Fabrication of Magnetic Silica Nanomaterials and Their Effects on Algal Harvesting
Abstract
1. Introduction
2. Materials and Methods
2.1. Microalgal Cultivation
2.2. Preparation of Magnetic Folded Mesoporous Silica
2.2.1. Preparation of Magnetic Fe3O4
2.2.2. Preparation of Magnetic Mesoporous Silicon Materials
2.3. Characterization of the Physicochemical Properties of Magnetic Silica
2.4. Harvesting of Chlorella using Magnetic Mesoporous Silica
2.5. Data Analysis and Modeling Study
3. Results and Discussion
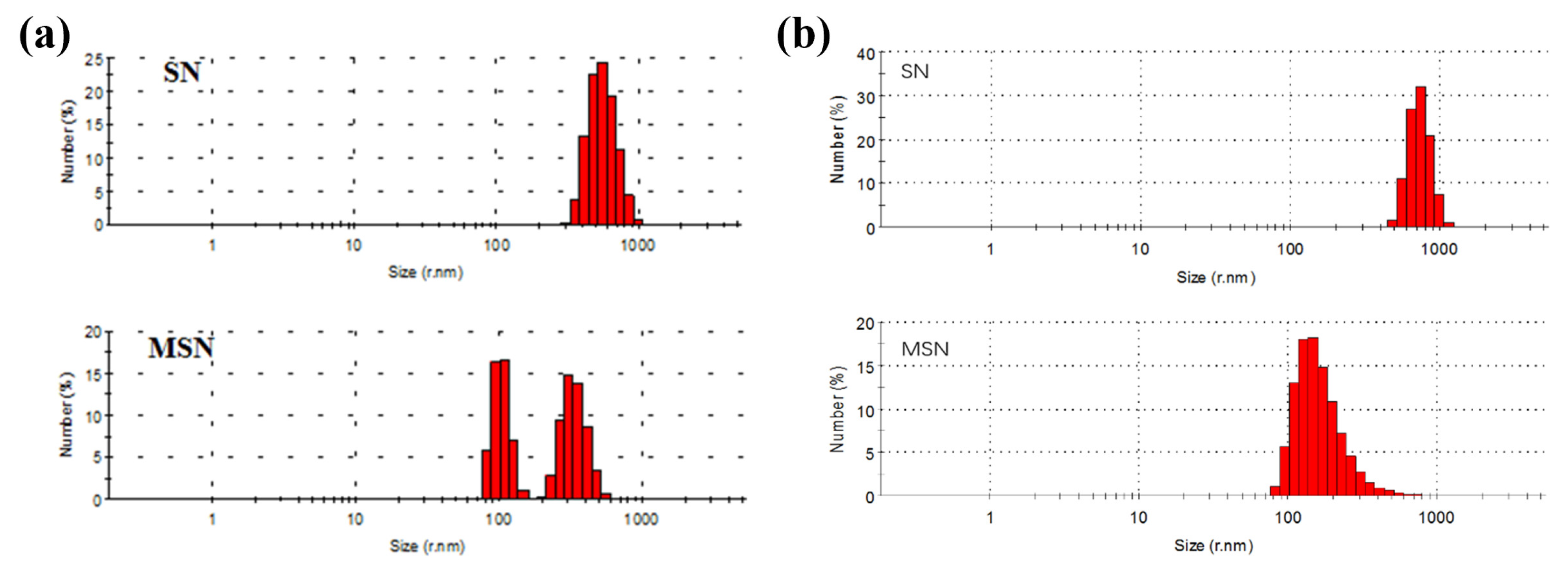
3.1. Material Characterization Morphology and Structure
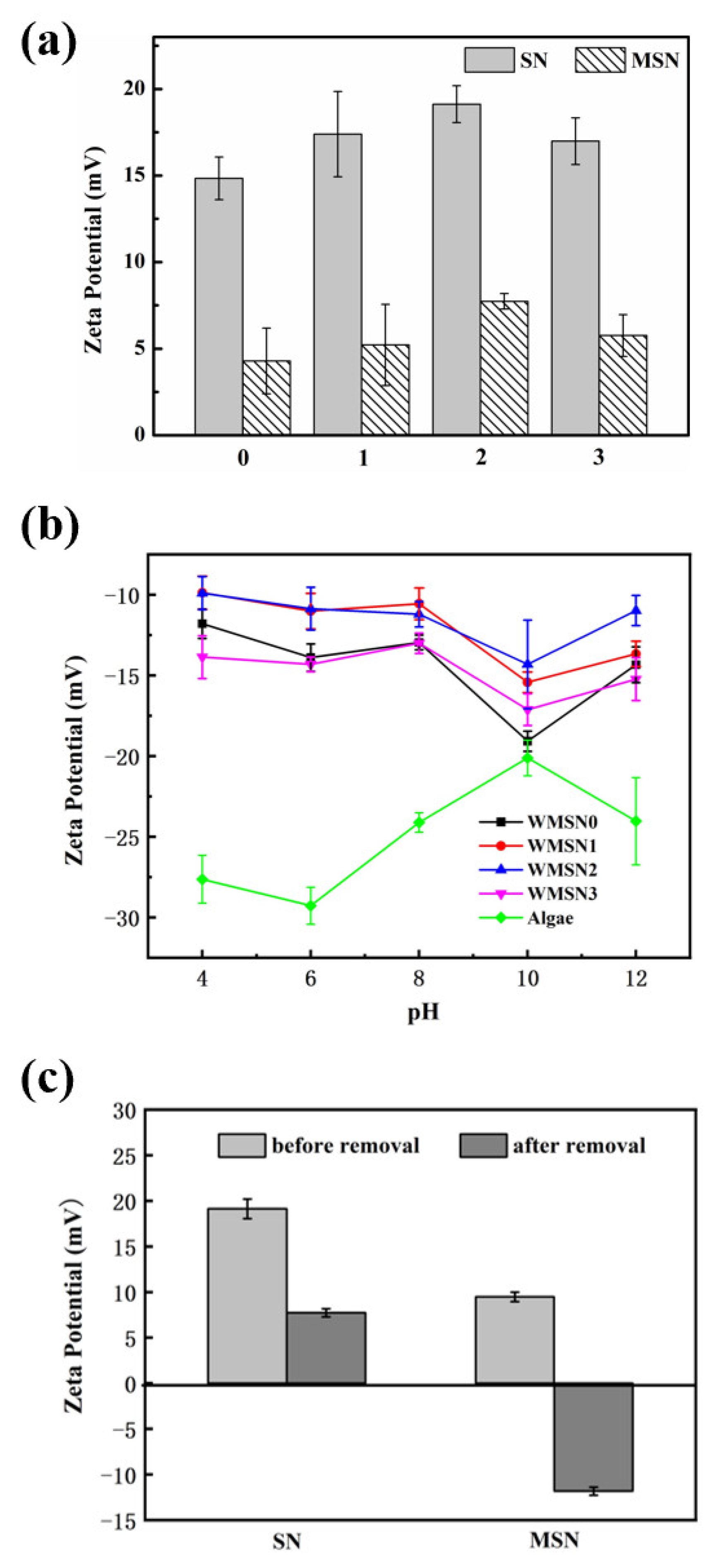
3.2. Zeta Potential of Materials under Different Experimental Conditions
3.3. Harvesting of Chlorella under Magnetic Mesoporous Silica
3.3.1. Effect of Active Agent and Stabilizer Ratios on the Harvesting of Chlorella
3.3.2. Effect of Mesoporous Structure Activator Removal Methods on the Harvesting of Chlorella
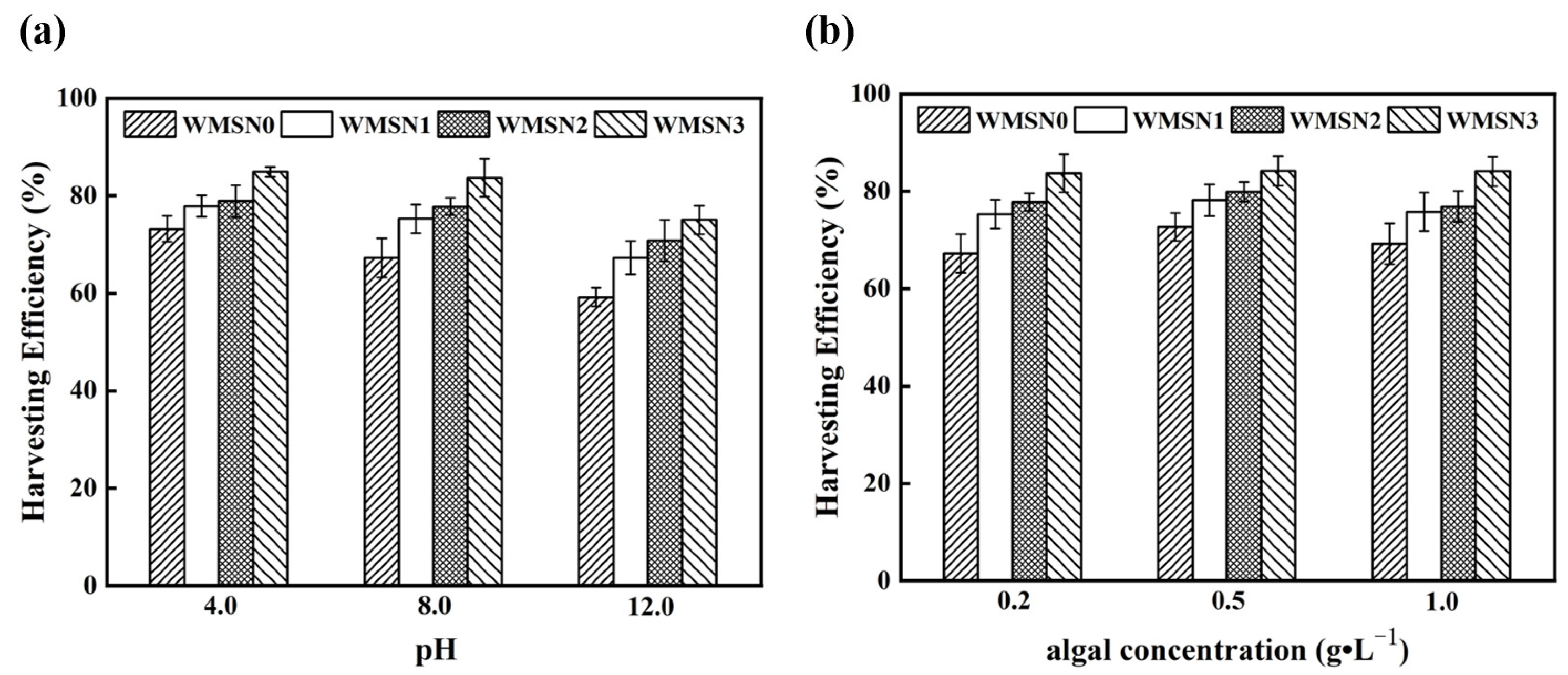
3.3.3. Effect of pH and Algal Concentration on the Harvesting of Chlorella
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- De Nicolás, A.P.; Molina-García, A.; García-Bermejo, J.T.; Vera-García, F. Reject brine management: Denitrification and zero liquid discharge (ZLD)—Current status, challenges and future prospects. J. Clean. Prod. 2022, 381 Pt 1, 135124. [Google Scholar]
- Lee, B.; Kang, H.; Kim, S. Using a novel coagulant as a practical and sustainable approach for cyanobacterial bloom control. Environ. Technol. Innov. 2023, 30, 103057. [Google Scholar]
- Zhan, M.; Liu, P.; Liu, X.; Hong, Y. Inactivation and removal technologies for algal-bloom control: Advances and challenges. Curr. Pollut. Rep. 2021, 7, 392–406. [Google Scholar]
- Hu, X.; Liu, W.J.; Ma, L.L.; Yu, H.Q. Sustainable Conversion of Harmful Algae Biomass into a CO2 Reduction Electrocatalyst for Two-Fold Carbon Utilization. Environ. Sci. Technol. 2023, 57, 1157–1166. [Google Scholar]
- Leon-Vaz, A.; Cubero-Cardoso, J.; Trujillo-Reyes, Á. Enhanced wastewater bioremediation by a sulfur-based copolymer as scaffold for microalgae immobilization (AlgaPol). Chemosphere 2023, 315, 137761. [Google Scholar] [PubMed]
- Singh, G.; Patidar, S.K. Microalgae harvesting techniques: A review. J. Environ. Manag. 2018, 217, 499–508. [Google Scholar]
- Matter, I.A.; Bui, V.K.H.; Jung, M.; Seo, J.Y.; Kim, Y.-E.; Lee, Y.-C.; Oh, Y.-K. Flocculation harvesting techniques for microalgae: A review. Appl. Sci. 2019, 9, 3069. [Google Scholar]
- Hola, K.; Markova, Z.; Zoppellaro, G.; Tucek, J.; Zboril, R. Tailored functionalization of iron oxide nanoparticles for MRI, drug delivery, magnetic separation and immobilization of biosubstances. Biotechnol. Adv. 2015, 33, 1162–1176. [Google Scholar]
- Chiang, Y.D.; Dutta, S.; Chen, C.; Huang, Y.T.; Lin, K.S.; Wu, J.C.S.; Suzuki, N.; Yamauchi, Y.; Wu, K.C.W. Functionalized Fe3O4@Silica Core-Shell Nanoparticles as Microalgae Harvester and Catalyst for Biodiesel Production. ChemSusChem 2015, 8, 789–794. [Google Scholar]
- Ge, S.J.; Agbakpe, M.; Wu, Z.Y.; Kuang, L.Y.; Zhang, W.; Wang, X.Q. Influences of surface coating, UV irradiation and magnetic field on the algae removal using magnetite nanoparticles. Environ. Sci. Technol. 2015, 49, 1190–1196. [Google Scholar] [CrossRef]
- Hu, Y.R.; Guo, C.; Wang, F.; Wang, S.K.; Pan, F.; Liu, C.Z. Improvement of microalgae harvesting by magnetic nanocomposites coated with polyethylenimine. Chem. Eng. J. 2014, 242, 341–347. [Google Scholar]
- Li, S.X.; Hu, T.Y.; Xu, Y.Z.; Wang, J.Y.; Chu, R.Y.; Yin, Z.H.; Mo, F.; Zhu, L.D. A review on flocculation as an efficient method to harvest energy microalgae: Mechanisms, performances, influencing factors and perspectives. Renew. Sust. Energ. Rev. 2020, 131, 110005. [Google Scholar]
- Wang, T.; Yang, W.L.; Hong, Y.; Hou, Y.L. Magnetic nanoparticles grafted with amino-riched dendrimer as magnetic flocculant for efficient harvesting of oleaginous microalgae. Chem. Eng. J. 2016, 297, 304–314. [Google Scholar]
- Jafarirad, S.; Ardehjani, P.H.; Movafeghi, A. Are the green synthesized nanoparticles safe for environment? A case study of aquatic plant Azolla filiculoides as an indicator exposed to magnetite nanoparticles fabricated using microwave hydrothermal treatment and plant extract. J. Environ. Sci. Health A 2019, 54, 516–527. [Google Scholar]
- Hui, C.; Shen, C.; Tian, J.; Bao, L.; Ding, H.; Li, C.; Tian, Y.; Shi, X.; Gao, H.J. Core-shell Fe3O4@SiO2 nanoparticles synthesized with well dispersed hydrophilic Fe3O4 seeds. Nanoscale 2011, 3, 701–705. [Google Scholar]
- Liu, P.R.; Zhang, H.L.; Wang, T.; Yang, W.L.; Hong, Y.; Hou, Y.-L. Functional graphene-based magnetic nanocomposites as magnetic flocculant for efficient harvesting of oleaginous microalgae. Algal Res. 2016, 19, 86–95. [Google Scholar]
- Liu, P.R.; Wang, T.; Yang, Z.Y.; Hong, Y.; Hou, Y.L. Long-chain poly-arginine functionalized porous Fe3O4 microspheres as magnetic flocculant for efficient harvesting of oleaginous microalgae. Algal Res. 2017, 27, 99–108. [Google Scholar]
- Gerulová, K.; Bartošová, A.; Blinová, L.; Bártová, K.; Dománková, M.; Garaiová, Z.; Palcut, M. Magnetic Fe3O4-polyethyleneimine nanocomposites for efficient harvesting of Chlorella zofingiensis, Chlorella vulgaris, Chlorella sorokiniana, Chlorella ellipsoidea and Botryococcus braunii. Algal Res. 2018, 33, 165–172. [Google Scholar]
- Gerulová, K.; Kucmanová, A.; Sanny, Z.; Garaiová, Z.; Seiler, E.; Čaplovičová, M.; Čaplovič, L.; Palcut, M. Fe3O4-PEI nanocomposites for magnetic harvesting of Chlorella vulgaris, Chlorella ellipsoidea, Microcystis aeruginosa, and Auxenochlorella protothecoides. Nanomaterials 2022, 12, 1786. [Google Scholar]
- Wei, Y.L.; Yang, W.; Yang, Z.W. An excellent universal catalyst support-mesoporous silica: Preparation, modification and applications in energy-related reactions. Int. J. Hydrogen Energy 2022, 47, 9537–9565. [Google Scholar]
- Wang, X.; Wang, H.; Lu, M.; Teng, R.; Du, X. Facile synthesis of phenyl-modified magnetic graphene/mesoporous silica with hierarchical bridge-pore structure for efficient adsorption of pesticides. Mater. Chem. Phys. 2017, 198, 393–400. [Google Scholar]
- Shah, S.M.; Makhdoom, A.; Su, X.; Faheem, M.; Irfan, M.; Irfan, M.; Wang, G.; Gao, Y. Synthesis of sulphonic acid functionalized magnetic mesoporous silica for Cu (II) and Co (II) adsorption. Microchem. J. 2019, 151, 104194. [Google Scholar]
- Fatimah, I.; Fadillah, G.; Purwiandono, G.; Sahroni, I.; Purwaningsih, D.; Riantana, H.; Avif, A.N.; Sagadevan, S. Magnetic-silica nanocomposites and the functionalized forms for environment and medical applications: A review. Inorg. Chem. Commun. 2022, 137, 109213. [Google Scholar]
- Kumar, P.; Patel, A.K.; Chen, C.W.; Nguyen, T.B.; Chang, J.S.; Pandey, A.; Dong, C.-D.; Singhania, R.R. Development of dopamine-based magnetite nanocomposite for effective harvesting of Chlorella sorokiniana Kh12 biomass. Environ. Technol. Innov. 2023, 29, 103008. [Google Scholar]
- Nie, X.; Zhang, H.; Cheng, S.; Mubashar, M.; Xu, C.; Li, Y.; Tan, D.; Zhang, X. Study on the cell-collector-bubble interfacial interactions during microalgae harvesting using foam flotation. Sci. Total Environ. 2022, 806, 150901. [Google Scholar] [CrossRef]
- Zhang, Q.; Hong, Y. Comparison in growth, lipid accumulation, and nutrient removal capacities of Chlorella sp. in secondary effluents under sterile and non-sterile conditions. Water Sci. Technol. 2014, 69, 573–579. [Google Scholar] [PubMed]
- Zhang, H.K.; Hong, Y.; Wang, X.Y.; Li, X.Y.; Zhao, G.P.; Wang, Q.; Zhai, Q.Y. Chlorella potential to purify domestic waste leachate for biomass production and community structure responses of wastewater-borne bacteria. Algal Res. 2022, 66, 102825. [Google Scholar]
- Liu, X.Y.; Hong, Y.; Liu, Y. Effect of light quality on Chlorella sp. HQ cultivation in inland saline-alkali leachate: Growth, lipid accumulation and fatty acid profile. Front. Environ. Sci. Eng. 2022, 16, 45. [Google Scholar]
- Zhang, Q.; Hong, Y. Effects of stationary phase elongation and initial nitrogen and phosphorus concentrations on the growth and lipid-producing potential of Chlorella sp. HQ. J. Appl. Phycol. 2013, 26, 141–149. [Google Scholar]
- Hu, Y.R.; Wang, F.; Wang, S.K.; Liu, C.Z.; Guo, C. Efficient harvesting of marine microalgae Nannochloropsis maritima using magnetic nanoparticles. Bioresour. Technol. 2013, 138, 387–390. [Google Scholar]
- Pang, J.L.; Zhou, G.W.; Liu, R.R.; Li, T.D. Esterification of oleic acid with methanol by immobilized lipase on wrinkled silica nanoparticles with highly ordered, radially oriented mesochannels. Mater. Sci. Eng. C 2016, 59, 35–42. [Google Scholar]
- Ndikubwimana, T.; Zeng, X.H.; Liu, Y.; Chang, J.S.; Lu, Y.H. Harvesting of microalgae Desmodesmus sp. F51 by bioflocculation with bacterial bioflocculant. Algal Res. 2014, 6, 186–193. [Google Scholar]
- Martin, P.; Jana, N.; Tomas, B. The impact of interactions between algal organic matter and humic substances on coagulation. Water Res. 2015, 84, 278–285. [Google Scholar]
- Liu, H.F.; Ji, S.F.; Yang, H.; Zhang, H.; Tang, M. Ultrasonic-assisted ultra-rapid synthesis of monodisperse meso-SiO2@Fe3O4 microspheres with enhanced mesoporous structure. Ultrason. Sonochem. 2014, 21, 505–512. [Google Scholar] [PubMed]
- Yamaguchi, A.; Kobayashi, M.; Adachi, Y. Yield stress of mixed suspension of silica particles and lysozymes: The effect of zeta potential and adsorbed amount. Colloids Surf. A 2019, 578, 123575. [Google Scholar]
- Seo, J.Y.; Lee, K.; Praveenkumar, R.; Kim, B.; Lee, S.Y.; Oh, Y.-K.; Bin Park, S. Trifunctionality of Fe3O4-embedded carbon microparticles in microalgae harvesting. Chem. Eng. J. 2015, 280, 206–214. [Google Scholar]
- Li, J.; Zhang, J.; Chen, L.X.; Wen, Z.Q.; Luo, J.; Yu, C.; Wang, P.P. Binary cationic-anionic surfactant templated synthesis of organic functionalized porous silica nanomaterials. J. Porous Mater. 2020, 27, 29–35. [Google Scholar]
- Vandamme, D.; Foubert, I.; Muylaert, K. Flocculation as a low-cost method for harvesting microalgae for bulk biomass production. Trends Biotechnol. 2013, 31, 233–239. [Google Scholar]
- Zhang, X.; Amendola, P.; Hewson, J.C.; Sommerfeld, M.; Hu, Q. Influence of growth phase on harvesting of Chlorella zofingiensis by dissolved air flotation. Bioresour. Technol. 2012, 116, 477–484. [Google Scholar]
- Fuentes-García, J.A.; Alavarse, A.C.; de Castro, C.E.; Giacomelli, F.C.; Ibarra, M.R.; Bonvent, J.-J.; Goya, G.-F. Sonochemical route for mesoporous silica-coated magnetic nanoparticles towards pH-triggered drug delivery system. J. Mater. Sci. Technol. 2021, 15, 52–67. [Google Scholar]
- Rong, H.; Gao, T.; Zheng, Y.; Li, L.; Xu, D.; Zhang, X.; Hou, Y.; Yan, M. Fe3O4@ silica nanoparticles for reliable identification and magnetic separation of Listeria monocytogenes based on molecular-scale physiochemical interactions. J. Mater. Sci. Technol. 2021, 84, 116–123. [Google Scholar]
- Niksefat, M.; Rahimi, J.; Maleki, A.; Nia, A.S. N-rich porous triazine based organic polymer composed with magnetic for high-efficiency removal of blue-green microalgae from wastewater. Algal Res. 2023, 70, 103003. [Google Scholar] [CrossRef]
- Cheng, Y.-S.; Zheng, Y.; Labavitch, J.M.; VanderGheynst, J.S. The impact of cell wall carbohydrate composition on the chitosan flocculation of Chlorella. Process. Biochem. 2011, 46, 1927–1933. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, X.; Jiang, X.; Fan, Q.; Li, X.; Jiao, L.; Liang, W. Harvesting of Chlorella vulgaris using Fe3O4 coated with modified plant polyphenol. Environ. Sci. Pollut. Res. 2018, 25, 26246–26258. [Google Scholar]
- Wang, X.; Zhao, Y.; Jiang, X.; Liu, L.; Li, X.; Li, H.; Liang, W. In-situ self-assembly of plant polyphenol-coated Fe3O4 particles for oleaginous microalgae harvesting. J. Environ. Manag. 2018, 214, 335–345. [Google Scholar]
- Seo, J.Y.; Jeon, H.J.; Kim, J.W.; Lee, J.; Oh, Y.K.; Ahn, C.W.; Lee, J.W. Simulated-sunlight-driven cell lysis of magnetophoretically separated microalgae using ZnFe2O4 octahedrons. Ind. Eng. Chem. Res. 2018, 57, 1655–1661. [Google Scholar] [CrossRef]
- Hao, N.; Jayawardana, K.W.; Chen, X.; Yan, M. One-step synthesis of amine-functionalized hollow mesoporous silica nanoparticles as efficient antibacterial and anticancer materials. ACS Appl. Mater. Interfaces 2015, 7, 1040–1045. [Google Scholar] [CrossRef]
- Cerff, M.; Morweiser, M.; Dillschneider, R.; Michel, A.; Menzel, K.; Posten, C. Harvesting fresh water and marine algae by magnetic separation: Screening of separation parameters and high gradient magnetic filtration. Bioresour. Technol. 2012, 118, 289–295. [Google Scholar]
- Dai, C.; Wang, S.; Li, Y.; Gao, M.; Liu, Y.; Sun, Y.; Zhao, M. The first study of surface modified silica nanoparticle in pressure decreasing application. RSC Adv. 2015, 5, 61838–61845. [Google Scholar]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Liu, P.; Hong, Y. Fabrication of Magnetic Silica Nanomaterials and Their Effects on Algal Harvesting. Water 2023, 15, 2823. https://doi.org/10.3390/w15152823
Zhang Y, Liu P, Hong Y. Fabrication of Magnetic Silica Nanomaterials and Their Effects on Algal Harvesting. Water. 2023; 15(15):2823. https://doi.org/10.3390/w15152823
Chicago/Turabian StyleZhang, Yuewen, Peirui Liu, and Yu Hong. 2023. "Fabrication of Magnetic Silica Nanomaterials and Their Effects on Algal Harvesting" Water 15, no. 15: 2823. https://doi.org/10.3390/w15152823
APA StyleZhang, Y., Liu, P., & Hong, Y. (2023). Fabrication of Magnetic Silica Nanomaterials and Their Effects on Algal Harvesting. Water, 15(15), 2823. https://doi.org/10.3390/w15152823





