eds.; Balkema: Rotterdam, The Netherlands, 1994. [Google Scholar]
- Re, V.; Sacchi, E.; Mas-Pla, J.; Menció, A.; El Amrani, N. Identifying the Effects of Human Pressure on Groundwater. Quality to Support Water Management Strategies in Coastal Regions: A Multi-Tracer and Statistical Approach (Bouareg Region, Morocco). Sci. Total Environ. 2014, 500–501, 211–223. [Google Scholar] [CrossRef] [Green Version]
- Carreira, P.M.; Lobo de Pina, A.; da Mota Gomes, A.; Marques, J.M.; Monteiro Santos, F. Radiocarbon Dating and Stable Isotopes Content in the Assessment of Groundwater Recharge at Santiago Island, Republic of Cape Verde. Water 2022, 14, 2339. [Google Scholar] [CrossRef]
- Simler, R. Diagrammes: Free Distribution Multi-Language Hydrochemistry Software; Hydrogeology Laboratory of Avignon, Avignon University: Avignon, France, 2009. [Google Scholar]
- Rashad, M.; Dultz, S.; Guggenberger, G. Dissolved Organic Matter Release and Retention in an Alkaline Soil from the Nile River Delta in Relation to Surface Charge and Electrolyte Type. Geoderma 2010, 158, 385–391. [Google Scholar] [CrossRef]
- Abdulameer, A.; Thabit, J.M.; Kanoua, W.; Wiche, O.; Merkel, B. Possible Sources of Salinity in the Upper Dibdibba Aquifer, Basrah, Iraq. Water 2021, 13, 578. [Google Scholar] [CrossRef]
- Yurtsever, Y. Role of Environmental Isotopes in Studies Related to Salinization Processes and Salt Water Intrusion Dynamics. In Proceedings of the 13th Saltwater Intrusion Meeting, Cagliari, Italy, 5–10 June 1994; pp. 177–185. [Google Scholar]
- Modibo Sidibé, A.; Lin, X.; Koné, S. Assessing Groundwater Mineralization Process, Quality, and Isotopic Recharge Origin in the Sahel Region in Africa. Water 2019, 11, 789. [Google Scholar] [CrossRef] [Green Version]
- Sankoh, A.A.; Derkyi, N.S.A.; Frazer-williams, R.A.D.; Laar, C.; Kamara, I. A Review on the Application of Isotopic Techniques to Trace Groundwater Pollution Sources within Developing Countries. Water 2022, 14, 35. [Google Scholar] [CrossRef]
- Carreira, P.M.; Marques, J.M.; Nunes, D. Source of Groundwater Salinity in Coastline Aquifers Based on Environmental Isotopes (Portugal): Natural vs. Human Interference. A Review and Reinterpretation. Appl. Geochem. 2014, 41, 163–175. [Google Scholar] [CrossRef]
- Kammoun, S.; Re, V.; Trabelsi, R.; Zouari, K.; Daniele, S. Assessing seasonal variations and aquifer vulnerability in coastal aquifers of semi-arid regions using a multi-tracer isotopic approach: The case of Grombalia (Tunisia). Hydro Geol. J. 2018, 26, 2575–2594. [Google Scholar] [CrossRef]
- GNIP. 2023. Available online: https://nucleus.iaea.org/wiser/index.aspx (accessed on 1 October 2022).
- Bath, A.H. Stable Isotopic Evidence for Palaeo-Recharge Conditions of Groundwater; International Atomic Energy Agency (IAEA): Vienna, Austria, 1983; ISBN 978-92-0-141083-2. [Google Scholar]
- Rozanski, K. Deuterium and Oxygen-18 in European Groundwaters—Links to Atmospheric Circulation in the Past. Chem. Geol. Isot. Geosci. Sect. 1985, 52, 349–363. [Google Scholar] [CrossRef]
- Négrel, P.; Petelet-Giraud, E. Isotopes in Groundwater as Indicators of Climate Changes. TrAC Trends Anal. Chem. 2011, 30, 1279–1290. [Google Scholar] [CrossRef] [Green Version]
- Rozanski, K.; Araguás-Araguás, L.; Gonfiantini, R. Relation Between Long-Term Trends of Oxygen-18 Isotope Composition of Precipitation and Climate. Science 1992, 258, 981–985. [Google Scholar] [CrossRef]
- Blavoux, B.; Dray, M.; Fehri, A.; Olive, P.; Groening, M.; Sonntag, C.; Hauquin, J.P.; Pelissier, G.; Pouchan, P. Paleoclimatic and hydrodynamic approach to the Aquitaine Basin deep aquifer (France) by means of environmental isotopes and noble gases. In Isotope Techniques in the Study of Past and Current Environmental Changes in the Hydrosphere and the Atmosphere; International Atomic Energy Agency: Vienna, Austria, 1993; pp. 293–305. [Google Scholar]
- Edmunds, W.M. Palaeowaters in Coastal Europe: Evolution of Groundwater Since the Late Pleistocene; Milne, C.J., Ed.; Geological Society of London: London, UK, 2001; ISBN 978-1-86239-086-7. [Google Scholar]
- Stute, M.; Deak, J. Environmental isotope study (14C, 13C, 18O, 2H, noble gases) on deep groundwater circulation systems in Hungary with reference to palaeoclimate. Radiocarbon 1989, 31, 902–918. [Google Scholar] [CrossRef] [Green Version]
- Stute, M.; Clark, J.F.; Schlosser, P.; Broecker, W.S.; Bonani, G. A 30,000 Yr Continental Paleotemperature Record Derived from Noble Gases Dissolved in Groundwater from the San Juan Basin, New Mexico. Quat. Res. 1995, 43, 209–220. [Google Scholar] [CrossRef]
- Stute, M.; Forster, M.; Frischkorn, H.; Serejo, A.; Clark, J.F.; Schlosser, P.; Broecker, W.S.; Bonani, G. Cooling of Tropical Bra-zil (5°C) During the Last Glacial Maximum. Science 1995, 269, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Eggenkamp, H.G.M.; Louvat, P.; Agrinier, P.; Bonifacie, M.; Bekker, A.; Krupenik, V.; Griffioen, J.; Horita, J.; Brocks, J.J.; Bagheri, R. The Bromine and Chlorine Isotope Composition of Primary Halite Deposits and Their Significance for the Secular Isotope Composition of Seawater. Geochim. Cosmochim. Acta 2019, 264, 13–29. [Google Scholar] [CrossRef]
- Bagheri, R.; Nadri, A.; Raeisi, E.; Eggenkamp, H.G.M.; Kazemi, G.A.; Montaseri, A. Hydrochemical and Isotopic (Δ18O, Δ2H, 87Sr/86Sr, Δ37Cl and Δ81Br) Evidence for the Origin of Saline Formation Water in a Gas Reservoir. Chem. Geol. 2014, 384, 62–75. [Google Scholar] [CrossRef]
- Fontes, J.-C. Dating of Groundwater. In Guidebook on Nuclear Techniques in Hydrology; Technical Report Series No. 91; IAEA: Vienna, Austria, 1983; pp. 285–317. [Google Scholar]
- Fontes, J.-C. Chemical and Isotopic Constraints on 14C Dating of Groundwater. In Radiocarbon After Four Decades; Taylor, R.E., Long, A., Kra, R.S., Eds.; Springer: New York, NY, USA, 1992; pp. 242–261. ISBN 978-1-4757-4251-0. [Google Scholar]
- Zouari, K.; Trabelsi, R.; Chkir, N. Using Geochemical Indicators to Investigate Groundwater Mixing and Residence Time in the Aquifer System of Djeffara of Medenine (Southeastern Tunisia). Hydrogeol. J. 2011, 19, 209–219. [Google Scholar] [CrossRef]
- Tamers, M.A. Validity of Radiocarbon Dates on Ground Water. Geophys. Surv. 1975, 2, 217–239. [Google Scholar] [CrossRef]
- Ingerson, E.; Pearson, F. Estimation of Age and Rate of Motion of Groundwater by the 14C Method. In Recent Researches in the Fields of Hydrosphere, Atmosphere and Nuclear Geochemistry; Miyake, Y., Koyama, T., Eds.; Maruzen: Tokyo, Japan, 1964; pp. 263–283. [Google Scholar]
- Mook, W.G. Carbon-14 in hydrogeological studies. In The Terrestrial Environment; Elsevier: Amsterdam, The Netherlands, 1980; pp. 49–74. ISBN 978-0-444-41780-0. [Google Scholar]
- Fontes, J.-C.; Garnier, J.-M. Determination of the Initial 14C Activity of the Total Dissolved Carbon: A Review of the Existing Models and a New Approach. Water Resour. Res. 1979, 15, 399–413. [Google Scholar] [CrossRef]
- Salem, O.; Visser, J.H.; Dray, M.; Gonfiantini, R. Groundwater Flow Patterns in the Western Libyan Arab Jamahiriya Evalu-ated from Isotopic Data. In Arid Zone Hydrology. Investigations with Isotope Techniques; International Atomic Energy Agency: Vienna, Austria, 1980; pp. 165–179. ISBN 978-92-0-141180-8. [Google Scholar]
- Evans, G.V.; Otlet, R.L.; Downing, R.A.; Monkhouse, R.A.; Rae, G. Some Problems in the Interpretation of Isotope Measurements in United Kingdom Aquifers; International Atomic Energy Agency (IAEA): Vienna, Austria, 1979; ISBN 978-92-0-040179-4. [Google Scholar]
- Eichinger, L. A Contribution to the Interpretation of 14 C Groundwater Ages Considering the Example of a Partially Con-fined Sandstone Aquifer. Radiocarbon 1983, 25, 347–356. [Google Scholar] [CrossRef] [Green Version]
- Lassoued, S. Hydrogeology and Isotopic Geochemistry of the Plain of El Haouaria (Northern Tunisia). Ph.D. Thesis, University of South Paris, Paris, France, 1982. (In French). [Google Scholar]
- Daoud, D. Geochemical and Isotopic Characterization of Groundwater and Estimation of the Evaporation Rate in the Chott-Chergui Basin (Semiarid Zone, Algeria). Ph.D. Thesis, University of Paris 11, Paris, France, 1995. [Google Scholar]
- Vogel, J.C. Investigation of Groundwater Flow with Radiocarbon. In Proceeding of the Symposium on Isotopes in Hydrology, Vienna, Austria, 14–18 November 1966; IAEA: Vienna, Austria, 1966; pp. 355–369. [Google Scholar]
- Cook, P.G.; Herczeg, A.L. Environmental Tracers in Subsurface Hydrology; Springer: Boston, MA, USA, 2000; pp. 111–144. ISBN 978-1-4613-7057-4. [Google Scholar]



| N° | WPT | PL (m) | pH | EC mS/cm | Ca | Mg | Na | K | SO4 | Cl | NO3 | HCO3 | Br | SI Gypsum | δ18O | δ2H | 3H | δ37Cl | δ13C | 14C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mg/L | ‰ V-SMOW | TU | ‰ SMOC | ‰ PDB | PMC | |||||||||||||||
| 1 | DW | 28.8 | 7.9 | 7.89 | 273.2 | 129.84 | 666.7 | 13.294 | 1152.0 | 1070.6 | 68.2 | 129.93 | 0.48 | |||||||
| 2 | DW | 29.72 | 7.7 | 10.14 | 140 | 180 | 1809.3 | 5.865 | 2112.0 | 1655.5 | 11.78 | 567.3 | 1.38 | |||||||
| 3 | DW | 27.9 | 7.7 | 8.9 | 322.4 | 141.36 | 1459.9 | 4.692 | 1680.0 | 1721.1 | 43.4 | 434.93 | 0.67 | 0.3 | ||||||
| 4 | B | 7.3 | 5.98 | 240 | 112.8 | 823.0 | 2.737 | 912.0 | 1198.9 | 69.44 | 292.8 | 0.78 | ||||||||
| 5 | DW | 37.46 | 7.3 | 10.91 | 440 | 216 | 1517.3 | 5.865 | 1680.0 | 2268.8 | 22.94 | 264.13 | 1.46 | |||||||
| 6 | DW | 36.9 | 8.9 | 7.85 | 248 | 63.84 | 1269.0 | 77.027 | 1180.8 | 1595.3 | 4.96 | 274.5 | 4.49 | −0.34 | −4.91 | −31.23 | 4.3 | 0.25 | −0.2 | 11.72 |
| 7 | B | 7.9 | 4.7 | 270.4 | 111.6 | 687.4 | 6.647 | 1227.8 | 886.3 | 48.36 | 223.87 | 2.84 | −0.23 | 0 | −10 | 0.51 | ||||
| 8 | B | 7 | 6.72 | 399.2 | 209.28 | 1565.6 | 7.038 | 2157.6 | 2197.9 | 34.72 | 276.33 | 6.45 | 0.01 | 0 | 0.30 | −11.4 | 20.01 | |||
| 9 | DW | 33.55 | 8.2 | 7.56 | 234 | 117.84 | 1951.9 | 42.619 | 3446.4 | 1063.5 | 0.62 | 268.4 | 3.39 | −0.02 | −0.04 | |||||
| 10 | DW | 30.4 | 7.6 | 1.87 | 152 | 67.2 | 158.6 | 1.955 | 584.6 | 177.3 | 44.02 | 134.2 | 0.57 | −0.6 | −5.05 | −29.77 | −0.23 | −12.3 | 71.5 | |
| 11 | DW | 27.9 | 7.3 | 5.69 | 508 | 210 | 712.7 | 5.474 | 2399.0 | 779.9 | 124 | 211.67 | 3.07 | 0.22 | 0 | −0.49 | ||||
| 12 | B | 7.4 | 4.34 | 266 | 100.32 | 570.2 | 5.083 | 1053.6 | 602.7 | 213.28 | 278.77 | 2.20 | −0.27 | −5.13 | −30.57 | 0 | 0.01 | |||
| 13 | DW | 28.95 | 7.7 | 3.12 | 258 | 107.28 | 395.4 | 4.301 | 1459.2 | 248.2 | 20.46 | 254.98 | 0.81 | −0.14 | 0 | −0.35 | −11.5 | 37.71 | ||
| 14 | B | 7.3 | 5.64 | 448.4 | 132.24 | 896.6 | 28.543 | 2853.6 | 567.2 | 45.88 | 168.97 | 2.67 | 0.24 | −4.86 | −28.71 | 0 | −11.5 | 55.61 | ||
| 15 | DW | 31.63 | 7.6 | 2.6 | 211.2 | 83.28 | 457.5 | 3.128 | 1436.6 | 141.8 | 36.58 | 128.1 | 0.38 | −0.21 | −4.37 | −27.16 | 0.3 | −13.2 | 30.65 | |
| 16 | DW | 31.25 | 8.1 | 1.58 | 113.6 | 36.72 | 165.5 | 5.083 | 454.6 | 35.5 | 9.3 | 258.03 | 0.19 | −0.78 | −5.03 | −29.4 | 0 | −0.16 | −14.2 | 41.67 |
| 17 | DW | 23.6 | 7 | 6.5 | 350.4 | 182.88 | 1521.9 | 4.692 | 2222.4 | 1347.1 | 87.42 | 349.53 | 4.54 | 0 | −5.39 | −32.66 | 2.6 | 0.20 | −13.3 | 79.64 |
| 18 | DW | 24.59 | 7.1 | 12.7 | 796 | 325.92 | 3002.5 | 93.058 | 2067.8 | 5707.5 | 17.98 | 195.2 | 16.93 | 0.15 | −4.43 | −27.57 | 1.6 | |||
| 19 | DW | 19.05 | 7.4 | 2.36 | 120 | 62.4 | 250.6 | 3.128 | 200.2 | 425.4 | 24.8 | 295.24 | 1.74 | −1.15 | −4.21 | −26.34 | −0.03 | −14.6 | 63.22 | |
| 20 | DW | 18.35 | 7.3 | 9.67 | 536 | 237.6 | 2193.2 | 86.02 | 1936.3 | 3048.7 | 814.68 | 189.1 | 7.72 | 0.04 | −5.2 | −31.61 | −0.31 | |||
| 21 | DW | 1.69 | 7.5 | 2.18 | 80 | 46.56 | 206.9 | 1.173 | 187.7 | 283.6 | 34.1 | 204.35 | 1.41 | −1.3 | 0 | −0.17 | −13.4 | 72.3 | ||
| 22 | DW | 27.22 | 7.2 | 3.47 | 160 | 76.8 | 462.1 | 3.91 | 355.2 | 744.5 | 51.46 | 207.4 | 3.46 | −0.86 | −4.89 | −29.59 | 0.05 | −11.8 | 69.97 | |
| 23 | DW | 23.3 | 7.3 | 4.08 | 216 | 81.6 | 586.2 | 1.955 | 562.6 | 921.7 | 25.42 | 196.42 | 3.62 | −0.59 | −3.98 | −25.14 | 0 | 0.04 | −12.5 | 74.81 |
| 24 | DW | 21.69 | 7.8 | 3.8 | 184 | 91.2 | 492.0 | 5.474 | 1102.6 | 425.4 | 11.16 | 283.04 | 1.74 | −0.38 | −0.02 | |||||
| 25 | DW | 4.15 | 7.1 | 4.1 | 194 | 79.56 | 542.6 | 3.128 | 423.8 | 815.4 | 246.14 | 198.86 | 3.46 | −0.74 | −0.15 | |||||
| 26 | DW | 7.3 | 4.52 | 224.6 | 92.28 | 731.1 | 7.038 | 487.2 | 1205.3 | 169.26 | 210.45 | 3.68 | −0.67 | −0.22 | ||||||
| 27 | B | 7 | 4.75 | 241.8 | 97.68 | 784.0 | 34.799 | 481.0 | 709.0 | 1190.4 | 250.1 | 1.21 | −0.66 | −0.30 | ||||||
| 28 | DW | 2.72 | 7.5 | 3.38 | 156 | 68.16 | 475.9 | 23.069 | 569.3 | 638.1 | 138.88 | 262.3 | 1.71 | −0.69 | −0.20 | |||||
| 29 | DW | 2.87 | 7.2 | 8.18 | 449.8 | 214.8 | 1873.7 | 39.1 | 2326.6 | 2339.7 | 54.56 | 346.48 | 7.03 | 0.07 | −0.21 | |||||
| 30 | DW | 10 | 7.6 | 1.38 | 68 | 34.8 | 121.8 | 8.602 | 192.5 | 212.7 | 6.2 | 126.88 | 0.32 | −1.31 | ||||||
| 31 | B | 7.2 | 4.48 | 265.2 | 104.4 | 650.6 | 10.166 | 913.4 | 1028.1 | 182.9 | 234.24 | 2.90 | −0.35 | −0.06 | ||||||
| 32 | DW | 20.86 | 7.4 | 5.29 | 285.6 | 113.52 | 938.0 | 4.692 | 836.6 | 1418.0 | 40.92 | 277.55 | 5.09 | −0.39 | −0.07 | |||||
| 33 | DW | 18 | 7.2 | 7.21 | 396 | 301.2 | 1115.0 | 20.723 | 1766.4 | 1800.9 | 47.74 | 531.92 | 0.72 | −3.89 | −25.17 | |||||
| 34 | B | 7.3 | 7.97 | 428 | 224.4 | 1220.8 | 9.384 | 1824.0 | 2034.8 | 68.2 | 220.82 | 0.68 | −4.69 | −31.42 | 0 | |||||
| 35 | B | 7.4 | 7.94 | 490 | 225.6 | 1135.7 | 10.166 | 1536.0 | 2114.9 | 91.76 | 227.53 | 0.85 | −4.8 | −31.18 | 0.5 | |||||
| 36 | DW | 25 | 7.4 | 12.01 | 708 | 423.12 | 2643.9 | 64.515 | 3600.0 | 3885.3 | 53.32 | 313.54 | 1.31 | −4.35 | −27.36 | |||||
| 37 | DW | 37.7 | 7.4 | 10.63 | 576.4 | 182.16 | 1584.0 | 21.505 | 1536.0 | 2853.7 | 65.1 | 254.98 | 0.85 | −4.58 | −27.99 | 0.3 | ||||
 Shallow aquifer;
Shallow aquifer;  Deep aquifer.
Deep aquifer.| Sample No | Age Ao = 100 | Age Tamers | Age Pearson | Age Mook | Age F. & G. | Age F. &G. eq | Age AIEA | Age Evans | Age Eichinger |
|---|---|---|---|---|---|---|---|---|---|
| Years | |||||||||
| 2 | 2876 | −2157 | −2560 | −4576 | −2573 | 1167 | 1747 | −2807 | −1972 |
| 3 | 2271 | −2607 | −2494 | −2017 | −2417 | 1136 | 1821 | −2698 | −2171 |
| 4 | 9046 | 3774 | 3479 | 2107 | 3469 | 7417 | 7757 | 3216 | 4037 |
| 7 | 11,620 | 6512 | 5512 | −553 | 5475 | 9208 | 9746 | 5197 | 6434 |
| 8 | 13,302 | 8050 | 8223 | 9059 | 8345 | 12,352 | 12,703 | 8039 | 8409 |
| 10 | 2773 | −2713 | −1635 | 1997 | −963 | 2601 | 2717 | −1813 | −1831 |
| 13 | 8062 | 2883 | 3098 | 3852 | 3235 | 6846 | 7239 | 2840 | 3498 |
| 14 | 4851 | −637 | −163 | 1645 | 148 | 4037 | 4160 | −380 | −20 |
| 15 | 9775 | 4421 | 5931 | 10,572 | 6835 | 10,070 | 10,283 | 5782 | 5492 |
| 16 | 7236 | 1956 | 3973 | 8947 | 5070 | 7898 | 8142 | 3826 | 3359 |
| 17 | 1882 | −3635 | −1901 | 2820 | −913 | 2259 | 2341 | −2065 | −2383 |
| 19 | 3791 | −1256 | 786 | 5875 | 1902 | 4569 | 4977 | 656 | 123 |
| 21 | 2681 | −2746 | −1014 | 4084 | 3 | 3186 | 3338 | −1156 | −1574 |
| 22 | 2952 | −2766 | −1807 | 1515 | −1202 | 2620 | 2546 | −2003 | −1923 |
| 23 | 2399 | −2769 | −1923 | 835 | −1400 | 1940 | 2319 | −2118 | −1930 |
| 35 | 7647 | 2280 | 3198 | 5983 | 3749 | 7157 | 7367 | 2980 | 3211 |
| 37 | 4416 | −457 | −39 | 1619 | 238 | 3644 | 4313 | −219 | 99 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nefzaoui, F.; Ben Hamouda, M.F.; Carreira, P.M.; Marques, J.M.; Eggenkamp, H.G.M. Evidence for Groundwater Salinity Origin Based on Hydrogeochemical and Isotopic (2H, 18O, 37Cl, 3H, 13C, 14C) Approaches: Sousse, Eastern Tunisia. Water 2023, 15, 1242. https://doi.org/10.3390/w15061242
Nefzaoui F, Ben Hamouda MF, Carreira PM, Marques JM, Eggenkamp HGM. Evidence for Groundwater Salinity Origin Based on Hydrogeochemical and Isotopic (2H, 18O, 37Cl, 3H, 13C, 14C) Approaches: Sousse, Eastern Tunisia. Water. 2023; 15(6):1242. https://doi.org/10.3390/w15061242
Chicago/Turabian StyleNefzaoui, Farah, Mohamed Fethi Ben Hamouda, Paula Maria Carreira, José Manuel Marques, and Hans G. M. Eggenkamp. 2023. "Evidence for Groundwater Salinity Origin Based on Hydrogeochemical and Isotopic (2H, 18O, 37Cl, 3H, 13C, 14C) Approaches: Sousse, Eastern Tunisia" Water 15, no. 6: 1242. https://doi.org/10.3390/w15061242







