Abstract
Magnetic nanomaterials (MNPs) have been widely used in the detection of pollutants in the environment because of their excellent nano effect and magnetic properties. These intrinsic properties of MNPs have diversified their application in environmental contaminant detection. In this paper, the research status quo of the use of MNPs in detecting organic and inorganic contaminants from wastewater and soil is reviewed. The preparation method and modification technology of magnetic nanoparticles are also described in detail. The application prospect of magnetic nanoparticle composites in the detection of contaminants in water and soil is discussed. Compared with traditional detection methods, MNPs are more accurate and efficient in pollutant enrichment. Moreover, the biological synthesis of MNPs was proven to be eco-friendly and aided in sustainable development. The study shows that MNPs have good application prospects in soil pollution detection, but the mechanism still needs to be investigated to realize their popularization and application.
1. Introduction
Since their discovery, nanomaterials have attracted much attention due to their unique properties. MNPs refer to magnetic particles with a scale of 0.1~100 nm, which have special nanoparticle properties and strong surface chemical activity [1]. MNPs are characterized by small particle size, large specific surface area, and good dispersion, etc. [2]. In addition, MNPs can be modified by copolymerization and surface modification [3]. In recent years, the detection of pollutants in the environment by the modification or functionalization of nanomaterials has become a research hotspot [4]. MNPs are the most commonly used adsorbent among nanomaterials, which can be used as an adsorbent for wastewater pollution detection to absorb trace organic pollutants, heavy metals, and inorganic salt pollutants [5,6]. These characteristics of MNPs make them better at pollutant detection. The small particle size characteristic gives MNPs a small size effect. The large specific surface area allows MNPs to adsorb more of the substance to be tested, increasing their activity. The strong magnetic responsiveness enables efficient separation of MNPs under an external magnetic field [7,8]. Currently, the widely used MNPs mainly include nano zero-valent iron (nZVI) [9,10], magnetite (Fe3O4) [11,12], and ferric oxide (γ-Fe2O3) nanoparticles [13,14]. The main principle of MNPs detection is that when MNPs are added to the sample, the substance to be measured can be extracted from the sample by attracting and concentrating under certain conditions, and then separated and detected under the action of an external magnetic field [15]. The use of MNPs has attracted great attention in the detection of water and soil pollution [16,17,18]. Modifying MNPs to enrich specific pollutants more efficiently, or using the characteristics of MNPs and a microbial composition biosensor to directly measure the biological toxicity of samples, has become a current research hotspot [19,20]. There have been many articles introducing the preparation, modification, and other processes of magnetic nanoparticles. However, there is still a lack of comprehensive articles on the application of magnetic nanoparticles in environmental detection. In this review, different methods to synthesize MNPs with different properties are discussed. Meanwhile, the modification methods of MNPs and the principles and methods of its functional application are reviewed, as well as the application examples of MNPs in environmental detection in water and soil. The progress and advantages of MNPs in environmental detection are introduced, and the future application prospect is presented.
We comprehensively searched the Web of Science Core Collection (WoSCC) database from inception (1986) to 21 March 2023, using the following terms: “Magnetic nanoparticles” OR “magnetic nanoparticle preparation” OR “magnetic nanoparticle modification” OR “magnetic nanoparticle application”. And through the China National Knowledge Infrastructure database with “Magnetic nanoparticles” OR “magnetic nanoparticle preparation” OR “magnetic nanoparticle modification” OR “magnetic nanoparticle application” as the search terms. There were no restrictions on language, document type, data category, or document’s year. Then we made a preliminary selection based on the title and abstract of the paper. Although magnetic nanoparticles have been extensively studied, the use of magnetic nanoparticles for environmental detection accounts for only a small fraction of the published literature. For papers that could not be clearly judged by reading the title and abstract, secondary screening was conducted by checking the content of the identified records.
2. Synthesis of Magnetic Nanoparticles
There are many types of MNPs, and the common ones are as follows: ① iron oxides, such as Fe3O4, γ-Fe2O3; ② metals and metal alloys, such as Fe, Co, Ni, etc.; ③ ferrate, such as MgFe2O4, MnFe2O4, CoFe2O4, etc. In addition, there are many ways to prepare MNPs. Common preparation methods include the co-precipitation method [21], high-temperature pyrolysis [22], microemulsion method [23], suspension polymerization method [24], ultrasonic-assisted co-precipitation method [25], and bionic mineralization method [26]. The following four commonly used methods for preparing MNPs are briefly introduced.
The co-precipitation method is a simple and effective method for the preparation of MNPs, which can be used to prepare most MNPs, such as ferric oxide (Fe3O4, Fe2O3, etc.) and ferritic (Zn-Mn ferritic, Ni-Zn ferritic, Co-Zn ferritic), etc. [27]. This method is to add alkaline solution (ammonia, sodium, sodium hydroxide solution) to the metal salt solution as a precipitating agent, so that metal ions precipitate from the solution. The process of preparing Fe3O4 magnetic particles by chemical co-precipitation is as follows. The Fe3O4 magnetic particles can be obtained by adding alkaline solution precipitant of a certain pH to the co-existing solution of Fe2+ and Fe3+, reacting under protective gas, and then separating by external magnetic field [28]. Chemical co-precipitation is widely used because of its easy availability of materials and simple preparation method.
High-temperature pyrolysis is a method for the preparation of magnetic nanoparticles by decomposition at high temperature in non-aqueous solution, which refers to a method for the preparation of magnetic nanoparticles by heating organic metal compounds in a high boiling point organic solvent under high temperature and high pressure [29]. In this method, metal complexes such as iron pentacarbonyl and ferric acetylacetonate are usually used as precursors to produce metal nanoparticles through high-temperature cracking. If these metal nanoparticles are further oxidized, metal oxide nanoparticles can be prepared. Bellaïd Sarah and Tomar Dimpal, respectively, prepared monodisperse ferric oxide nanoparticles and CoFe2O4 nanoparticles using this method. These samples all exhibit good morphology and dimensional characteristics [22,30].
Microemulsion is a transparent, isotropic, and thermodynamically stable system with low viscosity composed of oil (hydrocarbon), water (aqueous electrolyte solution), and surfactant (sometimes alcohol as co-surfactant) [23]. Microemulsion reaction types are divided into water in oil type (W/O) and oil in water type (O/W). The microemulsion droplet size is the nanometer scale, the droplets are separated from each other, and the reaction space is confined to the microreactor droplet. When two microemulsions with reactants were mixed, the exchange or transfer of substances in the water core occurred due to the collision between micelles particles. This process causes chemical reactions in the nucleus to produce magnetic nanoparticles. In addition, the microemulsion method can inhibit agglomeration of MNPs in the preparation process and control the particle size of products [31,32]. For example, Xie Yijun et al. prepared CoFe2O4 magnetic nanoparticles with high coercivity by the microemulsion method [33]. Zhao Shuchun et al. synthesized magnetic FeCoB amorphous nanoparticles by using the borohydride reduction method in water/n-hexane (W/He) microemulsion [34].
The biomimetic mineralization method is used to control the generation of inorganic mineral materials by imitating the organic biological tissues in the process of biological mineralization, and magneto tactic bacteria are used to react to generate magnetic nanoparticles under appropriate conditions [26]. Liu et al. used a 14-mer bi-functional copolypeptide as a template for biomimetic mineralization of magnetite [35]. Zhou Yanhong et al. designed temperature-responsive elastic polypeptides to participate in biomimetic mineralization, and successfully prepared elastin-like polypeptides-MNPs with multiple responses [36]. The MNPs synthesized by this method have fine and uniform particle size, high crystallinity and purity, and special crystal shape and chain arrangement, which have great advantages in biomedical applications. However, the cost of preparing magnetic nanoparticles of the same size using the biomineralization method is 18 times higher than that using the co-precipitation method. At the same time, due to the high reproductive conditions of microorganisms such as Magnetospirillum magneticum, they cannot be cultured on a large scale, and there is a high maintenance cost of related production equipment; the above problems have led to low production capacity of the biomineralization method. How to solve these problems is the key to breaking the technical barriers of the biomimetic mineralization method [37,38].
As shown in Table 1, each of the four methods has advantages and disadvantages in the preparation of magnetic nanoparticles. The preparation process of the co-precipitation method is relatively simple and suitable for large-scale production. However, the prepared MNPs are prone to agglomeration, uneven particle size distribution, and poor biocompatibility, so the surface needs to be modified to be used. The pyrolysis method and microemulsion method have advantages in the preparation of monodisperse MNPs, but the product yield is low and the cost is high. MNPs prepared by the biomimetic mineralization method have good biocompatibility and bioactivity, and the preparation process is green and environmentally friendly, so they are widely used in the field of medicine. It is also a hot research direction to combine with microorganisms to prepare sensors. The modification and functionalization of MNPs should be further studied.

Table 1.
Comparison of advantages and disadvantages of MNPs preparation methods.
3. Modification and Functionalization of MNPs
MNPs have high specific surface energy and dipole interaction. MNPs tend to agglomerate and lose magnetism [42]. Modified MNPs can prevent the aggregation of nanoparticles and maintain a certain stability of the colloidal system. Meanwhile, MNPs have biocompatibility, water solubility, biological coupling, and cell non-specificity, so that MNPs can be used to realize the application of fixed load, biomolecule binding, and biosensors [43]. The modification methods and functionalization applications of magnetic nanoparticles are mainly as follows.
3.1. Magnetic Nanomaterials Functionalized with Metal-Organic Frameworks
The organic framework, also known as porous coordination polymers, is a widely used porous crystal material assembled by metal ions or metal clusters and the coordination of organic ligands [44]. The main characteristics of the organic framework are organic ligand as the framework, and metal ions or their clusters as the coordination center, in the form of a coordination bond sum connected into a three-dimensional space network structure. Metal-organic frameworks (MOFs) have the characteristics of large specific surface area, adjustable cavity, good stability, and adjustable chemical properties. In addition, MOFs are relatively easy to synthesize and have been studied in depth and widely used in adsorption separation, catalysis, drug loading, and other fields [45]. Therefore, magnetic metal organic skeleton composites with a wider range of applications can be prepared by combining the advantages of magnetic nanomaterials and metal organic frameworks.
Magnetic nanomaterials functionalized with MOFs are commonly used in water environmental organic pollutants detection. Cai Dandan et al. prepared a metal organic framework material Cobalt 2-methylimidazole as the precursor, synthesized magnetic Co/C nanocomposites by high-temperature pyrolysis, and applied them to the detection of the content of organic dyes in water, such as Congo red [46]. Lian Lili et al. constructed a unique coil-shell titanium fund organic skeleton functionalized magnetic microsphere Fe3O4@Cys@MIL125-NH2 and used it as a magnetic adsorbent to enrich five fluoroquinolones in water samples, which can be used to detect fluoroquinolones in tap water and environmental water samples [47].
3.2. Ionic Liquid Functionalized Magnetic Nanomaterials
Ionic liquids are molten salts that are liquid at room temperature and typically consist of organic cations, and organic or inorganic anions. Ionic liquids are widely used in the fields of extraction and separation due to their non-volatile properties, low melting point, and good solubility [48]. In order to improve the application ability of ionic liquids in solid-phase extraction, they can be combined with magnetic nanoparticles based on phase separation as extractant carriers to form functional magnetic nanoparticles of ionic liquids for the analysis of trace components in biological, environmental, and food complex samples [49].
Lu Dingkun et al. coated hydrophobic carboxyl functionalized ionic liquid (IL-COOH) in the prepared Fe3O4@Zr-MOFs, synthesized a novel water-stable IL-COOH/Fe3O4@ZR-MOF nanocomposite for the first time, and applied it in the selective adsorption and detection of fluoroquinolone antibiotics [50]. Ping Wenhui et al. synthesized room temperature ionic liquid loaded cyclodextrin magnetic nanomaterials and used them to perform magnetic solid phase extraction on samples, which can be used for extraction analysis of organic pollutants in water [51].
3.3. Magnetic Nanomaterials Functionalized by Molecularly Imprinted Polymers
Molecularly imprinted polymer (MIP) contains holes with a specific spatial structure and has the advantages of being tailor-made, having a simple preparation, and being stable and reusable [52]. Magnetic molecularly imprinted nanoparticles (MMIP NPs) developed on this basis are a kind of material that can be selectively enriched and separated by an external magnetic field [53].
Chen Fangfang et al. used superparamagnetic Fe3O4 nanoparticles functionalized with silica layer as the carrier, water-soluble 4-[(4-methacryloyloxy)phenylazo]benzenesulfonic acid as the functional monomer, and sulfadiazine as the template to successfully prepare a dual-response molecule-imprinted polymer sensitive to both photon and magnetic stimulation. It was used for the detection of sulfonamides in aqueous media [54]. Because MMIP NPs recognition sites are established on or near the surface of magnetic nanoparticles, rapid adsorption and elution of the object to be tested can be achieved. Therefore, it is not only simple to operate, but also greatly saves the time of sample pretreatment when it is applied to the enrichment and separation of the substance to be tested [55].
There are currently multiple methods for modifying magnetic nanoparticles and enhancing their properties. However, further exploration is needed to find a stable, inexpensive, and easy to prepare method for modifying magnetic nanoparticles.
4. Application of MNPs in Environment Detection
MNPs are widely used because of their characteristics, such as easy separation, high stability, and easy surface encapsulation [47,50,56]. The main application ways of MNPs in environmental detection include the enrichment of environmental pollution substances, or combining with bacteria to make biosensors for environmental quality detection. At present, MNPs are commonly used in water and soil detection [44,49,57]. Table 2 introduces several environmental samples detected using MNPs.

Table 2.
Comparison of analytical methods for various environmental samples.
4.1. Application in Water Environment Detection
MNPs are widely used in water environment detection, mainly combined with other ornaments to detect or enrich various pollutants in water [60,61,62]. The detected pollutants in water are mainly divided into organic matter and inorganic matter. Organic matters include dyes [58], pesticides [63], drugs [64], chemical raw materials [65], etc. Inorganic substances are mainly heavy metal ions and acid ions [66,67,68,69], etc.
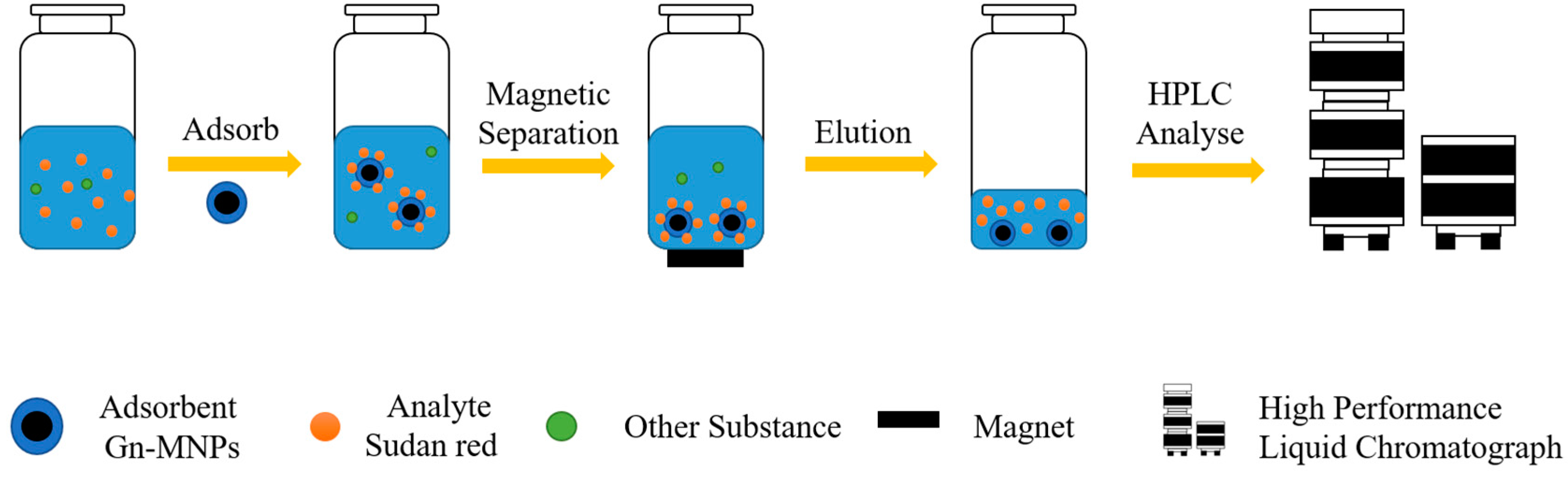
Wu Yalin et al. synthesized and characterized a new type of MNP, polyamide amine dendrimer-modified magnetic nanoparticles (Gn-MNPs) [58]. As shown in Figure 1, under the adsorption action of the adsorbent Gn-MNP, the Sudan red in the solution was adsorbed by stirring for 1 h. The adsorbent is separated from the original solution by magnetic separation of the magnet. Sudan red is then desorbed from the adsorbent with acetone. After the eluent is dried with nitrogen, the residue is eluted with methanol and dispersed. The dispersed samples are taken and sent for inspection. The nanomaterial has good adsorption performance for Sudan red, methyl green, and Congo red dye in natural water. The detection limit of Congo red is in the range of 1.8~5.5 ng·L−1, and the precision is less than 3.0%. The nanomaterial has significant application potential in the enrichment of trace environmental pollutants in water samples.
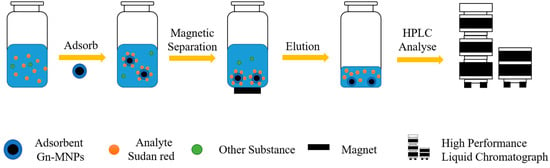
Figure 1.
Schematic diagram of Gn-MNP usage [58].
MNPs are also used to enrich and detect pesticides in environmental water samples. In practical application, the recovery rate of adding samples can reach 90%. Cao et al. synthesized the Cupric organic skeleton/ferrioxide (MOF-199/Fe3O4) complex by in situ method at room temperature, and applied it to the separation and detection of nicotinic insecticides in environmental water samples [70]. Turiel et al. modified the surface of MNPs by molecular imprinting technology, and tested it as a selective adsorbent for extracting triazine compounds from environmental water [71]. In addition, Kouhestani et al. synthesized cysteine-functionalized chitosan-coated magnetic nanoparticles and used them as an effective adsorbent for the extraction of casine and benzene sulfomethyl [63]. Using imidacloprid as a template and dopamine as a functional monomer, Cui et al. prepared MMIP through the nucleation process of magnetic nanoparticles Fe3O4 and the polymerization process of molecularly imprinted polymers [72]. Under the best conditions, imidacloprid was extracted from water samples by MMIP and HPLC.
MNPs can also enrich drugs in water. Gamal et al. prepared MNPs modified with cetyltrimethyl ammonium bromide (CTAB), and then carried out solid phase extraction of valsartan in water samples, pre-enrichment, and detection [64]. The working principle of this method is to use MNPs as adsorbents to adsorb the tested substance, and then use methanol for desorption. A mobile phase composed of phosphate buffer (0.03 M), acetonitrile, and methanol (40:40:20%) is used to separate and desorb the solution. Finally, chromatographic analysis is performed on the separated solution to obtain the concentration of the substance to be tested. These MNPs have a good linear response within the concentration range of 10–150 ng/mL, and the detection limit can reach 2.02 ng/mL.
Since MNPs can rapidly extract the substance to be tested in the extraction solution, they are widely used in the detection of organic reagents. Wu synthesized Fe3O4 magnetic nanoparticles modified with polyamide dendrimers. The magnetic solid phase extraction (MSE) of trace Tetrabromobisphenol A and 4-Nonylphenol in environmental water samples was carried out using modified magnetic particles as effective adsorbents [73]. After this process, magnetic nanoparticles are applied to facilitate extraction of the extraction solvent containing bisphenol A. At the same time, Tian et al. prepared Fe3O4@SiO2@CTS for the detection of polychlorinated biphenyls (PCBS) by combining modified natural polysaccharide chitosan (CTS) with silica on the surface of magnetic Fe3O4 nanoparticles [66]. These results indicate that MNPs have good selectivity, easy magnetic separation, and can be reused.
At the same time, there are many new nanotechnology applications for the detection and removal of pollutants in water. Zheng Yanmei et al. developed synthesizing a novel SCN/rGO PNs via a supramolecular self-assembling followed by a solvothermal treatment [74]. Under the action of this material, the photocatalytic activity of Cr(VI) and Rhodamine B enhance 17 times compared to ordinary materials. Bharatraj Singh Rathore et al. used a batch adsorption technique to prepare chitosan-PANI-Fe2O3. This material can adsorb and remove dyes such as methyl orange from water, with a removal efficiency of 91.5% [75]. In addition, Sadaf Bashir Khan et al. summarized the research progress of 3D printed nanosheet membranes for water purification, providing a new approach for water treatment [76].
Magnetic solid phase extraction (M-SPE) is a dispersible solid phase extraction technique in which magnetic or magnetized materials are used as adsorbent substrate [77,78,79]. Compared with conventional solid phase extraction (SPE) fillers, nanoparticles have larger specific surface area and shorter diffusion distance, and only need to use a small amount of adsorbent and a short equilibrium time to achieve low concentration of microextraction, with very high extraction capacity and extraction efficiency. This method has the following advantages: ① fast extraction speed; ② high pre-enrichment factor; ③ sensitive detection; ④ small relative standard deviation.
John Kong et al. synthesized Mangan-based MNPs by the microemulsion method, coated the surface with gold, and fixed cysteine through the Au-S covalent bond, for the capture and detection of heavy metal Hg2+ [80]. These magnetic MNPs are highly efficient nano-adsorbents for heavy metals in aqueous solutions, thus contributing to the determination of Hg2+ ion content in aqueous solutions in environmental tests. The magnetic adsorbent has good selectivity for Pb2+, fast adsorption kinetics, and large adsorption capacity. Using 4 nm ZnO nanoparticles as a sacrificial template, Zhao et al. prepared double-imprinted polymer-coated MNPs with template Pb2+ ion co-imprinting, and detected trace Pb2+ by graphite furnace atomic absorption spectrometry [79]. Jagirani et al. extracted trace lead from environmental samples by solid phase microextraction (SPME) using magnetic cellulose nanoparticles (Cell-MNPs) as adsorbent [61]. By optimizing the analysis parameters, Cell-MNPs were successfully used for the solid phase extraction of Pb2+ in water samples. Wu et al. used magnetic solid phase extraction of 1-octanol with hydrophilic magnetic nanoparticles [62]. Schiff base was used as the complex agent, acetonitrile/water (60:40, v/v) was used to treat environmental samples, acetonitrile/water was used as the dispersive solvent, and 1-octanol was used as the extractant for dispersive liquid-liquid microextraction to detect Cr3+, Ni2+, Co2+, and Hg2+ in environmental water samples. This method has a lower detection limit and can achieve a recovery rate of over 90% for metal ions in water.
Maleki et al. synthesized the second generation of amino-dendrimer functionalized MNPs (Fe3O4@G2-PAD) and applied it to the measurement of Pb2+ and Cd2+ ions in environmental water bodies (river, wastewater, and lake) [81]. Wu et al. used Selective Trace analysis of Pb2+ and Cd2+ based on glutathione-modified silver nanoparticles and Fe3O4 magnetic nanoparticles [82]. The presence of Pb2+ and Cd2+ induces AgNPs aggregation through co-metal ligand interaction and loads the aggregation onto magnetic Fe3O4 nanoparticles as adsorbent. The detection limits of Cd and Pb were 0.13 and 1.25 g·L−1, respectively, and the relative standard deviations were 1.5% and 1.8% at the concentration level of 10 g·L−1, respectively. Wang et al. used magnetic Fe3O4/ZrO2/Ag composite microspheres to explore the sensitive surface-enhanced Raman Scattering (SERS) substrate for rapid detection of Cr6+, and adopted the optimized Fe3O4/ZrO2/Ag system for quantitative detection of Cr6+ in aqueous solution [83]. The results showed that there was a good linear relationship between SERS intensity and logarithmic concentration of Cr6+ (R2 = 0.98), and the detection limit could be as low as 10−7 M. Sulfhydryl-amino functionalized magnetic nanoparticles Fe3O4@SiO2@MPTMS and Fe3O4@SiO2@APTES were used as adsorbents for magnetic solid phase extraction (MSPE) to directly extract As(III) and As(V), respectively, and were detected by inductively coupled plasma mass spectrometry (ICP-MS) [84]. Sulfhydryl and amino extracts As(III) and As(V) were recovered through coordination and electrostatic interactions, respectively. The recoveries of As(III) and As(V) are 89~96% and 90~102%, respectively. These methods had been successfully applied to the evaluation of heavy metal content in environmental water samples with satisfactory results.
The colorimetric or optical technique-based MNPs can be used for qualitative and quantitative detection of heavy metals in water, and this method has the advantages of high stability, strong selectivity, and easy operation [85,86]. Mehmet Oguz et al. modified fluorescent compounds into Fe3O4 nanoparticles, and detected mercury ion (Hg2+) in the water environment according to the luminescence intensity in MNPs water [87]. Liu et al. synthesized gold nanoparticles (AuNPs) on the surface of Fe3O4 NPs to achieve visual detection of Au@ Fe3O4 NPs with a high concentration of Hg2+ in industrial pollutants [88]. This method has high sensitivity and selectivity, and can be used for qualitative analysis of Hg2+ above 5 μM by the naked eye. The fluorescence technique is an effective method for detecting pollutants. Li synthesized magnetic Fe3O4@SiO2-TbDPA nanoprobes [89]. Synthetic Fe3O4@SiO2-TbDPA aqueous solution exhibits strong green luminescence. However, when different concentrations of NO2− (0–100 µM) are added, the fluorescence intensity is inhibited. According to the pre-measured inhibition luminescence curve, the corresponding NO2− ion content of the solution to be tested can be obtained. With good linearity in the range of 5~80 µM and a detection limit of 1.03 µM, it can be extended to a wide range of environmental monitoring and biomedical fields.
The research on magnetic nanoparticles used for detecting pollutants in water has been very in-depth, and the magnetic nanoparticles have shown good results in the detection process of different types of pollutants. However, the preparation of most modified MNPs is difficult and cannot be reused multiple times. In the future, MNPs that can be reused multiple times and prepared easily should be developed.
4.2. Application in Soil Environmental Detection
The comprehensive biological toxicity of heavy metals and organic matter in polluted soil will cause human health risks, lead to human poisoning, and even affect the whole ecosystem [90,91]. With the attention paid to soil biotoxicity, accurate detection of soil biotoxicity in the presence of multiple types and forms of pollutants has become the focus of research [92]. Due to MNPs’ fast adsorption rate and easy magnetic separation, they have been widely used in soil environmental detection in recent years.
The main advantage of magnetic nanoparticles is that they can be easily and efficiently removed from the treated solution using a common magnet. Singh synthesized magnetic iron oxide (Fe3O4) nanoparticles by chemical co-precipitation, which were used to remove heavy metals (Cd, Cr, Cu, Fe, Ni, Pb, and Zn) from soil samples [18]. When the pH of soil leaching solution is 0.7, the adsorption efficiency of heavy metals is between 69.6 and 99.6%. The adsorption efficiency of MNPs on various metals was determined by atomic absorption spectrometry. The results showed that the adsorption effect of lead MNPs was the best and the detection efficiency was the highest in soil. Kim et al. used bare Fe3O4 nanoparticles to selectively separate Cs-contaminated clay particles from soil and detect Cs content in soil [93]. When the mass ratio of MNPs to clay is close to 0.1, and at low pH, the recovery rate of Cs can reach more than 90%. The reason is that the increase of electrostatic attraction and dispersion is conducive to magnetic separation of clay minerals at low pH. Kasa et al. established a fast and effective method for dispersive solid phase microextraction (d-SPME) of Pd in soil using MNPs [94]. Pd can be directly separated from soil sample solution without complexation. The grooved quartz tube-flame atomic absorption spectrometry (SQT-FAAS) was used. The limits of detection and quantification of Pd by this method were 6.4 ng/mL and 21.4 ng/mL, respectively, and the relative standard deviation was 6.6%. The recoveries of this method are 90~101%, which proves the accuracy and applicability of this method.
Some scholars use modified magnetic nanoparticles to separate pesticides from soil and detect them. For example, Maria Jose et al. synthesized a promazine-imprinted polymer on the surface of modified MNPs for solid phase extraction of triazine from soil samples. The obtained MNPs exhibit high selectivity for triazine compounds and are easily collected and separated by external magnetic fields without additional centrifugation or filtration steps [95]. The recoveries of triazines ranged from 5.4% to 40.6% with relative standard deviations less than 7.0% (n = 3) and detection limits of 0.1 to 3 ng·g−1, depending on the triazines and the type of soil used. Lei Sun et al. synthesized alumina-coated magnetite nanoparticles (Fe3O4/Al2O3 NPs) and applied them to the analysis of sulfonamides based on magnetic solid phase extraction of sulfadimethylate and sulfaquinoxaline in different soil samples [59]. The extraction and concentration process are completed in one step by mixing extraction solvent, magnetic adsorbent, and a soil sample under the action of an ultrasonic wave. The adsorbent is then easily separated from the complex substrate under the action of an applied magnetic field, and the sulfonamides desorbed from the adsorbent are then determined by liquid chromatography-tandem mass spectrometry. Kyung Tae Kim et al. fixed naphthalimide DPA(2) on the surface of iron oxide nanoparticles to prepare hybrid nanomagnetic 1-Fe3O4 [96]. The binding of naphthalimide DPA(2) and 1-Fe3O4 to Zn2+ resulted in a significant increase in fluorescence intensity at 527 nm. The nanomagnetite 1-Fe3O4 can be used for the selective detection and removal of Zn2+ from soil samples. Lu Yang synthesized a polyionic liquid (PIL) and fixed it on the prepared SiO2-coated MNPs, used it as a magnetic solid phase (MSPE) adsorbent, and used it to extract sulfonylurea herbicides (SUHs) from soil samples prior to HPLC analysis [97]. The relative standard deviation of repeated assay analytes ranges from 3.2% to 4.5%. The limits of detection and quantification were 1.62~2.94 ng·mL−1 and 5.4~9.8 ng·mL−1, respectively. These results indicate that Fe3O4@SiO2@PIL can effectively extract sulfonylurea herbicides from soil samples.
Pang used MNPs to enrich and detect sulfonylurea herbicides and heavy metal ions in contaminated soil [98,99]. He doped Fe3O4 magnetic nanoparticles into a monolithic capillary microextraction column (MCMC) of polyethylene imidazole dimethyl acrylate polymer. Concentrate sulfonylurea herbicides (SUHs) under the action of a magnetic field, and the extraction efficiency can reach 82.6–94.5%. It can detect the content of trace SUHs in soil samples with a detection limit (S/N = 3) of 0.30~1.5 µg/kg. In addition, he coordinated Cr3+ and Cr6+ with pyrrolidine ammonium dithiocarbamate (APD) to form Cr3+/APD and Cr6+/APD complexes, respectively. Then, a porous monomer microextraction column doped with magnetic nanoparticles was prepared in situ in a capillary tube. The results showed that applying a magnetic field helped improve the extraction efficiency of Cr3+/APD (80.4%) and Cr6+/APD (86.2%) complexes. The detection limits of Cr3+ and Cr6+ in soil samples, respectively, were 0.47 and 0.057 µg/kg. The enrichment factors of Cr3+ and Cr6+, respectively, were 59 and 72.
Li et al. synthesized MOF-1210 (Zr/Cu)-MNPs-modified MNPs by the solvothermal method in one step for magnetic solid phase extraction (MSPE) of benzophenone [100]. It was successfully applied to the extraction and detection of benzophenone in soil samples. The recoveries were 87.6–113.8%, RSDS < 11.12%. Hubeyska et al. established the QuEChERS extraction and purification process based on hydrophobic MNPs (C18/GCB/Fe3O4/Fe3O4@Triton), and combined it with gas chromatography-mass spectrometry for the simultaneous determination of 16 organochlorine pesticides, and applied this method to the determination of pesticide residues in agricultural soil samples [101]. Kim’s application of Fe3O4 nanocomposites coated with polyethylenimide (PEI) for selective separation of clay particles from Cs-contaminated soils was used. The PEI coating on the surface of nano Fe3O4 enhances the binding force between magnetic nano Fe3O4 and clay minerals by electrostatic attraction [102]. The Fe3O4-PEI nanocomposite magnetically separates approximately 100% of the clay particles from the solution at a low dose (0.04 nanocomposite/g clay).
MNPs that are directly used to adsorb pollutants in soil have poorer adsorption effects and lower recovery rates compared to water. The reason is that MNPs are difficult to separate from soil effectively. Therefore, in the future, we need to explore MNPs with higher recovery rates in soil and explore new directions for their application in soil.
In soil environmental testing, accurate detection of soil biotoxicity is the starting point and even the end point of contaminated soil evaluation and remediation [103]. In recent years, there have been problems in the method of using analytical chemical detection technology to determine the content of pollutants and calculate their biotoxicity through models [104,105]: When using chemical extraction and other force methods to extract pollutants, the use of chemical agents, centrifugal shock, and other treatments will destroy the physical and chemical properties of pollutants, resulting in the inability to truly quantify the biological toxicity of contaminated soil. In particular, when multiple pollutants have synergistic and impedance effects, the model often cannot accurately and quantitatively describe the comprehensive biological toxicity. However, the detection of biological toxicity by traditional analytical chemical methods is essentially the analysis of pollutant content, rather than the direct reflection of biological toxicity. Compared with the analytical chemical method, the microbial detection method is a direct detection of the biotoxicity of harmful elements transferred to the liquid phase in contaminated soil, which is considered as a very promising technology for the detection of biotoxicity in contaminated soil. MNPs prepared by biofriendly methods and modified and functionalized can be combined with microorganisms to form biosensors, which can well establish a microbial detection method of MNP-based biosensors and nanosensors. This method with high sensitivity and good reproducibility can accurately characterize the comprehensive biotoxicity of composite contaminated soil.
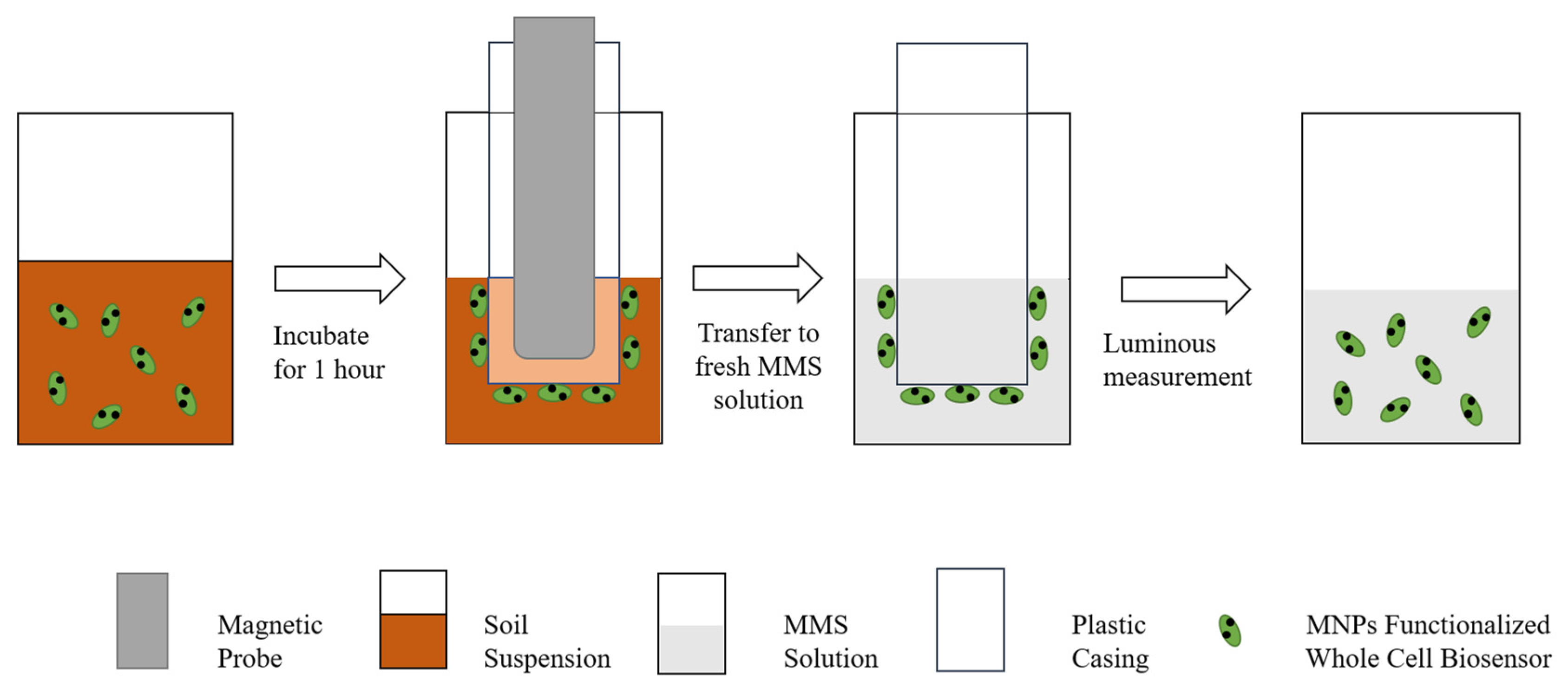
Jia Jianli et al. developed a whole-cell bioreporter ADPWH recA, which combines magnetic nanoparticles with genetically modified Escherichia coli and can judge soil ecotoxicity by luminescence intensity. The application of magnetic nanoparticles allows permanent magnets to recover sensors from soil samples to reduce soil particle interference [106]. Figure 2 shows the use process of the sensor. After the sensor reacts in the soil diluent, the magnetic probe is used to separate the sensor from the solution, and the luminescence intensity is measured after the MMS solution to characterize the soil eco-toxicity. Compared with the traditional treatment of applying a bioreporter directly to a soil-water mixture (SW-M treatment) or supernatant (SW-S treatment), the bioreporter functionalized with MNPs by a magnetic device (MFB) has higher sensitivity and better reproducibility to evaluate the toxicity and bioavailability of Cr contamination in soil. Zhang et al. used MNPs to combine with Bright Luminescent Bacillus to synthesize a magnetic nanobacterial sensor for detecting soil biological toxicity [107]. The characteristic of this sensor is that it does not require changing the physical and chemical properties of pollutants in the soil, thus obtaining more accurate results for determining the biological toxicity of contaminated soil [108].
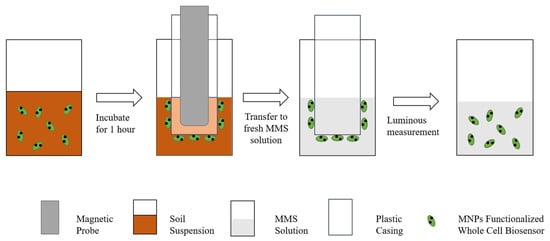
Figure 2.
Schematic diagram of the magnetic biosensor device [106].
5. Conclusions and Outlook
This article reviews the research progress of magnetic nanomaterials from two aspects: preparation and modification. In addition, this article reviews the preparation methods of magnetic nanomaterials and their applications in the field of environmental detection. In the current research, researchers have developed a series of magnetic nanomaterials for soil and water environment detection. These materials have achieved good results in environmental testing, but there are still issues that need to be addressed, such as the complex preparation methods for MNPs and the low recovery rates of MNPs in soil. Although magnetic nanoparticles can directly extract pollutants from water and soil for quantitative detection, they cannot directly detect the biological toxicity of environmental samples. The article mentions that combining MNPs with microorganisms to prepare an MNPs-microbe sensor can effectively measure the biological toxicity of environmental samples. This method separates the sensor from the contaminated sample through the action of an external magnetic field, and can directly measure the biological toxicity of the sample without other operations. However, due to the easy aggregation and poor biological activity of ordinary MNPs, the performance of synthetic sensors is unstable. How to prepare magnetic nanoparticles with regular shapes, good dispersion performance, good biocompatibility and biological activity, good stability under physiological conditions, and green environmental protection is currently a research hotspot. Biomimetic mineralization is the most successful alternative method for producing nanomaterials in a biologically friendly manner, but this method currently has problems such as high preparation costs and low yield. In the future, further research should be conducted on biomimetic mineralization methods to facilitate the widespread utilization of bio-friendly MNPs in industries such as healthcare, environment, and food.
Author Contributions
Conceptualization, K.Z. and X.S.; methodology, K.Z. and X.S.; software, M.L.; validation, K.Z. and X.S.; formal analysis, X.S. and M.L.; investigation, M.L. and J.L.; resources, K.Z. and X.S.; data curation, K.Z. and X.S.; writing—original draft preparation, K.Z. and X.S.; writing—review and editing, K.Z. and X.S.; visualization, J.H.; supervision, J.L. and M.C.; project administration, K.Z.; funding acquisition, K.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (42177037), Key Research and Development Program of Autonomous Region (2022B03028-1), and the Fundamental Research Funds for the Central Universities (2023JCCXHH02).
Data Availability Statement
Not applicable.
Acknowledgments
We acknowledge all the authors for their contributions. We sincerely thank the anonymous reviewers and the editor for their effort to review this manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef]
- Zhang, Y.; Cheng, J.; Liu, W. Characterization and relaxation properties of a series of Monodispersed magnetic nanoparticles. Sensors 2019, 19, 3396. [Google Scholar] [CrossRef]
- Zhu, N.; Ji, H.N.; Yu, P.; Niu, J.Q.; Faroop, M.U.; Akram, M.W.; Udego, I.O.; Li, H.; Niu, X. Surface modification of magnetic iron oxide nanoparticles. Nanomaterials 2018, 8, 810. [Google Scholar] [CrossRef]
- Lu, R.; Wang, C.; Chen, Y.; Tan, L.; Wang, P.; Feng, S. IL-functionalized Mn(II)-doped core-shell Fe3O4@Zr-MOF nanomaterials for the removal of MB from wastewater based on dual adsorption/Fenton catalysis. New J. Chem. 2022, 46, 8534–8544. [Google Scholar] [CrossRef]
- Chai, Z.; Yuan, C.; Qian, Y.; Zhang, Y.; Wang, J. Developed on the Adsorption Properties of 9 Organophosphorus Pesticides in Spinach by Magnetic Nanoparticles Modified Carbon Nanotube Composites. Food Ferment. Sci. Technol. 2019, 54, 70–75. (In Chinese) [Google Scholar]
- Lou, X.Y.; Boada, R.; Verdugo, V.; Simonelli, L.; Pérez, G.; Valiente, M. Decoupling the adsorption mechanisms of arsenate at molecular level on modified cube-shaped sponge loaded superparamagnetic iron oxide nanoparticles. J. Environ. Sci. 2022, 121, 1–12. [Google Scholar] [CrossRef]
- Yang, J.; Zhou, L.; Ma, F.; Zhao, H.; Deng, F.; Pi, S.; Tang, A. Magnetic nanocomposite microbial extracellular polymeric substances@Fe3O4 supported nZVI for Sb(V) reduction and adsorption under aerobic and anaerobic conditions. Environ. Res. 2020, 189, 109950. [Google Scholar] [CrossRef]
- Plastiras, O.-E.; Deliyanni, E.; Samanidou, V. Synthesis and Application of the Magnetic Nanocomposite GO-Chm for the Extraction of Benzodiazepines from Surface Water Samples Prior to HPLC-PDA Analysis. Appl. Sci. 2021, 11, 7828. [Google Scholar] [CrossRef]
- Zhou, Z.; Huang, J.; Xu, Z.; Ali, M.; Shan, A.; Fu, R.; Lyu, S. Mechanism of contaminants degradation in aqueous solution by persulfate in different Fe(II)-based synergistic activation environments: Taking chlorinated organic compounds and benzene series as the targets. Sep. Purif. Technol. 2021, 273, 118990–118998. [Google Scholar] [CrossRef]
- Vilardi, G.; Rodríguez-Rodríguez, J.; Ochando-Pulido, J.M.; Verdone, N.; Martinez-Ferez, A.; Palma, L.D. Large Laboratory-Plant application for the treatment of a Tannery wastewater by Fenton oxidation: Fe(II) and nZVI catalysts comparison and kinetic modelling. Process Saf. Environ. Prot. 2018, 117, 629–638. [Google Scholar] [CrossRef]
- Al-Qasmi, N.; Almughem, F.A.; Jarallah, S.J.; Almaabadi, A. Efficient Green Synthesis of (Fe3O4) and (NiFe2O4) Nanoparticles Using Star Anise (Illicium verum) Extract and Their Biomedical Activity against Some Cancer Cells. Materials 2022, 15, 4832. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.D.; Tran, H.-V.; Xu, S.; Lee, T.R. Fe3O4 Nanoparticles: Structures, Synthesis, Magnetic Properties, Surface Functionalization, and Emerging Applications. Appl. Sci. 2021, 11, 11301. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Peng, X.; Ni, F.; Li, J.; Wang, D.; Luan, Z. Arsenite removal from aqueous solutions by γ-Fe2O3-TiO2 magnetic nanoparticles through simultaneous photocatalytic oxidation and adsorption. J. Hazard. Mater. 2013, 246–247, 10–17. [Google Scholar] [CrossRef]
- Akhbarizadeh, R.; Shayestefar, M.R.; Darezereshki, E. Competitive Removal of Metals from Wastewater by Maghemite Nanoparticles: A Comparison Between Simulated Wastewater and AMD. Mine Water Environ. 2014, 33, 89–96. [Google Scholar] [CrossRef]
- Wierucka, M.; Biziuk, M. Application of magnetic nanoparticles for magnetic solid-phase extraction in preparing biological, environmental and food samples. Trends Anal. Chem. 2014, 59, 50–58. [Google Scholar] [CrossRef]
- Kim, J.-H.; Kim, S.-M.; Yoon, I.-H.; Choi, S.-J.; Kim, I. Selective separation of Cs-contaminated clay from soil using polyethylenimine-coated magnetic nanoparticles. Sci. Total Environ. 2020, 706, 136020. [Google Scholar] [CrossRef]
- Li, T.; Yu, Z.Y.; Yang, T.L.; Xu, G.; Guan, Y.; Guo, C. Modified Fe3O4 magnetic nanoparticles for COD removal in oil field produced water and regeneration. Environ. Technol. Innov. 2021, 23, 101630. [Google Scholar] [CrossRef]
- Singh, A.; Chaudhary, S.; Dehiya, B.S. Fast removal of heavy metals from water and soil samples using magnetic Fe3O4 nanoparticles. Environ. Sci. Pollut. Res. 2021, 28, 3942–3952. [Google Scholar] [CrossRef]
- Heo, Y.J.; Lee, E.-H.; Lee, S.-W. Adsorptive removal of micron-sized polystyrene particles using magnetic iron oxide nanoparticles. Chemosphere 2022, 307, 135672. [Google Scholar] [CrossRef]
- Guo, M.; Huang, K.; Xu, W. Third generation whole-cell sensing systems: Synthetic biology inside, nanomaterial outside. Trends Biotechnol. 2021, 39, 550–559. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, J.; Gu, L.; Wang, L.; Zhang, Q. Preparation and electromagnetic properties of nanosized Co0.5Zn0.5Fe2O4 ferrite. Rare Met. 2022, 41, 3228–3232. [Google Scholar] [CrossRef]
- Tomar, D.; Jeevanandam, P. Synthesis of cobalt ferrite nanoparticles with different morphologies via thermal decomposition approach and studies on their magnetic properties. J. Alloys Compd. 2020, 843, 155815. [Google Scholar] [CrossRef]
- Beygi, H.; Babakhani, A. Microemulsion synthesis and magnetic properties of FexNi(1-x) alloy nanoparticles. J. Magn. Magn. Mater. 2017, 421, 177–183. [Google Scholar] [CrossRef]
- Fatima, S.; Imran, M.; Kanwal, F.; Javaid, A.; Latif, S.; Boczkaj, G. Design and Preparation of Magnetically-Oriented Poly(styr-co-MMA)-3MPS Capped Fe(ZnO) Hybrid Microspheres for Ion Exchange Removal of Toxic Pollutants from Wastewater. Water 2023, 15, 1761. [Google Scholar] [CrossRef]
- Darwish, M.S.A.; Al-Harbi, L.M. Self-heating properties of iron oxide nanoparticles prepared at room temperature via ultrasonic-assisted co-precipitation process. Soft Mater. 2022, 10, 35–44. [Google Scholar] [CrossRef]
- Mao, Y.; Yang, Y.; He, S.; Li, Y.; Gu, N. Biomimetic synthesis of magnetic nanoparticles mediated by magnetosome proteins. Chin. Sci. Bull. 2020, 65, 1476–1485. (In Chinese) [Google Scholar] [CrossRef]
- Gandhi, V.; Ganesan, R.; Syedahamed, H.H.A.; Thaiyan, M. Effect of cobalt doping on structural, optical, and magnetic properties of ZnO nanoparticles synthesized by coprecipitation method. J. Phys. Chem. C 2014, 118, 9715–9725. [Google Scholar] [CrossRef]
- Rajput, S.; Pittman, C.U.; Mohan, D. Magnetic magnetite (Fe3O4) nanoparticle synthesis and applications for lead (Pb2+) and chromium (Cr6+) removal from water. J. Colloid. Interface Sci. 2016, 468, 334–346. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Deng, G.; Wang, D.; Chen, M.; Zhou, Z.; Yang, H.; Yang, S.P. Renal-clearable zwitterionic conjugated hollow ultrasmall Fe3O4 nanoparticles for T-1-weighted MR imaging in vivo. J. Mater. Chem. B 2020, 8, 3087. [Google Scholar] [CrossRef]
- Belaïd, S.; Stanicki, D.; Elst, L.V.; Muller, R.N.; Laurent, S. Influence of experimental parameters on iron oxide nanoparticle properties synthesized by thermal decomposition: Size and nuclear magnetic resonance studies. Nanotechnology 2018, 29, 165603. [Google Scholar] [CrossRef]
- Salvador, M.; Gutiérrez, G.; Noriega, S.; Moyano, A.; Blanco-López, M.C.; Matos, M. Microemulsion Synthesis of Superparamagnetic Nanoparticles for Bioapplications. Int. J. Mol. Sci. 2021, 22, 427. [Google Scholar] [CrossRef]
- Morán, D.; Gutiérrez, G.; Mendoza, R.; Rayner, M.; Blanco-Lopez, C.; Matos, M. Synthesis of controlled-size starch nanoparticles and superparamagnetic starch nanocomposites by microemulsion method. Carbohydr. Polym. 2023, 299, 120223. [Google Scholar] [CrossRef]
- Xie, Y.; Vincent, A.H.; Chang, H.; Rinehart, J.D. Strengthening nanocomposite magnetism through microemulsion synthesis. Nano Res. 2018, 11, 4133–4141. [Google Scholar] [CrossRef]
- Zhao, S.; Bian, X.; Yang, C.; Yu, M.; Wang, T. Synthesis of FeCoB amorphous nanoparticles and application in ferrofluids. Appl. Surf. Sci. 2018, 435, 1314–1321. [Google Scholar] [CrossRef]
- Liu, L.; Pu, X.; Yin, G.; Chen, X.; Yin, J.; Wu, Y. Biomimetic mineralization of magnetic iron oxide nanoparticles mediated by bi-functional copolypeptides. Molecules 2019, 24, 1401. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zeng, B.; Zhou, R.; Li, X.; Zhang, G. One-pot synthesis of multiple stimuli-responsive magnetic nanomaterials based on the biomineralization of elastin-like polypeptides. ACS Omega 2021, 6, 27946–27954. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Zhao, H.; Zheng, X.; Sun, H.; Hu, L.; Zhu, L.; Shen, Y.; Luo, T.; Dai, H.; Wang, J. NMR studies of the interactions between AMB-1 Mms6 protein and magnetosome Fe3O4 nanoparticles. J. Mater. Chem. B 2017, 5, 2888. [Google Scholar] [CrossRef] [PubMed]
- Correa, T.; Presciliano, R.; Abreu, F. Why Does Not Nanotechnology Go Green? Bioprocess Simulation and Economics for Bacterial-Origin Magnetite Nanoparticles. Front. Microbiol. 2021, 12, 718232. [Google Scholar] [CrossRef]
- Li, S.; Wang, Z. Adsorption Performance of Reactive Red 2BF onto Magnetic NiFe2O4 Nanoparticles Prepared via the Coprecipitation Process. J. Nanosci. Nanotechnol. 2020, 20, 2832–2839. [Google Scholar] [CrossRef]
- He, X.; Zhong, W.; Du, Y. Controllable synthesis and performance of magnetic nanocomposites with core/shell structure. Acta Phys. Sin. 2018, 67, 227501. [Google Scholar]
- Liu, W.; Yin, S.; Hu, Y.; Deng, T.; Li, J. Microemulsion-Confined Biomineralization of PEGylated Ultrasmall Fe3O4 Nanocrystals for T2-T1 Switchable MRI of Tumors. Anal. Chem. 2021, 93, 14223–14230. [Google Scholar] [CrossRef] [PubMed]
- Ling, D.; Hyeon, T. Chemical design of biocompatible iron oxide nanoparticles for medical applications. Small 2013, 9, 1450–1466. [Google Scholar] [CrossRef]
- Radoń, A.; Drygała, A.; Hawełek, Ł.; Łukowiec, D. Structure and optical properties of Fe3O4 nanoparticles synthesized by co-precipitation method with different organic modifiers. Mater. Charact. 2017, 131, 148–156. [Google Scholar] [CrossRef]
- Abu-Dief, A.M.; Abdelbaky, M.S.M.; Martínez-Blanco, D.; Amghouz, Z.; García-Granda, S. Effect of chromium substitution on the structural and magnetic properties of nanocrystalline zinc ferrite. Mater. Chem. Phys. 2016, 174, 164–171. [Google Scholar] [CrossRef]
- Abu-Dief, A.M.; Nassar, I.F.; Elsayed, W.H. Magnetic NiFe2O4 nanoparticles: Efficient, heterogeneous and reusable catalyst for synthesis of acetylferrocene chalcones and their anti-tumour activity. Appl. Organometal. Chem. 2016, 30, 917–923. [Google Scholar] [CrossRef]
- Cai, D.; Ruan, C.; Qiu, J.; Wu, L.; Zhang, C.; Zhang, Y. Synthesis of Magnetic Co/C Nanocomposite and Its Adsorption Performance for Congo Red in Aqueous Systems. Univ. Chem. 2020, 35, 212–220. [Google Scholar]
- Lian, L.; Zhang, X.; Hao, J.; Lv, J.; Wang, X.; Zhu, B.; Lou, D. Magnetic solid-phase extraction of fluoroquinolones from water samples using titanium-based metal-organic framework functionalized magnetic microspheres. J. Chromatogr. A 2018, 1579, 1–8. [Google Scholar] [CrossRef]
- Yao, X.; Yue, Y.; Tang, F. Application of Ionic Liquids in Separation and Analysis of Natural Products: A Review. Chem. Ind. For. Prod. 2013, 33, 143–148. (In Chinese) [Google Scholar]
- He, Y.; Wang, Y.; Zhao, W.; Gao, R.; Lv, D. Preparation of magnetic nano-composite adsorbents and their application in water treatment. Ind. Water Treat. 2021, 41, 1–6. (In Chinese) [Google Scholar]
- Lu, D.; Qin, M.; Liu, C.; Deng, J.; Shi, G.; Zhou, T. Ionic Liquid-Functionalized Magnetic Metal-Organic Framework Nanocomposites for Efficient Extraction and Sensitive Detection of Fluoroquinolone Antibiotics in Environmental Water. ACS Appl. Mater. Interfaces 2021, 13, 5357–5367. [Google Scholar] [CrossRef]
- Ping, W.; Shi, Y.; Yang, J.; Yang, Q.; Xie, C. HPLC Determination of Bisphenol A in Beverages with Its Separationand Enrichment by Solid Phase Extraction Using Room Temperature-Ionic Liquid Loaded Magnetic Nano-composite of Cyclodextrin Polymer/Fe3O4. Phys. Test. Chem. Anal. 2019, 55, 1091–1094. (In Chinese) [Google Scholar]
- Fan, W.; Yang, D.; Ding, N.; Chen, P.; Wang, L.; Tao, G.; Zheng, F.; Ji, S. Application of core-satellite polydopamine-coated Fe3O4 nanoparticles-hollow porous molecularly imprinted polymer combined with HPLC-MS/MS for the quantification of macrolide antibiotics. Anal. Methods 2021, 13, 1412. [Google Scholar] [CrossRef]
- Chen, F.; Dong, Y.; Zhao, Y. Synthesis and characterization of photo-responsive magnetic molecularly imprinted microspheres for the detection of sulfonamides in aqueous solution. J. Sep. Sci. 2016, 39, 4866–4875. [Google Scholar] [CrossRef]
- Cao, Y.; Li, J.; Luo, L.; Li, P.; Liu, X. Preparation of Magnetic Molecularly Imprinted Nanoparticles for Enrichment and Separation of 6-Benzylaminopurine. Food Res. Dev. 2021, 42, 87–93. (In Chinese) [Google Scholar]
- Kaabipour, M.; Khodadoust, S.; Zeraatpisheh, F. Preparation of magnetic molecularly imprinted polymer for dispersive solid-phase extraction of valsartan and its determination by high-performance liquid chromatography: Box-Behnken design. J. Sep. Sci. 2020, 43, 912–919. [Google Scholar] [CrossRef] [PubMed]
- Serantes, D.; Chantrell, R.; Gavilán, H.; Morales, M.D.P.; Chubykalo-Fesenko, O.; Baldomir, D.; Satoh, A. Anisotropic magnetic nanoparticles for biomedicine: Bridging frequency separated AC-field controlled domains of actuation. Phys. Chem. Chem. Phys. 2018, 20, 30445. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Hu, L.; Niu, C.; Huang, D.; Zeng, G. A magnetic separation fluorescent aptasensor for highly sensitive detection of bisphenol A. Sens. Actuators B Chem. 2018, 266, 805–811. [Google Scholar] [CrossRef]
- Wu, Y.; Bai, H.; Zhou, Q.; Li, S.; Tong, Y.; Guo, J.; Zhou, B.; Li, Z.; Zhan, Y.; Liu, M.; et al. Preparation of polyamidoamine dendrimer modified magnetic nanoparticles and its application for reliable measurement of Sudan red contaminants in natural waters at parts-per-billion levels. Front. Chem. 2021, 9, 708995. [Google Scholar] [CrossRef]
- Sun, L.; Sun, X.; Du, X.; Yue, Y.; Chen, L.; Xu, H.; Zeng, Q.; Wang, H.; Ding, L. Determination of sulfonamides in soil samples based on alumina-coated magnetite nanoparticles as adsorbents. Anal. Chim. Acta 2010, 665, 185–192. [Google Scholar] [CrossRef]
- Kumar, A.; Joshi, H.; Kumar, A. Arsenate Removal from the Groundwater Employing Maghemite Nanoparticles. Water 2022, 14, 3617. [Google Scholar] [CrossRef]
- Jagirani, M.; Uzcan, F.; Soylak, M. A selective and sensitive procedure for magnetic solid-phase microextraction of lead(II) on magnetic cellulose nanoparticles from environmental samples prior to its flame atomic absorption spectrometric detection. J. Iran. Chem. Soc. 2021, 18, 1005–1013. [Google Scholar] [CrossRef]
- Wu, H.; Meng, L. Liquid chromatography-UV determination of heavy metal ions in environmental samples using dispersive liquid-liquid microextraction coupled with magnetic nanoparticles. Appl. Ecol. Environ. Res. 2018, 17, 1571–1584. [Google Scholar] [CrossRef]
- Kouhestani, H.; Ebrahimi, P. Extraction of carboxin and bensulfuron-methyl using cysteine-functionalized chitosan-coated magnetic nanoparticles and response surface methodology. Sep. Sci. Technol. 2021, 56, 2385–2397. [Google Scholar] [CrossRef]
- Ragab, G.H.; Bahgat, E.A. Magnetic iron oxide nanoparticles for HPLC determination of valsartan in aqueous environmental samples, biological samples and tablet dosage forms. Int. J. Environ. Anal. Chem. 2021, 101, 2612–2628. [Google Scholar] [CrossRef]
- Tian, Y.; Xu, Z.; Yang, Y.; Wang, D.; Liu, Z.; Si, X. Magnetic solid phase extraction based on Fe3O4@SiO2@CTS nano adsorbent for the sensitive detection of trace polychlorinated biphenyls in environmental water samples. Microchem. J. 2022, 172, 106947. [Google Scholar] [CrossRef]
- García-Merino, B.; Bringas, E.; Ortiz, I. Robust system for the regenerative capture of aqueous pollutants with continuously synthesized and functionalized magnetic nanoparticles. J. Environ. Chem. Eng. 2022, 10, 108417. [Google Scholar] [CrossRef]
- Yağci, Ö.; Akkaya, E.; Bakirdere, S. Nano-sized magnetic Ni particles based dispersive solid-phase extraction of trace Cd before the determination by flame atomic absorption spectrometry with slotted quartz tube: A new, accurate, and sensitive quantification method. Env. Monit. Assess. 2020, 192, 583. [Google Scholar] [CrossRef] [PubMed]
- Leal, M.; Alonso, E.V.; Guerrero, M.M.L.; Cordero, M.T.S.; Pavón, J.M.C.; Torres, A.G.D. Speciation analysis of inorganic arsenic by magnetic solid phase extraction on-line with inductively coupled mass spectrometry determination. Talanta 2018, 184, 251–259. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Su, S.; Chen, B.; Hu, B. Simultaneous speciation of inorganic selenium and tellurium in environmental water samples by polyaniline functionalized magnetic solid phase extraction coupled with ICP-MS detection. Talanta 2020, 207, 120314. [Google Scholar] [CrossRef]
- Cao, X.; Liu, G.; She, Y.; Jiang, Z.; Jin, F.; Jin, M.; Du, P.; Zhao, F.; Zhang, Y.; Wang, J. Preparation of magnetic metal organic framework composites for the extraction of neonicotinoid insecticides from environmental water samples. RSC Adv. 2016, 6, 113144–113151. [Google Scholar] [CrossRef]
- Turiel, E.; Díaz-Álvarez, M.; Martín-Esteban, A. Surface modified-magnetic nanoparticles by molecular imprinting for the dispersive solid-phase extraction of triazines from environmental waters. J. Sep. Sci. 2020, 43, 3304–3314. [Google Scholar] [CrossRef] [PubMed]
- Cui, Z.; Xiang, L.; Tang, J. Facile one-pot synthesis of magnetic molecular imprinting polymers as a novel adsorbent for the enrichment of imidacloprid based on a magnetic dispersive micro-solid-phase extraction in water samples. Chem. Pap. 2021, 75, 3787–3795. [Google Scholar] [CrossRef]
- Wu, Y.; Chen, C.; Zhou, Q.; Li, Q.; Yuan, Y.; Tong, Y.; Wang, H.; Zhou, X.; Sun, Y.; Sheng, X. Polyamidoamine dendrimer decorated nanoparticles as an adsorbent for magnetic solid-phase extraction of tetrabromobisphenol A and 4-nonylphenol from environmental water samples. J. Colloid. Interface Sci. 2019, 539, 361–369. [Google Scholar] [CrossRef]
- Zheng, Y.; Liu, Y.; Guo, X.; Chen, Z.; Zhang, W.; Wang, Y.; Tang, X.; Zhang, Y.; Zhao, Y. Sulfur-doped g-C3N4/rGO porous nanosheets for highly efficient photocatalytic degradation of refractory contaminants. J. Mater. Sci. Technol. 2020, 41, 117–126. [Google Scholar] [CrossRef]
- Singh Rathore, B.; Pal Singh Chauhan, N.; Panneerselvam, P.; Jadoun, S.; Barani, M.; Ameta, S.C.; Ameta, R. Synthesis and Characterization of Ch-PANI-Fe2O3 Nanocomposite and Its Water Remediation Applications. Water 2022, 14, 3615. [Google Scholar] [CrossRef]
- Khan, S.B.; Irfan, S.; Lam, S.S.; Sun, X.; Chen, S. 3D printed nanofiltration membrane technology for waste water distillation. J. Water Process Eng. 2022, 49, 102958. [Google Scholar] [CrossRef]
- Ali, N.; Riead, M.M.H.; Bilal, M.; Yang, Y.; Khan, A.; Ali, F.; Karim, S.; Zhou, G.; Wenjie, Y.; Sher, F.; et al. Adsorptive remediation of environmental pollutants using magnetic hybrid materials as platform adsorbents. Chemosphere 2021, 284, 131279. [Google Scholar] [CrossRef]
- Ibrahim, W.A.W.; Nodeh, H.R.; Aboul-Enein, H.Y.; Sanagi, M.M. Magnetic solid-phase extraction based on modified ferum oxides for enrichment, preconcentration, and isolation of pesticides and selected pollutants. Crit. Rev. Anal. Chem. 2015, 45, 270–287. [Google Scholar] [CrossRef]
- Zhao, B.; He, M.; Chen, B.; Hu, B. Fe3O4 nanoparticles coated with double imprinted polymers for magnetic solid phase extraction of lead(II). Microchim. Acta 2019, 186, 775. [Google Scholar] [CrossRef]
- Kong, J.; Coolahan, K.; Mugweru, A. Manganese based magnetic nanoparticles for heavy metal detection and environmental remediation. Anal. Methods 2013, 5, 5128. [Google Scholar] [CrossRef]
- Maleki, B.; Baghayeri, M.; Ghanei-Motlagh, M.; Zonoz, F.M.; Amiri, A.; Hajizadeh, F.; Hosseinifar, A.; Esmaeilnezhad, E. Polyamidoamine dendrimer functionalized iron oxide nanoparticles for simultaneous electrochemical detection of Pb2+ and Cd2+ ions in environmental waters. Measurement 2019, 140, 81–88. [Google Scholar] [CrossRef]
- Wu, H.; Meng, L.; Song, W. Glutathione-stabilized silver nanoparticles and magnetic nanoparticles combination for determination of lead and cadmium in environmental waters. Sci. Adv. Mater. 2019, 11, 1133–1139. [Google Scholar] [CrossRef]
- Wang, X.; Yang, J.; Zhou, L.; Song, G.; Lu, F.; You, L.; Li, J. Rapid and ultrasensitive surface enhanced Raman scattering detection of hexavalent chromium using magnetic Fe3O4/ZrO2/Ag composite microsphere substrates. Colloids Surf. A Physicochem. Eng. Asp. 2021, 610, 125414. [Google Scholar] [CrossRef]
- Faiz, F.; Qiao, J.; Lian, H.; Mao, L.; Cui, X. A combination approach using two functionalized magnetic nanoparticles for speciation analysis of inorganic arsenic. Talanta 2022, 237, 122939. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Chen, S.; Ye, F.; Su, L.; Zhang, C.; Shen, S.; Zhao, S. Preparation of magnetic core–shell nanoflower Fe3O4@MnO2 as reusable oxidase mimetics for colorimetric detection of phenol. Anal. Methods 2015, 7, 1300. [Google Scholar] [CrossRef]
- Christus, A.A.B.; Panneerselvam, P.; Ravikumar, A.; Morad, N.; Sivanesan, S. Colorimetric determination of Hg (II) sensor based on magnetic nanocomposite (Fe3O4@ZIF-67) acting as peroxidase mimics. J. Photochem. Photobiol. A Chem. 2018, 364, 715–724. [Google Scholar] [CrossRef]
- Oguz, M.; Bhatti, A.A.; Yilmaz, M. Surface coating of magnetite nanoparticles with fluorescence derivative for the detection of mercury in water environments. Mater. Lett. 2020, 267, 127548. [Google Scholar] [CrossRef]
- Liu, Y.; Cai, Z.; Sheng, L.; Ma, M.; Wang, X. A magnetic relaxation switching and visual dual-mode sensor for selective detection of Hg2+ based on aptamers modified Au@Fe3O4 nanoparticles. J. Hazard. Mater. 2020, 388, 121728. [Google Scholar] [CrossRef]
- Li, X.; Wen, Q.; Chen, J.; Sun, W.; Zheng, Y.; Long, C.; Wang, Q. Lanthanide Molecular Species Generated Fe3O4@SiO2-TbDPA Nanosphere for the Efficient Determination of Nitrite. Molecules 2022, 27, 4431. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Zheng, X.; Li, H.; Zhao, Z. Human health risk assessment and early warning of heavy metal pollution in soil of a coal chemical Plant in Northwest China. Soil. Sediment. Contam. Int. J. 2020, 5, 481–502. [Google Scholar] [CrossRef]
- Li, Q.; Dai, L.; Wang, M.; Su, G.; Wang, T.; Zhao, X.; Liu, X.; Xu, Y.; Meng, J.; Shi, B. Distribution, influence factors, and biotoxicity of environmentally persistent free radical in soil at a typical coking plant. Sci. Total Environ. 2022, 835, 155493. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, X.; Si, X.; Yang, R.; Zhou, J.; Quan, X. Environmentally persistent free radical generation on contaminated soil and their potential biotoxicity to luminous bacteria. Sci. Total Environ. 2019, 687, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.; Kim, J.-H.; Kim, S.-M.; Park, C.W.; Yoon, I.-H.; Yang, H.-M.; Sihn, Y. Enhanced selective separation of fine particles from Cs-contaminated soil using magnetic nanoparticles. J. Soils Sediments 2021, 21, 346–354. [Google Scholar] [CrossRef]
- Kasa, N.A.; Sel, S.; Özkan, B.Ç.; Bakırdere, S. Determination of palladium in soil samples by slotted quartz tube-flame atomic absorption spectrophotometry after vortex-assisted ligandless preconcentration with magnetic nanoparticle–based dispersive solid-phase microextraction. Env. Monit. Assess. 2019, 191, 692. [Google Scholar] [CrossRef]
- Patiño-Ropero, M.J.; Díaz-Álvarez, M.; Martín-Esteban, A. Molecularly imprinted core-shell magnetic nanoparticles for selective extraction of triazines in soils. J. Mol. Recognit. 2017, 30, e2593. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.T.; Yoon, S.A.; Ahn, J.; Choi, Y.; Lee, M.H.; Jung, J.H.; Park, J. Synthesis of fluorescent naphthalimide-functionalized Fe3O4 nanoparticles and their application for the selective detection of Zn2+ present in contaminated soil. Sens. Actuators B Chem. 2017, 243, 1034–1041. [Google Scholar] [CrossRef]
- Yang, L.; Su, P.; Chen, X.; Zhang, R.; Yang, Y. Microwave-assisted synthesis of poly (ionic liquid)-coated magnetic nanoparticles for the extraction of sulfonylurea herbicides from soil for HPLC. Anal. Methods 2015, 7, 3246–3252. [Google Scholar] [CrossRef]
- Pang, J.; Song, X.; Huang, X.; Yuan, D. Porous monolith-based magnetism-reinforced in-tube solid phase microextraction of sulfonylurea herbicides in water and soil samples. J. Chromatogr. A 2020, 1613, 460672. [Google Scholar] [CrossRef]
- Pang, J.; Chen, H.; Huang, X. Magnetism-assisted in-tube solid phase microextraction for the on-line chromium speciation in environmental water and soil samples. Microchem. J. 2021, 164, 105956. [Google Scholar] [CrossRef]
- Li, W.; Wang, R.; Chen, Z. Metal-organic framework-1210(zirconium/cuprum) modified magnetic nanoparticles for solid phase extraction of benzophenones in soil samples. J. Chromatogr. A 2019, 1607, 460403. [Google Scholar] [CrossRef]
- Hubetska, T.S.; Kobylinska, N.G.; Menendez, J.R.G. Application of hydrophobic magnetic nanoparticles as cleanup adsorbents for pesticide residue analysis in fruit, vegetable, and various soil samples. J. Agric. Food Chem. 2020, 68, 13550–13561. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, S.M.; Yoon, I.H.; Kim, I. Application of polyethylenimine-coated magnetic nanocomposites for the selective separation of Cs-enriched clay particles from radioactive soil. RSC Adv. 2020, 10, 21822. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; Ma, A.; Wang, T.; Lin, J.; Wang, H.; Du, B.; Zhuang, X.; Zhuang, G. Detection of bioavailable cadmium, lead, and arsenic in polluted soil by tailored multiple Escherichia coli whole-cell sensor set. Anal. Bioanal. Chem. 2015, 407, 6865–6871. [Google Scholar] [CrossRef] [PubMed]
- Fedotov, P.S.; Kördel, W.; Miró, M.; Peijnenburg, W.J.G.M.; Wennrich, R.; Huang, P. Extraction and fractionation methods for exposure assessment of trace metals, metalloids, and hazardous organic compounds in terrestrial environments. Crit. Rev. Environ. Sci. Technol. 2012, 42, 1117–1171. [Google Scholar] [CrossRef]
- Meng, D.; Zhao, N.; Ma, M.; Fang, L.; Gu, Y.; Yao, J.; Liu, J.; Liu, W. Application of a mobile laser-induced breakdown spectroscopy system to detect heavy metal elements in soil. Appl. Opt. 2017, 56, 5204–5210. [Google Scholar] [CrossRef]
- Jia, J.; Li, H.; Zong, S.; Jiang, B.; Li, G.; Ejenavi, O.; Zhu, J.; Zhang, D. Magnet bioreporter device for ecological toxicity assessment on heavy metal contamination of coal cinder sites. Sens. Actuators B Chem. 2016, 222, 290–299. [Google Scholar] [CrossRef]
- Zhang, K.; Bao, K.; Xiong, Z.; Li, T. Didactic experimental design for characterization of biotoxicity in contaminated soils with MNPs-P. phosphoreum biosensor. Exp. Technol. Manag. 2022, 39, 209–213. (In Chinese) [Google Scholar]
- Zhang, K.; Liu, M.; Song, X.; Wang, D. Application of Luminescent Bacteria Bioassay in the Detection of Pollutants in Soil. Sustainability 2023, 15, 7351. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).