1. Introduction
Chromium is commonly used for the production of steel and other alloys, bricks, dyes and pigments, leather tanning, electroplating and wood preserving [
1]. It was reported that occupational exposure to trivalent chromium can lead to oxidative stress and DNA damage in tannery workers [
2]. Due to extensive use in industrial processes, large quantities of Cr compounds are discharged into the environment and contaminate the air, soil, surface water and groundwater. Chromium occurs in the environment in two oxidation states, as Cr(III) and Cr(VI) compounds, which differ in terms of chemical behavior, biological activity and toxic effects [
3]. Chromium(VI) has a mutagenic and carcinogenic effect on living organisms [
2,
3]. On the other hand, chromium(III) has been recommended for years as a trace element necessary for the proper functioning of organisms. However, recent studies indicate that chromium, currently, can only be considered as pharmacologically active and not as an essential element [
4]. Despite this, some national authorities have left the adequate intake (AI) values of Cr in their Dietary Reference Values, e.g., 35 µg/d for men and 25 µg/d for women in USA, 70 µg/d for men and 55 µg/d for women in France and 30–100 µg/d for adults in Germany [
5]. Cr(III) is widely used in dietary supplements that either support weight loss or improve glucose metabolism in type 2 diabetes [
6]. However, the safety of consuming chromium compounds in food is being disputed, as high dietary doses of Cr(III) can probably limit the absorption of iron and zinc ions, as well as affect calcium metabolism in bones. Cr(III), as a metalloestrogen, is also associated with induction of oxidative stress associated with the excessive production of reactive oxygen species which damage cell biomolecules [
2]. It has been reported that exposure to CrO
3 and CrCl
3 induce genetic mutation in cultured in vitro yeast cells and cause DNA damage within yeast cells and human leukemic T cell lymphoblast (Jurkat cells). In addition, a significantly greater ability of Cr(III) to generate DNA damage than that of Cr(VI) was observed [
7]. Due to the growing consumption of Cr(III) in the form of dietary supplements and doubts of its safety use, assessing the genotoxic risk of Cr(III) is especially important and requires further research [
2].
In natural waters, the presence and concentration ratio of Cr(III) and Cr(VI) forms depend on the following: chemical and photochemical redox reactions; precipitation/dissolution; adsorption/desorption or complexation processes, and chromium concentration, pH and redox potential [
8]. Under environmental conditions, more mobile species of Cr(VI) can undergo redox transformation and form a labile and more stable species of Cr(III), which are predominant in water. These Cr(III) aqua/hydroxo complexes have tendency to be adsorbed on naturally occurring solid material, whereas organic complexes can be immobilized by macromolecular compounds, which leads to the accumulation of Cr(III) in bottom sediments [
8,
9]. Therefore, the contamination of surface waters with chromium, mainly in the form of Cr(III), has become a global problem. It was found that Cr(III) may have an adverse effect on freshwater fish [
10], meaning its determination in the aquatic environment is important for assessing the risk to human health. According to the European Council Directive 98/83/EC, the maximum concentration of total chromium in drinking water intended for human consumption cannot exceed 50 µg/L [
11].
The determination of chromium species in water samples requires reliable and accurate analytical procedures. Due to the high toxicity of Cr(VI), most researchers are focused on the development of analytical methods for its selective determination in various samples [
12,
13]. Additionally, selective determination of Cr(III) is relevant because of its recently reported adverse effects on living organisms [
2,
10]. Despite recent advances in instrumental analysis, direct determination of chromium species, especially Cr(III), in environmental water samples is still difficult. This is due to the insufficient sensitivity of instrumental techniques and low concentrations of analytes in complex matrices, which can be often a source of interference during measurements. Therefore, special sample preparation methods that allow for the separation/preconcentration of various forms of chromium and for the removal of interfering substances are needed [
14,
15]. In recent years, solid phase extraction (SPE) has become one of the most popular techniques for the separation of chromium species, as it can be easily coupled with electrothermal atomic absorption spectrometry (ETAAS), inductively coupled plasma mass spectrometry (ICP-MS) or inductively coupled plasma optical emission spectrometry (ICP-OES). Although commercially available materials are often used as solid sorbents, there has been an increasing interest in recent years to design special, new materials that are characterized by high selectivity for a given analyte, e.g., functionalized graphene and carbon nanotubes [
16], nanocomposites [
17,
18], nanoparticles [
15,
19] or ion imprinted polymers [
20,
21,
22,
23,
24,
25,
26,
27,
28,
29,
30,
31,
32,
33].
Ion imprinted polymers (IIP) are prepared based on the concept of molecular imprinting, a modern method of synthesizing sorbents with desired properties [
20,
21,
22]. IIP synthesis consists of immobilizing the matrix molecule, i.e., a complex of a metal ion with an appropriate ligand, in the polymer structure through a copolymerization reaction with functional and cross-linking monomers. The physicochemical properties of IIPs, their binding properties and their ability of selective recognition largely depend on both the specific interactions of the ligand with metal ions and the reagents used in the polymerization process. The advantages of IIPs are their specific affinity for a given ion, their stability and reusability, and their relative ease and low cost of preparation.
A review of the literature has shown that Cr(III)- ion imprinted polymers were prepared using Cr(III) complexes with 2-methacryloylamidohistidine (MAH) [
23], nicotinic acid [
24], 8-hydroxyquinoline [
25], 1,5-diphenylcarbazide [
26] and ammonium pyrrolidinedithiocarbamate [
27] as templates. The polymeric structure was mainly formed by the cross-linking of methacrylic acid [
26], acrylamide [
24,
27] with ethylene glycol dimethacrylate [
24,
27] or divinylbenzene [
25]. The sorption properties of the prepared IIPs were tested in model solutions [
23,
29], and a few of them were used for the analysis of chromium species in various environmental water samples [
23,
24,
25,
27,
28,
30,
33] [(
Table 1).
The prepared Cr(III)-IIPs were used, mostly in a static mode, as solid sorbents in SPE due to the slow kinetics of analyte sorption and desorption process [
23,
29,
31,
32,
33]. For example, sorption of Cr(III) from a solution of pH 6 on the Cr(III)-MAH-EGDMA polymer occurred within 30 min, whereas its desorption with 0.1 mol/L succinic acid lasted 2 h [
23]. The Cr(III)-IIPs, characterized by faster sorption kinetics, were applied in a dynamic mode [
24,
25,
26,
27], which allowed for the minimization of sample/reagent consumption and waste production. This reduced the risk of sample contamination as well as increased the sampling frequency [
25]. Most of the described Cr(III)-IIPs showed good selectivity towards Cr(III) ions in the presence of foreign ions as well as Cr(VI) ions. This, therefore, allowed for their use in the speciation analysis of chromium in wastewater [
24,
25,
31,
32] and surface water samples [
27,
28,
30,
33]. However, the Cr(III)-MAH-EGDMA polymer [
23] and silica imprinted with Cr(III)-AAPTS [
30], which had been synthesized in the presence of Cr(III) complexes, simultaneously retained both forms of chromium. As the successful separation of chromium species strongly depends on the properties of the sorbent used, research on the development of new selective and efficient sorbents is still needed.
The aim of this work was to develop a new dynamic SPE procedure for selective separation and determination of Cr(III) ions in water samples. We paid special attention to the design and preparation of solid sorbents characterized by good sorption properties including high efficiency and fast kinetics of the sorption and desorption process of the analyte, and versatile in practice and application. Based on our previous experience [
34], we have prepared two ion imprinted polymers based on the Cr(III)-1,10-phenanthroline complex, containing aromatic rings that can be easily arranged in the polymer matrix. For the synthesis of both IIPs, neutral functional monomer—styrene, aprotic solvent—acetonitrile, divinylbenzene as a cross-linking monomer and AIBN as an initiator were used in bulk polymerization. This resulted in the first polymer, later referred to as Cr(III)-phen-ST. For the second polymer obtained, referred to as Cr(III)-phen-ST-4VP, an additional functional monomer of 4-vinylpyridine (4VP) was applied. The physicochemical properties of both solid sorbents were evaluated and compared. The ability of these IIPs to separate and determine Cr(III) from water samples was assessed.
2. Materials and Methods
2.1. Instrumentation
For the determination of chromium, an atomic absorption spectrometer with electrothermal atomization and Zeeman background correction system Solaar M6 (Thermo Electron Corporation, Cambridge, UK) was used. The measurements of chromium were performed at wavelength λ = 357.9 nm with a 0.5 nm spectral bandpass using a chromium hollow cathode lamp (Photron, Narre Warren, Australia) operating at a 5.4 mA current. Integrated absorbances were measured using standard pyrolytically coated graphite tubes with the use of the following optimized time/temperature furnace program: drying at 110 °C for 20 s, ashing at 1200 °C for 8 s, and atomization at 2600 °C for 3 s. Sample aliquots of 10 μL volumes were dispensed from autosampler cups into the graphite furnace.
The FT-IR absorption spectra of polymers (4000–500 cm−1) in KBr were recorded using the Nicolet FT-IR 6700 spectrometer (Thermo Scientific, Newington, CT, USA). Surface areas of the polymers were measured using the surface area analyzer Gemini VII 2390 (Micrometrics, Norcross, GA, USA). Nitrogen sorption analysis was carried out using approximately 0.3 g portions of polymers degassed for 24 h at 80 °C. A scanning electron microscope SEM Inspect (FEI, Hillsboro, OR, USA) was used to study the morphology of polymer particles.
A solid phase extraction (SPE) system consisting of laboratory made glass columns (i.d. 3.15 mm) filled with 0.1 g of the polymer, PTFE tubing of i.d. 0.8 mm and a peristaltic pump Minipuls 3 (Gilson, Villiers-le Bel, France) was used. The two ends of the columns were blocked with PTFE membranes.
For pH measurements, an inoLab pH Level 1 (WTW, Weiheim, Germany) pH meter equipped with an electrode SenTix 21 (WTW, Weiheim, Germany) was used.
2.2. Reagents and Materials
A stock solution (20 mg/mL) of Cr(III) as CrCl3 was prepared from Titrisol solution (Merck, Darmstadt, Germany). A stock solution (1.001 mg/mL) of Cr(VI) as K2Cr2O7 (Sigma Aldrich, Steinheim, Germany) was used. Working standard solutions of chromium were prepared daily by diluting the stock solutions. Sodium hydroxide (p.a.) supplied by POCh (Gliwice, Poland) was used to adjust the pH of the samples and standards. Acetic acid (80%, POCh, Gliwice, Poland) and ethylenediaminetetraacetic acid (EDTA) (POCh, Gliwice, Poland) were used as desorption agents. 1, 10-phenanthroline (phen), supplied by Sigma Aldrich (Steinheim, Germany), was used to prepare the Cr(III)-1,10-phenanthroline complex. Nitric acid (69.5%, Trace Select, Fluka, Seelze, Germany) and hydrochloric acid (37%, fuming, Trace Select, Fluka, Seelze, Germany) were used for digestion of the samples. Styrene (ST, 99%, Merck, Darmstadt, Germany), 4-vinylpyridine (4VP, Sigma Aldrich, Steinheim, Germany), divinylbenzene (DVB, 80%, mixture of isomers, Merck, Darmstadt, Germany), 2, 2′-azobisisobutyronitrile (AIBN, Fluka, Steinheim, Germany) and acetonitrile (Sigma Aldrich, Steinheim, Germany) were used for the synthesis of the ion-imprinted polymers. High-purity argon was used to de-aerate the solutions. For interference studies, solutions of copper(II) nitrate(V), manganese(II) nitrate(V) and iron(III) nitrate(V) (all supplied by Fluka, Steinheim, Germany) were used. Deionized water with 18.2 MΩ/cm conductivity obtained from the Milli-Q water purification system (Millipore, Burlington, VT, USA) was used to prepare all of the solutions.
Standard reference material of surface water SRM 1643e (NIST, Gaithersburg, MD, USA) was used for accuracy control.
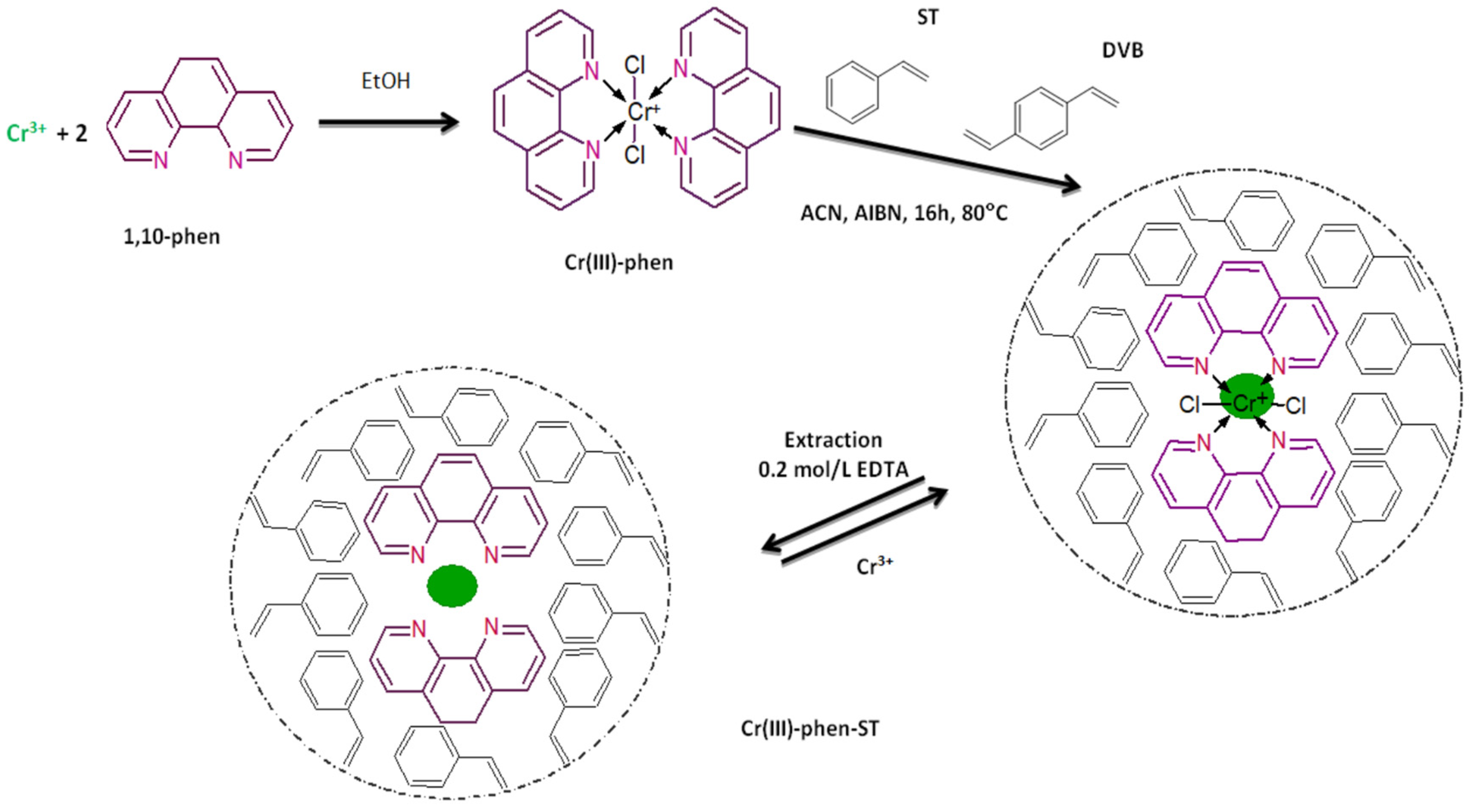
2.3. Preparation of Ion Imprinted Polymers
The IIPs were prepared in two steps. First, the binary complex of Cr(III) with 1,10-phenanthroline (template) was formed. Then, the copolymerization of this complex was performed with suitable reagents using the bulk method. The complex of
cis-dichloridobis(1, 10-phenanthroline)chromium(III) chloride (Cr(III)Cl
2(phen)
2+Cl
−), designated here as Cr(III)-phen, was prepared according to the procedure described in the work of Gao [
35]. A Cr(III)-phen complex, containing 0.002 g (0.04 mmol) of Cr(III) was used for the synthesis of the IIPs. The Cr(III)-phen-ST polymer was prepared using styrene (2.3 mL, 20 mmol), divinylbenzene (11.2 mL, 80 mmol), 2, 2′-azobisisobutyronitrile (0.050 g) and acetonitrile (4 mL), whereas for the Cr(III)-phen-ST-4VP polymer, an addition of 4VP (1.1 mL, 10 mmol) was included. In summary, all reagents were mixed in glass polymerization ampoules and purged for 10 min with argon. Next, they were left for 16 h at 80 °C. Monolithic blocks of polymers were crushed and washed with dichloromethane, and then dried and sieved. Control polymers (CP) were synthesized in a similar way but without Cr(III) ions. The schematic illustration of bulk polymer preparation is presented in
Figure 1. The efficiency of Cr(III) imprinting in the polymeric structure was evaluated as the ratio of the Cr content in IIPs determined after digestion of the polymer in a microwave oven (0.1 g IIP + 5 mL of HNO
3 + 1 mL of HCl) to the initial mass of Cr(III) used for polymers’ synthesis.
The imprinted Cr(III) ions were leached from IIPs by passing 100 mL of 0.5 mol/L CH3COOH and 100 mL of 0.2 mol/L EDTA solutions through column filled with portions of the polymers (0.5 g) at a flow rate of 0.5 mL/ min. The total amount of removed Cr(III) ions was evaluated by analyzing effluents from the IIPs. The efficiency of Cr(III) ions removal was calculated as the ratio of the total amount of leached Cr(III) ions to the amount of Cr imprinted in IIPs. In the final step, the polymers were rinsed with deionized water and dried at room temperature. A fraction 120–200 µm in diameter was used as a column filling.
The FT-IR spectra of the Cr(III)-phen-ST and the Cr(III)-phen-ST-4VP polymers (
Figure S1) were registered and the following characteristic bands were observed:—ν(=C-H) band at 3100–2849 cm
−1,—ν(-C = C) band at 1600–1510 cm
−1,—ν(-C-H) band at 1487—1446 cm
−1 derived from the polymer matrix,—ν(-C = N) band at 1635–1629 cm
−1 and—ν(-C-N) band at 1350–1150 cm
−1 from Cr(III)-phen complex. In the spectrum of the Cr(III)-phen-ST-4VP polymer, the last two characteristic bands have a higher intensity as they can also originate from 4-vinylpyridine groups. The CPs have similar FT-IR spectra as the IIPs, thus indicating a similarity in their backbone structure. Some differences in the fingerprint region were observed.
2.4. Procedures
For Cr(III) adsorption, IIPs were conditioned by passing 1 mL of deionized water through glass columns packed with 0.1 g of the sorbents at a flow rate of 1 mL/min. For retention of the analyte, either the Cr(III) standards or sample solutions were adjusted to pH 4.5 with NaOH solution and then were passed through the columns at a flow rate of 1 mL/min. In order to remove Cr(VI) ions and retained matrix components, the columns were rinsed with 2 mL of deionized water before the elution step. Elution of Cr(III) ions was performed with 1 mL of 0.1 mol/L EDTA at a flow rate of 1 mL/min. The concentration of chromium in all solutions was determined using the ETAAS technique. All results are based on at least three parallel replications.
The retention efficiency was calculated as the ratio of the difference between the initial mass of Cr(III) in the solution loaded on the column and the mass of Cr(III) in the effluent, derived from the calibration graph, to the initial mass of Cr(III) in the solution loaded on the column. The recovery of Cr(III) was calculated as the ratio of analyte mass in the eluate to the initial mass of Cr(III) in the loaded solution. The concentration of Cr(III) in the eluate was calculated from the calibration graph obtained after separation of Cr(III) on the IIPs column by developed procedure.
In order to study the selectivity of IIPs towards Cr(III) ions in the presence of Cu(II), Mn(II) and Fe(III) ions, solutions containing equivalent amounts of Cr(III) and competitive metal (Me) ions (10 ng/mL) were passed through columns filled with IIPs. Then, the columns were rinsed with 2 mL of water and Cr(III) was eluted with 1 mL of a 0.1 mol L
−1 EDTA solution. The concentrations of Cr(III) and other metal ions were measured in both the column effluent and the eluate. The distribution coefficient (D), selectivity coefficient (α) and relative selectivity coefficient (α’) were calculated according to the following equations:
where C
0 and C
f are the initial and final concentrations of Cr(III) or competitive Me ions in the solution, [ng/mL, V—volume of the solution, mL, and m—mass of sorbent, g.
The sorption capacity of the IIPs was determined by passing an aqueous Cr(III) solution (10 ng/mL) through columns packed with 0.1 g of sorbents and quantifying the content of chromium in each of the 0.3 mL effluent fractions using ETAAS. The experiment was completed when the efficiency of the Cr(III) retention decreased by 20%.
2.5. Sample Preparation
Samples of tap water were filtered through a cation-exchange membrane Neosepta (Astom Corporation, Japan), spiked with Cr(III) ions at concentration of 10 ng/mL and left for equilibration. Before the analysis, samples were adjusted to pH 4.5 using NaOH and subjected to the IIPs’ analyte separation procedures.
Green tea infusion was obtained by immersing the teabag in 200 mL of Milli Q water or tap water at 80 °C for 5 min. Then, the infusion was cooled, spiked with 10 ng/mL of Cr(III) ions and left for equilibration. After adjusting to pH 4.5, the samples were subjected to the analyte separation procedures on the IIPs.
3. Results and Discussion
New solid sorbents—ion imprinted polymers were designed and prepared for selective separation process of Cr(III) ions from water to determine the analyte by ETAAS. For this purpose Cr(III)-phen complex, in which the Cr(III) ion is bonded via a nitrogen donor atom, was imprinted into the polymeric structure through the trapping method using styrene, the neutral functional monomer. As the template molecule was physically immobilized in the polymeric structure, there was a risk of leaching out of the complex during the washing step, which could affect the stability of the binding cavities. Therefore, for preparation of the second polymer, the additional functional monomer, namely 4-vinylpyridine (4VP), was introduced to the reaction mixture along with styrene. It was expected that as bifunctional monomer, 4VP was capable of interacting with the 1,10-phenentroline ligand’s aromatic rings to form π-π bonds with the polymer matrix. This would allow for the stabilization the specific binding sites and influence the selectivity of prepared polymer. The physicochemical properties of both IIPs were compared and their suitability as SPE material was assessed.
3.1. The Physicochemical Characteristics of Cr(III) Ion Imprinted Polymers
The physicochemical properties of the sorbent determine the possibility of its multiuse potential in analyte sorption/desorption cycles in dynamic systems. Sorbents used as a column fillings in the SPE technique should have the appropriate particle size, porosity, mechanical stability and chemical resistance. The particle size affects the degree of sorbent packing in the column and forces the reagent flow rate thus impacting the analysis time. The sorbent porosity determines the specific surface area and the availability of binding sites for the analyte. The stability of sorbents and their resistance to chemical reagents affects the cost and time of analysis.
The particles of IIPs were characterized using scanning electron microscopy (SEM). The particles displayed both irregular shapes and various sizes (120–200 µm) and were very hard and, therefore, difficult to crush. The particle surfaces of both IIPs were very smooth (
Figure S2). The surface areas of the imprinted and control polymers were derived from adsorption isotherms using the BET (Brunauer, Emmett and Teller) method (
Table S1). The BET surface area of the Cr(III)-phen-ST polymer was 365 m
2/g, which is twice as high as that of the Cr(III)-phen-ST-4VP polymer, 177 m
2/g. The pore size of the Cr(III)-phen-ST polymer obtained using the BET method was 1.29 nm, whereas the pore size of the Cr(III)-phen-ST-4VP polymer was lower, 1.21 nm. The values of pore size obtained using BJH (Barrett-Joyner-Halenda) method for IIPs were similar: 2.12 nm and 2.14 nm, respectively. Therefore, both polymers can be considered as microporous materials. However, the pore volume of the Cr(III)-phen-ST-4VP polymer, equal to 0.091 cm
3/g, was 2.6-times lower than that of the Cr(III)-phen-ST polymer, 0.241 cm
3/g. It indicates that introduction of 4VP, an additional functional monomer, generates a steric barrier, resulting in both greater space congestion and denser sorbent packing.
The Cr(III) ions were efficiently imprinted into the structure of polymers. About 80% of Cr(III) ions were imprinted in the Cr(III)-phen-ST polymer, whereas 70% were imprinted in the Cr(III)-phen-ST-4VP polymer (
Table S1). However, the amount of imprinted Cr(III) ions removed from IIPs during the washing step was very small, as only 6% and 1% of the entrapped Cr(III) ions were leached from the Cr(III)-phen-ST and the Cr(III)-phen-ST-4VP polymers, respectively. The significant difference in the leaching of Cr(III) ions indicates the stronger binding of the template (Cr(III)-phen complex) in the polymer structure when 4VP is present, mostly likely due to its interaction with the ligand aromatic rings. Moreover, such low efficiency removal of the imprinted Cr(III) ions may result in the limited availability of active sites of the polymer for the analyte during the sorption step.
3.2. The Characteristic of Sorption Properties of Ion Imprinted Polymers
Sorption of Cr(III) on a solid sorbent results from the combined effect of the chemical form of the analyte in the sample and of the strength of its interaction with the sorbent’s active sites. In order to test the efficiency of separation of Cr(III) ions on IIPs the retention and elution conditions of the analyte, i.e., the sorbent conditioning procedure, pH and flow rate of sample, type of eluent, its concentration and flow rate were optimized. Because both IIPs tested have the same template, the same chemical interactions should be responsible for the retention of the analyte. For this reason, the optimization of the analyte separation conditions was performed only on the Cr(III)-phen-ST polymer. Subsequently, the separation of Cr(III) ions on the Cr(III)-phen-ST-4VP polymer was checked under the previously established optimal conditions. The selectivity of the polymers towards Cr(III) ions in the presence of Cr(VI) and other metal ions coexisting in water as well as the sorption capacity and stability of both tested IIPs were also assessed.
3.2.1. Optimization of Separation Procedure of Cr(III) Ions on IIPs
Optimization of Cr(III) Retention Conditions
The pH of a sample has the greatest influence on the amount of the analyte retained on the IIPs. This is related to the presence of various forms of Cr(III) in the water environment. In aqueous solutions at a pH < 4, Cr(III) is present in the form of a kinetically inert aqua-complexes Cr(H
2O)
63+. When the pH increases to pH 6, coordinated water molecules are replaced by more reactive hydroxyl groups, forming more labile neutral or cationic complexes, i.e., Cr(OH)
3aq, Cr(H
2O)
5OH
2+, Cr(H
2O)(OH)
2+ or Cr(H
2O)(OH)
2+. In the neutral and slightly alkaline environment, Cr(III) precipitates in the form Cr(OH)
3(s), while at a pH > 9, the precipitate is transformed into the soluble Cr(OH)
4− complex [
36].
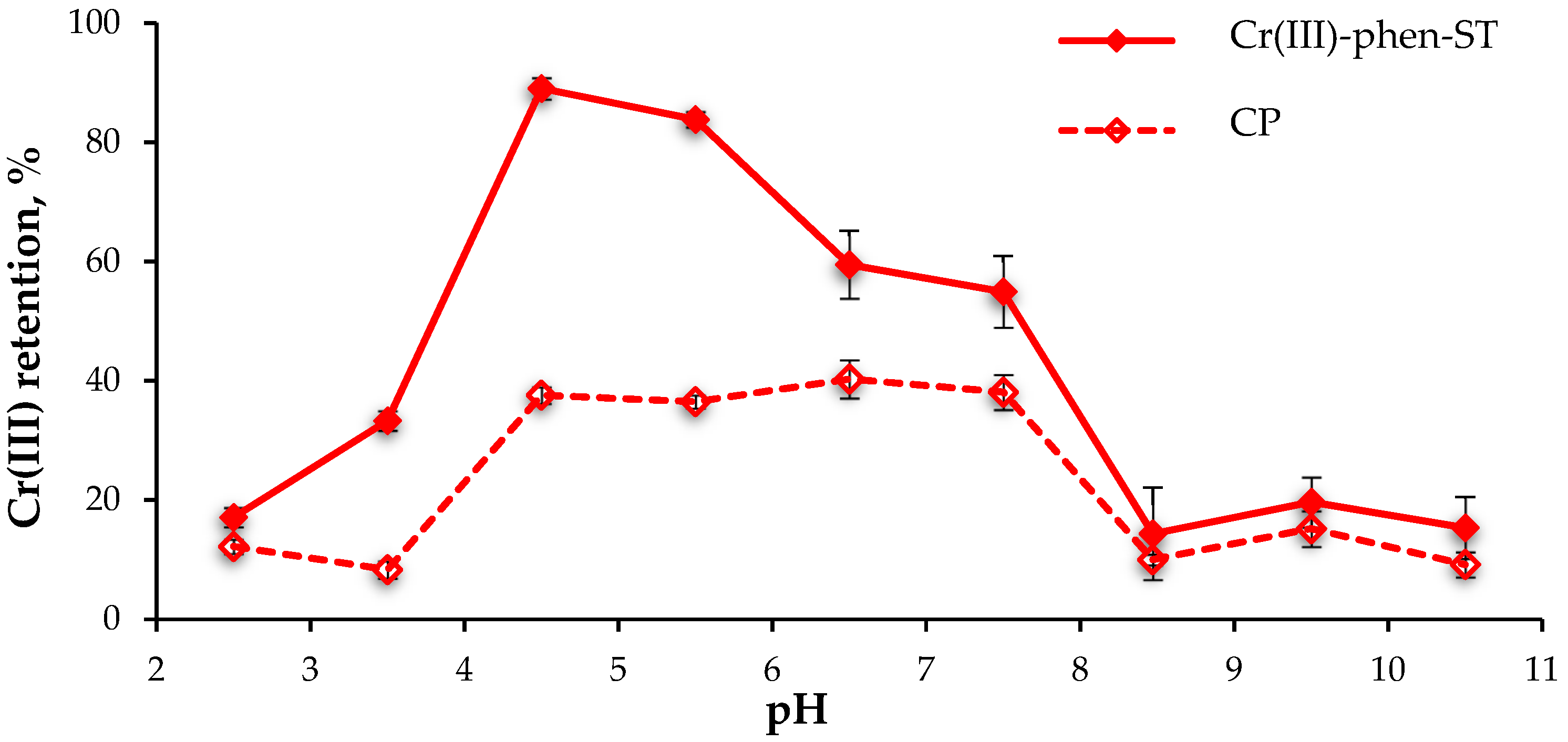
The effect of the pH of a sample on the retention of analyte (10 ng of Cr(III)) on the Cr(III)-phen-ST polymer was tested in the pH range from 2.5 to 10.5 (adjusted with HCl or NaOH solution) at a flow rate of 1 mL/min. The absorbance of chromium was measured in the column effluents using the ETAAS technique, and based on this, the efficiency of its sorption was calculated (
Figure 2). The sorption of Cr(VI) ions on the tested sorbent was also studied in order to evaluate its selectivity towards Cr(III) ions. The study of the retention of Cr (III) and Cr (VI) ions was also carried out on the control polymer.
The highest retention of Cr(III) ions on the Cr(III)-phen-ST polymer occurred in the solutions of pH 4.5 (89.1 ± 1.8%, n = 3). This may indicate that Cr(III) ions react with the ligand imprinted into the structure of the polymer under conditions similar to those that form the Cr(III)-phen complex in ethanol. The lower efficiency of Cr(III) sorption from solutions at a pH < 4.5 probably results from the protonation of the nitrogen atom present in the 1,10-phenantroline ligand, which weakened the interaction of active polymer sites with the cationic form of Cr(III). A significant decrease of analyte retention from solutions with a pH > 7.5 was caused by the lack of interactions between precipitated Cr(OH)3 and the ligand imprinted into polymeric structure. For further studies, pH 4.5 was chosen as the optimal sample solution pH. Under such conditions, the retention of analyte on the Cr(III)-phen-ST-4VP polymer, prepared with the additional functional monomer, was also high (87.8 ± 1.3%, n = 3).
The retention of the analyte on the control polymers was significantly lower and measured 37.6 ± 1.4%, n = 3 on CP-ST and 30.3 ± 1.4%, n = 3 on CP-ST-4VP. This indicates a good imprinting effect of both of the polymers prepared with Cr(III)-phen complex.
The retention of Cr(VI) from the solution of pH 4.5 was only 12.3 ± 0.6% (n = 3) on the Cr(III)-phen-ST polymer and 16.7 ± 0.9% (n = 3) on the Cr(III)-fen-ST-4VP polymer, respectively. This demonstrates high selectivity of synthesized sorbents towards the Cr(III) ions. It was also observed that more than 90% of the Cr(VI), which had been retained on the IIPs, was removed with 2 mL of deionized water. This indicates nonspecific sorption of these ions on the sorbent. The losses of Cr(III) ions sorbed on IIP during the rinsing of the column with deionized water were negligible (<2%).
The method of conditioning the solid sorbent also affects the efficiency of Cr(III) retention. Most often for conditioning, the solution in which the sample is prepared is recommended. In this work we checked the effect of the Cr(III)-phen-ST polymer conditioning with deionized water, deionized water of pH 4.5 (HCl) and 0.1 mol/L acetate buffer of the pH 4.5. For this purpose, 1 mL of conditioning solution at a flow rate of 1 mL/min was passed through the polymer columns, followed by 1 mL of 10 ng/mL Cr(III) standard solution. The highest efficiency of Cr(III) retention, equal to 86.7 ± 2.9% (n = 3), was obtained on the Cr(III)-phen-ST polymer conditioned with deionized water. The same analyte retention efficiency was obtained on sorbent conditioning with deionized water at pH 4.5, but, in that case, the repeatability was lower (86.6 ± 5.7%, n = 3). When a 0.1 mol/L acetate buffer solution was used, the Cr(III) retention on the sorbent bed decreased by 25%. This could be due to the presence of acetate ions in the column, which compete in the interaction between Cr(III) ions and the active sites of sorbents. In further studies, the columns were conditioned with 1 mL of deionized water.
The influence of sample flow rate on the retention of Cr(III) on the Cr(III)-phen-ST polymer was investigated in a range from 0.25 to 2.0 mL/min. The highest Cr(III) retention (90%) was observed at the sample flow rate of 1.0 mL/min. The increase of a sample flow rate to 1.5 mL/min and 2 mL/min caused a decrease of Cr(III) retention by a maximum of 9%, whereas reduction of a flow rate did not influence the retention of analyte. The optimal flow rates of samples on the Cr(III)-phen-ST and the Cr(III)-phen-ST-4VP polymers was assumed to be 1.0 mL/min.
Optimization of Elution of Cr(III) Conditions
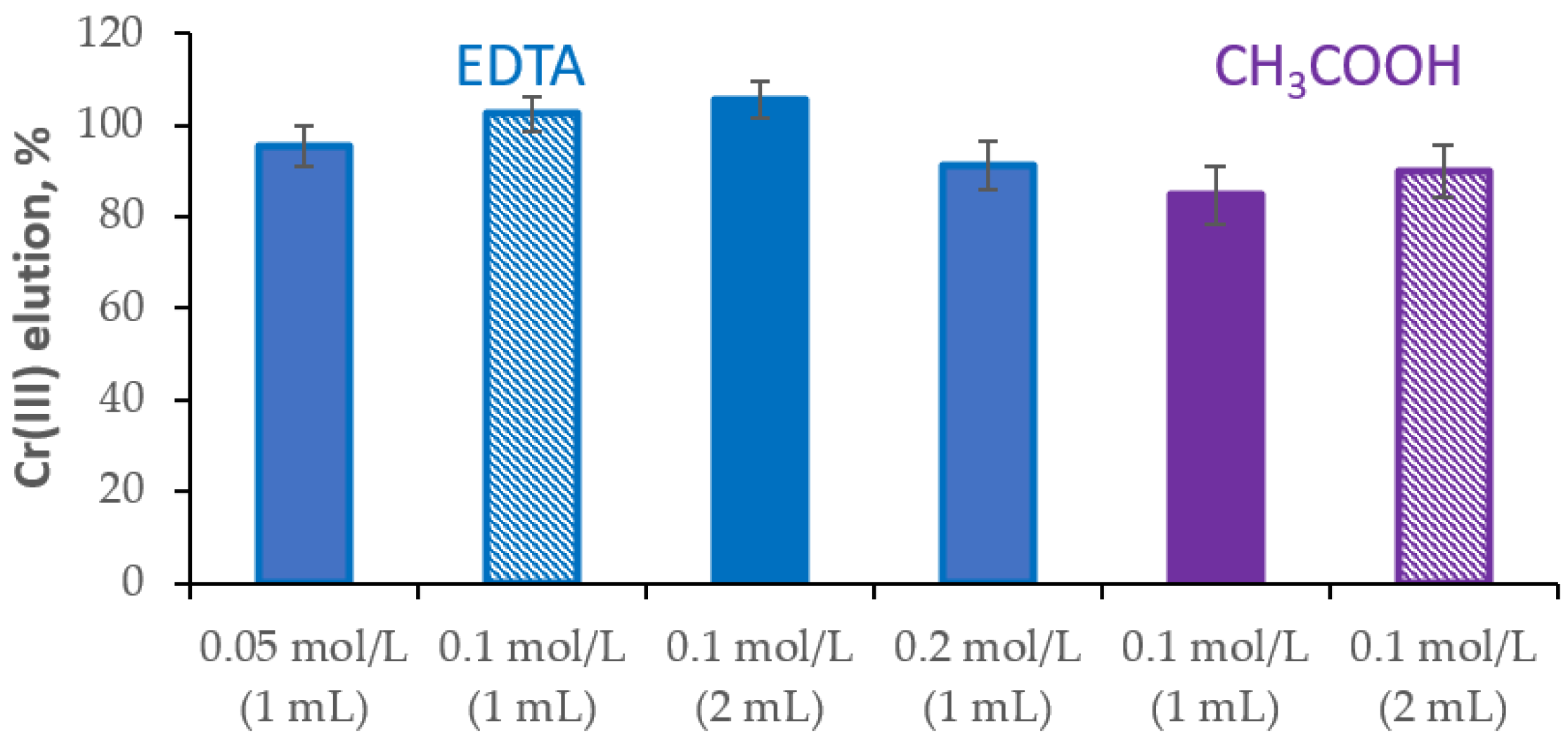
The efficient elution of the analyte retained on the IIP polymer enables multiple uses. Therefore, it is essential to find a suitable eluent that will ensure a quantitative elution of Cr(III) ions and will not affect the properties of the sorbent. In order to optimize the Cr(III) elution conditions, the eluent solutions of various concentrations, volumes and flow rates were tested.
Reagents’ complexing Cr(III) ions, namely EDTA (log β
1 = 23.4) and acetic acid (log β
1 = 4.63), were tested as eluents. For elution of the analyte retained on the Cr(III)-phen-ST polymer, either 1 mL and 2 mL of EDTA solution at concentration of 0.05–0.2 mol/L or CH
3COOH solution at concentration of 0.1 mol/L was used. It was observed that the elution of Cr(III) ions with 1 mL of the 0.1 mol/L EDTA solution was 102.5 ± 3.9% (n = 3), while the elution with 2 mL of the 0.1 mol/L CH
3COOH solution was 90.0 ± 5.7% (n = 3) (
Figure 3).
Increasing the EDTA concentration to 0.2 mol/L adversely affected the elution of Cr(III) from the sorbent, reducing it by 11%. The use of a smaller volume of CH3COOH reduced the efficiency of analyte elution by 5%. Therefore, 1 mL of the 0.1 mol/L EDTA solution was selected for further studies, as it resulted in the optimal elution of Cr(III) ions from the Cr(III)-phen-ST polymer.
The influence of the eluent flow rate on the elution efficiency of Cr(III) from the Cr(III)-phen-ST polymer was investigated in a range from 0.25 to 2.0 mL/min. It was found that the highest analyte elution (96.6 ± 1.3%, n = 3) was obtained using the eluent flow rate of 1.0 mL/min. Therefore, it was selected as the optimal rate. The increase of the flow rate up to 2 mL/min reduced the elution of Cr(III) ions by 15%. The use of a lower flow rate did not significantly influence the elution efficiency of Cr(III) from IIP. Under optimal conditions, the elution efficiency of the analyte from the Cr(III)-phen-ST-4VP polymers was equal to 93.9 ± 3.4% (n = 3).
3.2.2. Selectivity of IIPs towards Cr(III) Ions
The selectivity of the sorbent determines the sorbent’s ability to retain one form of chromium (analyte) in the presence of other ions, also Cr(VI), present in the sample. This can be expressed as a selectivity coefficient (α) and relative selectivity coefficient (α’) (for definitions see
Section 2.4). The selectivity of tested IIPs towards Cr(III) ions was studied in the presence of Cu(II), Mn(II) and Fe(III) ions, which have an ionic radius similar to Cr(III) and therefore can compete with Cr(III) ions in the formation of complexes with the 1,10-phenenthroline ligand imprinted into the polymer structure. It was observed that the Cr(III)-phen-ST polymer showed good selectivity towards Cr(III) ions in the presence of Cu(II) and Fe(III) ions, but lower selectivity in the presence of Mn(II) ions (
Table 2). The Cr(III)-phen-ST-4VP polymer turned out to be more selective for Cr(III) ions. The selectivity coefficients α of the Cr(III)-phen-ST-4VP polymer in the presence of Cu(II), Fe(III) and Mn(II) ions increased by 1.3 times, 1.6 times and 40 times, respectively. The introduction of 4VP, an additional functional monomer, resulted both in better entrapment of the complex in the polymer structure and in the formation of more selective binding sites. The smaller BET surface area and pore volume of this polymer indicate a lower availability of active sites for other metal ions; therefore, only the analyte ions can be bound at these sites.
The relative selectivity coefficients showed that the Cr(III)-phen-ST (α’ = 1.2–9.0) and the Cr(III)-phen-ST-4VP (α’ = 10.2–26.2) polymers were more selective towards Cr(III) ions with respect to control polymers. The obtained values of selectivity coefficients (α, α’) for Cr(III) were similar to these reported for Cr(III)-IIPs in the literature (see
Table 1) [
23,
24,
32].
3.2.3. Sorption Capacity and Stability of IIPs
Due to the physicochemical properties of IIPs, each portion of the polymers required up to 15 cycles of preliminary conditioning in order to activate the binding sites in the polymer structure and to achieve the appropriate sorption properties.
The sorption capacity obtained for the Cr(III)-phen-ST polymer was 1.18 µg/g. The introduction of 4VP, an additional functional monomer, decreased the sorption capacity of the Cr(III)-phen-ST-4VP polymer three fold (to 0.41 µg/g). As mentioned above (
Section 2.3), the lower removal efficiency of imprinted Cr(III) ions from the polymer in the washing step led to the formation of a porous structure with a low pore volume. This, in turn, caused limited availability of active sites for analyte sorption. The sorption capacities obtained for the tested IIPs were low but were comparable with other IIPs used for the separation and determination of trace amounts of Cr(III) by the ETAAS technique [
27].
The stability and reusability of the sorbents were studied in repeated sorption and desorption cycles performed at optimal conditions. On the Cr(III)-phen-ST polymer, the retention (86.5 ± 3.3%) and recovery (85.6 ± 4.0%) of the analyte remained constant for up to 107 successive separation cycles. This indicates the high stability of that polymer and its possibility for multiple uses. The Cr(III)-phen-ST-4VP polymer can be used continuously for 75 cycles without losing its sorption properties, as the average retention of Cr(III) during that time was 83.9 ± 4.7%. The reproducibility of analyte retention rate during the stability studies of IIPs, expressed as the RSD (relative standard deviation), ranged from 3.8% to 5.6%. In respect to stability, the tested IIPs were superior to other Cr-imprinted polymers, which retained their sorption/desorption properties for only five or ten cycles (see
Table 1) [
23,
33]. However, the Cr(III)-IIPs previously prepared in our group are characterized by similar [
24] or better stability [
25,
27] to the tested polymers (see
Table 1). The lower stability of the Cr(III)-phen-ST-4VP polymer as compared to the Cr(III)-phen-ST polymer was likely caused by the presence of lower amounts of initial active sites in the polymeric structure due to weaker leaching of the imprinted ions during the washing step (
Table S1). The elution of Cr(III) ions with the eluent solution in subsequent cycles led to the removal of the ligand from the polymeric structure and loss of its sorption properties. Thus, the stronger binding of the ligand to the polymer structure affects the stability of the polymer, while the weaker interaction of the imprinted ions with the ligand allows for multiple retention/elution of the analyte.
3.3. Analytical Performance of Methods
The analytical performance of the developed methods, based on the separation of Cr(III) ions on the Cr(III)-phen-ST and the Cr(III)-phen-ST-4VP polymers prior to determination of Cr by ETAAS, was evaluated in optimized experimental conditions. The separation of Cr(III) ions on both polymers was characterized by high retention efficiency and recovery of the analyte (
Table 3). The repeatability of the Cr(III) ions’ separation on the IIPs for six subsequent cycles was better than 3.6%, expressed as the RSD of the analyte recovery.
Calibration graphs were prepared by passing standard Cr(III) solutions of different concentrations through columns filled with IIPs under optimal conditions. It was found that calibration graphs of Cr(III) were linear in the concentration range from 0.5 ng/mL to 20 ng/mL given the following regression equation: y = 0.0153x + 0.0251, R
2 = 0.9992 and y = 0.0104x + 0.0304, R
2 = 0.9989. The limits of detection (LOD) and quantification (LOQ) of the methods were calculated according to IUPAC recommendation [
37] as LOD = 3 SD
blank/a, and LOQ = 10 SD
blank/a, where SD
blank is the standard deviation of the absorbance of blank sample, while
a is the slope of the calibration graph. The values of LOD: 0.35 ng/mL and 0.47 ng/mL, and LOQ: 1.18 ng/mL and 1.55 ng/mL obtained using the Cr(III)-phen-ST and the Cr(III)-phen-ST-4VP polymers, respectively.
The ability of the IIPs to preconcentrate Cr(III) (1 ng/mL) from various sample volumes (1–20 mL) was also studied (
Table 4). It was found that for 20 mL of sample, the recovery of analyte on the Cr(III)-phen-ST polymer was 87.4 ± 4.3% (n = 5), while on the Cr(III)-phen-ST-4VP polymer it was 84.8 ± 1.4% (n = 5). This results in a 20-fold preconcentration of Cr(III) ions and decreases the LOD of the method to 0.018 ng/mL and 0.024 ng/mL, respectively. The obtained values of LOD were comparable to those found in literature, when ETAAS or a very sensitive ICP-MS technique was applied for determination of analyte (see
Table 1). The LOD values after separation of Cr(III) on tested IIPs seems to be sufficient for analysis of natural water samples.
The procedures that use the Cr(III)-phen-ST and the Cr(III)-phen-ST-4VP polymers were applied to separate the Cr(III) and Cr(VI) ions from solutions containing different concentrations of these ions (listed in
Table 4). It was found that using the Cr(III)-phen-ST polymer allowed for the recovery of Cr(III) in the range of 83–100% in mixtures containing various ratios of Cr(III) and Cr(VI) ions. In the case of the Cr(III)-phen-ST-4VP polymer, recovery values of Cr(III) ranged from 85% to 99% in solutions with an excess of Cr(VI) ions. It was observed that with an increase in the excess of Cr(VI) ions, the recovery of Cr(III) ions on the Cr(III)-phen-ST-4VP polymer slightly decreased. The specific binding sites of Cr(III) ions were, potentially, partially blocked by Cr(VI) ions, due to the lower sorption capacity of this sorbent. The obtained results are characterized by good repeatability (RSD < 5%). Both polymers demonstrated good characteristic for the separation of Cr(III) in the presence of Cr(VI) ions, which is an advantage of the prepared IIPs over other imprinted polymers, where Cr(III) and Cr(VI) ions were retained simultaneously [
23,
30].
The accuracy of the proposed methods was evaluated by analyzing the standard reference materials of surface water SRM 1643e (NIST, USA) with the certified value for total concentration of chromium. The presence of only Cr(III) ions in this material was previously confirmed in the literature [
38,
39]. The concentrations of Cr(III) in SRM 1643e (2- and 4-fold diluted) determined after separation of analyte on both polymers were in good agreement with the certified value for the total concentration of chromium (recovery in the range of 93.3 to 98.6%,
Table 5), thus indicating the good accuracy of the proposed SPE—ETAAS methods.
3.4. Testing of the Method for Analysis of Water Samples
Methods were applied in determining of Cr(III) ions in surface water, tap water and green tea infusions. Green tea was chosen as a popular drink consumed by people, because it is rich in antioxidants and minerals and has a positive effect on the human body. However, there are reports in the literature [
40,
41,
42] about the presence of heavy metals, including chromium, in tea leaves.
Samples were adjusted to pH 4.5, left for equilibration and then submitted to the separation on both IIPs. As can be seen in
Table 5, the recovery of Cr(III) from surface water (SRM 1643e) was quantitative. Because the concentration of Cr(III) in tap water samples and green tea infusions was below the LOQ of the methods, the procedures were applied to samples spiked with Cr(III) at a concentration of 10 ng/mL. However, the recovery of Cr(III) in tap water was below 40% (
Table 5), which indicates a significant effect of sample constituents. Since the destruction of the organic matrix of the tap water sample with UV radiation did not improve the Cr(III) recovery, we assumed that this effect was due to the presence of inorganic ions in the water. Therefore, the columns were filled with bigger masses of polymers (0.4 g and 1 g vs. 0.1 g), which resulted in higher, but still not quantitative (60–75%), recovery of the analyte from tap water (
Table 5). Next, we compared the total content of following elements: Ca, Mg, Cu, Fe, Mn, Zn and Ni, in tap water, determined by ICP-MS technique, with their content in SRM surface water (taken from the certificate). We found that the sample of tap water loaded on the column contained 3.2 times higher content of these metals than sample of surface water (after 2-fold dilution) (63 mg/L vs. 20 mg/L). In order to check whether the reason for the incomplete recovery was an excess of other ions, water was filtered through the cation-exchange membrane. It allowed to reduce the content of other metal ion in the sample by 37% (to 40 mg/L) and resulted in quantitative recovery of Cr(III) on 0.1 g mass of Cr(III)-phen-ST polymer (93.2 ± 8.5%). Analogous experiments were performed for infusions of green tea prepared in Milli Q water and filtered tap water showing good recovery of Cr(III) from such samples on Cr(III)-phen-ST polymer (98.6 ± 3.2% and 94.2 ± 2.0%).
As shown in
Table 5, lower Cr(III) recovery from almost all samples was observed on the Cr(III)-phen-ST-4VP polymer, which is most likely the result of its lower sorption capacity than the Cr(III)-fen-ST polymer (0.4 µg/g versus 1.18 µg/g). It can be assumed that metal cations remaining in the water sample after filtration underwent non-specific sorption on the Cr(III)-phen-ST-4VP polymer, overloading of the capacity of this sorbent and decreasing its sorption ability for Cr(III) ions. This effect was not observed during separation of Cr(III) ions from surface water SRM 1643e which contained from 3 to 6 -times lower content of metals than tap water samples.
Summarizing, the developed method using the Cr(III)-phen-ST polymer is characterized by good separation properties of Cr(III) ions, exceeding some procedures published in the literature (see
Table 1) [
23,
33]. The prepared polymer allows for efficient of Cr(III) retention and elution with a fast sorption and desorption process of the analyte, making it suitable for working in a dynamic mode. The high stability of the Cr(III)-phen-ST polymer is its additional advantage compared to the sorbents used in the static mode [
23,
33]. This allows for multiple uses of the sorbent, which reduces the time and cost of the analysis. The Cr (III)-phen-ST polymer is also characterized by good selectivity towards Cr(III) ions in the presence of Fe(III), Mn(II), Cu(II) and Cr(VI) ions. Among the limitations of the procedure, the most serious is its susceptibility to the high content of other metal ions (mainly Ca and Mg) commonly present in water samples, which reduces the sorption capacity of the polymer towards Cr(III) ions, probably due to blocking the active site of the sorbent. The procedure could be useful for studies of chromium speciation in tea infusions.