Co-Pyrolysis of Fenton Sludge and Pomelo Peel for Heavy Metal Stabilization: Speciation Mechanism and Risk Evaluation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Feedstocks and Chemicals
2.2. Co-Pyrolysis of FS and PP
2.3. Heavy Metals Analysis
2.3.1. Sequential Extraction of Heavy Metals
2.3.2. Leaching Experiment
2.3.3. Determination of Heavy Metals
2.4. Ecological Risk Assessment
3. Results and Discussion
3.1. Properties of Sludge and Biochars
3.1.1. General Properties
3.1.2. Elemental Analysis and Specific Surface Area
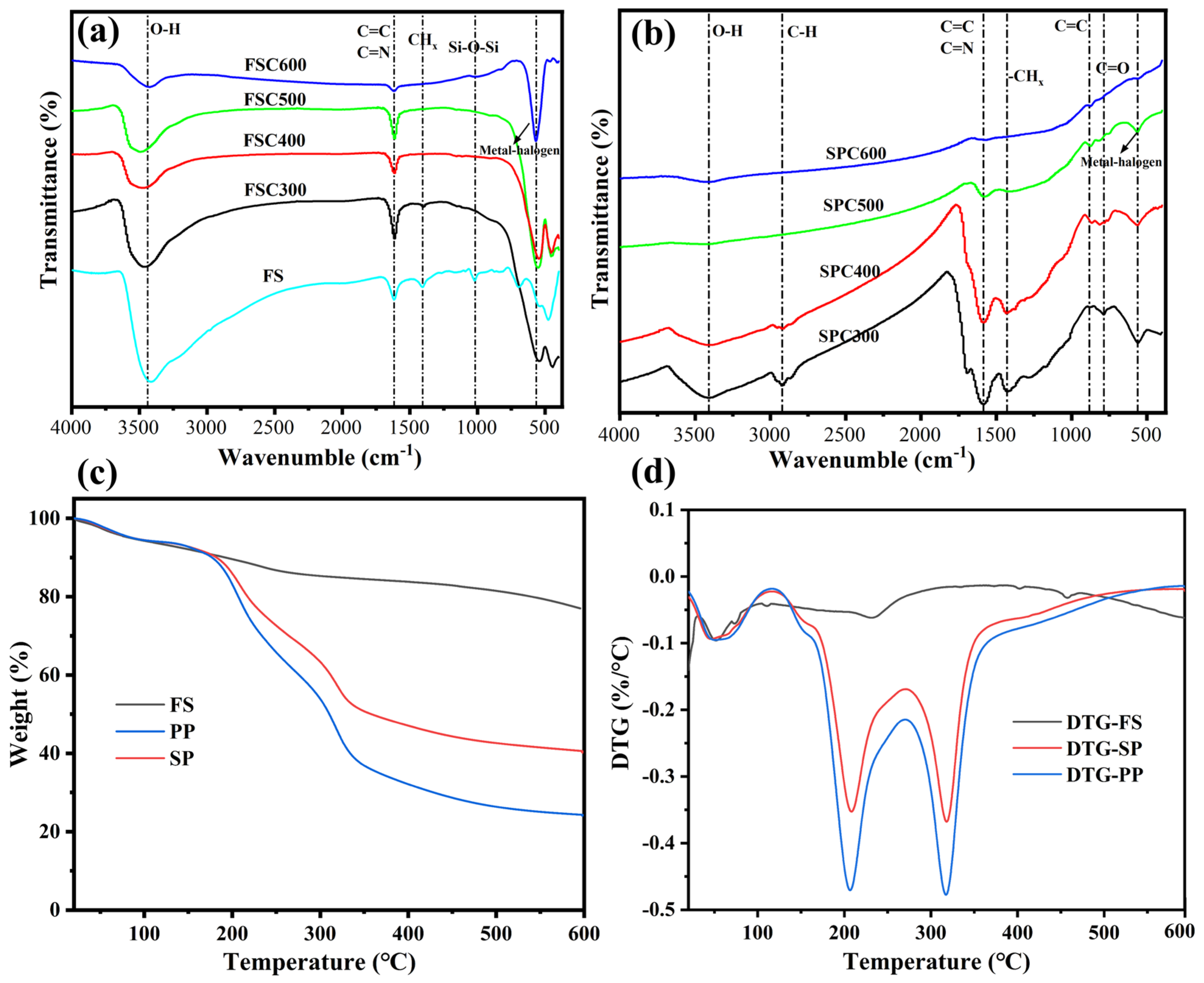
3.1.3. FTIR Spectra Analysis
3.1.4. Thermogravimetric Analysis
3.2. The Total Contents of HMs in the Biochars
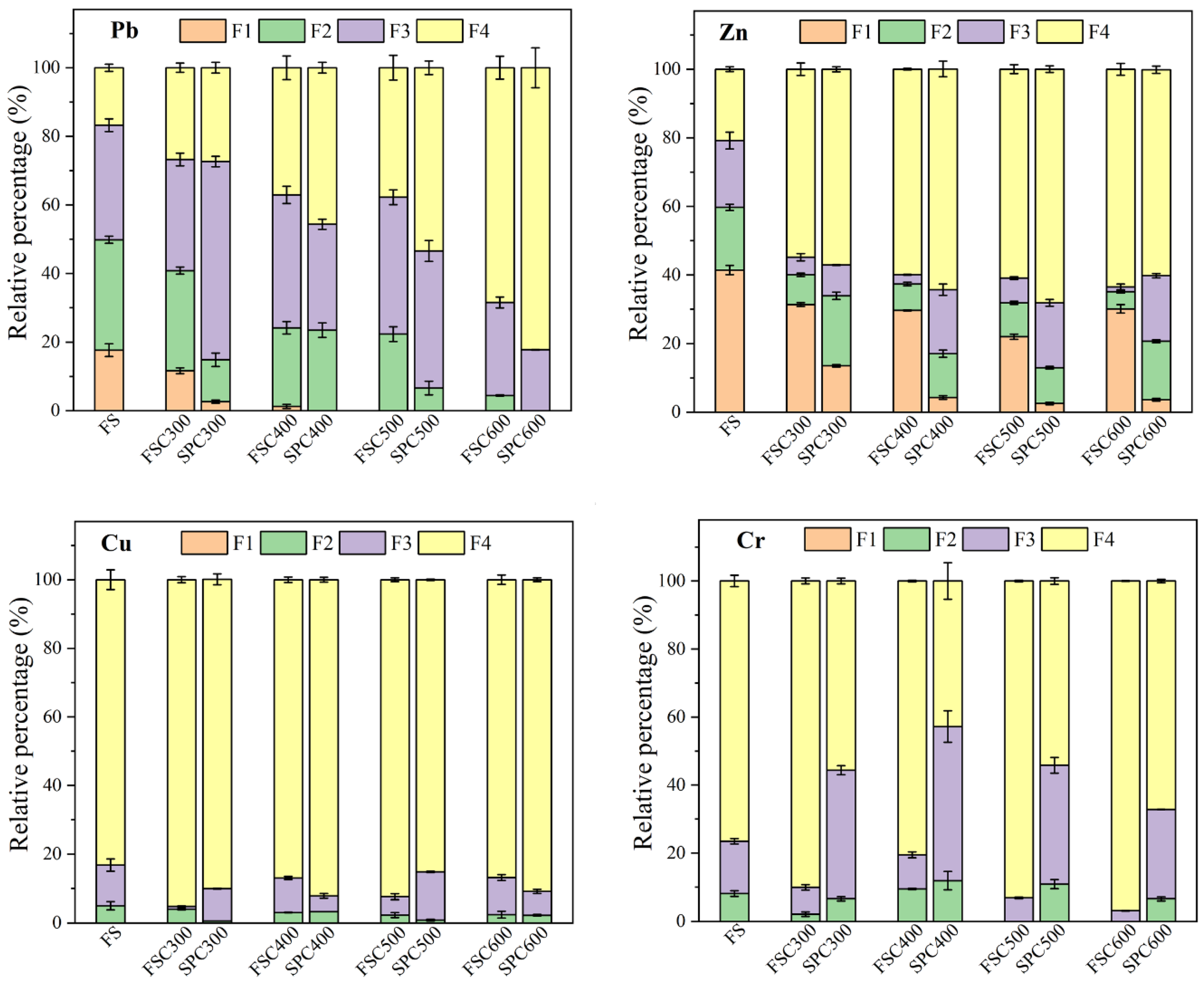
3.3. The Speciation of HMs in the Biochars
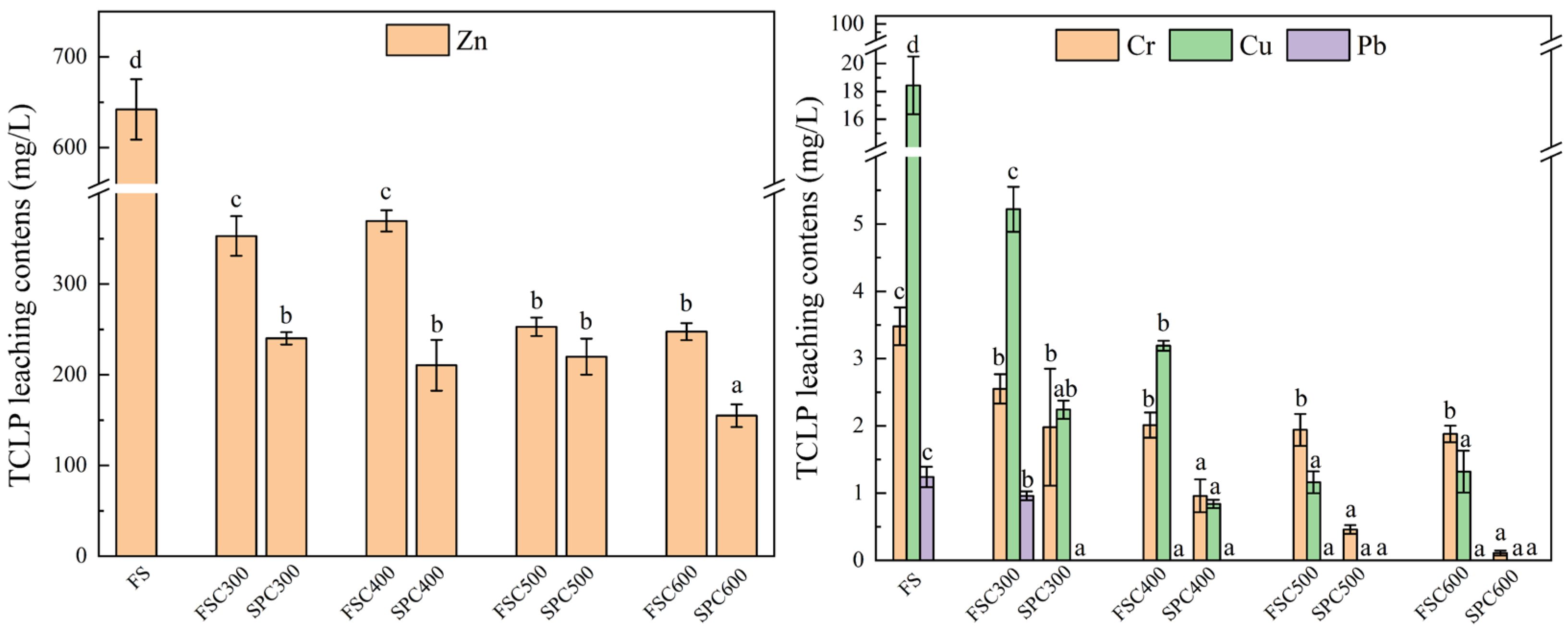
3.4. The Leaching Toxicity of HMs in the Biochars
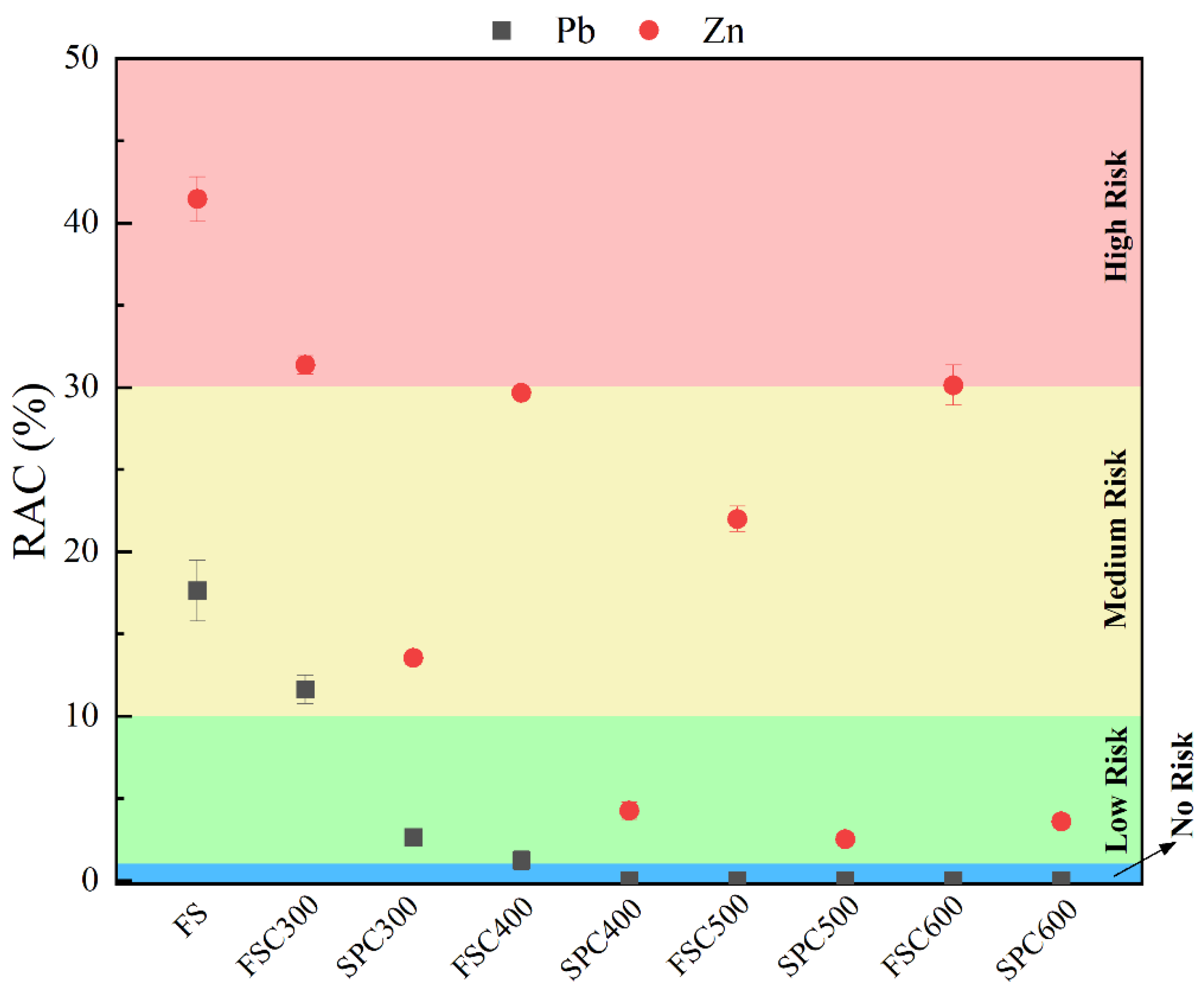
3.5. The Environmental Risk Evaluation of HMs in the Biochars
4. Conclusions and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gao, L.; Cao, Y.; Wang, L.; Li, S. A review on sustainable reuse applications of Fenton sludge during wastewater treatment. Front. Environ. Sci. Eng. 2021, 16, 77. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ahmad, H.; Parveen, T.; Ahmad, A.; Oves, M.; Ismail, I.M.I.; Qari, H.A.; Umar, K.; Mohamad Ibrahim, M.N. Recent Advances in Metal Decorated Nanomaterials and Their Various Biological Applications: A Review. Front. Chem. 2020, 8, 341. [Google Scholar] [CrossRef] [PubMed]
- Chai, Y.; Huang, C.; Sui, M.; Yin, Y.; Sun, N.; Chen, Y.; Liao, Z.; Sun, X.; Shen, W.; Tang, S. Fe-loaded alginate hydrogel beads activating peroxymonosulfate for enhancing anaerobic fermentation of waste activated sludge: Performance and potential mechanism. J. Environ. Manag. 2023, 341, 118079. [Google Scholar] [CrossRef]
- He, M.; Xu, Z.; Hou, D.; Gao, B.; Cao, X.; Ok, Y.S.; Rinklebe, J.; Bolan, N.S.; Tsang, D.C.W. Waste-derived biochar for water pollution control and sustainable development. Nat. Rev. Earth Environ. 2022, 3, 444–460. [Google Scholar] [CrossRef]
- Hossain, M.K.; Strezov, V.; Chan, K.Y.; Ziolkowski, A.; Nelson, P.F. Influence of pyrolysis temperature on production and nutrient properties of wastewater sludge biochar. J. Environ. Manag. 2011, 92, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Li, Y.; Zhang, J.; Wu, S.; Cao, Y.; Liang, P.; Zhang, J.; Wong, M.H.; Wang, M.; Shan, S.; et al. Influence of pyrolysis tem-perature on properties and environmental safety of heavy metals in biochars derived from municipal sewage sludge. J. Hazard. Mater. 2016, 320, 417–426. [Google Scholar] [CrossRef]
- Zhang, J.; Shao, J.; Jin, Q.; Li, Z.; Zhang, X.; Chen, Y.; Zhang, S.; Chen, H. Sludge-based biochar activation to enhance Pb(II) adsorption. Fuel 2019, 252, 101–108. [Google Scholar] [CrossRef]
- Mian, M.M.; Alam, N.; Ahommed, M.S.; He, Z.; Ni, Y. Emerging applications of sludge biochar-based catalysts for environ-mental remediation and energy storage: A review. J. Clean. Prod. 2022, 360, 132131. [Google Scholar] [CrossRef]
- Li, D.; Shan, R.; Jiang, L.; Gu, J.; Zhang, Y.; Yuan, H.; Chen, Y. A review on the migration and transformation of heavy metals in the process of sludge pyrolysis. Resour. Conserv. Recy. 2022, 185, 106452. [Google Scholar] [CrossRef]
- Udayanga, W.D.C.; Veksha, A.; Giannis, A.; Liang, Y.N.; Lisak, G.; Hu, X.; Lim, T.-T. Insights into the speciation of heavy metals during pyrolysis of industrial sludge. Sci. Total Environ. 2019, 691, 232–242. [Google Scholar]
- Feng, S.; Zhang, G.; Yuan, D.; Li, Y.; Zhou, Y.; Lin, F. Co-pyrolysis of paper mill sludge and textile dyeing sludge with high calorific value solid waste: Pyrolysis kinetics, products distribution, and pollutants transformation. Fuel 2022, 329, 125433. [Google Scholar] [CrossRef]
- Ma, M.; Xu, D.; Zhi, Y.; Yang, W.; Duan, P.; Wu, Z. Co-pyrolysis re-use of sludge and biomass waste: Development, kinetics, synergistic mechanism and industrialization. J. Anal. Appl. Pyrol. 2022, 168, 105746. [Google Scholar] [CrossRef]
- Li, Y.; Yu, H.; Liu, L.; Yu, H. Application of co-pyrolysis biochar for the adsorption and immobilization of heavy metals in contaminated environmental substrates. J. Hazard. Mater. 2021, 420, 126655. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Liu, Q.; Li, X.; Zhou, Z.; Wu, C.; Zhang, H. Co-pyrolysis of industrial sludge and rice straw: Synergistic effects of biomass on reaction characteristics, biochar properties and heavy metals solidification. Fuel Process. Technol. 2022, 230, 107211. [Google Scholar] [CrossRef]
- Dong, Q.; Zhang, S.; Wu, B.; Pi, M.; Xiong, Y.; Zhang, H. Co-pyrolysis of Sewage Sludge and Rice Straw: Thermal Behavior and Char Characteristic Evaluations. Energy Fuels 2020, 34, 607–615. [Google Scholar] [CrossRef]
- Lu, J.; Kumar Mishra, P.; Hunter, T.N.; Yang, F.; Lu, Z.; Harbottle, D.; Xu, Z. Functionalization of mesoporous carbons de-rived from pomelo peel as capacitive electrodes for preferential removal/recovery of copper and lead from contaminated water. Chem. Eng. J. 2022, 433, 134508. [Google Scholar] [CrossRef]
- Wang, W.; Chen, M. Catalytic degradation of sulfamethoxazole by peroxymonosulfate activation system composed of nitrogen-doped biochar from pomelo peel: Important roles of defects and nitrogen, and detoxification of intermediates. J. Colloid Interf. Sci. 2022, 613, 57–70. [Google Scholar] [CrossRef]
- Geng, Y.; Sun, W.; Ying, P.; Zheng, Y.; Ding, J.; Sun, K.; Li, L.; Li, M. Bioinspired Fractal Design of Waste Biomass-Derived Solar–Thermal Materials for Highly Efficient Solar Evaporation. Adv. Funct. Mater. 2021, 31, 2007648. [Google Scholar] [CrossRef]
- Wang, X.; Wei-Chung Chang, V.; Li, Z.; Song, Y.; Li, C.; Wang, Y. Co-pyrolysis of sewage sludge and food waste digestate to synergistically improve biochar characteristics and heavy metals immobilization. Waste Manag. 2022, 141, 231–239. [Google Scholar] [CrossRef]
- Wang, X.; Chang, V.W.-C.; Li, Z.; Chen, Z.; Wang, Y. Co-pyrolysis of sewage sludge and organic fractions of municipal solid waste: Synergistic effects on biochar properties and the environmental risk of heavy metals. J. Hazard. Mater. 2021, 412, 125200. [Google Scholar] [CrossRef]
- Cui, Z.; Xu, G.; Ormeci, B.; Liu, H.; Zhang, Z. Transformation and stabilization of heavy metals during pyrolysis of organic and inorganic-dominated sewage sludges and their mechanisms. Waste Manag. 2022, 150, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, W.; Sun, X.; Shen, J.; Wang, L. A novel acetogenic bacteria isolated from waste activated sludge and its potential application for enhancing anaerobic digestion performance. J. Environ. Manag. 2020, 255, 109842. [Google Scholar] [CrossRef]
- Zhang, P.; Zhang, X.; Li, Y.; Han, L. Influence of pyrolysis temperature on chemical speciation, leaching ability, and environmental risk of heavy metals in biochar derived from cow manure. Bioresour. Technol. 2020, 302, 1228500. [Google Scholar] [CrossRef]
- Gu, W.; Guo, J.; Bai, J.; Dong, B.; Hu, J.; Zhuang, X.; Zhang, C.; Shih, K. Co-pyrolysis of sewage sludge and Ca(H2PO4)2: Heavy metal stabilization, mechanism, and toxic leaching. J. Environ. Manag. 2022, 305, 114292. [Google Scholar] [CrossRef]
- Qi, Q.; Hu, C.; Lin, J.; Wang, X.; Tang, C.; Dai, Z.; Xu, J. Contamination with multiple heavy metals decreases microbial diver-sity and favors generalists as the keystones in microbial occurrence networks. Environ. Pollut. 2022, 306, 119406. [Google Scholar] [CrossRef]
- Fan, Z.; Zhou, X.; Peng, Z.; Wan, S.; Gao, Z.F.; Deng, S.; Tong, L.; Han, W.; Chen, X. Co-pyrolysis technology for enhancing the functionality of sewage sludge biochar and immobilizing heavy metals. Chemosphere 2023, 317, 137929. [Google Scholar] [CrossRef]
- Chen, J.; Liu, J.; He, Y.; Huang, L.; Sun, S.; Sun, J.; Chang, K.; Kuo, J.; Huang, S.; Ning, X. Investigation of co-combustion characteristics of sewage sludge and coffee grounds mixtures using thermogravimetric analysis coupled to artificial neural net-works modeling. Bioresour. Technol. 2017, 225, 234–245. [Google Scholar] [CrossRef]
- Shen, X.; Zeng, J.; Zhang, D.; Wang, F.; Li, Y.; Yi, W. Effect of pyrolysis temperature on characteristics, chemical speciation and environmental risk of Cr, Mn, Cu, and Zn in biochars derived from pig manure. Sci. Total Environ. 2020, 704, 135283. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, B.; Liu, H.; Zhao, Y.; Li, L. Effects of pyrolysis temperature on biochar’s characteristics and speciation and environmental risks of heavy metals in sewage sludge biochars. Environ. Technol. Inno. 2022, 26, 102288. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, H.; Zhang, Y.; Lichtfouse, E. Efficient phosphate recycling by adsorption on alkaline sludge biochar. Environ. Chem. Lett. 2023, 21, 21–30. [Google Scholar] [CrossRef]
- Zhang, S.; Gu, W.; Geng, Z.; Bai, J.; Dong, B.; Zhao, J.; Zhuang, X.; Shih, K. Immobilization of heavy metals in biochar by co-pyrolysis of sludge and CaSiO3. J. Environ. Manag. 2023, 326, 116635. [Google Scholar] [CrossRef] [PubMed]
- Xiaorui, L.; Longji, Y.; Xudong, Y. Evolution of chemical functional groups during torrefaction of rice straw. Bioresour. Technol. 2021, 320, 124328. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Ma, R.; Luo, J.; Huang, W.; Fang, L.; Sun, S.; Lin, J. Co-microwave pyrolysis of electroplating sludge and municipal sewage sludge to synergistically improve the immobilization of high-concentration heavy metals and an analysis of the mechanism. J. Hazard. Mater. 2021, 417, 126099. [Google Scholar] [CrossRef]
- Tang, F.; Yu, H.; Yassin Hussain Abdalkarim, S.; Sun, J.; Fan, X.; Li, Y.; Zhou, Y.; Chiu Tam, K. Green acid-free hydrolysis of wasted pomelo peel to produce carboxylated cellulose nanofibers with super absorption/flocculation ability for environmental remediation materials. Chem. Eng. J. 2020, 395, 125070. [Google Scholar] [CrossRef]
- Fang, S.; Yu, Z.; Lin, Y.; Hu, S.; Liao, Y.; Ma, X. Thermogravimetric analysis of the co-pyrolysis of paper sludge and municipal solid waste. Energ. Convers. Manag. 2015, 101, 626–631. [Google Scholar] [CrossRef]
- Yang, Y.-Q.; Cui, M.-H.; Guo, J.-C.; Du, J.-J.; Zheng, Z.-Y.; Liu, H. Effects of co-pyrolysis of rice husk and sewage sludge on the bioavailability and environmental risks of Pb and Cd. Environ. Technol. 2021, 42, 2304–2312. [Google Scholar] [CrossRef]
- Huang, H.; Yao, W.; Li, R.; Ali, A.; Du, J.; Guo, D.; Xiao, R.; Guo, Z.; Zhang, Z.; Awasthi, M.K. Effect of pyrolysis temperature on chemical form, behavior and environmental risk of Zn, Pb and Cd in biochar produced from phytoremediation residue. Bioresour. Technol. 2018, 249, 487–493. [Google Scholar] [CrossRef]
- Chen, G.; Tian, S.; Liu, B.; Hu, M.; Ma, W.; Li, X. Stabilization of heavy metals during co-pyrolysis of sewage sludge and ex-cavated waste. Waste Manag. 2020, 103, 268–275. [Google Scholar] [CrossRef]
- Xu, Y.; Qi, F.; Bai, T.; Yan, Y.; Wu, C.; An, Z.; Luo, S.; Huang, Z.; Xie, P. A further inquiry into co-pyrolysis of straws with manures for heavy metal immobilization in manure-derived biochars. J. Hazard. Mater. 2019, 380, 120870. [Google Scholar] [CrossRef]
- Wang, Z.; Xie, L.; Liu, K.; Wang, J.; Zhu, H.; Song, Q.; Shu, X. Co-pyrolysis of sewage sludge and cotton stalks. Waste Manag. 2019, 89, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Huang, Y.; Zhu, Z.; Yu, M.; Cheng, H.; Shi, H.; Zuo, W.; Zhou, H.; Wang, S. Co-pyrolysis of industrial sludge with phytoremediation residue: Improving immobilization of heavy metals at high temperature. Fuel 2023, 351, 128942. [Google Scholar] [CrossRef]
| Parameter | FS | Pomelo Peel |
|---|---|---|
| pH | 5.2 ± 0.1 | 6.9 ± 0.1 |
| Total organic matter (TOC) (%) | 4.58 ± 0.47 | 92.1 ± 1.05 |
| Cu (mg/kg) | 818 ± 21 | ND a |
| Zn (mg/kg) | 16,380 ± 231 | ND |
| Cr (mg/kg) | 128 ± 17 | ND |
| Pb (mg/kg) | 483 ± 24 | ND |
| Sample | Yield (%) | pH | C (%) | H (%) | H/C | SSA (m2/g) |
|---|---|---|---|---|---|---|
| FS | / | 5.25 ± 0.05 | 4.37 | 1.66 | 4.56 | / |
| PP | / | 6.95 ± 0.05 | 58.34 | 13.67 | 2.81 | / |
| FSC300 | 83.36 ± 4.15 | 5.29 ± 0.08 | 2.29 ± 0.12 | 0.84 ± 0.03 | 4.40 | 2.15 |
| FSC400 | 82.18 ± 3.18 | 5.27 ± 0.04 | 1.79 ± 0.18 | 0.63 ± 0.05 | 4.22 | 3.18 |
| FSC500 | 79.46 ± 2.87 | 5.28 ± 0.07 | 1.77 ± 0.07 | 0.62 ± 0.08 | 4.20 | 3.74 |
| FSC600 | 74.32 ± 4.43 | 5.80 ± 0.13 | 1.67 ± 0.09 | 0.53 ± 0.02 | 3.80 | 4.17 |
| SPC300 | 71.83 ± 2.79 | 7.28 ± 0.06 | 40.67 ± 0.16 | 2.52 ± 0.10 | 0.75 | 18.54 |
| SPC400 | 60.31 ± 4.63 | 8.59 ± 0.04 | 37.14 ± 0.14 | 3.07 ± 0.08 | 0.74 | 22.92 |
| SPC500 | 52.43 ± 3.98 | 9.81 ± 0.11 | 34.52 ± 010 | 1.73 ± 0.06 | 0.60 | 27.25 |
| SPC600 | 47.32 ± 3.53 | 10.92 ± 0.08 | 31.03 ± 0.08 | 1.14 ± 0.02 | 0.44 | 31.26 |
| Sample | Pb | Zn | Cr | Cu | ||||
|---|---|---|---|---|---|---|---|---|
| Total Content (mg/kg) | Recovery Rate (%) | Total Content (mg/kg) | Recovery Rate (%) | Total Content (mg/kg) | Recovery Rate (%) | Total Content (mg/kg) | Recovery Rate (%) | |
| FS | 483.45 ± 24.29 | / | 16,380 ± 231 | / | 128.55 ± 17.07 | / | 818.68 ± 21.85 | / |
| FSC300 | 519.65 ±21.67 | 89.68 ± 3.74 | 17,771 ± 373 | 90.4 ± 1.90 | 145.76 ± 2.76 | 94.9 ± 1.80 | 704.25 ± 45.50 | 71.77 ± 4.64 |
| FSC400 | 513.18 ± 26.40 | 87.31 ± 4.49 | 18,661 ± 265 | 93.6 ± 1.33 | 144.95 ± 6.83 | 93.06 ± 4.38 | 765.13 ± 20.38 | 76.87 ± 2.05 |
| FSC500 | 530.29 ± 28.66 | 87.24 ± 4.71 | 18,939 ± 736 | 91.9 ± 3.57 | 144.63 ± 3.58 | 89.78 ± 2.21 | 796.50 ± 11.50 | 77.37 ± 1.12 |
| FSC600 | 611.43 ± 2.76 | 94.08 ± 0.42 | 20,573 ± 891 | 93.3 ± 4.05 | 163.80 ± 3.25 | 95.11 ± 1.89 | 873.12 ± 17.63 | 79.33 ± 0.69 |
| SPC300 | 304.64 ± 3.92 | 90.61 ± 1.17 | 10,930 ± 495 | 95.8 ± 4.34 | 94.94 ± 4.81 | 94.94 ± 5.40 | 450.63 ± 13.13 | 81.88 ± 2.38 |
| SPC400 | 356.31 ± 16.65 | 88.98 ± 4.16 | 12,854 ± 379 | 94.3 ± 2.79 | 116.25 ± 4.25 | 104.25 ± 4.00 | 539.50 ± 35.50 | 79.55 ± 5.23 |
| SPC500 | 469.38 ± 12.62 | 101.90 ± 2.74 | 13,121 ± 208 | 84.0 ± 1.33 | 128.94 ± 2.94 | 103.94 ± 1.69 | 677.45 ± 27.50 | 86.85 ± 3.53 |
| SPC600 | 503.21 ± 7.69 | 98.60 ± 1.51 | 15,960 ± 603 | 95.7 ± 3.49 | 139.25 ± 0.75 | 99.25 ± 0.55 | 748.81 ± 26.19 | 86.64 ± 3.03 |
| Sample | Pi | PN | Classification Level | |||
|---|---|---|---|---|---|---|
| Pb | Zn | Cr | Cu | |||
| FS | 12.84 | 4.64 | 0.37 | 0.12 | 9.62 | Serious |
| FSC300 | 7.06 | 3.40 | 0.10 | 0.10 | 5.33 | Severe |
| FSC400 | 4.80 | 2.64 | 0.04 | 0 | 3.64 | Severe |
| FSC500 | 7.39 | 2.68 | 0.06 | 0 | 5.53 | Severe |
| FSC600 | 4.21 | 1.28 | 0.02 | 0 | 3.13 | Severe |
| SPC300 | 5.06 | 2.59 | 0.02 | 0 | 3.82 | Severe |
| SPC400 | 4.40 | 0.61 | 0 | 0 | 3.23 | Severe |
| SPC500 | 4.95 | 2.51 | 0.03 | 0 | 3.74 | Severe |
| SPC600 | 3.10 | 0.15 | 0 | 0 | 2.26 | Moderate |
| Pollution degree | Pi/PN < 1, low contamination; 1 < Pi/PN < 3, moderate contamination; 3 < Pi/PN < 6, severe contamination; Pi/PN > 6, serious contamination. | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, C.; Wang, L.; Fan, L.; Chen, Y. Co-Pyrolysis of Fenton Sludge and Pomelo Peel for Heavy Metal Stabilization: Speciation Mechanism and Risk Evaluation. Water 2023, 15, 3733. https://doi.org/10.3390/w15213733
Huang C, Wang L, Fan L, Chen Y. Co-Pyrolysis of Fenton Sludge and Pomelo Peel for Heavy Metal Stabilization: Speciation Mechanism and Risk Evaluation. Water. 2023; 15(21):3733. https://doi.org/10.3390/w15213733
Chicago/Turabian StyleHuang, Cheng, Lixian Wang, Lingyi Fan, and Yong Chen. 2023. "Co-Pyrolysis of Fenton Sludge and Pomelo Peel for Heavy Metal Stabilization: Speciation Mechanism and Risk Evaluation" Water 15, no. 21: 3733. https://doi.org/10.3390/w15213733




