Heavy Metals Removal from Domestic Sewage in Batch Mesocosm Constructed Wetlands using Tropical Wetland Plants
Abstract
:1. Introduction
2. Materials and Methods
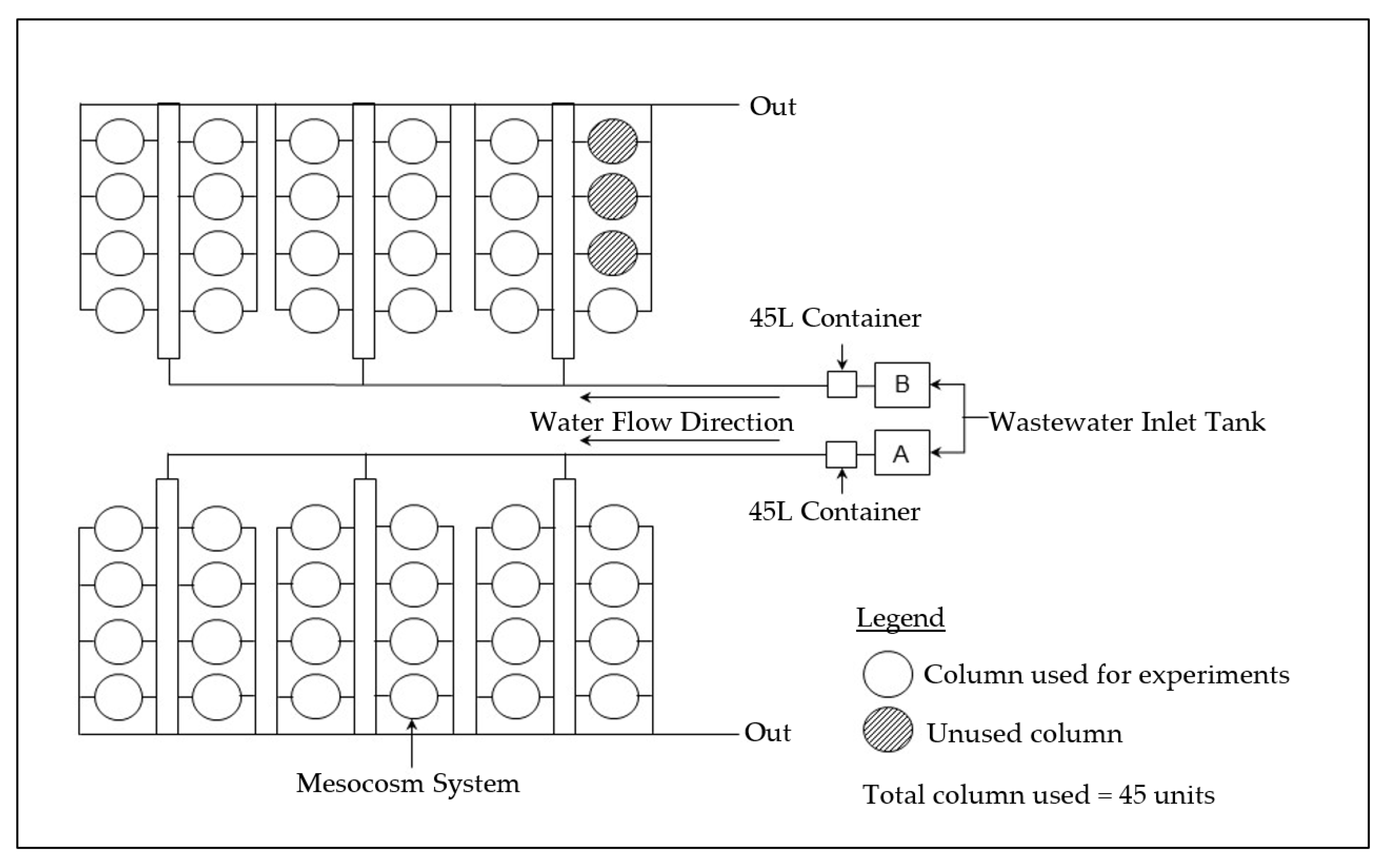
2.1. Experimental Design
2.2. Tropical Wetland Plant Selection and Cultivation
2.3. Sample Collection and Preservation
2.4. Plant and Soil Digestion
2.5. Bioconcentration Factor (BCF) and Translocation Factor (TF) for Plant Data Analysis
2.6. Enrichment Factor (EF) and Geoaccumulation Index (Igeo) for Soil Data Analysis
2.7. Statistical Analysis
3. Results and Discussion
3.1. Bioconcentration Factor (BCF)
3.2. Translocation Factor (TF)
3.3. Phytoextration and Phytostabilisation Property
3.4. Enrichment Factor (EF) and Geoaccumulation Index (Igeo)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boutin, C.; Eme, C. Domestic Wastewater Characterization by Emission Source. In Proceedings of the 13eme congress spécialisé IWA on Small Water and Wastewater Systems, Athènes, Greece, 14–16 September 2016. [Google Scholar]
- Hernández Leal, L.; Temmink, H.; Zeeman, G.; Buisman, C.J.N. Comparison of Three Systems for Biological Greywater Treatment. Water 2010, 2, 155–169. [Google Scholar] [CrossRef]
- De Graaff, M.S.; Temmink, H.; Zeeman, G.; Buisman, C.J.N. Anaerobic Treatment of Concentrated Black Water in a UASB Reactor at a Short HRT. Water 2010, 2, 101–119. [Google Scholar] [CrossRef]
- Jiao, Y. Chapter 2 Waste to biohydrogen: Potential and feasibility. In Waste to Renewable Biohydrogen; Zhang, Q., He, C., Ren, J., Goodsite, M., Eds.; Academic Press: London, UK, 2021; pp. 33–53. [Google Scholar]
- Wang, C.; Feng, B.; Wang, P.; Guo, W.; Li, X.; Gao, H. Revealing factors influencing spatial variation in the quantity and quality of rural domestic sewage discharge across China. Process Saf. Environ. Prot. 2022, 162, 200–210. [Google Scholar] [CrossRef]
- Speight, J.G. Sources of water pollution. In Natural Water Remediation; Speight, J.G., Ed.; Butterworth-Heinemann: Oxford, UK, 2020; pp. 165–198. [Google Scholar]
- Wear, S.L.; Acuña, V.; McDonald, R.; Font, C. Sewage pollution, declining ecosystem health, and cross-sector collaboration. Biol. Conserv. 2021, 255, 109010. [Google Scholar] [CrossRef]
- Ariffin, M.; Sulaiman, S.N. Regulating sewage pollution of Malaysian rivers and its challenges. International Conference on Environmental Forensics 2015 (iENFORCE2015). Procedia Environ. Sci. 2015, 30, 168–173. [Google Scholar] [CrossRef] [Green Version]
- Rivera, C.; Quirogac, E.; Mezad, V.; Pastenec, M. Evaluation of water quality and heavy metal concentrations in the RAMSAR Wetland El Yali (Central Chile, 33°45′ S). Mar. Pollut. Bull. 2019, 145, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Sanfilippo, M.; Albano, M.; Manganaro, A.; Capillo, G.; Spanò, N.; Savoca, S. Spatiotemporal Organic Carbon Distribution in the Capo Peloro Lagoon (Sicily, Italy) in Relation to Environmentally Sustainable Approaches. Water 2022, 14, 108. [Google Scholar] [CrossRef]
- Wear, S.L.; Thurber, R.V. Sewage pollution: Mitigation is key for coral reef stewardship. Ann. N. Y. Acad. Sci. 2015, 1355, 15–30. [Google Scholar] [CrossRef]
- Reef Check Malaysia (RCM). Status of Coral Reef in Malaysia 2019. 2019. Available online: https://static1.squarespace.com/static/5c9c815e348cd94acf3b352e/t/5e4e461750bc066ba303bbc5/1582188086832/2019+Annual+Survey+Report.pdf (accessed on 21 June 2020).
- Department of Marine Park Malaysia. Pulau Tioman Marine Park Management Plan. 2013. Available online: https://wdpa.s3.amazonaws.com/Country_informations/MYS/Pulau_Tioman_3150.pdf (accessed on 7 September 2021).
- DOE. Environmental Quality Report (EQR) 2018; Department of Environment: Putrajaya, Malaysia, 2018. [Google Scholar]
- Tangahu, B.V.; Sheikh Abdullah, S.R.; Basri, H.; Idris, M.; Anuar, N.; Mukhlisin, M. A review on heavy metals (As, Pb, and Hg) uptake by plants through phytoremediation. Int. J. Chem. Eng. 2011, 2011, 939161. [Google Scholar] [CrossRef]
- Moriyama, K.; Mori, T.; Arayashiki, H.; Saitot, H.; Chino, M. The amount of heavy metals derived from domestic wastewater. Water Sci. Technol. 1989, 21, 1913–1916. [Google Scholar] [CrossRef]
- Drozdova, J.; Raclavsky, H.; Raclavsky, K.; Skrobankova, H. Heavy metals in domestic wastewater with respect to urban population in Ostrava, Czech Republic. Water Environ. J. 2019, 33, 77–85. [Google Scholar] [CrossRef] [Green Version]
- Rule, K.L.; Comber, S.D.; Ross, D.; Thornton, A.; Makropoulos, C.K.; Rautiu, R. Diffuse sources of heavy metals entering an urban wastewater catchment. Chemosphere 2006, 63, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdiscip. Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef] [Green Version]
- D’Souza, H.S.; Menezes, G.; Venkatesh, T. Role of essential trace minerals on the absorption of heavy metals with special reference to lead. Indian J. Clin. Biochem. 2003, 18, 154–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agoro, M.A.; Adeniji, A.O.; Adefisoye, M.A.; Okoh, O.O. Heavy metals in wastewater and sewage sludge from selected municipal treatment plants in Eastern Cape Province, South Africa. Water 2020, 12, 2746. [Google Scholar] [CrossRef]
- Jyothi, N.R. Heavy Metal Sources and Their Effects on Human Health. In Heavy Metals-Their Environmental Impacts and Mitigation; Nazal, M.K., Zhao, H., Eds.; IntechOpen: London, UK, 2020. [Google Scholar]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy metal toxicity and the environment. Exp. Suppl. 2012, 101, 133–164. [Google Scholar]
- Titilawo, Y.; Adeniji, A.; Adeniyi, M.; Okoh, A. Determination of levels of some metal contaminants in the freshwater environments of Osun State, Southwest Nigeria: A risk assessment approach to predict health threat. Chemosphere 2018, 211, 834–843. [Google Scholar] [CrossRef]
- Alloway, B.J. Heavy Metals and Metalloids as Micronutrients for Plants and Animals. In Heavy Metals in Soils; Environmental Pollution; Alloway, B., Ed.; Springer: Dordrecht, The Netherlands, 2013; Volume 22, pp. 195–209. [Google Scholar]
- Schück, M.; Greger, M. Screening the capacity of 34 wetland plant species to remove heavy metals from water. Int. J. Environ. Res. Pub. Health 2020, 17, 4623. [Google Scholar] [CrossRef]
- Rahman, M.E.; Bin Halmi, M.; Bin Abd Samad, M.Y.; Uddin, M.K.; Mahmud, K.; Abd Shukor, M.Y.; Sheikh Abdullah, S.R.; Shamsuzzaman, S.M. Design, Operation and Optimisation of Constructed Wetland for Removal of Pollutant. Int. J. Environ. Res. Pub. Health 2020, 17, 8339. [Google Scholar] [CrossRef]
- Valipour, A.; Hamnabard, N.; Woo, K.S.; Ahn, Y.H. Performance of high-rate constructed phytoremediation process with attached growth for domestic wastewater treatment: Effect of high TDS and Cu. J. Environ. Manag. 2014, 145, 1–8. [Google Scholar] [CrossRef]
- Mustafa, A. Constructed wetland for wastewater treatment and reuse: A case study of developing country. Int. J. Environ. Sci. Dev. 2013, 4, 20. [Google Scholar] [CrossRef] [Green Version]
- Aydın Temel, F.; Avcı, E.; Ardalı, Y. Full scale horizontal subsurface flow constructed wetlands to treat domestic wastewater by Juncus acutus and Cortaderia selloana. Int. J. Phytoremediat. 2018, 20, 264–273. [Google Scholar] [CrossRef] [PubMed]
- ElZein, Z.; Abdou, A.; ElGawad, I.A. Constructed Wetlands as a Sustainable Wastewater Treatment Method in Communities. Procedia Environ. Sci. 2016, 34, 605–617. [Google Scholar] [CrossRef] [Green Version]
- Stottmeister, U.; Wießner, A.; Kuschk, P.; Kappelmeyer, U.; Kästner, M.; Bederski, O.; Müller, R.A.; Moormann, H. Effects of plants and microorganisms in constructed wetlands for wastewater treatment. Biotechnol. Adv. 2003, 22, 93–117. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, H.M.; Hayder, G. Recent studies on applications of aquatic weed plants in phytoremediation of wastewater: A review article. Ain Shams Eng. J. 2021, 12, 355–365. [Google Scholar] [CrossRef]
- Brix, H.; Koottatep, T.; Fryd, O.; Laugesen, C.H. The flower and the butterfly constructed wetland system at Koh Phi Phi—System design and lessons learned during implementation and operation. Ecol. Eng. 2011, 37, 729–735. [Google Scholar] [CrossRef] [Green Version]
- Fu, X.X.; Wu, X.F.; Zhou, S.Y.; Chen, Y.H.; Chen, M.L.; Chen, R.H. A Constructed Wetland System for Rural Household Sewage Treatment in Subtropical Regions. Water 2018, 10, 716. [Google Scholar] [CrossRef] [Green Version]
- Sandoval, L.; Zamora-Castro, S.A.; Vidal-Álvarez, M.; Marín-Muñiz, J.L. Role of wetland plants and use of ornamental flowering plants in constructed wetlands for wastewater treatment: A review. Appl. Sci. 2019, 9, 685. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.M.; Zhang, J.; Guo, W.S.; Liang, S.; Fan, J.L. Secondary effluent purification by a large-scale multi-stage surface-flow constructed wetland: A case study in northern China. Bioresour. Technol. 2018, 249, 1092–1096. [Google Scholar] [CrossRef]
- Md. Akhir, M.S.A.; Amir, A.A.; Mokhtar, M.; Wong, A.K.H. Constructed Wetland for Wastewater Treatment: A Case Study at Frangipani Resort, Langkawi. Int. J. Malay World Civilis. (Iman) 2016, 4, 21–28. [Google Scholar]
- Mustafa, H.M.; Hayder, G. Cultivation of S. molesta plants for phytoremediation of secondary treated domestic wastewater. Ain Shams Eng. J. 2021, 12, 2585–2592. [Google Scholar] [CrossRef]
- Mustafa, H.M.; Hayder, G. Performance of Salvinia molesta plants in tertiary treatment of domestic wastewater. Heliyon 2021, 7, e06040. [Google Scholar] [CrossRef] [PubMed]
- Ojoawa, S.O.; Udayakumar, G.; Naik, P. Phytoremediation of Phosphorus and nitrogen with Canna x generalis Reeds in Domestic Wastewater through NMAMIT Constructed Wetland. Aquat. Procedia 2015, 4, 349–356. [Google Scholar] [CrossRef]
- Patil, Y.M.; Munavalli, G.R. Performance evaluation of an Integrated On-site Greywater Treatment System in a tropical region. Ecol. Eng. 2016, 95, 492–500. [Google Scholar] [CrossRef]
- Cho-Ruk, K.; Kurukote, J.; Supprung, P.; Vetayasuporn, S. Perennial plants in the phytoremediation of lead-contaminated soils. Biotechnology 2006, 5, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Mantovi, P.; Marmiroli, M.; Maestri, E.; Tagliavini, S.; Piccinini, S.; Marmiroli, N. Application of a horizontal subsurface flow constructed wetland on treatment of dairy parlor wastewater. Bioresour. Technol. 2003, 88, 85–94. [Google Scholar] [CrossRef]
- Walaszek, M.; Bois, P.; Laurent, J.; Lenormand, E.; Wanko, A. Urban stormwater treatment by a constructed wetland: Seasonality impacts on hydraulic efficiency, physico-chemical behavior and heavy metal occurrence. Sci. Total Environ. 2018, 637, 443–454. [Google Scholar] [CrossRef]
- Mustapha, H.I.; van Bruggen, J.J.A.; Lens, P.N.L. Fate of heavy metals in vertical subsurface flow constructed wetlands treating secondary treated petroleum refinery wastewater in Kaduna, Nigeria. Int. J. Phytoremediation 2018, 20, 44–53. [Google Scholar] [CrossRef]
- Yeh, T.Y.; Chou, C.C.; Pan, C.T. Heavy metal removal within pilot-scale constructed wetlands receiving river water contaminated by confined swine operations. Desalination 2009, 249, 368–373. [Google Scholar] [CrossRef]
- DID. Urban Stormwater Management for Malaysia (MSMA), 2nd ed.; Department of Irrigation (DID) Malaysia: Kuala Lumpur, Malaysia, 2012; pp. 11–19. [Google Scholar]
- Radojevic, M.; Bashkin, V.N. Practical Environmental Analysis, 2nd ed.; RSC Publishing: Cambridge, UK, 2006. [Google Scholar]
- Che Abdullah, M.I.; Md Shah, A.S.R.; Haris, H. Geoaccumulation index and enrichment factor of Arsenic in surface sediment of Bukit Merah Reservoir, Malaysia. Trop. Life Sci. Res. 2020, 31, 109–125. [Google Scholar] [CrossRef]
- Mishra, P.; Pandey, V.C. Chapter 16: Phytoremediation of red mud deposits through natural succession. In Phytomanagement of Polluted Sites; Pandey, V.C., Bauddh, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 409–424. [Google Scholar]
- Zhang, W.; Cai, Y.; Tu, C.; Ma, L.Q. Arsenic speciation and distribution in an arsenic hyperaccumulating plant. Sci. Total Environ. 2002, 300, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.C. Phytoremediation of heavy metals from fly ash pond by Azolla caroliniana. Ecotoxicol. Environ. Saf. 2012, 82, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Usman, A.R.A.; Alkredaa, R.S.; Al-Wabel, M.I. Heavy metal contamination in sediment sand mangroves from the coast of Red Sea: Avicennia marina as potential metal bioaccumulator. Ecotoxicol. Environ. Saf. 2013, 97, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Takarina, N.D.; Tjiong, G.P. Bioconcentration Factor (BCF) and Translocation Factor (TF) of Heavy Metals in Mangrove Trees of Blanakan Fish Farm. Makara J. Sci. 2017, 21, 77–81. [Google Scholar] [CrossRef]
- Shaari, H.; Mohamad Azmi, S.N.H.; Sultan, K.; Bidai, J.; Mohamad, Y. Spatial distribution of selected heavy metals in surface sediments of the EEZ of the east coast of Peninsular Malaysia. Int. J. Oceanogr. 2015, 2015, 618074. [Google Scholar] [CrossRef] [Green Version]
- Looi, L.J.; Aris, A.Z.; Md. Yusoff, F.; Isa, N.M.; Haris, H. Application of enrichment factor, geoaccumulation index, and ecological risk index in assessing the elemental pollution status of surface sediments. Environ. Geochem. Health 2019, 41, 27–42. [Google Scholar] [CrossRef]
- Sutherland, R.A. Bed sediment-associated trace metals in an urban stream, Oahu, Hawaii. Environ. Geol. 2000, 39, 611–627. [Google Scholar] [CrossRef]
- Müller, G. Index of geoaccumulation in sediments of the Rhine River. Geojournal 1969, 2, 108–118. [Google Scholar]
- Haris, H.; Aris, A.Z. The geoaccumulation index and enrichment factor of mercury in mangrove sediment of Port Klang, Selangor, Malaysia. Arab. J. Geosci. 2012, 6, 4119–4128. [Google Scholar] [CrossRef]
- Chen, C.W.; Kao, C.M.; Chen, C.F.; Dong, C.D. Distribution and accumulation of heavy metals in the sediments of Kaohsiung Harbor, Taiwan. Chemosphere 2007, 66, 1431–1440. [Google Scholar] [CrossRef]
- Zahra, A.; Hashmi, M.Z.; Malik, R.N.; Ahmed, Z. Enrichment and geo-accumulation of heavy metals and risk assessment of sediments of the Kurang Nallah--feeding tributary of the Rawal Lake Reservoir, Pakistan. Sci. Total Environ. 2014, 470, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Olubunmi, F.E.; Olorunsola, O.E. Evaluation of the status of heavy metal pollution of sediment of Agbabu bitumen deposit area, Nigeria. Eur. J. Sci. Res. 2010, 3, 373–382. [Google Scholar]
- Rudnick, R.L.; Gao, S. Composition of the continental crust. In The Crust; Rudnick, R.L., Ed.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 3, pp. 1–64. [Google Scholar]
- Ismail, A.; Badri, M.A.; Ramlan, M.N. The background levels of heavy metal concentration in sediments of the west coast of Peninsular Malaysia. Sci. Total Environ. 1993, 134, 315–323. [Google Scholar] [CrossRef]
- Soda, S.; Hamada, T.; Yamaoka, Y.; Ike, M.; Nakazato, H.; Saeki, Y.; Kasamatsu, T.; Sakurai, Y. Constructed wetlands for advanced treatment of wastewater with a complex matrix from a metal-processing plant: Bioconcentration and translocation factors of various metals in Acorus gramineus and Cyperus alternifolius. Ecol. Eng. 2012, 39, 63–70. [Google Scholar] [CrossRef]
- Tu, C.; Ma, L.Q. Effects of arsenic concentrations and forms on arsenic uptake by the hyperaccumulator ladder brake. J. Environ. Qual. 2002, 31, 641–647. [Google Scholar] [CrossRef]
- Mishra, T.; Singh, N.B.; Singh, N. Restoration of red mud deposits by naturally growing vegetation. Int. J. Phytoremediation 2017, 19, 439–445. [Google Scholar] [CrossRef]
- Morrissey, J.; Guerinot, M.L. Iron uptake and transport in plants: The good, the bad, and the ionome. Chem. Rev. 2009, 109, 4553–4567. [Google Scholar] [CrossRef] [Green Version]
- López-Millán, A.; Grusak, M.A.; Abadia, A.; Abadia, J. Iron deficiency in plants: An insight from proteomic approaches. Front. Plant Sci. 2013, 4, 254. [Google Scholar] [CrossRef] [Green Version]
- Whiting, D.; Card, A.; Wilson, C.; Reeder, J. CMG GardenNotes #223: Iron Chlorosis of Woody Plants. Colorado State University Extension. 2015. Available online: https://cmg.extension.colostate.edu/Gardennotes/223.pdf (accessed on 18 April 2022).
- Shanker, A.K.; Cervantes, C.; Loza-Tavera, H.; Avudainayagam, S. Chromium toxicity in plants. Environ. Int. 2005, 31, 739–753. [Google Scholar] [CrossRef]
- Baker, A.J.M. Accumulators and excluders—Strategies in the response of plants to heavy metals. J. Plant Nutr. 1981, 3, 643–654. [Google Scholar] [CrossRef]


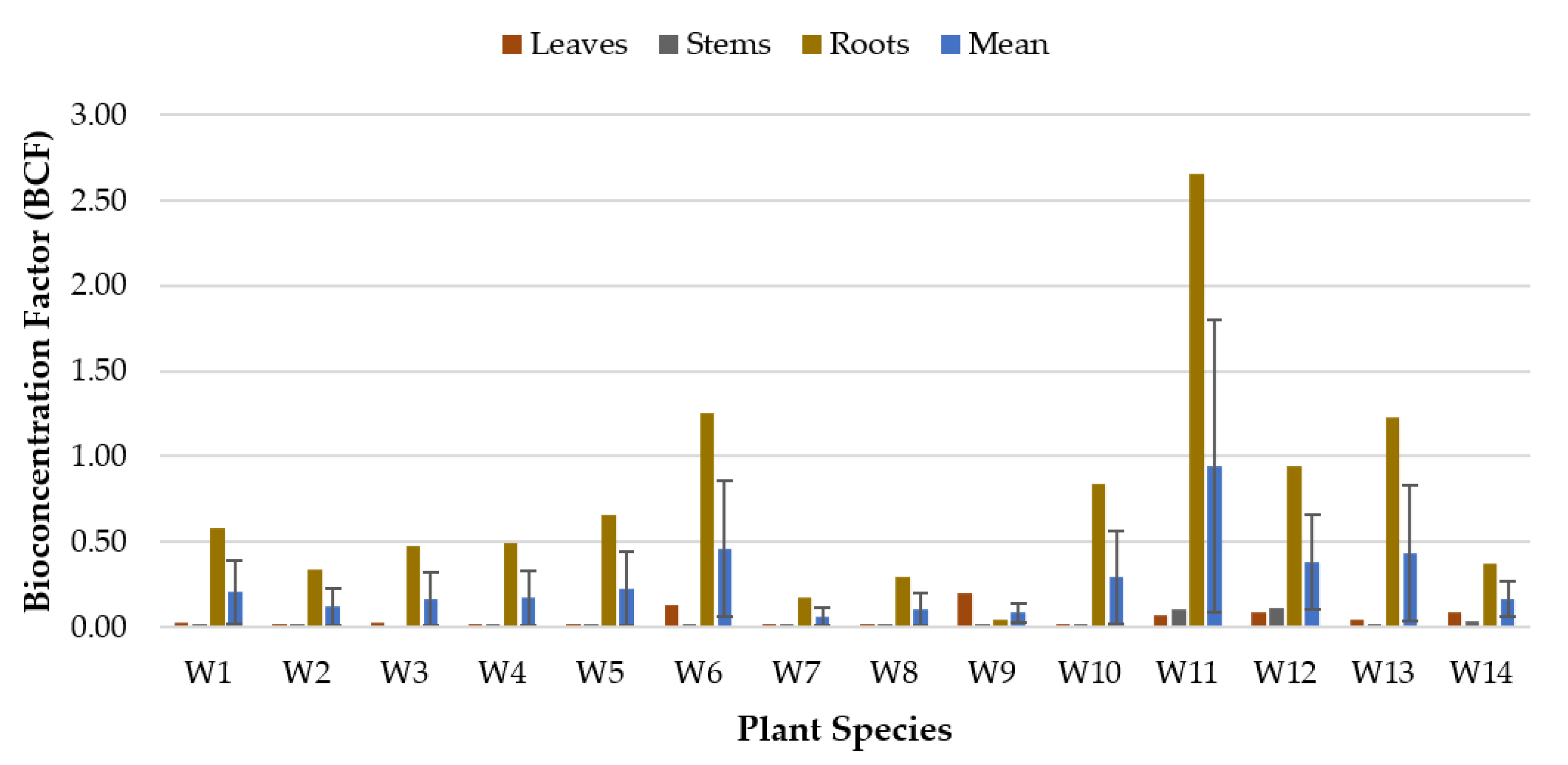






| Code No. | Common Name | Scientific Name | Plant Type |
|---|---|---|---|
| W1 | Tall Sedge | Carex appressa | Sedge |
| W2 | Vetiver Grass | Chrysopogon zizanioides | Perennial Grass |
| W3 | Common Spikerush | Eleocharis dulcis | Perennial Herb |
| W4 | Cattail | Typha angustifolia | Perennial |
| W5 | False Bird of Paradise | Heliconia psittacorum | Perennial |
| W6 | Blue Water Hyssop | Bacopa caroliniana | Perennial (Emergent/Submerged) |
| W7 | Alligator-flag | Thalia geniculata | Perennial |
| W8 | Canna Lily | Canna x generalis | Perennial |
| W9 | Water Mimosa * | Neptunia oleracea | Floating Leaves |
| W10 | Yam | Colocasia esculenta | Perennial |
| W11 | Mexican Sword | Echinodorus palifolius | Perennial |
| W12 | Water Hyacinth * | Eichhornia crassipes | Free-floating |
| W13 | Water Spinach * | Ipomoea aquatica | Floating Leaves |
| W14 | Giant Salvinia * | Salvinia molesta | Free-floating |
| Enrichment Factor (EF) | Degree of Enrichment |
|---|---|
| EF < 2 | Depletion to minimal enrichment (no or minimal pollution) |
| 2 ≤ EF < 5 | Moderate enrichment (moderate pollution) |
| 5 ≤ EF < 20 | Significant enrichment (significant pollution) |
| 20 ≤ EF < 40 | Very high enrichment (very strong pollution) |
| EF > 40 | Extreme enrichment (extreme pollution) |
| Igeo | Igeo Class | Description of Soil Quality |
|---|---|---|
| <0 | 0 | Uncontaminated |
| 0–1 | 1 | Uncontaminated to moderately contaminated |
| 1–2 | 2 | Moderately contaminated |
| 2–3 | 3 | Moderately to strongly contaminated |
| 3–4 | 4 | Strongly contaminated |
| 4–5 | 5 | Strongly to extremely strongly contaminated |
| >5 | 6 | Extremely contaminated |
| Metals | Phytoextraction | Phytostabilisation | ||||
|---|---|---|---|---|---|---|
| BCFmean | TFmean | Plant Species | BCFmean | TFmean | Plant Species | |
| Calcium (Ca) | 21.10 ± 5.04 | 1.42 ± 0.18 | Water Hyacinth (Eichhornia crassipes) | 9.97 ± 5.72 | 0.40 ± 0.02 | Water Mimosa (Neptunia oleracea) |
| 17.14 ± 3.33 | 1.36 ± 0.12 | Blue Water Hyssop (Bacopa caroliniana) | 6.54 ± 1.61 | 0.86 ± 0.21 | Alligator-flag (Thalia geniculata) | |
| 12.40 ± 4.65 | 1.87 ± 0.32 | Cattail (Typha angustifolia) | 6.26 ± 3.52 | 0.49 ± 0.22 | Vetiver Grass (Chrysopogon zizanioides) | |
| 11.69 ± 2.12 | 1.32 ± 0.12 | False Bird of Paradise (Heliconia psittacorum) | 1.99 ± 1.81 | 0.34 ± 0.34 | Common Spikerush (Eleocharis dulcis) | |
| 10.47 ± 6.63 | 2.16 ± 0.90 | Mexican Sword (Echinodorus palifolius) | - | - | - | |
| 10.32 ± 2.05 | 1.26 ± 0.18 | Canna Lily (Canna x generalis) | - | - | - | |
| 9.18 ± 1.29 | 1.11 ± 0.14 | Water Spinach (Ipomoea aquatica) | - | - | - | |
| 8.75 ± 4.03 | 2.25 ± 0.44 | Yam (Colocasia esculenta) | - | - | - | |
| 7.42 ± 3.08 | 1.62 ± 0.47 | Giant Salvinia (Salvinia molesta) | - | - | -- | |
| 1.41 ± 0.20 | 1.17 ± 0.12 | Tall Sedge (Carex appressa) | - | - | ||
| Iron (Fe) | - | - | - | 2.652 * | 0.03 ± 0.01 | Mexican Sword (Echinodorus palifolius) |
| - | - | - | 1.252 * | 0.05 ± 0.05 | Blue Water Hyssop (Bacopa caroliniana) | |
| - | - | - | 1.231 * | 0.03 ± 0.01 | Water Spinach (Ipomoea aquatica) | |
| Magnesium (Mg) | 13.73 ± 15.63 | 1.47 ± 1.47 | Alligator-flag (Thalia geniculata) | 80.80 ± 127.01 | 0.03 ± 0.01 | Mexican Sword (Echinodorus palifolius) |
| 6.07 ± 2.83 | 1.29 ± 0.53 | Vetiver Grass (Chrysopogon zizanioides) | 37.04 ± 59.35 | 0.03 ± 0.01 | Water Spinach (Ipomoea aquatica) | |
| 3.06 ± 0.65 | 1.12 ± 0.22 | Tall Sedge (Carex appressa) | 33.68 ± 54.06 | 0.03 ± 0.00 | Yam (Colocasia esculenta) | |
| - | - | - | 32.81 ± 41.66 | 0.11 ± 0.22 | Water Hyacinth (Eichhornia crassipes) | |
| - | - | - | 31.96 ± 1.34 | 0.97 ± 0.04 | False Bird of Paradise (Heliconia psittacorum) | |
| - | - | - | 27.13 ± 7.16 | 0.65 ± 0.01 | Canna Lily (Canna x generalis) | |
| - | - | 14.08 ± 15.36 | 0.17 ± 0.07 | Giant Salvinia (Salvinia molesta) | ||
| - | - | - | 11.53 ± 5.83 | 0.54 ± 0.22 | Cattail (Typha angustifolia) | |
| - | - | - | 7.40 ± 7.89 | 0.00 ± 0.00 | Blue Water Hyssop (Bacopa caroliniana) | |
| - | - | - | 5.23 ± 4.66 | 0.67 ± 0.67 | Common Spikerush (Eleocharis dulcis) | |
| - | - | - | 3.67 ± 1.14 | 0.67 ± 0.14 | Water Mimosa (Neptunia oleracea) | |
| Manganese (Mn) | 21.70 ± 13.38 | 1.81 ± 0.83 | False Bird of Paradise (Heliconia psittacorum) | 3.00 ± 2.81 | 0.81 ± 0.81 | Common Spikerush (Eleocharis dulcis) |
| 6.04 ± 4.94 | 4.26 ± 1.79 | Cattail (Typha angustifolia) | 2.59 ± 1.53 | 0.44 ± 0.18 | Vetiver grass (Chrysopogon zizanioides) | |
| 3.08 ± 2.02 | 1.36 ± 0.79 | Tall Sedge (Carex appressa) | 2.44 ± 2.38 | 0.27 ± 0.27 | Mexican Sword (Echinodorus palifolius) | |
| Plant Species | Enrichment Factor (EF) | |||||||
|---|---|---|---|---|---|---|---|---|
| Calcium (Ca) | Iron (Fe) | Magnesium (Mg) | Manganese (Mn) | |||||
| Before | After | Before | After | Before | After | Before | After | |
| W1 | 0.086 | 0.026 | 1.000 | 1.000 | 0.070 | 0.006 | 0.354 | 0.038 |
| W2 | 0.086 | 0.008 | 1.000 | 1.000 | 0.070 | 0.004 | 0.354 | 0.023 |
| W3 | 0.086 | 0.026 | 1.000 | 1.000 | 0.070 | 0.006 | 0.354 | 0.038 |
| W4 | 0.086 | 0.026 | 1.000 | 1.000 | 0.070 | 0.006 | 0.354 | 0.038 |
| W5 | 0.086 | 0.026 | 1.000 | 1.000 | 0.070 | 0.006 | 0.354 | 0.038 |
| W6 | 0.086 | 0.026 | 1.000 | 1.000 | 0.070 | 0.006 | 0.354 | 0.038 |
| W7 | 0.086 | 0.026 | 1.000 | 1.000 | 0.070 | 0.006 | 0.354 | 0.038 |
| W8 | 0.086 | 0.026 | 1.000 | 1.000 | 0.070 | 0.006 | 0.354 | 0.038 |
| W9 | 0.086 | 0.074 | 1.000 | 1.000 | 0.070 | 0.018 | 0.354 | 0.053 |
| W10 | 0.086 | 0.074 | 1.000 | 1.000 | 0.070 | 0.018 | 0.354 | 0.053 |
| W11 | 0.086 | 0.063 | 1.000 | 1.000 | 0.070 | 0.024 | 0.354 | 0.095 |
| W12 | 0.086 | 0.063 | 1.000 | 1.000 | 0.070 | 0.024 | 0.354 | 0.095 |
| W13 | 0.086 | 0.063 | 1.000 | 1.000 | 0.070 | 0.024 | 0.354 | 0.095 |
| W14 | 0.086 | 0.063 | 1.000 | 1.000 | 0.070 | 0.024 | 0.354 | 0.095 |
| Plant Species | Geoaccumulation Index (Igeo) | |||||||
|---|---|---|---|---|---|---|---|---|
| Calcium (Ca) | Iron (Fe) | Magnesium (Mg) | Manganese (Mn) | |||||
| Before | After | Before | After | Before | After | Before | After | |
| W1 | −11.25 | −13.35 | −7.72 | −8.08 | −11.56 | −15.54 | −9.22 | −12.80 |
| W2 | −11.25 | −14.41 | −7.72 | −7.49 | −11.56 | −15.64 | −9.22 | −12.95 |
| W3 | −11.25 | −13.35 | −7.72 | −8.08 | −11.56 | −15.54 | −9.22 | −12.80 |
| W4 | −11.25 | −13.35 | −7.72 | −8.08 | −11.56 | −15.54 | −9.22 | −12.80 |
| W5 | −11.25 | −13.35 | −7.72 | −8.08 | −11.56 | −15.54 | −9.22 | −12.80 |
| W6 | −11.25 | −13.35 | −7.72 | −8.08 | −11.56 | −15.54 | −9.22 | −12.80 |
| W7 | −11.25 | −13.35 | −7.72 | −8.08 | −11.56 | −15.54 | −9.22 | −12.80 |
| W8 | −11.25 | −13.35 | −7.72 | −8.08 | −11.56 | −15.54 | −9.22 | −12.80 |
| W9 | −11.25 | −12.34 | −7.72 | −8.57 | −11.56 | −14.39 | −9.22 | −12.80 |
| W10 | −11.25 | −12.34 | −7.72 | −8.57 | −11.56 | −14.39 | −9.22 | −12.80 |
| W11 | −11.25 | −13.10 | −7.72 | −9.10 | −11.56 | −14.50 | −9.22 | −12.49 |
| W12 | −11.25 | −13.10 | −7.72 | −9.10 | −11.56 | −14.50 | −9.22 | −12.49 |
| W13 | −11.25 | −13.10 | −7.72 | −9.10 | −11.56 | −14.50 | −9.22 | −12.49 |
| W14 | −11.25 | −13.10 | −7.72 | −9.10 | −11.56 | −14.50 | −9.22 | −12.49 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ang, S.Y.; Goh, H.W.; Mohd Fazli, B.; Haris, H.; Azizan, N.A.; Zakaria, N.A.; Johar, Z. Heavy Metals Removal from Domestic Sewage in Batch Mesocosm Constructed Wetlands using Tropical Wetland Plants. Water 2023, 15, 797. https://doi.org/10.3390/w15040797
Ang SY, Goh HW, Mohd Fazli B, Haris H, Azizan NA, Zakaria NA, Johar Z. Heavy Metals Removal from Domestic Sewage in Batch Mesocosm Constructed Wetlands using Tropical Wetland Plants. Water. 2023; 15(4):797. https://doi.org/10.3390/w15040797
Chicago/Turabian StyleAng, Shin Ying, Hui Weng Goh, Bashirah Mohd Fazli, Hazzeman Haris, Nor Ariza Azizan, Nor Azazi Zakaria, and Zubaidi Johar. 2023. "Heavy Metals Removal from Domestic Sewage in Batch Mesocosm Constructed Wetlands using Tropical Wetland Plants" Water 15, no. 4: 797. https://doi.org/10.3390/w15040797






