Greenhouse Gases Trade-Off from Ponds: An Overview of Emission Process and Their Driving Factors
Abstract
:1. Introduction
2. Methodology Adopted
3. Ponds in the Landscape
4. Mechanism of Greenhouse Gas Emission from Ponds
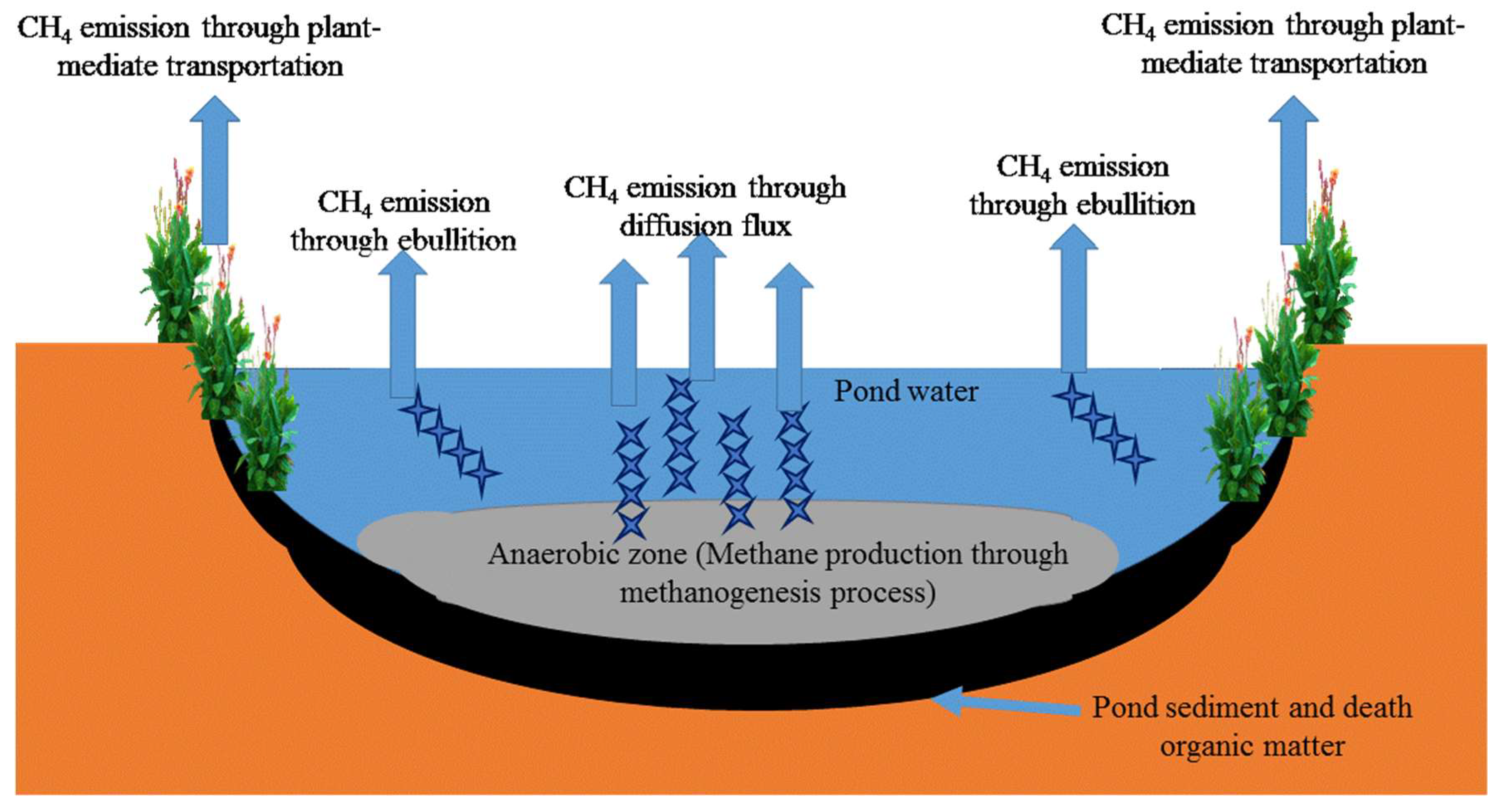
4.1. Methane Emission from Ponds
4.2. Carbon Dioxide Emissions from Ponds
4.3. Nitrous Oxide Emissions from Ponds
5. Methods for Greenhouse Gas Emissions Measurement from Ponds
5.1. Floating Chamber Method
5.2. Headspace Sampling
5.3. Bubbles Trapped Using Inverted Funnels
6. Factors Affecting Greenhouse Gas Emissions from Ponds
6.1. Effect of Water pH on GHG Emissions
6.1.1. Methane
6.1.2. Carbon Dioxide
6.1.3. Nitrous Oxide
6.2. Effect of Temperature on GHG
6.2.1. Methane
6.2.2. Carbon Dioxide
6.2.3. Nitrous Oxide
6.3. Effect of Nutrients Concentration on GHG Emissions
6.3.1. Methane
6.3.2. Carbon Dioxide
6.3.3. Nitrous Oxide
6.4. Effect of Eutrophication on CH4, CO2, and N2O Emission
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hoverman, J.T.; Johnson, P.T.J. Ponds and Lakes: A Journey Through the Life Aquatic. Nat. Educ. Knowl. 2012, 3, 17. [Google Scholar]
- Downing, J.A.; Duarte, C.M. Abundance and Size Distribution of Lakes, Ponds and Impoundments. Encycl. Inl. Waters 2009, 51, 469–478. [Google Scholar] [CrossRef]
- Kumar, M.; Padhy, P. Environmental Perspectives of Pond Ecosystems: Global Issues, Services and Indian Scenarios. Curr. World Environ. 2015, 10, 848–867. [Google Scholar] [CrossRef] [Green Version]
- Kumar, A.; Sharma, M.P. Assessment of risk of GHG emissions from Tehri hydropower reservoir, India. Hum. Ecol. Risk Assess Int. J. 2015, 22, 71–85. [Google Scholar] [CrossRef]
- Gorsky, A.L.; Racanelli, G.A.; Belvin, A.C.; Chambers, R.M. Greenhouse gas flux from stormwater ponds in southeastern Virginia (USA). Anthropocene 2019, 28, 100218. [Google Scholar] [CrossRef]
- Kumar, A.; Yu, Z.G.; Klemeš, J.J.; Bokhari, A. A state-of-the-art review of greenhouse gas emissions from Indian hydropower reservoirs. J. Clean. Prod. 2021, 320, 128806. [Google Scholar] [CrossRef]
- Malyan, S.K.; Bhatia, A.; Kumar, A.; Gupta, D.K.; Singh, R.; Kumar, S.S.; Tomer, R.; Kumar, O.; Jain, N. Methane production, oxidation and mitigation: A mechanistic understanding and comprehensive evaluation of influencing factors. Sci. Total Environ. 2016, 572, 874–896. [Google Scholar] [CrossRef]
- Malyan, S.K.; Bhatia, A.; Fagodiya, R.K.; Kumar, S.S.; Kumar, A.; Gupta, D.K.; Tomer, R.; Harit, R.C.; Kumar, V.; Jain, N.; et al. Plummeting global warming potential by chemicals interventions in irrigated rice: A lab to field assessment. Agric. Ecosyst. Environ. 2021, 319, 107545. [Google Scholar] [CrossRef]
- Malyan, S.K.; Yadav, S.; Sonkar, V.; Goyal, V.C.; Singh, O.; Singh, R. Mechanistic understanding of the pollutant removal and transformation processes in the constructed wetland system. Water Environ. Res. 2021, 93, 1882–1909. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, M.; Pandey, R.; Yu, Z.; Cabral-Pinto, M. Forest soil nutrient stocks along with an altitudinal range of Uttarakhand Himalayas: An aid to Nature Based Climate Solutions. Catena 2021, 207, 105667. [Google Scholar] [CrossRef]
- Kumar, A.; Tomer, R.; Bhatia, A.; Jain, N.; Pathak, H. Greenhouse gas mitigation technologies in Indian Agriculture. In Climate Change and Agriculture Technologies for Enhancing Resilience; ICARIARI: New Delhi, India, 2016; pp. 137–149. [Google Scholar]
- Baron, A.A.P.; Dyck, L.T.; Amjad, H.; Bragg, J.; Kroft, E.; Newson, J.; Oleson, K.; Casson, N.J.; North, R.L.; Venkiteswaran, J.J.; et al. Differences in ebullitive methane release from small, shallow ponds present challenges for scaling. Sci. Total Environ. 2022, 802, 149685. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Yang, T.; Sharma, M.P. Greenhouse Gas Measurement from Chinese Freshwater Bodies: A Review. J. Cleaner Prod. 2019, 233, 368–378. [Google Scholar] [CrossRef]
- Bastviken, D.; Cole, J.; Pace, M.; Tranvik, L. Methane emissions from lakes: Dependence of lake characteristics, two regional assessments, and a global estimate. Global Biogeochem. Cycles 2004, 18, 1–12. [Google Scholar] [CrossRef]
- Panneer Selvam, B.; Natchimuthu, S.; Arunachalam, L.; Bastviken, D. Methane and carbon dioxide emissions from inland waters in India–implications for large scale greenhouse gas balances. Glob. Chang. Biol. 2014, 20, 3397–3407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grinham, A.; Albert, S.; Deering, N.; Dunbabin, M.; Bastviken, D.; Sherman, B.; Lovelock, C.; Evans, C. The importance of small artificial water bodies as sources of methane emissions in Queensland, Australia. Hydrol. Earth Syst. Sci. 2018, 22, 5281–5298. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Zhang, M.; Xiao, W.; Jia, L.; Zhang, X.; Wang, J.; Zhang, Z.; Xie, Y.; Pu, Y.; Liu, S.; et al. Large methane emission from freshwater aquaculture ponds revealed by long-term eddy covariance observation. Agric. For. Meteorol. 2021, 308–309, 108600. [Google Scholar] [CrossRef]
- Yang, P.; Zhang, Y.; Yang, H.; Guo, Q.; Lai, D.Y.F.; Zhao, G.; Li, L.; Tong, C. Ebullition was a major pathway of methane emissions from the aquaculture ponds in southeast China. Water Res. 2020, 184, 116176. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Liu, D.; Xiang, J.; He, T.; Kang, H.; Ding, W. Methane and nitrous oxide have separated production zones and distinct emission pathways in freshwater aquaculture ponds. Water Res. 2021, 190, 116739. [Google Scholar] [CrossRef]
- Holgerson, M.A.; Raymond, P.A. Large contribution to inland water CO2 and CH4 emissions from very small ponds. Nat. Geosci. 2016, 9, 222–226. [Google Scholar] [CrossRef]
- Herrero Ortega, S.; Romero González-Quijano, C.; Casper, P.; Singer, G.A.; Gessner, M.O. Methane emissions from contrasting urban freshwaters: Rates, drivers, and a whole-city footprint. Glob. Chang. Biol. 2019, 25, 4234–4243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davidson, T.A.; Audet, J.; Jeppesen, E.; Landkildehus, F.; Lauridsen, T.L.; Søndergaard, M.; Syväranta, J. Synergy between nutrients and warming enhances methane ebullition from experimental lakes. Nat. Clim. Chang. 2018, 8, 156–160. [Google Scholar] [CrossRef]
- Prairie, Y.T.; Alm, J.; Beaulieu, J.; Barros, N.; Battin, T.; Cole, J.; del Giorgio, P.; DelSontro, T.; Guérin, F.; Harby, A.; et al. Greenhouse Gas Emissions from Freshwater Reservoirs: What Does the Atmosphere See? Ecosystems 2018, 21, 1058–1071. [Google Scholar] [CrossRef] [PubMed]
- Vachon, D.; Solomon, C.T.; del Giorgio, P.A. Reconstructing the seasonal dynamics and relative contribution of the major processes sustaining CO2 emissions in northern lakes. Limnol. Oceanogr. 2017, 62, 706–722. [Google Scholar] [CrossRef]
- Thottathil, S.D.; Reis, P.C.J.; Prairie, Y.T. Methane oxidation kinetics in northern freshwater lakes. Biogeochemistry 2019, 143, 105–116. [Google Scholar] [CrossRef]
- Fagodiya, R.K.; Pathak, H.; Bhatia, A.; Jain, N.; Gupta, D.K.; Kumar, A.; Malyan, S.K.; Dubey, R.; Radhakrishanan, S.; Tomer, R. Nitrous oxide emission and mitigation from maize–wheat rotation in the upper Indo-Gangetic Plains. Carbon Manag. 2019, 10, 489–499. [Google Scholar] [CrossRef]
- Xiao, Q.; Hu, Z.; Fu, C.; Bian, H.; Lee, X.; Chen, S.; Shang, D. Surface nitrous oxide concentrations and fluxes from water bodies of the agricultural watershed in Eastern China. Environ. Pollut. 2019, 251, 185–192. [Google Scholar] [CrossRef]
- Audet, J.; Carstensen, M.V.; Hoffmann, C.C.; Lavaux, L.; Thiemer, K.; Davidson, T.A. Greenhouse gas emissions from urban ponds in Denmark. Inl. Waters 2020, 10, 373–385. [Google Scholar] [CrossRef]
- Peacock, M.; Audet, J.; Bastviken, D.; Cook, S.; Evans, C.D.; Grinham, A.; Holgerson, M.A.; Högbom, L.; Pickard, A.E.; Zieliński, P.; et al. Small artificial waterbodies are widespread and persistent emitters of methane and carbon dioxide. Glob. Chang. Biol. 2021, 27, 5109–5123. [Google Scholar] [CrossRef] [PubMed]
- Peacock, M.; Audet, J.; Jordan, S.; Smeds, J.; Wallin, M.B. Greenhouse gas emissions from urban ponds are driven by nutrient status and hydrology. Ecosphere 2019, 10, e02643. [Google Scholar] [CrossRef]
- Pickard, A.; White, S.; Bhattacharyya, S.; Carvalho, L.; Dobel, A.; Drewer, J.; Jamwal, P.; Helfter, C. Greenhouse gas budgets of severely polluted urban lakes in India. Sci. Total Environ. 2021, 798, 149019. [Google Scholar] [CrossRef] [PubMed]
- Webb, J.R.; Leavitt, P.R.; Simpson, G.L.; Baulch, H.M.; Haig, H.A.; Hodder, K.R.; Finlay, K. Regulation of carbon dioxide and methane in small agricultural reservoirs: Optimizing potential for greenhouse gas uptake. Biogeosciences 2019, 16, 4211–4227. [Google Scholar] [CrossRef] [Green Version]
- Natchimuthu, S.; Panneer Selvam, B.; Bastviken, D. Influence of weather variables on methane and carbon dioxide flux from a shallow pond. Biogeochemistry 2014, 119, 403–413. [Google Scholar] [CrossRef]
- Singh, V.P.; Dass, P.; Kaur, K.; Billore, S.K.; Gupta, P.K.; Parashar, D.C. Nitrous oxide fluxes in a tropical shallow urban pond under influencing factors. Curr. Sci. 2005, 88, 478–483. [Google Scholar]
- Wang, G.; Xia, X.; Liu, S.; Zhang, L.; Zhang, S.; Wang, J.; Xi, N.; Zhang, Q. Intense methane ebullition from urban inland waters and its significant contribution to greenhouse gas emissions. Water Res. 2021, 189, 116654. [Google Scholar] [CrossRef]
- Schrier-Uijl, A.P.; Veraart, A.J.; Leffelaar, P.A.; Berendse, F.; Veenendaal, E.M. Release of CO2 and CH4 from lakes and drainage ditches in temperate wetlands. Biogeochemistry 2011, 102, 265–279. [Google Scholar] [CrossRef] [Green Version]
- Wik, M.; Varner, R.K.; Anthony, K.W.; MacIntyre, S.; Bastviken, D. Climate-sensitive northern lakes and ponds are critical components of methane release. Nat. Geosci. 2016, 9, 99–105. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, M.; Xiao, W.; Wang, W.; Zhang, Z.; Yu, Z.; Xiao, Q.; Cao, Z.; Xu, J.; Zhang, X.; et al. An evaluation of the flux-gradient and the eddy covariance method to measure CH4, CO2, and H2O fluxes from small ponds. Agric. For. Meteorol. 2019, 275, 255–264. [Google Scholar] [CrossRef]
- Kumar, A.; Bhatia, A.; Sehgal, V.K.; Tomer, R.; Jain, N.; Pathak, H. Net Ecosystem Exchange of Carbon Dioxide in Rice-Spring Wheat System of Northwestern Indo-Gangetic Plains. Land 2021, 10, 701. [Google Scholar] [CrossRef]
- Yang, S.S.; Chen, I.C.; Liu, C.P.; Liu, L.Y.; Chang, C.H. Carbon dioxide and methane emissions from Tanswei River in Northern Taiwan. Atmos. Pollut. Res. 2015, 6, 52–61. [Google Scholar] [CrossRef] [Green Version]
- Protocol, G.H.G. Required Greenhouse Gases in Inventories: Accounting and reporting standard amendment. Greenh. Gas Protoc. 2013, 8, 1–9. [Google Scholar]
- Halbedel, S. Protocol for CO2 sampling in waters by the use of the headspace equilibration technique, based on the simple gas equation; second update. Protocol Exch. 2015, 10, 1709–1727. [Google Scholar] [CrossRef] [Green Version]
- Koschorreck, M.; Prairie, Y.T.; Kim, J.; Marcé, R. Technical note: CO2 is not like CH4- Limits of and corrections to the headspace method to analyse pCO2 in fresh water. Biogeosciences 2021, 18, 1619–1627. [Google Scholar] [CrossRef]
- Egorov, A.V.; Nigmatulin, R.I.; Rimskii-Korsakov, N.A.; Rozhkov, A.N.; Sagalevich, A.M.; Chernyaev, E.S. Breakup of deep-water methane bubbles. Oceanology 2010, 50, 469–478. [Google Scholar] [CrossRef]
- McClure, R.P.; Lofton, M.E.; Chen, S.; Krueger, K.M.; Little, J.C.; Carey, C.C. The Magnitude and Drivers of Methane Ebullition and Diffusion Vary on a Longitudinal Gradient in a Small Freshwater Reservoir. J. Geophys. Res. Biogeosci. 2020, 125, e2019JG005205. [Google Scholar] [CrossRef]
- Hao, X.; Yu, R.; Zhang, Z.; Qi, Z.; Lu, X.; Liu, T.; Gao, R. Greenhouse gas emissions from the water–air interface of a grassland river: A case study of the Xilin River. Sci. Rep. 2021, 11, 2659. [Google Scholar] [CrossRef]
- van Bergen, T.J.H.M.; Barros, N.; Mendonça, R.; Aben, R.C.H.; Althuizen, I.H.J.; Huszar, V.; Lamers, L.P.M.; Lürling, M.; Roland, F.; Kosten, S. Seasonal and diel variation in greenhouse gas emissions from an urban pond and its major drivers. Limnol. Oceanogr. 2019, 64, 2129–2139. [Google Scholar] [CrossRef]
- DelSontro, T.; Boutet, L.; St-Pierre, A.; del Giorgio, P.A.; Prairie, Y.T. Methane ebullition and diffusion from northern ponds and lakes regulated by the interaction between temperature and system productivity. Limnol. Oceanogr. 2016, 61, S62–S77. [Google Scholar] [CrossRef]
- Beaulieu, J.J.; DelSontro, T.; Downing, J.A. Eutrophication will increase methane emissions from lakes and impoundments during the 21st century. Nat. Commun. 2019, 10, 3–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marotta, H.; Pinho, L.; Gudasz, C.; Bastviken, D.; Tranvik, L.J.; Enrich-Prast, A. Greenhouse gas production in low-latitude lake sediments responds strongly to warming. Nat. Clim. Chang. 2014, 4, 467–470. [Google Scholar] [CrossRef]
- Stadmark, J.; Leonardson, L. Greenhouse gas production in a pond sediment: Effects of temperature, nitrate, acetate and season. Sci. Total Environ. 2007, 387, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Khoiyangbam, R.S.; Chingangbam, S.S. Assessing seasonal variation of diffusive nitrous oxide emission from freshwater wetland in Keibul Lamjao National Park, Manipur Northeast India. Atmos. Environ. X 2022, 13, 100147. [Google Scholar] [CrossRef]
- Borges, A.V.; Darchambeau, F.; Teodoru, C.R.; Marwick, T.R.; Tamooh, F.; Geeraert, N.; Omengo, F.O.; Guérin, F.; Lambert, T.; Morana, C.; et al. Globally significant greenhouse-gas emissions from African inland waters. Nat. Geosci. 2015, 8, 637–642. [Google Scholar] [CrossRef] [Green Version]
- Paudel, S.R.; Choi, O.; Khanal, S.K.; Chandran, K.; Kim, S.; Lee, J.W. Effects of temperature on nitrous oxide (N2O) emission from intensive aquaculture system. Sci. Total Environ. 2015, 518–519, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Stadmark, J.; Leonardson, L. Emissions of greenhouse gases from ponds constructed for nitrogen removal. Ecol. Eng. 2005, 25, 542–551. [Google Scholar] [CrossRef]
- Zheng, Y.; Yu, K.; Freeman, C. Global methane and nitrous oxide emissions from terrestrial ecosystems due to multiple environmental changes. Ecosyst. Health Sustain. 2007, 1, 1–20. [Google Scholar] [CrossRef]
- Köhn, D.; Welpelo, C.; Günther, A.; Jurasinski, G. Drainage Ditches Contribute Considerably to the CH4 Budget of a Drained and a Rewetted Temperate Fen. Wetlands 2021, 41, 71. [Google Scholar] [CrossRef]
- Obrador, B.; Von Schiller, D.; Marcé, R.; Gómez-Gener, L.; Koschorreck, M.; Borrego, C.; Catalán, N. Dry habitats sustain high CO2 emissions from temporary ponds across seasons. Sci. Rep. 2018, 8, 3015. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.; Tong, W.; Chen, H.; Sun, J.; Wu, Z.; He, F. Quantification of N2O and NO emissions from a small-scale pond-ditch circulation system for rural polluted water treatment. Sci. Total Environ. 2018, 619–620, 946–956. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Sharma, M.P. Impact of water quality on GHG emissions from Hydropower Reservoir. J. Mater. Environ. Sci. 2014, 5, 95–100. [Google Scholar]
- DelSontro, T.; Beaulieu, J.J.; Downing, J.A. Greenhouse gas emissions from lakes and impoundments: Upscaling in the face of global change. Limnol. Oceanogr. Lett. 2018, 3, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Shang, J.; Zhang, C.; Zhang, W.; Niu, L.; Wang, L.; Zhang, H. The role of freshwater eutrophication in greenhouse gas emissions: A review. Sci. Total Environ. 2021, 768, 144582. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Lu, X.; Yu, R.; Yang, J.; Liu, X.; Cao, Z.; Zhang, Z.; Li, M.; Geng, Y. Eutrophication decreased CO2 but increased CH4 emissions from lake: A case study of a shallow Lake Ulansuhai. Water Res. 2021, 201, 117363. [Google Scholar] [CrossRef] [PubMed]
- Goyal, V.C.; Singh, O.; Singh, R.; Chhoden, K.; Kumar, J.; Yadav, S.; Singh, N.; Shrivastava, N.G.; Carvalho, L. Ecological health and water quality of village ponds in the subtropics limiting their use for water supply and groundwater recharge. J. Environ. Manag. 2021, 277, 111450. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, S.; Kanungo, V.K. Physico-Chemical Analysis of Pond Water of Surguja District, Chhattishgarh, India. Int. J. Herb. Med. 2013, 1, 35–43. [Google Scholar]
- Pandit, D.N.; Kumari, R.; Shitanshu, S.K. A comparative assessment of the status of Surajkund and Rani Pond, Aurangabad, Bihar, India using overall Index of Pollution and Water Quality Index. Acta Ecol. Sin. 2022, 18, 1–7. [Google Scholar] [CrossRef]
- Meenakshi, P.; Sriram, G. Water Quality Index and Correlation Study of Temple Ponds in Kanchipuram, Tamil Nadu, India. In Advances in Materials and Manufacturing Engineering; Springer Proceedings in Materials; Rajmohan, T., Palanikumar, K., Davim, J.P., Eds.; Springer: Singapore, 2021; Volume 7. [Google Scholar] [CrossRef]
- Sarkar, R.; Ghosh, A.R.; Mondal, N.K. Comparative study on physicochemical status and diversity of macrophytes and zooplanktons of two urban ponds of Chandannagar, WB, India. Appl. Water Sci. 2020, 10, 63. [Google Scholar] [CrossRef] [Green Version]
- Majeed, A.; Mandokhail, B.M.; Masood, Z.; Rehman, A.H.; Ullah, N.G. Assessment study about the water quality criteria and heavy metals concentrations in different fish ponds of four districts of Balochistan Province, Pakistan. Glob. Vet. 2015, 14, 351–357. [Google Scholar] [CrossRef]
- Ashraf, M.; Oweis, T.Y.; Razzaq, A.; Hussain, B.; Majid, A. Spatial and Temporal Analyses of Water Quality in the Dhrabi Watershed of Pakistan: Issues and Options. J. Environ. Sci. Eng. A 2012, 1, 329–340. [Google Scholar]
- Perron, M.A.C.; Pick, F.R. Water quality effects on dragonfly and damselfly nymph communities: A comparison of urban and natural ponds. Environ. Pollut. 2020, 263, 114472. [Google Scholar] [CrossRef] [PubMed]
- Perron, M.A.C.; Richmond, I.C.; Pick, F.R. Plants, water quality and land cover as drivers of Odonata assemblages in urban ponds. Sci. Total Environ. 2021, 773, 145467. [Google Scholar] [CrossRef] [PubMed]
- Serder, M.F.; Islam, M.S.; Hasan, M.R.; Yeasmin, M.S.; Mostafa, M.G. Assessment of coastal surface water quality for irrigation purpose. Water Pract. Technol. 2020, 15, 960–972. [Google Scholar] [CrossRef]
- Wurts, W.A.; Durborow, R.M. Interactions of pH, Carbon Dioxide, Alkalinity and Hardness in Fish Ponds; Publication ID SRAC 464; KSU State Extension Specialist for Aquaculture: Paducah, Kentucky, 1992; pp. 1–4. [Google Scholar]
- Malyan, S.K.; Bhatia, A.; Tomer, R.; Harit, R.C.; Jain, N.; Bhowmik, A.; Kaushik, R. Mitigation of yield-scaled greenhouse gas emissions from irrigated rice through Azolla, Blue-green algae, and plant growth–promoting bacteria. Environ. Sci. Pollut. Res. 2021, 28, 51425–51439. [Google Scholar] [CrossRef] [PubMed]
- Samarkin, V.A.; Madigan, M.T.; Bowles, M.W.; Casciotti, K.L.; Priscu, J.C.; McKay, C.P.; Joye, S.B. Abiotic nitrous oxide emission from the hypersaline Don Juan Pond in Antarctica. Nat. Geosci. 2010, 3, 341–344. [Google Scholar] [CrossRef]
- Mendonça, R.; Müller, R.A.; Clow, D.; Verpoorter, C.; Raymond, P.; Tranvik, L.J.; Sobek, S. Organic carbon burial in global lakes and reservoirs. Nat. Commun. 2017, 8, 1694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, S.S.; Malyan, S.K.; Basu, S.; Bishnoi, N.R. Syntrophic association and performance of Clostridium, Desulfovibrio, Aeromonas and Tetrathiobacter as anodic biocatalysts for bioelectricity generation in dual chamber microbial fuel cell. Environ. Sci. Pollut. Res. 2017, 24, 16019–16030. [Google Scholar] [CrossRef] [PubMed]
- Badiou, P.; Page, B.; Ross, L. A comparison of water quality and greenhouse gas emissions in constructed wetlands and conventional retention basins with and without submerged macrophyte management for storm water regulation. Ecol. Eng. 2019, 127, 292–301. [Google Scholar] [CrossRef]
- Malyan, S.K.; Kumar, S.S.; Fagodiya, R.K.; Ghosh, P.; Kumar, A.; Singh, R.; Singh, L. Biochar for environmental sustainability in the energy-water-agroecosystem nexus. Renew. Sustain. Energy Rev. 2021, 149, 111379. [Google Scholar] [CrossRef]
- Bao, Q.; Liu, Z.; Zhao, M.; Hu, Y.; Li, D.; Han, C.; Zeng, C.; Chen, B.; Wei, Y.; Ma, S.; et al. Role of carbon and nutrient exports from different land uses in the aquatic carbon sequestration and eutrophication process. Sci. Total Environ. 2022, 813, 151917. [Google Scholar] [CrossRef] [PubMed]
| Reference | Location Detail | Sampling Methodology | Methane | Carbon Dioxide | Nitrous Oxide | Remarks |
|---|---|---|---|---|---|---|
| Selvam et al. [15] | Different water bodies of Tamil Nadu, Andhra Pradesh, and Kerala | Floating chamber | 0.1–52.1 mmol m−2 day−1 | −28.2 to 262.4 mmol m−2 day−1 | - | In this study, 45 different water bodies, such as ponds, open wells, lakes, and channels were studied |
| Audet et al. [28] | Silkeborg, Denmark | Headspace sampling | 44 µg L−1 | 1938 µg L−1 | 0.8 µg L−1 | In this study, authors reported the mean concentration of GHG emissions from urban ponds |
| Peacock et al. [29] | Uppsala, Sweden | Floating chamber | 0.1–44.3 g CH4 m−2 year−1 | −36 to 4421 g CO2 m−2 year−1 | - | Small constructed ponds and ditches |
| Peacock et al. [30] | Uppsala, Sweden | Headspace method | 0.4–174 mg CH4 m−2 year−1 | −187 to 3449 mg CH4 m−2 year−1 | - | Small urban ponds |
| Pickard et al. [31] | Bengaluru, India | Headspace sampling | 0.33–3413 ton CH4-C evasion year−1 | 24–5711 CO2-C evasion tone year−1 | - | Inland polluted urban lakes near industrial areas |
| Webb et al. [32] | Saskatchewan, Canada | Headspace method | 0.14–92 mmol m−2 day−1 | 21–466 mmol m−2 d−1 | - | Small agricultural farm reservoir |
| Natchimthu et al. [33] | Linkoping University, Sweden | Floating chamber | 3.3–15.1 mmol m−2 day−1 | −9.8 to 16.0 mmol m−2 day−1 | - | Freshwater shallow pond |
| Singh et al. [34] | Ujjain City, India | Floating chamber | - | - | 0.00–0.51 mg m−2 day−1 | Urban pond receiving domestic and agricultural runoff |
| Wang et al. [35] | Beijing, China | Headspace sampling | 0.08–8.3 mmol m−2 day−1 | −24.2 to 37.9 mmol m−2 day−1 | - | Urban inland water bodies such as lakes |
| Schrier-Uijl et al. [36] | Different peats drainage ditches of Netherlands | Floating method | 33.7 mg m−2 h−1 | 129.1 mg m−2 h−1 | - | In this study, mean CH4 and CO2 emissions were reported from shallow freshwater bodies |
| Ortega et al. [21] | Berlin, Germany | Floating chamber | 385 Mg CH4 year−1 | - | - | Emissions from urban ponds |
| Grinham et al. [16] | Queensland, Australia | Floating chamber | 1.6 Mt CO2 eq. year-1 | - | - | Small artificial ponds, lakes, etc., were investigated |
| Wik et al. [37] | 733 lakes and ponds of Northern regional | - | 1.0 Tg CH4 year−1 | - | - | Findings for Beaver ponds |
| 3.1 Tg CH4 year−1 | - | - | Results of peatland ponds | |||
| 16.5 Tg CH4 year−1 | - | - | Total CH4 emissions from Beaver ponds, Peatland ponds, Glacial/post-glacial lakes, and Thermokarst bodies | |||
| Zhao et al. [38] | Anhui Province, China | Eddy covariance | 1.05–1.66 µg m−2 s−1 | 0.011–0.024 mg m−2 s−1 | - | Two small fish ponds |
| Factors | Correlation with CH4 Flux (References) | Correlation with CO2 Flux (References) | Correlation with N2O Flux (References) |
|---|---|---|---|
| Water temperature | Increased exponentially up to certain limit [22,28,29,33,35,47,48,50,51] | Positive [28,29,35] | Positive [28,50,52] |
| No correlation [12,53] | No correlation [47,48] | - | |
| - | Negative [33] | Negative [54] | |
| Nitrate concentration | No impact [51] | Positive [55] | Positive [51,55,56] |
| Negative [28,57] | - | - | |
| Dissolved organic matter (DOM) | Positive [5,30,35,40,56,58] | Positive [5,53,58] | Positive correlation [5,59] |
| Water pH | - | Positive [5,60] | Positive [59] |
| Non-significant correlation [40,46] | - | Non-clear correlation [52] | |
| - | Negative [4,5,30,40,46] | Negative [56] | |
| Dissolved oxygen (DO) | - | - | Positive [46,59] |
| No correlation [46] | - | - | |
| Negative [28,29,40,56] | Negative [40,46] | Negative [56] | |
| Surface area of pond | Negative [5,29] | - | Positive [61] |
| Total nitrogen (TN) | - | Positive [46] | Positive [52,56] |
| Total phosphorus (TP) | Positive [29,30,46,57] | Positive [46] | Positive [46] |
| Sulphate | Negative [12,28] | - | - |
| Eutrophication | Enhanced CH4 flux [22,49,62,63] | Positive [62] | - |
| - | Negative [63] | Negative [62] |
| Location of the Study/Reference | Range of Water pH | Remarks |
|---|---|---|
| Uttar Pradesh, India [64] | 7.2–8.2 | Out of 12 studied ponds, only at 1 location, the pond water pH was 9.2 (No GHG) |
| Chhattisgarh, India [65] | 6.93–7.55 | The authors investigated the water quality of 10 ponds (No GHG) |
| Bihar, India [66] | 6.35–7.57 | Pond water quality of two districts was investigated in this study. (No GHG) |
| Tamil Nadu, India [67] | 7.25–8.85 | Out of a total of 17 investigated temple ponds, only 2 ponds (pH 9.07 and 8.42) had a pH above 8. (No GHG) |
| West Bengal, India [68] | 7.5–7.9 | In this study, water quality of both rural and urban ponds was conducted. (No GHG) |
| Balochistan province, Pakistan [69] | 7.11–7.96 | This study conducted the assessment of the water quality of fish farming ponds of four districts of Balochistan province, Pakistan. (No GHG) |
| Dhrabi Watershed, Pakistan [70] | 7.2 | The study was conducted in Dhrabi reservoir of Pakistan. |
| Ontario, Canada [71] | 5.77–7.74 | In this study, water quality of 51 natural ponds was investigated. |
| National Capital Region of Canada, Canada [72] | 5.77–9.23 | In this study, water quality of 10 natural ponds and 40 stormwater constructed ponds was investigated. The pH of all-natural ponds was below 8 while higher pH was observed in stormwater constructed ponds. |
| Patuakhali district, coastal watershed, Bangladesh [73] | 7.6–7.9 | Water quality of coastal ponds during pre-monsoon, monsoon, and post-monsoon was reported. |
| Silkeborg, Denmark [28] | 7.5–8.0 | The water quality of four urban ponds was reported in this study. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malyan, S.K.; Singh, O.; Kumar, A.; Anand, G.; Singh, R.; Singh, S.; Yu, Z.; Kumar, J.; Fagodiya, R.K.; Kumar, A. Greenhouse Gases Trade-Off from Ponds: An Overview of Emission Process and Their Driving Factors. Water 2022, 14, 970. https://doi.org/10.3390/w14060970
Malyan SK, Singh O, Kumar A, Anand G, Singh R, Singh S, Yu Z, Kumar J, Fagodiya RK, Kumar A. Greenhouse Gases Trade-Off from Ponds: An Overview of Emission Process and Their Driving Factors. Water. 2022; 14(6):970. https://doi.org/10.3390/w14060970
Chicago/Turabian StyleMalyan, Sandeep K., Omkar Singh, Amit Kumar, Gagan Anand, Rajesh Singh, Sandeep Singh, Zhiguo Yu, Jhlaesh Kumar, Ram K. Fagodiya, and Amit Kumar. 2022. "Greenhouse Gases Trade-Off from Ponds: An Overview of Emission Process and Their Driving Factors" Water 14, no. 6: 970. https://doi.org/10.3390/w14060970









