Screening for Antibiotics and Their Degradation Products in Surface and Wastewaters of the POCTEFA Territory by Solid-Phase Extraction-UPLC-Electrospray MS/MS
Abstract
:1. Introduction
2. Experimental
2.1. Sampling
2.2. Standards and Chemicals
2.3. Solid-Phase Extraction (SPE)
2.4. Targeted Analysis by LC-MS/MS
2.5. Detection and Identification of Antibiotic Degradation Products by LC-High Resolution MS/MS
2.6. Method Validation
2.7. ANOVA Test
3. Results
3.1. Quantitative Analysis of Antibiotics: Method Development and Validation
3.2. Quantification of Antibiotics in Surface Waters
3.3. Concentrations of Antibiotics Measured near to Wastewater Treatment Plants (WWTP)
3.4. Analysis for Antibiotic Degradation Products
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Group | Antibiotic | Recovery (%) | Matrix Effect (%) | Linearity | ||||
|---|---|---|---|---|---|---|---|---|
| 0.01 | 0.05 | 0.1 | River Water | Outlet-WWTP * | Inlet-WWTP * | |||
| florfenicol metabolite | florfenicol amine | 70 ± 18 | 75 ± 14 | 72 ± 17 | 90 | 101 | 106 | 0.998 |
| ß-Lactamase | amoxicillin | 51 ± 18 | 55 ± 12 | 58 ± 15 | 85 | 78 | 70 | 0.998 |
| ampicillin | 51 ± 15 | 55 ± 12 | 55 ± 11 | 77 | 75 | 71 | 0.998 | |
| ß-Lactamase AMX degradation product | diketopiperazine | 60 ± 11 | 62 ± 13 | 63 ± 11 | 76 | 78 | 70 | 0.998 |
| penicilloic acid | 68 ± 18 | 61 ± 12 | 61 ± 12 | 86 | 83 | 80 | 0.998 | |
| diaminopyrimidine | trimethoprim | 51 ± 12 | 49 ± 12 | 59 ± 16 | 77 | 81 | 75 | 0.998 |
| fluoroquinolone | ciprofloxacin | 90 ± 8 | 91± 12 | 99 ± 11 | 99 | 112 | 125 | 0.998 |
| enrofloxacin | 77± 11 | 80± 15 | 81 ± 11 | 86 | 80 | 75 | 0.998 | |
| moxifloxacin | 83 ± 15 | 81 ± 11 | 87 ± 7 | 90 | 85 | 80 | 0.998 | |
| norfloxacin | 69 ± 11 | 77 ± 15 | 81 ± 9 | 83 | 80 | 81 | 0.998 | |
| lincosamide | lincomycin | 71 ± 17 | 81 ± 15 | 82 ± 11 | 91 | 87 | 85 | 0.998 |
| macrolides | azithromycin | 69 ± 11 | 71 ± 13 | 68 ± 13 | 86 | 80 | 75 | 0.998 |
| clarithromycin | 99 ± 11 | 104 ± 9 | 114 ± 9 | 100 | 113 | 122 | 0.998 | |
| clarithromycin N-oxide | 40 ± 16 | 44 ± 11 | 44 ± 14 | 76 | 80 | 75 | 0.999 | |
| erythromycin | 43 ± 11 | 44 ± 12 | 50 ± 9 | 80 | 80 | 76 | 0.998 | |
| sulfonamide | dapsone | 70 ± 18 | 75 ± 14 | 72 ± 17 | 90 | 100 | 107 | 0.998 |
| sulfacetamide | 91 ± 7 | 92 ± 11 | 91 ± 8 | 89 | 90 | 78 | 0.998 | |
| sulfadiazine | 92 ± 7 | 93 ± 11 | 94 ± 10 | 87 | 88 | 80 | 0.998 | |
| sulfadoxine | 93 ± 12 | 93 ± 10 | 90 ± 10 | 87 | 80 | 78 | 0.998 | |
| sulfamerazine | 90 ± 7 | 89 ± 10 | 89 ± 7 | 91 | 90 | 87 | 0.998 | |
| sulfapyridine | 95 ± 5 | 90 ± 11 | 93 ± 13 | 92 | 89 | 77 | 0.998 | |
| sulfamethoxazole | 91 ± 10 | 95 ± 5 | 95 ± 10 | 97 | 90 | 91 | 0.998 | |
| sulfamethoxypyridazine | 99 ± 7 | 95 ± 11 | 94 ± 13 | 95 | 90 | 89 | 0.998 | |
| tetracycline | oxytetracycline | 91 ± 11 | 90 ± 10 | 90 ± 10 | 88 | 85 | 80 | 0.998 |
| Group | Antibiotic | LOD µg L−1 | LOQ µg L−1 |
|---|---|---|---|
| florfenicol metabolite | florfenicol amine | 0.093 | 0.306 |
| ß-Lactamase | amoxicillin | 0.152 | 0.500 |
| ampicillin | 0.021 | 0.071 | |
| ß-Lactamase AMX degradation product | diketopiperazine | 0.202 | 0.610 |
| penicilloic acid | 0.151 | 0.460 | |
| diaminopyrimidine | trimethoprim | 0.026 | 0.085 |
| fluoroquinolone | ciprofloxacin | 0.285 | 0.094 |
| enrofloxacin | 0.041 | 0.135 | |
| moxifloxacin | 0.011 | 0.035 | |
| norfloxacin | 0.264 | 0.871 | |
| lincosamide | lincomycin | 0.025 | 0.082 |
| macrolides | azithromycin | 0.020 | 0.067 |
| clarithromycin | 0.018 | 0.059 | |
| clarithromycin N-oxide | 0.012 | 0.038 | |
| erythromycin | 0.024 | 0.072 | |
| sulfonamide | dapsone | 0.029 | 0.095 |
| sulfacetamide | 0.049 | 0.163 | |
| sulfadiazine | 0.046 | 0.152 | |
| sulfadoxine | 0.012 | 0.038 | |
| sulfamerazine | 0.230 | 0.750 | |
| sulfapyridine | 0.029 | 0.097 | |
| sulfamethoxazole | 0.008 | 0.025 | |
| sulfamethoxypyridazine | 0.060 | 0.197 | |
| tetracycline | oxytetracycline | 0.028 | 0.094 |
| Sources of Variation | Sum of Squares | Degree of Freedom | Mean Square | Factor F | p-Value |
|---|---|---|---|---|---|
| Between rivers | 21.642.135 | 1 | 21.642.135 | F = 5.27929 | 0.024 |
| Within rivers | 344.353.214 | 84 | 4.099.443 | ||
| Total | 365.995.349 | 85 |
| Group | Antibiotic | Cmax Spain (ng L−1) | Reference | Cmax France (ng L−1) | Reference |
|---|---|---|---|---|---|
| ß-Lactamase | amoxicillin | n/d | 68 | [71] | |
| ampicillin | n/d | n/d | |||
| fluoroquinolone | ciprofloxacin | 740 | [44] | 9660 | [42] |
| enrofloxacin | 178 | [31] | n/d | ||
| moxifloxacin | 205 | [72] | n/d | ||
| norfloxacin | n/d | n/d | 163 | [45] | |
| lincosamide | lincomycin | 47 | [44] | n/d | |
| macrolides | azithromycin | 28 | [71] | n/d | |
| erythromycin | 70 | [72] | 4 | [71] | |
| clarithromycin | 91 | [44] | 2330 | [42] | |
| sulfonamide | sulfadiazine | 2312 | [73] | n/d | |
| sulfamerazine | n/d | n/d | |||
| sulfamethoxazole | 11,000 | [73] | 544 | [45] | |
| sulfapyridine | 12,000 | [74] | 1 | [75] | |
| diaminopyrimidine | trimethoprim | 252 | [76] | 20 | [75] |
References
- Cacace, D.; Fatta-Kassinos, D.; Manaia, C.M.; Cytryn, E.; Kreuzinger, N.; Rizzo, L.; Karaolia, P.; Schwartz, T.; Alexander, J.; Merlin, C.; et al. Antibiotic resistance genes in treated wastewater and in the receiving water bodies: A pan-European survey of urban settings. Water Res. 2019, 162, 320–330. [Google Scholar] [CrossRef]
- World Health Organization. WHO Global Strategy for Containment of Antimicrobial Resistance; WHO/CDS/CS; World Health Organisatin: Geneva, Switzerland, 2001; pp. 1–105. [Google Scholar]
- Gothwal, R.; Shashidhar, T. Antibiotic Pollution in the Environment: A Review. Clean Soil Air Water 2015, 43, 479–489. [Google Scholar] [CrossRef]
- He, Y.; Yuan, Q.; Mathieu, J.; Stadler, L.; Senehi, N.; Sun, R.; Alvarez, P.J.J. Antibiotic resistance genes from livestock waste: Occurrence, dissemination, and treatment. Npj Clean Water 2020, 3, 4. [Google Scholar] [CrossRef] [Green Version]
- Matthiessen, P.; Arnold, D.; Johnson, A.C.; Pepper, T.J.; Pottinger, T.G.; Pulman, K.G.T. Contamination of headwater streams in the United Kingdom by oestrogenic hormones from livestock farms. Sci. Total Environ. 2006, 367, 616–630. [Google Scholar] [CrossRef]
- Quaik, S.; Embrandiri, A.; Ravindran, B.; Hossain, K.; Al-Dhabi, N.A.; Arasu, M.V.; Ignacimuthu, S.; Ismail, N. Veterinary antibiotics in animal manure and manure laden soil: Scenario and challenges in Asian countries. J. King Saud Univ. Sci. 2020, 32, 1300–1305. [Google Scholar] [CrossRef]
- Kim, H.; Hong, Y.; Park, J.E.; Sharma, V.K.; Cho, S. Il Sulfonamides and tetracyclines in livestock wastewater. Chemosphere 2013, 91, 888–894. [Google Scholar] [CrossRef]
- Ghauch, A.; Tuqan, A.M.; Kibbi, N. Ibuprofen removal by heated persulfate in aqueous solution: A kinetics study. Chem. Eng. J. 2012, 197, 483–492. [Google Scholar] [CrossRef]
- Saitoh, T.; Shibata, K.; Fujimori, K.; Ohtani, Y. Rapid removal of tetracycline antibiotics from water by coagulation-flotation of sodium dodecyl sulfate and poly(allylamine hydrochloride) in the presence of Al(III) ions. Sep. Purif. Technol. 2017, 187, 76–83. [Google Scholar] [CrossRef]
- Yuan, S.F.; Liu, Z.H.; Yin, H.; Dang, Z.; Wu, P.X.; Zhu, N.W.; Lin, Z. Trace determination of sulfonamide antibiotics and their acetylated metabolites via SPE-LC-MS/MS in wastewater and insights from their occurrence in a municipal wastewater treatment plant. Sci. Total Environ. 2019, 653, 815–821. [Google Scholar] [CrossRef]
- Barbosa, M.O.; Ribeiro, A.R.; Ratola, N.; Hain, E.; Homem, V.; Pereira, M.F.R.; Blaney, L.; Silva, A.M.T. Spatial and seasonal occurrence of micropollutants in four Portuguese rivers and a case study for fluorescence excitation-emission matrices. Sci. Total Environ. 2018, 644, 1128–1140. [Google Scholar] [CrossRef]
- Díaz-Bao, M.; Barreiro, R.; Miranda, J.M.; Cepeda, A.; Regal, P. Fast HPLC-MS/MS method for determining penicillin antibiotics in infant formulas using molecularly imprinted solid-phase extraction. J. Anal. Methods Chem. 2015, 2015, 959675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moles, S.; Mosteo, R.; Gómez, J.; Szpunar, J.; Gozzo, S.; Castillo, J.R.; Ormad, M.P. Towards the removal of antibiotics detected in wastewaters in the POCTEFA territory: Occurrence and TiO2 photocatalytic pilot-scale plant performance. Water 2020, 12, 1453. [Google Scholar] [CrossRef]
- European Medicines Agency. Sales of Veterinary Antimicrobial Agents in 31 European Countries in 2018: Trends from 2010–2018; European Medicines Agency: Amsterdam, The Netherlands, 2019; ISBN 9789291550685. [Google Scholar]
- European Medicines Agency. Sales of Veterinary Antimicrobial Agents in 30 European Countries in 2015. Seventh ESVAC Report. Seventh ESVAC Rep., 2017. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Report/2017/10/WC500236750.pdf%0Ahttps://bi.ema.europa.eu/analyticsSOAP/saw.dll?PortalPages (accessed on 4 May 2022).
- EMA. Sales of veterinary antimicrobial agents in 31 European countries in 2017 Trends from 2010 to 2017; Ema/294674/2019; EMA: Amsterdam, The Netherlands, 2019. [Google Scholar]
- López Navas, A.; Muñoz Madero, C.; Aguilera Moyano, C.; Alonso Herreras, M.; Bueno Parralo, R.; Sacristán Álvarez, S.; Toboso Escribano, S. Plan Nacional Frente a la Resistencia a los Antibióticos 2019–2021. Agencia Española Medicam. y Prod. Sanit. 2019, 37. Available online: http://resistenciaantibioticos.es/es/system/files/field/files/pran_2019-2021_0.pdf?file=1&type=node&id=497&force=0 (accessed on 7 August 2022).
- French Agency for Food Environmental and Occupational Health & Safet (ANSES). Sales Survey of Veterinary Medicinal Products Containing Antimicrobials in France—2013; French Agency for Food Environmental and Occupational Health & Safet (ANSES): Maisons-Alfort, France, 2014; p. 78. [Google Scholar]
- ECDC. Antimicrobial Consumption in the EU/EEA—Annual Epidemiological Report 2019; ECDC: Solna, Sweden, 2020; pp. 1–25. [Google Scholar]
- European Commission. COMMISION IMPLEMENTING DECISION (EU) 2022/1307 of 22 July 2022; European Commission: Brussels, Belgium, 2022; Volume 2016. [Google Scholar]
- Mirzaei, R.; Yunesian, M.; Nasseri, S.; Gholami, M.; Jalilzadeh, E.; Shoeibi, S.; Bidshahi, H.S.; Mesdaghinia, A. An optimized SPE-LC-MS/MS method for antibiotics residue analysis in ground, surface and treated water samples by response surface methodology- central composite design. J. Environ. Health Sci. Eng. 2017, 15, 21. [Google Scholar] [CrossRef]
- Mirzaei, R.; Mesdaghinia, A.; Hoseini, S.S.; Yunesian, M. Antibiotics in urban wastewater and rivers of Tehran, Iran: Consumption, mass load, occurrence, and ecological risk. Chemosphere 2019, 221, 55–66. [Google Scholar] [CrossRef]
- Biošić, M.; Mitrevski, M.; Babić, S. Environmental behavior of sulfadiazine, sulfamethazine, and their metabolites. Environ. Sci. Pollut. Res. 2017, 24, 9802–9812. [Google Scholar] [CrossRef]
- Reis, A.C.; Kolvenbach, B.A.; Nunes, O.C.; Corvini, P.F.X. Biodegradation of antibiotics: The new resistance determinants—Part II. N. Biotechnol. 2020, 54, 13–27. [Google Scholar] [CrossRef]
- Reis, A.C.; Kolvenbach, B.A.; Nunes, O.C.; Corvini, P.F.X. Biodegradation of antibiotics: The new resistance determinants—Part I. N. Biotechnol. 2020, 54, 34–51. [Google Scholar] [CrossRef]
- Nieto-Juárez, J.I.; Torres-Palma, R.A.; Botero-Coy, A.M.; Hernández, F. Pharmaceuticals and environmental risk assessment in municipal wastewater treatment plants and rivers from Peru. Environ. Int. 2021, 155, 106674. [Google Scholar] [CrossRef]
- Huidobro-López, B.; López-Heras, I.; Alonso-Alonso, C.; Martínez-Hernández, V.; Nozal, L.; de Bustamante, I. Analytical method to monitor contaminants of emerging concern in water and soil samples from a non-conventional wastewater treatment system. J. Chromatogr. A 2022, 1671, 463006. [Google Scholar] [CrossRef]
- Maia, A.S.; Paíga, P.; Delerue-Matos, C.; Castro, P.M.L.; Tiritan, M.E. Quantification of fluoroquinolones in wastewaters by liquid chromatography-tandem mass spectrometry. Environ. Pollut. 2020, 259, 113927. [Google Scholar] [CrossRef] [PubMed]
- Goessens, T.; Huysman, S.; De Troyer, N.; Deknock, A.; Goethals, P.; Lens, L.; Vanhaecke, L.; Croubels, S. Multi-class analysis of 46 antimicrobial drug residues in pond water using UHPLC-Orbitrap-HRMS and application to freshwater ponds in Flanders, Belgium. Talanta 2020, 220, 121326. [Google Scholar] [CrossRef] [PubMed]
- USEPA. Method 1694: Pharmaceuticals and Personal Care Products in Water, Soil, Sediment, and Biosolids by HPLC/MS/MS; USEPA: Washington, DC, USA, 2007; p. 77. [Google Scholar]
- López-Serna, R.; Petrović, M.; Barceló, D. Development of a fast instrumental method for the analysis of pharmaceuticals in environmental and wastewaters based on ultra high performance liquid chromatography (UHPLC)–tandem mass spectrometry (MS/MS). Chemosphere 2011, 85, 1390–1399. [Google Scholar] [CrossRef] [PubMed]
- US Food & Drug Administration. Acceptance Criteria for Confirmation of Identity of Chemical Residues using Exact Mass Data for the FDA Foods and Veterinary Medicine Program; US Food & Drug Administration: Silver Spring, MD, USA, 2015; pp. 1–17.
- Pfeifer, T.; Tuerk, J.; Fuchs, R. Structural characterization of sulfadiazine metabolites using H/D exchange combined with various MS/MS experiments. J. Am. Soc. Mass Spectrom. 2005, 16, 1687–1694. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Guo, C.; Ai, L.; Dou, C.; Wang, G.; Sun, H. Research on degradation of penicillins in milk by β-lactamase using ultra-performance liquid chromatography coupled with time-of-flight mass spectrometry. J. Dairy Sci. 2014, 97, 4052–4061. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Zhang, B.; Timári, I.; Somogyi, Á.; Li, D.W.; Adcox, H.E.; Gunn, J.S.; Bruschweiler-Li, L.; Brüschweiler, R. Accurate and Efficient Determination of Unknown Metabolites in Metabolomics by NMR-Based Molecular Motif Identification. Anal. Chem. 2019, 91, 15686–15693. [Google Scholar] [CrossRef]
- Chiesa, L.M.; Di Cesare, F.; Nobile, M.; Villa, R.; Decastelli, L.; Martucci, F.; Fontana, M.; Pavlovic, R.; Arioli, F.; Panseri, S. Antibiotics and non-targeted metabolite residues detection as a comprehensive approach toward food safety in raw milk. Foods 2021, 10, 544. [Google Scholar] [CrossRef]
- Schymanski, E.L.; Jeon, J.; Gulde, R.; Fenner, K.; Ruff, M.; Singer, H.P.; Hollender, J. Identifying small molecules via high resolution mass spectrometry: Communicating confidence. Environ. Sci. Technol. 2014, 48, 2097–2098. [Google Scholar] [CrossRef]
- Kaliszan, R.; Wiczling, P. Theoretical opportunities and actual limitations of pH gradient HPLC. Anal. Bioanal. Chem. 2005, 382, 718–727. [Google Scholar] [CrossRef]
- Kubik, Ł.; Wiczling, P.; Kaliszan, R. pH-Gradient Liquid Chromatography: Fundamentals and Examples. J. AOAC Int. 2017, 100, 1590–1598. [Google Scholar] [CrossRef]
- Kasprzyk-Hordern, B.; Dinsdale, R.M.; Guwy, A.J. The removal of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs during wastewater treatment and its impact on the quality of receiving waters. Water Res. 2009, 43, 363–380. [Google Scholar] [CrossRef] [PubMed]
- Christian, T.; Schneider, R.J.; Färber, H.A.; Skutlarek, D.; Meyer, M.T.; Goldbach, H.E. Determination of Antibiotic Residues in Manure, Soil, and Surface Waters. Acta Hydrochim. Hydrobiol. 2003, 31, 36–44. [Google Scholar] [CrossRef]
- Feitosa-Felizzola, J.; Chiron, S. Occurrence and distribution of selected antibiotics in a small Mediterranean stream (Arc River, Southern France). J. Hydrol. 2009, 364, 50–57. [Google Scholar] [CrossRef]
- Seifrtová, M.; Pena, A.; Lino, C.M.; Solich, P. Determination of fluoroquinolone antibiotics in hospital and municipal wastewaters in Coimbra by liquid chromatography with a monolithic column and fluorescence detection. Anal. Bioanal. Chem. 2008, 391, 799–805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gracia-Lor, E.; Sancho, J.V.; Hernández, F. Multi-class determination of around 50 pharmaceuticals, including 26 antibiotics, in environmental and wastewater samples by ultra-high performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2011, 1218, 2264–2275. [Google Scholar] [CrossRef]
- Tamtam, F.; Mercier, F.; Le Bot, B.; Eurin, J.; Tuc Dinh, Q.; Clément, M.; Chevreuil, M. Occurrence and fate of antibiotics in the Seine River in various hydrological conditions. Sci. Total Environ. 2008, 393, 84–95. [Google Scholar] [CrossRef]
- Andreozzi, R.; Caprio, V.; Ciniglia, C.; Champdoré, M.; Lo Giudice, R.; Marotta, R.; Zuccato, E. Antibiotics in the environment: Occurrence in Italian STPs, fate, and preliminary assessment on algal toxicity of amoxicillin. Environ. Sci. Technol. 2004, 38, 6832–6838. [Google Scholar] [CrossRef]
- Bielen, A.; Šimatović, A.; Kosić-Vukšić, J.; Senta, I.; Ahel, M.; Babić, S.; Jurina, T.; González Plaza, J.J.; Milaković, M.; Udiković-Kolić, N. Negative environmental impacts of antibiotic-contaminated effluents from pharmaceutical industries. Water Res. 2017, 126, 79–87. [Google Scholar] [CrossRef]
- Hirsch, R.; Ternes, T.; Haberer, K.; Kratz, K.L. Occurrence of antibiotics in the aquatic environment. Sci. Total Environ. 1999, 225, 109–118. [Google Scholar] [CrossRef]
- Conde-Cid, M.; Álvarez-Esmorís, C.; Paradelo-Núñez, R.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Álvarez-Rodríguez, E.; Fernández-Sanjurjo, M.J.; Núñez-Delgado, A. Occurrence of tetracyclines and sulfonamides in manures, agricultural soils and crops from different areas in Galicia (NW Spain). J. Clean. Prod. 2018, 197, 491–500. [Google Scholar] [CrossRef]
- García-Galán, M.J.; Díaz-Cruz, M.S.; Barceló, D. Determination of 19 sulfonamides in environmental water samples by automated on-line solid-phase extraction-liquid chromatography–tandem mass spectrometry (SPE-LC–MS/MS). Talanta 2010, 81, 355–366. [Google Scholar] [CrossRef] [PubMed]
- García-Galán, M.J.; Díaz-Cruz, M.S.; Barceló, D. Occurrence of sulfonamide residues along the Ebro river basin. Removal in wastewater treatment plants and environmental impact assessment. Environ. Int. 2011, 37, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Lei, K.; Zhu, Y.; Chen, W.; Pan, H.Y.; Cao, Y.X.; Zhang, X.; Guo, B.B.; Sweetman, A.; Lin, C.Y.; Ouyang, W.; et al. Spatial and seasonal variations of antibiotics in river waters in the Haihe River Catchment in China and ecotoxicological risk assessment. Environ. Int. 2019, 130, 104919. [Google Scholar] [CrossRef]
- Pujar, P.M.; Kenchannavar, H.H.; Kulkarni, R.M.; Kulkarni, U.P. Real-time water quality monitoring through Internet of Things and ANOVA-based analysis: A case study on river Krishna. Appl. Water Sci. 2020, 10, 22. [Google Scholar] [CrossRef] [Green Version]
- European Centre for Disease Prevention and Control. Antimicrobial Consumption and Resistance in Bacteria from Humans and Animals: Third Joint Inter-Agency Report on Integrated Analysis of Antimicrobial Agent Consumption and Occurrence of Antimicrobial Resistance in Bacteria from Humans and Food-Producing; European Centre for Disease Prevention and Control: Stockholm, Sweden, 2021. [Google Scholar]
- European Centre for Disease Prevention and Control. Antimicrobial Consumption. In ECDC. Annual Epidemiological Report for 2017; European Centre for Disease Prevention and Control: Stockholm, Sweden, 2018. [Google Scholar]
- Danner, M.C.; Robertson, A.; Behrends, V.; Reiss, J. Antibiotic pollution in surface fresh waters: Occurrence and effects. Sci. Total Environ. 2019, 664, 793–804. [Google Scholar] [CrossRef]
- Papageorgiou, M.; Kosma, C.; Lambropoulou, D. Seasonal occurrence, removal, mass loading and environmental risk assessment of 55 pharmaceuticals and personal care products in a municipal wastewater treatment plant in Central Greece. Sci. Total Environ. 2016, 543, 547–569. [Google Scholar] [CrossRef]
- Verlicchi, P.; Al Aukidy, M.; Galletti, A.; Petrovic, M.; Barceló, D. Hospital effluent: Investigation of the concentrations and distribution of pharmaceuticals and environmental risk assessment. Sci. Total Environ. 2012, 430, 109–118. [Google Scholar] [CrossRef]
- Rodriguez-Mozaz, S.; Vaz-Moreira, I.; Della Giustina, S.V.; Llorca, M.; Barceló, D.; Schubert, S.; Berendonk, T.U.; Michael-Kordatou, I.; Fatta-Kassinos, D.; Martinez, J.L.; et al. Antibiotic residues in final effluents of European wastewater treatment plants and their impact on the aquatic environment. Environ. Int. 2020, 140, 105733. [Google Scholar] [CrossRef]
- Lamm, A.; Gozlan, I.; Rotstein, A.; Avisar, D. Detection of amoxicillin-diketopiperazine-2’, 5’ in wastewater samples. J. Environ. Sci. Health Part A Toxic/Hazardous Subst. Environ. Eng. 2009, 44, 1512–1517. [Google Scholar] [CrossRef]
- Wang, K.; Zhuang, T.; Su, Z.; Chi, M.; Wang, H. Antibiotic residues in wastewaters from sewage treatment plants and pharmaceutical industries: Occurrence, removal and environmental impacts. Sci. Total Environ. 2021, 788, 147811. [Google Scholar] [CrossRef]
- Santos, L.H.M.L.M.; Gros, M.; Rodriguez-mozaz, S.; Delerue-matos, C.; Pena, A.; Barceló, D.; Montenegro, M.C.B.S.M. Science of the Total Environment Contribution of hospital effluents to the load of pharmaceuticals in urban wastewaters: Identification of ecologically relevant pharmaceuticals. Sci. Total Environ. 2013, 461–462, 302–316. [Google Scholar] [CrossRef] [PubMed]
- Jewell, K.S.; Castronovo, S.; Wick, A.; Falås, P.; Joss, A.; Ternes, T.A. New insights into the transformation of trimethoprim during biological wastewater treatment. Water Res. 2016, 88, 550–557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tong, L.; Eichhorn, P.; Pérez, S.; Wang, Y.; Barceló, D. Photodegradation of azithromycin in various aqueous systems under simulated and natural solar radiation: Kinetics and identification of photoproducts. Chemosphere 2011, 83, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Sturini, M.; Speltini, A.; Maraschi, F.; Profumo, A.; Pretali, L.; Irastorza, E.A.; Fasani, E.; Albini, A. Photolytic and photocatalytic degradation of fluoroquinolones in untreated river water under natural sunlight. Appl. Catal. B Environ. 2012, 119–120, 32–39. [Google Scholar] [CrossRef]
- Szymańska, U.; Wiergowski, M.; Sołtyszewski, I.; Kuzemko, J.; Wiergowska, G.; Woźniak, M.K. Presence of antibiotics in the aquatic environment in Europe and their analytical monitoring: Recent trends and perspectives. Microchem. J. 2019, 147, 729–740. [Google Scholar] [CrossRef]
- Boy-Roura, M.; Mas-Pla, J.; Petrovic, M.; Gros, M.; Soler, D.; Brusi, D.; Menció, A. Towards the understanding of antibiotic occurrence and transport in groundwater: Findings from the Baix Fluvià alluvial aquifer (NE Catalonia, Spain). Sci. Total Environ. 2018, 612, 1387–1406. [Google Scholar] [CrossRef] [PubMed]
- Paul, T.; Dodd, M.C.; Strathmann, T.J. Photolytic and photocatalytic decomposition of aqueous ciprofloxacin: Transformation products and residual antibacterial activity. Water Res. 2010, 44, 3121–3132. [Google Scholar] [CrossRef] [PubMed]
- Kovalova, L.; Siegrist, H.; Von Gunten, U.; Eugster, J.; Hagenbuch, M.; Wittmer, A.; Moser, R.; McArdell, C.S. Elimination of micropollutants during post-treatment of hospital wastewater with powdered activated carbon, ozone, and UV. Environ. Sci. Technol. 2013, 47, 7899–7908. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dinh, Q.T.; Alliot, F.; Guigon, E.M.; Eurin, J.; Chevreuil, M.; Labadie, P. Measurement of trace levels of antibiotics in river water using on-line enrichment and triple-quadrupole LC-MS/MS. Talanta 2011, 85, 1238–1245. [Google Scholar] [CrossRef]
- Gros, M.; Petrovic, M.; Ginebreda, A.; Barceló, D. Sources, Occurrence, and Environmental Risk Assessment of Pharmaceuticals in the Ebro River Basin. In The Ebro River Basin; Springer: Berlin/Heidelberg, Germany, 2010; pp. 209–237. [Google Scholar]
- Ginebreda, A.; Guasch, H.; de Alda, M.J.L.; Romaní, A.M.; Sabater, S.; Barceló, D.; Muñoz, I.; López-Doval, J.C.; Ricart, M.; Villagrasa, M.; et al. Bridging Levels of Pharmaceuticals in River Water With Biological Community Structure in the Llobregat River Basin (Northeast Spain). Environ. Toxicol. Chem. 2009, 28, 2706–2714. [Google Scholar]
- Díaz-Cruz, M.S.; García-Galán, M.J.; Barceló, D. Highly sensitive simultaneous determination of sulfonamide antibiotics and one metabolite in environmental waters by liquid chromatography-quadrupole linear ion trap-mass spectrometry. J. Chromatogr. A 2008, 1193, 50–59. [Google Scholar] [CrossRef]
- Jurado, A.; Walther, M.; Díaz-Cruz, M.S. Occurrence, fate and environmental risk assessment of the organic microcontaminants included in the Watch Lists set by EU Decisions 2015/495 and 2018/840 in the groundwater of Spain. Sci. Total Environ. 2019, 663, 285–296. [Google Scholar] [CrossRef]
- Oberlé, K.; Capdeville, M.J.; Berthe, T.; Budzinski, H.; Petit, F. Evidence for a complex relationship between antibiotics and antibiotic-resistant escherichia coli: From medical center patients to a receiving environment. Environ. Sci. Technol. 2012, 46, 1859–1868. [Google Scholar] [CrossRef]
- López-Roldán, R.; de Alda, M.L.; Gros, M.; Petrovic, M.; Martín-Alonso, J.; Barceló, D. Advanced monitoring of pharmaceuticals and estrogens in the Llobregat River basin (Spain) by liquid chromatography-triple quadrupole-tandem mass spectrometry in combination with ultra performance liquid chromatography-time of flight-mass spectrometry. Chemosphere 2010, 80, 1337–1344. [Google Scholar] [CrossRef]


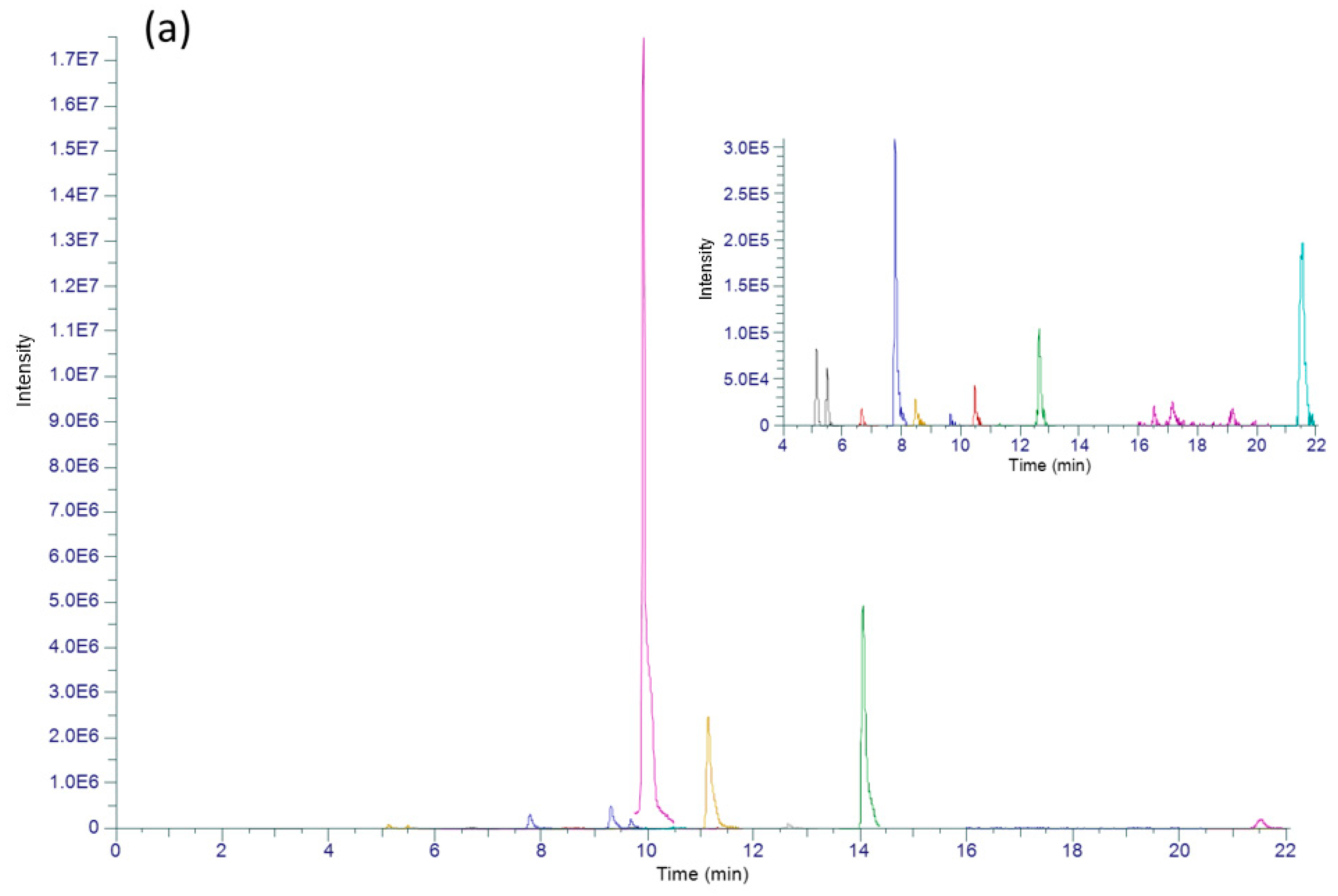

| Group | Antibiotic | CAS | RT (min) | Parent Ion (m/z) [M + H]+ | CE (eV) | Product Ion 1 | Product Ion 2 | LOD (ng L−1) | LOQ (ng L−1) |
|---|---|---|---|---|---|---|---|---|---|
| ß-Lactamase | amoxicillin | 26,787-78-0 | 8.99 | 366.1118 | 10 | 349.0853 | 208.0427 | 0.152 | 0.500 |
| ampicillin | 69-53-4 | 5.93 | 350.1169 | 20 | 192.0478 | 106.051 | 0.021 | 0.071 | |
| diketopiperazine | 94,659-47-9 | 9.36 | 366.1118 | 10 | 207.0764 | 160.0427 | 0.300 | 0.100 | |
| penicilloic acid | 210,289-72-8 | 5.50 | 384.1224 | 10 | 367.0959 | 323.1058 | 0.015 | 0.05 | |
| diaminopyrimidine | trimethoprim | 738-70-5 | 9.06 | 291.1452 | 50 | 261.0982 | 230.1162 | 0.026 | 0.085 |
| fluoroquinolone | ciprofloxacin | 85,721-33-1 | 9.91 | 332.1405 | 65 | 249.0670 | 231.0564 | 0.028 | 0.094 |
| enrofloxacin | 93,106-60-6 | 10.47 | 360.1718 | 35 | 316.1820 | 245.1085 | 0.041 | 0.135 | |
| moxifloxacin | 354,812-41-2 | 12.64 | 402.1824 | 40 | 384.1718 | 341.1534 | 0.011 | 0.035 | |
| norfloxacin | 70,458-96-7 | 9.66 | 320.1405 | 35 | 276.1507 | 233.1085 | 0.015 | 0.050 | |
| lincosamide | lincomycin | 154-21-2 | 8.44 | 407.2210 | 25 | 359.2177 | 126.1277 | 0.025 | 0.082 |
| macrolide | azithromycin | 83,905-01-5 | 13.99 | 749.5158 | 25 | 591.4215 | 158.1176 | 0.020 | 0.067 |
| clarithromycin | 81,103-11-9 | 20.91 | 748.4842 | 20 | 590.3899 | 158.1176 | 0.018 | 0.059 | |
| clarithromycin N-oxide | 118,074-07-0 | 20.87 | 764.4791 | 28 | 606.3848 | 123.0804 | 0.012 | 0.038 | |
| sulfonamide | dapsone | 80-08-0 | 9.94 | 249.0692 | 35 | 156.0114 | 108.0444 | 0.029 | 0.095 |
| sulfacetamide | 144-80-9 | 5.69 | 215.0485 | 35 | 156.0114 | 108.0444 | 0.049 | 0.163 | |
| sulfadiazine | 68-35-9 | 6.71 | 251.0597 | 30 | 156.0114 | 108.0444 | 0.015 | 0.050 | |
| sulfadoxine | 2447-57-6 | 11.31 | 311.0809 | 40 | 156.0114 | 108.0444 | 0.012 | 0.038 | |
| sulfamerazine | 127-79-7 | 8.14 | 265.0754 | 35 | 190.0274 | 156.0114 | 0.023 | 0.075 | |
| sulfapyridine | 144-83-2 | 6.82 | 250.0645 | 35 | 184.0869 | 156.0114 | 0.015 | 0.050 | |
| sulfamethoxazole | 723-46-6 | 11.19 | 254.0594 | 40 | 156.0114 | 108.0444 | 0.008 | 0.025 | |
| sulfamethoxypyridazine | 80-35-3 | 10.73 | 281.0703 | 30 | 156.0114 | 126.0662 | 0.058 | 0.197 | |
| tetracycline | oxytetracycline | 79-57-2 | 9.75 | 461.1555 | 20 | 426.1183 | 381.0605 | 0.028 | 0.094 |
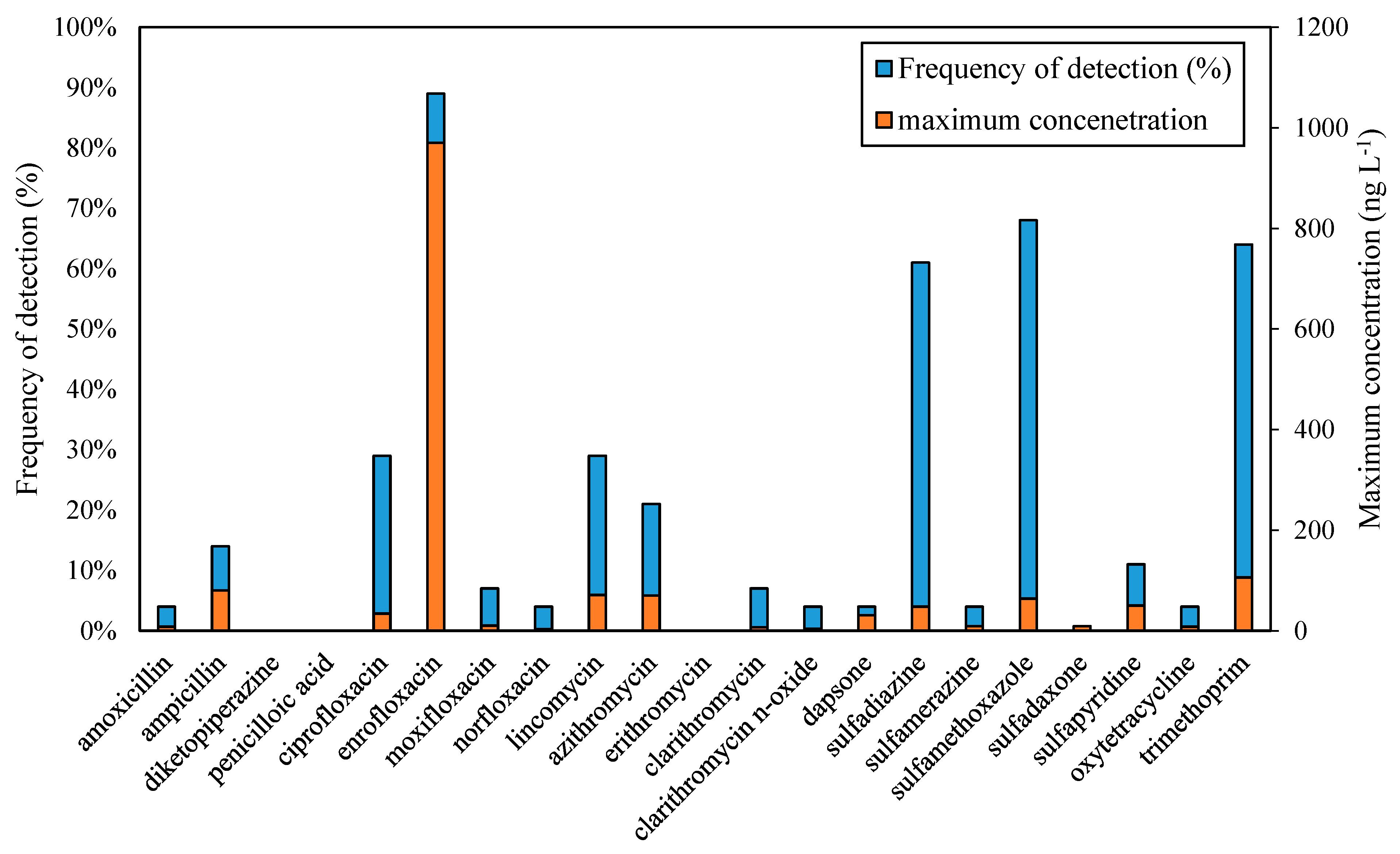
| Group | Antibiotic | Frequency of Detection (%) | Concentration Range (ng L−1) | Cmax (ng L−1) * |
|---|---|---|---|---|
| ß-lactamase | amoxicillin | 4 | LOQ-8.0 | 522 (UK) [40] |
| ampicillin | 14 | 15.7–79.6 | 26 (Germany) [41] | |
| diketopiperazine | - | <LOQ | not given | |
| penicilloic acid | - | <LOQ | not given | |
| fluoroquinolone | ciprofloxacin | 29 | LOQ-33.6 | 9660 (France) [42] |
| enrofloxacin | 89 | 11.8–970.0 | 210 (Portugal) [43] | |
| moxifloxacin | 7 | 1.4–9.8 | 210 (Spain) [44] | |
| norfloxacin | 4 | LOQ-3.2 | 160 (France) [45] | |
| lincosamide | lincomycin | 29 | 0.9–70.7 | 250 (Italy) [46] |
| macrolides | azithromycin | 21 | 1.7–67.0 | 1600(Croatia) [47] |
| erythromycin | - | <LOQ | 1700 (Germany) [48] | |
| clarithromycin | 7 | 3.8–7.0 | 2330 (France) [42] | |
| clarithromycin n-oxide | 4 | LOQ-4.0 | not given | |
| sulfonamide | dapsone | 4 | 1.1–30.7 | not given |
| sulfadiazine | 61 | 0.4–48.3 | 2400 (Croatia) [47] | |
| sulfamerazine | 4 | LOQ-8.6 | 11,000 (Croatia) [47] | |
| sulfamethoxazole | 68 | LOQ-63.9 | 11,000 (Spain) [49] | |
| sulfadaxone | - | <LOQ | not given | |
| sulfapyridine | 11 | 1.4–48.8 | 12,000 (Spain) [50,51] | |
| tetracycline | oxytetracycline | 4 | LOQ-7.8 | not given |
| diaminopyrimidine | trimethoprim | 64 | 2.5–106.0 | 11,000 (Croatia) [47] |
| Group | Antibiotic | Frequency of Detection (%) | Concentration Range (ng L−1) |
|---|---|---|---|
| ß-lactamase | amoxicillin | 8 | 7.0–15.0 |
| ampicillin | 25 | LOQ-26.0 | |
| diketopiperazine | - | <LOQ | |
| penicilloic acid | - | <LOQ | |
| fluoroquinolone | ciprofloxacin | 75 | 3.8–172.7 |
| enrofloxacin | 61 | 48.3 | |
| moxifloxacin | 42 | 2.6–46.9 | |
| norfloxacin | 25 | LOQ-3.2 | |
| lincosamide | lincomycin | 50 | LOQ-26.0 |
| macrolides | azithromycin | 92 | 1.8–144.0 |
| erythromycin | 8 | LOQ-43.0 | |
| clarithromycin | 50 | 6.6–62.7 | |
| clarithromycin N-oxide | - | <LOQ | |
| sulfonamide | dapsone | 8 | LOQ-30.7 |
| sulfadiazine | 42 | LOQ-103.0 | |
| sulfamerazine | - | <LOQ | |
| sulfamethoxazole | 92 | 5.9–256.0 | |
| sulfapyridine | 42 | 2.8–12.6 | |
| tetracycline | oxytetracycline | 8 | 535–2670 |
| diaminopyrimidine | trimethoprim | 75 | 1.4–122.9 |
| Group | Antibiotic | Cmax (ng L−1) | Country |
|---|---|---|---|
| fluoroquinolones | ciprofloxacin | 1436 | Portugal |
| enrofloxacin | 176 | Spain | |
| macrolides | azithromycin | 1577 | Portugal |
| clarithromycin | 337 | Ireland | |
| sulfonamides | sulfamethoxazole | 177 | Spain |
| diaminopyrimidine | trimethoprim | 330 | Finland |
| Group | Antibiotic | Degradation Product | RT (min) | Formula | Exact Mass | Δ ppm | Ref. |
|---|---|---|---|---|---|---|---|
| diaminopyrimidine | trimethoprim | 4-desmethyl-TMP | 4.16 | C13H17N4O3 | 277.1294 | −0.5 | [64] |
| fluoroquinolone | enrofloxacin | enrofloxacin 5Bwt | 5.80 | C17H21FN3O3 | 334.1558 | −1.37 | [66] |
| macrolides | azithromycin | azithromycin double cleavage | 5.90 | C22H44NO7 | 434.3110 | −0.38 | [65] |
| sulphonamide | sulfadiazine | N-acetyl sulfadiazine | 4.98 | C12H13N4O3S | 293.0701 | −0.73 | [23,33] |
| sulfamethoxazole | N-acetyl sulfamethoxazole | 6.63 | C12H14N3O4S | 296.0698 | −0.6 | [23] |
| Group | Antibiotic | Degradation Product | Frequency of Detection (%) | |
|---|---|---|---|---|
| Surface Water | Wastewater | |||
| -lactamates | amoxicillin | penicilloic acid | - | - |
| diketopiperazine | - | - | ||
| macrolides | azithromycin | azithromycin double cleavage | 57 | 50 |
| diaminopyrimidines | trimethoprim | 4-desmethyl-TMP | 7 | 50 |
| fluoroquinolones | enrofloxacin | enrofloxacin 5Bwt | 14 | 58 |
| sulphonamides | sulfadiazine | N-acetyl sulfadiazine | 11 | 8 |
| sulfamethoxazole | N-acetyl sulfamethoxazole | 11 | 67 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gozzo, S.; Moles, S.; Kińska, K.; Ormad, M.P.; Mosteo, R.; Gómez, J.; Laborda, F.; Szpunar, J. Screening for Antibiotics and Their Degradation Products in Surface and Wastewaters of the POCTEFA Territory by Solid-Phase Extraction-UPLC-Electrospray MS/MS. Water 2023, 15, 14. https://doi.org/10.3390/w15010014
Gozzo S, Moles S, Kińska K, Ormad MP, Mosteo R, Gómez J, Laborda F, Szpunar J. Screening for Antibiotics and Their Degradation Products in Surface and Wastewaters of the POCTEFA Territory by Solid-Phase Extraction-UPLC-Electrospray MS/MS. Water. 2023; 15(1):14. https://doi.org/10.3390/w15010014
Chicago/Turabian StyleGozzo, Sebastiano, Samuel Moles, Katarzyna Kińska, Maria P. Ormad, Rosa Mosteo, Jairo Gómez, Francisco Laborda, and Joanna Szpunar. 2023. "Screening for Antibiotics and Their Degradation Products in Surface and Wastewaters of the POCTEFA Territory by Solid-Phase Extraction-UPLC-Electrospray MS/MS" Water 15, no. 1: 14. https://doi.org/10.3390/w15010014
APA StyleGozzo, S., Moles, S., Kińska, K., Ormad, M. P., Mosteo, R., Gómez, J., Laborda, F., & Szpunar, J. (2023). Screening for Antibiotics and Their Degradation Products in Surface and Wastewaters of the POCTEFA Territory by Solid-Phase Extraction-UPLC-Electrospray MS/MS. Water, 15(1), 14. https://doi.org/10.3390/w15010014







