Stormwater Treatment Using Natural and Engineered Options in an Urban Growth Area: A Case Study in the West of Melbourne
Abstract
:1. Introduction
- A reverse osmosis (RO)-based treatment process primarily consisting of microfiltration (MF), RO, advanced oxidation (UV/H2O2), and chlorination in a storage reservoir (Cl2).
- An ozone-and-biologically active filtration (O3/BAF)-based process primarily consisting of O3/BAF, ultrafiltration (UF), advanced oxidation (UV/H2O2), and chlorination in a storage reservoir.
2. Methods
2.1. Treatment Trains
2.2. Estimation of Key Microbial Parameter Removal
2.3. Estimation of Key Chemical Parameter Removal
2.3.1. PAH Removal
| Treatment Train Stage | Chemical | Water Type | Key Conditions | Comment and Removal | Reference and Reference Type |
|---|---|---|---|---|---|
| Wetland | PAH | Stormwater | Feed concentration of 786 ± 514 ng/L; 42 m2 wetland; southern Greece (35°19__N and 25°10__E); mixed cultures of two species of reed, Phragmites australis and Arudo donax; mean water temperature: 12.1 and 34.1 °C | Free-water-surface constructed wetland; 68% removal | [18], experimental |
| Wetland | Heavy metals | Stormwater | Various constructions and conditions | Typical removal: 30–60% removal, 30% used in modelling | [19], literature review |
| Wetland | BOD, COD | Stormwater | No media, with floating, submerged, and emergent plants; continuous water supply | Free-water-surface constructed wetland; around 89% BOD removal and 72% COD removal | [11], literature review |
| RO | PAH | Wastewater | Validation of specific application and operational conditions required | Indicative, >80% removal; intended to be informative and not to be used as the design basis for schemes | [6] (Table 4.10 of water recycling guidelines) |
| RO | As(III) | Synthetic brackish water | Feed concentration between 0.36 and 1.2 mg/L As(III), spiked drinking water, Dow 5K membranes, tests at manufacturer operating specifications | 85% removal | [20] |
| RO | Cd | Drinking water | Feed concentrations between 0.47 mg/L and 1.9 mg/L, spiked drinking water, Dow 5K membranes, tests at manufacturer operating specifications | 98% removal | [20] |
| RO | Pb | Municipal and industrial wastewater | Laboratory and on-site pilot-scale tests, feed concentration of 1.5 mg/L, various conditions | 89 to 100% removal, 95% used in modelling | [21], experimental |
| RO | COD, BOD | Hospital wastewater | Feed concentrations: 200–235 mg/L COD, 95–115 mg/L BOD; feed rate: 10–14 L/h; variable pressure to 13.6 Bar maximum; specific flux: 90–190 L/m2/h/bar | More than 99% removal for both COD and BOD | [22], experimental |
| UV | PAH | Natural water | Feed concentration: 3.9 to 5.6 µg/L; 3 different PAHs; 3 different water matrices; fluence between 40 and 1500 mJ/cm2 | Negligible PAH removal at 40 mJ/cm2 | [23], experimental |
| UV | COD, BOD | Raw and biotreated textile dye bath effluent | Feed concentrations: 760 mg/L COD, 261 mg/L BOD; UV dose: 5 mW/cm2; exposure time between 5 and 25 min | Negligible COD and BOD removal at 12 mJ/cm2, 35% BOD and 25% COD removal at 7500 mJ/cm2 | [24], experimental |
| UV/H2O2 | PAH | Natural water | Feed concentration: 3 µg/L; 30 min contact time; UV radiation: 170 µW/cm2; 10 mg/L H2O2 | 99% removal at 306 mJ/cm2 | [25], experimental |
| UV/H2O2 | COD, BOD | Raw and biotreated textile dye bath effluent | Feed concentrations: 760 mg/L COD, 261 mg/L BOD; UV intensity: 5 mW/cm2 and 254 nm; 150–200 mg/L H2O2 for raw wastewater, 100–150 mg/L for biotreated wastewater | Negligible BOD and COD removal at 800 mJ/cm2 Raw wastewater: 35% COD removal, 44% BOD removal at 7500 mJ/cm2 Biotreated: ~85% COD removal and ~90% BOD removal at 7500 mJ/cm2 | [24], experimental |
| Cl2 | PAH | Wastewater | Various conditions | Indicative, >80% removal | [6] |
| Cl2 | COD | Industrial wastewater | Feed concentration: 39 mg/L; chlorination after coagulation and flocculation; 1.2 mg/L free chlorine; 30 min contact time | 10% removal | [33], experimental |
| Cl2 | BOD | Secondary-effluent wastewater | Various feed concentrations: 12–30 mg/L, 5 mg/L residual Cl2; 15 min contact time | 67% to 20% depending on starting concentration; conservative setting of 30% removal chosen for modelling | [32], experimental |
| O3 + biological treatment | PAH | Contaminated water | Feed PAH concentration: ~5000 µg/L; 0.5 mg/L ozone; 30 min ozone treatment; 24 h biological treatment in flask | 91% PAH removal overall | [36], experimental |
| BAC | PAH | Diesel and petrol Synthetic wastewater | Petroleum content: 5 mg/L; ~1100 µg/L PAH; 8 L BAC to 300 L contaminated water; aerobic conditions; 12–24 h contact time | 97% PAH removal | [27], experimental |
| O3 + BAC | As | Groundwater | Feed As concentration: 14–27 µg/L; 43 min contact time | 99% removal | [28], experimental |
| BAC | Pb, Cd | Wastewater | Feed Pb and Cd concentrations: ~ 200 µg/L; 50–150 mg/L activated carbon; 2 h contact time | 99% Pb and 86% Cd removal | [29], experimental |
| O3 | COD, BOD | Secondary-effluent wastewater | Full scale, variable feed concentrations (~10–80 mgO2/L COD, ~2–10 mgO2/L BOD), 11–13 mg/L ozone | 8%–88% COD removal with most results in 10–20% removal range, ~0% BOD removal | [37], experimental |
| O3 + BAF | COD | Surface water | Biological sand filter, full-scale plant, 30–60 min contact time, 17 mg/L O3 concentration | Two different plants: one achieved ~50% COD removal, the other, ~20% COD removal | [38], experimental |
| O3 + BAF | BOD, COD | Textile effluent | Biological aerated filtration; feed COD ≤ 110 mg/L, BOD ≤ 30 mg/L; 20–25 mg/L ozone dose; 3.3 h hydraulic retention time; 6 air-to-water flow ratio | Approximately 64% COD removal, 67% BOD removal | [26], experimental |
| UF | PAH | Biologically treated wastewater | Feed PAH concentration: 22–38 µg/L; 0.04 µm pore size | 67% removal | [30], experimental |
| UF | COD, BOD | Hospital wastewater | Feed concentrations: 200–235 mg/L COD, 95–115 mg/L BOD; 0.01 µm pore size (1 kDa molecular-weight cutoff) | 97% removal for both COD and BOD | [22], experimental |
| COD, BOD | Stormwater | Feed concentrations: 11–32 mg/L BOD, 28–60 mg/L COD; 50 kDa molecular-weight cutoff UF | [39], experimental |
2.3.2. Arsenic, Cadmium, and Lead Removal
2.3.3. BOD and COD Removal
2.4. Cost Estimates
3. Results and Discussion
3.1. Microbial Removal
3.2. Chemical Removal
3.2.1. PAH Removal
3.2.2. BOD and COD
3.2.3. Arsenic, Cadmium, and Lead Removal
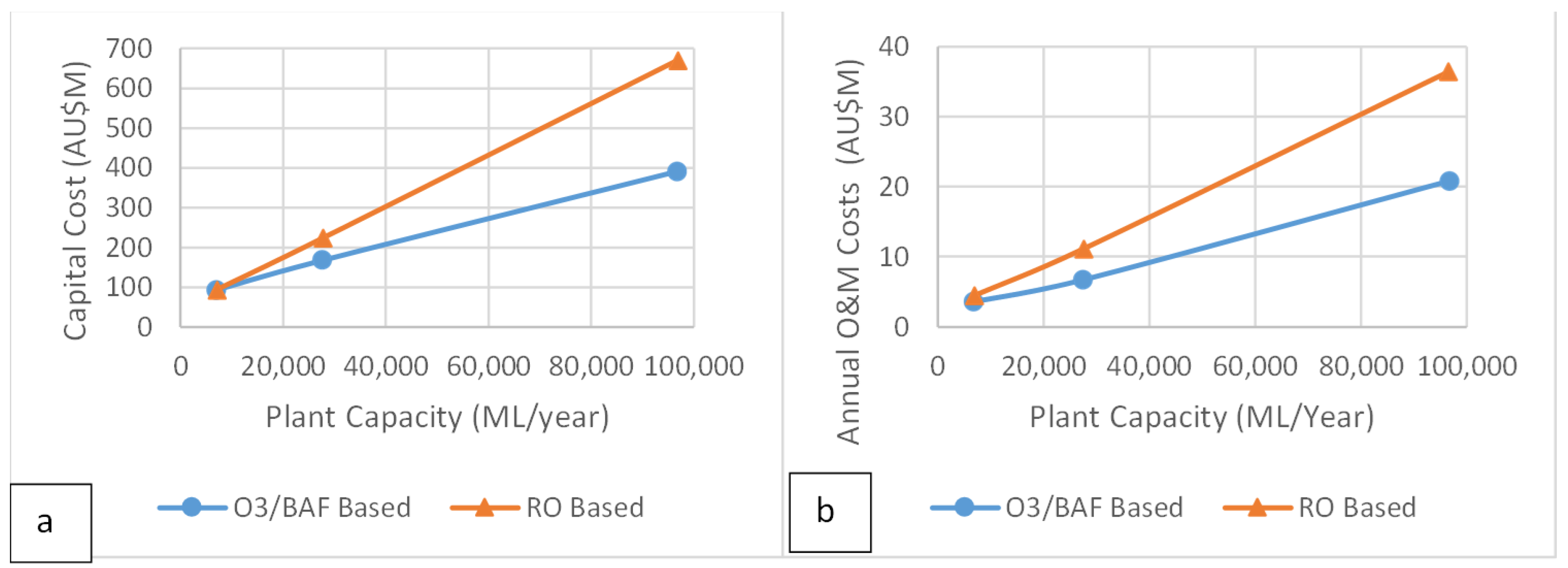
3.3. Cost Estimates
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- State Government of Victoria. Werribee Catchment Integrated Water Management Plan, Targets Driving Outcomes, Summary, September 2022. Available online: https://www.water.vic.gov.au/__data/assets/word_doc/0031/677407/werribee-catchment-iwm-plan-summary-accessible.docx (accessed on 13 November 2023).
- DELWP. Guidelines for Assessing the Impact of Climate Change on Water Supplies in Victoria Final Report. December 2016. Available online: https://nla.gov.au/nla.obj-385683871/view (accessed on 25 March 2023).
- Wiwoho, B.S.; Astuti, I.S.; Purwanto, P.; Deffinika, I.; Alfarizi, I.A.G.; Sucahyo, H.R.; Gusti, R.; Herwanto, M.T.; Herlambang, G.A. Assessing long-term rainfall trends and changes in a tropical watershed Brantas, Indonesia: An approach for quantifying the agreement among satellite-based rainfall data, ground rainfall data, and small-scale farmers questionnaires. Nat. Hazards 2023, 117, 2835–2862. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.K.; Sanciolo, P.; Behroozi, A.; Navaratna, D.; Muthukumaran, S. Stormwater Harvesting Potential for Local Reuse in an Urban Growth Area: A Case Study of Melton Growth Area in the West of Melbourne. Water 2023, 15, 2093. [Google Scholar] [CrossRef]
- NRMMC; EPHC; NHMRC. Australian Guidelines for Water Recycling (Phase 2): Stormwater Harvesting and Reuse; Natural Resource Management Ministerial Council: Canberra, Australia; Environment Protection and Heritage Council: Canberra, Australia; National Health and Medical Research Council: Canberra, Australia, 2009. Available online: www.waterquality.gov.au/sites/default/files/documents/water-recycling-guidelines-stormwater-23.pdf (accessed on 20 April 2023).
- EPHC; NHMRC; NRMMC. Australian Guidelines for Water Recycling: Managing Health and Environmental Risks (Phase 2)—Augmentation of Drinking Water Supplies: Natural Resource Management Ministerial Council, the Environment Protection and Health Council, and the National Health and Medical Research Council. 2008. Available online: www.waterquality.gov.au/sites/default/files/documents/water-recycling-guidelines-augmentation-drinking-22.pdf (accessed on 20 April 2023).
- Orange City Council. Orange Stormwater to Potable: Building Urban Water Supply Diversity. Prepared by Cooperative Research Centre for Water Sensitive Cities, City of Orange, NSW, May 2018. Available online: https://watersensitivecities.org.au/wp-content/uploads/2018/05/Orange-stormwater-to-potable-Case-Study-180503-V8_WEB.pdf (accessed on 20 April 2023).
- McCallum, T. Kalkallo: A Case Study in Technological Innovation Amidst Complex Regulation; Cooperative Research Centre for Water Sensitive Cities: Melbourne, Australia, 2015; ISBN 978-1-921912-24-5; Available online: https://watersensitivecities.org.au/wp-content/uploads/2016/05/TMR_A3-2_KalkalloCaseStudy.pdf (accessed on 20 April 2023).
- NWRI (National Water Research Institute). Framework for Direct Potable Reuse; Sponsored by the American Water Works Association (AWWA), Water Environment Foundation (WEF), and Wate Reuse Research Foundation; WRRF: Alexandria, VA, USA, 2015; Available online: https://watereuse.org/wp-content/uploads/2015/09/14-20.pdf (accessed on 5 November 2023).
- Schoen, M.E.; Ashbolt, N.J.; Jahne, M.A.; Garland, J. Risk-based enteric pathogen reduction targets for non-potable and direct potable use of roof runoff, stormwater, and greywater. Microb. Risk Anal. 2017, 5, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Varma, M.; Gupta, A.K.; Ghosal, P.S.; Majumder, A. A review on performance of constructed wetlands in tropical and cold climate: Insights of mechanism, role of influencing factors, and system modification in low temperature. Sci. Total Environ. 2021, 755, 142540. [Google Scholar] [CrossRef] [PubMed]
- Soller, J.A.; Eftim, S.E.; Warren, I.; Nappier, S.P. Evaluation of microbiological risks associated with direct potable reuse. Microb. Risk Anal. 2017, 5, 3–14. [Google Scholar] [CrossRef]
- WHO. Potable Reuse: Guidance for Producing Safe Drinking-Water. Licence: CC BY-NC-SA 3.0 IGO. 2017. Available online: https://apps.who.int/iris/handle/10665/258715 (accessed on 13 November 2023).
- USEPA. Potable Reuse Compendium; Report No: EPA/810/R-17/002; USEPA: Washington, DC, USA, 2017. Available online: https://www.epa.gov/sites/default/files/2018-01/documents/potablereusecompendium_3.pdf (accessed on 5 November 2023).
- Ahmed, W.; Hamilton, K.; Toze, S.; Cook, S.; Page, D. A review on microbial contaminants in stormwater runoff and outfalls: Potential health risks and mitigation strategies. Sci. Total Environ. 2019, 692, 1304–1321. [Google Scholar] [CrossRef]
- WSAA (Water Services Association of Australia). Manual for the Application of Health Based Targets for Drinking Water Safety, September; WSA 202—2015-1.2; WRRF: Denver, CO, USA, 2015; Available online: https://www.wsaa.asn.au/sites/default/files/publication/download/Health%20Based%20Targets%20Manual_0_0.pdf (accessed on 20 April 2023).
- NHMRC; NRMMC. Australian Drinking Water Guidelines. Commonwealth of Australia. 2011. Available online: www.nhmrc.gov.au/sites/default/files/documents/reports/aust-drinking-water-guidelines.pdf (accessed on 20 April 2023).
- Fountoulakis, M.; Terzakis, S.; Kalogerakis, N.; Manios, T. Removal of polycyclic aromatic hydrocarbons and linear alkylbenzene sulfonates from domestic wastewater in pilot constructed wetlands and a gravel filter. Ecol. Eng. 2009, 35, 1702–1709. [Google Scholar] [CrossRef]
- Haarstad, K.; Bavor, H.J.; Maehlum, T. Organic and metallic pollutants in water treatment and natural wetlands: A review. Water Sci. Technol. 2012, 65, 76–99. [Google Scholar] [CrossRef]
- USEPA. Reverse Osmosis Treatment to Remove Inorganic Contaminants from Drinking Water; Report No: EPA/600/S2-87/109; USEPA: Washington, DC, USA, 1987. Available online: https://nepis.epa.gov/Exe/ZyPDF.cgi/94001878.PDF?Dockey=94001878.PDF (accessed on 13 November 2023).
- Ozbey-Unal, B.; Omwene, P.I.; Yagcioglu, M.; Balcik-Canbolat, Ç.; Karagunduz, A.; Keskinler, B.; Dizge, N. Treatment of organized industrial zone wastewater by microfiltration/reverse osmosis membrane process for water recovery: From lab to pilot scale. J. Water Process. Eng. 2020, 38, 101646. [Google Scholar] [CrossRef]
- Jadhao, R.K.; Dawande, S.D. Reverse osmosis and ultrafiltration membrane for hospital wastewater treatment. Int. J. Chem. Sci. Appl. 2012, 3, 283–288. [Google Scholar]
- Sanches, S.; Leitão, C.; Penetra, A.; Cardoso, V.; Ferreira, E.; Benoliel, M.; Crespo, M.B.; Pereira, V. Direct photolysis of polycyclic aromatic hydrocarbons in drinking water sources. J. Hazard. Mater. 2011, 192, 1458–1465. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, A.; Shafeeq, A.; Butt, M.A.; Rizvi, Z.H.; Chughtai, M.A.; Rehman, S. Decolorization and removal of COD and BOD from raw and biotreated textile dye bath effluent through advanced oxidation process (AOP). Braz. J. Chem. Eng. 2008, 25, 453–459. [Google Scholar] [CrossRef]
- Rubio-Clemente, A.; Chica, E.; Peñuela, G.A. Photovoltaic array for powering advanced oxidation processes: Sizing, application and investment costs for the degradation of a mixture of anthracene and benzo[a]pyrene in natural water by the UV/H2O2 system. J. Environ. Chem. Eng. 2018, 6, 2751–2761. [Google Scholar] [CrossRef]
- He, Y.; Wang, X.; Xu, J.; Yan, J.; Ge, Q.; Gu, X.; Jian, L. Application of integrated ozone biological aerated filters and membrane filtration in water reuse of textile effluents. Bioresour. Technol. 2013, 133, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Augulyte, L.; Kliaugaite, D.; Racys, V.; Jankunaite, D.; Zaliauskiene, A.; Bergqvist, P.-A.; Andersson, P.L. Multivariate analysis of a biologically activated carbon (BAC) system and its efficiency for removing PAHs and aliphatic hydrocarbons from wastewater polluted with petroleum products. J. Hazard. Mater. 2009, 170, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Pokhrel, D.; Viraraghavan, T.; Braul, L. Evaluation of Treatment Systems for the Removal of Arsenic from Groundwater. Pract. Period. Hazard. Toxic Radioact. Waste Manag. 2005, 9, 152–157. [Google Scholar] [CrossRef]
- Dong, L.; Hou, L.; Wang, Z.; Gu, P.; Chen, G.; Jiang, R. A new function of spent activated carbon in BAC process: Removing heavy metals by ion exchange mechanism. J. Hazard. Mater. 2018, 359, 76–84. [Google Scholar] [CrossRef]
- Smol, M.; Włodarczyk-Makuła, M. Effectiveness in the Removal of Polycyclic Aromatic Hydrocarbons From Industrial Wastewater by Ultrafiltration Technique. Arch. Environ. Prot. 2012, 38, 49–58. [Google Scholar] [CrossRef]
- Kumar, V.; Dwivedi, S.; Oh, S. A critical review on lead removal from industrial wastewater: Recent advances and future outlook. J. Water Process. Eng. 2022, 45, 102518. [Google Scholar] [CrossRef]
- Ishihara, M.; Ogawa, H.; Iwahori, K. Effects of Nitrification and Chlorine Disinfection on BOD of Effluent from Johkasou Systems. Jpn. J. Water Treat. Biol. 2009, 45, 115–122. [Google Scholar] [CrossRef]
- Üstün, G.E.; Solmaz, S.K.A.; Çiner, F.; Başkaya, H.S. Tertiary treatment of a secondary effluent by the coupling of coagulation–flocculation–disinfection for irrigation reuse. Desalination 2011, 277, 207–212. [Google Scholar] [CrossRef]
- USEPA. Separation of Dilute Hazardous Organics by Low Pressure Composite Membranes; Report No: EPA/600/S2-87/053; USEPA: Washington, DC, USA, 1987. Available online: https://nepis.epa.gov/Exe/ZyPDF.cgi/2000TLJI.PDF?Dockey=2000TLJI.PDF (accessed on 13 November 2023).
- USEPA. Treatability Manual Volume III: Technologies for Control/Removal of Pollutants; Report No.: EPA-600/2-82-00lc; Office of Research and Development, US Environmental Protection Agency: Washington, DC, USA, 1980.
- Yerushalmi, L.; Nefil, S.; Hausler, R.; Guiot, S.R. Removal of Pyrene and Benzo(a)Pyrene from Contaminated Water by Sequential and Simultaneous Ozonation and Biotreatment. Water Environ. Res. 2006, 78, 2286–2292. [Google Scholar] [CrossRef] [PubMed]
- Martínez, S.B.; Pérez-Parra, J.; Suay, R. Use of Ozone in Wastewater Treatment to Produce Water Suitable for Irrigation. Water Resour. Manag. 2011, 25, 2109–2124. [Google Scholar] [CrossRef]
- Zanacic, E.; Stavrinides, J.; McMartin, D.W. Field-analysis of potable water quality and ozone efficiency in ozone-assisted biological filtration systems for surface water treatment. Water Res. 2016, 104, 397–407. [Google Scholar] [CrossRef]
- Sandoval, A.D.O.; Brião, V.B.; Fernandes, V.M.C.; Hemkemeier, A.; Friedrich, M.T. Stormwater management by microfiltration and ultrafiltration treatment. J. Water Process Eng. 2019, 30, 100453. [Google Scholar] [CrossRef]
- Warsinger, D.M.; Chakraborty, S.; Tow, E.W.; Plumlee, M.H.; Bellona, C.; Loutatidou, S.; Karimi, L.; Mikelonis, A.M.; Achilli, A.; Ghassemi, A.; et al. A review of polymeric membranes and processes for potable water reuse. Prog. Polym. Sci. 2018, 81, 209–237. [Google Scholar] [CrossRef] [PubMed]
- Yangali-Quintanilla, V.; Verliefde, A.; Kim, T.-U.; Sadmani, A.; Kennedy, M.; Amy, G. Artificial neural network models based on QSAR for predicting rejection of neutral organic compounds by polyamide nanofiltration and reverse osmosis membranes. J. Membr. Sci. 2009, 342, 251–262. [Google Scholar] [CrossRef]
- Fujioka, T.; Tu, K.L.; Khan, S.J.; McDonald, J.A.; Roux, A.; Poussade, Y.; Drewes, J.E.; Nghiem, L.D. Rejection of small solutes by reverse osmosis membranes for water reuse applications: A pilot-scale study. Desalination 2014, 350, 28–34. [Google Scholar] [CrossRef]
- Berego, Y.S.; Sota, S.S.; Ulsido, M.D.; Beyene, E.M. Treatment Performance Assessment of Natural and Constructed Wetlands on Wastewater from Kege Wet Coffee Processing Plant in Dale Woreda, Sidama Regional State, Ethiopia. Environ. Health Insights 2022, 16, 1–17. [Google Scholar] [CrossRef]
- Lenntech Website. Available online: https://www.lenntech.com/services/mwco.htm (accessed on 20 April 2023).
- El-Rehaili, A.M. Response of BOD, COD and TOC of secondary effluents to chlorination. Water Res. 1995, 29, 1571–1577. [Google Scholar] [CrossRef]
- WRRF (Water Reuse Research Foundation). Fit for Purpose Water: The Cost of Overtreating Reclaimed Water; WRRF Product No.: 10-01-1; WRRF: Denver, CO, USA, 2014; ISBN 978-1-941242-03-2. Available online: https://watereuse.org/watereuse-research/fit-for-purpose-water/ (accessed on 20 April 2023).
- WRRF (Water Reuse Research Foundation). The Opportunities and Economics of Direct Potable Reuse; WRRF Product No: 14-08-1; WRRF: Denver, CO, USA, 2014; ISBN 978-1-941242-17-9. Available online: https://watereuse.org/watereuse-research/the-opportunities-and-economics-of-direct-potable-reuse/ (accessed on 20 April 2023).
- Melbourne Water. Melbourne Water System Strategy; Melbourne Water: Melbourne, Australia, 2017; ISBN 978-1-925541-07-6. Available online: https://www.melbournewater.com.au/media/1801/download (accessed on 20 April 2023).
- Melbourne Water. Melbourne’s Water Outlook 2021. 2020. Available online: https://www.melbournewater.com.au/media/15436/download (accessed on 20 April 2023).
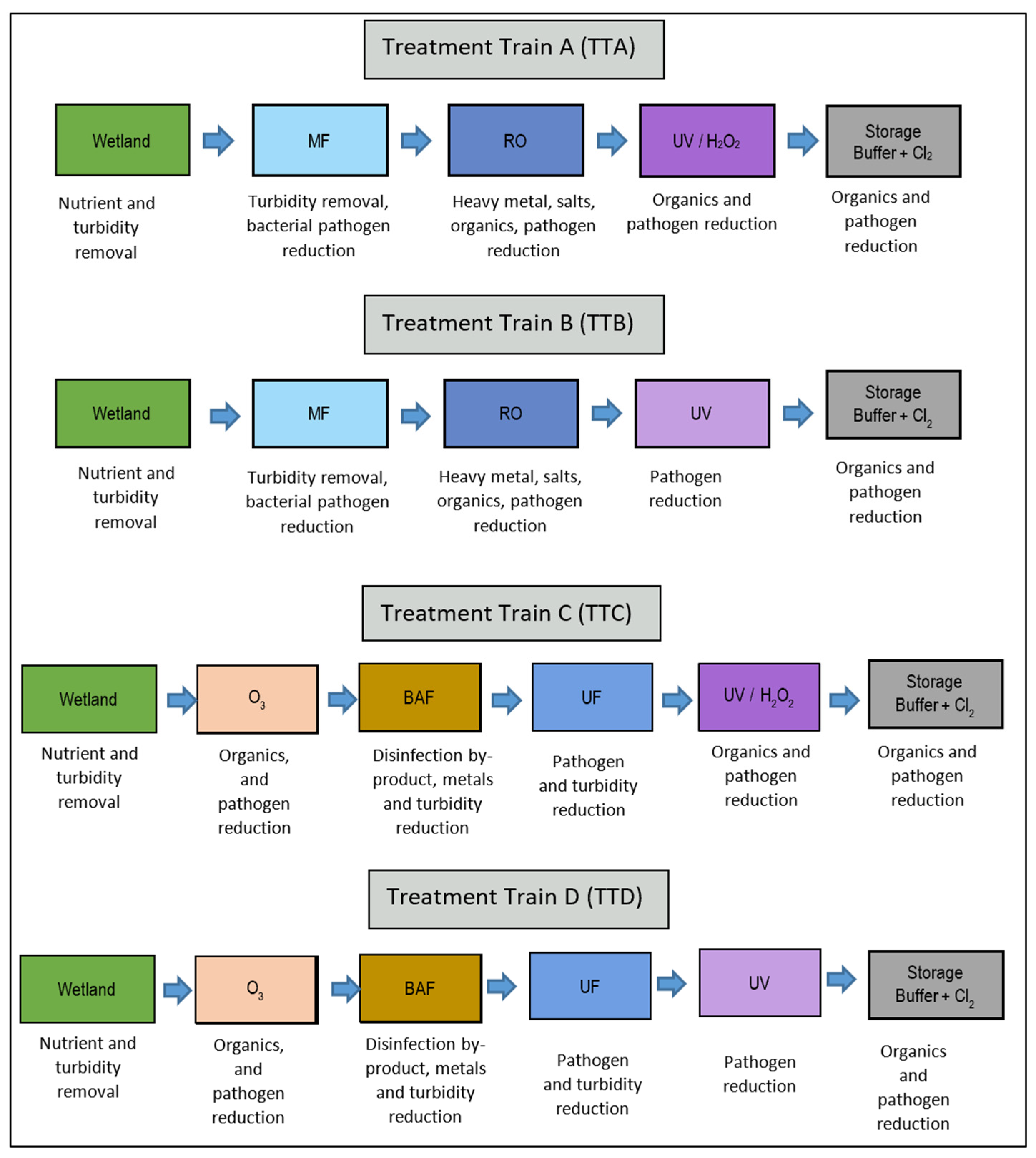

| Process Stage | Bacteria (Campylobacter) | Viruses (Adenovirus) | Cryptosporidium | Reference |
|---|---|---|---|---|
| Wetland | 0.2 | 0 | 0 | [15] |
| MF | 3 | 1 | 3 | [16] |
| RO | 3 | 3 | 3 | [13] a |
| UV (12 mJ/cm2) | 4 | 1 | 2 | [12] |
| UV/H2O2 (800 mJ/cm2) | 4 | 4 | 4 | [9,12] b |
| O3 + BAF | 4 | 4 | 1 | [12] |
| UF | 3 | 2.5 | 3 | [16] |
| Cl2 (Ct > 15 mg/L·min) | 4 | 4 | 0 | [16] |
| Process Stage | Parameter | |||||
|---|---|---|---|---|---|---|
| PAHs (Benzo(a)pyrene) | As | Cd | Pb | BOD | COD | |
| Wetlands | 68 (A) | 30 (B) | 30 (B) | 30 (B) | 89 (C) | 72 (C) |
| MF | 0 | 0 | 0 | 0 | 0 | 0 |
| RO | 80 (D) | 85 (E) | 98 (E) | 95 (F) | 99 (G) | 99 (G) |
| UV | 0 (H) | 0 | 0 | 0 | 0(I) | 0 (I) |
| UV/H2O2 | 99 (J) | 0 | 0 | 0 | 0 (I) | 0 (I) |
| O3 | * | 0 | 0 | 0 | 6711 (K) | 6411 (K) |
| BAF | 97 (L) | 99 (M) | 86 (N) | 95 (N) | - | - |
| UF | 67 (O) | 0 (P) | 0 (P) | 0 (P) | 97 (G) | 97 (G) |
| Chlorination in engineered storage buffer | 80 (D) | 0 | 0 | 0 | 30 (Q) | 10 (R) |
| Bacteria (Campylobacter) | Viruses (Adenovirus) | Cryptosporidium | |
|---|---|---|---|
| TTA | 14.2 | 12 | 10 |
| TTB | 14.2 | 9 | 8 |
| TTC | 15.2 | 14.5 | 8 |
| TTD | 15.2 | 11.5 | 6 |
| Default LRTs for wastewater 1 | 8.1 | 9.5 | 8.0 |
| Conservative literature LRTs for stormwater 2 | 8.2 | 8.9 | 8.5 |
| Less conservative literature LRTs for stormwater 3 | 6.2 | 6.9 | 6.5 |
| Soller et al. [12] TT | Corresponding Current Study TT | Approximate Cumulative Annual Risk of Infection |
|---|---|---|
| TT1a: MF-RO-AOP-Cl2 in storage | TTA | 2.5 × 10−9 |
| TT1b: MF-RO-UV-Cl2 in storage | TTB | 5.0 × 10−4 |
| TT3a: O3-BAF-UF-AOP-Cl2 in storage | TTC | 1.2 × 10−8 |
| TT3b: O3-BAF-UF-UV–Cl2 in storage | TTD | 3.9 × 10−5 |
| Literature Stormwater Quality A | Wetland Analysis Results (Current Project) | Comparison 1 | Comparison 2 | Comparison 3 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Contaminant | Unit | Mean | SD | Median | 95th Percentile | Wetlands, In (Mean) | Wetlands, Out (Mean) | Guideline Values B | 95th Percentile Literature Data vs. Guidelines | Wetlands (Out) vs. Guidelines | Wetlands (Out) Mean vs. Literature Data Mean |
| Metal | |||||||||||
| Aluminium | mg/L | 1.19 | 0.6 | 1.07 | 2.29 | 0.2380 | 0.318 | N/A | Under | ||
| Arsenic (III) | mg/L | 0.009 | 0.001 | 0.009 | 0.011 | 0.0012 | 0.0015 | 0.01 | Over | Under | Under |
| Barium | mg/L | 0.028 | 0.005 | 0.028 | 0.038 | 0.0576 | 0.0364 | 2 | Under | Under | Over |
| Cadmium | mg/L | 0.0198 | 0.0242 | 0.0127 | 0.0606 | <0.0002 | <0.0002 | 0.002 | Over | Under | Under |
| Chromium | mg/L | 0.009 | 0.001 | 0.008 | 0.017 | <0.001 | 0.0015 | 0.05 | Under | Under | Under |
| Copper | mg/L | 0.055 | 0.047 | 0.041 | 0.141 | 0.0020 | 0.001 | 2 | Under | Under | Under |
| Iron | mg/L | 2.842 | 1.246 | 2.674 | 5.1 | 1.2680 | 0.87 | N/A | - | - | Under |
| Lead | mg/L | 0.073 | 0.048 | 0.063 | 0.162 | <0.001 | <0.001 | 0.01 | Over | Under | Under |
| Manganese | mg/L | 0.111 | 0.046 | 0.103 | 0.197 | 0.1780 | 0.0284 | 0.5 | Under | Under | Under |
| Mercury | mg/L | 0.000218 | 0.000105 | 0.000201 | 0.000411 | <0.0001 | <0.0001 | 0.001 | Under | Under | Under |
| Nickel | mg/L | 0.009 | 0.004 | 0.009 | 0.017 | 0.003 | 0.003 | 0.02 | Under | Under | Under |
| Zinc | mg/L | 0.293 | 0.153 | 0.272 | 0.57 | 0.0072 | 0.0102 | N/A | Under | ||
| Nutrient | |||||||||||
| Total nitrogen | mg/L | 3.09 | 2.33 | 2.51 | 7.46 | 1.3 | 0.54 | N/A | Under | ||
| Total dissolved nitrogen | mg/L | 3.28 | 2.61 | 2.59 | 8.22 | 0.52 | 0.27 | N/A | Under | ||
| Total phosphorus | mg/L | 0.48 | 0.413 | 0.367 | 1.261 | 0.16 | 0.06 | ID | Under | ||
| Ammonia | mg/L | 1.135 | 1.187 | N/A | 3.281 | 0.200 | 0.400 | N/A | Under | ||
| Organic | |||||||||||
| PAHs (benzo(a)pyrene) | μg/L | 0.262 | 0.306 | 0.168 | 0.811 | <1 | <1 | 0.01 | Over | (a) | (a) |
| Physicochemical | |||||||||||
| Chloride | mg/L | 11.4 | 1.05 | 11.35 | 13.2 | 37.4 | 14.4 | 250 (b) | Under | Under | Over |
| Sodium | mg/L | 10.63 | 2.82 | 10.31 | 15.72 | 39.4 | 23.6 | 180 (b) | Under | Under | Over |
| Total dissolved solids | mg/L | 139.6 | 17.3 | 138.54 | 169.6 | 214 | 126.2 | 600 (b) | Under | Under | Over |
| Total organic carbon | mg/L | 16.9 | 3.33 | 16.6 | 22.8 | 6.4 | 4.26 | ID | Under | ||
| BOD | mg/L | 54.28 | 45.58 | 42.53 | 140.77 | 4.6 | <2 | ID | Under | ||
| COD | mg/L | 57.67 | 17.22 | 55.75 | 88.72 | 33.3 | 28.5 | ID | Under | ||
| Turbidity | NTU | 50.93 | 40.46 | 40.74 | 127.79 | 7.2 | 5.84 | 5 (b) | Over | Over | Under |
| pH | - | 6.35 | 0.54 | 6.33 | 7.27 | 7.7 | 7.5 | 6.5–8.5 | Over | ||
| Parameter | ||||||
|---|---|---|---|---|---|---|
| PAHs (Benzo(a)pyrene) | As | Cd | Pb | BOD | COD | |
| μg/L | mg/L | mg/L | mg/L | mg/L | mg/L | |
| Feed | 0.811 | 0.011 | 0.0606 | 0.162 | 140.7 | 88.7 |
| TTA | 0.00010 | 0.0011 | 0.00085 | 0.0057 | 0.11 | 0.22 |
| TTB | 0.010 | 0.001155 | 0.00085 | 0.0057 | 0.11 | 0.22 |
| TTC | 5.1 × 10−6 | 0.00008 | 0.0059 | 0.0057 | 0.11 | 0.24 |
| TTD | 0.00051 | 0.00008 | 0.0059 | 0.0057 | 0.11 | 0.24 |
| ADWG value | 0.01 | 0.01 | 0.002 | 0.01 | ||
| Cost/Impact | RO-Based TT | Ozone–BAF-Based TT |
|---|---|---|
| Capital cost (millions) | AUD 216 | AUD 164 |
| Annual O&M cost (millions) | AUD 11 | AUD 8 |
| Annual environmental costs (millions) (1) | AUD 3 | AUD 1 |
| Total TBL NPV (millions) | AUD 482 | AUD 312 |
| Cost of water (including environmental costs, AUD/m3) | AUD 0.87 | AUD 0.56 |
| Power consumption (MWh/yr) | 16,000 | 4400 |
| Chemical consumption (dry tons/yr) | 1860 | 1770 |
| CO2 emissions (tons/yr) | 13,400 | 2900 |
| Other air emissions (tons/yr) (2) | 30 | 11 |
| TT Stage | Plant Capacity (ML/yr) | |||
|---|---|---|---|---|
| Cost Type | 6908 | 27,630 | 96,707 | |
| Capital | UF | 11.3 | 26.4 | 114.1 |
| UV | 1.5 | 1.6 | 4.5 | |
| UV/H2O2 | 5.2 | 18.0 | 43.3 | |
| O&M | UF | 2.5 | 5.2 | 18.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanciolo, P.; Sharma, A.K.; Navaratna, D.; Muthukumaran, S. Stormwater Treatment Using Natural and Engineered Options in an Urban Growth Area: A Case Study in the West of Melbourne. Water 2023, 15, 4047. https://doi.org/10.3390/w15234047
Sanciolo P, Sharma AK, Navaratna D, Muthukumaran S. Stormwater Treatment Using Natural and Engineered Options in an Urban Growth Area: A Case Study in the West of Melbourne. Water. 2023; 15(23):4047. https://doi.org/10.3390/w15234047
Chicago/Turabian StyleSanciolo, Peter, Ashok K. Sharma, Dimuth Navaratna, and Shobha Muthukumaran. 2023. "Stormwater Treatment Using Natural and Engineered Options in an Urban Growth Area: A Case Study in the West of Melbourne" Water 15, no. 23: 4047. https://doi.org/10.3390/w15234047
APA StyleSanciolo, P., Sharma, A. K., Navaratna, D., & Muthukumaran, S. (2023). Stormwater Treatment Using Natural and Engineered Options in an Urban Growth Area: A Case Study in the West of Melbourne. Water, 15(23), 4047. https://doi.org/10.3390/w15234047








