Assessment of Sewage Molecular Markers: Linear Alkylbenzenes in Sediments of an Industrialized Region in Peninsular Malaysia
Abstract
:1. Introduction
2. Methods and Materials
2.1. Experimental Design
2.2. Chemical Analysis
2.3. Quality Control and Assurance
2.4. Statistical Analysis
2.5. Evaluation of Total Organic Carbon (TOC)
3. Results and Discussion
3.1. Differences in the Distribution of LABs Concentrations
3.2. Assessing the Efficiency of LABs Removal from Wastewaters
3.3. Investigation of TOC
3.4. Source Apportionment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adyasari, D.; Pratama, M.A.; Teguh, N.A.; Sabdaningsih, A.; Kusumaningtyas, M.A.; Dimova, N. Anthropogenic Impact on Indonesian Coastal Water and Ecosystems: Current Status and Future Opportunities. Mar. Pollut. Bull. 2021, 171, 112689. [Google Scholar] [CrossRef]
- Linghu, K.; Wu, Q.; Zhang, J.; Wang, Z.; Zeng, J.; Gao, S. Occurrence, Distribution and Ecological Risk Assessment of Antibiotics in Nanming River: Contribution from Wastewater Treatment Plant and Implications of Urban River Syndrome. Process Saf. Environ. Prot. 2023, 169, 428–436. [Google Scholar] [CrossRef]
- Alkhadher, S.A.A.; Suratman, S.; Al-Hazmi, H.E.; Zakaria, M.P.; Szeląg, B.; Majtacz, J.; Drewnowski, J. Unlocking the Secrets of River Pollution: Analyzing Organic Pollutants in Sediments—Experimental Study. Water 2023, 15, 2216. [Google Scholar] [CrossRef]
- Afify, A.A.; Hassan, G.K.; Al-Hazmi, H.E.; Kamal, R.M.; Mohamed, R.M.; Drewnowski, J.; Majtacz, J.; Mąkinia, J.; El-Gawad, H.A. Electrochemical Production of Sodium Hypochlorite from Salty Wastewater Using a Flow-by Porous Graphite Electrode. Energies 2023, 16, 4754. [Google Scholar] [CrossRef]
- Bogacki, J.P.; Al-Hazmi, H. Automotive Fleet Repair Facility Wastewater Treatment Using Air/ZVI and Air/ZVI/H2O2 Processes. Arch. Environ. Prot. 2017, 43, 24–31. [Google Scholar] [CrossRef]
- Al-Hazmi, H.E.; Shokrani, H.; Shokrani, A.; Jabbour, K.; Abida, O.; Mousavi Khadem, S.S.; Habibzadeh, S.; Sonawane, S.H.; Saeb, M.R.; Bonilla-Petriciolet, A.; et al. Recent Advances in Aqueous Virus Removal Technologies. Chemosphere 2022, 305, 135441. [Google Scholar] [CrossRef] [PubMed]
- Al-Hazmi, H.E.; Kot-Wasik, A.; Shokrani, A.; Majtacz, J.; Vatanpour, V.; Munir, M.T.; Habibzadeh, S.; Hejna, A.; Hasanpour, M.; Mohammadi, A.; et al. Diving Boldly into COVID-19 Contaminated Wastewater: Eyes at Nanotechnology-Assisted Solutions. Clin. Transl. Discov. 2023, 3, e195. [Google Scholar] [CrossRef]
- Al-Hazmi, H.; Namiesnik, J.; Tobiszewski, M. Application of TOPSIS for Selection and Assessment of Analytical Procedures for Ibuprofen Determination in Wastewater. Curr. Anal. Chem. 2016, 12, 261–267. [Google Scholar] [CrossRef]
- Pereira, T.D.S.; Al-Hazmi, H.E.; Majtacz, J.; Pires, E.C. Effect of Operating Conditions on N2O Production in an Anammox Sequencing Batch Reactor Containing Granular Sludge. Water Sci. 2019, 880, 37–47. [Google Scholar] [CrossRef]
- Feng, Y.; Lu, X.; Al-Hazmi, H.; Mąkinia, J. An Overview of the Strategies for the Deammonification Process Start-up and Recovery after Accidental Operational Failures. Rev. Environ. Sci. Biotechnol. 2017, 16, 541–568. [Google Scholar] [CrossRef]
- Al-Hazmi, H.; Lu, X.; Grubba, D.; Majtacz, J.; Kowal, P.; Mąkinia, J. Achieving Efficient and Stable Deammonification at Low Temperatures—Experimental and Modeling Studies. Energies 2021, 14, 3961. [Google Scholar] [CrossRef]
- Lu, X.; Pereira, T.D.S.; Al-Hazmi, H.E.; Majtacz, J.; Zhou, Q.; Xie, L.; Makinia, J. Model-Based Evaluation of N2O Production Pathways in the Anammox-Enriched Granular Sludge Cultivated in a Sequencing Batch Reactor. Environ. Sci. Technol. 2018, 52, 2800–2809. [Google Scholar] [CrossRef] [PubMed]
- Maktabifard, M.; Al-Hazmi, H.E.; Szulc, P.; Mousavizadegan, M.; Xu, X.; Zaborowska, E.; Li, X.; Mąkinia, J. Net-Zero Carbon Condition in Wastewater Treatment Plants: A Systematic Review of Mitigation Strategies and Challenges. Renew. Sustain. Energy Rev. 2023, 185, 113638. [Google Scholar] [CrossRef]
- Majtacz, J.B.; Kowal, P.; Lu, X.; Al-Hazmi, H.; Makinia, J. Adaptation of the activated sludge to the digestate liquors during the nitrification and denitrification processes. J. Ecol. Eng. 2017, 18, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Al-Hazmi, H.E.; Lu, X.; Grubba, D.; Majtacz, J.; Badawi, M.; Mąkinia, J. Sustainable Nitrogen Removal in Anammox-Mediated Systems: Microbial Metabolic Pathways, Operational Conditions and Mathematical Modelling. Sci. Total Environ. 2023, 868, 161633. [Google Scholar] [CrossRef] [PubMed]
- Derwis, D.; Majtacz, J.; Kowal, P.; Al-Hazmi, H.E.; Zhai, J.; Ciesielski, S.; Piechota, G.; Mąkinia, J. Integration of the Sulfate Reduction and Anammox Processes for Enhancing Sustainable Nitrogen Removal in Granular Sludge Reactors. Bioresour. Technol. 2023, 383, 129264. [Google Scholar] [CrossRef]
- Al-Hazmi, H.E.; Maktabifard, M.; Grubba, D.; Majtacz, J.; Hassan, G.K.; Lu, X.; Piechota, G.; Mannina, G.; Bott, C.B.; Mąkinia, J. An Advanced Synergy of Partial Denitrification-Anammox for Optimizing Nitrogen Removal from Wastewater: A Review. Bioresour. Technol. 2023, 381, 129168. [Google Scholar] [CrossRef]
- Grubba, D.; Yin, Z.; Majtacz, J.; Al-Hazmi, H.E.; Mąkinia, J. Incorporation of the Sulfur Cycle in Sustainable Nitrogen Removal Systems—A Review. J. Clean. Prod. 2022, 372, 133495. [Google Scholar] [CrossRef]
- Al-Hazmi, H.; Grubba, D.; Majtacz, J.; Kowal, P.; Makinia, J. Evaluation of Partial Nitritation/Anammox (PN/A) Process Performance and Microorganisms Community Composition under Different C/N Ratio. Water 2019, 11, 2270. [Google Scholar] [CrossRef]
- Al-Hazmi, H.E.; Yin, Z.; Grubba, D.; Majtacz, J.B.; Mąkinia, J. Comparison of the Efficiency of Deammonification under Different DO Concentrations in a Laboratory-Scale Sequencing Batch Reactor. Water 2022, 14, 368. [Google Scholar] [CrossRef]
- Al-Hazmi, H.E.; Mohammadi, A.; Hejna, A.; Majtacz, J.; Esmaeili, A.; Habibzadeh, S.; Saeb, M.R.; Badawi, M.; Lima, E.C.; Mąkinia, J. Wastewater Reuse in Agriculture: Prospects and Challenges. Environ. Res. 2023, 236, 116711. [Google Scholar] [CrossRef]
- Soleimani-Gorgani, A.; Al-Hazmi, H.E.; Esmaeili, A.; Habibzadeh, S. Screen-Printed Sn-Doped TiO2 Nanoparticles for Photocatalytic Dye Removal from Wastewater: A Technological Perspective. Environ. Res. 2023, 237, 117079. [Google Scholar] [CrossRef] [PubMed]
- Masood, N.; Alkhadher, S.A.A.; Magam, S.M.; Halimoon, N.; Alsukaibi, A.; Zakaria, M.P.; Vaezzadeh, V.; Keshavarzifard, M.; Maisara, S.; Khaled Bin Break, M. Monitoring of Linear Alkyl Benzenes (LABs) in Riverine and Estuarine Sediments in Malaysia. Env. Geochem. Health 2022, 44, 3687–3702. [Google Scholar] [CrossRef] [PubMed]
- Alkhadher, S.A.A.; Kadir, A.A.; Zakaria, M.P.; Adel, A.-G.; Magam, S.M.S.; Masood, N. Monitoring of Sewage Pollution in the Surface Sediments of Coastal Ecosystems Using Linear Alkylbenzenes (LABs) as Molecular Markers. J. Soils Sediments 2020, 20, 3230–3242. [Google Scholar] [CrossRef]
- Cabral, A.C.; Dauner, A.L.L.; Xavier, F.C.B.; Garcia, M.R.D.; Wilhelm, M.M.; dos Santos, V.C.G.; Netto, S.A.; Martins, C.C. Tracking the Sources of Allochthonous Organic Matter along a Subtropical Fluvial-Estuarine Gradient Using Molecular Proxies in View of Land Uses. Chemosphere 2020, 251, 126435. [Google Scholar] [CrossRef] [PubMed]
- Alkhadher, S.A.A.; Zakaria, M.P.; Yusoff, F.M.; Kannan, N.; Suratman, S.; Keshavarzifard, M.; Magam, S.M.; Masood, N.; Vaezzadeh, V.; Sani, M.S.A. Baseline Distribution and Sources of Linear Alkyl Benzenes (LABs) in Surface Sediments from Brunei Bay, Brunei. Mar. Pollut. Bull. 2015, 101, 397–403. [Google Scholar] [CrossRef]
- Al-Hazmi, H.E.; Grubba, D.; Majtacz, J.; Ziembińska-Buczyńska, A.; Zhai, J.; Mąkinia, J. Combined Partial Denitrification/Anammox Process for Nitrogen Removal in Wastewater Treatment. J. Environ. Chem. Eng. 2023, 11, 108978. [Google Scholar] [CrossRef]
- Alkhadher, S.A.A.; Pauzi Zakaria, M.; Suratman, S.; Alanazi, T.Y.A.; Al-Bagawi, A.H.; Magam, S.M.; Masood, N.; Abdul Kadir, A.; Al-Gheethi, A. Assessment of Sewage Molecular Markers in Port Dickson Coast and Kim Kim River with Sediment Linear Alkylbenzenes. Polycycl. Aromat. Compd. 2023, 43, 343–355. [Google Scholar] [CrossRef]
- Sakai, N.; Shirasaka, J.; Matsui, Y.; Ramli, M.R.; Yoshida, K.; Ali Mohd, M.; Yoneda, M. Occurrence, Fate and Environmental Risk of Linear Alkylbenzene Sulfonate in the Langat and Selangor River Basins, Malaysia. Chemosphere 2017, 172, 234–241. [Google Scholar] [CrossRef]
- Lin, L.; Yang, H.; Xu, X. Effects of Water Pollution on Human Health and Disease Heterogeneity: A Review. Front. Environ. Sci. 2022, 10, 880246. [Google Scholar] [CrossRef]
- Sallan, M.I.B.M.; Al-Hazmi, H.E.; Suratman, S.; Alkhadher, S.A.A.; Szeląg, B.; Majtacz, J.; Kowal, P.; Kurniawan, T.A.; Piechota, G. Anthropogenic Trace Metals in Setiu Wetland: Spatial and Seasonal Distribution and Implications for Environmental Health. J. Water Process Eng. 2023, 55, 104172. [Google Scholar] [CrossRef]
- Al-Gheethi, A.; Rahman, J.A.; Mohamed, R.M.S.R.; Talip, B.; Abdullah, M.E. Bacteria Load Assessment at Sungai Benut in Simpang Renggam, Johor. IOP Conf. Ser. Earth Environ. Sci. 2020, 498, 012061. [Google Scholar] [CrossRef]
- Zakaria, M.P.; Takada, H.; Tsutsumi, S.; Ohno, K.; Yamada, J.; Kouno, E.; Kumata, H. Distribution of Polycyclic Aromatic Hydrocarbons (PAHs) in Rivers and Estuaries in Malaysia: A Widespread Input of Petrogenic PAHs. Environ. Sci. Technol. 2002, 36, 1907–1918. [Google Scholar] [CrossRef] [PubMed]
- Masood, N.; Zakaria, M.P.; Halimoon, N.; Aris, A.Z.; Magam, S.M.; Kannan, N.; Mustafa, S.; Ali, M.M.; Keshavarzifard, M.; Vaezzadeh, V.; et al. Anthropogenic Waste Indicators (AWIs), Particularly PAHs and LABs, in Malaysian Sediments: Application of Aquatic Environment for Identifying Anthropogenic Pollution. Mar. Pollut. Bull. 2016, 102, 160–175. [Google Scholar] [CrossRef]
- Eganhouse, R.P. Molecular Markers and Their Use in Environmental Organic Geochemistry. In The Geochemical Society Special Publications; Hill, R.J., Leventhal, J., Aizenshtat, Z., Baedecker, M.J., Claypool, G., Eganhouse, R., Goldhaber, M., Peters, K., Eds.; Geochemical Investigations in Earth and Space Science: A Tribute to Isaac R. Kaplan; Elsevier: Amsterdam, The Netherlands, 2004; Volume 9, pp. 143–158. [Google Scholar]
- Tsutsumi, S.; Yamaguchi, Y.; Nishida, I.; Akiyama, K.-I.; Zakaria, M.P.; Takada, H. Alkylbenzenes in Mussels from South and South East Asian Coasts as a Molecular Tool to Assess Sewage Impact. Mar. Pollut. Bull. 2002, 45, 325–331. [Google Scholar] [CrossRef]
- Nelson, D.; Sommers, L. Total carbon, organic carbon and organic matter. In Methods of Soil Analysis. Part 3: Chemical Methods; Sparks, D.L., Ed.; Soil Science Society of America: Madison, WI, USA, 1996; pp. 961–1010. [Google Scholar]
- Zeng, E.Y.; Vista, C.L. Organic Pollutants in the Coastal Environment off San Diego, California. 1. Source Identification and Assessment by Compositional Indices of Polycyclic Aromatic Hydrocarbons. Environ. Toxicol. Chem. 1997, 16, 179–188. [Google Scholar] [CrossRef]
- Cabral, A.C.; Martins, C.C. Insights about Sources, Distribution, and Degradation of Sewage and Biogenic Molecular Markers in Surficial Sediments and Suspended Particulate Matter from a Human-Impacted Subtropical Estuary. Environ. Pollut. 2018, 241, 1071–1081. [Google Scholar] [CrossRef]
- Shahbazi, A.; Zakaria, M.P.; Yap, C.K.; Tan, S.G.; Surif, S.; Mohamed, C.A.R.; Sakari, M.; Bakhtiari, A.R.; Bahry, P.S.; Chandru, K.; et al. Use of Different Tissues of Perna Viridis as Biomonitors of Polycyclic Aromatic Hydrocarbons (PAHs) in the Coastal Waters of Peninsular Malaysia. Environ. Forensics 2010, 11, 248–263. [Google Scholar] [CrossRef]
- Sakai, N.; Mohd Yusof, R.; Sapar, M.; Yoneda, M.; Ali Mohd, M. Spatial Analysis and Source Profiling of Beta-Agonists and Sulfonamides in Langat River Basin, Malaysia. Sci. Total Environ. 2016, 548–549, 43–50. [Google Scholar] [CrossRef]
- Luo, X.-J.; Chen, S.-J.; Ni, H.-G.; Yu, M.; Mai, B.-X. Tracing Sewage Pollution in the Pearl River Delta and Its Adjacent Coastal Area of South China Sea Using Linear Alkylbenzenes (LABs). Mar. Pollut. Bull. 2008, 56, 158–162. [Google Scholar] [CrossRef]
- Zacchi, F.L.; Flores-Nunes, F.; Mattos, J.J.; Lima, D.; Lüchmann, K.H.; Sasaki, S.T.; Bícego, M.C.; Taniguchi, S.; Montone, R.C.; de Almeida, E.A.; et al. Biochemical and Molecular Responses in Oysters Crassostrea Brasiliana Collected from Estuarine Aquaculture Areas in Southern Brazil. Mar. Pollut. Bull. 2018, 135, 110–118. [Google Scholar] [CrossRef]
- Isobe, K.O.; Zakaria, M.P.; Chiem, N.H.; Minh, L.Y.; Prudente, M.; Boonyatumanond, R.; Saha, M.; Sarkar, S.; Takada, H. Distribution of Linear Alkylbenzenes (LABs) in Riverine and Coastal Environments in South and Southeast Asia. Water Res. 2004, 38, 2448–2458. [Google Scholar] [CrossRef] [PubMed]
- Bakhtiari, A.R.; Javedankherad, I.; Mohammadi, J.; Taghizadeh, R. Distribution of Linear Alkylbenzenes as a Domestic Sewage Molecular Marker in Surface Sediments of International Anzali Wetland in the Southwest of the Caspian Sea, Iran. Environ.Sci. Pollut. Res. 2018, 25, 20920–20929. [Google Scholar] [CrossRef] [PubMed]
- Alkhadher, S.A.A.; Kadir, A.A.; Zakaria, M.P.; Al-Gheethi, A.; Asghar, B.H.M. Determination of Linear Alkylbenzenes (LABs) in Mangrove Ecosystems Using the Oyster Crassostrea Belcheri as a Biosensor. Mar. Pollut. Bull. 2020, 154, 111115. [Google Scholar] [CrossRef] [PubMed]
- Alkhadher, S.A.A.; Suratman, S.; Zakaria, M.P. Lateral Distribution, Environmental Occurrence and Assessment of Organic Pollutants in Surface Sediments of the West and South Peninsular Malaysia. Water Air Soil Pollut. 2023, 234, 124. [Google Scholar] [CrossRef]
- Dauner, A.L.L.; Hernández, E.A.; MacCormack, W.P.; Martins, C.C. Molecular Characterisation of Anthropogenic Sources of Sedimentary Organic Matter from Potter Cove, King George Island, Antarctica. Sci. Total Environ. 2015, 502, 408–416. [Google Scholar] [CrossRef]
- Alkhadher, S.A.A.; Kadir, A.A.; Zakaria, M.P.; Al-Gheethi, A.; Keshavarzifard, M.; Masood, N.; Alenezi, K.M.; Magam, S.M. Linear Alkylbenzenes in Surface Sediments of an Estuarine and Marine Environment in Peninsular Malaysia. Mar. Pollut. Bull. 2020, 153, 111013. [Google Scholar] [CrossRef]
- Sherblom, P.M.; Gschwend, P.M.; Eganhouse, R.P. Aqueous Solubilities, Vapor Pressures, and 1-Octanol-Water Partition Coefficients for C9-C14 Linear Alkylbenzenes. J. Chem. Eng. Data 1992, 37, 394–399. [Google Scholar] [CrossRef]
- Abdo Alkhadher, S.A.; Zakaria, M.P.; Yusoff, F.M.; Kannan, N.; Suratman, S.; Magam, S.M.; Masood, N.; Keshavarzifard, M.; Vaezzadeh, V.; Sani, M.S.A. Distribution and Sources of Linear Alkyl Benzenes (LABs) in Surface Sediments from Johor Bahru Coast and the Kim Kim River, Malaysia. Environ. Forensics 2016, 17, 36–47. [Google Scholar] [CrossRef]
- Magam, S.M.; Zakaria, M.P.; Halimoon, N.; Aris, A.Z.; Kannan, N.; Masood, N.; Mustafa, S.; Alkhadher, S.; Keshavarzifard, M.; Vaezzadeh, V.; et al. Evaluation of Distribution and Sources of Sewage Molecular Marker (LABs) in Selected Rivers and Estuaries of Peninsular Malaysia. Environ. Sci. Pollut. Res. 2016, 23, 5693–5704. [Google Scholar] [CrossRef]
- Alkhadher, S.A.A.; Suratman, S.; Zakaria, M.P. Occurrence and Assessment of Organic Pollutants Residues in the Aquatic Environment of the Coastal Sediments. Sustainability 2023, 15, 8365. [Google Scholar] [CrossRef]
- Alkhadher, S.A.A.; Suratman, S.; Zakaria, M.P. Lateral Distribution, Environmental Occurrence, and Assessment of Organic Pollutants in Surface Sediments of the East Malaysia. Environ. Monit. Assess. 2023, 195, 720. [Google Scholar] [CrossRef]
- Gustafsson, Ö.; Long, C.M.; MacFarlane, J.; Gschwend, P.M. Fate of Linear Alkylbenzenes Released to the Coastal Environment near Boston Harbor. Environ. Sci. Technol. 2001, 35, 2040–2048. [Google Scholar] [CrossRef] [PubMed]
- Ni, H.-G.; Lu, F.-H.; Wang, J.-Z.; Guan, Y.-F.; Luo, X.-L.; Zeng, E.Y. Linear Alkylbenzenes in Riverine Runoff of the Pearl River Delta (China) and Their Application as Anthropogenic Molecular Markers in Coastal Environments. Environ. Pollut. 2008, 154, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Hamzah, A.; Kipli, S.H.; Ismail, S.R.; Una, R.; Sarmani, S. Microbiological Study in Coastal Water of Port Dickson, Malaysia. Sains Malays. 2011, 40, 93–99. [Google Scholar]
- Thomes, M.W.; Vaezzadeh, V.; Zakaria, M.P.; Bong, C.W. Use of Sterols and Linear Alkylbenzenes as Molecular Markers of Sewage Pollution in Southeast Asia. Environ. Sci. Pollut. Res. 2019, 26, 31555–31580. [Google Scholar] [CrossRef]
- Cabral, A.C.; Stark, J.S.; Kolm, H.E.; Martins, C.C. An Integrated Evaluation of Some Faecal Indicator Bacteria (FIB) and Chemical Markers as Potential Tools for Monitoring Sewage Contamination in Subtropical Estuaries. Environ. Pollut. 2018, 235, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Wang, J.-Z.; Liang, B.; Shen, R.-L.; Zeng, E.Y. Assessment of Aquatic Wastewater Pollution in a Highly Industrialized Zone with Sediment Linear Alkylbenzenes. Environ. Toxicol. Chem. 2012, 31, 724–730. [Google Scholar] [CrossRef]
- Harwood, J.J. Molecular Markers for Identifying Municipal, Domestic and Agricultural Sources of Organic Matter in Natural Waters. Chemosphere 2014, 95, 3–8. [Google Scholar] [CrossRef]
- Wang, X.-C.; Zhang, Y.-X.; Chen, R.F. Distribution and Partitioning of Polycyclic Aromatic Hydrocarbons (PAHs) in Different Size Fractions in Sediments from Boston Harbor, United States. Mar. Pollut. Bull. 2001, 42, 1139–1149. [Google Scholar] [CrossRef]
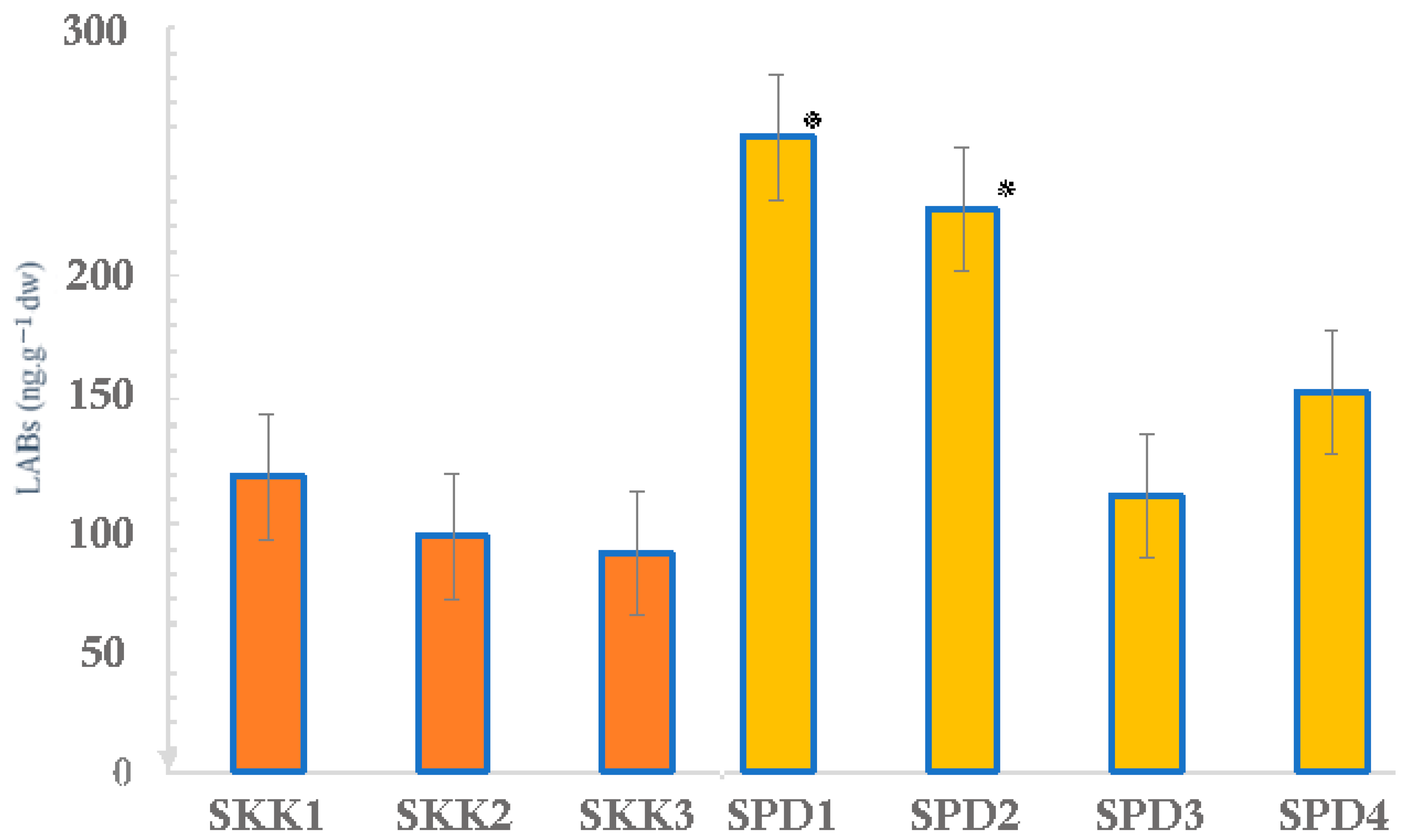
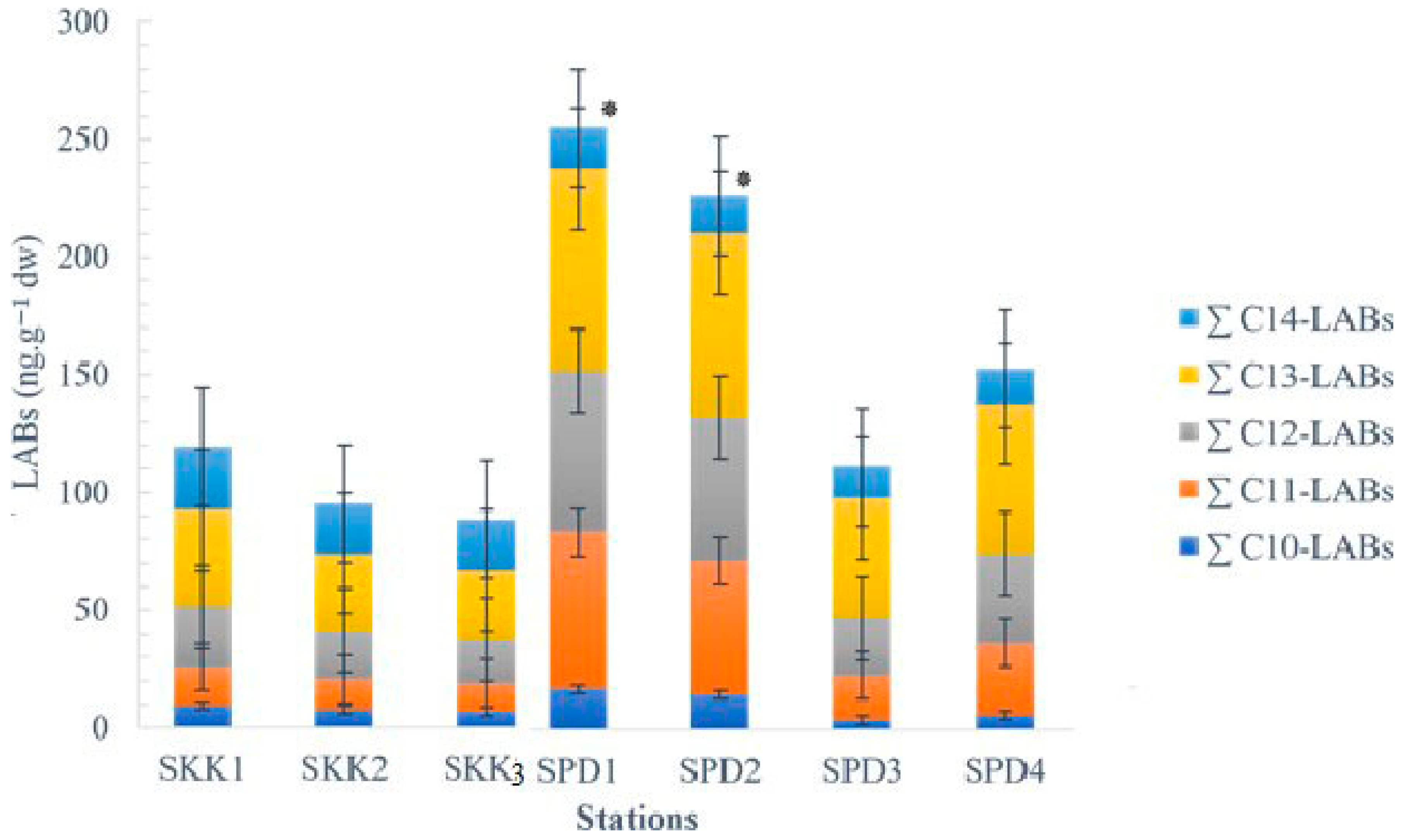




| Compound | SKK1 b | SKK2 | SKK3 | SPD1 | SPD2 | SPD3 | SPD4 |
|---|---|---|---|---|---|---|---|
| C10-LABs (ng·g−1 dw) a | 9.4 | 7.7 | 6.8 | 17.2 | 15.0 | 4.2 | 6.2 |
| C11-LABs (ng·g−1 dw) | 16.3 | 13.0 | 12.0 | 66.7 | 57.0 | 19.0 | 30.7 |
| C12-LABs (ng·g−1 dw) | 25.6 | 20.7 | 18.6 | 68.6 | 61.0 | 24.0 | 38.0 |
| C13-LABs (ng·g−1 dw) | 41.0 | 32.6 | 29.5 | 86.0 | 78.4 | 51.2 | 63.6 |
| C14-LABs (ng·g−1 dw) | 26.8 | 21.1 | 21.3 | 17.4 | 15.6 | 13.2 | 14.8 |
| LABs (ng·g−1 dw) | 119 | 95.2 | 88.3 | 256 | 227 | 112 | 153 |
| LC-LABs (ng·g−1 dw) c | 67.8 | 53.8 | 51.0 | 103 | 94.0 | 64.5 | 78.4 |
| SC-LABs (ng·g−1 dw) d | 25.8 | 20.7 | 19.0 | 84.0 | 71.8 | 23.2 | 37.0 |
| I/E e | 2.0 | 1.8 | 1.7 | 4.1 | 3.7 | 2.0 | 2.6 |
| L/S g | 2.4 | 2.5 | 2.6 | 1.6 | 1.6 | 1.7 | 1.6 |
| C13/C12 h | 5.1 | 5.2 | 5.2 | 9.7 | 9.5 | 13.7 | 14.3 |
| LAB degradation (%) f | 38 | 35 | 34 | 64 | 61 | 40 | 48 |
| TOC (%) i | 1.2 | 1.0 | 8.1 | 2.0 | 2.3 | 1.5 | 1.9 |
| TOC (mg/g) | 11.5 | 9.0 | 81 | 19.7 | 22.7 | 14.7 | 18.9 |
| (a) ANOVA | ||||||
|---|---|---|---|---|---|---|
| Sum of Squares | df | Mean Square | F | Sig. * | ||
| Between groups | 8410 | 4 | 210 | 7.3 | 0.000 | |
| Within groups | 8680 | 30 | 289 | |||
| Total | 17,100 | 34 | ||||
| (b) Post Hoc Tests | ||||||
| Dependent Variable: LAB Concentration | ||||||
| LSD | ||||||
| (I) Location | (J) Location | Mean Difference (I-J) | Std. Error | Sig. | 95% Confidence Interval | |
| Lower Bound | Upper Bound | |||||
| 1.00 | 2.00 | −21.2 * | 9.1 | 0.03 | −39.7 | −2.6 |
| 3.00 | −27.2 * | 9.1 | 0.01 | −45.7 | −8.6 | |
| 4.00 | −45.1 * | 9.1 | 0.00 | −63.7 | −26.6 | |
| 5.00 | −9.1 | 9.1 | 0.32 | −27.7 | 9.5 | |
| 2.00 | 1.00 | 21.2 * | 9.1 | 0.03 | 2.6 | 39.7 |
| 3.00 | −6.0 | 9.1 | 0.52 | −24.6 | 12.6 | |
| 4.00 | −24.0 * | 9.1 | 0.01 | −42.5 | −5.3 | |
| 5.00 | 12.1 | 9.1 | 0.20 | −6.5 | 30.6 | |
| 3.00 | 1.00 | 27.2 * | 9.1 | 0.01 | 8.6 | 45.7 |
| 2.00 | 6.0 | 9.1 | 0.52 | −12.6 | 24.6 | |
| 4.00 | −18.0 | 9.1 | 0.06 | −36.5 | 0.6 | |
| 5.00 | 18.0 | 9.1 | 0.06 | −0.53 | 36.6 | |
| 4.00 | 1.00 | 45.1 * | 9.1 | 0.00 | 26.6 | 63.7 |
| 2.00 | 24.0 * | 9.1 | 0.01 | 5.4 | 42.5 | |
| 3.00 | 18.0 | 9.1 | 0.06 | −0.6 | 36.5 | |
| 5.00 | 36.0 * | 9.1 | 0.00 | 17.4 | 54.6 | |
| 5.00 | 1.00 | 9.1 | 9.1 | 0.32 | −9.5 | 27.7 |
| 2.00 | −12.1 | 9.1 | 0.20 | −30.6 | 6.5 | |
| 3.00 | −18.0 | 9.1 | 0.06 | −36.6 | 0.53 | |
| 4.00 | −36.0 * | 9.1 | 0.00 | −54.6 | −17.4 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alkhadher, S.A.A.; Al-Hazmi, H.E.; Suratman, S.; Zakaria, M.P.; Masood, N.; Szeląg, B.; Magam, S.M.; Al-Qadami, E.H.H.; Majtacz, J.; Kowal, P.; et al. Assessment of Sewage Molecular Markers: Linear Alkylbenzenes in Sediments of an Industrialized Region in Peninsular Malaysia. Water 2023, 15, 3301. https://doi.org/10.3390/w15183301
Alkhadher SAA, Al-Hazmi HE, Suratman S, Zakaria MP, Masood N, Szeląg B, Magam SM, Al-Qadami EHH, Majtacz J, Kowal P, et al. Assessment of Sewage Molecular Markers: Linear Alkylbenzenes in Sediments of an Industrialized Region in Peninsular Malaysia. Water. 2023; 15(18):3301. https://doi.org/10.3390/w15183301
Chicago/Turabian StyleAlkhadher, Sadeq A. A., Hussein E. Al-Hazmi, Suhaimi Suratman, Mohamad P. Zakaria, Najat Masood, Bartosz Szeląg, Sami M. Magam, Ebrahim H. H. Al-Qadami, Joanna Majtacz, Przemysław Kowal, and et al. 2023. "Assessment of Sewage Molecular Markers: Linear Alkylbenzenes in Sediments of an Industrialized Region in Peninsular Malaysia" Water 15, no. 18: 3301. https://doi.org/10.3390/w15183301






