Adsorption of Pb2+ by Activated Carbon Produced by Microwave-Assisted K2CO3 Activation of Date Palm Leaf Sheath Fibres
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Activated Carbon (LSDPFAC) from Leaf Sheath Fibres from Date Palm
2.3. Characterisation Methods
2.4. Equilibrium Study
2.5. Isotherm Study
2.6. Kinetic Study
2.7. Thermodynamic Study
3. Results and Discussion
3.1. Characteristics of Samples
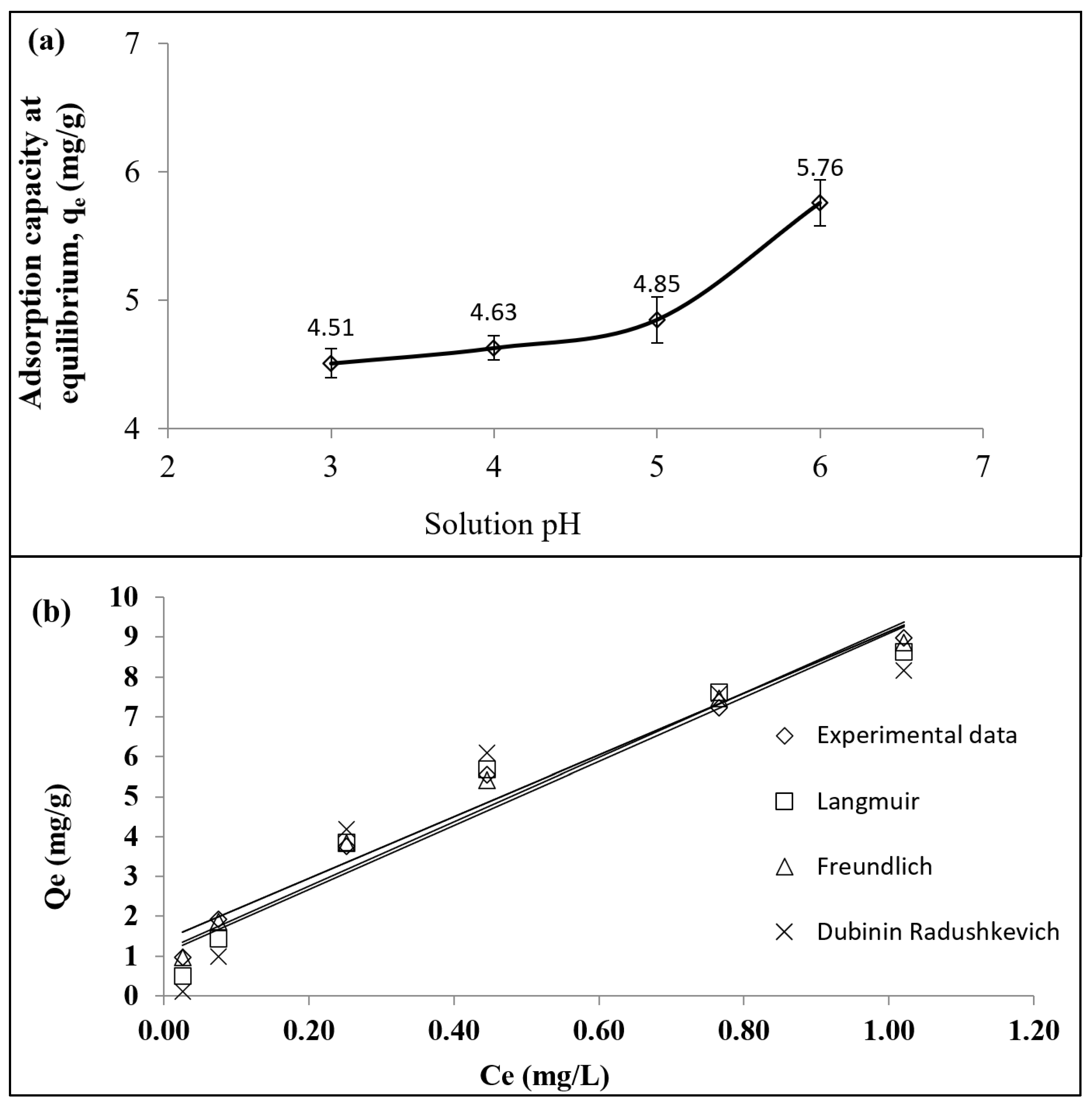
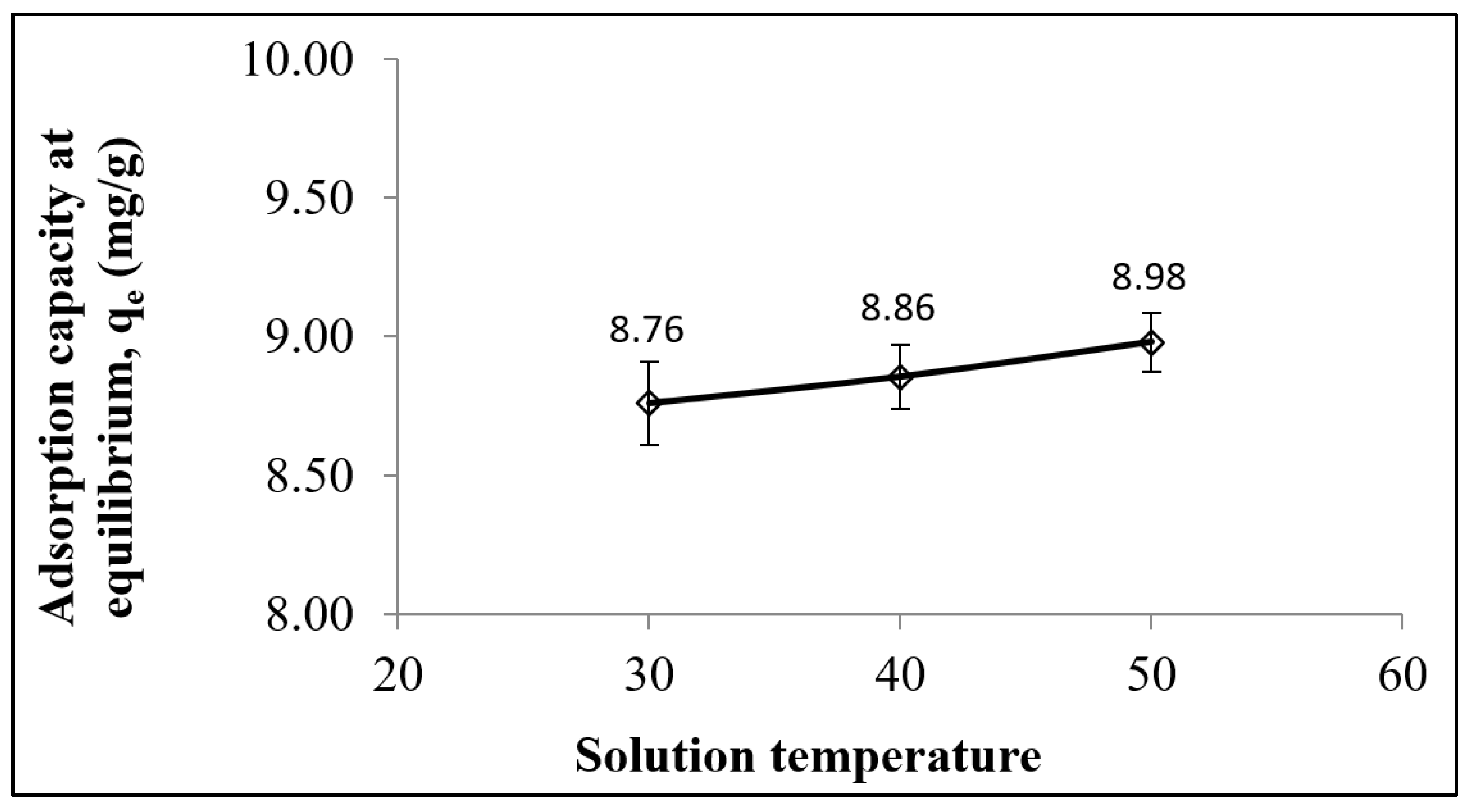
3.2. Adsorption Equilibrium
3.3. Adsorption Isotherm
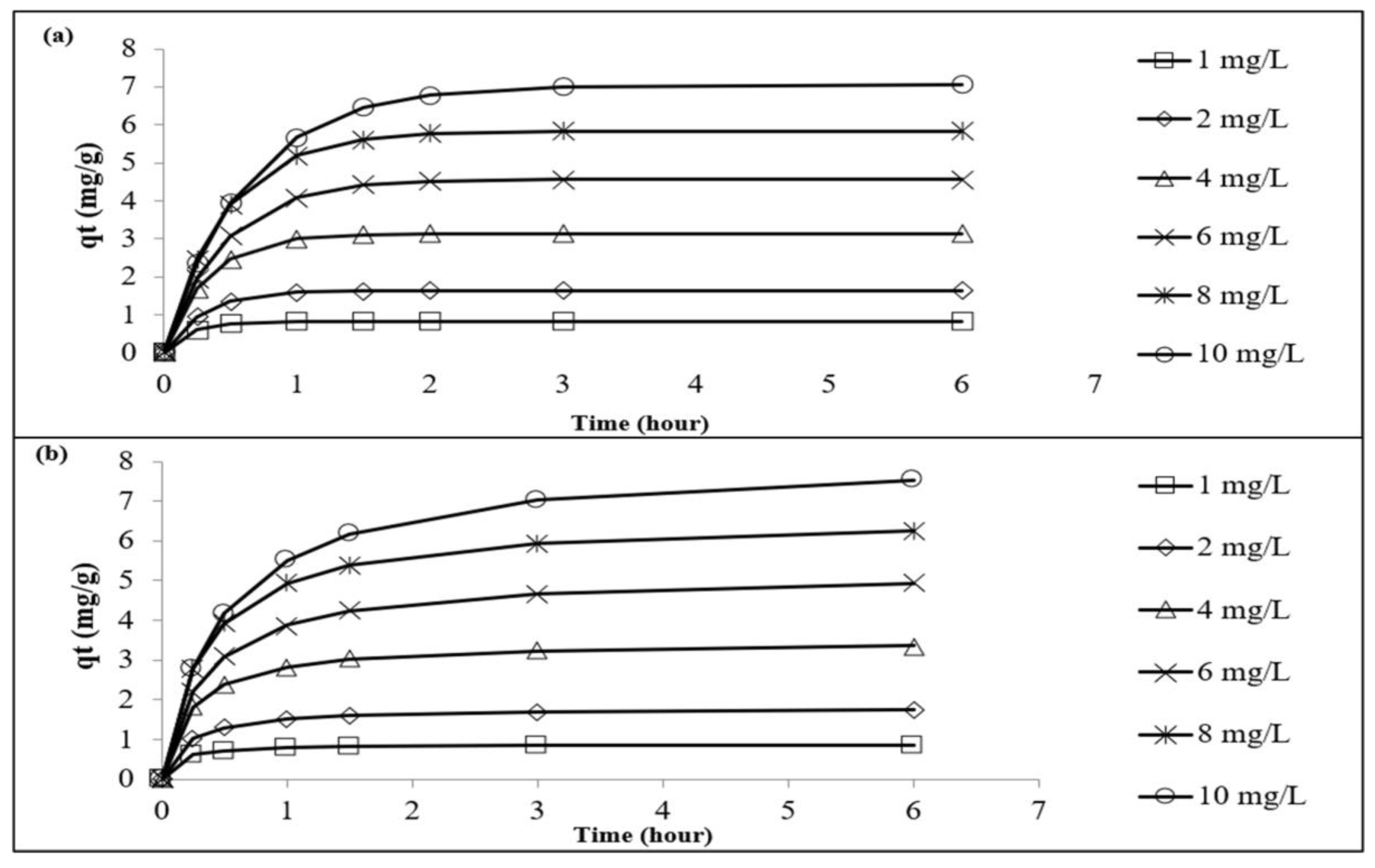
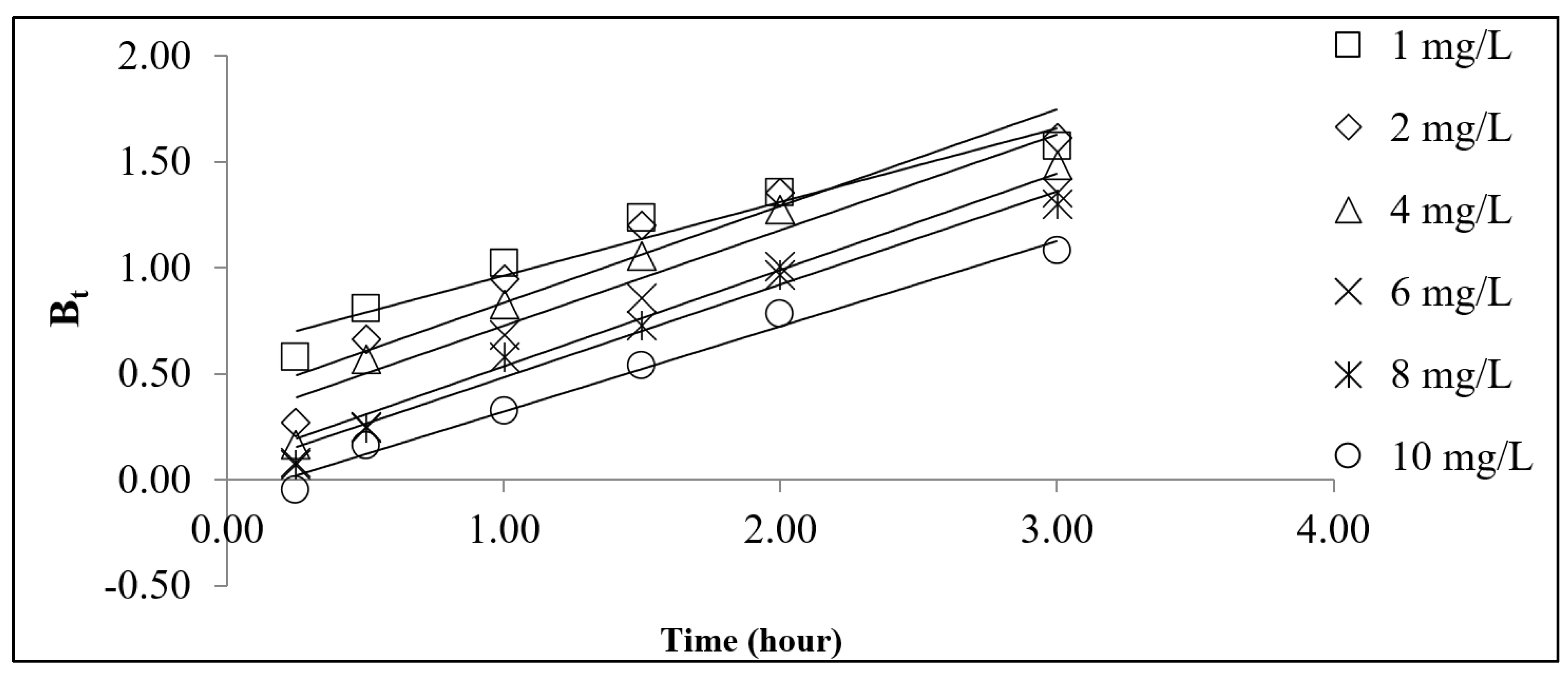
3.4. Adsorption Kinetics
3.5. Adsorption Thermodynamics
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rajendran, S.; Priya, A.K.; Senthil Kumar, P.; Hoang, T.K.A.; Sekar, K.; Chong, K.Y.; Khoo, K.S.; Ng, H.S.; Show, P.L. A critical and recent developments on adsorption technique for removal of heavy metals from wastewater—A review. Chemosphere 2022, 303, 135146. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, Y.; Chang, J. A review on the incorporation and potential mechanism of heavy metals on the recovered struvite from wastewater. Water Res. 2021, 207, 117823. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Min, X.; Tang, C.-J.; Sillanpää, M.; Zhao, F. Recent advances in membrane filtration for heavy metal removal from wastewater: A mini review. J. Water Process Eng. 2022, 49, 103023. [Google Scholar] [CrossRef]
- Iqbal, M.O.; Yahya, E.B. In vivo assessment of reversing aminoglycoside antibiotics nephrotoxicity using Jatropha mollissima crude extract. Tissue Cell 2021, 72, 101525. [Google Scholar] [CrossRef] [PubMed]
- Yusop, M.F.M.; Jaya, E.M.J.; Ahmad, M.A. Single-stage microwave assisted coconut shell based activated carbon for removal of Zn(II) ions from aqueous solution—Optimization and batch studies. Arab. J. Chem. 2022, 15, 104011. [Google Scholar] [CrossRef]
- Mirzabeygi, M.; Abbasnia, A.; Yunesian, M.; Nodehi, R.N.; Yousefi, N.; Hadi, M.; Mahvi, A.H. Heavy metal contamination and health risk assessment in drinking water of Sistan and Baluchistan, Southeastern Iran. Hum. Ecol. Risk Assess. Int. J. 2017, 23, 1893–1905. [Google Scholar] [CrossRef]
- Afroze, S.; Sen, T.K. A Review on Heavy Metal Ions and Dye Adsorption from Water by Agricultural Solid Waste Adsorbents. Water Air Soil Pollut. 2018, 229, 225. [Google Scholar] [CrossRef]
- Zeng, G.; Wan, J.; Huang, D.; Hu, L.; Huang, C.; Cheng, M.; Xue, W.; Gong, X.; Wang, R.; Jiang, D. Precipitation, adsorption and rhizosphere effect: The mechanisms for Phosphate-induced Pb immobilization in soils—A review. J. Hazard. Mater. 2017, 339, 354–367. [Google Scholar] [CrossRef]
- Rezania, S.; Mojiri, A.; Park, J.; Nawrot, N.; Wojciechowska, E.; Marraiki, N.; Zaghloul, N.S.S. Removal of lead ions from wastewater using lanthanum sulfide nanoparticle decorated over magnetic graphene oxide. Environ. Res. 2022, 204, 111959. [Google Scholar] [CrossRef]
- Goswami, L.; Arul Manikandan, N.; Pakshirajan, K.; Pugazhenthi, G. Simultaneous heavy metal removal and anthracene biodegradation by the oleaginous bacteria Rhodococcus opacus. 3 Biotech 2017, 7, 37. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Zhou, A.; Lin, X.; Bu, Y. Adsorption mechanism of Pb2+ in montmorillonite nanopore under various temperatures and concentrations. Environ. Res. 2022, 209, 112817. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Lin, Q.; Luo, S.; Ruan, K.; Peng, K. Preparation of novel oxidized mesoporous carbon with excellent adsorption performance for removal of malachite green and lead ion. Appl. Surf. Sci. 2018, 442, 322–331. [Google Scholar] [CrossRef]
- Gupta, K.M.; Zhang, K.; Jiang, J. Efficient removal of Pb2+ from aqueous solution by an ionic covalent−organic framework: Molecular simulation study. Ind. Eng. Chem. Res. 2018, 57, 6477–6482. [Google Scholar] [CrossRef]
- Xu, J.; Cao, Z.; Zhang, Y.; Yuan, Z.; Lou, Z.; Xu, X.; Wang, X. A review of functionalized carbon nanotubes and graphene for heavy metal adsorption from water: Preparation, application, and mechanism. Chemosphere 2018, 195, 351–364. [Google Scholar] [CrossRef]
- Gherasim, C.V.; Křivčík, J.; Mikulášek, P. Investigation of batch electrodialysis process for removal of lead ions from aqueous solutions. Chem. Eng. J. 2014, 256, 324–334. [Google Scholar] [CrossRef]
- Azamat, J.; Sardroodi, J.J.; Poursoltani, L.; Jahanshahi, D. Functionalized boron nitride nanosheet as a membrane for removal of Pb2+ and Cd2+ ions from aqueous solution. J. Mol. Liq. 2021, 321, 114920. [Google Scholar] [CrossRef]
- Lystvan, K.; Listvan, V.; Shcherbak, N.; Kuchuk, M. Rhizoextraction potential of convolvulus tricolor hairy roots for Cr6+, Ni2+, and Pb2+ removal from aqueous solutions. Appl. Biochem. Biotechnol. 2021, 193, 1215–1230. [Google Scholar] [CrossRef]
- Manfrin, J.; Gonçalves, A.C., Jr.; Schwantes, D.; Conradi, E., Jr.; Zimmermann, J.; Ziemer, G.L. Development of biochar and activated carbon from cigarettes wastes and their applications in Pb2+ adsorption. J. Environ. Chem. Eng. 2021, 9, 104980. [Google Scholar] [CrossRef]
- Yusop, M.F.M.; Mohd Johan Jaya, E.; Mohd Din, A.T.; Bello, O.S.; Ahmad, M.A. Single-stage optimized microwave-induced activated carbon from coconut shell for cadmium adsorption. Chem. Eng. Technol. 2022, 45, 1943–1951. [Google Scholar] [CrossRef]
- Kongsune, P.; Rattanapan, S.; Chanajaree, R. The removal of Pb2+ from aqueous solution using mangosteen peel activated carbon: Isotherm, kinetic, thermodynamic and binding energy calculation. Groundw. Sustain. Dev. 2021, 12, 100524. [Google Scholar] [CrossRef]
- Yusop, M.F.M.; Ahmad, M.A.; Rosli, N.A.; Gonawan, F.N.; Abdullah, S.J. Scavenging malachite green dye from aqueous solution using durian peel based activated carbon. Malays. J. Fundam. Appl. Sci. 2021, 17, 95–103. [Google Scholar] [CrossRef]
- Alharbi, H.A.; Hameed, B.H.; Alotaibi, K.D.; Al-Oud, S.S.; Al-Modaihsh, A.S. Recent methods in the production of activated carbon from date palm residues for the adsorption of textile dyes: A review. Front. Environ. Sci. 2022, 10, 1943–1951. [Google Scholar] [CrossRef]
- Bello, O.S.; Ahmad, M.A. Removal of remazol brilliant violet-5R dye using periwinkle shells. Chem. Ecol. 2011, 27, 481–492. [Google Scholar] [CrossRef]
- Quesada, H.B.; de Araújo, T.P.; Cusioli, L.F.; de Barros, M.A.S.D.; Gomes, R.G.; Bergamasco, R. Caffeine removal by chitosan/activated carbon composite beads: Adsorption in tap water and synthetic hospital wastewater. Chem. Eng. Res. Des. 2022, 184, 1–12. [Google Scholar] [CrossRef]
- Aziz, A.; Khan, M.N.N.; Yusop, M.F.M.; Jaya, M.J.J.; Jaya, M.A.T.; Ahmad, M.A. Single-stage microwave-assisted coconut-shell-based activated carbon for removal of dichlorodiphenyltrichloroethane (DDT) from aqueous solution: Optimization and batch studies. Int. J. Chem. Eng. 2021, 2021, 9331386. [Google Scholar] [CrossRef]
- Li, S.; Cho, M.-K.; Lee, K.; Deng, S.; Zhao, L.; Yuan, X.; Wang, J. Diamond in the rough: Polishing waste polyethylene terephthalate into activated carbon for CO2 capture. Sci. Total Environ. 2022, 834, 155262. [Google Scholar] [CrossRef]
- Mohamad Yusop, M.F.; Abdullah, A.Z.; Ahmad, M.A. Malachite green dye adsorption by jackfruit based activated carbon: Optimization, mass transfer simulation and surface area prediction. Diam. Relat. Mater. 2023, 136, 109991. [Google Scholar] [CrossRef]
- Mbarki, F.; Selmi, T.; Kesraoui, A.; Seffen, M. Low-cost activated carbon preparation from Corn stigmata fibers chemically activated using H3PO4, ZnCl2 and KOH: Study of methylene blue adsorption, stochastic isotherm and fractal kinetic. Ind. Crops Prod. 2022, 178, 114546. [Google Scholar] [CrossRef]
- Ramutshatsha-Makhwedzha, D.; Mavhungu, A.; Moropeng, M.L.; Mbaya, R. Activated carbon derived from waste orange and lemon peels for the adsorption of methyl orange and methylene blue dyes from wastewater. Heliyon 2022, 8, e09930. [Google Scholar] [CrossRef]
- Firdaus, M.Y.M.; Aziz, A.; Azmier Ahmad, M. Conversion of teak wood waste into microwave-irradiated activated carbon for cationic methylene blue dye removal: Optimization and batch studies. Arab. J. Chem. 2022, 15, 104081. [Google Scholar] [CrossRef]
- Haghbin, M.R.; Niknam Shahrak, M. Process conditions optimization for the fabrication of highly porous activated carbon from date palm bark wastes for removing pollutants from water. Powder Technol. 2021, 377, 890–899. [Google Scholar] [CrossRef]
- Ahmad, F.; Daud, W.M.A.W.; Ahmad, M.A.; Radzi, R. The effects of acid leaching on porosity and surface functional groups of cocoa (Theobroma cacao)-shell based activated carbon. Chem. Eng. Res. Des. 2013, 91, 1028–1038. [Google Scholar] [CrossRef]
- Yusop, M.F.M.; Ahmad, M.A.; Rosli, N.A.; Manaf, M.E.A. Adsorption of cationic methylene blue dye using microwave-assisted activated carbon derived from acacia wood: Optimization and batch studies. Arab. J. Chem. 2021, 14, 103122. [Google Scholar] [CrossRef]
- Lai, H.J. Adsorption of remazol brilliant violet 5R (RBV-5R) and remazol brilliant blue R (RBBR) from aqueous solution by using agriculture waste. Trop. Aquat. Soil Pollut. 2021, 1, 11–23. [Google Scholar] [CrossRef]
- Bouchelkia, N.; Mouni, L.; Belkhiri, L.; Bouzaza, A.; Bollinger, J.-C.; Madani, K.; Dahmoune, F. Removal of lead(II) from water using activated carbon developed from jujube stones, a low-cost sorbent. Sep. Sci. Technol. 2016, 51, 1645–1653. [Google Scholar] [CrossRef]
- Chedri Mammar, A.; Mouni, L.; Bollinger, J.-C.; Belkhiri, L.; Bouzaza, A.; Assadi, A.A.; Belkacemi, H. Modeling and optimization of process parameters in elucidating the adsorption mechanism of Gallic acid on activated carbon prepared from date stones. Sep. Sci. Technol. 2020, 55, 3113–3125. [Google Scholar] [CrossRef]
- Mohamad Yusop, M.F.; Nasehir Khan, M.N.; Zakaria, R.; Abdullah, A.Z.; Ahmad, M.A. Mass transfer simulation on remazol brilliant blue R dye adsorption by optimized teak wood Based activated carbon. Arab. J. Chem. 2023, 16, 104780. [Google Scholar] [CrossRef]
- Mohamad Yusop, M.F.; Tamar Jaya, M.A.; Idris, I.; Abdullah, A.Z.; Ahmad, M.A. Optimization and mass transfer simulation of remazol brilliant blue R dye adsorption onto meranti wood based activated carbon. Arab. J. Chem. 2023, 16, 104683. [Google Scholar] [CrossRef]
- Al Harthi, S.; Mavazhe, A.; Al Mahroqi, H.; Khan, S.A. Quantification of phenolic compounds, evaluation of physicochemical properties and antioxidant activity of four date (Phoenix dactylifera L.) varieties of Oman. J. Taibah Univ. Med. Sci. 2015, 10, 346–352. [Google Scholar] [CrossRef]
- Alotaibi, K.D.; Alharbi, H.A.; Yaish, M.W.; Ahmed, I.; Alharbi, S.A.; Alotaibi, F.; Kuzyakov, Y. Date palm cultivation: A review of soil and environmental conditions and future challenges. Land Degrad. Dev. 2023, 34, 2431–2444. [Google Scholar] [CrossRef]
- Rambabu, K.; AlYammahi, J.; Bharath, G.; Thanigaivelan, A.; Sivarajasekar, N.; Banat, F. Nano-activated carbon derived from date palm coir waste for efficient sequestration of noxious 2,4-dichlorophenoxyacetic acid herbicide. Chemosphere 2021, 282, 131103. [Google Scholar] [CrossRef] [PubMed]
- Alharbi, H.A.; Hameed, B.H.; Alotaibi, K.D.; Aloud, S.S.; Al-Modaihsh, A.S. Mesoporous Activated Carbon from Leaf Sheath Date Palm Fibers by Microwave-Assisted Phosphoric Acid Activation for Efficient Dye Adsorption. ACS Omega 2022, 7, 46079–46089. [Google Scholar] [CrossRef] [PubMed]
- Zamouche, M.; Mouni, L.; Ayachi, A.; Merniz, I. Use of commercial activated carbon for the purification of synthetic water polluted by a pharmaceutical product. Desalin. Water Treat. 2019, 172, 86–95. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Over the adsorption in solution. J. Phys. Chem. 1906, 57, 385–471. [Google Scholar]
- Parmanbek, N.; Sütekin, D.S.; Barsbay, M.; Mashentseva, A.A.; Zheltov, D.A.; Aimanova, N.A.; Jakupova, Z.Y.; Zdorovets, M.V. Hybrid PET track-etched membranes grafted by well-defined poly(2-(dimethylamino)ethyl methacrylate) brushes and loaded with silver nanoparticles for the removal of as(iii). Polymers 2022, 14, 4026. [Google Scholar] [CrossRef]
- Marrakchi, F.; Hameed, B.H.; Bouaziz, M. Mesoporous and high-surface-area activated carbon from defatted olive cake by-products of olive mills for the adsorption kinetics and isotherm of methylene blue and acid blue 29. J. Environ. Chem. Eng. 2020, 8, 104199. [Google Scholar] [CrossRef]
- Lagergren, S.K. About the theory of so-called adsorption of soluble substances. Sven. Vetensk. Handingarl 1898, 24, 1–39. [Google Scholar]
- Ho, Y.S.; McKay, G. Sorption of dye from aqueous solution by peat. Chem. Eng. J. 1998, 70, 115–124. [Google Scholar] [CrossRef]
- Lima, E.C.; Hosseini-Bandegharaei, A.; Moreno-Piraján, J.C.; Anastopoulos, I. A critical review of the estimation of the thermodynamic parameters on adsorption equilibria. Wrong use of equilibrium constant in the Van’t Hoof equation for calculation of thermodynamic parameters of adsorption. J. Mol. Liq. 2019, 273, 425–434. [Google Scholar] [CrossRef]
- Hijab, M.; Parthasarathy, P.; Mackey, H.R.; Al-Ansari, T.; McKay, G. Minimizing adsorbent requirements using multi-stage batch adsorption for malachite green removal using microwave date-stone activated carbons. Chem. Eng. Process.—Process Intensif. 2021, 167, 108318. [Google Scholar] [CrossRef]
- Canales-Flores, R.A.; Prieto-García, F. Taguchi optimization for production of activated carbon from phosphoric acid impregnated agricultural waste by microwave heating for the removal of methylene blue. Diam. Relat. Mater. 2020, 109, 108027. [Google Scholar] [CrossRef]
- Liu, H.; Liu, J.; Huang, H.; Evrendilek, F.; Wen, S.; Li, W. Optimizing bioenergy and by-product outputs from durian shell pyrolysis. Renew. Energy 2021, 164, 407–418. [Google Scholar] [CrossRef]
- Martinez, C.L.M.; Rocha, E.P.A.; Carneiro, A.O.; Gomes, F.J.B.; Batalha, L.A.R.; Vakkilainen, E. Characterization of residual biomasses from the coffee production chain and assessment the potential for energy purposes. Biomass Bioenergy 2019, 120, 68–76. [Google Scholar] [CrossRef]
- Tan, Y.L.; Abdullah, A.Z.; Hameed, B.H. Product distribution of the thermal and catalytic fast pyrolysis of karanja (Pongamia pinnata) fruit hulls over a reusable silica-alumina catalyst. Fuel 2019, 245, 89–95. [Google Scholar] [CrossRef]
- Neme, I.; Gonfa, G.; Masi, C. Activated carbon from biomass precursors using phosphoric acid: A review. Heliyon 2022, 8, e11940. [Google Scholar] [CrossRef]
- Maršálek, R.; Švidrnoch, M. The adsorption of amitriptyline and nortriptyline on activated carbon, diosmectite and titanium dioxide. Environ. Chall. 2020, 1, 100005. [Google Scholar] [CrossRef]
- Joshi, N.C.; Rawat, B.S.; Kumar, P.; Kumar, N.; Upadhyay, S.; Chetana, S.; Gururani, P.; Kimothi, S. Sustainable synthetic approach and applications of ZnO/r-GO in the adsorption of toxic Pb2+ and Cr6+ ions. Inorg. Chem. Commun. 2022, 145, 110040. [Google Scholar] [CrossRef]
- Liu, L.; Huang, Y.; Cao, J.; Hu, H.; Dong, L.; Zha, J.; Su, Y.; Ruan, R.; Tao, S. Qualitative and relative distribution of Pb2+ adsorption mechanisms by biochars produced from a fluidized bed pyrolysis system under mild air oxidization conditions. J. Mol. Liq. 2021, 323, 114600. [Google Scholar] [CrossRef]
- Mashentseva, A.A.; Aimanova, N.A.; Parmanbek, N.; Temirgaziyev, B.S.; Barsbay, M.; Zdorovets, M.V. Serratula coronata L. Mediated synthesis of ZnO nanoparticles and their application for the removal of alizarin yellow R by photocatalytic degradation and adsorption. Nanomaterials 2022, 12, 3293. [Google Scholar] [CrossRef]
- Saleh, T.A.; Adio, S.O.; Asif, M.; Dafalla, H. Statistical analysis of phenols adsorption on diethylenetriamine-modified activated carbon. J. Clean. Prod. 2018, 182, 960–968. [Google Scholar] [CrossRef]
- Altynbaeva, L.S.; Mashentseva, A.A.; Aimanova, N.A.; Zheltov, D.A.; Shlimas, D.I.; Nurpeisova, D.T.; Barsbay, M.; Abuova, F.U.; Zdorovets, M.V. Eco-friendly electroless template synthesis of cu-based composite track-etched membranes for sorption removal of lead(II) ions. Membranes 2023, 13, 495. [Google Scholar] [CrossRef] [PubMed]
- Sepehri, S.; Kanani, E.; Abdoli, S.; Rajput, V.D.; Minkina, T.; Asgari Lajayer, B. Pb(II) Removal from aqueous solutions by adsorption on stabilized zero-valent iron nanoparticles & mdash;a green approach. Water 2023, 15, 222. [Google Scholar]
- Wang, Q.; Wang, Y.; Yang, Z.; Han, W.; Yuan, L.; Zhang, L.; Huang, X. Efficient removal of Pb(II) and Cd(II) from aqueous solutions by mango seed biosorbent. Chem. Eng. J. Adv. 2022, 11, 100295. [Google Scholar] [CrossRef]
- Gusain, R.; Kumar, N.; Fosso-Kankeu, E.; Ray, S.S. Efficient removal of Pb(II) and Cd(II) from industrial mine water by a hierarchical MoS2/SH-MWCNT Nanocomposite. ACS Omega 2019, 4, 13922–13935. [Google Scholar] [CrossRef]
- Khoso, W.A.; Haleem, N.; Baig, M.A.; Jamal, Y. Synthesis, characterization and heavy metal removal efficiency of nickel ferrite nanoparticles (NFN’s). Sci. Rep. 2021, 11, 3790. [Google Scholar] [CrossRef]
- Mashentseva, A.A.; Seitzhapar, N.; Barsbay, M.; Aimanova, N.A.; Alimkhanova, A.N.; Zheltov, D.A.; Zhumabayev, A.M.; Temirgaziev, B.S.; Almanov, A.A.; Sadyrbekov, D.T. Adsorption isotherms and kinetics for Pb(II) ion removal from aqueous solutions with biogenic metal oxide nanoparticles. RSC Adv. 2023, 13, 26839–26850. [Google Scholar] [CrossRef]
- Vázquez-Sánchez, A.Y.; Lima, E.C.; Abatal, M.; Tariq, R.; Santiago, A.A.; Alfonso, I.; Aguilar, C.; Vazquez-Olmos, A.R. Biosorption of Pb(II) using natural and treated Ardisia compressa K. leaves: Simulation framework extended through the application of artificial neural network and genetic algorithm. Molecules 2023, 28, 6387. [Google Scholar] [CrossRef]
- Nyirenda, J.; Kalaba, G.; Munyati, O. Synthesis and characterization of an activated carbon-supported silver-silica nanocomposite for adsorption of heavy metal ions from water. Results Eng. 2022, 15, 100553. [Google Scholar] [CrossRef]
- Preeti; Banerjee, S.; Debnath, A.; Singh, V. Gum ghatti-alginate hybrid bead derived titania spheres for deep removal of toxic dye Remazol Brilliant Violet from aqueous solutions. Environ. Nanotechnol. Monit. Manag. 2021, 15, 100459. [Google Scholar] [CrossRef]

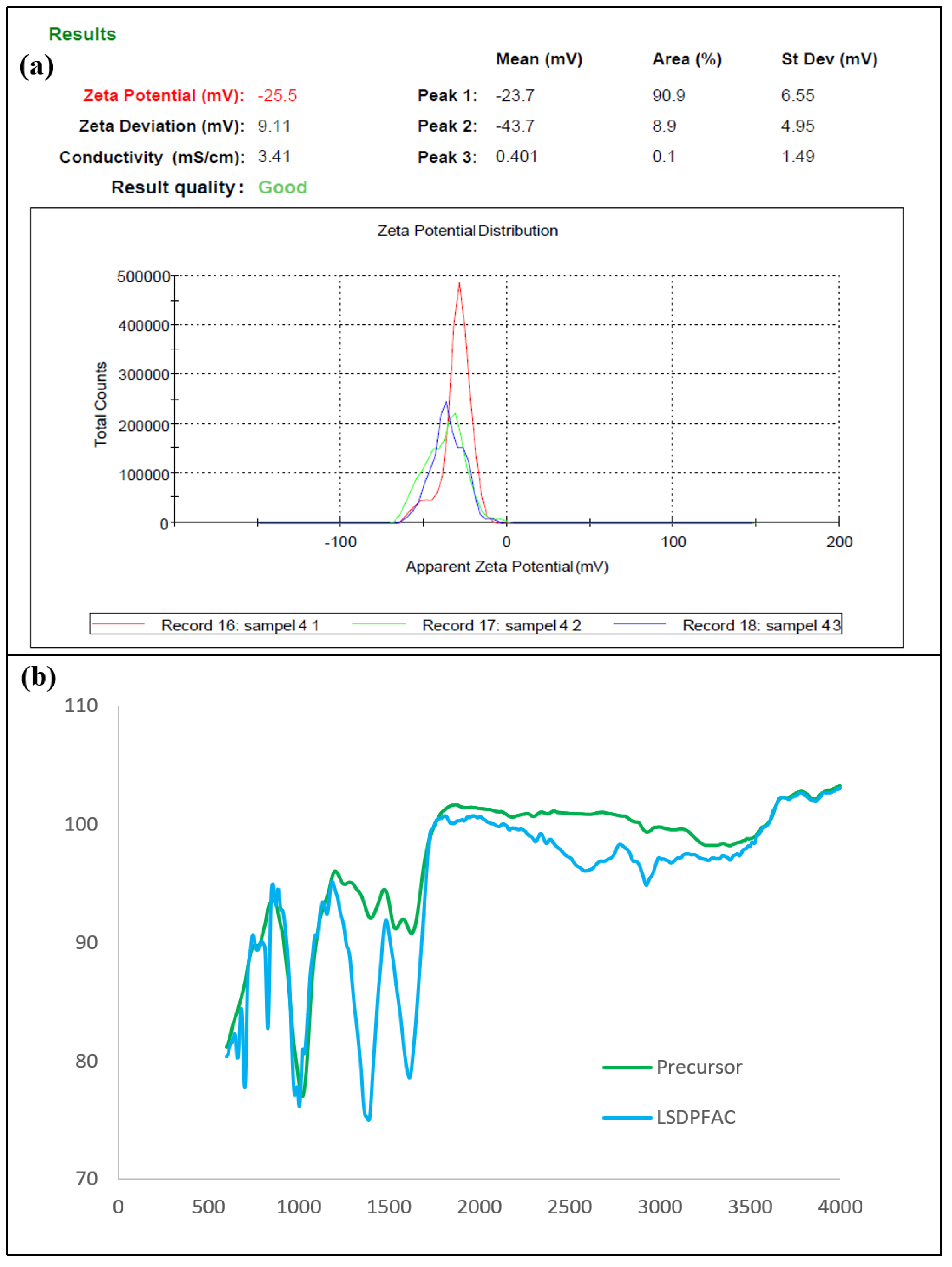

| Samples | Elemental Analysis | Proximate Analysis | |||||||
|---|---|---|---|---|---|---|---|---|---|
| C | H | N | S | Others | Moisture | Volatile Matter | Fixed Carbon | Ash | |
| Precursor | 33.45 | 3.85 | 0.97 | 0.37 | 61.36 | 11.92 | 66.27 | 19.92 | 1.90 |
| LSDPFAC | 55.67 | 5.44 | 0.74 | 0.37 | 37.78 | 14.26 | 5.99 | 76.52 | 3.23 |
| Precursor | LSDPFAC | ||
|---|---|---|---|
| Peak (cm−1) | Functional Groups | Peak (cm−1) | Functional Groups |
| 748 | Methylene –(CH2)n | 648 | Alkyne, C-H, bending |
| 860 | Peroxides, C-O-O-, stretching | 677 | Alkyne, C-H, bending |
| 1199 | Phenol, C-O, stretching | 856 | Peroxides, C-O-O-, stretching |
| 1583 | C=C-C Aromatic ring stretching | 881 | Peroxides, C-O-O-, stretching |
| 2970 | Methyl, C-H, asymmetric stretching | 1148 | Tertiary alcohol, C-O, stretching |
| 3645 | Nonbonded hydroxy group, OH, stretching | 1198 | Phenol, C-O, stretching |
| 1477 | Carbonate ions | ||
| Isotherm | Parameters | 30 °C |
|---|---|---|
| Langmuir | Qm | 14.10 |
| KL | 1.21 | |
| RL | 0.064–0.41 | |
| R2 | 0.9976 | |
| RMSE | 0.35 | |
| Freundlich | K | 7.65 |
| n | 1.69 | |
| R2 | 0.9972 | |
| RMSE | 0.11 | |
| Dubinin–Radushkevich | QDR | 9.46 |
| BDR | 0.05 | |
| EDR | 3.17 | |
| R2 | 0.9939 | |
| RMSE | 0.48 |
| Adsorbent | Isotherm | Kinetic | Langmuir Monolayer Adsorption Capacity, Qm (mg/g) | Rate Constant | Mechanism | Experimental Conditions | References |
|---|---|---|---|---|---|---|---|
| Leaf sheath fibre of date-palm-based activated carbon | Freundlich | PSO | 14.10 | 0.1735 to 0.0043 g mg−1 min−1 | Physisorption | Concentration: 1–10 mg/L pH: 3–9 | This study |
| Composite track-etched membranes | Freundlich | PSO | 0.08–0.21 | 0.09 × 10−4–0.55 × 10−4 g µg−1 min−1 | Ion exchange and chemisorption | Concentration: 50 mg/L pH: 3–8 | [62] |
| Biomass-derived activated carbon supported by nano zero-valent iron particles | Langmuir–Freundlich | PSO | 23.30–140.80 | 0.006 g mg−1 min−1 | Surface and redox reaction | Concentration: 10–1000 mg/L pH: 2–10 | [63] |
| Mango seed biosorbent | Redlich–Peterson | PFO | 283.20 | 0.461 min−1 | Electrostatic attraction, microprecipitation, complexation and ion exchange | Concentration: 500 mg/L pH: 2–7.5 | [64] |
| Molybdenum sulphide (MoS2)/thiol-functionalised multiwalled carbon nanotube | Freundlich | PSO | 90.00 | 0.014 g mg−1 min−1 | Ion exchange and electrostatic interactions | Concentration: 100 mg/L pH: 2–6 | [65] |
| Magnetic nickel-ferrite nanoparticles | - | PSO | - | 0.0148 g mg−1 min−1 | - | Concentration: 5 mg/L pH: 2–6 | [66] |
| Zinc oxide (ZnO) and copper (II) oxide (CuO) nanoparticles | Freundlich | PSO | - | 1.97 × 10−4–2.09 × 10−4 g mg−1 min−1 | Chemical interactions | Concentration: 100 mg/L pH: 3–9 | [67] |
| A. compressa K.-based activated carbon | Langmuir | PSO | 170.00 | 0.488–3.175 to 0.022–0.032 g mg−1 min−1 | Chemisorption | Concentration: 25–250 mg/L pH: 2–6 | [68] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aloud, S.S.; Hameed, B.H.; Yusop, M.F.M.; Alharbi, H.A.; Giesy, J.P.; Alotaibi, K.D. Adsorption of Pb2+ by Activated Carbon Produced by Microwave-Assisted K2CO3 Activation of Date Palm Leaf Sheath Fibres. Water 2023, 15, 3905. https://doi.org/10.3390/w15223905
Aloud SS, Hameed BH, Yusop MFM, Alharbi HA, Giesy JP, Alotaibi KD. Adsorption of Pb2+ by Activated Carbon Produced by Microwave-Assisted K2CO3 Activation of Date Palm Leaf Sheath Fibres. Water. 2023; 15(22):3905. https://doi.org/10.3390/w15223905
Chicago/Turabian StyleAloud, Saud S., Bassim H. Hameed, Mohamad F. M. Yusop, Hattan A. Alharbi, John P. Giesy, and Khaled D. Alotaibi. 2023. "Adsorption of Pb2+ by Activated Carbon Produced by Microwave-Assisted K2CO3 Activation of Date Palm Leaf Sheath Fibres" Water 15, no. 22: 3905. https://doi.org/10.3390/w15223905
APA StyleAloud, S. S., Hameed, B. H., Yusop, M. F. M., Alharbi, H. A., Giesy, J. P., & Alotaibi, K. D. (2023). Adsorption of Pb2+ by Activated Carbon Produced by Microwave-Assisted K2CO3 Activation of Date Palm Leaf Sheath Fibres. Water, 15(22), 3905. https://doi.org/10.3390/w15223905







