Age, Growth and Reproduction of Schizothorax pseudaksaiensis of the Turks River
Abstract
:1. Introduction
2. Materials and Methods
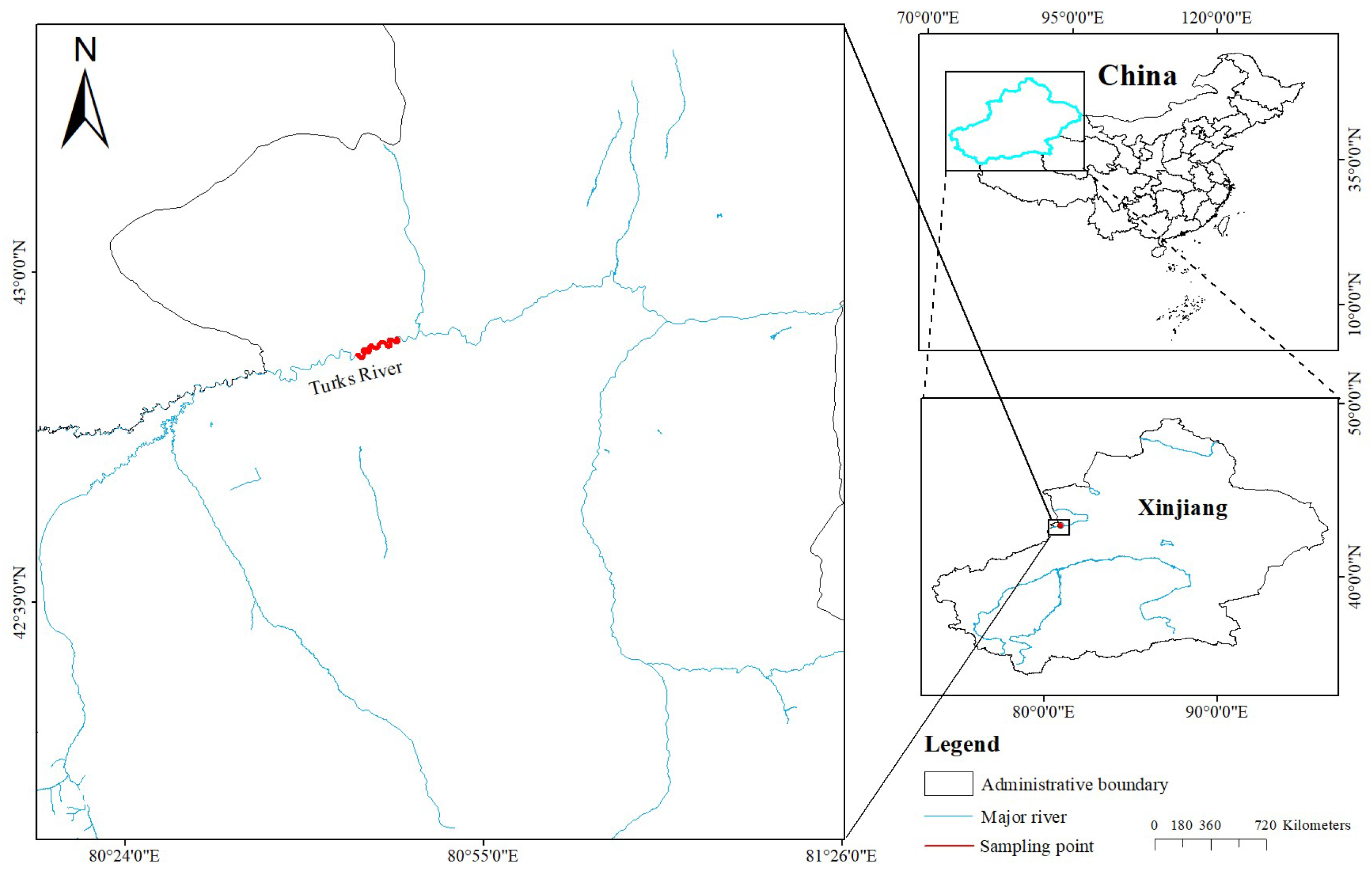
2.1. Sample Collection
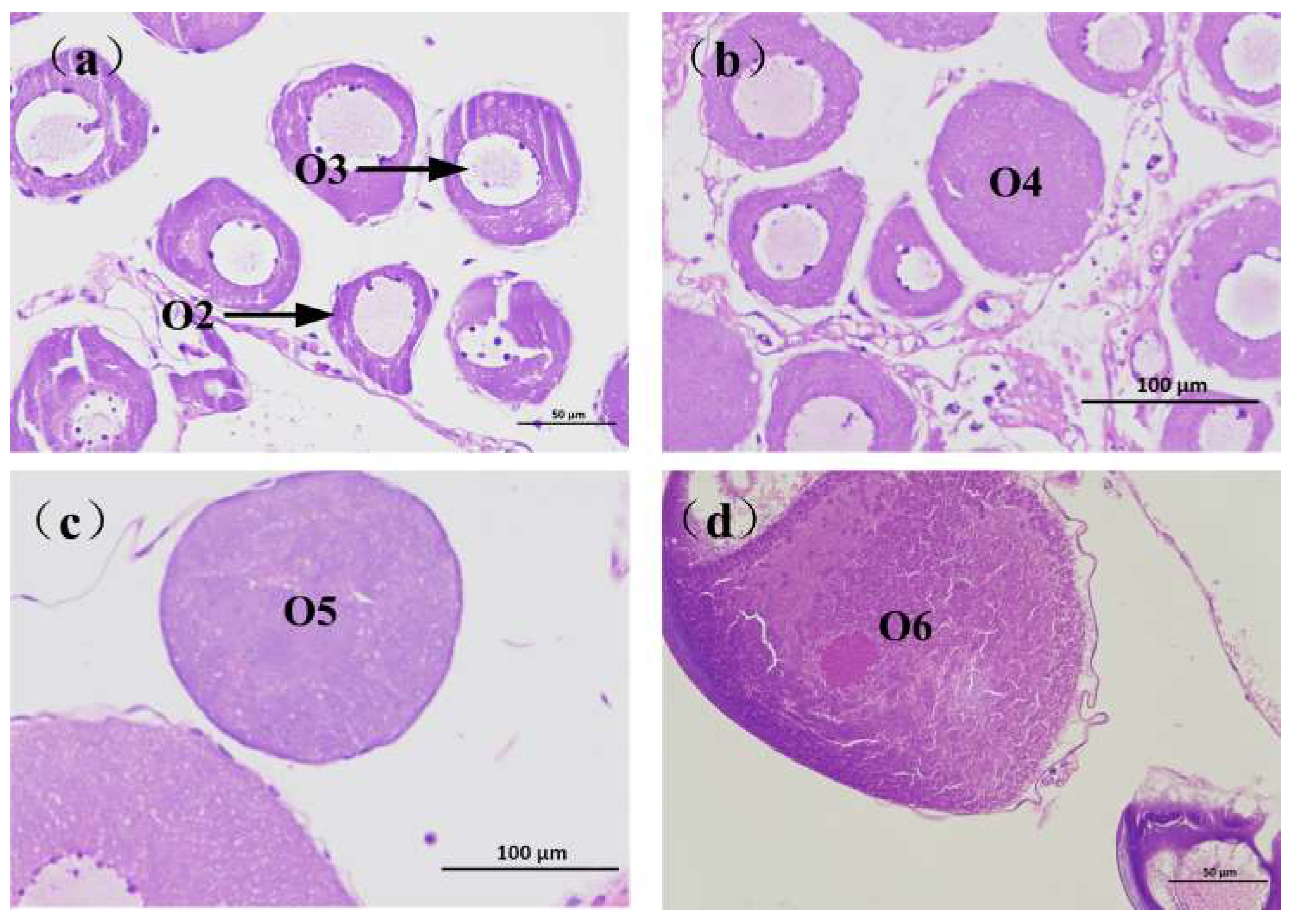
2.2. Otolith Characteristics
2.3. Age Determination
2.4. Growth Modeling
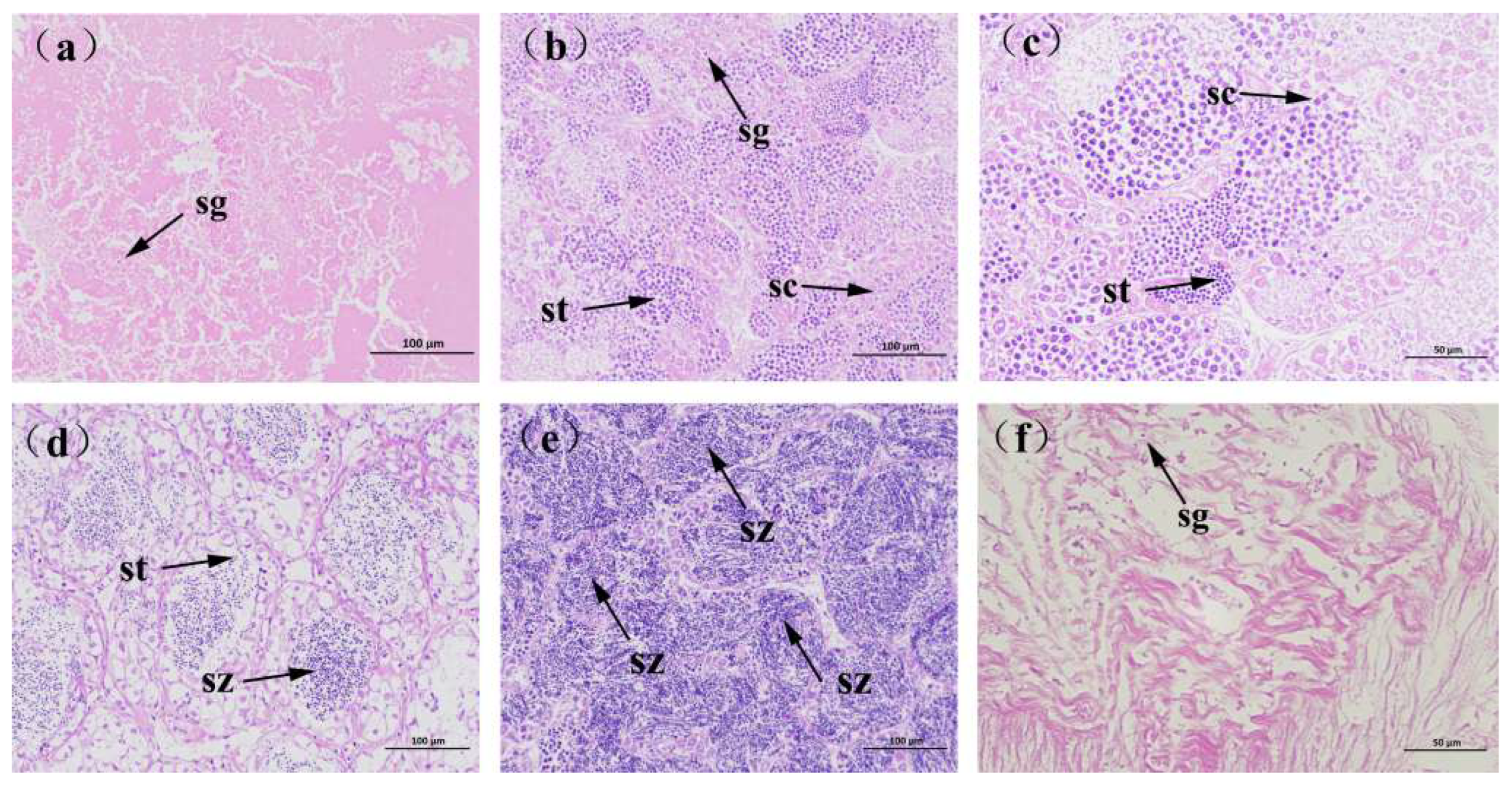
2.5. Reproductive Characteristics
3. Results
3.1. Size and Age Structure
3.2. Fish Growth
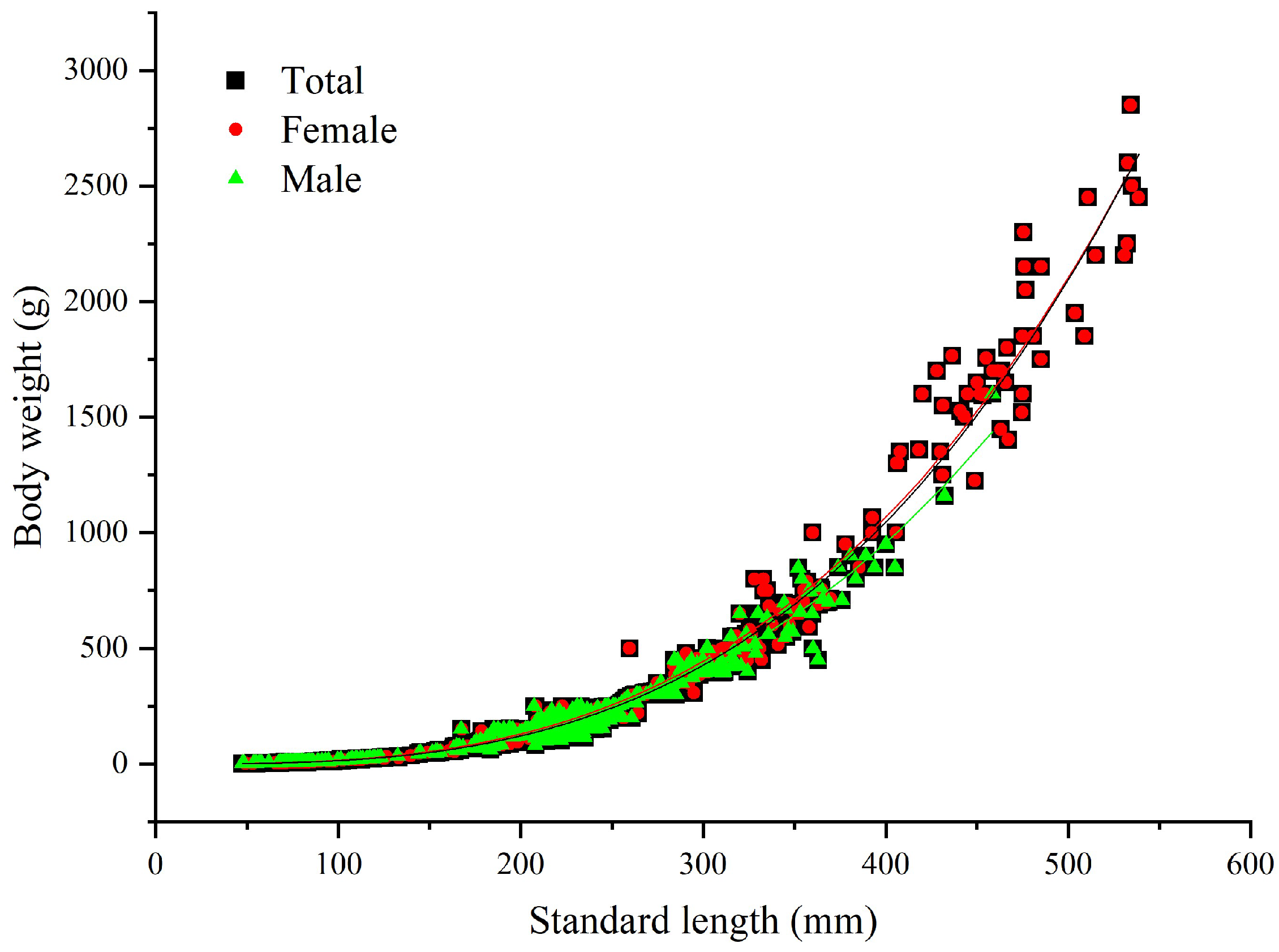
3.2.1. Length–Weight Relationship
3.2.2. Fulton’s Condition Index
3.2.3. Otolith Morphology
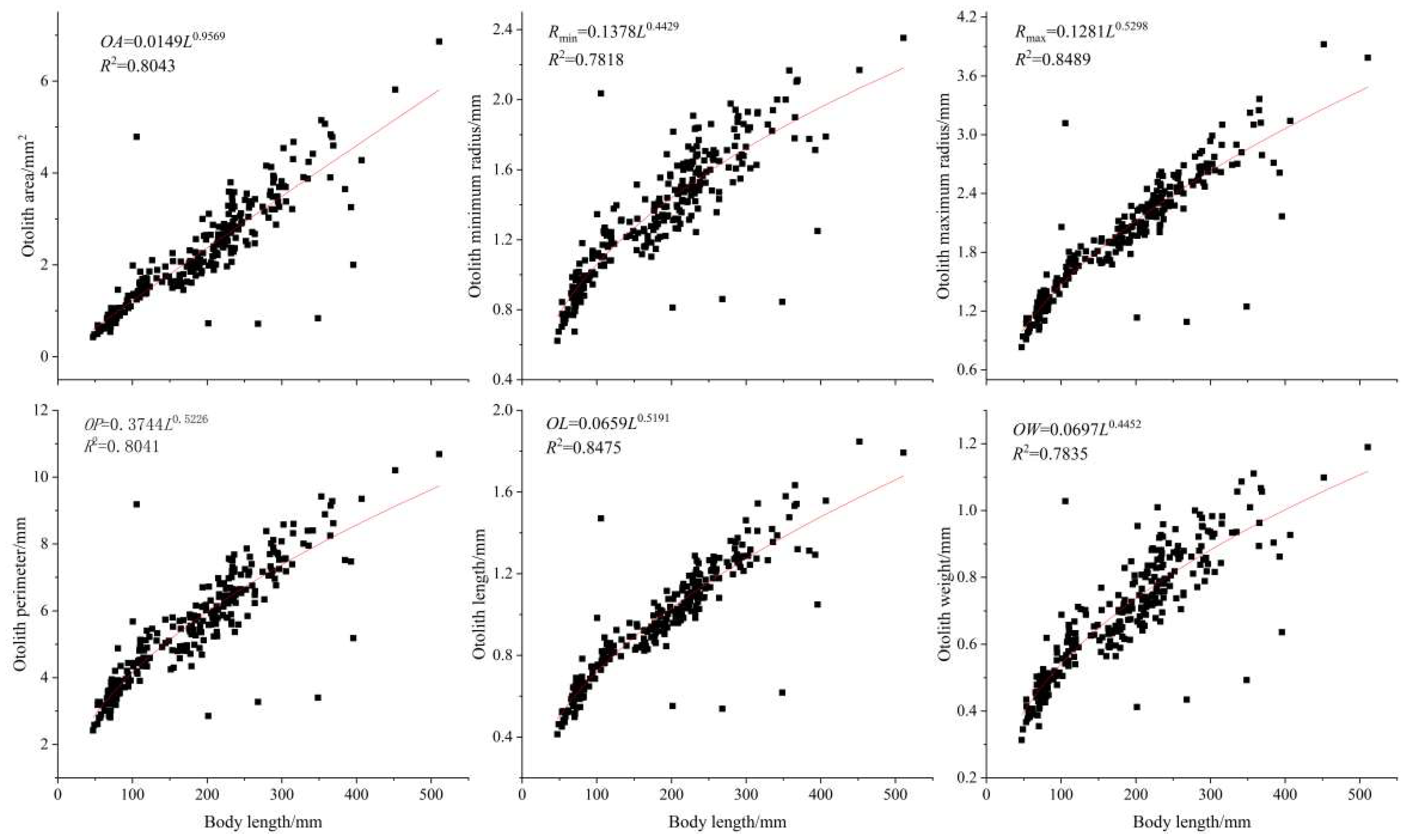
3.2.4. Relationship between the Morphology of Otoliths and Standard Length/Weight
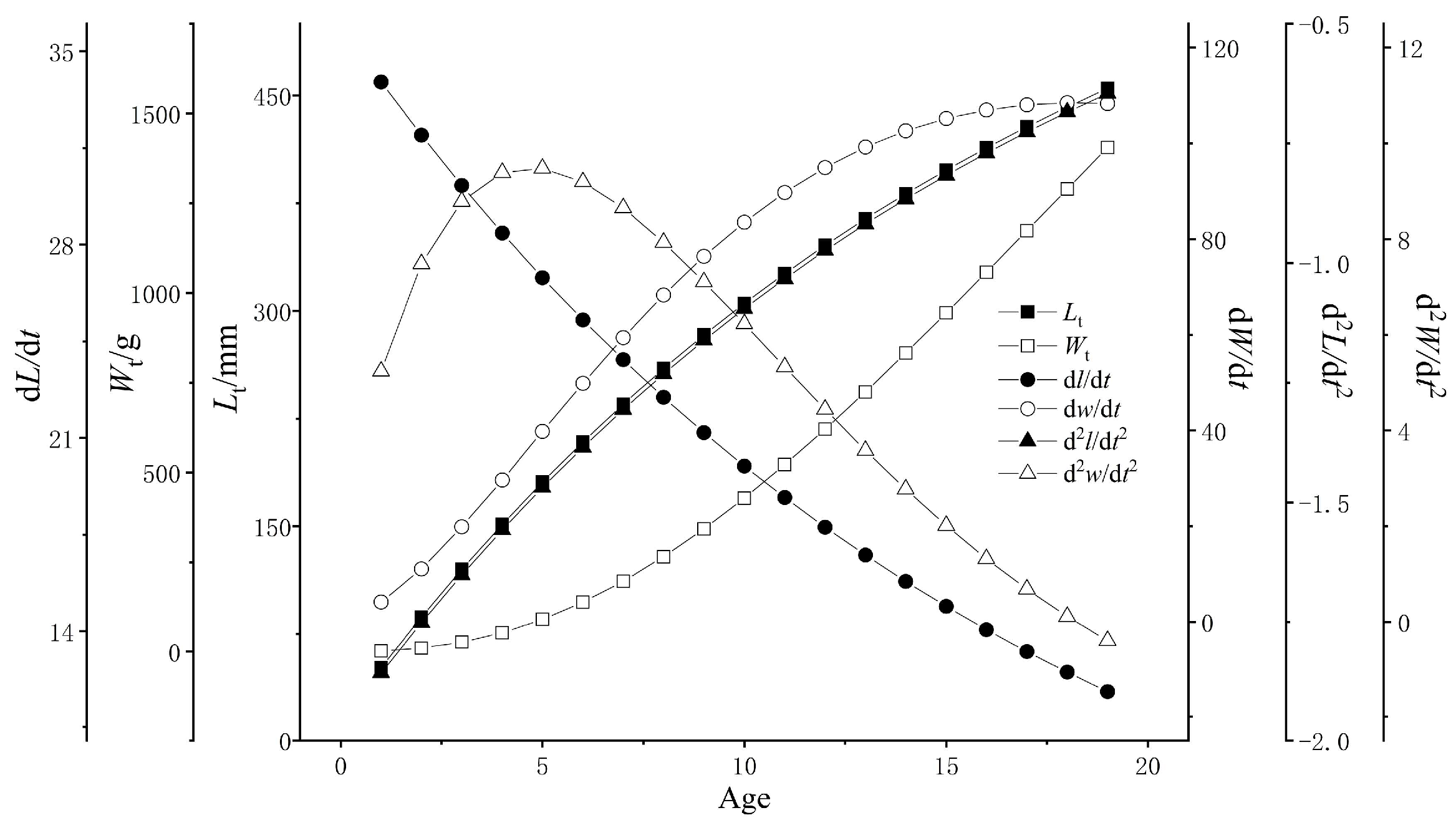
3.2.5. Growth Equation
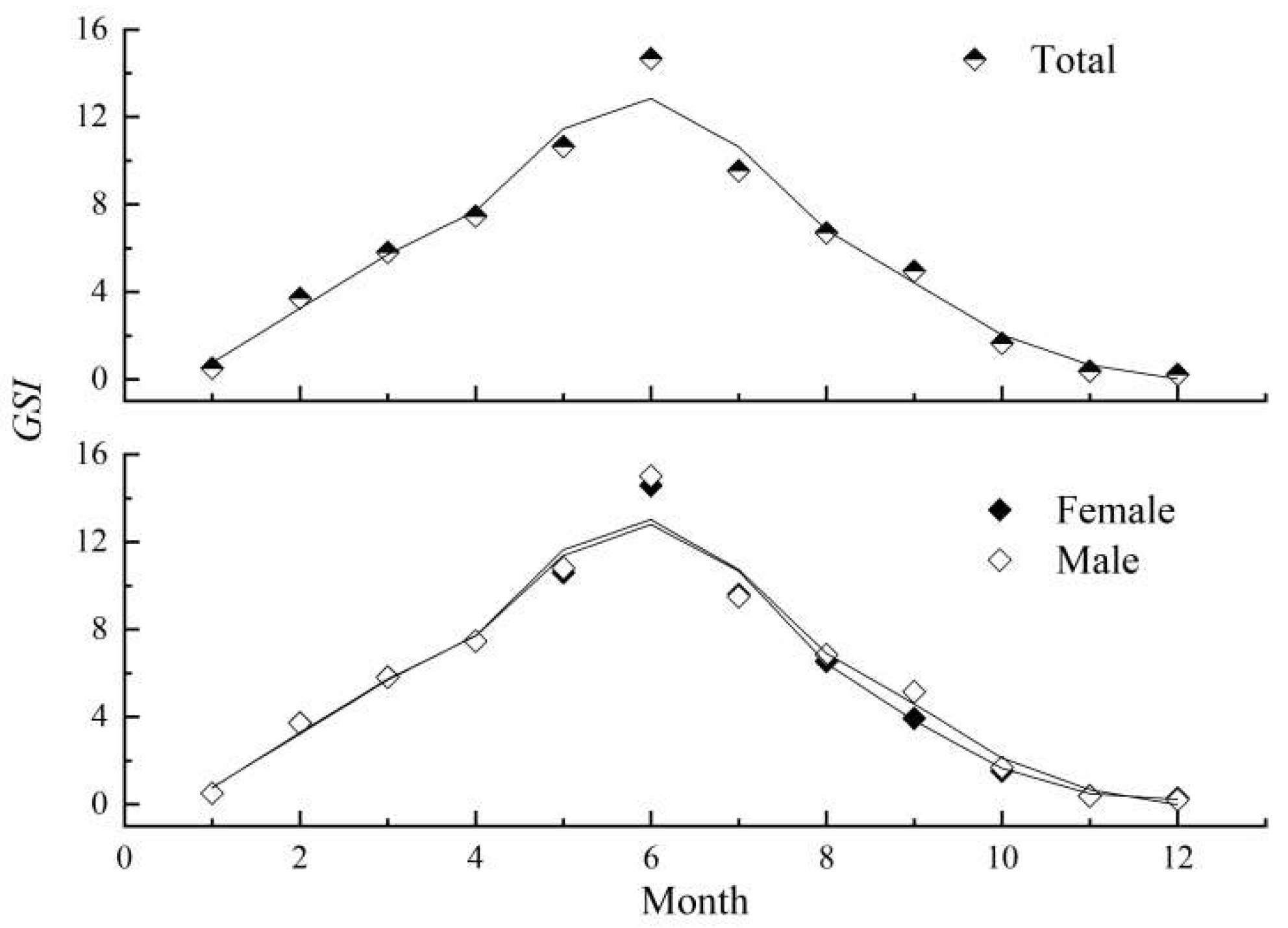
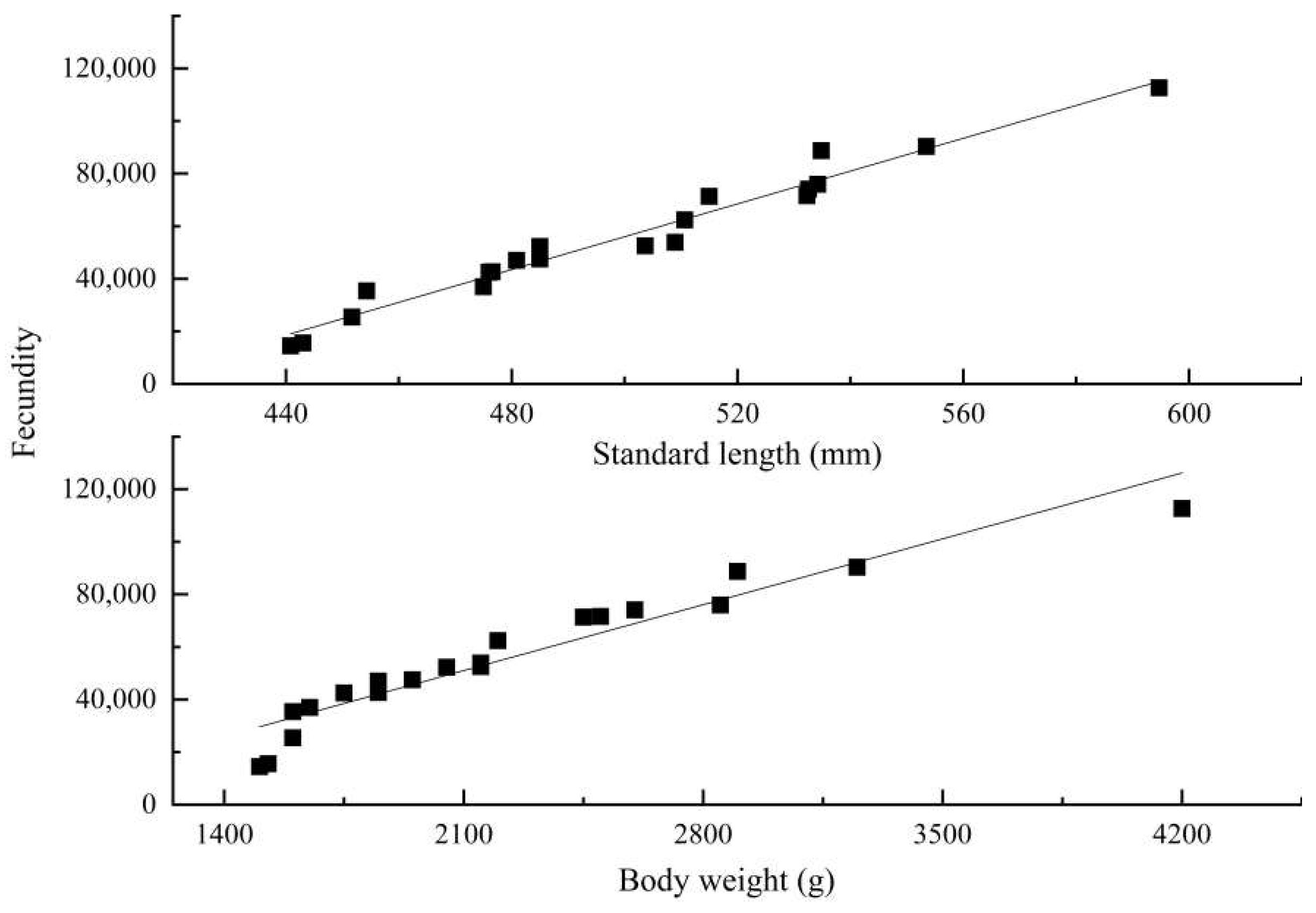
3.3. Reproductive Biology
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ren, M.L.; Guo, Y.; Zhang, Q.L.; Zhang, R.M.; Li, H.; A, D.K.; Cai, L.G.; Yong, W.D.; Ren, B.; Gao, H.; et al. Fish Resources and Fishery of Ili River; Heilongjiang Science and Technology Press: Harbin, China, 1998; pp. 13–310. ISBN 7-5388-3369-2. [Google Scholar]
- Guo, Y.; Zhang, R.M.; Cai, L.G. Xinjiang of Fishery; Xinjiang Science and Technology Press: Urumqi, China, 2012; ISBN 9787546615295. [Google Scholar]
- Notice on the Release of the List of Wildlife under Key Protection in Xinjiang Uygur Autonomous Region (Revised). Available online: https://www.xinjiang.gov.cn/xinjiang/c112543/202209/aaaf1dd3516e46d99086b7e378c8243b.shtml?cnName=%C3%A6%E2%80%9D%C2%BF%C3%A5%C2%BA%C5%93%C3%A4%C2%BB%C2%A4l (accessed on 21 September 2022).
- Campana, S.E.; Smoliński, S.; Black, B.A.; Morrongiello, J.R.; Alexandroff, S.J.; Andersson, C.; Bogstad, B.; Butler, P.G.; Denechaud, C.; Frank, D.C.; et al. Growth portfolios buffer climate-linked environmental change in marine systems. Ecology 2022, 104, e3918. [Google Scholar] [CrossRef] [PubMed]
- Zhan, B.Y. Fisheries Resource Assessment; China Agriculture Press: Beijing, China, 1995; pp. 8–14. [Google Scholar]
- Cai, L.G.; Meng, W.; Yang, T.Y.; Niu, J.G. Genetic differentiation of Ili Schizothorax (Schizothorax pseudaksaiensis) populations in Ili river basin. J. Aqua. Sci. 2014, 27, 25–29. [Google Scholar] [CrossRef]
- Cai, L.G.; Niu, J.G.; Tu, E.X.; Guo, Y. Annuli characteristics of the different ageing materials of Schizothorax (Racoma) pseudaksaiensis (Herzenstein). J. Water Ecol. 2011, 3, 78–81. [Google Scholar] [CrossRef]
- Cai, L.G.; Niu, J.G.; Zhang, B.P.; Tu, E.X.; Xie, C.G.; Tang, W.J.; Guo, Y. Observation on embryonic and larval development of Schizothorax pseudaksaiensis Herzenstein. Fresh. Fish. 2011, 41, 74–79. [Google Scholar] [CrossRef]
- Wang, C.X.; Liu, F.; Chen, S.A.; Yang, H.K.; Xie, C.X.; Wei, Q. Biological characteristics of Schizothorax pseudaksaiensis. Xinjiang Agric. Sci. 2022, 59, 2065–2072. [Google Scholar] [CrossRef]
- Li, Y.Y.; Zhang, H.L.; Zhang, F.H.; Qu, F.; Lai, X.Q. Analysis on the potentialof the available agriculture water resources in Manas river valley, Xinjiang. J. Natu. Reso. 2007, 22, 44–50. [Google Scholar] [CrossRef]
- Wang, Z.; Yan, Y.C.; Jiang, P.A.; Yan, A. Effects of different factors on soil moisture content of farmland in Manas river basin. Xinjiang Agric. Sci. 2013, 50, 1879–1886. [Google Scholar]
- Sui, F.G. Tectonic evolution and its relationship with hydrocarbon accumulation in the northwest margin of Junggar basin. Acta Geol. Sin. 2015, 89, 779–793. [Google Scholar]
- Li, L.; Ma, B.; Jin, X. Quantitative assessment on priority conservation of Schizothoracinae fishes in the middle Yarlung Zangbo River, Tibet. J. Fish. Sci. China 2019, 26, 914–924. [Google Scholar]
- Yang, H.Y.; Huang, D.M. A preliminary investigation on fish fauna and resources of the upper and middle Yarlung Zangbo River. J. Cent. China Norm. Univ. Nat. Sci. Ed. 2011, 45, 629–633. [Google Scholar] [CrossRef]
- Campana, S.E.; Annand, C.M.; Millan, J.I. Graphical and statistical methods for determining the consistency of age determinations. Trans. Am. Fish. Soc. 1995, 124, 131–138. [Google Scholar] [CrossRef]
- Aibibulla, T.; Abdushalih, N.; Kuerban, A. Evaluation of the status quo of animal and plants resources after reservoir construction in Turks river and protection measures. Anhui Agric. Sci. 2011, 39, 16688–16690. [Google Scholar] [CrossRef]
- Shen, J.Y. Influence of storm decline index on storm flood calculation of mountain flood gully in middle reaches of Turks River. Jilin Water Conserv. 2012, 9–11+18. [Google Scholar] [CrossRef]
- Zheng, Z.W. Spatial Variation of Hydraulic Conductivities of Yili-Kunes River Streambed Sediments and the Transformation Relationship between River and Groundwater. Master’s Thesis, Chang’an University, Xi’an, China, 2017. [Google Scholar]
- Wu, Y.F.; Wu, C.Z. The Fishes of the Qinghai-Xizang Plateau; Sichuan Science and Technology Press: Chengdu, China, 1991; pp. 1–599. ISBN 9787536421684. [Google Scholar]
- Yin, M.C. Fish Ecology; China Agriculture Press: Beijing, China, 1995; pp. 31–219. ISBN 7-109-03143-8. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multi-Model Inference: A Practical Information—Theoretical Approach; Springer: Berlin/Heidelberg, Germany, 2002. [Google Scholar]
- Liu, J.Y. Study on Biology and Population Dynamics of Schizothorax macropogon in the Yarlung Tsangpo River. Master’s Thesis, Tarim University, Alar, China, 2016. [Google Scholar]
- Liu, Y.C.; Liu, S.Y.; Liu, H.P. Values of eight structures as age determination of Ptychobarbus dipogon, Tibet Autonomous Region. Acta Hydrobiol. Sin. 2019, 43, 579–588. [Google Scholar] [CrossRef]
- Ma, B.S. Study on the Biology and Population Dynamics of Schizothorax oconnori. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2011. [Google Scholar] [CrossRef]
- Ren, B.; Ma, Y.W.; Tu, E.X.; Guo, Y.; Zhang, R.M.; A, B.D.; Au, Z.Z.; Liu, Y. The study on the biology of Triplophysa (Hedinichthys) yarkandensis (Day) in Akesu River. Chin. J. Fish. 2004, 17, 46–52. [Google Scholar]
- Li, X. Histoembryology of Aquatic Animals; China Agriculture Press: Beijing, China, 2006; ISBN 7-109-09815-X. [Google Scholar]
- Chen, S.A.; Ma, C.H.; Ding, H.P.; Zhou, X.J.; Xie, C.X. The reproductive biology of Triplophysa (Hedinichthys) yarkandensis (Day) in Tarim river. Acta Hydrobiol. Sin. 2013, 37, 810–816. [Google Scholar] [CrossRef]
- Wang, J.W. Spawning performance and development of oocytes in Gobiocypris rarus. Acta Hydrobiol. Sin. 1999, 23, 161–166. [Google Scholar]
- Chen, F.; Yuan, Y.J.; Wei, W.S.; Yu, S.L.; Fan, Z.A.; Zhang, R.B.; Zhang, T.W.; Shang, H.M. Variation and prediction trend of precipitation series for theTekes river basin during the last 236 years. J. Mt. Sci.-Engl. 2010, 28, 545–551. [Google Scholar] [CrossRef]
- Niu, Y.J.; Ren, D.Q.; Chen, S.A.; Cai, L.G.; Niu, J.G.; Xie, C.X. Growth characteristics of Gymnodipty chusdybowskii Kessler in three tributaries of the Ili river in Xinjiang, China. J. Hydroecol. 2015, 36, 59–65. [Google Scholar] [CrossRef]
- Borwn, M.E. Experimental Studies on Growth in the Physiology of Fishes; Academic Press: London, UK, 1957; pp. 391–400. [Google Scholar]
- Gislason, H.; Daan, N.; Rice, J.C.; Pope, J.G. Size, growth, temperature and the natural mortality of marine fish. Fish Fish. 2010, 11, 149–158. [Google Scholar] [CrossRef]
- Gauldie, R.W. Function, form and time-keeping properties of fish otoliths. Comp. Biochem. Physiol. A Physiol. 1988, 91, 395–402. [Google Scholar] [CrossRef]
- Battaglia, P.; Malara, D.; Romeo, T.; Andaloro, F. Relationships between otolith size and fish size in some mesopelagic and bathypelagic species from the Mediterranean Sea (Strait of Messina, Italy). Sci. Mar. 2010, 74, 605–612. [Google Scholar] [CrossRef]
- Souza, A.T.; Soukalová, K.; Děd, V.; Šmejkal, M.; Blabolil, P.; Říha, M.; Jůza, T.; Vašek, M.; Čech, M.; Peterka, J.; et al. Ontogenetic and interpopulation differences in otolith shape of the European perch (Perca fluviatilis). Fish. Res. 2020, 230, 105673. [Google Scholar] [CrossRef]
- Reñones, O.; Piñeiro, C.; Mas, X.; Goñi, R. Age and growth of the dusky grouper Epinephelus marginatus (Lowe 1834) in an exploited population of the western Mediterranean Sea. J. Fish Biol. 2007, 71, 346–362. [Google Scholar] [CrossRef]
- Larid, A.K. Postnatal growth of birds and mammals. Grozeth 1966, 30, 349–363. [Google Scholar]
- Shatunovskii, I.M.; Ruban, I.G. Ecological aspects of age-related dynamics of fish reproductive parameters. Russ. J. Ecol. 2009, 40, 339–347. [Google Scholar] [CrossRef]
- Zhou, X.J. Study on the Biology and Population Dynamics of Schizothorax waltoni. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2014. [Google Scholar] [CrossRef]
- Chen, S.A. Study on Population Ecology of Triplophysa yarkandensis (Day) in Tarim River. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2012. [Google Scholar]
- Branstetter, S.A. Age and growth estimates for blacktip, Carcharhinus limbatus, and Spinner, Carcharhinus brevipinna, sharks from the northwestern Gulf of Mexico. Copeia 1987, 1987, 964–974. [Google Scholar] [CrossRef]
- Musick, J.A. Ecology and conservation of long-lived marine animals. Am. Fish. Soc. Symp. 1999, 23, 1–10. [Google Scholar]
- Fei, J.H.; Shao, X.Y. Studies on the growth characteristics and morphological differences of fish in plateau lakes. Oceanol. Limnol. Sin. 2012, 43, 789–796. [Google Scholar] [CrossRef]
- Froyman, W.; Timmerman, D. Methods of assessing ovarian masses: International ovarian tumor analysis approach. Obstet. Gyn. Clin. N. Am. 2019, 46, 625–641. [Google Scholar] [CrossRef]
- Alves, M.M.; Leme Dos Santos, H.S.; Lopes, R.A.; Petenusci, S.O.; Haiyashi, C. Rhythm of development in the oocyte of the tilapia Oreochromis niloticus L. (Pisces: Cichlidae); a morphometric and histochemical study. Gegenbaurs Morphol. Jahrb. 1983, 129, 575–592. [Google Scholar] [PubMed]
- Li, H.J.; Liu, H.Y.; Fan, Q.X.; Xie, C.X. Individual fecundities of Glyptosternum maculatum. Chin. J. Appl. Environ. Biol. 2008, 14, 499–502. [Google Scholar] [CrossRef]
- Tang, Z.S.; Lin, Y.; Yang, H.Z.; Zhang, Y.D.; Chen, Z.; Huang, Y.; Peng, T.; Zhang, Y. Growth model of GIFT strain tilapia (Oreochromis niloticus). Guangdong Agric. Sci. 2011, 38, 104–107. [Google Scholar] [CrossRef]
- Shubhadeep, G.; Mohanraj, G.; Asokan, P.K.; Dhokia, H.K.; Zala, M.S.; Bhint, H.M. Trophodynamics and reproductive biology of Otolithoides biauritus (Cantor) landed by trawlers at Vanakbara, Diu along the west coast of india. Indian J. Fish. 2009, 56, 261–265. [Google Scholar] [CrossRef]
- Yamaguchi, A.; Kume, G. Reproductive biology of the fanray, Platyrhina sinensis, (Batoidea: Platyrhinidae) in Ariake Bay, Japan. Ichthyol Res. 2009, 56, 133–139. [Google Scholar] [CrossRef]
- Zhang, L.; Li, J.; Ding, Y.X.; Zheng, J.H. Study on Individual Fecundity of Pelteobagrus fulvidraco. Anhui Agri. Sci. Bull. 2012, 18, 164–167+171. [Google Scholar] [CrossRef]
- Yang, Y.; Li, S.Y.; Zhou, X.N.; Duan, Y.L. Biology and culture of Schizothoracinae. Water Conserv. Fish. 2003, 4, 22–23. [Google Scholar] [CrossRef]
- Zhou, C.P. Reproduce Biology of Schizopygopsis malacanthus baoxingensis. Master’s Thesis, Sichuan Agricultural University, Yaan, China, 2007. [Google Scholar]
- Le, P.Q. Chinese Zoology Teleoichthys Cypriniformes (Part II); Science Press: Beijing, China, 2000; pp. 277–305. ISBN 7030075757. [Google Scholar]
- Huo, B. Study on the Biology and Population Dynamics of Oxygymnocypris stewartii. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2014. [Google Scholar] [CrossRef]
- Wootton, R.J. Ecology of Teleost Fishes; Chapman and Hall: London, UK; New York, NY, USA, 1990; pp. 1–404. ISBN 978-94-010-6859-8. [Google Scholar]
- Chen, M.K.; Tong, H.Y.; Chen, Z.X.; Gao, J. On the growth rate of some commercial fishes in the Qiantang Jiang River. Acta Ecol. Sin. 1984, 4, 181–187. [Google Scholar]








| Time | Site | Number | Standard Length/mm | Body Weight/g | ||
|---|---|---|---|---|---|---|
| Range | Mean ± S.D. | Range | Mean ± S.D. | |||
| Apr. 2021 (Spring) | E 80°57′, N 42°57′—E 80°96′, N 42°95′ | 192 | 48.41~534.84 | 244.01 ± 94.04 | 1.59~2500.00 | 344.31 ± 452.61 |
| Jun. 2021 (Summer) | 196 | 47.30~532.60 | 187.04 ± 112.13 | 1.83~2600.00 | 228.12 ± 412.48 | |
| Oct. 2021 (Autumn) | 173 | 69.82~538.60 | 256.20 ± 110.45 | 5.00~2850.00 | 422.50 ± 515.65 | |
| Jan. 2022 (Winter) | 174 | 76.05~515.01 | 247.96 ± 77.07 | 8.84~2450.00 | 324.59 ± 349.15 | |
| Age | Turks River | ||||
|---|---|---|---|---|---|
| Number | Standard Length/mm | Body Weight/g | |||
| Range | Mean ± S.D. | Range | Mean ± S.D. | ||
| 1 | 23 | 47.30~70.44 | 61.03 ± 7.59 | 1.59~4.98 | 3.52 ± 1.09 |
| 2 | 84 | 53.82~97.57 | 79.348.16 ± 7.02 | 2.69~11.51 | 7.69 ± 1.86 |
| 3 | 38 | 86.30~133.12 | 110.39 ± 10.32 | 10.00~27.43 | 20.56 + 4.54 |
| 4 | 17 | 126.33~166.84 | 148.79 ± 10.54 | 30.10~58.11 | 46.97 ± 8.01 |
| 5 | 52 | 155.17~198.44 | 177.92 ± 9.11 | 59.30~150.00 | 83.61 ± 14.41 |
| 6 | 92 | 167.61~235.00 | 208.02 ± 13.37 | 81.82~200.00 | 131.37 ± 18.16 |
| 7 | 124 | 204.00~264.17 | 231.28 ± 11.55 | 137.82~250.00 | 192.99 ± 24.58 |
| 8 | 68 | 242.00~285.06 | 256.38 ± 10.01 | 237.47~350.00 | 270.04 ± 27.77 |
| 9 | 57 | 252.00~301.91 | 278.71 ± 11.07 | 211.42~501.98 | 336.63 ± 47.11 |
| 10 | 49 | 284.06~324.45 | 301.12 ± 11.80 | 306.54~550.00 | 423.04 ± 37.04 |
| 11 | 26 | 302.41~340.96 | 322.30 ± 9.14 | 450.00~650.00 | 522.03 ± 41.32 |
| 12 | 23 | 320.00~359.73 | 342.26 ± 10.80 | 548.42~800.00 | 652.01 ± 70.36 |
| 13 | 20 | 344.37~369.77 | 358.67 ± 7.30 | 449.94~848.00 | 721.74 ± 76.78 |
| 14 | 9 | 360.00~394.00 | 378.08 ± 11.90 | 500.00~1000.00 | 817.65 ± 142.81 |
| 15 | 7 | 378.00~406.82 | 497.22 ± 10.35 | 850.00~1300.00 | 1016.60 ± 141.39 |
| 16 | 2 | 406.00~408.00 | 407.00 ± 1.41 | 1300.00~1350.00 | 1325.00 ± 35.36 |
| 17 | 7 | 418.18~432.18 | 427.25 ± 5.75 | 1159.38~1700.00 | 1423.99 ± 197.10 |
| 18 | 7 | 436.38~451.70 | 445.11 ± 5.48 | 1224.95~1764.74 | 1552.97 ± 168.03 |
| 19 | 10 | 453.06~467.10 | 459.97 ± 4.88 | 1403.16~1755.68 | 1614.93 ± 113.78 |
| 20 | 4 | 466.50~475.00 | 472.78 ± 4.19 | 1520.36~1850.00 | 1692.59 ± 157.640 |
| 21 | 6 | 475.53~485.00 | 479.81 ± 4.45 | 1750.00~2300.00 | 2041.67 ± 205.95 |
| 22 | 4 | 503.67~515.01 | 509.58 ± 4.69 | 1850.00~2450.00 | 2112.50 ± 268.87 |
| 23 | 6 | 530.74~538.60 | 533.89 ± 2.73 | 2200.00~2850.00 | 2475.00 ± 238.22 |
| Total | 735 | 47.30~538.60 | 232.62 ± 103.32 | 1.59~2850.00 | 327.06 ± 440.90 |
| Parameter | Linear Equation | Logarithmic Equation | Power Equation | Exponential Equation |
|---|---|---|---|---|
| OA | −388.81959 | −326.00457 | −837.21834 | −727.98510 |
| Rmin | −988.28599 | −1010.62538 | −1188.16460 | −1100.23007 |
| Rmax | −818.08214 | −807.90184 | −1217.95391 | −1107.54899 |
| OP | −164.87986 | −150.30386 | −1141.61319 | −1065.05038 |
| OL | −1230.35127 | −1221.03375 | −1224.43525 | −1117.13393 |
| OW | −1363.38737 | −1387.88444 | −1195.11009 | −1104.32348 |
| Parameter | Linear Equation | Logarithmic Equation | Power Equation | Exponential Equation |
|---|---|---|---|---|
| OA | −376.26244 | −376.26244 | −887.08062 | −452.85771 |
| Rmin | −758.20313 | −1060.49461 | −1233.47752 | −860.31769 |
| Rmax | −520.14663 | −869.60714 | −1274.17292 | −806.49466 |
| OP | 72.37525 | −211.98641 | −1198.64829 | −806.26546 |
| OL | −930.36584 | −1274.79386 | −1273.24277 | −816.98914 |
| OW | −1130.29426 | −1434.92895 | −1238.52020 | −858.82629 |
| Population | Female (n = 350) | Male (n = 389) |
|---|---|---|
| Lt | 737.7 [1 − e−0.05(t + 0.59)] (R2 = 0.9924) | 667.0 [1 − e−0.05(t + 0.36)] (R2 = 0.9829) |
| Wt | 6853.3[1 − e−0.05(t + 0.59)]3.0436 (R2 = 0.9651) | 4447.2[1 − e−0.05(t + 0.36)]2.9759 (R2 = 0.9707) |
| dL/dt | 35.9 e−0.05(t + 0.59) | 36.7 e−0.05(t + 0.36) |
| dW/dt | 1015.8 e−0.05(t + 0.59) [1 − e−0.05(t + 0.59)]2.0436 | 725.3 e−0.05(t + 0.36) [1 − e−0.05(t + 0.36)]1.9759 |
| dL2/dt2 | −1.7 e−0.05(t + 0.59) | −2.0 e−0.05(t + 0.36) |
| dW2/dt2 | 49.5 e−0.05(t + 0.59) [1 − e−0.05(t + 0.59)]1.0436 [3.0436 e−0.05(t + 0.59) − 1] | 39.7 e−0.05(t + 0.36) [1 − e−0.05(t + 0.36)]0.9759 [2.9759 e−0.05(t + 0.36) − 1] |
| ti | 22.28 | 19.55 |
| Standard length/mm corresponding to ti | 520.17 | 507.26 |
| Body weight/g corresponding to ti | 2366.32 | 1943.33 |
| Parameter | Linear Equation | Logarithmic Equation | Power Equation | Exponential Equation |
|---|---|---|---|---|
| L | −63.14150848 | 340.538227 | 340.0967467 | −66.56823424 |
| W | −49.30208045 | 357.2521667 | 339.9725195 | −57.66593688 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Yao, N.; Xia, L.; Wang, X.; Song, Y.; Serekbol, G.; Zi, F.; Lin, X.; Yan, J.; Chen, S. Age, Growth and Reproduction of Schizothorax pseudaksaiensis of the Turks River. Water 2023, 15, 4044. https://doi.org/10.3390/w15234044
Wang C, Yao N, Xia L, Wang X, Song Y, Serekbol G, Zi F, Lin X, Yan J, Chen S. Age, Growth and Reproduction of Schizothorax pseudaksaiensis of the Turks River. Water. 2023; 15(23):4044. https://doi.org/10.3390/w15234044
Chicago/Turabian StyleWang, Chengxin, Na Yao, Liwei Xia, Xinyue Wang, Yong Song, Gulden Serekbol, Fangze Zi, Xuyuan Lin, Jin Yan, and Shengao Chen. 2023. "Age, Growth and Reproduction of Schizothorax pseudaksaiensis of the Turks River" Water 15, no. 23: 4044. https://doi.org/10.3390/w15234044
APA StyleWang, C., Yao, N., Xia, L., Wang, X., Song, Y., Serekbol, G., Zi, F., Lin, X., Yan, J., & Chen, S. (2023). Age, Growth and Reproduction of Schizothorax pseudaksaiensis of the Turks River. Water, 15(23), 4044. https://doi.org/10.3390/w15234044






