Continuous Flow Photocatalytic Degradation of Phenol Using Palladium@Mesoporous TiO2 Core@Shell Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of 2%Pd@TiO2 Nanoparticles
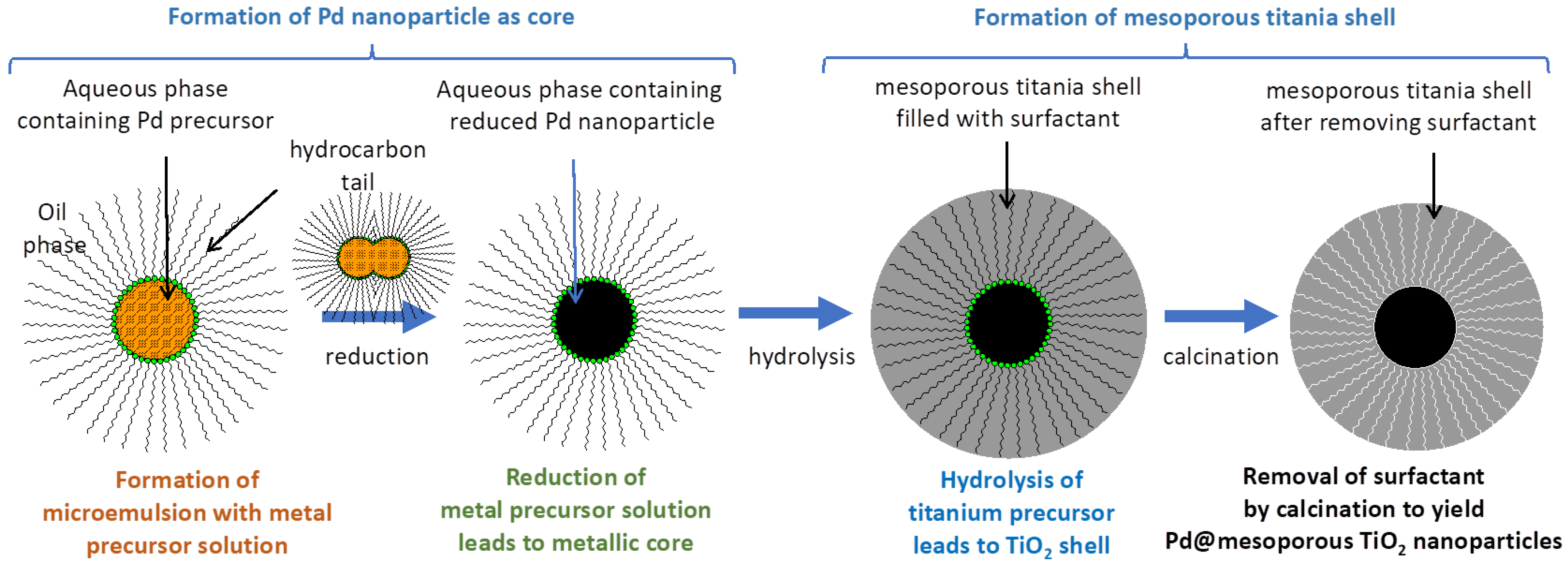
2.2.1. Microemulsion Preparation
2.2.2. Preparation of 2%Pd@mTiO2 Core@shell Nanoparticles
2.2.3. Preparation of 0.5%Pd/TiO2 P25 Catalyst
2.3. Characterisation of 2%Pd@TiO2 Nanoparticles
2.4. Evaluation of Photocatalytic Activity
3. Results and Discussion
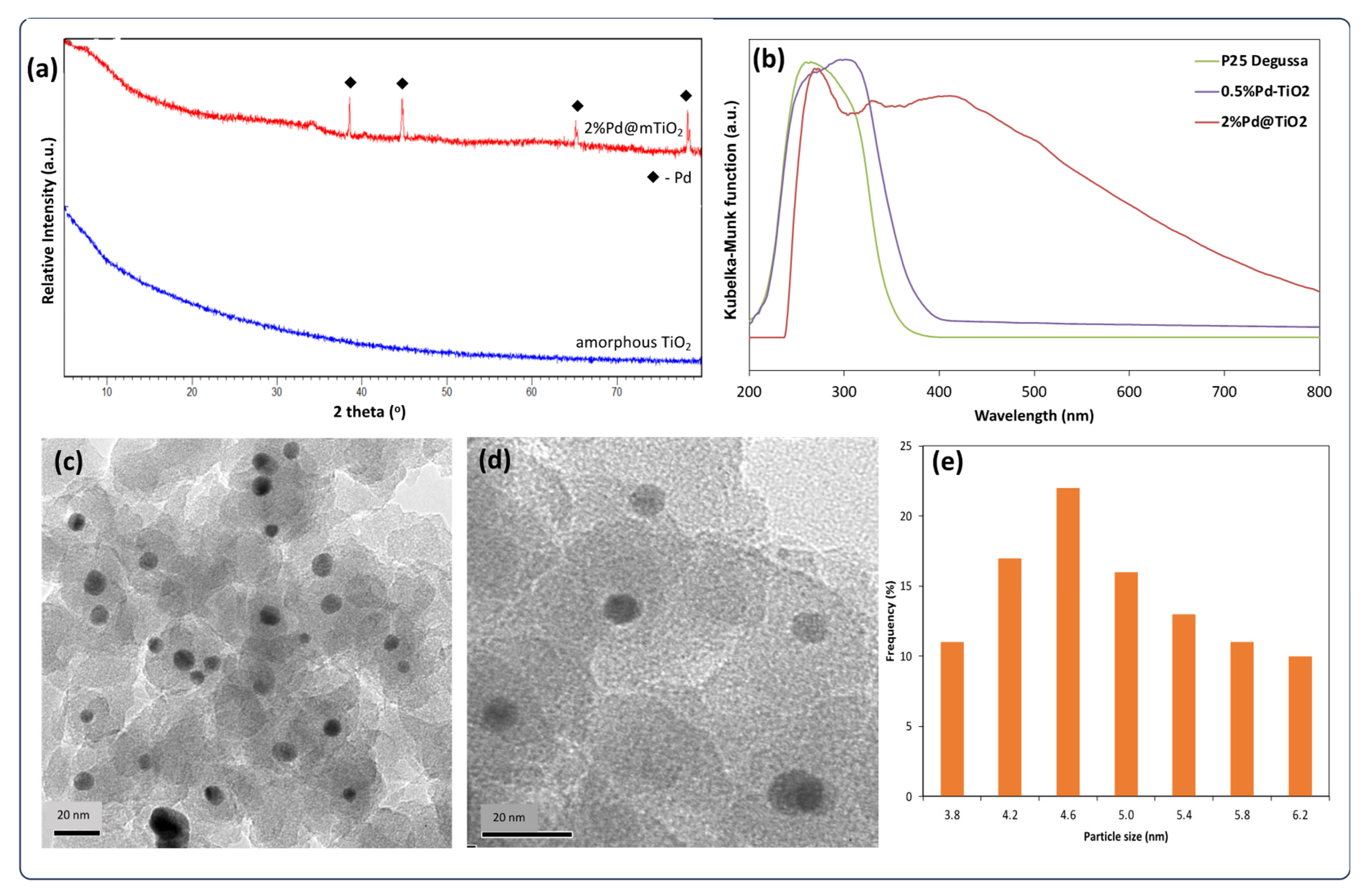
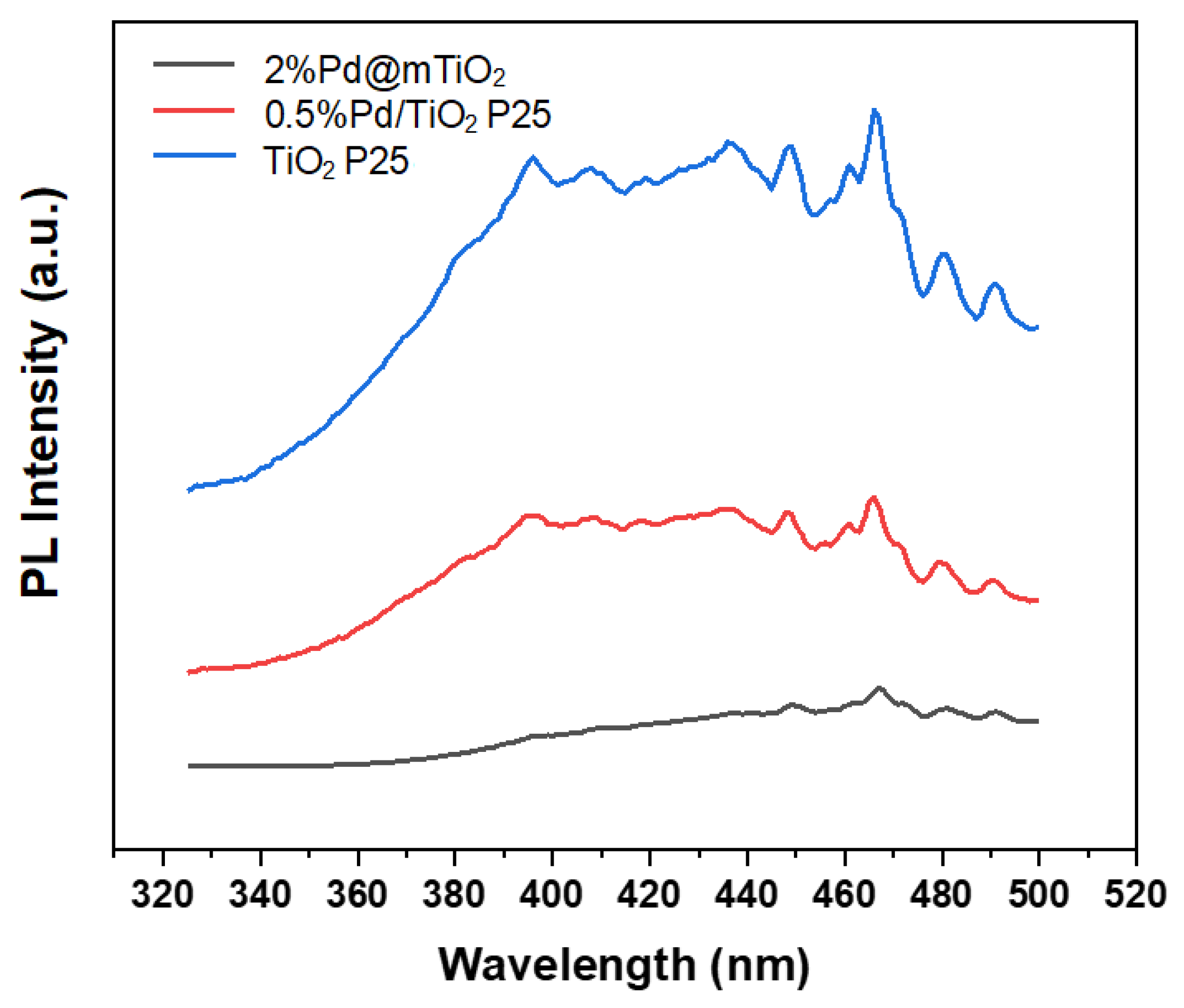
3.1. Catalyst Characterisation
3.2. Photocatalytic Degradation of Phenol Using Batch Photoreactor
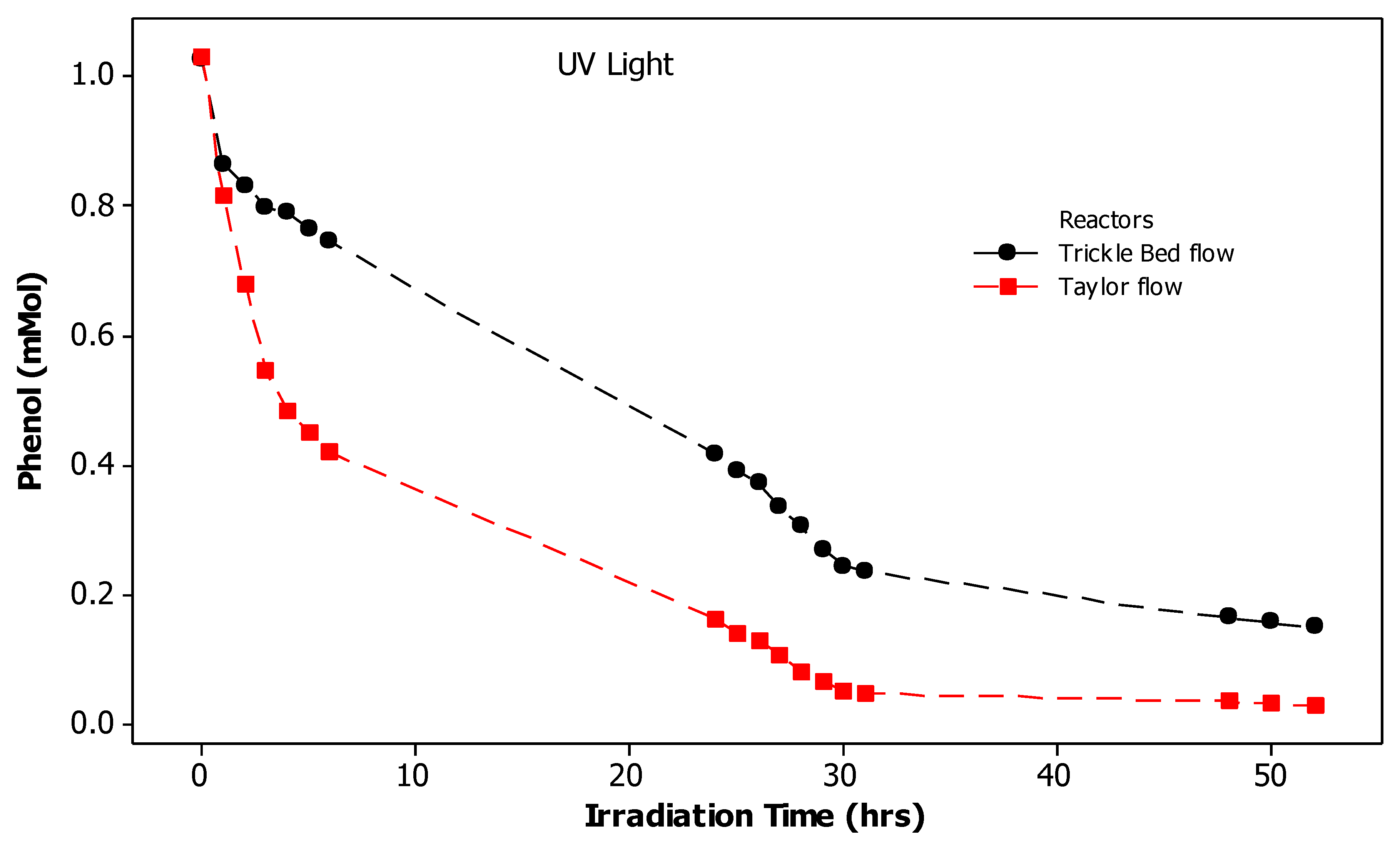
3.3. Photocatalytic Degradation of Phenol Using Continuous Flow Trickle Bed and Taylor Flow Photoreactors
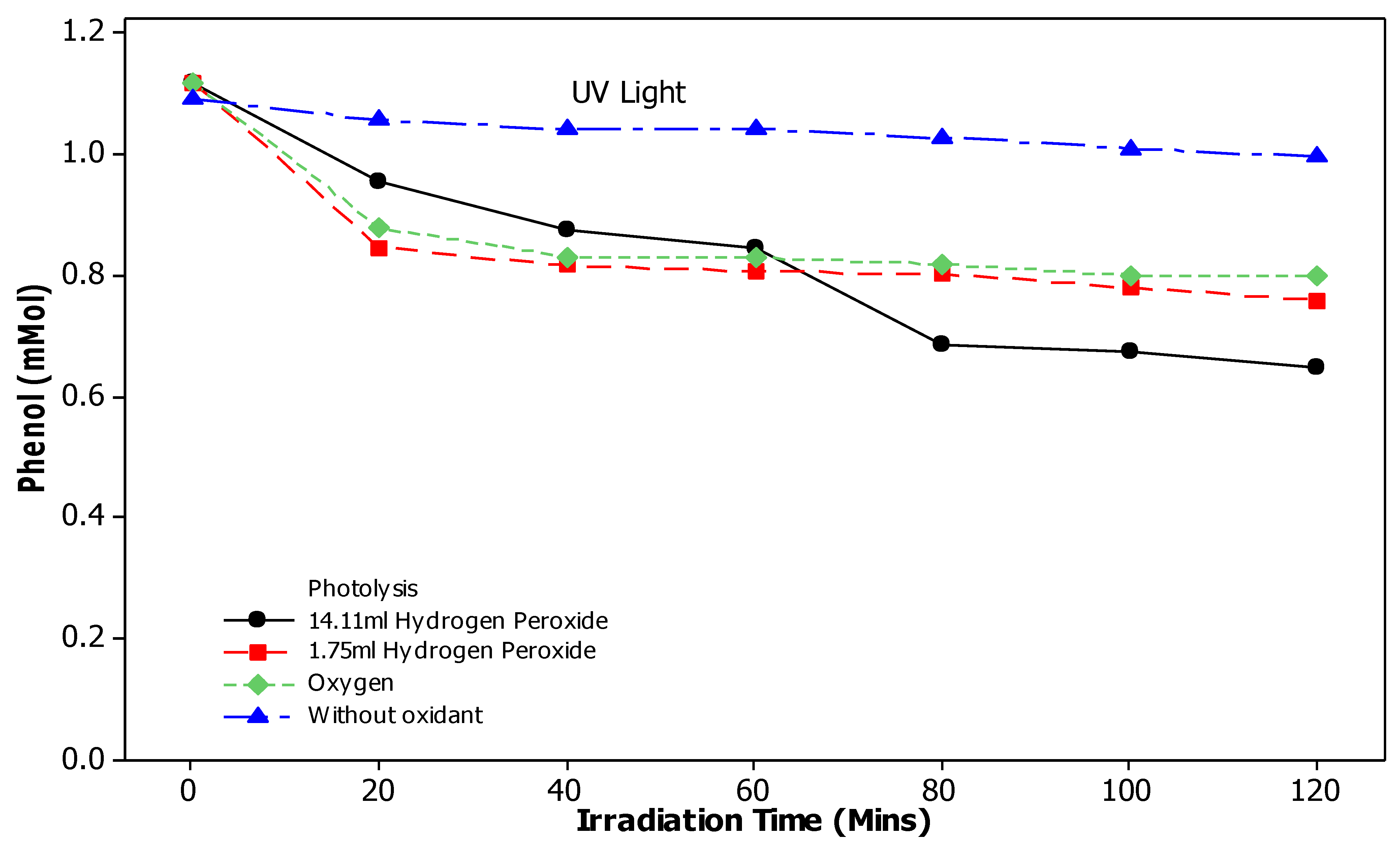
3.4. Effect of Photolysis
3.5. Reusability of Catalyst
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Beltran, F.J.; Rivas, F.J.; Gimeno, O. Comparison between photocatalytic ozonation and other oxidation processes for the removal of phenols from water. J. Chem. Technol. Biotechnol. 2005, 80, 973–984. [Google Scholar] [CrossRef]
- Adan, C.; Coronado, J.M.; Bellod, R.; Soria, J.; Yamaoka, H. Photochemical and photocatalytic degradation of salicylic acid with hydrogen peroxide over TiO2/SiO2 fibres. Appl. Catal. A Gen. 2006, 303, 199–206. [Google Scholar] [CrossRef]
- Su, R.; Tiruvalam, R.; He, Q.; Dimitratos, N.; Kesavan, L.; Hammond, C.; Lopez-Sanchez, J.A.; Bechstein, R.; Kiely, C.J.; Hutchings, G.J.; et al. Promotion of phenol photodecomposition over TiO2 using Au, Pd and Au-Pd Nanoparticles. ACS Nano 2012, 6, 6284–6292. [Google Scholar] [CrossRef] [PubMed]
- Dionysiou, D.D.; Khodadoust, A.P.; Kern, A.M.; Suidan, M.T.; Baudin, I.; Laine, J.-M. Continuous-mode photocatalytic degradation of chlorinated phenols and pesticides in water using a bench-scale TiO2 rotating disk reactor. Appl. Catal. B Environ. 2000, 24, 139–155. [Google Scholar] [CrossRef]
- Pérez, M.H.; Peñuela, G.; Maldonado, M.I.; Malato, O.; Pilar, F.-I.; Oller, I.; Gernjak, W.; Malato, S. Degradation of pesticides in water using solar advanced oxidation processes. Appl. Catal. B Environ. 2006, 64, 272–281. [Google Scholar] [CrossRef]
- Ghumro, S.S.; Lal, B.; Pirzada, T. Visible-Light-Driven Carbon-Doped TiO2-Based Nanocatalysts for Enhanced Activity toward Microbes and Removal of Dye. ACS Omega 2022, 7, 4333–4341. [Google Scholar] [CrossRef]
- Park, B.G. Photocatalytic activity of TiO2-doped Fe, Ag, and Ni with N under visible light irradiation. Gels 2022, 8, 14. [Google Scholar] [CrossRef]
- Sirivallop, A.; Areerob, T.; Chiarakorn, S. Enhanced Visible Light Photocatalytic Activity of N and Ag Doped and Co-Doped TiO2 Synthesized by Using an In-Situ Solvothermal Method for Gas Phase Ammonia Removal. Catalysts 2020, 10, 251. [Google Scholar] [CrossRef]
- Al-Hamoud, K.; Shaik, M.R.; Khan, M.; Alkhathlan, H.Z.; Adil, S.F.; Kuniyil, M.; Assal, M.E.; Al-Warthan, A.; Siddiqui, M.R.H.; Tahir, M.N.; et al. Pulicaria undulata Extract-Mediated Eco-Friendly Preparation of TiO2 Nanoparticles for Photocatalytic Degradation of Methylene Blue and Methyl Orange. ACS Omega 2022, 7, 4812–4820. [Google Scholar] [CrossRef]
- Shubha, J.P.; Adil, S.F.; Khan, M.; Hatshan, M.R.; Khan, A. Facile Fabrication of a ZnO/Eu2O3/NiO-Based Ternary Heterostructure Nanophotocatalyst and Its Application for the Degradation of Methylene Blue. ACS Omega 2021, 5, 3866–3874. [Google Scholar] [CrossRef]
- Gerven, T.V.; Stankiewicz, A. Structure, energy, synergy, time-The fundamentals of process intensification. Ind. Eng. Chem. Res. 2009, 48, 2465–2474. [Google Scholar] [CrossRef]
- Cassano, A.E.; Martin, C.A.; Brandi, R.J.; Alfano, O.M. Photoreactor analysis and design: Fundamentals and applications. Ind. Eng. Chem. Res. 1995, 34, 2155–2201. [Google Scholar] [CrossRef]
- Yue, P.L. Modelling, scale-up and design of multiphasic photoreactors. In Photocatalytic Purification and Treatment of Water and Air; Ollis, D.F., Al-Ekabi, H., Eds.; Elsevier Science Publishers BV: Amsterdam, The Netherlands, 1993; pp. 495–510. [Google Scholar]
- Sclafani, A.; Brucato, A.; Rizzuti, L. Mass transfer limitations in a packed bed photoreactor used for phenol removal. In Photocatalytic Purification and Treatment of Water and Air; Ollis, D.F., Al-Ekabi, H., Eds.; Elsevier Science Publishers BV: Amsterdam, The Netherlands, 1993; pp. 533–545. [Google Scholar]
- Sczechowski, J.G.; Koval, C.A.; Noble, R.D. A Taylor vortex reactor for heterogeneous photocatalysis. Chem. Eng. Sci. 1995, 50, 3163–3173. [Google Scholar] [CrossRef]
- Haarstrick, A.; Kut, O.M.; Heinzle, E. TiO2 assisted degradation of environmentally relevant organic compounds in wastewater using a novel fluidized bed photoreactor. Environ. Sci. Technol. 1996, 30, 817–824. [Google Scholar] [CrossRef]
- Ollis, D.F.; Pelizzetti, E.; Serpone, N. Photocatalysis: Fundamentals and Applications; Wiley: New York, NY, USA, 1989. [Google Scholar]
- Peill, N.J.; Hoffmann, M.R. Development and optimization of a TiO2-coated fiber--optic cable reactor: Photocatalytic degradation of 4-chlorophenol. Environ. Sci. Technol. 1995, 29, 2974–2981. [Google Scholar] [CrossRef]
- Ray, A.K.; Beenackers, A.A.C.M. Novel swirl flow reactor for kinetic studies of semiconductor photocatalysis. AIChE J. 1997, 43, 2571–2578. [Google Scholar] [CrossRef]
- Amoli-Diva, M.; Anvari, A.; Sadighi-Bonabi, R. Synthesis of magneto-plasmonic Au-Ag NPs-decorated TiO2-modified Fe3O4 nanocomposite with enhanced laser/solar-driven photocatalytic activity for degradation of dye pollutant in textile wastewater. Ceram. Int. 2019, 45, 17837–17846. [Google Scholar] [CrossRef]
- Mondal, K.; Sharma, A. Recent advances in the synthesis and application of photocatalytic metal-metal oxide core-shell nanoparticles for environmental remediation and their recycling process. RSC Adv. 2016, 6, 83589–83612. [Google Scholar] [CrossRef]
- Lu, F.F.; Dong, A.Q.; Ding, G.J.; Xu, K.; Li, J.M.; You, L.J. Magnetic porous polymer composite for high performance adsorption of acid red 18 based on melamine resin and chitosan. J. Mol. Liq. 2019, 294, 111515. [Google Scholar] [CrossRef]
- Du, D.; Shi, W.; Wang, L.Z.; Zhang, J.L. Yolk-shell structured Fe3O4@void@TiO2 as a photo-Fenton-like catalyst for the extremely efficient elimination of tetracycline. Appl. Catal. B 2017, 200, 484–492. [Google Scholar] [CrossRef]
- Min, Y.; Song, G.; Zhou, L.; Wang, X.; Liu, P.; Li, J. Silver@mesoporous Anatase TiO2 Core-Shell Nanoparticles and Their Application in Photocatalysis and SERS Sensing. Coatings 2022, 12, 64. [Google Scholar] [CrossRef]
- Bayles, A.; Tian, S.; Zhou, J.; Yuan, L.; Yuan, Y.; Jacobson, C.R.; Farr, C.; Zhang, M.; Swearer, D.F.; Solti, D.; et al. Al@TiO2 Core–Shell Nanoparticles for Plasmonic Photocatalysis. ACS Nano 2022, 16, 5839–5850. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, G.; Manyar, H. Advanced Oxidation Processes for Wastewater Treatment, Advanced Materials and Technologies for Wastewater Treatment; CRC Press: Boca Raton, FL, USA, 2021; pp. 153–164. [Google Scholar]
- Morrison, G.; Bannon, R.; Wharry, S.; Moody, T.S.; Mase, N.; Hattori, M.; Manyar, H.; Smyth, M. Continuous flow photooxidation of alkyl benzenes using fine bubbles for mass transfer enhancement. Tetrahed. Lett. 2022, 90, 153613. [Google Scholar] [CrossRef]
- Yadav, G.D.; Mewada, R.K.; Wagh, D.W.; Manyar, H. Advances and future trends in selective oxidation catalysis: A critical review. Catal. Sci. Technol. 2022, 12, 7245. [Google Scholar] [CrossRef]
- Ethiraj, J.; Wagh, D.; Manyar, H. Advances in upgrading biomass to biofuels and oxygenated fuel additives using metal oxide catalysts. Energy Fuels 2022, 36, 1189–1204. [Google Scholar] [CrossRef]
- Skillen, N.; Ralphs, K.; Craig, D.; McCalmont, S.; Muzio, A.F.V.; O’Rourke, C.; Manyar, H.; Robertson, P.K.J. Photocatalytic reforming of glycerol to H2 in a thin film Pt-TiO2 recirculating photoreactor. J. Chem. Technol. Biotech. 2020, 95, 2619–2627. [Google Scholar]
- Yilleng, M.T.; Gimba, E.C.; Ndukwe, G.I.; Bugaje, I.M.; Rooney, D.W.; Manyar, H.G. Batch to continuous photocatalytic degradation of phenol using TiO2 and Au-Pd nanoparticles supported on TiO2. J. Environ. Chem. Eng. 2018, 6, 6382–6389. [Google Scholar] [CrossRef]
- Yadav, G.D.; Manyar, H.G. Synthesis of a novel redox material UDCaT-3: An efficient and versatile catalyst for selective oxidation, hydroxylation and hydrogenation reactions. Adv. Syn. Catal. 2008, 350, 2286–2294. [Google Scholar] [CrossRef]
- Manyar, H.G.; Chaure, G.S.; Kumar, A. The green catalytic oxidation of alcohols in water by using highly efficient manganosilicate molecular sieves. Green Chem. 2006, 8, 344–348. [Google Scholar] [CrossRef]
- Jakubek, T.; Hudy, C.; Gryboś, J.; Manyar, H.; Kotarba, A. Thermal transformation of birnessite (OL) towards highly active cryptomelane (OMS-2) catalyst for soot oxidation. Catal. Lett. 2019, 149, 2218–2225. [Google Scholar] [CrossRef]
- Jakubek, T.; Ralphs, K.; Kotarba, A.; Manyar, H. Nanostructured potassium-manganese oxides decorated with Pd nanoparticles as efficient catalysts for low-temperature soot oxidation. Catal. Lett. 2019, 149, 100–106. [Google Scholar] [CrossRef]
- McManus, I.J.; Daly, H.; Manyar, H.G.; Taylor, S.F.R.; Thompson, J.M.; Hardacre, C. Selective hydrogenation of halogenated arenes using porous manganese oxide (OMS-2) and platinum supported OMS-2 catalysts. Faraday Discuss. 2016, 188, 451–466. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, R.; Ralphs, K.; Grolleau, M.; Manyar, H.; Artioli, N. Doping Manganese Oxides with Ceria and Ceria Zirconia Using a One-Pot Sol–Gel Method for Low Temperature Diesel Oxidation Catalysts. Top. Catal. 2020, 63, 351–362. [Google Scholar] [CrossRef]
- Coney, C.; Hardacre, C.; Morgan, K.; Artioli, N.; York, A.P.E.; Millington, P.; Kolpin, A.; Goguet, A. Investigation of the oxygen storage capacity behaviour of three way catalysts using spatio-temporal analysis. Appl. Catal. B Environ. 2019, 258, 117918. [Google Scholar] [CrossRef]
- Castoldi, L.; Matarrese, R.; Kubiak, L.; Daturi, M.; Artioli, N.; Pompa, S.; Lietti, L. In-depth insights into N2O formation over Rh- and Pt-based LNT catalysts. Catal. Today 2019, 320, 141–151. [Google Scholar] [CrossRef]
- Salisu, J.; Gao, N.; Quan, C.; Yanik, J.; Artioli, N. Co-gasification of rice husk and plastic in the presence of CaO using a novel ANN model-incorporated Aspen plus simulation. J. Energy Inst. 2023, 108, 101239. [Google Scholar] [CrossRef]
- Quan, C.; Zhang, G.; Gao, N.; Su, S.; Artioli, N.; Feng, D. Behavior Study of Migration and Transformation of Heavy Metals during Oily Sludge Pyrolysis. Energy Fuels 2022, 36, 8311–8322. [Google Scholar] [CrossRef]
- Byrne, E.L.; O’Donnell, R.; Gilmore, M.; Artioli, N.; Holbrey, J.D.; Swadzba-Kwasny, M. Hydrophobic functional liquids based on trioctylphosphine oxide (TOPO) and carboxylic acids. Phys. Chem. Chem. Phys. 2020, 22, 24744–24763. [Google Scholar] [CrossRef]
- Pandit, K.; Jeffrey, C.; Keogh, J.; Tiwari, M.S.; Artioli, N.; Manyar, H.G. Techno-Economic Assessment and Sensitivity Analysis of Glycerol Valorization to Biofuel Additives via Esterification. Ind. Eng. Chem. Res. 2023, 62, 9201–9210. [Google Scholar] [CrossRef]
- Manyar, H.G.; Iliade, P.; Bertinetti, L.; Coluccia, S.; Berlier, G. Structural and spectroscopic investigation of ZnS nanoparticles grown in quaternary reverse micelles. J. Coll. Interface Sci. 2011, 354, 511–516. [Google Scholar] [CrossRef]
- Yaemsunthorn, K.; Kobielusz, M.; Macyk, W. TiO2 with Tunable Anatase-to-Rutile Nanoparticles Ratios: How Does the Photoactivity Depend on the Phase Composition and the Nature of Photocatalytic Reaction. ACS Appl. Nano Mater. 2021, 4, 633–643. [Google Scholar] [CrossRef]
- Ohde, H.; Wai, C.M.; Kim, H.; Kim, J.; Ohde, M. Hydrogenation of olefins in supercritical CO2 catalyzed by palladium nanoparticles in a water-in-CO2 microemulsion. J. Am. Chem. Soc. 2002, 124, 4540–4541. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Zeng, G.; Huang, D.; Liu, L.; Lai, C.; Chen, M.; Zhang, C.; He, X.; Lai, M.; He, Y. Photocatalytic degradation of phenol by the heterogeneous Fe3O4 nanoparticles and oxalate complex system. RSC Adv. 2014, 4, 40828–40836. [Google Scholar] [CrossRef]
- Hamza, A.; Fatuase, J.T.; Waziri, S.M.; Ajayi, O.A. Solar Photocatalytic degradation of phenol using nanosized ZnO and α-Fe2O3. J. Chem. Eng. and Mat. Sci. 2013, 4, 87–92. [Google Scholar]
- Qiu, R.; Song, L.; Mo, Y.; Zhang, D.; Brewer, E. Visible light induced photocatalytic degradation of phenol by polymer- modified semiconductors: Study of the influencing factors and the kinetics. Reac. Kinet. Catal. Lett. 2008, 94, 183–189. [Google Scholar] [CrossRef]
- Ilisz, A.; La, Z. Investigation of the photodecomposition of phenol in near UV irradiated aqueous TiO2 suspensions. I: Effect of charge-trapping species on the degradation kinetics. Appl. Catal. A Gen. 1999, 180, 25–33. [Google Scholar] [CrossRef]
- Park, H.; Choi, W. Photocatalytic conversion of benzene to phenol using modified TiO2 and polyoxometalates. Catal. Today 2005, 101, 291–297. [Google Scholar] [CrossRef]





| Catalysts | ICP Analysis (%) | |
|---|---|---|
| Fresh (%Pd) | Used (%Pd) | |
| 0.5%Pd-TiO2 | 0.43 | 0.33 |
| 2%Pd@TiO2 | 1.8 | 1.78 |
| Catalysts | UV Light | Visible Light | |||
|---|---|---|---|---|---|
| Rate Constant kabs (10−4 s−1) | Phenol Converted (%) | Quantum Efficiency | Rate Constant kabs (10−4 s−1) | Phenol Converted (%) | |
| TiO2 P25 | 1.37 | 64.6 | 0.73 | 0.27 | 18.3 |
| 0.5%Pd/TiO2 | 2.45 | 79.7 | 0.94 | 0.42 | 25.1 |
| 2%Pd@mTiO2 | 1.1 | 53.7 | 0.61 | 0.9 | 50.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yilleng, M.T.; Artioli, N.; Rooney, D.; Manyar, H. Continuous Flow Photocatalytic Degradation of Phenol Using Palladium@Mesoporous TiO2 Core@Shell Nanoparticles. Water 2023, 15, 2975. https://doi.org/10.3390/w15162975
Yilleng MT, Artioli N, Rooney D, Manyar H. Continuous Flow Photocatalytic Degradation of Phenol Using Palladium@Mesoporous TiO2 Core@Shell Nanoparticles. Water. 2023; 15(16):2975. https://doi.org/10.3390/w15162975
Chicago/Turabian StyleYilleng, Moses T., Nancy Artioli, David Rooney, and Haresh Manyar. 2023. "Continuous Flow Photocatalytic Degradation of Phenol Using Palladium@Mesoporous TiO2 Core@Shell Nanoparticles" Water 15, no. 16: 2975. https://doi.org/10.3390/w15162975
APA StyleYilleng, M. T., Artioli, N., Rooney, D., & Manyar, H. (2023). Continuous Flow Photocatalytic Degradation of Phenol Using Palladium@Mesoporous TiO2 Core@Shell Nanoparticles. Water, 15(16), 2975. https://doi.org/10.3390/w15162975







