PFAS in the Drinking Water Source: Analysis of the Contamination Levels, Origin and Emission Rates
Abstract
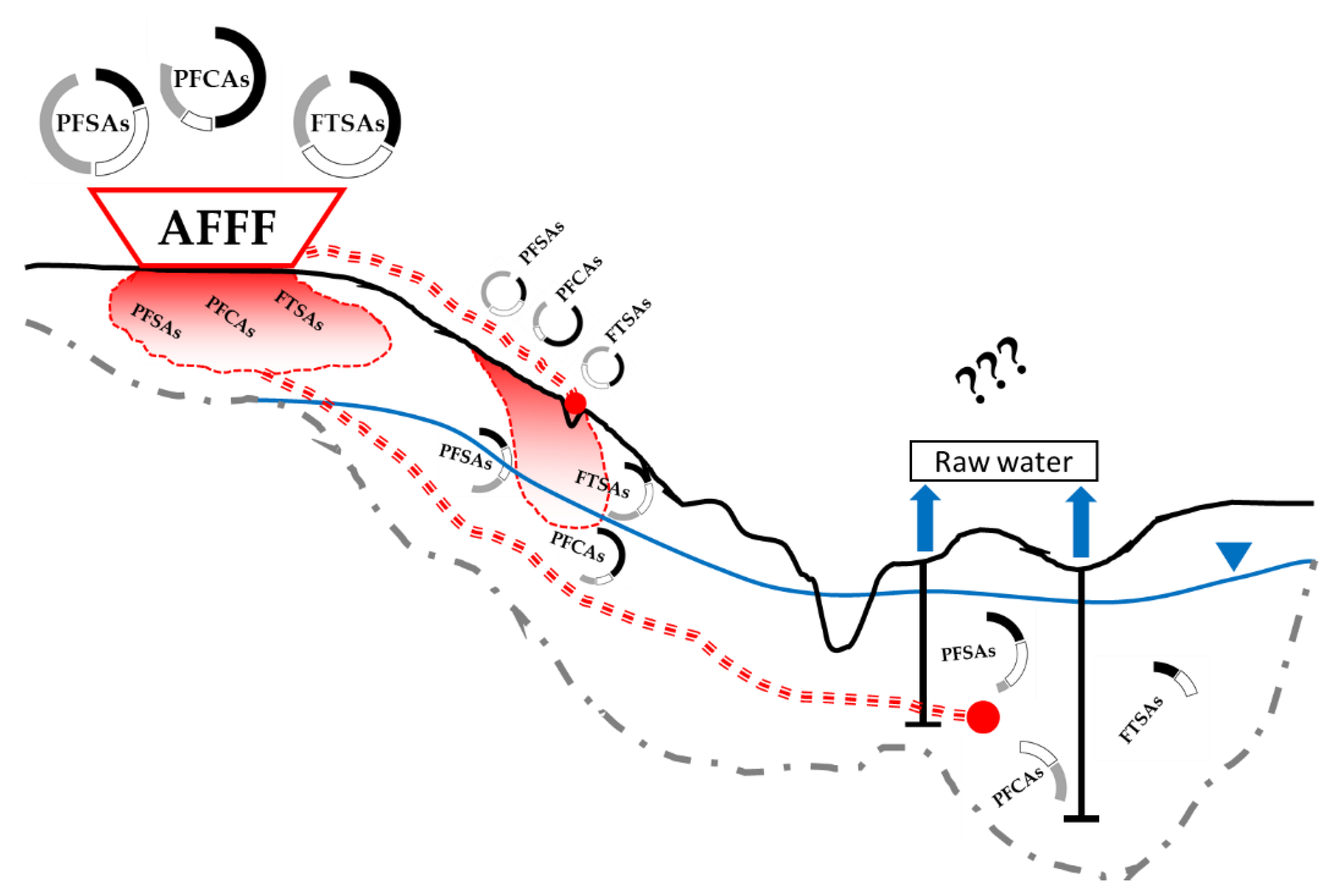
1. Introduction
2. Materials and Methods
2.1. Study Site
2.1.1. Area Description
2.1.2. Surface Water and Hydrogeology of the Area
2.1.3. PFAS Emission Source
2.2. PFAS Measurements in Groundwater
2.3. PFAS Emission Estimates
3. Results and Discussion
3.1. Groundwater Contamination
3.2. Assessment of the PFAS Emission
3.3. PFAS Transport Considerations
3.4. Remarks on Regulations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cousins, I.T.; Vestergren, R.; Wang, Z.Y.; Scheringer, M.; McLachlan, M.S. The precautionary principle and chemicals management: The example of perfluoroalkyl acids in groundwater. Environ. Int. 2016, 94, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Gramstad, T.; Haszeldine, R.N. 512. Perfluoroalkyl derivatives of sulphur. Part VI. Perfluoroalkanesulphonic acids CF3·[CF2]·SO3H (n = 1–7). J. Chem. Soc. 1957, 6, 2640–2645. [Google Scholar] [CrossRef]
- Kissa, E. Fluorinated Surfactants and Repellents; Marcel Dekker: New York, NY, USA, 2001; Volume 97, p. 640. [Google Scholar]
- Kleiner, E.; Jho, C. Recent developments in 6: 2 fluorotelomer surfactants and fosam stabilizers. In Proceedings of the 4th Reebok Foam Seminar, Bolton, UK, 6–7 July 2009; pp. 6–7. [Google Scholar]
- Levine, A.D.; Libelo, E.; Bugna, G.; Shelley, T.; Mayfield, H.; Stauffer, T. Biogeochemical assessment of natural attenuation of JP-4 contaminated ground water in the presence of fluorinated surfactants. Sci. Total Environ. 1998, 208, 179–195. [Google Scholar] [CrossRef]
- Moody, C.; Field, J. Determination of Perfluorocarboxylates in Groundwater Impacted by Fire-Fighting Activity. Environ. Sci. Technol. 1999, 33, 2800–2806. [Google Scholar] [CrossRef]
- Moody, C.A.; Martin, J.W.; Kwan, W.C.; Muir, D.C.G.; Mabury, S.C. Monitoring perfluorinated surfactants in biota and surface water samples following an accidental release of fire-fighting foam into Etobicoke Creek. Environ. Sci. Technol. 2002, 36, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Awad, E.; Zhang, X.M.; Bhavsar, S.P.; Petro, S.; Crozier, P.W.; Reiner, E.J.; Fletcher, R.; Tittemier, S.A.; Braekevelt, E. Long-Term Environmental Fate of Perfluorinated Compounds after Accidental Release at Toronto Airport. Environ. Sci. Technol. 2011, 45, 8081–8089. [Google Scholar] [CrossRef] [PubMed]
- Karrman, A.; Elgh-Dalgren, K.; Lafossas, C.; Moskeland, T. Environmental levels and distribution of structural isomers of perfluoroalkyl acids after aqueous fire-fighting foam (AFFF) contamination. Environ. Chem. 2011, 8, 372–380. [Google Scholar] [CrossRef]
- Kwadijk, C.J.A.F.; Kotterman, M.; Koelmans, A.A. Partitioning of Perfluorooctanesulfonate and Perfluorohexanesulfonate in the Aquatic Environment after an Accidental Release of Aqueous Film Forming Foam at Schiphol Amsterdam Airport. Environ. Toxicol. Chem. 2014, 33, 1761–1765. [Google Scholar] [CrossRef]
- Banzhaf, S.; Filipovic, M.; Lewis, J.; Sparrenbom, C.J.; Barthel, R. A review of contamination of surface-, ground-, and drinking water in Sweden by perfluoroalkyl and polyfluoroalkyl substances (PFASs). Ambio 2017, 46, 335–346. [Google Scholar] [CrossRef]
- Andersson, E.M.; Scott, K.; Xu, Y.; Li, Y.; Olsson, D.S.; Fletcher, T.; Jakobsson, K. High exposure to perfluorinated compounds in drinking water and thyroid disease. A cohort study from Ronneby, Sweden. Environ. Res. 2019, 176, 108540. [Google Scholar] [CrossRef]
- Ryota, S. Kyoto University Professors Detect PFOS Concentration of 4 Times National Average in Ginowan Residents’ Blood. Ryukyu Shimpo, 17 May 2019. Available online: http://english.ryukyushimpo.jp/2019/05/21/30452/ (accessed on 20 December 2022).
- Shin, H.M.; Vieira, V.M.; Ryan, P.B.; Steenland, K.; Bartell, S.M. Retrospective Exposure Estimation and Predicted versus Observed Serum Perfluorooctanoic Acid Concentrations for Participants in the C8 Health Project. Environ. Health Perspect. 2013, 121, A113. [Google Scholar] [CrossRef]
- Kerger, B.D.; Copeland, T.L.; DeCaprio, A.P. Tenuous dose-response correlations for common disease states: Case study of cholesterol and perfluorooctanoate/sulfonate (PFOA/PFOS) in the C8 Health Project. Drug Chem. Toxicol. 2011, 34, 396–404. [Google Scholar] [CrossRef]
- Li, Y.; Fletcher, T.; Mucs, D.; Scott, K.; Lindh, C.H.; Tallving, P.; Jakobsson, K. Half-lives of PFOS, PFHxS and PFOA after end of exposure to contaminated drinking water. Occup. Environ. Med. 2018, 75, 46–51. [Google Scholar] [CrossRef]
- Oberg, M.; Da Silva, A.; Ringblom, J.; Scott, K.; Lindh, C.; Jakobsson, K. Probabilistic risk of decreased levels of triiodothyronine following chronic exposure to PFOS and PFHxS. Toxicol. Lett. 2018, 295, S224. [Google Scholar] [CrossRef]
- Ahrens, L. Polyfluoroalkyl compounds in the aquatic environment: A review of their occurrence and fate. J. Environ. Monit. 2011, 13, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Moller, A.; Ahrens, L.; Surm, R.; Westerveld, J.; van der Wielen, F.; Ebinghaus, R.; de Voogt, P. Distribution and sources of polyfluoroalkyl substances (PFAS) in the River Rhine watershed. Environ. Pollut. 2010, 158, 3243–3250. [Google Scholar] [CrossRef] [PubMed]
- Filipovic, M.; Berger, U.; McLachlan, M. Mass Balance of Perfluoroalkyl Acids in the Baltic Sea. Environ. Sci. Technol. 2013, 47, 4088–4095. [Google Scholar] [CrossRef] [PubMed]
- Filipovic, M.; Laudon, H.; McLachlan, M.; Berger, U. Mass Balance of Perfluorinated Alkyl Acids in a Pristine Boreal Catchment. Environ. Sci. Technol. 2015, 49, 12127–12135. [Google Scholar] [CrossRef]
- Weber, A.K.; Barber, L.B.; LeBlanc, D.R.; Sunderland, E.M.; Vecitis, C.D. Geochemical and Hydrologic Factors Controlling Subsurface Transport of Poly- and Perfluoroalkyl Substances, Cape Cod, Massachusetts. Environ. Sci. Technol. 2017, 51, 4269–4279. [Google Scholar] [CrossRef]
- Shin, H.M.; Steenland, K.; Ryan, P.B.; Vieira, V.M.; Bartell, S.M. Biomarker-Based Calibration of Retrospective Exposure Predictions of Perfluorooctanoic Acid. Environ. Sci. Technol. 2014, 48, 5636–5642. [Google Scholar] [CrossRef]
- Gobelius, L.; Persson, C.; Wiberg, K.; Ahrens, L. Calibration and application of passive sampling for per- and polyfluoroalkyl substances in a drinking water treatment plant. J. Hazard. Mater. 2019, 362, 230–237. [Google Scholar] [CrossRef]
- Sinclair, E.; Kannan, K. Mass loading and fate of perfluoroalkyl surfactants in wastewater treatment plants. Environ. Sci. Technol. 2006, 40, 1408–1414. [Google Scholar] [CrossRef]
- Busch, J.; Ahrens, L.; Sturm, R.; Ebinghaus, R. Polyfluoroalkyl compounds in landfill leachates. Environ. Pollut. 2010, 158, 1467–1471. [Google Scholar] [CrossRef]
- Hellsing, M.S.; Josefsson, S.; Hughes, A.V.; Ahrens, L. Sorption of perfluoroalkyl substances to two types of minerals. Chemosphere 2016, 159, 385–391. [Google Scholar] [CrossRef]
- Jeon, J.; Kannan, K.; Lim, B.J.; An, K.G.; Kim, S.D. Effects of salinity and organic matter on the partitioning of perfluoroalkyl acid (PFAs) to clay particles. J. Environ. Monitor. 2011, 13, 1803–1810. [Google Scholar] [CrossRef]
- Nouhi, S.; Ahrens, L.; Pereira, H.C.; Hughes, A.V.; Campana, M.; Gutfreund, P.; Palsson, G.K.; Vorobiev, A.; Hellsing, M.S. Interactions of perfluoroalkyl substances with a phospholipid bilayer studied by neutron reflectometry. J. Colloid Interface Sci. 2018, 511, 474–481. [Google Scholar] [CrossRef]
- Pereira, H.C.; Ullberg, M.; Kleja, D.B.; Gustafsson, J.P.; Ahrens, L. Sorption of perfluoroalkyl substances (PFASs) to an organic soil horizon—Effect of cation composition and pH. Chemosphere 2018, 207, 183–191. [Google Scholar] [CrossRef]
- McCarthy, C.; Kappleman, W.; DiGuiseppi, W. Ecological Considerations of Per- and Polyfluoroalkyl Substances (PFAS). Curr. Pollut. Rep. 2017, 3, 289–301. [Google Scholar] [CrossRef]
- Zareitalabad, P.; Siemens, J.; Hamer, M.; Amelung, W. Perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) in surface waters, sediments, soils and wastewater—A review on concentrations and distribution coefficients. Chemosphere 2013, 91, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Filipovic, M.; Woldegiorgis, A.; Norstrom, K.; Bibi, M.; Lindberg, M.; Osteras, A.H. Historical usage of aqueous film forming foam: A case study of the widespread distribution of perfluoroalkyl acids from a military airport to groundwater, lakes, soils and fish. Chemosphere 2015, 129, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Place, B.J.; Field, J.A. Identification of Novel Fluorochemicals in Aqueous Film-Forming Foams Used by the US Military. Environ. Sci. Technol. 2012, 46, 7120–7127. [Google Scholar] [CrossRef]
- Benskin, J.P.; De Silva, A.O.; Martin, J.W. Isomer Profiling of Perfluorinated Substances as a Tool for Source Tracking: A Review of Early Findings and Future Applications. Rev. Environ. Contam. Toxicol. 2010, 208, 111–160. [Google Scholar] [CrossRef]
- Buck, R.C.; Franklin, J.; Berger, U.; Conder, J.M.; Cousins, I.T.; de Voogt, P.; Jensen, A.A.; Kannan, K.; Mabury, S.A.; van Leeuwen, S.P. Perfluoroalkyl and polyfluoroalkyl substances in the environment: Terminology, classification, and origins. Integr. Environ. Assess. Manag. 2011, 7, 513–541. [Google Scholar] [CrossRef]
- F17. Available online: https://www.forsvarsmakten.se/sv/organisation/blekinge-flygflottilj-f-17/ (accessed on 20 December 2022).
- Rupert, W.; Verdonik, D.; Hanauska, C. Environmental Impacts of Fire Fighting Foams; Hughes Associates Inc.: Baltimore, MD, USA, 2005. [Google Scholar]
- Kemikalieinspektionen. Chemical Analysis of Selected Fire-Fighting Foams on the Swedish Market 2014; Swedish Chemicals Agency: Sundbyberg, Sweden, 2015. [Google Scholar]
- Kärrman, A.; Bjurlid, F.; Hagberg, J.; Ricklund, N.; Larsson, M.; Stubleski, J.; Hollert, H. Study of Environmental and Human Health Impacts of Firefighting Agents; Technical Report; MTM Research Centre, Örebro University: Örebro, Sweden, 2016. [Google Scholar]
- Johnson, R.L.; Anschutz, A.J.; Smolen, J.M.; Simcik, M.F.; Penn, R.L. The adsorption of perfluorooctane sulfonate onto sand, clay, and iron oxide surfaces. J. Chem. Eng. Data 2007, 52, 1165–1170. [Google Scholar] [CrossRef]
- Moody, C.A.; Field, J.A. Perfluorinated surfactants and the environmental implications of their use in fire-fighting foams. Environ. Sci. Technol. 2000, 34, 3864–3870. [Google Scholar] [CrossRef]
- Turekova, I.; Karol, B. The Environmental Impacts of Fire-Fighting Foams. Res. Pap. Fac. Mater. Sci. Technol. Slovak Univ. Technol. 2010, 18, 111–120. [Google Scholar] [CrossRef][Green Version]
- Bock, A.R.; Laird, B.E. CHAPTER 1 PFAS Regulations: Past and Present and Their Impact on Fluoropolymers. In Perfluoroalkyl Substances: Synthesis, Applications, Challenges and Regulations; The Royal Society of Chemistry: London, UK, 2022; pp. 1–21. [Google Scholar]
- Brennan, N.M.; Evans, A.T.; Fritz, M.K.; Peak, S.A.; von Holst, H.E. Trends in the Regulation of Per- and Polyfluoroalkyl Substances (PFAS): A Scoping Review. Int. J. Environ. Res. Public Health 2021, 18, 10900. [Google Scholar] [CrossRef]
- Reisch, M. 3M supports PFAS regulation. Chem. Eng. News 2019, 97, 12. [Google Scholar]
- Kemikalieinspektionen. Per- and Polyfluoroalkyl Substances (PFAS). Available online: https://www.kemi.se/en/chemical-substances-and-materials/highly-fluorinated-substances (accessed on 19 December 2022).
- EU. Commission Regulation (EU) 2021/1297 of 4 August 2021 Amending Annex XVII to Regulation (EC) No 1907/2006 of the European Parliament and of the Council as Regards Perfluorocarboxylic Acids Containing 9 to 14 Carbon Atoms in the Chain (C9-C14 PFCAs), Their Salts and C9-C14 PFCA-Related Substances; EU: Maastricht, The Netherlands, 2021. [Google Scholar]
- ECHA. Proposal to Ban ‘Forever Chemicals’ in Firefighting Foams Throughout the EU (ECHA/NR/22/05); ECHA: Helsinki, Finland, 2022. [Google Scholar]
- Swedish Civil Contingencies Agency (MSB). Släckmedel för Räddningstjänst. Available online: https://www.msb.se/sv/amnesomraden/skydd-mot-olyckor-och-farliga-amnen/raddningstjanst-och-raddningsinsatser/slackmedel-for-raddningstjanst/ (accessed on 20 December 2022).
- Livsmedelsverket. PFAS i Dricksvatten och Livsmedel—Kontroll. Available online: https://www.livsmedelsverket.se/foretagande-regler-kontroll/dricksvattenproduktion/riskhantering-pfas-i-dricksvatten-egenfangad-fisk/ (accessed on 20 December 2022).
- Livsmedelsverket; Lindfeldt, E.; Gyllenhammar, I.; Strandh, S.; Halldin Ankarberg, E. Kartläggning av Per- och Polyfluorerade Alkylsubstanser (PFAS) i Sveriges Kommunala rå- Och Dricksvatten; Livsmedelsverket: Uppsala, Sweden, 2021. [Google Scholar]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mussabek, D.; Söderman, A.; Imura, T.; Persson, K.M.; Nakagawa, K.; Ahrens, L.; Berndtsson, R. PFAS in the Drinking Water Source: Analysis of the Contamination Levels, Origin and Emission Rates. Water 2023, 15, 137. https://doi.org/10.3390/w15010137
Mussabek D, Söderman A, Imura T, Persson KM, Nakagawa K, Ahrens L, Berndtsson R. PFAS in the Drinking Water Source: Analysis of the Contamination Levels, Origin and Emission Rates. Water. 2023; 15(1):137. https://doi.org/10.3390/w15010137
Chicago/Turabian StyleMussabek, Dauren, Anna Söderman, Tomomi Imura, Kenneth M. Persson, Kei Nakagawa, Lutz Ahrens, and Ronny Berndtsson. 2023. "PFAS in the Drinking Water Source: Analysis of the Contamination Levels, Origin and Emission Rates" Water 15, no. 1: 137. https://doi.org/10.3390/w15010137
APA StyleMussabek, D., Söderman, A., Imura, T., Persson, K. M., Nakagawa, K., Ahrens, L., & Berndtsson, R. (2023). PFAS in the Drinking Water Source: Analysis of the Contamination Levels, Origin and Emission Rates. Water, 15(1), 137. https://doi.org/10.3390/w15010137








