A Floatable and Highly Water-Durable TiO2-Coated Net for Photocatalytic Antibacterial Water Treatment in Developing Countries
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Surface Modification of TiO2
2.3. Photocatalyst Coating
2.4. Durability Test
2.5. Photocatalytic Sterilization of Water Containing Bacteria
3. Results and Discussion
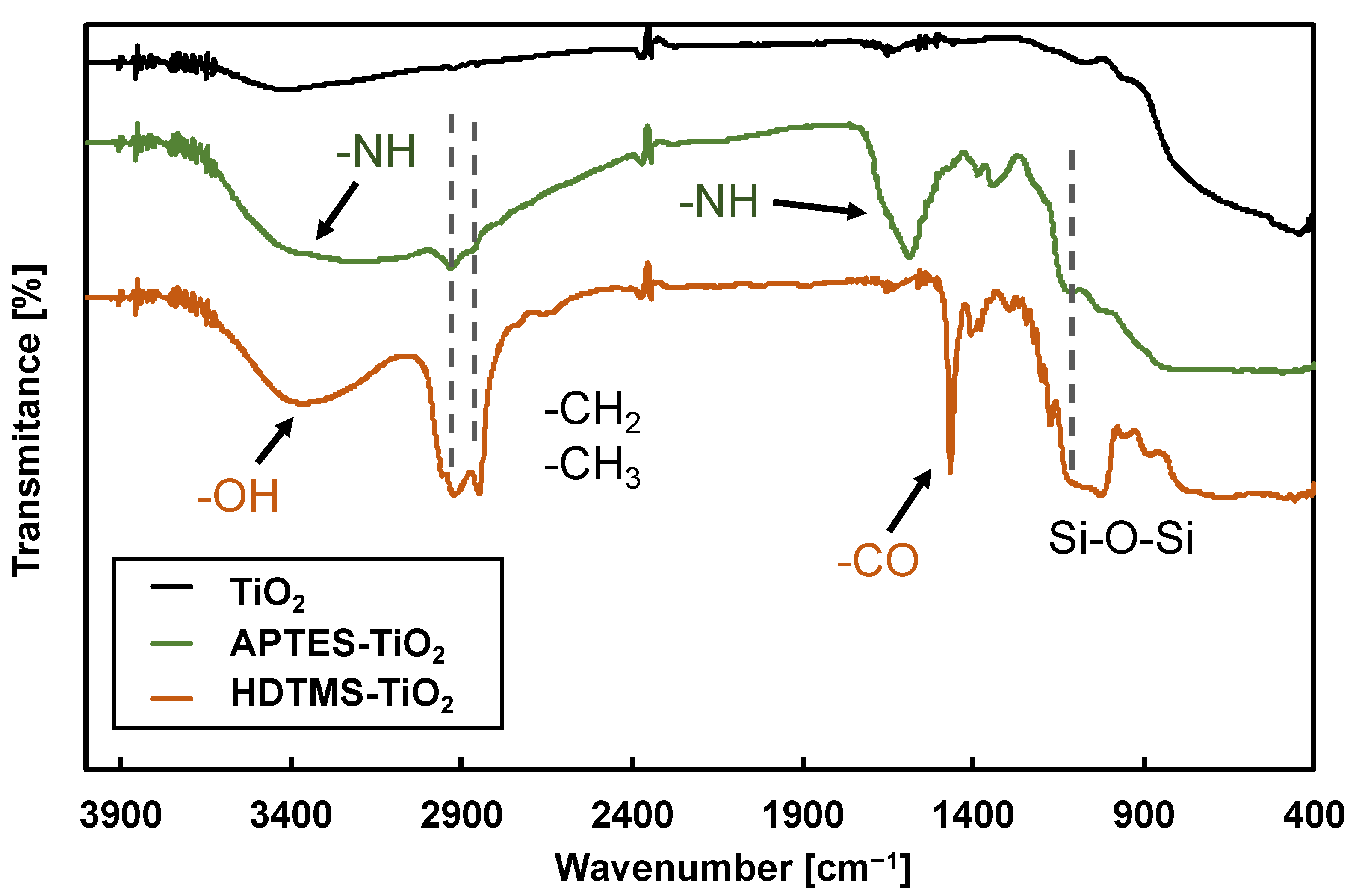
3.1. FTIR Spectra of Surface-Modified TiO2
3.2. Evaluation of Coated Material
3.3. Lab-Scale Durability Test
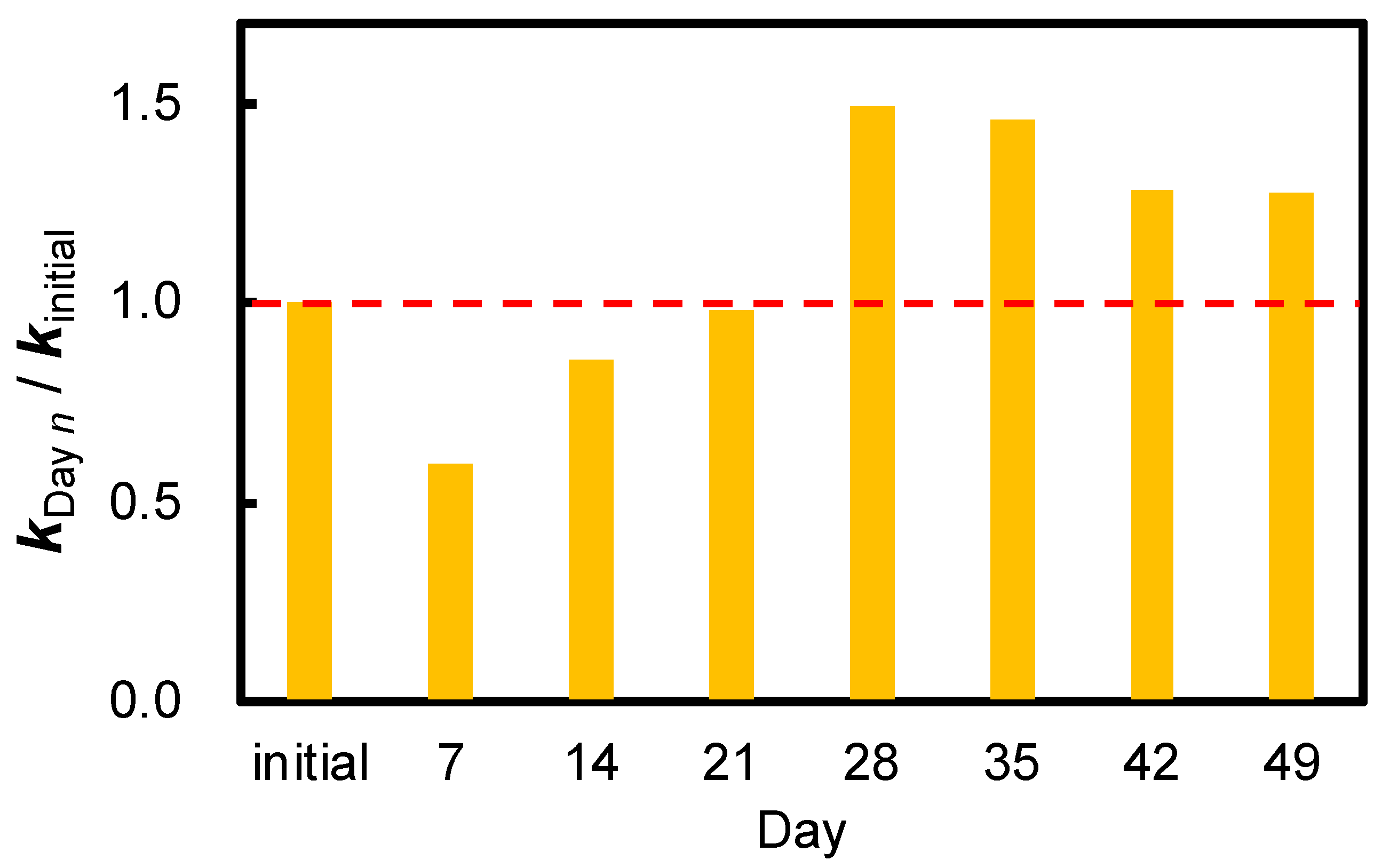
3.4. Durability in the Natural Environment
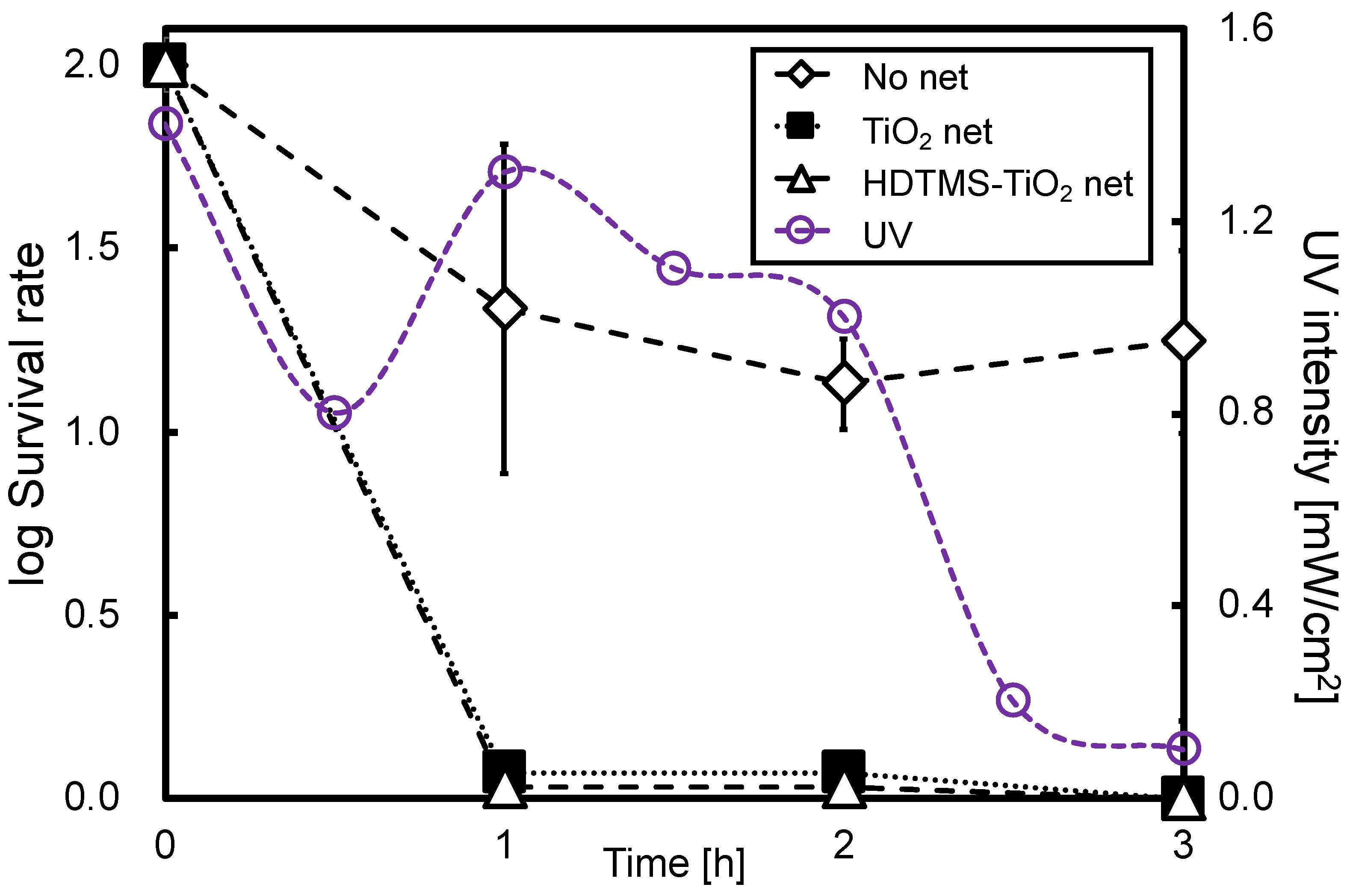
3.5. Photocatalytic Sterilization of Water Containing Bacteria
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Negishi, N.; Chawengkijwanich, C.; Pimpha, N.; Larpkiattaworn, S.; Charinpanitkul, T. Performance verification of the photocatalytic solar water purification system for sterilization using actual drinking water in Thailand. J. Water. Process. Eng. 2019, 31, 100835. [Google Scholar] [CrossRef]
- World Health Organization (WHO) and the United Nations Children’s Fund (UNICEF). Progress on Drinking Water, Sanitation and Hygiene: 2017 Update and SDG Baselines. Available online: https://apps.who.int/iris/bitstream/handle/10665/258617/9789241512893-eng.pdf (accessed on 10 November 2022).
- WHO. The World Health Report: Making a Difference 1999. Available online: https://apps.who.int/iris/bitstream/handle/10665/42167/WHR_1999.pdf (accessed on 10 November 2022).
- National Research Council. Drinking Water and Health, Volume 7: Disinfectants and Disinfectant By-Products; The National Academic Press: Washington DC, USA, 1987. [Google Scholar]
- Xie, Y. Disinfection Byproducts in Drinking Water: Formation, Analysis, and Control; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Palmucci, H.E.; Premuzic, Z.; Mascarini, L.; Campetella, C.; Lopez, V. The influence of chlorination on the phytotoxicity and the production of Zinnia elegans. J. Appl. Hortic. 2010, 12, 161–164. [Google Scholar] [CrossRef]
- Arnal, J.M.; Garcia-Fayos, B.; Verdu, G.; Lora, J. Ultrafiltration as an alternative membrane technology to obtain safe drinking water from surface water: 10 years of experience on the scope of the AQUAPOT project. Desalination 2009, 248, 34–41. [Google Scholar] [CrossRef]
- Conroy, R.M.; Meegan, M.E.; Joyce, T.; McGuigan, K.; Barnes, J. Solar disinfection of drinking water protects against cholera in children under 6 years of age. Arch. Dis. Child. 2001, 85, 293–295. [Google Scholar] [CrossRef] [PubMed]
- Acra, A.; Jurdi, M.; Mu’allem, H.; Karahagopian, Y.; Raffoul, Z. Water Disinfection by Solar Radiation Assessment and Application; International Development Research Center: Ottawa, ON, Canada, 2003. [Google Scholar]
- Keogh, M.B.; Elmusharaf, K.; Borde, P.; McGuian, K.G. Evaluation of the natural coagulant Moringa oleifera as a pretreatment for SODIS in contaminated turbid water. Sol. Energy 2017, 158, 448–454. [Google Scholar] [CrossRef]
- Blake, D.M.; Maness, P.-C.; Huang, Z.; Wolfrum, E.J.; Huang, J.; Jacoby, W.A. Application of the Photocatalytic Chemistry of Titanium Dioxide to Disinfection and the Killing of Cancer Cells. Sep. Purif. Method 1999, 28, 1–50. [Google Scholar] [CrossRef]
- Matsuura, R.; Kawamura, A.; Matsumoto, Y.; Fukushima, T.; Fujimoto, K.; Ochiai, H.; Somei, J.; Aida, Y. Rutile-TiO2/PtO2 Glass Disinfects Aquatic Legionella pneumophila via Morphology Change and Endotoxin Degradation under LED Irradiation. Catalysts 2022, 12, 856. [Google Scholar] [CrossRef]
- Saikachi, A.; Sugasawara, K.; Suzuki, T. Analyses of the Effect of Peptidoglycan on Photocatalytic Bactericidal Activity Using Different Growth Phases Cells of Gram-Positive Bacterium and Spheroplast Cells of Gram-Negative Bacterium. Catalysts 2021, 11, 147. [Google Scholar] [CrossRef]
- Braham, R.J.; Harris, A.T. Review of Major Design and Scale-up Considerations for Solar Photocatalytic Reactors. Ind. Eng. Chem. Res. 2009, 48, 8890–8905. [Google Scholar] [CrossRef]
- Yamada, N.; Suzumura, M.; Koiwa, F.; Negishi, N. Differences in elimination efficiencies of Escherichia coli in freshwater and seawater as a result of TiO2 photocatalysis. Water Res. 2013, 47, 2770–2776. [Google Scholar] [CrossRef]
- Farrow, G. Crystallinity, ‘crystallite size’ and melting point of polypropylene. Polymer 1963, 4, 191–197. [Google Scholar] [CrossRef]
- Wang, R.; Hashimoto, K.; Fujishima, A.; Chikuni, M.; Kojima, E.; Kitamura, A.; Shimohigoshi, M.; Watanabe, T. Light-induced amphiphilic surfaces. Nature 1997, 388, 431–432. [Google Scholar] [CrossRef]
- García-Montelongo, X.L.; Martínez-de la Cruz, A.; Vázquez-Rodríguez, S.; Torres-Martínez, L.M. Photo-oxidative degradation of TiO2/polypropylene films. Mater. Res. Bull. 2014, 51, 56–62. [Google Scholar] [CrossRef]
- Mihailović, D.; Šaponjić, Z.; Molina, R.; Radoičić, M.; Esquena, J.; Jovančić, P.; Nedeljković, J.; Radetić, M. Multifunctional Properties of Polyester Fabrics Modified by Corona Discharge/Air RF Plasma and Colloidal TiO2 Nanoparticles. Polym. Compos. 2011, 32, 390–397. [Google Scholar] [CrossRef]
- Machida, M.; Norimoto, K.; Yamamoto, M. Control of the oxidizing and hydrophilicizing properties of photocatalytic TiO2 thin films coated on a polyethylene-terephthalate substrate. J. Ceram. Soc. Jpn. 2017, 125, 168–174. [Google Scholar] [CrossRef] [Green Version]
- Neyman, E.; Dillard, J.G.; Dillard, D.A. Plasma and Silane Surface Modification of SiC/Si: Adhesion and Durability for the Epoxy-SiC System. J. Adhes. 2006, 82, 331–353. [Google Scholar] [CrossRef]
- Lam, S.W.; Soetanto, A.; Amal, R. Self-cleaning performance of polycarbonate surfaces coated with titania nanoparticles. J. Nanopart. Res. 2009, 11, 1971. [Google Scholar] [CrossRef]
- Latthe, S.S.; Liu, S.; Terashima, C.; Nakata, K.; Fujishima, A. Transparent, Adherent, and Photocatalytic SiO2-TiO2 Coatings on Polycarbonate for Self-Cleaning Applications. Coatings 2014, 4, 497–507. [Google Scholar] [CrossRef] [Green Version]
- Chen, Q.; Yakovlev, N.L. Adsorption and interaction of organosilanes on TiO2 nanoparticles. Appl. Surf. Sci. 2010, 257, 1395–1400. [Google Scholar] [CrossRef]
- Ohtani, B.; Prieto-Mahaney, O.O.; Li, D.; Abe, R. What is Degussa (Evonik) P25? Crystalline composition analysis, reconstruction from isolated pure particles and photocatalytic activity test. J. Photochem. Photobiol. A Chem. 2010, 216, 179–182. [Google Scholar] [CrossRef]
- Japan Meteorological Agency. Monthly Total of Sunshine Duration (h). Available online: https://www.data.jma.go.jp/obd/stats/etrn/view/monthly_s3_en.php?block_no=47682&view=12 (accessed on 10 November 2022).
- Xu, B.; Zhang, Q. Preparation and Properties of Hydrophobically Modified Nano-SiO2 with Hexadecyltrimethoxysilane. ACS Omega 2021, 6, 9764–9770. [Google Scholar] [CrossRef] [PubMed]
- Ukaji, E.; Furusawa, T.; Sato, M.; Suzuki, N. The effect of surface modification with silane coupling agent on suppressing the photo-catalytic activity of fine TiO2 particles as inorganic UV filter. Appl. Surf. Sci. 2007, 254, 563–569. [Google Scholar] [CrossRef]
- Zhang, L.; Rao, L.; Wang, P.; Guo, X.; Wang, Y. Fabrication and photocatalytic performance evaluation of hydrodynamic erosion-resistant nano-TiO2-silicone resin composite films. Environ. Sci. Pollut. Res. 2019, 26, 4997–5007. [Google Scholar] [CrossRef] [PubMed]
- Dao, P.H.; Nguyen, T.D.; Nguyen, T.C.; Nguyen, A.H.; Mac, V.P.; Tran, H.T.; Phung, T.L.; Vu, Q.T.; Vu, D.H.; Ngo, T.C.Q.; et al. Assessment of some characteristics, properties of a novel waterborne acrylic coating incorporated TiO2 nanoparticles modified with silane coupling agent and Ag/Zn zeolite. Prog. Org. Coat. 2022, 163, 106641. [Google Scholar] [CrossRef]
- Gomes, A.I.; Santos, J.C.; Vilar, V.J.P.; Boaventura, R.A.R. Inactivation of Bacteria E. Coli and photodegradation of humic acids using natural sunlight. Appl. Catal. B Environ. 2009, 88, 283–291. [Google Scholar] [CrossRef]
- Rincón, A.G.; Pulgarin, C. Photocatalytical inactivation of E. coli: Effect of (continuous-intermittent) light intensity and of (suspended-fixed) TiO2 concentration. Appl. Catal. B Environ. 2003, 44, 263–284. [Google Scholar] [CrossRef]
- Valtin, H. “Drink at least eight glasses of water a day.” Really? Is there scientific evidence for “8 × 8”? Am. J. Physiol. Regul. Integr. Comp. Physiol. 2002, 283, R993–R1287. [Google Scholar] [CrossRef]



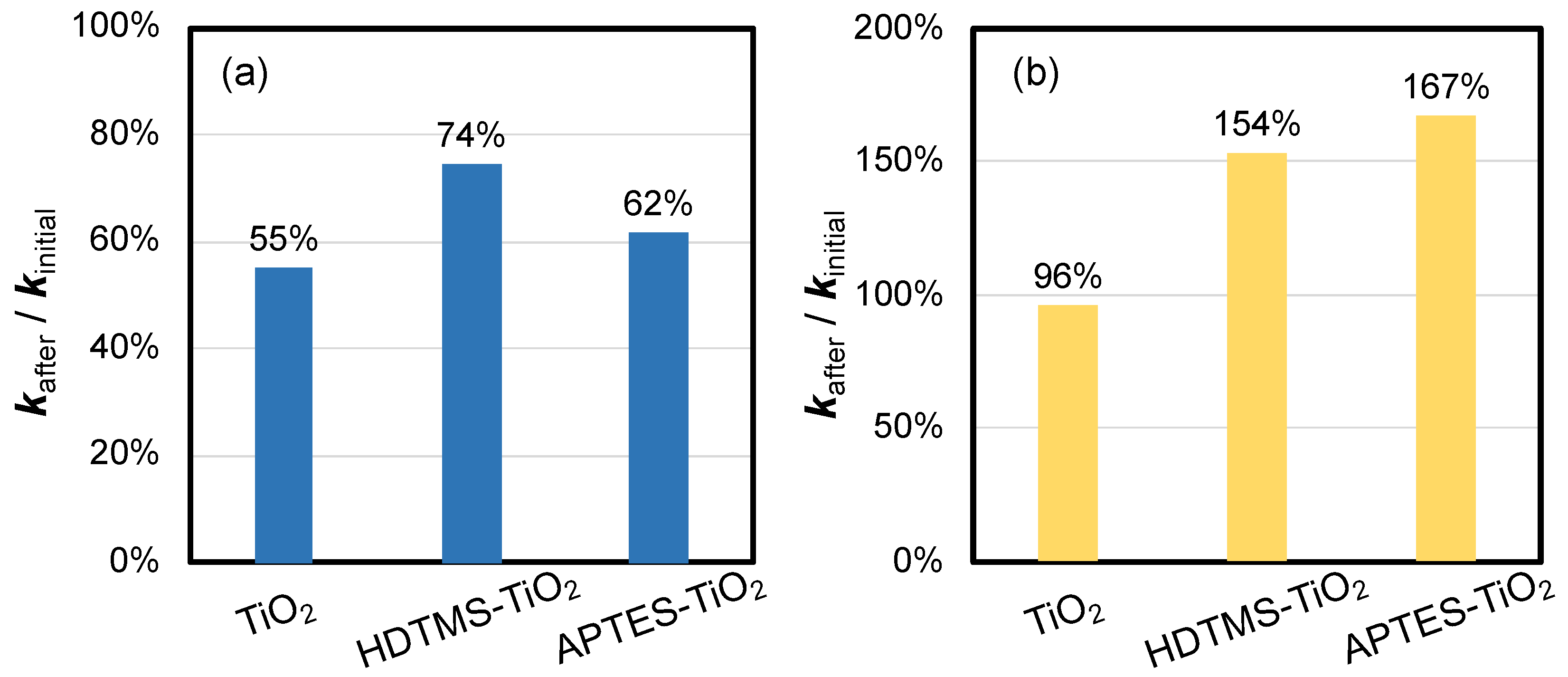




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Izuma, D.S.; Suzuki, N.; Suzuki, T.; Motomura, H.; Ando, S.; Fujishima, A.; Teshima, K.; Terashima, C. A Floatable and Highly Water-Durable TiO2-Coated Net for Photocatalytic Antibacterial Water Treatment in Developing Countries. Water 2023, 15, 320. https://doi.org/10.3390/w15020320
Izuma DS, Suzuki N, Suzuki T, Motomura H, Ando S, Fujishima A, Teshima K, Terashima C. A Floatable and Highly Water-Durable TiO2-Coated Net for Photocatalytic Antibacterial Water Treatment in Developing Countries. Water. 2023; 15(2):320. https://doi.org/10.3390/w15020320
Chicago/Turabian StyleIzuma, Dylan Shun, Norihiro Suzuki, Tomonori Suzuki, Haruka Motomura, Shiro Ando, Akira Fujishima, Katsuya Teshima, and Chiaki Terashima. 2023. "A Floatable and Highly Water-Durable TiO2-Coated Net for Photocatalytic Antibacterial Water Treatment in Developing Countries" Water 15, no. 2: 320. https://doi.org/10.3390/w15020320
APA StyleIzuma, D. S., Suzuki, N., Suzuki, T., Motomura, H., Ando, S., Fujishima, A., Teshima, K., & Terashima, C. (2023). A Floatable and Highly Water-Durable TiO2-Coated Net for Photocatalytic Antibacterial Water Treatment in Developing Countries. Water, 15(2), 320. https://doi.org/10.3390/w15020320








