Comparative Analysis of Metabolites between Different Altitude Schizothorax nukiangensis (Cyprinidae, Schizothoracine) on the Qinghai-Tibet Plateau in Nujiang River
Abstract
:1. Introduction
2. Materials and Methods
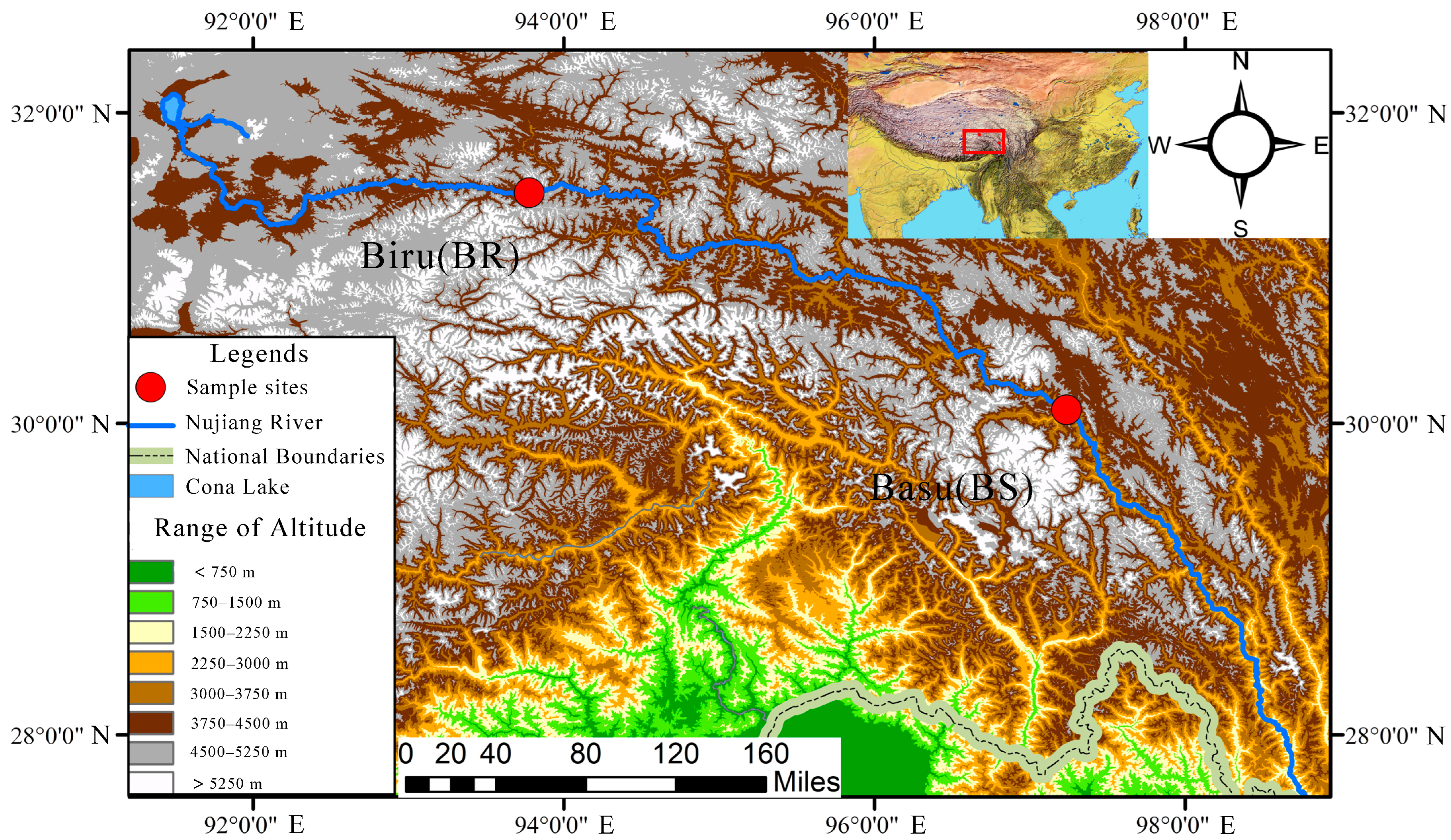
2.1. Fish Sampling
2.2. Metabolite Extraction and UPLC-MS Sample Preparation
2.3. LC-MS/MS Analysis
2.4. Data Processing and Statistical Analysis
3. Results
3.1. Environment Indicators and Metabolomics
3.2. PCA and OPLS-DA Analysis
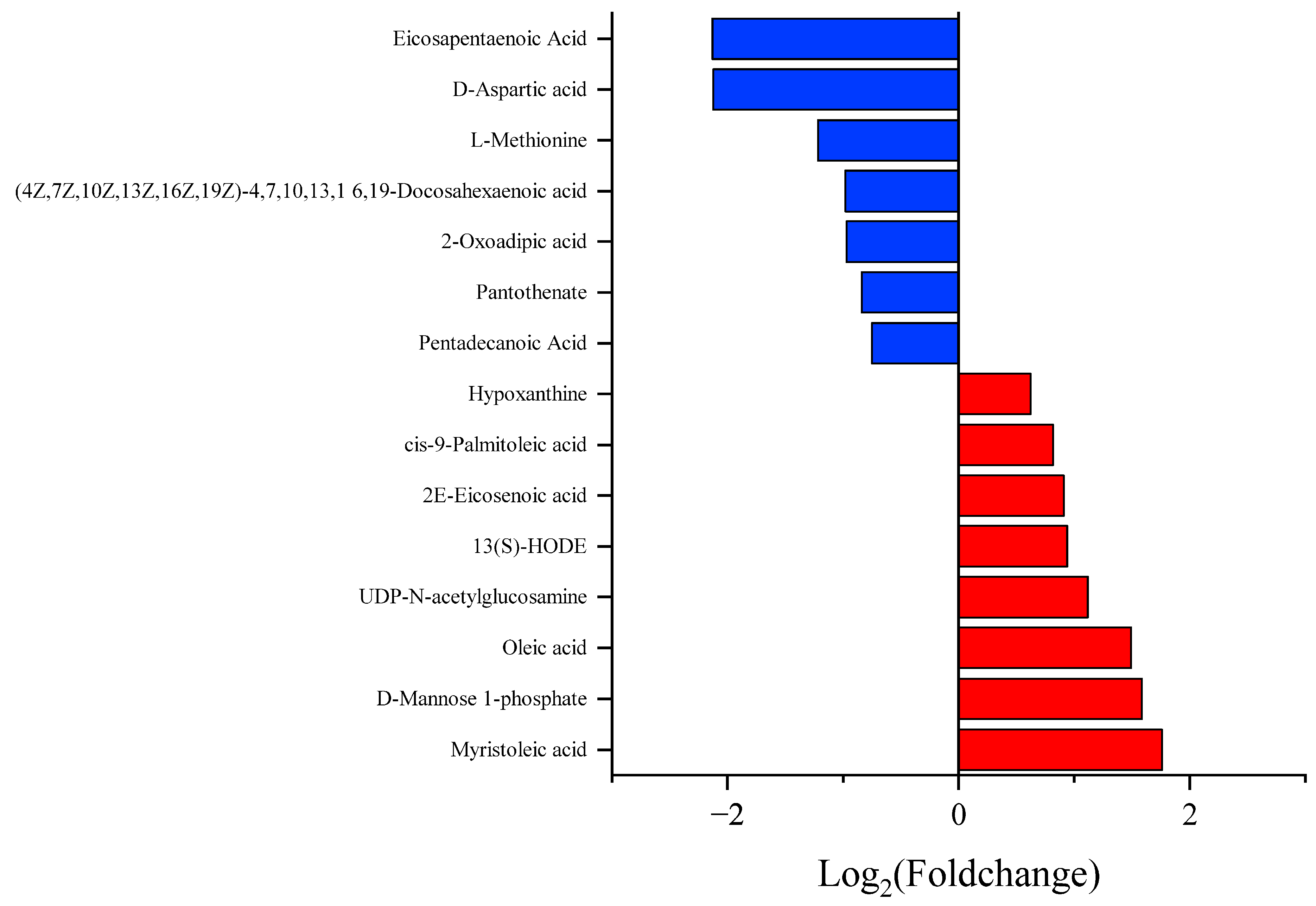
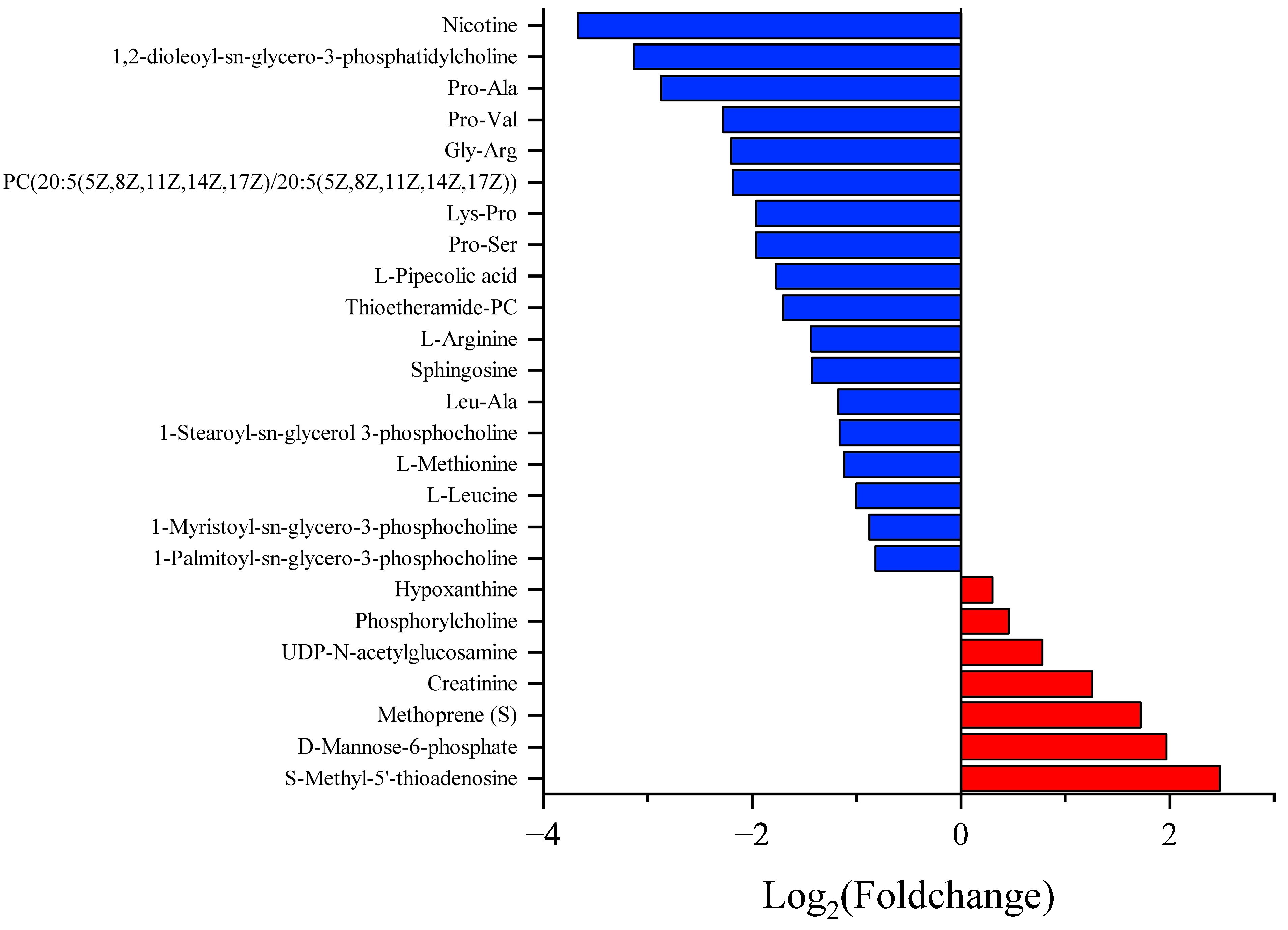
3.3. Different Metabolite Profiles
3.4. KEGG Pathway
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cheviron, Z.A.; Brumfield, R.T. Genomic insights into adaptation to high-altitude environments. Heredity 2012, 108, 354–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, X.; Meng, X.; Liu, Z.; Chang, J.; Wang, B.; Li, M.; Wengel, P.O.; Tian, S.; Wen, C.; Wang, Z.; et al. Population genomics reveals low genetic diversity and adaptation to hypoxia in snub-nosed monkeys. Mol. Biol. Evol. 2016, 33, 2670–2681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, G.D.; Fan, R.X.; Zhai, W.; Liu, F.; Wang, L.; Zhong, L.; Wu, H.; Yang, H.C.; Wu, S.F.; Zhu, C.L.; et al. Genetic convergence in the adaptation of dogs and humans to the high-altitude environment of the Tibetan Plateau. Genome Biol. Evol. 2014, 6, 2122–2128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miao, B.P.; Wang, Z.; Li, Y.X. Genomic analysis reveals hypoxia adaptation in the Tibetan Mastiff by introgression of the gray wolf from the Tibetan Plateau. Mol. Phylogenet. Evol. 2017, 34, 734–743. [Google Scholar]
- Yang, L.; Wang, Y.; Zhang, Z.; He, S. Comprehensive transcriptome analysis reveals accelerated genic evolution in a Tibet fish, Gymnodiptychus pachycheilus. Genome Biol. Evol. 2015, 7, 251–261. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.; Yang, H.; Xu, Q.; Wang, Y.; Sang, Z.; Yuan, H. Comparative metabolomics analysis of the response to cold stress of resistant and susceptible Tibetan hulless barley (Hordeum distichon). Phytochemistry 2020, 174, 112346. [Google Scholar] [CrossRef]
- Liu, F.; Liu, P.; Zhang, Y.; Sun, L.; Zhang, P.; Cao, M.; Zhou, H.; Wang, W.; Xu, J. Comparative metabolomics reveals that Agaricus bisporus fairy ring modulates the growth of alpine meadow plant on the Qinghai-Tibet Plateau. Ecol. Indic. 2021, 129, 107865. [Google Scholar] [CrossRef]
- Fiehn, O. Metabolomics—The link between genotypes and phenotypes. In Functional Genomics; Springer: Berlin/Heidelberg, Germany, 2002; pp. 155–171. [Google Scholar]
- Kell, D.B. Metabolomics and systems biology: Making sense of the soup. Curr. Opin. Microbiol. 2004, 7, 296–307. [Google Scholar] [CrossRef]
- Griffin, J.L.; Rosiers, C.D. Applications of metabolomics and proteomics to the mdx mouse model of Duchenne muscular dystrophy: Lessons from downstream of the transcriptome. Genome Med. 2009, 1, 32. [Google Scholar] [CrossRef] [Green Version]
- Holmes, E.; Wilson, I.D.; Nicholson, J.K. Metabolic phenotyping in health and disease. Cell 2008, 134, 714–717. [Google Scholar] [CrossRef] [Green Version]
- Wilson, I.D. Drugs, bugs, and personalized medicine: Pharmacometabonomics enters the ring. Proc. Natl. Acad. Sci. USA 2009, 106, 14187–14188. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.S.; da Costa, A.M.R.; Conceicao, L.E.C.; Dias, J.P.; Rodrigues, P.M.L.; Richard, N. Metabolic fingerprinting of gilthead seabream (Sparus aurata) liver to track interactions between dietary factors and seasonal temperature variations. PeerJ 2014, 2, e527. [Google Scholar] [CrossRef] [Green Version]
- Alfaro, A.C.; Young, T. Showcasing metabolomic applications in aquaculture: A review. Rev. Aquacult. 2016, 10, 135–152. [Google Scholar] [CrossRef]
- Van der Oost, R.; Beyer, J.; Vermeulen, N.P.E. Fish bioaccumulation and biomarkers in environmental risk assessment: A review. Environ. Toxicol. Phar. 2003, 13, 57–149. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhang, J.; Fu, J.; Shi, J.; Jiang, G. Biomonitoring: An appealing tool for assessment of metal pollution in the aquatic ecosystem. Anal. Chim. Acta 2008, 606, 135–150. [Google Scholar] [CrossRef]
- He, D.K.; Chen, Y.F.; Chen, Y.Y.; Chen, Z.M. Molecular phylogeny of the specialized schizothoracine fishes (Teleostei: Cyprinidae), with their implications for the uplift of the Qinghai-Tibetan Plateau. Chin. Sci. Bull. 2004, 49, 39–48. [Google Scholar] [CrossRef]
- He, D.K.; Chen, Y.F. Biogeography and molecular phylogeny of the genus Schizothorax (Teleostei: Cyprinidae) in China inferred from cytochrome b sequences. J. Biogeogr. 2006, 33, 1448–1460. [Google Scholar] [CrossRef]
- Chen, W.; Du, K.; He, S. Genetic structure and historical demography of Schizothorax nukiangensis (Cyprinidae) in continuous habitat. Ecol. Evol. 2015, 5, 984–995. [Google Scholar] [CrossRef]
- Chen, W.; Yue, X.; He, S. Genetic differentiation of the Schizothorax species complex (Cyprinidae) in the Nujiang River (upper Salween). Sci. Rep. 2017, 7, 5944. [Google Scholar] [CrossRef] [Green Version]
- Claireaux, G.; Lagardère, J.P. Influence of temperature, oxygen and salinity on the metabolism of the European sea bass. J. Sea Res. 1999, 42, 157–168. [Google Scholar] [CrossRef]
- Meador, J.P.; Bettcher, L.F.; Ellenberger, M.C.; Senn, T.D. Metabolomic profiling for juvenile Chinook salmon exposed to contaminants of emerging concern. Sci. Total Environ. 2020, 747, 141097. [Google Scholar] [CrossRef] [PubMed]
- Izral, N.M.; Brua, R.B.; Culp, J.M.; Yates, A.G. Crayfish tissue metabolomes effectively distinguish impacts of wastewater and agriculture in aquatic ecosystems. Sci. Total Environ. 2021, 760, 143322. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Nutrient Requirements of Fish and Shrimp; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Wang, Q.; Liu, M.D.; Zhu, F.Y.; Li, Z.; Liu, S.P.; Duan, X.B.; Chen, D.Q.; Yang, R.B. Comparative study of three species of Schizothoracine on feeding and digestive organs in upper Nujiang River. Chinese J. Zool. 2019, 54, 207–221, (In Chinese with English Abstract). [Google Scholar]
- Fortin, M.C.; Medeiros, A.S.; Gajewski, K.; Barley, E.M.; Larocque-Tobler, I.; Porinchu, D.F.; Wilson, S.E. Chironomid-environment relations in northern north America. J. Paleolimnol. 2015, 54, 223–237. [Google Scholar] [CrossRef]
- Kurihara, T.; Adachi, Y.; Yamagata, M.; Abe, K.; Akimoto, M.; Hashimoto, H.; Ishiguro, H.; Niimi, A.; Maeda, A.; Shigemoto, M. Role of eicosapentaenoic acid in lipid metabolism in the liver, with special reference to experimental fatty liver. Clin. Ther. 1994, 16, 830–837. [Google Scholar]
- Flachs, P.; Rossmeisl, M.; Kopecky, J. The effect of n-3 fatty acids on glucose homeostasis and insulin sensitivity. Physiol. Res. 2014, 63, S93–S118. [Google Scholar] [CrossRef]
- Masoodi, M.; Kuda, O.; Rossmeisl, M.; Flachs, P.; Kopecky, J. Lipid signaling in adipose tissue: Connecting inflammation & metabolism. BBA-Mol. Cell Biol. Lipids 2015, 1851, 503–518. [Google Scholar]
- Kong, L.; Cheng, S.; Xiang, X.; Liu, W.; Yu, D.; Yang, Y.; Zhou, J.; Huang, F.; Dong, G. Dietary conjugated linoleic acid modulates morphology, selective immune parameters, and gene expressions in the intestine of grass carp. Fish Shellfish Immun. 2019, 86, 536–548. [Google Scholar] [CrossRef]
- Hughes, R. Definitions for public health nutrition: A developing consensus. Public Health Nutr. 2003, 6, 615–620. [Google Scholar] [CrossRef] [Green Version]
- Brevik, A.; Veierød, M.B.; Drevon, C.A.; Andersen, L.F. Evaluation of the odd fatty acids 15:0 and 17:0 in serum and adipose tissue as markers of intake of milk and dairy fat. Eur. J. Clin. Nutr. 2005, 59, 1417–1422. [Google Scholar] [CrossRef]
- Dąbrowski, G.; Konopka, I. Update on food sources and biological activity of odd-chain, branched and cyclic fatty acids—A review. Trends Food Sci. Technol. 2021, 119, 514–529. [Google Scholar] [CrossRef]
- Holman, R.T.; Adams, C.E.; Nelson, R.A.; Grater, S.J.; Jaskiewicz, J.A.; Johnson, S.B.; Erdman, J.W., Jr. Patients with anorexia nervosa demonstrate deficiencies of selected essential fatty acids, compensatory changes in nonessential fatty acids and decreased fluidity of plasma lipids. J. Nutr. 1995, 125, 901–907. [Google Scholar] [PubMed]
- Abdullah, M.; Kornegay, J.N.; Honcoop, A.; Parry, T.L.; Balog-Alvarez, C.J.; O’Neal, S.K.; Bain, J.R.; Muehlbauer, M.J.; Newgard, C.B.; Patterson, C.; et al. Non-targeted metabolomics analysis of golden retriever muscular dystrophy-affected muscles reveals alterations in arginine and proline metabolism, and elevations in glutamic and oleic acid in vivo. Metabolites 2017, 7, 38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, Y.; Wang, G.; Hao, H.; Huang, Q.; Yan, B.; Zha, W.; Gu, S.; Ren, H.; Zhang, Y.; Fan, X.; et al. Gas chromatography/time-of-flight mass spectrometry based metabonomic approach to differentiating hypertension- and age- related metabolic variation in spontaneously hypertensive rats. Rapid Commun. Mass Spectrom. 2008, 22, 2882–2888. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.G.; Lee, J.H.; Lee, J. Antibiofilm activities of fatty acids including myristoleic acid against Cutibacterium acnes via reduced cell hydrophobicity. Phytomedicine 2021, 91, 153710. [Google Scholar] [CrossRef] [PubMed]
- Piccinin, E.; Cariello, M.; De Santis, S.; Ducheix, S.; Sabbà, C.; Ntambi, J.M.; Moschetta, A. Role of oleic acid in the gut-liver axis: From diet to the regulation of its synthesis via stearoyl-CoA desaturase 1 (SCD1). Nutrients 2019, 11, 2283. [Google Scholar] [CrossRef] [Green Version]
- Sokolowska, M.; Stefanska, J. Cytosolic phospholipase A2 group IVA influence on GM-CSF expression in human lung cells: A pilot study. Med. Sci. Monitor 2010, 16, BR300–BR306. [Google Scholar]
- Gao, E.M.; Turathum, B.; Wang, L.; Zhang, D.; Liu, Y.B.; Tang, R.X.; Chian, R.C. The differential metabolomes in cumulus and mural granulosa cells from Human preovulatory follicles. Reprod. Sci. 2022, 29, 1343–1356. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, Y.; Bai, Y.; Xu, S.; Yang, X.; Cheng, B. Intestinal metabolomics of juvenile lenok (Brachymystax lenok) in response to heat stress. Fish Physiol. Biochem. 2022, 48, 1389–1400. [Google Scholar] [CrossRef]
- Shimura, M.; Shindou, H.; Szyrwiel, L.; Tokuoka, S.M.; Hamano, F.; Matsuyama, S.; Okamoto, M.; Matsunaga, A.; Kita, Y.; Ishizaka, Y.; et al. Imaging of intracellular fatty acids by scanning X-ray fluorescence microscopy. FASEB J. 2016, 30, 4149–4158. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Song, Y.; Chai, T.T.; Liao, G.Q.; Zhang, L.; Jia, Q.; Qian, Y.Z.; Qiu, J. Lipidomics perturbations in the brain of adult zebrafish (Danio rerio) after exposure to chiral ibuprofen. Sci. Total Environ. 2020, 713, 136565. [Google Scholar] [CrossRef]
- Melvin, S.D.; Lanctot, C.M.; Doriean, N.J.C.; Bennett, W.; Carroll, A.R. NMR-based lipidomics of fish from a metal (loid) contaminated wetland show differences consistent with effects on cellular membranes and energy storage. Sci. Total Environ. 2019, 654, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Lushchak, V.I. Environmentally induced oxidative stress in aquatic animals. Aquat. Toxicol. 2011, 101, 13–30. [Google Scholar] [CrossRef] [PubMed]
- Birnie-Gauvin, K.; Costantini, D.; Cooke, S.J.; Willmore, W.G. A comparative and evolutionary approach to oxidative stress in fish: A review. Fish Fish. 2017, 18, 928–942. [Google Scholar] [CrossRef]
- Hurem, S.; Fraser, T.W.K.; Gomes, T.; Mayer, I.; Christensen, T. Sub-lethal UV radiation during early life stages alters the behaviour, heart rate and oxidative stress parameters in zebrafish (Danio rerio). Ecotoxicol. Environ. Safe. 2018, 166, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Harding, H.P.; Zhang, Y.; Zeng, H.; Novoa, I.; Lu, P.D.; Calfon, M.; Sadri, N.; Yun, C.; Popko, B.; Paules, R.; et al. An integrated stress response regulates amino acid metabolism and resistance to oxidative stress. Mol. Cell 2003, 11, 619–633. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Gui, X.; Wu, L.; Tian, S.; Wang, H.; Xie, L.; Wu, W. Amino acid metabolism, lipid metabolism, and oxidative stress are associated with post-stroke depression: A metabonomics study. BMC Neurol. 2020, 20, 250. [Google Scholar] [CrossRef]
- Gu, Z.; Li, L.; Tang, S.; Liu, C.; Fu, X.; Shi, Z.; Mao, H. Metabolomics reveals that crossbred dairy buffaloes are more thermotolerant than Holstein cows under chronic heat stress. J. Agr. Food Chem. 2018, 66, 12889–12897. [Google Scholar] [CrossRef]
- Williams-Ashman, H.G.; Seidenfeld, J.; Galletti, P. Trends in the biochemical pharmacology of 5’-deoxy-5’-methylthioadenosine. Biochem. Pharmacol. 1982, 31, 277–288. [Google Scholar] [CrossRef]
- Mato, J.M.; Corrales, F.J.; Lu, S.C.; Avila, M.A. S-Adenosylmethionine: A control switch that regulates liver function. Faseb J. 2002, 16, 15–26. [Google Scholar] [CrossRef] [Green Version]
- Sahlin, K.; Harris, R.C. The creatine kinase reaction: A simple reaction with functional complexity. Amino Acids 2011, 40, 1363–1367. [Google Scholar] [CrossRef] [PubMed]
- Ballantyne, J.S. Amino acid metabolism. Fish Physiol. 2001, 20, 77–107. [Google Scholar]
- Wyss, M.; Kaddurah-Daouk, R. Creatine and creatinine metabolism. Physiol. Rev. 2000, 80, 1107–1213. [Google Scholar] [CrossRef]
- Hu, X.; Shi, Y.; Zhang, P.; Miao, M.; Zhang, T.; Jiang, B. D-Mannose: Properties, production, and applications: An overview. Comprehen. Rev. Food Sci. Food Saf. 2016, 15, 773–785. [Google Scholar] [CrossRef] [Green Version]
- Freeze, H.H. Genetic defects in the human glycome. Nat. Rev. Genet. 2006, 7, 537–551. [Google Scholar] [CrossRef] [PubMed]
- McElroy, G.S.; Reczek, C.R.; Reyfman, P.A.; Mithal, D.S.; Horbinski, C.M.; Chandel, N.S. NAD+ regeneration rescues lifespan, but not ataxia, in a mouse model of brain mitochondrial complex I dysfunction. Cell Metab. 2020, 32, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.F.; Qiang, J.; He, J.; Zhu, H.J.; Bao, J.W.; Xu, P. Untargeted LC-MS metabolomics approach reveals metabolic changes in genetically improved farmed tilapia (Oreochromis niloticus) with fatty liver induced by a high-fat diet. Aquac. Res. 2021, 52, 724–735. [Google Scholar] [CrossRef]
- Wu, W.; Ashley, D.L.; Watson, C.H. Determination of nicotine and other minor alkaloids in international cigarettes by solid-phase microextraction and gas chromatography/mass spectrometry. Anal. Chem. 2002, 74, 4878–4884. [Google Scholar] [CrossRef]
- Siegmund, B.; Leitner, E.; Pfannhauser, W. Determination of the nicotine content of various edible nightshades (Solanaceae) and their products and estimation of the associated dietary nicotine intake. J. Agr. Food Chem. 1999, 47, 3113–3120. [Google Scholar] [CrossRef]
- Berlin, I.; Singleton, E.G.; Pedarriosse, A.M.; Lancrenon, S.; Rames, A.; Aubin, H.; Niaura, R. The modified reasons for smoking scale: Factorial structure, gender effects and relationship with nicotine dependence and smoking cessation in French smokers. Addiction 2003, 98, 1575–1583. [Google Scholar] [CrossRef]
- Ebele, A.J.; Oluseyi, T.; Drage, D.S.; Harrad, S.; Abdallah, M.A. Occurrence, seasonal variation and human exposure to pharmaceuticals and personal care products in surface water, groundwater and drinking water in Lagos State, Nigeria. Emerg. Contam. 2020, 6, 124–132. [Google Scholar] [CrossRef]
- Pal, R.; Megharaj, M.; Kirkbride, K.P.; Naidu, R. Illicit drugs and the environment-a review. Sci. Total Environ. 2013, 463, 1079–1092. [Google Scholar] [CrossRef] [PubMed]
- Verovšek, T.; Heath, D.; Heath, E. Occurrence, fate and determination of tobacco (nicotine) and alcohol (ethanol) residues in waste-and environmental waters. Trends Environ. Anal. 2022, 34, e00164. [Google Scholar] [CrossRef]
- Lawler, S.P. Environmental safety review of methoprene and bacterially-derived pesticides commonly used for sustained mosquito control. Ecotoxicol. Environ. Safe. 2017, 139, 335–343. [Google Scholar] [CrossRef]
- Henrick, C.A. Methoprene. J. Am. Mosq. Control Assoc. 2007, 23, 225–239. [Google Scholar] [CrossRef]
- Ross, D.H.; Judy, D.; Jacobson, B.; Howell, R. Methoprene concentrations in freshwater microcosms treated with sustained-release Altosid (r) formulations. J. Am. Mosq. Control Assoc. 1994, 10, 202–210. [Google Scholar]
- Henrick, C.A.; Ko, J.; Nguyen, J.; Burleson, J.; Lindahl, G.; Van Gundy, D.; Edge, J.M. Investigation of the relationship between s-methoprene and deformities in anurans. J. Am. Mosq. Control Assoc. 2002, 18, 214–221. [Google Scholar]
- Hurst, T.P.; Kay, B.H.; Ryan, P.A.; Brown, M.D. Sublethal effects of mosquito larvicides on swimming performance of larvivorous fish Melanotaenia duboulayi (Atheriniformes: Melanotaeniidae). J. Econ. Entomol. 2007, 100, 61–65. [Google Scholar] [CrossRef]
- Ren, J.; Wang, X.; Wang, C.; Gong, P.; Yao, T. Atmospheric processes of organic pollutants over a remote lake on the central Tibetan Plateau: Implications for regional cycling. Atmos. Chem. Phys. 2017, 17, 1401–1415. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Wang, C.; Zhu, T.; Gong, P.; Fu, J.; Cong, Z. Persistent organic pollutants in the polar regions and the Tibetan Plateau: A review of current knowledge and future prospects. Environ. Pollut. 2019, 248, 191–208. [Google Scholar] [CrossRef]
- Villa, S.; Migliorati, S.; Monti, G.S.; Holoubek, I.; Vighi, M. Risk of POP mixtures on the Arctic food chain. Environ. Toxicol. Chem. 2017, 36, 1181–1192. [Google Scholar] [CrossRef] [PubMed]
- Spongberg, A.L.; Witter, J.D. Pharmaceutical compounds in the wastewater process stream in Northwest Ohio. Sci. Total Environ. 2008, 397, 148–157. [Google Scholar] [CrossRef]
- Laplante, M.; Sabatini, D.M. mTOR signaling in growth control and disease. Cell 2012, 149, 274–293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Düvel, K.; Yecies, J.L.; Menon, S.; Raman, P.; Lipovsky, A.I.; Souza, A.L.; Triantafellow, E.; Ma, Q.; Gorski, R.; Cleaver, S.; et al. Activation of a metabolic gene regulatory network downstream of mTOR complex 1. Mol. Cell 2010, 39, 171–183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mooren, F.C.; Krüger, K. Exercise, autophagy, and apoptosis. Prog. Mol. Biol. Transl. 2015, 135, 407–422. [Google Scholar]
- Levine, B.; Kroemer, G. Biological functions of autophagy genes: A disease perspective. Cell 2019, 176, 11–42. [Google Scholar] [CrossRef] [Green Version]
- Han, J.; Wang, Y. mTORC1 signaling in hepatic lipid metabolism. Protein Cell 2018, 9, 145–151. [Google Scholar] [CrossRef] [Green Version]
- Pfeffer, C.M.; Singh, A.T.K. Apoptosis: A target for anticancer therapy. Int. J. Mol. Sci. 2018, 19, 448. [Google Scholar] [CrossRef] [Green Version]
- Perotti, M.; Toddei, F.; Mirabelli, F.; Vairetti, M.; Bellomo, G.; McConkey, D.J.; Orrenius, S. Calcium-dependent DNA fragmentation in human synovial cells exposed to cold shock. FEBS Lett. 1990, 259, 331–334. [Google Scholar] [CrossRef]
- Lopez, J.; Tait, S.W.G. Mitochondrial apoptosis: Killing cancer using the enemy within. Brit. J. Cancer 2015, 112, 957–962. [Google Scholar] [CrossRef] [Green Version]
- Guicciardi, M.E.; Leist, M.; Gores, G.J. Lysosomes in cell death. Oncogene 2004, 23, 2881–2890. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, W.; Zhu, F.; Wang, D.; Chen, D.; Duan, X.; Liu, M.; Li, D. Comparative Analysis of Metabolites between Different Altitude Schizothorax nukiangensis (Cyprinidae, Schizothoracine) on the Qinghai-Tibet Plateau in Nujiang River. Water 2023, 15, 284. https://doi.org/10.3390/w15020284
Xu W, Zhu F, Wang D, Chen D, Duan X, Liu M, Li D. Comparative Analysis of Metabolites between Different Altitude Schizothorax nukiangensis (Cyprinidae, Schizothoracine) on the Qinghai-Tibet Plateau in Nujiang River. Water. 2023; 15(2):284. https://doi.org/10.3390/w15020284
Chicago/Turabian StyleXu, Weitong, Fengyue Zhu, Dengqiang Wang, Daqing Chen, Xinbin Duan, Mingdian Liu, and Dapeng Li. 2023. "Comparative Analysis of Metabolites between Different Altitude Schizothorax nukiangensis (Cyprinidae, Schizothoracine) on the Qinghai-Tibet Plateau in Nujiang River" Water 15, no. 2: 284. https://doi.org/10.3390/w15020284
APA StyleXu, W., Zhu, F., Wang, D., Chen, D., Duan, X., Liu, M., & Li, D. (2023). Comparative Analysis of Metabolites between Different Altitude Schizothorax nukiangensis (Cyprinidae, Schizothoracine) on the Qinghai-Tibet Plateau in Nujiang River. Water, 15(2), 284. https://doi.org/10.3390/w15020284






