Geochemistry of Thermal and Cold Mineral Water and Gases of the Tien Shan and the Pamir
Abstract
:1. Introduction
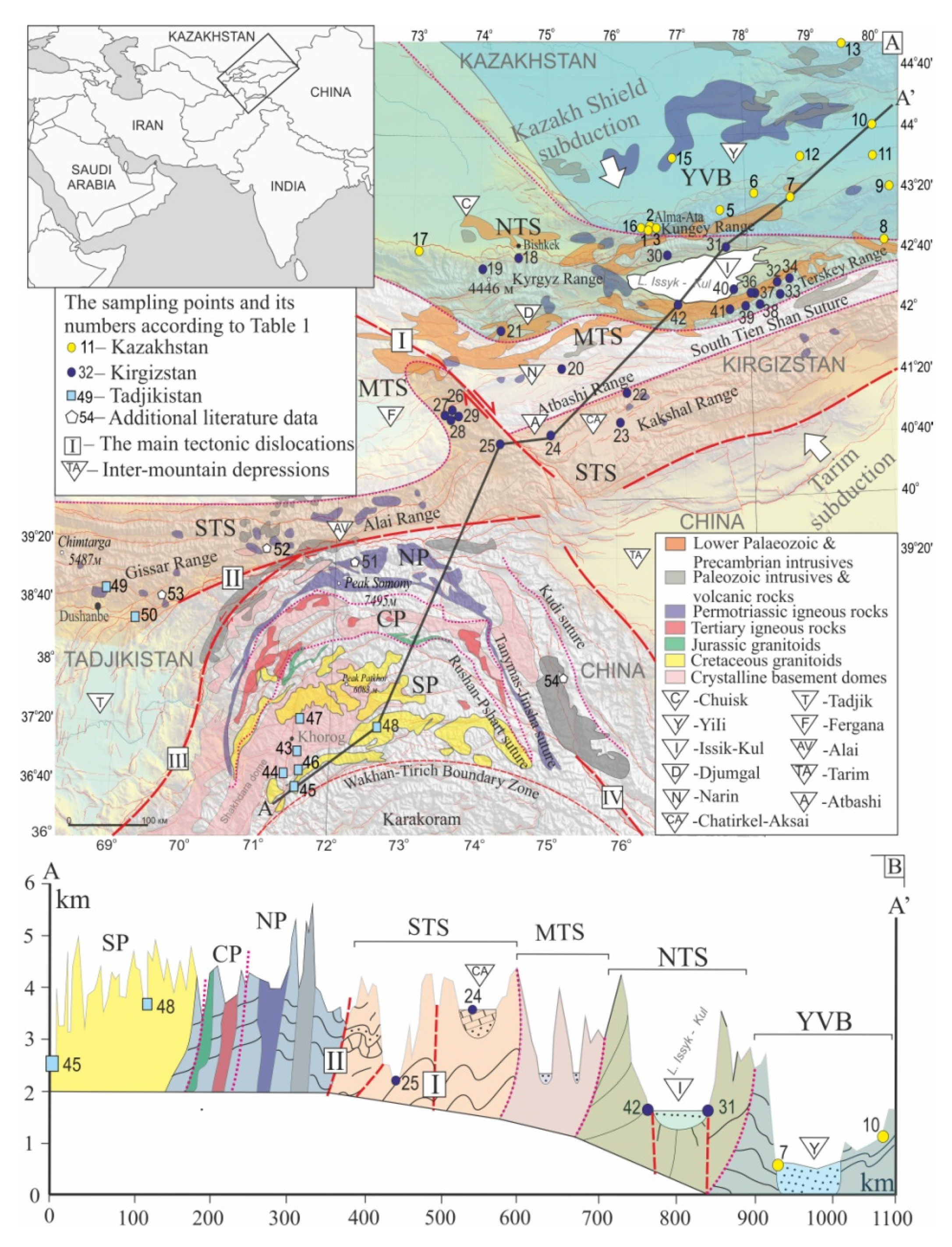
2. Geological and Tectonic Framework
3. Materials and Methods
- TSiO2 = [1032/(4.69 − lgSiO2)] − 273.15, [37];
- TSiO2 = −44.119 + 0.24469 SiO2 − 1.7414 × 10−4 SiO22 + 79.305 lgSiO2; Improved SiO2 [38]
- TNa–Li = 1049/[lg(Na/Li) + 0.44] − 273.15, [39];
- TNa–K–Ca = 1647/(lg(Na/K) + (4/3)[lg(Ca0.5/Na) + 2.06] + 2.47) − 273.15, [40].
- TCa–Mg = 979.8/3.1170 − log(Ca/Mg) + 0.07003 logΣeq − 273.15, [41]
- TK–Mg = 4410/14 − log(K2/Mg) − 273.15, [42]
4. Results and Discussion
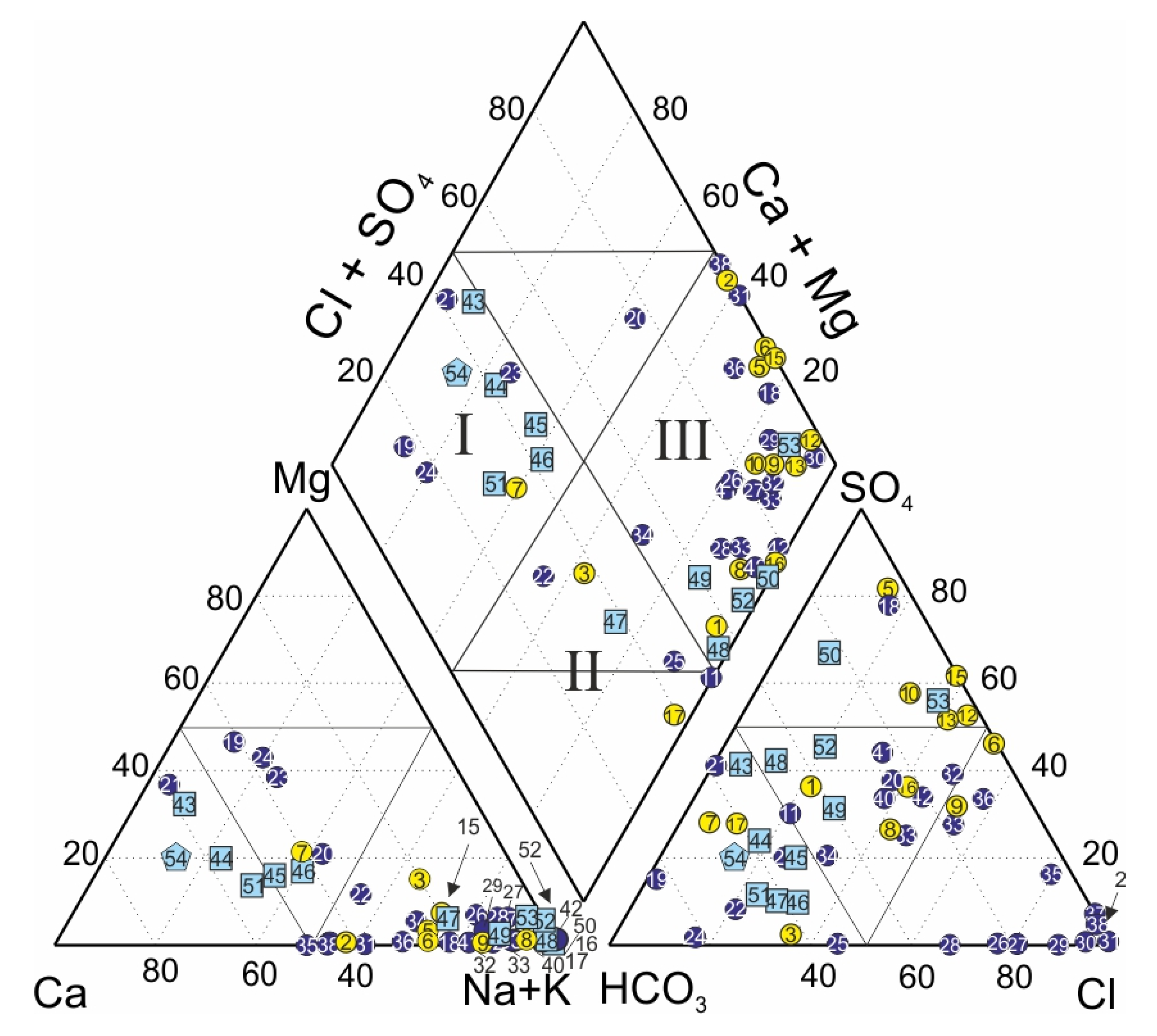
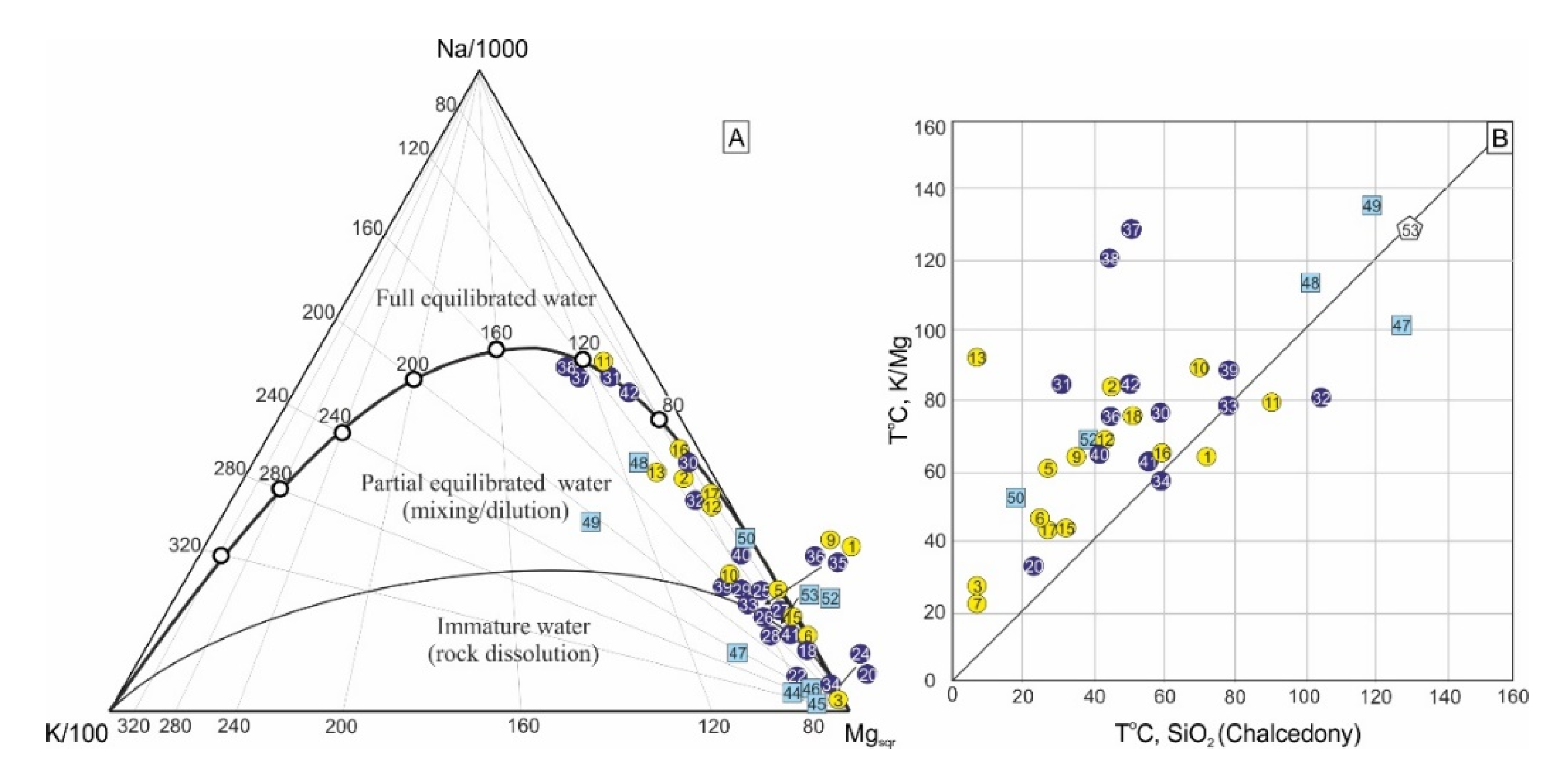
4.1. Water Chemistry
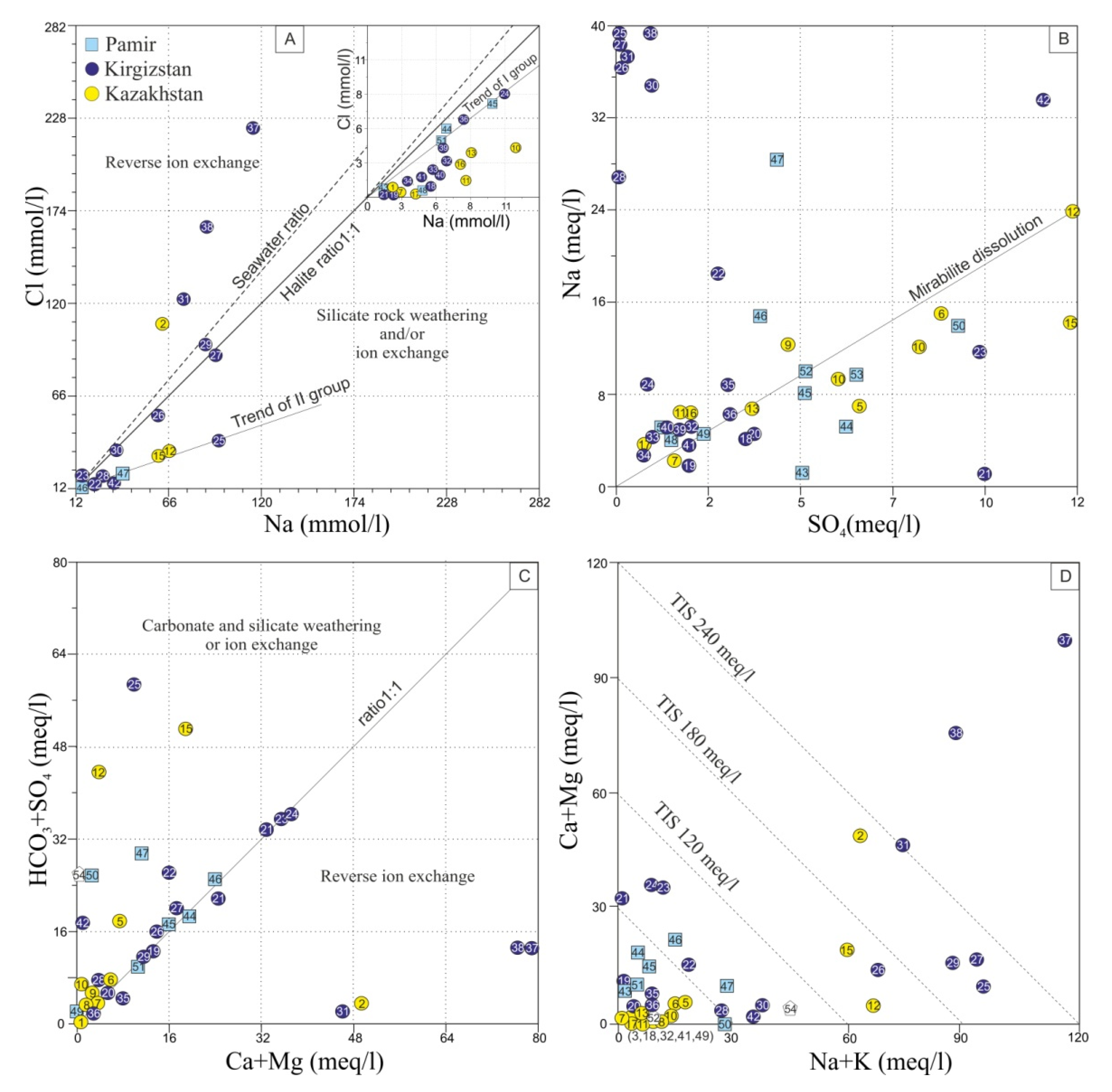
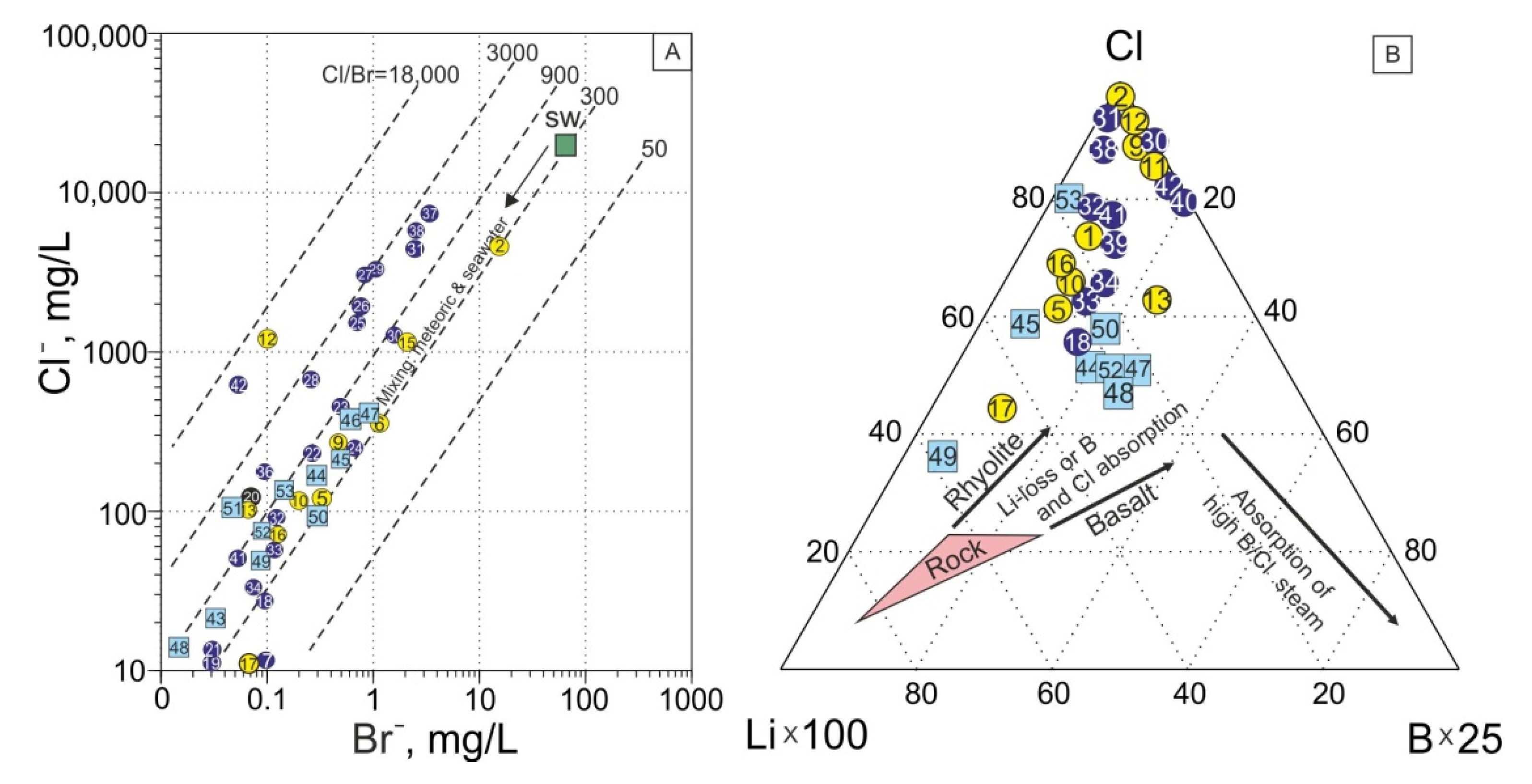
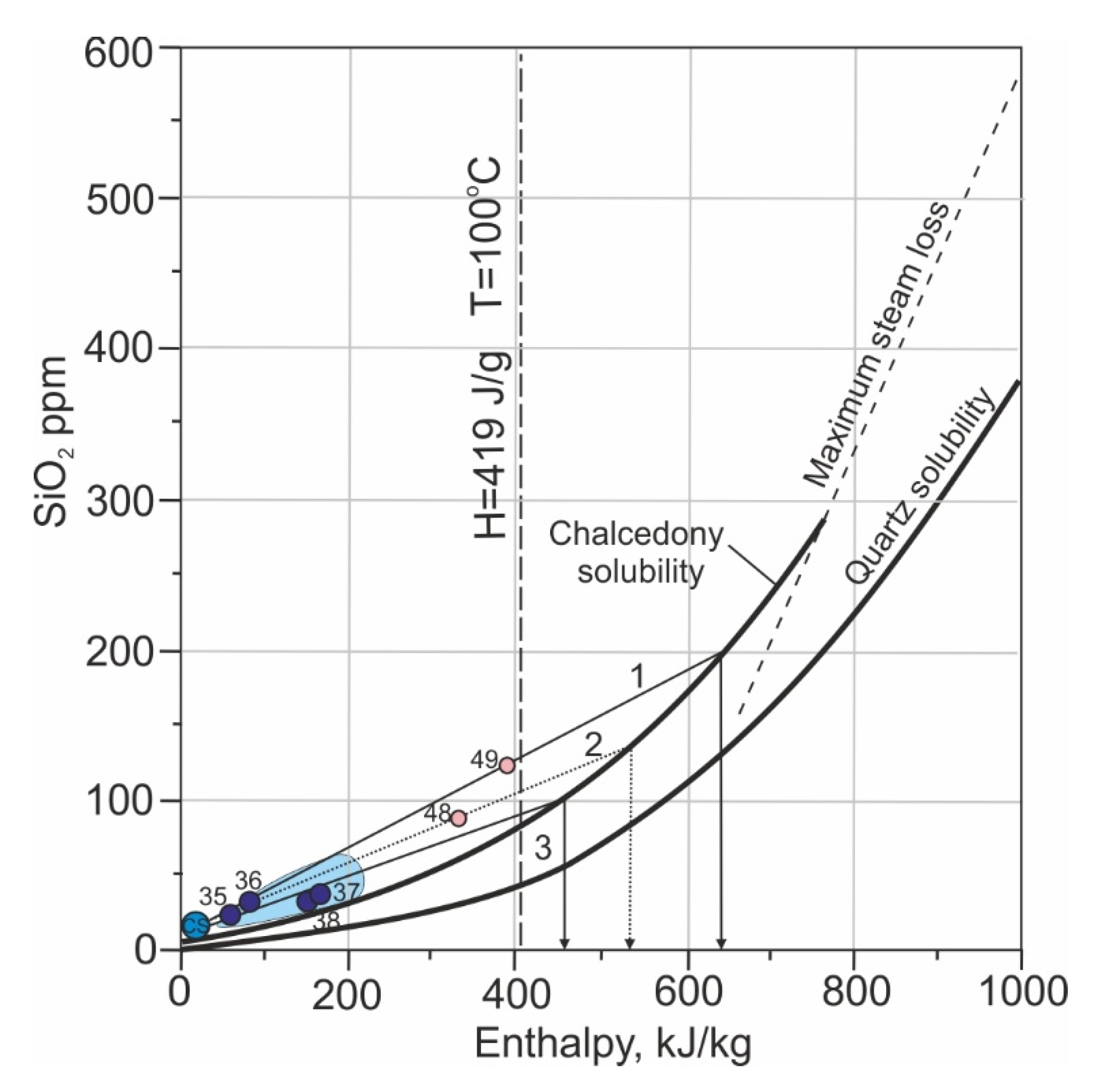
4.2. Fate of Chemical Heterogeneity
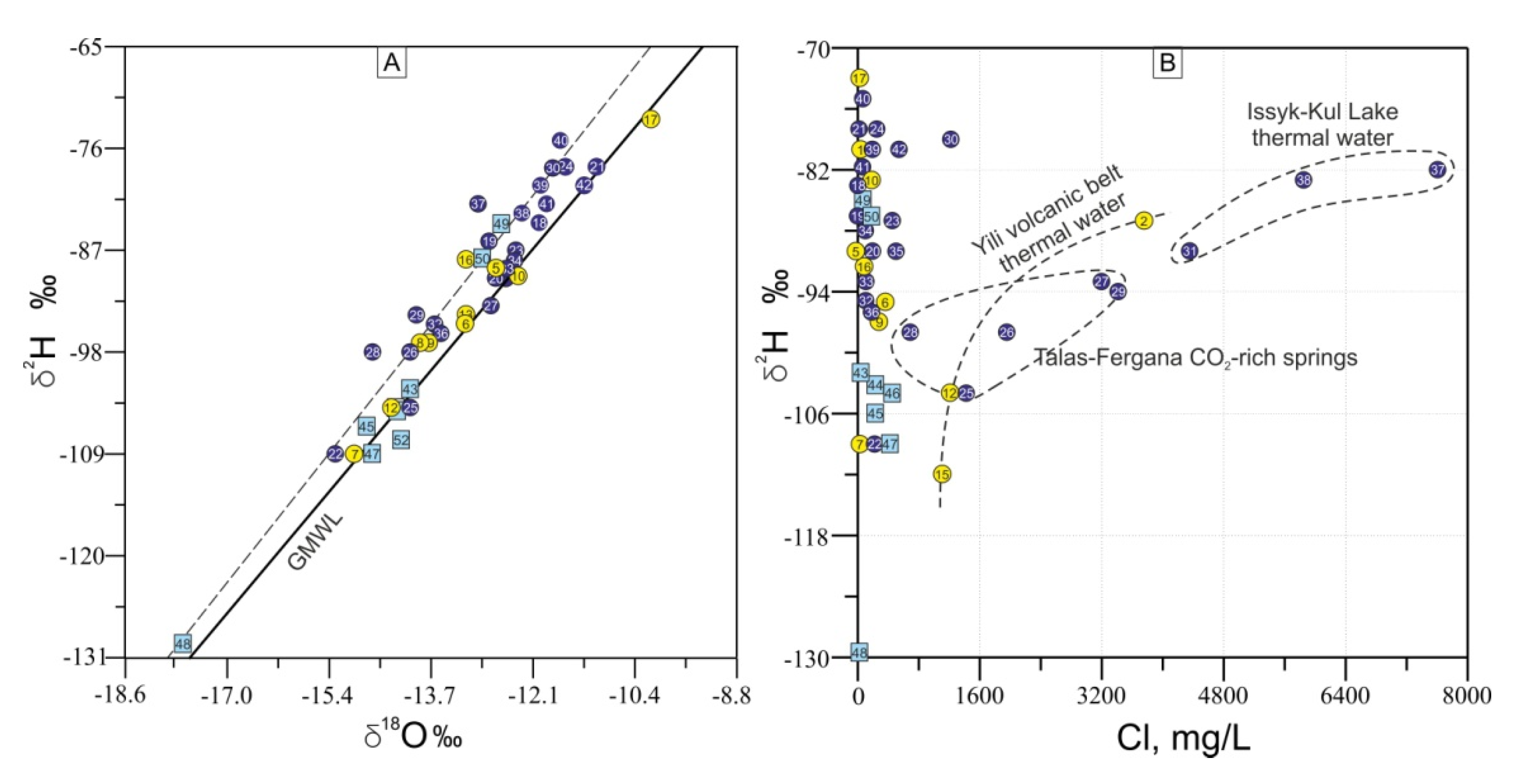
4.3. Isotopic Composition of Waters
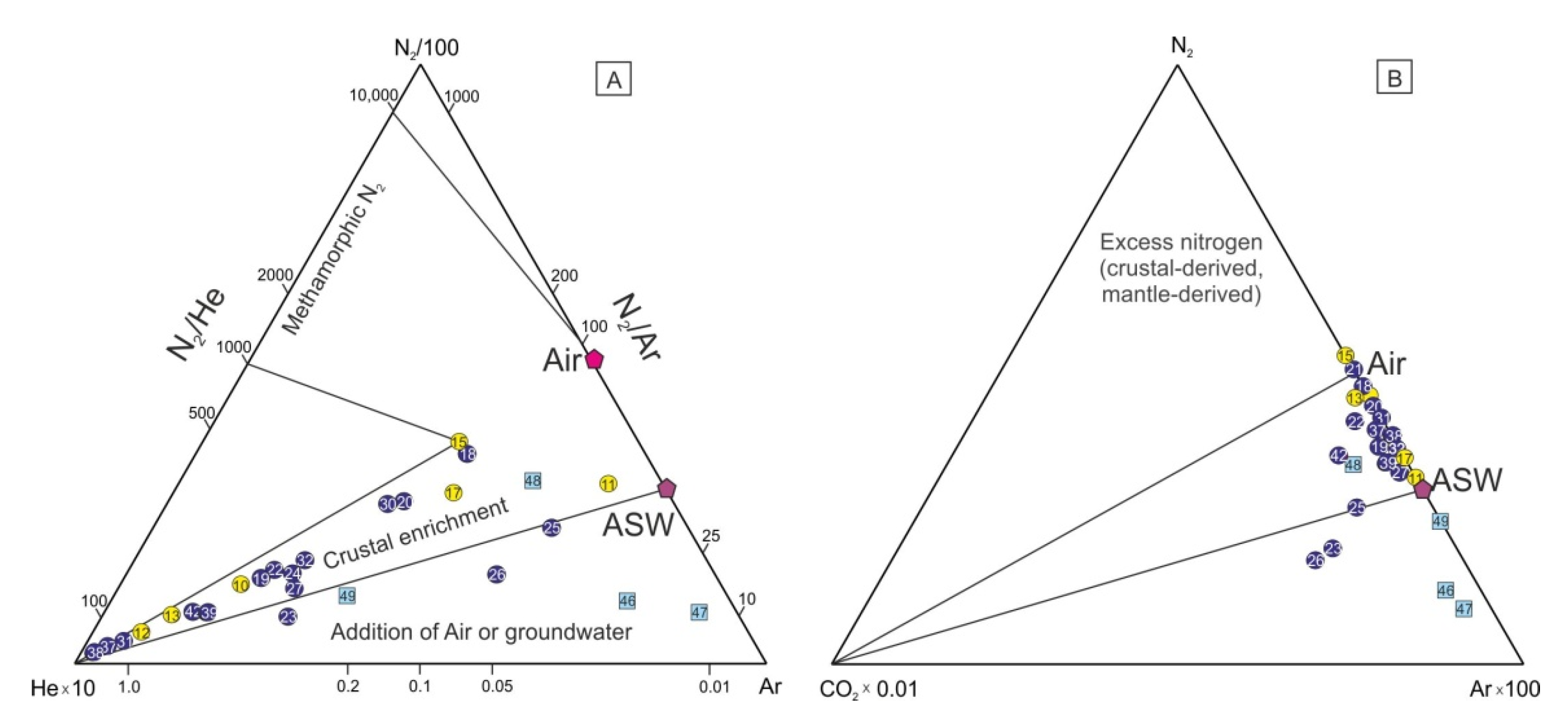
4.4. Chemical and Isotopic Composition of Gases
4.5. The Overview of Obtained Regional Tectonic and Geochemical Results
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Krainov, S.P.; Petrova, N.G. Trace elements of mineral waters pf Pamir. J. Geochem. 1962, 4, 356–366. (In Russian) [Google Scholar]
- Churshina, N.M. Geothermal Investigations in Central Asia and Kazakhstan; M: Nauka: Moscow, Russia, 1985; pp. 95–101. (In Russian) [Google Scholar]
- Zhevago, V.S. Geothermy and Thermal Waters of Kazakhstan; Nauka: Alma-Ata, Kazakhstan, 1972; p. 225. [Google Scholar]
- Matychenkov, V.E.; Imankulov, B.I. Mineral Waters in Kirghizia; Llim: Frunze, Kirghizia, 1987; p. 250. (In Russian) [Google Scholar]
- Volkenshtein, S.K. Ak−su hot springs. In Mineral Waters of the Semirechensk Region; B. И. AндpУcoв: Kazan, Russia, 1901; pp. 3–19. (In Russian) [Google Scholar]
- Prokopenko, N.M. Thermal Water of Pamir; Pamir Expedition: Moscow, Russia, 1932; Volume 1. [Google Scholar]
- Prochukhan, D.P. Ak-su and Dzhety-Ogyz hot springs (Kirghizia). In Geology and Geochemistry of the Tien Shan; Gatalskiy, M.M., Ed.; Proceedings of Kirgizstan. Special Expedition of 1932−1933; Izd. AN SSSR: Moscow, Russia, 1935; Volume II, Part V; pp. 155–184. (In Russian) [Google Scholar]
- Vasilieva, V.N. Formation of Dzhety−Oguz radioactive thermal springs. In Formation and Distribution of Mineral Wafers in the USSR Territory; Ivanov, V.V., Ed.; Gosgeoltehizdat: Moscow, Russia, 1960; pp. 47–50. (In Russian) [Google Scholar]
- Disler, V.N. High pCO2 Mineral Waters of the Tien Shan and Pamir. Hydrogeological Questions of the Mineral Waters; Research Institute of Balneology and Physiotherapy, Nauka: Moscow, Russia, 1977; Volume 34, pp. 192–218. (In Russian) [Google Scholar]
- Rybin, A.K.; Batalev, V.Y.; Bataleva, Е.A. 3He/4He Isotopic Composition in Gases of Thermal Springs of the Tian Shan; VII Int. Symposium: Bishkek, Kyrgyzstan, 2018; pp. 128–134. [Google Scholar]
- Demonova, A.Y.; Kharitonova, N.A.; Korzun, A.V.; Sardorov, A.I.; Chelnokov, G.A. The chemical composition of the nitrogen thermal waters of the balneoclimatic of Khoja-Obi-Garm Spa (Tajikistan). Mosc. Univ. Bulletin. S 4. Geol. 2017, 5, 77–84. (In Russian) [Google Scholar] [CrossRef]
- Demonova, A.; Kharitonova, N.; Bragin, I.; Chelnokov, G.; Ivanov, V. Low-enthalpy thermal waters within Khoja-Obi-Garm field (Republic of Tajikistan). E3S Web Conf. 2019, 98, 01011. [Google Scholar] [CrossRef]
- Kireeva, T.K.; Salikhov, F.S.; Bychkov, A.Y.; Kharitonova, N.A. Chemical Composition and Parameters of Formation of the Waters of Some Thermal Springs in Tajikistan. Geochem. Int. 2020, 58, 423–434. [Google Scholar] [CrossRef]
- Voitov, G.I.; Kucher, M.I.; Denisov, S.A. Features of the water and gas chemistry fate and isotopic composition of CO2 and CH4 in mineral springs of Djete-Oguz. Doklady AN SSSR 1988, 302, 1212–1216. (In Russian) [Google Scholar]
- Abdullaev, A.U.; Voitov, G.I.; Imanbaeva, M.D. Chemical, isotopical and heat flow instabilities of thermal springs of Kirgizstan. In Geothermy of Seismic and Non−Seismic Zones; Nauka: Moscow, Russia, 1993; p. 400. [Google Scholar]
- Matychenkov, V.E. Hydromineral resources of the Issyk−Kul region In Lake Issyk−Kul: Its Natural Environment; Klerkx, J., Imanackunov, B., Eds.; NATO Science Series; Earth and Environmental Sciences; Kluwer Academic Publishers: Dordrecht, The Netherlands; Springer: Amsterdam, The Netherlands, 2002; Volume 13, pp. 59–70. [Google Scholar]
- Bucher, K.; Zhang, L.; Stober, I. A hot spring in granite of the Western Tianshan, China. Appl. Geochem. 2009, 24, 402–410. [Google Scholar] [CrossRef]
- Li, Y.; Pang, Z.; Yang, F.; Yuan, L.; Tang, P. Hydrogeochemical characteristics and genesis of the high−temperature geothermal system in the Tashkorgan basin of the Pamir syntax, western China. J. Asian Earth Sci. 2017, 149, 134–144. [Google Scholar] [CrossRef]
- Krestnikov, V.N.; Nersesov, I.L.; Stange, D.V. The relationship between the deep structure and quaternary tectonics of the Pamir and Tien-Shan. Tectonophysics 1984, 104, 67–83. [Google Scholar] [CrossRef]
- Brunet, M.-F.; Sobel, E.R.; McCann, T. Geological evolution of Central Asian Basins and the western Tien Shan Range. Geol. Soc. Lond. Spec. Publ. 2017, 427, 1–17. [Google Scholar] [CrossRef]
- Medved, I.; Bataleva, E.; Buslov, M. Studying the Depth Structure of the Kyrgyz Tien Shan by Using the Seismic Tomography and Magnetotelluric Sounding Methods. Geosciences 2021, 11, 122. [Google Scholar] [CrossRef]
- Khan, N.G.; Bai, L.; Zhao, J.; Li, G.; Moklesur Rahman, M.; Cheng, C.; Yang, J. Crustal structure beneath Tien Shan orogenic belt and its adjacent regions from multi-scale seismic data. Sci. China Earth Sci. 2017, 60, 1769–1782. [Google Scholar] [CrossRef]
- Mechie, J.; Yuan, X.; Schurr, B.; Schneider, F.; Sippl, C.; Ratschbacher, L.; Minaev, V.; Gadoev, M.; Oimahmadov, I.; Abdybachaev, U.; et al. Crustal and uppermost mantle velocity structure along a profile across the Pamir and southern Tien Shan as derivedfrom project TIPAGE wide-angle seismic data. Geophys. J. Int. 2012, 188, 385–407. [Google Scholar] [CrossRef] [Green Version]
- Molnar, P.; Tapponnier, P. Cenozoic tectonics of Asia: Effects of a continental collision. Science 1975, 189, 419–426. [Google Scholar] [CrossRef]
- Burtman, V.S.; Skobelev, S.F.; Molnar, P. Late Cenozoic slip on the Talas-Ferghana fault, the Tien Shan, central Asia. Geol. Soc. Am. Bull. 1996, 108, 1004–1021. [Google Scholar] [CrossRef]
- Safonova, I.; Seltmann, R.; Kröner, A.; Gladkochub, D.; Schulmann, K.; Xiao, W.; Kim, J.; Komiya, T.; Sun, M. A new concept of continental construction in the Central Asian Orogenic Belt. Episodes 2011, 34, 186–196. [Google Scholar] [CrossRef] [Green Version]
- Schneider, F.M.; Yuan, X.; Schurr, B.; Mechie, J.; Sippl, C.; Kufner, S.K.; Murodkulov, S. The Crust in the Pamir: Insights from Receiver Functions. J. Geoph. Res. Solid Earth 2019, 124, 9313–9331. [Google Scholar] [CrossRef] [Green Version]
- Alekseev, D.V.; Kroner, A.; Hegner, E.; Rojas-Agramonte, Y.; Biske, Y.S.; Wong, J.; Geng, H.Y.; Ivleva, E.A.; Mühlberg, M.; Mikolaichuk, A.V.; et al. Middle to Late Ordovician arc system in the Kyrgyz Middle Tian Shan: From arc-continent collision to subsequent evolution of a Palaeozoic continental margin. Gond. Res. 2016, 39, 261–291. [Google Scholar]
- Biske, Y.S.; Seltmann, R. Paleozoic Tian Shan as a transitional region between the Rheic and Urals−Turkestan oceans. Gond. Res. 2010, 17, 602–613. [Google Scholar] [CrossRef]
- Thomas, J.-C.; Cobbold, P.R.; Shein, V.S.; Le Douaran, S. Sedimentary record of late Paleozoic to Recent tectonism in central Asia—Analysis of subsurface data from the Turan and south Kazak domains. Tectonophysics 1999, 313, 243–263. [Google Scholar] [CrossRef]
- Korjenkov, A.M. General Peculiarities of Neotectonic Structural Forms of the Northeastern Tien Shan. Ph.D. Thesis, Institute of Seismology, AS Kirghiz SSR, Frunze, Russia, 1988; p. 261. (In Russian). [Google Scholar]
- Seltmann, R.; Konopelko, D.; Biske, G.; Divaev, F.; Sergeev, S. Hercynian post−collisional magmatism in the context of Paleozoic magmatic evolution of the Tien Shan orogenic belt. J. Asian Earth Sci. 2011, 42, 821–838. [Google Scholar] [CrossRef]
- Hamburger, M.W.; Sarewitz, D.R.; Pavlis, T.L.; Popandopulo, G.A. Structural and seismic evidence for intracontinental subduction in the Peter the First Range, Central Asia. GSA Bull. 1992, 104, 397–408. [Google Scholar] [CrossRef]
- Yogibekov, D.; Sang, M.; Xiao, W.; Windley, B.F.; Mamadjonov, Y.; Yang, H.; Huang, P.; Aminov, J.; Vatanbekov, F. Late Palaeozoic to Late Triassic northward accretion and incorporation of seamounts along the northern South Pamir: Insights from the anatomy of the Pshart accretionary complex. Geol. J. 2020, 55, 7837–7857. [Google Scholar] [CrossRef]
- Nicholson, K. Geothermal Fluids: Chemistry and Exploration Techniques, 1st ed.; Springer: Berlin/Heidelberg, Germany, 1993; pp. 19–194. [Google Scholar]
- Parkhurst, D.L.; Appelo, C.A.J. A computer program for speciation, batch−reaction, one−dimensional transport, and inverse geochemical calculations. In Water−Resources Investigations Report 99–4259; U.S. Geological Survey: Denver, CO, USA, 1999; 312p. [Google Scholar]
- Fournier, R.O. Chemical geothermometers and mixing models for geothermal systems. Geothermics 1977, 5, 41–50. [Google Scholar] [CrossRef]
- Verma, S.P.; Santoyo, E. New improved equations for, and SiO2 geothermometers by outlier detection and rejection. J. Volcanol. Geotherm. Res. 1997, 79, 9–23. [Google Scholar] [CrossRef]
- Fouillac, C.; Micard, G. Sodium/Lithium ratios in water applied to geothermometry of geothermal reservoirs. Geothermics 1981, 10, 55–70. [Google Scholar] [CrossRef]
- Fournier, R.O. Truesdel, Empirical Na−K−Ca geothermometer for natural waters. Geochim. Cosmochim. Acta 1973, 37, 1255–1275. [Google Scholar] [CrossRef]
- Chiodini, G.; Frondini, F.; Marini, L. Theoretical geothermometers and pCO2 indicators for aqueous solutions coming from hydrothermal systems of medium−low temperature hosted in carbonate−evaporite rocks. Application to the thermal springs of the Etruscan Swell. Italy. Appl. Geochem. 1995, 10, 337–346. [Google Scholar] [CrossRef]
- Giggenbach, W.F. Geothermal solute equilibria. Derivation of Na−K−Mg−Ca geoindicators. Geochim. Cosmochim. Acta 1988, 52, 2749–2765. [Google Scholar] [CrossRef]
- Giggenbach, W.F.; Goguel, R.L. Collection and analysis of geothermal and volcanic water and gas discharges. NZ DSIR Chem. Rep. 1989, 2401, 1–82. [Google Scholar]
- Arnorsson, S.; Bjarnasson, J.O.; Giroud, N.; Gunnarsson, I.; Stefansson, A. Sampling and analysis of geothermal fluids. Geofluids 2006, 6, 203–216. [Google Scholar] [CrossRef]
- Apollaro, C.; Vespasiano, G.; De Rosa, R.; Marini, L. Use of mean residence time and flowrate of thermal waters to evaluate the volume of reservoir water contributing to the natural discharge and the related geothermal reservoir volume. Application to Northern Thailand hot springs. Geothermics 2015, 58, 62–74. [Google Scholar] [CrossRef]
- Magaritz, M.; Nadler, A.; Koyumdjisky, H.; Dan, J. The use of Na−Cl ratios to trace solute sources in a semi−arid zone. Water Resour. Res. 1981, 17, 602–608. [Google Scholar] [CrossRef]
- Dixon, W.; Chiswell, B. The use of hydrochemical sections to identify recharge areas and saline intrusions in alluvial aquifers, southeast Queensland, Australia. J. Hydrol. 1992, 135, 259–274. [Google Scholar] [CrossRef]
- Lloyd, J.A.; Heathcote, J.A. Natural Inorganic Hydrochemistry in Relation to Groundwater: An Introduction; Oxford University Press: New York, NY, USA, 1985; p. 296. [Google Scholar]
- Alfaifi, H.J. Combined graphical and geostatistical technique to determine the hydrochemical processes affecting groundwater chemistry in coastal areas, Western Saudi Arabia. Arab. J. Geosci. 2019, 12, 65. [Google Scholar] [CrossRef]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater and Pollution; Balkema: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Glover, E.T.; Akiti, T.T.; Osae, S. Major ion chemistry and identification of hydro geochemical processes of groundwater in the Accra Plains Elixir. Geoscience 2012, 50, 10279–10288. [Google Scholar]
- Zhu, G.F.; Li, Z.Z.; Su, Y.H.; Ma, J.Z.; Zhang, Y.Y. Hydrogeochemical and isotope evidence of groundwater evolution and recharge in Minqin Basin, Northwest China. J. Hydrol. 2007, 33, 239–251. [Google Scholar] [CrossRef]
- Schoeller, H. Geochemistry of groundwater. In Groundwater Studies—An International Guide for Research and Practice; UNESCO: Paris, France, 1977; Chapter 15; pp. 1–18. [Google Scholar]
- Rittenhouse, G. Bromine in oil−field waters and its use in determining possibilities of origin of these waters. AAPG Bull. 1967, 51, 2430–2440. [Google Scholar]
- Stober, I.; Zhong, J.; Zhang, L.; Bucher, K. Deep hydrothermal fluid−rock interaction: The thermal springs of Da Qaidam, China. Geofluids 2016, 16, 711–728. [Google Scholar] [CrossRef]
- Truesdell, A.H.; Haizlip, J.R.; Armansson, H.; D’Amore, F. Origin and transport of chloride in superheated geothermal steam. Geothermics 1989, 18, 295–304. [Google Scholar] [CrossRef]
- Arnórsson, S.; Andresdottir, A. Processes controlling the distribution of boron and chlorine in natural waters in Iceland. Geochim. Cosmichim. Acta 1995, 59, 4125–4146. [Google Scholar] [CrossRef]
- Drüppel, K.; Stober, I.; Grimmer, J.C.; Mertz-Kraus, R. Experimental alteration of granitic rocks: Implications for the evolution of geothermal brines in the Upper Rhine Graben, Germany. Geothermics 2020, 88, 101903. [Google Scholar] [CrossRef]
- Earle, S.; Krogh, E. Elevated fluoride and boron levels in groundwater from the Nanaimo group, Vancover Island, Sea to Sky. Geotechnique 2006, 43, 1584–1591. [Google Scholar]
- Kharitonova, N.A.; Lyamina, L.A.; Chelnokov, G.A.; Bragin, I.V.; Karabtsov, A.A.; Tarasenko, I.A.; Nakamura, H.; Iwamori, H. The Chemical and Isotope Composition of Nitrogen Thermal Groundwaters of the Kuldur Spa (Jewish Autonomous Region, Russia). Mosc. Univ. Geol. Bull. 2020, 75, 621–635. [Google Scholar] [CrossRef]
- Bragin, I.V.; Zippa, E.V.; Chelnokov, G.A.; Kharitonova, N.A. Estimation of the Deep Geothermal Reservoir Temperature of the Thermal Waters of the Active Continental Margin (Okhotsk Sea coast, Far East of Asia). Water 2021, 13, 1140. [Google Scholar] [CrossRef]
- Chelnokov, G.A.; Bragin, I.V.; Kharitonova, N.A.; Aleksandrov, I.A.; Ivin, V.V.; Chelnokova, B.I. Geochemistry and Conditions of the Formation of the Ulsk Thermal Spring (Coasts of the Sea of Okhotsk, Khabarovsk Krai). Russ. J. Pac. Geol. 2019, 13, 163–175. [Google Scholar] [CrossRef]
- Jenne, E.A.; Ball, J.W.; Buchard, J.M.; Vivit, D.V.; Barks, J.H. Geochemical modeling: Apparent solubility controls of Ba, Zn, Cd, Pb. and F in waters of the Missouri Tri-State Mining Area. In Trace Substances in Environmental; Hemphill, D.D., Ed.; University of Missouri: Columbia, MO, USA, 1980; pp. 353–361. [Google Scholar]
- Langmuir, D.; Melchior, D. The geochemistry of Ca, Sr, Ba andrea sulfates in some deep brines from the Paulo Duro Basin, Texas. Geochim. Cosmochim. Acta 1985, 49, 2423–2432. [Google Scholar] [CrossRef]
- Plummer, N.; Busby, J.; Lee, R.; Hanshaw, B. Geochemical Modelling of the Madison Aquifer in Parts of Montana, Wyoming, and South Dakota. Water Resour. Res. 1990, 26, 1981–2014. [Google Scholar] [CrossRef]
- Apollaro, C.; Caracausi, A.; Paternoster, M.; Randazzo, P.; Aiuppa, A.; De Rosa, R.; Fuoco, I.; Mongelli, G.; Muto, F.; Vanni, E.; et al. Fluid geochemistry in a low-enthalpy geothermal field along a sector of southern Apennines chain (Italy). J. Geochem. Explor. 2020, 219, 106618. [Google Scholar] [CrossRef]
- Drever, J.F. The Geochemistry of Natural Waters: Englewood Cliffs; Prentice-Hall: Hoboken, NJ, USA, 1982; p. 388. [Google Scholar]
- Minissale, A.; Corti, G.; Tassi, F.; Darrahc, T.H.; Vaselli, O.; Montanari, D.; Montegrossi, G.; Yirgud, G.; Selmo, E.; Tecluf, A. Geothermal potential and origin of natural thermal fluids in the northern Lake Abaya area, Main Ethiopian Rift, East Africa. J. Volcanol. Geotherm. Res. 2017, 336, 1–18. [Google Scholar] [CrossRef]
- Truesdell, A.H.; Fournier, R.O. Procedure for estimating the temperature of a hot water component in a mixed water using a plot of dissolved silica vs. enthalpy. J. Res. US Geol. Surv. 1977, 5, 49–52. [Google Scholar]
- Arnórsson, S.; Gunnlaugsson, E.; Svavarsson, H. The chemistry of geothermal waters in Iceland III. Chemical geothermometry in geothermal investigations. Geochim. Cosmochim. Acta 1983, 47, 567–577. [Google Scholar] [CrossRef]
- Bershaw, J.; Lechler, A. The isotopic composition of meteoric water along altitudinal transects in the Tian Shan of Central Asia. Chem. Geol. 2019, 516, 68–78. [Google Scholar] [CrossRef]
- Liu, Q.; Tian, L.D.; Wang, J.L.; Wen, R.; Weng, Y.B.; Shen, Y.P.; Vladislav, M.; Kanaev, E. A study of longitudinal and altitudinal variations in surface water stable isotopes in West Pamir, Tajikistan. Atmos. Res. 2015, 153, 10–18. [Google Scholar] [CrossRef]
- Truesdell, A.H.; Frye, G.A. Isotope Geochemistry in Geothermal Reservoir Studies. In Proceedings of the SPE California Regional Meeting, Bakersfield, CA, USA, 13 April 1977. [Google Scholar]
- Taran, Y.A.; Bernard, A.; Gavilanes, J.-C.; Lunezheva, E.; Cortés, A.; Armienta, M.A. Chemistry and mineralogy of high−temperature gas discharges from Colima volcano, Mexico. Implications for magmatic gas–atmosphere interaction. J. Volcanol. Geotherm. Res. 2001, 108, 245–264. [Google Scholar] [CrossRef]
- Ohba, T.; Hirabayashi, J.; Nogami, K. D/H and 18 O/16 O Ratios of Water in the Crater lake at Kusatsu−Shirane Volcano, Japan. J. Volcanol. Geotherm. Res. 2000, 97, 329–346. [Google Scholar] [CrossRef]
- Ma, L.; Jilili, A.; Li, Y.M. Spatial differentiation in stable isotope compositions of surface waters and its environmental significance in the Issyk-Kul Lake region of Central Asia. J. Mt. Sci. 2018, 15, 254–263. [Google Scholar] [CrossRef]
- Giggenbach, W.F. The composition of gases in geothermal and volcanic systems as a function of tectonic setting. Proc. Int. Symp. Water-Rock Interact. 1992, 8, 873–878. [Google Scholar]
- Minissale, A.; Vaselli, O.; Chandrasekharam, D.; Magro, G.; Tassi, F.; Casiglia, A. Origin and evolution of ‘intracratonic’ thermal fluids from central−western peninsular India. Earth Planet. Sci. Lett. 2000, 181, 377–394. [Google Scholar] [CrossRef]
- Marty, B.; Gunnlaugsson, E.; Jambon, A.; Oskarsson, N.; Ozima, M.; Pineau, F.; Torrsander, P. Gas geochemistry of geothermal fluids at a spreading center: The Hengill area, SW-Iceland. Chem. Geol. 1991, 91, 207–225. [Google Scholar] [CrossRef]
- Hamme, R.C.; Emerson, S.R. The solubility of neon, nitrogen and argon in distilled water and seawater. Deep. Sea Res. Part I Oceanogr. Res. Pap. 2004, 51, 1517–1528. [Google Scholar] [CrossRef]
- Jenden, P.D.; Kaplan, I.R.; Poreda, R.J.; Craig, H. Origin of nitrogen−rich natural gases in the California Great Valley: Evidence for helium, carbon and nitrogen isotope ration. Geochim. Cosmochim. Acta 1988, 52, 851–861. [Google Scholar] [CrossRef]
- Ballentine, C.J.; Lollar, B.S. Regional groundwater focusing of nitrogen and noble gases into the Hugoton−Panhandle giant gas field, USA. Geochim. Cosmochim. Acta 2002, 66, 2483–2497. [Google Scholar] [CrossRef]
- Barabanov, L.N.; Disler, V.N. Nitrogenous Termae of the USSR Ministry of Health Public; Nauka: Moscow, Russia, 1968; 120p. (In Russian) [Google Scholar]
- Sharif-Zade, V.B.; Verkhovskii, A.B.; Loktev, V.A.; Markov, I.M.; Meshik, A.P.; Shukolyukov, Y.A. Isotopes of noble gases in the nitrogen thermal waters and carbon dioxide waters of the southern Pamir. Geokhimiya 1988, 8, 1187–1198. (In Russian) [Google Scholar]
- Polyak, B.G.; Prasolov, E.M.; Kamenskii, I.L.; Elmanova, N.M.; Sultankhodzhaev, A.A.; Chernov, I.G. The isotopic composition of He, Ne, and Ar in the underground fluids of the Tien Shan. Geokhimiya 1989, 1, 87–97. (In Russian) [Google Scholar]
- Polyak, B.G.; Kamenskii, I.L.; Sultankhodzhaev, A.A.; Chernov, I.G.; Barabanov, L.N.; Lisitsyn, A.N.; Khabarovskaya, M.V. Mantle-derived helium in fluids from south-eastern Tien-Shan. Dok. AN SSSR 1990, 312, 721–725. (In Russian) [Google Scholar]
- Polyak, B.G.; Prasolov, E.M.; Tolstikhin, I.N.; Yakovlev, L.E.; Ioffe, A.I.; Kikvadze, O.E.; Vereina, O.B.; Vetrina, M.A. Noble Gas Isotope Data Base. 2015. Available online: http://data.deepcarbon.net/ckan/dataset/523f4bf3−5de4−4c30−9c62−34af0dc62f70 (accessed on 2 February 2022).
- O’Nions, R.K.; Oxburgh, E.R. Helium, volatile fluxes and the development of the continental crust. Earth Planet. Sci. Lett. 1988, 90, 315–331. [Google Scholar] [CrossRef]
- Ueda, A.; Nagao, K.; Shibata, T.; Suzuki, T. Stable and noble gas isotopic study of thermal and groundwaters in northwestern Hokkaido, Japan and the occurrence of geopressured fluids. Geochem. J. 2010, 44, 545–560. [Google Scholar] [CrossRef] [Green Version]
- Álvarez-Valero, A.M.; Sumino, H.; Burgess, R.; Núñez-Guerrero, E.; Okumura, S.; Borrajo, J.; Lozano Rodríguez, J.A. Noble gas variation during partial crustal melting and magma ascent processes. Chem. Geol. 2022, 588, 120–635. [Google Scholar] [CrossRef]
- Whiticar, M.J.; Faber, E.; Whelan, J.K.; Simoneit, B.R.T. Thermogenic and bacterial hydrocarbon gases (free and sorbed) in Middle Valley, Juan de Fuca Ridge, Leg 139. In Proceedings of the Ocean Drilling Programme, Scientific Results, V. 139: College Station, TX (Ocean Drilling Program); Texas A&M University: College Station, TX, USA, 1994; pp. 467–477. [Google Scholar]
- Mook, W.G.; Bommerson, J.C.; Staverman, W.H. Carbon isotope fractionation between dis-solved bicarbonate and gaseous carbon dioxide. Earth Planet. Sci. Lett. 1974, 22, 169–176. [Google Scholar] [CrossRef]
- Giammanco, S.; Krajnc, B.; Kotnik, J.; Ogrinc, N. Temporal analysis of d13C CO2 and CO2 efflux in soil gas emissions at Mt. Etna: A new tool for volcano monitoring. Ann. Geophys. 2017, 60, S0663. [Google Scholar] [CrossRef] [Green Version]
- Cartigny, P.; Marty, B. Nitrogen Isotopes and Mantle Geodynamics: The Emergence of Life and the Atmosphere−Crust−Mantle Connection. Elements 2013, 9, 359–366. [Google Scholar] [CrossRef]
- Bebout, G.E.; Fogel, M.L. Nitrogen−isotope compositions of metasedimentary rocks in the Catalina Schist, California: Implications for metamorphic devolatilization history. Geochim. Cosmochim. Acta 1992, 56, 2839–2849. [Google Scholar] [CrossRef]
- Zhu, Y.; Shi, B.; Fang, C. The isotopic compositions of molecular nitrogen: Implications on their origins in natural gas accumulations. Chem. Geol. 2000, 164, 321–330. [Google Scholar] [CrossRef]
- Mingram, B.; Bräuer, K. Ammonium concentration and nitrogen isotope composition in metasedimentary rocks from different tectonometamorphic units of the European Variscan Belt. Geochim. Cosmochim. Acta 2001, 65, 273–287. [Google Scholar] [CrossRef]
- Agusto, M.; Tassi, F.; Caselli, A.T.; Vaselli, O.; Rouwet, D.; Capaccioni, B.; Caliro, S.; Chiodini, G.; Darrah, T. Gas geochemistry of the magmatic−hydrothermal fluid reservoir in the Copahue–Caviahue Volcanic Complex (Argentina). J. Volcanol. Geotherm. Res. 2013, 257, 44–56. [Google Scholar] [CrossRef]
- Zelenski, M.; Taran, Y. Geochemistry of volcanic and hydrothermal gases of Mutnovsky volcano, Kamchatka: Evidence for mantle, slab and atmosphere contributions to fluids of a typical arc volcano. Bull. Volcanol. 2011, 73, 373–394. [Google Scholar] [CrossRef]
- Akhmedsafin, U.M.; Sadykov, Z.S. Underground Water Flow in Kazakhstan; Nauka: Alma-Ata, Kazakhstan, 1964. (In Russian) [Google Scholar]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chelnokov, G.; Lavrushin, V.; Bragin, I.; Abdullaev, A.; Aidarkozhina, A.; Kharitonova, N. Geochemistry of Thermal and Cold Mineral Water and Gases of the Tien Shan and the Pamir. Water 2022, 14, 838. https://doi.org/10.3390/w14060838
Chelnokov G, Lavrushin V, Bragin I, Abdullaev A, Aidarkozhina A, Kharitonova N. Geochemistry of Thermal and Cold Mineral Water and Gases of the Tien Shan and the Pamir. Water. 2022; 14(6):838. https://doi.org/10.3390/w14060838
Chicago/Turabian StyleChelnokov, Georgy, Vasily Lavrushin, Ivan Bragin, Abdulaziz Abdullaev, Altyn Aidarkozhina, and Natalya Kharitonova. 2022. "Geochemistry of Thermal and Cold Mineral Water and Gases of the Tien Shan and the Pamir" Water 14, no. 6: 838. https://doi.org/10.3390/w14060838






