Restoration of Marine Sponges—What Can We Learn from over a Century of Experimental Cultivation?
Abstract
1. Introduction
- compile information on the methodologies that have been applied previously for the transplantation and cultivation of marine sponges,
- synthesize the results of the compiled literature to identify potential best techniques in consideration of taxonomic differences and skeletal features,
- provide guidelines and recommendations on in situ transplantation and cultivation methods for sponges with different skeletal consistency.
2. Materials and Methods
3. Results
3.1. Geographical Distribution and Commercial Interests
3.2. Transplantation Techniques
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| References | Technique | Species | Environment |
|---|---|---|---|
| Wilkinson & Vacelet (1979) [52] | Mesh systems; Mounted on artificial substrate | Aplysina aerophoba Aplysina cavernicola Chondrilla nucula Chondrosia reniformis Petrosia ficiformis | In-situ |
| Barthel & Theede (1986) [53] | In-vitro | Halichondria (Halichondria) panicea | Ex-situ |
| Verdenal & Vacelet (1990) [48] | Rope system | Spongia agaricina Spongia nitens Spongia officinales | In-situ |
| Kaandorp & De Kluijver (1992) [54] | Mounted on artificial substrate | Haliclona (Haliclona) oculata | In-situ |
| Duckworth et al. (1997) [7] | Mesh systems; Mounted on artificial substrate | Psammocinia hawere Raspailia topsenti Raspailia (Clathriodendron) arbuscula | In-situ |
| Müller et al. (1999) [55] | In-vitro | Geodia cydonium | Ex-situ |
| Pronzato et al. (1999) [30] | Rope systems | Agelas oroides Axinella damicornis Cacospongia mollior Chondrosia reniformis Hippospongia communis Ircinia variabilis Petrosia ficiformis Spongia agaricina Spongia officinales | In-situ |
| Nickel & Brümmer (2002) [24] | In-vitro | Chondrosia reniformis | Ex-situ |
| Belarabi et al. (2003) [25] | In-vitro | Crambe crambe | Ex-situ |
| De Caralt et al. (2003) [19] | In-vitro | Corticium candelabrum | Ex-situ |
| Duckworth & Battershill (2003) [56] | Mesh systems; Rope systems | Latrunculia sp. nov. Polymastia crocea | In-situ |
| Duckworth et al. (2003) [57] | In-vitro | Axinella corrugate | Ex-situ |
| Thoms et al. (2003) [58] | Mounted on natural substrate | Aplysina cavernicola | In-situ |
| Van Treeck et al. (2003) [13] | Mesh systems | Axinella damicornis Axinella verrucose Chondrosia reniformis Ircinia variabilis | In-situ |
| Corriero et al. (2004) [9] | Rope systems | Spongia officinalis | In-situ |
| Duckworth et al. (2004) [59] | Mesh systems | Latrunculia (Biannulata) wellingtonensis Polymastia crocea | In-situ |
| Kelly et al. (2004) [14] | Mesh systems | Spongia (Heterofibria) manipulates | In-situ |
| Hadas et al. (2005) [60] | Mounted on artificial substrate; Rope systems | Negombata magnifica | In-situ |
| Hausmann et al. (2006) [26] | In-vitro | Aplysina aerophoba | Ex-situ |
| De Caralt et al. (2007) [61] | In-vitro | Crambe crambe Dysidea avara Hippospongia communis Ircinia oros | Ex-situ |
| De Voogd (2007) [10] | Mounted on artificial substrate; Rope systems | Callyspongia (Euplacella) biru | In-situ |
| De Voogd (2007) [62] | Rope systems | Aaptos suberitoides Amphimedon paraviridis Callyspongia (Euplacella) biru Hyrtios reticulatus Ircinia ramose | In-situ |
| Duckworth & Wolff (2007) [15] | Mesh systems; Mesh-rope systems; Rope systems | Coscinoderma sp. | In-situ |
| Duckworth et al. (2007) [63] | Mesh systems; Mounted on artificial substrate; Rope systems | Coscinoderma sp. Rhopaloeides odorabile | In-situ |
| Johnston & Clark (2007) [64] | Mounted on artificial substrate | Tedania (Tedania) anhelans | In-situ |
| Louden et al. (2007) [11] | In-vitro; Rope systems | Coscinoderma sp. Rhopaloeides odorabile | Ex-situ; In-situ |
| Lipton & Sunith (2009) [65] | In-vitro; Cage systems | Callyspongia (Chladochalina) diffusa Callyspongia (Chladochalina) subarmigera Clathria (Clathria) gorgonoides | Ex-situ; In-situ |
| Baldacconi et al. (2010) [51] | Mounted on artificial substrate | Spongia officinalis | In-situ |
| Carballo et al. (2010) [66] | In-vitro; Mounted on artificial substrate | Mycale (Carmia) Cecilia | Ex-situ; In-situ |
| De Caralt et al. (2010) [67] | Mesh systems; Mounted on artificial substrate, Rope systems | Dysidea avara | In-situ |
| Osinga et al. (2010) [12] | Cage systems; Rope systems | Chondrosia reniformis Dysidea avara | In-situ |
| Bergman et al. (2011) [68] | Mounted on artificial substrate | Diacarnus erythraenus | In-situ |
| Page et al. (2011) [69] | Mesh systems | Mycale (Clamia) hentscheli | In-situ |
| Webster et al. (2011) [27] | In-vitro; Mesh systems | Rhopaloeides odorabile | Ex-situ; In-situ |
| Oronti et al. (2012) [49] | Rope systems | Hyatella pertusa Spongia (Spongia) tubulifera | In-situ |
| Schiefenhövel & Kunzmann (2012) [17] | Mounted on artificial substrate; Mounted on natural substrate; Rope systems | Neopetrosia sp. Stylissa massa | Ex-situ; In-situ |
| Biggs (2013) [70] | Mounted on natural substrate | Aplysina cauliformis Aplysina sp. | In-situ |
| Ruiz et al. (2013) [50] | Mesh systems | Discoderma dissolute | In-situ |
| Di Bari et al. (2014) [71] | In-vitro | Tethya citrina | Ex-situ |
| Ledda et al. (2014) [31] | Rope system | Agelas oroides Ircinia variabilis | In-situ |
| Kiruba-Sankar et al. (2016) [18] | Mounted on artificial substrate; Mounted on natural substrate; Rope system | Liosina paradoxa Stylissa massa | Ex-situ; In-situ |
| Meyer et al. (2016) [16] | Cage systems; Mesh systems | Ecionemia alata | Ex-situ; In-situ |
| Avila & Briceno-Vera (2018) [72] | Mounted on artificial substrate | Halichondria (Halichondria) melanadocia | In-situ |
| Padiglia et al. (2018) [73] | Mounted on artificial substrate; Mounted on natural substrate | Crambe crambe | In-situ |
| Gökalp et al. (2019) [21] | Mounted on artificial substrate; Mesh system | Chondrosia reniformis | In-situ |
| Santiago et al. (2019) [74] | Mounted on artificial substrate; Mesh systems | Xestospongia sp. | In-situ |
| Giangrande et al. (2020) [32] | Mesh systems | Sarcotragus spinosulus | In-situ |
| Gökalp et al. (2021) [33] | Cage systems | Chondrosia reniformis | In-situ |
References
- Turner, E.C. Possible Poriferan Body Fossils in Early Neoproterozoic Microbial Reefs. Nature 2021, 596, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Moitinho-Silva, L.; Nielsen, S.; Amir, A.; Gonzalez, A.; Ackermann, G.L.; Cerrano, C.; Astudillo-Garcia, C.; Easson, C.; Sipkema, D.; Liu, F.; et al. The sponge microbiome project. Gigascience 2017, 6, gix077. [Google Scholar] [CrossRef] [PubMed]
- Cerrano, C.; Calcinai, B.; Pinca, S.; Bavestrello, G. Reef sponges as hosts of biodiversity: Cases from North Sulawesi. In Xth International Coral Reef Symposium Proceedings; Suzuki, Y., Nakamori, T., Hidaka, M., Kayanne, H., Casareto, B.E., Nadao, K., Yamano, H., Tsuchiya, M., Eds.; Japanese Coral Reef Societey: Okinawa, Japan, 2006; pp. 208–213. [Google Scholar]
- Wulff, J.L. Ecological interactions of marine sponges. Can. J. Zool. 2006, 84, 146–166. [Google Scholar] [CrossRef]
- Pronzato, R.; Manconi, R. Mediterranean Commercial Sponges: Over 5000 Years of Natural History and Cultural Heritage. Mar. Ecol. 2008, 29, 146–166. [Google Scholar] [CrossRef]
- Webster, N.S. Minireview. Sponge disease: A global threat? Environ. Microbiol. 2007, 9, 1363–1375. [Google Scholar] [CrossRef]
- Duckworth, A.R.; Battershill, C.N.; Bergquist, P.R. Influence of Explant Procedures and Environmental Factors on Culture Success of Three Sponges. Aquaculture 1997, 156, 251–267. [Google Scholar] [CrossRef]
- Pronzato, R. Sponge-Fishing, Disease and Farming in the Mediterranean. Aquat. Conserv. Mar. Freshw. Ecosyst. 1999, 9, 485–493. [Google Scholar] [CrossRef]
- Corriero, G.; Longo, C.; Mercurio, M.; Nonnis Marzano, C.; Lembo, G.; Spedicato, M.T. Rearing Performance of Spongia officinalis on Suspended Ropes off the Southern Italian Coast (Central Mediterranean Sea). Aquaculture 2004, 238, 195–205. [Google Scholar] [CrossRef]
- De Voogd, N.J. An Assessment of Sponge Mariculture Potential in the Spermonde Archipelago, Indonesia. J. Mar. Biol. Assoc. U. K. 2007, 87, 1777–1784. [Google Scholar] [CrossRef]
- Louden, D.; Whalan, S.; Evans-Illidge, E.; Wolff, C.; de Nys, R. An Assessment of the Aquaculture Potential of the Tropical Sponges Rhopaloeides odorabile and Coscinoderma sp. Aquaculture 2007, 270, 57–67. [Google Scholar] [CrossRef]
- Osinga, R.; Sidri, M.; Cerig, E.; Gokalp, S.Z.; Gokalp, M. Sponge Aquaculture Trials in the East–Mediterranean Sea: New Approaches to Earlier Ideas. Open Mar. Biol. J. 2010, 4, 74–81. [Google Scholar] [CrossRef]
- Van Treeck, P.; Eisinger, M.; Müller, J.; Paster, M.; Schuhmacher, H. Mariculture Trials with Mediterranean Sponge Species: The Exploitation of an Old Natural Resource with Sustainable and Novel Methods. Aquaculture 2003, 218, 439–455. [Google Scholar] [CrossRef]
- Kelly, M.; Handley, S.; Page, M.; Butterfield, P.; Hartill, B.; Kelly, S. Aquaculture Trials of the New Zealand Bath-Sponge Spongia (Heterofibria) Manipulates Using Lanterns. N. Zeal. J. Mar. Freshw. Res. 2004, 38, 231–241. [Google Scholar] [CrossRef]
- Duckworth, A.R.; Wolff, C. Bath Sponge Aquaculture in Torres Strait, Australia: Effect of Explant Size, Farming Method and the Environment on Culture Success. Aquaculture 2007, 271, 188–195. [Google Scholar] [CrossRef]
- Meyer, K.M.; Deines, P.; Schupp, P.J.; Taylor, M.W. Impact of Explantation Techniques on the Microbiota of the Marine Sponge Ecionemia Alata. J. Exp. Mar. Biol. Ecol. 2016, 484, 11–15. [Google Scholar] [CrossRef]
- Schiefenhövel, K.; Kunzmann, A. Sponge Farming Trials: Survival, Attachment, and Growth of Two Indo-Pacific Sponges, Neopetrosia sp. and Stylissa massa. J. Mar. Biol. 2012, 2012, 417360. [Google Scholar] [CrossRef][Green Version]
- Kiruba Sankar, R.; Chadha, N.K.; Dam Roy, S.; Banerjee, P.; Saharan, N.; Krishnan, P. Growth and Survival of Marine Sponges, Stylissa massa (Carter, 1887) and Liosina paradoxa (Thiele, 1899) in Sea and Land Based Culture Systems. Indian J. Fish. 2016, 63, 55–60. [Google Scholar] [CrossRef][Green Version]
- De Caralt, S.; Agell, G.; Uriz, M.J. Long-term culture of sponge explants: Conditions enhancing survival and growth, and assessment of bioactivity. Biomol. Eng. 2003, 20, 339–347. [Google Scholar] [CrossRef]
- Duckworth, A. Farming Sponges to Supply Bioactive Metabolites and Bath Sponges: A Review. Mar. Biotechnol. 2009, 11, 669–679. [Google Scholar] [CrossRef]
- Gökalp, M.; Wijgerde, T.; Sarà, A.; De Goeij, J.M.; Osinga, R. Development of an Integrated Mariculture for the Collagen-Rich Sponge Chondrosia reniformis. Mar. Drugs 2019, 17, 29. [Google Scholar] [CrossRef] [PubMed]
- Schippers, K.J.; Sipkema, D.; Osinga, R.; Smidt, H.; Pomponi, S.A.; Martens, D.E.; Wijffels, R.H. Cultivation of Sponges, Sponge Cells and Symbionts. Achievements and Future Prospects. Adv. Mar. Biol. 2012, 62, 273–337. [Google Scholar] [CrossRef]
- Leal, M.C.; Puga, J.; Serôdio, J.; Gomes, N.C.M.; Calado, R. Trends in the Discovery of New Marine Natural Products from Invertebrates over the Last Two Decades—Where and What Are We Bioprospecting? PLoS ONE 2012, 7, e30580. [Google Scholar] [CrossRef] [PubMed]
- Nickel, M.; Brümmer, F. In Vitro Sponge Fragment Culture of Chondrosia reniformis (Nardo, 1847). J. Biotechnol. 2003, 100, 147–159. [Google Scholar] [CrossRef]
- Belarbi, E.H.; Ramírez Domínguez, M.; Cerón García, M.C.; Contreras Gómez, A.; García Camacho, F.; Molina Grima, E. Cultivation of Explants of the Marine Sponge Crambe crambe in Closed Systems. Biomol. Eng. 2003, 20, 333–337. [Google Scholar] [CrossRef]
- Hausmann, R.; Vitello, M.P.; Leitermann, F.; Syldatk, C. Advances in the Production of Sponge Biomass Aplysina aerophoba-A Model Sponge for Ex Situ Sponge Biomass Production. J. Biotechnol. 2006, 124, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Webster, N.S.; Cobb, R.E.; Soo, R.; Anthony, S.L.; Battershill, C.N.; Whalan, S.; Evans-Illidge, E. Bacterial Community Dynamics in the Marine Sponge Rhopaloeides odorabile Under In Situ and Ex Situ Cultivation. Mar. Biotechnol. 2011, 13, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Pronzato, R.; Cerrano, C.; Cubeddu, T.; Lanza, S.; Magnino, G.; Manconi, R.; Pantelis, J.; Sarà, A.; Sidri, M. Aquaculture and water: Fish culture, shellfish culture and water usage sustainable development in coastal areas: Role of sponge farming in integrated aquaculture. In Aquaculture Europe; European Aquaculture Society: Bordeaux, France, 1998. [Google Scholar] [CrossRef]
- Gökalp, M. The Multifunctional Role of Marine Sponges in Multi-Trophic Mariculture Systems. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2021. [Google Scholar]
- Pronzato, R.; Bavestrello, G.; Cerrano, C.; Magnino, G.; Manconi, R.; Pantelis, J.; Sarà, A.; Sidri, M. Sponge Farming in the Mediterranean Sea: New Perspectives. Mem. Queensl. Mus. 1999, 44, 485–491. [Google Scholar]
- Ledda, F.D.; Pronzato, R.; Manconi, R. Mariculture for Bacterial and Organic Waste Removal: A Field Study of Sponge Filtering Activity in Experimental Farming. Aquac. Res. 2014, 45, 1389–1401. [Google Scholar] [CrossRef]
- Giangrande, A.; Pierri, C.; Arduini, D.; Borghese, J.; Licciano, M.; Trani, R.; Corriero, G.; Basile, G.; Cecere, E.; Petrocelli, A.; et al. An Innovative IMTA System: Polychaetes, Sponges and Macroalgae Co-Cultured in a Southern Italian in-Shore Mariculture Plant (Ionian Sea). J. Mar. Sci. Eng. 2020, 8, 733. [Google Scholar] [CrossRef]
- Gökalp, M.; Wijgerde, T.; Murk, A.; Osinga, R. Design for Large-Scale Maricultures of the Mediterranean Demosponge Chondrosia Reniformis Nardo, 1847 for Collagen Production. Aquaculture 2022, 548, 737702. [Google Scholar] [CrossRef]
- Sipkema, D.; Osinga, R.; Schatton, W.; Mendola, D.; Tramper, J.; Wijffels, R.H. Large-Scale Production of Pharmaceuticals by Marine Sponges: Sea, Cell, or Synthesis? Biotechnol. Bioeng. 2005, 90, 201–222. [Google Scholar] [CrossRef]
- Maslin, M.; Gaertner-Mazouni, N.; Debitus, C.; de Voogd, N.J.; Ho, R. Marine Sponge Aquaculture towards Drug Development: An Ongoing History of Technical, Ecological, Chemical Considerations and Challenges. Aquac. Rep. 2021, 21, 100813. [Google Scholar] [CrossRef]
- Osinga, R.; Tramper, J.; Wijffels, R.H. Cultivation of Marine Sponges. Mar. Biotechnol. 1999, 1, 509–532. [Google Scholar] [CrossRef] [PubMed]
- Spalding, M.D.; Fox, H.E.; Allen, G.R.; Davidson, N.; Ferdaña, Z.A.; Finlayson, M.; Halpern, B.S.; Jorge, M.A.; Lombana, A.; Lourie, S.A.; et al. Marine Ecoregions of the World: A Bioregionalization of Coastal and Shelf Areas. Bioscience 2007, 57, 573–583. [Google Scholar] [CrossRef]
- Cebrian, E.; Tamburello, L.; Verdura, J.; Guarnieri, G.; Medrano, A.; Linares, C.; Hereu, B.; Garrabou, J.; Cerrano, C.; Galobart, C.; et al. A Roadmap for the Restoration of Mediterranean Macroalgal Forests. Front. Mar. Sci. 2021, 8, 709219. [Google Scholar] [CrossRef]
- Bekkby, T.; Papadopoulou, N.; Fiorentino, D.; McOwen, C.J.; Rinde, E.; Boström, C.; Carreiro-Silva, M.; Linares, C.; Andersen, G.S.; Bengil, E.G.T.; et al. Habitat Features and Their Influence on the Restoration Potential of Marine Habitats in Europe. Front. Mar. Sci. 2020, 7, 184. [Google Scholar] [CrossRef]
- Manconi, R.; Cubeddu, T.; Corriero, G.; Pronzato, R. Commercial sponges farming as natural control of floating cages pollution. In New Species for Mediterranean Aquaculture; Enne, G., Greppi, G.F., Eds.; Elsevier: Amsterdam, The Netherlands, 1999; pp. 269–274. [Google Scholar]
- Pronzato, R. Mediterranean Sponge Fauna: A Biological, Historical and Cultural Heritage. Biogeogr.–J. Integr. Biogeogr. 2003, 24, 91–98. [Google Scholar] [CrossRef]
- Gaino, E.; Pronzato, R. Disease and overfishing drive commercial sponges to the brink of extinction in the Mediterranean basin. Boll. Mus. Ist. Biol. Univ. Genova 1992, 56–57, 209–224. [Google Scholar]
- Cerrano, C.; Bavestrello, G.; Bianchi, C.N.; Cattaneo-Vietti, R.; Bava, S.; Morganti, C.; Morri, C.; Picco, P.; Sara, G.; Schiaparelli, S.; et al. A Catastrophic Mass-Mortality Episode of Gorgonians and Other Organisms in the Ligurian Sea (North-Western Mediterranean), Summer 1999. Ecol. Lett. 2000, 3, 284–293. [Google Scholar] [CrossRef]
- Voultsiadou, E.; Gerovasileiou, V.; Dailianis, T. Extinction Trends of Marine Species and Populations in the Aegean Sea and Adjacent Ecoregions. CIESM Work. Monogr. 2013, 45, 58–73. [Google Scholar]
- Butler Iv, M.J. Cascading Disturbances in Florida Bay, USA: Cyanobacteria Blooms, Sponge Mortality, and Implications for Juvenile Spiny Lobsters Panulirus argus. Mar. Ecol. Prog. Ser. 1995, 129, 119–125. [Google Scholar] [CrossRef]
- Berman, J. Patterns of Temporal and Spatial Variability of Sponge Assemblages. Ph.D. Thesis, Victoria University of Wellington, Wellington, New Zealand, 2012. [Google Scholar]
- Giangrande, A.; Gravina, M.F.; Rossi, S.; Longo, C.; Pierri, C. Aquaculture and Restoration: Perspectives from Mediterranean Sea Experiences. Water 2021, 13, 991. [Google Scholar] [CrossRef]
- Verdenal, B.; Vacelet, J. Sponge Culture on Vertical Ropes in the Northwestern Mediterranean Sea. In New Perspectives in Sponge Biology; Rützler, K., Ed.; Smithsonian Institution Press: Washington, DC, USA, 1990; pp. 416–424. [Google Scholar]
- Oronti, A.; Danylchuk, A.J.; Elmore, C.E.; Auriemma, R.; Pesle, G. Assessing the Feasibility of Sponge Aquaculture as a Sustainable Industry in The Bahamas. Aquac. Int. 2012, 20, 295–303. [Google Scholar] [CrossRef]
- Ruiz, C.; Valderrama, K.; Zea, S.; Castellanos, L. Mariculture and Natural Production of the Antitumoural (+)-Discodermolide by the Caribbean Marine Sponge Discodermia dissoluta. Mar. Biotechnol. 2013, 15, 571–583. [Google Scholar] [CrossRef] [PubMed]
- Baldacconi, R.; Cardone, F.; Longo, C.; Mercurio, M.; Marzano, C.N.; Gaino, E.; Corriero, G. Transplantation of Spongia officinalis L. (Porifera, Demospongiae): A Technical Approach for Restocking This Endangered Species. Mar. Ecol. 2010, 31, 309–317. [Google Scholar] [CrossRef]
- Wilkinson, C.R.; Vacelet, J. Transplantation of Marine Sponges to Different Conditions of Light and Current. J. Exp. Mar. Biol. Ecol. 1979, 37, 91–104. [Google Scholar] [CrossRef]
- Barthel, D. and Theede, H. A new method for the culture of marine sponges and its application for experimental studies. Ophelia 1986, 25, 75–82. [Google Scholar] [CrossRef]
- Kaandorp, J.A.; de Kluijver, M.J. Verification of Fractal Growth Models of the Sponge Haliclona oculata (Porifera) with Transplantation Experiments. Mar. Biol. 1992, 113, 133–143. [Google Scholar] [CrossRef]
- Müller, W.E.; Wimmer, W.; Schatton, W.; Böhm, M.; Batel, R.; Filic, Z. Initiation of an aquaculture of sponges for the sustainable production of bioactive metabolites in open systems: Example, Geodia cydonium. Mar. Biotechnol. 1999, 1, 569–579. [Google Scholar] [CrossRef]
- Duckworth, A.R.; Battershill, C.N. Developing Farming Structures for Production of Biologically Active Sponge Metabolites. Aquaculture 2003, 217, 139–156. [Google Scholar] [CrossRef]
- Duckworth, A.R.; Samples, G.A.; Wright, A.E.; Pomponi, S.A. In Vitro Culture of the Tropical Sponge Axinella corrugata (Demospongiae): Effect of Food Cell Concentration on Growth, Clearance Rate, and Biosynthesis of Stevensine. Mar. Biotechnol. 2003, 5, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Thoms, C.; Horn, M.; Wagner, M.; Hentschel, U.; Proksch, P. Monitoring Microbial Diversity and Natural Product Profiles of the Sponge Aplysina cavernicola Following Transplantation. Mar. Biol. 2003, 142, 685–692. [Google Scholar] [CrossRef]
- Duckworth, A.R.; Battershill, C.N.; Schiel, D.R. Effects of Depth and Water Flow on Growth, Survival and Bioactivity of Two Temperate Sponges Cultured in Different Seasons. Aquaculture 2004, 242, 237–250. [Google Scholar] [CrossRef]
- Hadas, E.; Shpigel, M.; Ilan, M. Sea Ranching of the Marine Sponge Negombata magnifica (Demospongiae, Latrunculiidae) as a First Step for Latrunculin B Mass Production. Aquaculture 2005, 244, 159–169. [Google Scholar] [CrossRef]
- De Caralt, S.; Otjens, H.; Uriz, M.J.; Wijffels, R.H. Cultivation of Sponge Larvae: Settlement, Survival, and Growth of Juveniles. Mar. Biotechnol. 2007, 9, 592–605. [Google Scholar] [CrossRef]
- de Voogd, N.J. The Mariculture Potential of the Indonesian Reef-Dwelling Sponge Callyspongia (Euplacella) biru: Growth, Survival and Bioactive Compounds. Aquaculture 2007, 262, 54–64. [Google Scholar] [CrossRef]
- Duckworth, A.R.; Wolff, C.; Evans-Illidge, E. Developing Methods for Commercially Farming Bath Sponges in Tropical Australia. In Porifera Research: Biodiversity, Innovation and Sustainability; Custodio, M.R., Lobo-Hajdu, G., Hajdu, E., Muricy, G., Eds.; Museu Nacional: Rio de Janeiro, Brazil, 2007; pp. 297–302. [Google Scholar]
- Johnston, E.L.; Clark, G.F. Recipient Environment More Important than Community Composition in Determining the Success of an Experimental Sponge Transplant. Restor. Ecol. 2007, 15, 638–651. [Google Scholar] [CrossRef]
- Lipton, A.; Sunith, S. Mariculture of Marine Sponges for Drug Development: Bioactivity Potentials of Cultured Sponges, Callyspongia subarmigera (Ridley) and Echinodictyum gorgonoides (Dendy). Mar. Fish. Inf. Serv. T&E Ser. 2009, 202, 7–10. [Google Scholar]
- Carballo, J.L.; Yañez, B.; Zubía, E.; Ortega, M.J.; Vega, C. Culture of Explants from the Sponge Mycale Cecilia to Obtain Bioactive Mycalazal-Type Metabolites. Mar. Biotechnol. 2010, 12, 516–525. [Google Scholar] [CrossRef] [PubMed]
- De Caralt, S.; Sánchez-Fontenla, J.; Uriz, M.J.; Wijffels, R.H. In Situ Aquaculture Methods for Dysidea Avara (Demospongiae, Porifera) in the Northwestern Mediterranean. Mar. Drugs 2010, 8, 1731–1742. [Google Scholar] [CrossRef]
- Bergman, O.; Haber, M.; Mayzel, B.; Anderson, M.A.; Shpigel, M.; Hill, R.T.; Ilan, M. Marine-Based Cultivation of Diacarnus Sponges and the Bacterial Community Composition of Wild and Maricultured Sponges and Their Larvae. Mar. Biotechnol. 2011, 13, 1169–1182. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; Handley, S.J.; Northcote, P.T.; Cairney, D.; Willan, R.C. Successes and Pitfalls of the Aquaculture of the Sponge Mycale hentscheli. Aquaculture 2011, 312, 52–61. [Google Scholar] [CrossRef]
- Biggs, B.C. Harnessing Natural Recovery Processes to Improve Restoration Outcomes: An Experimental Assessment of Sponge-Mediated Coral Reef Restoration. PLoS ONE 2013, 8, e64945. [Google Scholar] [CrossRef] [PubMed]
- Di Bari, G.; Cardone, F.; Gaino, E.; Liuzzi, G.M.; Nonnis Marzano, C.; Scoccia, F.; Corriero, G. Biological Variations in a Long-Term Ex Situ Cultivation: A Mediterranean Demosponge as Model System. Mediterr. Mar. Sci. 2015, 16, 73–81. [Google Scholar] [CrossRef]
- Ávila, E.; Briceño-Vera, A.E. A Reciprocal Inter-Habitat Transplant Reveals Changes in the Assemblage Structure of Macroinvertebrates Associated with the Sponge Halichondria melanadocia. Estuaries Coasts 2018, 41, 1397–1409. [Google Scholar] [CrossRef]
- Padiglia, A.; Ledda, F.D.; Padedda, B.M.; Pronzato, R.; Manconi, R. Long-term experimental in situ farming of Crambe crambe (Demospongiae: Poecilosclerida). PeerJ 2018, 6, e4964. [Google Scholar] [CrossRef]
- Santiago, V.S.; Manzano, G.G.; Yu, C.C.; Aliño, P.M.; Salvador-Reyes, L.A. Mariculture Potential of Renieramycin-Producing Philippine Blue Sponge Xestospongia sp. (Porifera: Haplosclerida). Aquaculture 2019, 502, 356–364. [Google Scholar] [CrossRef]
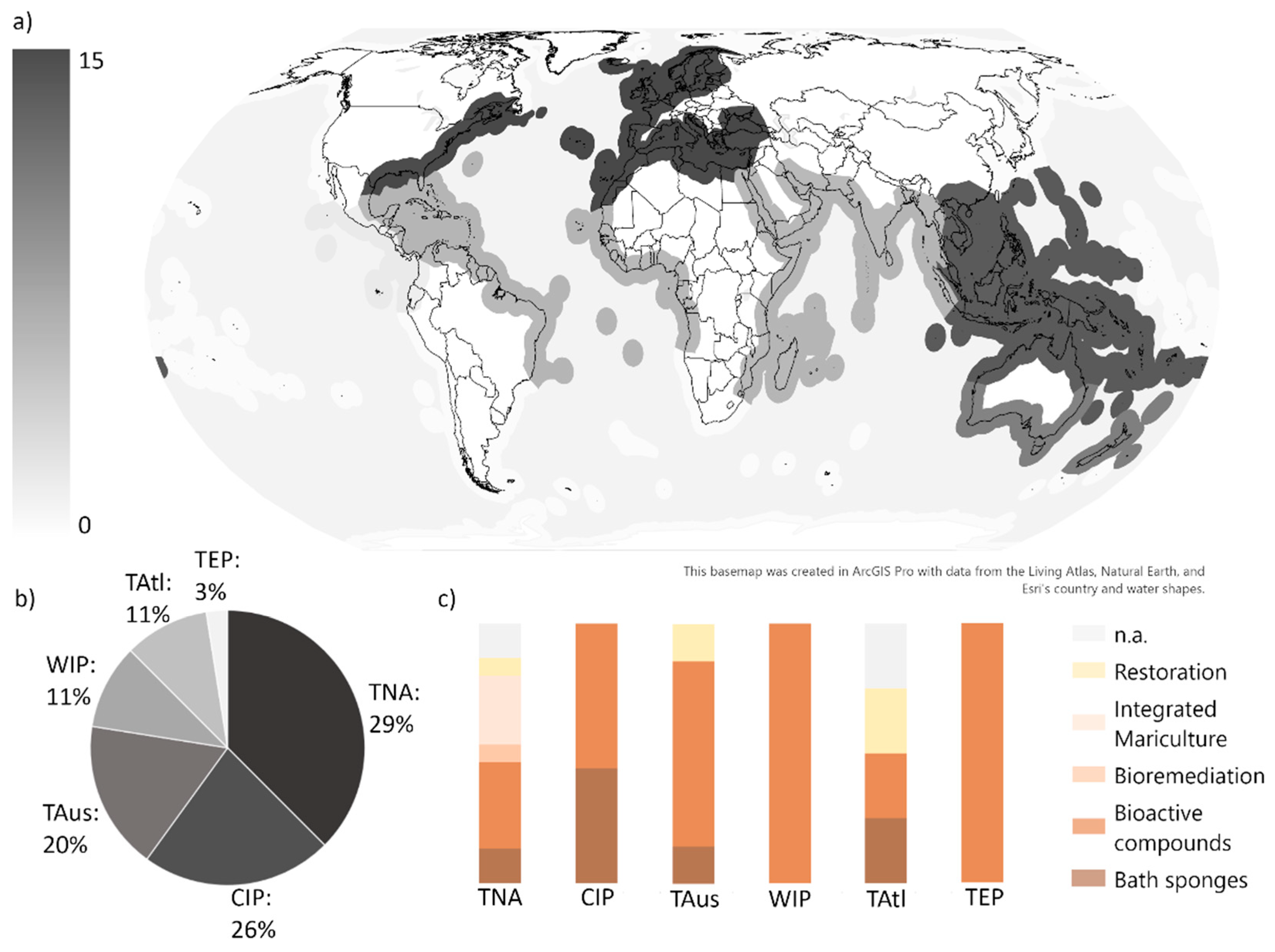
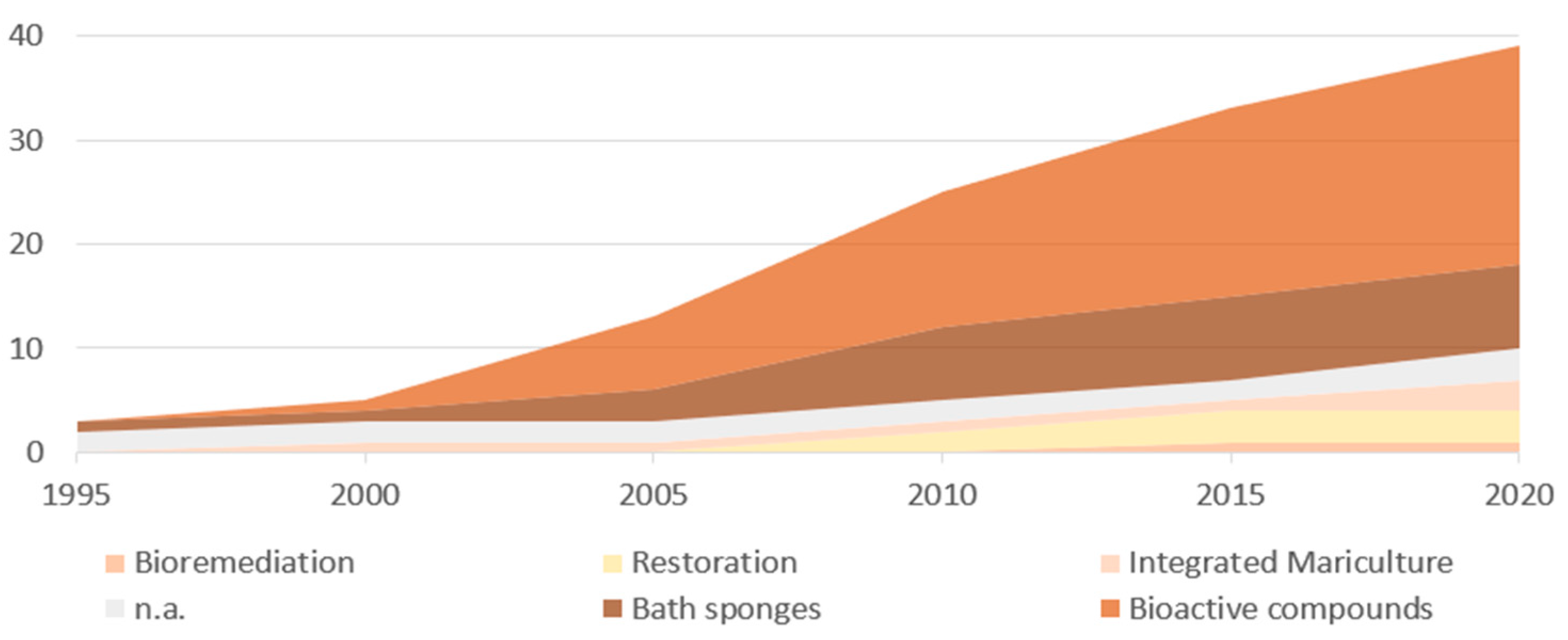

| Criteria | Specification |
|---|---|
| Years | 1950–2022 |
| Search terms | “marine sponge transplant(-ation)” |
| “marine sponge cultivation” | |
| “marine sponge explant(s)” | |
| “restoration marine sponge(s) | |
| “sponge aquaculture” | |
| “sponge farm(-ing)” | |
| Databases | Web of Science |
| Google Scholar | |
| ResearchGate | |
| Inclusion criteria | Studies published between the period 1950–2021 |
| Studies including information about transplantation of marine sponges | |
| Studies experimenting transplantation methods | |
| Peer-reviewed studies | |
| Published in English, French, or German language | |
| Available in selected databases | |
| Exclusion criteria | Studies outside marine sponge transplantation-cultivation domain |
| No peer-reviewed studies | |
| Duplicated among databases |
| Technique | Description |
|---|---|
| Cage systems | Sponges cultivated inside a cage structure with a solid (not mesh) bottom part. Cage may be covered or uncovered on the top. |
| Mesh systems | Sponges cultivated in enclosed mesh arrays (completely surrounded by mesh). Can be “squeezed” between panels or held loosely in more spacious arrays (e.g., oyster/mussel panels or lanterns). |
| Mesh-rope systems | Sponges initially cultivated in mesh arrays (see Mesh systems) before transferred to a rope array (see Rope systems) |
| Mounted on artificial substrate | Sponges cultivated attached/on top of artificial substrates (e.g., cement blocks, plastic trays). Category also assigned when farmed on top of mesh arrays (e.g., mesh grid) instead of enclosed by it. |
| Mounted on natural substrate | Sponges are farmed attached/on top of artificial substrates (e.g., live rock, coral boulder, oyster shells) |
| Rope systems | Sponges cultivated using ropes. Can either be single lines or large arrays spanning across artificial frameworks. No differentiation between vertical or horizontal orientation. Sponges may be threaded directly on ropes, attached to ropes (e.g., using zip ties or thin ropes) or hung on ropes using small pieces of rope (or other material such as zip ties) forming a loop |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bierwirth, J.; Mantas, T.P.; Villechanoux, J.; Cerrano, C. Restoration of Marine Sponges—What Can We Learn from over a Century of Experimental Cultivation? Water 2022, 14, 1055. https://doi.org/10.3390/w14071055
Bierwirth J, Mantas TP, Villechanoux J, Cerrano C. Restoration of Marine Sponges—What Can We Learn from over a Century of Experimental Cultivation? Water. 2022; 14(7):1055. https://doi.org/10.3390/w14071055
Chicago/Turabian StyleBierwirth, Jan, Torcuato Pulido Mantas, Juliette Villechanoux, and Carlo Cerrano. 2022. "Restoration of Marine Sponges—What Can We Learn from over a Century of Experimental Cultivation?" Water 14, no. 7: 1055. https://doi.org/10.3390/w14071055
APA StyleBierwirth, J., Mantas, T. P., Villechanoux, J., & Cerrano, C. (2022). Restoration of Marine Sponges—What Can We Learn from over a Century of Experimental Cultivation? Water, 14(7), 1055. https://doi.org/10.3390/w14071055







