Revealing the Extent of Pesticide Runoff to the Surface Water in Agricultural Watersheds
Abstract
:1. Introduction
2. Materials and Methods
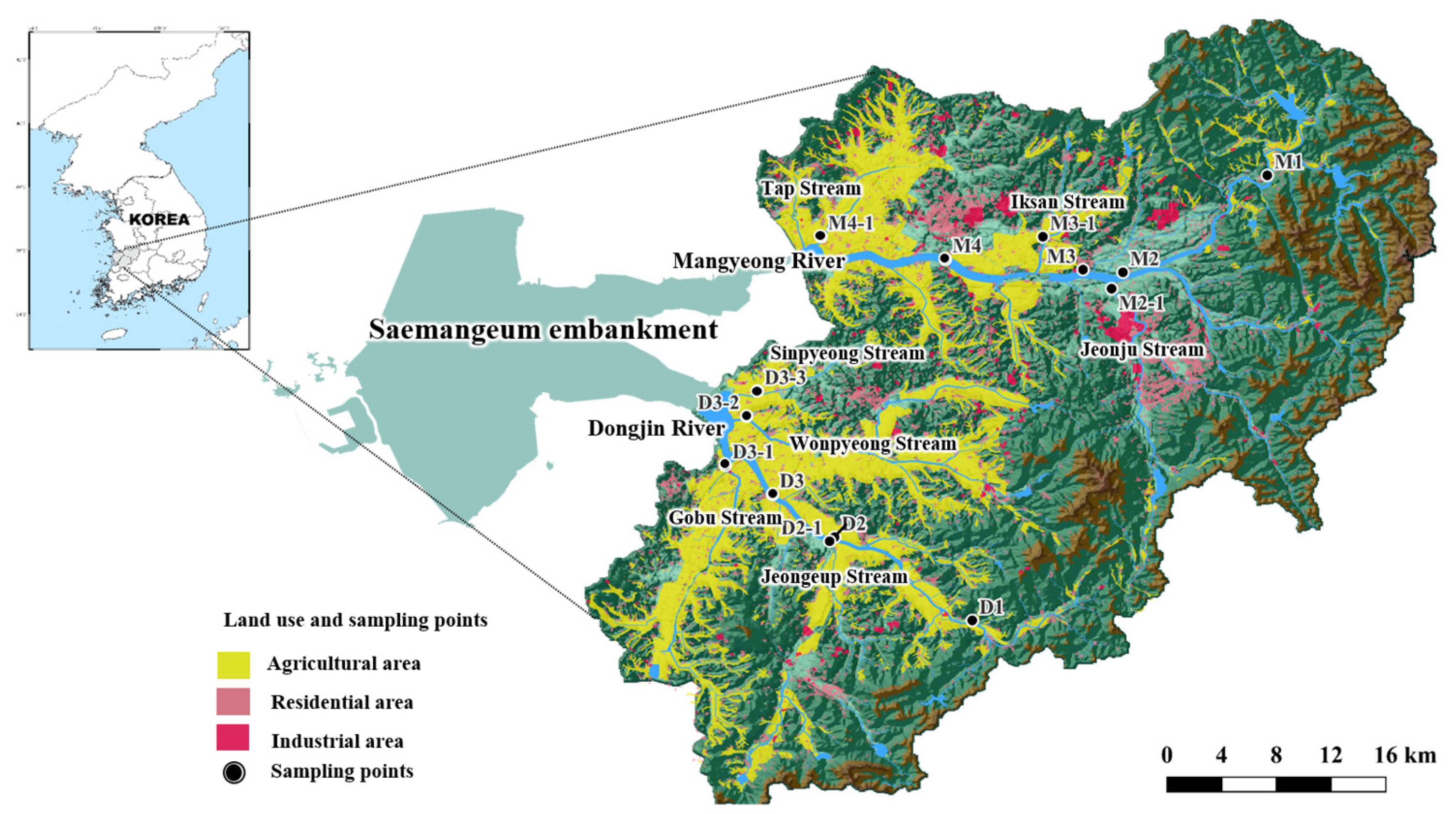
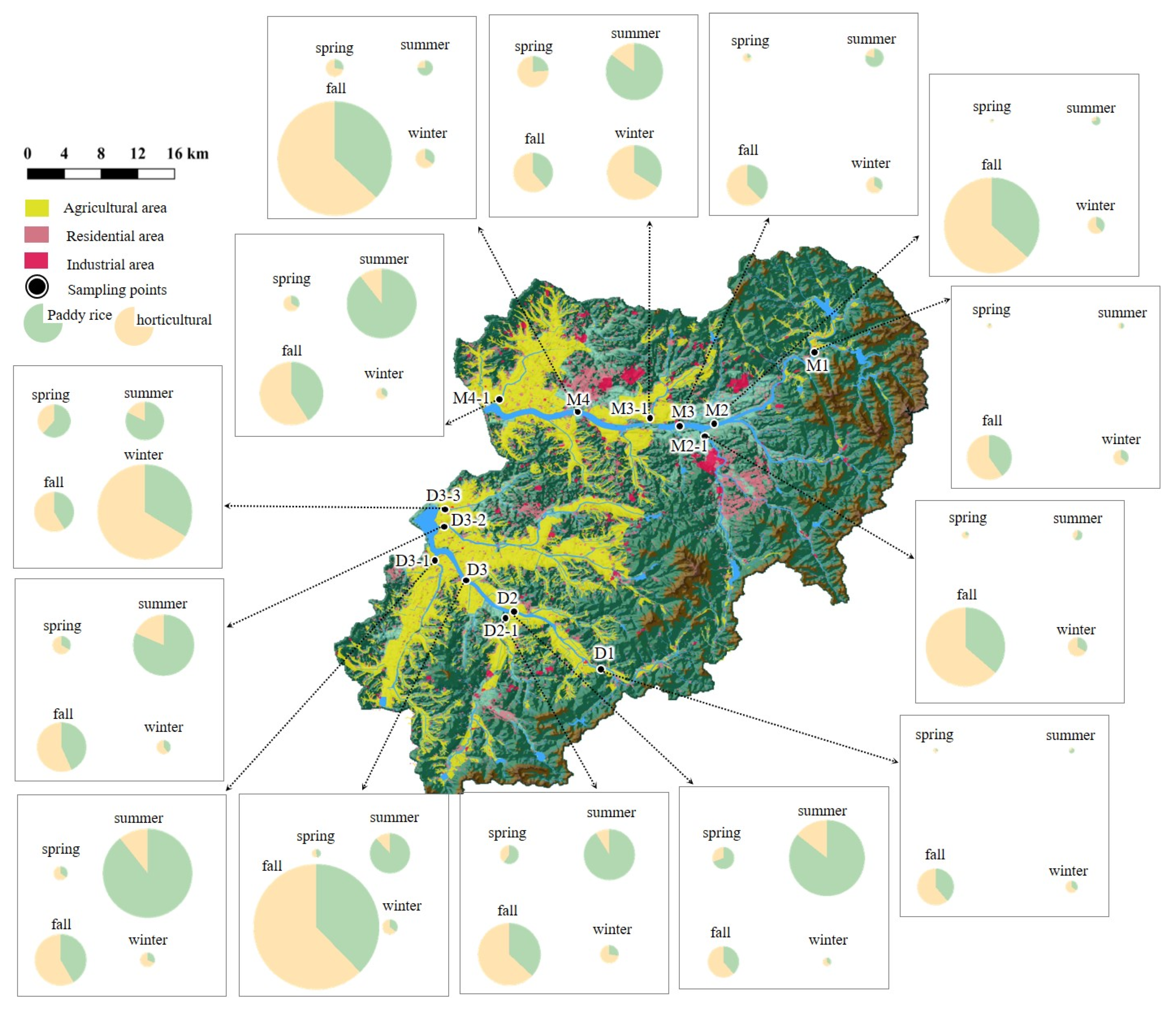
2.1. Study Area
2.2. Sample Collection and Preservation
2.3. Sample Analysis
2.4. Quality Assurance and Control
2.5. Environmental Risk Assessment of a Mixture of Pesticides
3. Results and Discussion
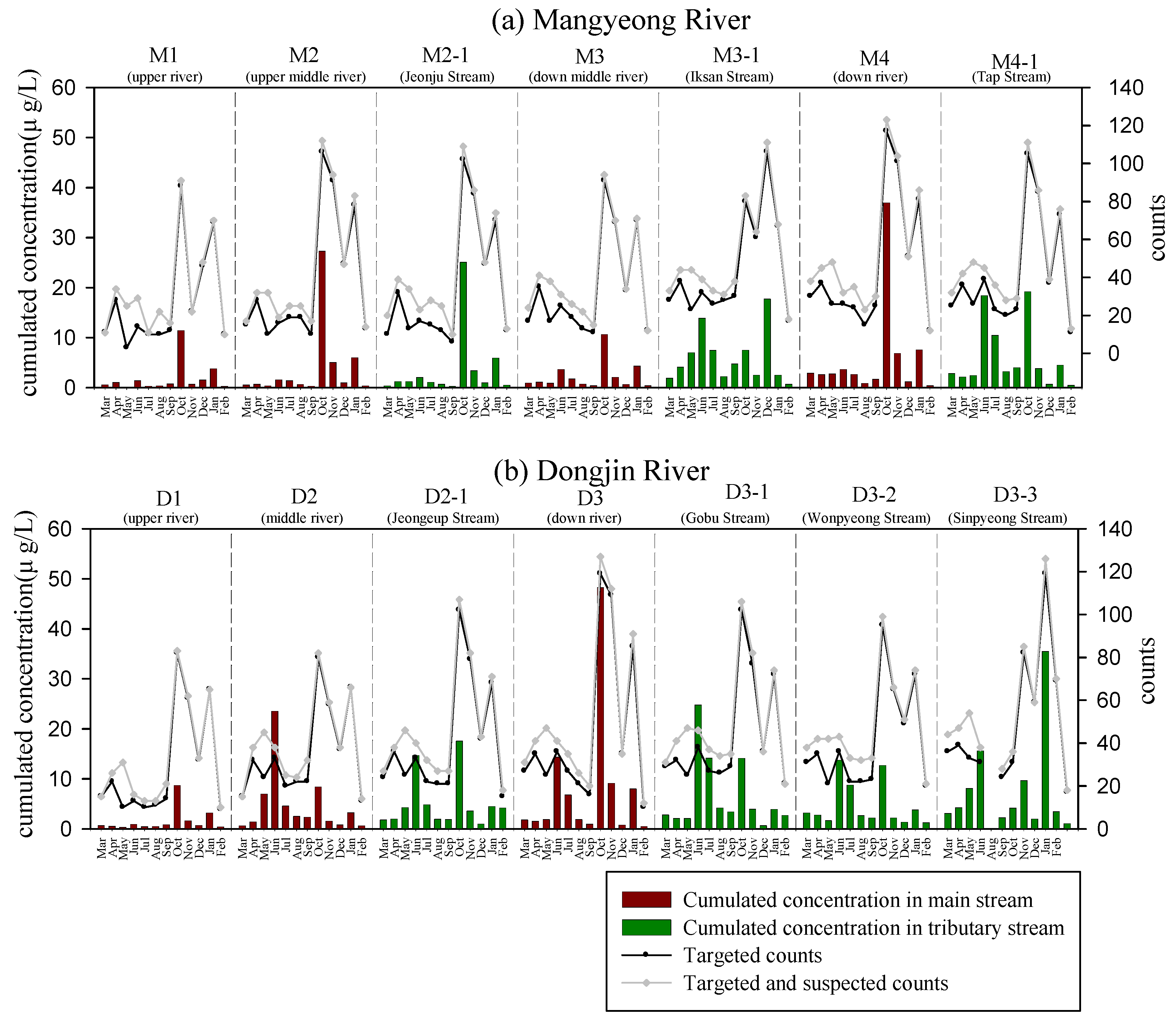
3.1. Occurrence of Pesticides
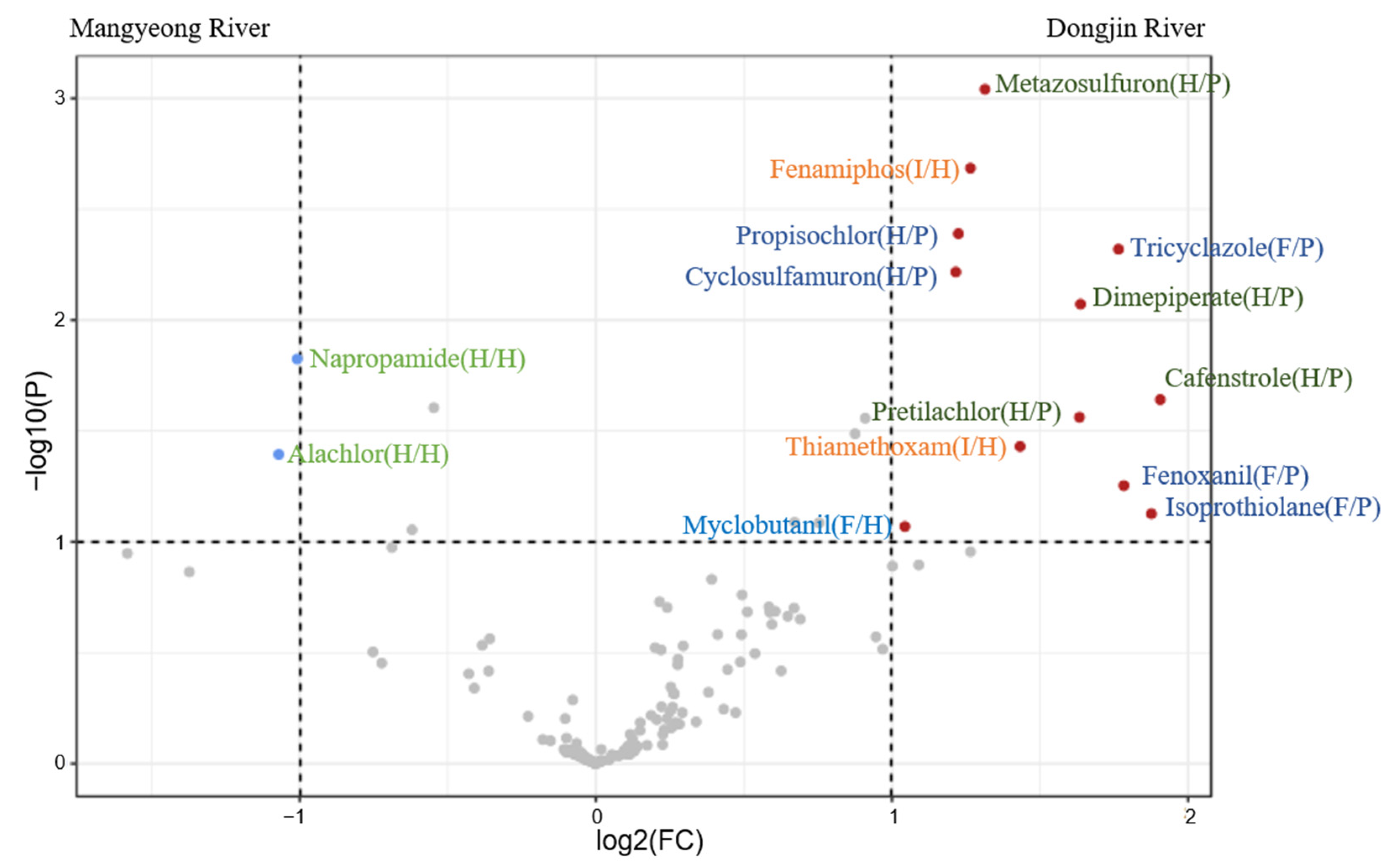
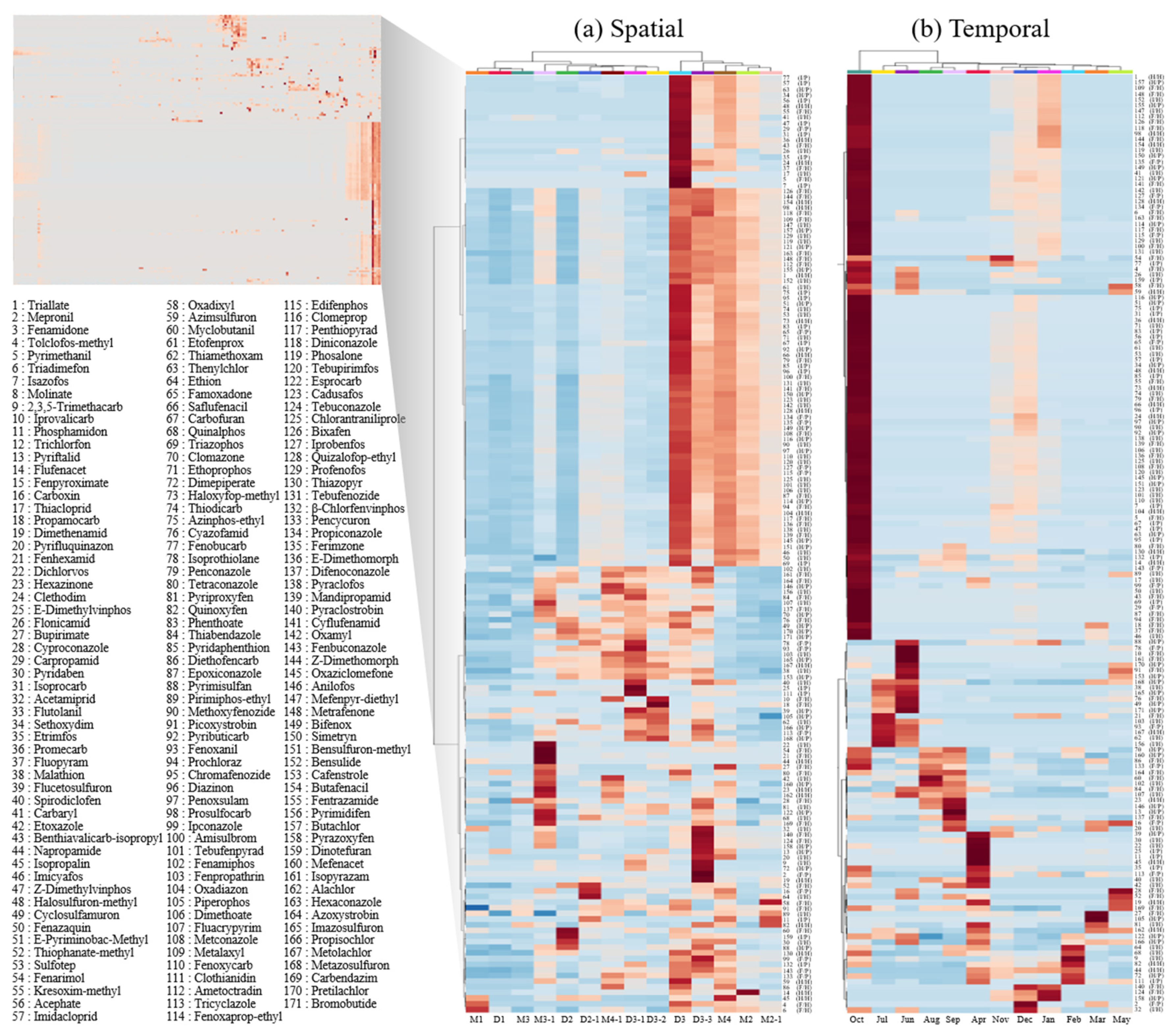
3.2. Tracking Pesticide Sources
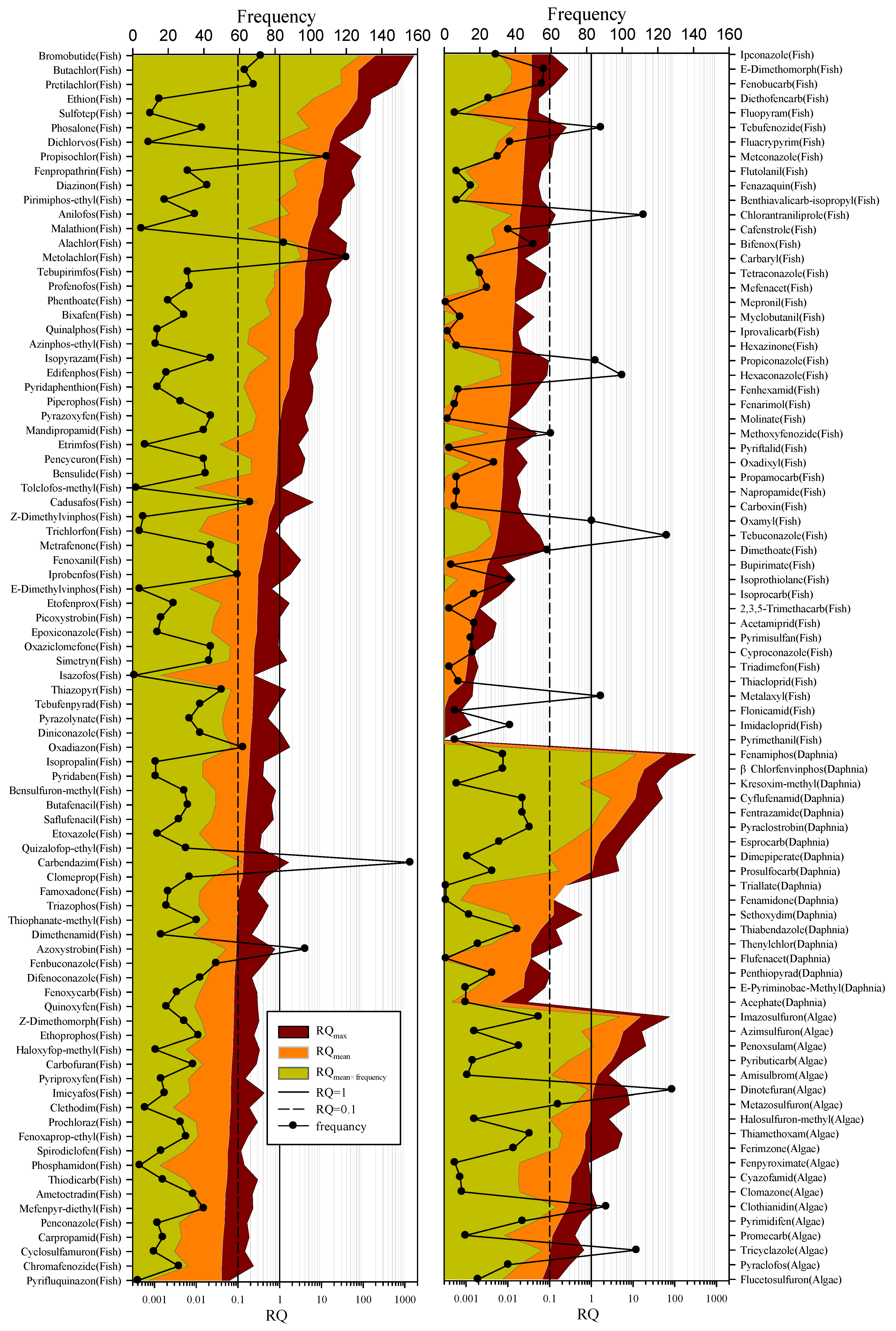
3.3. Environmental Risk Assessment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ndolo, D.; Njuguna, E.; Adetunji, C.O.; Harbor, C.; Rowe, A.; Den Breeyen, A.; Sangeetha, J.; Singh, G.; Szewczyk, B.; Anjorin, T.S.; et al. Research and development of biopesticides: Challenges and prospects. Outlooks Pest Manag. 2019, 30, 267–276. [Google Scholar] [CrossRef]
- Pathak, V.M.; Verma, V.K.; Rawat, B.S.; Kaur, B.; Babu, N.; Sharma, A.; Dewali, S.; Yadav, M.; Kumari, R.; Singh, S.; et al. Current status of pesticide effects on environment, human health and it’s eco-friendly management as bioremediation: A comprehensive review. Front. Microbiol. 2022, 13, 962619. [Google Scholar] [CrossRef] [PubMed]
- Pironti, C.; Ricciardi, M.; Proto, A.; Bianco, P.M.; Montano, L.; Motta, O. Endocrine-Disrupting Compounds: An Overview on Their Occurrence in the Aquatic Environment and Human Exposure. Water 2021, 13, 1347. [Google Scholar] [CrossRef]
- Shefali, G.; Kumar, R.; Sankhla, M.S.; Kumar, R.; Sonone, S.S. Impact of pesticide toxicity in aquatic environment. Biointerface Res. Appl. Chem. 2021, 11, 10131–10140. [Google Scholar]
- Sishu, F.K.; Tilahun, S.A.; Schmitter, P.; Assefa, G.; Steenhuis, T.S. Pesticide Contamination of Surface and Groundwater in an Ethiopian Highlands’ Watershed. Water 2022, 14, 3446. [Google Scholar] [CrossRef]
- Im, S.; Park, D. Standard safety policy: A retrospect of the Korean chicken egg crisis in 2017. J. Consum. Prot. Food Saf. 2019, 14, 341–348. [Google Scholar] [CrossRef]
- Rajmohan, K.S.; Chandrasekaran, R.; Varjani, S. A Review on Occurrence of Pesticides in Environment and Current Technologies for Their Remediation and Management. Indian J. Microbiol. 2020, 60, 125–138. [Google Scholar] [CrossRef]
- Hertz-Picciotto, I.; Sass, J.B.; Engel, S.; Bennett, D.H.; Bradman, A.; Eskenazi, B.; Lanphear, B.; Whyatt, R. Organophosphate Exposures during Pregnancy and Child Neurodevelopment: Recommendations for Essential Policy Reforms. PLoS Med. 2018, 15, e1002671. [Google Scholar] [CrossRef]
- Borsuah, J.F.; Messer, T.L.; Snow, D.D.; Comfort, S.D.; Mittelstet, A.R. Literature Review: Global Neonicotinoid Insecticide Occurrence in Aquatic Environments. Water 2020, 12, 3388. [Google Scholar] [CrossRef]
- Zhou, Y.; Wu, J.; Wang, B.; Duan, L.; Zhang, Y.; Zhao, W.; Wang, F.; Sui, Q.; Chen, Z.; Xu, D.; et al. Occurrence, source and ecotoxicological risk assessment of pesticides in surface water of Wujin District (northwest of Taihu Lake), China. Environ. Pollut. 2020, 265, 114953. [Google Scholar] [CrossRef]
- Kaushal, J.; Khatri, M.; Arya, S.K. A Treatise on Organophosphate Pesticide Pollution: Current Strategies and Advancements in Their Environmental Degradation and Elimination. Ecotoxicol. Environ. Saf. 2021, 207, 111483. [Google Scholar] [CrossRef] [PubMed]
- Olisah, C.; Rubidge, G.; Human, L.R.D.; Adams, J.B. Organophosphate pesticides in South African eutrophic estuaries: Spatial distribution, seasonal variation, and ecological risk assessment. Environ. Pollut. 2022, 306, 119446. [Google Scholar] [CrossRef]
- Aleseyyed, S.B.; Rafati, L.; Heidarimoghadam, R.; Khodabakhshi, M.; Zafarmirmohammadi, S.A.; Heidari, S. Investigation of residual concentration of organochlorine, organophosphorus, and carbamate pesticides in urban drinking water networks of Hamadan Province, Iran. Avicenna J. Environ. Health Eng. 2021, 8, 9–16. [Google Scholar] [CrossRef]
- Mali, H.; Shah, C.; Raghunandan, B.H.; Prajapati, A.S.; Patel, D.H.; Trivedi, U.; Subramanian, R.B. Organophosphate pesticides an emerging environmental contaminant: Pollution, toxicity, bioremediation progress, and remaining challenges. J. Environ. Sci. 2023, 127, 234–250. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kumar, V.; Shahzad, B.; Tanveer, M.; Sidhu, G.P.S.; Handa, N.; Kohli, S.K.; Yadav, P.; Bali, A.S.; Parihar, R.D. Worldwide pesticide usage and its impacts on ecosystem. SN Appl. Sci. 2019, 1, 1446. [Google Scholar] [CrossRef]
- Ore, O.T.; Adeola, A.O.; Bayode, A.A.; Adedipe, D.T.; Nomngongo, P.N. Organophosphate pesticide residues in environmental and biological matrices: Occurrence, distribution and potential remedial approaches. Environ. Chem. Ecotoxicol. 2023, 5, 9–23. [Google Scholar] [CrossRef]
- FAOSTAT. Pesticides. Food and Agriculture Organization, Rome, Italy. Available online: http://www.fao.org/faostat/en/#data/RP (accessed on 10 May 2023).
- Corrêa, M.G.; Barbosa, S.C.; dos Santos, G.B.; Collares, G.L.; Primel, E.G. Assessment of Pesticides in the Chasqueiro Irrigation District, Southern Brazil, an Agricultural Area of International Importance. Water Air Soil Pollut. 2022, 233, 517. [Google Scholar] [CrossRef]
- Papadakis, E.-N.; Tsaboula, A.; Kotopoulou, A.; Kintzikoglou, K.; Vryzas, Z.; Papadopoulou-Mourkidou, E. Pesticides in the surface waters of Lake Vistonis Basin, Greece: Occurrence and environmental risk assessment. Sci. Total Environ. 2015, 536, 793–802. [Google Scholar] [CrossRef]
- Syafrudin, M.; Kristanti, R.A.; Yuniarto, A.; Hadibarata, T.; Rhee, J.; Al-onazi, W.A.; Algarni, T.S.; Almarri, A.H.; Al-Mohaimeed, A.M. Pesticides in Drinking Water—A Review. Int. J. Environ. Res. Public Health 2021, 18, 468. [Google Scholar] [CrossRef]
- Kruć-Fijałkowska, R.; Dragon, K.; Drożdżyński, D.; Górski, J. Seasonal variation of pesticides in surface water and drinking water wells in the annual cycle in western Poland, and potential health risk assessment. Sci. Rep. 2022, 12, 3317. [Google Scholar] [CrossRef]
- Pietrzak, D.; Kania, J.; Malina, G.; Kmiecik, E.; Wątor, K. Pesticides from the EU First and Second Watch Lists in the Water Environment. CLEAN—Soil Air Water 2019, 47, 1800376. [Google Scholar] [CrossRef]
- Carazo-Rojas, E.; Pérez-Rojas, G.; Pérez-Villanueva, M.; Chinchilla-Soto, C.; Chin-Pampillo, J.S.; Aguilar-Mora, P.; Alpízar-Marín, M.; Masís-Mora, M.; Rodríguez-Rodríguez, C.E.; Vryzas, Z. Pesticide monitoring and ecotoxicological risk assessment in surface water bodies and sediments of a tropical agro-ecosystem. Environ. Pollut. 2018, 241, 800–809. [Google Scholar] [CrossRef]
- Climent, M.J.; Herrero-Hernández, E.; Sánchez-Martín, M.J.; Rodríguez Cruz, M.S.; Pedreros, P.; Urrutia, R. Residues of pesticides and some metabolites in dissolved and particulate phase in surface stream water of Cachapoal River basin, central Chile. Environ. Pollut. 2019, 251, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Kapsi, M.; Tsoutsi, C.; Paschalidou, A.; Albanis, T. Environmental monitoring and risk assessment of pesticide residues in surface waters of the Louros River (N.W. Greece). Sci. Total Environ. 2019, 650, 2188–2198. [Google Scholar] [CrossRef] [PubMed]
- Kamata, M.; Matsui, Y.; Asami, M. National trends in pesticides in drinking water and water sources in Japan. Sci. Tot. Environ. 2020, 744, 140930. [Google Scholar] [CrossRef]
- Li, W.; Xin, S.; Deng, W.; Wang, B.; Liu, X.; Yuan, Y.; Wang, S. Occurrence, spatiotemporal distribution patterns, partitioning and risk assessments of multiple pesticide residues in typical estuarine water environments in eastern China. Water Res. 2023, 245, 120570. [Google Scholar] [CrossRef]
- Sun, X.; Liu, M.; Meng, J.; Wang, L.; Chen, X.; Peng, S.; Rong, X.; Wang, L. Residue level, occurrence characteristics and ecological risk of pesticides in typical farmland-river interlaced area of Baiyang Lake upstream, China. Sci. Rep. 2022, 12, 12049. [Google Scholar] [CrossRef] [PubMed]
- Togola, A.; Baran, N.; Coureau, C. Advantages of online SPE coupled with UPLC/MS/MS for determining the fate of pesticides and pharmaceutical compounds. Anal. Bioanal. Chem. 2014, 406, 1181–1191. [Google Scholar] [CrossRef] [PubMed]
- Sang, D.; Cimetiere, N.; Giraudet, S.; Tan, R.; Wolbert, D.; Le Cloirec, P. Online SPE-UPLC-MS/MS for herbicides and pharmaceuticals compounds’ determination in water environment: A case study in France and Cambodia. Environ. Adv. 2022, 8, 100212. [Google Scholar] [CrossRef]
- Courant, F.; Antignac, J.-P.; Dervilly-Pinel, G.; Le Bizec, B. Basics of Mass Spectrometry Based Metabolomics. Proteomics 2014, 14, 2369–2388. [Google Scholar] [CrossRef]
- Brock, T.C. Priorities to improve the ecological risk assessment and management for pesticides in surface water. Integr. Environ. Assess. Manag. 2013, 9, e64–e74. [Google Scholar] [CrossRef] [PubMed]
- Jeon, D.; Huh, Y.; Kim, B.-K.; Yoo, E.-J.; Lee, H.; Chung, H.M.; Choi, J.-W. Simultaneous and Rapid Analysis of Multi-pesticide Residues using On-line SPE-LC-Orbitrap Mass Spectrometry. J. Environ. Anal. Health Toxicol. 2019, 22, 268–276. [Google Scholar] [CrossRef]
- Lee, J.; Shin, Y.; Lee, J.; Lee, J.; Kim, B.J.; Kim, J.-H. Simultaneous analysis of 310 pesticide multiresidues using UHPLC-MS/MS in brown rice, orange, and spinach. Chemosphere 2018, 207, 519–526. [Google Scholar] [CrossRef] [PubMed]
- EMEA. Guideline on the Environmental Risk Assessment of Medicinal Products for Human Use; EMEA/CHMP/SWP/4447/00; The European Agency for the Evaluation of Medicinal Products, Committee for Medical Products for Human Use: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Heys, K.A.; Shore, R.F.; Pereira, M.G.; Jones, K.C.; Martin, F.L. Risk assessment of environmental mixture effects. RSC Adv. 2016, 6, 47844–47857. [Google Scholar] [CrossRef]
- Babu, V.; Unnikrishnan, P.; Anu, G.; Nair, S.M. Distribution of organophosphorus pesticides in the bed sediments of a backwater system located in an agricultural watershed: Influence of seasonal intrusion of seawater. Arch. Environ. Contam. Toxicol. 2011, 60, 597–609. [Google Scholar] [CrossRef] [PubMed]
- Vryzas, Z. Pesticide fate in soil-sediment-water environment in relation to contamination preventing actions. Curr. Opin. Environ. Sci. Health 2018, 4, 5–9. [Google Scholar] [CrossRef]
- Chen, C.; Guo, W.; Ngo, H.H. Pesticides in stormwater runoff—A mini review. Front. Environ. Sci. Eng. 2019, 13, 72. [Google Scholar] [CrossRef]
- Zhao, B.; Wong, Y.; Ihara, M.; Nakada, N.; Yu, Z.; Sugie, Y.; Miao, Y.; Tanaka, H.; Guan, Y. Characterization of nitrosamines and nitrosamine precursors as non-point source pollutants during heavy rainfall events in an urban water environment. J. Hazard. Mater. 2022, 424, 127552. [Google Scholar] [CrossRef]
- Chaudhary, N.; Choudhary, K.K.; Agrawal, S.; Agrawal, M. Pesticides Usage, Uptake and Mode of Action in Plants with Special Emphasis on Photosynthetic Characteristics. In Pesticides in Crop Production; Wiley: Hoboken, NJ, USA, 2020; pp. 159–180. [Google Scholar]
- de Souza, R.M.; Seibert, D.; Quesada, H.B.; de Jesus Bassetti, F.; Fagundes-Klen, M.R.; Bergamasco, R. Occurrence, impacts and general aspects of pesticides in surface water: A review. Process. Saf. Environ. Prot. 2020, 135, 22–37. [Google Scholar] [CrossRef]
- Bloomfield, J.P.; Williams, R.; Gooddy, D.; Cape, J.; Guha, P. Impacts of climate change on the fate and behaviour of pesticides in surface and groundwater—A UK perspective. Sci. Total Environ. 2006, 369, 163–177. [Google Scholar] [CrossRef]
- Tudi, M.; Daniel Ruan, H.; Wang, L.; Lyu, J.; Sadler, R.; Connell, D.; Chu, C.; Phung, D.T. Agriculture Development, Pesticide Application and Its Impact on The Environment. Int. J. Environ. Res. Public Health 2021, 18, 1112. [Google Scholar] [CrossRef]
- Patnaik, L.; Patra, A.K. Haemoatopoietic alterations induced by carbaryl in Clarias batrachus (LINN). J. Appl. Sci. Environ. Manag. 2006, 10, 5–7. [Google Scholar] [CrossRef]
- Boran, M.; Altinok, I.; Capkin, E. Acute toxicity of carbaryl, methiocarb, and carbosulfan to the rainbow trout (Oncorhynchus mykiss) and guppy (Poecilia reticulata). Turkish J. Vet. Anim. Sci. 2007, 31, 39–45. [Google Scholar]
- Helfrich, L.A.; Weigmann, D.L.; Hipkins, P.A.; Stinson, E.R. Pesticides and Aquatic Animals: A Guide to Reducing Impacts on Aquatic Systems; Virginia Cooperative Extension: Blacksburg, VA, USA, 2009. [Google Scholar]
- Relyea, R.A. The lethal impact of roundup on aquatic and terrestrial amphibians. Ecol. Appl. 2005, 15, 1118–1124. [Google Scholar] [CrossRef]
- Samanic, C.M.; De Roos, A.J.; Stewart, P.A.; Rajaraman, P.; Waters, M.A.; Inskip, P.D. Occupational exposure to pesticides and risk of adult brain tumors. Am. J. Epidemiol. 2008, 167, 976–985. [Google Scholar] [CrossRef] [PubMed]
- Rani, L.; Thapa, K.; Kanojia, N.; Sharma, N.; Singh, S.; Grewal, A.S.; Srivastav, A.L.; Kaushal, J. An Extensive Review on the Consequences of Chemical Pesticides on Human Health and Environment. J. Clean. Prod. 2021, 283, 124657. [Google Scholar] [CrossRef]
- Thomaidi, V.S.; Matsoukas, C.; Stasinakis, A.S. Risk assessment of triclosan released from sewage treatment plants in European rivers using a combination of risk quotient methodology and Monte Carlo simulation. Sci. Total. Environ. 2017, 603, 487–494. [Google Scholar] [CrossRef]
- Noutsopoulos, C.; Koumaki, E.; Sarantopoulos, V.; Mamais, D. Analytical and mathematical assessment of emerging pollutants fate in a river system. J. Hazard. Mater. 2019, 364, 48–58. [Google Scholar] [CrossRef] [PubMed]
- European Commission EC. Technical Guidance Document on Risk Assessment in Support of Commission Directive 93/67/EEC on Risk Assessment for New Notified Substances Part II. In Commission Regulation (EC) No 1488/94 on Risk Assessment for Existing Substances and of Directive 98/8/EC of the European Parliament and of the Council Concerning the Placing of Biocidal Products on the Market; Technical Report EUR 20418 EN/2; European Commission EC: Luxembourg, 2003. [Google Scholar]
- Tsuda, T.; Nakamura, T.; Inoue, A.; Tanaka, K. Pesticides in water, fish and shellfish from littoral area of Lake Biwa. Bull. Environ. Contam. Toxicol. 2009, 82, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Morohashi, M.; Nagasawa, S.; Enya, N.; Suzuki, K.; Kose, T.; Kawata, K. Behavior of bromobutide in paddy water and soil after application. Bull. Environ. Contam. Toxicol. 2012, 88, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Hwang, I.-S.; Oh, Y.-J.; Kwon, H.-Y.; Ro, J.-H.; Kim, D.-B.; Moon, B.-C.; Oh, M.-S.; Noh, H.-H.; Park, S.-W.; Choi, G.-H.; et al. Monitoring of Pesticide Residues Concerned in Stream Water. Korean J. Environ. Agric. 2019, 38, 173–184. [Google Scholar] [CrossRef]
- Kim, C.-S.; Lee, H.-D.; Ihm, Y.-B.; Son, K.-A. Temporal patterns of pesticide residues in the Keum, Mangyung and Dongjin Rivers in 2002. Korean J. Environ. Agric. 2017, 36, 230–240. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.-E.; Jeon, D.; Lee, H.; Huh, Y.; Lee, S.; Kim, J.G.; Kim, H.S. Revealing the Extent of Pesticide Runoff to the Surface Water in Agricultural Watersheds. Water 2023, 15, 3984. https://doi.org/10.3390/w15223984
Kim Y-E, Jeon D, Lee H, Huh Y, Lee S, Kim JG, Kim HS. Revealing the Extent of Pesticide Runoff to the Surface Water in Agricultural Watersheds. Water. 2023; 15(22):3984. https://doi.org/10.3390/w15223984
Chicago/Turabian StyleKim, Young-Eun, Darae Jeon, Hyeri Lee, Yujeong Huh, Soohyung Lee, Jong Guk Kim, and Hyoung Seop Kim. 2023. "Revealing the Extent of Pesticide Runoff to the Surface Water in Agricultural Watersheds" Water 15, no. 22: 3984. https://doi.org/10.3390/w15223984
APA StyleKim, Y.-E., Jeon, D., Lee, H., Huh, Y., Lee, S., Kim, J. G., & Kim, H. S. (2023). Revealing the Extent of Pesticide Runoff to the Surface Water in Agricultural Watersheds. Water, 15(22), 3984. https://doi.org/10.3390/w15223984





