Health Risks to Communities and Athletes Associated with Swimming, Wading, and Sailing in Water Bodies of Brazil’s Guanabara Bay Basin
Abstract
:1. Background
2. Methods
2.1. Study Area
2.2. Population and Exposure Scenarios
2.3. Microorganisms Considered
2.4. Exposure Route Assumptions
2.5. Indicator Concentrations
2.6. Estimation of Pathogen Dose from Indicator Data
2.7. Dose–Response Model
| Microorganisms | Parameters | Model Form | References |
|---|---|---|---|
| E. coli (saltwater) | k = 1.4 × 10−4 | Exponential | Derived here based on data from Cabelli et al. (1982) [34] |
| E. coli (freshwater) | k = 5.1 × 10−5 | Exponential | Sunger (2013) [35] |
| Enterococci (saltwater) | k = 1.8 × 10−4 | Exponential | Sunger (2015) [36] |
| Campylobacter | α = 1.6 × 10−1 N50 = 8.9 × 102 PDI = 0.3 | beta-Poisson | Haas (2014) [22] |
| Salmonella | α = 3.1 × 10−1 N50 = 2.4 × 104 PDI = 3.0 × 10−1 | beta-Poisson | Haas (2014) [22] |
| Rotavirus | α = 2.5 × 10−1 N50 = 6.2 PDI = 5.0 × 10−1 | beta-Poisson | Haas (2014) [22] |
| Cryptosporidium | k = 4.2 × 10−3 PDI = 7.0 × 10−1 | Exponential | Teunis (1996) [38] |
| Ascaris | k = 3.9 × 10−2 PDI = 3.9 × 10−1 | Exponential | Navarro (2009) [39] |
2.8. Disease Risk Estimation
3. Results
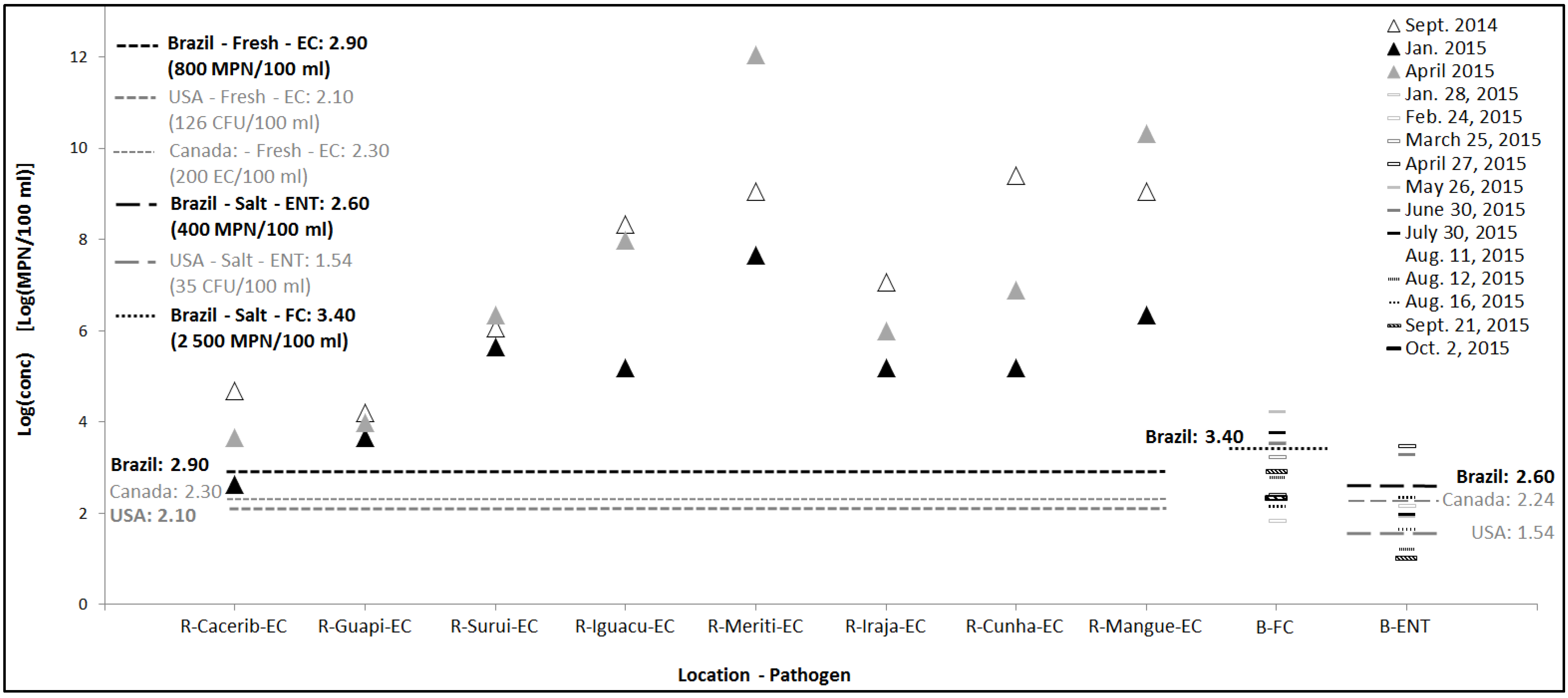
3.1. Concentration of Indicator Organisms in Environmental Samples
3.2. Risk Estimation
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fistarol, G.O.; Coutinho, F.H.; Moreira, A.P.B.; Venas, T.; Cánovas, A.; De Paula, S.E.M.; Coutinho, R.; De Moura, R.L.; Valentin, J.L.; Tenenbaum, D.R.; et al. Environmental and sanitary conditions of Guanabara Bay, Rio de Janeiro. Front. Microbiol. 2015, 6, 1232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- CDC Healthy Swimming—Diarrheal Illness. Available online: http://www.cdc.gov/healthywater/swimming/swimmers/rwi/diarrheal-illness.html (accessed on 21 June 2016).
- Dufour, A.; Cabelli, V.; Ericksen, T.; Ballantine, R.; Goldberg, M.; Fox, W. Ambient Water Quality Criteria for Bacteria. In Bacteriological Ambient Water Quality Criteria for Marine and Fresh Recreational Waters; United States Environmental Protection Agency: Washington, DC, USA, 1986. [Google Scholar]
- Korajkic, A.; McMinn, B.R.; Harwood, V.J. Relationships between Microbial Indicators and Pathogens in Recreational Water Settings. Int. J. Environ. Res. Public Health 2018, 15, 2842. [Google Scholar] [CrossRef] [Green Version]
- Directive 2006/7/EC of the European Parliament and of the Council of 15 February 2006 concerning the management of bathing water quality and repealing Directive 76/160/EEC. Off. J. Eur. Union 2006, 64, 37–51.
- EPA. 2012 Recreational Water Quality Criteria; United States Environmental Protection Agency: Washington, DC, USA, 2012. [Google Scholar]
- Federal-Provincial-Territorial Working Group on Recreational Water Quality (Canada); Health Canada. Guidelines for Canadian Recreational Water Quality; Water, Air and Climate Change Bureau Healthy Environments and Consumer Safety Branch; Health Canada: Ottawa, ON, Canada, 2012. [Google Scholar]
- WHO. Guidelines for Safe Recreational Water Environments. Available online: http://www.who.int/water_sanitation_health/publications/srwe1/en/ (accessed on 30 September 2020).
- EPA. Total Coliform Rule: A Quick Reference Guide; United States Environmental Protection Agency: Washington, DC, USA, 2013. [Google Scholar]
- CONAMA. Current CONAMA Resolutions Published between September 1984 and January 2012; Conselho Nacional do Meio Ambiente: Brasilia, Brasil, 2012. [Google Scholar]
- CONAMA. CONAMA Resolution No 274/2000; Conselho Nacional do Meio Ambiente: Brasilia, Brasil, 2000; Volume 53. [Google Scholar]
- CONAMA. CONAMA Resolution No 357/2005; Conselho Nacional do Meio Ambiente: Brasilia, Brasil, 2005. [Google Scholar]
- Ford, B. Rio Olympic Games—The Promise Rio Couldn’t Keep; ESPN: Bristol, CT, USA, 2016. [Google Scholar]
- Balch, O. Funding Problems Hit Plan to Clean Rio’s Polluted Waterways Ahead of Olympics. Guardian Sustainable Business. The Guardian, 1 February 2016. Available online: https://www.theguardian.com/sustainable-business/2016/feb/01/funding-problems-hit-plan-clean-rios-polluted-waterways-olympics (accessed on 8 July 2023).
- Fidalgo, M. Rio’s “Bay of All Delights”: The Polluted Waters of the 2016 Olympics. Vice, 2 March 2016. [Google Scholar]
- INEA. Boletim de Qualidade das Aguas da Baia de Guanabara (No 02/16). Area de Provas Olimpicas—2016 (Bacteriologia); INEA: Rio De Janeiro, Brazil, 2016. [Google Scholar]
- Demographia. World Urban Areas & Population Projections; Demographia: Belleville, IL, USA, 2015. [Google Scholar]
- SEMADS. Bacias Hidrográficas e Rios Fluminenses: Síntese Informativa por Macroregião Ambiental; Secretaria de Estado de Meio Ambiente e Desenvolvimento Sustentável: Rio de Janeiro, Brazil, 2001; ISBN 8587206109. [Google Scholar]
- Ministério da Saúde. Guia de Vigilância Epidemiológica; Ministério da Saúde: Brasilia, Brazil, 2009; ISBN 9788533416321. [Google Scholar]
- Evison, L.M. Comparative Studies on the Survival of Indicator Organisms and Pathogens in Fresh and Sea Water. Water Sci. Technol. 1988, 20, 309–315. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking-Water Quality: First Addendum to the Third Edition; Volume 1: Recommendations; WHO: Geneva, Switzerland, 2006; ISBN 978-92-4-154696-6. [Google Scholar]
- Haas, C.N.; Rose, J.B.; Gerba, C.P. Quantitative Microbial Risk Assessment, 2nd ed.; John Wiley & Sons: New York, NY, USA, 2014; ISBN 978-1-118-14529-6. [Google Scholar]
- Katukiza, A.Y.; Ronteltap, M.; Niwagaba, C.B.; Foppen, J.W.A.; Kansiime, F.; Lens, P.N.L. Sustainable sanitation technology options for urban slums. Biotechnol. Adv. 2012, 30, 964–978. [Google Scholar] [CrossRef]
- Kundu, A.; Poma, H.R.; Jenkins, M.W.; Rajal, V.B.; Wuertz, S. QMRA of intestinal nematode infection via multimedia exposure pathways. In Proceedings of the7th International Congress on Environmental Modelling and Software, San Diego, CA, USA, 6 June 2014. [Google Scholar]
- Shuval, H.; Lampert, Y.; Fattal, B. Development of a risk assessment approach for evaluating wastewater reuse standards for agriculture. Water Sci. Technol. 1997, 35, 15–20. [Google Scholar] [CrossRef]
- Mara, D.D.; Sleigh, P.A.; Blumenthal, U.J.; Carr, R.M. Health risks in wastewater irrigation: Comparing estimates from quantitative microbial risk analyses and epidemiological studies. J. Water Health 2007, 5, 39–50. [Google Scholar] [CrossRef] [Green Version]
- Labite, H.; Lunani, I.; Van Der Steen, P.; Vairavamoorthy, K.; Drechsel, P.; Lens, P. Quantitative microbial risk analysis to evaluate health effects of interventions in the urban water system of Accra, Ghana. J. Water Health 2010, 8, 417–430. [Google Scholar] [CrossRef] [Green Version]
- Howard, G.; Ahmed, F.M.; Teunis, P.; Gaifur Mahmud, S.; Davison, A.; Deere, D. Disease burden estimation to support policy decision-making and research prioritization for arsenic mitigation. J. Water Health 2007, 5, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Fattal, B.; Peleg-olevsky, E.; Agursky, T.; Shuvsl, P.H.I. The association between seawater pollution as measured by bacterial indicators and morbidity among bathers at Mediterranean bathing beaches of Israel. Chemosphere 1987, 16, 565–570. [Google Scholar] [CrossRef]
- Schets, F.M.; Schijven, J.F.; De Roda Husman, A.M. Exposure assessment for swimmers in bathing waters and swimming pools. Water Res. 2011, 45, 2392–2400. [Google Scholar] [CrossRef]
- Dorevitch, S.; Panthi, S.; Huang, Y.; Li, H.; Michalek, A.M.; Pratap, P.; Wroblewski, M.; Liu, L.; Scheff, P.A.; Li, A. Water ingestion during water recreation. Water Res. 2011, 45, 2020–2028. [Google Scholar] [CrossRef]
- Westrell, T.; Schönning, C.; Stenström, T.A.; Ashbolt, N.J. QMRA (quantitative microbial risk assessment) and HACCP (hazard analysis and critical points) for management of pathogens in wastewater and sewage sludge treatment and reuse. Water Sci. Technol. 2004, 50, 23–30. [Google Scholar] [CrossRef] [Green Version]
- Aquece-Rio. International Sailing Regatta 2015; Notice of Race; Aquece-Rio: Rio de Janeiro, Brazil, 2015. [Google Scholar]
- Dufour, A.P. Health Effects Criteria for Fresh Recreational Waters; United States Environmental Protection Agency: Washington, DC, USA, 1984. [Google Scholar]
- Cabelli, V.J.; Dufour, A.P.; McCABE, L.J.; Levin, M.A. Swimming-Associated Gastroenteritis and Water Quality. Am. J. Epidemiol. 1982, 115, 606–616. [Google Scholar] [CrossRef]
- Sunger, N. Quantitative Health Risk Assessment of Recreational Water Users in Philadelphia. Ph.D. Thesis, Department of Civil, Architectural, and Environmental Engineering, Drexel University, Philadelphia, PA, USA, 2013. [Google Scholar]
- Anderson, I.C.; Rhodes, M.; Kator, H. Sublethal stress in Escherichia coli: A function of salinity. Appl. Environ. Microbiol. 1979, 38, 1147–1152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teunis, P.F.M.; Van der Heijden, O.G.; Van der Giessen, J.W.B.; Havelaar, A.H. The Dose-Response Relation in Human Volunteers for Gastro-Intestinal Pathogens; Rijksinstituut voor Volksgezondheid en Milieu: Bilthoven, The Netherlands, 1996. [Google Scholar]
- Navarro, I.; Jiménez, B.; Cifuentes, E.; Lucario, S. Application of Helminth ova infection dose curve to estimate the risks associated with biosolid application on soil. J. Water Health 2009, 7, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Coster, T.S.; Wolf, M.K.; Hall, E.R.; Cassels, F.J.; Taylor, D.N.; Liu, C.T.; Trespalacios, F.C.; DeLorimier, A.; Angleberger, D.R.; McQueen, C.E. Immune response, ciprofloxacin activity, and gender differences after human experimental challenge by two strains of enterotoxigenic Escherichia coli. Infect. Immun. 2007, 75, 252–259. [Google Scholar] [CrossRef] [Green Version]
- Levine, M.M.; Rennels, M.B.; Cisneros, L.; Hughes, T.P.; Nalin, D.R.; Young, C.R. Lack of person-to-person transmission of enterotoxigenic Escherichia coli despite close contact. Am. J. Epidemiol. 1980, 111, 347–355. [Google Scholar] [CrossRef]
- Rendtorff, R.C. The experimental transmission of human intestinal protozoan parasites. Am. J. Hyg. 1954, 9, 196–208. [Google Scholar]
- López-Pila, J.M.; Szewzyk, R. Estimating the infection risk in recreational waters from the faecal indicator concentration and from the ratio between pathogens and indicators. Water Res. 2000, 34, 4195–4200. [Google Scholar] [CrossRef]
- Pouillot, R.; Delignette-Muller, M.L. Evaluating variability and uncertainty separately in microbial quantitative risk assessment using two R packages. Int. J. Food Microbiol. 2010, 142, 330–340. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Guidelines for Drinking-Water Quality, 3rd ed.; Volume 1—Recommendations; WHO: Geneva, Switzerland, 2008; Volume 38. [Google Scholar]
- Hamilton, A.J.; Stagnitti, F.; Xiong, X.; Kreidl, S.L.; Benke, K.K.; Maher, P. Wastewater Irrigation: The State of Play. Vadose Zone J. 2007, 6, 823–840. [Google Scholar] [CrossRef] [Green Version]
- Sklar, R.S. Microbial Health Risks to Sanitation Workers in Low-Resource Settings: Incorporation of Field, Molecular, and Modeling Approaches. Ph.D. Thesis, UC Berkeley, Berkeley, CA, USA, 2020. Available online: https://escholarship.org/uc/item/11t4t1qs (accessed on 17 May 2023).

| Parameter | Description | Units | Distribution | References |
|---|---|---|---|---|
| CEC fresh | Concentration of E. coli in freshwater (from 8 rivers) | E. coli/100 mL | Lognormal μ = 3.6 × 109 σ =1.2 × 1011 | Field measurement (this study) |
| CFC salt Cent salt | Concentration of fecal coliforms and enterococci in saltwater bay, at Marina de Gloria | fecal coliform/100 mL | Lognormal distribution μ = 3.2 × 103 σ = 1.0 × 104 | INEA (2016) [17] |
| RFC:EC salt | E. coli/fecal coliform ratio in saltwater | -- | Point-value μ = 3.0 × 10−1 | Fattal (1987) [29] |
| REC:Pathogen | E. coli:pathogen ratios | -- | Lognormal distribution μ EC:Campylobacter = 105 μ EC:Salmonella = 105 μ EC:rotavirus = 105 μ EC:Cryptosporidium = 105 μ EC:Ascaris = 106 | Ratios used are from Labite (2010) [27], These agree with the upper bounds provided by Mara (2007) [26] for rotavirus, Cryptosporidium, and Campylobacter. They also agree with the general ratios for virus, bacteria, and protozoa provided by Howard (2007) [28]. |
| VIngestion Swimming | Mean volume of water ingested during Swimming | mL/event | Gamma distribution r = 0.45 λ = 60 | Schets (2011) [30] |
| VIngestion Wading | Mean volume of water ingested during Wading | mL/event | Point value μ = 3.2 mL | Dorevitch (2011) [31] Westrell (2004) [32] Labite (2010) [27] |
| VIngestion Sailing | Mean volume of water ingested during Sailing | mL/hour | Point value μ = 3.9 mL | Dorevitch (2011) [31] |
| t Sailing | Duration of the sailing event (time) | hours | Point value μ = 0.5 h | Aquece Rio (2015) [33] |
| Organisms– Water Type (Source) | N | Percentiles (MPN/100 mL) | Geometric Mean | Standard Deviation | ||
|---|---|---|---|---|---|---|
| 5th | 50th | 95th | ||||
| E. coli—freshwater (measured here) | 24 | 4.3 × 103 | 1.6 × 106 | 1.74 × 1010 | 3.20 × 106 | 2.20 × 1011 |
| Fecal coliform—bay (INEA 2016 [17]) | 12 | 9.9 × 101 | 7.9 × 102 | 9.72 × 103 | 8.61 × 102 | 4.24 × 103 |
| ENT—Saltwater (INEA 2016 [17]) | 11 | 1.0 × 101 | 7.9 × 102 | 2.26 × 103 | 7.7 × 101 | 9.1 × 101 |
| Exposure Scenarios (Water Type, Location) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Swimming (Fresh Water, Rivers) | Wading (Fresh Water, Rivers) | Sailing (Saltwater, Bay) | |||||||
| Risk Percentiles | Risk Percentiles | Risk Percentiles | |||||||
| Organisms | 5th | 50th | 95th | 5th | 50th | 95th | 5th | 50th | 95th |
| E. coli | 1.9 × 10−2 | 1.00 | 1.00 | 4.7 × 10−4 | 9.9 × 10−1 | 1.0 | 1.5 × 10−5 | 8.2 × 10−4 | 3.6 × 10−2 |
| Enterococci | - | - | - | - | - | - | 8.0 × 10−6 | 3.4 × 10−4 | 1.1 × 10−2 |
| ∑ Organisms | 1.8 × 10−1 | 5.9 × 10−1 | 8.8 × 10−1 | 1.1 × 10−2 | 3.6 × 10−1 | 7.5 × 10−1 | 4.2 × 10−5 | 1.0 × 10−3 | 3.5 × 10−2 |
| Campylobacter | 2.3 × 10−5 | 6.9 × 10−2 | 2.4 × 10−1 | 5.6 × 10−7 | 5.5 × 10−3 | 2.0 × 10−1 | 6.2 × 10−9 | 3.5 × 10−7 | 1.5 × 10−5 |
| Salmonella | 1.9 × 10−7 | 1.5 × 10−3 | 2.4 × 10−1 | 3.8 × 10−9 | 3.9 × 10−5 | 1.2 × 10−1 | 3.3 × 10−11 | 1.8 × 10−9 | 8.0 × 10−8 |
| Rotavirus | 1.4 × 10−3 | 3.5 × 10−1 | 4.9 × 10−1 | 3.1 × 10−5 | 1.4 × 10−1 | 4.6 × 10−1 | 2.9 × 10−7 | 1.8 × 10−5 | 8.3 × 10−4 |
| Cryptosporidium | 1.4 × 10−6 | 1.2 × 10−2 | 7.0 × 10−1 | 3.0 × 10−8 | 2.8 × 10−4 | 6.9 × 10−1 | 3.1 × 10−10 | 1.7 × 10−8 | 7.1 × 10−7 |
| Ascaris | 6.8 × 10−6 | 6.7 × 10−2 | 3.9 × 10−1 | 1.6 × 10−7 | 1.6 × 10−3 | 3.9 × 10−1 | 1.6 × 10−9 | 9.2 × 10−8 | 3.8 × 10−6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sklar, R.; Chabrelie, A.E.; Carreira, R.S.; Gurian, P.L.; Mitchell, J. Health Risks to Communities and Athletes Associated with Swimming, Wading, and Sailing in Water Bodies of Brazil’s Guanabara Bay Basin. Water 2023, 15, 2509. https://doi.org/10.3390/w15142509
Sklar R, Chabrelie AE, Carreira RS, Gurian PL, Mitchell J. Health Risks to Communities and Athletes Associated with Swimming, Wading, and Sailing in Water Bodies of Brazil’s Guanabara Bay Basin. Water. 2023; 15(14):2509. https://doi.org/10.3390/w15142509
Chicago/Turabian StyleSklar, Rachel, Alexander E. Chabrelie, Renato S. Carreira, Patrick L. Gurian, and Jade Mitchell. 2023. "Health Risks to Communities and Athletes Associated with Swimming, Wading, and Sailing in Water Bodies of Brazil’s Guanabara Bay Basin" Water 15, no. 14: 2509. https://doi.org/10.3390/w15142509







