Ecological and Health Risk Assessment of Potential Toxic Elements from a Mining Area (Water and Sediments): The San Juan-Taxco River System, Guerrero, Mexico
Abstract
:1. Introduction
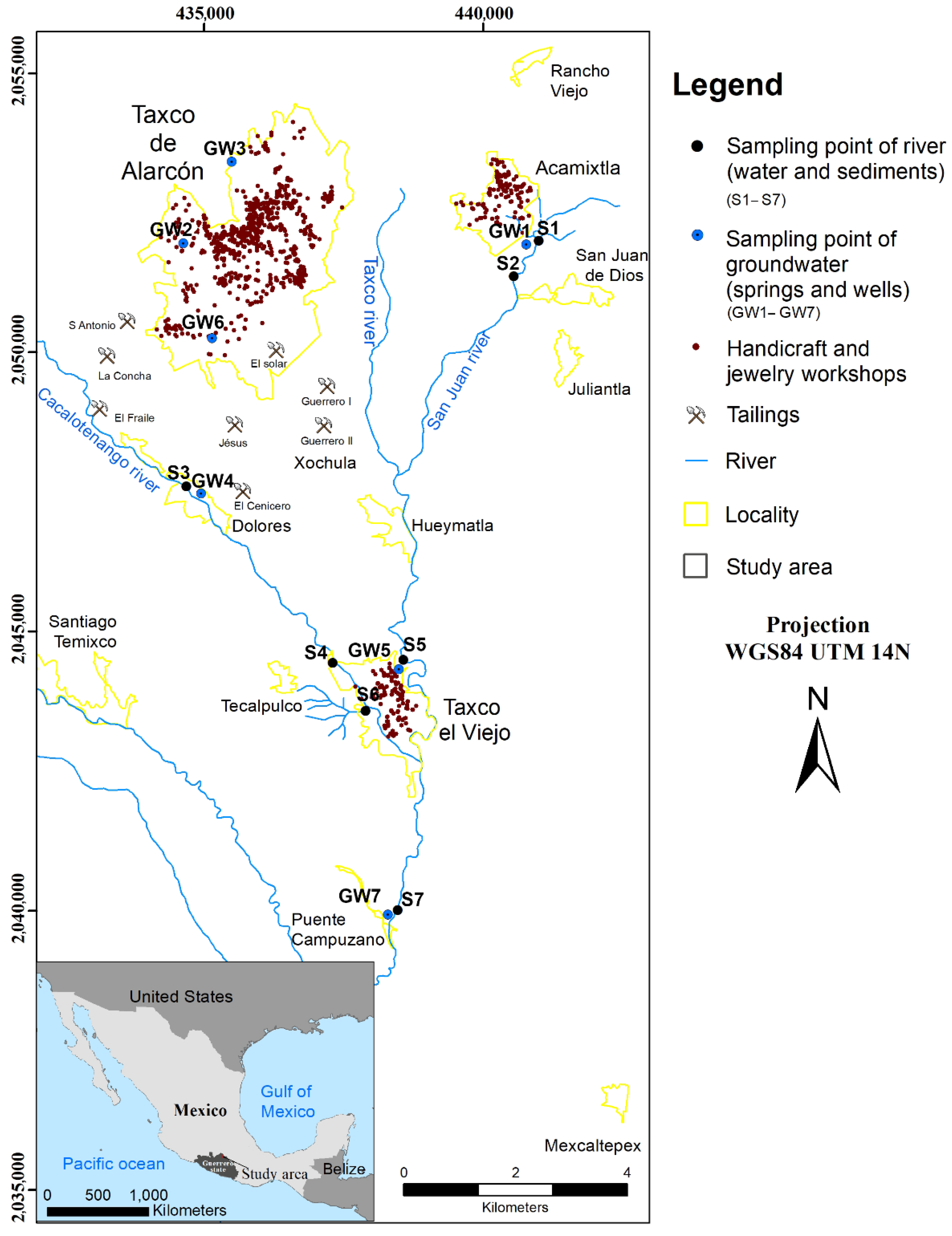
2. Study Area
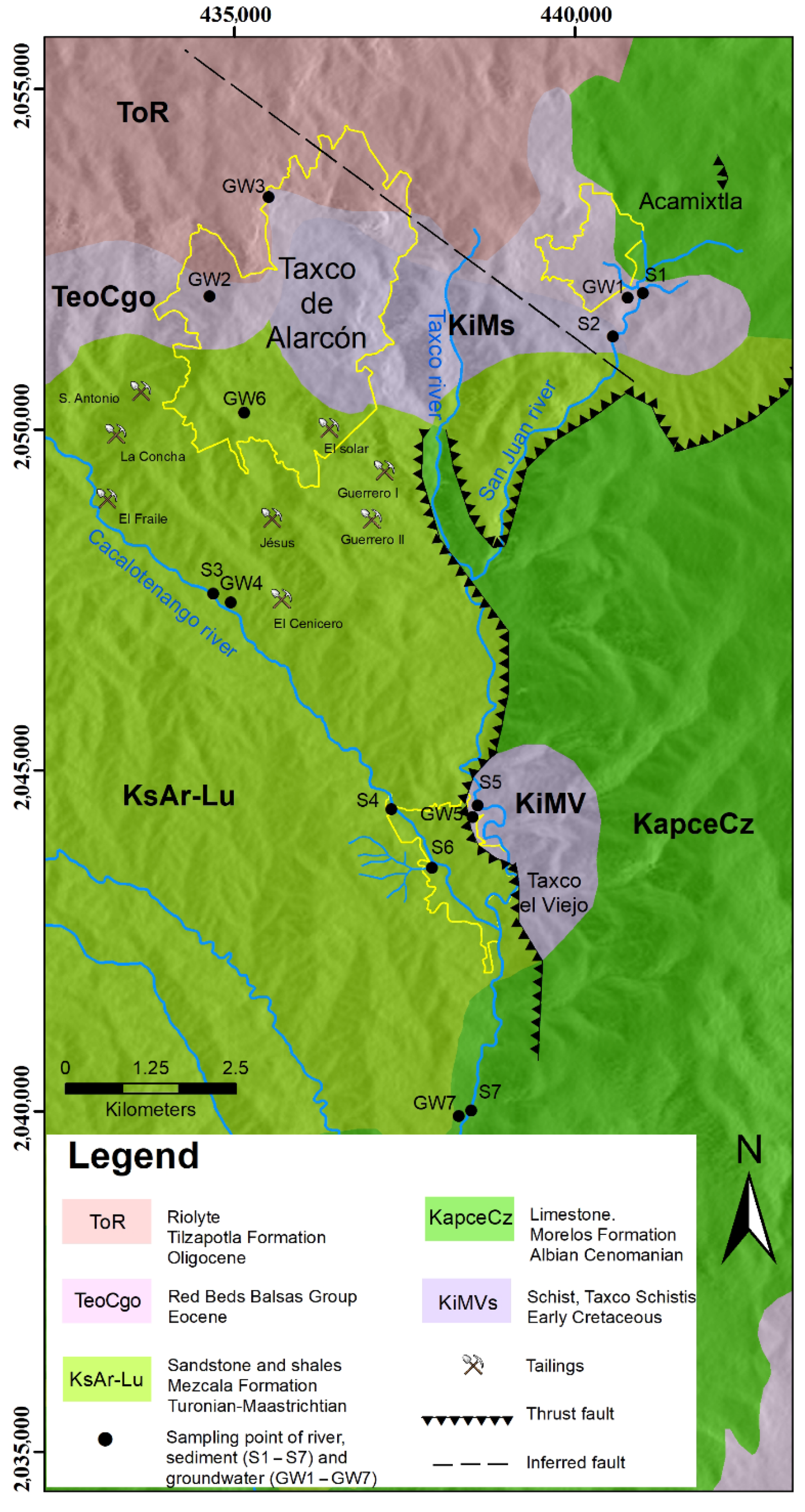
Geological and Hydrogeological Setting
3. Materials and Methods
3.1. Sample Collection and Analytical Procedure
3.2. Quality Controls and Calibration Curves for Analytical Equipment
3.3. Non-Parametric Statistics
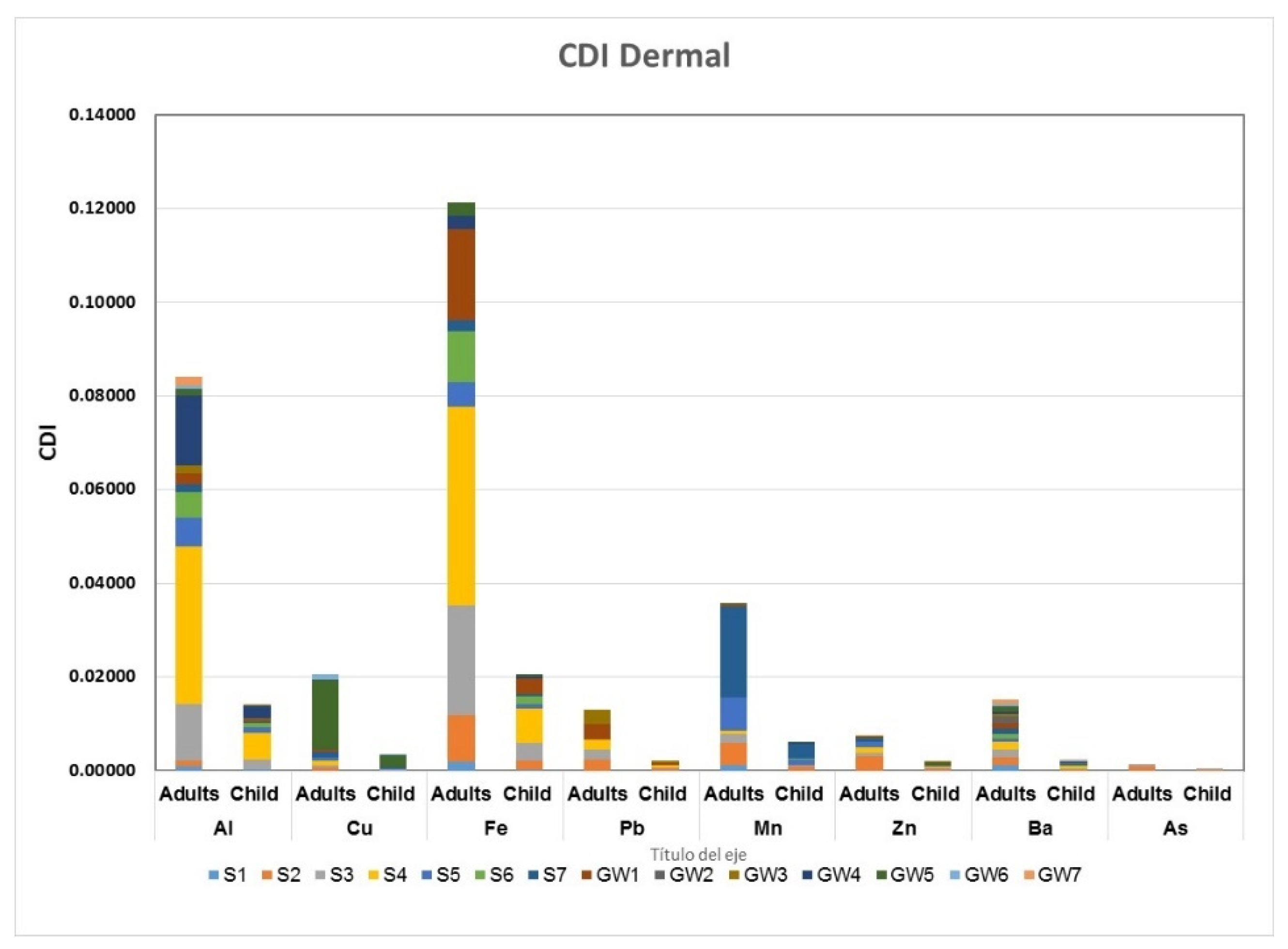
3.4. Health Risk Assessment
3.5. Assessment of PTE in the San Juan-Taxco River System (Water and Sediment)
3.6. Assessment of PTE Enrichment in Sediments
3.6.1. Geoaccumulation Index (Igeo)
3.6.2. Enrichment Factor (EF)
3.6.3. Contamination Factor (CF)
3.6.4. Pollution Load Index (PLI)
3.6.5. Potential Ecological Risk Index (RI)
4. Results
4.1. Non-Parametric Statistics
4.2. Water
4.2.1. Hydrochemical Characterization
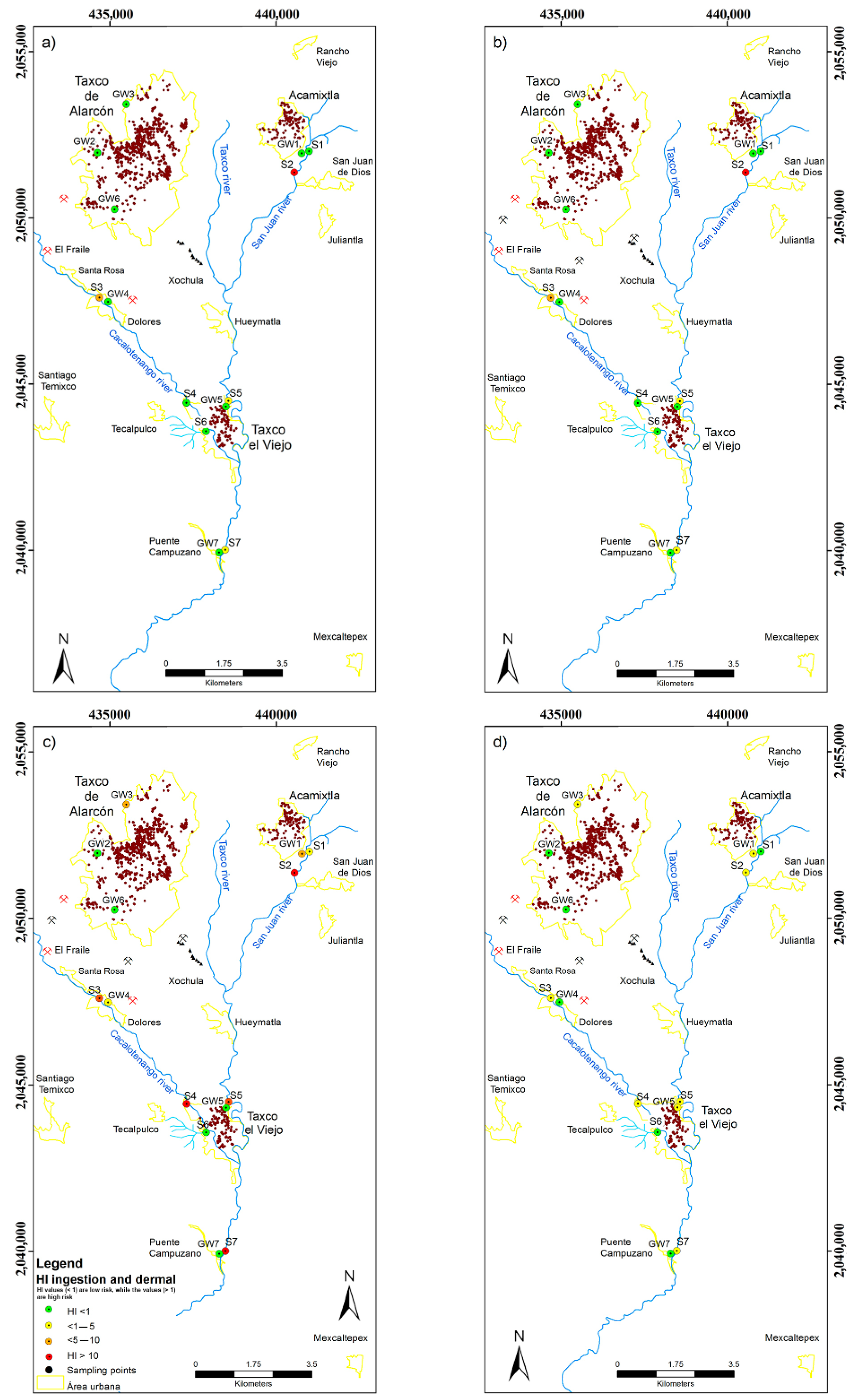
4.2.2. Health Risk Assessment in Water Samples
4.3. Sediments
4.3.1. PTE Concentrations in Sediments
4.3.2. Grain Size
4.3.3. Sediment Mineralographic Composition
4.3.4. Degree of PTE Enrichment and Ecological Risk
4.3.5. Comparison of PTE Concentrations with Other Riverine Environments Worldwide
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, J.; Yin, P.; Chen, B.; Gao, F.; Song, H.; Li, M. Distribution and contamination assessment of heavy metals in surface sediments of the Luanhe River Estuary, northwest of the Bohai Sea. Mar. Pollut. Bull. 2016, 109, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Malvandi, H. Preliminary evaluation of heavy metal contamination in the Zarrin-Gol River sediments, Iran. Mar. Pollut. Bull. 2017, 117, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Espinosa, P.; Shruti, V.; Jonathan, M.; Tavera, E.M. Metal concentrations and their potential ecological risks in fluvial sediments of Atoyac River basin, Central Mexico: Volcanic and anthropogenic influences. Ecotoxicol. Environ. Saf. 2018, 148, 1020–1033. [Google Scholar] [CrossRef]
- Morales-García, S.; Rodríguez-Espinosa, P.; Shruti, V.; Jonathan, M.; Martínez-Tavera, E. Metal concentrations in aquatic environments of Puebla River basin, Mexico: Natural and industrial influences. Environ. Sci. Pollut. Res. 2016, 24, 2589–2604. [Google Scholar] [CrossRef]
- Singh, U.K.; Kumar, B. Pathways of heavy metals contamination and associated human health risk in Ajay River basin, India. Chemosphere 2017, 174, 183–199. [Google Scholar] [CrossRef]
- Loredo-Portales, R.; Bustamante-Arce, J.; González-Villa, H.N.; Moreno-Rodríguez, V.; Del Rio-Salas, R.; Molina-Freaner, F.; González-Méndez, B.; Archundia-Peralta, D. Mobility and accessibility of Zn, Pb, and As in abandoned mine tailings of northwestern Mexico. Environ. Sci. Pollut. Res. 2020, 27, 26605–26620. [Google Scholar] [CrossRef]
- Hidayati, N.V.; Asia, L.; Khabouchi, I.; Torre, F.; Widowati, I.; Sabdono, A.; Doumenq, P.; Syakti, A.D. Ecological risk assessment of persistent organic pollutants (POPs) in surface sediments from aquaculture system. Chemosphere 2020, 263, 128372. [Google Scholar] [CrossRef]
- Wang, X.; Cui, L.; Li, J.; Zhang, C.; Gao, X.; Fan, B.; Liu, Z. Water quality criteria for the protection of human health of 15 toxic metals and their human risk in surface water, China. Environ. Pollut. 2021, 276, 116628. [Google Scholar] [CrossRef]
- Wang, X.; Fan, B.; Fan, M.; Belanger, S.; Li, J.; Chen, J.; Gao, X.; Liu, Z. Development and use of interspecies correlation estimation models in China for potential application in water quality criteria. Chemosphere 2020, 240, 124848. [Google Scholar] [CrossRef]
- Łuczyńska, J.; Paszczyk, B.; Łuczyński, M.J. Fish as a bioindicator of heavy metals pollution in aquatic ecosystem of Pluszne Lake, Poland, and risk assessment for consumer’s health. Ecotoxicol. Environ. Saf. 2018, 153, 60–67. [Google Scholar] [CrossRef]
- Prasad, S.; Saluja, R.; Joshi, V.; Garg, J.K. Heavy metal pollution in surface water of the Upper Ganga River, India: Human health risk assessment. Environ. Monit. Assess. 2020, 192, 742. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, E.; Pandey, J. Assessment of heavy metal pollution in water and surface sediment and evaluation of ecological risks associated with sediment contamination in the Ganga River: A basin-scale study. Environ. Sci. Pollut. Res. 2019, 26, 10926–10940. [Google Scholar] [CrossRef] [PubMed]
- Corami, F.; Morabito, E.; Gambaro, A.; Cescon, P.; Libralato, G.; Picone, M.; Ghirardini, A.V.; Barbante, C. Geospeciation, toxicological evaluation, and hazard assessment of trace elements in superficial and deep sediments. Environ. Sci. Pollut. Res. Int. 2020, 27, 15565–15583. [Google Scholar] [CrossRef] [PubMed]
- Dótor, A.A.; Armienta, M.A.; Arcega, F.; Talavera, M.O. Procesos de transporte de arsénico y metales en aguas superficiales del distrito minero de Taxco, Mexico: Aplicación de isótopos estables (Transport processes of arsenic and metals in surface waters in the mining district of Taxco, Mexico). Hidrobiológica 2014, 24, 256. [Google Scholar]
- Dótor, A.; Armienta, H.M.; Talavera, M.O.; Ruiz, J. Geochemical behavior of Cu and sulfur isotopes in the tropical mining region of Taxco, Guerrero (southern Mexico). Chem. Geol. 2017, 471, 1–12. [Google Scholar] [CrossRef]
- Talavera, M.O.; Ruiz, J.; Díaz, V.E.; Ramírez, G.A.; Cortés, A.; Salgado SS, A.; Rivera, B.R. Water-rock-tailings interactions and sources of sulfur and metals in the subtropical mining region of Taxco, Guerrero (Southern Mexico): A multi-isotopic approach. Appl. Geochem. 2016, 66, 73–81. [Google Scholar] [CrossRef]
- Árcega–Cabrera, F.; Armienta, M.A.; Daesslé, L.W.; Castillo-Blum, S.E.; Talavera, O.; Dótor, A. Variations of Pb in a mine-impacted tropical river, Taxco, Mexico: Use of geochemical, isotopic and statistical tools. Appl. Geochem. 2009, 24, 162–171. [Google Scholar] [CrossRef]
- Vázquez, B.A.; Talavera, M.O.; Moreno, G.M.; Salgado, S.S.; Ruiz, J.; Huerta, B.G. Source apportionment of lead in the blood of women of reproductive age living near tailings in Taxco, Guerrero, Mexico: An isotopic study. Sci. Total Environ. 2017, 583, 104–114. [Google Scholar] [CrossRef]
- INEGI. Censo y Conteos de Población y Vivienda of Guerrero; INEGI: Mexico City, Mexico, 2020. [Google Scholar]
- Clausell, A.M. Historia de la Plata Mexicana; AAPAUNAM Academia, Ciencia y Cultura: Mexico City, Mexico, 2010; Volume II, pp. 24–34. Available online: https://medigraphic.com/pdfs/aapaunam/pa-2010/pa101f.pdf (accessed on 7 June 2021).
- INEGI. La Minería en México; Serie estadísticas sectoriales No. 24; INEGI: Mexico City, Mexico, 2010. [Google Scholar]
- Ramírez, M. Acuerdos Comunitarios Para la Realización del Programa de Saneamiento de las Aguas Residuales de la Microcuenca del Río San Juan Taxco; Pueblo Sa-bio AC: San Juan de Dios, Guerrero, Mexico, 2013. [Google Scholar]
- Romero, F.M.; Armienta, M.A.; González-Hernández, G. Solid-phase control on the mobility of potentially toxic elements in an abandoned lead/zinc mine tailings impoundment, Taxco, Mexico. App. Geochem. 2007, 22, 109–127. [Google Scholar] [CrossRef]
- CONAGUA. Actualización de la Disponibilidad Media Anual de Agua en el Acuífero de Iguala Estado de Guerrero. México: Comisión Nacional del Agua. DOF, 20 de abril de 2015. Available online: https://www.gob.mx/cms/uploads/attachment/file/103668/DR_1205.pdf (accessed on 23 December 2021).
- Tessier, A.; Campbell, P.G.C.; Bisson, M. Sequential Extraction Procedure for the Speciation of Particulate Trace Metals. Anal. Chem. 1979, 51, 844–851. [Google Scholar] [CrossRef]
- ASTM D2487-06; Standard Practice for Classification of Soils for Engineering Purposes (Unified Soil Classification System); ASTM International: West Conshohocken, PA, USA, 2006.
- USEPA. Guidelines for Carcinogen Risk Assessment; EPA/630/P-03/001F; Risk Assessment Forum: Washington, DC, USA, 2005. [Google Scholar]
- USEPA. Exposure Factors Handbook; EPA/600/R-09/052F; National Center for Environmental Assessment: Washington, DC, USA, 2011. Available online: http://www.epa.gov/ncea/efh (accessed on 15 October 2021).
- WHO. Guidelines for Drinking-Water Quality, 3rd ed.; Incorporating 1st and 2nd Addenda; WHO: Geneva, Switzerland, 2008; Volume 1. [Google Scholar]
- Canadian Council of Ministers of the Environment CCME. Canadian Environmental Quality Guidelines; CCME: Winnipeg, MB, Canada, 1999. [Google Scholar]
- Environmental Protection Agency. 2015. Available online: https://www.epa.gov/sites/production/files/2015-09/documents/r3_btag_fw_sediment_benchmarks_8-06.pdf (accessed on 4 September 2021).
- Macdonald, D.D.; Ingersoll, C.G.; Berger, T.A. Development and evaluation of Consensus-Based Sediment Quality Guidelines for freshwater ecosystems. Arch. Environ. Contam. Toxicol 2000, 39, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Müller, G. Index of geoaccumulation in sediments of the Rhine River. Geo. J. 1969, 2, 108–118. [Google Scholar]
- Varol, M. Assessment of heavymetal contamination in sediments of the Tigris River (Turkey) using pollution indices and multivariate statistical techniques. J. Hazard. Mater. 2011, 195, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Rudnick, R.L.; Gao, S. Composition of the Continental Crust. Treatise Geochem. 2014, 3, 1–64. [Google Scholar] [CrossRef]
- Barats, A.; Renac, C.; Orani, A.M.; Durrieu, G.; Saint Martin, H.; Esteller, A.M.; Garrido, H.S. Tracing source and mobility of arsenic and trace elements in a hydrosystem impacted by past mining activities (Morelos state, Mexico). Sci. Total Environ. 2019, 712, 135565. [Google Scholar] [CrossRef]
- Sakan, S.M.; Ðorđević, D.S.; Manojlović, D.D.; Predrag, P.S. Assessment of heavy metal pollutants accumulation in the Tisza river sediments. J. Environ. Manag. 2009, 90, 3382–3390. [Google Scholar] [CrossRef]
- Al Rashdi, S.; Arabi, A.A.; Howari, F.M.; Siad, A. Distribution of heavy metals in the coastal area of Abu Dhabi in the United Arab Emirates. Mar. Pollut. Bull. 2015, 97, 494–498. [Google Scholar] [CrossRef]
- Hakanson, L. An ecological risk assessment index for aquatic contamination control, a sedimentological approach. Water Res. 1980, 14, 975–1001. [Google Scholar] [CrossRef]
- Tomlinson, D.L.; Wilson, J.G.; Harris, C.R.; Jeffrey, D.W. Problems in the assessment of heavy-metal levels in estuaries and the formation of a pollution index. Helgol. Meeresunters. 1980, 33, 566–575. [Google Scholar] [CrossRef] [Green Version]
- Hadzi, G.Y.; Essumang, D.K.; Ayoko, G.A. Assessment of contamination and health risk of heavy metals in selected water bodies around gold mining areas in Ghana. Environ. Monit. Assess. 2018, 190, 406. [Google Scholar] [CrossRef]
- Bodrud-Doza, M.; Didar-Ul Islam, S.M.; Rume, T.; Quraishi, S.B.; Rahman, M.S.; Bhuiyan, M.A.H. Groundwater quality and human health risk assessment for safe and sustainable water supply of Dhaka City dwellers in Bangladesh Groundw. Sustain. Dev. 2020, 10, 100374. [Google Scholar] [CrossRef]
- Armah, F.A.; Gyeabour, E.K. Health risks to children and adults residing in riverine environments where surficial sediments contain metals generated by active gold mining in Ghana. Toxicol. Res. 2013, 29, 69–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nkoom, M.; Jerry, S.C.; Michael, K. Assessment of endocrine disrupting trace metals in river Samre at Samreboi in the Wassa Amenfi West District of the western region of Ghana. J. Water Resour. Prot. 2013, 5, 983–992. [Google Scholar] [CrossRef] [Green Version]
- Obasi, P.N.; Akudinobi, B.B. Potential health risk and levels of heavy metals in water resources of lead–zinc mining communities of Abakaliki, southeast Nigeria. Appl. Water Sci. 2020, 10, 184. [Google Scholar] [CrossRef]
- Kim, S.; Lee, K.C.; Kim, J.E.; Jung, K.Y.; Ahn, J.M.; Kim, H.G.; Lee, I.J.; Shin, D.S.; Yang, D.S. Change in geochemical characteristics of surface sediments in the Nakdong river main stream. J. Environ. Sci. Int. 2017, 26, 311–324. [Google Scholar] [CrossRef]
- Farmer, V.C. Sources and speciation of aluminium and silicon in natural waters. Ciba Found. Symp. 1986, 121, 4–23. [Google Scholar] [CrossRef]
- Jonathan, M.P.; Roy, P.D.; Rodríguez-Espinosa, P.F.; Sarkar, S.K.; Muñoz-Sevilla, N.P.; Navarrete-López, M.; Srinivasalu, S.; Thangadurai, N. Evaluation of trace element concentration (acid leachable) in sediments from River Pánuco and its adjacent lagoon areas, NE México. Environ. Earth Sci. 2012, 68, 2239–2252. [Google Scholar] [CrossRef]
- Ruiz-Fernández, A.C.; Sanchez-Cabeza, J.A.; Alonso-Hernández, C.; Martínez-Herrera, V.; Pérez-Bernal, L.H.; Preda, M.; Hillaire-Marcel, C.; Gastaud, J.; Quejido-Cabezas, A.J. Effects of land use change and sediment mobilization on coastal contamination (Coatzacoalcos River, Mexico). Cont. Shelf Res. 2012, 37, 57–65. [Google Scholar] [CrossRef]
- Wang, H.T.; Wang, J.W.; Liu, R.M.; Yu, W.W.; Shen, Z.Y. Spatial variation, environmental risk and biological hazard assessment of heavy metals in surface sediments of the Yangtze River estuary. Mar. Pollut. Bull. 2015, 93, 250–258. [Google Scholar] [CrossRef]
- Ma, X.L.; Zuo, H.; Tian, M.J.; Zhang, L.Y.; Meng, J.; Zhou, X.N.; Min, N.; Chang, X.Y.; Liu, Y. Assessment of heavy metals contamination in sediments from three adjacent regions of the of the Yellow River using metal chemical fractions and multivariate analysis techniques. Chemosphere 2015, 144, 264–272. [Google Scholar] [CrossRef]
- García, J.; Aguado, J.; Morillo, I. Heavy metals in sediments from the Tinto River (Spain). Mine Water Environ. 2012, 1, 243–246. [Google Scholar]
- Varol, M.; Sen, B. Assessment of nutrient and heavy metal contamination in surface water and sediments of the upper Tigris River, Turkey. Catena 2012, 92, 1–10. [Google Scholar] [CrossRef]
- Pandey, J.; Singh, R. Heavy metals in sediments of Ganga River: Up- and downstream urban influences. Appl. Water Sci. 2017, 7, 669–1678. [Google Scholar] [CrossRef] [Green Version]
- Salah, E.A.M.; Zaidan, T.A.; Al-Rawi, A.S. Assessment of heavy metals pollution in the sediments of Euphrates River, Iraq. J. Water Res. Prot. 2012, 4, 1009–1023. [Google Scholar] [CrossRef] [Green Version]
- Armienta, M.A.; Talavera, O.; Villaseñor, G.; Espinosa, E.; Pérez-Martínez, I.; Cruz, O.; Ceniceros, N.; Aguayo, A. Environmental behaviour of metals from tailings in shallow rivers: Taxco, central Mexico. Appl. Earth Sci. 2004, 113, B76–B82. [Google Scholar] [CrossRef]
- Talavera, M.O.; Moreno, T.R.; Dotor-Almazán, A.; Flores-Mundo, N.; Duarte Gutiérrez, C. Mineralogy and geochemistry of sulfide-bearing tailings from silver mines in the Taxco, Mexico Area to evaluate their potential environmental impact. Geofís. Int. 2005, 44, 49–64. [Google Scholar] [CrossRef]


| Symbol | Name | Unit | Recommended | Symbol | Name | Unit | Recommended |
|---|---|---|---|---|---|---|---|
| Values | Values (Adult) | ||||||
| Cw | Element concentration | mg L−1 | SA | exposed skin area | cm2 | adult 5700, | |
| child 28,00 * | |||||||
| IR | Ingestion rate | L day−1 | adult 2.5 | AF | adherence factor | - | 0.07 * |
| child: 0.78 | |||||||
| EF | Exposure frequency | day y−1 | 350 | ABSd | dermal absorption fraction | - | 0.03 |
| ED | Total exposure duration | year | adult 30; | ET | exposure time | h day−1; | 0.58 |
| child 6 | |||||||
| BW | Average body weight | Adult 52 kg children 15 kg | CF | conversion factor | kg mg−1 | 10−2 + | |
| AT | Average exposure time | day | adult 10,950, child 2190 | RfDingestion | Reference Dose of PTEs | mg kg−1 day−1 | RfDingestion |
| Al = 1, Cu = 0.04, Fe = 0.7, Mn = 0.00096, Pb = 0.00042 | |||||||
| RfDD | Zn = 0.06, As = 0.0003 | ||||||
| RfDDerlmal | |||||||
| Al = 0.01, Cu = 0.00184, Fe = 0.14, Mn = 0.024, Pb = 0.0035 | |||||||
| Zn = 0.3, As = 0.003, Ba = 20 |
| Geoaccumulation Index (Igeo) | Enrichment Factor (EF) | Contamination Factor (CF) | |||
|---|---|---|---|---|---|
| Value | Categorization | Value | Categorization | Value | Categorization |
| <0 | Unpolluted | <1 | indicates no enrichment | <1 | low contamination |
| 0–1 | Pristine to moderately contaminated | 3–5 | moderate enrichment | 1–3 | moderate contamination |
| 1–2 | Moderately contaminated | 5–10 | moderately severe enrichment | 3–6 | considerable contamination |
| 2–3 | Moderately to strongly contaminated | 10–25 | severe enrichment | >6 | very high contamination |
| 3–4 | Strongly contaminated | 25–50 | Very severe enrichment | ||
| 4–5 | Strongly to extremely strongly contaminated | >50 | Extremely severe enrichment | ||
| >5 | extremely strongly contaminated | ||||
| Potential ecological risk factor ( | Potential ecological risk index (RI) | Pollution Load Index (PLI) | |||
| Value | Categorization | Value | Categorization | Value | Categorization |
| <40 | low ecological risk | <150 | low ecological risk | <1 | Unpolluted |
| 40–80 | moderate ecological risk | 150–300 | moderate ecological risk | 1–2 | Moderately polluted |
| 80–60 | appreciable ecological risk | 300–600 | considerable ecological risk | 2–3 | Heavily polluted |
| 160–320 | high ecological risk | >600 | very high ecological risk | >3 | Extremely polluted. |
| >320 | serious ecological risk | ||||
| Sites | Nature | pH | Al | As | B | Ba | Cd | Co | Cr | Cu | Fe | Mn | Ni | Pb | Zn | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Waters (mg L−1) | S1 | River water | 6.8 | 0.0071 | <l.q. | 0.0223 | 0.0942 | <l.q. | <l.q. | <l.q. | <l.q. | 0.0137 | 0.0822 | <l.q. | <l.q. | 0.0191 |
| S2 | 8.1 | 0.0084 | 0.0725 | 0.1173 | 0.1217 | <l.q. | <l.q. | <l.q. | 0.0046 | 0.0755 | 0.3622 | <l.q. | 0.0182 | 0.2335 | ||
| S3 | 7.9 | 0.0912 | 0.0291 | 0.0159 | 0.1162 | <l.q. | <l.q. | <l.q. | 0.0044 | 0.1759 | 0.1346 | <l.q. | 0.0149 | 0.0573 | ||
| S4 | 7.3 | 0.2525 | <l.q. | <l.q. | 0.1224 | <l.q. | <l.q. | <l.q. | 0.0061 | 0.3164 | 0.0541 | <l.q. | 0.0165 | 0.0858 | ||
| S5 | 7.2 | 0.0455 | <l.q. | 0.5284 | 0.0646 | <l.q. | <l.q. | <l.q. | 0.0070 | 0.0396 | 0.5343 | 0.0181 | <l.q. | 0.0843 | ||
| S6 | 7.2 | 0.0397 | <l.q. | <l.q. | 0.0722 | <l.q. | <l.q. | <l.q. | <l.q. | 0.0818 | 0.0073 | <l.q. | <l.q. | 0.0069 | ||
| S7 | 8.1 | 0.0125 | <l.q. | 0.5791 | 0.0914 | <l.q. | <l.q. | <l.q. | 0.0078 | 0.0175 | 1.4482 | 0.0174 | <l.q. | 0.0503 | ||
| GW1 | Dug well | 7.0 | 0.0183 | <l.q. | <l.q. | 0.081 | <l.q. | <l.q. | <l.q. | 0.004 | 0.146 | 0.032 | <l.q. | 0.0245 | 0.0770 | |
| GW2 | Springs | 6.6 | <l.q. | <l.q. | <l.q. | 0.111 | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | |
| GW3 | 6.7 | 0.0125 | <l.q. | <l.q. | 0.023 | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | 0.0235 | 0.0053 | ||
| GW4 | 8.2 | 0.1127 | <l.q. | 0.017 | 0.048 | <l.q. | <l.q. | <l.q. | <l.c. | 0.021 | 0.009 | <l.q. | <l.q. | 0.0149 | ||
| GW5 | 7.2 | 0.0097 | <l.q. | 0.068 | 0.081 | <l.q. | <l.q. | <l.q. | 0.111 | 0.022 | 0.006 | <l.q. | <l.q. | 0.2442 | ||
| GW6 | 6.9 | 0.0059 | <l.q. | <l.q. | 0.040 | <l.c. | <l.c. | <l.c. | 0.010 | <l.q. | <l.q. | <l.q. | <l.q. | 0.0130 | ||
| GW7 | Dug well | 7.9 | 0.0135 | <l.q. | <l.q. | 0.069 | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | <l.q. | 0.0277 | |
| WHO (2008) | 6.5–8.5 | 0.200 | 0.025 | 0.300 | 0.700 | 0.050 | 1.300 | 0.300 | 0.400 | 0.010 | 3.000 | |||||
| sediment (mg kg−1) | S1 | River sediment | 16,771.0 | <l.q. | 15.8 | 154.0 | 2.1 | 5.2 | 5.0 | 209.0 | 18,637.0 | 231.0 | 18.7 | 35.6 | 351.0 | |
| S2 | 16,610.0 | 39.3 | 16.8 | 151.0 | 1.8 | 6.1 | 2.5 | 68.3 | 20,009.0 | 362.0 | 16.6 | 94.2 | 144.0 | |||
| S3 | 18,313.0 | 48.8 | 12.8 | 173.0 | 16.2 | 7.2 | <l.q. | 78.4 | 20,526.0 | 795.0 | 15.7 | 265.0 | 2008.0 | |||
| S4 | 20,303.0 | 25.5 | 31.2 | 278.0 | 50.4 | 7.8 | 5.5 | 393.0 | 20,614.0 | 2710.0 | 111.0 | 244.0 | 4509.0 | |||
| S5 | 19,032.0 | 24.7 | 15.9 | 104.0 | 5.5 | 7.2 | <l.q. | 41.8 | 23,223.0 | 334.0 | 18.6 | 160.0 | 967.0 | |||
| S6 | 22,982.0 | <l.q. | 19.9 | 143.0 | 2.0 | 9.0 | 9.4 | 117.0 | 20,930.0 | 105.0 | 24.2 | 13.9 | 142.0 | |||
| S7 | 12,086.0 | 21.3 | 13.8 | 88.0 | 9.4 | 6.3 | <l.q. | 55.9 | 22,301.0 | 660.0 | 20.0 | 189.0 | 1763.0 | |||
| PEL a | 17 a | 3.53 a | 90 a | 196.6 a | 75 a | 91.3 a | 314.8 a | |||||||||
| TEC b | 9.79 b | 0.99 b | 43.4 b | 31.6 b | 22.7 b | 35.8 b | 121 b | |||||||||
| PEC c | 33 c | 4.98 c | 111 c | 149 c | 48.6 c | 128 c | 459 c | |||||||||
| Background values | 81,500 | 4.8 | 17 | 628 | 0.09 | 17.3 | 92 | 28 | 50,400 | 774 | 47 | 17 | 67 | |||
| River Water | Groundwater | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Oral | Dermal | Oral | Dermal | |||||||||||
| Mean | Min | Max | Mean | Min | Max | Mean | Min | Max | Mean | Min | Max | |||
| Adult | ||||||||||||||
| HQ | Al | 0.0044 | 0.0005 | 0.0171 | 0.8715 | 0.0943 | 3.3710 | HQ | 0.0017 | 0.0000 | 0.0076 | 0.3290 | 0.0000 | 1.5041 |
| Cu | 0.0072 | 0.0000 | 0.0132 | 0.0143 | 0.0000 | 0.0259 | 0.0303 | 0.0000 | 0.1883 | 0.0596 | 0.0000 | 0.3702 | ||
| Fe | 0.0100 | 0.0013 | 0.0307 | 0.0981 | 0.0130 | 0.3017 | 0.0026 | 0.0000 | 0.0141 | 0.0257 | 0.0000 | 0.1388 | ||
| Pb | 0.1375 | 0.0000 | 0.3536 | 2.2537 | 0.0000 | 5.7946 | 0.1330 | 0.0000 | 0.4747 | 2.1805 | 0.0000 | 7.7800 | ||
| Mn | 1.0599 | 0.0208 | 4.0964 | 5.2114 | 0.1022 | 20.141 | 0.0189 | 0.0000 | 0.0902 | 0.0930 | 0.0000 | 0.4436 | ||
| Zn | 0.0174 | 0.0016 | 0.0528 | 0.0165 | 0.0004 | 0.0519 | 0.0123 | 0.0000 | 0.0553 | 0.0012 | 0.0000 | 0.0054 | ||
| Ba | 0.1750 | 0.0000 | 0.5616 | 0.2657 | 0.1760 | 0.3334 | 0.0628 | 0.0221 | 0.1078 | 0.1765 | 0.0620 | 0.3029 | ||
| As | 3.2849 | 0.0000 | 16.400 | 0.6461 | 0.0000 | 3.2255 | ||||||||
| HI | 4.6963 | 0.0330 | 17.960 | 9.3773 | 0.9090 | 20.610 | HI | 0.2617 | 0.0588 | 0.6838 | 2.8656 | 0.2214 | 8.8439 | |
| Children | ||||||||||||||
| Al | 0.0034 | 0.0004 | 0.0135 | 0.1484 | 0.0161 | 0.5741 | 0.0013 | 0.0000 | 0.0060 | 0.0560 | 0.0000 | 0.2561 | ||
| HQ | Cu | 0.0042 | 0.0000 | 0.0094 | 0.0528 | 0.0000 | 0.0959 | HQ | 0.0238 | 0.0000 | 0.1479 | 0.2207 | 0.0000 | 1.3706 |
| Fe | 0.0078 | 0.0010 | 0.0241 | 0.0167 | 0.0022 | 0.0514 | 0.0021 | 0.0000 | 0.0111 | 0.0044 | 0.0000 | 0.0236 | ||
| Pb | 0.1080 | 0.0000 | 0.2778 | 0.3838 | 0.0000 | 0.9868 | 0.1045 | 0.0000 | 0.3729 | 0.3713 | 0.0000 | 1.3249 | ||
| Mn | 0.8327 | 0.0163 | 3.2183 | 0.8624 | 0.0174 | 3.4299 | 0.0149 | 0.0000 | 0.0709 | 0.0061 | 0.0000 | 0.0214 | ||
| Zn | 0.0136 | 0.0012 | 0.0415 | 0.0028 | 0.0001 | 0.0088 | 0.0097 | 0.0000 | 0.0434 | 0.0021 | 0.0003 | 0.0042 | ||
| Ba | 0.0743 | 0.0492 | 0.0932 | 0.0396 | 0.0044 | 0.0568 | 0.0494 | 0.0173 | 0.0847 | 0.0021 | 0.0003 | 0.0042 | ||
| As | 2.5808 | 0.0000 | 12.884 | 0.1100 | 0.0000 | 0.5493 | ||||||||
| HI | 3.6249 | 0.0873 | 14.102 | 1.6166 | 0.0422 | 3.6014 | HI | 0.2056 | 0.0462 | 0.5372 | 0.6623 | 0.0042 | 1.4523 | |
| Geoaccumulation Indices (Igeo) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| S1 | S2 | S3 | S4 | S5 | S6 | S7 | Mean | Max | Min | |
| Al | −2.87 | −2.88 | −2.7 | −2.59 | −2.68 | −2.41 | −3.34 | −2.787 | −2.41 | −3.34 |
| As | - | 2.45 | 2.76 | 1.82 | 1.78 | - | 1.56 | 2.074 | 2.76 | 1.56 |
| B | −0.69 | −0.6 | −1 | 0.29 | −0.68 | −0.36 | −0.89 | −0.56 | 0.29 | −0.99 |
| Ba | −2.61 | −2.64 | −2.4 | −1.76 | −3.18 | −2.72 | −3.42 | −2.681 | −1.76 | −3.42 |
| Cd | 3.96 | 3.74 | 6.91 | 8.54 | 5.35 | 3.89 | 6.12 | 5.5014 | 8.54 | 3.74 |
| Co | −2.32 | −2.09 | −1.9 | −1.73 | −1.85 | −1.53 | −2.04 | −1.916 | −1.53 | −2.32 |
| Cr | −4.79 | −5.79 | - | −4.65 | - | −3.88 | - | −4.778 | −3.88 | −5.79 |
| Cu | 2.32 | 0.7 | 0.9 | 3.23 | −0.01 | 1.48 | 0.41 | 1.29 | 3.23 | −0.01 |
| Fe | −2.02 | −1.92 | −1.9 | −1.87 | −1.7 | −1.85 | −1.76 | −1.857 | −1.7 | −2.02 |
| Mn | −2.33 | −1.68 | −0.6 | 1.22 | −1.8 | −3.47 | −0.81 | −1.346 | 1.22 | −3.47 |
| Ni | −1.91 | −2.09 | −2.2 | 0.65 | −1.92 | −1.54 | −1.82 | −1.543 | 0.65 | −2.17 |
| Pb | 0.48 | 1.89 | 3.38 | 3.26 | 2.65 | −0.88 | 2.89 | 1.9529 | 3.38 | −0.88 |
| Zn | 1.8 | 0.52 | 4.32 | 5.49 | 3.27 | 0.5 | 4.13 | 2.8614 | 5.49 | 0.5 |
| Enrichment factor (EF) | ||||||||||
| Al | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| As | - | 40.17 | 45.3 | 21.33 | 22.04 | - | 29.92 | 31.742 | 45.25 | 21.33 |
| B | 4.52 | 4.85 | 3.35 | 7.37 | 4.01 | 4.15 | 5.47 | 4.8171 | 7.37 | 3.35 |
| Ba | 1.19 | 1.18 | 1.23 | 1.78 | 0.71 | 0.81 | 0.94 | 1.12 | 1.78 | 0.71 |
| Cd | 113.39 | 98.13 | 801 | 2248 | 261.7 | 78.81 | 704.3 | 615.05 | 2247.9 | 78.81 |
| Co | 1.46 | 1.73 | 1.85 | 1.81 | 1.78 | 1.84 | 2.46 | 1.8471 | 2.46 | 1.46 |
| Cr | 0.26 | 0.13 | - | 0.24 | - | 0.36 | - | 0.2475 | 0.36 | 0.13 |
| Cu | 36.27 | 11.97 | 12.5 | 56.34 | 6.39 | 14.82 | 13.46 | 21.673 | 56.34 | 6.39 |
| Fe | 1.8 | 1.95 | 1.81 | 1.64 | 1.97 | 1.47 | 2.98 | 1.9457 | 2.98 | 1.47 |
| Mn | 1.45 | 2.29 | 4.57 | 14.05 | 1.85 | 0.48 | 5.75 | 4.3486 | 14.05 | 0.48 |
| Ni | 1.93 | 1.73 | 1.49 | 9.48 | 1.69 | 1.83 | 2.87 | 3.0029 | 9.48 | 1.49 |
| Pb | 10.18 | 27.19 | 69.4 | 57.62 | 40.3 | 2.9 | 74.97 | 40.366 | 74.97 | 2.9 |
| Zn | 25.46 | 10.55 | 133 | 270.2 | 61.81 | 7.52 | 177.4 | 98.044 | 270.15 | 7.52 |
| Sites | Contamination Factors (CFs) | PLI | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Al | As | B | Ba | Cd | Co | Cr | Cu | Fe | Mn | Ni | Pb | Zn | ||
| S1 | 0.21 | - | 0.93 | 0.25 | 23.33 | 0.3 | 0.05 | 7.46 | 0.37 | 0.3 | 0.4 | 2.09 | 5.24 | 0.80 |
| S2 | 0.2 | 8.19 | 0.99 | 0.24 | 20 | 0.35 | 0.03 | 2.44 | 0.4 | 0.47 | 0.35 | 5.54 | 2.15 | 0.86 |
| S3 | 0.22 | 10.17 | 0.75 | 0.28 | 180 | 0.42 | - | 2.8 | 0.41 | 1.03 | 0.33 | 15.59 | 29.97 | 2.08 |
| S4 | 0.25 | 5.31 | 1.84 | 0.44 | 560 | 0.45 | 0.06 | 14.04 | 0.41 | 3.5 | 2.36 | 14.35 | 67.3 | 2.83 |
| S5 | 0.23 | 5.15 | 0.94 | 0.17 | 61.11 | 0.42 | - | 1.49 | 0.46 | 0.43 | 0.4 | 9.41 | 14.43 | 1.43 |
| S6 | 0.28 | - | 1.17 | 0.23 | 22.22 | 0.52 | 0.1 | 4.18 | 0.42 | 0.14 | 0.51 | 0.82 | 2.12 | 0.72 |
| S7 | 0.15 | 4.44 | 0.81 | 0.14 | 104.44 | 0.36 | - | 2 | 0.44 | 0.85 | 0.43 | 11.12 | 26.31 | 1.59 |
| Mean | 0.22 | 6.652 | 1.06 | 0.25 | 138.73 | 0.4 | 0.06 | 4.916 | 0.42 | 0.96 | 0.68 | 8.417 | 21.07 | 1.47 |
| Max | 0.28 | 10.17 | 1.84 | 0.44 | 560 | 0.52 | 0.1 | 14.04 | 0.46 | 3.5 | 2.36 | 15.59 | 67.3 | 2.83 |
| Min | 0.15 | 4.44 | 0.75 | 0.14 | 20 | 0.3 | 0.03 | 1.49 | 0.37 | 0.14 | 0.33 | 0.82 | 2.12 | 0.72 |
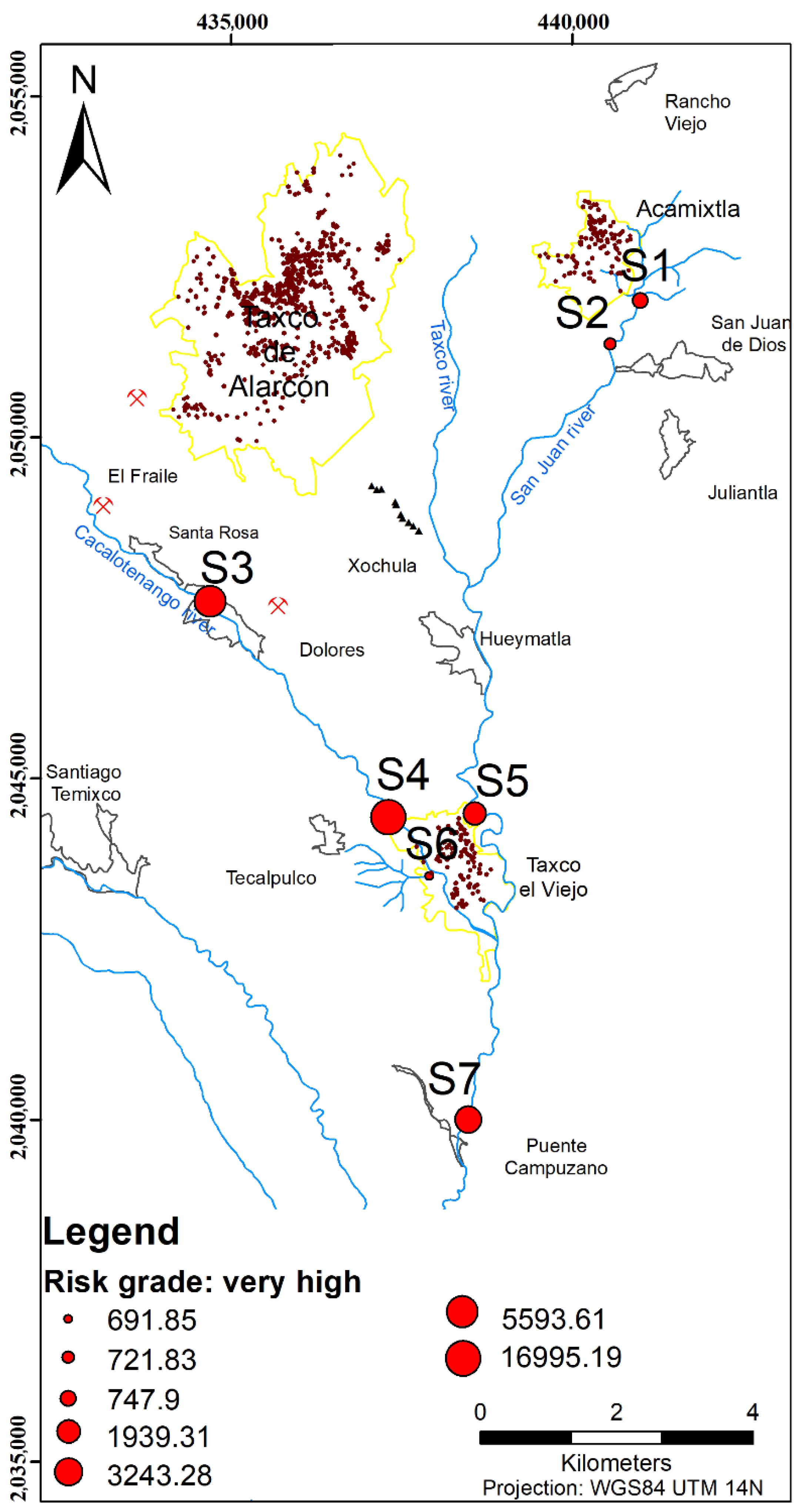
| Sites | RI | Risk Grade | |||||
|---|---|---|---|---|---|---|---|
| As | Cd | Cr | Cu | Pb | |||
| S1 | 700 | 0.109 | 37.32 | 10.47 | 747.9 | very high | |
| S2 | 81.88 | 600 | 0.054 | 12.2 | 27.71 | 721.83 | very high |
| S3 | 101.67 | 5400 | 14 | 77.94 | 5593.61 | very high | |
| S4 | 53.13 | 16,800 | 0.12 | 70.18 | 71.76 | 16,995.19 | very high |
| S5 | 51.46 | 1833.3 | 7.46 | 47.06 | 1939.31 | very high | |
| S6 | 666.7 | 0.204 | 20.89 | 4.09 | 691.85 | very high | |
| S7 | 44.38 | 3133.3 | 9.98 | 55.59 | 3243.28 | very high | |
| Mean | 66.504 | 4161.9 | 0.12175 | 24.576 | 42.0886 | 4276.1386 | |
| Max | 101.67 | 16,800 | 0.204 | 70.18 | 77.94 | 16,995.19 | |
| Min | 44.38 | 600 | 0.054 | 7.46 | 4.09 | 691.85 | |
| PTE | As | Ba | Cd | Cr | Cu | Ni | Pb | Zn | Fe |
|---|---|---|---|---|---|---|---|---|---|
| (mg kg−1) | |||||||||
| Present study | 17.1–48.8 | 1.8–50.4 | 2.5–17.1 | 4.3–502 | 15.4–111 | 2.5–13.9 | 45.09–142 | 15,366–23,223 | |
| Yangtze River [50] | 9.1 | 0.19 | 79.1 | 24.7 | 31.9 | 23.8 | 82.9 | - | |
| Yellow River [51] | - | 0.1–0.3 | 61.3–139.5 | 14.1–30.3 | 19.3–34.6 | 15.5–24.6 | 39.9–74.6 | - | |
| Tinto River [52] | 1130 | 2.75 | 56 | 805 | 17 | 2230 | 901 | 11,500 | |
| Tigris River [53] | 7.9 | - | 2860 | 66 | 1061 | - | |||
| Ganga River [54] | - | 1.7 | 69.9 | 29.8 | 372 | 26.7 | 67.8 | 31,988.60 | |
| Euphrates River [55] | - | 1.9 | 58.9 | 18.9 | 67.1 | 22.6 | 48 | 2249.50 | |
| Zarrin-Gol River [7] | 21.91 | - | 37.67 | - | 12.39 | - | 32.68 | 13,751 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salcedo Sánchez, E.R.; Martínez, J.M.E.; Morales, M.M.; Talavera Mendoza, O.; Alberich, M.V.E. Ecological and Health Risk Assessment of Potential Toxic Elements from a Mining Area (Water and Sediments): The San Juan-Taxco River System, Guerrero, Mexico. Water 2022, 14, 518. https://doi.org/10.3390/w14040518
Salcedo Sánchez ER, Martínez JME, Morales MM, Talavera Mendoza O, Alberich MVE. Ecological and Health Risk Assessment of Potential Toxic Elements from a Mining Area (Water and Sediments): The San Juan-Taxco River System, Guerrero, Mexico. Water. 2022; 14(4):518. https://doi.org/10.3390/w14040518
Chicago/Turabian StyleSalcedo Sánchez, Edith Rosalba, Juan Manuel Esquivel Martínez, Manuel Martínez Morales, Oscar Talavera Mendoza, and María Vicenta Esteller Alberich. 2022. "Ecological and Health Risk Assessment of Potential Toxic Elements from a Mining Area (Water and Sediments): The San Juan-Taxco River System, Guerrero, Mexico" Water 14, no. 4: 518. https://doi.org/10.3390/w14040518








