Invasive and Rare Aquatic Invertebrates of Taiwan with a Focus on Their Dormancy
Abstract
:1. Introduction
2. Materials and Methods
3. Results
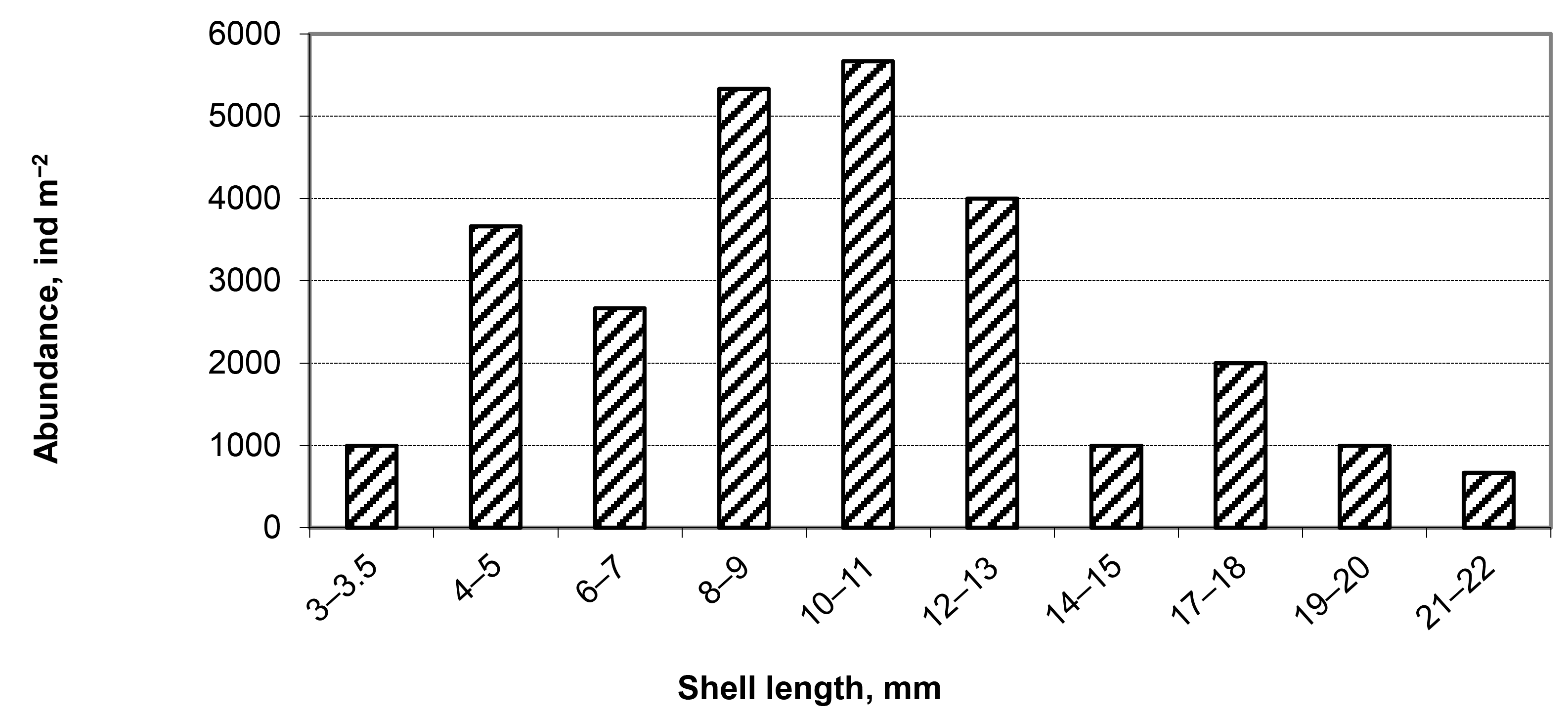
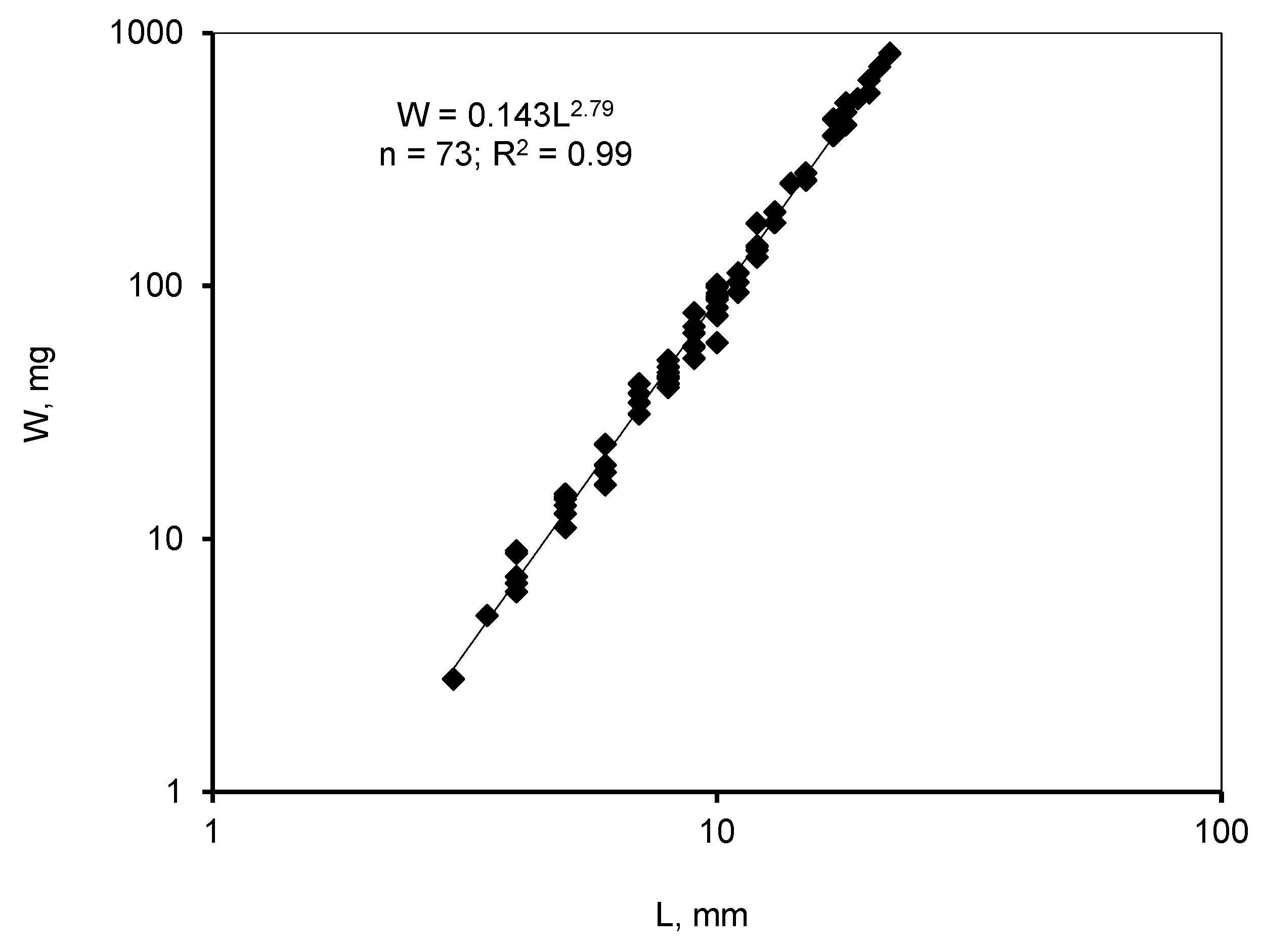
3.1. Invasive and New Species of Aquatic Invertebrates in Water Bodies of Taiwan
3.2. Accumulation of Resting Stages in the Sediments of Inland Water Bodies and Coastal Areas of Taiwan
3.2.1. Marine Ecosystems
- Shallow biotopes (sediments from mangroves and shallow coral reefs)
- B.
- Deeper marine waters along the west coast
3.2.2. Freshwater Biotopes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brown, J.H. Why are there so many species in the tropics? J. Biogeogr. 2014, 41, 8–22. [Google Scholar] [CrossRef]
- O’Shea, S.; Cangelosi, A. Trojan Horses in Our Harbors: Biological Contamination from Ballast Water Discharge. Univ. Toledo Law Rev. 1995, 27, 381. [Google Scholar]
- Ricciardi, A.; MacIsaac, H.J. Recent mass invasion of the North American Great Lakes by Ponto–Caspian species. Trends Ecol. Evol. 2000, 15, 62–65. [Google Scholar] [CrossRef]
- Leppäkoski, E.; Gollasch, S. Risk Assessment of Ballast Water Mediated Species Introductions—A Baltic Sea Approach. Report Prepared for HELCOM, Helsinki, Finland. 2006, p. 111. Available online: https://www.reabic.net/publ/leppakoski_gollasch_helcom.pdf (accessed on 10 July 2023).
- Wang, Z.; Saebi, M.; Grey, E.K.; Corbett, J.J.; Chen, D.; Yang, D.; Wan, Z. Ballast water-mediated species spread risk dynamics and policy implications to reduce the invasion risk to the Mediterranean Sea. Mar. Pollut. Bull. 2022, 174, 113285. [Google Scholar] [CrossRef]
- Liang, S.; Chuang, L.; Chang, M. The pet trade as a source of invasive fish in Taiwan. Taiwania 2006, 51, 93–98. [Google Scholar]
- Liang, S.H.; Walther, B.A.; Shieh, B.S. Determinants of establishment success: Comparing alien and native freshwater fishes in Taiwan. PLoS ONE 2020, 15, e0236427. [Google Scholar] [CrossRef]
- Olenin, S. Invasive Aquatic Species in the Baltic States; Klaipëda University Press: Klaipeda, Lithuania, 2005. [Google Scholar]
- Hairston, N.G.; Fox, J.A.; Yamamichi, M. Dormancy and Diapause; Elsevier: Amsterdam, The Netherlands, 2022. [Google Scholar] [CrossRef]
- Zadereev, E.; Lopatina, T.S.; Oskina, N. Resistance of Dormant Eggs of Cladocera to Anthropogenic Pollutants. In Dormancy in Aquatic Organisms. Theory, Human Use and Modeling; Alekseev, V.R., Pinel-Alloul, B., Eds.; Springer: Cham, Switzerland, 2019; pp. 121–135. [Google Scholar]
- Alekseev, V.; Sukhikh, N. Ust-Luga Seaport of Russia: Biological Invasions and Resting Stages Accumulation. Life 2022, 13, 117. [Google Scholar] [CrossRef] [PubMed]
- Danilevsky, A.S. Photoperiodism and Seasonal Development of Insects; Len. State Univ.: St-Petersburg, Russia, 1961. (In Russian) [Google Scholar]
- Andrusov, N. Iskopayemyya i zhivushchiya Dreissensidae Yevrazii. Dopolneniye pervoye [Fossile und lebende Dreissenidae Eurasiens. Erstes Supplement]. Trav. Société Nat. St. Pétersbourg 1897, 29, 59–132. [Google Scholar]
- Claus, C. Die Frei Lebenden Copepoden mit Besonderer Berucksichtigung der Fauna Deutschlands, der Nordsee und des Mittelmeeres; Verlag von Wilhelm Engelmann: Leipzig, Germany, 1863. [Google Scholar]
- Sukhikh, N.; Alekseev, V. Genetic and morphological heterogeneity within Eucyclops serrulatus (Fischer, 1851) (Crustacea: Copepoda: Cyclopidae). J. Nat. Hist. 2015, 49, 2929–2953. [Google Scholar] [CrossRef]
- Tsalolikhin, S.Y. To the fauna of free-living freshwater nematodes of South-East Asia. Tr. Zool. Inst. Ross. Akad. Nauk 2009, 313, 427–434. [Google Scholar] [CrossRef]
- Tsalolikhin, S.Y.; Shoshin, Y. Review of the genus Neotobrilus Tsalolikhin, 1981 (Nematoda, Enoplida: Tobrilidae). Russ. J. Nematol. 2009, 17, 59–72. [Google Scholar]
- Tsalolikhin, S.Y. Crassolabium alekseevi spn. (Nematoda, Dorylaimida, Qudsianematidae) from Taiwan Island. Zool. Zhurnal 2017, 96, 740–744. [Google Scholar]
- Vogler, R.E.; Beltramino, A.A. Achatina fulica (giant African land snail). CABI Compend. 2022. [Google Scholar] [CrossRef]
- Thakur, S.; Kumari, R. Seasonal behaviour of giant African snail Achatina fulica in Bihar. J. Ecotoxicol. Environ. Monit. 1998, 8, 153–160. [Google Scholar]
- Meyer, W.M.; Hayes, K.A.; Meyer, A.L. Giant African snail, Achatina fulica, as a snail predator. Am. Malacol. Bull. 2008, 24, 117–119. [Google Scholar] [CrossRef]
- Vijayan, K.; Suganthasakthivel, R.; Sajeev, T.V.; Soorae, P.S.; Naggs, F.; Wade, C.M. Genetic variation in the Giant African Snail Lissachatina fulica (Bowdich, 1822) in its invasive ranges of Asia and West Africa. Biol. J. Linn. Soc. 2020, 131, 973–985. [Google Scholar] [CrossRef]
- Mead, A.R. The Giant African Snail: A Problem in Economic Malacology; University of Chicago Press: Chicago, IL, USA, 1961. [Google Scholar]
- Odermatt, P.; Lv, S.; Sayasone, S. Less common parasitic infections in Southeast Asia that can produce outbreaks. Adv. Parasitol. 2010, 72, 409–435. [Google Scholar]
- Mochida, O. Spread of freshwater Pomacea snails (Pilidae, Mollusca) from Argentina to Asia. Micronesica 1991, 3, 51–62. [Google Scholar]
- Cowie, R. Pomacea canaliculata (invasive apple snail). CABI Compend. 2022. [Google Scholar] [CrossRef]
- Wada, T.; Matsukura, K. Seasonal changes in cold hardiness of the invasive freshwater apple snail, Pomacea canaliculata (Lamarck) (Gastropoda: Ampullariidae). Malacologia 2007, 49, 383–392. [Google Scholar] [CrossRef]
- Heath, R.T.; Fahnenstiel, G.L.; Gardner, W.S.; Cavaletto, J.F.; Hwang, S.J. Ecosystem-level effects of zebra mussels (Dreissena polymorpha): An enclosure experiment in Saginaw Bay, Lake Huron. J. Great Lakes Res. 1995, 21, 501–516. [Google Scholar] [CrossRef]
- Karatayev, A.Y.; Boltovskoy, D.; Padilla, D.K.; Burlakova, L.E. The invasive bivalves Dreissena polymorpha and Limnoperna fortunei: Parallels, contrasts, potential spread and invasion impacts. J. Shellfish Res. 2007, 26, 205–213. [Google Scholar] [CrossRef]
- Spidle, A.P.; May, B.; Mills, E.L. Limits to tolerance of temperature and salinity in the quagga mussel (Dreissena bugensis) and the zebra mussel (Dreissena polymorpha). Can. J. Fish. Aquat. Sci. 1995, 52, 2108–2119. [Google Scholar] [CrossRef]
- Dussart, B.H.; Defaye, D. World Directory of Crustacea Copepoda of Inland Waters. II. Cyclopiformes; Backhuys: Leiden, The Netherlands, 2006. [Google Scholar]
- Alekseev, V.R. Rol’ fotoperrioda i drugikh faktorov vneshne’ i sredy v induktsii diapauzy u tsiklopov vremennykh vodoemov na primere Metacyclops minutus (Claus). Ekologiya 1989, 5, 51–57. (In Russian) [Google Scholar]
- Zhao, Z.; Hou, Z.E.; Li, S.Q. Cenozoic Tethyan changes dominated Eurasian animal evolution and diversity patterns. Zool. Res. 2022, 43, 3. [Google Scholar] [CrossRef] [PubMed]
- Berg, L.S. Climate and Life; GosIzdat: Moscow, Russia, 1922. (In Russian) [Google Scholar]
- Artamonova, V.S.; Bolotov, I.N.; Vinarski, M.V.; Makhrov, A.A. Fresh-and Brackish-Water Cold-Tolerant Species of Southern Europe: Migrants from the Paratethys That Colonized the Arctic. Water 2021, 13, 1161. [Google Scholar] [CrossRef]
- Alekseev, V. The modern distribution of two Eucyclops (Eucyclops) Claus, 1893 species (Cyclopoida: Copepoda) as a reflection of the Tethys Sea evolution. Hydrobiologia 2023, 1–14. [Google Scholar] [CrossRef]
- Uye, S.I.; Fleminger, A. Effects of various environmental factors on egg development of several species of Acartia in southern California. Mar. Biol. 1976, 38, 253–262. [Google Scholar] [CrossRef]
- Sabia, L.; Zagami, G.; Mazzocchi, M.G.; Zambianchi, E.; Uttieri, M. Spreading factors of a globally invading coastal copepod. Mediterr. Mar. Sci. 2015, 16, 460–471. [Google Scholar] [CrossRef]
- Marcus, N.H.; Lutz, R.; Burnett, W.; Cable, P. Age, viability, and vertical distribution of zooplankton resting eggs from an anoxic basin: Evidence of an egg bank. Limnol. Oceanogr. 1994, 39, 154–158. [Google Scholar] [CrossRef]
- Hairston, N.G.; Van Brunt, R.A.; Kearns, C.M.; Engstrom, D.R. Age and survivorship of diapausing eggs in a sediment egg bank. Ecology 1995, 76, 1706–1711. [Google Scholar] [CrossRef]
- Marcus, N.H.; Lutz, R.V.; Chanton, J.P. Impact of anoxia and sulfide on the viability of eggs of three planktonic copepods. Mar. Ecol. Prog. Ser. 1997, 146, 291–295. [Google Scholar] [CrossRef]
- Marcus, N.H. Ecological and evolutionary significance of resting eggs in marine copepods: Past, present, and future studies. Hydrobiologia 1996, 320, 141–152. [Google Scholar] [CrossRef]
- Marcus, N.H.; Fuller, C.M. Distribution and abundance of eggs of Labidocera aestiva (Copepoda: Calanoida) in the bottom sediments of Buzzards Bay, Massachusetts, USA. Mar. Biol. 1989, 100, 319–326. [Google Scholar] [CrossRef]
- Lindley, J.A. Distribution of overwintering calanoid copepod eggs in sea-bed sediments around southern Britain. Mar. Biol. 1990, 104, 209–217. [Google Scholar] [CrossRef]
- Uye, S.; Yoshiya, M.; Ueda, K.; Kasahara, S. The effect of organic sea-bottom pollution on survivability of resting eggs of neritic calanoids. In Proceedings of the First International Conference on Copepoda, Amsterdam, The Netherlands, 24–28 August 1981; pp. 390–403. [Google Scholar]



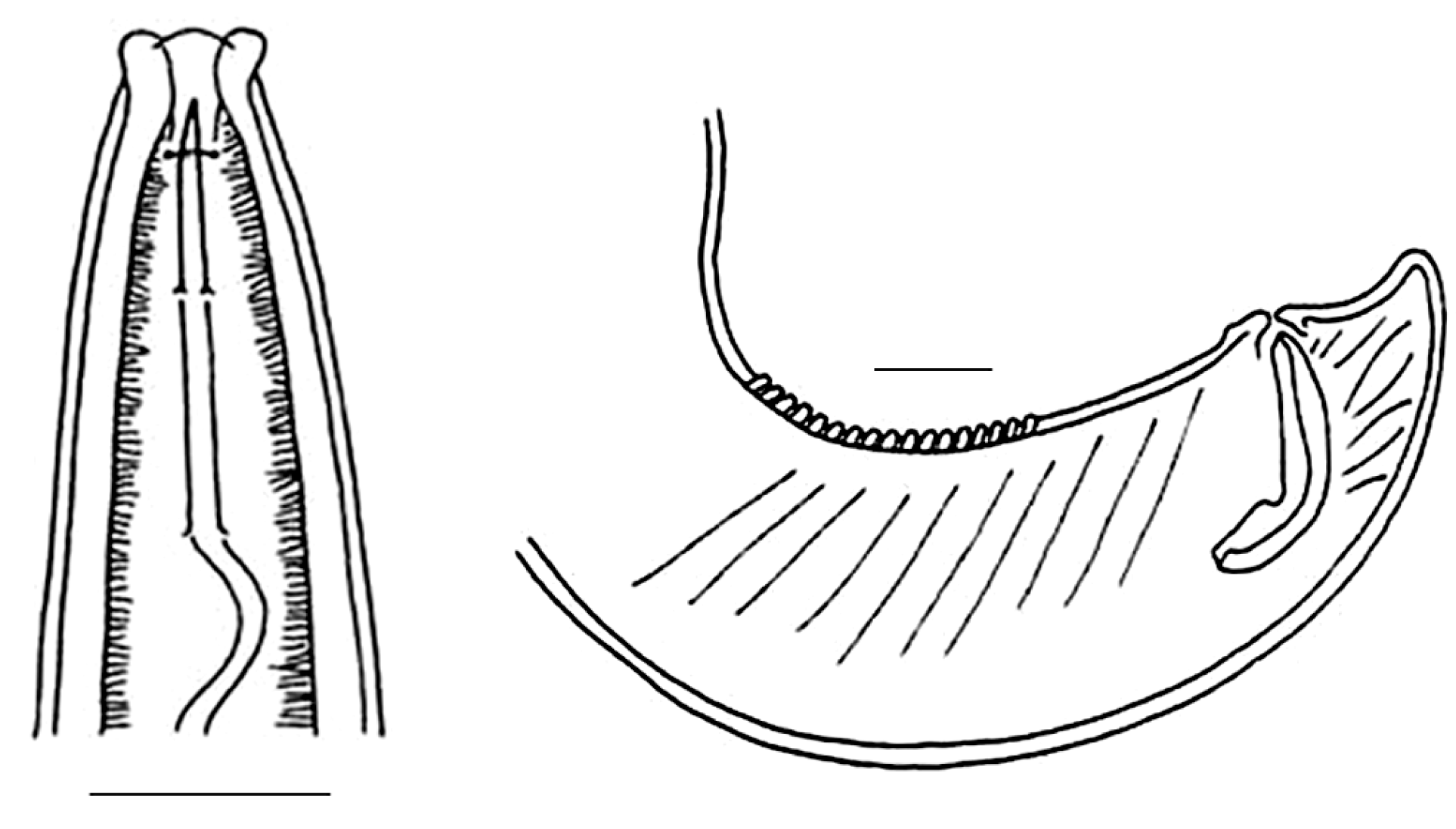
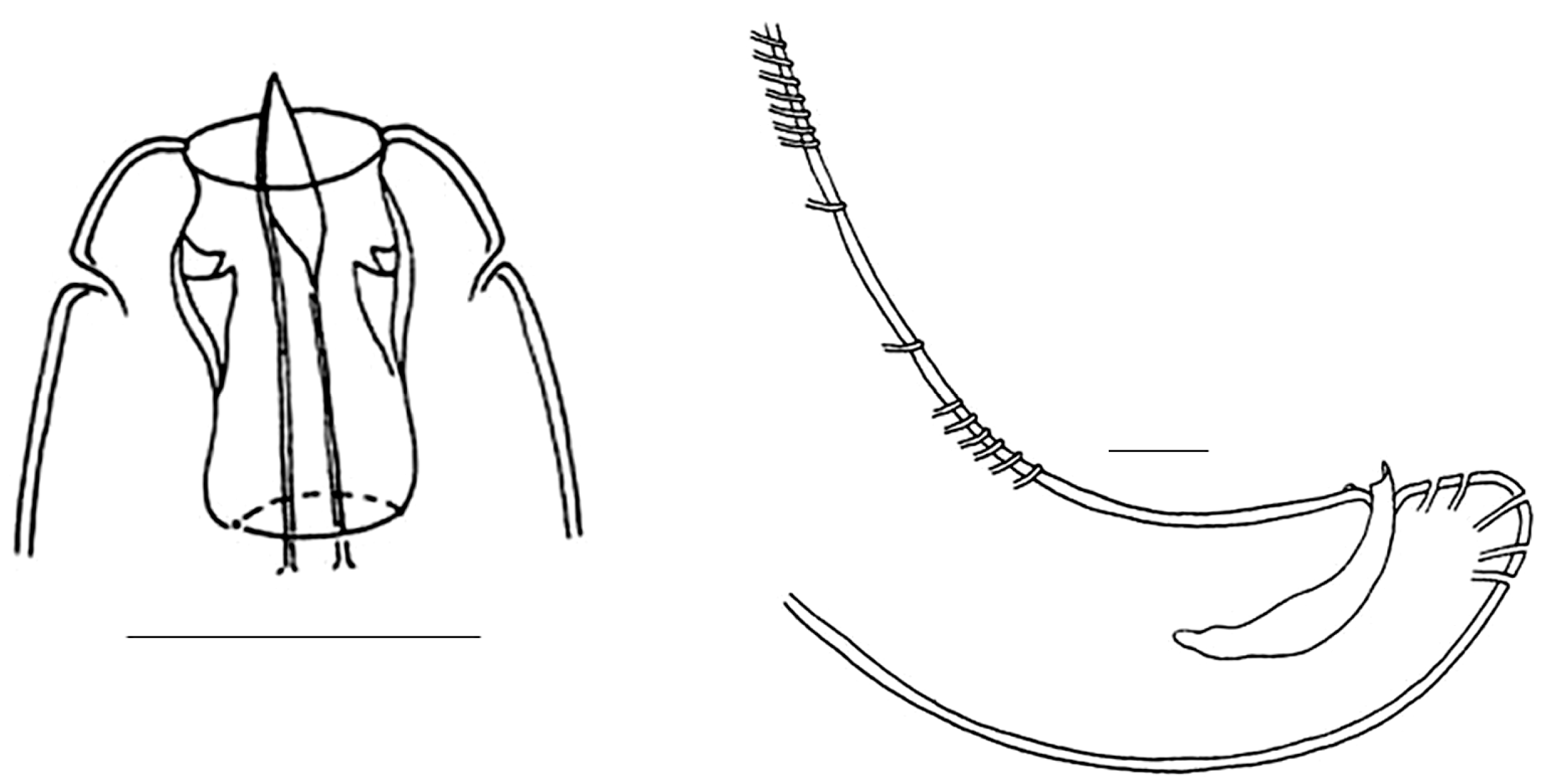
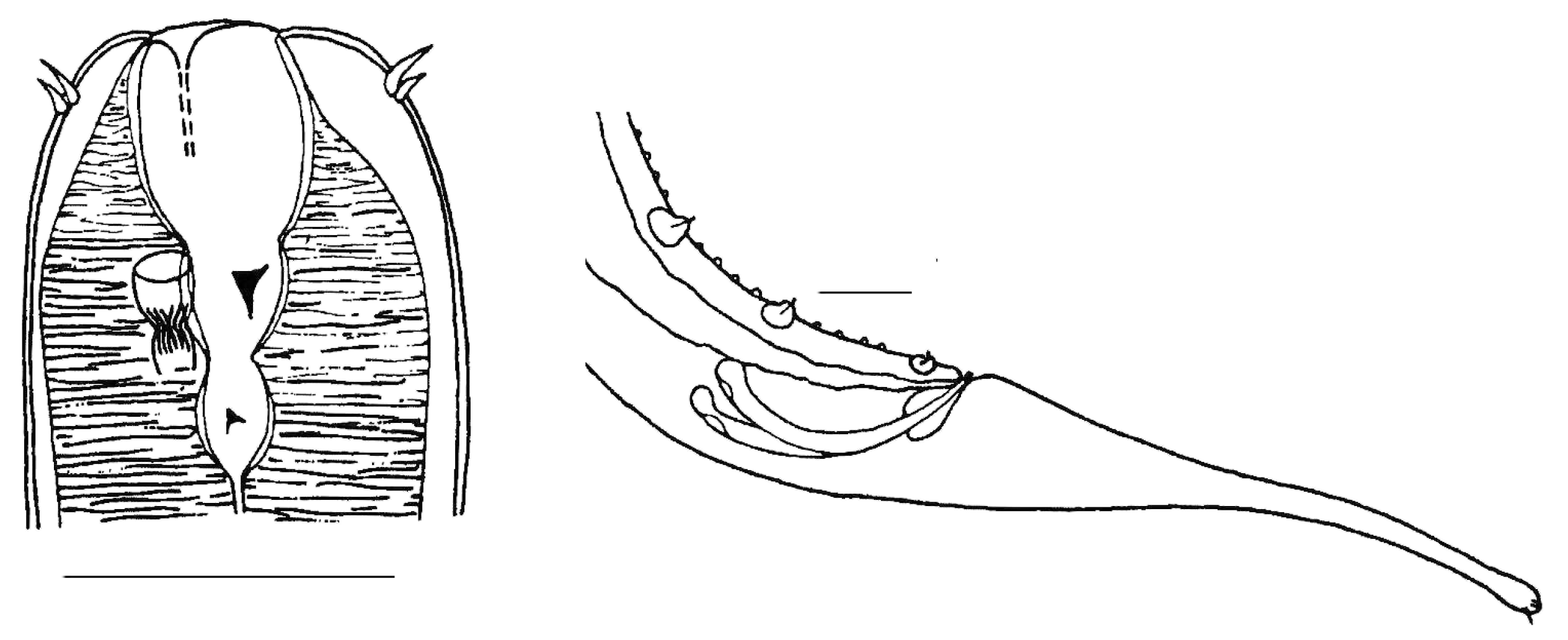
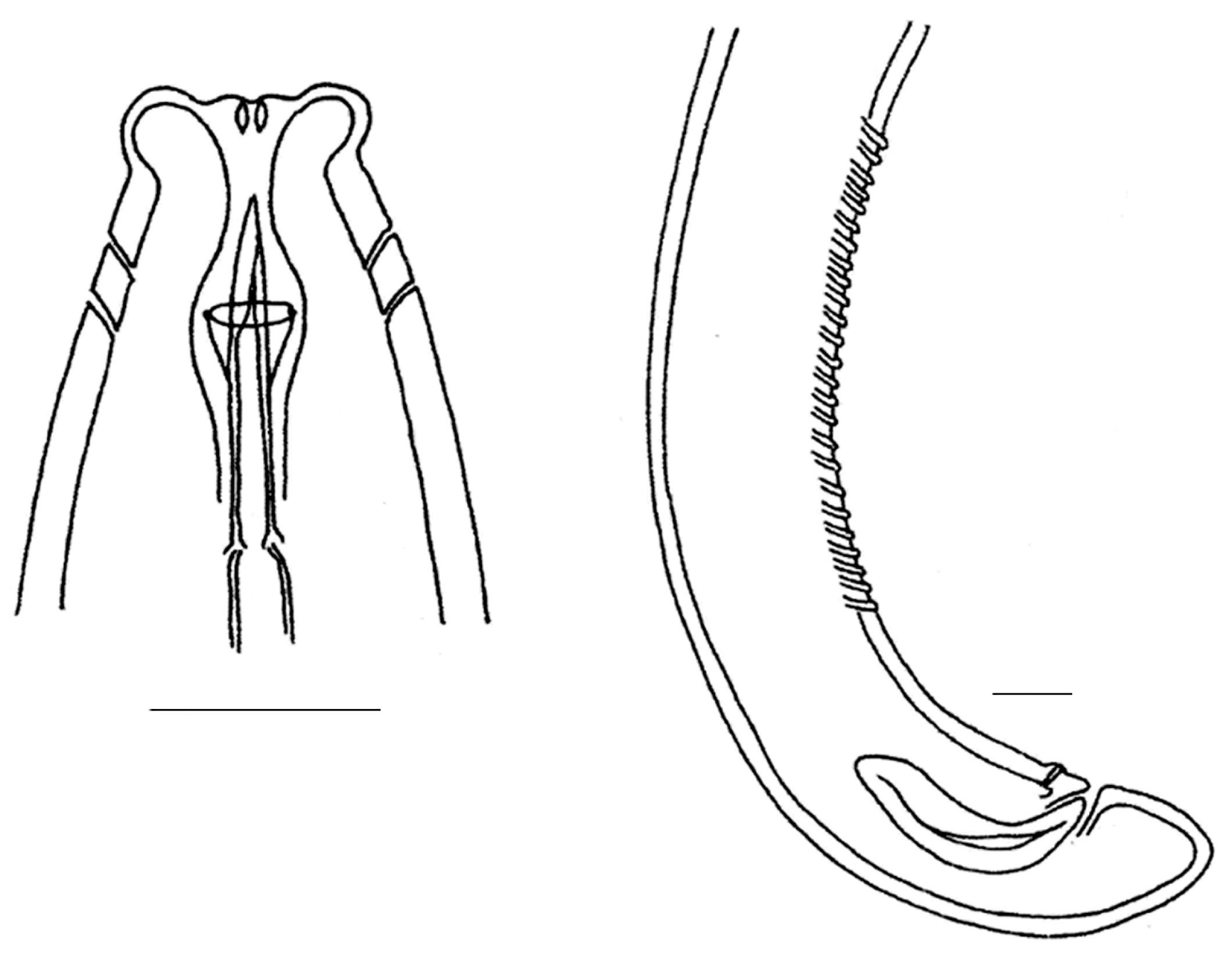
| Sampling Sites | Resting Stages Density (ind/m2) | Taxon |
|---|---|---|
| St. 1 Port area, 66 m | 2222 ± 2890 (0–5000) | Acartia cf. negligens Dana, 1849 Tortanus cf. forcipatus (Giesbrecht, 1889) Centropages cf. calaninus (Dana, 1849) C3 |
| St. 2 Kaohsiung River estuary, 63 m | 4442 ± 3852 (1666–8330) | Tortanus cf. forcipatus (Giesbrecht, 1889) Acartia cf. negligens Dana, 1849 |
| St. 3 Kaohsiung River estuary, 68 m | 2776 ± 2889 (0–4998) | Tortanus cf. forcipatus (Giesbrecht, 1889) Calanopia cf. elliptica (Dana, 1849) C2 Acartia cf. negligens Dana, 1849 |
| Locality and Depth | Resting Stages Density (ind/m2) | Taxon |
|---|---|---|
| Sun Moon Lake, Kaohsiung River system (9–11 m) | 1000 ± 714 (500–1500) | Copepoda |
| Taipei River (2 m) | 0 | – |
| Temporary water body, Keelong vicinity (0.15 m) | 32,000 ± 23,265 (1250–41,500) | Cladocera Ostracoda Rotifera Nematoda |
| Rice field near Kaohsiung (0.2 m) | 87,500 ± 67,341 (3500–120,000) | Copepoda Cladocera Ostracoda Rotifera Nematoda |
| Agricultural canal near Kaohsiung (1 m) | 45,000 ± 45,664 (1000–80,000) | Copepoda Cladocera Rotifera |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alekseev, V.; Dahms, H.-U.; Hwang, J.-S.; Sukhikh, N. Invasive and Rare Aquatic Invertebrates of Taiwan with a Focus on Their Dormancy. Water 2023, 15, 3155. https://doi.org/10.3390/w15173155
Alekseev V, Dahms H-U, Hwang J-S, Sukhikh N. Invasive and Rare Aquatic Invertebrates of Taiwan with a Focus on Their Dormancy. Water. 2023; 15(17):3155. https://doi.org/10.3390/w15173155
Chicago/Turabian StyleAlekseev, Victor, Hans-Uwe Dahms, Jiang-Shiou Hwang, and Natalia Sukhikh. 2023. "Invasive and Rare Aquatic Invertebrates of Taiwan with a Focus on Their Dormancy" Water 15, no. 17: 3155. https://doi.org/10.3390/w15173155







