Construction of Novel Electro-Fenton Systems by Magnetically Decorating Zero-Valent Iron onto RuO2-IrO2/Ti Electrode for Highly Efficient Pharmaceutical Wastewater Treatment
Abstract
:1. Introduction
2. Materials and Characterizations
2.1. Materials
2.2. Analysis and Characterization Methods
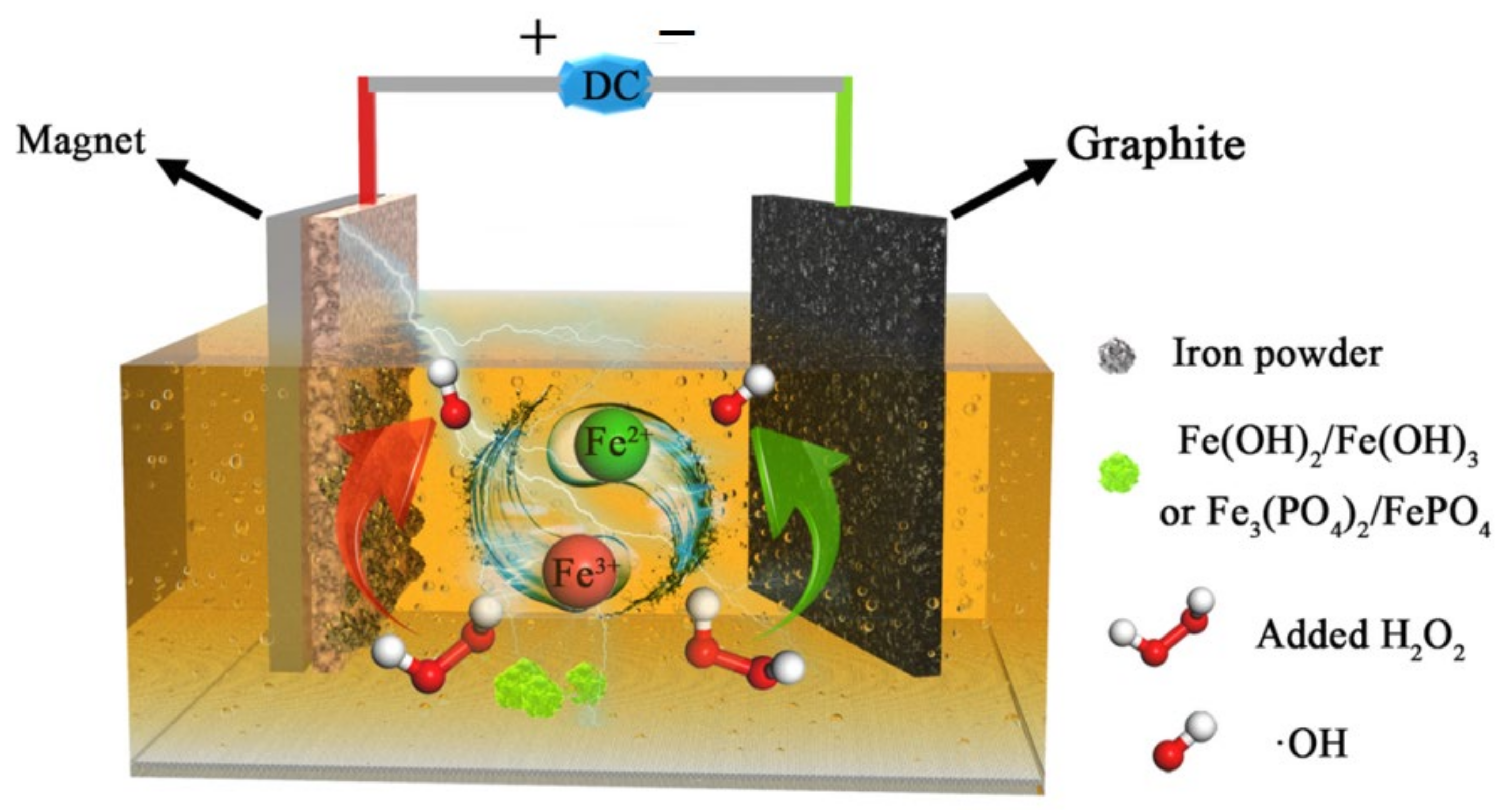
2.3. Experimental Setup and Operation
3. Results and Discussion
3.1. Influencing Factors of the Constructed Novel E-Fenton System
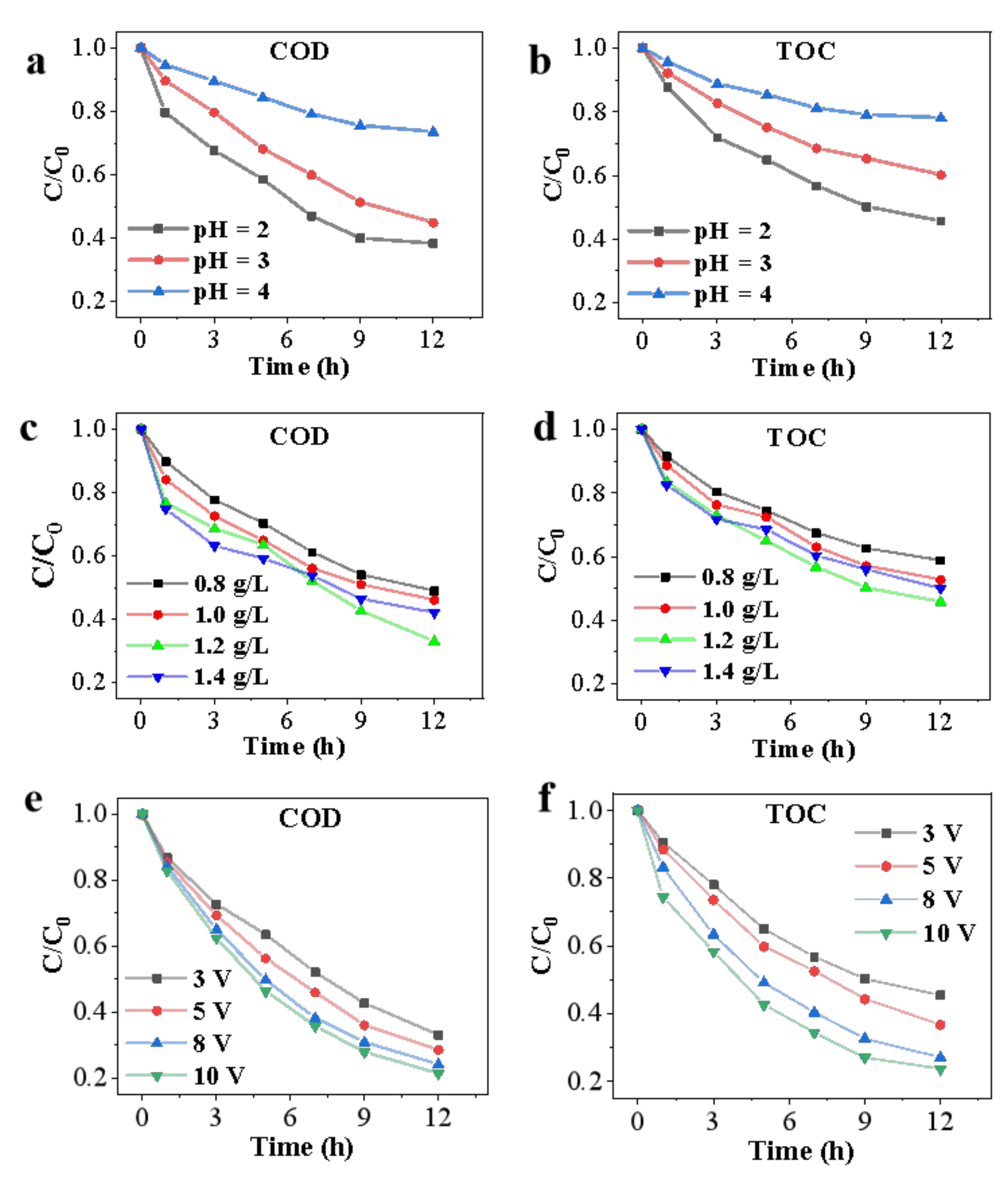
3.1.1. Initial pH Value
3.1.2. ZVI Dosage
3.1.3. Applied Voltage
3.1.4. Na2SO4 Concentration
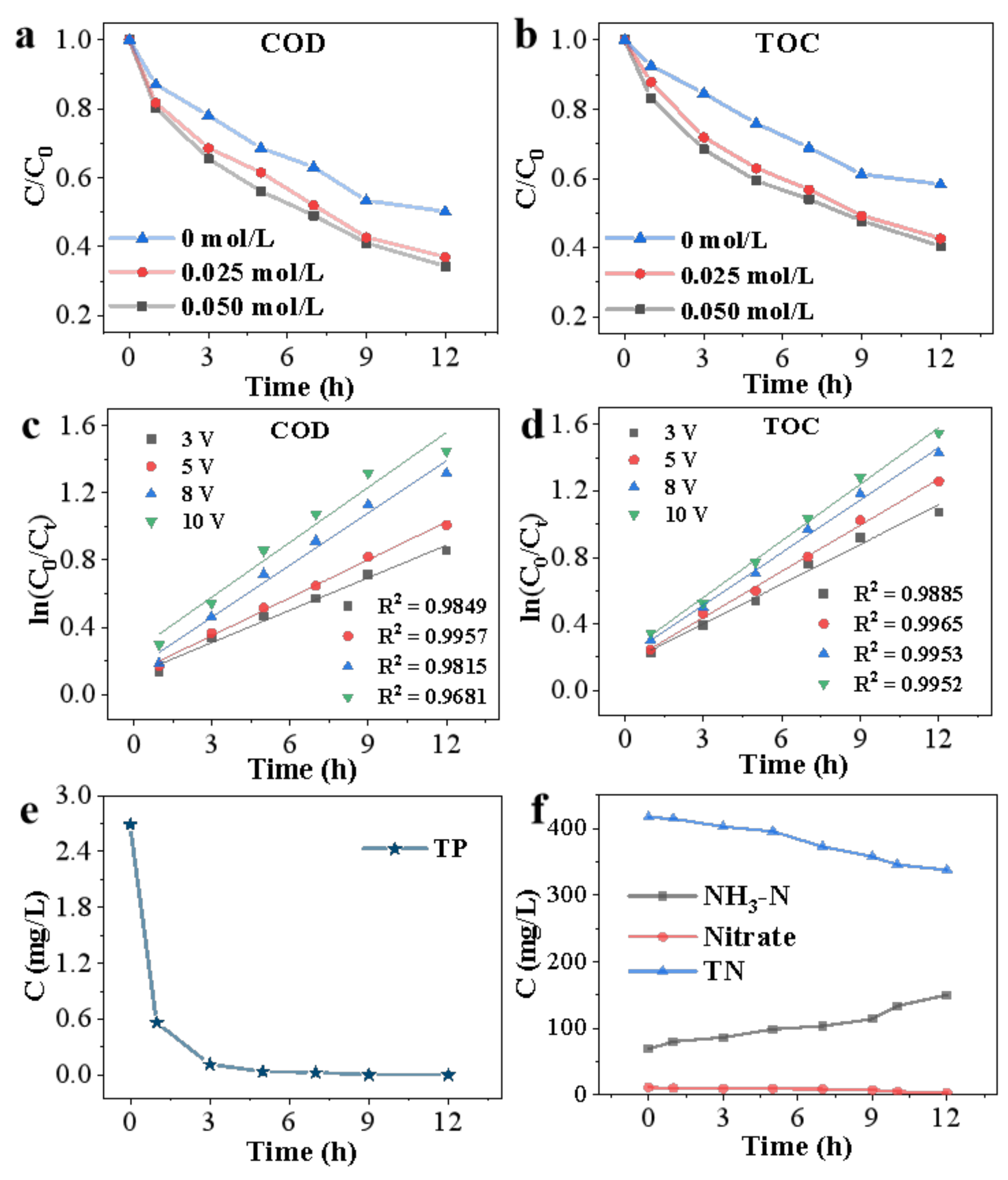
3.2. Kinetics of COD and TOC Removal
3.3. Overall Treatment Efficiency under Optimal Conditions
3.3.1. Total Phosphorous Removal
3.3.2. Transformation of Nitrogen and Its Species
3.3.3. Removal Performance of Tetracycline
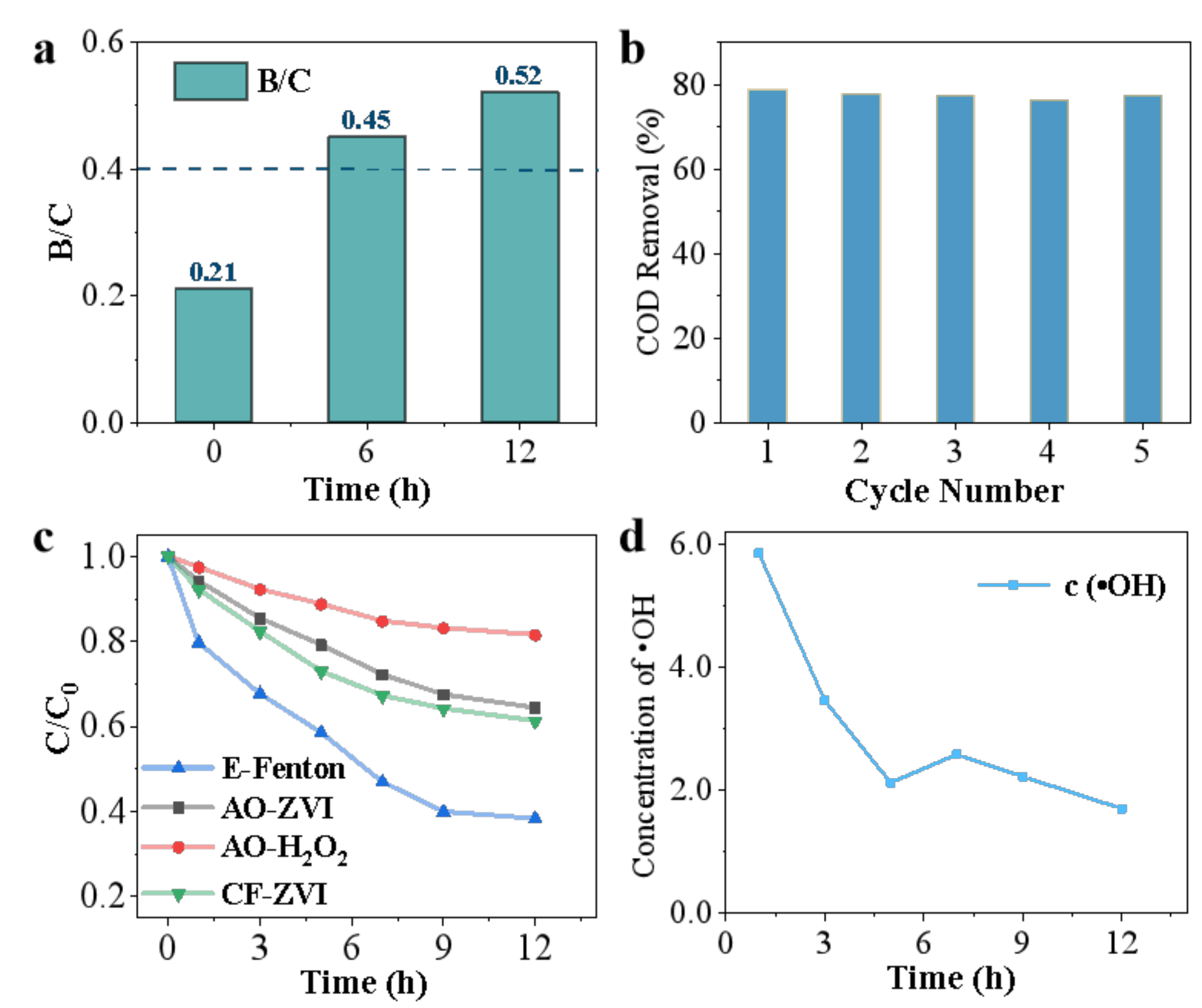
3.4. Biodegradability Analysis
3.5. Reusability of Anode Material
3.6. Treatment Mechanism Clarifications
3.6.1. Role of Additional H2O2
3.6.2. The Concentration of ·OH
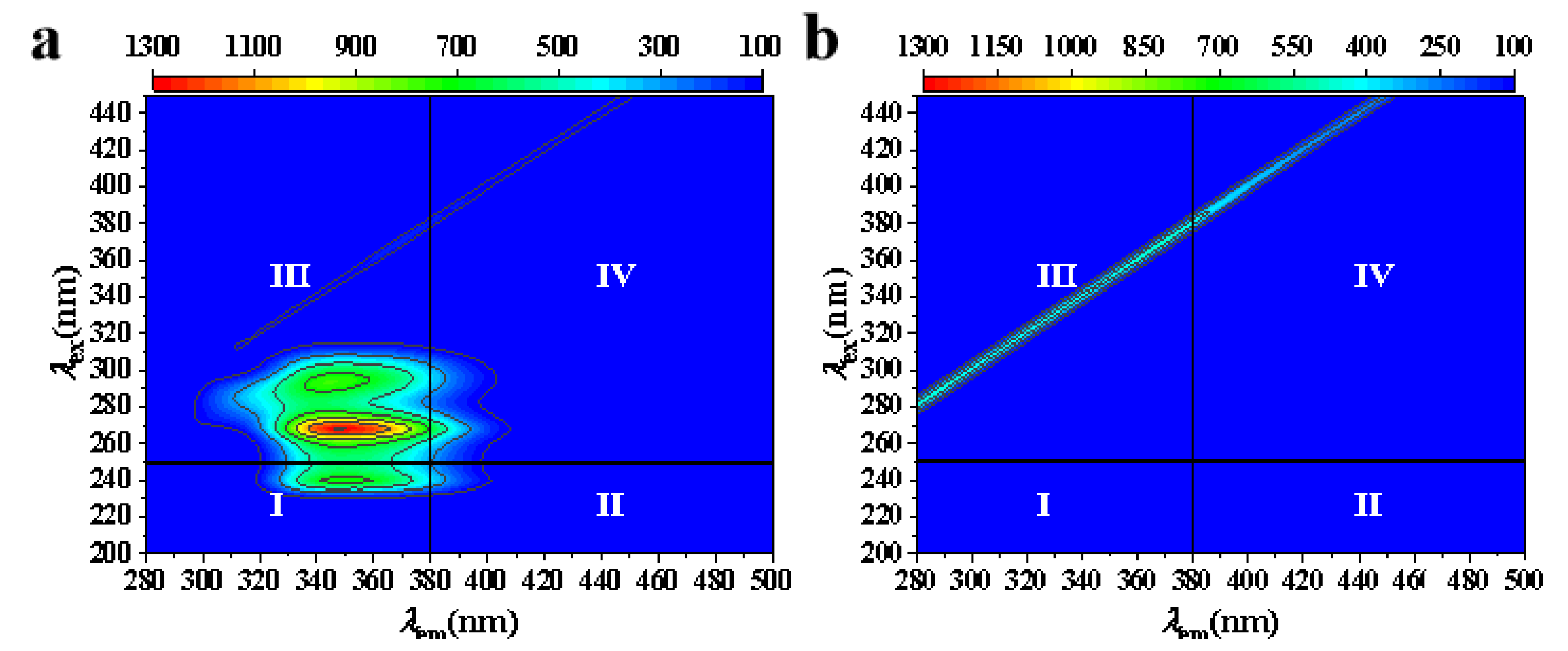
3.6.3. Three-Dimensional Fluorescence Analysis
3.6.4. Precipitate Characterization
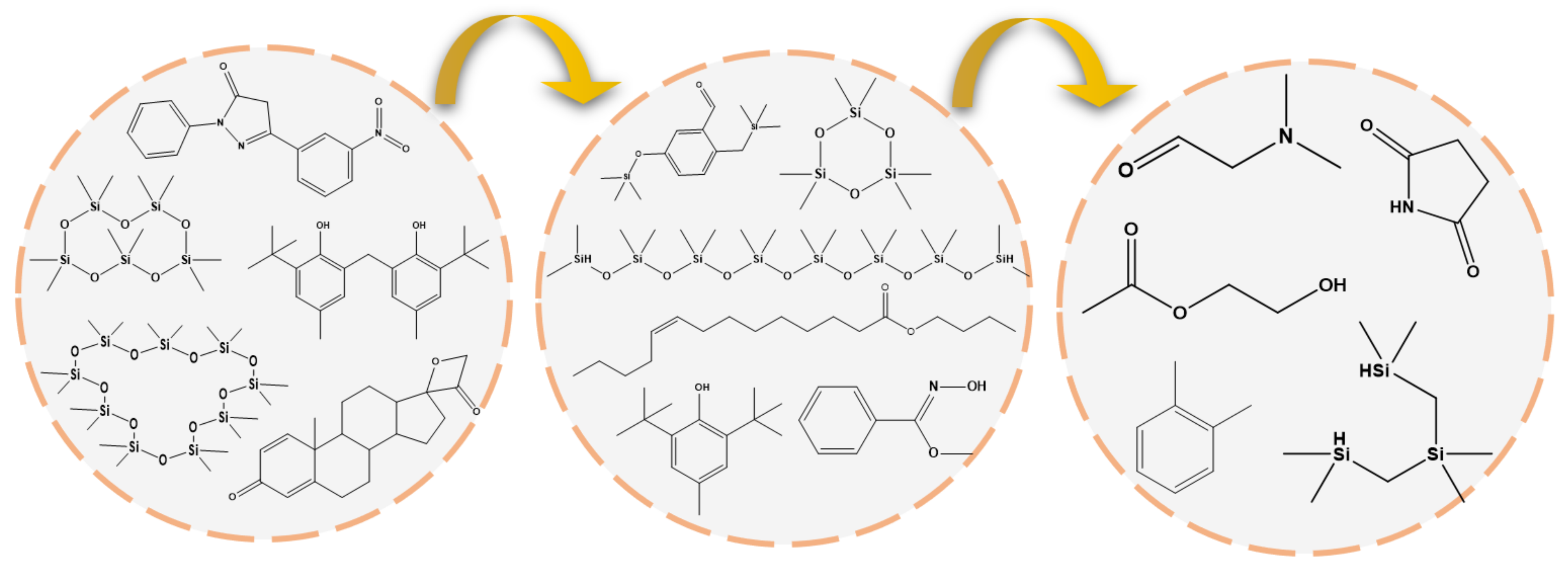
3.6.5. Byproducts and Possible Degradation Pathway
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jiang, Y.; Shi, X.; Ng, H.Y. Aerobic granular sludge systems for treating hypersaline pharmaceutical wastewater: Start-up, long-term performances and metabolic function. J. Hazard. Mater. 2021, 412, 125229. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Gao, T.; Salama, E.-S.; El-Dalatony, M.M.; Peng, L.; Gong, Y.; Liu, P.; Li, X. Using Aspergillus niger whole-cell biocatalyst mycelial aerobic granular sludge to treat pharmaceutical wastewater containing β-lactam antibiotics. Chem. Eng. J. 2021, 412, 128665. [Google Scholar] [CrossRef]
- Ao, X.-W.; Eloranta, J.; Huang, C.-H.; Santoro, D.; Sun, W.-J.; Lu, Z.-D.; Li, C. Peracetic acid-based advanced oxidation processes for decontamination and disinfection of water: A review. Water Res. 2021, 188, 116479. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Jiang, J.; Ma, L.; Wang, Y.; Liang, M.; Zhang, Z.; Li, L. Iron foam combined ozonation for enhanced treatment of pharmaceutical wastewater. Environ. Res. 2020, 183, 109205. [Google Scholar] [CrossRef]
- Li, C.; Mei, Y.; Qi, G.; Xu, W.; Zhou, Y.; Shen, Y. Degradation characteristics of four major pollutants in chemical pharmaceutical wastewater by Fenton process. J. Environ. Chem. Eng. 2021, 9, 104564. [Google Scholar] [CrossRef]
- Ling, L.; Liu, Y.; Pan, D.; Lyu, W.; Xu, X.; Xiang, X.; Lyu, M.; Zhu, L. Catalytic detoxification of pharmaceutical wastewater by Fenton-like reaction with activated alumina supported CoMnAl composite metal oxides catalyst. Chem. Eng. J. 2020, 381, 122607. [Google Scholar] [CrossRef]
- Kantar, C.; Oral, O.; Oz, N.A. Ligand enhanced pharmaceutical wastewater treatment with Fenton process using pyrite as the catalyst: Column experiments. Chemosphere 2019, 237, 124440. [Google Scholar] [CrossRef]
- Chávez, A.M.; Gimeno, O.; Rey, A.; Pliego, G.; Oropesa, A.L.; Álvarez, P.M.; Beltrán, F. Treatment of highly polluted industrial wastewater by means of sequential aerobic biological oxidation-ozone based AOPs. Chem. Eng. J. 2019, 361, 89–98. [Google Scholar] [CrossRef]
- Xie, Z.-H.; Zhou, H.-Y.; He, C.-S.; Pan, Z.-C.; Yao, G.; Lai, B. Synthesis, application and catalytic performance of layered double hydroxide based catalysts in advanced oxidation processes for wastewater decontamination: A review. Chem. Eng. J. 2021, 414, 128713. [Google Scholar] [CrossRef]
- Kilic, M.Y.; Abdelraheem, W.H.; He, X.; Kestioglu, K.; Dionysiou, D.D. Photochemical treatment of tyrosol, a model phenolic compound present in olive mill wastewater, by hydroxyl and sulfate radical-based advanced oxidation processes (AOPs). J. Hazard. Mater. 2019, 367, 734–742. [Google Scholar] [CrossRef]
- Bansal, P.; Verma, A.; Talwar, S. Detoxification of real pharmaceutical wastewater by integrating photocatalysis and photo-Fenton in fixed-mode. Chem. Eng. J. 2018, 349, 838–848. [Google Scholar] [CrossRef]
- Li, D.; Zheng, T.; Liu, Y.; Hou, D.; He, H.; Song, H.; Zhang, J.; Tian, S.; Zhang, W.; Wang, L.; et al. A cost-effective Electro-Fenton process with graphite felt electrode aeration for degradation of dimethyl phthalate: Enhanced generation of H2O2 and iron recycling that simultaneously regenerates the electrode. Chem. Eng. J. 2020, 394, 125033. [Google Scholar] [CrossRef]
- Monteil, H.; Péchaud, Y.; Oturan, N.; Oturan, M.A. A review on efficiency and cost effectiveness of electro- and bio-electro-Fenton processes: Application to the treatment of pharmaceutical pollutants in water. Chem. Eng. J. 2019, 376, 119577. [Google Scholar] [CrossRef]
- Ye, Z.; Schukraft, G.E.M.; L’Hermitte, A.; Xiong, Y.; Brillas, E.; Petit, C.; Sirés, I. Mechanism and stability of an Fe-based 2D MOF during the photoelectro-Fenton treatment of organic micropollutants under UVA and visible light irradiation. Water Res. 2020, 184, 115986. [Google Scholar] [CrossRef]
- Divyapriya, G.; Srinivasan, R.; Nambi, I.M.; Senthilnathan, J. Highly active and stable ferrocene functionalized graphene encapsulated carbon felt array—A novel rotating disc electrode for electro-Fenton oxidation of pharmaceutical compounds. Electrochim. Acta 2018, 283, 858–870. [Google Scholar] [CrossRef]
- Mousset, E.; Pontvianne, S.; Pons, M.-N. Fate of inorganic nitrogen species under homogeneous Fenton combined with electro-oxidation/reduction treatments in synthetic solutions and reclaimed municipal wastewater. Chemosphere 2018, 201, 6–12. [Google Scholar] [CrossRef]
- Qi, H.; Sun, X.; Sun, Z. Porous graphite felt electrode with catalytic defects for enhanced degradation of pollutants by electro-Fenton process. Chem. Eng. J. 2021, 403, 126270. [Google Scholar] [CrossRef]
- Ouarda, Y.; Trellu, C.; Lesage, G.; Rivallin, M.; Drogui, P.; Cretin, M. Electro-oxidation of secondary effluents from various wastewater plants for the removal of acetaminophen and dissolved organic matter. Sci. Total Environ. 2020, 738, 140352. [Google Scholar] [CrossRef]
- He, Y.; Lin, H.; Guo, Z.; Zhang, W.; Li, H.; Huang, W. Recent developments and advances in boron-doped diamond electrodes for electrochemical oxidation of organic pollutants. Sep. Purif. Technol. 2019, 212, 802–821. [Google Scholar] [CrossRef]
- García-Gómez, C.; Drogui, P.; Zaviska, F.; Seyhi, B.; Gortáres-Moroyoqui, P.; Buelna, G.; Neira-Sáenz, C.; Estrada-Alvarado, M.; Ulloa-Mercado, R. Experimental design methodology applied to electrochemical oxidation of carbamazepine using Ti/PbO2 and Ti/BDD electrodes. J. Electroanal. Chem. 2014, 732, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Kurt, U.; Apaydin, O.; Gönüllü, M.T. Reduction of COD in wastewater from an organized tannery industrial region by Electro-Fenton process. J. Hazard. Mater. 2007, 143, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Chai, Y.; Qin, P.; Wu, Z.; Bai, M.; Li, W.; Pan, J.; Cao, R.; Chen, A.; Jin, D.; Peng, C. A coupled system of flow-through electro-Fenton and electrosorption processes for the efficient treatment of high-salinity organic wastewater. Sep. Purif. Technol. 2021, 267, 118683. [Google Scholar] [CrossRef]
- Ismail, S.A.; Ang, W.L.; Mohammad, A.W. Electro-Fenton technology for wastewater treatment: A bibliometric analysis of current research trends, future perspectives and energy consumption analysis. J. Water Process Eng. 2021, 40, 101952. [Google Scholar] [CrossRef]
- Zhang, Z.; Meng, H.; Wang, Y.; Shi, L.; Wang, X.; Chai, S. Fabrication of graphene@graphite-based gas diffusion electrode for improving H2O2 generation in Electro-Fenton process. Electrochim. Acta 2018, 260, 112–120. [Google Scholar] [CrossRef]
- Yang, W.; Zhou, M.; Oturan, N.; Li, Y.; Oturan, M.A. Electrocatalytic destruction of pharmaceutical imatinib by electro-Fenton process with graphene-based cathode. Electrochim. Acta 2019, 305, 285–294. [Google Scholar] [CrossRef]
- Orimolade, B.O.; Zwane, B.N.; Koiki, B.A.; Rivallin, M.; Bechelany, M.; Mabuba, N.; Lesage, G.; Cretin, M.; Arotiba, O.A. Coupling cathodic electro-fenton with anodic photo-electrochemical oxidation: A feasibility study on the mineralization of paracetamol. J. Environ. Chem. Eng. 2020, 8, 104394. [Google Scholar] [CrossRef]
- Guo, W.; Zhao, Q.; Du, J.; Wang, H.; Li, X.; Ren, N. Enhanced removal of sulfadiazine by sulfidated ZVI activated persulfate process: Performance, mechanisms and degradation pathways. Chem. Eng. J. 2020, 388, 124303. [Google Scholar] [CrossRef]
- Li, W.; Zhang, Y.; Zhao, P.; Zhou, P.; Liu, Y.; Cheng, X.; Wang, J.; Yang, B.; Guo, H. Enhanced kinetic performance of peroxymonosulfate/ZVI system with the addition of copper ions: Reactivity, mechanism, and degradation pathways. J. Hazard. Mater. 2020, 393, 122399. [Google Scholar] [CrossRef]
- Fu, S.; Jia, H.; Meng, X.; Guo, Z.; Wang, J. Fe-C micro-electrolysis-electrocoagulation based on BFDA in the pre-treatment of landfill leachate: Enhanced mechanism and electrode decay monitoring. Sci. Total Environ. 2021, 781, 146797. [Google Scholar] [CrossRef]
- Chen, R.-F.; Wu, L.; Zhong, H.-T.; Liu, C.-X.; Qiao, W.; Wei, C.-H. Evaluation of electrocoagulation process for high-strength swine wastewater pretreatment. Sep. Purif. Technol. 2021, 272, 118900. [Google Scholar] [CrossRef]
- Sánchez, M.; Gonzalo, O.G.; Yáñez, S.; Ruiz, I.; Soto, M. Influence of nutrients and pH on the efficiency of vertical flow constructed wetlands treating winery wastewater. J. Water Process Eng. 2021, 42, 102103. [Google Scholar] [CrossRef]
- Hufschmid, A.; Becker-Van Slooten, K.; Strawczynski, A.; Vioget, P.; Parra, S.; Péringer, P.; Pulgarin, C. BOD5 measurements of water presenting inhibitory Cu2+.: Implications in using of BOD to evaluate biodegradability of industrial wastewaters. Chemosphere 2003, 50, 171–176. [Google Scholar] [CrossRef]
- Rachidi, L.; Kaichouh, G.; Khachani, M.; Zarrouk, A.; El Karbane, M.; Chakchak, H.; Warad, I.; EL Hourch, A.; El Kacemi, K.; Guessous, A. Optimization and modeling of the electro-Fenton process for treatment of sertraline hydrochloride: Mineralization efficiency, energy cost and biodegradability enhancement. Chem. Data Collect. 2021, 35, 100764. [Google Scholar] [CrossRef]
- García-Espinoza, J.D.; Robles, I.; Durán-Moreno, A.; Godínez, L.A. Study of simultaneous electro-Fenton and adsorption processes in a reactor containing porous carbon electrodes and particulate activated carbon. J. Electroanal. Chem. 2021, 895, 115476. [Google Scholar] [CrossRef]
- Radjenovic, J.; Bagastyo, A.; Rozendal, R.A.; Mu, Y.; Keller, J.; Rabaey, K. Electrochemical oxidation of trace organic contaminants in reverse osmosis concentrate using RuO2/IrO2-coated titanium anodes. Water Res. 2011, 45, 1579–1586. [Google Scholar] [CrossRef] [Green Version]
- Santos, M.J.R.; Medeiros, M.C.; Oliveira, T.M.B.F.; Morais, C.C.O.; Mazzetto, S.E.; Martínez-Huitle, C.A.; Castro, S.S.L. Electrooxidation of cardanol on mixed metal oxide (RuO2-TiO2 and IrO2-RuO2-TiO2) coated titanium anodes: Insights into recalcitrant phenolic compounds. Electrochim. Acta 2016, 212, 95–101. [Google Scholar] [CrossRef]
- Estrada, A.L.; Li, Y.-Y.; Wang, A. Biodegradability enhancement of wastewater containing cefalexin by means of the electro-Fenton oxidation process. J. Hazard. Mater. 2012, 227–228, 41–48. [Google Scholar] [CrossRef]
- Deng, F.; Olvera-Vargas, H.; Garcia-Rodriguez, O.; Qiu, S.; Ma, F.; Chen, Z.; Lefebvre, O. Unconventional electro-Fenton process operating at a wide pH range with Ni foam cathode and tripolyphosphate electrolyte. J. Hazard. Mater. 2020, 396, 122641. [Google Scholar] [CrossRef]
- Brillas, E.; Sirés, I.; Oturan, M.A. Electro-Fenton process and related electrochemical technologies based on Fenton’s reaction chemistry. Chem. Rev. 2009, 109, 6570–6631. [Google Scholar] [CrossRef]
- Zheng, Y.; Qiu, S.; Deng, F.; Zhu, Y.; Li, G.; Ma, F. Three-dimensional electro-Fenton system with iron foam as particle electrode for folic acid wastewater pretreatment. Sep. Purif. Technol. 2019, 224, 463–474. [Google Scholar] [CrossRef]
- Huo, S.; Necas, D.; Zhu, F.; Chen, D.; An, J.; Zhou, N.; Liu, W.; Wang, L.; Cheng, Y.; Liu, Y.; et al. Anaerobic digestion wastewater decolorization by H2O2-enhanced electro-Fenton coagulation following nutrients recovery via acid tolerant and protein-rich Chlorella production. Chem. Eng. J. 2021, 406, 127160. [Google Scholar] [CrossRef]
- Dindaş, G.B.; Çalışkan, Y.; Çelebi, E.E.; Tekbaş, M.; Bektaş, N.; Yatmaz, H.C. Treatment of pharmaceutical wastewater by combination of electrocoagulation, electro-fenton and photocatalytic oxidation processes. J. Environ. Chem. Eng. 2020, 8, 103777. [Google Scholar] [CrossRef]
- Senthilnathan, J.; Younis, S.A.; Kwon, E.E.; Surenjana, A.; Kimb, K.-H.; Yoshimurae, M. An efficient system for electro-Fenton oxidation of pesticide by a reduced graphene oxide-aminopyrazine@3DNi foam gas diffusion electrode. J. Hazard. Mater. 2020, 400, 123323. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.; Zhou, M.; Zhang, Q.; Xu, X.; Li, Y.; Su, P. A novel stacked flow-through electro-Fenton reactor as decentralized system for the simultaneous removal of pollutants (COD, NH3-N and TP) and disinfection from domestic sewage containing chloride ions. Chem. Eng. J. 2020, 387, 124037. [Google Scholar] [CrossRef]
- McQuillan, R.V.; Stevens, G.W.; Mumford, K.A. Assessment of the electro-Fenton pathway for the removal of naphthalene from contaminated waters in remote regions. Sci. Total Environ. 2021, 762, 143155. [Google Scholar] [CrossRef]
- Midassi, S.; Bedoui, A.; Bensalah, N. Efficient degradation of chloroquine drug by electro-Fenton oxidation: Effects of operating conditions and degradation mechanism. Chemosphere 2020, 260, 127558. [Google Scholar] [CrossRef]
- Lacasa, E.; Cañizares, P.; Llanos, J.; Rodrigo, M.A. Removal of nitrates by electrolysis in non-chloride media: Effect of the anode material. Sep. Purif. Technol. 2011, 80, 592–599. [Google Scholar] [CrossRef]
- Wang, D.; Hu, J.; Liu, B.; Hou, H.; Yang, J.; Li, Y.; Zhu, Y.; Liang, S.; Xiao, K. Degradation of refractory organics in dual-cathode electro-Fenton using air-cathode for H2O2 electrogeneration and microbial fuel cell cathode for Fe2+ regeneration. J. Hazard. Mater. 2021, 412, 125269. [Google Scholar] [CrossRef]
- Zhang, J.; Qiu, S.; Feng, H.; Hu, T.; Wu, Y.; Luo, T.; Tang, W.; Wang, D. Efficient degradation of tetracycline using core–shell Fe@Fe2O3-CeO2 composite as novel heterogeneous electro-Fenton catalyst. Chem. Eng. J. 2022, 428, 131403. [Google Scholar] [CrossRef]
- Barhoumi, N.; Olvera-Vargas, H.; Oturan, N.; Huguenot, D.; Gadri, A.; Ammar, S.; Brillas, E.; Oturan, M.A. Kinetics of oxidative degradation/mineralization pathways of the antibiotic tetracycline by the novel heterogeneous electro-Fenton process with solid catalyst chalcopyrite. Appl. Catal. B Environ. 2017, 209, 637–647. [Google Scholar] [CrossRef]
- Zhang, Y.; Zuo, S.; Zhou, M.; Liang, L.; Ren, G. Removal of tetracycline by coupling of flow-through electro-Fenton and in-situ regenerative active carbon felt adsorption. Chem. Eng. J. 2018, 335, 685–692. [Google Scholar] [CrossRef]
- Luo, T.; Feng, H.; Tang, L.; Lu, Y.; Tang, W.; Chen, S.; Yu, J.; Xie, Q.; Ouyang, X.; Chen, Z. Efficient degradation of tetracycline by heterogeneous electro-Fenton process using Cu-doped Fe@Fe2O3: Mechanism and degradation pathway. Chem. Eng. J. 2020, 382, 122970. [Google Scholar] [CrossRef]
- Zwane, B.N.; Orimolade, B.O.; Koiki, B.A.; Mabuba, N.; Gomri, C.; Petit, E.; Bonniol, V.; Lesage, G.; Rivallin, M.; Cretin, M.; et al. Combined Electro-Fenton and Anodic Oxidation Processes at a Sub-Stoichiometric Titanium Oxide (Ti4O7) Ceramic Electrode for the Degradation of Tetracycline in Water. Water 2021, 13, 2772. [Google Scholar] [CrossRef]
- Olvera-Vargas, H.; Gore-Datar, N.; Garcia-Rodriguez, O.; Mutnuri, S.; Lefebvre, O. Electro-Fenton treatment of real pharmaceutical wastewater paired with a BDD anode: Reaction mechanisms and respective contribution of homogeneous and heterogeneous OH. Chem. Eng. J. 2021, 404, 126524. [Google Scholar] [CrossRef]
- Feng, Y.; Yang, S.; Xia, L.; Wang, Z.; Suo, N.; Chen, H.; Long, Y.; Zhou, B.; Yu, Y. In-situ ion exchange electrocatalysis biological coupling (i-IEEBC) for simultaneously enhanced degradation of organic pollutants and heavy metals in electroplating wastewater. J. Hazard. Mater. 2019, 364, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Salles, N.A.; Fourcade, F.; Geneste, F.; Floner, D.; Amrane, A. Relevance of an electrochemical process prior to a biological treatment for the removal of an organophosphorous pesticide, phosmet. J. Hazard. Mater. 2010, 181, 617–623. [Google Scholar] [CrossRef]
- Yao, B.; Luo, Z.; Yang, J.; Zhi, D.; Zhou, Y. FeIIFeIII layered double hydroxide modified carbon felt cathode for removal of ciprofloxacin in electro-Fenton process. Environ. Res. 2021, 197, 111144. [Google Scholar] [CrossRef]
- Johnson, M.B.; Mehrvar, M. Treatment of Actual Winery Wastewater by Fenton-like Process: Optimization to Improve Organic Removal, Reduce Inorganic Sludge Production and Enhance Co-Treatment at Municipal Wastewater Treatment Facilities. Water 2021, 14, 39. [Google Scholar] [CrossRef]
- Dung, N.T.; Duong, L.T.; Hoa, N.T.; Thao, V.D.; Ngan, L.V.; Huy, N.N. A comprehensive study on the heterogeneous electro-Fenton degradation of tartrazine in water using CoFe2O4/carbon felt cathode. Chemosphere 2022, 287, 132141. [Google Scholar] [CrossRef]
- Ma, B.; Lv, W.; Li, J.; Yang, C.; Tang, Q.; Wang, D. Promotion removal of aniline with electro-Fenton processes utilizing carbon nanotube 3D morphology modification of an Ag-loaded copper foam cathode. J. Water Process Eng. 2021, 43, 102295. [Google Scholar] [CrossRef]
- Thor, S.-H.; Ho, L.-N.; Ong, S.-A.; Abidin, C.Z.A.; Heah, C.-Y.; Nordin, N.; Ong, Y.-P.; Yap, K.-L. Advanced oxidation treatment of amaranth dye synchronized with electricity generation using carbon-based cathodes in a sustainable photocatalytic fuel cell integrated electro-fenton system. J. Environ. Chem. Eng. 2021, 9, 106439. [Google Scholar] [CrossRef]
- Dong, P.; Liu, W.; Wang, S.; Wang, H.; Wang, Y.; Zhao, C. In suit synthesis of Fe3O4 on carbon fiber paper@polyaniline substrate as novel self-supported electrode for heterogeneous electro-Fenton oxidation. Electrochim. Acta 2019, 308, 54–63. [Google Scholar] [CrossRef]
- Deng, J.; Lu, J.; Yan, Q.; Pan, J. Basic research on chemical mechanical polishing of single-crystal SiC—Electro–Fenton: Reaction mechanism and modelling of hydroxyl radical generation using condition response modelling. J. Environ. Chem. Eng. 2021, 9, 104954. [Google Scholar] [CrossRef]
- Sun, M.; Zhang, Z.; Liu, G.; Lv, M.; Feng, Y. Enhancing methane production of synthetic brewery water with granular activated carbon modified with nanoscale zero-valent iron (NZVI) in anaerobic system. Sci. Total Environ. 2021, 760, 143933. [Google Scholar] [CrossRef]
- Wang, J.; Chen, R.; Fan, L.; Cui, L.; Zhang, Y.; Cheng, J.; Wu, X.; Zeng, W.; Tian, Q.; Shen, L. Construction of fungi-microalgae symbiotic system and adsorption study of heavy metal ions. Sep. Purif. Technol. 2021, 268, 118689. [Google Scholar] [CrossRef]
- Zhang, A.; Wang, S.; Yang, M.; Li, H.; Wang, H.; Fang, F.; Guo, J. Influence of NaCl salinity on the aggregation performance of anammox granules. J. Water Process Eng. 2020, 39, 101687. [Google Scholar] [CrossRef]
- Sun, D.; Hong, X.; Cui, Z.; Du, Y.; Hui, K.S.; Zhu, E.; Wu, K.; Hui, K.N. Treatment of landfill leachate using magnetically attracted zero-valent iron powder electrode in an electric field. J. Hazard. Mater. 2020, 388, 121768. [Google Scholar] [CrossRef]
- Chu, Y.; Miao, B.; Zhang, X.; Lv, R. Heterogeneous electro-Fenton-like oxidation for the degradation of 4-nitrophenol characterized by immobilized Fe(III): Performance, mechanism and chlorinated organic compounds formation. J. Water Process Eng. 2020, 38, 101662. [Google Scholar] [CrossRef]
- Kaspar, P.; Sobola, D.; Dallaev, R.; Ramazanov, S.; Nebojsa, A.; Rezaee, S.; Grmela, L. Characterization of Fe2O3 thin film on highly oriented pyrolytic graphite by AFM, Ellipsometry and XPS. Appl. Surf. Sci. 2019, 493, 673–678. [Google Scholar] [CrossRef]
- Liang, J.; Xiang, Q.; Lei, W.; Zhang, Y.; Sun, J.; Zhu, H.; Wang, S. Ferric iron reduction reaction electro-Fenton with gas diffusion device: A novel strategy for improvement of comprehensive efficiency in electro-Fenton. J. Hazard. Mater. 2021, 412, 125195. [Google Scholar] [CrossRef]
- Ye, Z.; Padilla, J.A.; Xuriguera, E.; Brillas, E.; Sirés, I. Magnetic MIL(Fe)-type MOF-derived N-doped nano-ZVI@C rods as heterogeneous catalyst for the electro-Fenton degradation of gemfibrozil in a complex aqueous matrix. Appl. Catal. B Environ. 2020, 266, 118604. [Google Scholar] [CrossRef]
- Wang, R.; Shu, J.; Chen, M.; Wang, R.; He, D.; Wang, J.; Tang, C.; Han, Y.; Luo, Z. An innovative method for fractionally removing high concentrations of Ni2+, PO43−, TP, COD, and NH4+-N from printed-circuit-board nickel plating wastewater. Sep. Purif. Technol. 2021, 260, 118241. [Google Scholar] [CrossRef]
- Kumar, N.; Sreenivasa, S.; Prashant, A.; Kumar, V.; Holla, B.S.; Chandramohan, V.; Vishwantha, P.; Yadav, A.K. N′-((3-(substituted phenyl)-1-phenyl-1H-Pyrazol-4-yl)methylene)-(substituted) benzhydrazide: Synthesis, characterization and pharmacological evaluation. Chem. Data Collect. 2021, 32, 100665. [Google Scholar] [CrossRef]
- Mojsiewicz-Pieńkowska, K. Size exclusion chromatography with evaporative light scattering detection as a method for speciation analysis of polydimethylsiloxanes. III. Identification and determination of dimeticone and simeticone in pharmaceutical formulations. J. Pharm. Biomed. Anal. 2012, 58, 200–207. [Google Scholar] [CrossRef]
- Yang, X.; Sun, Z.; Wang, W.; Zhou, Q.; Shi, G.; Wei, F.; Jiang, G. Developmental toxicity of synthetic phenolic antioxidants to the early life stage of zebrafish. Sci. Total Environ. 2018, 643, 559–568. [Google Scholar] [CrossRef]
- Wang, W.; Xiong, P.; Zhang, H.; Zhu, Q.; Liao, C.; Jiang, G. Analysis, occurrence, toxicity and environmental health risks of synthetic phenolic antioxidants: A review. Environ. Res. 2021, 201, 111531. [Google Scholar] [CrossRef]
- Zhang, X.; Lu, X.; Li, H. Isolation and identification of a novel allelochemical from Ruppia maritima extract against the cyanobacteria Microcystis aeruginosa. Environ. Technol. Innov. 2021, 21, 101301. [Google Scholar] [CrossRef]
- Seibert, D.; Borba, F.H.; Bueno, F.; Inticher, J.J.; Módenes, A.N.; Espinoza-Quiñones, F.R.; Bergamasco, R. Two-stage integrated system photo-electro-Fenton and biological oxidation process assessment of sanitary landfill leachate treatment: An intermediate products study. Chem. Eng. J. 2019, 372, 471–482. [Google Scholar] [CrossRef]


| Parameters | Values | Average |
|---|---|---|
| COD | 5388–6292 mg/L | 5840 mg/L |
| TOC | 4154–4720 mg/L | 4437 mg/L |
| BOD5 | 1190–2300 mg/L | 1945 mg/L |
| BOD5/COD | 0.21–0.35 mg/L | 0.28 mg/L |
| Cl− | 0.10–0.18 mg/L | 0.14 mg/L |
| TP | 2.26–2.72 mg/L | 2.49 mg/L |
| Phosphates | 1.98–2.51 mg/L | 2.25 mg/L |
| TN | 396–422 mg/L | 409 mg/L |
| NO3− | 9.2–11.0 mg/L | 10.1 mg/L |
| NO2− | <0.016 mg/L | <0.016 mg/L |
| NH4+ | 66.38–75.28 mg/L | 70.83 mg/L |
| pH | 4.69–4.86 | 4.77 |
| Fe | <0.1 μg/L | <0.1 μg/L |
| Cu | <0.1 μg/L | <0.1 μg/L |
| Conductivity | 2.25–2.28 mS/cm | 2.26 mS/cm |
| Molecular Structures | Substance Names | Retention Time (Min) |
|---|---|---|
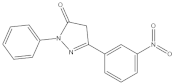 | 5-(3-nitrophenyl)-2-phenyl-2,4-dihydro-3H-pyrazol-3-one | 6.021 |
 | Decamethylcyclopentasiloxane | 7.950 |
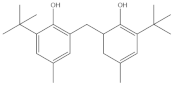 | 6,6′-methylenebis(2-(tert-butyl)-4-methylphenol) | 13.545 |
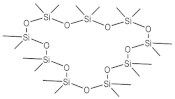 | 2,2,4,4,6,6,8,8,10,10,12,12,14,14,16,16,18,18-octadecamethyl-1,3,5,7,9,11,13,15,17-nonaoxa-2,4,6,8,10,12,14,16,18-nonasilacyclooctadecane | 14.095 |
 | 10,13-dimethyl-7,8,9,10,11,12,13,14,15,16-decahydrospiro[cyclopenta[a]phenanthrene-17,2′-oxetane]-3,3′(6H)-dione | 15.995 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deng, M.; Wu, K.; Yang, T.; Yu, D.; Liu, G.; Gong, S.; Sun, D.; Petru, M. Construction of Novel Electro-Fenton Systems by Magnetically Decorating Zero-Valent Iron onto RuO2-IrO2/Ti Electrode for Highly Efficient Pharmaceutical Wastewater Treatment. Water 2022, 14, 1044. https://doi.org/10.3390/w14071044
Deng M, Wu K, Yang T, Yu D, Liu G, Gong S, Sun D, Petru M. Construction of Novel Electro-Fenton Systems by Magnetically Decorating Zero-Valent Iron onto RuO2-IrO2/Ti Electrode for Highly Efficient Pharmaceutical Wastewater Treatment. Water. 2022; 14(7):1044. https://doi.org/10.3390/w14071044
Chicago/Turabian StyleDeng, Miao, Keming Wu, Tao Yang, Deyou Yu, Gaojie Liu, Shuai Gong, Dongni Sun, and Michal Petru. 2022. "Construction of Novel Electro-Fenton Systems by Magnetically Decorating Zero-Valent Iron onto RuO2-IrO2/Ti Electrode for Highly Efficient Pharmaceutical Wastewater Treatment" Water 14, no. 7: 1044. https://doi.org/10.3390/w14071044
APA StyleDeng, M., Wu, K., Yang, T., Yu, D., Liu, G., Gong, S., Sun, D., & Petru, M. (2022). Construction of Novel Electro-Fenton Systems by Magnetically Decorating Zero-Valent Iron onto RuO2-IrO2/Ti Electrode for Highly Efficient Pharmaceutical Wastewater Treatment. Water, 14(7), 1044. https://doi.org/10.3390/w14071044









