Spinel-Ferrite-Decorated Graphene-Based Nanocomposites for Enhanced Photocatalytic Detoxification of Organic Dyes in Aqueous Medium: A Review
Abstract
:1. Introduction
2. Synthesis Protocols
3. Role of Decoration of Spinel Ferrites on Graphene in MFGN for Enhanced Photocatalytic-Dye Detoxification
3.1. Textural and Microstructural Properties of MFGN
3.2. Separation and Migration of Photoinduced Charge Carriers
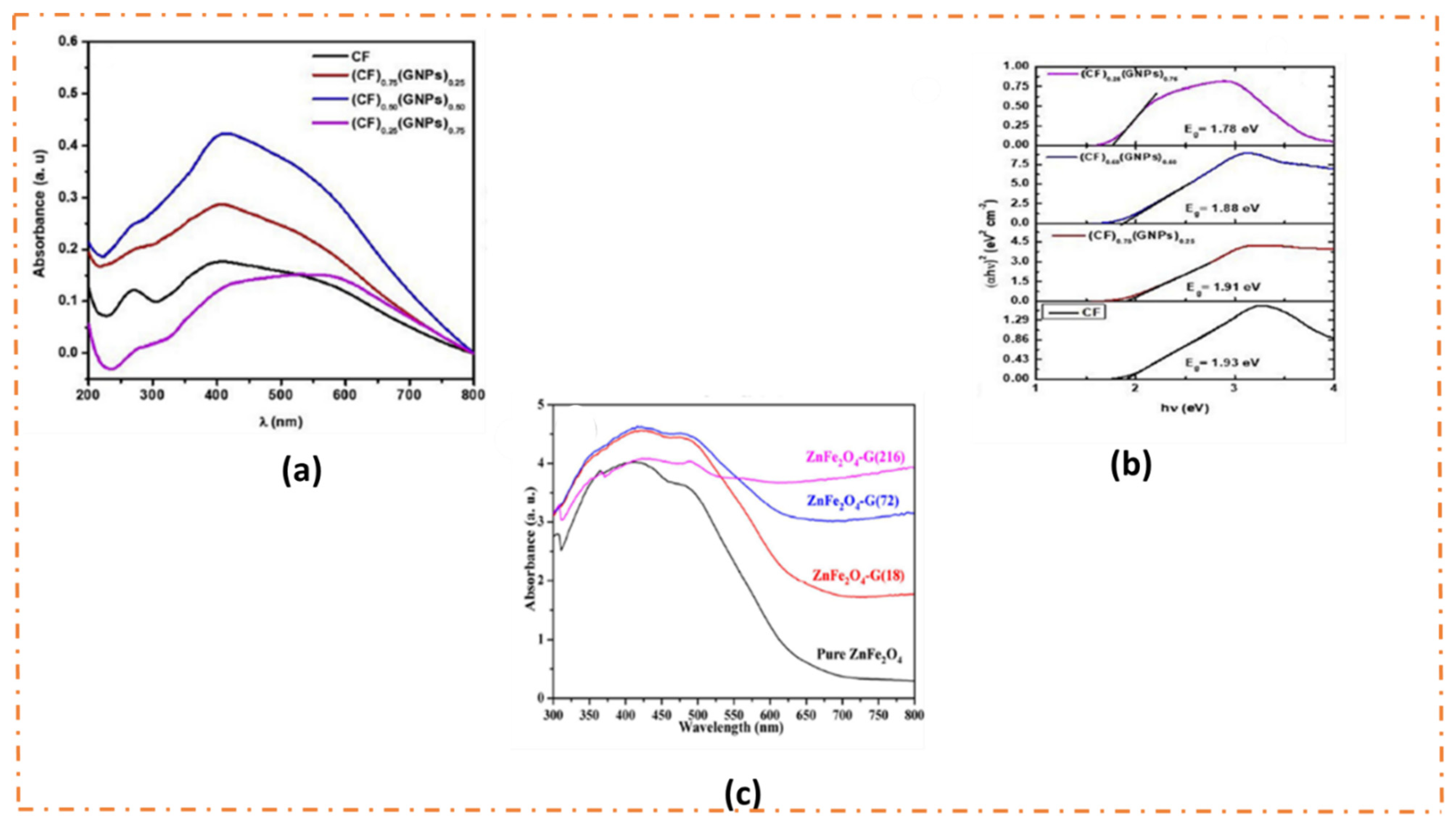
3.3. Light Absorption Range
4. Fundamental Mechanisms for Photocatalytic-Dye Detoxification over MFGN
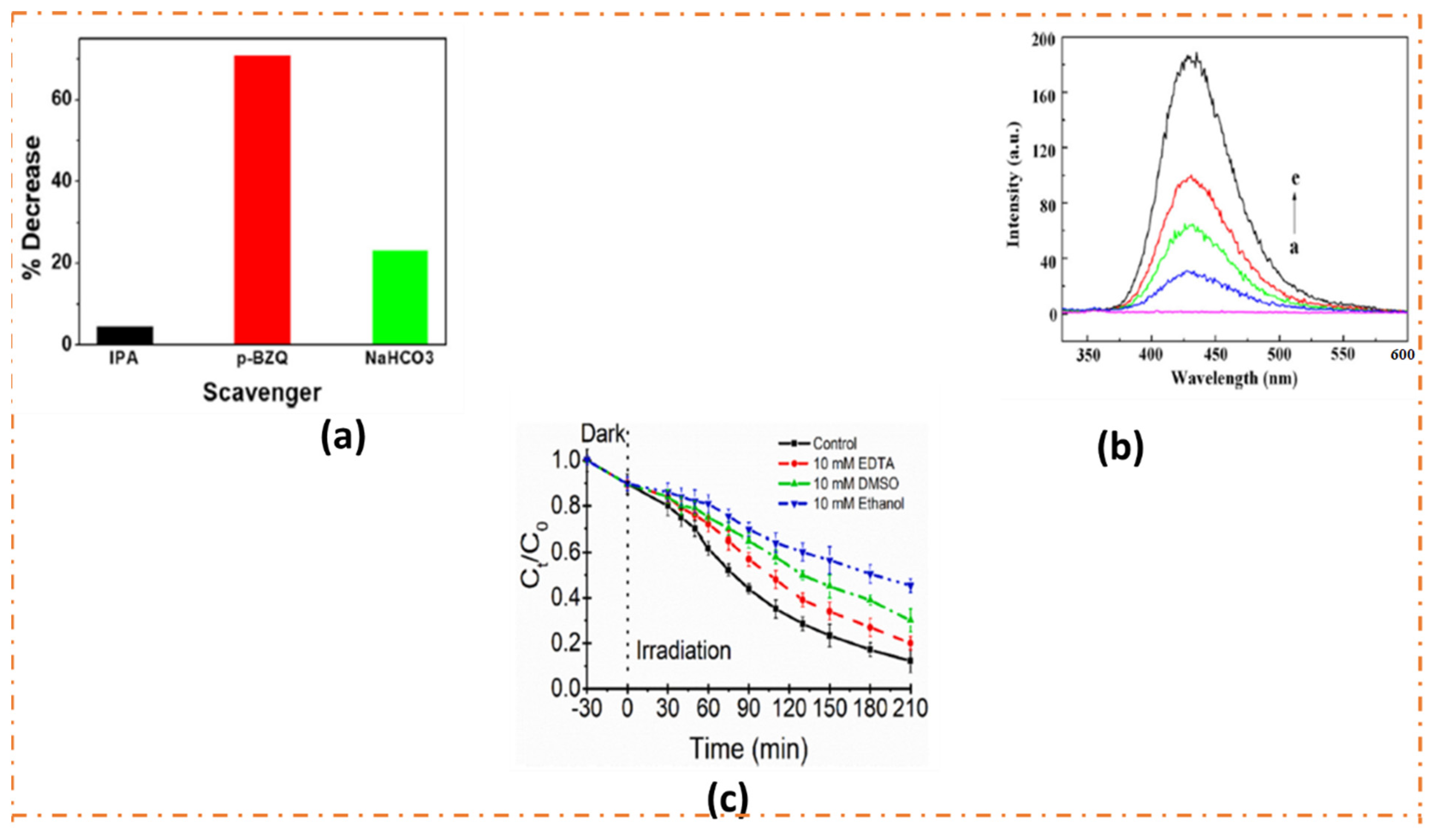
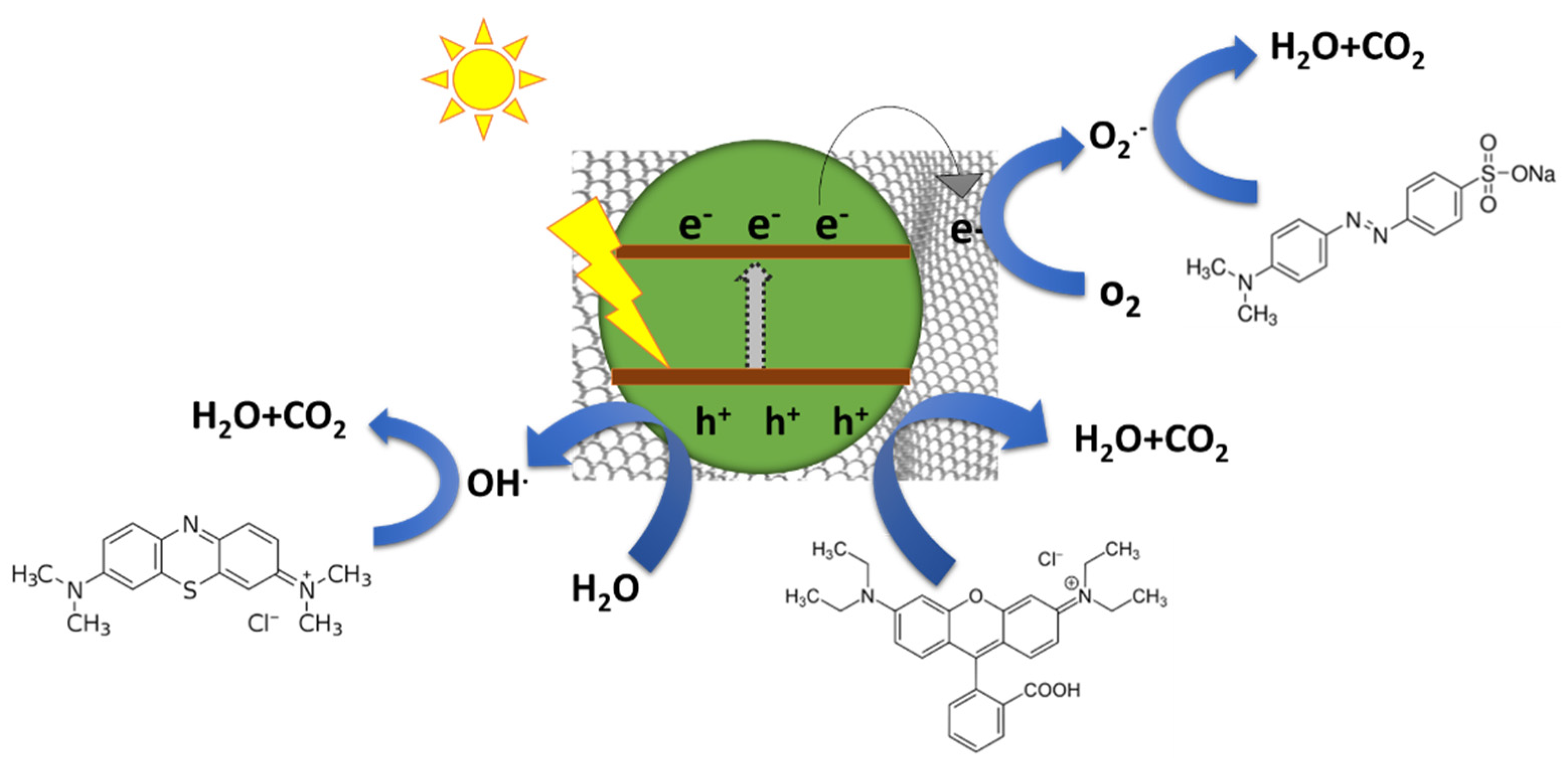
4.1. Mechanism for Photodetoxification of Dyes over MFGNs with Strong Reduction Ability
4.2. Mechanism for Photodetoxification of Dyes over MFGNs with Strong Oxidation Ability
4.3. Mechanism for Photodegradation of Dyes over MFGNs with Moderate Redox Ability
5. Reaction Kinetics for Photocatalytic-Dye Degradation over MFGN
6. Impact of Reaction Parameters on Photocatalytic-Dye Detoxification by MFGNs
6.1. pH of the Solution
6.2. Temperature
6.3. Catalyst Dosage
6.4. Effect of Dye Concentration
7. Comparison of Photocatalytic Performance of MFGNs for Dye Detoxification
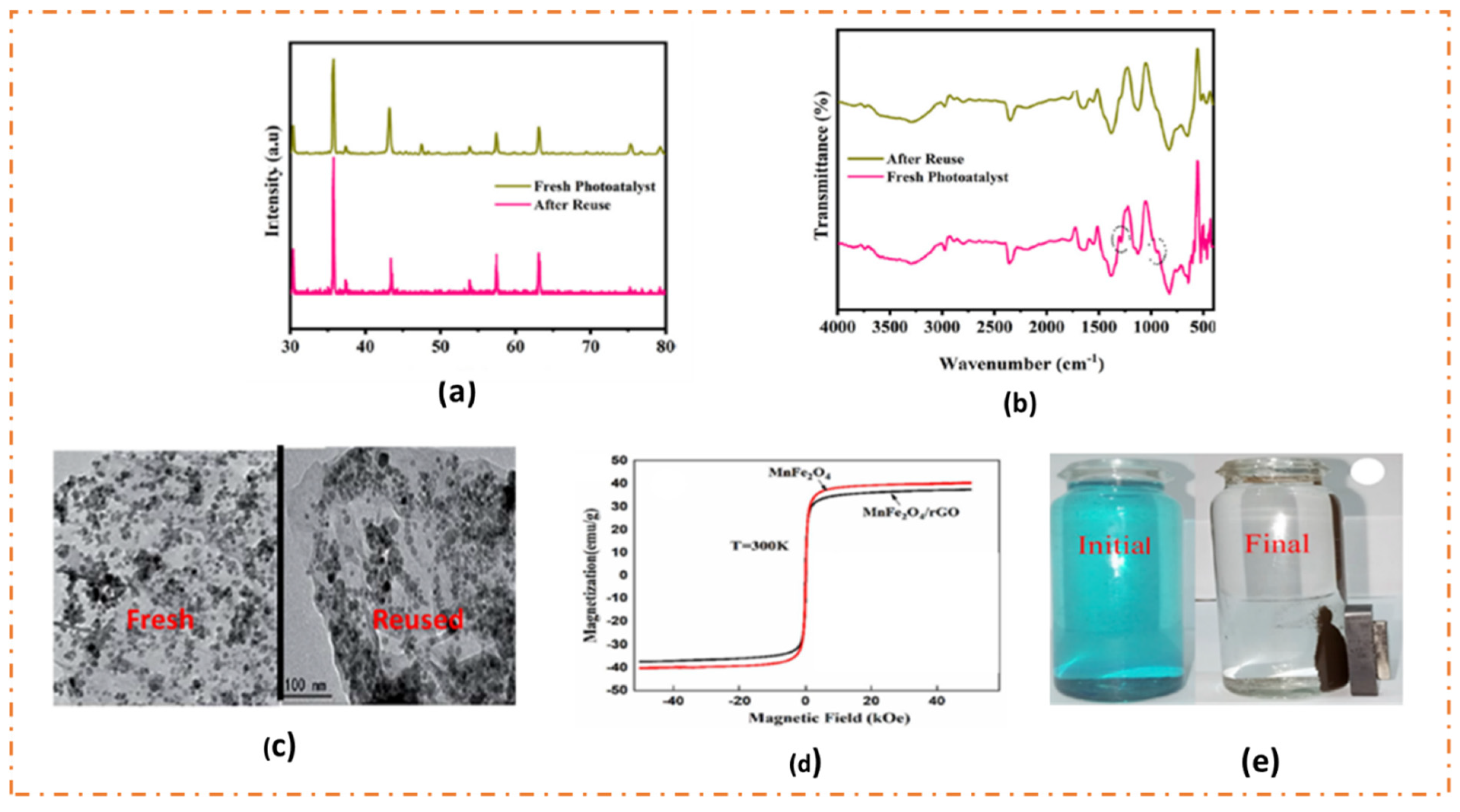
8. Stability and Recoverability
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, C.; Ma, W.; Zhao, J. Semiconductor-mediated photodegradation of pollutants under visible-light irradiation. Chem. Soc. Rev. 2010, 39, 4206. [Google Scholar] [CrossRef] [PubMed]
- Adeleke, J.T.; Theivasanthi, T.; Thiruppathi, M.; Swaminathan, M.; Akomolafe, T.; Alabi, A.B. Photocatalytic degradation of methylene blue by ZnO/NiFe2O4 nanoparticles. Appl. Surf. Sci. 2018, 455, 195–200. [Google Scholar] [CrossRef] [Green Version]
- Rafiq, A.; Ikram, M.; Ali, S. Photocatalytic degradation of dyes using semiconductor photocatalysts to clean industrial water pollution. J. Indust. Eng. Chem. 2021, 97, 111–128. [Google Scholar] [CrossRef]
- Katheresan, V.; Kansedo, J.; Lau, S.Y. Efficiency of various recent wastewater dye removal methods: A review. J. Environ. Chem. Eng. 2018, 6, 4676–4697. [Google Scholar] [CrossRef]
- Ahmad, A.; Hamidah, S.; Mohd-Setapar, S.H.; Chuong, C.S.; Khatoon, A.; Wani, W.A.; Kumard, R.; Rafatullah, M. Recent advances in new generation dye removal technologies: Novel search for approaches to reprocess wastewater. RSC Adv. 2015, 5, 30801. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interf. Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef]
- Dutta, S.; Gupta, B.; Srivastava, S.K.; Gupta, A.K. Recent advances on the removal of dyes from wastewater using various adsorbents: A critical review. Adv. Mater. 2021, 2, 4497–4531. [Google Scholar] [CrossRef]
- Pattnaik, S.P.; Behera, A.; Acharya, R.; Parida, K. Green exfoliation of graphitic carbon nitride towards decolourization of Congo-Red under solar irradiation. J. Environ. Chem. Eng. 2019, 7, 103456. [Google Scholar] [CrossRef]
- Pattnaik, S.P.; Behera, A.; Martha, S.; Acharya, R.; Parida, K. Synthesis, photoelectrochemical properties and solar light-induced photocatalytic activity of bismuth ferrite nanoparticles. J. Nanopart. Res. 2018, 20, 10. [Google Scholar] [CrossRef]
- Frank, S.N.; Bard, A.J. Heterogeneous photocatalytic oxidation of cyanide ion in aqueous solutions at TiO2 powder. J. Am. Chem. Soc. 1977, 99, 303–304. [Google Scholar] [CrossRef]
- Rauf, M.A.; Ashraf, S.S. Fundamental principles and application of heterogeneous photocatalytic degradation of dyes in solution. Chem. Eng. J. 2009, 151, 10–18. [Google Scholar] [CrossRef]
- Friedmann, D. A General Overview of Heterogeneous Photocatalysis as a Remediation Technology for Wastewaters Containing Pharmaceutical Compounds. Water 2022, 14, 3588. [Google Scholar] [CrossRef]
- Acharya, R.; Naik, B.; Parida, K. Cr(VI) remediation from aqueous environment through modified-TiO2-mediated photocatalytic reduction. Beilstein J. Nanotechnol. 2018, 9, 1448–1470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mansingh, S.; Sultana, S.; Acharya, R.; Ghosh, M.K.; Parida, K. Efficient photon conversion via double charge dynamics CeO2–BiFeO3 p–n heterojunction photocatalyst promising toward N2 fixation and Phenol–Cr(VI) detoxification. Inorg. Chem. 2020, 59, 3856–3873. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.M.; Lai, C.W.; Ngai, K.S.; Juan, J.C. Recent developments of zinc oxide based photocatalyst in water treatment technology: A review. Water Res. 2015, 88, 428–448. [Google Scholar] [CrossRef] [PubMed]
- Acharya, L.; Swain, G.; Mishra, B.P.; Acharya, R.; Parida, K. Development of MgIn2S4 microflower-embedded exfoliated B-doped g-C3N4 nanosheets: P–n heterojunction photocatalysts toward photocatalytic water reduction and H2O2 production under visible-light irradiation. ACS Appl. Energy Mater. 2022, 5, 2838–2852. [Google Scholar] [CrossRef]
- Antoniadou, M.; Arfanis, M.K.; Ibrahim, I.; Falaras, P. Bifunctional g-C3N4/WO3 thin films for photocatalytic water purification. Water 2019, 11, 2439. [Google Scholar] [CrossRef] [Green Version]
- Lei, D.; Xue, J.; Peng, X.; Lia, S.; Bi, Q.; Tang, C. Oxalate enhanced synergistic removal of chromium(VI) and arsenic(III) over ZnFe2O4/g-C3N4: Z-scheme charge transfer pathway and photo-Fenton like reaction. Appl. Catal. B. 2021, 282, 119578. [Google Scholar] [CrossRef]
- Jo, W.-K.; Moru, S.; Tonda, S. Magnetically responsive SnFe2O4/g-C3N4 hybrid photocatalysts with remarkable visible-light-induced performance for degradation of environmentally hazardous substances and sustainable hydrogen production. Appl. Surf. Sci. 2020, 506, 144939. [Google Scholar] [CrossRef]
- Kefeni, K.K.; Msagatia, T.A.M.; Nkambule, T.T.; Mamba, B.B. Spinel ferrite nanoparticles and nanocomposites for biomedical applications and their toxicity. Mater. Sci. Eng. C. 2020, 107, 110314. [Google Scholar] [CrossRef]
- Kefeni, K.K.; Mamba, B.B.; Msagatia, T.A.M. Application of spinel ferrite nanoparticles in water and wastewater treatment: A review. Separ. Purif. Technol. 2017, 188, 399–422. [Google Scholar] [CrossRef]
- Xiang, Q.; Yu, J.; Jaroniec, M. Graphene-based semiconductor photocatalysts. Chem. Soc. Rev. 2012, 41, 782–796. [Google Scholar] [CrossRef]
- Li, X.; Yu, J.; Wageh, S.; Al-ghamdi, A.A.; Xie, J. Graphene in Photocatalysis: A Review. Small 2016, 12, 6640. [Google Scholar] [CrossRef]
- Yang, D.; Feng, J.; Jiang, L.; Wu, X.; Sheng, L.; Jiang, Y.; Wei, T.; Fan, Z. Photocatalyst interface engineering: Spatially confined growth of ZnFe2O4 within graphene networks as excellent visible-light-driven photocatalysts. Adv. Funct. Mater. 2015, 25, 7080–7087. [Google Scholar] [CrossRef]
- Fu, Y.S.; Wang, X. Magnetically separable ZnFe2O4-graphene catalyst and its high photocatalytic performance under visible light irradiation. Ind. Eng. Chem. Res. 2011, 50, 7210–7218. [Google Scholar] [CrossRef]
- Hou, Y.; Li, X.; Zhao, Q.; Chen, G. ZnFe2O4 multi-porous microbricks/graphene hybrid photocatalyst: Facile synthesis, improved activity and photocatalytic mechanism. Appl. Catal. B 2013, 142–143, 80–88. [Google Scholar] [CrossRef]
- Behera, A.; Kandi, D.; Mansingh, S.; Martha, S.; Parida, K. Facile synthesis of ZnFe2O4@rGO nanocomposites towards photocatalytic ciprofloxacin degradation and H2 energy production. J. Coll. Interf. Sci. 2019, 556, 667–679. [Google Scholar] [CrossRef]
- Karim, K.M.R.; Tarek, M.; Ong, H.R.; Abdullah, H.; Yousuf, A.; Cheng, C.K.; Khan, M.M.R. Photoelectrocatalytic Reduction of Carbon Dioxide to Methanol Using CuFe2O4 Modified with Graphene Oxide under Visible Light Irradiation. Ind. Eng. Chem. Res. 2019, 58, 563–572. [Google Scholar] [CrossRef]
- Mekasuwandumronga, O.; Pawinrat, P.; Praserthdamb, P.; Panpranot, J. Effects of synthesis conditions and annealing post-treatment on the photocatalytic activities of ZnO nanoparticles in the degradation of methylene blue dye. Chem. Eng. J. 2020, 164, 77–84. [Google Scholar] [CrossRef]
- Fei, P.; Wang, Q.; Zhong, M.; Su, B. Preparation and adsorption properties of enhanced magnetic zinc ferrite-reduced graphene oxide nanocomposites via a facile one-pot solvothermal method. J. Alloy. Compd. 2016, 685, 411–417. [Google Scholar] [CrossRef]
- Yin, W.; Haoab, S.; Cao, H. Solvothermal synthesis of magnetic CoFe2O4/rGO nanocomposites for highly efficient dye removal in wastewater. RSC Adv. 2017, 7, 4062. [Google Scholar] [CrossRef] [Green Version]
- Hu, L.; Li, M.; Cheng, L.; Jiang, A. Solvothermal synthesis of octahedral and magnetic CoFe2O4–reduced graphene oxide hybrids and their photo-Fenton-like behavior under visible light irradiation. RSC Adv. 2021, 11, 22250. [Google Scholar] [CrossRef]
- Meidanchi, A.; Akhavan, O. Superparamagnetic zinc ferrite spinel–graphene nanostructures for fast wastewater purification. Carbon 2014, 69, 230–238. [Google Scholar] [CrossRef]
- Kumar, S.; Nair, R.R.; Pillai, P.B.; Gupta, S.N.; Iyengar, M.A.R.; Sood, A.K. Graphene oxide−MnFe2O4 magnetic nanohybrids for efficient removal of lead and arsenic from water. ACS Appl. Mater. Interf. 2014, 6, 17426–17436. [Google Scholar] [CrossRef] [PubMed]
- Rostami, M.; Zamani, R.M.; Aghajanzadeh, K.M.; Danafar, H. Sol–gel synthesis and characterization of zinc ferrite–graphene nano-hybrids for photo-catalytic degradation of the paracetamol. J. Pharm. Investig. 2016, 48, 657–664. [Google Scholar] [CrossRef]
- He, G.; Ding, J.; Zhang, J.; Hao, Q.; Chen, H. One-step ball-milling preparation of highly photocatalytic active CoFe2O4-reduced graphene oxide heterojunctions for organic dye removal. Ind. Eng. Chem. Res. 2015, 54, 2862–2867. [Google Scholar] [CrossRef]
- Kumar, S.; Kumar, A.; Bahuguna, A.; Sharma, V.; Krishnan, V. Two-dimensional carbon-based nanocomposites for photocatalytic energy generation and environmental remediation applications. Beilstein J. Nanotechnol. 2017, 8, 1571–1600. [Google Scholar] [CrossRef] [Green Version]
- Mansingh, S.; Acharya, R.; Martha, S.; Parida, K. Pyrochlore Ce2Zr2O7 decorated over rGO: A photocatalyst that proves to be efficient towards the reduction of 4-nitrophenol and degradation of ciprofloxacin under visible light. Phys. Chem. Chem. Phys. 2018, 20, 9872–9885. [Google Scholar] [CrossRef]
- Moussa, H.; Girot, E.; Mozet, K.; Alem, H.; Medjahdi, G.; Schneider, R. ZnO rods/reduced graphene oxide composites prepared via a solvothermal reaction for efficient sunlight-driven photocatalysis. Appl. Catal. B. 2016, 185, 11. [Google Scholar] [CrossRef]
- Mahdikhah, V.; Saadatkia, S.; Sheibani, S.; Ataie, A. Outstanding photocatalytic activity of CoFe2O4 /rGO nanocomposite in degradation of organic dyes. Opt. Mater. 2020, 108, 110193. [Google Scholar] [CrossRef]
- Baynosa, M.L.; Mady, A.H.; Nguyen, V.Q.; Kumar, D.R.; Sayed, M.S.; Tuma, D.; Shim, J.-J. Eco-friendly synthesis of recyclable mesoporous zinc ferrite @ reduced graphene oxide nanocomposite for efficient photocatalytic dye degradation under solar radiation. J. Colloid Interface Sci. 2020, 561, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Low, J.; Yu, J.; Jaroniec, M.; Wageh, S.; Al-Ghamdi, A.A. Heterojunction photocatalysts. Adv. Mater. 2017, 29, 1601694. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Shen, R.; Ma, S.; Chen, X.; Xie, J. Graphene-based heterojunction photocatalysts. Appl. Surf. Sci. 2018, 430, 53–107. [Google Scholar] [CrossRef]
- Liu, S.-Q.; Zhu, X.-L.; Zhou, Y.; Meng, Z.-D.; Chen, Z.-G.; Liu, C.-B.; Chen, F.; Wu, Z.-Y.; Qian, J.-C. Smart photocatalytic removal of ammonia through molecular recognition of zinc ferrite/reduced graphene oxide hybrid catalyst under visible-light irradiation. Catal. Sci. Technol. 2017, 7, 3210–3219. [Google Scholar] [CrossRef]
- Mokhtar, M.; Islam, M.; Tarek, I.; Salama, M. Rational design of manganese ferrite-graphene hybrid photocatalysts: Efficient water splitting and effective elimination of organic pollutants. Appl. Catal. A Gen. 2016, 524, 182–191. [Google Scholar]
- Nazim, S.; Kousar, T.; Shahid, M.; Khan, M.A.; Nasara, G.; Sher, M.; Warsia, M.F. New graphene-CoxZn1−xFe2O4 nano-heterostructures: Magnetically separable visible light photocatalytic materials. Ceram. Int. 2016, 42, 7647–7654. [Google Scholar] [CrossRef]
- Fu, Y.; Xiong, P.; Chen, H.; Sun, X.; Wang, X. High photocatalytic activity of magnetically separable manganese ferrite-graphene hetero-architectures. Ind. Eng. Chem. Res. 2012, 51, 725–731. [Google Scholar] [CrossRef]
- Zhang, H.; Lv, X.; Li, Y.; Wang, Y.; Li, J. P25-graphene composite as a high performance photocatalyst. ACS Nano 2010, 4, 380–386. [Google Scholar] [CrossRef]
- Mady, A.H.; Baynosa, M.L.; Tumac, D.; Shim, J.-J. Facile microwave-assisted green synthesis of Ag-ZnFe2O4@rGO nanocomposites for efficient removal of organic dyes under UV- and visible-light irradiation. Appl. Catal. B 2017, 203, 416–427. [Google Scholar] [CrossRef]
- Soltani, T.; Lee, B.-K. Sono-synthesis of nanocrystallized BiFeO3/reduced graphene oxide composites for visible photocatalytic degradation improvement of bisphenol A. Chem. Eng. J. 2016, 306, 204–213. [Google Scholar] [CrossRef]
- Israr, M.; Iqbal, J.; Arshad, A.; Gomez-Romero, P. Sheet-on-sheet like calcium ferrite and graphene nanoplatelets nanocomposite: A multifunctional nanocomposite for high-performance supercapacitor and visible light driven photocatalysis. J. Solid State Chem. 2021, 293, 121646. [Google Scholar] [CrossRef]
- Ai, J.; Hu, L.; Zhou, Z.; Cheng, L.; Liu, W.; Su, K.; Zhang, R.; Chen, Z.; Li, W. Surfactant-free synthesis of a novel octahedral ZnFe2O4/graphene composite with high adsorption and good photocatalytic activity for efficient treatment of dye wastewater. Ceram. Int. 2020, 46, 11786–11798. [Google Scholar] [CrossRef]
- Liang, J.; Wei, Y.; Zhang, J.; Yao, Y.; He, G.; Tang, B.; Chen, H. Scalable green method to fabricate magnetically separable NiFe2O4-reduced graphene oxide nanocomposites with enhanced photocatalytic performance driven by visible light. Ind. Eng. Chem. Res. 2018, 57, 4311–4319. [Google Scholar] [CrossRef]
- Moitra, D.; Chandel, M.; Ghosh, B.K.; Jani, R.K.; Patra, M.K.; Vadera, S.R.; Ghosh, N.N. A simple ‘in situ’ co-precipitation method for preparation of multifunctional CoFe2O4-reduced graphene oxide nanocomposites: Excellent microwave absorber and highly efficient magnetically separable recyclable photocatalyst for dye degradation. RSC Adv. 2016, 6, 76759–76772. [Google Scholar] [CrossRef]
- Dong, J.; Chen, G.; Sun, J.; Li, C.; Yu, Y.; Chen, D. A novel high-efficiency visible-light sensitive Ag2CO3 photocatalyst with universal photodegradation performances: Simple synthesis, reaction mechanism and first-principles study. Appl. Catal. B 2013, 134–135, 46–54. [Google Scholar] [CrossRef]
- Acharya, R.; Parida, K. A review on TiO2/g-C3N4 visible-light- responsive photocatalysts for sustainable energy generation and environmental remediation. J. Environ. Chem. Eng. 2020, 8, 103896. [Google Scholar] [CrossRef]
- Zou, L.; Wang, H.; Yuan, G.; Wang, X. Magnetically separable CdS/ZnFe2O4 composites with highly efficient photocatalytic activity and photostability under visible light. ACS Appl. Nano Mater. 2018, 1, 831–838. [Google Scholar] [CrossRef]
- Costa, R.C.C.; Lelis, M.F.F.; Oliveira, L.C.A. Novel active heterogeneous Fenton system based on Fe3-xMxO4 (Fe, Co, Mn, Ni): The role of M2+ species on the reactivity towards H2O2 reactions. J. Hazard. Mater. B 2006, 129, 171–178. [Google Scholar] [CrossRef]
- Sharma, R.; Bansal, S.; Singhal, S. Tailoring the photo- fenton activity of spinel ferrites (MFe2O4) by incorporating different cations (M¼ Cu, Zn, Ni and Co) in the structure. RSC Adv. 2015, 5, 6006–6018. [Google Scholar] [CrossRef]
- Chen, P.; Xing, X.; Xie, H.; Sheng, Q.; Qu, H. High catalytic activity of magnetic CuFe2O4/graphene oxide composite for the degradation of organic dyes under visible light irradiation. Chem. Phys. Let. 2016, 660, 176–181. [Google Scholar] [CrossRef]
- Lu, D.; Zhang, Y.; Lin, S.; Wang, L.; Wang, C. Synthesis of magnetic ZnFe2O4/graphene composite and its application in photocatalytic degradation of dyes. J. Alloys Compd. 2013, 579, 336–342. [Google Scholar] [CrossRef]
- Jiang, R.; Zhu, H.; Fu, Y.; Jiang, S.; Zong, E.; Yao, J. Photocatalytic decolorization of congo red wastewater by magnetic ZnFe2O4/graphene nanosheets composite under simulated solar light irradiation. Ozone Sci. Eng. 2020, 42, 174–182. [Google Scholar] [CrossRef]
- Tamilselvi, R.; Lekshmi, G.S.; Padmanathan, N.; Selvaraj, V.; Bazaka, O.; Levchenko, I.; Bazaka, K.; Mandhakini, M. NiFe2O4/rGO nanocomposites produced by soft bubble assembly for energy storage and environmental remediation. Renew. Energy 2022, 181, 1386–1401. [Google Scholar] [CrossRef]
- Rahman, A.; Warsia, M.F.; Shakir, I.; Shahid, M.; Zulfiqar, S. Fabrication of Ce3+ substituted nickel ferrite-reduced graphene oxide heterojunction with high photocatalytic activity under visible light irradiation. J. Hazard. Mater. 2020, 394, 122593. [Google Scholar] [CrossRef]
- Wei, Z.; Huang, S.; Zhang, X.; Lu, C.; He, Y. Hydrothermal synthesis and photo-Fenton degradation of magnetic MnFe2O4/rGO nanocomposites. J. Mater. Sci. Mater. Electron. 2020, 31, 5176–5186. [Google Scholar] [CrossRef]
- He, H.-Y.; Lu, J. Highly photocatalytic activities of magnetically separable reduced graphene oxide-CoFe2O4 hybrid nanostructures in dye photodegradation. Sep. Purif. Technol. 2017, 172, 374–381. [Google Scholar] [CrossRef]
- Mazarji, M.; Esmaili, H.; Bidhendi, G.N.; Mahmoodi, N.M.; Minkina, T.; Sushkova, S.; Mandzhieva, S.; Barakhov, A.; Moghtaderi, H.; Bhatnagar, A. Green synthesis of reduced graphene oxide-CoFe2O4 nanocomposite as a highly efficient visible-light-driven catalyst in photocatalysis and photo Fenton-like reaction. Mater. Sci. Eng. B 2021, 270, 115223. [Google Scholar] [CrossRef]
- Klamerth, N.; Malato, S.; Agüera, A.; Fernández-Alba, A.; Mailhot, G. Treatment of municipal wastewater treatment plant effluents with modified photo-fenton as a tertiary treatment for the degradation of micro pollutants and disinfection. Environ. Sci. Technol. 2012, 46, 2885–2892. [Google Scholar] [CrossRef]
- Guan, Y.-H.; Ma, J.; Ren, Y.-M.; Liu, Y.-L.; Xiao, J.-Y.; Lin, L.-Q.; Zhang, C. Efficient degradation of atrazine by magnetic porous copper ferrite catalyzed peroxymonosulfate oxidation via the formation of hydroxyl and sulfate radicals. Water Res. 2013, 47, 5431–5438. [Google Scholar] [CrossRef]
- Ren, F.; Zhu, W.; Zhao, J.; Liu, H.; Zhang, X.; Zhang, H.; Zhu, H.; Peng, Y.; Wang, B. Nitrogen-doped graphene oxide aerogel anchored with spinel CoFe2O4 nanoparticles for rapid degradation of tetracycline. Sep. Purif. Technol. 2020, 241, 116690. [Google Scholar] [CrossRef]
- Zhang, Y.; Cheng, Y.; Qi, H. Synergistic degradation of organic pollutants on CoFe2O4/rGO nanocomposites by peroxymonosulfate activation under LED irradiation. Appl. Surf. Sci. 2022, 579, 152151. [Google Scholar] [CrossRef]
- Fu, Y.; Chen, H.; Sun, X.; Wang, X. Combination of cobalt ferrite and graphene: High-performance and recyclable visible-light photocatalysis. Appl. Catal. B Environ. 2012, 111–112, 280–287. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, Q.; Wang, L.; Zhang, L. Magnetically separable CdFe2O4/graphene catalyst and its enhanced photocatalytic properties. J. Mater. Chem. A 2015, 3, 3576–3585. [Google Scholar] [CrossRef]
- Yao, Y.; Cai, Y.; Lu, F.; Wei, F.; Wang, X.; Wang, S. Magnetic recoverable MnFe2O4 and MnFe2O4-graphene hybrid as heterogeneous catalysts of peroxymonosulfate activation for efficient degradation of aqueous organic pollutants. J. Hazard. Mater. 2014, 270, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Zou, J.; Lin, J.; Lia, J.; Huang, H.; Zhang, S.; Yuan, B.; Ma, J. Sodium hydroxide-enhanced acetaminophen elimination in heat/peroxymonosulfate system: Production of singlet oxygen and hydroxyl radical. Chem. Eng. J. 2022, 429, 132438. [Google Scholar] [CrossRef]
- Lin, J.; Zou, J.; Cai, H.; Huang, Y.; Li, J.; Xiao, J.; Yuan, B.; Ma, J. Hydroxylamine enhanced Fe(II)-activated peracetic acid process for diclofenac degradation: Efficiency, mechanism and effects of various parameters. Water Res. 2021, 207, 117796. [Google Scholar] [CrossRef]
- Huang, Y.; Zou, J.; Lin, J.; Yang, H.; Wang, M.; Li, J.; Cao, W.; Yuan, B.; Ma, J. ABTS as Both Activator and Electron Shuttle to Activate Persulfate for Diclofenac Degradation: Formation and Contributions of ABTS•+, SO4•–, and •OH. Environ. Sci. Technol. 2022. [Google Scholar] [CrossRef]
- Huang, Y.; Lin, Y.; Zou, J.; Xu, J.; Wang, M.; Cai, H.; Yuan, B.; Ma, J. ABTS as an electron shuttle to accelerate the degradation of diclofenac with horseradish peroxidase-catalyzed hydrogen peroxide oxidation. Sci. Total Environ. 2021, 798, 149276. [Google Scholar] [CrossRef]
- Daneshvar, N.; Salari, D.; Khataee, A.R. Photocatalytic degradation of azo dye acid red 14 in water: Investigation of the effect of operational parameters. J. Photochem. Photobiol. A 2003, 157, 111–116. [Google Scholar] [CrossRef]
- You-ji, L.; Wei, C. Photocatalytic degradation of rhodamine B using nanocrystalline TiO2–zeolite surface composite catalysts: Effects of photocatalytic condition on degradation efficiency. Catal. Sci. Technol. 2011, 1, 802–809. [Google Scholar] [CrossRef]
- Bayantong, A.R.B.; Shih, Y.-J.; Dong, C.D.; Garcia-Segura, S.; Luna, M.D.G. Nickel ferrite nanoenabled graphene oxide (NiFe2O4@GO) as photoactive nanocomposites for water treatment. Environ Sci. Pollut. Res. 2020, 28, 5472–5481. [Google Scholar] [CrossRef] [PubMed]
- Mozia, S.; Morawski, A.W.; Toyoda, M.; Tsumura, T. Effect of process parameters on photodegradation of Acid Yellow 36 in a hybrid photocatalysis–membrane distillation system. Chem. Eng. J. 2009, 150, 152. [Google Scholar] [CrossRef]
- Kodasma, R.; Palas, B.; Ersöz, G.; Atalay, S. Photocatalytic activity of copper ferrite graphene oxide particles for an efficient catalytic degradation of Reactive Black 5 in water. Ceram. Int. 2020, 46, 6284–6292. [Google Scholar] [CrossRef]
- Krishnan, S.; Murugesan, S.; Vasanthakumar, V.; Priyadharsan, A.; Alsawalha, M.; Alomayri, T.; Yuan, B. Facile green synthesis of ZnFe2O4/rGO nanohybrids and evaluation of its photocatalytic degradation of organic pollutant, photo antibacterial and cytotoxicity activities. Colloids Surf. A: Physicochem. Eng. Asp. 2021, 611, 125835. [Google Scholar]
- Xu, L.; Wang, J. Magnetic nanoscaled Fe3O4/CeO2 composite as an efficient Fenton-like heterogeneous catalyst for degradation of 4-chlorophenol. Environ. Sci. Technol. 2012, 46, 10145–10153. [Google Scholar] [CrossRef]
- Lu, H.; Salabas, E.E.; Schüth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244. [Google Scholar] [CrossRef]
- Gan, L.; Shang, S.; Wah, C.; Yuen, M.; Jiang, S.-X.; Hu, E. Hydrothermal synthesis of magnetic CoFe2O4/graphene nanocomposites with improved photocatalytic activity. Appl. Surf. Sci. 2015, 351, 140–147. [Google Scholar] [CrossRef]
- Rani, G.J.; Jothirajan, M.A.; Gnanakumar, G. Reduced graphene oxide/ZnFe2O4 nanocomposite as an efficient catalyst for the photocatalytic degradation of methylene blue dye. Res. Chem. Intermed. 2017, 43, 2669–2690. [Google Scholar] [CrossRef]
- Mandal, B.; Panda, J.; Paul, P.K.; Sarkar, R.; Tudu, B. MnFe2O4 decorated reduced graphene oxide heterostructures: Nanophotocatalyst for methylene blue dye degradation. Vacuum 2019, 173, 109150. [Google Scholar] [CrossRef]
- Rahman, A.; Aadil, M.; Akhtar, M.; Warsi, M.F.; Jamil, A.; Shakir, I.; Shahid, M. Magnetically recyclable Ni1−xCdxCeyFe2−yO4-rGO nanocomposite photocatalyst for visible light driven photocatalysis. Ceram. Int. 2020, 46, 13517–13526. [Google Scholar] [CrossRef]








| SFGNs | Targeted Dye | Catalyst Dosage (gL−1) | Initial Dye Conc. (mgL−1) | Degradation Efficiency (%) | Number of Times Increase in Activity w.r.t. Pristine SF | References |
|---|---|---|---|---|---|---|
| CoFe2O4/rGO | BR46 | 0.02 | 20 | 62 | 1.45 | [67] |
| CoFe2O4/rGO | BR18 | 0.02 | 20 | 79 | 2.25 | [67] |
| ZnFe2O4/rGO | MB | 0.5 | 10 | 98.0 | 7.6 | [41] |
| MnFe2O4/GO | MB | 0.3 | 10 | 97 | 1.15 | [89] |
| MnFe2O4/GO | MB | 0.25 | 20 | 100 | 1.2 | [47] |
| CaFe2O4/G | MB | 0.2 | 10 | 99.4 | 1.7 | [51] |
| CdFe2O4/G | MB | 1 | 10 | 89.22 | 2.2 | [73] |
| CoFe2O4/GO | MB | 0.25 | 10 | ~100 | 2.9 | [87] |
| ZnFe2O4/G | Congo red | 0.5 | 15 | 92.21 | 2.3 | [62] |
| MnFe2O4/rGO | Orange II | 0.05 | 20 | 90 | 1.6 | [74] |
| Ce-doped NiFe2O4/rGO | MB | 0.1 | 10 | 94.67 | 1.8 | [64] |
| ZnFe2O4/G | RhB | 1 | 20 | 100 | 3.3 | [61] |
| ZnFe2O4/rGO | MB | 0.5 | 50 | 92.4 | 1.04 | [84] |
| MnFe2O4/rGO | MB | 0.4 | - | 62 | 1.7 | [65] |
| MnFe2O4/GO | MB | 1 | 10 | 100 | 4 | [45] |
| ZnFe2O4/G | MB | 0.2 | 50 | 84 | 1.7 | [24] |
| CoFe2O4/rGO | MB | 0.5 | 5 | 74 | ~1.4 | [40] |
| NiFe2O4/rGO | MB | 0.25 | 20 | 99.1 | ~90 | [53] |
| CoFe2O4/rGO | MB | 0.25 | 20 | 100 | - | [36] |
| CoFe2O4/rGO | RhB | 0.25 | 20 | 72.2 | - | [36] |
| CoFe2O4/rGO | MO | 0.25 | 20 | 37.5 | - | [36] |
| CoFe2O4/G | MB | 0.25 | 20 | 100 | ~10 | [72] |
| CoFe2O4/G | RhB | 0.25 | 20 | 94 | - | [72] |
| CoFe2O4/G | MO | 0.25 | 20 | 71 | - | [72] |
| CoFe2O4/G | Active-black BL-G | 0.25 | 20 | 66 | - | [72] |
| CoFe2O4/G | Active-red RGB | 0.25 | 20 | 61 | - | [72] |
| CoFe2O4/rGO | MB | 1 | 10 | 73 | ~20 | [32] |
| CoFe2O4/rGO | MO | 0.5 | 20 | 100 | - | [54] |
| CoFe2O4/rGO | MB | 0.5 | 30 | 100 | - | [54] |
| CoFe2O4/rGO | RhB | 0.5 | 25 | 100 | - | [54] |
| CoFe2O4/rGO | RhB | 0.2 | 10 | ~100 | ~4 | [71] |
| NiFe2O4/rGO | MG | 0.2 | - | 96.5 | ~1.5 | [63] |
| ZnFe2O4/Graphene | MO | 1 | 15 | 5 | - | [52] |
| ZnFe2O4/Graphene | MB | 1 | 15 | 56 | - | [52] |
| ZnFe2O4/Graphene | RhB | 1 | 15 | 15 | - | [52] |
| NiFe2O4/GO | MB | 0.5 | - | ~40 | - | [81] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mishra, S.; Acharya, R.; Parida, K. Spinel-Ferrite-Decorated Graphene-Based Nanocomposites for Enhanced Photocatalytic Detoxification of Organic Dyes in Aqueous Medium: A Review. Water 2023, 15, 81. https://doi.org/10.3390/w15010081
Mishra S, Acharya R, Parida K. Spinel-Ferrite-Decorated Graphene-Based Nanocomposites for Enhanced Photocatalytic Detoxification of Organic Dyes in Aqueous Medium: A Review. Water. 2023; 15(1):81. https://doi.org/10.3390/w15010081
Chicago/Turabian StyleMishra, Subhasish, Rashmi Acharya, and Kulamani Parida. 2023. "Spinel-Ferrite-Decorated Graphene-Based Nanocomposites for Enhanced Photocatalytic Detoxification of Organic Dyes in Aqueous Medium: A Review" Water 15, no. 1: 81. https://doi.org/10.3390/w15010081
APA StyleMishra, S., Acharya, R., & Parida, K. (2023). Spinel-Ferrite-Decorated Graphene-Based Nanocomposites for Enhanced Photocatalytic Detoxification of Organic Dyes in Aqueous Medium: A Review. Water, 15(1), 81. https://doi.org/10.3390/w15010081









