Integrated Process of Immediate One-Step Lime Precipitation, Atmospheric Carbonation, Constructed Wetlands, or Adsorption for Industrial Wastewater Treatment: A Review
Abstract
:1. Introduction
| Types of Industrial Effluent | pH | COD (mg O2 L−1) | BOD (mg O2 L−1) | TSS (mg L−1) | TN (mg N L−1) | NH3 (mg N L−1) | TKN (mg N L−1) | TP (mg P L−1) | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Winery wastewater | 3–12 | 320–296,119 | 125–130,000 | 0–30,300 | NR | NR | NR | NR | [1] |
| Brewery wastewater | 3–12 | 2000–6000 | 1200–3600 | 2901–3000 | NR | NR | 25–80 | NR | [2] |
| Rubber processing wastewater | 3.7–5.5 | 3500–14,000 | 1500–7000 | 200–700 | 200–1800 | NR | NR | NR | [3] |
| Distillery industry wastewater | 3.8–4.4 | 70,000–98,000 | 45,000–60,000 | 2000–14,000 | 1000–1200 | NR | NR | NR | [4] |
| Olive mill wastewater | 4–6 | 40,000–220,000 | 35,000–110,000 | NR | NR | NR | NR | NR | [5] |
| Dairy wastewater | 7.2–8.8 | 1900–2700 | 1200–1800 | 500–740 | NR | NR | NR | NR | [6] |
| Textile industry wastewater | 5.5–10.5 | 350–700 | 150–350 | 200–1100 | NR | NR | NR | NR | [14] |
| Seafood processing wastewater | 6.1–7.1 | 1147–8313 | 463–4569 | 324–3150 | 21–471 | 3.2–1059 | NR | 13–47 | [15] |
| Swine wastewater | 7.4–7.9 | 2050–33,860 | 287–5820 | 1000–27,800 | NR | 321–1129 | 483–2502 | 148–1039 | [16] |
| Pulp and paper mill wastewater | 3.9–8.2 | 1314–4100 | 480–1353 | 83–605 | NR | NR | NR | NR | [17] |
| Slaughterhouse wastewater | 4.9–8.1 | 1250–15,900 | 610–4635 | 300–2800 | 50–841 | NR | NR | 25–200 | [18] |
| Tannery wastewater | 7–8.5 | 3000–6000 | 1200–2700 | 2000–3000 | NR | 100–300 | 250–400 | NR | [19] |
| Pharmaceutical wastewater | 6.7–7.2 | 616–4750 | 322–2440 | 120–354 | NR | NR | 24.6–82.7 | 1.2–3.4 | [20] |
| Petroleum refinery wastewater | 7.5–9.4 | 744–1673 | 205–448 | 280–340 | NR | 40–45 | 82–95 | 1.67–1.73 | [21] |
2. Industrial Wastewater Treatment Strategies
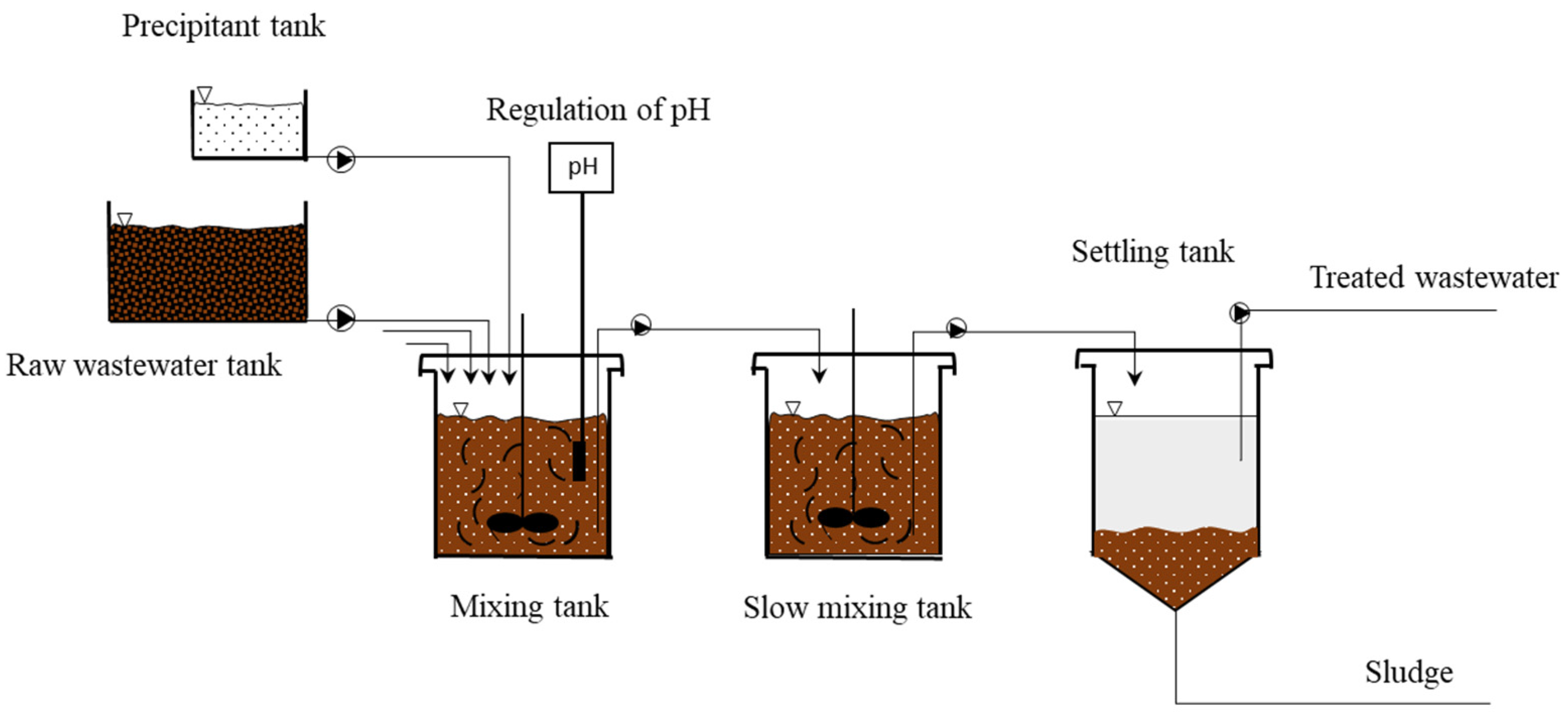
3. Immediate One-Step Lime Precipitation Process
3.1. Theory and Applications
3.2. Operating Variables and Removal Mechanisms
| Type of Wastewater | Reagent and Applied Dose/pH | Operation Mode | Optimum Removal Efficiency | Reference |
|---|---|---|---|---|
| Landfill leachates | Lime (4 g L−1), pH 11.85 | Rapid mixing (300 rpm) for 5 min, slow mixing (30 rpm) for 30 min, followed by settling for 30 min. | COD (25.5%), Ca2+ (93.5%), Mg2+ (98.5%), NH4+ (56.2%), total alkalinity (87.7%), and Fe (75.4%). | [63] |
| Lime (2.0 g L−1), pH = 11.20 | COD (18.0%), Ca2+ (65.0%), Mg2+ (65.0%), NH4+ (29.7%), and total alkalinity (80.0%). | |||
| Lime (6.0 g L−1), pH 10, 40 | COD (0.4%), Ca2+ (91.5%), Mg2+ (95.5%), NH4+ (24.7%), and total alkalinity (90.9%). | |||
| CaO (27.6 g L−1) | 300 rpm stirring speed for 2–60 min, followed by settling for 2 h. | Stirring time has a small influence on organic load and NH4+ removal. | [47] | |
| CaO (18.2–33.3 g L−1) | 300 rpm stirring speed for 40 min, followed by settling for 2 h. | COD (64%) at 27.6 g L−1 of CaO. | ||
| Plywood industry wastewater | Lime (1.5 g L−1) | Rapid mixing and settling for 2 h. | COD (40%), TSS (36.8%), phenol (41%), and TKN (48.1%). | [64] |
| Olive mill wastewater | Lime (10 g L−1), pH 12 | Rapid mixing (200 rpm) for 5 min, slow mixing (60 rpm) for 10 min, and filtration (11 μm) after a resting period. | COD (72%), TSS (73%), and Phenol (60%). | [68] |
| Textile wastewater | Lime (0.8 g L−1), pH 13–13.5 | Rapid mixing for 1–2 min, slow mixing for 15–20 min, followed by settling for 45 min. | COD (50–60%) and color (70–90%). | [65] |
| Tannery wastewater | CaO and Ca(OH)2 (0.3 to 3.2 g alkali/g Cr3+) | 10 min of vigorous stirring, 200 rpm, and a settling time of 24 h. | Cr (99.8%), SO42− (66.9%), ZnSO4 (99.6%), FeSO4 (21.4%), CN−1 (70.9%), NiSO4 (52.8%), and Fe2[Fe(CN)6] (76.4%) for CaO, Cr (99.8%), SO42− (61.6%), ZnSO4 (99.9%), FeSO4 (7.1%), CN−1 (84.0%), NiSO4 (54.4%), and Fe2[Fe(CN)6] (90.5%) for Ca(OH)2 | [67] |
| Cheese whey wastewater | Lime, pH 6 to 13 | 700–800 rpm for 1 min, 300–400 rpm for 1 min, followed by settling for 24 h. | The highest COD removal (29.7%) was obtained at pH 11.0. Total phosphorus (61.9–95.6%) only occurred at pH ≥ 8.0. Highest total phenols removals (63.2–65.5%) at pH 12.0 and 13.0. | [78] |
| Ca(OH)2, pH 8.57 to 12.37 | Vigorous agitation. Then the agitation system was switched off. | Under optimum conditions: COD (90%), turbidity (99.8%), TSS (98–99%), oils and fats (82–96%), phosphorus (98–99%), potassium (96–97%), and total coliforms (100%) for 80% cheese whey recovery and lime application. | [54] | |
| Vinasse wastewater | Ca(OH)2 (12–100 g L−1), pH 10.14–12.49. | Rapid agitation. | COD (51%), absorbances, magnesium, nitrogen, and phosphorus had depletions (≥70%) at pH 12.13 and 12.49. | [70] |
| Olive oil mill wastewater | Ca(OH)2, pH 11.0 to 12.75. | Rapid agitation followed a settling time of 24 h. | COD (11.4–17.8%), total phosphorus (23.6–42.2%), turbidity (60.9–100%), total phenols (25.9–48.0%), and absorbances at 220 nm (10.3–33.5%), 254 nm (18.5–45.9%), 410 nm (34.2–81.6%), and 600 nm (22.1–77.3%). | [80] |
| Winery wastewater | Quicklime, slaked lime, Calcium hydroxide, using 1–50 mL L−1 for slaked lime and calcium hydroxide, and 5–40 mL L−1 for the quicklime. | Vigorous stirring for 2 min followed by a settling time of 1 h. | High removal levels of BOD5 (77.9%), turbidity (98.7%), total phosphorus (87.1%), total phenols (99.9%), fecal coliforms, and Enterococcus (100%) at 25 mL L−1 of slaked lime. | [46] |
| Urban wastewater | Hydrated lime, reagent-grade Ca(OH)2 and quick lime, pH 9.5 to 12.5. | Vigorous mechanical stirring (magnetically agitated, rotation speed of 300 rpm) followed by a settling time of 120 min. | COD (88%), BOD5 (86%), TP (89%), N-organic (75%), and total coliform count (100%) at 0.7 g L−1 (reaction pH of 11.5) of hydrated lime. | [48] |
| Explosives wastewater | Ca(OH)2 (2–19 g L−1), pH 9–12. | Rapid mixing (3 s−1) for 1 min, followed by settling for 46 min. | COD (92.1%), oils and fats (98.2%), organic nitrogen (100%) at 7.76 g L−1 (reaction pH of 10) of hydrated lime. | [51] |
| Slaughterhouse wastewater | Ca(OH)2, pH 9.5–12. | Rapid mixing (3 s−1) for 1 min, followed by settling for 60 min. | COD (7–91%), BOD5 (80–86%), TP (98–99%), TSS (52–99%), 254 nm (87–96%), 410 nm (83–96%), oils and fats (47–92%), turbidity (62–97%) at reaction pH of 12. | [52] |
| Lime (100–600 mg L−1) | Stirring at 100 rpm for 1 min, slow mixing at 40 rpm for 30 min, followed by a settling time of 30 min. | TSS (41.9%), BOD (38.9%), and COD (36.1%) at 400 mg L−1 of lime. | [81] |
3.3. Main Challenges and Recent Advances
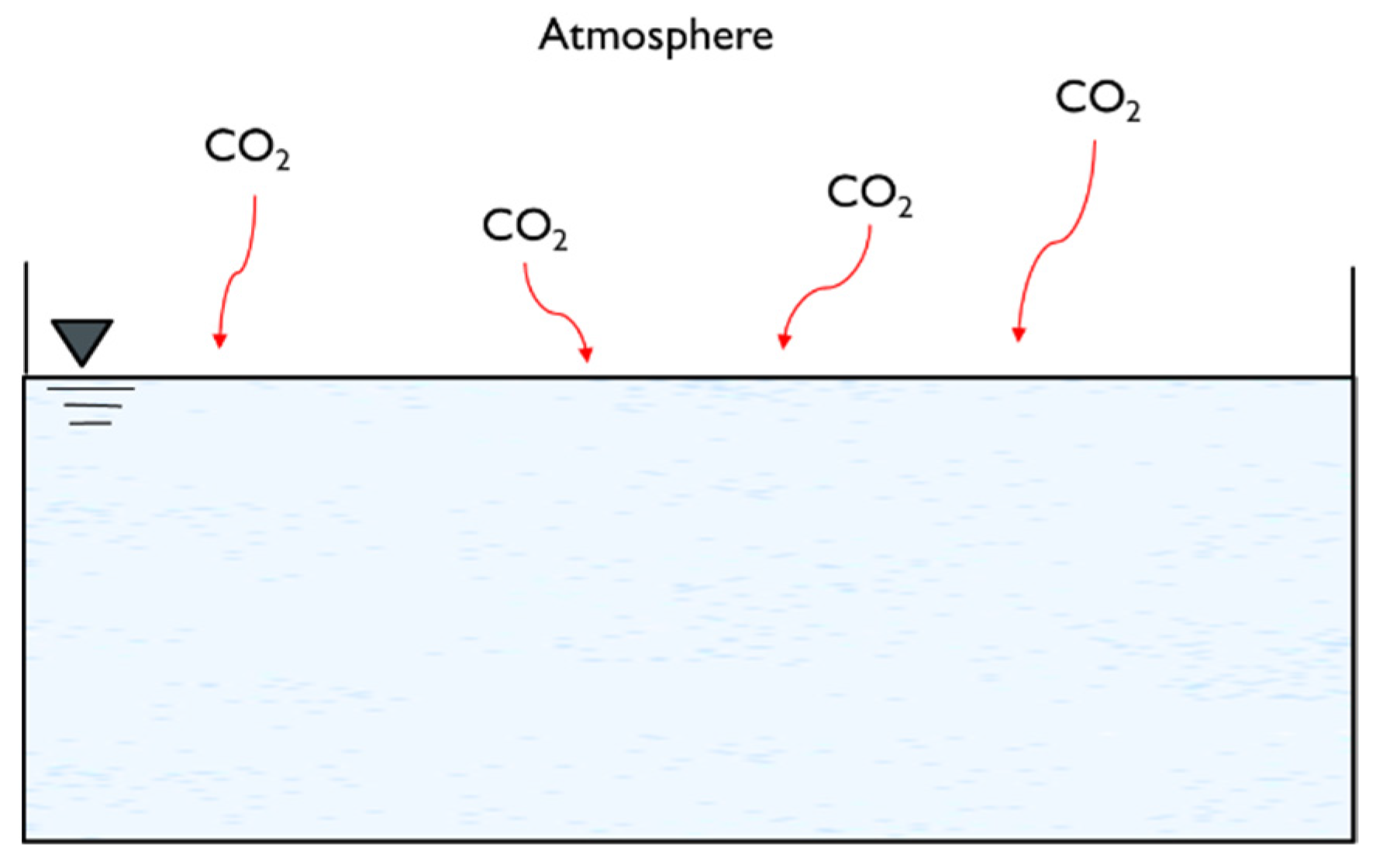
4. Atmospheric Carbonation
4.1. Theory and Applications
4.2. Operating Variables and Removal Mechanisms
4.3. Main Challenges and Recent Advances
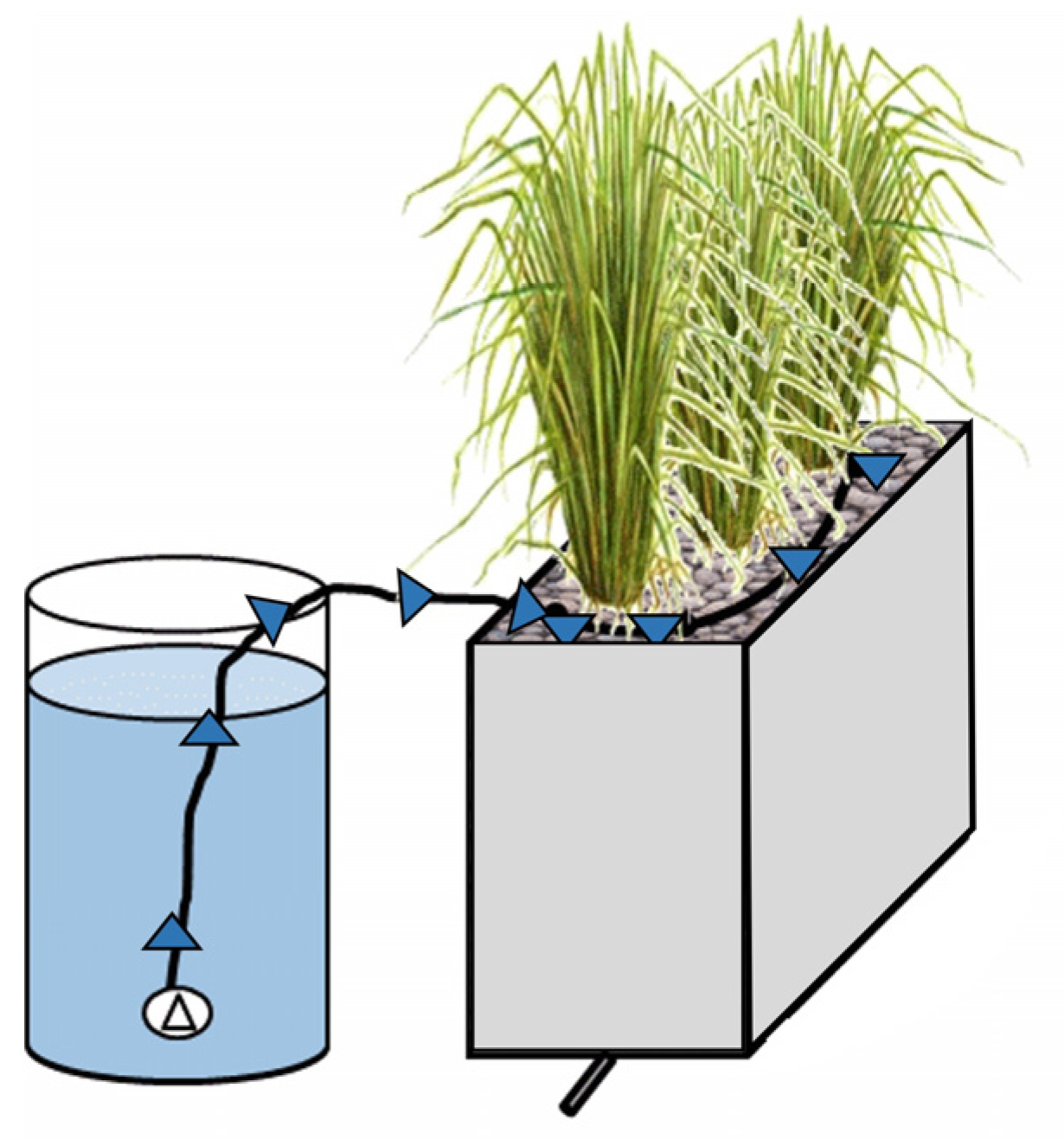
5. Constructed Wetlands
5.1. Theory and Applications
5.2. Operating Variables and Removal Mechanisms
5.3. Main Challenges and Recent Advances
6. Adsorption
6.1. Theory and Applications
6.2. Operating Variables and Removal Mechanisms
6.3. Main Challenges and Recent Advances
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mosse, K.P.M.; Patti, A.F.; Christen, E.W.; Cavagnaro, T.R. Review: Winery wastewater quality and treatment options in Australia. Aust. J. Grape Wine Res. 2011, 17, 111–122. [Google Scholar] [CrossRef]
- Rao, A.G.; Reddy, T.S.K.; Prakash, S.S.; Vanajakshi, J.; Joseph, J.; Sarma, P.N. pH regulation of alkaline wastewater with carbon dioxide: A case study of treatment of brewery wastewater in UASB reactor coupled with absorber. Bioresour. Technol. 2007, 98, 2131–2136. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, M.; Man, H.C.; Hassan, M.A.; Yee, P.L. Treatment of wastewater from rubber industry in Malaysia. African J. Biotechnol. 2010, 9, 6233–6243. [Google Scholar]
- Saha, N.K.; Balakrishnan, M.; Batra, V.S. Improving industrial water use: Case study for an Indian distillery. Resour. Conserv. Recycl. 2005, 43, 163–174. [Google Scholar] [CrossRef]
- Khdair, A.; Abu-Rumman, G. Sustainable environmental management and valorization options for olive mill byproducts in the Middle East and North Africa (MENA) region. Processes 2020, 8, 671. [Google Scholar] [CrossRef]
- Deshannavar, U.B.; Basavaraj, R.K.; Naik, N.M. High rate digestion of dairy industry effluent by upflow anaerobic fixed-bed reactor. J. Chem. Pharm. Res. 2012, 2012, 2895–2899. [Google Scholar]
- Paulino, A.T.; Santos, L.B.; Nozaki, J. Removal of Pb2+, Cu2+, and Fe3+ from battery manufacture wastewater by chitosan produced from silkworm chrysalides as a low-cost adsorbent. React. Funct. Polym. 2008, 68, 634–642. [Google Scholar] [CrossRef]
- Metcalf, E.; Eddy, H. Wastewater Engineering, Treatment, Disposal and Reuse, 4th ed.; Mc Graw-Hill: New York, NY, USA, 2003. [Google Scholar]
- Edokpayi, J.N.; Odiyo, J.O.; Durowoju, O.S.; Edokpayi, J.N.; Odiyo, J.O.; Durowoju, O.S. Impact of Wastewater on Surface Water Quality in Developing Countries: A Case Study of South Africa. Water Qual. 2017, 10, 401–416. [Google Scholar] [CrossRef]
- Singh, S.; Sharma, A.; Malviya, R. Industrial Wastewater: Health Concern and Treatment Strategies. Open Biol. J. 2021, 9, 1–10. [Google Scholar] [CrossRef]
- Fabricius, K. Nutrient pollution/eutrophication. Encycl. Earth Sci. Ser. Part 2011, 2, 722–731. [Google Scholar] [CrossRef]
- Heredia, C.; Guédron, S.; Point, D.; Perrot, V.; Campillo, S.; Verin, C.; Espinoza, M.E.; Fernandez, P.; Duwig, C.; Achá, D. Anthropogenic eutrophication of Lake Titicaca (Bolivia) revealed by carbon and nitrogen stable isotopes fingerprinting. Sci. Total Environ. 2022, 845, 157286. [Google Scholar] [CrossRef] [PubMed]
- Sukmana, H.; Bellahsen, N.; Pantoja, F.; Hodur, C. Adsorption and coagulation in wastewater treatment—Review. Prog. Agric. Eng. Sci. 2021, 17, 49–68. [Google Scholar] [CrossRef]
- Priya, E.S.; Selvan, P.S. Water hyacinth (Eichhornia crassipes)—An efficient and economic adsorbent for textile effluent treatment—A review. Arab. J. Chem. 2017, 10, S3548–S3558. [Google Scholar] [CrossRef]
- Cristóvão, R.O.; Botelho, C.M.; Martins, R.J.E.; Loureiro, J.M.; Boaventura, R.A.R. Primary treatment optimization of a fish canning wastewater from a Portuguese plant. Water Resour. Ind. 2014, 6, 51–63. [Google Scholar] [CrossRef]
- Hunt, P.G.; Matheny, T.A.; Vanotti, M.B.; Stone, K.C.; Szogi, A.A. Denitrification Enzyme Activity in Swine Wastewater Effluent of a Nitrification/Denitrification Treatment System. Trans. ASABE 2012, 55, 159–165. [Google Scholar] [CrossRef]
- Amor, C.; Marchão, L.; Lucas, M.S.; Peres, J.A. Application of advanced oxidation processes for the treatment of recalcitrant agro-industrial wastewater: A review. Water 2019, 11, 205. [Google Scholar] [CrossRef]
- Bustillo-Lecompte, C.; Mehrvar, M.; Quiñones-Bolaños, E. Slaughterhouse wastewater characterization and treatment: An economic and public health necessity of the meat processing industry in Ontario, Canada. J. Geosci. Environ. Prot. 2016, 4, 175–186. [Google Scholar] [CrossRef]
- Sabumon, P. Perspectives on Biological Treatment of Tannery Effluent. Waste Manag. Adv. Recycl. Waste Manag. 2016, 1, 3–10. [Google Scholar] [CrossRef]
- Fawzy, M.E.; Abdelfattah, I.; Abuarab, M.E.; Mostafa, E.; Aboelghait, K.M.; El-Awady, M.H. Sustainable approach for pharmaceutical wastewater treatment and reuse: Case study. J. Environ. Sci. Technol. 2018, 11, 209–219. [Google Scholar] [CrossRef]
- Ishak, S.; Malakahmad, A. Optimization of Fenton process for refinery wastewater biodegradability augmentation. Korean J. Chem. Eng. 2013, 30, 1083–1090. [Google Scholar] [CrossRef]
- Elkarrach, K.; Atia, F.; Omor, A.; Laidi, O.; Biyada, S.; Benlmelih, M.; Merzouki, M. Biological versus Physicochemical Technologies for Industrial Sewage Treatment: Which Is the Most Efficient and Inexpensive. In Sewage—Recent Advances, New Perspectives and Applications? IntechOpen: Rijeka, Croatia, 2022. [Google Scholar] [CrossRef]
- Samer, M. Biological and Chemical Wastewater Treatment Processes. Wastewater Treat. Eng. 2015, 150, 212. [Google Scholar] [CrossRef]
- Hamza, R.A.; Zaghloul, M.S.; Iorhemen, O.T.; Sheng, Z.; Tay, J.H. Optimization of organics to nutrients (COD:N:P) ratio for aerobic granular sludge treating high-strength organic wastewater. Sci. Total Environ. 2019, 650, 3168–3179. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Gupta, R.; Sethi, S.; Biswas, R. Enhancing the efficiency of nitrogen removing bacterial population to a wide range of C:N ratio (1.5:1 to 14:1) for simultaneous C & N removal. Front. Environ. Sci. Front. Environ. Sci. Eng. 2022, 16, 101. [Google Scholar] [CrossRef]
- Wang, H.; Song, Q.; Wang, J.; Zhang, H.; He, Q.; Zhang, W.; Song, J.; Zhou, J.; Li, H. Simultaneous nitrification, denitrification and phosphorus removal in an aerobic granular sludge sequencing batch reactor with high dissolved oxygen: Effects of carbon to nitrogen ratios. Sci. Total Environ. 2018, 642, 1145–1152. [Google Scholar] [CrossRef] [PubMed]
- Derakhshan, M.; Fazeli, M. Improved biodegradability of hardly-decomposable wastewaters from petrochemical industry through photo-Fenton method and determination of optimum operational conditions by response surface methodology. J. Biol. Eng. 2018, 12, 10. [Google Scholar] [CrossRef]
- Mangkoedihardjo, S. Biodegradability improvement of industrial wastewater using hyacinth. J. Appl. Sci. 2006, 6, 1409–1414. [Google Scholar] [CrossRef]
- Cheng, S.; Ran, X.; Ren, G.; Wei, Z.; Wang, Z.; Rao, T.; Li, R.; Ma, X. Comparison of Fenton and Ozone Oxidation for Pretreatment of Petrochemical Wastewater: COD Removal and Biodegradability Improvement Mechanism. Separations 2022, 9, 179. [Google Scholar] [CrossRef]
- Devda, V.; Chaudhary, K.; Varjani, S.; Pathak, B.; Patel, A.K.; Singhania, R.R.; Taherzadeh, M.J.; Ngo, H.H.; Wong, J.W.C.; Guo, W.; et al. Recovery of resources from industrial wastewater employing electrochemical technologies: Status, advancements and perspectives. Bioengineered 2021, 12, 4697–4718. [Google Scholar] [CrossRef]
- Ma, C.; Ran, Z.; Yang, Z.; Wang, L.; Wen, C.; Zhao, B.; Zhang, H. Efficient pretreatment of industrial estate wastewater for biodegradability enhancement using a micro-electrolysis-circulatory system. J. Environ. Manag. 2019, 250, 109492. [Google Scholar] [CrossRef]
- Saravanathamizhan, R.; Perarasu, V.T. Improvement of Biodegradability Index of Industrial Wastewater Using Different Pretreatment Techniques. In Wastewater Treatment: Cutting-Edge Molecular Tools, Techniques and Applied Aspects; Elsevier: Amsterdam, The Netherlands, 2021; pp. 103–136. [Google Scholar] [CrossRef]
- Kokkinos, P.; Venieri, D.; Mantzavinos, D. Advanced Oxidation Processes for Water and Wastewater Viral Disinfection. A Systematic Review. Food Environ. Virol. 2021, 13, 283. [Google Scholar] [CrossRef]
- Pandey, N.; Keshavkant, S. Mechanisms of heavy metal removal using microorganisms as biosorbents. In New Trends in Removal of Heavy Metals from Industrial Wastewater; Elsevier: Amsterdam, The Netherlands, 2021; pp. 1–21. [Google Scholar] [CrossRef]
- Ding, R.; Wang, Y.; Chen, X.; Gao, Y.; Yang, M. Extended Fenton’s process: Toward improving biodegradability of drilling wastewater. Water Sci. Technol. 2019, 79, 1790–1797. [Google Scholar] [CrossRef] [PubMed]
- Waly, M.M.; Ahmed, T.; Abunada, Z.; Mickovski, S.B.; Thomson, C. Constructed Wetland for Sustainable and Low-Cost Wastewater Treatment: Review Article. Land 2022, 11, 1388. [Google Scholar] [CrossRef]
- Almazán-Sánchez, P.T.; Linares-Hernández, I.; Solache-Río, M.J.; Martínez-Miranda, V. Textile wastewater treatment using iron-modified clay and copper-modified carbon in batch and column systems. Water Air Soil Pollut. 2016, 227, 100. [Google Scholar] [CrossRef]
- Álvarez-Torrellas, S.; Peres, J.A.; Gil-Álvarez, V.; Ovejero, G.; García, J. Effective adsorption of non-biodegradable pharmaceuticals from hospital wastewater with different carbon materials. Chem. Eng. J. 2017, 320, 319–329. [Google Scholar] [CrossRef]
- Rodríguez, J.; Castrillón, L.; Marañón, E.; Sastre, H.; Fernández, E. Removal of non-biodegradable organic matter from landfill leachates by adsorption. Water Res. 2004, 38, 3297–3303. [Google Scholar] [CrossRef] [PubMed]
- Skrzypiec, K.; Gajewska, M.H. The use of constructed wetlands for the treatment of industrial wastewater. J. Water L. Dev. 2017, 34, 233–240. [Google Scholar] [CrossRef]
- Keerthana, K.; Thivyatharsan, R. Evaluation of the efficiency of constructed wetland and activated charcoal for the treatment of slaughterhouse wastewater. Earth Energy Environ. 2018, 6, 12. [Google Scholar]
- Achak, M.; Barka, N.; Lamy, E. Constructed Wetlands Process for Treating Sewage to Improve the Quantitative and Qualitative Management of Groundwater Resources; IntechOpen: Rijeka, Croatia, 2023. [Google Scholar] [CrossRef]
- Wang, H.; Sheng, L.; Xu, J. Clogging mechanisms of constructed wetlands: A critical review. J. Clean. Prod. 2021, 295, 126455. [Google Scholar] [CrossRef]
- Karungamye, P. Wastewater pretreatment methods for constructed wetland: Review. J. Mater. Environ. Sci. 2021, 12, 1614–1626. [Google Scholar]
- Ying, Z.; Li, X.; Zhang, L.; Xi, B.; Xia, X.; Zhang, Z.; Zhan, J. Coagulation pretreatment for constructed Wetlands. Fresenius Environ. Bull. 2011, 20, 2326–2334. [Google Scholar]
- Luz, S.; Rivas, J.; Afonso, A.; Carvalho, F. Immediate one-step lime precipitation process for the valorization of winery wastewater to agricultural purposes. Environ. Sci. Pollut. Res. 2021, 28, 18382–18391. [Google Scholar] [CrossRef] [PubMed]
- Ramalho, M.; Jovanović, T.; Afonso, A.; Baía, A.; Lopes, A.; Fernandes, A.; Almeida, A.; Carvalho, F. Landfill leachate treatment by immediate one-step lime precipitation, carbonation, and phytoremediation fine-tuning. Environ. Sci. Pollut. Res. 2023, 30, 8647–8656. [Google Scholar] [CrossRef] [PubMed]
- Correia, T.; Regato, M.; Almeida, A.; Santos, T.; Amaral, L.; Carvalho, F. Manual Treatment of Urban Wastewater by Chemical Precipitation for Production of Hydroponic Nutrient Solutions. Ecol. Eng. J. Ecol. Eng. 2020, 21, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Madeira, L.; Almeida, A.; da Costa, A.M.R.; Mestre, A.S.; Carvalho, F.; Ribau Teixeira, M. Tunning processes for organic matter removal from slaughterhouse wastewater treated by immediate one-step lime precipitation and atmospheric carbonation. J. Environ. Chem. Eng. 2023, 11, 110450. [Google Scholar] [CrossRef]
- Madeira, L.; Carvalho, F.; Teixeira, M.R.; Ribeiro, C.; Almeida, A. Vertical flow constructed wetland as a green solution for low biodegradable and high nitrogen wastewater: A case study of explosives industry. Chemosphere 2021, 272, 129871. [Google Scholar] [CrossRef]
- Madeira, L.; Almeida, A.; Ribau Teixeira, M.; Prazeres, A.; Chaves, H.; Carvalho, F. Immediate one-step lime precipitation and atmospheric carbonation as pre-treatment for low biodegradable and high nitrogen wastewaters: A case study of explosives industry. J. Environ. Chem. Eng. 2020, 8, 103808. [Google Scholar] [CrossRef]
- Madeira, L.; Carvalho, F.; Almeida, A.; Ribau Teixeira, M. Optimization of atmospheric carbonation in the integrated treatment immediate one-step lime precipitation and atmospheric carbonation. The case study of slaughterhouse effluents. Results Eng. 2023, 17, 100807. [Google Scholar] [CrossRef]
- De Jesus, M.A.S.; Aguiar Dutra, A.R.D.; Cirani, C.B.S.; Jesus, K.R.E.; Neto, R.C.S.; Guerra, J.B.A. Eco-innovation assessment of biodigesters technology: An application in cassava processing industries in the south of Brazil, Parana state. Clean Technol. Environ. Policy 2022, 24, 931–948. [Google Scholar] [CrossRef]
- Prazeres, A.R.; Rivas, J.; Paulo, Ú.; Ruas, F.; Carvalho, F. Sustainable treatment of different high-strength cheese whey wastewaters: An innovative approach for atmospheric CO2 mitigation and fertilizer production. Environ. Sci. Pollut. Res. 2016, 23, 13062–13075. [Google Scholar] [CrossRef]
- Aguilar, M.I.; Sáez, J.; Lloréns, M.; Soler, A.; Ortuño, J.F.; Meseguer, V.; Fuentes, A. Improvement of coagulation-flocculation process using anionic polyacrylamide as coagulant aid. Chemosphere 2005, 58, 47–56. [Google Scholar] [CrossRef]
- Mahedi, M.; Cetin, B.; Dayioglu, A.Y. Leaching behavior of aluminum, copper, iron and zinc from cement activated fly ash and slag stabilized soils. Waste Manag. 2019, 95, 334–355. [Google Scholar] [CrossRef]
- Serra, A.P.; Marchetti, M.E.; Dupas, E.; Carducci, C.E.; da Silva, E.F.; Pinheiro, E.R. Phosphorus in Forage Production. In New Perspectives in Forage Crops; InTech: Rijeka, Croatia, 2018. [Google Scholar] [CrossRef]
- Zhao, Z.L.; Li, W.W.; Wang, F.; Zhang, Y.Q. Using of hydrated lime water as a novel degumming agent of silk and sericin recycling from wastewater. J. Clean. Prod. 2018, 172, 2090–2096. [Google Scholar] [CrossRef]
- Wang, W.; Hua, Y.; Li, S.; Yan, W.; Zhang, W. xian Removal of Pb(II) and Zn(II) using lime and nanoscale zero-valent iron (nZVI): A comparative study. Chem. Eng. J. 2016, 304, 79–88. [Google Scholar] [CrossRef]
- Semerjian, L.; Ayoub, G.M. High-pH–magnesium coagulation–flocculation in wastewater treatment. Adv. Environ. Res. 2003, 7, 389–403. [Google Scholar] [CrossRef]
- An, B.H.; Xu, D.M.; Geng, R.; Cheng, Y.; Qian, R.B.; Tang, X.C.; Fan, Z.Q.; Chen, H. Bin The pretreatment effects of various target pollutant in real coal gasification gray water by coupling pulse electrocoagulation with chemical precipitation methods. Chemosphere 2023, 311, 136898. [Google Scholar] [CrossRef] [PubMed]
- Niaounakis, M.; Halvadakis, C.P. Olive Processing Waste Management: Literature Review and Patent Survey, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Renou, S.; Poulain, S.; Givaudan, J.G.; Sahut, C.; Moulin, P. Lime treatment of stabilized leachates. Water Sci. Technol. 2009, 59, 673–685. [Google Scholar] [CrossRef]
- Prasad, H.; Lohchab, R.K.; Singh, B.; Nain, A.; Kumari, M. Lime treatment of wastewater in a plywood industry to achieve the zero liquid discharge. J. Clean. Prod. 2019, 240, 118176. [Google Scholar] [CrossRef]
- Georgiou, D.; Aivazidis, A.; Hatiras, J.; Gimouhopoulos, K. Treatment of cotton textile wastewater using lime and ferrous sulfate. Water Res. 2003, 37, 2248–2250. [Google Scholar] [CrossRef]
- Ayoub, G.M.; Hamzeh, A.; Semerjian, L. Post treatment of tannery wastewater using lime/bittern coagulation and activated carbon adsorption. Desalination 2011, 273, 359–365. [Google Scholar] [CrossRef]
- Reyes-Serrano, A.; López-Alejo, J.E.; Hernández-Cortázar, M.A.; Elizalde, I. Removing contaminants from tannery wastewater by chemical precipitation using CaO and Ca(OH)2. Chin. J. Chem. Eng. 2020, 28, 1107–1111. [Google Scholar] [CrossRef]
- Boukhoubza, F.; Jail, A.; Korchi, F.; Idrissi, L.L.; Hannache, H.; Duarte, J.C.; Hassani, L.; Nejmeddine, A. Application of lime and calcium hypochlorite in the dephenolisation and discolouration of olive mill wastewater. J. Environ. Manag. 2009, 91, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Folkman, Y.; Wachs, A.M. Removal of algae from stabilization pond effluents by lime treatment. Water Res. 1973, 7, 419–435. [Google Scholar] [CrossRef]
- Prazeres, A.R.; Lelis, J.; Alves-Ferreira, J.; Carvalho, F. Treatment of vinasse from sugarcane ethanol industry: H2SO4, NaOH and Ca(OH)2 precipitations, FeCl3 coagulation-flocculation and atmospheric CO2 carbonation. J. Environ. Chem. Eng. 2019, 7, 103203. [Google Scholar] [CrossRef]
- Wang, Y. Zero Discharge of wastewater Treatment Technology of Wet Desulfurization in Coal-fired Power Plants. In Proceedings of the 5th International Conference on Environment, Materials, Chemistry and Power Electronics, Zhengzhou, China, 11–12 April 2016; Atlantis Press: Zhengzhou, China, 2016. [Google Scholar] [CrossRef]
- Forouzesh, M.; Fatehifar, E.; Khoshbouy, R.; Daryani, M. Experimental investigation of iron removal from wet phosphoric acid through chemical precipitation process. Chem. Eng. Res. Des. 2023, 189, 308–318. [Google Scholar] [CrossRef]
- Spellman, F.R. Handbook of Water and Wastewater Treatment Plant Operations, 4th ed.; CRC Press: Boca Raton, FL, USA, 2020. [Google Scholar] [CrossRef]
- Ayoub, G.M.; Merhebi, F. Characteristics and quantities of sludge produced by coagulating wastewater with seawater bittern, lime and caustic. Adv. Environ. Res. 2002, 6, 277–284. [Google Scholar] [CrossRef]
- Ayeche, R. Treatment by Coagulation-Flocculation of Dairy Wastewater with the Residual Lime of National Algerian Industrial Gases Company (NIGC-Annaba). Energy Procedia 2012, 18, 147–156. [Google Scholar] [CrossRef]
- Liu, D.H.F.; Lipták, B.G. Wastewater Treatment; Boca Raton: London, UK, 2000. [Google Scholar]
- Li, Y.; Nan, X.; Li, D.; Wang, L.; Xu, R.; Li, Q. Advances in the treatment of phosphorus-containing wastewater. IOP Conf. Ser. Earth Environ. Sci. 2021, 647, 012163. [Google Scholar] [CrossRef]
- Prazeres, A.R.; Luz, S.; Fernandes, F.; Jerónimo, E. Cheese wastewater treatment by acid and basic precipitation: Application of H2SO4, HNO3, HCl, Ca(OH)2 and NaOH. J. Environ. Chem. Eng. 2020, 8, 103556. [Google Scholar] [CrossRef]
- Habte, L.; Shiferaw, N.; Thriveni, T.; Mulatu, D.; Lee, M.H.; Jung, S.H.; Ahn, J.W. Removal of Cd(II) and Pb(II) from wastewater via carbonation of aqueous Ca(OH)2 derived from eggshell. Process Saf. Environ. Prot. 2020, 141, 278–287. [Google Scholar] [CrossRef]
- Prazeres, A.R.; Afonso, A.; Guerreiro, R.; Jerónimo, E. Contamination reduction of real olive oil mill wastewater using innovative acid and basic chemical precipitation processes. Int. J. Environ. Sci. Technol. 2021, 18, 799–808. [Google Scholar] [CrossRef]
- Satyanarayan, S.; Ramakant; Vanerkar, A.P. Conventional approach for abattoir wastewater treatment. Environ. Technol. 2005, 26, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Luo, Z.; Hills, C.; Xue, G.; Tyrer, M. Precipitation of heavy metals from wastewater using simulated flue gas: Sequent additions of fly ash, lime and carbon dioxide. Water Res. 2009, 43, 2605–2614. [Google Scholar] [CrossRef] [PubMed]
- Mirbagheri, S.A.; Hosseini, S.N. Pilot plant investigation on petrochemical wastewater treatmentfor the removal of copper and chromium with the objective of reuse. Desalination 2005, 171, 85–93. [Google Scholar] [CrossRef]
- Cheng, Q.; Wu, Y.; Huang, Y.; Li, F.; Liu, Z.; Nengzi, L.; Bao, L. An integrated process of calcium hydroxide precipitation and air stripping for pretreatment of flue gas desulfurization wastewater towards zero liquid discharge. J. Clean. Prod. 2021, 314, 128077. [Google Scholar] [CrossRef]
- Martínez-Cruz, A.; Fernandes, A.; Ramos, F.; Soares, S.; Correia, P.; Baía, A.; Lopes, A.; Carvalho, F. An Eco-Innovative Solution for Reuse of Leachate Chemical Precipitation Sludge: Application to Sanitary Landfill Coverage. Ecol. Eng. Environ. Technol. 2021, 22, 52–58. [Google Scholar] [CrossRef]
- Bal Krishna, K.C.; Niaz, M.R.; Sarker, D.C.; Jansen, T. Phosphorous removal from aqueous solution can be enhanced through the calcination of lime sludge. J. Environ. Manag. 2017, 200, 359–365. [Google Scholar] [CrossRef]
- Madeira, L.; Ribau Teixeira, M.; Almeida, A.; Santos, T.; Carvalho, F. Reuse of lime sludge from immediate one-step lime precipitation process as a coagulant (aid) in slaughterhouse wastewater treatment. J. Environ. Manag. 2023, 342, 118278. [Google Scholar] [CrossRef]
- Viswanaathan, S.; Perumal, P.K.; Sundaram, S. Integrated Approach for Carbon Sequestration and Wastewater Treatment Using Algal-Bacterial Consortia: Opportunities and Challenges. Sustainability 2022, 14, 1075. [Google Scholar] [CrossRef]
- Mollica, N.R.; Guo, W.; Cohen, A.L.; Huang, K.F.; Foster, G.L.; Donald, H.K.; Solow, A.R. Ocean acidification affects coral growth by reducing skeletal density. Proc. Natl. Acad. Sci. USA 2018, 115, 1754–1759. [Google Scholar] [CrossRef]
- Mihelcic, J.R.; Zimmerman, J.B.; Zhang, Q. Water Treatment and Design. In Environmental Engineering: Fundamentals, Sustainability, Design; Mihelcic, J.R., Zimmerman, J.B., Eds.; Wiley: Hoboken, NJ, USA, 2002; pp. 397–455. [Google Scholar]
- Lin, L.; Yuan, S.; Lin, L.; Yuan, S.; Chen, J.; Xu, Z.; Lu, X. Removal of ammonia nitrogen in wastewater by microwave radiation. J. Hazard. Mater. 2009, 161, 1063–1068. [Google Scholar] [CrossRef]
- Wang, L.; Hung, Y.T.; Shammas, N.K. Physicochemical Treatment Processes; Humana Press: Totowa, NJ, USA, 2005. [Google Scholar] [CrossRef]
- Folino, A.; Zema, D.A.; Calabrò, P.S. Environmental and economic sustainability of swine wastewater treatments using ammonia stripping and anaerobic digestion: A short review. Sustainability 2020, 12, 4971. [Google Scholar] [CrossRef]
- Madeira, L.; Ribau, M.; Almeida, A.; Carvalho, F. A Process for Treating Wastewater Using Atmospheric CO2. Provisional Patent Application nr. PT 118724, 15 June 2023. [Google Scholar]
- Dinesh Kumar, M.; Gopikumar, S.; Uan, D.K.; Adishkumar, S.; Rajesh Banu, J. Constructed Wetlands: An Emerging Green Technology for the Treatment of Industrial Wastewaters. In Emerging Eco-Friendly Green Technologies for Wastewater Treatment; Springer: Singapore, 2020; pp. 21–44. [Google Scholar] [CrossRef]
- Hota, A.; Gopal Krishna Patro, S.; Obaid, A.J.; Khatak, S.; Kumar, R. Constructed wetland challenges for the treatment of industrial wastewater in smart cities: A sensitive solution. Sustain. Energy Technol. Assess. 2023, 55, 102967. [Google Scholar] [CrossRef]
- Lajayer, B.A.; Nobaharan, K.; Moghiseh, E.; Biglari Quchan Atigh, Z.; Delangiz, N.; Mosaferi, M.; Aftab, T.; Astatkie, T. Removal of pharmaceuticals and personal care products from water and wastewater through biological processes: An overview. In Hazardous and Trace Materials in Soil and Plants: Sources, Effects, and Management; Academic Press: Cambridge, MA, USA, 2022; pp. 87–96. [Google Scholar] [CrossRef]
- Kadlec, R.H.; Wallace, S. Treatment Wetlands, Treatment Wetlands; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar] [CrossRef]
- Fitch, M.W. 3.14 Constructed Wetlands. In Comprehensive Water Quality and Purification; Elsevier: Amsterdam, The Netherlands, 2013; pp. 268–295. [Google Scholar] [CrossRef]
- Saeed, T.; Khan, T. Constructed wetlands for industrial wastewater treatment: Alternative media, input biodegradation ratio and unstable loading. J. Environ. Chem. Eng. 2019, 7, 103042. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, J.; Ngo, H.H.; Guo, W.; Hu, Z.; Liang, S.; Fan, J.; Liu, H. A review on the sustainability of constructed wetlands for wastewater treatment: Design and operation. Bioresour. Technol. 2015, 175, 594–601. [Google Scholar] [CrossRef] [PubMed]
- Mlih, R.; Bydalek, F.; Klumpp, E.; Yaghi, N.; Bol, R.; Wenk, J. Light-expanded clay aggregate (LECA) as a substrate in constructed wetlands—A review. Ecol. Eng. 2020, 148, 105783. [Google Scholar] [CrossRef]
- Masoud, A.M.N.; Alfarra, A.; Sorlini, S. Constructed Wetlands as a Solution for Sustainable Sanitation: A Comprehensive Review on Integrating Climate Change Resilience and Circular Economy. Water 2022, 14, 3232. [Google Scholar] [CrossRef]
- Stefanakis, A.I. Constructed wetlands for sustainable wastewater treatment in hot and arid climates: Opportunities, challenges and case studies in the Middle East. Water 2020, 12, 1665. [Google Scholar] [CrossRef]
- Hassan, I.; Chowdhury, S.R.; Prihartato, P.K.; Razzak, S.A. Wastewater treatment using constructed wetland: Current trends and future potential. Processes 2021, 9, 1917. [Google Scholar] [CrossRef]
- Gorgoglione, A.; Torretta, V. Sustainable management and successful application of constructed wetlands: A critical review. Sustainability 2018, 10, 3910. [Google Scholar] [CrossRef]
- Pundlik, R.C.; Dash, R.R.; Bhunia, P. Constructed wetland system for the treatment of wastewater in a circular bioeconomy. In Biomass, Biofuels, Biochemicals: Circular Bioeconomy: Technologies for Waste Remediation; Elsevier: Amsterdam, The Netherlands, 2022; pp. 365–386. [Google Scholar] [CrossRef]
- Singh, A.; Katoch, S.S.; Bajpai, M.; Rawat, A. Constructed Wetlands: A sustainable way of Treating Wastewater in Cold Climate—A review. Pollution 2022, 8, 373–396. [Google Scholar] [CrossRef]
- Almeida, A.; Ribeiro, C.; Carvalho, F.; Durao, A.; Bugajski, P.; Kurek, K.; Pochwatka, P.; Jóźwiakowski, K. Phytoremediation potential of Vetiveria zizanioides and Oryza sativa to nitrate and organic substance removal in vertical flow constructed wetland systems. Ecol. Eng. 2019, 138, 19–27. [Google Scholar] [CrossRef]
- Vymazal, J. Constructed wetlands for treatment of industrial wastewaters: A review. Ecol. Eng. 2014, 73, 724–751. [Google Scholar] [CrossRef]
- Ky, N.M.; Hung, N.T.Q.; Manh, N.C.; Lap, B.Q.; Dang, H.T.T.; Ozaki, A. Assessment of nutrients removal by constructed wetlands using reed grass (Phragmites australis L.) and vetiver grass (Vetiveria zizanioides L.). J. Fac. Agric. Kyushu Univ. 2020, 65, 149–156. [Google Scholar] [CrossRef]
- Vymazal, J.; Zhao, Y.; Mander, Ü. Recent research challenges in constructed wetlands for wastewater treatment: A review. Ecol. Eng. 2021, 169, 106318. [Google Scholar] [CrossRef]
- García-Ávila, F.; Avilés-Añazco, A.; Cabello-Torres, R.; Guanuchi-Quito, A.; Cadme-Galabay, M.; Gutiérrez-Ortega, H.; Alvarez-Ochoa, R.; Zhindón-Arévalo, C. Application of ornamental plants in constructed wetlands for wastewater treatment: A scientometric analysis. Case Stud. Chem. Environ. Eng. 2023, 7, 100307. [Google Scholar] [CrossRef]
- Billore, S.K.; Singh, N.; Ram, H.K.; Sharma, J.K.; Singh, V.P.; Nelson, R.M.; Dass, P. Treatment of a molasses based distillery effluent in a constructed wetland in central India. Water Sci. Technol. 2001, 44, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Sarabia, A.; Fernández-Villagómez, G.; Martínez-Pereda, P.; Rinderknecht-Seijas, N.; Poggi-Varaldo, H.M. Slaughterhouse wastewater treatment in a full-scale system with constructed wetlands. Water Environ. Res. 2004, 76, 334–343. [Google Scholar] [CrossRef]
- Mustapha, H.I.; van Bruggen, J.J.A.; Lens, P.N.L. Optimization of Petroleum Refinery Wastewater Treatment by Vertical Flow Constructed Wetlands Under Tropical Conditions: Plant Species Selection and Polishing by a Horizontal Flow Constructed Wetland. Water Air Soil Pollut. 2018, 229, 137. [Google Scholar] [CrossRef]
- Engida, T.; Alemu, T.; Wu, J.; Xu, D.; Zhou, Q.; Wu, Z. Analysis of constructed wetlands technology performance efficiency for the treatment of floriculture industry wastewater, in Ethiopia. J. Water Process Eng. 2020, 38, 101586. [Google Scholar] [CrossRef]
- Serrano, L.; de la Varga, D.; Ruiz, I.; Soto, M. Winery wastewater treatment in a hybrid constructed wetland. Ecol. Eng. 2011, 37, 744–753. [Google Scholar] [CrossRef]
- Abira, M.A.; van Bruggen, J.J.A.; Denny, P. Potential of a tropical subsurface constructed wetland to remove phenol from pre-treated pulp and papermill wastewater. Water Sci. Technol. 2005, 51, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Hussain, Z.; Arslan, M.; Malik, M.H.; Mohsin, M.; Iqbal, S.; Afzal, M. Integrated perspectives on the use of bacterial endophytes in horizontal flow constructed wetlands for the treatment of liquid textile effluent: Phytoremediation advances in the field. J. Environ. Manag. 2018, 224, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Bulc, T.G.; Ojstršek, A. The use of constructed wetland for dye-rich textile wastewater treatment. J. Hazard. Mater. 2008, 155, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.C.; Silva, L.; Albuquerque, A.; Simões, R.; Stefanakis, A.I. Investigation of lab-scale horizontal subsurface flow constructed wetlands treating industrial cork boiling wastewater. Chemosphere 2018, 207, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Schulz, C.; Gelbrecht, J.; Rennert, B. Treatment of rainbow trout farm effluents in constructed wetland with emergent plants and subsurface horizontal water flow. Aquaculture 2003, 217, 207–221. [Google Scholar] [CrossRef]
- Kapellakis, I.E.; Paranychianakis, N.V.; Tsagarakis, K.P.; Angelakis, A.N. Treatment of olive mill wastewater with constructed wetlands. Water 2012, 4, 260–271. [Google Scholar] [CrossRef]
- Saeed, T.; Muntaha, S.; Rashid, M.; Sun, G.; Hasnat, A. Industrial wastewater treatment in constructed wetlands packed with construction materials and agricultural by-products. J. Clean. Prod. 2018, 189, 442–453. [Google Scholar] [CrossRef]
- Gholipour, A.; Zahabi, H.; Stefanakis, A.I. A novel pilot and full-scale constructed wetland study for glass industry wastewater treatment. Chemosphere 2020, 247, 125966. [Google Scholar] [CrossRef]
- Agarry, S.E.; Oghenejoboh, K.M.; Latinwo, G.K.; Owabor, C.N. Biotreatment of petroleum refinery wastewater in vertical surface-flow constructed wetland vegetated with Eichhornia crassipes: Lab-scale experimental and kinetic modelling. Environ. Technol. 2020, 41, 1793–1813. [Google Scholar] [CrossRef]
- Yirong, C.; Puetpaiboon, U. Performance of constructed wetland treating wastewater from seafood industry. Water Sci. Technol. 2004, 49, 289–294. [Google Scholar] [CrossRef]
- Klomjek, P. Swine wastewater treatment using vertical subsurface flow constructed wetland planted with Napier grass. Sustain. Environ. Res. 2016, 26, 217–223. [Google Scholar] [CrossRef]
- Bojcevska, H.; Tonderski, K. Impact of loads, season, and plant species on the performance of a tropical constructed wetland polishing effluent from sugar factory stabilization ponds. Ecol. Eng. 2007, 29, 66–76. [Google Scholar] [CrossRef]
- Vymazal, J.; Sochacki, A.; Fučík, P.; Šereš, M.; Kaplická, M.; Hnátková, T.; Chen, Z. Constructed wetlands with subsurface flow for nitrogen removal from tile drainage. Ecol. Eng. 2020, 155, 105943. [Google Scholar] [CrossRef]
- Almeida, A.; Jóźwiakowski, K.; Kowalczyk-Juśko, A.; Bugajski, P.; Kurek, K.; Carvalho, F.; Durao, A.; Ribeiro, C.; Gajewska, M. Nitrogen removal in vertical flow constructed wetlands: Influence of bed depth and high nitrogen loadings. Environ. Technol. 2020, 41, 2196–2209. [Google Scholar] [CrossRef]
- Saeed, T.; Sun, G. A review on nitrogen and organics removal mechanisms in subsurface flow constructed wetlands: Dependency on environmental parameters, operating conditions and supporting media. J. Environ. Manag. 2012, 112, 429–448. [Google Scholar] [CrossRef]
- Vymazal, J.; Březinová, T. Accumulation of heavy metals in aboveground biomass of Phragmites australis in horizontal flow constructed wetlands for wastewater treatment: A review. Chem. Eng. J. 2016, 290, 232–242. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, T.; Xu, Z.; Zhang, L.; Dai, Y.; Tang, X.; Tao, R.; Li, R.; Yang, Y.; Tai, Y. Effect of heavy metals in mixed domestic-industrial wastewater on performance of recirculating standing hybrid constructed wetlands (RSHCWs) and their removal. Chem. Eng. J. 2020, 379, 122363. [Google Scholar] [CrossRef]
- Biswal, B.K.; Balasubramanian, R. Constructed Wetlands for Reclamation and Reuse of Wastewater and Urban Stormwater: A Review. Front. Environ. Sci. 2022, 10, 201. [Google Scholar] [CrossRef]
- Vymazal, J. Removal of nutrients in various types of constructed wetlands. Sci. Total Environ. 2007, 380, 48–65. [Google Scholar] [CrossRef]
- Cooper, P.; Job, G.; Green, M.; Shutes, R. Reed Beds and Constructed Wetlands for Wastewater Treatment; Work Research Centre Publications: Buckinghamshire, UK, 1996. [Google Scholar]
- Kafle, A.; Timilsina, A.; Gautam, A.; Adhikari, K.; Bhattarai, A.; Aryal, N. Phytoremediation: Mechanisms, plant selection and enhancement by natural and synthetic agents. Environ. Adv. 2022, 8, 100203. [Google Scholar] [CrossRef]
- Sohsalam, P.; Englande, A.J.; Sirianuntapiboon, S. Seafood wastewater treatment in constructed wetland: Tropical case. Bioresour. Technol. 2008, 99, 1218–1224. [Google Scholar] [CrossRef] [PubMed]
- Saeed, T.; Afrin, R.; Al Muyeed, A.; Sun, G. Treatment of tannery wastewater in a pilot-scale hybrid constructed wetland system in Bangladesh. Chemosphere 2012, 88, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.F.; Ling, J.; Xu, J.C.; Feng, Y.; Li, G.M. Advanced treatment of wastewater from an iron and steel enterprise by a constructed wetland/ultrafiltration/reverse osmosis process. Desalination 2011, 269, 41–49. [Google Scholar] [CrossRef]
- Ji, G.D.; Sun, T.H.; Ni, J.R. Surface flow constructed wetland for heavy oil-produced water treatment. Bioresour. Technol. 2007, 98, 436–441. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Zhang, G.; Liu, J.; Zhu, Y.; Xu, J. Performance of a constructed wetland in treating brackish wastewater from commercial recirculating and super-intensive shrimp growout systems. Bioresour. Technol. 2011, 102, 9416–9424. [Google Scholar] [CrossRef]
- Fibbi, D.; Doumett, S.; Colzi, I.; Coppini, E.; Pucci, S.; Gonnelli, C.; Lepri, L.; Del Bubba, M. Total and hexavalent chromium removal in a subsurface horizontal flow (h-SSF) constructed wetland operating as post-treatment of textile wastewater for water reuse. Water Sci. Technol. 2011, 64, 826–831. [Google Scholar] [CrossRef]
- Soroko, M. Treatment of wastewater from small slaughterhouse in hybrid constructed wetlands systems. In Ecohydrology and Hydrobiology; Polish Academy of Sciences: Warsaw, Poland, 2007; pp. 339–343. [Google Scholar] [CrossRef]
- Olguín, E.J.; Sánchez-Galván, G.; González-Portela, R.E.; López-Vela, M. Constructed wetland mesocosms for the treatment of diluted sugarcane molasses stillage from ethanol production using Pontederia sagittata. Water Res. 2008, 42, 3659–3666. [Google Scholar] [CrossRef]
- Alayu, E.; Leta, S. Post treatment of anaerobically treated brewery effluent using pilot scale horizontal subsurface flow constructed wetland system. Bioresour. Bioprocess. 2021, 8, 8. [Google Scholar] [CrossRef]
- Comino, E.; Riggio, V.; Rosso, M. Mountain cheese factory wastewater treatment with the use of a hybrid constructed wetland. Ecol. Eng. 2011, 37, 1673–1680. [Google Scholar] [CrossRef]
- Kumar, S.; Choudhary, A.K. Constructed Wetland Technology for Pulp and Paper Mill Wastewater Treatment. In Constructed Wetlands for Industrial Wastewater Treatment; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2018; pp. 309–325. [Google Scholar] [CrossRef]
- Justin, M.Z.; Vrhovšek, D.; Stuhlbacher, A.; Bulc, T.G. Treatment of wastewater in hybrid constructed wetland from the production of vinegar and packaging of detergents. Desalination 2009, 246, 100–109. [Google Scholar] [CrossRef]
- Avellan, C.T.; Ardakanian, R.; Gremillion, P. The role of constructed wetlands for biomass production within the water-soil-waste nexus. Water Sci. Technol. 2017, 75, 2237–2245. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.E.; Bin Halmi MI, E.; Bin Abd Samad, M.Y.; Uddin, M.K.; Mahmud, K.; Abd Shukor, M.Y.; Abdullah, S.R.S.; Shamsuzzaman, S.M. Design, Operation and Optimization of Constructed Wetland for Removal of Pollutant. Int. J. Environ. Res. Public Health 2020, 17, 8339. [Google Scholar] [CrossRef] [PubMed]
- Stefanakis, A.; Akratos, C.S.; Tsihrintzis, V.A. Introduction. In Vertical Flow Constructed Wetlands; Elsevier: Amsterdam, The Netherlands, 2014; pp. 1–16. [Google Scholar] [CrossRef]
- Makopondo, R.O.B.; Rotich, L.K.; Kamau, C.G. Potential Use and Challenges of Constructed Wetlands for Wastewater Treatment and Conservation in Game Lodges and Resorts in Kenya. Sci. World J. 2020, 2020, 9184192. [Google Scholar] [CrossRef] [PubMed]
- Milani, M.; Marzo, A.; Toscano, A.; Consoli, S.; Cirelli, G.L.; Ventura, D.; Barbagallo, S. Evapotranspiration from horizontal subsurface flow constructed wetlands planted with different perennial plant species. Water 2019, 11, 2159. [Google Scholar] [CrossRef]
- Ramos-Arcos, S.A.; González-Mondragón, E.G.; López-Hernández, E.S.; Rodríguez-Luna, A.R.; Morales-Bautista, C.M.; Lagunas-Rivera, S.; López-Martínez, S. Phytoremediation Potential of Chrysopogon zizanioides for Toxic Elements in Contaminated Matrices. In Biodegradation Technology of Organic and Inorganic Pollutants; IntechOpen: Rijeka, Croatia, 2022. [Google Scholar] [CrossRef]
- Danh, L.T.; Truong, P.; Mammucari, R.; Tran, T.; Foster, N. Vetiver grass, Vetiveria zizanioides: A choice plant for phytoremediation of heavy metals and organic wastes. Int. J. Phytoremediation 2009, 11, 664–691. [Google Scholar] [CrossRef]
- Fasani, E.; DalCorso, G.; Zerminiani, A.; Ferrarese, A.; Campostrini, P.; Furini, A. Phytoremediatory efficiency of Chrysopogon zizanioides in the treatment of landfill leachate: A case study. Environ. Sci. Pollut. Res. Int. 2019, 26, 10057–10069. [Google Scholar] [CrossRef]
- Goren, A.Y.; Yucel, A.; Sofuoglu, S.C.; Sofuoglu, A. Phytoremediation of olive mill wastewater with Vetiveria zizanioides (L.) Nash and Cyperus alternifolius L. Environ. Technol. Innov. 2021, 24, 102071. [Google Scholar] [CrossRef]
- Seroja, R.; Effendi, H.; Hariyadi, S. Tofu wastewater treatment using vetiver grass (Vetiveria zizanioides) and zeliac. Appl. Water Sci. 2018, 8, 2. [Google Scholar] [CrossRef]
- Burger, P.; Landreau, A.; Watson, M.; Janci, L.; Cassisa, V.; Kempf, M.; Azoulay, S.; Fernandez, X. Vetiver Essential Oil in Cosmetics: What Is New? Medicines 2017, 4, 41. [Google Scholar] [CrossRef]
- Darajeh, N.; Truong, P.; Rezania, S.; Alizadeh, H.; Leung, D.W.M. Effectiveness of Vetiver grass versus other plants for phytoremediation of contaminated water. J. Environ. Treat. Tech. 2019, 7, 485–500. [Google Scholar]
- Nguyen, M.K.; Hung, N.T.Q.; Nguyen, C.M.; Lin, C.; Nguyen, T.A.; Nguyen, H.-L. Application of vetiver grass (Vetiveria zizanioides L.) for organic matter removal from contaminated surface water. Bioresour. Technol. Reports 2023, 22, 101431. [Google Scholar] [CrossRef]
- Badejo, A.A.; Omole, D.O.; Ndambuki, J.M.; Kupolati, W.K. Municipal wastewater treatment using sequential activated sludge reactor and vegetated submerged bed constructed wetland planted with Vetiveria zizanioides. Ecol. Eng. 2017, 99, 525–529. [Google Scholar] [CrossRef]
- Hemalatha, G.; Uma, S.G.; Muthulakshmi, S. Sewage water treatment using vetiver grass. Mater. Today Proc. 2020, 46, 3795–3798. [Google Scholar] [CrossRef]
- Mudhiriza, T.; Mapanda, F.; Mvumi, B.M.; Wuta, M. Removal of nutrient and heavy metal loads from sewage effluent using vetiver grass, Chrysopogon zizanioides (L.) Roberty. Water SA 2015, 41, 457–463. [Google Scholar] [CrossRef]
- Banerjee, R.; Goswami, P.; Lavania, S.; Mukherjee, A.; Lavania, U.C. Vetiver grass is a potential candidate for phytoremediation of iron ore mine spoil dumps. Ecol. Eng. 2019, 132, 120–136. [Google Scholar] [CrossRef]
- Chintani, Y.S.; Butarbutar, E.S.; Nugroho, A.P.; Sembiring, T. Uptake and release of chromium and nickel by Vetiver grass (Chrysopogon zizanioides (L.) Roberty). SN Appl. Sci. 2021, 3, 285. [Google Scholar] [CrossRef]
- Kriti, B.N.; Singh, J.; Kumari, B.; Sinam, G.; Gautam, A.; Singh, G.; Swapnil, M.K.; Mallick, S. Nickel and cadmium phytoextraction efficiencies of vetiver and lemongrass grown on Ni–Cd battery waste contaminated soil: A comparative study of linear and nonlinear models. J. Environ. Manag. 2021, 295, 113144. [Google Scholar] [CrossRef]
- Raharjo, S.; Fitriyah Irmawati, E.S.; Manaf, M. Constructed Wetland with Flow Water Surface Type for Elimination of Aquaculture Wastewater from Catfish (Clarias gariepinus, var). IOP Conf. Ser. Earth Environ. Sci. 2018, 187, 012061. [Google Scholar] [CrossRef]
- Liao, X.; Luo, S.; Wu, Y.; Wang, Z. Studies on the abilities of Vetiveria zizanioides and Cyperus alternifolius for pig farm wastewater treatment. Int. Conf. Vetiver Exhib. 2003, 3, 174–181. [Google Scholar]
- Pongthornpruek, S. Treatment of Piggery Wastewater by Three Grass Species Growing in a Constructed Wetland. Appl. Environ. Res. 2017, 39, 75–83. [Google Scholar] [CrossRef]
- Yeboah, S.; Allotey, A.; Biney, E. Purification of industrial wastewater with vetiver grasses (Vetiveria zizanioides): The case of food and beverages wastewater in Ghana. Asian J. Basic Appl. Sci. 2015, 2, 1–14. [Google Scholar]
- Njau, K.N.; Mlay, H. Wastewater Treatment and other Research Initiatives with Vetiver Grass; University of Dar es Salaam: Ubungo, Tanzania, 2003. [Google Scholar]
- Roongtanakiat, N.; Tangruangkiat, S.; Meesat, R. Utilization of vetiver grass (Vetiveria zizanioides) for removal of heavy metals from industrial wastewaters. Sci. Asia 2007, 33, 397–403. [Google Scholar] [CrossRef]
- Davamani, V.; Indhu Parameshwari, C.; Arulmani, S.; Ezra John, J.; Poornima, R. Hydroponic phytoremediation of paperboard mill wastewater by using vetiver (Chrysopogon zizanioides). J. Environ. Chem. Eng. 2021, 9, 105528. [Google Scholar] [CrossRef]
- Panja, S.; Sarkar, D.; Datta, R. Vetiver grass (Chrysopogon zizanioides) is capable of removing insensitive high explosives from munition industry wastewater. Chemosphere 2018, 209, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Mohan, T.K.; Nancharaiah, Y.V.; Venugopalan, V.P.; Sai, P.S. Effect of C/N ratio on denitrification of high-strength nitrate wastewater in anoxic granular sludge sequencing batch reactors. Ecol. Eng. 2016, 91, 441–448. [Google Scholar] [CrossRef]
- Laber, J.; Perfler, R.; Haberl, R. Two strategies for advanced nitrogen elimination in vertical flow constructed wetlands. Water Sci. Technol. 1997, 35, 71–77. [Google Scholar] [CrossRef]
- Tee, H.C.; Lim, P.E.; Seng, C.E.; Nawi, M.A.M. Newly developed baffled subsurface-flow constructed wetland for the enhancement of nitrogen removal. Bioresour. Technol. 2012, 104, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Sadegh, H.; Ali, G.A.M.; Gupta, V.K.; Makhlouf, A.S.H.; Shahryari-ghoshekandi, R.; Nadagouda, M.N.; Sillanpää, M.; Megiel, E. The role of nanomaterials as effective adsorbents and their applications in wastewater treatment. J. Nanostructure Chem. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Crini, G. Non-conventional low-cost adsorbents for dye removal: A review. Bioresour. Technol. 2006, 97, 1061–1085. [Google Scholar] [CrossRef]
- Crini, G. Recent developments in polysaccharide-based materials used as adsorbents in wastewater treatment. Prog. Polym. Sci. 2005, 30, 38–70. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E.; Wilson, L.D.; Morin-Crini, N. Conventional and non-conventional adsorbents for wastewater treatment. Environ. Chem. Lett. 2019, 17, 195–213. [Google Scholar] [CrossRef]
- Ali, M.E.; Hoque, M.E.; Safdar Hossain, S.K.; Biswas, M.C. Nanoadsorbents for wastewater treatment: Next generation biotechnological solution. Int. J. Environ. Sci. Technol. 2020, 17, 4095–4132. [Google Scholar] [CrossRef]
- Dlamini, D.S.; Tesha, J.M.; Vilakati, G.D.; Mamba, B.B.; Mishra, A.K.; Thwala, J.M.; Li, J. A critical review of selected membrane- and powder-based adsorbents for water treatment: Sustainability and effectiveness. J. Clean. Prod. 2020, 277, 123497. [Google Scholar] [CrossRef]
- Renu, A.M.; Singh, K. Heavy metal removal from wastewater using various adsorbents: A review. J. Water Reuse Desalin. 2017, 7, 387–419. [Google Scholar] [CrossRef]
- Phothong, K.; Tangsathitkulchai, C.; Lawtae, P. The analysis of pore development and formation of surface functional groups in bamboo-based activated carbon during CO2 activation. Molecules 2021, 26, 5641. [Google Scholar] [CrossRef] [PubMed]
- Vasiraja, N.; Saravana Sathiya Prabhahar, R.; Joshua, A. Preparation and Physio–Chemical characterisation of activated carbon derived from Prosopis juliflora stem for the removal of methylene blue dye and heavy metal containing textile industry effluent. J. Clean. Prod. 2023, 397, 136579. [Google Scholar] [CrossRef]
- Hatt, J.W.; Germain, E.; Judd, S.J. Granular activated carbon for removal of organic matter and turbidity from secondary wastewater. Water Sci. Technol. 2013, 67, 846–853. [Google Scholar] [CrossRef]
- Lakdawala, M.M.; Lakdawala, J.M. Comparative Study of Effect of PAC and GAC on Removal of COD Contributing Component of Sugar Industry waste water. Res. J. Recent Sci. 2013, 2, 90–97. [Google Scholar]
- Subki, N.S.; Akhir, N.M.; Abdul Halim, N.S.; Nik Yusoff, N.R. COD Reduction in Industrial Wastewater Using Activated Carbon Derived from Wodyetia Bifurcata Fruit. IOP Conf. Ser. Earth Environ. Sci. 2020, 549, 012066. [Google Scholar] [CrossRef]
- Almanassra, I.W.; Kochkodan, V.; Mckay, G.; Atieh, M.A.; Al-Ansari, T. Review of phosphate removal from water by carbonaceous sorbents. J. Environ. Manag. 2021, 287, 112245. [Google Scholar] [CrossRef]
- Zhao, Q.; Han, H.; Xu, C.; Zhuang, H.; Fang, F.; Zhang, L. Effect of powdered activated carbon technology on short-cut nitrogen removal for coal gasification wastewater. Bioresour. Technol. 2013, 142, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Ani, J.U.; Okoro, U.C.; Aneke, L.E.; Onukwuli, O.D.; Obi, I.O.; Akpomie, K.G.; Ofomatah, A.C. Application of response surface methodology for optimization of dissolved solids adsorption by activated coal. Appl. Water Sci. 2019, 9, 60. [Google Scholar] [CrossRef]
- Kuptajit, P.; Sano, N.; Nakagawa, K.; Suzuki, T. A study on pore formation of high surface area activated carbon prepared by microwave-induced plasma with KOH (MiWP-KOH) activation: Effect of temperature-elevation rate. Chem. Eng. Process.—Process Intensif. 2021, 167, 108511. [Google Scholar] [CrossRef]
- Radhi, A.A. Comparison of granulated and powdered activated carbon in the removal of organic matter from river water. Int. Res. J. Adv. Eng. Sci. 2020, 5, 191–197. [Google Scholar]
- Newcombe, G. Chapter 8: Removal of natural organic material and algal metabolites using activated carbon. Interface Sci. Technol. 2006, 10, 133–153. [Google Scholar] [CrossRef]
- Upadhyayula, S.; Chaudhary, A. Advanced Materials and Technologies for Wastewater Treatment, 1st ed.; CRC Press: Boca Raton, FL, USA, 2021. [Google Scholar] [CrossRef]
- Soni, R.; Bhardwaj, S.; Shukla, D.P. Various water-treatment technologies for inorganic contaminants: Current status and future aspects. In Inorganic Pollutants in Water; Elsevier: Amsterdam, The Netherlands, 2020; pp. 273–295. [Google Scholar] [CrossRef]
- Wilcox, D.P.; Chang, E.; Dickson, K.L.; Johansson, K.R. Microbial growth associated with granular activated carbon in a pilot water treatment facility. Appl. Environ. Microbiol. 1983, 46, 406. [Google Scholar] [CrossRef]
- Marczewski, A.W.; Seczkowska, M.; Deryło-Marczewska, A.; Blachnio, M. Adsorption equilibrium and kinetics of selected phenoxyacid pesticides on activated carbon: Effect of temperature. Adsorption 2016, 22, 777–790. [Google Scholar] [CrossRef]
- Nizam, N.U.M.; Hanafiah, M.M.; Mahmoudi, E.; Halim, A.A.; Mohammad, A.W. The removal of anionic and cationic dyes from an aqueous solution using biomass-based activated carbon. Sci. Rep. 2021, 11, 8623. [Google Scholar] [CrossRef]
- Lee, S.H.; Nishijima, W.; Lee, C.H.; Okada, M. Calcium accumulation on activated carbon deteriorates synthetic organic chemicals adsorption. Water Res. 2003, 37, 4631–4636. [Google Scholar] [CrossRef]
- Soltani, R.D.C.; Safari, M.; Rezaee, A.; Godini, H. Application of a compound containing silica for removing ammonium in aqueous media. Environ. Prog. Sustain. Energy 2015, 34, 105–111. [Google Scholar] [CrossRef]
- Mokhatab, S.; Poe, W.A.; Mak, J.Y. Natural Gas Dehydration and Mercaptans Removal. In Handbook of Natural Gas Transmission and Processing; Elsevier: Amsterdam, The Netherlands, 2019; pp. 307–348. [Google Scholar] [CrossRef]
- Kalam, S.; Abu-Khamsin, S.A.; Kamal, M.S.; Patil, S. Surfactant Adsorption Isotherms: A Review. ACS Omega 2021, 6, 32342. [Google Scholar] [CrossRef] [PubMed]
- Ribas, M.C.; Adebayo, M.A.; Prola, L.D.T.; Lima, E.C.; Cataluña, R.; Feris, L.A.; Puchana-Rosero, M.J.; Machado, F.M.; Pavan, F.A.; Calvete, T. Comparison of a homemade cocoa shell activated carbon with commercial activated carbon for the removal of reactive violet 5 dye from aqueous solutions. Chem. Eng. J. 2014, 248, 315–326. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Largitte, L.; Pasquier, R. A review of the kinetics adsorption models and their application to the adsorption of lead by an activated carbon. Chem. Eng. Res. Des. 2016, 109, 495–504. [Google Scholar] [CrossRef]
- Kajjumba, G.W.; Emik, S.; Öngen, A.; Kurtulus Özcan, H.; Aydın, S. Modelling of Adsorption Kinetic Processes—Errors, Theory and Application. In Advanced Sorption Process Applications; IntechOpen: Rijeka, Croatia, 2019. [Google Scholar] [CrossRef]
- Patel, H. Charcoal as an adsorbent for textile wastewater treatment. Sep. Sci. Technol. 2018, 53, 2797–2812. [Google Scholar] [CrossRef]
- Hsieh, C.T.; Teng, H. Influence of mesopore volume and adsorbate size on adsorption capacities of activated carbons in aqueous solutions. Carbon N. Y. 2000, 38, 863–869. [Google Scholar] [CrossRef]
- Rashed, M.N. Adsorption Technique for the Removal of Organic Pollutants from Water and Wastewater. In Organic Pollutants—Monitoring, Risk and Treatment; IntechOpen: Rijeka, Croatia, 2013. [Google Scholar] [CrossRef]
- Sadegh, H.; Ali, G.A.M. Potential Applications of Nanomaterials in Wastewater Treatment. In Advanced Treatment Techniques for Industrial Wastewater; IGI Global: Hershey, PA, USA, 2018; pp. 51–61. [Google Scholar] [CrossRef]
- Yegane, B.M.; Azari, A.; Pasalari, H.; Esrafili, A.; Farzadkia, M. Modification of activated carbon with magnetic Fe3O4 nanoparticle composite for removal of ceftriaxone from aquatic solutions. J. Mol. Liq. 2018, 261, 146–154. [Google Scholar] [CrossRef]
- Do, M.H.; Phan, N.H.; Nguyen, T.D.; Pham, T.T.S.; Nguyen, V.K.; Vu, T.T.T.; Nguyen, T.K.P. Activated carbon/Fe3O4 nanoparticle composite: Fabrication, methyl orange removal and regeneration by hydrogen peroxide. Chemosphere 2011, 85, 1269–1276. [Google Scholar] [CrossRef]
- Borghi, C.C.; Fabbri, M. Magnetic recovery of modified activated carbon powder used for removal of endocrine disruptors present in water. Environ. Technol. 2014, 35, 1018–1026. [Google Scholar] [CrossRef]
- Khajeh, M.; Laurent, S.; Dastafkan, K. Nanoadsorbents: Classification, preparation, and applications (with emphasis on aqueous media). Chem. Rev. 2013, 113, 7728–7768. [Google Scholar] [CrossRef]
- Aragaw, T.A.; Bogale, F.M.; Aragaw, B.A. Iron-based nanoparticles in wastewater treatment: A review on synthesis methods, applications, and removal mechanisms. J. Saudi Chem. Soc. 2021, 25, 101280. [Google Scholar] [CrossRef]
- Besenhard, M.O.; LaGrow, A.P.; Hodzic, A.; Kriechbaum, M.; Panariello, L.; Bais, G.; Loizou, K.; Damilos, S.; Margarida Cruz, M.; Thanh, N.T.K.; et al. Co-precipitation synthesis of stable iron oxide nanoparticles with NaOH: New insights and continuous production via flow chemistry. Chem. Eng. J. 2020, 399, 125740. [Google Scholar] [CrossRef]
- Kumari, P.; Alam, M.; Siddiqi, W.A. Usage of nanoparticles as adsorbents for waste water treatment: An emerging trend. Sustain. Mater. Technol. 2019, 22, e00128. [Google Scholar] [CrossRef]
- Shen, J.; He, R.; Han, W.; Sun, X.; Li, J.; Wang, L. Biological denitrification of high-nitrate wastewater in a modified anoxic/oxic-membrane bioreactor (A/O-MBR). J. Hazard. Mater. 2009, 172, 595–600. [Google Scholar] [CrossRef]
- Barasarathi, J.; Abdullah, P.S.; Uche, E.C. Application of magnetic carbon nanocomposite from agro-waste for the removal of pollutants from water and wastewater. Chemosphere 2022, 305, 135384. [Google Scholar] [CrossRef]
- Vinayagam, R.; Pai, S.; Murugesan, G.; Varadavenkatesan, T.; Narayanasamy, S.; Selvaraj, R. Magnetic activated charcoal/Fe2O3 nanocomposite for the adsorptive removal of 2,4-Dichlorophenoxyacetic acid (2,4-D) from aqueous solutions: Synthesis, characterization, optimization, kinetic and isotherm studies. Chemosphere 2022, 286, 131938. [Google Scholar] [CrossRef]
- Abdullah, N.H.; Shameli, K.; Abdullah, E.C.; Abdullah, L.C. Solid matrices for fabrication of magnetic iron oxide nanocomposites: Synthesis, properties, and application for the adsorption of heavy metal ions and dyes. Compos. Part B Eng. 2019, 162, 538–568. [Google Scholar] [CrossRef]
- Jain, M.; Yadav, M.; Kohout, T.; Lahtinen, M.; Garg, V.K.; Sillanpää, M. Development of iron oxide/activated carbon nanoparticle composite for the removal of Cr(VI), Cu(II) and Cd(II) ions from aqueous solution. Water Resour. Ind. 2018, 20, 54–74. [Google Scholar] [CrossRef]
- Safarik, I.; Horska, K.; Pospiskova, K.; Safarikova, M. Magnetically Responsive Activated Carbons for Bio—And Environmental Applications. Int. Rev. Chem. Eng. 2012, 4, 346–352. [Google Scholar]
- Lompe, K.M.; Menard, D.; Barbeau, B. The influence of iron oxide nanoparticles upon the adsorption of organic matter on magnetic powdered activated carbon. Water Res. 2017, 123, 30–39. [Google Scholar] [CrossRef]
- Park, H.S.; Koduru, J.R.; Choo, K.H.; Lee, B. Activated carbons impregnated with iron oxide nanoparticles for enhanced removal of bisphenol A and natural organic matter. J. Hazard. Mater. 2015, 286, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Vargues, F.; Brion, M.A.; Rosa da Costa, A.M.; Moreira, J.A.; Ribau Teixeira, M. Development of a magnetic activated carbon adsorbent for the removal of common pharmaceuticals in wastewater treatment. Int. J. Environ. Sci. Technol. 2021, 18, 2805–2818. [Google Scholar] [CrossRef]

| Type of Effluent | Operating Conditions | Initial Physicochemical Characteristics | Final Physicochemical Characteristics | References |
|---|---|---|---|---|
| Vinasse from the sugarcane ethanol industry | V = 2.6 L A = 162 cm2 Room temperature = 16.8 ± 2.3 °C Without agitation, air injection, and reagent addition With or without precipitate | Without precipitate: pH = 10.6 Conductivity ≈ 10 mS cm−1 Ca ≈ 2400 mg L−1 | After 15 days: pH ≈ 8 Conductivity ≈ 10.8 mS cm−1 Ca ≈ 2400 mg L−1 | [70] |
| With precipitate: pH = 12.34 | After 20 days: pH ≈ 8 | |||
| Without precipitate: pH = 12.05 | After 9.2 days: pH ≈ 8 | |||
| Winery wastewater | V = 1 L Without agitation, air injection, and reagent addition Without precipitate | pH =12.4 Conductivity = 6.5 mS cm−1 Ca = 499.8 mg L−1 Mg = 179.9 mg L−1 | After 15 days: pH = 7.46 Conductivity = 1.805 mS cm−1 Ca = 426.5 mg L−1 Mg = 6.6 mg L−1 | [46] |
| Cheese whey wastewater | V = 3.5 L A = 162 cm2 Without agitation, air injection, and reagent addition Without precipitate | pH ≈ 12 Conductivity ≈ 4.75 mS cm−1 Ca ≈ 300 mg L−1 Mg ≈ 13 mg L−1 | After 7.3 days: pH ≈ 8 Conductivity ≈ 4 mS cm−1 Ca ≈ 200 mg L−1 Mg ≈ 2 mg L−1 | [54] |
| Landfill leachate | V = 3 to 4 L A = 200 cm2 Without agitation, air injection, and reagent addition Without precipitate | pH = 12.5 NH4+ = 889 mg N L−1 Conductivity = 23.1 mS cm−1 Calcium Hardness = 490 mg CaCO3 L−1 P. alkalinity = 6600 mg CaCO3 L−1 Total alkalinity = 7530 mg CaCO3 L−1 COD = 460 mg O2 L−1 | After 32 days: pH = 10.10 NH4+ < 0.1 mg N L−1 Conductivity = 15.0 mS cm−1 Calcium Hardness < 0.1 mg CaCO3 L−1 P. alkalinity = 2350 mg CaCO3 L−1 Total alkalinity = 4480 mg CaCO3 L−1 COD = 474 mg O2 L−1 | [47] |
| Urban wastewater | V = 4 L A = 200 cm2 Without agitation and reagent addition With precipitate With or without air injection | Without air injection: pH ≈ 11.5 Conductivity = 1.144 mS cm−1 | After 9.2 days: pH = 8.4 Conductivity ≈ 1.050 mS cm−1 | [48] |
| With air injection (85 L h−1): pH ≈ 11.5 Conductivity ≈ 1.300 mS cm−1 | After 4.2 days: pH ≈ 8 Conductivity ≈ 1.170 mS cm−1 | |||
| Explosives wastewater | V = 5 L A = 189 cm2 Room temperature = 24.5 ± 2.0 °C Without agitation, air injection, and reagent addition With precipitate | pH = 10.3 NH4+ = 1505 mg N L−1 Conductivity = 9 mS cm−1 Ca = 1500 mg L−1 Mg = 52.5 mg L−1 P. alkalinity = 3674 mg CaCO3 L−1 Total alkalinity = 3923 mg CaCO3 L−1 | After 10 days: pH = 8 NH4+ = 578 mg N L−1 Conductivity = 10.3 mS cm−1 Ca = 1626 mg L−1 Mg = 36.2 mg L−1 P. Alkalinity = 62 mg CaCO3 L−1 Total alkalinity = 125 mg CaCO3 L−1 | [51] |
| Slaughterhouse wastewater | A/V ratio = 5 and 155.4 m2/m3 Without agitation, air injection, and reagent addition Without precipitate | With A/V = 5 m2/m3: pH = 11.9 Conductivity = 3.12 mS cm−1 Ca = 297.6 mg L−1 Mg = 43.3 mg L−1 NH4+ = 69 mg N L−1 | After 13 days: pH = 7.9 Conductivity = 2.46 mS cm−1 Ca = 130.9 mg L−1 Mg = 20.2 mg L−1 NH4+ = 22 mg N L−1 | [52] |
| With A/V = 155.4 m2/m3: pH = 11.9 Conductivity = 3.12 mS cm−1 Ca = 297.6 mg L−1 Mg = 43.3 mg L−1 NH4+ = 69 mg N L−1 | After 1 day: pH = 8.2 Conductivity = 2.40 mS cm−1 Ca = 60.2 mg L−1 Mg = 31.4 mg L−1 NH4+ = 12 mg N L− |
| Wastewater Types | Removals | Reference |
|---|---|---|
| Seafood wastewater | BOD (91–99%), TSS (52–90%), TN (72–92%), and TP (72–77%) | [140] |
| Winery wastewater | BOD (70%), COD (71%), TSS (87%), TKN (52%), and PO43− (17%) | [118] |
| Tannery wastewater | BOD (98%), COD (98%), TSS (55%), TP (87%), and NH4+ (86%) | [141] |
| Steel industry wastewater | COD (77%), NH4+ (77%), Fe (94%), and Mn (81%) | [142] |
| Refinery wastewater | COD (80%), oil (93%), BOD (88%), and TKN (86%) | [143] |
| Aquaculture wastewater | COD (27%), TSS (66%), TN (67%), TP (24%), and NO3− (59%) | [144] |
| Textile wastewater | Cr (40–50%) | [145] |
| Abattoir wastewater | BOD (97%), COD (97%), TSS (94%), TN (74%), and NH4+ (99%) | [146] |
| Distillery wastewater | BOD (85%), COD (80%), TKN (75%), NO3− (57%), NH4+ (2–10%), and SO42− (69%) | [147] |
| Brewery wastewater | TSS (89%), COD (92%), TN (83.6%), NH4+ (92.9%), TP (74.4%), and PO43− (79.5%) | [148] |
| Cheese wastewater | BOD (55%), COD (72%), TSS (60%), TP (30%), and TN (50%) | [149] |
| Olive mill wastewater | COD (85%), TSS (90%), TP (83%), TKN (83%), Phenol (80%), NH4+ (54%), and NO3− (46%) | [124] |
| Pulp and paper wastewater | COD (88%), color (96%), BOD (93%), and chlorophenols (90%) | [150] |
| Mixed industrial wastewater | BOD (66%), COD (67%), NH4+ (24%), organic-N (83%), and PO43− (62%) | [151] |
| Wastewater Types | CW Type | Media | HRT (d) | HL (m3 d−1) | Initial Concentrations | Removal Performance (%) | Country | References |
|---|---|---|---|---|---|---|---|---|
| Aquaculture Wastewater | FWSCW (2 m × 1 m × 0.5 m) | Coarse sand and corals | 1 | 0.576 | NH3 (0.1–0.2 mg L−1), PO43− (6–10 mg L−1) | NH3 (2–67%), PO43− (0–75%) | Indonesia | [171] |
| Piggery Wastewater | FWSCW (1 m × 3 m × 1 m) | Soil | 5 | 0.18 | BOD (767.90 mg L−1), COD (1330.25 mg L−1), TKN (158.67 mg L−1), TP (69.90 mg L−1) | BOD (74%), COD (70%), TKN (88%), TP (83%) | Thailand | [173] |
| Pinora Wastewater | FWSCW | - | 120 | - | TSS (385 mg L−1), BOD (4836 mg L−1), COD (6296 mg L−1), NO3− (0.721 mg L−1), NH3 (9.69 mg L−1) | TSS (82%), BOD (94%), COD (86%), NO3− (10%), NH3 (41%) | Ghana | [174] |
| Palm Oil Mill Wastewater | FWSCW | - | 120 | - | TSS (278,600 mg L−1), BOD (44,520 mg L−1), COD (128,911 mg L−1), NO3− (0.80 mg L−1), NH3 (20.40 mg L−1) | TSS (71%), BOD (51%), COD (10%), NO3− (6%), NH3 (40%) | Ghana | [174] |
| Biogas Wastewater | FWSCW | - | 120 | - | TSS (330 mg L−1), BOD (492 mg L−1), COD (1952 mg L−1), NO3− (0.122 mg L−1), NH3 (17.3 mg L−1) | TSS (95%), BOD (91%), COD (82%), NO3− (99%), NH3 (42%) | Ghana | [174] |
| Pig farm Wastewater | FWSCW (50 cm × 38.5 cm × 23 cm) | - | 4 | - | COD (825 mg L−1), BOD5 (500 mg L−1), NH3 (130 mg N L−1), TP (23 mg L−1) | COD (64%), BOD (69%), NH3 (20%), TP (27%) | China | [172] |
| Olive mill wastewater | FWSCW | - | 67 | - | TOC (1132 mg L−1), TN (26.6 mg L−1) | TOC (85%), TN (93%), TP (39%) | Turkey | [160] |
| TOC (3168 mg L−1), TN (72.6 mg L−1) | TOC (89%), TN (24%), TP (92%) | |||||||
| Milk factory wastewater | FWSCW | - | 120 | - | Mn (0.49 mg L−1), Fe (16.15 mg L−1), Zn (4.09 mg L−1) Pb (0.05 mg L−1) | Mn (34%), Fe (28%), Zn (53%), Pb (9%) | Thailand | [176] |
| Tofu wastewater | FWSCW | Zeliac | 15 | - | COD (5759 mg L−1), BOD (580 mg L−1), TSS (552 mg L−1) | COD (76%), BOD (72%), TSS (75%) | Indonesia | [161] |
| Paperboard mill wastewater (raw and treated) | FWSCW | - | 40 | - | TSS (1000 mg L−1), BOD (156 mg L−1), COD (512 mg L−1), TN (39 mg L−1), TP (9.25 mg L−1), Lead (2.01 mg L−1), cadmium (1.90 mg L−1) | TSS (60%), BOD (96%), COD (50%), TN (64%), TP (65%), Lead (51%), cadmium (27%) | India | [177] |
| TSS (200 mg L−1), BOD (44 mg L−1), COD (256 mg L−1), TN (25 mg L−1) TP (8.50 mg L−1), Lead (0.96 mg L−1), cadmium (0.42 mg L−1) | TSS (75%), BOD (72%), COD (56%), TN (70%), TP (43%), Lead (91%), cadmium (81%) | |||||||
| Explosives wastewater | VFCW (40 cm × 60 cm × 70 cm) with flooding level at 25% | LECA | - | 0.02 | COD (361 mg L−1), NO3− (145 mg N L−1), NH4+ (4.8 mg N L−1) | COD (>90%), NO3− (55%), NH4+ (75%) | Portugal | [50] |
| Slaughterhouse wastewater | VFCW (40 cm × 60 cm × 70 cm) | LECA | 0.29 | 0.02 | COD (2648 mg L−1), NH4+ (48.8 mg N L−1) | COD (59–83%), NH4+ (52–65%) | Portugal | [49] |
| Textile wastewater | HFCW (1 m × 0.6 m × 0.3 m) | Limestone soil | 25 | - | TSS (100–120 mg L−1), COD (820–1200 mg L−1), BOD (226–282 mg L−1) | TSS (81%), COD (46.2%), Cu (73.6%), color (78.2%) | Tanzania | [175] |
| Munition industry wastewater | FWSCW | - | 100 | - | Nitroguanidine (3996 mg L−1) | Nitroguanidine (79%) | United States of America | [178] |
| 100 | NO3− (352,734 mg N L−1) | NO3− (95%) | ||||||
| 100 | Dinitroanisole (120 mg L−1) | Dinitroanisole (96%) | ||||||
| 20 | RDX (7.8 mg L−1), HMX (12 mg L−1) | RDX (100%), HMX (100%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Madeira, L.; Carvalho, F.; Almeida, A.; Ribau Teixeira, M. Integrated Process of Immediate One-Step Lime Precipitation, Atmospheric Carbonation, Constructed Wetlands, or Adsorption for Industrial Wastewater Treatment: A Review. Water 2023, 15, 3929. https://doi.org/10.3390/w15223929
Madeira L, Carvalho F, Almeida A, Ribau Teixeira M. Integrated Process of Immediate One-Step Lime Precipitation, Atmospheric Carbonation, Constructed Wetlands, or Adsorption for Industrial Wastewater Treatment: A Review. Water. 2023; 15(22):3929. https://doi.org/10.3390/w15223929
Chicago/Turabian StyleMadeira, Luís, Fátima Carvalho, Adelaide Almeida, and Margarida Ribau Teixeira. 2023. "Integrated Process of Immediate One-Step Lime Precipitation, Atmospheric Carbonation, Constructed Wetlands, or Adsorption for Industrial Wastewater Treatment: A Review" Water 15, no. 22: 3929. https://doi.org/10.3390/w15223929





