Plant Growth-Promoting Rhizobacteria (PGPR): A Rampart against the Adverse Effects of Drought Stress
Abstract
:1. Introduction
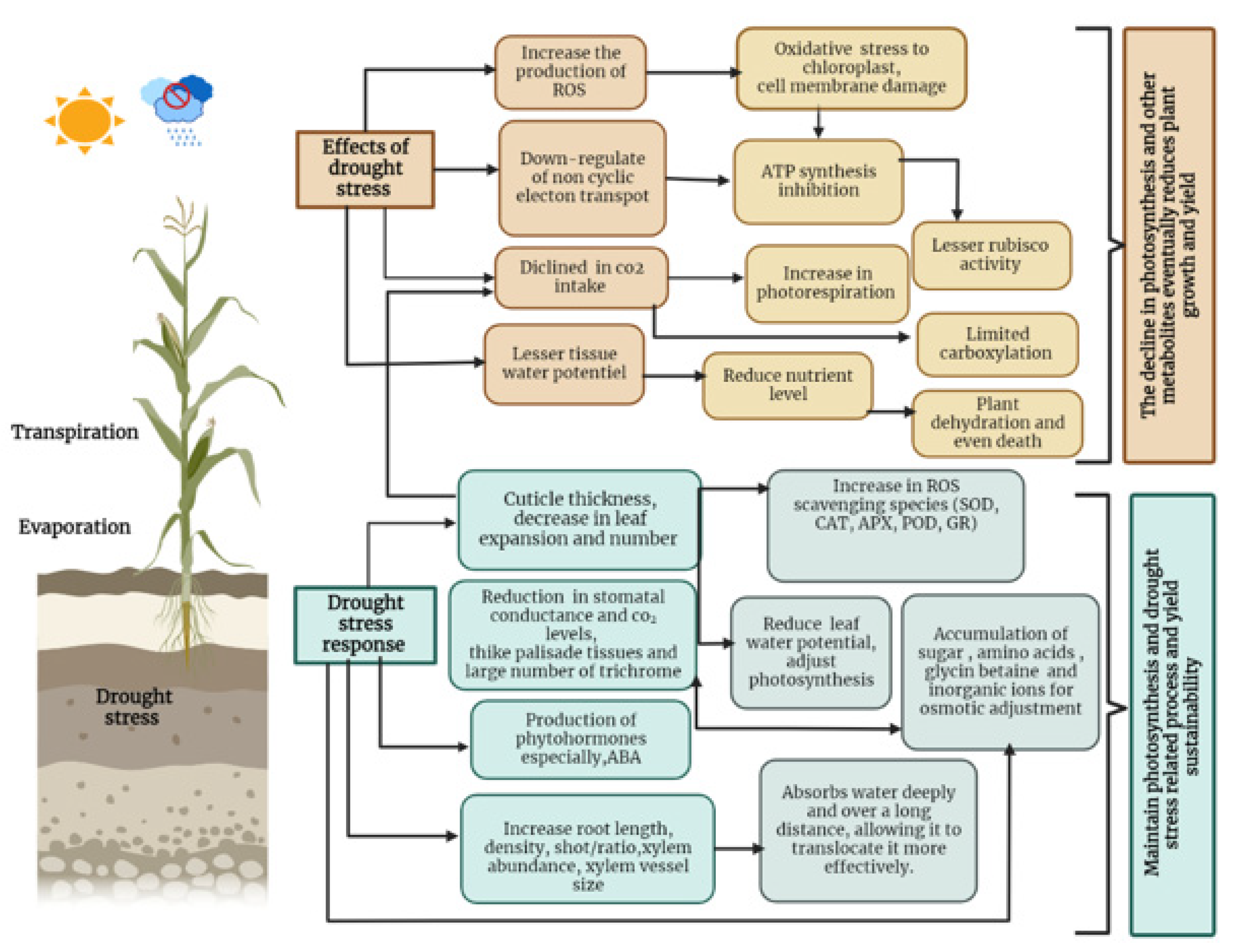
2. Effects of Water Stress on Plant Growth
2.1. Effects of Water Stress on Germination
2.2. Mineral Nutrition
2.3. Plant Morphology and Anatomy
2.4. Photosynthesis
2.5. Hormonal Balance
2.6. Relative Water Content (RWC) and Membrane Stability
2.7. Oxidative Stress: Production of ROS
3. Water Stress Tolerance Mechanisms Involved by Plants
3.1. Morphological Adaptation
3.2. Osmolyte Accumulation: Osmotic Adjustment
3.3. Phytohormones
3.4. Antioxidant Mechanisms: Reduction in ROS Generation
3.5. Molecular Responses: Stress Proteins, Transcription Factors, and Resistance Genes
4. Osmotolerance through Plant Growth-Promoting Rhizobacteria
4.1. Improvements in Physiological and Morphological Processes
4.1.1. Germination Enhancement
4.1.2. PGPR Improve Root Architecture
4.1.3. PGPR Improve Shoot Growth
4.2. PGPR Improve Relative Water Content RWC
4.3. Osmotic Adjustment through the Synthesis and Accumulation of Solute Compatible
4.4. Exopolysaccharide Production
4.5. Modification of Phytohormone Activity by PGPR under Drought
4.6. 1-aminocyclopropane-1-carboxylic Acid (ACC) Deaminase
4.7. Volatile Organic Compounds (VOCs)
4.8. Modification of the Plant Antioxidant Defense System by PGPR
4.9. Molecular Studies in Drought Stress Mitigation by PGPR/Alteration of Stress Responsive Gene Expression
5. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Goswami, M.; Suresh, D.E.K.A. Plant growth-promoting rhizobacteria—Alleviators of abiotic stresses in soil: A review. Pedosphere 2020, 30, 40–61. [Google Scholar] [CrossRef]
- Koza, N.A.; Adedayo, A.A.; Babalola, O.O.; Kappo, A.P. Microorganisms in Plant Growth and Development: Roles in Abiotic Stress Tolerance and Secondary Metabolites Secretion. Microorganisms 2022, 10, 1528. [Google Scholar] [CrossRef] [PubMed]
- Chandra, P.; Wunnava, A.; Verma, P.; Chandra, A.; Sharma, R.K. Strategies to mitigate the adverse effect of drought stress on crop plants—Influences of soil bacteria: A review. Pedosphere 2021, 31, 496–509. [Google Scholar] [CrossRef]
- Kapoor, D.; Bhardwaj, S.; Landi, M.; Sharma, A.; Ramakrishnan, M.; Sharma, A. The impact of drought in plant metabolism: How to exploit tolerance mechanisms to increase crop production. Appl. Sci. 2020, 10, 5692. [Google Scholar] [CrossRef]
- Akhtar, N.; Ilyas, N.; Hayat, R.; Yasmin, H.; Noureldeen, A.; Ahmad, P. Synergistic effects of plant growth promoting rhizobacteria and silicon dioxide nano-particles for amelioration of drought stress in wheat. Plant Physiol. Biochem. 2021, 166, 160–176. [Google Scholar] [CrossRef]
- Siddique, S.; Naveed, M.; Yaseen, M.; Shahbaz, M. Exploring Potential of Seed Endophytic Bacteria for Enhancing Drought Stress Resilience in Maize (Zea mays L.). Sustainability 2022, 14, 673. [Google Scholar] [CrossRef]
- Pequeno, D.N.; Hernandez-Ochoa, I.M.; Reynolds, M.; Sonder, K.; MoleroMilan, A.; Robertson, R.D.; Lopes, M.S.; Xiong, W.; Kropff, M.; Asseng, S. Climate impact and adaptation to heat and drought stress of regional and global wheat production. Environ. Res. Lett. 2021, 16, 054070. [Google Scholar] [CrossRef]
- Bhandari, P.; Garg, N. Plant-Microbe Communication: New Facets for Sustainable Agriculture. In Microbial Interventions in Agriculture and Environment; Singh, D., Gupta, V., Prabha, R., Eds.; Springer: Singapore, 2019. [Google Scholar] [CrossRef]
- Lalarukh, I.; Al-Dhumri, S.A.; Al-Ani, L.K.T.; Hussain, R.; Al Mutairi, K.A.; Mansoora, N.; Galal, T.M. A combined use of rhizobacteria and moringa leaf extract mitigates the adverse effects of drought stress in wheat (Triticum aestivum L.). Front. Microbiol. 2022, 1087, 813415. [Google Scholar] [CrossRef]
- Ojuederie, O.B.; Olanrewaju, O.S.; Babalola, O.O. Plant growth promoting rhizobacterial mitigation of drought stress in crop plants: Implications for sustainable agriculture. Agronomy 2019, 9, 712. [Google Scholar] [CrossRef]
- Azeem, M.; Haider, M.Z.; Javed, S.; Saleem, M.H.; Alatawi, A. Drought Stress Amelioration in Maize (Zea mays L.) by Inoculation of Bacillus spp. Strains under Sterile Soil Conditions. Agriculture 2022, 12, 50. [Google Scholar] [CrossRef]
- Ahluwalia, O.; Singh, P.C.; Bhatia, R. A review on drought stress in plants: Implications, mitigation and the role of plant growth promoting rhizobacteria. Resour. Environ. Sustain. 2021, 5, 100032. [Google Scholar] [CrossRef]
- Barnawal, D.; Singh, R.; Singh, R.P. Role of plant growth promoting rhizobacteria in drought tolerance: Regulating growth hormones and osmolytes. In PGPR Amelioration in Sustainable Agriculture; Woodhead Publishing: Sawston, UK, 2019; pp. 107–128. [Google Scholar] [CrossRef]
- Kour, D.; Yadav, A.N. Bacterial Mitigation of Drought Stress in Plants: Current Perspectives and Future Challenges. Curr. Microbiol. 2022, 79, 248. [Google Scholar] [CrossRef] [PubMed]
- Vurukonda, S.S.K.P.; Vardharajula, S.; Shrivastava, M.; SkZ, A. Enhancement of drought stress tolerance in crops by plant growth promoting rhizobacteria. Microbiol. Res. 2016, 184, 13–24. [Google Scholar] [CrossRef]
- Yang, L.I.U.; Hong, X.U.; Wen, X.X.; Liao, Y.C. Effect of polyamine on seed germination of wheat under drought stress is related to changes in hormones and carbohydrates. J. Integr. Agric. 2016, 15, 2759–2774. [Google Scholar] [CrossRef] [Green Version]
- Zhu, G.; Gu, L.; Shi, Y.; Wang, Y. Plant hydraulic conductivity determines photosynthesis in rice under PEG-induced drought stress. Pakistan J. Bot. 2021, 53, 409–417. [Google Scholar] [CrossRef]
- Zhang, S.H.; Xu, X.F.; Sun, Y.M.; Zhang, J.L.; Li, C.Z. Influence of drought hardening on the resistance physiology of potato seedlings under drought stress. J. Integr. Agr. 2018, 17, 336–347. [Google Scholar] [CrossRef] [Green Version]
- Xu, C.; He, C.G.; Wang, Y.J.; Bi, Y.F.; Jiang, H. Effect of drought and heat stresses on photosynthesis, pigments, and xanthophyll cycle in alfalfa (Medicago sativa L.). Photosynthetica 2020, 58, 1226–1236. [Google Scholar] [CrossRef]
- Niu, J.; Zhang, S.; Liu, S.; Ma, H.; Chen, J.; Shen, Q.; Ge, C.; Zhang, X.; Pang, C.; Zhao, X. The compensation effects of physiology and yield in cotton after drought stress. J. Plant Physiol. 2018, 224, 30–48. [Google Scholar] [CrossRef] [PubMed]
- Naderi, S.; Fakheri, B.A.; Maali-Amiri, R.; Mahdinezhad, N. Tolerance responses in wheat landrace Bolani are related to enhanced metabolic adjustments under drought stress. Plant Physiol. Biochem. 2020, 150, 244–253. [Google Scholar] [CrossRef]
- Wasaya, A.; Manzoor, S.; Yasir, T.A.; Sarwar, N.; Mubeen, K.; Ismail, I.A.; Raza, A.; Rehman, A.; Hossain, A.; EL Sabagh, A. Evaluation of Fourteen Bread Wheat (Triticum aestivum L.) Genotypes by Observing Gas Exchange Parameters, Relative Water and Chlorophyll Content, and Yield Attributes under Drought Stress. Sustainability 2021, 13, 4799. [Google Scholar] [CrossRef]
- Mustafa, H.; Ilyas, N.; Akhtar, N.; Raja, N.I.; Zainab, T.; Shah, T.; Ahmad, A.; Ahmad, P. Biosynthesis and characterization of titanium dioxide nanoparticles and its effects along with calcium phosphate on physicochemical attributes of wheat under drought stress. Ecotoxicol. Environ. Safety 2021, 223, 112519. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.N.; Zhang, J.; Luo, T.; Liu, J.; Ni, F.; Rizwan, M.; Fahad, S.; Hu, L. Morpho-physiological and biochemical responses of tolerant and sensitive rapeseed cultivars to drought stress during early seedling growth stage. Acta Physiol. Plant 2019, 41, 25. [Google Scholar] [CrossRef]
- Ghaffari, H.; Tadayon, M.R.; Nadeem, M.; Cheema, M.; Razmjoo, J. Proline-mediated changes in antioxidant enzymatic activities and the physiology of sugar beet under drought stress. Acta Physiol. Plant. 2019, 41, 23. [Google Scholar] [CrossRef]
- Gowtham, H.G.; Singh, B.; Murali, M.; Shilpa, N.; Prasad, M.; Aiyaz, M.; Amruthesh, K.N.; Niranjana, S.R. Induction of drought tolerance in tomato upon the application of ACC deaminase producing plant growth promoting rhizobacterium Bacillus subtilis Rhizo SF 48. Microbiol. Res. 2020, 234, 126422. [Google Scholar] [CrossRef]
- Li, L.; Gu, W.; Li, J.; Li, C.; Xie, T.; Qu, D.; Meng, Y.; Li, C.; Wei, S. Exogenously applied spermidine alleviates photosynthetic inhibition under drought stress in maize (Zea mays L.) seedlings associated with changes in endogenous polyamines and phytohormones. Plant Physiol. Biochem. 2018, 129, 35–55. [Google Scholar] [CrossRef]
- Mukami, A.; Ngetich, A.; Mweu, C.; Oduor, R.O.; Muthangya, M.; Mbinda, W.M. Differential characterization of physiological and biochemical responses during drought stress in finger millet varieties. Physiol. Mol. Biol. Plants. 2019, 25, 837–846. [Google Scholar] [CrossRef]
- Zia, R.; Nawaz, M.S.; Siddique, M.J.; Hakim, S.; Imran, A. Plant survival under drought stress: Implications, adaptive responses, and integrated rhizosphere management strategy for stress mitigation. Microbiol. Res. 2021, 242, 126626. [Google Scholar] [CrossRef]
- Islam, M.M.; Kayesh, E.; Zaman, E.; Urmi, T.A.; Haque, M.M. Evaluation of rice (Oryza sativa L.) genotypes for drought tolerance at germination and early seedling stage. Agriculturists 2018, 16, 44–54. [Google Scholar] [CrossRef]
- Toscano, M.; Grandi, R.D.; Giampietro, D.; Grossi, E.; Facchin, V.; Comberiati, P.; Drago, L. Impact of delivery mode on the colostrum microbiota composition. BMC Microbiol. 2017, 17, 205. [Google Scholar] [CrossRef]
- Queiroz, M.S.; Oliveira, C.E.; Steiner, F.; Zuffo, A.M.; Zoz, T.; Vendruscolo, E.P.; Silva, M.V.; Mello, B.F.F.R.; Cabral, R.C.; Menis, F.T. Drought stresses on seed germination and early growth of maize and sorghum. J. Agr. Sci. 2019, 11, 310–318. [Google Scholar] [CrossRef]
- Vandana, U.K.; Singha, B.; Gulzar, A.B.M.; Mazumder, P.B. Molecular mechanisms in plant growth promoting bacteria (PGPR) to resist environmental stress in plants. In Molecular Aspects of Plant Beneficial Microbes in Agriculture; Academic Press: Cambridge, MA, USA, 2020; pp. 221–233. [Google Scholar] [CrossRef]
- García-Caparrós, P.; Romero, M.J.; Llanderal, A.; Cermeño, P.; Lao, M.T.; Segura, M.L. Effects of Drought Stress on Biomass, Essential Oil Content, Nutritional Parameters, and Costs of Production in Six Lamiaceae Species. Water 2019, 11, 573. [Google Scholar] [CrossRef] [Green Version]
- Etesami, H.; Adl, S.M. Plant Growth-Promoting Rhizobacteria (PGPR) and Their Action Mechanisms in Availability of Nutrients to Plants. In Phyto-Microbiome in Stress Regulation; Environmental and Microbial Biotechnology, Kumar, M., Kumar, V., Prasad, R., Eds.; Springer: Singapore, 2020. [Google Scholar] [CrossRef]
- Fattahi, M.; Mohammadkhani, A.; Shiran, B.; Baninasab, B.; Ravash, R.; Gogorcena, Y. Beneficial effect of mycorrhiza on nutritional uptake and oxidative balance in pistachio (Pistacia spp.) rootstocks submitted to drought and salinity stress. Sci. Hortic. 2021, 281, 109937. [Google Scholar] [CrossRef]
- Salehi-Lisar, S.Y.; Bakhshayeshan-Agdam, H. Drought Stress in Plants: Causes, Consequences, and Tolerance. In Drought Stress Tolerance in Plants; Hossain, M., Wani, S., Bhattacharjee, S., Burritt, D., Tran, L.S., Eds.; Springer: Cham, Switzerland, 2016; Volume 1. [Google Scholar] [CrossRef]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant Drought Stress: Effects, Mechanisms and Management. In Sustainable Agriculture; Lichtfouse, E., Navarrete, M., Debaeke, P., Véronique, S., Alberola, C., Eds.; Springer: Dordrecht, The Netherlands, 2009. [Google Scholar] [CrossRef] [Green Version]
- Ahanger, M.A.; Morad-Talab, N.; Abd-Allah, E.F.; Ahmad, P.; Hajiboland, R. Plant growth under drought stress: Significance of mineral nutrients. In Water Stress and Crop Plants: A Sustainable Approach; Ahmad, P., Ed.; Wiley Blackwell: Weinheim, Germany, 2016; pp. 649–668. [Google Scholar] [CrossRef]
- Bashir, S.S.; Hussain, A.; Hussain, S.J.; Wani, O.A.; Zahid Nabi, S.; Dar, N.A.; Baloch, F.S.; Mansoor, S. Plant drought stress tolerance: Understanding its physiological, biochemical and molecular mechanisms. Biotechnol. Biotechnol. Equip. 2021, 35, 1912–1925. [Google Scholar] [CrossRef]
- Tadayyon, A.; Nikneshan, P.; Pessarakli, M. Effects of drought stress on concentration of macro-and micro-nutrients in Castor (Ricinus communis L.) plant. J. Plant Nutr. 2018, 41, 304–310. [Google Scholar] [CrossRef]
- Liu, C.; Duan, N.; Chen, X.; Li, H.; Zhao, X.; Duo, P.; Wang, J.; Li, Q. Metabolic Pathways Involved in the Drought Stress Response of Nitraria tangutorum as Revealed by Transcriptome Analysis. Forests 2022, 13, 509. [Google Scholar] [CrossRef]
- Hussain, H.A.; Hussain, S.; Khaliq, A.; Ashraf, U.; Anjum, S.A.; Men, S.; Wang, L. Chilling and drought stresses in crop plants: Implications, cross talk, and potential management opportunities. Front. Plant Sci. 2018, 9, 393. [Google Scholar] [CrossRef]
- Singh, A.D.; Kour, J.; Dhiman, S.; Khanna, K.; Kumar, P.; Kaur, R.; Kapoor, N.; Bhardwaj, R. Prospects of PGPR-Mediated Antioxidants and S and P Metabolism in Plants Under Drought Stress. In Antioxidants in Plant-Microbe Interaction; Singh, H.B., Vaishnav, A., Sayyed, R., Eds.; Springer: Singapore, 2021. [Google Scholar] [CrossRef]
- Chiappero, J.; del Rosario Cappellari, L.; Alderete, L.G.S.; Palermo, T.B.; Banchio, E. Plant growth promoting rhizobacteria improve the antioxidant status in Mentha piperita grown under drought stress leading to an enhancement of plant growth and total phenolic content. Ind. Crops Prod. 2019, 139, 111553. [Google Scholar] [CrossRef]
- Yasmin, H.; Nosheen, A.; Naz, R.; Keyani, R.; Anjum, S. Regulatory Role of Rhizobacteria to Induce Drought and Salt Stress Tolerance in Plants. In Field Crops: Sustainable Management by PGPR; Sustainable Development and Biodiversity, Maheshwari, D., Dheeman, S., Eds.; Springer: Cham, Switzerland, 2019; Volume 23. [Google Scholar] [CrossRef]
- Bhattacharya, A. Effect of Soil Water Deficit on Growth and Development of Plants: A Review. In Soil Water Deficit and Physiological Issues in Plants; Springer: Singapore, 2021. [Google Scholar] [CrossRef]
- Farooqi, Z.U.R.; Ayub, M.A.; Rehman, M.Z.; Sohail, M.I.; Usman, M.; Khalid, H.; Naz, K. Regulation of drought stress in plants. In Plant Life under Changing Environment; Academic Press: Cambridge, MA, USA, 2020; pp. 77–104. [Google Scholar] [CrossRef]
- Jain, M.; Kataria, S.; Hirve, M.; Prajapati, R. Water Deficit Stress Effects and Responses in Maize. In Plant Abiotic Stress Tolerance; Hasanuzzaman, M., Hakeem, K., Nahar, K., Alharby, H., Eds.; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Khatun, M.; Sarkar, S.; Era, F.M.; Islam, A.K.M.M.; Anwar, M.P.; Fahad, S.; Datta, R.; Islam, A.K.M.A. Drought Stress in Grain Legumes: Effects, Tolerance Mechanisms and Management. Agronomy 2021, 11, 2374. [Google Scholar] [CrossRef]
- Raja, V.; Qadir, S.U.; Alyemeni, M.N.; Ahmad, P. Impact of drought and heat stress individually and in combination on physio-biochemical parameters, antioxidant responses, and gene expression in Solanum lycopersicum. 3 Biotech 2020, 10, 208. [Google Scholar] [CrossRef]
- Iqbal, M.S.; Singh, A.K.; Ansari, M.I. Effect of Drought Stress on Crop Production. In New Frontiers in Stress Management for Durable Agriculture; Rakshit, A., Singh, H., Singh, A., Singh, U., Fraceto, L., Eds.; Springer: Singapore, 2020. [Google Scholar] [CrossRef]
- El-Sabagh, A.E.; Islam, M.S.; Hossain, A.; Iqbal, M.A.; Mubeen, M.; Waleed, M.; Reginato, M.; Battaglia, M.; Ahmed, S.; Rehman, A.; et al. Phytohormones as growth regulators during abiotic stress tolerance in plants. Front. Agron. 2022. [Google Scholar] [CrossRef]
- Zhang, C.; Shi, S.; Liu, Z.; Yang, F.; Yin, G. Drought tolerance in alfalfa (Medicago sativa L.) varieties is associated with enhanced antioxidative protection and declined lipid peroxidation. J. Plant Physiol. 2019, 232, 226–240. [Google Scholar] [CrossRef] [PubMed]
- Chhaya, Y.B.; Jogawat, A.; Gnanasekaran, P.; Kumari, P.; Lakra, N.; Lal, S.K.; Pawar, J.; Narayan, O.P. An overview of recent advancement in phytohormones-mediated stress management and drought tolerance in crop plants. Plant Gene 2021, 25, 100264. [Google Scholar] [CrossRef]
- Ullah, A.; Manghwar, H.; Shaban, M.; Khan, A.H.; Akbar, A.; Ali, U.; Ali, E.; Fahad, S. Phytohormones enhanced drought tolerance in plants: A coping strategy. Environ. Sci. Pollut. Res. 2018, 25, 33103–33118. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A.; Nisar, M.; Ali, H.; Hazrat, A.; Hayat, K.; Keerio, A.A.; Ihsan, M.; Laiq, M.; Ullah, S.; Fahad, S.; et al. Drought tolerance improvement in plants: An endophytic bacterial approach. Appl. Microbiol. Biotechnol. 2019, 103, 7385–7397. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K.; Asami, T. Chemical regulators of plant hormones and their applications in basic research and agriculture. Biosci. Biotechnol. Biochem. 2018, 82, 1265–1300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nolan, T.M.; Vukašinović, N.; Liu, D.; Russinova, E.; Yin, Y. Brassinosteroids: Multidimensional regulators of plant growth, development, and stress responses. The Plant Cell 2020, 32, 295–318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghafoor, R.; Akram, N.A.; Rashid, M.; Ashraf, M.; Iqbal, M.; Lixin, Z. Exogenously applied proline induced changes in key anatomical features and physio-biochemical attributes in water stressed oat (Avena sativa L.) plants. Physiol. Mol. Biol. Plants 2019, 25, 1121–1135. [Google Scholar] [CrossRef]
- Jamshidi Goharrizi, K.; Moosavi, S.S.; Amirmahani, F.; Salehi, F.; Nazari, M. Assessment of changes in growth traits, oxidative stress parameters, and enzymatic and non-enzymatic antioxidant defense mechanisms in Lepidium draba plant under osmotic stress induced by polyethylene glycol. Protoplasma 2020, 257, 459–473. [Google Scholar] [CrossRef]
- El-Beltagi, H.S.; Shah, S.; Ullah, S.; Sulaiman; Mansour, A.T.; Shalaby, T.A. Impacts of Ascorbic Acid and Alpha-Tocopherol on Chickpea (Cicer arietinum L.) Grown in Water Deficit Regimes for Sustainable Production. Sustainability 2022, 14, 8861. [Google Scholar] [CrossRef]
- Laxa, M.; Liebthal, M.; Telman, W.; Chibani, K.; Dietz, K.-J. The Role of the Plant Antioxidant System in Drought Tolerance. Antioxidants 2019, 8, 94. [Google Scholar] [CrossRef] [Green Version]
- Ansari, F.A.; Jabeen, M.; Ahmad, I. Pseudomonas azotoformans FAP5, a novel biofilm-forming PGPR strain, alleviates drought stress in wheat plant. Int. J. Environ. Sci. Technol. 2021, 18, 3855–3870. [Google Scholar] [CrossRef]
- Kirova, E.; Pecheva, D.; Simova-Stoilova, L. Drought response in winter wheat: Protection from oxidative stress and mutagenesis effect. Acta Physiol. Plant. 2021, 43, 8. [Google Scholar] [CrossRef]
- He, W.; Yan, K.; Zhang, Y.; Bian, L.; Mei, H.; Han, G. Contrasting photosynthesis, photoinhibition and oxidative damage in honeysuckle (Lonicera japonica Thunb.) under iso-osmotic salt and drought stresses. Environ. Exp. Bot. 2021, 182, 104313. [Google Scholar] [CrossRef]
- Zenda, T.; Liu, S.; Duan, H. Adapting cereal grain crops to drought stress: 2020 and beyond. In Abiotic Stress Plants; IntechOpen: London, UK, 2020; pp. 1–27. [Google Scholar]
- Carlson, R.; Tugizimana, F.; Steenkamp, P.A.; Dubery, I.A.; Hassen, A.I.; Labuschagne, N. Rhizobacteria-induced systemic tolerance against drought stress in, Sorghum bicolor (L.) Moench. Microbiol. Res. 2019, 232, 126388. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, N.; Mumtaz, K.; Akhtar, N.; Yasmin, H.; Sayyed, R.Z.; Khan, W.; Enshasy, H.A.E.; Dailin, D.J.; Elsayed, E.A.; Ali, Z. Exopolysaccharides Producing Bacteria for the Amelioration of Drought Stress in Wheat. Sustainability 2020, 12, 8876. [Google Scholar] [CrossRef]
- Fang, Y.; Xiong, L. General mechanisms of drought response and their application in drought resistance improvement in plants. Cell. Mol. Life Sci. 2015, 72, 673–689. [Google Scholar] [CrossRef] [PubMed]
- Hazman, M.; Brown, K.M. Progressive drought alters architectural and anatomical traits of rice roots. Rice 2018, 11, 62. [Google Scholar] [CrossRef]
- Chun, H.C.; Sanghun, L.E.E.; Choi, Y.D.; Gong, D.H.; Jung, K.Y. Effects of drought stress on root morphology and spatial distribution of soybean and adzuki bean. J. Integr. Agr. 2021, 20, 2639–2651. [Google Scholar] [CrossRef]
- Lamaoui, M.; Jemo, M.; Datla, R.; Bekkaoui, F. Heat and drought stresses in crops and approaches for their mitigation. Front. Chem. 2018, 6, 26. [Google Scholar] [CrossRef]
- Dutta, P.; Chakraborti, S.; Chaudhuri, K.M.; Mondal, S. Physiological Responses and Resilience of Plants to Climate Change. In New Frontiers in Stress Management for Durable Agriculture; Rakshit, A., Singh, H., Singh, A., Singh, U., Fraceto, L., Eds.; Springer: Singapore, 2020. [Google Scholar] [CrossRef]
- Men, Y.; Wang, D.; Li, B.; Su, Y.; Chen, G. Effects of drought stress on the antioxidant system, osmolytes and secondary metabolites of Saposhnikovia divaricata seedlings. Acta Physiol. Plant. 2018, 40, 191. [Google Scholar] [CrossRef]
- Farooq, M.; Almamari, S.A.D.; Rehman, A.; Al-Busaidi, W.M.; Wahid, A.; Al-Ghamdi, S.S. Morphological, physiological and biochemical aspects of zinc seed priming-induced drought tolerance in faba bean. Sci. Hortic. 2021, 281, 109894. [Google Scholar] [CrossRef]
- Li, B.; Feng, Y.; Zong, Y.; Zhang, D.; Hao, X.; Li, P. Elevated CO2-induced changes in photosynthesis, antioxidant enzymes and signal transduction enzyme of soybean under drought stress. Plant Physiol. Biochem. 2020, 154, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.M.; Nahar, K.; Hasanuzzaman, M.; Fujita, M. Exogenous jasmonic acid modulates the physiology, antioxidant defense and glyoxalase systems in imparting drought stress tolerance in different Brassica species. Plant Biotechnol. Rep. 2014, 8, 279–293. [Google Scholar] [CrossRef]
- Singh, M.; Rani, S.; Malhotra, N.; Katna, G.; Sarker, A. Transgressive segregations for agronomic improvement using interspecific crosses between C. arietinum L. x C. reticulatum Ladiz. and C. arietinum L. x C. echinospermum Davis species. PLoS ONE 2018, 13, e0203082. [Google Scholar] [CrossRef] [Green Version]
- Javadipour, Z.; Balouchi, H.; Movahhedi Dehnavi, M.; Yadavi, A. Physiological Responses of Bread Wheat (Triticum aestivum) Cultivars to Drought Stress and Exogenous Methyl Jasmonate. J. Plant Growth Regul. 2021, 41, 3433–3448. [Google Scholar] [CrossRef]
- Zamani, S.; Naderi, M.R.; Soleymani, A.; Nasiri, B.M. Sunflower (Helianthus annuus L.) biochemical properties and seed components affected by potassium fertilization under drought conditions. Ecotoxicol. Environ. Safety 2020, 190, 110017. [Google Scholar] [CrossRef]
- Anjum, S.A.; Ashraf, U.; Tanveer, M.; Khan, I.; Hussain, S.; Shahzad, B.; Zohaib, A.; Abbas, F.; Saleem, M.F.; Ali, I.; et al. Drought induced changes in growth, osmolyte accumulation and antioxidant metabolism of three maize hybrids. Front Plant Sci. 2017, 8, 69. [Google Scholar] [CrossRef] [Green Version]
- Mäkelä, P.S.A.; Jokinen, K.; Himanen, K. Roles of Endogenous Glycinebetaine in Plant Abiotic Stress Responses. In Osmoprotectant-Mediated Abiotic Stress Tolerance in Plants; Hossain, M., Kumar, V., Burritt, D., Fujita, M., Mäkelä, P., Eds.; Springer: Cham, Switzerland; Geneva, Switzerland, 2019. [Google Scholar] [CrossRef]
- Yang, X.; Lu, M.; Wang, Y.; Wang, Y.; Liu, Z.; Chen, S. Response Mechanism of Plants to Drought Stress. Horticulturae 2021, 7, 50. [Google Scholar] [CrossRef]
- Singh, A.; Kumar, A.; Yadav, S.; Singh, I.K. Reactive oxygen species-mediated signaling during abiotic stress. Plant Gene 2019, 18, 100173. [Google Scholar] [CrossRef]
- Annunziata, M.G.; Ciarmiello, L.F.; Woodrow, P.; Dell’Aversana, E.; Carillo, P. Spatial and temporal profile of glycine betaine accumulation in plants under abiotic stresses. Front. Plant Sci. 2019, 10, 230. [Google Scholar] [CrossRef] [Green Version]
- Bai, X.; Dai, L.; Sun, H.; Chen, M.; Sun, Y. Effects of moderate soil salinity on osmotic adjustment and energy strategy in soybean under drought stress. Plant Physiol. Biochem. 2019, 139, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Barnawal, D.; Bharti, N.; Pandey, S.S.; Pandey, A.; Chanotiya, C.S.; Kalra, A. Plant growth-promoting rhizobacteria enhance wheat salt and drought stress tolerance by altering endogenous phytohormone levels and TaCTR1/TaDREB2 expression. Physiol. Plant. 2017, 161, 502–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdelaal, K.; AlKahtani, M.; Attia, K.; Hafez, Y.; Király, L.; Künstler, A. The Role of Plant Growth-Promoting Bacteria in Alleviating the Adverse Effects of Drought on Plants. Biology 2021, 10, 520. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yuan, W.; Wang, Q.; Cao, Y.; Xu, F.; Dodd, I.C.; Xu, W. ABA regulation of root growth during soil drying and recovery can involve auxin response. Plant Cell Environ. 2022, 45, 871–883. [Google Scholar] [CrossRef]
- Yadav, R.; Juneja, S.; Kumar, S. Cross priming with drought improves heat-tolerance in chickpea (Cicer arietinum L.) by stimulating small heat shock proteins and antioxidative defense. Environ. Sustain. 2021, 4, 171–182. [Google Scholar] [CrossRef]
- Daszkowska-Golec, A. The Role of Abscisic Acid in Drought Stress: How ABA Helps Plants to Cope with Drought Stress. In Drought Stress Tolerance in Plant; Hossain, M., Wani, S., Bhattacharjee, S., Burritt, D., Tran, L.S., Eds.; Springer: Cham, Switzerland; Geneva, Switzerland, 2016; Volume 2. [Google Scholar] [CrossRef]
- Ghosh, D.; Gupta, A.; Mohapatra, S. Dynamics of endogenous hormone regulation in plants by phytohormone secreting rhizobacteria under water-stress. Symbiosis 2019, 77, 265–278. [Google Scholar] [CrossRef]
- Iqbal, S.; Wang, X.; Mubeen, I.; Kamran, M.; Kanwal, I.; Díaz, G.A.; Abbas, A.; Parveen, A.; Atiq, M.N.; Alshaya, H.; et al. Phytohormones trigger drought tolerance in crop plants: Outlook and future perspectives. Front. Plant Sci. 2022, 3378, 799318. [Google Scholar] [CrossRef]
- Hai, N.N.; Chuong, N.N.; Tu, N.H.C.; Kisiala, A.; Hoang, X.L.T.; Thao, N.P. Role and Regulation of Cytokinins in Plant Response to Drought Stress. Plants 2020, 9, 422. [Google Scholar] [CrossRef] [Green Version]
- Pavlović, I.; Petřík, I.; Tarkowská, D.; Lepeduš, H.; Vujčić Bok, V.; Radić Brkanac, S.; Novák, O.; Salopek-Sondi, B. Correlations between Phytohormones and Drought Tolerance in Selected Brassica Crops: Chinese Cabbage, White Cabbage and Kale. Int. J. Mol. Sci. 2018, 19, 2866. [Google Scholar] [CrossRef] [Green Version]
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Zulfiqar, F.; Raza, A.; Mohsin, S.M.; Mahmud, J.A.; Fujita, M.; Fotopoulos, V. Reactive Oxygen Species and Antioxidant Defense in Plants under Abiotic Stress: Revisiting the Crucial Role of a Universal Defense Regulator. Antioxidants 2020, 9, 681. [Google Scholar] [CrossRef]
- Khedia, J.; Agarwal, P.; Agarwal, P.K. Deciphering hydrogen peroxide-induced signaling towards stress tolerance in plants. 3 Biotech 2019, 9, 395. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Sachdeva, S.; Bhat, K.V.; Vats, S. Plant Responses to Drought Stress: Physiological, Biochemical and Molecular Basis. In Biotic and Abiotic Stress Tolerance in Plants; Vats, S., Ed.; Springer: Singapore, 2018. [Google Scholar] [CrossRef]
- Abdelaal, K.A.A.; Attia, K.A.; Alamery, S.F.; El-Afry, M.M.; Ghazy, A.I.; Tantawy, D.S.; Al-Doss, A.A.; El-Shawy, E.-S.E.; Abu-Elsaoud, A.M.; Hafez, Y.M. Exogenous Application of Proline and Salicylic Acid can Mitigate the Injurious Impacts of Drought Stress on Barley Plants Associated with Physiological and Histological Characters. Sustainability 2020, 12, 1736. [Google Scholar] [CrossRef] [Green Version]
- Vuković, R.; Čamagajevac, I.Š.; Vuković, A.; Šunić, K.; Begović, L.; Mlinarić, S.; Sekulić, R.; Sabo, N.; Španić, V. Physiological, Biochemical and Molecular Response of Different Winter Wheat Varieties under Drought Stress at Germination and Seedling Growth Stage. Antioxidants 2022, 11, 693. [Google Scholar] [CrossRef]
- Rajput, V.D.; Harish; Singh, R.K.; Verma, K.K.; Sharma, L.; Quiroz-Figueroa, F.R.; Meena, M.; Gour, V.S.; Minkina, T.; Sushkova, S.; et al. Recent Developments in Enzymatic Antioxidant Defense Mechanism in Plants with Special Reference to Abiotic Stress. Biology 2021, 10, 267. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Mishra, R.; Rai, S.; Bano, A.; Pathak, N.; Fujita, M.; Kumar, M.; Hasanuzzaman, M. Mechanistic Insights of Plant Growth Promoting Bacteria Mediated Drought and Salt Stress Tolerance in Plants for Sustainable Agriculture. Int. J. Mol. Sci. 2022, 23, 3741. [Google Scholar] [CrossRef]
- Batth, R.; Singh, K.; Kumari, S.; Mustafiz, A. Transcript profiling reveals the presence of abiotic stress and developmental stage specific ascorbate oxidase genes in plants. Front. Plant Sci. 2017, 8, 198. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Liu, H.; Yu, F.; Hu, B.; Jia, Y.; Sha, H.; Zhao, H. Differential activity of the antioxidant defense system and alterations in the accumulation of osmolyte and reactive oxygen species under drought stress and recovery in rice (Oryza sativa L.) tillering. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Shafiq, S.; Akram, N.A.; Ashraf, M.; García-Caparrós, P.; Ali, O.M.; Latef, A.A.H.A. Influence of Glycine Betaine (Natural and Synthetic) on Growth, Metabolism and Yield Production of Drought-Stressed Maize (Zea mays L.) Plants. Plants 2021, 10, 2540. [Google Scholar] [CrossRef]
- Nawaz, M.; Wang, X.; Saleem, M.H.; Khan, M.H.U.; Afzal, J.; Fiaz, S.; Ali, S.; Ishaq, H.; Khan, A.H.; Rehman, N.; et al. Deciphering Plantago ovata Forsk Leaf Extract Mediated Distinct Germination, Growth and Physio-Biochemical Improvements under Water Stress in Maize (Zea mays L.) at Early Growth Stage. Agronomy 2021, 11, 1404. [Google Scholar] [CrossRef]
- Shah, T.M.; Imran, M.; Atta, B.M.; Ashraf, M.Y.; Hameed, A.; Waqar, I.; Shafiq, M.; Hussain, K.; Naveed, M.; Aslam, M.; et al. Selection and screening of drought tolerant high yielding chickpea genotypes based on physio-biochemical indices and multi-environmental yield trials. BMC Plant Biol. 2020, 20, 171. [Google Scholar] [CrossRef]
- Marwein, R.; Debbarma, J.; Sarki, Y.N.; Baruah, I.; Saikia, B.; Boruah, H.D.; Velmurgan, N.; Chikkaputtaiah, C. Genetic engineering/Genome editing approaches to modulate signaling processes in abiotic stress tolerance. In Plant signaling molecules; Woodhead Publishing: Sawston, UK, 2019; pp. 63–82. [Google Scholar] [CrossRef]
- Arora, S.; Jha, P.N. Impact of Plant-Associated Microbial Communities on Host Plants Under Abiotic Stresses. In Microbial Interventions in Agriculture and Environment; Singh, D., Gupta, V., Prabha, R., Eds.; Springer: Singapore, 2019. [Google Scholar] [CrossRef]
- Mukarram, M.; Choudhary, S.; Kurjak, D.; Petek, A.; Khan, M.M.A. Drought: Sensing, signaling, effects and tolerance in higher plants. Physiol. Plant. 2021, 172, 1291–1300. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.; Wani, S.H.; Singh, B.; Bohra, A.; Dar, Z.A.; Lone, A.A.; Singla-Pareek, S.L. Transcription factors and plants response to drought stress: Current understanding and future directions. Front. Plant Sci. 2016, 7, 1029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramegowda, V.; Da Costa, M.V.J.; Harihar, S.; Karaba, N.N.; Sreeman, S.M. Abiotic and biotic stress interactions in plants: A cross-tolerance perspective. In Priming-Mediated Stress and Cross-Stress Tolerance in Crop Plants; Academic Press: Cambridge, MA, USA, 2020; pp. 267–302. [Google Scholar] [CrossRef]
- Priya, M.; Dhanker, O.P.; Siddique, K.H.M.; HanumanthaRao, B.; Nair, R.M.; Pandey, S.; Singh, S.; Varshney, R.K.; Prasad, P.V.V.; Nayyar, H. Drought and heat stress-related proteins: An update about their functional relevance in imparting stress tolerance in agricultural crops. Theor. Appl. Genet. 2019, 132, 1607–1638. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Zhang, S.; Liang, Y.; Qi, Y.; Chen, J.; Zhu, W.; Zhang, L. Label-free quantitative proteomic analysis of drought stress-responsive late embryogenesis abundant proteins in the seedling leaves of two wheat (Triticum aestivum L.) genotypes. J. Proteom. 2018, 172, 122–142. [Google Scholar] [CrossRef] [PubMed]
- Razi, K.; Muneer, S. Drought stress-induced physiological mechanisms, signaling pathways and molecular response of chloroplasts in common vegetable crops. Crit. Rev. Biotechnol. 2021, 41, 669–691. [Google Scholar] [CrossRef] [PubMed]
- Riyazuddin, R.; Nisha, N.; Singh, K.; Verma, R.; Gupta, R. Involvement of dehydrin proteins in mitigating the negative effects of drought stress in plants. Plant Cell Rep. 2022, 41, 519–533. [Google Scholar] [CrossRef]
- Abdel-Ghany, S.E.; Ullah, F.; Ben-Hur, A.; Reddy, A.S.N. Transcriptome Analysis of Drought-Resistant and Drought-Sensitive Sorghum (Sorghum bicolor) Genotypes in Response to PEG-Induced Drought Stress. Int. J. Mol. Sci. 2020, 21, 772. [Google Scholar] [CrossRef] [Green Version]
- Magwanga, R.O.; Lu, P.; Kirungu, J.N.; Lu, H.; Wang, X.; Cai, X.; Zhou, Z.; Zhang, Z.; Salih, H.; Wang, K.; et al. Characterization of the late embryogenesis abundant (LEA) proteins family and their role in drought stress tolerance in upland cotton. BMC Genet. 2018, 19, 1–31. [Google Scholar] [CrossRef] [Green Version]
- Cao, J.; Li, X. Identification and phylogenetic analysis of late embryogenesis abundant proteins family in tomato (Solanum lycopersicum). Planta 2015, 241, 757–772. [Google Scholar] [CrossRef]
- Shafqat, W.; Jaskani, M.J.; Maqbool, R.; Chattha, W.S.; Ali, Z.; Naqvi, S.A.; Haider, M.S.; Khan, I.A.; Vincent, C.I. Heat shock protein and aquaporin expression enhance water conserving behavior of citrus under water deficits and high temperature conditions. Environ. Exp. Bot. 2021, 181, 104270. [Google Scholar] [CrossRef]
- Afzal, Z.; Howton, T.C.; Sun, Y.; Mukhtar, M.S. The Roles of Aquaporins in Plant Stress Responses. J. Dev. Biol. 2016, 4, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chávez-Arias, C.C.; Ligarreto-Moreno, G.A.; Ramírez-Godoy, A.; Restrepo-Díaz, H. Maize responses challenged by drought, elevated daytime temperature and arthropod herbivory stresses: A physiological, biochemical and molecular view. Front. Plant Sci. 2021, 1512, 702841. [Google Scholar] [CrossRef] [PubMed]
- Shivaraj, S.M.; Sharma, Y.; Chaudhary, J.; Rajora, N.; Sharma, S.; Thakral, V.; Ram, H.; Sonah, H.; Singh-Pareek, S.L.; Sharma, T.R.; et al. Dynamic role of aquaporin transport sys-tem under drought stress in plants. Environ. Exp. Bot. 2021, 184, 104367. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, V.K.; Tripathi, V.; Singh, P.P.; Singh, A.K. Plant growth-promoting rhizobacteria (PGPR): Perspective in agriculture under biotic and abiotic stress. In Crop Improvement through Microbial Biotechnology; Elsevier: Amsterdam, The Netherlands, 2018; pp. 333–342. [Google Scholar] [CrossRef]
- Bi, H.; Miao, J.; He, J.; Chen, Q.; Qian, J.; Li, H.; Xu, Y.; Ma, D.; Zhao, Y.; Tian, X.; et al. Characterization of the Wheat Heat Shock Factor TaHsfA2e-5D Conferring Heat and Drought Tolerance in Arabidopsis. Int. J. Mol. Sci. 2022, 23, 2784. [Google Scholar] [CrossRef]
- Piveta, L.B.; Roma-Burgos, N.; Noldin, J.A.; Viana, V.E.; Oliveira, C.d.; Lamego, F.P.; Avila, L.A.d. Molecular and Physiological Responses of Rice and Weedy Rice to Heat and Drought Stress. Agriculture 2021, 11, 9. [Google Scholar] [CrossRef]
- Khan, Z.; Shahwar, D. Role of Heat Shock Proteins (HSPs) and Heat Stress Tolerance in Crop Plants. In Sustainable Agriculture in the Era of Climate Change; Roychowdhury, R., Choudhury, S., Hasanuzzaman, M., Srivastava, S., Eds.; Springer: Cham, Switzerland, 2020. [Google Scholar] [CrossRef]
- Kumar, R.R.; Goswami, S.; Singh, K.; Dubey, K.; Rai, G.K.; Singh, B.; Singh, S.; Grover, M.; Mishra, D.; Kumar, S.; et al. Characterization of novel heat-responsive transcription factor (TaHSFA6e) gene involved in regulation of heat shock proteins (HSPs)—A key member of heat stress-tolerance network of wheat. J. Biotechnol. 2018, 279, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, R.; Liu, W.; Zhang, H.; Guo, Y.; Wen, R. Genome-Wide Characterization of Heat-Shock Protein 70s from Chenopodium quinoa and Expression Analyses of Cqhsp70s in Response to Drought Stress. Genes 2018, 9, 35. [Google Scholar] [CrossRef] [Green Version]
- Ngumbi, E.; Kloepper, J. Bacterial-mediated drought tolerance: Current and future prospects. Appl. Soil Ecol. 2016, 105, 109–125. [Google Scholar] [CrossRef]
- Cherif-Silini, H.; Thissera, B.; Chenari Bouket, A.; Saadaoui, N.; Silini, A.; Eshelli, M.; Alenezi, F.N.; Vallat, A.; Luptakova, L.; Yahiaoui, B. Durum Wheat Stress Tolerance Induced by Endophyte Pantoea agglomerans with Genes Contributing to Plant Func-tions and Secondary Metabolite Arsenal. Int. J. Mol. Sci. 2019, 20, 3989. [Google Scholar] [CrossRef] [Green Version]
- Cherif-Silini, H.; Silini, A.; Chenari Bouket, A.; Alenezi, F.N.; Luptakova, L.; Bouremani, N.; Nowakowska, J.N.; Oszako, T.; Belbahri, L. Tailoring Next Generation Plant Growth Promoting Microorganisms as Versatile Tools beyond Soil Desalinization:A Road Map towards Field Application. Sustainability 2021, 13, 4422. [Google Scholar] [CrossRef]
- Kerbab, S.; Silini, A.; Chenari Bouket, A.; Cherif-Silini, H.; Eshelli, M.; El Houda Rabhi, N.; Belbahri, L. Mitigation of NaCl Stressin Wheat by Rhizosphere Engineering Using Salt Habitat Adapte d PGPR Halotolerant Bacteria. Appl. Sci. 2021, 11, 1034. [Google Scholar] [CrossRef]
- Ghosh, D.; Gupta, A.; Mohapatra, S. A comparative analysis of exopolysaccharide and phytohormone secretions by four drought-tolerant rhizobacterial strains and their impact on osmotic-stress mitigation in Arabidopsis thaliana. World J. Microbiol. Biotechnol. 2019, 35, 90. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.-C.; Jiang, C.-H.; Zhang, L.-N.; Chen, L.; Zhang, X.-Y.; Guo, J.-H. Biofilms Positively Contribute to Bacillus amyloliquefaciens 54-induced Drought Tolerance in Tomato Plants. Int. J. Mol. Sci. 2019, 20, 6271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azizi, S.; Kouchaksaraei, M.T.; Hadian, J.; Abad, A.R.F.N.; Sanavi, S.A.M.M.; Ammer, C.; Bader, M.K.F. Dual inoculations of arbuscular mycorrhizal fungi and plant growth-promoting rhizobacteria boost drought resistance and essential oil yield of common myrtle. For. Ecol. Manag. 2021, 497, 119478. [Google Scholar] [CrossRef]
- Danish, S.; Zafar-ul-Hye, M.; Fahad, S.; Saud, S.; Brtnicky, M.; Hammerschmiedt, T.; Datta, R. Drought Stress Alleviation by ACC Deaminase Producing Achromobacter xylosoxidans and Enterobacter cloacae, with and without Timber Waste Biochar in Maize. Sustainability 2020, 12, 6286. [Google Scholar] [CrossRef]
- Agami, R.A.; Ghramh, H.A.; Hasheem, M. Seed inoculation with Azospirillum lipoferum alleviates the adverse effects of drought stress on wheat plants. J. Appl. Bot. Food Qual. 2017, 90, 165–173. [Google Scholar] [CrossRef]
- Mariotti, L.; Scartazza, A.; Curadi, M.; Picciarelli, P.; Toffanin, A. Azospirillum baldaniorum Sp245 Induces Physiological Responses to Alleviate the Adverse Effects of Drought Stress in Purple Basil. Plants 2021, 10, 1141. [Google Scholar] [CrossRef]
- Li, H.; Guo, Q.; Jing, Y.; Liu, Z.; Zheng, Z.; Sun, Y.; Xue, Q.; Lai, H. Application of Streptomyces pactum Act12 enhances drought resistance in wheat. J. Plant Growth Regul. 2020, 39, 122–132. [Google Scholar] [CrossRef]
- He, A.; Niu, S.; Yang, D.; Ren, W.; Zhao, L.; Sun, Y.; Meng, L.; Zhao, Q.; Pare, P.W.; Zhang, J. Two PGPR strains from the rhizosphere of Haloxylon ammodendron promoted growth and enhanced drought tolerance of ryegrass. Plant Physiol. Biochem. 2021, 161, 74–85. [Google Scholar] [CrossRef]
- Sood, G.; Kaushal, R.; Sharma, M. Significance of inoculation with Bacillus subtilis to alleviate drought stress in wheat (Triticum aestivum L.). Vegetos 2020, 33, 782–792. [Google Scholar] [CrossRef]
- Joshi, B.; Chaudhary, A.; Singh, H.; Kumar, P.A. Prospective evaluation of individual and consortia plant growth promoting rhizobacteria for drought stress amelioration in rice (Oryza sativa L.). Plant Soil. 2020, 457, 225–240. [Google Scholar] [CrossRef]
- Saleem, M.; Nawaz, F.; Hussain, M.B.; Ikram, R.M. Comparative effects of individual and consortia Plant Growth Promoting Bacteria on physiological and enzymatic mechanisms to confer drought tolerance in maize (Zea mays L.). J. Soil Sci. Plant Nutr. 2021, 21, 3461–3476. [Google Scholar] [CrossRef]
- Khan, N.; Zandi, P.; Ali, S.; Mehmood, A.; Adnan Shahid, M.; Yang, J. Impact of salicylic acid and PGPR on the drought tolerance and phytoremediation potential of Helianthus annus. Front. Microbiol. 2018, 9, 2507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Batool, T.; Ali, S.; Seleiman, M.F.; Naveed, N.H.; Ali, A.; Ahmed, K. Plant growth promoting rhizobacteria alleviates drought stress in potato in response to suppressive oxidative stress and antioxidant enzymes activities. Sci. Rep. 2020, 10, 16975. [Google Scholar] [CrossRef] [PubMed]
- Zarei, T.; Moradi, A.; Kazemeini, S.A.; Akhgar, A.; Rahi, A.A. The role of ACC deaminase producing bacteria in improving sweet corn (Zea mays L. var saccharata) productivity under limited availability of irrigation water. Sci. Rep. 2020, 10, 20361. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Xin, K.; Liu, H.; Cheng, J.; Shen, X.; Wang, Y.; Zhang, L. Pantoea alhagi, a novel endophytic bacterium with ability to improve growth and drought tolerance in wheat. Sci. Rep. 2017, 7, 41564. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Astorga-Eló, M.; Gonzalez, S.; Acuña, J.J.; Sadowsky, M.J.; Jorquera, M.A. Rhizobacteria from ‘flowering desert’ events contribute to the mitigation of water scarcity stress during tomato seedling germination and growth. Sci. Rep. 2021, 11, 13745. [Google Scholar] [CrossRef]
- Chandra, D.; Srivastava, R.; Glick, B.R.; Sharma, A.K. Rhizobacteria producing ACC deaminase mitigate water-stress response in finger millet (Eleusine coracana (L.) Gaertn.). 3 Biotech 2020, 10, 65. [Google Scholar] [CrossRef]
- Khan, A.; Singh, A.V. Multifarious effect of ACC deaminase and EPS producing Pseudomonas sp. and Serratia marcescens to augment drought stress tolerance and nutrient status of wheat. World J. Microbiol. Biotechnol. 2021, 37, 198. [Google Scholar] [CrossRef]
- Dubey, A.; Saiyam, D.; Kumar, A.; Hashem, A.; Abd_Allah, E.F.; Khan, M.L. Bacterial Root Endophytes: Characterization of Their Competence and Plant Growth Promotion in Soybean (Glycine max (L.) Merr.) under Drought Stress. Int. J. Environ. Res. Public Health 2021, 18, 931. [Google Scholar] [CrossRef]
- Saikia, J.; Sarma, R.K.; Dhandia, R.; Yadav, A.; Bharali, R.; Gupta, V.K.; Saikia, R. Alleviation of drought stress in pulse crops with ACC deaminase producing rhizobacteria isolated from acidic soil of Northeast India. Sci. Rep. 2018, 8, 3560. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Yang, L.; Hao, R.; Bai, X.; Wang, Y.; Yu, X. Drought-tolerant plant growth-promoting rhizobacteria isolated from jujube (Ziziphus jujuba) and their potential to enhance drought tolerance. Plant Soil. 2020, 452, 423–440. [Google Scholar] [CrossRef]
- Murali, M.; Gowtham, H.G.; Singh, S.B.; Shilpa, N.; Aiyaz, M.; Niranjana, S.R.; Amruthesh, K.N.a. Bio-prospecting of ACC deaminase producing Rhizobacteria towards sustainable agriculture: A special emphasis on abiotic stress in plants. Appl. Soil Ecol. 2021, 168, 104142. [Google Scholar] [CrossRef]
- Yasmin, H.; Bano, A.; Wilson, N.L.; Nosheen, A.; Naz, R.; Hassan, M.N.; Illyas, N.; Saleem, M.; Noureldeen, A.; Ahmad, P.; et al. Drought-tolerant Pseudomonas sp. showed differential expression of stress-responsive genes and induced drought tolerance in Arabidopsis thaliana. Physiol. Plant. 2022, 174, e13497. [Google Scholar] [CrossRef]
- Puthiyottil, P.; Akkara, Y. Pre-treatment with Bacillus subtilis mitigates drought induced photo-oxidative damages in okra by modulating antioxidant system and photochemical activity. Physiol. Mol. Biol. Plants. 2021, 27, 945–957. [Google Scholar] [CrossRef]
- Narayanasamy, S.; Thangappan, S.; Uthandi, S. Plant growth-promoting Bacillus sp. cahoots moisture stress alleviation in rice genotypes by triggering antioxidant defense system. Microbiol. Res. 2020, 239, 126518. [Google Scholar] [CrossRef]
- Singh, B.; Gowtham, H.G.; Niranjana, S.R. ACC deaminase producing PGPR invoke changes in antioxidant systems to minimize the adverse effects of salt in sunflower. RJLBPCS 2019, 5, 1. [Google Scholar] [CrossRef]
- Gontia-Mishra, I.; Sapre, S.; Sharma, A.; Tiwari, S. Amelioration of drought tolerance in wheat by the interaction of plant growth-promoting rhizobacteria. Plant Biol. 2016, 18, 992–1000. [Google Scholar] [CrossRef]
- Chukwuneme, C.F.; Babalola, O.O.; Kutu, F.R.; Ojuederie, O.B. Characterization of Actinomycetes Isolates for Plant Growth Promoting Traits and Their Effects on Drought Tolerance in Maize. J. Plant Interact. 2020, 15, 93–105. [Google Scholar] [CrossRef] [Green Version]
- Latif, M.; Bukhari, S.A.H.; Alrajhi, A.A.; Alotaibi, F.S.; Ahmad, M.; Shahzad, A.N.; Dewidar, A.Z.; Mattar, M.A. Inducing Drought Tolerance in Wheat through E-xopolysaccharide-Producing Rhizobacteria. Agronomy 2022, 12, 1140. [Google Scholar] [CrossRef]
- Han, L.; Zhang, M.; Du, L.; Zhang, L.; Li, B. Effects of Bacillus amyloliquefaciens QST713 on Photosynthesis and Antioxidant Characteristics of Alfalfa (Medicago sativa L.) under Drought Stress. Agronomy 2022, 12, 2177. [Google Scholar] [CrossRef]
- Silva, R.; Filgueiras, L.; Santos, B.; Coelho, M.; Silva, M.; Estrada-Bonilla, G.; Vidal, M.; Baldani, J.I.; Meneses, C. Gluconacetobacter diazotrophicus Changes The Molecular Mechanisms of Root Development in Oryza sativa L. Growing Under Water Stress. Int. J. Mol. Sci. 2020, 21, 333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petrillo, C.; Vitale, E.; Ambrosino, P.; Arena, C.; Isticato, R. Plant Growth-Promoting Bacterial Consortia as a Strategy to Alleviate Drought Stress in Spinacia oleracea. Microorganisms 2022, 10, 1798. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Bano, A.; Babar, M.A. The stimulatory effects of plant growth promoting rhizobacteria and plant growth regulators on wheat physiology grown in sandy soil. Arch. Microbiol. 2019, 201, 769–785. [Google Scholar] [CrossRef]
- Mahmood Aulakh, A.; Qadir, G.; Hassan, F.U.; Hayat, R.; Sultan, T.; Billah, M.; Hussain, M.; Khan, N. Desert Soil Microbes as a Mineral Nutrient Acquisition Tool for Chickpea (Cicer arietinum L.) Productivity at Different Moisture Regimes. Plants. 2020, 9, 1629. [Google Scholar] [CrossRef]
- Govindasamy, V.; George, P.; Kumar, M.; Aher, L.; Raina, S.; Rane, J.; Annapurna, K.; Minhas, P.S. Multi-trait PGP rhizobacterial endophytes alleviate drought stress in a senescent genotype of sorghum [Sorghum bicolor (L.) Moench]. 3 Biotech. 2020, 10, 13. [Google Scholar] [CrossRef]
- Devarajan, A.K.; Muthukrishanan, G.; Truu, J.; Truu, M.; Ostonen, I.; Kizhaeral, S.S.; Panneerselvam, P.; Kuttalingam Gopalasubramanian, S. The Foliar Application of Rice Phyllosphere Bacteria induces Drought-Stress Tolerance in Oryza sativa (L.). Plants. 2021, 10, 387. [Google Scholar] [CrossRef]
- Karimzadeh, J.; Alikhani, H.A.; Etesami, H.; Pourbabaei, A.A. Improved Phosphorus Uptake by Wheat Plant (Triticum aestivum L.) with Rhizosphere Fluorescent Pseudomonads Strains Under Water-Deficit Stress. J. Plant Growth Regul. 2021, 40, 162–178. [Google Scholar] [CrossRef]
- Silambarasan, S.; Logeswari, P.; Vangnai, A.S.; Kamaraj, B.; Cornejo, P. Plant Growth-Promoting Actinobacterial Inoculant Assisted Phytoremediation Increases Cadmium Uptake in Sorghum bicolor under Drought and Heat Stresses. Environ. Pollut. 2022, 307, 119489. [Google Scholar] [CrossRef]
- Abbasi, S.; Sadeghi, A.; Safaie, N. Streptomyces alleviate drought stress in tomato plants and modulate the expression of transcription factors ERF1 and WRKY70 genes. Sci. Hortic. 2020, 265, 109206. [Google Scholar] [CrossRef]
- Rashid, U.; Yasmin, H.; Hassan, M.N.; Naz, R.; Nosheen, A.; Sajjad, M.; Ilyas, N.; Keyani, R.; Jabeen, Z.; Mumtaz, S.; et al. Drought-tolerant Bacillus megaterium isolated from semi-arid conditions induces systemic tolerance of wheat under drought conditions. Plant Cell Rep. 2022, 41, 549–569. [Google Scholar] [CrossRef] [PubMed]
- Bakhshandeh, E.; Gholamhosseini, M.; Yaghoubian, Y.; Pirdashti, H. Plant growth promoting microorganisms can improve germination, seedling growth and potassium uptake of soybean under drought and salt stress. Plant Growth Regul. 2020, 90, 123–136. [Google Scholar] [CrossRef]
- Tiwari, S.; Lata, C.; Chauhan, P.S.; Nautiyal, C.S. Pseudomonas putida attunes morphophysiological, biochemical and molecular responses in Cicer arietinum L. during drought stress and recovery. Plant Physiol Biochem. 2016, 99, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Saadaoui, N.; Silini, A.; Cherif-Silini, H.; Bouket, A.C.; Alenezi, F.N.; Luptakova, L.; Boulahouat, S.; Belbahri, L. Semi-Arid-Habitat-Adapted Plant-Growth-Promoting RhizobacteriaAllows Efficient Wheat GrowthPromotion. Agronomy 2022, 12, 2221. [Google Scholar] [CrossRef]
- Bhatt, R.M.; Selvakumar, G.; Upreti, K.K.; Boregowda, P.C. Effect of Biopriming with Enterobacter Strains on Seed Germination and Seedling Growth of Tomato (Solanum lycopersicum L.) Under Osmotic Stress. Proc. Natl. Acad. Sci. USA 2015, 85, 63–69. [Google Scholar] [CrossRef]
- Selvakumar, G.; Bhatt, R.M.; Upreti, K.K.; Bindu, H.; Kademani, S. Citricoccus zhacaiensis B-4 (MTCC 12119) a novel osmotolerant plant growth promoting actinobacterium enhances onion (Allium cepa L.) seed germination under osmotic stress conditions. World J. Microbiol. Biotechnol. 2015, 31, 833–839. [Google Scholar] [CrossRef]
- Galeano, R.M.S.; de Russo Godoy, F.M.; Duré, L.M.M.; Fernandes-Júnior, P.I.; Baldani, J.I.; Paggi, G.M.; Zanoelo, F.F.; Brasil, M.S. Potential of Bacterial Strains Isolated from Ironstone Outcrops Bromeliads to Promote Plant Growth Under Drought Conditions. Curr. Microbiol. 2021, 78, 2741–2752. [Google Scholar] [CrossRef]
- Kumar, A.; Usmani, Z.; Kumar, V. Biochar and flyash inoculated with plant growth promoting rhizobacteria act as potential biofertilizer for luxuriant growth and yield of tomato plant. J. Environ. Manag. 2017, 190, 20–27. [Google Scholar] [CrossRef]
- Li, Y.; Shi, H.; Zhang, H.; Chen, S. Amelioration of drought effects in wheat and cucumber by the combined application of super absorbent polymer and potential biofertilizer. Peer J. 2019, 7, e6073. [Google Scholar] [CrossRef]
- Huang, X.F.; Zhou, D.; Lapsansky, E.R.; Reardon, K.F.; Guo, J.; Andales, M.J.; Vivanco, J.M.; Manter, D.K. Mitsuaria sp. and Burkholderia sp. from Arabidopsis rhizosphere enhance drought tolerance in Arabidopsis thaliana and maize (Zea mays L.). Plant Soil 2017, 419, 523–539. [Google Scholar] [CrossRef]
- Grover, M.; Bodhankar, S.; Sharma, A.; Sharma, P.; Singh, J.; Nain, L. PGPR mediated alterations in root traits: Way toward sustainable crop production. Front. Sustain. Food Sys. 2021, 4, 618230. [Google Scholar] [CrossRef]
- Timmusk, S.; Abd El-Daim, I.A.; Copolovici, L.; Tanilas, T.; Kännaste, A.; Behers, L.; Nevo, E.; Seisenbaeva, G.; Niinemets, Ü. Drought-tolerance of wheat improved by rhizosphere bacteria from harsh environments: Enhanced biomass production and reduced emissions of stress volatiles. PLoS ONE 2014, 9, e96086. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ambreetha, S.; Chinnadurai, C.; Marimuthu, P.; Balachandar, D. Plant-associated Bacillus modulates the expression of auxin-responsive genes of rice and modifies the root architecture. Rhizosphere 2018, 5, 57–66. [Google Scholar] [CrossRef]
- Turan, M.; Arjumend, T.; Argın, S.; Yıldırım, E.; Katırcıoğlu, H.; Gürkan, B.; Ekinci, M.; Gunes, A.; Kocaman, A.; Bolouri, P. Plant root enhancement by plant growth promoting rhizobacteria. Plant Roots 2021, 1–19. [Google Scholar] [CrossRef]
- Jochum, M.D.; McWilliams, K.L.; Borrego, E.J.; Kolomiets, M.V.; Niu, G.; Pierson, E.A.; Jo, Y.K. Bioprospecting plant growth-promoting rhizobacteria that mitigate drought stress in grasses. Front. Microbiol. 2019, 10, 2106. [Google Scholar] [CrossRef]
- Mishra, S.K.; Khan, M.H.; Misra, S.; Dixit, V.K.; Gupta, S.; Tiwari, S.; Gupta, S.C.; Chauhan, P.S. Drought tolerant Ochrobactrum sp. inoculation performs multiple roles in maintaining the homeostasis in Zea mays L. subjected to deficit water stress. Plant Physiol. Biochem. 2020, 150, 1–14. [Google Scholar] [CrossRef]
- Woo, O.G.; Kim, H.; Kim, J.S.; Keum, H.L.; Lee, K.C.; Sul, W.J.; Lee, J.H. Bacillus subtilis strain GOT9 confers enhanced tolerance to drought and salt stresses in Arabidopsis thaliana and Brassica campestris. Plant Physiol. Biochem. 2020, 148, 359–367. [Google Scholar] [CrossRef]
- Mohanty, P.; Singh, P.K.; Chakraborty, D.; Mishra, S.; Pattnaik, R. Insight into the role of PGPR in sustainable agriculture and environment. Front. Sustain. Food Sys. 2021, 5, 667150. [Google Scholar] [CrossRef]
- Naveed, M.; Hussain, M.B.; Zahir, Z.A.; Mitter, B.; Sessitsch, A. Drought stress amelioration in wheat through inoculation with Burkholderia phytofirmans strain PsJN a. Plant Growth Regul. 2014, 73, 121–131. [Google Scholar] [CrossRef]
- Naveed, M.; Mitter, B.; Reichenauer, T.G.; Wieczorek, K.; Sessitsch, A. Increased drought stress resilience of maize through endophytic colonization by Burkholderia phytofirmans PsJN and Enterobacter sp. FD17. Environ. Exp. Bot. 2014, 97, 30–39. [Google Scholar] [CrossRef]
- García, J.E.; Maroniche, G.; Creus, C.; Suárez-Rodríguez, R.; Ramirez-Trujillo, J.A.; Groppa, M.D. In vitro PGPR properties and osmotic tolerance of different Azospirillum native strains and their effects on growth of maize under drought stress. Microbiol. Res. 2017, 202, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Mishra, S.; Dixit, V.; Kumar, M.; Agarwal, L.; Chauhan, P.S.; Nautiyal, C.S. Synergistic effect of Pseudomonas putida and Bacillus amyloliquefaciens ameliorates drought stress in chickpea (Cicer arietinum L.). Plant signal. Behav. 2016, 11, e1071004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Araújo, V.L.V.P.; Junior, M.A.L.; de Souza Júnior, V.S.; de Araújo Filho, J.C.; Fracetto, F.J.C.; Andreote, F.D.; Pereira, A.P.A.; Junior, J.P.M.; Barros, F.M.R.; Fracetto, G.G.M. Bacteria from tropical semiarid temporary ponds promote maize growth under hydric stress. Microbiol. Res. 2020, 240, 126564. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.C.; Bottini, R.; Pontin, M.; Berli, F.J.; Moreno, D.; Boccanlandro, H.; Travaglia, C.N.; Piccoli, P.N. Azospirillum brasilense ameliorates the response of Arabidopsis thaliana to drought mainly via enhancement of ABA levels. Physiol. Plant. 2015, 153, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Khalilpour, M.; Mozafari, V.; Abbaszadeh-Dahaji, P. Tolerance to salinity and drought stresses in pistachio (Pistacia vera L.) seedlings inoculated with indigenous stress-tolerant PGPR isolates. Sci. Hortic. 2021, 289, 110440. [Google Scholar] [CrossRef]
- Sandhya, V.; Sk, Z.A.; Grover, M.; Reddy, G.; Venkateswarlu, B. Alleviation of drought stress effects in sunflower seedlings by the exopolysaccharides producing Pseudomonas putida strain GAP-P45. Biol. Fertil. Soils. 2009, 46, 17–26. [Google Scholar] [CrossRef]
- Ma, Y.; Rajkumar, M.; Moreno, A.; Zhang, C.; Freitas, H. Serpentine endophytic bacterium Pseudomonas azotoformans ASS1 accelerates phytoremediation of soil metals under drought stress. Chemosphere. 2017, 185, 75–85. [Google Scholar] [CrossRef]
- Asghari, B.; Khademian, R.; Sedaghati, B. Plant growth promoting rhizobacteria (PGPR) confer drought resistance and stimulate biosynthesis of secondary metabolites in pennyroyal (Mentha pulegium L.) under water shortage condition. Sci. Hortic. 2020, 263, 109132. [Google Scholar] [CrossRef]
- Yasmin, H.; Rashid, U.; Hassan, M.N.; Nosheen, A.; Naz, R.; Ilyas, N.; Sajjad, M.; Azmat, A.; Alyemeni, M. Volatile organic compounds produced by Pseudomonas pseudoalcaligenes alleviated drought stress by modulating defense system in maize (Zea mays L.). Physiol. Plant. 2021, 172, 896–911. [Google Scholar] [CrossRef]
- Ahmad, H.M.; Fiaz, S.; Hafeez, S.; Zahra, S.; Shah, A.N.; Gul, B.; Aziz, O.; Ur-Rahman, M.; Fakhar, A.; Rafique, M. Plant growth-promoting rhizobacteria eliminate the effect of drought stress in plants: A review. Front. Plant Sci. 2022, 1965, 875774. [Google Scholar] [CrossRef]
- Oleńska, E.; Małek, W.; Wójcik, M.; Swiecicka, I.; Thijs, S.; Vangronsveld, J. Beneficial features of plant growth-promoting rhizobacteria for improving plant growth and health in challenging conditions: A methodical review. Sci. Total Environ. 2020, 743, 140682. [Google Scholar] [CrossRef] [PubMed]
- Abdela, A.A.; Barka, G.D.; Degefu, T. Co-inoculation effect of Mesorhizobium ciceri and Pseudomonas fluorescens on physiological and biochemical responses of Kabuli chickpea (Cicer arietinum L.) during drought stress. Plant Physiol. Rep. 2020, 25, 359–369. [Google Scholar] [CrossRef]
- Asaf, S.; Khan, A.L.; Khan, M.A.; Imran, Q.M.; Yun, B.W.; Lee, I.J. Osmoprotective functions conferred to soybean plants via inoculation with Sphingomonas sp. LK11 and exogenous trehalose. Microbiol. Res. 2017, 205, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Kaushal, M.; Wani, S.P. Rhizobacterial-plant interactions: Strategies ensuring plant growth promotion under drought and salinity stress. Agr. Ecosys. Environ. 2016, 231, 68–78. [Google Scholar] [CrossRef]
- Gou, W.E.I.; Tian, L.I.; Ruan, Z.H.I.; Zheng, P.E.N.G.; Chen, F.U.C.A.I.; Zhang, L.; Cui, Z.; Li, Z.; Gao, M.; Shi, W. Accumulation of choline and glycinebetaine and drought stress tolerance induced in maize (Zea mays) by three plant growth promoting rhizobacteria (PGPR) strains. Pak. J. Bot. 2015, 47, 581–586. [Google Scholar]
- Rabhi, N.E.H.; Cherif-Silini, H.; Silini, A.; Alenezi, F.N.; Chenari Bouket, A.; Oszako, T.; Belbahri, L. Alleviation of Salt Stress via Habitat-Adapted Symbiosis. Forests 2022, 13, 586. [Google Scholar] [CrossRef]
- Suárez, R.; Wong, A.; Ramírez, M.; Barraza, A.; Orozco, M.D.C.; Cevallos, M.A.; Lara, M.; Hernandez, G.; Iturriaga, G. Improvement of drought tolerance and grain yield in common bean by overexpressing trehalose-6-phosphate synthase in rhizobia. Mol. Plant-Microbe Interact. 2008, 21, 958–966. [Google Scholar] [CrossRef] [Green Version]
- Vílchez, J.I.; García-Fontana, C.; Román-Naranjo, D.; González-López, J.; Manzanera, M. Plant drought tolerance enhancement by trehalose production of desiccation-tolerant microorganisms. Front. Microbiol. 2016, 7, 1577. [Google Scholar] [CrossRef] [Green Version]
- Shameer, S.; Prasad, T.N.V.K.V. Plant growth promoting rhizobacteria for sustainable agricultural practices with special reference to biotic and abiotic stresses. Plant Growth Regul. 2018, 84, 603–615. [Google Scholar] [CrossRef]
- Lu, X.; Liu, S.-F.; Yue, L.; Zhao, X.; Zhang, Y.-B.; Xie, Z.-K.; Wang, R.-Y. Epsc Involved in the Encoding of Exopolysaccharides Produced by Bacillus amyloliquefaciens FZB42 Act to Boost the Drought Tolerance of Arabidopsis thaliana. Int. J. Mol. Sci. 2018, 19, 3795. [Google Scholar] [CrossRef] [Green Version]
- Pereira, S.I.A.; Abreu, D.; Moreira, H.; Vega, A.; Castro, P.M.L. Plant growth-promoting rhizobacteria (PGPR) improve the growth and nutrient use efficiency in maize (Zea mays L.) under water deficit conditions. Heliyon 2020, 6, e05106. [Google Scholar] [CrossRef] [PubMed]
- Dodd, I.C.; Zinovkina, N.Y.; Safronova, V.I.; Belimov, A.A. Rhizobacterial mediation of plant hormone status. Ann. Appl. Biol. 2010, 157, 361–379. [Google Scholar] [CrossRef]
- Vaishnav, A.; Choudhary, D.K. Regulation of Drought-Responsive Gene Expression in Glycine max L. Merrill is Mediated Through Pseudomonas simiae Strain AU. J. Plant Growth Regul. 2019, 38, 333–342. [Google Scholar] [CrossRef]
- Curá, J.A.; Franz, D.R.; Filosofía, J.E.; Balestrasse, K.B.; Burgueño, L.E. Inoculation with Azospirillum sp. and Herbaspirillum sp. Bacteria Increases the Tolerance of Maize to Drought Stress. Microorganisms 2017, 5, 41. [Google Scholar] [CrossRef] [Green Version]
- Kang, S.M.; Radhakrishnan, R.; Khan, A.L.; Kim, M.J.; Park, J.M.; Kim, B.R.; Shin, D.-H.; Lee, I.-J. Gibberellin secreting rhizobacterium, Pseudomonas putida H-2-3 modulates the hormonal and stress physiology of soybean to improve the plant growth under saline and drought conditions. Plant Physiol. Biochem. 2014, 84, 115–124. [Google Scholar] [CrossRef]
- Cohen, A.C.; Travaglia, C.N.; Bottini, R.; Piccoli, P.N. Participation of abscisic acid and gibberellins produced by endophytic Azospirillum in the alleviation of drought effects in maize. Botany. 2009, 87, 455–462. [Google Scholar] [CrossRef]
- Mohammadipanah, F.; Zamanzadeh, M. Bacterial Mechanisms Promoting the Tolerance to Drought Stress in Plants. In Secondary Metabolites of Plant Growth Promoting Rhizomicroorganisms; Singh, H., Keswani, C., Reddy, M., Sansinenea, E., García-Estrada, C., Eds.; Springer: Singapore, 2019. [Google Scholar] [CrossRef]
- Eke, P.; Kumar, A.; Sahu, K.P.; Wakam, L.N.; Sheoran, N.; Ashajyothi, M.; Patel, A.; Fekam, F.B. Endophytic bacteria of desert cactus (Euphorbia trigonas Mill) confer drought tolerance and induce growth promotion in tomato (Solanum lycopersicum L.). Microbiol. Res. 2019, 228, 126302. [Google Scholar] [CrossRef]
- Glick, B.R. Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol. Res. 2014, 169, 30–39. [Google Scholar] [CrossRef]
- Glick, B.R.; Nascimento, F.X. Pseudomonas 1-Aminocyclopropane-1-carboxylate (ACC) Deaminase and Its Role in Beneficial Plant-Microbe Interactions. Microorganisms 2021, 9, 2467. [Google Scholar] [CrossRef]
- Murali, M.; Singh, S.B.; Gowtham, H.G.; Shilpa, N.; Prasad, M.; Aiyaz, M.; Amruthesh, K.N. Induction of drought tolerance in Pennisetum glaucum by ACC deaminase producing PGPR-Bacillus amyloliquefaciens through Antioxidant defense system. Microbiol. Res. 2021, 253, 126891. [Google Scholar] [CrossRef]
- Ansari, F.A.; Ahmad, I. Alleviating Drought Stress of Crops Through PGPR: Mechanism and Application. In Microbial Interventions in Agriculture and Environment; Singh, D., Gupta, V., Prabha, R., Eds.; Springer: Singapore, 2019. [Google Scholar] [CrossRef]
- Zafar-ul-Hye, M.; Danish, S.; Abbas, M.; Ahmad, M.; Munir, T.M. ACC Deaminase Producing PGPR Bacillus amyloliquefaciens and Agrobacterium fabrum along with Biochar Improve Wheat Productivity under Drought Stress. Agronomy 2019, 9, 343. [Google Scholar] [CrossRef] [Green Version]
- Mhlongo, M.I.; Piater, L.A.; Dubery, I.A. Profiling of Volatile Organic Compounds from Four Plant Growth-Promoting Rhizobacteria by SPME–GC–MS: A Metabolomics Study. Metabolites 2022, 12, 763. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, D.; Garladinne, M.; Lee, Y.H. Volatile Indole Produced by Rhizobacterium Proteus vulgaris JBLS202 Stimulates Growth of Arabidopsis thaliana Through Auxin, Cytokinin, and Brassinosteroid Pathways. J. Plant Growth Regul. 2015, 34, 158–168. [Google Scholar] [CrossRef]
- Cho, S.M.; Kang, B.R.; Han, S.H.; Anderson, A.J.; Park, J.Y.; Lee, Y.H.; Cho, B.H.; Yang, K.Y.; Ryu, C.M.; Kim, Y.C. 2R, 3R-butanediol, a bacterial volatile produced by Pseudomonas chlororaphis O6, is involved in induction of systemic tolerance to drought in Arabidopsis thaliana. Mol. Plant-Microbe Interact. 2008, 21, 1067–1075. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.; Wang, J.; Hui, W.; Zhao, F.; Wang, P.; Su, C.; Gong, W. Physiology of Plant Responses to Water Stress and Related Genes: A Review. Forests 2022, 13, 324. [Google Scholar] [CrossRef]
- Saakre, M.; Baburao, T.M.; Salim, A.P.; Ffancies, R.M.; Achuthan, V.P.; Thomas, G.; Sivarajan, S.R. Identification and characterization of genes responsible for drought tolerance in rice mediated by Pseudomonas fluorescens. Rice Sci. 2017, 24, 291–298. [Google Scholar] [CrossRef]
- Kaushal, M. Portraying rhizobacterial mechanisms in drought tolerance: A way forward toward sustainable agriculture. In PGPR Amelioration in Sustainable Agriculture; Woodhead Publishing: Sawston, UK, 2019; pp. 195–216. [Google Scholar] [CrossRef]
- Vargas, L.; Santa Brigida, A.B.; Mota Filho, J.P.; de Carvalho, T.G.; Rojas, C.A.; Vaneechoutte, D.; Bel, M.V.; Farrinelli, L.; Ferreira, P.C.G.; Vandepoel, K.; et al. Drought tolerance conferred to sugarcane by association with Gluconacetobacter diazotrophicus: A transcriptomic view of hormone pathways. PLoS ONE 2014, 9, e114744. [Google Scholar] [CrossRef] [Green Version]
- Sukweenadhi, J.; Kim, Y.J.; Choi, E.S.; Koh, S.C.; Lee, S.W.; Kim, Y.J.; Yang, D.C. Paenibacillus yonginensis DCY84T induces changes in Arabidopsis thaliana gene expression against aluminum, drought, and salt stress. Microbiol. Res. 2015, 172, 7–15. [Google Scholar] [CrossRef]
- Sen, S.; Ghosh, D.; Mohapatra, S. Modulation of polyamine biosynthesis in Arabidopsis thaliana by a drought mitigating Pseudomonas putida strain. Plant Physiol. Biochem. 2018, 129, 180–188. [Google Scholar] [CrossRef]
- Sonbarse, P.P.; Kiran, K.; Sharma, P.; Parvatam, G. Biochemical and molecular insights of PGPR application for the augmentation of carotenoids, tocopherols, and folate in the foliage of Moringa oleifera. Phytochemistry 2020, 179, 112506. [Google Scholar] [CrossRef]
- Omara, A.E.-D.; Hafez, E.M.; Osman, H.S.; Rashwan, E.; El-Said, M.A.A.; Alharbi, K.; Abd El-Moneim, D.; Gowayed, S.M. Collaborative Impact of Compost and Beneficial Rhizobacteria on Soil Properties, Physiological Attributes, and Productivity of Wheat Subjected to Deficit Irrigation in Salt Affected Soil. Plants 2022, 11, 877. [Google Scholar] [CrossRef] [PubMed]

| Plant Species | Effectiveness of Stress | Reference |
|---|---|---|
| Wheat (Triticum aestivum L.) | -Inhibition of seed germination. -Reduction in seed germination rate, coleoptile weight length, and seed radicle length. | [16] |
| Rice (Oryza sativa L.) | -Limitations in photosynthesis, stomatal conductance, transpiration rate, maximum electron transport rate, and carboxylation efficiency. | [17] |
| Potato (Solanum tuberosum L.) | -Decrease in relative water content (RWC), photosynthetic rate, and chlorophyll a, b content. -Increase in the content of electrical conductivity and the content of MDA, absissic acid (ABA), proline, soluble sugars, and polyamines. | [18] |
| Alfalfa (Medicago sativa) | -Decrease in photosynthesis rate, chlorophyll a and b, a + b content, and Rubisco activity. | [19] |
| Cotton (Gossypium sp.) | -Increase in ABA, proline, soluble sugar content, and antioxidant enzyme activities. -Decrease in root size and biomass. -Decrease in cotton yield and quality. | [20] |
| Wheat (Triticum aestivum L.) | -Decrease in photosynthetic pigments, chlorophyll a and b, and carotenoids. -Increase in H2O2, loss of electrolyte, MDA, proline, and protein content. | [21] |
| Wheat (Triticum aestivum L.) | -Reduction in photosynthesis, stomatal conductance, relative water content (RWC), grain weight, and grain yield. | [22] |
| Wheat (Triticum aestivum L.) | -Decrease in length, fresh weight, and dry weight of the plant shoots. -Decrease in membrane stability, chlorophyll a, b, relative water content (RWC), K, P, and N content in shoots. - Increase in proline, sugar, and antioxidant enzyme activity (SOD, POD, CAT). -Decrease in weight and number of grains per ear. | [23] |
| Canola (Brassica napus L.) | -Inhibition and decrease in germination percentage. -Decrease in chlorophyll a and b content. -Strong increase in H2O2 and MDA content, loss of electrolytes (EL). - Increase in proline, antioxidant enzyme activity of SOD, POD, and CAT, concentrations of non-enzymatic antioxidants such as ascorbic acid (AsA), and glutathione (GSH). | [24] |
| Sugar beet (Beta vulgaris L.) | -Decrease in chlorophyll a, b, chl a + b carotenoids, and anthocyanins. -Increase in proline content and synthesis, and accumulation of antioxidant activities (CAT, APX, POX) and radicals (ROS) that damage the cell membrane. -Increase in MDA, H2O2, and membrane degradation, and decrease in membrane stability. -Decrease in relative leaf water content (RWC) and root yield. | [25] |
| Tomato (Solanum lycopersicum L.) | -Decrease in height, shoot weight, chl a, b, a + b, and relative leaf water content (RWC). -Increase in MDA and H2O2 content, proline accumulation, and SOD and APX enzyme activity. | [26] |
| Maize (Zea mays L.) | -Reduction in size and fresh and dry weight of shoots and roots. -Decrease in chl a, b, a + b, and relative leaf water content (RWC). -Decrease in net photosynthetic, transpiration rate, and stomatal conductance. -Increase in ABA concentrations. -Causes changes in the ultra-structure of chloroplasts and thylakoids. | [27] |
| PGPR Strains | Plant Species | Mechanisms | Beneficial Features Related to Drought Tolerance | Reference |
|---|---|---|---|---|
| Pseudomonas fluorescens; P. putida | Myrtle (Myrtus communis L.) | IAA ,Nitrogen fixation | -Enhancement of plant biomass (shoot and root trimming), greater water and nutrient absorption. -Increase in chlorophyll a, b, and carotenoid concentration while decreasing electrolyte loss and MDA. -Stimulation of enzymatic (CAT, SOD, POD) and non-enzymatic (Phenol and flavonoids) antioxidant defense. | [137] |
| Achromobacter xylosoxidans; Enterobacter cloacae | Maize (Zea mays L.) | ACC deaminase IAA, | -Increase in plant size, height, shoot dry weight, grain production, photosynthetic rate, transpiration rate, chlorophyll a, b, and a + b, and stomatal conductance. | [138] |
| Bacillus sp.; Azospirillum lipoferum; Azospirillum brasilense | Wheat (Triticum turgidum. L) | IAA, ABA, CK | -Increase in seed germination and root system growth. -Improved plant biomass (fresh and dry weight), RWC, photosynthetic pigment concentration (Chl a, b, and carotenoids), nutrient absorption, and plant yield. -Decrease in EL while maintaining membrane stability. -Increase the amount of osmolytes (proline and sugar) and antioxidant enzymes (CAT, SOD, APX). | [5] |
| Bacillus amyloliquefaciens 54 | Tomato (Solanum lycopersicum.L) | EPS | -Improved RWC, root vigor, antioxidant enzyme activity (SOD, APX, CAT, POD). -Decrease in MDA content and increase in nced1 gene transcription and ABA content. | [136] |
| Azospirillum lipoferum | Wheat (Triticum aestivum. L) | - | -Decrease in oxidative membrane damage. Improved plant growth, RWC, grain yield, and leaf photosynthesis. -Increase in antioxidant enzymes such as SOD, POD, and non-enzymatic phenolics. | [139] |
| Azospirillum baldaniorum Sp245 | Basilic (Ocimum basilicum L.) | ISR | -Increase in the content of phytohormones in plant xylem (SA, JA, ABA, etc.) -Increase in chlorophyll a, b, a + b, and anthocyanin concentration, as well as photosynthesis. | [140] |
| Streptomyces pactum Act12 | Wheat (Triticum aestivum L.) | - | -Increase in root and shoot growth and fresh weight, as well as osmolyte sugar and proline accumulation, RWC. -Decrease in MDA content and increase in ABA content and non-enzymatic antioxidant. -Stress resistance gene expression SnRK2, EXPA2, EXPA6, P5CS | [141] |
| Bacillus sp. WM13-24; Pseudomonas sp. M30- 35 | Ray-grass (Lolium perenne L.) | IAA, ACC deaminase, EPS | -Improved root architecture (RSA), shoot trimming, and fresh and dry pea shoots and roots -Increase in photosynthetic rate, RWC, total chlorophyll, total nitrogen, and phosphorus levels in shoots. -Decrease in MDA content, relative membrane permeability (RMP), and H2O2 buildup. -Increase in antioxidant enzyme activity (CAT, POD) and proline accumulation. | [142] |
| Pseudomonas fluorescens WCS417; Bacillus amyloliquefaciens GB03 | Peppermint (Mentha piperita L.) | IAA, ACC deaminase | -Increase in growth through increasing leaf area, fresh and dry weight of shoots and roots. -Decrease in MDA levels and increase in both enzymatic (PX, SOD) and non-enzymatic (Phenol, ascorbic acid) antioxidant activities. | [45] |
| Pseudomonas azotoformans FAP5 | Wheat (Triticum aestivum L.) | EPS ,ACC deaminase, IAA | Increase in the rate of germination. Increase in plant biomass (root and leaf size, dried and fresh root and leaf weight). -Increase in RAS/RT ratio, soil aggregation, photosynthesis, and chlorophyll content. -Reduction in MDA levels and antioxidant enzyme activity. | [64] |
| Bacillus subtilis | Wheat (Triticum aestivum L.) | ACC deaminase | -Improved plant development in terms of size, fresh peas, and dry weight of shoots and roots. -Increase in RWC, chlorophyll levels, minerals, and antioxidant enzyme activity (SOD, CAT, POD) | [143] |
| Bacillus spp. | Rice (Oryza sativa L.) | ACC deaminase, IAA, EPS. | -Improved germination, plant biomass, and yield while reducing membrane and oxidative damage. | [144] |
| Bacillus sp. MN-54; Enterobacter sp. FD-17; Pseudomonas fluorescens 1 | Maize (Zea mays L.) | ACC deaminase ,ABA | -Increase in RWC, chlorophyll a, b, and carotenoid concentration, and photosynthesis. Increase in the activity of antioxidant enzymes (SOD, GPX, CAT). | [145] |
| Planomicrobium chinense P1; Bacillus cereus P2 | Sunflower (Helianthus annuus L.) | Phosphate solubilization Production of phytohormone (GA, AIA) | -Improved shoot length, root length, fresh and dried root biomass. -Increase in quantity of chlorophyll, carotenoids, leaf proteins, sugar, and phenolic substances. -Reduction in MDA levels. | [146] |
| Bacillus subtilis; Azospirillum brasilense | Wheat (Triticum aestivum L.) | EPS | -Increase in germination rate, seedling vigor index, and promptitude index. -Improved plant biomass (height, dry weight, fresh weight of shoots and roots, leaf area). -Increase in water potential and chlorophyll content (Chl a, b, and carotenoids). -Increase in osmolyte content (proline, amino acids, sugars and proteins) and antioxidant enzymes (SOD, CAT, and POD). | [69] |
| Pseudomonas sp. N66 | Sorghum bicolor L. | ACC deaminase | -Improved growth and root system architecture (RSA) via regulation of phytohormones (GA, AIA, CK). -Activates systemic tolerance induced (ISR) by the signaling hormones brassinolides, SA, and JA and the signaling molecules sphingosine and psychosine. -Reduced ethylene levels. -Increase in antioxidant activity and osmolyte accumulation (proline, glutamic acid, choline). | [68] |
| Bacillus subtilis HAS31 | Potato (Solanum tuberosum L.) | ACC deaminase | -Increase in chlorophyll concentration, soluble proteins, total soluble carbohydrates, and antioxidant enzyme activity (CAT, POD, SOD). | [147] |
| Pseudomonas fluorescens | Maize (Zea mays L.) | ACC deaminase | -Enhanced chl a, b, and total chl content, as well as F0 and Fm photosynthetic activities and yield. -Increase in proline accumulation, total soluble sugars, and antioxidant activity (CAT, POD). | [148] |
| Pantoea alhagi LTYR-11ZT | Wheat (Triticum aestivum L.) | IAA, EPS | -Improved growth and fresh weight of shoots and roots. -Increase in the amount of sugar and chlorophyll and reduction in MDA content. | [149] |
| Bacillus sp.; Pseudomonas sp. | Tomato (Solanum lycopersicum L.) | ACC deaminase, EPS, IAA. | -Increase in the rate of germination. -Increase in plant biomass and photosynthesis. -Improved resilience to drought and recovery. | [150] |
| Ochrobactrum anthropi DPB13; Pseudomonas palleroniana DPB15 Pseudomonas fluorescens DPB16; Pseudomonas palleroniana | Finger millet (Eleusine coracana L.) | ACC deaminase, IAA. | -Increase in growth metrics (shoot and root size and weight), total chlorophyll, and nutrient supply (N, P, K Na+, Ca2+). -Reduction in H2O2 and MDA levels and improved membrane stability. -Increase in proline, phenol, and antioxidant enzyme activity accumulation (SOD, GPX, APX). | [151] |
| Pseudomonas sp.; Serratia marcescens | Wheat (Triticum aestivum L.) | ACC deaminase, IAA, EPS | -Decrease in reactive oxygen species. -Enhanced osmolyte accumulation, chlorophyll, and carotenoid content in plant leaves, and Zn and Fe content in leaves are all benefits of improved plant water status. | [152] |
| Bacillus cereus AKAD A1-1, Pseudomonas otitidis AKAD A1-2; Pseudomonas sp. AKAD A1-16 | Soybean (Glycine max L.) | ACC deaminase, IAA, EPS | -Improved plant biomass (height, fresh weight, dried shoot and root weight), RWC, and chlorophyll content. -Increase in proline, soluble carbohydrates, and protein content. -Decrease in MDA and H2O2 levels. | [153] |
| Ochrobactrum pseudogrignonense RJ12; Pseudomonas sp. RJ15; Bacillus subtilis RJ46 | Pea (Pea sativum L.) Black gam (Vigna mungo L.) | ACC deaminase. | -Improved plant biomass, root and shoot length, and seed germination. -Increase in the accumulation of osmolytes (proline, phenol), chlorophyll content, and RWC. -Decrease in ACC accumulation in plants and decrease in production of the ACC oxidase gene. -Increase in antioxidant enzyme activity (CAT, POD). | [154] |
| Pseudomonas lini DT6; Serratia plymuthica DT8 | Jujubier (Ziziphus jujuba) | ACC deaminase, IAA EPS | -Increase in plant biomass via pruning (fresh and dry peas from branches and roots) ,RWC. -Increase in the phosphate and nitrogen content of the leaves. -Reduction in the amount of MDA and ABA in the leaves. -Increase in SOD and POD antioxidant enzymes. | [155] |
| Bacillus amyloliquefaciens MMR0 | Candle millet (Pennisetum glaucum) | ACC deaminase | -Increase in seedling vigor and germination rate. -Enhanced plant growth factors such as total chlorophyll and RWC. -Reduction in MDA content, whereas proline, APX, and SOD levels were elevated. -Significant reduction in the relative expression of drought response marker genes (DREB-1E) and ethylene response factor (ERF-1B), with enhanced APX1 and SOD1 gene expression. | [156] |
| Pseudomonas SP2 | Arabidopsis thaliana | ACC deaminase, ABA ,IAA | -Improved plant biomass, RWC, and chlorophyll levels and reduction in MDA. -Improved SP2 colonization and survival. -Increase in osmolyte, glycine-betaine, and proline accumulation. | [157] |
| Bacillus subtilis | Okra (Abelmoschus esculentus L.) | - | -Increase in osmolyte (sugar and proline) accumulation, non-enzymatic antioxidants (glutathione GSH, ascorbate AsA), and enzymatic antioxidants (SOD, CAT, APX, GR, DHAR, MDHAR). -Increase in photosynthesis and reduction in MDA and O2− levels. | [158] |
| Bacillus altitudinis FD48; Bacillus methylotrophicus RABA6 | Rice (Oryza sativa L.) | IAA, CK, GA, ACC deaminase | -Increase in root and shoot growth, root to shoot ratio R/S, RWC, photosynthetic pigments (Chl a and b), and proline content. -Reduction in oxidative stress by increasing the activity of antioxidant enzymes (CAT, SOD, POD, APX). -Increase in the number of productive tillers, the quantity of seeds, the grain weight, and the harvest index. | [159] |
| Bacillus thuringiensis Rhizo SF 23; Bacillus subtilis Rhizo SF 48 | Sunflower (Helianthus annuus) | ACC deaminase. | -Improved plant growth, proline content, and antioxidant enzyme activity (APX, SOD). -Reduction in MDA content. | [160] |
| Klebsiella sp.; Enterobacter ludwigii; Flavobacterium sp | Wheat (Triticum aestivum L.) | EPS, ACC deaminase, IAA. | -Increase in the quantity, size, and dry weight of roots and shoots, as well as RWC and RAS/RT. -Reduction in H2O2, MDA, and EL while preserving membrane integrity and stability. -Decrease in proline and soluble sugars accumulation. -Enhanced expression of resistance genes (DREB2A, CAT1). | [161] |
| Arthrobacter arilaitensis; Streptomyces pseudovenezuelae | Maize (Zea mays L.) | ACC deaminase, phosphate solubilization, IAA, siderophore production and ammonia | -Increase in leaf number, height, fresh weight, dry weight of shoots and roots, and chlorophyll content. | [162] |
| Bacillus spp. | Maize (Zea mays L.) | IAA, nitrogen fixation, Phosphate solubilization | -Enhanced growth parameters (height, fresh weight, dry weight). -Increase in the amount of photosynthetic pigments (chl a, b, and total chl). -Decrease in MDA, H2O2, and antioxidant enzyme activity (SOD, APX, POD, CAT). | [11] |
| Klebsiella sp. (LEW16) | Wheat (Triticum aestivum L.) | EPS, IAA | -Increase in germination rate, root morphology, and seedling growth parameters (height, fresh and dry weight, and root diameter). | [163] |
| Bacillus amyloliquefaciens QST713 | Alfalfa (Medicago sativa L.) | IAA, EPS | -Increase in plant growth and biomass (dry, fresh weight of shoots and roots), RWC, photosynthesis, chlorophyll a, b, carotenoid content, and antioxidant enzyme activities (SOD, CAT, POD, APX). -Reduction in H2O2, O2− content. -Induction of systemic resistance. | [164] |
| Gluconacetobacter diazotrophicus Pal5 | Rice (Oryza sativa L.) | Nitrogen fixation, IAA | -Improved plant development (leaf area, root and shoot size), root system architecture (RSA), and RWC. -Increase in photosynthetic activity (Fv/Fm), photosynthetic pigment (Chll a, b, a + b, and carotenoids), phytohormone concentration (AIA, GA1, GA3, and zeatin), and osmolyte accumulation (trehalose, -tocopherol). -Activation of the expression of genes involved in root formation. | [165] |
| B. amyloliquefaciens RHF6; B. amyloliquefaciens LMG9814; Bacillus sp. AGS84 | Spinach (Spinacia oleracea L.) | IAA, phosphate solubilization, NH3 | -Increase in plant biomass and germination rate. -Increase in the content of photosynthetic pigments (Chll a, b, a + b, and carotenoids), as well as Fv/Fm photosynthesis. | [166] |
| Planomicrobium chinense strain P1; Bacillus cereus strain P2 | Wheat (Triticum aestivum L.) | EPS, IAA | -Increase in plant biomass (fresh weight, dry weight of roots and shoots), RWC, and micro/macronutrient accumulation (such as Ca, Mg, Na, K, Cu, Cr, Zn, and Fe). -Decrease in MDA content, antioxidant enzyme activity (POX, CAT, POD), and proline content. | [167] |
| Mesorhizobium cicero.; B. subtilis; B. mojavenss | Chickpea (Cicer arietinum L.) | IAA, ACC deaminase | -Improved plant growth (height, fresh peas, shoot and root dry weight), nutrient accumulation in seeds and leaves, root colonization, and survival. -Improvement in seed yield. | [168] |
| Ochrobactrum sp. EB-165; Microbacterium sp. EB-65; Enterobacter sp. EB-14; Enterobacter cloacae strain EB-48 | Sorghum (Sorghum bicolor.L.) | IAA, N2 fixation, phosphate solubilization, siderophore synthesis, ACC deaminase | -Increase in proline accumulation, relative water content (RWC), and membrane stability index. -Improved root system development and topology (height, fresh weight and dry weight). -Induction of particular gene upregulation: sbP5CS2, sbP5CS1. | [169] |
| Bacillus endophyticus PB3; Bacillus altitudinis PB46; Bacillus megaterium PB50 | Rice (Oryza sativa L.) | ACC deaminase, IAA, EPS, GA | -Increase in (RWC), total sugars, proteins, proline, phenolics, potassium, calcium, and ABA. -Increased the expression of stress-related genes (LEA, RAB16B, HSP70, SNAC1, and bZIP23). | [170] |
| Pseudomona helmanticensis; Pseudomonas | Wheat (Triticum aestivum L.) | Phosphate solubilization, IAA. | -Increase in seed and plant growth (height, fresh weight, and dry weight of shoots and roots). -Increase in the availability of phosphorus in the soil as well as its uptake by the shoots. | [171] |
| Streptomyces sp. RA04; Nocardiopsis sp RA07 | Sorghum (Sorghum bicolor L.) | ACC deaminas, Phosphate solubilization, IAA, sederophore synthesis | -Increase in plant growth, photosynthesis, and chlorophyll levels. -Reduction in MDA level while increasing the activity of antioxidant enzymes SOD, CAT, and APX. | [172] |
| Streptomyces strains IT25; Streptomyces C-2012 | Tomato (Solanum lycopersicum L.) | ACC deaminas, Phosphate solubilization, sederophore synthesis | -Increase in plant growth and fruit output. -Reduction in CAT and GPX activity while increasing relative water, proline, and sugar content. -Inhibition of ERF1 and WRKY70 stress transcript genes. | [173] |
| Bacillus megaterium; B. licheniformis | Wheat (Triticum turgidum L.) | ACC deaminase IAA, | -Increase in seed germination. -Increase in plant development (shoot and root), RWC, chlorophyll a, b, and carotenoids, and osmolyte accumulation (proline, sugar, protein). -Decrease in MDA and electrolytes (EL). -Increase in the activity of antioxidant enzymes (SOD, APX, PODL, CAT, GR). -Stimulation of water stress-related protein production. | [174] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouremani, N.; Cherif-Silini, H.; Silini, A.; Bouket, A.C.; Luptakova, L.; Alenezi, F.N.; Baranov, O.; Belbahri, L. Plant Growth-Promoting Rhizobacteria (PGPR): A Rampart against the Adverse Effects of Drought Stress. Water 2023, 15, 418. https://doi.org/10.3390/w15030418
Bouremani N, Cherif-Silini H, Silini A, Bouket AC, Luptakova L, Alenezi FN, Baranov O, Belbahri L. Plant Growth-Promoting Rhizobacteria (PGPR): A Rampart against the Adverse Effects of Drought Stress. Water. 2023; 15(3):418. https://doi.org/10.3390/w15030418
Chicago/Turabian StyleBouremani, Naoual, Hafsa Cherif-Silini, Allaoua Silini, Ali Chenari Bouket, Lenka Luptakova, Faizah N. Alenezi, Oleg Baranov, and Lassaad Belbahri. 2023. "Plant Growth-Promoting Rhizobacteria (PGPR): A Rampart against the Adverse Effects of Drought Stress" Water 15, no. 3: 418. https://doi.org/10.3390/w15030418






