Cladocera Responses to the Climate-Forced Abrupt Environmental Changes Related to the Late Glacial/Holocene Transition
Abstract
:1. Introduction
2. Materials and Methods
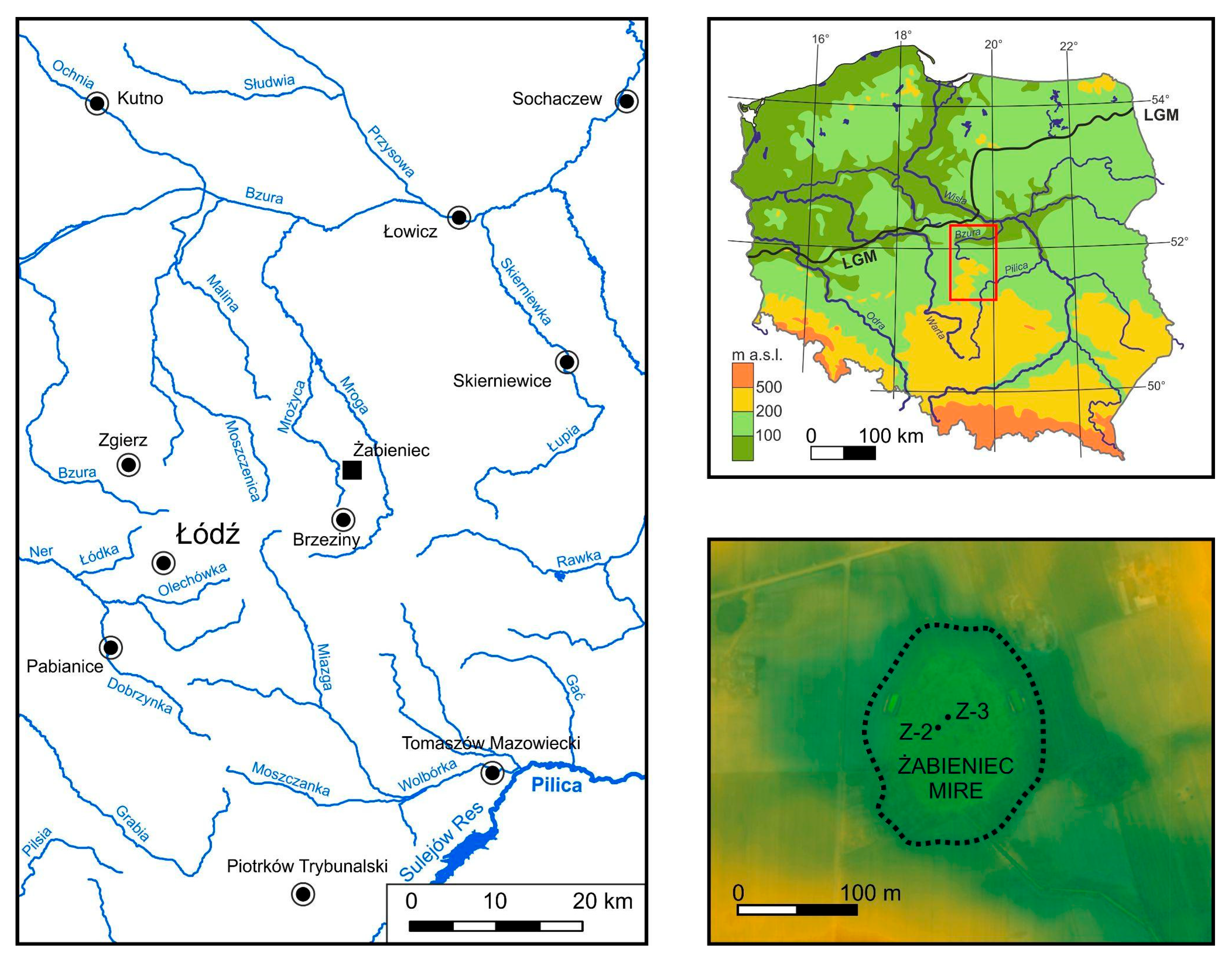
2.1. Study Site
2.2. Coring and Subsampling
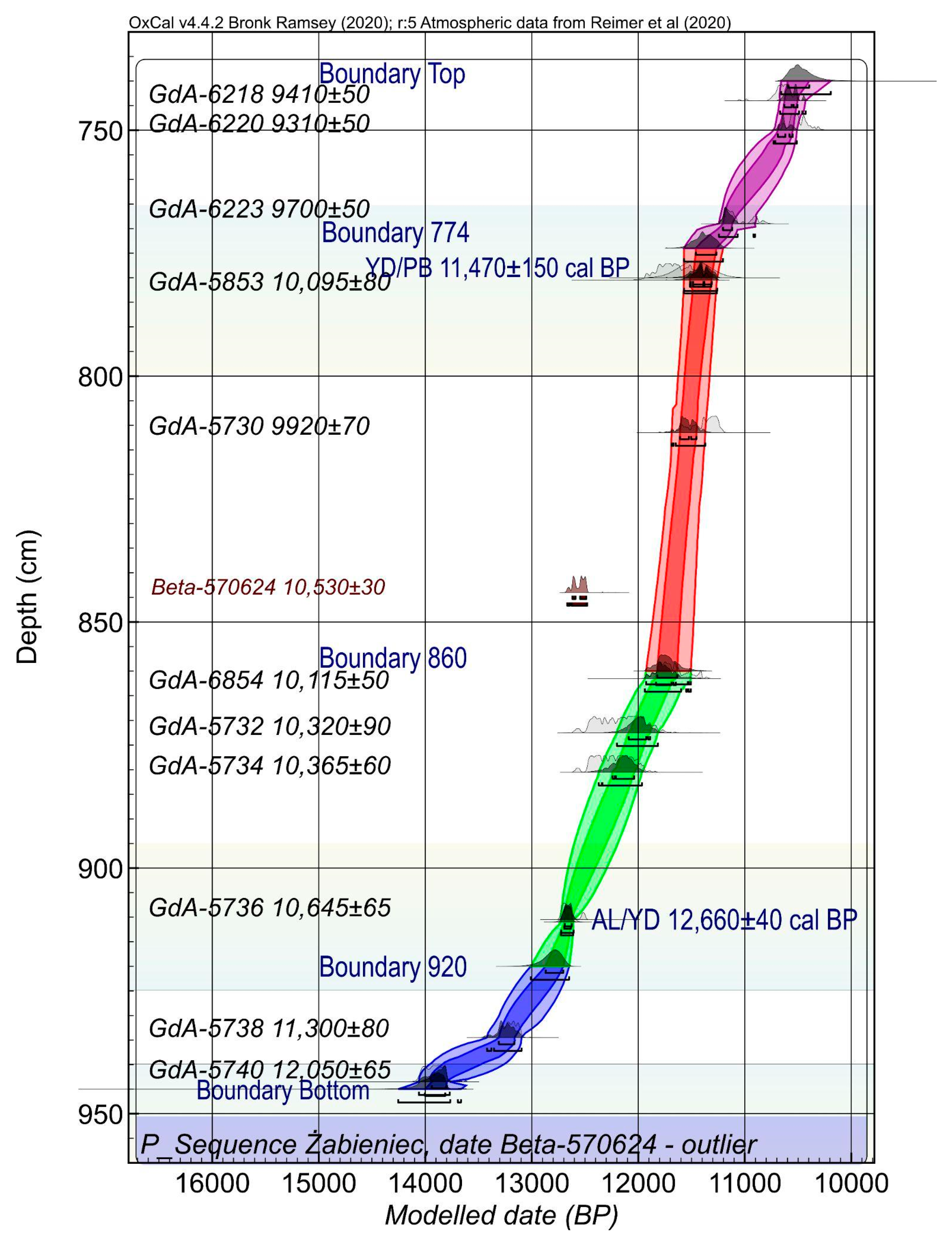
2.3. Geochronology
2.4. Cladocera Analysis
2.5. Geochemical Analysis
2.6. Palynological Analysis
2.7. Statistical Analysis
3. Results
3.1. Chronology
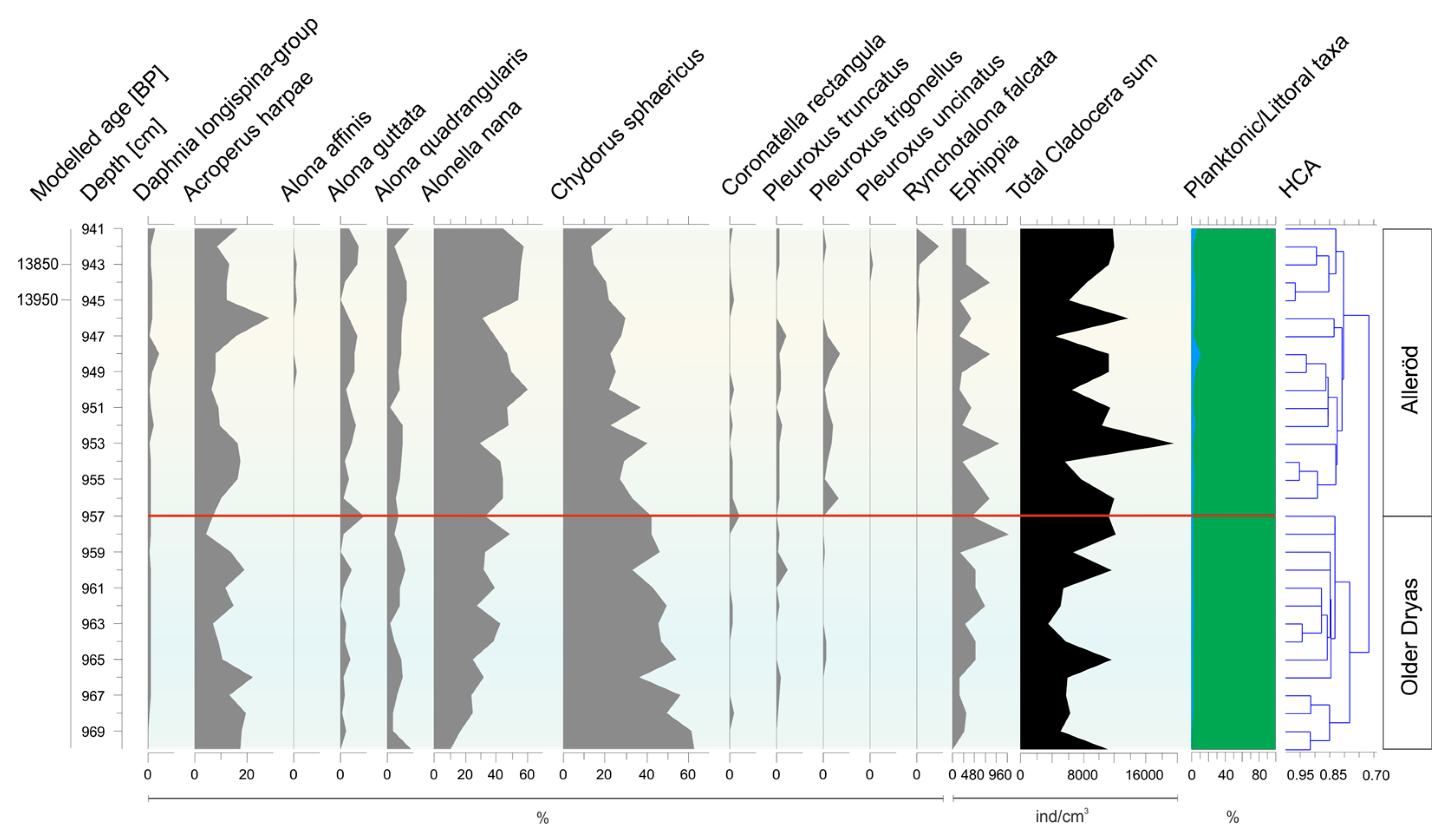
3.2. Subfossil Cladocera Assemblages
- Older Dryas/Alleröd transition (970–940 cm)
- Alleröd/Younger Dryas transition (895–925 cm)
- Younger Dryas/Holocene transition (799–765 cm)
3.3. Geochemistry
3.4. Pollen Record
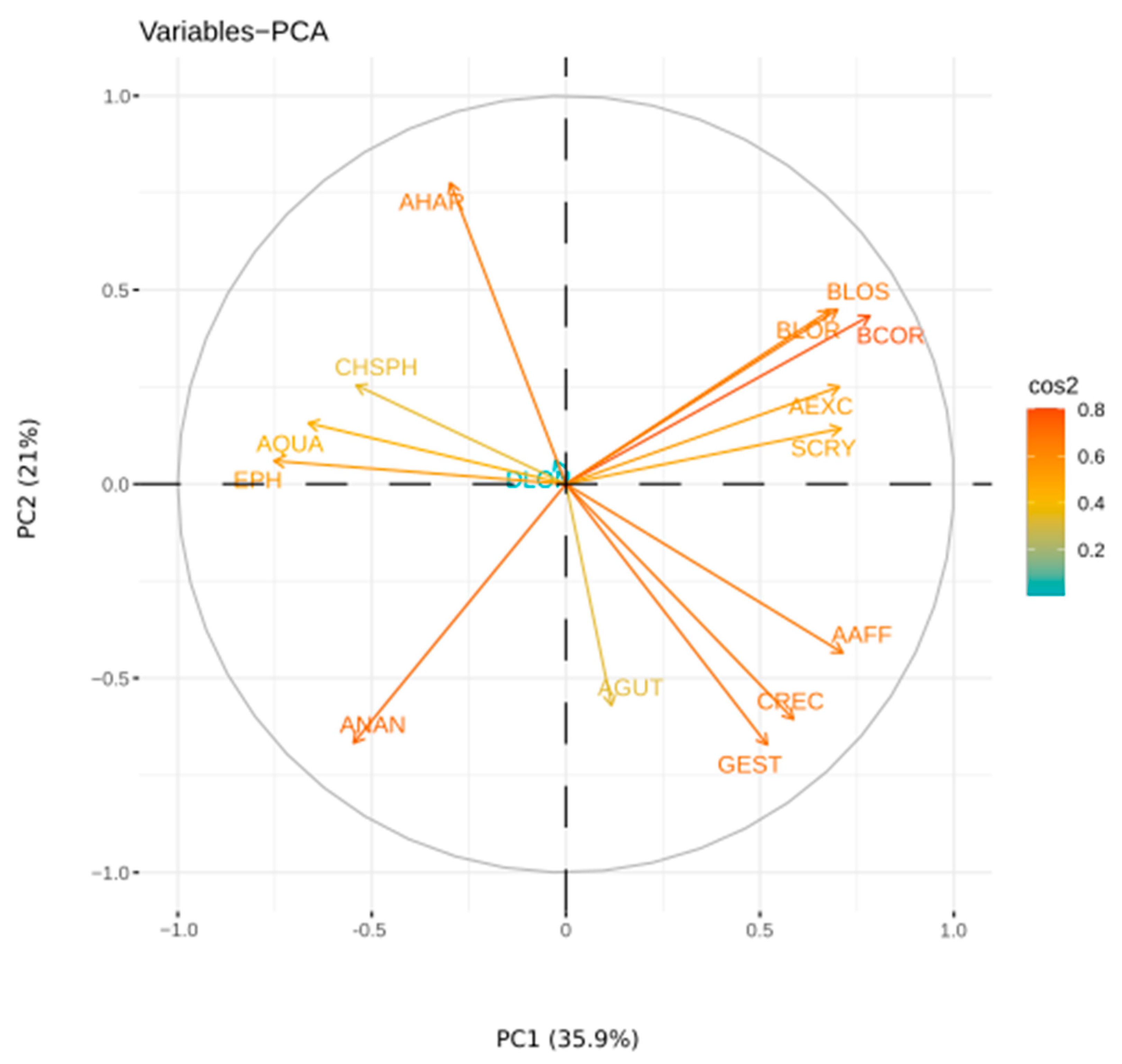
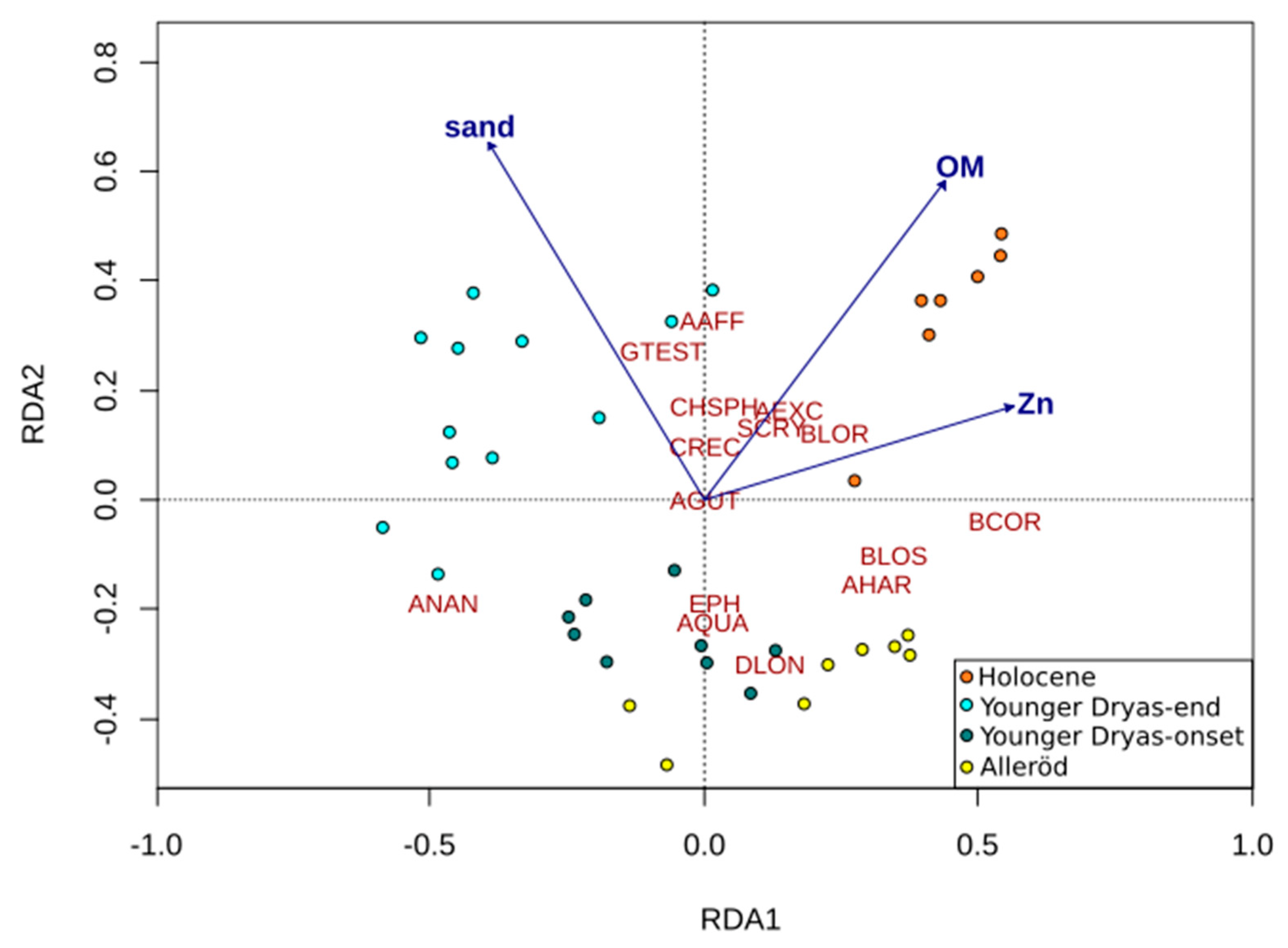
3.5. Numerical Analysis
4. Discussion
4.1. Environment Alteration during the Rapid Climate Changes Related to LGM/Holocene Transition
4.1.1. Older Dryas/Alleröd Transition
4.1.2. Alleröd/Younger Dryas Transition
4.1.3. Younger Dryas/Early Holocene Transition
4.2. Comparison of Low-Resolution and High-Resolution Cladocera Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shurin, J.B.; Clasen, J.L.; Greig, H.S.; Kratina, P.; Thompson, P.L. Warming Shifts Top-down and Bottom-up Control of Pond Food Web Structure and Function. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 3008–3017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kozarski, S. Paleogeografia Polski w vistulianie. In Geografia Polski Środowisko przyrodnicze; Starkel, L., Ed.; Wydawnictwo Naukowe PWN: Warszawa, Poland, 1991; p. 80. [Google Scholar]
- Marks, L. Last Glacial Maximum in Poland. Quat. Sci. Rev. 2002, 21, 103–110. [Google Scholar] [CrossRef]
- Kolstrup, E. Climate and Stratigraphy in Northwestern Europe between 30,000 B.P. and 13,000 B.P. with Special Reference to the Netherlands. Mededelingen Rijk Geologische Dienst. 1980, 32, 181–253. [Google Scholar]
- Dzieduszyńska, D.A.; Forysiak, J. Chronostratigraphy of the Late Vistulian in Central Poland and the Correlation with Vistulian Glacial Phases. Stud. Quat. 2019, 36, 126385. [Google Scholar] [CrossRef]
- Wasylikowa, K. Vegetation and Climate of the Late-Glacial in Central Poland Based on Investigations Made at Witów near Łęczyca. Biul. Peryglac. 1964, 13, 261–417. [Google Scholar]
- Walanus, A.; Nalepka, D. Calibration of Mangerud’S Boundaries. Radiocarbon 2010, 52, 1639–1644. [Google Scholar] [CrossRef] [Green Version]
- Marks, L. Timing of the Late Vistulian (Weichselian) Glacial Phases in Poland. Quat. Sci. Rev. 2012, 44, 81–88. [Google Scholar] [CrossRef]
- Starkel, L.; Michczyńska, D.J.; Krąpiec, M.; Margielewski, W.; Nalepka, D.; Pazdur, A. Progress in the Holocene Chrono-Climatostratigraphy of Polish Territory. Geochronometria 2013, 40, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Rasmussen, S.O.; Bigler, M.; Blockley, S.P.; Blunier, T.; Buchardt, S.L.; Clausen, H.B.; Cvijanovic, I.; Dahl-Jensen, D.; Johnsen, S.J.; Fischer, H.; et al. A Stratigraphic Framework for Abrupt Climatic Changes during the Last Glacial Period Based on Three Synchronized Greenland Ice-Core Records: Refining and Extending the INTIMATE Event Stratigraphy. Quat. Sci. Rev. 2014, 106, 14–28. [Google Scholar] [CrossRef] [Green Version]
- Helmens, K.F. The Last Interglacial–Glacial Cycle (MIS 5–2) Re-Examined Based on Long Proxy Records from Central and Northern Europe. Quat. Sci. Rev. 2014, 86, 115–143. [Google Scholar] [CrossRef]
- Gkinis, V.; Vinther, B.M.; Popp, T.J.; Quistgaard, T.; Faber, A.-K.; Holme, C.T.; Jensen, C.-M.; Lanzky, M.; Lütt, A.-M.; Mandrakis, V.; et al. A 120,000-Year Long Climate Record from a NW-Greenland Deep Ice Core at Ultra-High Resolution. Sci. Data 2021, 8, 141. [Google Scholar] [CrossRef]
- Kattel, G.R.; Augustinus, P.C. Cladoceran-Inferred Environmental Change during the LGM to Holocene Transition from Onepoto Maar Paleolake, Auckland, New Zealand. N. Z. J. Geol. Geophys. 2010, 53, 31–42. [Google Scholar] [CrossRef]
- Korponai, J.; Magyari, E.K.; Buczkó, K.; Iepure, S.; Namiotko, T.; Czakó, D.; Kövér, C.; Braun, M. Cladocera Response to Late Glacial to Early Holocene Climate Change in a South Carpathian Mountain Lake. Hydrobiologia 2011, 676, 223. [Google Scholar] [CrossRef]
- Pawłowski, D. Early Development of Late Vistulian (Weichselian) Lacustrine Sediments in the Żabieniec Swamp (Central Poland). Geochronometria 2012, 39, 197–211. [Google Scholar] [CrossRef] [Green Version]
- Suchora, M. Late-Glacial Cladoceran Succession in Three Lakes of the Chełm Hills Region (Łęczna-Włodawa Lake Group, SE Poland). Stud. Quat. 2013, 29, 9–21. [Google Scholar]
- Nováková, K.; van Hardenbroek, M.; van der Knaap, W.O. Response of Subfossil Cladocera in Gerzensee (Swiss Plateau) to Early Late Glacial Environmental Change. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2013, 391, 84–89. [Google Scholar] [CrossRef]
- Ralska-Jasiewiczowa, M.; Goslar, T.; Różański, K.; Wacnik, A.; Czernik, J.; Chróst, L. Very Fast Environmental Changes at the Pleistocene/Holocene Boundary, Recorded in Laminated Sediments of Lake Gościż, Poland. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2003, 193, 225–247. [Google Scholar] [CrossRef]
- Müller, D.; Tjallingii, R.; Płóciennik, M.; Luoto, T.P.; Kotrys, B.; Plessen, B.; Ramisch, A.; Schwab, M.J.; Błaszkiewicz, M.; Słowiński, M.; et al. New Insights into Lake Responses to Rapid Climate Change: The Younger Dryas in Lake Gościąż, Central Poland. Boreas 2021, 50, 535–555. [Google Scholar] [CrossRef]
- Frey, D.G. Cladocera Analysis. In Handbook of Holocene Palaeoecology and Palaeohydrology; Berglund, B.E., Ed.; The Blackburn Press: Caldwell, NJ, USA, 1986; pp. 667–692. ISBN 0471906913. [Google Scholar]
- Korhola, A.; Rautio, M. Cladocera and Other Branchiopod Crustaceans. In Tracking Environmental Change Using Lake Sediments: Volume 4: Zoological Indicators; Smol, J.P., Birks, H.J.B., Last, W.M., Eds.; Springer: Dordrecht, The Netherlands, 2001; pp. 5–41. ISBN 978-0-306-47671-6. [Google Scholar]
- Korhola, A.; Tikkanen, M.; Weckström, J. Quantification of Holocene Lake-Level Changes in Finnish Lapland Using a Cladocera–Lake Depth Transfer Model. J. Paleolimnol. 2005, 34, 175–190. [Google Scholar] [CrossRef]
- Locke, A.; Sprules, W.G. Effects of Acidic PH and Phytoplankton on Survival and Condition of Bosmina Longirostris and Daphnia Pulex. Hydrobiologia 2000, 437, 187–196. [Google Scholar] [CrossRef]
- Szeroczyńska, K. Impact of Prehistoric Settlements on the Cladocera in the Sediments of Lakes Suszek, Błȩdowo, and Skrzetuszewskie. In Biology of Cladocera; Kořínek, V., Frey, D.G., Eds.; Springer: Dordrecht, The Netherlands, 1991; pp. 105–114. ISBN 9789048141012. [Google Scholar]
- Lotter, A.F.; Birks, H.J.B.; Hofmann, W.; Marchetto, A. Modern Diatom, Cladocera, Chironomid, and Chrysophyte Cyst Assemblages as Quantitative Indicators for the Reconstruction of Past Environmental Conditions in the Alps. I. Climate. J. Paleolimnol. 1997, 18, 395–420. [Google Scholar] [CrossRef] [Green Version]
- Pawłowski, D. Warunki ekologiczne rozwoju późnovistuliańskiego zbiornika Koźmin Las w świetle analizy Cladocera. Acta Geogr. Lodz. 2014, 102, 65–70. [Google Scholar]
- Pawłowski, D. Analiza Cladocera z torfowiska Żabieniec. In Torfowisko Żabieniec: Warunki Naturalne, Rozwój i Zapis Zmian Paleoekologicznych w Jego Osadach; Twardy, J., Żurek, S., Forysiak, J., Eds.; Bogucki Wydawnictwo Naukowe: Poznań, Poland, 2010; pp. 129–139. [Google Scholar]
- Słowiński, M.; Marcisz, K.; Płóciennik, M.; Obremska, M.; Pawłowski, D.; Okupny, D.; Słowińska, S.; Borówka, R.; Kittel, P.; Forysiak, J.; et al. Drought as a Stress Driver of Ecological Changes in Peatland—A Palaeoecological Study of Peatland Development between 3500 BCE and 200 BCE in Central Poland. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2016, 461, 272–291. [Google Scholar] [CrossRef]
- Forysiak, J.; Obremska, M.; Pawłowski, D.; Kittel, P. Late Vistulian and Holocene Changes in the Ner River Valley in Light of Geological and Palaeoecological Data from the Ner-Zawada Peatland. Geologija 2010, 52, 25–33. [Google Scholar] [CrossRef]
- Twardy, J.; Żurek, S.; Forysiak, J. Torfowisko Żabieniec: Warunki Naturalne, Rozwój i Zapis Zmian Paleoekologicznych w Jego Osadach; Bogucki Wydawnictwo Naukowe: Poznań, Poland, 2010. [Google Scholar]
- Lamentowicz, M.; Balwierz, Z.; Forysiak, J.; Płóciennik, M.; Kittel, P.; Kloss, M.; Twardy, J.; Żurek, S.; Pawlyta, J. Multiproxy Study of Anthropogenic and Climatic Changes in the Last Two Millennia from a Small Mire in Central Poland. In Palaeolimnological Proxies as Tools of Environmental Reconstruction in Fresh Water; Buczkó, K., Korponai, J., Padisák, J., Starratt, S.W., Eds.; Springer: Dordrecht, The Netherlands, 2009; pp. 213–230. ISBN 9789048133864. [Google Scholar]
- Petera-Zganiacz, J.; Dzieduszyńska, D.A.; Milecka, K.; Okupny, D.; Słowiński, M.; Michczyńska, D.J.; Forysiak, J.; Twardy, J. Climate and Abiotic Landscape Controls of Younger Dryas Environmental Variability Based on a Terrestrial Archive (the Żabieniec Mire, Central Poland). CATENA 2022, 219, 106611. [Google Scholar] [CrossRef]
- Forysiak, J.; Twardy, J. Budowa Geologiczna i Paleogeografia Torfowiska Żabieniec i Jego Otoczenia. In Torfowisko Żabieniec: Warunki Naturalne, Rozwój i Zapis Zmian Paleoekologicznych w Jego Osadach; Twardy, J., Żurek, S., Forysiak, J., Eds.; Bogucki Wydawnictwo Naukowe: Poznań, Poland, 2010; pp. 17–42. [Google Scholar]
- Majecka, A.; Balwierz, Z.; Forysiak, J.; Twardy, J. Eemian and Vistulian (Weichselian) Development of the Meltout Depression on the Watershed between the Mroga and Mrożyca Rivers (Central Poland) Based on Lithological and Pollen Analysis. Quat. Int. 2018, 467, 79–95. [Google Scholar] [CrossRef]
- Bronk Ramsey, C. Deposition Models for Chronological Records. Quat. Sci. Rev. 2008, 27, 42–60. [Google Scholar] [CrossRef] [Green Version]
- Reimer, P.J.; Austin, W.E.N.; Bard, E.; Bayliss, A.; Blackwell, P.G.; Bronk Ramsey, C.; Butzin, M.; Cheng, H.; Edwards, R.L.; Friedrich, M.; et al. The IntCal20 Northern Hemisphere Radiocarbon Age Calibration Curve (0–55 Cal KBP). Radiocarbon 2020, 62, 725–757. [Google Scholar] [CrossRef]
- Kurek, J.; Korosi, J.B.; Jeziorski, A.; Smol, J.P. Establishing Reliable Minimum Count Sizes for Cladoceran Subfossils Sampled from Lake Sediments. J. Paleolimnol. 2010, 44, 603–612. [Google Scholar] [CrossRef]
- Szeroczyńska, K. Sarmaja-Korjonen Atlas of Subfossil Cladocera from Central and Northern Europe—Towarzystwo Przyjaciół Dolnej Wisły; Friends of the Lower Vistula Society: Poznań, Poland, 2007. [Google Scholar]
- Juggins, S. C2 Version 1.5: Software for Ecological and Palaeoecological Data Analysis and Visualisation. 2007. Available online: https://eprints.ncl.ac.uk (accessed on 24 November 2022).
- Bengtsson, L.; Enell, M. Chemical Analysis. In Handbook of Holocene Palaeoecology and Palaeohydrology; Berglund, B.E., Ed.; John Wiley and Sons: Chichester, UK, 1986. [Google Scholar]
- Heiri, O.; Lotter, A.F.; Lemcke, G. Loss on Ignition as a Method for Estimating Organic and Carbonate Content in Sediments: Reproducibility and Comparability of Results. J. Paleolimnol. 2001, 25, 101–110. [Google Scholar] [CrossRef]
- Berglund, B.E.; Ralska-Jasiewiczowa, M. Pollen Analysis and Pollen Diagrams. In Handbook of Holocene palaeoecology and palaeohydrology; Berglund, B.E., Ed.; The Blackburn Press: Caldwell, NJ, USA, 1986; pp. 455–484. [Google Scholar]
- R Core Team. European Environment Agency. 2020. Available online: https://www.eea.europa.eu/data-and-maps/indicators/oxygen-consuming-substances-in-rivers/r-development-core-team-2006 (accessed on 24 November 2022).
- Ter Braak, C.J.F.; Smilauer, P. Canoco Reference Manual and User’s Guide: Software for Ordination; Version 5.0; Microcomputer Power: Ithaca, NY, USA, 2012. [Google Scholar]
- Okupny, D.; Borówka, R.K.; Forysiak, J.; Twardy, J.; Kloss, M.; Żurek, S. The Relationship between the Chemical Composition and Lithology of Late Glacial and Holocene Biogenic Deposits of the Żabieniec Mire (Central Poland). Geol. Q. 2021, 65, 1579. [Google Scholar] [CrossRef]
- Azouzi, R.; Charef, A.; Hamzaoui, A.H. Assessment of Effect of PH, Temperature and Organic Matter on Zinc Mobility in a Hydromorphic Soil. Environ. Earth Sci. 2015, 74, 2967–2980. [Google Scholar] [CrossRef]
- Bennike, O.; Sarmaja-Korjonen, K.; SeppÄnen, A. Reinvestigation of the Classic Late-Glacial Bølling Sø Sequence, Denmark: Chronology, Macrofossils, Cladocera and Chydorid Ephippia. J. Quat. Sci. 2004, 19, 465–478. [Google Scholar] [CrossRef]
- Szeroczyńska, K. Cladocera Analysis in the Late-Glacial Sediments of Lake Gościąż. In Lake Gościąż, Central Poland. A Monographic Study; Ralska-Jasiewiczowa, M., Starkel, L., Eds.; Library of the W. Szafer Institute of Botany PAS: Kraków, Poland, 1998; pp. 148–158. ISBN 9788385444640. [Google Scholar]
- Zawiska, I. Wioślarki. In Mikroprzeszłość. Badania Specjalistyczne w Archeologii; Kurzawska, A., Sobkowiak-Tabaka, I., Eds.; Wydział Archeologii UAM: Poznań, Poland, 2021; pp. 115–128. [Google Scholar]
- Błędzki, L.; Rybak, J.I. Freshwater Crustacean Zooplankton of Europe: Cladocera & Copepoda (Calanoida, Cyclopoida) Key to Species Identification, with Notes on Ecology, Distribution, Methods and Introduction to Data Analysis; Springer International Publishing: Cham, Switzerland, 2016; ISBN 9783319298702. [Google Scholar]
- Szeroczyńska, K. Cladocera jako wskaźnik ekologiczny w późnoczwartorzędowych osadach jeziornych Polski Północnej. Acta Palaeontol. Pol. 1985, 30, 3–69. [Google Scholar]
- Harmsworth, R.V. The Developmental History of Blelham Tarn (England) as Shown by Animal Microfossils, with Special Reference to the Cladocera. Ecol. Monogr. 1968, 38, 223–241. [Google Scholar] [CrossRef]
- Szeroczyńska, K.; Zawisza, E. Records of the 8200calBP Cold Event Reflected in the Composition of Subfossil Cladocera in the Sediments of Three Lakes in Poland. Quat. Int. 2011, 233, 185–193. [Google Scholar] [CrossRef]
- Rybak, J.I.; Błędzki, L.A. Słodkowodne Skorupiaki Planktonowe. Klucz Do Oznaczania Gatunków; Warsaw University Press: Warsaw, Poland, 2010; ISBN 9788323511632. [Google Scholar]
- Szeroczyńska, K.; Zawisza, E. Paleolimnologia—Historia rozwoju jezior w Polsce w świetle badań fauny wioślarek. Stud. Limnol. Telmatologica 2007, 1, 51–60. [Google Scholar]
- Rundgren, S. Earthworms and Soil Remediation: Liming of Acidic Coniferous Forest Soils in Southern Sweden. Pedobiologia 1994, 38, 519–529. [Google Scholar]
- Sarmaja-Korjonen, K.; Seppänen, A.; Bennike, O. Pediastrum Algae from the Classic Late Glacial Bølling Sø Site, Denmark: Response of Aquatic Biota to Climate Change. Rev. Palaeobot. Palynol. 2006, 138, 95–107. [Google Scholar] [CrossRef]
- Meyers, P.A.; Teranes, J.L. Sediment Organic Matter. In Tracking Environmental Change Using Lake Sediments; Springer: Dordrecht, The Netherlands, 2001; pp. 239–269. [Google Scholar]
- Rosén, P.; Hammarlund, D. Effects of Climate, Fire and Vegetation Development on Holocene Changes in Total Organic Carbon Concentration in Three Boreal Forest Lakes in Northern Sweden. Biogeosciences 2007, 4, 975–984. [Google Scholar] [CrossRef] [Green Version]
- Szeroczyńska, K. Wioślarki (Cladocera, Crustacea) jako źródło informacji w badaniach osadów jeziornych. Stud. Geol. Pol. 1998, 112, 9–28. [Google Scholar]
- Szeroczyńska, K. Cladoceran Succession in Lakes and Peat Bogs of Łęczna-Włodawa Lake District. Limnol. Rev. 2003, 3, 235–242. [Google Scholar]
- Szeroczyńska, K. The Significance of Subfossil Cladocera in Stratigraphy of Late Glacial and Holocene. Stud. Quat. 2006, 23, 37–45. [Google Scholar]
- Pražáková, M.; Veselý, J.; Fott, J.; Majer, V.; Kopáček, J. The Long-Term Succession of Cladoceran Fauna and Palaeoclimate Forcing: A 14,600-Year Record from Plešné Lake, the Bohemian Forest. Biologia 2006, 61, S387–S399. [Google Scholar] [CrossRef]
- Mirosław-Grabowska, J.; Zawisza, E. Reaction of the Lake Environment to the Holocene Warming Depending on the Distance to the Maximum Extent of the Vistulian Ice Sheet. CATENA 2018, 171, 494–504. [Google Scholar] [CrossRef]
- Niska, M.; Mirosław-Grabowska, J. Eemian Environmental Changes Recorded in Lake Deposits from Rzecino (NW Poland): Cladocera, Isotopic and Selected Geochemical Data. J. Paleolimnol. 2015, 53, 89–105. [Google Scholar] [CrossRef] [Green Version]
- Milecka, K.; Kowalewski, G.; Szeroczyńska, K. Climate-Related Changes during the Late Glacial and Early Holocene in Northern Poland, as Derived from the Sediments of Lake Sierzywk. Hydrobiologia 2011, 676, 187. [Google Scholar] [CrossRef]
- Forysiak, J. Zapis zmian środowiska przyrodniczego późnego vistulianu i holocenu w osadach torfowisk regionu łódzkiego. Acta Geogr. Lodz. 2012, 99, 1–163. [Google Scholar]
- Fajer, M.; Waga, J.M.; Rzetala, M.; Szymczyk, A.; Nita, M.; Machowski, R.; Rzetala, M.A.; Ruman, M. The Late Vistulian and Holocene Evolution of Jezioro Lake: A Record of Environmental Change in Southern Poland Found in Deposits and Landforms. J. Paleolimnol. 2012, 48, 651–667. [Google Scholar] [CrossRef] [Green Version]
- Ramrath, A.; Nowaczyk, N.R.; Negendank, J.F.W. Sedimentological Evidence for Environmental Changes since 34,000 Years BP from Lago Di Mezzano, Central Italy. J. Paleolimnol. 1999, 21, 423–435. [Google Scholar] [CrossRef]
- Goslar, T.; Kuc, T.; Ralska-Jasiewiczowa, M.; Rózánski, K.; Arnold, M.; Bard, E.; van Geel, B.; Pazdur, M.; Szeroczyńska, K.; Wicik, B.; et al. High-Resolution Lacustrine Record of the Late Glacial/Holocene Transition in Central Europe. Quat. Sci. Rev. 1993, 12, 287–294. [Google Scholar] [CrossRef]
- Szeroczyńska, K. Cladocera w Osadach Jeziora Biskupińskiego. In Zarys Zmian Środowiska Geograficznego Okolic Biskupina pod Wpływem Czynników Naturalnych i Antropogenicznych w Późnym Glacjale i Holocenie; O.W. Turpress: Toruń, Poland, 1995; pp. 181–198. [Google Scholar]
- Bińka, K.; Cieśla, A.; Lacka, B.; Marciniak, B.; Madeyska, T.; Szeroczyńska, K.; Więckowiski, K. Lake Evolution during the Last 12 000 Years: Blędowo Lake (Central Poland). Stud. Geol. Pol. 1991, 10, 100. [Google Scholar]
- Polcyn, I. Analiza Kopalnych Wioślarek. In Paleoekologiczne Studium Późnoglacjalnych Osadów Jeziora Lednica w Imiołkach; Tobolski, K., Ed.; Lednicki Park Krajobrazowy: Poznań, Poland, 1998; pp. 51–54. [Google Scholar]
- Milecka, K.; Szeroczynska, K. Changes in Macrophytic Flora and Planktonic Organisms in Lake Ostrowite, Poland, as a Response to Climatic and Trophic Fluctuations. Holocene 2005, 15, 74–84. [Google Scholar] [CrossRef]
- Zawisza, E.; Szeroczyńska, K. The Development History of Wigry Lake as Shown by Subfossil Cladocera. Geochronometria 2007, 27, 67–74. [Google Scholar] [CrossRef] [Green Version]
- Duigan, C.A.; Birks, H.H. The Late-Glacial and Early-Holocene Palaeoecology of Cladoceran Microfossil Assemblages at Kråkenes, Western Norway, with a Quantitative Reconstruction of Temperature Changes. J. Paleolimnol. 2000, 23, 67–76. [Google Scholar] [CrossRef]
- Pawłowski, D. The Usefulness of Subfossil Cladocera Remains in Younger Dryas Climatic Reconstructions in Central Poland. Acta Geol. Pol. 2017, 67, 567–584. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rudna, M.; Wojewódka-Przybył, M.; Forysiak, J.; Milecka, K.; Okupny, D. Cladocera Responses to the Climate-Forced Abrupt Environmental Changes Related to the Late Glacial/Holocene Transition. Water 2023, 15, 348. https://doi.org/10.3390/w15020348
Rudna M, Wojewódka-Przybył M, Forysiak J, Milecka K, Okupny D. Cladocera Responses to the Climate-Forced Abrupt Environmental Changes Related to the Late Glacial/Holocene Transition. Water. 2023; 15(2):348. https://doi.org/10.3390/w15020348
Chicago/Turabian StyleRudna, Marta, Marta Wojewódka-Przybył, Jacek Forysiak, Krystyna Milecka, and Daniel Okupny. 2023. "Cladocera Responses to the Climate-Forced Abrupt Environmental Changes Related to the Late Glacial/Holocene Transition" Water 15, no. 2: 348. https://doi.org/10.3390/w15020348
APA StyleRudna, M., Wojewódka-Przybył, M., Forysiak, J., Milecka, K., & Okupny, D. (2023). Cladocera Responses to the Climate-Forced Abrupt Environmental Changes Related to the Late Glacial/Holocene Transition. Water, 15(2), 348. https://doi.org/10.3390/w15020348





