Bioponics as a Promising Approach to Sustainable Agriculture: A Review of the Main Methods for Producing Organic Nutrient Solution for Hydroponics
Abstract
1. Introduction
2. Sources of Organic Fertilizers for Bioponics
| Organic Source | N Content | P Content | TK (% DM) | C/N Ratio | References | ||
|---|---|---|---|---|---|---|---|
| TN (% DM) | Inorganic N (%TN) | TP (% DM) | Inorganic P (% TP) | ||||
| Animal manure | |||||||
| Cattle | 0.2–5.3 | 9.0–72.0 | 0.1–1.5 | 48.0–75.0 | 0.2–6.2 | 16.0–31.0 | [51,53,54,55,72,73,74,75,76,77,78] |
| Equine/horse | 1.4–3.9 | 5.8 | 0.6–1.2 | 45.0–49.0 | 1.2–4.2 | 19.0–25.0 | [51,73,74,76] |
| Sheep | 1.2–2.9 | 20.0–20.8 | 0.6–1.1 | 48.0–49.0 | 1.4–3.4 | 9.0–29.0 | [51,74,75,79] |
| Goat | 0.7–3.6 | 38.2 | 0.5–2.7 | 36.0–47.0 | 0.6–5.9 | 16.0–65.1 | [73,74,76,80] |
| Swine | 0.4–7.5 | 34.0–41.9 | 0.1–4.9 | 18.0–92.0 | 0.2–7.8 | 10.0–14.0 | [51,53,54,55,59,73,74,81] |
| Poultry | 1.2–6.1 | 32.6–48.0 | 0.4–3.6 | 25.0–90.0 | 0.8–3.1 | 4.0–18.0 | [53,55,59,73,74,75,77,78,81,82,83] |
| Slaughterhouse waste | |||||||
| Blood meal | 11.8–15.8 | <1 | 0.1–0.7 | n.a. 1 | 0.1–0.7 | 3.0–5.0 | [51,78,84,85] |
| Bone meal | 4.1–4.2 | n.a. | 8.7–23.5 | n.a. | 1.6 | 4.0–7.0 | [51,78,86,87] |
| Compost | 0.7–3.1 | 5.0–29.0 | 0.2–2 | 15.0–84.0 | 0.8–2.4 | 11.3–64.0 | [51,59,75] |
| Vermicompost | 0.5–3.5 | 2.7–37.0 | 0.1–4.7 | n.a. | 0.2–2.1 | 7.1–36.7 | [51,88,89,90,91,92] |
| Agro-industrial waste | |||||||
| Corn steep liquor | 3.4 | 2.5–8.2 | 1.5 | n.a. | 2.7 | n.a. | [21,93] |
| Cottonseed meal | 4.5–7.4 | <1 | 1.2–2.8 | n.a. | 1.7–2 | 8.0 | [51,53,67,68] |
| Canola meal | 5.6–6.6 | <1 | 2.8 | n.a. | 1.2 | 8.0 | [67,68] |
| Molasses | 0.4–2 | n.a. | 1.7 ± 0.7 | n.a. | 0.5–3.8 | n.a. | [69,71,94] |
| Plant residues | |||||||
| Soybean residue | 1.0–3.1 | n.a. | 0.4–0.8 | n.a. | 1.5–0.8 | 12.3–30.0 | [55,82,95] |
| Wheat residue | 0.4–0.8 | n.a. | 0.1–0.4 | n.a. | 1.2–3.8 | 84.0–124.0 | [95,96,97,98] |
| Leaf litter residue | 0.5–1 | n.a. | 0.4–0.8 | n.a. | 0.0–1.0 | 40.0–80.0 | [51,76,77] |
| Algae | 0.3–17.5 | n.a. | 0.1–4.5 | n.a. | 0.1–8.5 | n.a. | [99,100,101] |
| Household waste | 0.9–5.8 | 1.4 | 0.4–1.0 | n.a. | 0.8–1 | 7.0–27.0 | [40,51,75,82] |
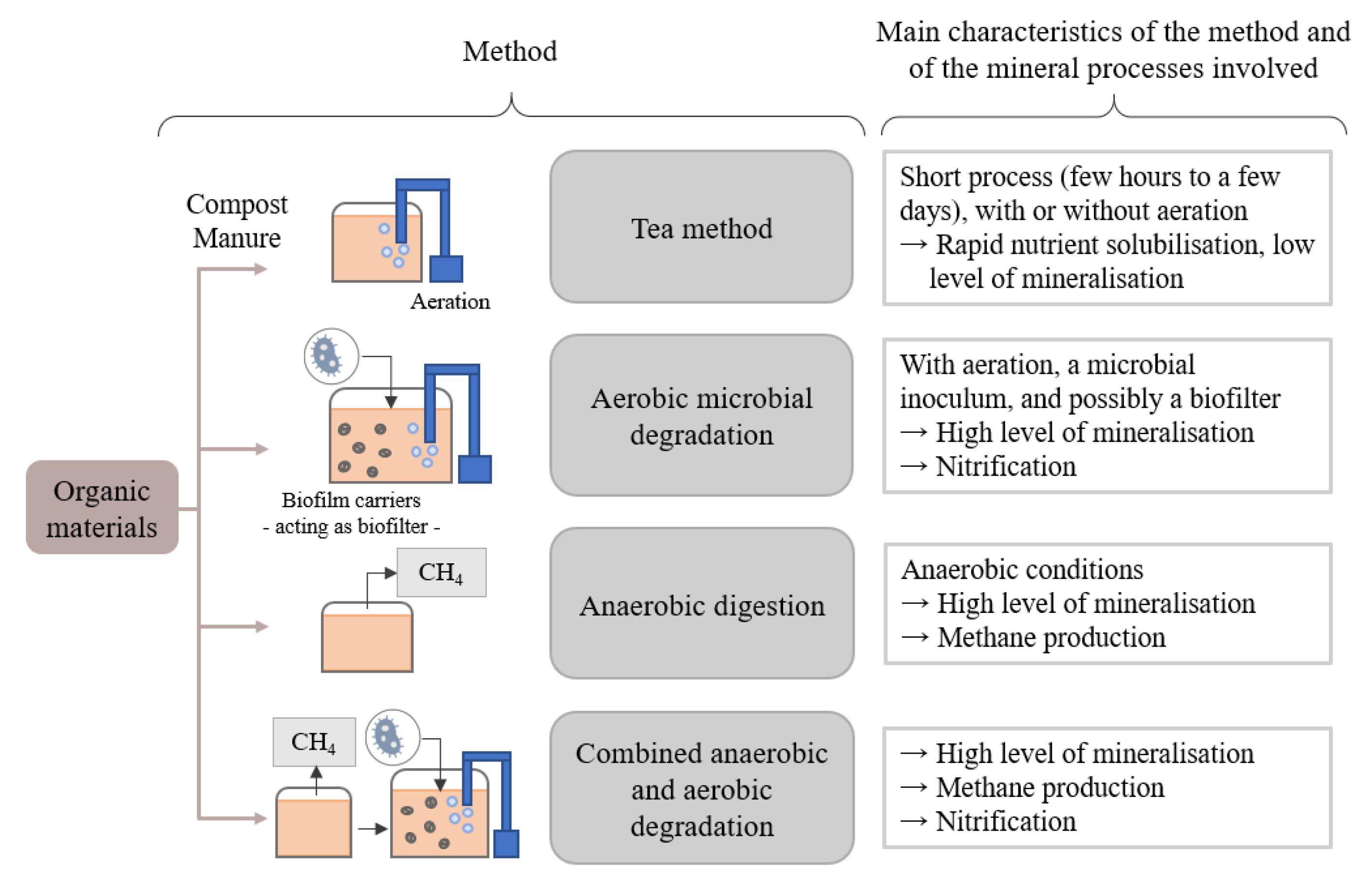
3. Methods Used for Producing Organic-Based Nutrient Solutions
3.1. The “Tea” Method
3.1.1. Definition and Principles
3.1.2. Use of the “Tea” Method in Bioponic Cultivation
Animal Manure Tea
Compost Tea
3.1.3. Drawbacks of the Method
Unbalanced Nutrient Composition and N-NH4+/N-NO3− Ratio
Phytotoxicity of Organic Compounds
3.1.4. Discussion on the Method
3.2. Aerobic Degradation Method with Microbial Processing
3.2.1. Principles and the Method Developed by Shinohara et al. (2011) [21]
3.2.2. Use of Aerobic Degradation for Bioponic Cultivation
3.2.3. A Parallel with Aquaponics
3.2.4. Discussion on the Method
3.3. Anaerobic Digestion Method
3.3.1. Principles and Contextualization of Anaerobic Digestion
3.3.2. Use of Anaerobic Digestates for Bioponic Cultivation
3.3.3. Drawbacks of the Method
3.3.4. Discussion on the Method and Potential Lines of Improvement
3.4. Method Combining Anaerobic Digestion and Microbial Aerobic Mineralization
3.4.1. Principles
3.4.2. Use of Combined Anaerobic Digestion and Aerobic Mineralization Methods for Bioponic Cultivation
3.4.3. Discussion on the Method
4. General Discussion and Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Resh, H.M. Hydroponic Food Production: A Definitive Guidebook for the Advanced Home Gardener and the Commercial Hydroponic Grower, 7th ed.; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar] [CrossRef]
- Bergstrand, K.-J.; Asp, H.; Hultberg, M. Utilizing Anaerobic Digestates as Nutrient Solutions in Hydroponic Production Systems. Sustainability 2020, 12, 10076. [Google Scholar] [CrossRef]
- Barman, N.C.; Hasan, M.M.; Islam, M.R.; Banu, N.A. A Review on Present Status and Future Prospective of Hydroponics Technique. Plant Environ. Dev. 2016, 5, 1–7. [Google Scholar]
- Gonnella, M.; Renna, M. The Evolution of Soilless Systems towards Ecological Sustainability in the Perspective of a Circular Economy. Is It Really the Opposite of Organic Agriculture? Agronomy 2021, 11, 950. [Google Scholar] [CrossRef]
- Arancon, N.Q.; Owens, J.D.; Converse, C. The effects of vermicompost tea on the growth and yield of lettuce and tomato in a non-circulating hydroponics system. J. Plant Nutr. 2019, 42, 2447–2458. [Google Scholar] [CrossRef]
- Velazquez-Gonzalez, R.S.; Garcia-Garcia, A.L.; Ventura-Zapata, E.; Barceinas-Sanchez, J.D.O.; Sosa-Savedra, J.C. A Review on Hydroponics and the Technologies Associated for Medium- and Small-Scale Operations. Agriculture 2022, 12, 646. [Google Scholar] [CrossRef]
- Salas, M.; Verdejo, M.; Sanchez, A.; Guzman, M.; Valenzuela, J.; Montero, J. Vertical gardening. Adaptation of hydroponic systems and ornamental species. Acta Hortic. 2012, 937, 1153–1160. [Google Scholar] [CrossRef]
- Basosi, R.; Spinelli, D.; Fierro, A.; Jez, S. Mineral Nitrogen Fertilizers: Environmental Impact of Production and Use; NOVA Science Publishers: New York, NY, USA, 2014; pp. 1–43. [Google Scholar]
- Cordell, D.; Drangert, J.-O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Chang. 2009, 19, 292–305. [Google Scholar] [CrossRef]
- U.S. Geological Survey. U.S. Mineral Commodity Summaries 2021; U.S. Geological Survey: Reston, VA, USA, 2021. [CrossRef]
- U.S. Geological Survey. USGS: Mineral Commodity Summaries, Phosphate Rock; U.S. Geological Survey: Reston, VA, USA, 2000.
- U.S. Geographical Survey. USGS: Mineral Commodity Summaries, Phosphate Rock; U.S. Geological Survey: Reston, VA, USA, 2011.
- Van Kauwenbergh, S.J. World Phosphate Rock Reserves and Resources; IFDC: Muscle Shoals, AL, USA, 2010. [Google Scholar]
- Kisinyo, P.O.; Opala, P.A. Depletion of phosphate rock reserves and world food crisis: Reality or hoax? Afr. J. Agric. Res. 2020, 16, 1223–1227. [Google Scholar] [CrossRef]
- Edixhoven, J.D.; Gupta, J.; Savenije, H.H.G. Recent revisions of phosphate rock reserves and resources: A critique. Earth Syst. Dyn. 2014, 5, 491–507. [Google Scholar] [CrossRef]
- Van Vuuren, D.; Bouwman, L.; Beusen, A. Phosphorus demand for the 1970–2100 period: A scenario analysis of resource depletion. Glob. Environ. Chang. 2010, 20, 428–439. [Google Scholar] [CrossRef]
- Hebebrand, C.; Laborde, D. High Fertilizer Prices Contribute to Rising Global Food Security Concerns. IFPRI: International Food Policy Research Institute. Available online: https://www.ifpri.org/blog/high-fertilizer-prices-contribute-rising-global-food-security-concerns (accessed on 6 October 2022).
- Chianu, J.N.; Chianu, J.N.; Mairura, F. Mineral Fertilizers in the Farming Systems of Sub-Saharan Africa. A Review. Agron. Sustain. Dev. 2012, 32, 545–566. [Google Scholar] [CrossRef]
- Sunaryo, Y.; Purnomo, D.; Darini, M.T.; Cahyani, V.R. Effects of goat manure liquid fertilizer combined with AB-MIX on foliage vegetables growth in hydroponic. IOP Conf. Ser. Earth Environ. Sci. 2018, 129, 012003. [Google Scholar] [CrossRef]
- Shubha, K.; Mukherjee, A.; Dubey, A.; Koley, T.K. Bioponics—A New Way to Grow Soilless Vegetable Cultivation. Agric. Food 2019, 1. [Google Scholar] [CrossRef]
- Shinohara, M.; Aoyama, C.; Fujiwara, K.; Watanabe, A.; Ohmori, H.; Uehara, Y.; Takano, M. Microbial mineralization of organic nitrogen into nitrate to allow the use of organic fertilizer in hydroponics. Soil Sci. Plant Nutr. 2011, 57, 190–203. [Google Scholar] [CrossRef]
- Giménez, A.; Fernández, J.A.; Pascual, J.A.; Ros, M.; Egea-Gilabert, C. Application of Directly Brewed Compost Extract Improves Yield and Quality in Baby Leaf Lettuce Grown Hydroponically. Agronomy 2020, 10, 370. [Google Scholar] [CrossRef]
- El-Shinawy, M.; Abd-Elmoniem, E.; Abou-Hadid, A. The use of organic manure for lettuce plants grown under NFT conditions. Acta Hortic. 1999, 491, 315–318. [Google Scholar] [CrossRef]
- Mowa, E.; Kalili, M.; Akundabweni, L.; Chimwamurombe, P. Impact of Organic Hydroponic Nutrient Solution on Tomato Fruit Quality. Int. Sci. Technol. J. Namib. 2018, 12, 62–77. [Google Scholar]
- Liu, W.; Du, L.; Yang, Q. Biogas slurry added amino acids decreased nitrate concentrations of lettuce in sand culture. Acta Agric. Scand. Sect. B—Soil Plant Sci. 2009, 59, 260–264. [Google Scholar] [CrossRef]
- Wang, L.; Guo, S.; Wang, Y.; Yi, D.; Wang, J. Poultry biogas slurry can partially substitute for mineral fertilizers in hydroponic lettuce production. Environ. Sci. Pollut. Res. 2019, 26, 659–671. [Google Scholar] [CrossRef]
- Gorenjak, A.H.; Cencic, A. Nitrate in vegetables and their impact on human health. A review. Acta Aliment. 2013, 42, 158–172. [Google Scholar] [CrossRef]
- Jin, E.; Cao, L.; Xiang, S.; Zhou, W.; Ruan, R.; Liu, Y. Feasibility of using pretreated swine wastewater for production of water spinach (Ipomoea aquatic Forsk.) in a hydroponic system. Agric. Water Manag. 2020, 228, 105856. [Google Scholar] [CrossRef]
- Botheju, D.; Svalheim, O.; Bakke, R. Digestate Nitrification for Nutrient Recovery. Open Waste Manag. J. 2010, 3, 1–12. [Google Scholar] [CrossRef]
- Lind, O.P.; Hultberg, M.; Bergstrand, K.-J.; Larsson-Jönsson, H.; Caspersen, S.; Asp, H. Biogas Digestate in Vegetable Hydroponic Production: pH Dynamics and pH Management by Controlled Nitrification. Waste Biomass-Valorization 2021, 12, 123–133. [Google Scholar] [CrossRef]
- Wongkiew, S.; Hu, Z.; Chandran, K.; Lee, J.W.; Khanal, S.K. Nitrogen transformations in aquaponic systems: A review. Aquac. Eng. 2017, 76, 9–19. [Google Scholar] [CrossRef]
- Williams, K.; Nelson, J.S. Challenges of using organic fertilizers in hydroponic production systems. Acta Hortic. 2016, 1112, 365–370. [Google Scholar] [CrossRef]
- Calvet, R.; Chenu, C.; Houot, S. Les Matières Organiques des Sols: Rôles Agronomiques et Environnementaux, 2nd ed.; Editions France Agricole: Paris, France, 2015. [Google Scholar]
- Tikasz, P.; MacPherson, S.; Adamchuk, V.; Lefsrud, M. Aerated chicken, cow, and turkey manure extracts differentially affect lettuce and kale yield in hydroponics. Int. J. Recycl. Org. Waste Agric. 2019, 8, 241–252. [Google Scholar] [CrossRef]
- Atkin, K.; Nichols, M. Organic Hydroponics. Acta Hortic. 2004, 648, 121–127. [Google Scholar] [CrossRef]
- Fang, W.; Chung, H. Bioponics for lettuce production in a plant factory with artificial lighting. Acta Hortic. 2018, 1227, 593–598. [Google Scholar] [CrossRef]
- Zandvakili, O.R.; Barker, A.V.; Hashemi, M.; Etemadi, F.; Autio, W.R. Comparisons of commercial organic and chemical fertilizer solutions on growth and composition of lettuce. J. Plant Nutr. 2019, 42, 990–1000. [Google Scholar] [CrossRef]
- Zhai, Z.; Ehret, D.L.; Forge, T.; Helmer, T.; Lin, W.; Dorais, M.; Papadopoulos, A.P. Organic Fertilizers for Greenhouse Tomatoes: Productivity and Substrate Microbiology. HortScience 2009, 44, 800–809. [Google Scholar] [CrossRef]
- Priadi, D.; Nuro, F. Seedling Production of Pak Choy (Brassica rapa L. var chinensis) using Organic and Inorganic Nutrients. Biosaintifika J. Biol. Biol. Educ. 2017, 9, 217–224. [Google Scholar] [CrossRef][Green Version]
- Kawamura-Aoyama, C.; Fujiwara, K.; Shinohara, M.; Takano, M. Study on the Hydroponic Culture of Lettuce with Microbially Degraded Solid Food Waste as a Nitrate Source. Jpn. Agric. Res. Quart. 2014, 48, 71–76. [Google Scholar] [CrossRef]
- Phibunwatthanawong, T.; Riddech, N. Liquid organic fertilizer production for growing vegetables under hydroponic condition. Int. J. Recycl. Org. Waste Agric. 2019, 8, 369–380. [Google Scholar] [CrossRef]
- Mowa, E. Organic Manure for Vegetable Production under Hydroponic Conditions in Arid Namibia. Int. Sci. Technol. J. Namib. 2015, 5, 3–12. [Google Scholar]
- Wongkiew, S.; Koottatep, T.; Polprasert, C.; Prombutara, P.; Jinsart, W.; Khanal, S.K. Bioponic system for nitrogen and phosphorus recovery from chicken manure: Evaluation of manure loading and microbial communities. Waste Manag. 2021, 125, 67–76. [Google Scholar] [CrossRef]
- Liedl, B.; Cummins, M.; Young, A.; Williams, M.; Chatfield, J. Hydroponic lettuce production using liquid effluent from poultry waste bioremediation as a nutrient source. Acta Hortic. 2004, 659, 721–728. [Google Scholar] [CrossRef]
- Leudtke, B. Use of Compost Tea as a Nutrient Amendment for Plant Growth in a Re-Circulating Hydroponic System; University of Wisconsin System: Madison, WI, USA, 2010. [Google Scholar]
- Latique, S.; Chernane, H.; El Kaoua, M. Seaweed Liquid Fertilizer Effect on Physiological and Biochemical Parameters of Bean Plant (Phaesolus Vulgaris Variety Paulista) under Hydroponic System. Eur. Sci. J. 2013, 9, 174–191. [Google Scholar]
- Yusuf, R.; Laude, S.; Alfiana; Syakur, A.; Ramli. The potential of seaweed used as hydroponic solution on the growth and yields of lettuce (Lactuca sativa L.). IOP Conf. Ser. Earth Environ. Sci. 2021, 653, 012065. [Google Scholar] [CrossRef]
- Steveni, C.M.; Norrington-Davies, J.; Hankins, S.D. Effect of seaweed concentrate on hydroponically grown spring barley. J. Appl. Phycol. 1992, 4, 173–180. [Google Scholar] [CrossRef]
- Saijai, S.; Ando, A.; Inukai, R.; Shinohara, M.; Ogawa, J. Analysis of microbial community and nitrogen transition with enriched nitrifying soil microbes for organic hydroponics. Biosci. Biotechnol. Biochem. 2016, 80, 2247–2254. [Google Scholar] [CrossRef]
- Baweja, P.; Kumar, S.; Kumar, G. Organic Fertilizer from Algae: A Novel Approach Towards Sustainable Agriculture. In Biofertilizers for Sustainable Agriculture and Environment; Springer: Amsterdam, The Netherlands, 2019; pp. 353–370. [Google Scholar] [CrossRef]
- Da Silva, E.F.; Melo, M.F.; Sombra, K.E.S.; Silva, T.S.; De Freitas, D.F.; da Costa, M.E.; Da Silva Santos, E.P.; da Silva, L.F.; Serra, A.P.; de Morais Cavalcante Neitzke, P.R. Organic Nitrogen in Agricultural Systems. In Nitrogen Fixation; IntechOpen: London, UK, 2020; pp. 1–22. [Google Scholar] [CrossRef]
- Shaji, H.; Chandran, V.; Mathew, L. Organic fertilizers as a route to controlled release of nutrients. In Controlled Release Fertilizers for Sustainable Agriculture; Lewu, F.B., Volova, T., Thomas, S., Rakhimol, K.R., Eds.; Academic Press: New York, NY, USA, 2020; pp. 231–245. [Google Scholar] [CrossRef]
- Green, B. Fertilizers in aquaculture. In Feed and Feeding Practices in Aquaculture; Woodhead Publishing: Sawston, Cambridge, UK, 2015; pp. 27–52. [Google Scholar] [CrossRef]
- Eghball, B.; Wienhold, B.J.; Gilley, J.E.; Eigenberg, R.A. Mineralization of Manure Nutrients. J. Soil Water Conserv. 2002, 57, 470–473. [Google Scholar]
- Goss, M.J.; Tubeileh, A.; Goorahoo, D. A Review of the Use of Organic Amendments and the Risk to Human Health. Adv. Agron. 2013, 120, 275–379. [Google Scholar] [CrossRef]
- Viskari, E.-L.; Grobler, G.; Karimäki, K.; Gorbatova, A.; Vilpas, R.; Lehtoranta, S. Nitrogen Recovery with Source Separation of Human Urine—Preliminary Results of Its Fertiliser Potential and Use in Agriculture. Front. Sustain. Food Syst. 2018, 2, 32. [Google Scholar] [CrossRef]
- Yang, L.; Giannis, A.; Chang, V.W.-C.; Liu, B.; Zhang, J.; Wang, J.-Y. Application of hydroponic systems for the treatment of source-separated human urine. Ecol. Eng. 2015, 81, 182–191. [Google Scholar] [CrossRef]
- Barnett, G. Phosphorus forms in animal manure. Bioresour. Technol. 1994, 49, 139–147. [Google Scholar] [CrossRef]
- Sharpley, A.; Moyer, B. Phosphorus Forms in Manure and Compost and Their Release during Simulated Rainfall. J. Environ. Qual. 2000, 29, 1462–1469. [Google Scholar] [CrossRef]
- Yadav, B.K.; Sidhu, A.S. Dynamics of Potassium and Their Bioavailability for Plant Nutrition. In Potassium Solubilizing Microorganisms for Sustainable Agriculture; Meena, V., Maurya, B., Verma, J., Meena, R., Eds.; Springer: New Delhi, India, 2016; pp. 187–201. [Google Scholar] [CrossRef]
- Mikkelsen, R.; Hartz, T.K. Nitrogen Sources for Organic Crop Production. Better Crops 2008, 92, 16–19. [Google Scholar]
- Chittora, D.; Meena, M.; Barupal, T.; Swapnil, P.; Sharma, K. Cyanobacteria as a source of biofertilizers for sustainable agriculture. Biochem. Biophys. Rep. 2020, 22, 100737. [Google Scholar] [CrossRef]
- Michalak, I.; Chojnacka, K. Algal compost–toward sustainable fertilization. Rev. Inorg. Chem. 2013, 33, 161–172. [Google Scholar] [CrossRef]
- Abdullahi, U.A.; Khandaker, M.M.; Alias, N.; Shaari, E.M.; Alam, A.; Badaluddin, N.A.; Mohd, K.S. Seaweed effects on plant growth and environmental remediation: A review. J. Phytol. 2021, 13, 122–129. [Google Scholar] [CrossRef]
- Nabti, E.; Jha, B.; Hartmann, A. Impact of seaweeds on agricultural crop production as biofertilizer. Int. J. Environ. Sci. Technol. 2016, 14, 1119–1134. [Google Scholar] [CrossRef]
- Michalak, I.; Chojnacka, K. Production of seaweed extracts by biological and chemical methods. In Marine Algae Extracts: Processes, Products, and Applications; Wiley: Weinheim, Germany, 2015; Volume 16, pp. 121–144. [Google Scholar] [CrossRef]
- Gale, E.S.; Sullivan, D.M.; Cogger, C.G.; Bary, A.I.; Hemphill, D.D.; Myhre, E.A. Estimating Plant-Available Nitrogen Release from Manures, Composts, and Specialty Products. J. Environ. Qual. 2006, 35, 2321–2332. [Google Scholar] [CrossRef] [PubMed]
- Fine, K.E.; Cole, J.C.; Penn, C.J. Nitrogen Mineralization from Canola Meal or Cottonseed Meal with or without Soapstock. HortScience 2013, 48, 891–896. [Google Scholar] [CrossRef]
- Jamir, L.; Kumar, V.; Kaur, J.; Kumar, S.; Singh, H. Composition, valorization and therapeutical potential of molasses: A critical review. Environ. Technol. Rev. 2021, 10, 131–142. [Google Scholar] [CrossRef]
- Kano, K.; Kitazawa, H.; Suzuki, K.; Widiastuti, A.; Odani, H.; Zhou, S.; Chinta, Y.; Eguchi, Y.; Shinohara, M.; Sato, T. Effects of Organic Fertilizer on Bok Choy Growth and Quality in Hydroponic Cultures. Agronomy 2021, 11, 491. [Google Scholar] [CrossRef]
- Hossain, M.Z.; von Fragstein, P.; Heß, J. Plant Origin Wastes as Soil Conditioner and Organic Fertilizer: A Review. J. Agric. Food Environ. Sci. 2016, 16, 1362–1371. [Google Scholar] [CrossRef]
- Weil, R.R.; Brady, N.C. The Nature and Properties of Soils, 15th ed.; Pearson: Columbus, OH, USA, 2017. [Google Scholar]
- Moreno-Caselles, J.; Moral, R.; Perez-Murcia, M.D.; Perez-Espinosa, A.; Rufete, B. Nutrient value of animal manures in front of environmental hazards. Commun. Soil Sci. Plant Anal. 2002, 33, 3023–3032. [Google Scholar] [CrossRef]
- Pagliari, P.H.; Laboski, C.A.M. Investigation of the Inorganic and Organic Phosphorus Forms in Animal Manure. J. Environ. Qual. 2012, 41, 901–910. [Google Scholar] [CrossRef]
- Mukai, S.; Oyanagi, W. Decomposition characteristics of indigenous organic fertilisers and introduced quick compost and their short-term nitrogen availability in the semi-arid Ethiopian Rift Valley. Sci. Rep. 2019, 9, 16000. [Google Scholar] [CrossRef]
- Agus, F.; Setyorini, D.; Hartatik, W.; Lee, S.-M.; Sung, J.-K.; Shin, J.-H. Nutrient Balance and Vegetable Crop Production as Affected by Different Sources of Organic Fertilizers. Korean J. Soil Sci. Fertil. 2009, 42, 1–13. [Google Scholar]
- Wortmann, C.S.; Shapiro, C.A. Composting Manure and Other Organic Materials. NebGuide G1315, University of Nebraska Extension. 2012, pp. 1–4. Available online: https://extensionpubs.unl.edu/publication/9000016363380/composting-manure-and-other-organic-materials/ (accessed on 1 December 2022).
- Nkoa, R. Agricultural benefits and environmental risks of soil fertilization with anaerobic digestates: A review. Agron. Sustain. Dev. 2014, 34, 473–492. [Google Scholar] [CrossRef]
- Sengupta, A.; Banerjee, H. Soil-Less Culture in Modern Agriculture. J. Sci. Technol. 2012, 2, 103–108. [Google Scholar]
- Cho, W.-M.; Ravindran, B.; Kim, J.K.; Jeong, K.-H.; Lee, D.J.; Choi, D.-Y. Nutrient status and phytotoxicity analysis of goat manure discharged from farms in South Korea. Environ. Technol. 2016, 38, 1191–1199. [Google Scholar] [CrossRef] [PubMed]
- Pratt, P.; Castellanos, J. Available Nitrogen from Animal Manures. Calif. Agric. 1981, 35, 24. [Google Scholar]
- Adediran, J.A.; Taiwo, L.B.; Sobulo, R.A. Effect of Organic Wastes and Method of Composting on Compost Maturity, Nutrient Composition of Compost and Yields of Two Vegetable Crops. J. Sustain. Agric. 2003, 22, 95–109. [Google Scholar] [CrossRef]
- Antil, R.S.; Singh, M. Effects of organic manures and fertilizers on organic matter and nutrients status of the soil. Arch. Agron. Soil Sci. 2007, 53, 519–528. [Google Scholar] [CrossRef]
- Hartz, T.K.; Johnstone, P.; Williams, E.; Smith, R. Establishing Lettuce Leaf Nutrient Optimum Ranges through DRIS Analysis. Hortscience 2007, 42, 143–146. [Google Scholar] [CrossRef]
- Basak, H.; Yigit, E. Comparative Effects of Ammonium Nitrate and Blood Meal on Plant Morphology and Leaf Coloring in Green Bean (Phaseolus Vulgaris L.) Seedlings under Salt Stress Condition. Int. J. Agric. Environ. Res. 2018, 4, 708–722. [Google Scholar]
- Genisel, M.; Erdal, S.; Turk, H.; Dumlupınar, R. The availability of bone powder as inorganic element source on growth and development in wheat seedlings. Toxicol. Ind. Health 2012, 28, 458–462. [Google Scholar] [CrossRef]
- Meng, X.; Huang, Q.; Xu, J.; Gao, H.; Yan, J. A review of phosphorus recovery from different thermal treatment products of sewage sludge. Waste Dispos. Sustain. Energy 2019, 1, 99–115. [Google Scholar] [CrossRef]
- Soobhany, N. Insight into the recovery of nutrients from organic solid waste through biochemical conversion processes for fertilizer production: A review. J. Clean. Prod. 2019, 241, 118413. [Google Scholar] [CrossRef]
- Mupambwa, H.A.; Mnkeni, P.N.S. Optimizing the vermicomposting of organic wastes amended with inorganic materials for production of nutrient-rich organic fertilizers: A review. Environ. Sci. Pollut. Res. 2018, 25, 10577–10595. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.-T.; Li, T.-P.; Zhang, Y.; Hu, J.; Bai, Y.-C.; Shan, Y.-H.; Ke, F. Effects of vermicompost amendment as a basal fertilizer on soil properties and cucumber yield and quality under continuous cropping conditions in a greenhouse. J. Soils Sediments 2017, 17, 2718–2730. [Google Scholar] [CrossRef]
- Lazcano, C.; Sampedro, L.; Zas, R.; Domínguez, J. Assessment of Plant Growth Promotion by Vermicompost in Different Progenies of Maritime Pine (Pinus pinaster Ait.). Compos. Sci. Util. 2010, 18, 111–118. [Google Scholar] [CrossRef]
- Suthar, S. Recycling of agro-industrial sludge through vermitechnology. Ecol. Eng. 2010, 36, 1028–1036. [Google Scholar] [CrossRef]
- Tan, J.P.; Jahim, J.M.; Wu, T.Y.; Harun, S.; Mumtaz, T. Use of corn steep liquor as an economical nitrogen source for biosuccinic acid production by Actinobacillus succinogenes. IOP Conf. Ser. Earth Environ. Sci. 2016, 36, 012058. [Google Scholar] [CrossRef]
- Roslan, M.; Sohedein, I.; Ling, P.; Sobri, Z.; Zuan, A.; Cheak, S.; Rahman, N. Sustainable Agronomic Valorization of Unsulfured Molasses and Defatted Soybean Meal as an Optimized Formulation of Bio-Organic Fertilizer Enriched with High Cell Density P-Solubilizing Bacteria. Agronomy 2021, 11, 996. [Google Scholar] [CrossRef]
- Andrews, E.; Kassama, S.; Smith, E.; Brown, P.; Khalsa, S. A Review of Potassium-Rich Crop Residues Used as Organic Matter Amendments in Tree Crop Agroecosystems. Agriculture 2021, 11, 580. [Google Scholar] [CrossRef]
- Damon, P.M.; Bowden, B.; Rose, T.; Rengel, Z. Crop residue contributions to phosphorus pools in agricultural soils: A review. Soil Biol. Biochem. 2014, 74, 127–137. [Google Scholar] [CrossRef]
- Mrad, F. Décomposition de Résidus de Culture et de Matériaux Biosourcés: Impact Sur Les Communautés Microbiennes des Sols Agricoles et Les Fonctions Associées. Ph.D. Thesis, Université de Rouen Normandie & UniLaSalle, Rouen, France, 2018. [Google Scholar]
- USDA; NRCS. Carbon to Nitrogen Ratios in Cropping Systems. 2011. Available online: https://soilhealthnexus.org/resources/soil-properties/soil-chemical-properties/carbon-to-nitrogen-ratio-cn/ (accessed on 7 March 2022).
- Raghunandan, B.L.; Vyas, R.V.; Patel, H.K.; Jhala, Y.K. Perspectives of Seaweed as Organic Fertilizer in Agriculture. In Soil Fertility Management for Sustainable Development; Springer: Singapore, 2019; pp. 267–289. [Google Scholar] [CrossRef]
- Olsson, J.; Toth, G.B.; Albers, E. Biochemical composition of red, green and brown seaweeds on the Swedish west coast. J. Appl. Phycol. 2020, 32, 3305–3317. [Google Scholar] [CrossRef]
- Cole, A.J.; Roberts, D.A.; Garside, A.L.; De Nys, R.; Paul, N.A. Seaweed compost for agricultural crop production. J. Appl. Phycol. 2016, 28, 629–642. [Google Scholar] [CrossRef]
- Charoenpakdee, S. Using Animal Manure to Grow Lettuce (Lactuca Sativa L.) in a Homemade Hydroponics System. Asia-Pac. J. Sci. Technol. 2014, 19, 256–261. [Google Scholar]
- Mowa, E.; Akundabweni, L.; Chimwamurombe, P.; Oku, E.; Mupanbwa, H.A. The influence of organic manure formulated from goat manure on growth and yield of tomato (Lycopersicum esculentum). Afr. J. Agric. Res. 2017, 12, 3061–3067. [Google Scholar] [CrossRef]
- Krishnasamy, K.; Nair, J.; Bäuml, B. Hydroponic system for the treatment of anaerobic liquid. Water Sci. Technol. 2012, 65, 1164–1171. [Google Scholar] [CrossRef]
- Kechasov, D.; Verheul, M.J.; Paponov, M.; Panosyan, A.; Paponov, I.A. Organic Waste-Based Fertilizer in Hydroponics Increases Tomato Fruit Size but Reduces Fruit Quality. Front. Plant Sci. 2021, 12, 680030. [Google Scholar] [CrossRef]
- Jones, J.B., Jr. Complete Guide for Growing Plants Hydroponically, 1st ed.; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Arancon, N.Q.; Pant, A.; Radovich, T.; Hue, N.V.; Potter, J.K.; Converse, C.E. Seed Germination and Seedling Growth of Tomato and Lettuce as Affected by Vermicompost Water Extracts (Teas). HortScience 2012, 47, 1722–1728. [Google Scholar] [CrossRef]
- Shaban, H.; Fazeli-Nasab, B.; Alahyari, H.; Alizadeh, G.; Shahpesandi, S. An Overview of the Benefits of Compost Tea on Plant and Soil Structure. Adv. Biores. 2015, 6, 154–158. [Google Scholar]
- Ingham, E.R. The Compost Tea Brewing Manual, 5th ed.; Soil Foodweb Institute: Corvallis, OR, USA, 2005. [Google Scholar]
- Carballo, T.; Gil, M.; Calvo, L.F.; Moran, A. The Influence of Aeration System, Temperature and Compost Origin on the Phytotoxicity of Compost Tea. Compos. Sci. Util. 2009, 17, 127–139. [Google Scholar] [CrossRef]
- St. Martin, C.C.; Brathwaite, R.A. Compost and compost tea: Principles and prospects as substrates and soil-borne disease management strategies in soil-less vegetable production. Biol. Agric. Hortic. 2012, 28, 1–33. [Google Scholar] [CrossRef]
- Radovich, T.; Pant, A.; Hue, N.; Sugano, J.; Arancon, N. Promoting Plant Growth with Compost Teas. Food Provid. 2011, 7, 1–3. [Google Scholar]
- Scheuerell, S.; Mahaffee, W. Compost Tea: Principles and Prospects for Plant Disease Control. Compos. Sci. Util. 2002, 10, 313–338. [Google Scholar] [CrossRef]
- Hargreaves, J.C.; Adl, M.S.; Warman, P.R. Are compost teas an effective nutrient amendment in the cultivation of strawberries? Soil and plant tissue effects. J. Sci. Food Agric. 2009, 89, 390–397. [Google Scholar] [CrossRef]
- Haghighi, M.; Barzegar, M.R.; da Silva, J.A.T. The effect of municipal solid waste compost, peat, perlite and vermicompost on tomato (Lycopersicum esculentum L.) growth and yield in a hydroponic system. Int. J. Recycl. Org. Waste Agric. 2016, 5, 231–242. [Google Scholar] [CrossRef]
- Haller, H.; Jonsson, A.; Rayo, K.M.; López, A.D. Microbial transport of aerated compost tea organisms in clay loam and sandy loam—A soil column study. Int. Biodeterior. Biodegrad. 2016, 106, 10–15. [Google Scholar] [CrossRef]
- Zaccardelli, M.; Pane, C.; Villecco, D.; Palese, A.M.; Celano, G. Compost tea spraying increases yield performance of pepper (Capsicum annuum L.) grown in greenhouse under organic farming system. Ital. J. Agron. 2018, 13, 229–234. [Google Scholar] [CrossRef]
- Liguori, L.; Pane, C.; Albanese, D.; Celano, G.; Zaccardelli, M.; Di Matteo, M. Compost and Compost Tea Management of Mini Watermelon Cultivations Affects the Chemical, Physical and Sensory Assessment of the Fruits. Agric. Sci. 2015, 6, 117–125. [Google Scholar] [CrossRef]
- Anjana, S.U.; Iqbal, M. Nitrate accumulation in plants, factors affecting the process, and human health implications. A review. Agron. Sustain. Dev. 2007, 27, 45–57. [Google Scholar] [CrossRef]
- Tian, X.; Fang, Y.; Jin, Y.; Yi, Z.; Li, J.; Du, A.; He, K.; Huang, Y.; Zhao, H. Ammonium detoxification mechanism of ammonium-tolerant duckweed (Landoltia punctata) revealed by carbon and nitrogen metabolism under ammonium stress. Environ. Pollut. 2021, 277, 116834. [Google Scholar] [CrossRef] [PubMed]
- Esteban, R.; Ariz, I.; Cruz, C.; Moran, J.F. Review: Mechanisms of ammonium toxicity and the quest for tolerance. Plant Sci. 2016, 248, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Britto, D.T.; Kronzucker, H. NH4+ toxicity in higher plants: A critical review. J. Plant Physiol. 2002, 159, 567–584. [Google Scholar] [CrossRef]
- Guo, S.; Bruck, H.; Sattelmacher, B. Effects of supplied nitrogen form on growth and water uptake of French bean (Phaseolus vulgaris L.) plants. Plant Soil 2002, 239, 267–275. [Google Scholar] [CrossRef]
- Ali, A.; Tucker, T.C.; Thompson, T.L.; Salim, M. Effects of Salinity and Mixed Ammonium and Nitrate Nutrition on the Growth and Nitrogen Utilization of Barley. J. Agron. Crop Sci. 2001, 186, 223–228. [Google Scholar] [CrossRef]
- Helali, S.M.; Nebli, H.; Kaddour, R.; Mahmoudi, H.; Lachaâl, M.; Ouerghi, Z. Influence of nitrate—Ammonium ratio on growth and nutrition of Arabidopsis thaliana. Plant Soil 2010, 336, 65–74. [Google Scholar] [CrossRef]
- Zhang, J.; Lv, J.; Dawuda, M.M.; Xie, J.; Yu, J.; Li, J.; Zhang, X.; Tang, C.; Wang, C.; Gan, Y. Appropriate Ammonium-Nitrate Ratio Improves Nutrient Accumulation and Fruit Quality in Pepper (Capsicum annuum L.). Agronomy 2019, 9, 683. [Google Scholar] [CrossRef]
- Hu, L.; Yu, J.; Liao, W.; Zhang, G.; Xie, J.; Lv, J.; Xiao, X.; Yang, B.; Zhou, R.; Bu, R. Moderate ammonium:nitrate alleviates low light intensity stress in mini Chinese cabbage seedling by regulating root architecture and photosynthesis. Sci. Hortic. 2015, 186, 143–153. [Google Scholar] [CrossRef]
- Tabatabaei, S.J.; Fatemi, L.S.; Fallahi, E. Effect of Ammonium: Nitrate Ratio on Yield, Calcium Concentration, and Photosynthesis Rate in Strawberry. J. Plant Nutr. 2007, 29, 1273–1285. [Google Scholar] [CrossRef]
- Liu, G.; Du, Q.; Li, J. Interactive effects of nitrate-ammonium ratios and temperatures on growth, photosynthesis, and nitrogen metabolism of tomato seedlings. Sci. Hortic. 2017, 214, 41–50. [Google Scholar] [CrossRef]
- Garland, J.; Mackowiak, C.; Sager, J. Hydroponic crop production using recycled nutrients from inedible crop residues. SAE Trans. 1993, 102, 1103–1110. [Google Scholar]
- Mackowiak, C.L.; Garland, J.L.; Strayer, R.F.; Finger, B.W.; Wheeler, R.M. Comparison of aerobically-treated and untreated crop residue as a source of recycled nutrients in a recirculating hydroponic system. Adv. Space Res. 1996, 18, 281–287. [Google Scholar] [CrossRef]
- Garland, J.L.; Mackowiak, C.L.; Strayer, R.F.; Finger, B.W. Integration of waste processing and biomass production systems as part of the KSC Breadboard project. Adv. Space Res. 1997, 20, 1821–1826. [Google Scholar] [CrossRef]
- Bohacz, J. Changes in mineral forms of nitrogen and sulfur and enzymatic activities during composting of lignocellulosic waste and chicken feathers. Environ. Sci. Pollut. Res. 2019, 26, 10333–10342. [Google Scholar] [CrossRef]
- Garland, J.L.; Mackowiak, C.L. Utilization of the Water Soluble Fraction of Wheat Straw as a Plant Nutrient Source. NASA Tech. Memo. 1990, 103497, 17–18. [Google Scholar]
- Finger, B.W.; Strayer, R.F. Development of an Intermediate-Scale Aerobic Bioreactor to Regenerate Nutrients from Inedible Crop Residues. SAE Trans. 1994, 103, 1365–1373. [Google Scholar] [CrossRef]
- Delaide, B.; Monsees, H.; Gross, A.; Goddek, S. Aerobic and Anaerobic Treatments for Aquaponic Sludge Reduction and Mineralisation. In Aquaponics Food Production Systems; Goddek, S., Joyce, A., Kotzen, B., Burnell, G.M., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 247–266. [Google Scholar] [CrossRef]
- Strayer, R.; Finger, B.; Alazraki, M. Evaluation of an anaerobic digestion system for processing CELSS crop residues for resource recovery. Adv. Space Res. 1997, 20, 2009–2015. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, K.; Aoyama, C.; Takano, M.; Shinohara, M. Suppression of Ralstonia solanacearum bacterial wilt disease by an organic hydroponic system. J. Gen. Plant Pathol. 2012, 78, 217–220. [Google Scholar] [CrossRef]
- Chinta, Y.D.; Eguchi, Y.; Widiastuti, A.; Shinohara, M.; Sato, T. Organic hydroponics induces systemic resistance against the air-borne pathogen, Botrytis cinerea (grey mould). J. Plant Interact. 2015, 10, 243–251. [Google Scholar] [CrossRef]
- Mowa, E.; Akundabweni, L.; Chimwamurombe, P.; Oku, E. Formulation of an Organic Hydroponic Nutrient Solution Using Nitrifying Microorganisms. Int. Sci. Technol. J. Namib. 2018, 12, 52–61. [Google Scholar]
- Somerville, C.; Cohen, M.; Pantanella, E.; Stankus, A.; Lovatelli, A. Small-Scale Aquaponic Food Production: Integrated Fish and Plant Farming; FAO Fisheries and Aquaculture Technical Paper; FAO: Rome, Italy, 2014; p. 262. [Google Scholar]
- Rakocy, J.E. Aquaponics—Integrating Fish and Plant Culture. In Aquaculture Production Systems; Wiley-Blackwell: Oxford, UK, 2012; pp. 344–386. [Google Scholar] [CrossRef]
- Vanotti, M.B.; Hunt, P.G. Nitrification treatment of swine wastewater with acclimated nitrifying sludge immobilized in polymer pellets. Trans. Am. Soc. Agric. Eng. 2000, 43, 405–413. [Google Scholar] [CrossRef]
- Cho, K.H.; Kim, J.-O.; Kang, S.; Park, H.; Kim, S.; Kim, Y.M. Achieving enhanced nitrification in communities of nitrifying bacteria in full-scale wastewater treatment plants via optimal temperature and pH. Sep. Purif. Technol. 2014, 132, 697–703. [Google Scholar] [CrossRef]
- Rochmah, W.N.; Mangkoedihardjo, S. Toxicity Effects of Organic Substances on Nitrification Efficiency. IOP Conf. Ser. Earth Environ. Sci. 2020, 506, 012011. [Google Scholar] [CrossRef]
- Prinčič, A.; Mahne, I.; Megušar, F.; Paul, E.A.; Tiedje, J.M. Effects of pH and Oxygen and Ammonium Concentrations on the Community Structure of Nitrifying Bacteria from Wastewater. Appl. Environ. Microbiol. 1998, 64, 3584–3590. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, J.; Liu, C.; Zhang, J.; Yang, J. Performance of a fixed-bed biofilm reactor with microbubble aeration in aerobic wastewater treatment. Water Sci. Technol. 2016, 74, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Ikeura, H.; Tsukada, K.; Tamaki, M. Effect of microbubbles in deep flow hydroponic culture on Spinach growth. J. Plant Nutr. 2017, 40, 2358–2364. [Google Scholar] [CrossRef]
- Park, J.-S.; Kurata, K. Application of Microbubbles to Hydroponics Solution Promotes Lettuce Growth. HortTechnology 2009, 19, 212–215. [Google Scholar] [CrossRef]
- Möller, K.; Müller, T. Effects of anaerobic digestion on digestate nutrient availability and crop growth: A review. Eng. Life Sci. 2012, 12, 242–257. [Google Scholar] [CrossRef]
- Alburquerque, J.A.; de la Fuente, C.; Bernal, M.P. Chemical properties of anaerobic digestates affecting C and N dynamics in amended soils. Agric. Ecosyst. Environ. 2012, 160, 15–22. [Google Scholar] [CrossRef]
- Singh, K.; Lee, K.; Worley, J.; Risse, L.M.; Das, K.C. Anaerobic Digestion of Poultry Litter: A Review. Appl. Eng. Agric. 2010, 26, 677–688. [Google Scholar] [CrossRef]
- Tuszynska, A.; Czerwionka, K.; Obarska-Pempkowiak, H. Phosphorus concentration and availability in raw organic waste and post fermentation products. J. Environ. Manag. 2021, 278 Pt 2, 111468. [Google Scholar] [CrossRef]
- Cioabla, A.E.; Ionel, I.; Dumitrel, G.-A.; Popescu, F. Comparative study on factors affecting anaerobic digestion of agricultural vegetal residues. Biotechnol. Biofuels 2012, 5, 39. [Google Scholar] [CrossRef]
- Ronga, D.; Setti, L.; Salvarani, C.; De Leo, R.; Bedin, E.; Pulvirenti, A.; Milc, J.; Pecchioni, N.; Francia, E. Effects of solid and liquid digestate for hydroponic baby leaf lettuce (Lactuca sativa L.) cultivation. Sci. Hortic. 2019, 244, 172–181. [Google Scholar] [CrossRef]
- Manyi-Loh, C.E.; Mamphweli, S.N.; Meyer, E.L.; Makaka, G.; Simon, M.; Okoh, A.I. An Overview of the Control of Bacterial Pathogens in Cattle Manure. Int. J. Environ. Res. Public Health 2016, 13, 843. [Google Scholar] [CrossRef]
- Chen, G.; Zhao, G.; Zhang, H.; Shen, Y.; Fei, H.; Cheng, W. Biogas slurry use as N fertilizer for two-season Zizania aquatica Turcz. in China. Nutr. Cycl. Agroecosyst. 2017, 107, 303–320. [Google Scholar] [CrossRef]
- Mupambwa, H.A.; Namwoonde, A.S.; Liswaniso, G.M.; Hausiku, M.K.; Ravindran, B. Biogas digestates are not an effective nutrient solution for hydroponic tomato (Lycopersicon esculentum L.) production under a deep water culture system. Heliyon 2019, 5, e02736. [Google Scholar] [CrossRef] [PubMed]
- Fahad, S.; Hussain, S.; Bano, A.; Saud, S.; Hassan, S.; Shan, D.; Khan, F.A.; Khan, F.; Chen, Y.; Wu, C.; et al. Potential role of phytohormones and plant growth-promoting rhizobacteria in abiotic stresses: Consequences for changing environment. Environ. Sci. Pollut. Res. 2015, 22, 4907–4921. [Google Scholar] [CrossRef] [PubMed]
- Kurepin, L.V.; Zaman, M.; Pharis, R.P. Phytohormonal basis for the plant growth promoting action of naturally occurring biostimulators. J. Sci. Food Agric. 2014, 94, 1715–1722. [Google Scholar] [CrossRef]
- Spaepen, S.; Vanderleyden, J.; Remans, R. Indole-3-acetic acid in microbial and microorganism-plant signaling. FEMS Microbiol. Rev. 2007, 31, 425–448. [Google Scholar] [CrossRef]
- Liedl, B.; Cummins, M.; Young, A.; Williams, M.; Chatfield, J. Liquid effluent from poultry waste bioremediation as a potential nutrient source for hydroponic tomato production. Acta Hortic. 2004, 659, 647–652. [Google Scholar] [CrossRef]
- Akl, I.A.; Savvas, D.; Papadantonakis, N.; Lydakis-Simantiris, N.; Kefalas, P. Influence of Ammonium to Total Nitrogen Supply Ratio on Growth, Yield and Fruit Quality of Tomato Grown in a Closed Hydroponic System. Eur. J. Hortic. 2003, 68, 204–211. [Google Scholar]
- Siddiqi, M.Y.; Malhotra, B.; Min, X.; Glass, A.D.M. Effects of ammonium and inorganic carbon enrichment on growth and yield of a hydroponic tomato crop. J. Plant Nutr. Soil Sci. 2002, 165, 191–197. [Google Scholar] [CrossRef]
- Chaudhry, A.; Nayab, S.; Hussain, S.; Ali, M.; Pan, Z. Current Understandings on Magnesium Deficiency and Future Outlooks for Sustainable Agriculture. Int. J. Mol. Sci. 2021, 22, 1819. [Google Scholar] [CrossRef]
- Schenk, M.; Wehrmann, J. The influence of ammonia in nutrient solution on growth and metabolism of cucumber plants. Plant Soil 1979, 52, 403–414. [Google Scholar] [CrossRef]
- Krupa, S. Effects of atmospheric ammonia (NH3) on terrestrial vegetation: A review. Environ. Pollut. 2003, 124, 179–221. [Google Scholar] [CrossRef]
- Hageman, R. Ammonium Versus Nitrate Nutrition of Higher Plants. In Nitrogen in Crop Production; American Society of Agronomy, Inc.: Madison, WI, USA, 1984; pp. 67–85. [Google Scholar] [CrossRef]
- Guo, S.; Zhou, Y.; Shen, Q.; Zhang, F. Effect of Ammonium and Nitrate Nutrition on Some Physiological Processes in Higher Plants—Growth, Photosynthesis, Photorespiration, and Water Relations. Plant Biol. 2007, 9, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Rafiullah, R.; Khan, M.J.; Muhammad, D. Foliar Application of Phosphorus to Enhance Phosphorus Utilization and Crop Growth: A Hydroponic Study. Sarhad J. Agric. 2018, 34, 47–53. [Google Scholar] [CrossRef]
- Cerozi, B.; Fitzsimmons, K. Phosphorus dynamics modeling and mass balance in an aquaponics system. Agric. Syst. 2017, 153, 94–100. [Google Scholar] [CrossRef]
- Da Silva Cerozi, B.; Fitzsimmons, K. The effect of pH on phosphorus availability and speciation in an aquaponics nutrient solution. Bioresour. Technol. 2016, 219, 778–781. [Google Scholar] [CrossRef]
- Sambo, P.; Nicoletto, C.; Giro, A.; Pii, Y.; Valentinuzzi, F.; Mimmo, T.; Lugli, P.; Orzes, G.; Mazzetto, F.; Astolfi, S.; et al. Hydroponic Solutions for Soilless Production Systems: Issues and Opportunities in a Smart Agriculture Perspective. Front. Plant Sci. 2019, 10, 923. [Google Scholar] [CrossRef]
- Ma, J.F. Role of silicon in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci. Plant Nutr. 2004, 50, 11–18. [Google Scholar] [CrossRef]
- Campos, C.N.S.; da Silva Júnior, G.B.; de Mello Prado, R.; de David, C.H.O.; de Souza Junior, J.P.; Teodoro, P.E. Silicon mitigates ammonium toxicity in plants. Agron. J. 2020, 112, 635–647. [Google Scholar] [CrossRef]
- Ashraf, M.; Rahmatullah; Ahmad, R.; Bhatti, A.; Afzal, M.; Sarwar, A.; Maqsood, M.; Kanwal, S. Amelioration of Salt Stress in Sugarcane (Saccharum officinarum L.) by Supplying Potassium and Silicon in Hydroponics. Pedosphere 2010, 20, 153–162. [Google Scholar] [CrossRef]
- Stamatakis, A.; Papadantonakis, N.; Savvas, D.; Lydakis-Simantiris, N.; Kefalas, P. Effects of silicon and salinity on fruit yield and quality of tomato grown hydroponically. Acta Hortic. 2003, 609, 141–147. [Google Scholar] [CrossRef]
- Bybordi, A.; Tabatabaei, S.J.; Ahmadov, A. Influence of salinity and ammonium: Nitrate ratio on growth, photosynthesis, fatty acid and the activity of antioxidative enzymes in canola. J. Plant Nutr. 2012, 35, 2089–2106. [Google Scholar] [CrossRef]
- Zhang, M.; Lawlor, P.G.; Wu, G.; Lynch, B.; Zhan, X. Partial nitrification and nutrient removal in intermittently aerated sequencing batch reactors treating separated digestate liquid after anaerobic digestion of pig manure. Bioprocess Biosyst. Eng. 2011, 34, 1049–1056. [Google Scholar] [CrossRef] [PubMed]
- Parravicini, V.; Svardal, K.; Hornek, R.; Kroiss, H. Aeration of anaerobically digested sewage sludge for COD and nitrogen removal: Optimization at large-scale. Water Sci. Technol. 2008, 57, 257–264. [Google Scholar] [CrossRef][Green Version]
- Oke, O.L. Nitrite Toxicity to Plants. Nature 1966, 212, 528. [Google Scholar] [CrossRef]

| Study | Primary Organic Material | pH | EC mS/cm | N Content | P | K | Ca | Mg | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | N-NO3− | N-NH4+ | N-NO2− | |||||||||
| Classical mineral solution 1 | 5.5–6.5 | n.a. | n.a. | 80.0–150.0 | 10.0–30.0 | n.a. | 15.0–30.0 | 100.0–150.0 | 150.0–200.0 | 50.0–80.0 | ||
| Tea method | ||||||||||||
| [23] | Chicken manure | 5.5–6.5 | 1.8–2.0 | 94.0 | n.a. 2 | n.a. | n.a. | 80.0 | 1596.0 | 106.0 | 81.6 | |
| [5] | Vermicompost | 7.9 | 0.5 | 1.3 | 1.0 | 0.3 | n.a. | 2.4 | 45.4 | 449.0 | 40.1 | |
| [34] | Chicken manure | 8.0–8.2 | 1.5–1.8 | n.a. | 0.6 | 284.6 | n.a. | n.a. | n.a. | 44.0 | 20.6 | |
| Cow manure | 8.0–8.3 | 0.5–0.8 | n.a. | 3.5 | 11.9 | n.a. | n.a. | n.a. | 62.4 | 45.4 | ||
| Turkey manure | 7.8–8.0 | 0.5–0.7 | n.a. | 11.7 | 35.2 | n.a. | n.a. | n.a. | 23.2 | 29.1 | ||
| [45] | Compost | 7.6–8.3 | n.a. | n.a. | 6.0 ± 0.7 | 2.0 | n.a. | 6.0 ± 0.6 | 84.0 ± 9.0 | 40.0 ± 5.0 | 19.0 ± 2.0 | |
| [22] | Compost | n.a. | 2.5–3.6 | n.a. | 13.5 | n.a. | n.a. | n.a. | 24.0 ± 3.0 | 77.0 ± 0.1 | 30.0 ± 0.1 | |
| Microbial aerobic degradation | ||||||||||||
| [21] | Fish-based | n.a. | n.a. | n.a. | 225.0 ± 13.0 | 0 | n.a. | n.a. | 28.8 | 66.3 | 10.1 | |
| [103] | Goat manure | 7.4 | 3.2 | n.a. | 44.7 | n.a. | n.a. | 42.1 | 360.0 | 250.0 | 67.0 | |
| [43] | Chicken manure | 8.4 ± 0.2 | n.a. | 19.6 ± 3.5 | 11.2 ± 7.5 | 7.0 ± 2.5 | 1.5 ± 0.8 | 53.9 ± 14.8 | n.a. | n.a. | n.a. | |
| Anaerobic digestion method | ||||||||||||
| [44] | Poultry litter | 5.3 | 1.3 | n.a. | 0 | 43.6 | 0 | 410.8 | 216.0 | 51.7 | 15.8 | |
| [104] 3 | Food waste | 7.7 | 1.7 | n.a. | 18.6 ± 0.1 | 42.7 ± 0.0 | n.a. | 1.9 ± 0.0 | 103 ± 0.1 | 40.9 ± 0.4 | 51.1 ± 1.3 | |
| [41] 4 | Distillery slop, sugarcane leaves | 7.0–8.0 | 2.5–3.0 | 160.0 | n.a. | n.a. | n.a. | 4.0 | 820.0 | n.a. | n.a. | |
| [25] 5 | N.D. | 7.5 | 2.5 | 182.5 | 2.2 | 175.0 | n.a. | 7.0 | 185.0 | 88.0 | 56.7 | |
| [26] 6 | Poultry manure | n.a. | 1.2 ± 0.1 | 104.1 | n.a. | n.a. | n.a. | 4.3 | 94.6 | 8.1 | 0.7 | |
| Combined anaerobic digestion and aerobic degradation | ||||||||||||
| [2] | Slaughterhouse and organic household waste, manure | n.a. | 2.0 | n.a. | 90.0 | 75.0 | 72.0 | 4.1 | 120.0 | 59.5 | 5.1 | |
| [30] | Plant-based residues | 5.0 | 1.8 | 182.0 | 90.0 | 14.0 | 78 | 41 | 250.0 | 76.0 | 21.0 | |
| [105] | Pig manure | 6.3 ± 1.0 | 0.5 ± 0.2 | n.a. | 21.0 ± 15.4 | 2.5 ± 4.7 | 0.4 ± 0.4 | 26.1 ± 16.7 | 30.9 ± 24.6 | 16.8 ± 4.4 | 4.8 ± 1.9 | |
| Study | Main Organic Materials | Main Method | Hydroponic System | Cultivated Plant |
|---|---|---|---|---|
| [34] | Chicken, cow or turkey manure | 2-day aerated tea (0.66, 1.66 or 3.33 g/L manure) | Ebb and flow | Lettuce (Lactuca sativa) and kale (Brassica napus) |
| [23] | Chicken, pigeon or buffalo manure | 2-day nonaerated tea (250 g/L manure) | NFT 1 | Lettuce (Lactuca sativa) |
| [102] | Bat, cattle or pig manure (dried and ground) | 1-day nonaerated tea (1:50 ratio manure:water) | DWC 2 | Lettuce (Lactuca sativa.) |
| [45] | Compost (from food waste products) | 2-day aerated tea (200 g/L compost) | Ebb and flow | Wisconsin Fast Plants™ (Brassica rapa) |
| Study | Main Organic Materials | Main Method | Hydroponic System | Cultivated Plant |
|---|---|---|---|---|
| [21] | Fish-based soluble fertilizer or corn steep liquor | (1) 12 to 170-day aeration of water with fertilizer (1–1.5 g/L) and bark compost (1 g/L) (2) Plant cultivation with solution aeration and fertilizer addition | DWC 1 (floating raft) and NFT 2 | Tomato (Solanum lycopersicum) and lettuce (Lactuca sativa) |
| [40] | Solid food waste (milled and dried) | (1) 64-day aeration of water with fertilizer (4 g/L) and bark compost (5 g/L); (2) Plant cultivation with solution aeration and fertilizer addition | DWC (floating raft) | Lettuce (Lactuca sativa) |
| [24] | Goat manure (dried and ground) | 30-day aeration of water with compost (10 g/L) and fertilizer (0.25 g/L/day during the first 15 days) | DWC (floating raft) | Tomato (Solanum lycopersicum) |
| [43] | Chicken manure | Aeration of water with manure placed in a biofilter (200, 300 or 400 g dry wt) and liquid compost (20 mL/L) within the hydroponic system | NFT | Lettuce (Lactuca sativa) |
| Study | Main Organic Materials | Main Method | Hydroponic System | Cultivated Plant |
|---|---|---|---|---|
| [44] | Broiler litter | Thermophilic (56 °C) anaerobic digestion (30-day HRT) | NFT 1 | Lettuce (Lactuca sativa) |
| [104] | Food and vegetable waste | Mesophilic anaerobic digestion (<36 °C) | DWC 2 | Silverbeet (Beta vulgaris L.) |
| [158] | Cow manure | Mesophilic anaerobic digestion (30 °C) | DWC | Tomato (Solanum lycopersicum) |
| [41] | Molasses, distillery slop, sugarcane leaves | Mesophilic anaerobic digestion (ambient temperature) for 30 days | NFT | Lettuce (Lactuca sativa) |
| [155] | Maize, silage, triticale silage, cow slurry, grape stalks | Anaerobic digestion; liquid digestate dilution as nutrient solution and solid digestate as substrate | DWC | Lettuce (Lactuca sativa) |
| Study | Main Organic Materials | Main Method | Hydroponic System | Cultivated Plant Species |
|---|---|---|---|---|
| [2] | Organic household and slaughterhouse waste, manure | (1) AD; (2) 14-day aeration of diluted digestate with bio-carriers; (3) Plant cultivation with digestate addition | NFT 1 | Pak Choi (Brassica campestris v. Chinensis) |
| [30] | Plant residues | (1) Mesophilic AD (80-day HRT); (2) Digestate aeration integrated or external to the hydroponic system with moving bed biofilm reactor (inoculated with active sludge); (3) Plant cultivation with digestate addition | NFT | Pak Choi (Brassica rapa v. Chinensis) |
| [105] | Pig manure | (1) Mesophilic AD (~37 °C, 10-day HRT); (2) Autoclavation of liquid digestate; (3) Digestate aeration integrated into the hydroponic system with a moving bed biofilm reactor; (4) Plant cultivation with digestate addition | Aeroponics (Low-pressure) | Tomato (Solanum lycopersicum) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szekely, I.; Jijakli, M.H. Bioponics as a Promising Approach to Sustainable Agriculture: A Review of the Main Methods for Producing Organic Nutrient Solution for Hydroponics. Water 2022, 14, 3975. https://doi.org/10.3390/w14233975
Szekely I, Jijakli MH. Bioponics as a Promising Approach to Sustainable Agriculture: A Review of the Main Methods for Producing Organic Nutrient Solution for Hydroponics. Water. 2022; 14(23):3975. https://doi.org/10.3390/w14233975
Chicago/Turabian StyleSzekely, Iris, and M. Haïssam Jijakli. 2022. "Bioponics as a Promising Approach to Sustainable Agriculture: A Review of the Main Methods for Producing Organic Nutrient Solution for Hydroponics" Water 14, no. 23: 3975. https://doi.org/10.3390/w14233975
APA StyleSzekely, I., & Jijakli, M. H. (2022). Bioponics as a Promising Approach to Sustainable Agriculture: A Review of the Main Methods for Producing Organic Nutrient Solution for Hydroponics. Water, 14(23), 3975. https://doi.org/10.3390/w14233975







