Electrochemical Recovery to Overcome Direct Osmosis Concentrate-Bearing Lead: Optimization of Treatment Process via RSM-CCD
Abstract
:1. Introduction
2. Materials and Methods
2.1. Direct Osmosis Concentrate Characteristics
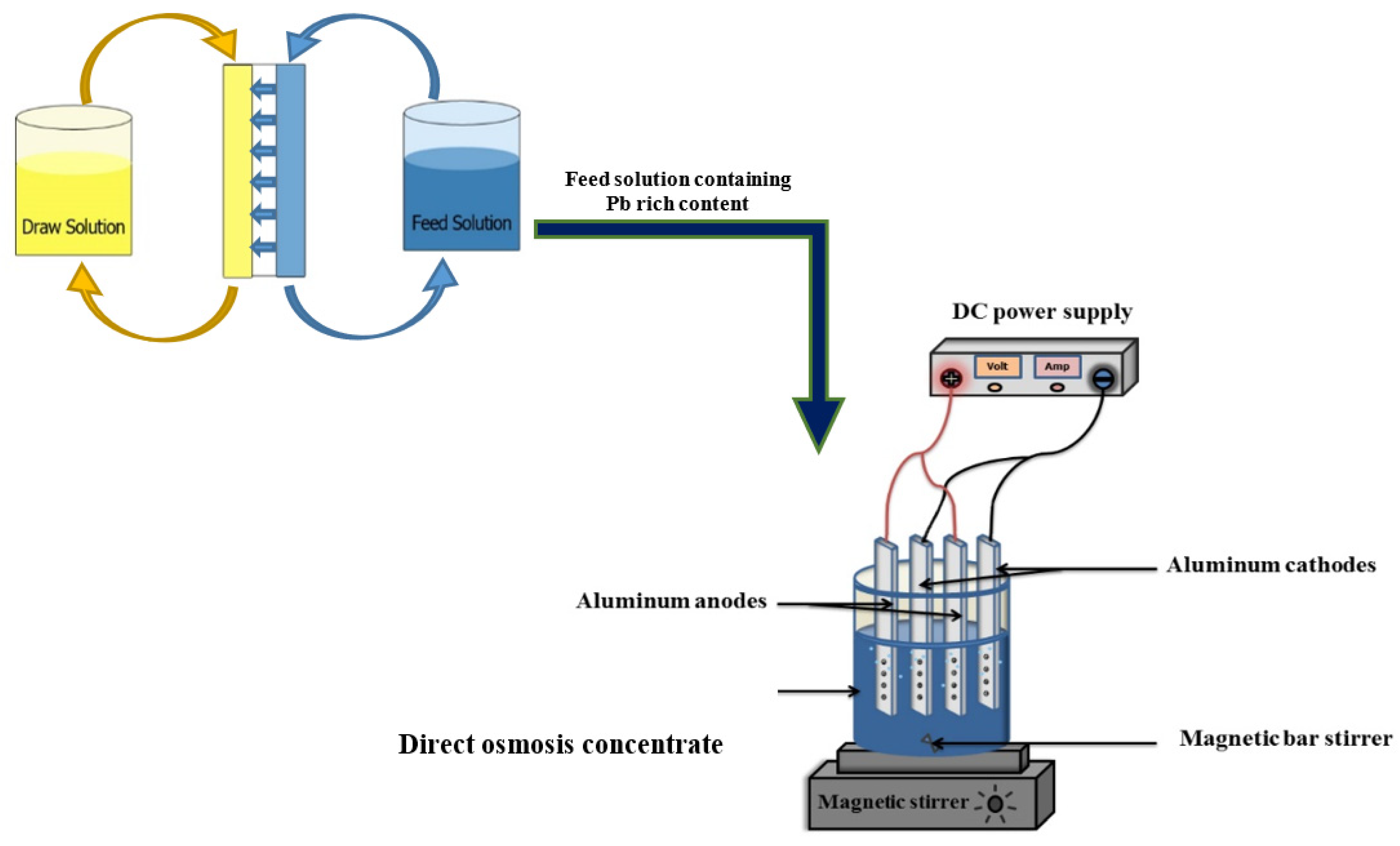
2.2. Perforated Aluminium–Electrocoagulation–Flotation (PA-ECF) Set Up
2.3. Analytical Methods
2.4. Experimental Statistical Design
3. Results and Discussion
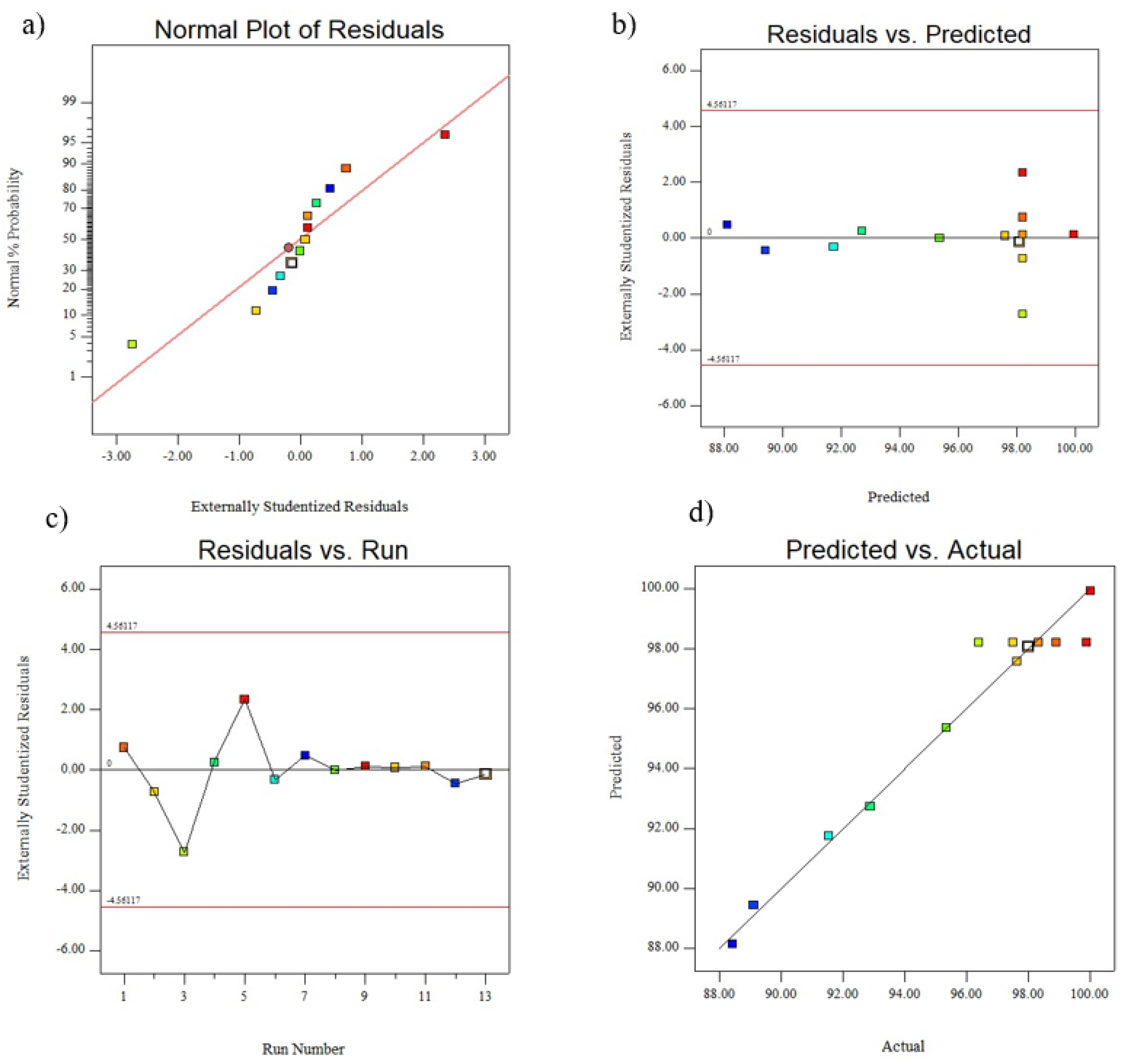
3.1. Modeling and Statistical Analysis via Central Composite Design (CCD)
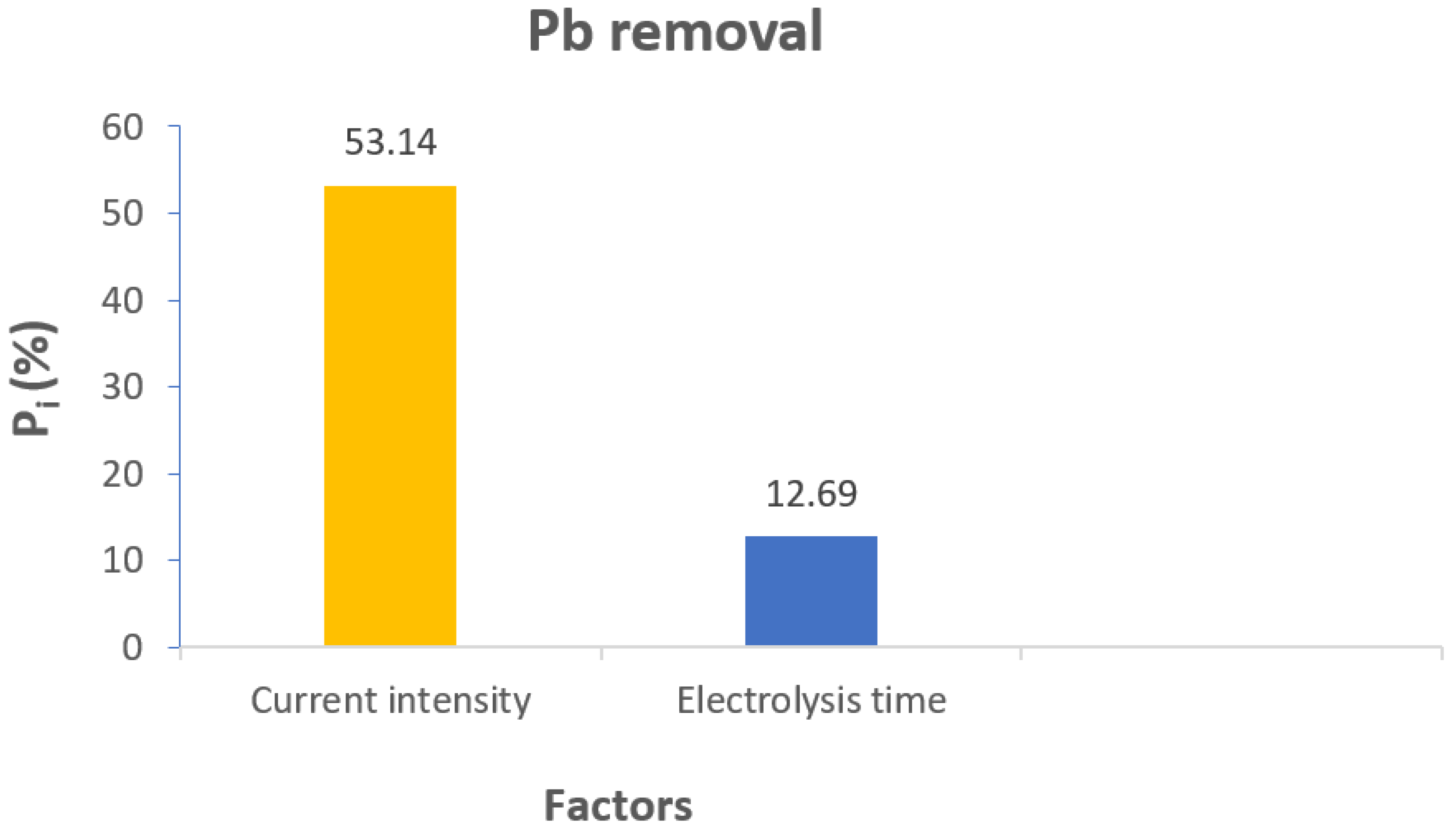
3.2. Importance of Influencing Parameters via Pareto Diagram Analysis
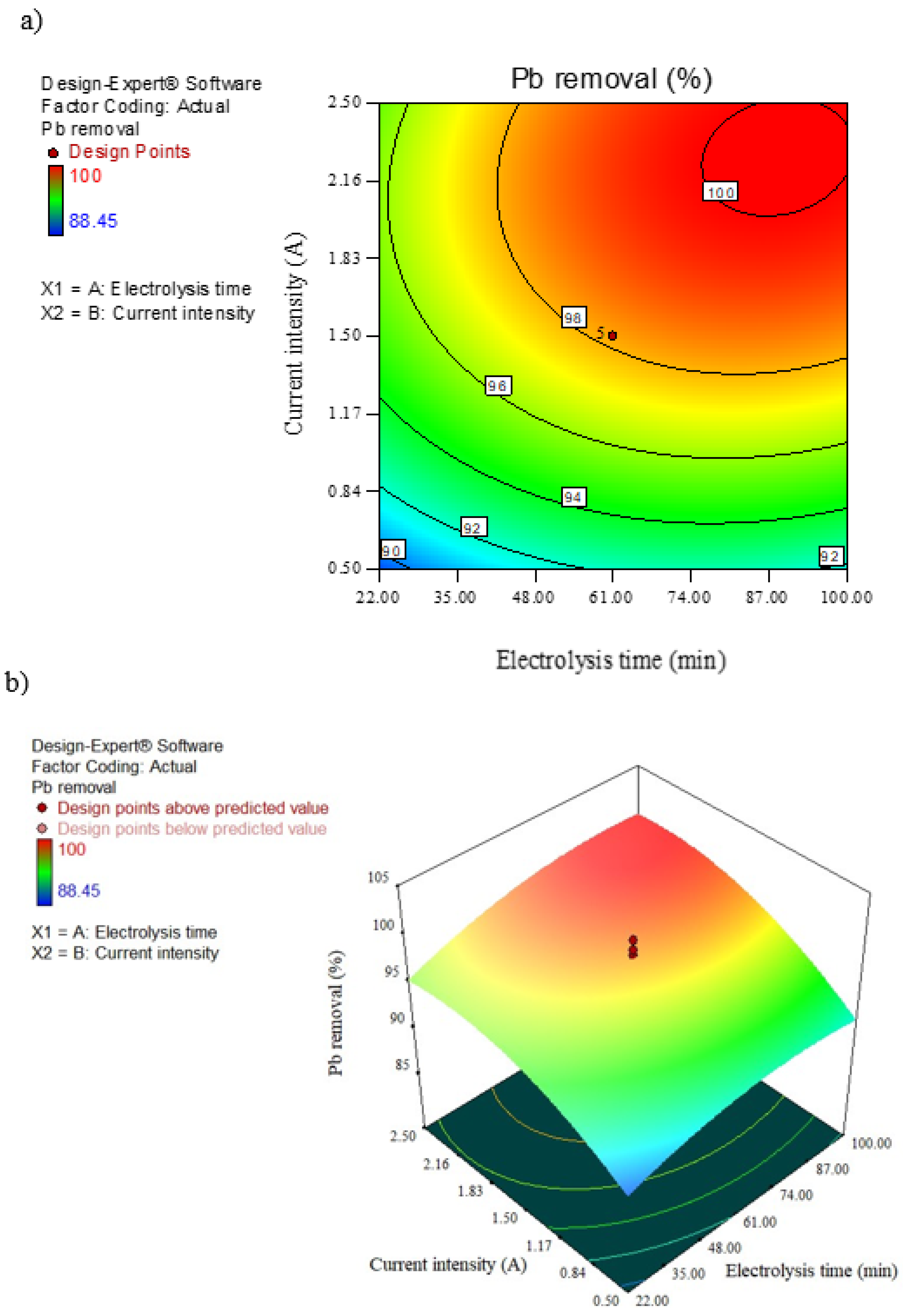
3.3. Interaction of Time and Current on Pb Removal
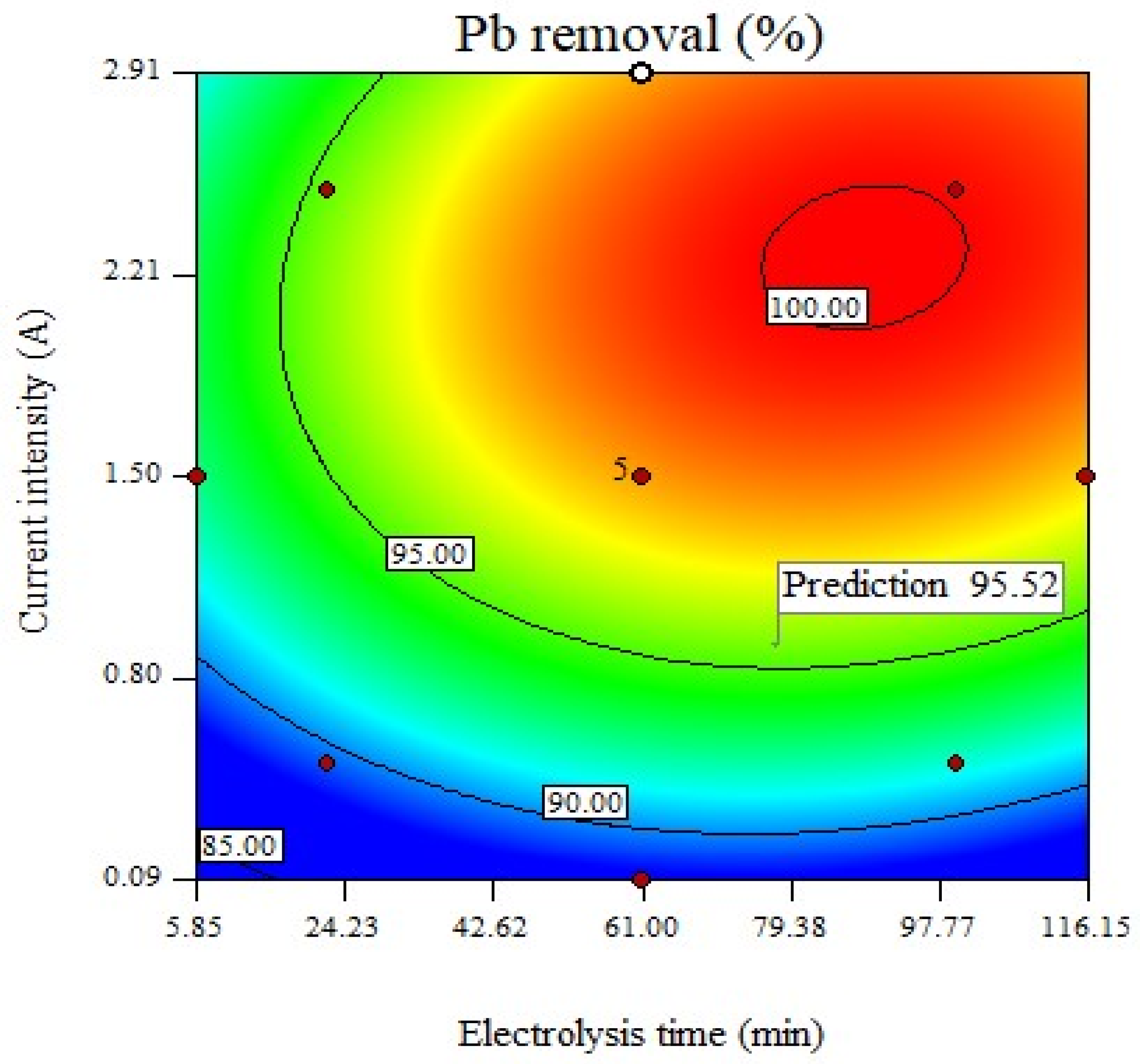
3.4. Optimization Process
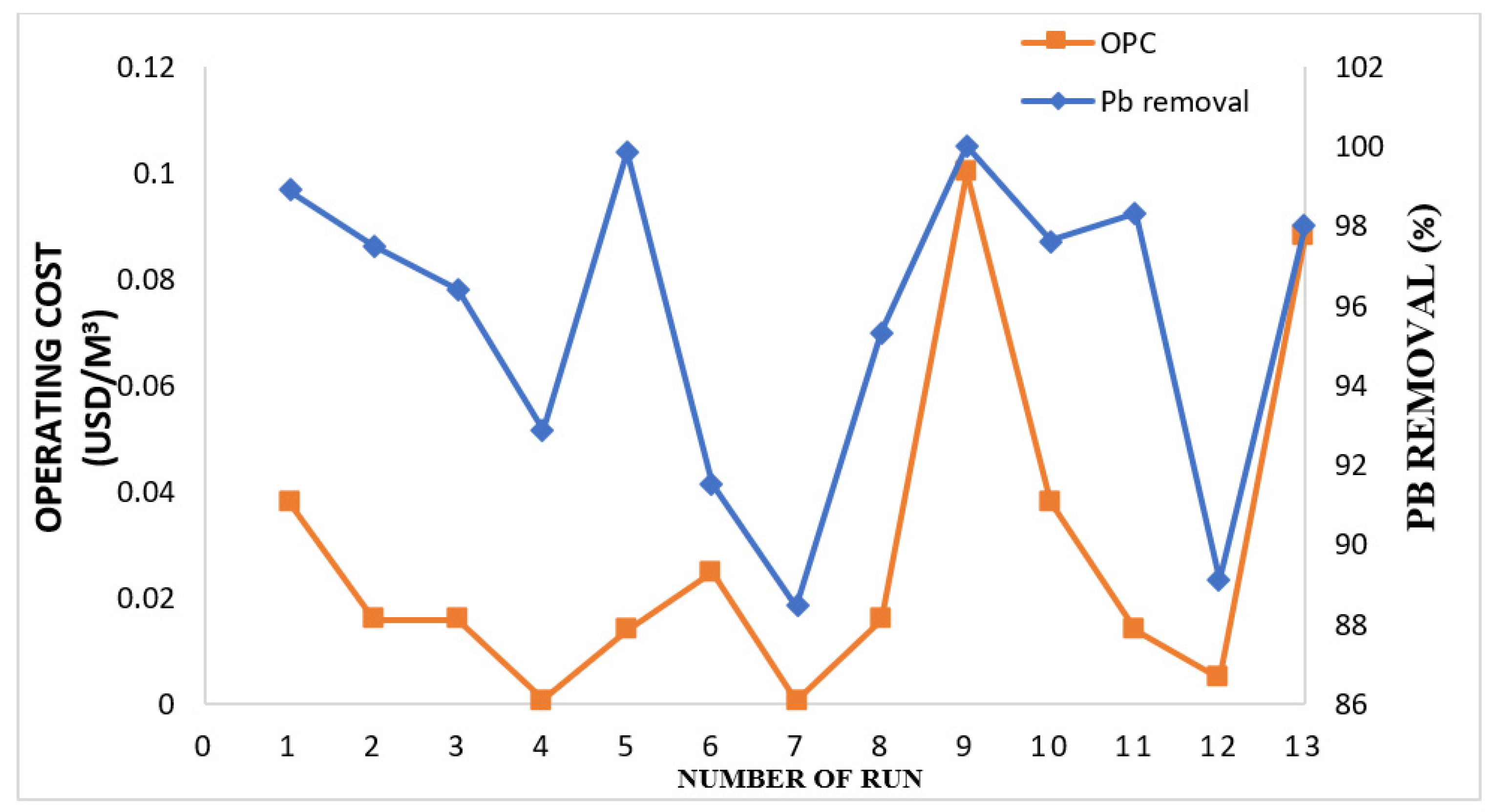
3.5. Cost-Effectiveness Estimation
3.6. Treatment of Direct Osmosis Concentrate from Metallurgical Industry
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fallah, S.; Mamaghani, H.R.; Yegani, R.; Hajinajaf, N.; Pourabbas, B. Use of graphene substrates for wastewater treatment of textile industries. Adv. Compos. Hybrid Mater. 2020, 3, 187–193. [Google Scholar] [CrossRef]
- Kinuthia, G.K.; Ngure, V.; Beti, D.; Lugalia, R.; Wangila, A.; Kamau, L. Levels of heavy metals in wastewater and soil samples from open drainage channels in Nairobi, Kenya: Community health implication. Sci. Rep. 2020, 10, 8434. [Google Scholar] [CrossRef]
- Bouguerra, W.; Barhoumi, A.; Ibrahim, N.; Brahmi, K.; Aloui, L.; Hamrouni, B. Optimization of the electrocoagulation process for the removal of lead from water using aluminium as electrode material. Desalination Water Treat. 2015, 56, 2672–2681. [Google Scholar] [CrossRef]
- Escobar, C.; Soto-Salazar, C.; Toral, M.I. Optimization of the electrocoagulation process for the removal of copper, lead and cadmium in natural waters and simulated wastewater. J. Environ. Manag. 2006, 81, 384–391. [Google Scholar] [CrossRef]
- Ahmad, N.; Sereshti, H.; Mousazadeh, M.; Nodeh, H.R.; Kamboh, M.A.; Mohamad, S. New magnetic silica-based hybrid organic-inorganic nanocomposite for the removal of lead (II) and nickel (II) ions from aqueous solutions. Mater. Chem. Phys. 2019, 226, 73–81. [Google Scholar] [CrossRef]
- Pohl, A. Removal of heavy metal ions from water and wastewaters by sulfur-containing precipitation agents. Water Air Soil Pollut. 2020, 231, 503. [Google Scholar] [CrossRef]
- EU, European Community. Council Directive 98/83/EC of 3 November 1998 on the quality of water intended for human consumption. Off. J. Eur. Communities 1998, 32–54. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A31998L0083 (accessed on 18 October 2021).
- Assadi, A.; Fazli, M.M.; Emamjomeh, M.M.; Ghasemi, M. Optimization of lead removal by electrocoagulation from aqueous solution using response surface methodology. Desalination Water Treat. 2016, 57, 9375–9382. [Google Scholar] [CrossRef]
- Mansoorian, H.J.; Mahvi, A.H.; Jafari, A.J. Removal of lead and zinc from battery industry wastewater using electrocoagulation process: Influence of direct and alternating current by using iron and stainless steel rod electrodes. Sep. Purif. Technol. 2014, 135, 165–175. [Google Scholar] [CrossRef]
- Lai, Y.-C.; Chang, Y.-R.; Chen, M.-L.; Lo, Y.-K.; Lai, J.-Y.; Lee, D.-J. Poly (vinyl alcohol) and alginate cross-linked matrix with immobilized Prussian blue and ion exchange resin for cesium removal from waters. Bioresour. Technol. 2016, 214, 192–198. [Google Scholar] [CrossRef]
- Verma, V.; Tewari, S.; Rai, J. Ion exchange during heavy metal bio-sorption from aqueous solution by dried biomass of macrophytes. Bioresour. Technol. 2008, 99, 1932–1938. [Google Scholar] [CrossRef] [PubMed]
- Cochrane, E.; Lu, S.; Gibb, S.; Villaescusa, I. A comparison of low-cost biosorbents and commercial sorbents for the removal of copper from aqueous media. J. Hazard. Mater. 2006, 137, 198–206. [Google Scholar] [CrossRef]
- Davarnejad, R.; Panahi, P. Cu (II) removal from aqueous wastewaters by adsorption on the modified Henna with Fe3O4 nanoparticles using response surface methodology. Sep. Purif. Technol. 2016, 158, 286–292. [Google Scholar] [CrossRef]
- Javid, A.B.; Barzanouni, H.; Younesi, A.; Amir, N.; Farahani, A.; Mousazadeh, M.; Soleimani, P. Performance of modified one-stage Phoredox reactor with hydraulic up-flow in biological removal of phosphorus from municipal wastewater. Desalination Water Treat. 2019, 171, 216–222. [Google Scholar]
- Sierra-Alvarez, R.; Karri, S.; Freeman, S.; Field, J.A. Biological treatment of heavy metals in acid mine drainage using sulfate reducing bioreactors. Water Sci. Technol. 2006, 54, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Akbal, F.; Camcı, S. Copper, chromium and nickel removal from metal plating wastewater by electrocoagulation. Desalination 2011, 269, 214–222. [Google Scholar] [CrossRef]
- Dharnaik, A.S.; Ghosh, P.K. Hexavalent chromium [Cr (VI)] removal by the electrochemical ion-exchange process. Environ. Technol. 2014, 35, 2272–2279. [Google Scholar] [CrossRef]
- Mohsen-Nia, M.; Montazeri, P.; Modarress, H. Removal of Cu2+ and Ni2+ from wastewater with a chelating agent and reverse osmosis processes. Desalination 2007, 217, 276–281. [Google Scholar] [CrossRef]
- Naghdali, Z.; Sahebi, S.; Ghanbari, R.; Mousazadeh, M.; Ali Jamali, H. Chromium removal and water recycling from electroplating wastewater through direct osmosis: Modeling and optimization by response surface methodology. Environ. Health Eng. Manag. J. 2019, 6, 113–120. [Google Scholar] [CrossRef]
- Li, Z.; Linares, R.V.; Bucs, S.; Fortunato, L.; Hélix-Nielsen, C.; Vrouwenvelder, J.S.; Ghaffour, N.; Leiknes, T.; Amy, G. Aquaporin based biomimetic membrane in forward osmosis: Chemical cleaning resistance and practical operation. Desalination 2017, 420, 208–215. [Google Scholar] [CrossRef]
- Naghdali, Z.; Sahebi, S.; Mousazadeh, M.; Jamali, H.A. Optimization of the forward osmosis process using aquaporin membranes in chromium removal. Chem. Eng. Technol. 2020, 43, 298–306. [Google Scholar] [CrossRef]
- Cui, Y.; Ge, Q.; Liu, X.-Y.; Chung, T.-S. Novel forward osmosis process to effectively remove heavy metal ions. J. Membr. Sci. 2014, 467, 188–194. [Google Scholar] [CrossRef]
- Mi, B.; Elimelech, M. Gypsum scaling and cleaning in forward osmosis: Measurements and mechanisms. Environ. Sci. Technol. 2010, 44, 2022–2028. [Google Scholar] [CrossRef]
- Labiadh, L.; Fernandes, A.; Ciríaco, L.; Pacheco, M.J.; Gadri, A.; Ammar, S.; Lopes, A. Electrochemical treatment of concentrate from reverse osmosis of sanitary landfill leachate. J. Environ. Manag. 2016, 181, 515–521. [Google Scholar] [CrossRef]
- Renou, S.; Givaudan, J.; Poulain, S.; Dirassouyan, F.; Moulin, P. Landfill leachate treatment: Review and opportunity. J. Hazard. Mater. 2008, 150, 468–493. [Google Scholar] [CrossRef]
- Emamjomeh, M.M.; Jamali, H.A.; Naghdali, Z.; Mousazadeh, M. Efficiency of Electrocoagulation, Sedimentation and Filtration Hybrid Process in Removing Chemical Oxygen Demand and Turbidity from Carwash Industrial Wastewater: Optimization by Response Surface Methodology. J. Maz. Univ. Med. Sci. 2019, 29, 106–120. [Google Scholar]
- Kabdaşlı, I.; Arslan-Alaton, I.; Ölmez-Hancı, T.; Tünay, O. Electrocoagulation applications for industrial wastewaters: A critical review. Environ. Technol. Rev. 2012, 1, 2–45. [Google Scholar] [CrossRef]
- Mousazadeh, M.; Alizadeh, S.; Frontistis, Z.; Kabdaşlı, I.; Karamati Niaragh, E.; Al Qodah, Z.; Naghdali, Z.; Mahmoud, A.E.D.; Sandoval, M.A.; Butler, E. Electrocoagulation as a Promising Defluoridation Technology from Water: A Review of State of the Art of Removal Mechanisms and Performance Trends. Water 2021, 13, 656. [Google Scholar] [CrossRef]
- Titchou, F.E.; Zazou, H.; Afanga, H.; El Gaayda, J.; Akbour, R.A.; Nidheesh, P.V.; Hamdani, M. An overview on the elimination of organic contaminants from aqueous systems using electrochemical advanced oxidation processes. J. Water Process Eng. 2021, 41, 102040. [Google Scholar] [CrossRef]
- Afanga, H.; Zazou, H.; Titchou, F.E.; Rakhila, Y.; Akbour, R.A.; Elmchaouri, A.; Ghanbaja, J.; Hamdani, M. Integrated electrochemical processes for textile industry wastewater treatment: System performances and sludge settling characteristics. Sustain. Environ. Res. 2020, 30, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Emamjomeh, M.; Kakavand, S.; Jamali, H.; Alizadeh, S.; Safdari, M.; Mousavi, S.E.S.; Hashim, K.S.; Mousazadeh, M. The treatment of printing and packaging wastewater by electrocoagulation–flotation: The simultaneous efficacy of critical parameters and economics. Desalination Water Treat. 2020, 205, 161–174. [Google Scholar] [CrossRef]
- Emamjomeh, M.M.; Jamali, H.A.; Naghdali, Z.; Mousazadeh, M. Carwash wastewater treatment by the application of an environmentally friendly hybrid system: An experimental design approach. Desalination Water Treat. 2019, 160, 171–177. [Google Scholar] [CrossRef] [Green Version]
- Bazrafshan, E.; Mohammadi, L.; Ansari-Moghaddam, A.; Mahvi, A.H. Heavy metals removal from aqueous environments by electrocoagulation process—A systematic review. J. Environ. Health Sci. Eng. 2015, 13, 74. [Google Scholar] [CrossRef] [Green Version]
- Titchou, F.E.; Afanga, H.; Zazou, H.; Akbour, R.A.; Hamdani, M. Batch elimination of cationic dye from aqueous solution by electrocoagulation process. Mediterr. J. Chem. 2020, 10, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Abdulgader, M.; Yu, Q.J.; Zinatizadeh, A.A.; Williams, P.; Rahimi, Z. Application of response surface methodology (RSM) for process analysis and optimization of milk processing wastewater treatment using multistage flexible fiber biofilm reactor. J. Environ. Chem. Eng. 2020, 8, 103797. [Google Scholar] [CrossRef]
- Nair, A.T.; Makwana, A.R.; Ahammed, M.M. The use of response surface methodology for modelling and analysis of water and wastewater treatment processes: A review. Water Sci. Technol. 2014, 69, 464–478. [Google Scholar] [CrossRef]
- Trinh, T.K.; Kang, L.S. Response surface methodological approach to optimize the coagulation–flocculation process in drinking water treatment. Chem. Eng. Res. Des. 2011, 89, 1126–1135. [Google Scholar] [CrossRef]
- Pandey, N.; Thakur, C. Statistical Comparison of Response Surface Methodology–Based Central Composite Design and Hybrid Central Composite Design for Paper Mill Wastewater Treatment by Electrocoagulation. Process Integr. Optim. Sustain. 2020, 4, 343–359. [Google Scholar] [CrossRef]
- Emamjomeh, M.M.; Mousazadeh, M.; Mokhtari, N.; Jamali, H.A.; Makkiabadi, M.; Naghdali, Z.; Hashim, K.S.; Ghanbari, R. Simultaneous removal of phenol and linear alkylbenzene sulfonate from automotive service station wastewater: Optimization of coupled electrochemical and physical processes. Sep. Sci. Technol. 2020, 55, 3184–3194. [Google Scholar] [CrossRef]
- Khan, S.U.; Mahtab, M.S.; Farooqi, I.H. Enhanced lead (II) removal with low energy consumption in an electrocoagulation column employing concentric electrodes: Process optimisation by RSM using CCD. Int. J. Environ. Anal. Chem. 2021, 1–18. [Google Scholar] [CrossRef]
- Khanniri, E.; Yousefi, M.; Mortazavian, A.M.; Khorshidian, N.; Sohrabvandi, S.; Arab, M.; Koushki, M.R. Effective removal of lead (II) using chitosan and microbial adsorbents: Response surface methodology (RSM). Int. J. Biol. Macromol. 2021, 178, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, S.S.; Moghaddam, M.A.; Arami, M. Coagulation/flocculation process for dye removal using sludge from water treatment plant: Optimization through response surface methodology. J. Hazard. Mater. 2010, 175, 651–657. [Google Scholar] [CrossRef]
- Mosaddeghi, M.R.; Pajoum Shariati, F.; Vaziri Yazdi, S.A.; Nabi Bidhendi, G. Application of response surface methodology (RSM) for optimizing coagulation process of paper recycling wastewater using Ocimum basilicum. Environ. Technol. 2020, 41, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Niazmand, R.; Jahani, M.; Sabbagh, F.; Rezania, S. Optimization of Electrocoagulation Conditions for the Purification of Table Olive Debittering Wastewater Using Response Surface Methodology. Water 2020, 12, 1687. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater; APHA American Public Health Association: Washington, DC, USA, 2003; Volume 2. [Google Scholar]
- Khan, S.U.; Islam, D.T.; Farooqi, I.H.; Ayub, S.; Basheer, F. Hexavalent chromium removal in an electrocoagulation column reactor: Process optimization using CCD, adsorption kinetics and pH modulated sludge formation. Process Saf. Environ. Prot. 2019, 122, 118–130. [Google Scholar] [CrossRef]
- Mousazadeh, M.; Naghdali, Z.; Al-Qodah, Z.; Alizadeh, S.; Niaragh, E.K.; Malekmohammadi, S.; Nidheesh, P.; Roberts, E.P.; Sillanpää, M.; Emamjomeh, M.M. A systematic diagnosis of state of the art in the use of electrocoagulation as a sustainable technology for pollutant treatment: An updated review. Sustain. Energy Technol. Assess. 2021, 47, 101353. [Google Scholar]
- Gönder, Z.B.; Arayici, S.; Barlas, H. Treatment of pulp and paper mill wastewater using utrafiltration process: Optimization of the fouling and rejections. Ind. Eng. Chem. Res. 2012, 51, 6184–6195. [Google Scholar] [CrossRef]
- Emamjomeh, M.M.; Sivakumar, M. An empirical model for defluoridation by batch monopolar electrocoagulation/flotation (ECF) process. J. Hazard. Mater. 2006, 131, 118–125. [Google Scholar] [CrossRef]
- Ait Ouaissa, Y.; Chabani, M.; Amrane, A.; Bensmaili, A. Removal of Cr (VI) from model solutions by a combined electrocoagulation sorption process. Chem. Eng. Technol. 2013, 36, 147–155. [Google Scholar] [CrossRef]
- Barışçı, S.; Turkay, O. Optimization and modelling using the response surface methodology (RSM) for ciprofloxacin removal by electrocoagulation. Water Sci. Technol. 2016, 73, 1673–1679. [Google Scholar] [CrossRef]
- Yoosefian, M.; Ahmadzadeh, S.; Aghasi, M.; Dolatabadi, M. Optimization of electrocoagulation process for efficient removal of ciprofloxacin antibiotic using iron electrode; kinetic and isotherm studies of adsorption. J. Mol. Liq. 2017, 225, 544–553. [Google Scholar] [CrossRef]
- Genawi, N.M.; Ibrahim, M.H.; El-Naas, M.H.; Alshaik, A.E. Chromium Removal from Tannery Wastewater by Electrocoagulation: Optimization and Sludge Characterization. Water 2020, 12, 1374. [Google Scholar] [CrossRef]
- Ano, J.; Briton, B.G.H.; Kouassi, K.E.; Adouby, K. Nitrate removal by electrocoagulation process using experimental design methodology: A techno-economic optimization. J. Environ. Chem. Eng. 2020, 8, 104292. [Google Scholar] [CrossRef]
- AlJaberi, F.Y. Operating cost analysis of a concentric aluminum tubes electrodes electrocoagulation reactor. Heliyon 2019, 5, e02307. [Google Scholar] [CrossRef] [PubMed]
- Gönder, Z.B.; Balcıoğlu, G.; Vergili, I.; Kaya, Y. Electrochemical treatment of carwash wastewater using Fe and Al electrode: Techno-economic analysis and sludge characterization. J. Environ. Manag. 2017, 200, 380–390. [Google Scholar] [CrossRef]
- Bakshi, A.; Verma, A.K.; Dash, A.K. Electrocoagulation for removal of phosphate from aqueous solution: Statistical modeling and techno-economic study. J. Clean. Prod. 2020, 246, 118988. [Google Scholar] [CrossRef]
- Bian, Y.; Ge, Z.; Albano, C.; Lobo, F.L.; Ren, Z.J. Oily bilge water treatment using DC/AC powered electrocoagulation. Environ. Sci. Water Res. Technol. 2019, 5, 1654–1660. [Google Scholar] [CrossRef]
| Independent | Code | Code Levels | ||||
|---|---|---|---|---|---|---|
| Variables | Variables | −1.41 | −1 | 0 | +1 | +1.41 |
| Electrolysis time (min) | A | 5.85 | 22 | 61 | 100 | 116.15 |
| Current intensity (A) | B | 0.09 | 0.5 | 1.5 | 2.5 | 2.91 |
| Runs | Standard Run No. | Experimental Matrix | Removal Efficiency (%) | ||
|---|---|---|---|---|---|
| Pb | |||||
| A | B | Actual | Predicted | ||
| 1 | 12 | 0 | 0 | 98.90 | 98.2 |
| 2 | 13 | 0 | 0 | 97.51 | 98.2 |
| 3 | 11 | 0 | 0 | 96.4 | 98.2 |
| 4 | 5 | −1.41 | 0 | 92.89 | 92.72 |
| 5 | 10 | 0 | 0 | 99.87 | 98.2 |
| 6 | 2 | +1 | −1 | 91.53 | 91.75 |
| 7 | 7 | 0 | −1.41 | 88.45 | 88.13 |
| 8 | 3 | −1 | +1 | 95.35 | 95.36 |
| 9 | 4 | +1 | +1 | 100.00 | 99.92 |
| 10 | 6 | +1.41 | 0 | 97.64 | 97.59 |
| 11 | 9 | 0 | 0 | 98.31 | 98.2 |
| 12 | 1 | −1 | −1 | 89.12 | 89.42 |
| 13 | 8 | 0 | +1.41 | 97.99 | 98.08 |
| Parameter | Equation for Real Variables |
|---|---|
| Υ1 (Pb removal efficiency, %) | +98.20 + 1.72 × A + 3.52 × B + 0.56 × AB − 1.52 × A2 − 2.55 × B2 |
| Source of Variations | Sum of Squares | Df | Mean Square | F-Value | p-Value Prob > F | Remarks |
|---|---|---|---|---|---|---|
| Model | 179.67 | 5 | 35.93 | 34.48 | <0.0001 | Highly significant |
| A-Electrolysis time | 23.73 | 1 | 23.73 | 22.77 | 0.002 | significant |
| B-Current intensity | 99.36 | 1 | 99.36 | 95.34 | <0.0001 | Highly significant |
| AB | 1.25 | 1 | 1.25 | 1.20 | 0.3089 | |
| A2 | 16.15 | 1 | 16.15 | 15.50 | 0.0056 | Significant |
| B2 | 45.23 | 1 | 45.23 | 43.41 | 0.0003 | Significant |
| Residual | 7.29 | 7 | 1.04 | |||
| Lack of Fit | 0.29 | 3 | 0.096 | 0.055 | 0.9809 | Not significant |
| Pure Error | 7.01 | 4 | 1.75 | |||
| Cor Total | 186.97 | 12 | ||||
| R2/R2adj (%) = 0.96/0.93 | ||||||
| Parameters | Electrolysis Time (min) | Current Intensity (A) | Removal (%) Predict | Removal (%) Experimental |
|---|---|---|---|---|
| Optimal value | 77.65 | 0.9 | 97.8 | 95.52 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moosazade, M.; Ashoori, R.; Moghimi, H.; Amani, M.A.; Frontistis, Z.; Taheri, R.A. Electrochemical Recovery to Overcome Direct Osmosis Concentrate-Bearing Lead: Optimization of Treatment Process via RSM-CCD. Water 2021, 13, 3136. https://doi.org/10.3390/w13213136
Moosazade M, Ashoori R, Moghimi H, Amani MA, Frontistis Z, Taheri RA. Electrochemical Recovery to Overcome Direct Osmosis Concentrate-Bearing Lead: Optimization of Treatment Process via RSM-CCD. Water. 2021; 13(21):3136. https://doi.org/10.3390/w13213136
Chicago/Turabian StyleMoosazade, Milaad, Razieh Ashoori, Hamid Moghimi, Mohammad Ali Amani, Zacharias Frontistis, and Ramezan Ali Taheri. 2021. "Electrochemical Recovery to Overcome Direct Osmosis Concentrate-Bearing Lead: Optimization of Treatment Process via RSM-CCD" Water 13, no. 21: 3136. https://doi.org/10.3390/w13213136
APA StyleMoosazade, M., Ashoori, R., Moghimi, H., Amani, M. A., Frontistis, Z., & Taheri, R. A. (2021). Electrochemical Recovery to Overcome Direct Osmosis Concentrate-Bearing Lead: Optimization of Treatment Process via RSM-CCD. Water, 13(21), 3136. https://doi.org/10.3390/w13213136







