Elevated River Inputs of the Total Alkalinity and Dissolved Inorganic Carbon in the Northern Adriatic Sea
Abstract
1. Introduction
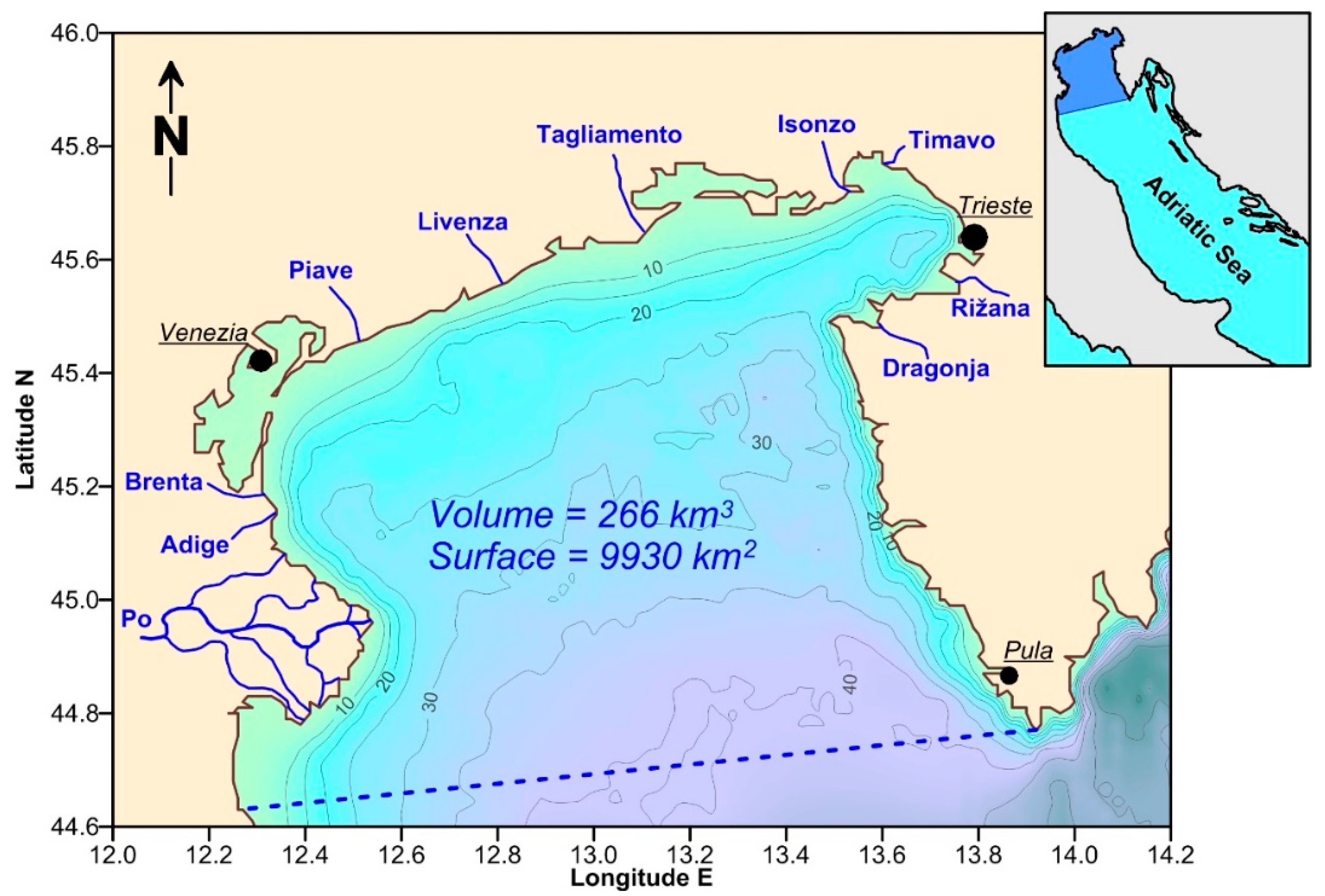
2. Study Area
3. Materials and Methods
3.1. Freshwater Discharges
3.2. Physical and Chemical Parameters
3.3. Data Processing
4. Results and Discussion
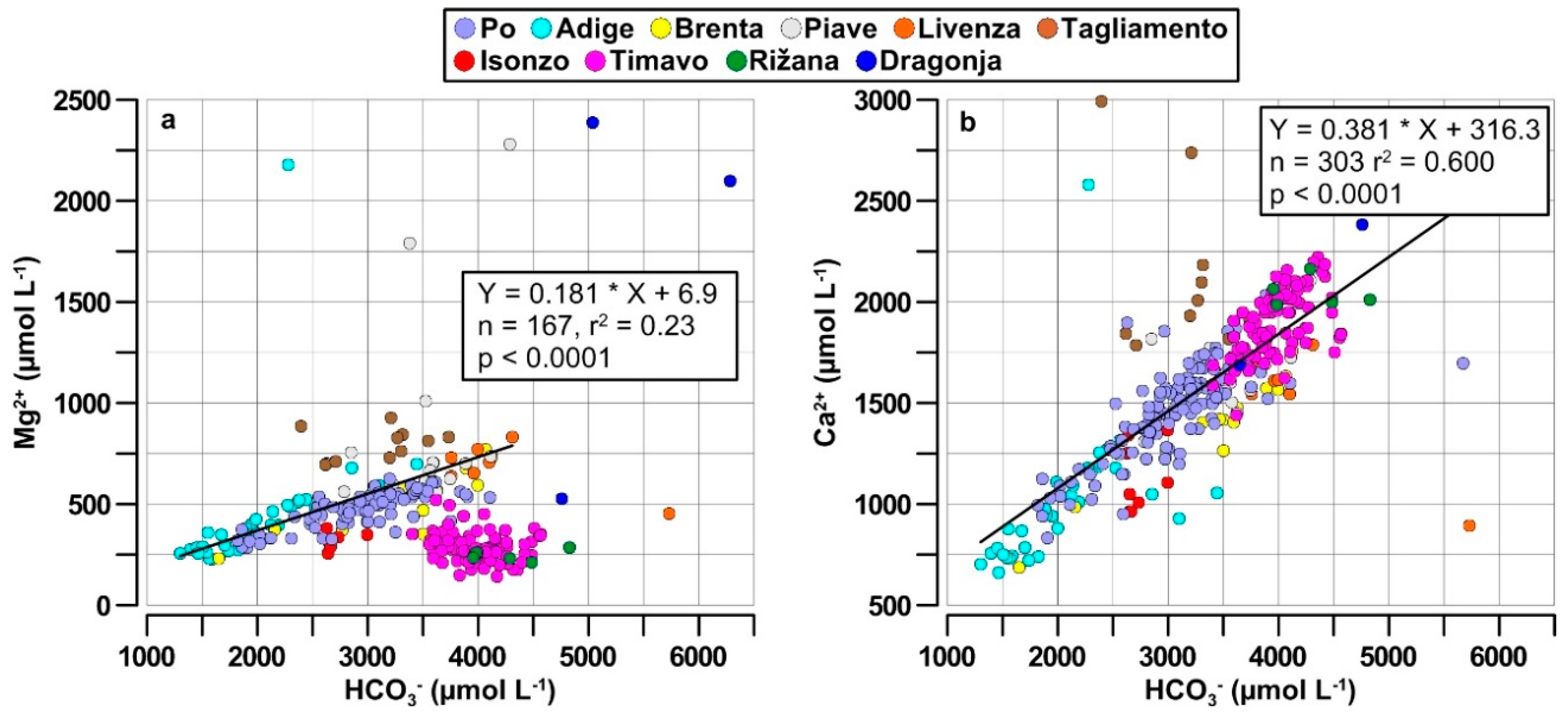
4.1. Chemical Characteristics of the NAd River Waters
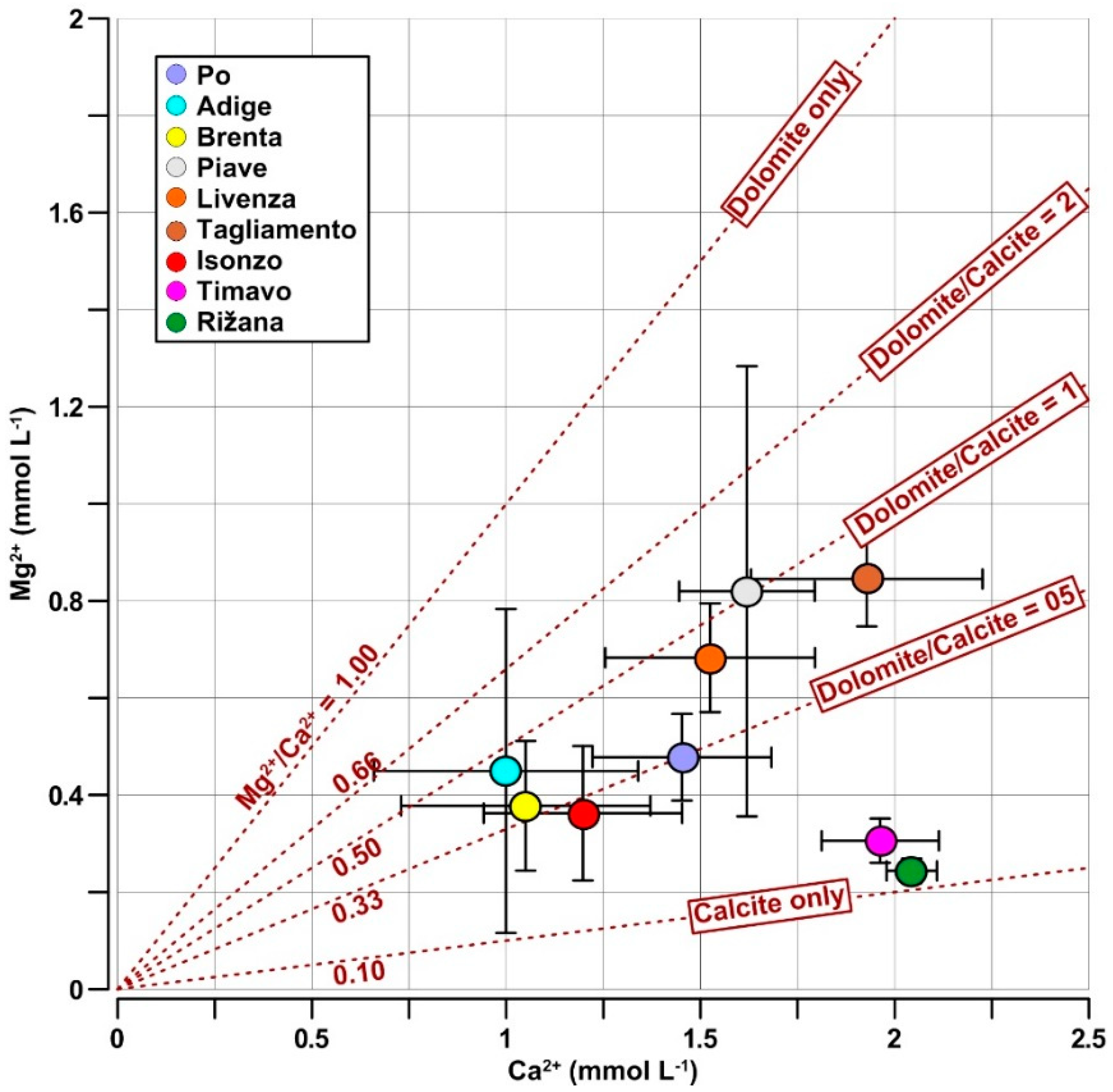
4.2. Mg2+/Ca2+ Molar Ratios in River Waters
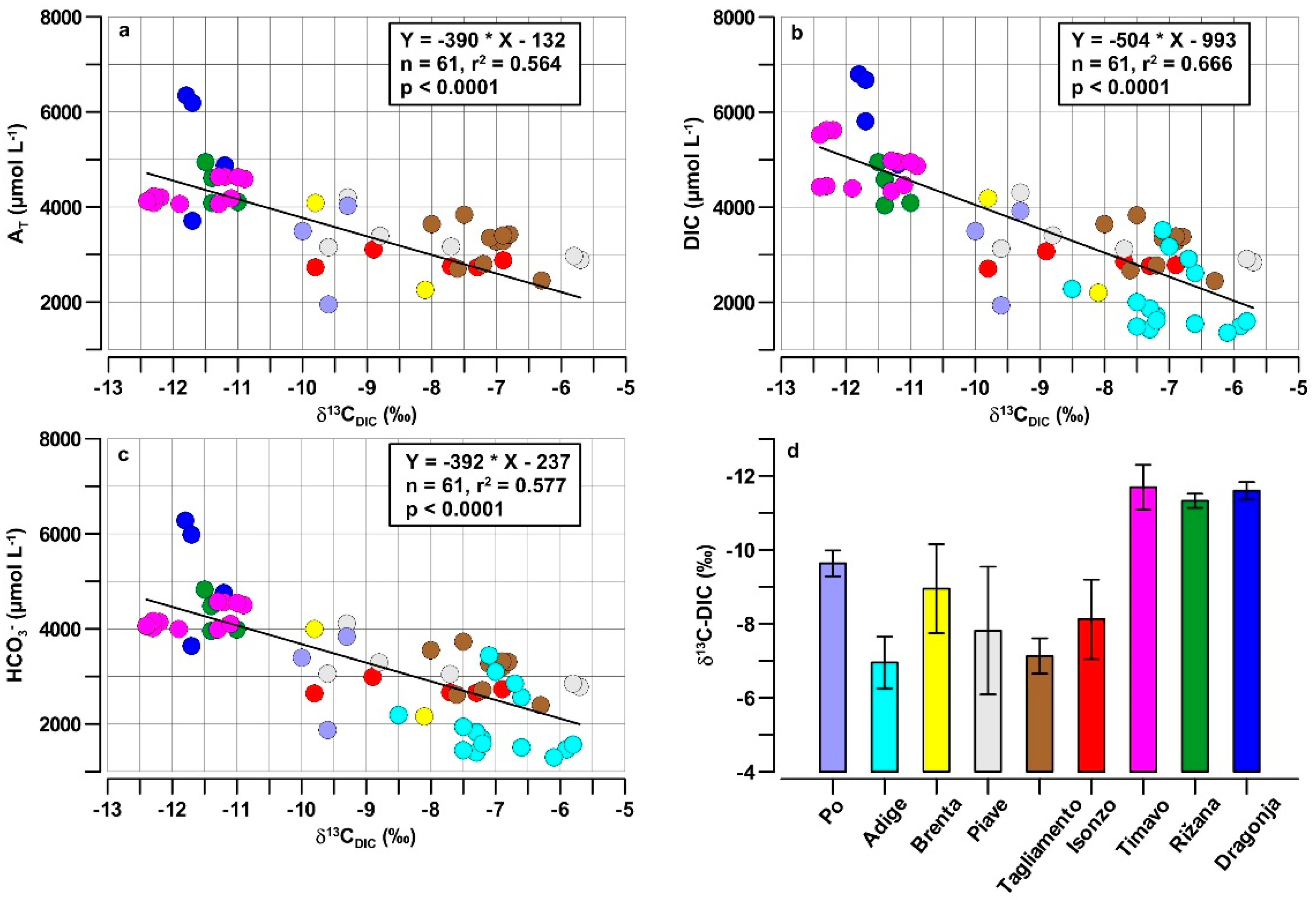
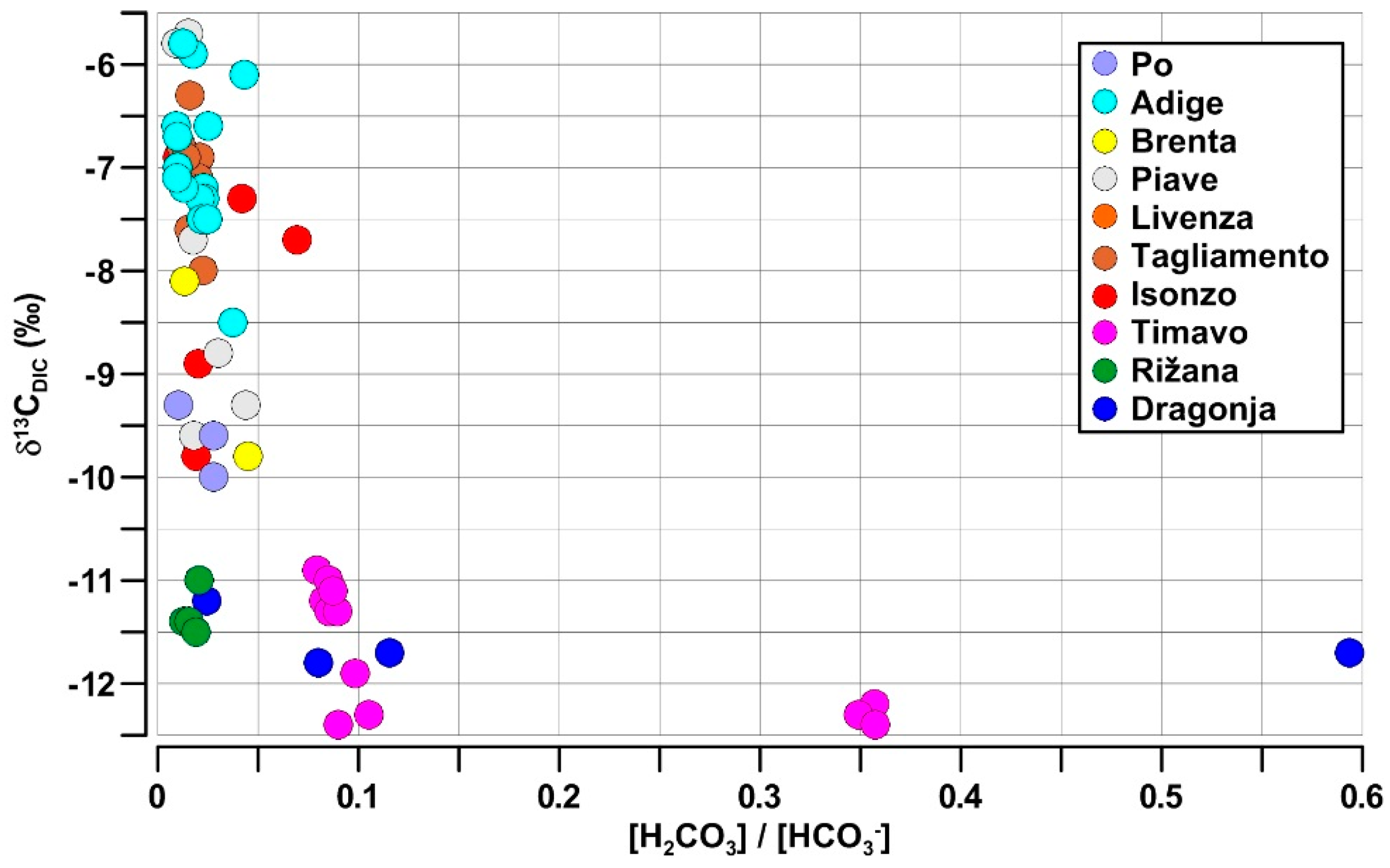
4.3. Isotopic Signature and Weathering Intensity of the Riverine DIC
- The first group had the highest AT, DIC and HCO3− and the most negative δ13CDIC values, including the Dragonja, Rižana and Timavo. In the small rivers flowing in the karst catchments, the δ13CDIC values from the weathering of carbonate rocks were significantly shifted toward the negative values by the contribution of the remineralization of organic matter, which also produced the highest DIC and HCO3− concentrations in freshwater.
- The second group had a highly variable AT, DIC and HCO3− and medium to low values of the δ13CDIC. This group included rivers such as the Brenta, Piave and Isonzo, which were probably influenced by a combination of processes along their river networks, such as the weathering of carbonate rocks, primary production and CO2 exchange with the atmosphere, which increased the variability of these parameters. Minimal information was available on the δ13CDIC values in the Po River, but the data showed that these were intermediate values compared to the other NAd rivers, at least at the mouth of this large basin.
- The third group presented a low AT, DIC and HCO3− and the least negative δ13CDIC values from the Adige and Tagliamento Rivers, which might have been due to the predominance of carbonate weathering in all the available sampling sites.
- Samples from the Timavo Rižana and Dragonja rivers, which show larger variations in the [H2CO3]/[HCO3−] ratio, ranging between 0 and 0.6 and low values of the δ13CDIC (−11 to −12.5‰), indicating no exchange with atmospheric CO2, but the presence of biogenic CO2.
- The second group, with a very low [H2CO3]/[HCO3−] ratio (<0.1), exhibits greater variations of the δ13CDIC (−10 to −5‰), indicating different biogeochemical processes in the rivers such as an exchange with atmospheric CO2, biogenic CO2 and carbonate weathering.
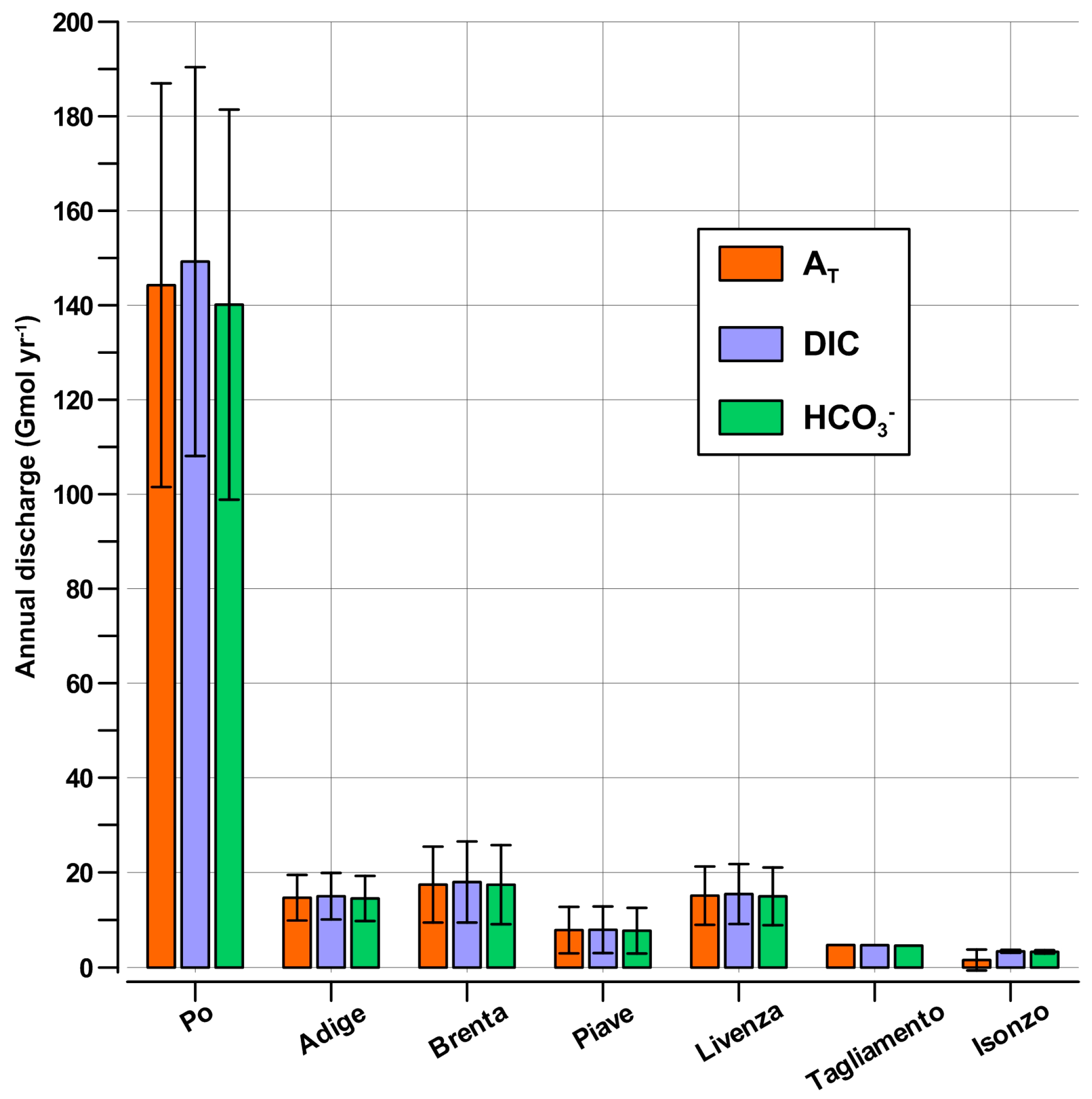
4.4. Impact of the Riverine AT and DIC in the NAd
5. Conclusions
- Strong bicarbonate weathering occurred in these mountainous watersheds.
- Calcite weathering was prevalent in the Po, Isonzo, Timavo and Rižana catchments, while dolomite weathering was more important in the Adige, Piave and Livenza rivers.
- The rivers with an AT higher than the DIC concentration have the potential to buffer the NAd waters, while those, such as the Po River, have the potential to enhance the acidification process in the marine environment.
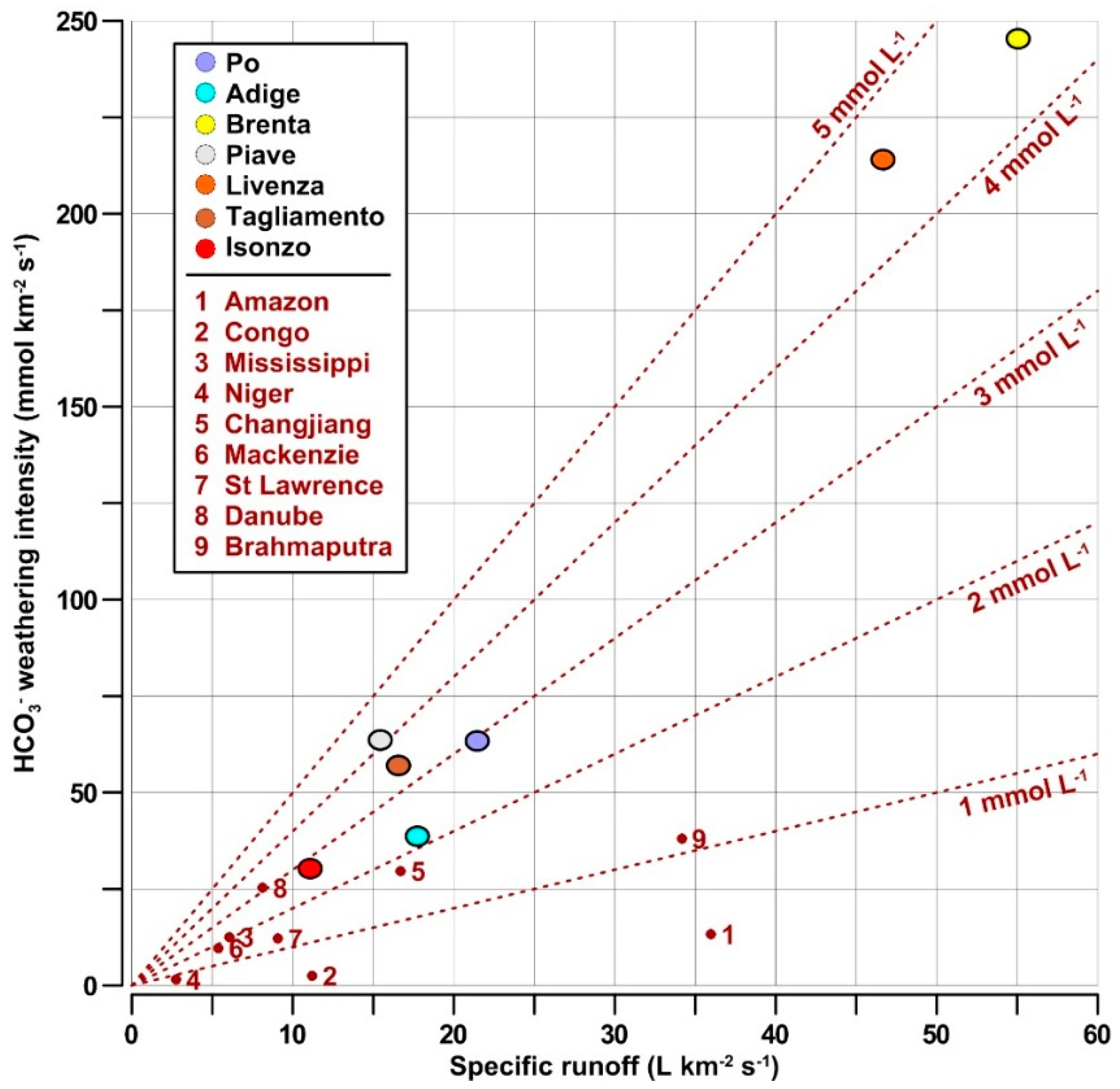
- The carbonate weathering intensity in the NAd watershed was among the highest and was approx. four to 35 times higher than the global average (7 meq km−2 s−1);
- Global climate models predicted that the precipitation pattern in southern Europe is likely to change. If the rivers in the NAd maintain a relatively unchanged concentration of carbonate mineral weathering products, the carbonate inputs to the coastal areas are expected to depend primarily on the overall changes in freshwater discharge.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| River | Sample Site | Sampling Date | Water Flow | Specific Runoff | T | pH | EC | AT | Ca2+ | Mg2+ | Cl− | δ13C-DIC | DIC | HCO3− | CO3 2− | H2CO3 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| dddd/mm/yy | m3 s−1 | L km2 s−1 | °C | NBS Scale | µS cm−1 | µmol L−1 | µmol L−1 | µmol L−1 | µmol L−1 | ‰ | µmol L−1 | µmol L−1 | µmol L−1 | µmol L−1 | ||
| Isonzo | Gradisca | 30/5/07 | 50.7 | 24.9 | 13.5 | 7.80 | 262 | 2736 | 1048 | 306 | 40 | −7.3 | 2769 | 2650 | 7 | 112 |
| Isonzo | Gradisca | 31/10/07 | 26.4 | 16.4 | 13.5 | 8.05 | 318 | 2715 | 1250 | 381 | 50 | - | 2699 | 2625 | 12 | 62 |
| Isonzo | Gradisca | 6/6/08 | 153.8 | 63.0 | 12.8 | 8.15 | 257 | 2732 | 1340 | 257 | 69 | −9.8 | 2704 | 2639 | 15 | 50 |
| Isonzo | Gradisca | 26/5/11 | 2.3 | 14.8 | 19.1 | 8.38 | 226 | 2877 | 1007 | 334 | 49 | −6.9 | 2788 | 2729 | 31 | 27 |
| Isonzo | Gradisca | 13/10/11 | 0.4 | 12.5 | 14.2 | 8.11 | 264 | 3103 | 1104 | 348 | 50 | −8.9 | 3070 | 2992 | 16 | 61 |
| Isonzo | Pieris | 30/5/07 | 50.7 | - | 15.0 | 7.57 | 274 | 2748 | 961 | 288 | 49 | −7.7 | 2852 | 2663 | 4 | 185 |
| Isonzo | Pieris | 31/10/07 | 26.4 | - | 11.3 | 8.03 | 336 | 3082 | 1365 | 411 | 63 | - | 3081 | 2990 | 12 | 77 |
| Dragonja | Mouth | 29/5/07 | 0.3 | 3.2 | 14.3 | 6.64 | 7490 | 3713 | 1688 | 5300 | 54,505 | −11.7 | 5809 | 3644 | 1 | 2164 |
| Dragonja | Mouth | 29/10/07 | 0.1 | 0.7 | 12.5 | 8.06 | 570 | 5119 | 2704 | 2387 | 22,085 | - | 5178 | 5035 | 24 | 117 |
| Dragonja | Mouth | 9/6/08 | 1.2 | 12.4 | 19.9 | 7.98 | 641 | 4871 | 2381 | 525 | 1395 | −11.2 | 4897 | 4757 | 23 | 116 |
| Dragonja | Mouth | 27/5/11 | 0.1 | 0.9 | 18.6 | 7.47 | 2225 | 6346 | 2846 | 2098 | 17,107 | −11.8 | 6794 | 6279 | 9 | 503 |
| Dragonja | Mouth | 13/10/11 | 0.0 | 0.1 | 17.6 | 7.33 | 12,560 | 6185 | - | - | - | −11.7 | 6674 | 5979 | 6 | 690 |
| Rižana | Bridge | 23/5/07 | 0.2 | 1.0 | 21.1 | 8.05 | 449 | 4108 | 1986 | 259 | 192 | −11.0 | 4087 | 3982 | 23 | 82 |
| Rižana | Bridge | 29/10/07 | 3.6 | 17.4 | 11.4 | 8.27 | 429 | 4397 | 2162 | 230 | 89 | - | 4377 | 4281 | 32 | 63 |
| Rižana | Bridge | 9/6/08 | 4.0 | 19.5 | 14.6 | 8.30 | 345 | 4081 | 2063 | 233 | 104 | −11.4 | 4040 | 3954 | 34 | 51 |
| Rižana | Bridge | 27/5/11 | 0.5 | 2.5 | 15.3 | 8.21 | 349 | 4612 | 1997 | 212 | 109 | −11.4 | 4585 | 4482 | 32 | 70 |
| Rižana | Bridge | 13/10/11 | 0.2 | 0.8 | 14.9 | 8.13 | 432 | 4948 | 2011 | 285 | 163 | −11.5 | 4946 | 4825 | 29 | 91 |
| Timavo | Spring 1 | 30/5/07 | 33.0 | 87.3 | 11.9 | 6.88 | 454 | 4200 | 2104 | 268 | 206 | −12.2 | 5625 | 4143 | 1 | 1479 |
| Timavo | Spring 1 | 31/10/07 | 10.0 | 26.5 | 13.6 | 7.36 | 464 | 4135 | 2155 | 282 | 195 | - | 4542 | 4072 | 4 | 465 |
| Timavo | Spring 1 | 10/6/08 | 23.0 | 60.8 | 12.1 | 7.41 | 373 | 4082 | 2065 | 267 | 190 | −12.3 | 4446 | 4018 | 4 | 422 |
| Timavo | Spring 1 | 26/5/11 | 15.0 | 39.7 | 12.5 | 7.51 | 332 | 4640 | 1843 | 352 | 208 | −11.2 | 4946 | 4561 | 6 | 378 |
| Timavo | Spring 1 | 13/10/11 | 12.0 | 31.7 | 13.6 | 7.52 | 397 | 4587 | 1749 | 380 | 236 | −10.9 | 4866 | 4502 | 6 | 356 |
| Timavo | Spring 2 | 30/5/07 | 33.0 | - | 11.9 | 6.89 | 455 | 4221 | 2080 | 262 | 203 | −12.3 | 5619 | 4163 | 1 | 1453 |
| Timavo | Spring 2 | 31/10/07 | 10.0 | - | 12.9 | 7.47 | 457 | 4121 | 2110 | 279 | 195 | −12.4 | 4427 | 4056 | 5 | 365 |
| Timavo | Spring 2 | 10/6/08 | 23.0 | - | 12.2 | 7.4 | 377 | 4034 | 2025 | 264 | 190 | - | 4400 | 3969 | 4 | 426 |
| Timavo | Spring 2 | 26/5/11 | 15.0 | - | 12.4 | 7.5 | 318 | 4631 | 1823 | 342 | 208 | −11.0 | 4945 | 4552 | 6 | 386 |
| Timavo | Spring 2 | 13/10/11 | 12.0 | - | 13.3 | 7.49 | 395 | 4065 | 1716 | 368 | 240 | −11.3 | 4333 | 3986 | 5 | 341 |
| Timavo | Spring 3 | 30/5/07 | 33.0 | - | 11.9 | 6.88 | 456 | 4124 | 2036 | 263 | 203 | −12.4 | 5520 | 4065 | 1 | 1453 |
| Timavo | Spring 3 | 31/10/07 | 10.0 | - | 13.0 | 7.39 | 467 | 4041 | 2124 | 280 | 198 | - | 4413 | 3979 | 4 | 429 |
| Timavo | Spring 3 | 10/6/08 | 23.0 | - | 15.5 | 7.41 | 392 | 4067 | 2019 | 263 | 190 | −11.9 | 4393 | 3995 | 5 | 392 |
| Timavo | Spring 3 | 26/5/11 | 15.0 | - | 12.0 | 7.48 | 321 | 4642 | 1839 | 346 | 212 | −11.3 | 4980 | 4564 | 6 | 409 |
| Timavo | Spring 3 | 13/10/11 | 12.0 | - | 13.4 | 7.48 | 393 | 4183 | 1752 | 372 | 241 | −11.1 | 4468 | 4104 | 5 | 358 |
| Tagliamento | Invilino | 3/6/08 | 35.0 | - | 15.8 | 8.19 | 598 | 2452 | 2993 | 886 | 52 | −6.3 | 2449 | 2393 | 17 | 39 |
| Tagliamento | Tolmezzo | 18/10/11 | 38.2 | - | 12.6 | 8.15 | 653 | 3275 | 2737 | 926 | 88 | −7.0 | 3286 | 3205 | 19 | 60 |
| Tagliamento | Pioverno | 2/6/11 | 23.0 | 16.0 | 14.8 | 8.09 | 429 | 3289 | 1930 | 729 | 98 | −6.9 | 3283 | 3198 | 17 | 67 |
| Tagliamento | Pioverno | 18/10/11 | 38.2 | 11.7 | 10.5 | 8.14 | 492 | 3352 | 2007 | 829 | 95 | −7.1 | 3351 | 3267 | 17 | 66 |
| Tagliamento | Dignano | 3/6/08 | 38.4 | - | 14.8 | 8.22 | 413 | 2704 | 1842 | 695 | 203 | −7.6 | 2675 | 2615 | 19 | 40 |
| Tagliamento | Dignano | 2/6/11 | 23.0 | - | 17.5 | 8.32 | 477 | 3426 | 2097 | 762 | 92 | −6.8 | 3375 | 3304 | 32 | 39 |
| Tagliamento | Dignano | 18/10/11 | 38.2 | - | 11.3 | 8.29 | 527 | 3408 | 2183 | 845 | 94 | −6.9 | 3384 | 3311 | 26 | 47 |
| Tagliamento | Madrisio | 6/6/08 | 36.0 | - | 16.0 | 8.20 | 428 | 2803 | 1785 | 710 | 94 | −7.2 | 2771 | 2709 | 19 | 43 |
| Tagliamento | Madrisio | 30/5/11 | 26.1 | - | 16.2 | 8.05 | 433 | 3836 | 1917 | 831 | 90 | −7.5 | 3836 | 3733 | 19 | 83 |
| Tagliamento | Madrisio | 20/10/11 | 91.0 | - | 13.6 | 8.07 | 494 | 3643 | 1817 | 812 | 74 | −8.0 | 3644 | 3546 | 18 | 79 |
| Piave | Provagna | 17/6/08 | 100.0 | - | 14.0 | 7.94 | 337 | 3392 | 1529 | 654 | 80 | −8.8 | 3411 | 3299 | 12 | 99 |
| Piave | Provagna | 1/6/11 | 26.7 | - | 15.0 | 8.23 | 290 | 2893 | 1308 | 562 | 44 | −5.7 | 2846 | 2783 | 20 | 42 |
| Piave | Provagna | 18/10/11 | 7.3 | - | 10.6 | 8.49 | 429 | 2969 | 1817 | 753 | 57 | −5.8 | 2910 | 2849 | 34 | 26 |
| Piave | San Dona | 6/6/08 | 168.0 | 42.8 | 15.4 | 8.15 | 336 | 3158 | 1531 | 542 | 88 | −9.6 | 3127 | 3053 | 19 | 55 |
| Piave | San Dona | 30/5/11 | 68.0 | 13.0 | 16.5 | 8.15 | 334 | 3162 | 1461 | 582 | 77 | −7.7 | 3125 | 3052 | 19 | 54 |
| Piave | San Dona | 20/10/11 | 13.8 | 9.5 | 13.2 | 7.78 | 457 | 4201 | 1724 | 735 | 107 | −9.3 | 4303 | 4111 | 10 | 180 |
| Brenta | Valstagna | 1/6/11 | 100.5 | - | 12.8 | 8.40 | 144 | 1742 | 686 | 230 | 48 | - | 1685 | 1651 | 16 | 18 |
| Brenta | Valstagna | 20/10/11 | 67.8 | - | 11.5 | 8.32 | 256 | 2255 | 985 | 373 | 66 | −8.1 | 2203 | 2157 | 17 | 29 |
| Brenta | Limena | 30/5/11 | 104.9 | 39.9 | 16.2 | 8.18 | 215 | 2766 | 963 | 316 | 86 | − | 2713 | 2651 | 17 | 44 |
| Brenta | Limena | 20/10/11 | 67.8 | 22.0 | 13.1 | 7.77 | 424 | 4086 | 1567 | 592 | 214 | −9.8 | 4183 | 3993 | 10 | 180 |
| Po | Pollesella | 8/6/08 | 3160.0 | 45.1 | 18.8 | 7.94 | 286 | 1949 | 1054 | 307 | 310 | −9.6 | 1932 | 1872 | 8 | 52 |
| Po | Pollesella | 30/5/11 | 1030.0 | 14.7 | 23.7 | 8.33 | 477 | 4024 | 1652 | 562 | 621 | −9.3 | 3921 | 3837 | 44 | 40 |
| Po | Pollesella | 20/10/11 | 873.0 | 12.5 | 14.5 | 7.97 | 469 | 3490 | 1518 | 540 | 557 | −10.0 | 3500 | 3392 | 14 | 94 |
| Adige | San Michele | 17/6/08 | 404.2 | - | 10.8 | 8.05 | 164 | 1566 | 751 | 287 | 86 | −6.6 | 1547 | 1502 | 6 | 38 |
| Adige | San Michele | 31/5/11 | 259.3 | - | 13.7 | 8.18 | 158 | 1533 | 662 | 257 | 82 | −5.9 | 1496 | 1462 | 9 | 26 |
| Adige | San Michele | 19/10/11 | 175.2 | - | 8.1 | 8.12 | 234 | 1739 | 867 | 347 | 116 | −7.2 | 1716 | 1670 | 8 | 38 |
| Adige | Mattarello | 15/6/08 | 484.7 | - | 10.8 | 7.82 | 151 | 1351 | 703 | 257 | 79 | −6.1 | 1358 | 1299 | 3 | 56 |
| Adige | Chizzola | 15/6/08 | 484.7 | - | 11.2 | 8.08 | 164 | 1455 | 756 | 275 | 98 | −7.3 | 1433 | 1394 | 6 | 33 |
| Adige | Chizzola | 1/6/11 | 260.8 | - | 13.4 | 8.11 | 166 | 1903 | 741 | 273 | 107 | −7.3 | 1870 | 1822 | 9 | 38 |
| Adige | Chizzola | 19/10/11 | 175.2 | - | 9.6 | 7.89 | 269 | 2266 | 1010 | 396 | 150 | −8.5 | 2280 | 2192 | 6 | 82 |
| Adige | Peri | 15/6/08 | 484.7 | - | 11.6 | 8.11 | 169 | 1515 | 780 | 285 | 93 | −7.5 | 1490 | 1451 | 7 | 31 |
| Adige | Peri | 30/5/11 | 312.6 | - | 13.9 | 8.14 | 174 | 1817 | 720 | 269 | 103 | - | 1780 | 1737 | 10 | 33 |
| Adige | Peri | 19/10/11 | 175.2 | - | 8.7 | 8.08 | 262 | 2018 | 941 | 399 | 138 | −7.5 | 2000 | 1943 | 8 | 48 |
| Adige | Avisio | 17/6/08 | 404.2 | - | 12.7 | 8.32 | 163 | 1666 | 743 | 228 | 147 | −7.2 | 1619 | 1585 | 13 | 21 |
| Adige | Avisio | 31/5/11 | 259.3 | - | 16.3 | 8.3 | 169 | 1647 | 732 | 229 | 117 | −5.8 | 1596 | 1563 | 13 | 20 |
| Adige | Avisio | 19/10/11 | 175.2 | - | 9.7 | 8.5 | 329 | 2687 | 1314 | 449 | 151 | −6.6 | 2619 | 2565 | 30 | 23 |
| Adige | Ala | 15/6/08 | 484.7 | - | 12.3 | 8.44 | 240 | 2993 | 1049 | 679 | 29 | −6.7 | 2914 | 2854 | 31 | 28 |
| Adige | Ala | 1/6/11 | 260.8 | - | 14.2 | 8.43 | 216 | 3257 | 929 | 593 | 27 | −7.0 | 3165 | 3100 | 35 | 30 |
| Adige | Ala | 19/10/11 | 175.2 | - | 9.5 | 8.48 | 293 | 3601 | 1055 | 696 | 28 | −7.1 | 3511 | 3440 | 38 | 33 |
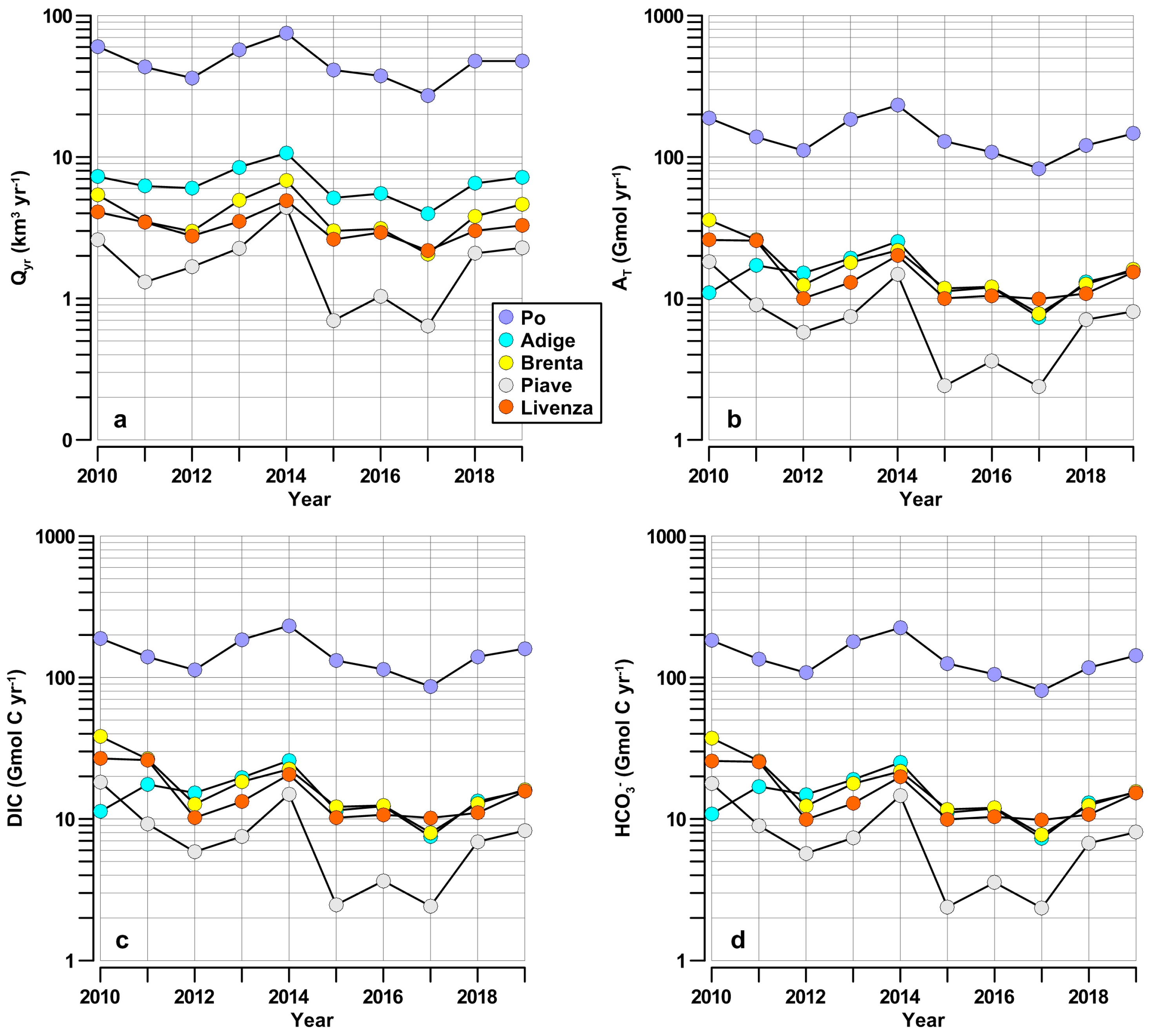
| Year | Freshwater | AT | DIC | HCO3− | Year | Freshwater | AT | DIC | HCO3− |
|---|---|---|---|---|---|---|---|---|---|
| km3 yr−1 | Gmol yr−1 | Gmol C yr−1 | Gmol C yr−1 | km3 yr−1 | Gmol yr−1 | Gmol C yr−1 | Gmol C yr−1 | ||
| Po River | Piave River | ||||||||
| 2010 | 60.47 | 188.52 | 188.99 | 182.90 | 2010 | 2.60 | 18.16 | 18.20 | 17.80 |
| 2011 | 43.26 | 138.46 | 139.74 | 134.43 | 2011 | 1.30 | 9.04 | 9.21 | 8.96 |
| 2012 | 36.21 | 111.32 | 113.07 | 108.11 | 2012 | 1.67 | 5.77 | 5.85 | 5.71 |
| 2013 | 57.16 | 184.26 | 185.38 | 178.91 | 2013 | 2.27 | 7.46 | 7.52 | 7.35 |
| 2014 | 75.09 | 232.17 | 232.63 | 224.85 | 2014 | 4.40 | 14.77 | 14.95 | 14.61 |
| 2015 | 41.20 | 129.24 | 131.83 | 125.60 | 2015 | 0.70 | 2.41 | 2.47 | 2.39 |
| 2016 | 37.44 | 108.25 | 114.35 | 105.27 | 2016 | 1.04 | 3.61 | 3.65 | 3.55 |
| 2017 | 27.23 | 82.79 | 86.19 | 80.65 | 2017 | 0.64 | 2.37 | 2.42 | 2.35 |
| 2018 | 47.68 | 120.92 | 140.20 | 117.63 | 2018 | 2.08 | 3.09 | 7.18 | 7.01 |
| 2019 | 47.70 | 146.61 | 160.06 | 142.96 | 2019 | 2.28 | 8.10 | 8.27 | 8.05 |
| AV | 47.34 | 144.25 | 149.25 | 140.13 | AV | 1.90 | 7.48 | 7.97 | 7.78 |
| SD | 13.11 | 42.73 | 41.15 | 41.29 | SD | 1.06 | 5.09 | 4.90 | 4.80 |
| Adige River | Livenza River | ||||||||
| 2010 | 7.27 | 10.93 | 11.30 | 10.85 | 2010 | 4.08 | 25.92 | 26.75 | 25.70 |
| 2011 | 6.23 | 17.14 | 17.53 | 16.96 | 2011 | 3.46 | 25.61 | 26.07 | 25.37 |
| 2012 | 6.03 | 15.08 | 15.27 | 14.88 | 2012 | 2.76 | 10.00 | 10.18 | 9.90 |
| 2013 | 8.47 | 19.20 | 19.61 | 19.00 | 2013 | 3.50 | 12.98 | 13.27 | 12.88 |
| 2014 | 10.67 | 25.29 | 25.90 | 25.06 | 2014 | 4.92 | 20.11 | 20.60 | 19.86 |
| 2015 | 5.14 | 11.23 | 11.50 | 11.09 | 2015 | 2.61 | 10.02 | 10.19 | 9.92 |
| 2016 | 5.51 | 11.96 | 12.31 | 11.85 | 2016 | 2.92 | 10.46 | 10.70 | 10.37 |
| 2017 | 3.98 | 7.35 | 7.50 | 7.29 | 2017 | 2.18 | 9.92 | 10.17 | 9.84 |
| 2018 | 6.51 | 13.10 | 13.34 | 12.97 | 2018 | 3.01 | 10.84 | 11.03 | 10.71 |
| 2019 | 7.19 | 15.55 | 15.79 | 15.36 | 2019 | 3.29 | 15.34 | 15.67 | 15.22 |
| AV | 6.70 | 14.68 | 15.00 | 14.53 | AV | 3.27 | 15.12 | 15.46 | 14.98 |
| SD | 1.77 | 4.79 | 4.89 | 4.74 | SD | 0.74 | 6.13 | 6.30 | 6.07 |
| Brenta River | |||||||||
| 2010 | 5.39 | 35.91 | 38.28 | 37.27 | |||||
| 2011 | 3.49 | 25.98 | 26.52 | 25.74 | |||||
| 2012 | 2.98 | 12.45 | 12.71 | 12.33 | |||||
| 2013 | 4.93 | 17.92 | 18.35 | 17.79 | |||||
| 2014 | 6.82 | 21.87 | 22.46 | 21.68 | |||||
| 2015 | 3.00 | 11.77 | 12.18 | 11.68 | |||||
| 2016 | 3.11 | 12.09 | 12.45 | 12.01 | |||||
| 2017 | 2.06 | 7.79 | 7.98 | 7.71 | |||||
| 2018 | 3.81 | 12.60 | 12.86 | 12.48 | |||||
| AV | 3.95 | 17.60 | 18.20 | 17.63 | |||||
| SD | 1.40 | 8.40 | 8.96 | 8.74 |
| River Name | Basin Area | Water Flow | Specific Runoff | Carbonate in Soil | Basin Latitude | HCO3— | DIC flux | HCO3− Flux | HCO3− Weathering Intensity | DIC Weathering Intensity | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|
| km2 | km3 yr−1 | L km−2 s−1 | % | Degree | µmol L−1 | Gmol yr−1 | Gmol yr−1 | mmol km−2 s−1 | mmol km−2 s−1 | ||
| Po | 70,091 | 47.30 | 21.4 | - | 45 N | 2929 | 148.0 | 139.82 | 63.25 | 66.98 | This study |
| Adige | 11,954 | 6.70 | 17.8 | - | 45 N | 2131 | 15.0 | 14.53 | 38.55 | 39.80 | This study |
| Brenta | 2280 | 3.95 | 55.0 | - | 45 N | 3325 | 18.2 | 17.63 | 245.23 | 253.11 | This study |
| Piave | 3899 | 1.90 | 15.4 | - | 46 N | 3629 | 8.0 | 7.78 | 63.27 | 64.84 | This study |
| Livenza | 2222 | 3.27 | 46.7 | - | 46 N | 4309 | 15.5 | 14.98 | 213.73 | 220.66 | This study |
| Tagliamento | 2582 | 1.35 | 16.5 | - | 46 N | 3231 | 4.7 | 4.62 | 56.70 | 57.89 | This study |
| Isonzo | 3452 | 1.20 | 11.0 | - | 46 N | 2723 | 3.4 | 3.30 | 30.29 | 31.30 | This study |
| Amazon | 5,854,000 | 6642 | 36.0 | 3.9 | 2 S | 369 | 2450 | 2450.90 | 13.28 | 13.27 | [5] |
| Congo | 3,699,000 | 1308 | 11.2 | 10.1 | 4 S | 224 | 293 | 292.99 | 2.51 | 2.51 | [5] |
| Mississippi | 3,203,000 | 610 | 6.0 | 18.1 | 36 N | 2074 | 1265 | 1265.14 | 12.52 | 12.52 | [5] |
| Niger | 2,240,000 | 193 | 2.7 | 6.3 | 10 N | 550 | 106 | 106.15 | 1.50 | 1.50 | [5] |
| Changjiang | 1,794,000 | 944 | 16.7 | 44.0 | 30 N | 1780 | 1680 | 1680.32 | 29.70 | 29.69 | [5] |
| Mackenzie | 1,713,000 | 290 | 5.4 | 20.6 | 64 N | 1800 | 522 | 522.00 | 9.66 | 9.66 | [5] |
| St Lawrence | 1,267,000 | 363 | 9.1 | 24.9 | 47 N | 1339 | 486 | 486.06 | 12.16 | 12.16 | [5] |
| Danube | 788,000 | 202 | 8.1 | 14.5 | 48 N | 3115 | 629 | 629.23 | 25.32 | 25.31 | [5] |
| Brahmaputra | 583,000 | 628 | 34.2 | 33.8 | 25 N | 1114 | 700 | 699.59 | 38.05 | 38.07 | [5] |
| Asian tropical rivers | 11,342,854 | 8694 | 24.3 | - | 30 N-30 S | 1064 * | 9241 | - | - | 25.85 | [57] |
| River | Period | N | Ca2+AV | Ca2+ SD | Mg2+AV | Mg2+ SD |
|---|---|---|---|---|---|---|
| mmol L−1 | mmol L−1 | mmol L−1 | mmol L−1 | |||
| Po | 2010–2019 | 82 | 1.45 | 0.23 | 0.48 | 0.09 |
| Adige | 2010–2019 | 28 | 1.00 | 0.34 | 0.45 | 0.33 |
| Brenta | 2010–2018 | 15 | 1.05 | 0.32 | 0.38 | 0.13 |
| Piave | 2010–2019 | 15 | 1.62 | 0.17 | 0.82 | 0.46 |
| Livenza | 2010–2019 | 7 | 1.52 | 0.27 | 0.68 | 0.11 |
| Tagliamento | 2008–2019 | 47 | 1.93 | 0.30 | 0.85 | 0.10 |
| Isonzo | 2007; 2008; 2011; 2016 | 18 | 1.20 | 0.26 | 0.36 | 0.14 |
| Timavo | 1998–2003;2006–2012; 2016 | 173 | 1.96 | 0.15 | 0.31 | 0.05 |
| Rižana | 2007, 2008, 2011 | 5 | 2.04 | 0.06 | 0.24 | 0.03 |
| Dragonja | 2007, 2008, 2011 | 3 | 2.40 | 0.45 | 2.58 | 1.72 |
References
- Probst, J.L.; Mortatti, J.; Tardy, Y. Carbon river fluxes and weathering CO2 consumption in the Congo and Amazon river basins. Appl. Geochem. 1994, 9, 1–13. [Google Scholar] [CrossRef]
- Salisbury, J.; Vandemark, D.; Hunt, C.; Campbell, J.; Jonsson, B.; Mahadevan, A.; McGillis, W.; Xue, H. Episodic riverine influence on surface DIC in the coastal Gulf of Maine. Estuar. Coast. Shelf Sci. 2009, 82, 108–118. [Google Scholar] [CrossRef]
- Drake, T.W.; Raymond, P.A.; Spencer, R.G.M. Terrestrial carbon inputs to inland waters: A current synthesis of estimates and uncertainty. Limnol Oceanogr. Lett. 2018, 3, 132–142. [Google Scholar] [CrossRef]
- Dickson, A.G. The development of the alkalinity concept in marine chemistry. Mar. Chem. 1992, 40, 49–63. [Google Scholar] [CrossRef]
- Cai, W.-J.; Guo, X.; Chen, C.-T.A.; Dai, M.; Zhang, L.; Zhai, W.; Lohrenz, S.E.; Yin, K.; Harrison, P.J.; Wang, Y. A comparative overview of weathering intensity and HCO3- flux in the world’s major rivers with emphasis on the Changjiang, Huanghe, Zhujiang (Pearl) and Mississippi Rivers. Cont. Shelf Res. 2008, 28, 1538–1549. [Google Scholar] [CrossRef]
- Berner, E.K.; Berner, R.A. Global Environment. Water, Air, and Geochemical Cycles; Princeton University Press: Princeton, NJ, USA, 2012; p. 444. ISBN 9780691136783. [Google Scholar]
- Kitidis, V.; Shutler, J.D.; Ashton, I.; Warren, M.; Brown, I.; Findlay, H.; Hartman, S.E.; Sanders, R.; Humphreys, M.; Kivimäe, C.; et al. Winter weather controls net influx of atmospheric CO2 on the north-west European shelf. Sci. Rep. 2019, 9, 20153. [Google Scholar] [CrossRef]
- Amiotte Suchet, P.; Probst, J.L.; Ludwig, W. Worldwide distribution of continental rock lithology: Implications for the atmospheric/soil CO2 uptake by continental weathering and alkalinity river transport to the oceans. Glob. Biogeochem. Cy. 2003, 17, 1038. [Google Scholar] [CrossRef]
- Ludwig, W.; Amiotte Suchet, P.; Munhoven, G.; Probst, J.L. Atmospheric CO2 consumption by continental erosion: Present-day controls and implications for the last glacial maximum. Glob. Planet. Chang. 1998, 16–17, 95–108. [Google Scholar] [CrossRef]
- Ludwig, W.; Amiotte Suchet, P.; Probst, J.-L. Enhanced chemical weathering of rocks during the last glacial maximum: A sink for atmospheric CO2? Chem. Geol. 1999, 159, 147–161. [Google Scholar] [CrossRef]
- Barnes, R.T.; Raymond, P.A. The contribution of agricultural and urban activities to inorganic carbon fluxes within temperate watersheds. Chem. Geol. 2009, 266, 318–327. [Google Scholar] [CrossRef]
- Xuan, Y.; Cao, Y.; Tang, C.; Li, M. Changes in dissolved inorganic carbon in river water due to urbanization revealed by hydrochemistry and carbon isotope in the Pearl River Delta, China. Environ. Sci. Pollut. Res. 2020, 27, 24542–24557. [Google Scholar] [CrossRef] [PubMed]
- das Neves Lopes, M.; Decarli, C.J.; Pinheiro-Silva, L.; Lima, T.C.; Leite, N.K.; Petrucio, M.M. Urbanization increases carbon concentration and pCO2 in subtropical streams. Environ. Sci. Pollut. Res. 2020, 27, 18371–18381. [Google Scholar] [CrossRef] [PubMed]
- Raymond, P.A.; Hamilton, S.K. Anthropogenic influences on riverine fluxes of dissolved inorganic carbon to the oceans. Limnol. Oceanogr. Letters. 2018, 3, 143–155. [Google Scholar] [CrossRef]
- Copin-Montegut, C. Alkalinity and carbon budgets in the Mediterranean Sea. Glob. Biogeochem. Cy. 1993, 7, 915–925. [Google Scholar] [CrossRef]
- Schneider, A.; Wallace, D.W.R.; Körtzinger, A. Alkalinity of the Mediterranean Sea. Geophys. Res. Lett. 2007, 34, L15608. [Google Scholar] [CrossRef]
- Álvarez, M.; Sanleón-Bartolomé, H.; Tanhua, T.; Mintrop, L.; Luchetta, A.; Cantoni, C.; Schroeder, K.; Civitarese, G. The CO2 system in the Mediterranean Sea: A basin wide perspective. Ocean Sci. 2014, 10, 69–92. [Google Scholar] [CrossRef]
- Cossarini, G.; Lazzari, P.; Solidoro, C. Spatiotemporal variability of alkalinity in the Mediterranean Sea. Biogeosciences 2015, 12, 1647–1658. [Google Scholar] [CrossRef]
- Rivaro, P.; Messa, R.; Massolo, S.; Frache, R. Distributions of carbonate properties along the water column in the Mediterranean Sea: Spatial and temporal variations. Mar. Chem. 2010, 121, 236–245. [Google Scholar] [CrossRef]
- Ingrosso, G.; Giani, M.; Cibic, T.; Karuza, A.; Kralj, M.; Del Negro, P. Carbonate chemistry dynamics and biological processes along a river-sea gradient (Gulf of Trieste, northern Adriatic Sea). J. Mar. Syst. 2016, 155, 35–49. [Google Scholar] [CrossRef]
- Luchetta, A.; Cantoni, C.; Catalano, G. New observations of CO2 induced acidification in the Northern Adriatic Sea, over the last quarter century. Chem. Ecol. 2010, 26, 1–17. [Google Scholar] [CrossRef]
- Urbini, L.; Ingrosso, G.; Djakovac, T.; Piacentino, S.; Giani, M. Temporal and spatial variability of the CO2 system in a riverine influenced area of the Mediterranean Sea, the northern Adriatic. Front. Mar. Sci. 2020, 7, 679. [Google Scholar] [CrossRef]
- Feely, R.A.; Doney, S.C.; Cooley, S.R. Ocean Acidification: Present conditions and future changes in a high-CO2 World. Oceanography 2009, 22, 36–47. [Google Scholar] [CrossRef]
- Tamše, S.; Ogrinc, N.; Walter, L.M.; Turk, D.; Faganeli, J. River Sources of Dissolved Inorganic Carbon in the Gulf of Trieste (N Adriatic): Stable Carbon Isotope Evidence. Estuaries Coast. 2014, 38, 151–164. [Google Scholar] [CrossRef]
- Cioce, F.; Stocco, G.; Toniolo, R. Hydrological and physical–chemical investigations on the Po river at Posella. February 1973-February 1975. Atti Dell’istituto Veneto Di Sci. Lett. Ed Arti 1977, 135, 119–132. [Google Scholar]
- Fossato, V.U. Hydrological, chemical and physical investigations on the Po river at Posella. June 1968-June 1970. Archo Oceanogr. Limnol. 1971, 17, 125–139. [Google Scholar]
- Fossato, V.U. Hydrological, chemical and physical investigations on the Adige river at Boara Pisani. June 1968-June 1970. Archo Oceanogr. Limnol. 1971, 17, 105–123. [Google Scholar]
- Cozzi, S.; Giani, M. River water and nutrient discharges in the Northern Adriatic Sea: Current importance and long term changes. Cont. Shelf Res. 2011, 31, 1881–1893. [Google Scholar] [CrossRef]
- Volf, G.; Atanasova, N.; Kompare, B.; Ožanić, N. Modelling nutrient loads to the northern Adriatic. J. Hydrol. 2013, 504, 182–193. [Google Scholar] [CrossRef]
- Cozzi, S.; Ibáñez, C.; Lazar, L.; Raimbault, P.; Giani, M. Flow regime and nutrient-loading trends from the largest South European watersheds: Implications for the productivity of Mediterranean and Black Sea’s coastal areas. Water 2019, 11, 1. [Google Scholar] [CrossRef]
- Viaroli, P.; Soana, E.; Pecora, S.; Laini, A.; Naldi, M.; Fano, E.A.; Nizzoli, D. Space and time variations of watershed N and P budgets and their relationships with reactive N and P loadings in a heavily impacted river basin (Po River, Northern Italy). Sci. Total Environ. 2018, 639, 1574–1587. [Google Scholar] [CrossRef]
- Zhang, Q.; Cozzi, S.; Palinkas, C.; Giani, M. Recent status and long-term trends in freshwater discharge and nutrient inputs. In Coastal Ecosystems in Transition: A Comparative Analysis of the Northern Adriatic and Chesapeake Bay, 1st ed.; Geophysical Monograph; Malone, T., Malej, A., Faganeli, J., Eds.; Wiley & Sons Ltd.: Hoboken, NJ, USA, 2021; p. 256. ISBN 978-1-119-54358-9. [Google Scholar]
- Grilli, F.; Accoroni, S.; Acri, F.; Bernardi Aubry, F.; Bergami, C.; Cabrini, M.; Campanelli, A.; Giani, M.; Guicciardi, S.; Marini, M.; et al. Seasonal and interannual trends of oceanographic parameters over 40 years in the northern Adriatic Sea in relation to nutrient loadings from EMODnet Chemistry data portal. Water 2020, 12, 2280. [Google Scholar] [CrossRef]
- Ravazzani, G.; Barbero, S.; Salandin, A.; Senatore, A.; Mancini, M. An integrated hydrological model for assessing climate change impacts on water resources of the upper Po River basin. Water Resour. Manage. 2015, 29, 1193–1215. [Google Scholar] [CrossRef]
- Giani, M.; Djakovac, T.; Degobbis, D.; Cozzi, S.; Solidoro, C.; Fonda Umani, S. Recent changes in the marine ecosystems of the northern Adriatic Sea. Estuar. Coast. Shelf Sci. 2012, 115, 1–13. [Google Scholar] [CrossRef]
- APAT, IRSA-CNR. Metodi analitici per le acque; Manuali e guide APAT, 29/2003; IRSA-CNR: Brugheri, Italy, 2003; Volume 2, pp. 493–839. ISBN 88-448-0083-7. [Google Scholar]
- Jarvie, P.; King, S.M.; Neal, C. Inorganic carbon dominates total dissolved carbon concentrations and fluxes in British rivers: Application of the THINCARB model—Thermodynamic modelling of inorganic carbon in freshwaters. Sci. Tot. Environ. 2017, 575, 496–512. [Google Scholar] [CrossRef] [PubMed]
- Brunet, F.; Gaiero, D.; Probst, J.L.; Depetris, P.J.; Gauthier Lafaye, F.; Stille, P. δ13C tracing of dissolved inorganic carbon sources in Patagonian rivers (Argentina). Hydrol. Process. 2005, 19, 3321–3344. [Google Scholar] [CrossRef]
- Ravaioli, M.; Alvisi, F.; Menegazzo Vitturi, L. Dolomite as a tracer for sediment transport and deposition on the northwestern Adriatic continental shelf (Adriatic Sea, Italy). Cont. Shelf Res. 2003, 23, 1359–1377. [Google Scholar] [CrossRef]
- Tesi, T.; Miserocchi, S.; Acri, F.; Langone, L.; Boldrin, A.; Hatten, J.A.; Albertazzi, S. Flood-driven transport of sediment, particulate organic matter, and nutrients from the Po River watershed to the Mediterranean Sea. J. Hydrol. 2013, 498, 144–152. [Google Scholar] [CrossRef]
- Szramek, K.; Walter, L.M.; Kanduč, T.; Ogrinc, N. Dolomite versus Calcite weathering in hydrogeochemically diverse watersheds established on bedded carbonates (Sava and Soča Rivers, Slovenia). Aquat. Geochem. 2011, 17, 357–396. [Google Scholar] [CrossRef]
- Barešić, J.; Horvatinčić, N.; Roller-Lutz, Z. Spatial and seasonal variations in the stable C isotope composition of dissolved inorganic carbon and in physico-chemical water parameters in the Plitvice Lakes system. Isot. Environ. Health Stud 2011, 47, 316–329. [Google Scholar] [CrossRef]
- Chaplot, V.; Mutema, M. Sources and main controls of dissolved organic and inorganic carbon in river basins: A worldwide meta-analysis. J. Hydrol. 2021, 603, 126941. [Google Scholar] [CrossRef]
- Ludwig, W.; Dumont, E.; Meybeck, M.; Heussner, S. River discharges of water and nutrients to the Mediterranean and Black Sea: Major drivers for ecosystem changes during past and future decades? Prog. Oceanogr. 2009, 80, 199–217. [Google Scholar] [CrossRef]
- Roy, S.; Gaillardet, J.; Allègre, C.J. Geochemistry of dissolved and suspended loads of the Seine River, France: Anthropogenic impact, carbonate and silicate weathering. Geochim. Cosmochim. Acta. 1999, 63, 1277–1292. [Google Scholar] [CrossRef]
- Fairchild, I.J.; Killawee, J.A.; Sharp, M.J.; Spiro, B.; Hubbard, B.; Lorrain, R.D.; Tison, J.-L. Solute generation and transfer from a chemically reactive alpine glacial-proglacial system. Earth Surf. Process. Landf. 1999, 24, 1189–1211. [Google Scholar] [CrossRef]
- Anderson, S.P.; Drever, J.I.; Frost, C.D.; Holden, P. Chemical weathering in the foreland of a retreating glacier. Geochim. Cosmochim. Acta 2000, 64, 1173–1189. [Google Scholar] [CrossRef]
- Jacobson, A.D.; Blum, J.D. Relationship between mechanical erosion and atmospheric CO2 consumption in the New Zealand Southern Alps. Geology 2003, 31, 865–868. [Google Scholar] [CrossRef]
- Amiotte Suchet, P.; Aubert, D.; Probst, J.L.; Gauthier-Lafaye, F.; Probst, A.; Andreux, F.; Viville, D. δ13C pattern of dissolved inorganic carbon in a small granitic catchment: The Strengbach case study (Vosges Mountain, France). Chem. Geol. 1999, 159, 129–145. [Google Scholar] [CrossRef]
- Huang, T.H.; Fu, Y.H.; Pan, P.Y.; Chen, C.T.A. Fluvial carbon fluxes in tropical rivers. Curr. Opin. Environ. Sustain. 2012, 4, 162–169. [Google Scholar] [CrossRef]
- Huang, T.H.; Chen, C.T.A.; Tseng, H.C.; Lou, J.Y.; Wang, S.L.; Yang, L.; Kandasamy, S.; Gao, X.; Wang, J.T.; Aldrian, E.; et al. Riverine carbon fluxes to the South China Sea. J. Geophys. Res. Biogeosci. 2017, 122, 1239–1259. [Google Scholar] [CrossRef]
- Maier, M.-S.; Teodoru, R.C.; Wehrli, B. Spatio-temporal variations in lateral and atmospheric carbon fluxes from the Danube Delta. Biogeosciences 2021, 18, 1417–1437. [Google Scholar] [CrossRef]
- Rodellas, V.; Garcia-Orellana, J.; Masqué, P.; Feldman, M.; Weinstein, Y. Submarine groundwater discharge as a major source of nutrients to the Mediterranean Sea. Proceeding Natl. Acad. Sci. 2015, 112, 3926–3930. [Google Scholar] [CrossRef]
- Trezzi, G.; Garcia-Orellana, J.; Rodellas, V.; Masqué, P.; Garcia-Solson, E.; Andersson, P.S. Assessing the role of submarine groundwater discharge as a source of Sr to the Mediterranean Sea Geochim. Cosmochim. Acta 2017, 200, 42–54. [Google Scholar] [CrossRef]
- Cantoni, C.; Luchetta, A.; Celio, M.; Cozzi, S.; Raicich, F.; Catalano, G. Carbonate system variability in the Gulf of Trieste (North Adriatic Sea). Estuar. Coast. Shelf Sci. 2012, 115, 51–62. [Google Scholar] [CrossRef]
- Sempéré, R.; Charriére, B.; Van Wambeke, F.; Cauwet, G. Carbon inputs of the Rhône River to the Mediterranean Sea: Biogeochemical implication. Glob. Biogeochem. Cy. 2000, 14, 669–681. [Google Scholar] [CrossRef]
- Stets, E.G.; Kelly, V.J.; Crawford, C.G. Long-term trends in alkalinity in large rivers of the conterminous US in relation to acidification, agriculture, and hydrologic modification. Sci.Tot. Environ. 2014, 488–489, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.; Cai, W.J. Total alkalinity minus dissolved inorganic carbon as a proxy for deciphering ocean acidification mechanisms. Mar. Chem. 2020, 222, 103791. [Google Scholar] [CrossRef]
- Brush, M.J.; Giani, M.; Totti, C.; Testa, J.M.; Faganeli, J.; Ogrinc, N.; Kemp, W.M.; Fonda Umani, S. Eutrophication, Harmful Algae, Oxygen Depletion, and Acidification. In Coastal Ecosystems in Transition: A Comparative Analysis of the Northern Adriatic and Chesapeake Bay, 1st ed.; Geophysical, Monograph; Malone, T., Malej, A., Faganeli, J., Eds.; Wiley & Sons Ltd.: Hoboken, NJ, USA, 2021; Volume 256, pp. 75–104. ISBN 978-1-119-54358-9. [Google Scholar]
| River | pH | AT | DIC | HCO3− | [AT−DIC] | Ca2+ | Mg2+ | δ 13CDIC | |
|---|---|---|---|---|---|---|---|---|---|
| μmol L−1 | μmol L−1 | μmol L−1 | μmol L−1 | μmol L−1 | μmol L−1 | (‰) | |||
| N | 122 | 122 | 122 | 122 | 122 | 121 | 82 | 3 | |
| Po | AV | 7.78 | 3115 | 3205 | 3023 | −89 | 1456 | 480 | −9.63 |
| SD | 0.32 | 536 | 534 | 531 | 164 | 229 | 88 | 0.35 | |
| N | 239 | 237 | 237 | 237 | 237 | 25 | 25 | 15 | |
| Adige | AV | 8.03 | 2305 | 2260 | 2190 | 46 | 1056 | 489 | −6.95 |
| SD | 0.21 | 901 | 887 | 865 | 58 | 357 | 361 | 0.70 | |
| N | 123 | 123 | 123 | 123 | 123 | 15 | 15 | 2 | |
| Brenta | AV | 7.98 | 4473 | 4578 | 4428 | −105 | 1346 | 519 | −8.95 |
| SD | 0.18 | 1823 | 1855 | 1804 | 70 | 272 | 160 | 1.20 | |
| n | 122 | 122 | 122 | 122 | 122 | 15 | 15 | 6 | |
| Piave | AV | 8.05 | 4271 | 4352 | 4224 | −80 | 1632 | 850 | −7.82 |
| SD | 0.17 | 1549 | 1561 | 1522 | 78 | 182 | 486 | 1.73 | |
| n | 88 | 88 | 88 | 88 | 88 | 7 | 7 | - | |
| Livenza | AV | 8.02 | 4804 | 4766 | 4614 | 38 | 1525 | 683 | - |
| SD | 0.15 | 1643 | 1670 | 1606 | 131 | 270 | 112 | - | |
| N | 47 | 7 | 7 | 7 | 7 | 44 | 44 | 10 | |
| Tagliamento | AV | 8.05 | 3461 | 3454 | 3369 | 8 | 1872 | 879 | −7.13 |
| SD | 0.16 | 190 | 193 | 185 | 19 | 207 | 109 | 0.48 | |
| N | 90 | 77 | 76 | 76 | 76 | 18 | 18 | 5 | |
| Isonzo | AV | 7.99 | 3695 | 3658 | 3537 | 84 | 1233 | 375 | −8.12 |
| SD | 0.18 | 548 | 561 | 533 | 419 | 236 | 119 | 1.20 | |
| N | 196 | 63 | 63 | 63 | 63 | 149 | 149 | 12 | |
| Timavo | AV | 7.49 | 4016 | 4408 | 3950 | −393 | 1843 | 281 | −11.69 |
| SD | 0.20 | 276 | 424 | 277 | 249 | 172 | 60 | 0.61 | |
| N | 5 | 5 | 5 | 5 | 5 | 4 | 4 | 4 | |
| Dragonja | AV | 7.50 | 5247 | 5875 | 5144 | −628 | 2405 | 2578 | −11.60 |
| SD | 0.51 | 959 | 766 | 939 | 761 | 447 | 1724 | 0.27 | |
| N | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | |
| Rižana | AV | 8.19 | 4429 | 4410 | 4309 | 19 | 2044 | 244 | −11.33 |
| SD | 0.09 | 325 | 335 | 326 | 13 | 65 | 25 | 0.22 |
| Water | AT | DIC | HCO3− | |
|---|---|---|---|---|
| Bulk NAd | 266 km3 | 729.2 ± 9.2 Gmol | 645.0 ± 11.9 Gmol | 579.5 ± 19.5 Gmol |
| River | 65.7 ± 24.5 km3 yr−1 | 205.2 ± 75.8 Gmol yr−1 | 212.8 ± 72.6 Gmol yr−1 | 202.6 ± 72.2 Gmol yr−1 |
| discharge | 24.7 ± 9.2% yr−1 | 28.1 ± 10.7% yr−1 | 33.0 ± 11.9% yr−1 | 35.0 ± 13.6% yr−1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giani, M.; Ogrinc, N.; Tamše, S.; Cozzi, S. Elevated River Inputs of the Total Alkalinity and Dissolved Inorganic Carbon in the Northern Adriatic Sea. Water 2023, 15, 894. https://doi.org/10.3390/w15050894
Giani M, Ogrinc N, Tamše S, Cozzi S. Elevated River Inputs of the Total Alkalinity and Dissolved Inorganic Carbon in the Northern Adriatic Sea. Water. 2023; 15(5):894. https://doi.org/10.3390/w15050894
Chicago/Turabian StyleGiani, Michele, Nives Ogrinc, Samo Tamše, and Stefano Cozzi. 2023. "Elevated River Inputs of the Total Alkalinity and Dissolved Inorganic Carbon in the Northern Adriatic Sea" Water 15, no. 5: 894. https://doi.org/10.3390/w15050894
APA StyleGiani, M., Ogrinc, N., Tamše, S., & Cozzi, S. (2023). Elevated River Inputs of the Total Alkalinity and Dissolved Inorganic Carbon in the Northern Adriatic Sea. Water, 15(5), 894. https://doi.org/10.3390/w15050894








