Investigating Freshwater Mullet Fisheries in Tunisian Reservoirs: Future Development Prospects
Abstract
:1. Introduction
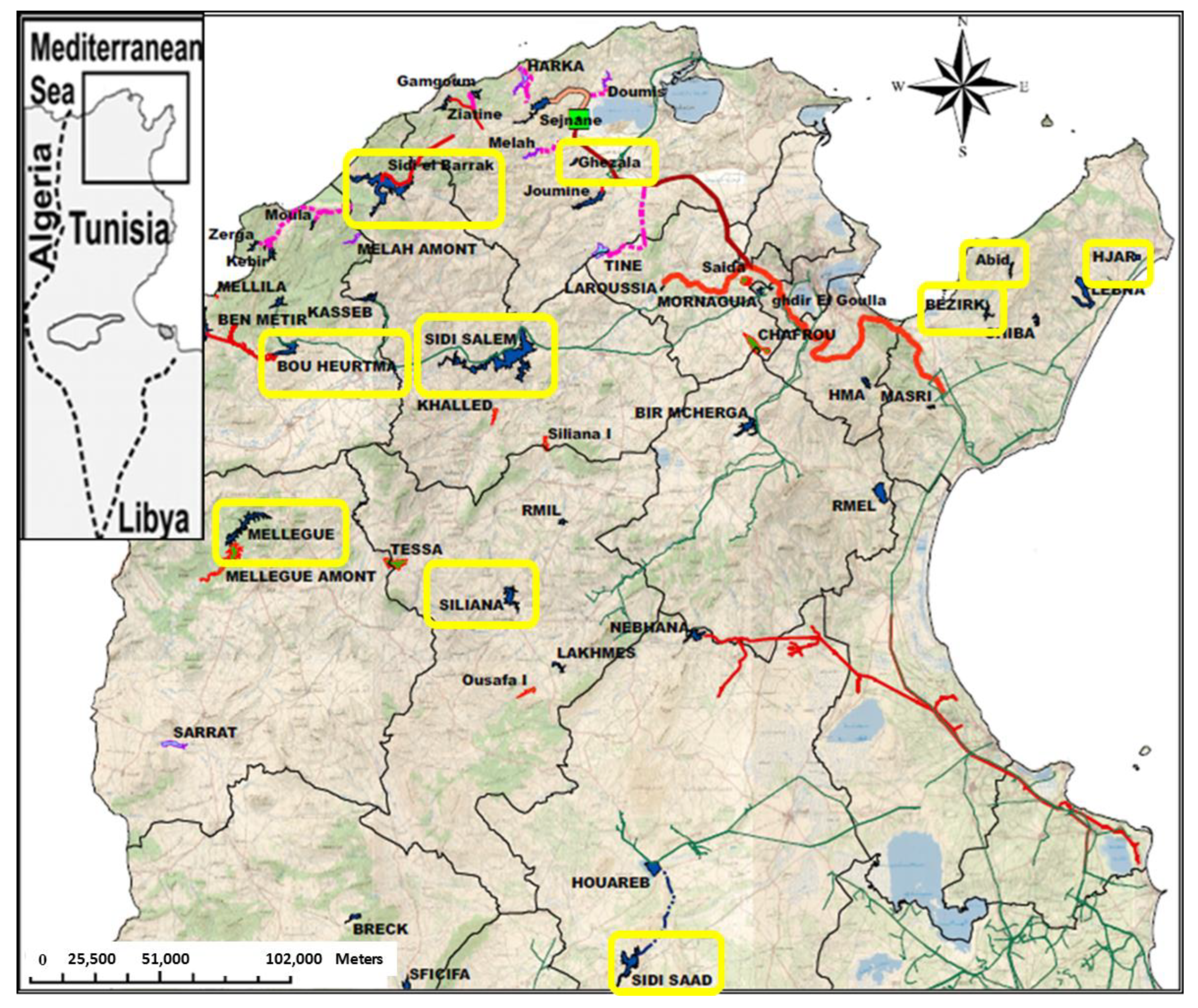
2. Materials and Methods
3. Results
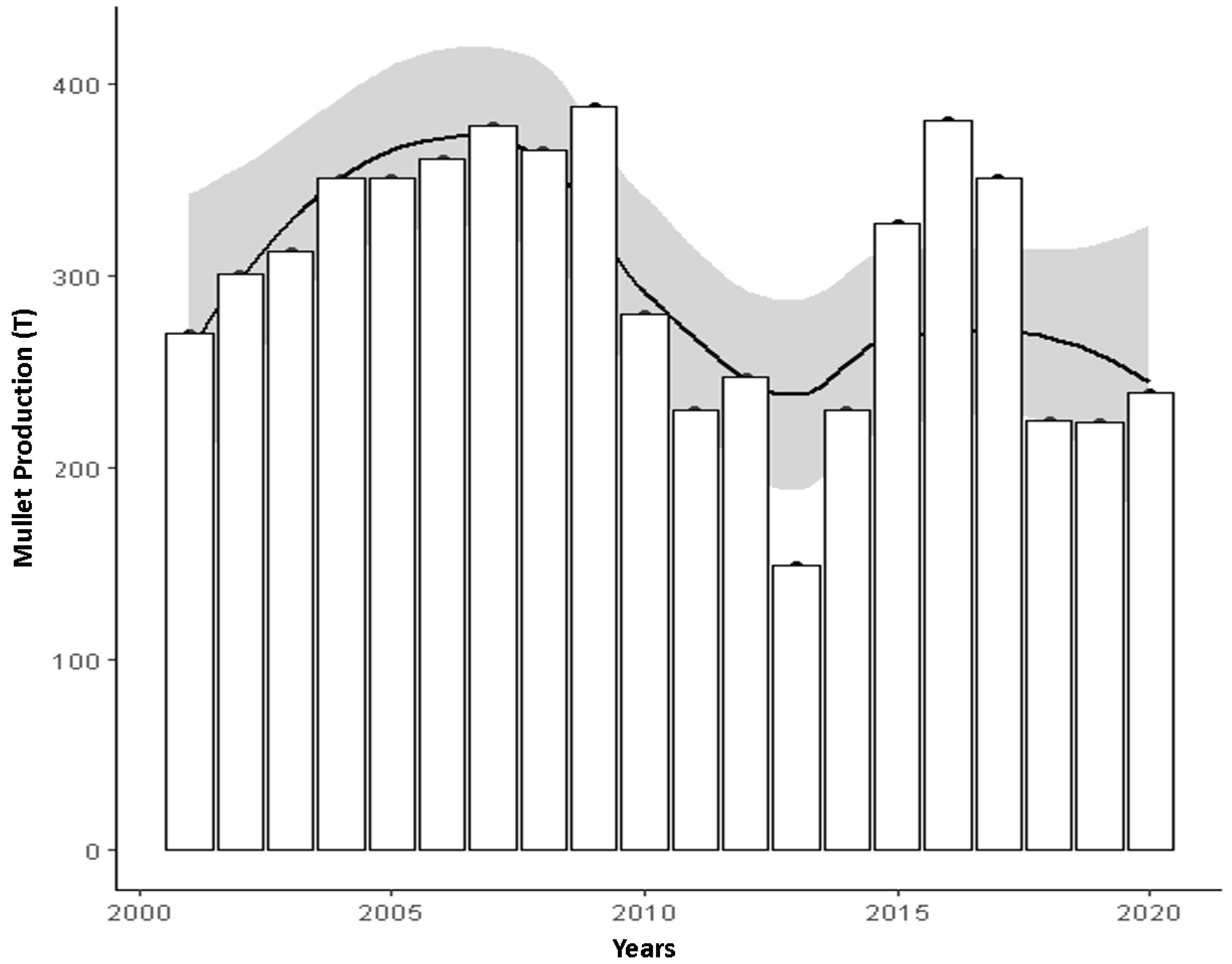
3.1. Mullet Production
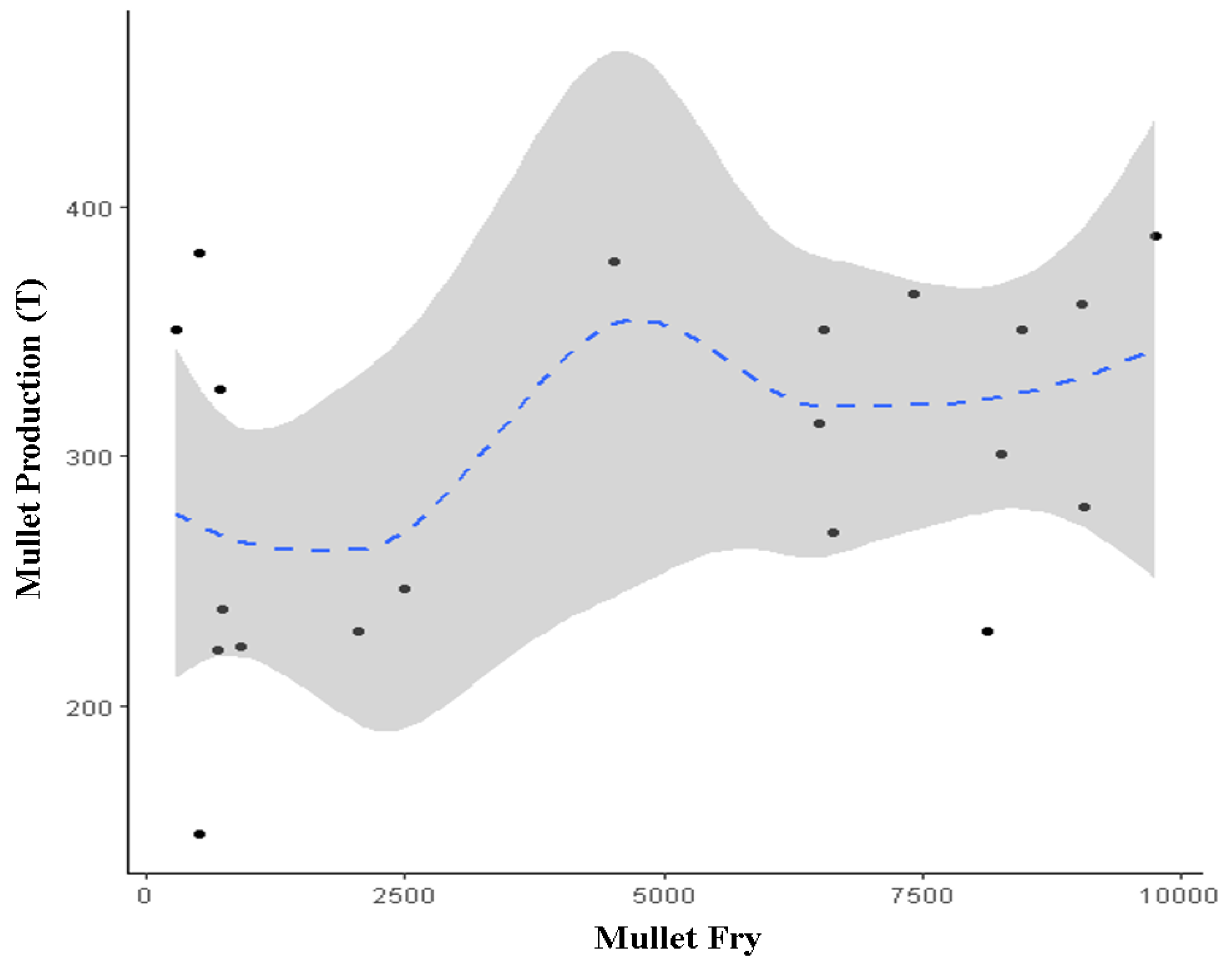
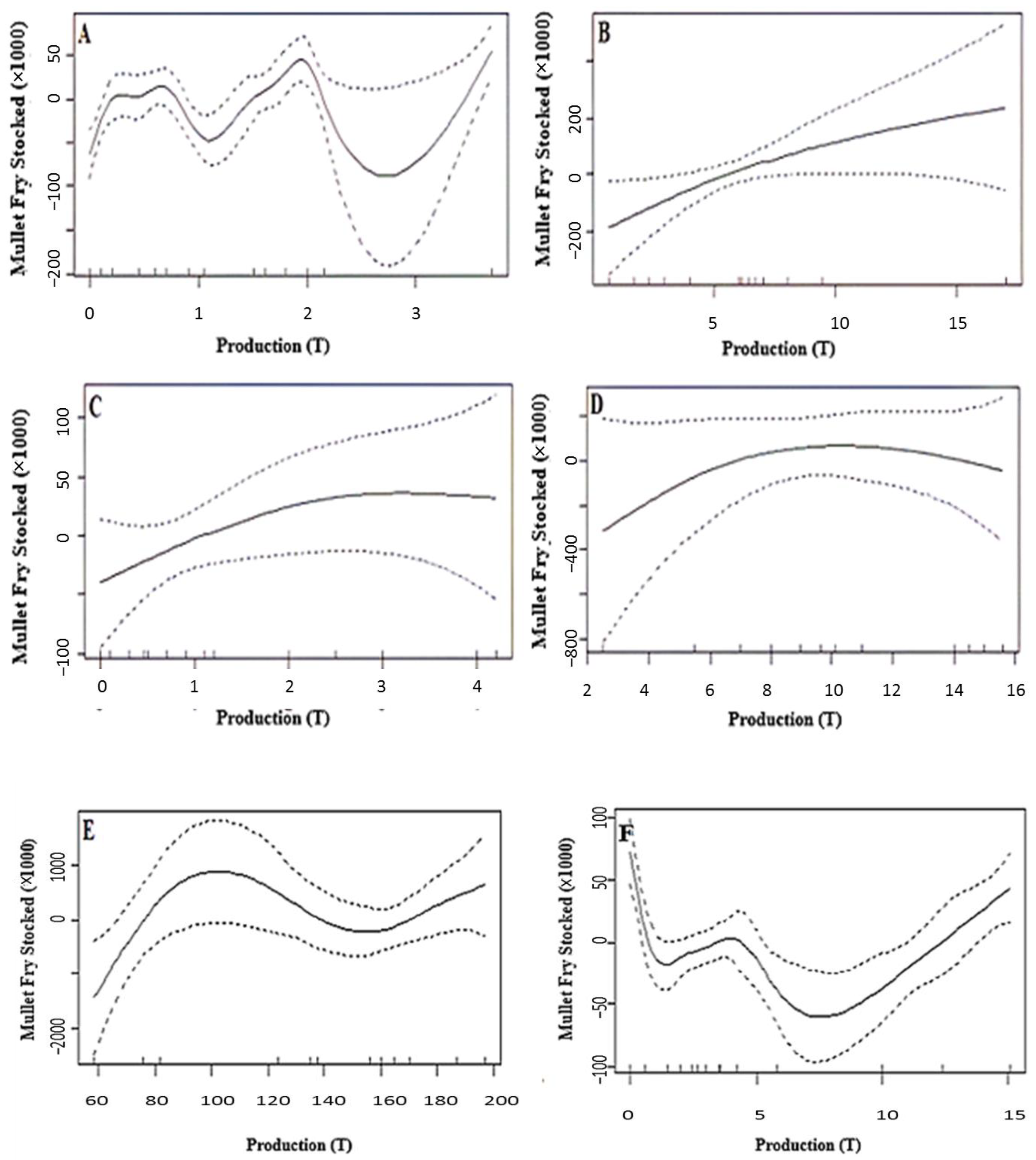
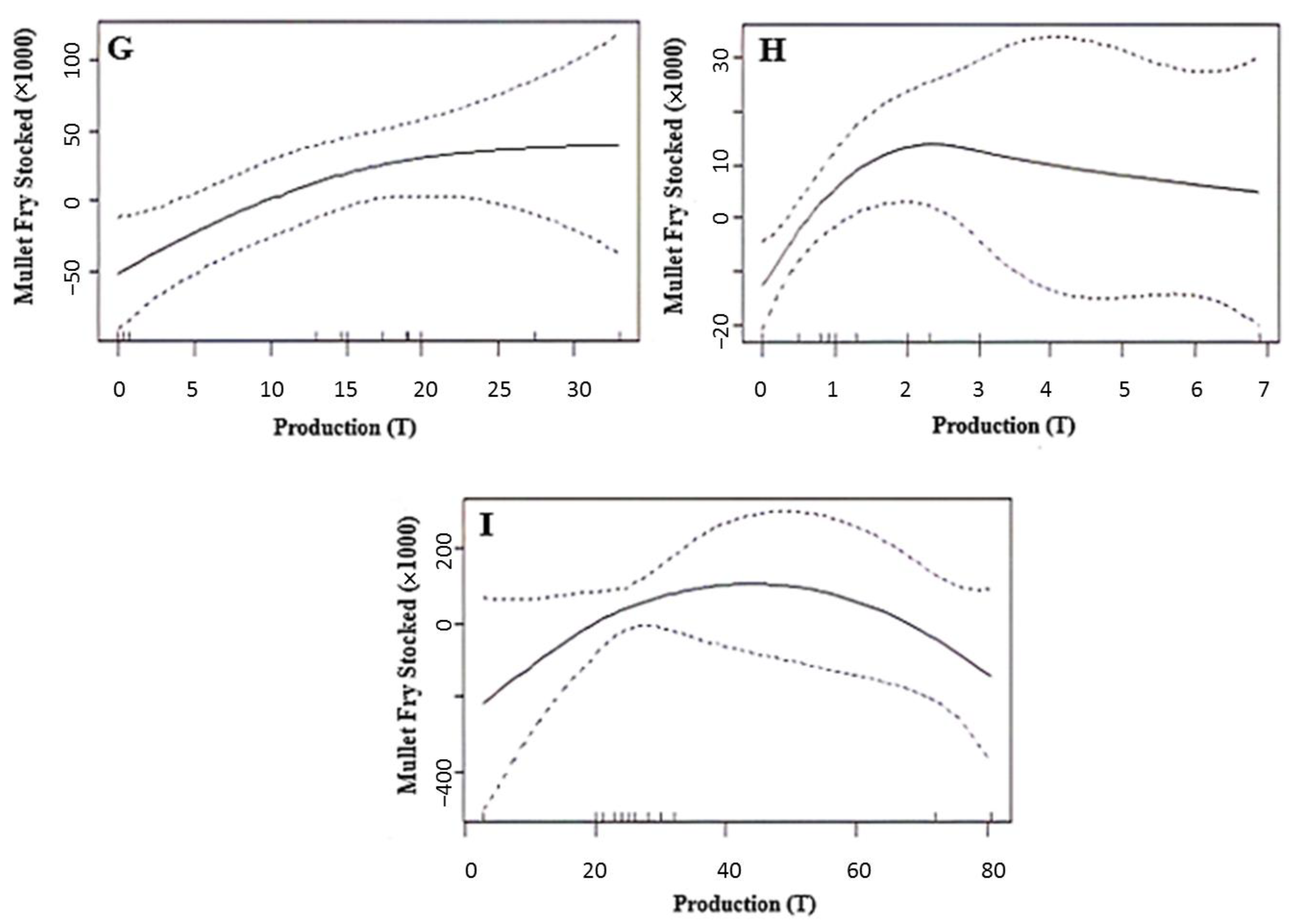
3.2. Correlation between the Introduced Fry and the Harvested Mullet
3.3. Numerical and Weight Yield
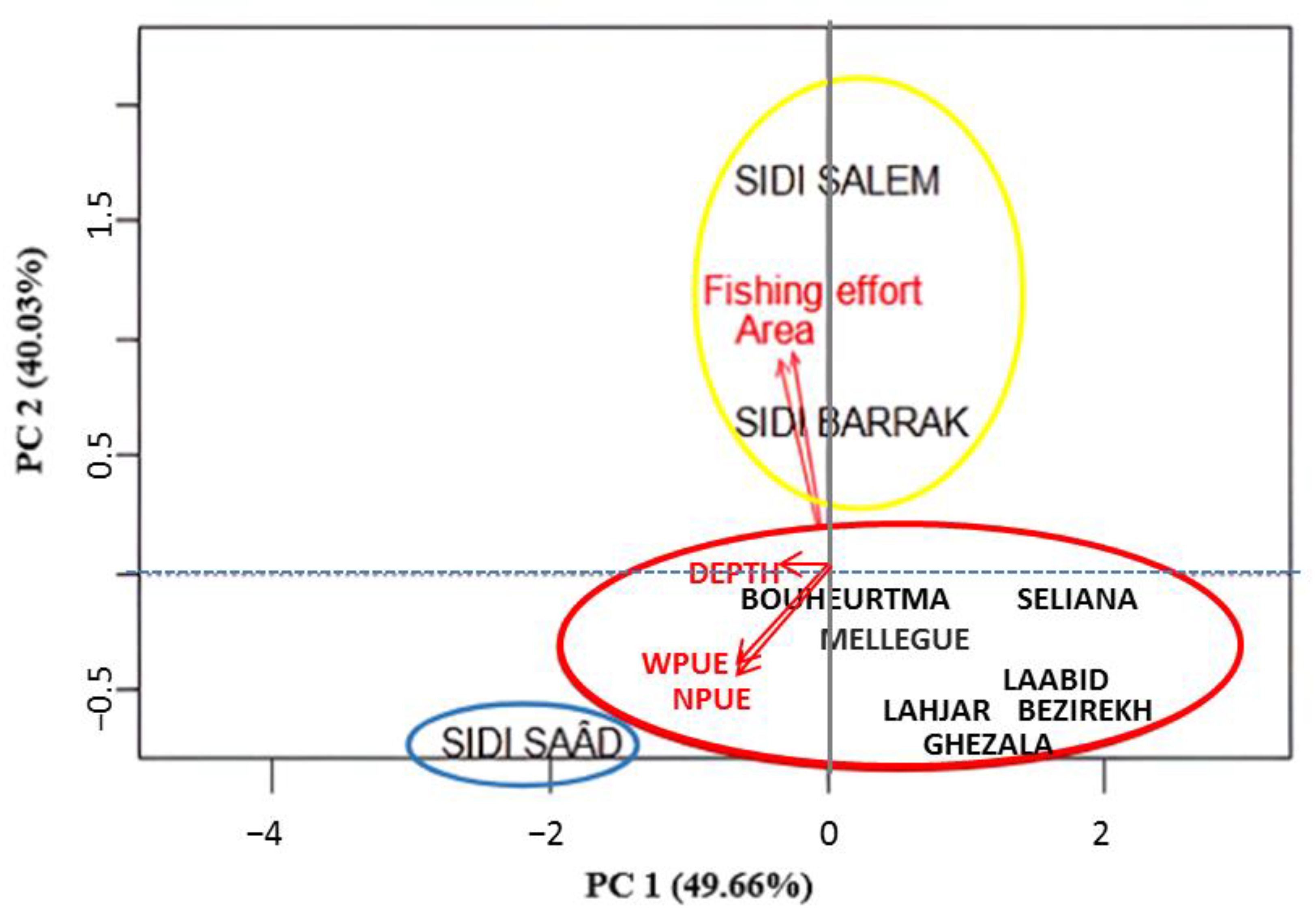
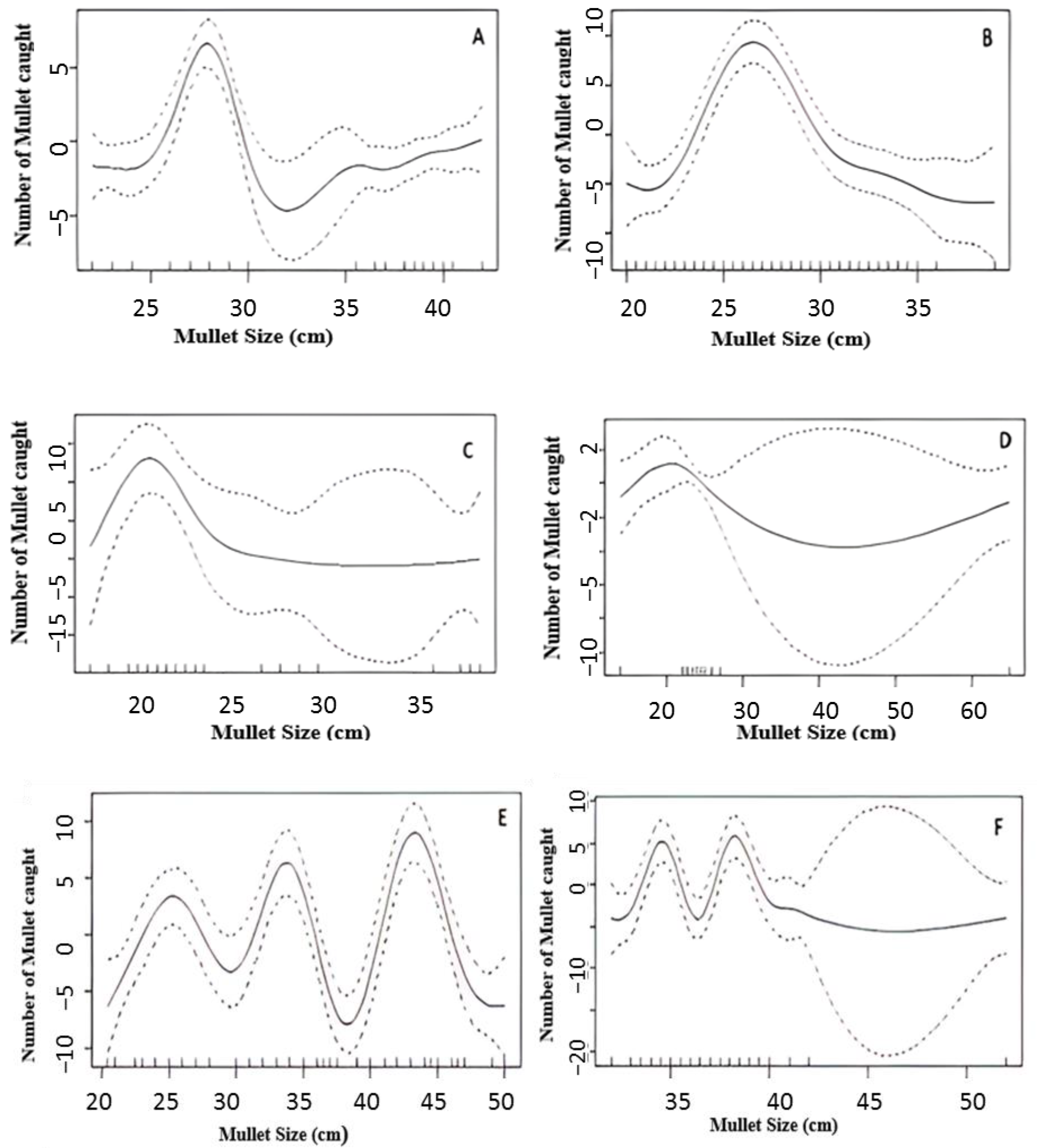
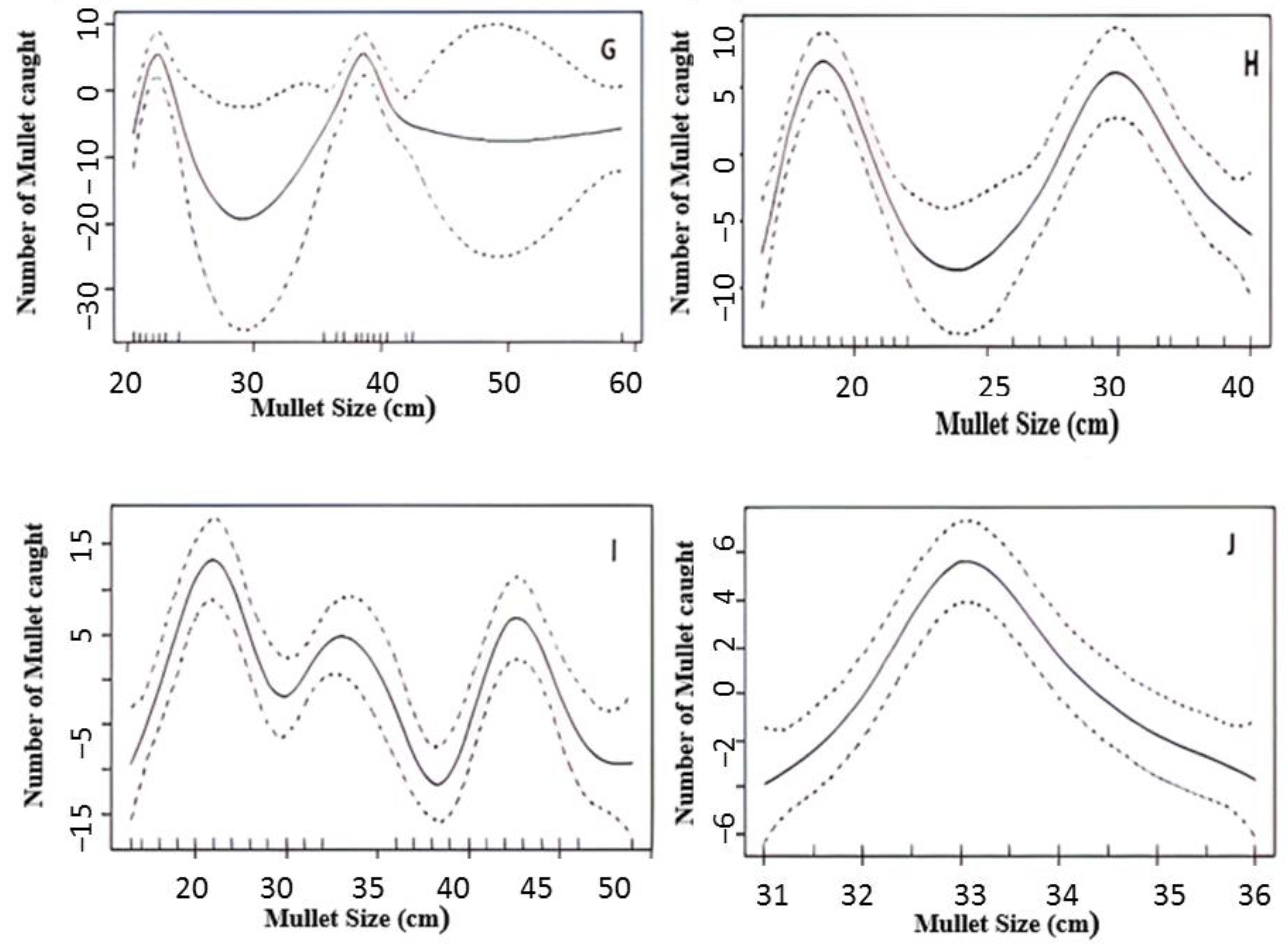
3.4. Population Status
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Losse, G.F.; Nau, W.; Winter, M. The Development of Freshwater Fishing in Northern Tunisia: Tunisian-German Technical Cooperation Project, Project Use of Dams for Fish Farming; GTZ and CGP: Tunis, Tunisia, 1991; 418p. [Google Scholar]
- ADCP. Etude du Potentiel Aquacole et Proposition Pour une Politique de Développement de L’aquaculture en Tunisie; Rapport d’une Mission Interdisciplinaire TCP/ADCP en Tunisie (Mars–Juin 1982); ADCP/MR/83-21; FAO: Rome, Italy, 1983; p. 248. [Google Scholar]
- Anonymous. Proposition de Projet: Projet Pilote Pour le Développement de L’Aquaculture Dans Les Retenues de Barrages; FAO: Rome, Italy, 1986; p. 49. [Google Scholar]
- Anonymous. American Peace Corps: Fish Farming of Dam Reservoirs; Study Project; National Agronomic Institute of Tunisia: Tunis, Tunisia, 1986; p. 122. [Google Scholar]
- Anonymous. Tunisia Country Profile 1989/90; The Economist Intelligence Unit: London, UK, 1988; p. 86. [Google Scholar]
- Mili, S.; Ennouri, R.; Laouar, H.; Khedhri, I.; Zarrouk, H.; Chargui, T.; Romdhane, N. Freshwater Fish Farming and Fishery Management in Tunisian Reservoirs: Limitations and Opportunities. In Agriculture Productivity in Tunisia under Stressed Environment; Allouche, K.F., Abu-hashim, M., Negm, A.M., Eds.; Springer: Cham, Switzerland, 2021; pp. 8–34. [Google Scholar]
- Loss, G.F.; Kraiem, M.M.; Boughedir, M. Preliminary Inventory of the Vertebrate Fauna of the Sidi Salem Dam with in Appendix a List of the Fish Fauna of the Nebhana, Bir M’chergua, Sidi Saâd, Ben Mtir, Bou Heurtma and Mellègue Reservoirs; Technical Reports; Tunisian-German Continental Fishing Project; GTZ Gmbh: Tunis, Tunisia, 1990; p. 403. [Google Scholar]
- Djemali, I.; Laouar, H.; Toujani, R. Distribution patterns of fish biomass by acoustic survey in three Tunisian man-made lakes. J. Appl. Ichthyol. 2010, 26, 390–396. [Google Scholar] [CrossRef]
- Mili, S.; Ennouri, R.; Dhib, A.; Laouar, H.; Missaoui, H.; Aleya, L. Characterization of fish assemblages and population structure of freshwater fish in two Tunisian reservoirs: Implications for fishery management. Environ. Monit. Assess. 2016, 188, 364. [Google Scholar] [CrossRef] [PubMed]
- Mili, S.; Ennouri, R.; Laouar, H. Population status of freshwater fish in Tunisian reservoirs. Sav. Freshw. Fishes Habitats 2016, 11, 32–34. [Google Scholar]
- De Silva, S.S.; Amarasinghe, U.S. Reservoir Fisheries in Asia. In Perspectives in Asian Fisheries, A Volume to Commemorate the 10th Anniversary of the Asian Fisheries Society; Asian Fisheries Society, Manila: Selangor, Malaysia, 1996; pp. 189–216. [Google Scholar]
- Hadj Salah, H. Etude Socio-Economique et de Rentabilité de L’activité de Pêche Dans Les Eaux Douces de Tunisie. Ph.D. Thesis, Rennes University, Rennes, France, 2002. [Google Scholar]
- Mili, S.; Ennouri, R.; Laouar, H.; Missaoui, H. Fisheries in the Tunisians dams: Diagnosis of the current situation and development opportunities. FAO Fish. Aqua. Proc. 2015, 39, 95–106. [Google Scholar]
- Mtimet, N. Socio-Economic Analysis of the Continental Fish Farming Sector in the North-West of Tunisia. In Project ‘Safeguarding and Socio-Economic Enhancement of Environmental Resources in the North-West Region of Tunisia’; COSPE/GIPP: Tabarka, Tunisia, 2010; p. 150. [Google Scholar]
- Linnæus, C. Systema Naturæ per Regna Tria Naturæ, Secundum Classes, Ordines, Genera, Species, Cum Characteribus, Differentiis, Synonymis, Locis, 10th ed.; Tom, I., Ed.; Laurentius Salvius: Stockholm, Sweden, 1758; 824p. [Google Scholar]
- Risso, A. Natural History of the Principal Productions of Southern Europe and Particularly of Those around Nice and the Alpes Maritimes; Book Series: Paris, France, 1827; pp. 1–5. [Google Scholar]
- Blel, H.; Chatti, N.; Besbes, R.; Farjallah, S.; Elouaer, A.; Guerbej, H.; Said, K. Phylogenetic relationships in grey mullets (Mugilidae) in a Tunisian lagoon. Aqua. Res. 2008, 39, 268–275. [Google Scholar] [CrossRef]
- EN 14757; Water Quality-Sampling of Fish with Multi-Mesh Gillnets. CEN (European Committee for Standardization): Brussels, Belgium, 2005.
- Wood, S.N. Generalized Additive Models: An Introduction with R; CRC Press: New York, NY, USA, 2006. [Google Scholar] [CrossRef] [Green Version]
- Züur, A.; Ieno, E.; Walker, N.; Saveliev, A.; Smith, G. Mixed Effects Models and Extensions in Ecology with R; Springer: New York, NY, USA, 2009; p. 547. [Google Scholar]
- Deceliere-Vergès, C.; Guillard, J. Assessment of the pelagic fish populations using CEN multi-mesh gill nets: Consequences for the characterization of the fish communities. Know. Manag. Aqua. Ecosys. 2008, 389, 4. [Google Scholar] [CrossRef]
- Argillier, C.; Caussé, S.; Gevrey, M.; Pédron, S.; De-Bortoli, J.; Brucet, S. Development of a fish-based index to assess the eutrophication status of European lakes. Hydrobiologia 2013, 704, 193–211. [Google Scholar] [CrossRef] [Green Version]
- Oksanen, J.; Blanchet, G.; Kindt, R.; Legendre, P.; O’Hara, R.; Simpson, G.; Solymos, P.; Stevens, H.; Wagner, H. Vegan: Community Ecology Package, R Package Version 1. 2011. Available online: http://CRAN.R-project.org/package0vegan (accessed on 2 January 2023).
- Giraudoux, P. Pgirmess: Data Analysis in Ecology. R Package Version ‘1.5.6′. 2012. Available online: http://CRAN.R-project.org/package=pgirmess (accessed on 3 January 2023).
- DGPA. Yearbook of Fishing Statistics from the General Directorate of Fisheries and Aquaculture; General Directorate of Fisheries and Aquaculture: Tunis, Tunisia, 2020; Available online: http://www.agridata.tn (accessed on 15 March 2023).
- Mili, S.; Ennouri, R.; Fatnassi, M.; Chargui, T.; Zarrouk, H.; Thabet, R.; Laouar, H.; Romdhane, N. Variability of Biological Features of a Mugilidae Chelon Ramada in Two Tunisian Reservoirs. J. Aqauc. Mar. Biol. 2022, 11, 41–48. [Google Scholar] [CrossRef]
- Rais, C.; Turki, I. Empoissonnement de la retenue du barrage de Bir M’Chergua par des mulets. Bull. Inst. Natl. Sci. Tech. Oceanogr. Pêche Salammbô 1989, 16, 43–53. [Google Scholar]
- Nau, W.; Ben Naceur, L. Socio-Economic Study of Mullet Fishing at the Bir M’Cherga dam (Technical Report of the Tunisian-German Inland Fisheries Project); GTZ: Bonn, Tunisia, 1990; Volume 3, pp. 1–51. [Google Scholar]
- Gophen, M.; Snovsky, G. Mugilids (Mugil cepalus, Linnaeus, 1758; Liza ramada, Risso, 1810) Stocking in Lake Kinneret. Open Jour. Ecol. 2015, 5, 389–399. [Google Scholar] [CrossRef] [Green Version]
- Hajlaoui, W.; Fatnassi, M.; Mili, S.; Ben Salem, S.; Troudi, D.; Missaoui, H. Characterization of socio-economic fishing activity in Tunisian reservoir: Sidi Saad (center of Tunisia) as a case study. J. Aquac. Mar. Biol. 2022, 11, 93–98. [Google Scholar]
- Farrugio, H. Mullets (Fish, Teleosts) of Tunisia: Distribution and Fishing, Contribution to Their Systematic and Biological Study. Ph.D. Thesis, Marine Biology and Océanography Diplomam, Faculty of Sciences of Tunis, Tunis, Tunisia, 1975. [Google Scholar]
- Chauvet, C. Exploitation of Fish in a Mediterranean Lagoon Environment. Dynamics of the Ichthyological Population of the Lagoon of Tunis and the Populations Exploited by Bordigues (Mullets, Wolves, Sea Bream). Ph.D. Thesis, University of Perpignan, Perpignan, France, 1986. [Google Scholar]
- Vidy, G.; Franc, J. Natural Resource in Mugilidae Fry in Tunisia; Scientific Report; Ministry of Agriculture (CGP-INSTOP); ORSTOM: Tunis, Tunisia, 1987; p. 213. [Google Scholar]
- Mili, S.; Bouriga, N.; Ennouri, R.; Laouer, H.; Missaoui, H. Identification, répartition spatio-temporelle et caractérisation génétique des alevins de Muges ensemencés dans les retenues de barrages tunisiennes. Cybium 2013, 37, 67–73. [Google Scholar]
- De Silva, S.S.; Funge-Smith, S.J. A Review of Stock Enhancement Practices in the Inland Water Fisheries of Asia; Asia-Pacific Fishery Commission—FAO: Bangkok, Thailand, 2005; p. 101. [Google Scholar]
- Bok, H. Extensive culture of two mullet species in freshwater impoundments in the eastern Cape, South. Afr. J. Zool. 1984, 19, 31–36. [Google Scholar]
- Besbes, R.; Besbes, B.A.; Kokokiris, L.; Changeux, T.; Hamza, A.; Kammoun, F.; Missaoui, H. Thicklip (Chelon labrosus) and flathead (Mugil cephalus) grey mullets fry production in Tunisian aquaculture. Aquac. Rep. 2020, 17, 100380. [Google Scholar] [CrossRef]
- El-Zarka, S.; Kamel, F. Mullet fry transportation and its contribution to the fisheries of inland brackish lakes in the United Arab Republic. Proc. Gen. Fish. Coun. Medit. 1965, 8, 209–226. [Google Scholar]
- Saleh, M. Capture-Based Aquaculture of Mullets in Egypt. Capture-Based Aquaculture: Global Overview; FAO Fisheries Technical Paper; FAO: Rome, Italy, 2008; Volume 508, pp. 109–126. [Google Scholar]
- Leber, K.M.; Lee, C.S.; Nathan, P.; Steve, M.B.; Arce, C.S.; Lee, T.H.; Blankenship Nishimoto, R.T. Biology, Ecology and Culture of Grey Mullets (Mugilidae), 1st ed.; CRC Press: Boca Raton, FL, USA, 2016; Volume 18, pp. 467–472. [Google Scholar]
- Jenhani, B.R.A.; Fathalli, A.; Djemali, I.; Changeux, T.; Romdhane, M.S. Tunisian reservoirs: Diagnosis and biological 1235 potentialities. Aquat. Living Resour. 2019, 32, 17. [Google Scholar] [CrossRef]
- Rhouma, A. Biological Study and Breeding of Mullet in Tunisia and Comparison with a Freshwater Species (Carp). Ph.D. Thesis, National Agronomic Institute of Tunisia, Tunis, Tunisia, 1975. [Google Scholar]
- Kraiem, M.M. Etude comparée de la croissance de différentes populations de Barbus callensis Valenciennes 1842, (Pisces, Cyprinidae), de Tunisie. Cybium 1989, 13, 365–374. [Google Scholar]
- Kraiem, M.M. Systématique, Biogéographie et Bioécologie de Barbus Callensis Valenciennes, 1842 (Poissons, Cyprinidae) de Tunisie. Ph.D. Thesis, University of Tunis, Tunis, Tunisia, 1994. [Google Scholar]
- Toujani, R. The Pike-Perch (Stizostedion lucioperca L.) from the Sidi-Salem Reservoir (Tunisia): Biology and Population Dynamics. Ph.D. Thesis, Claude Bernard University, Villeurbanne, France, 1998. [Google Scholar]
- Toujani, R.; Missaoui, H.; Romdhane, M.S. Sexual cycle of pike-perch females (Stizostedion lucioperca) in the reservoir of the Sidi Salem dam. Bull. Inst. Nat. Scien. Tech. Mer. 2000, 27, 85–96. [Google Scholar]
- M’Hetli, M. The Pike-Perch Stizostedion lucioperca (Linnaeus, 1758) Teleost Percidae. Allochthonous: Biological Study and Trial to Optimize Breeding Criteria. Ph.D. Thesis, University of Tunis, Tunis, Tunisia, 2001. [Google Scholar]
- Djemali, I. Assessment of Fish Biomass in Tunisian Freshwater Bodies: Analytical and Acoustic Approach. Ph.D. Thesis, National Agronomic Institute of Tunisia, Tunis, Tunisia, 2005. [Google Scholar]
- Tlili, S.; Jebali, J.; Banni, M.; Haouas, Z.; Mlayah, A.; Helal, A.N. Multimarker approach analysis in common carp Cyprinus carpio sampled from three freshwater sites. Environ. Monit. Assess. 2010, 168, 285–298. [Google Scholar] [CrossRef]
- Mili, S.; Ennouri, R.; Laouar, H.; Missaoui, H. Etude de l’âge et de la croissance chez deux espèces de Mugilidae (Mugil cephalus et Liza ramada) dans trois retenues de barrages en Tunisie. Bull. Soci. Zool. Fran. 2015, 140, 181–197. [Google Scholar]
- Instop. Programme de Recherche de L’INSTOP Sur La Retenue du Barrage de Bir M’Cherga; Ministère de l’agriculture, Institut National Scientifique et Technique d’Océanographie et de la Pêche: Tunis, Tunisia, 1989; p. 68. [Google Scholar]
- Djemali, I.; Toujani, R.; Guillard, J. Hydroacoustic fish biomass assessment in man-made lakes in Tunisia, horizontal beaming importance and diel effect. Aquat. Ecol. 2008, 43, 1121–1131. [Google Scholar] [CrossRef]
- Mili, S.; Ennouri, R.; Laouar, H.; Ben Romdhane, N.; Missaoui, H. Etude des peuplements piscicoles de la retenue du barrage de Sidi Salem. J. New Sci. Agri. Biotech. 2016, 27, 1454–1465. [Google Scholar]
- Mili, S.; Ennouri, R.; Laouar, H.; Ben Romdhane, N.; Djemali, I.; Toujani, R.; Missaoui, H. Etude des peuplements piscicoles de la retenue de barrage de Sidi Barrak. Bull. Inst. Natn. Tech. Mer De Salammbô 2016, 43, 55–69. [Google Scholar]
- McFarland, W.; Okubo, A. Animal Groups in Three Dimensions, Chapter Metabolic Models of Fish Behavior, the Need for Quantitative Observations; Cambridge University Press: Cambridge, UK, 1997; pp. 301–312. [Google Scholar]
- Quignard, J.P.; Farrugio, H. Age and Growth of Grey Mullet. In Aquaculture of Grey Mullets, International Biological Program 26 Cambridge; Cambridge University Press: Cambridge, UK, 1981; pp. 155–184. [Google Scholar]
- Fehri-Bedoui, R.; Gharbi, H. Age and growth of Liza aurata (Mugilidae) along Tunisian coasts. Cybium 2005, 29, 119–126. [Google Scholar]
- Gautier, D.; Hussenot, J. Les Mulets des Mers d’Europe: Synthèse des Connaissances Sur Les Bases Biologiques et Les Techniques d’Aquaculture, 1st ed.; Ifremer: Brest, France, 2005; p. 120. [Google Scholar]
- Kraiem, M.M.; Ben Hamza, C.; Ramdani, M.; Fathi, A.A.; Abdelzaher, H.M.A.; Flower, R.J. Some observations on the age and growth of thin-lipped grey mullet, Liza ramada Risso, 1826 (Pisces, Mugilidae) in three North African wetland lakes: Merja Zerga (Morocco), Garâat Ichkeul (Tunisia) and Edku lake (Egypt). Aqua. Ecol. 2001, 35, 335–345. [Google Scholar] [CrossRef]
- Albertini-Berhaut, J. Biology of the Juvenile Stages of Mugilidae in the Marseille Region: Growth, Diet and Digestive Enzymatic Activities. Ph.D. Thesis, Faculty of Sciences, University of Aix-Marseille II, Marseille, France, 1980. [Google Scholar]
- Bar-Ilan, M. Stocking of Mugil capito and Mugil cephalus and their commercial catch in Lake Kinneret. Aquaculture 1975, 5, 85–89. [Google Scholar] [CrossRef]
- Vidy, G.; Franc, J. Seasons of presence on the coast of mullet fry (Mugilidae) in Tunisia. Cybium 1992, 16, 53–71. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mili, S.; Ennouri, R.; Fatnassi, M.; Agrebi, S.; Louiz, I.; Khattab, Y.; Hedfi, A.; Ben Ali, M.; Laouar, H. Investigating Freshwater Mullet Fisheries in Tunisian Reservoirs: Future Development Prospects. Water 2023, 15, 2554. https://doi.org/10.3390/w15142554
Mili S, Ennouri R, Fatnassi M, Agrebi S, Louiz I, Khattab Y, Hedfi A, Ben Ali M, Laouar H. Investigating Freshwater Mullet Fisheries in Tunisian Reservoirs: Future Development Prospects. Water. 2023; 15(14):2554. https://doi.org/10.3390/w15142554
Chicago/Turabian StyleMili, Sami, Rym Ennouri, Manel Fatnassi, Siwar Agrebi, Ibtissem Louiz, Yassir Khattab, Amor Hedfi, Manel Ben Ali, and Houcine Laouar. 2023. "Investigating Freshwater Mullet Fisheries in Tunisian Reservoirs: Future Development Prospects" Water 15, no. 14: 2554. https://doi.org/10.3390/w15142554




