A Review of Recent Advances in Detection and Treatment Technology for Perfluorinated Compounds
Abstract
1. Introduction
2. The Hazards of PFCs
2.1. Pollution Status of PFCs
2.2. Toxicity Analysis of PFCs
3. Detection Techniques for PFCs
3.1. Sample Pretreatment Techniques for PFCs
3.1.1. Liquid–Liquid Extraction (LLE)
3.1.2. Solid-Phase Extraction (SPE)
3.1.3. Solid–Liquid Extraction (SLE)
3.1.4. Ultrasonic Extraction (USE)
3.1.5. Accelerated Solvent Extraction (ASE)
3.1.6. Microwave-Assisted Extraction
3.2. Detection Technologies for PFCs
3.2.1. Gas Chromatography–Tandem Mass Spectrometry (GC-MS/MS)
3.2.2. Liquid Chromatography–Tandem Mass Spectrometry (LC-MS/MS)
3.2.3. High Performance Liquid Chromatography–Tandem Mass Spectrometry (HPLC-MS/MS)
3.2.4. Ultra-Performance Liquid Chromatography–Tandem Mass Spectrometry (UPLC-MS/MS)
4. Treatment Technologies for PFCs
4.1. Physical Methods
4.1.1. Adsorption
4.1.2. Membrane Separation
4.2. Chemical Methods
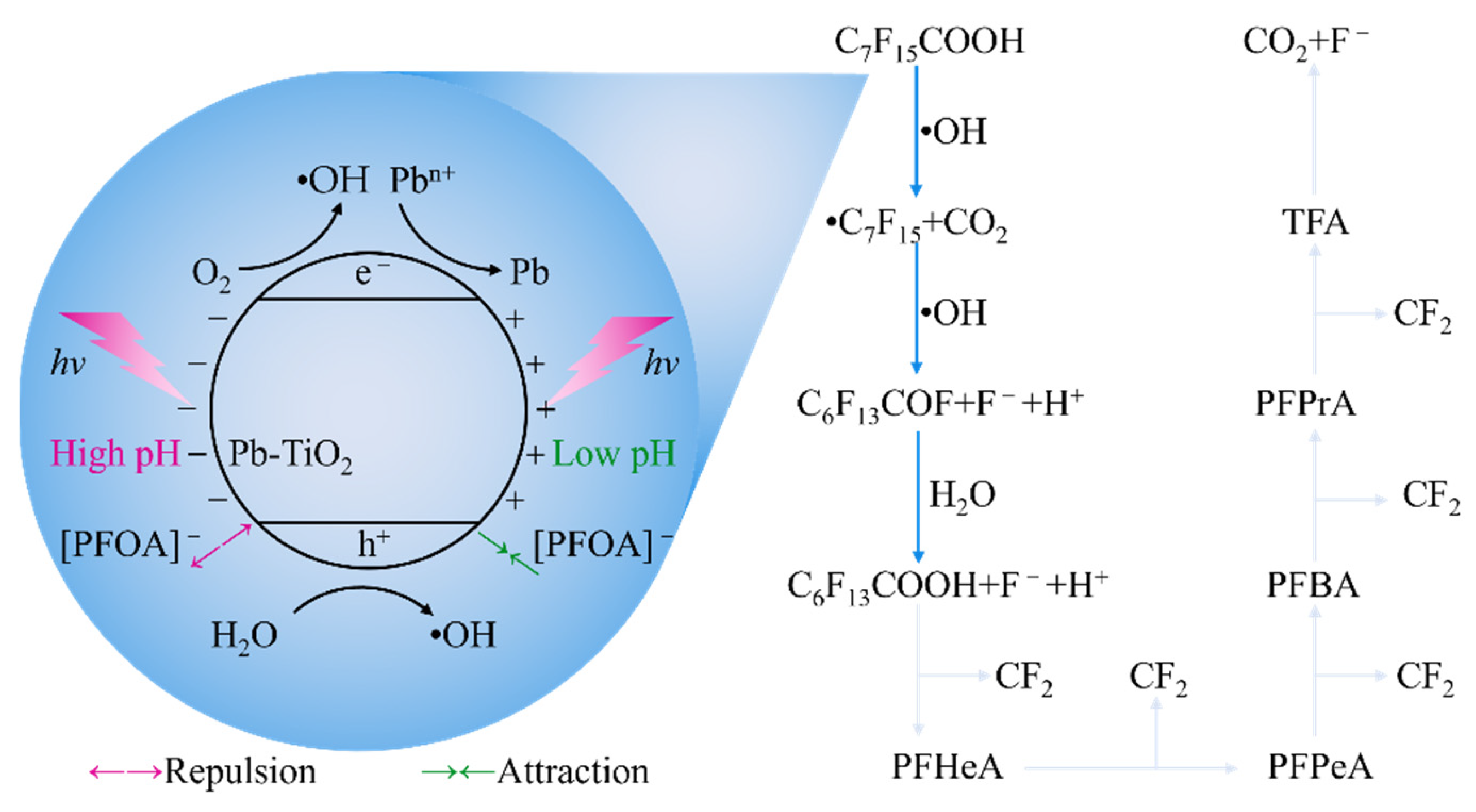
4.2.1. Photochemical Oxidation
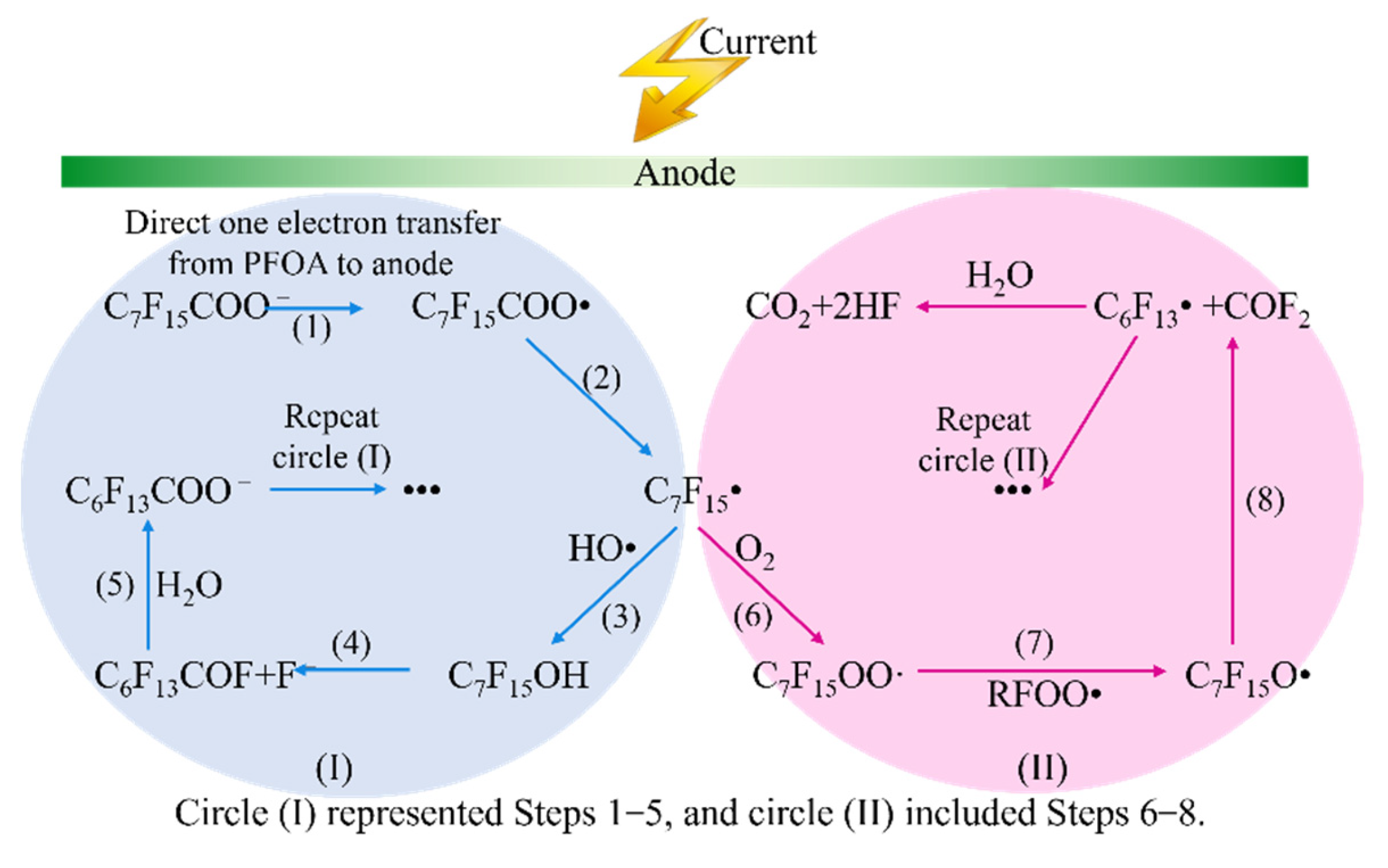
4.2.2. Electrochemical Oxidation
4.2.3. Sonochemical Oxidation
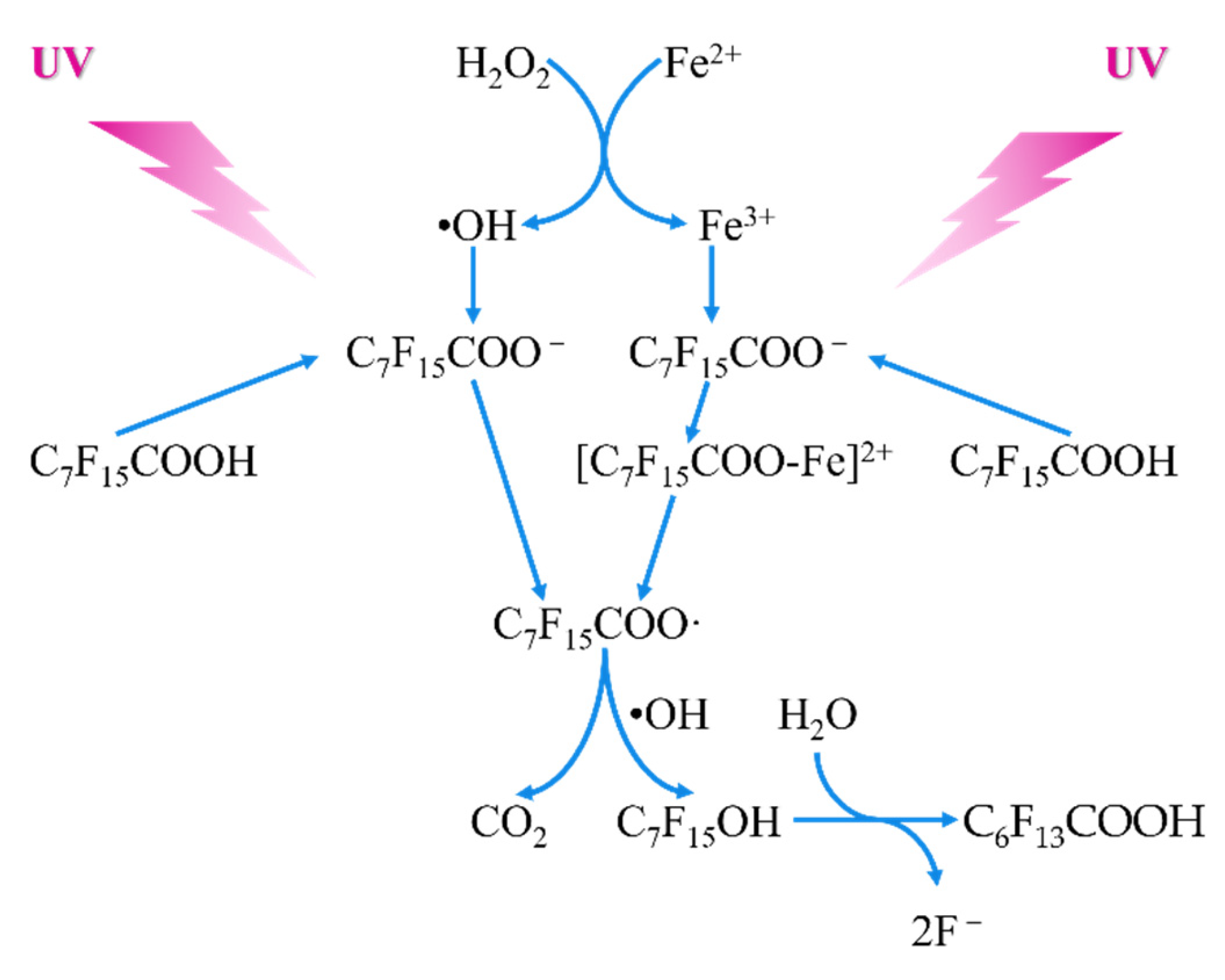
4.2.4. Fenton Oxidation
4.2.5. Other Chemical Methods
4.3. Biological Methods
5. Conclusions and Prospect
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Giesy, J.P.; Kannan, K. Perfluorochemical Surfactants in the Environment. Environ. Sci. Technol. 2002, 36, 146A–152A. [Google Scholar] [CrossRef] [PubMed]
- Senevirathna, S.; Tanaka, S.; Fujii, S.; Kunacheva, C.; Harada, H.; Shivakoti, B.R.; Okamoto, R. A Comparative Study of Adsorption of Perfluorooctane Sulfonate (Pfos) onto Granular Activated Carbon, Ion-Exchange Polymers and Non-Ion-Exchange Polymers. Chemosphere 2010, 80, 647–651. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Wang, Y.W.; Liao, C.Y.; Cai, Y.Q.; Jiang, G.B. Perspectives on the Inclusion of Perfluorooctane Sulfonate into the Stockholm Convention on Persistent Organic Pollutants. Environ. Sci. Technol. 2009, 43, 5171–5175. [Google Scholar] [CrossRef] [PubMed]
- Corsini, E.; Avogadro, A.; Galbiati, V.; dell’Agli, M.; Marinovich, M.; Galli, C.L.; Germolec, D.R. In Vitro Evaluation of the Immunotoxic Potential of Perfluorinated Compounds (Pfcs). Toxicol. Appl. Pharmacol. 2011, 250, 108–116. [Google Scholar] [CrossRef]
- Liu, B.; Zhang, H.; Yao, D.; Li, J.; Xie, L.; Wang, X.; Wang, Y.; Liu, G.; Yang, B. Perfluorinated Compounds (Pfcs) in the Atmosphere of Shenzhen, China: Spatial Distribution, Sources and Health Risk Assessment. Chemosphere 2015, 138, 511–518. [Google Scholar] [CrossRef]
- Yu, N.Y.; Shi, W.; Zhang, B.B.; Su, G.Y.; Feng, J.F.; Zhang, X.W.; Wei, S.; Yu, H.X. Occurrence of Perfluoroalkyl Acids Including Perfluorooctane Sulfonate Isomers in Huai River Basin and Taihu Lake in Jiangsu Province, China. Environ. Sci. Technol. 2013, 47, 710–717. [Google Scholar] [CrossRef]
- Thompson, J.; Eaglesham, G.; Reungoat, J.; Poussade, Y.; Bartkow, M.; Lawrence, M.; Mueller, J.F. Removal of Pfos, Pfoa and Other Perfluoroalkyl Acids at Water Reclamation Plants in South East Queensland Australia. Chemosphere 2011, 82, 9–17. [Google Scholar] [CrossRef]
- So, M.K.; Miyake, Y.; Yeung, W.Y.; Ho, Y.M.; Taniyasu, S.; Rostkowski, P.; Yamashita, N.; Zhou, B.S.; Shi, X.J.; Wang, J.X.; et al. Perfluorinated Compounds in the Pearl River and Yangtze River of China. Chemosphere 2007, 68, 2085–2095. [Google Scholar] [CrossRef]
- Skutlarek, D.; Exner, M.; Farber, H. Perfluorinated Surfactants in Surface and Drinking Water. Environ. Sci. Pollut. Res. 2006, 13, 299–307. [Google Scholar]
- Chen, S.; Jiao, X.C.; Gai, N.; Li, X.J.; Wang, X.C.; Lu, G.H.; Piao, H.T.; Rao, Z.; Yang, Y.L. Perfluorinated Compounds in Soil, Surface Water, and Groundwater from Rural Areas in Eastern China. Environ. Pollut. 2016, 211, 124–131. [Google Scholar] [CrossRef]
- Karaskova, P.; Venier, M.; Melymuk, L.; Becanova, J.; Vojta, S.; Prokes, R.; Diamond, M.L.; Klanova, J. Perfluorinated Alkyl Substances (Pfass) in Household Dust in Central Europe and North America. Environ. Int. 2016, 94, 315–324. [Google Scholar] [CrossRef]
- Yang, K.H.; Lin, Y.C.; Fang, M.D.; Wu, C.H.; Panchangam, S.C.; Hong, P.A.; Lin, C.F. Sorption of Perfluorooctanoic Acid (Pfoa) onto Sediment in the Presence of Dissolved Natural Organics. Sep. Sci. Technol. 2013, 48, 1473–1478. [Google Scholar] [CrossRef]
- Raymer, J.H.; Michael, L.C.; Studabaker, W.B.; Olsen, G.W.; Sloan, C.S.; Wilcosky, T.; Walmer, D.K. Concentrations of Perfluorooctane Sulfonate (Pfos) and Perfluorooctanoate (Pfoa) and Their Associations with Human Semen Quality Measurements. Reprod. Toxicol. 2012, 33, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.; Inoue, K.; Morikawa, A.; Yoshinaga, T.; Saito, N.; Koizumi, A. Renal Clearance of Perfluorooctane Sulfonate and Perfluorooctanoate in Humans and Their Species-Specific Excretion. Environ. Res. 2005, 99, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Bao, J.; Liu, W.; Liu, L.; Jin, Y.H.; Dai, J.Y.; Ran, X.R.; Zhang, Z.X.; Tsuda, S. Perfluorinated Compounds in the Environment and the Blood of Residents Living near Fluorochemical Plants in Fuxin, China. Environ. Sci. Technol. 2011, 45, 8075–8080. [Google Scholar] [CrossRef]
- Yu, J.; Hu, J.Y.; Tanaka, S.; Fujii, S. Perfluorooctane Sulfonate (Pfos) and Perfluorooctanoic Acid (Pfoa) in Sewage Treatment Plants. Water Res. 2009, 43, 2399–2408. [Google Scholar] [CrossRef]
- Loganathan, B.G.; Sajwan, K.S.; Sinclair, E.; Kumar, K.S.; Kannan, K. Perfluoroalkyl Sulfonates and Perfluorocarboxylates in Two Wastewater Treatment Facilities in Kentucky and Georgia. Water Res. 2007, 41, 4611–4620. [Google Scholar] [CrossRef]
- Mak, Y.L.; Taniyasu, S.; Yeung, L.W.Y.; Lu, G.H.; Jin, L.; Yang, Y.L.; Lam, P.K.S.; Kannan, K.; Yamashita, N. Perfluorinated Compounds in Tap Water from China and Several Other Countries. Environ. Sci. Technol. 2009, 43, 4824–4829. [Google Scholar] [CrossRef]
- O’Brien, J.M.; Crump, D.; Mundy, L.J.; Chu, S.; McLaren, K.K.; Vongphachan, V.; Letcher, R.J.; Kennedy, S.W. Pipping Success and Liver Mrna Expression in Chicken Embryos Exposed in Ovo to C8 and C11 Perfluorinated Carboxylic Acids and C10 Perfluorinated Sulfonate. Toxicol. Lett. 2009, 190, 134–139. [Google Scholar] [CrossRef]
- Kudo, N.; Bandai, N.; Suzuki, E.; Katakura, M.; Kawashima, Y. Induction by Perfluorinated Fatty Acids with Different Carbon Chain Length of Peroxisomal B-Oxidation in the Liver of Rats. Chem.-Biol. Interact. 2000, 124, 119–132. [Google Scholar] [CrossRef]
- Kmecick, M.; Vieira da Costa, M.C.; Oliveira Ribeiro, C.A.; Ortolani-Machado, C.F. Morphological Evidence of Neurotoxic Effects in Chicken Embryos after Exposure to Perfluorooctanoic Acid (Pfoa) and Inorganic Cadmium. Toxicology 2019, 427, 152286. [Google Scholar] [CrossRef] [PubMed]
- Mshaty, A.; Haijima, A.; Takatsuru, Y.; Ninomiya, A.; Yajima, H.; Kokubo, M.; Khairinisa, M.A.; Miyazaki, W.; Amano, I.; Koibuchi, N. Neurotoxic Effects of Lactational Exposure to Perfluorooctane Sulfonate on Learning and Memory in Adult Male Mouse. Food Chem. Toxicol. 2020, 145, 111710. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Fang, W.; Wang, D.; Gao, N.; Ding, Y.; Chen, C. The Role of Interleukin Family in Perfluorooctanoic Acid (Pfoa)-Induced Immunotoxicity. J. Hazard. Mater. 2014, 280, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Hong, Y.; Li, Z.; Yang, Z.; Lei, B.; Liu, J.; Cai, Z. Immunometabolism-Modulation and Immunotoxicity Evaluation of Perfluorooctanoic Acid in Macrophage. Ecotoxicol. Environ. Saf. 2021, 215, 112128. [Google Scholar] [CrossRef] [PubMed]
- Melzer, D.; Rice, N.; Depledge, M.H.; Henley, W.E.; Galloway, T.S. Association between Serum Perfluorooctanoic Acid (Pfoa) and Thyroid Disease in the Us National Health and Nutrition Examination Survey. Environ. Health Perspect. 2010, 118, 686–692. [Google Scholar] [CrossRef]
- Chaparro-Ortega, A.; Betancourt, M.; Rosas, P.; Vazquez-Cuevas, F.G.; Chavira, R.; Bonilla, E.; Casas, E.; Ducolomb, Y. Endocrine Disruptor Effect of Perfluorooctane Sulfonic Acid (Pfos) and Perfluorooctanoic Acid (Pfoa) on Porcine Ovarian Cell Steroidogenesis. Toxicol. In Vitro 2018, 46, 86–93. [Google Scholar] [CrossRef]
- Wang, M.Y.; Chen, J.F.; Lin, K.F.; Chen, Y.H.; Hu, W.; Tanguay, R.L.; Huang, C.J.; Dong, Q.X. Chronic Zebrafish Pfos Exposure Alters Sex Ratio and Maternal Related Effects in F1 Offspring. Environ. Toxicol. Chem. 2011, 30, 2073–2080. [Google Scholar] [CrossRef]
- Harada, K.; Xu, F.; Ono, K.; Iijima, T.; Koizumi, A. Effects of Pfos and Pfoa on L-Type Ca2+ Currents in Guinea-Pig Ventricular Myocytes. Biochem. Biophys. Res. Commun. 2005, 329, 487–494. [Google Scholar] [CrossRef]
- Valsecchi, S.; Conti, D.; Crebelli, R.; Polesello, S.; Rusconi, M.; Mazzoni, M.; Preziosi, E.; Carere, M.; Lucentini, L.; Ferretti, E.; et al. Deriving Environmental Quality Standards for Perfluorooctanoic Acid (Pfoa) and Related Short Chain Perfluorinated Alkyl Acids. J. Hazard. Mater. 2017, 323 Pt A, 84–98. [Google Scholar] [CrossRef]
- So, M.K.; Taniyasu, S.; Yamashita, N.; Giesy, J.P.; Zheng, J.; Fang, Z.; Im, S.H.; Lam, P.K.S. Perfluorinated Compounds in Coastal Waters of Hong Kong, South China, and Korea. Environ. Sci. Technol. 2004, 38, 4056–4063. [Google Scholar] [CrossRef]
- Simcik, M.F.; Dorweiler, K.J. Ratio of Perfluorochemical Concentrations as a Tracer of Atmospheric Deposition to Surface Waters. Environ. Sci. Technol. 2005, 39, 8678–8683. [Google Scholar] [CrossRef] [PubMed]
- Armitage, J.M.; Macleod, M.; Cousins, I.T. Comparative Assessment of the Global Fate and Transport Pathways of Long-Chain Perfluorocarboxylic Acids (Pfcas) and Perfluorocarboxylates (Pfcs) Emitted from Direct Sources. Environ. Sci. Technol. 2009, 43, 5830–5836. [Google Scholar] [CrossRef] [PubMed]
- Kirchgeorg, T.; Dreyer, A.; Gabrieli, J.; Kehrwald, N.; Sigl, M.; Schwikowski, M.; Boutron, C.; Gambaro, A.; Barbante, C.; Ebinghaus, R. Temporal Variations of Perfluoroalkyl Substances and Polybrominated Diphenyl Ethers in Alpine Snow. Environ. Pollut. 2013, 178, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, K.K.; Murugasamy, M.; Nikhil, N.P.; Elaiyaraja, A.; Sampath, S.; Krishnamoorthi, V.; He, H.; Ramaswamy, B.R. Investigation of Distribution, Sources and Flux of Perfluorinated Compounds in Major Southern Indian Rivers and Their Risk Assessment. Chemosphere 2021, 277, 130228. [Google Scholar] [CrossRef]
- Joyce Dinglasan-Panlilio, M.; Prakash, S.S.; Baker, J.E. Perfluorinated Compounds in the Surface Waters of Puget Sound, Washington and Clayoquot and Barkley Sounds, British Columbia. Mar. Pollut. Bull. 2014, 78, 173–180. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, L.; Yang, Q.; Li, G.; Zhang, F. Occurrence and Spatial Distribution of Perfluorinated Compounds in Groundwater Receiving Reclaimed Water through River Bank Infiltration. Chemosphere 2018, 211, 1203–1211. [Google Scholar] [CrossRef]
- Bossi, R.; Vorkamp, K.; Skov, H. Concentrations of Organochlorine Pesticides, Polybrominated Diphenyl Ethers and Perfluorinated Compounds in the Atmosphere of North Greenland. Environ. Pollut. 2016, 217, 4–10. [Google Scholar] [CrossRef]
- Gan, C.D.; Gan, Z.W.; Cui, S.F.; Fan, R.J.; Fu, Y.Z.; Peng, M.Y.; Yang, J.Y. Agricultural Activities Impact on Soil and Sediment Fluorine and Perfluorinated Compounds in an Endemic Fluorosis Area. Sci. Total Environ. 2021, 771, 144809. [Google Scholar] [CrossRef]
- Taniyasu, S.; Kannan, K.; Horii, Y.; Hanari, N.; Yamashita, N. A Survey of Perfluorooctane Sulfonate and Related Perfluorinated Organic Compounds in Water, Fish, Birds, and Humans from Japan. Environ. Sci. Technol. 2003, 37, 2634–2639. [Google Scholar] [CrossRef]
- Kadar, H.; Veyrand, B.; Barbarossa, A.; Pagliuca, G.; Legrand, A.; Bosher, C.; Boquien, C.Y.; Durand, S.; Monteau, F.; Antignac, J.P.; et al. Development of an Analytical Strategy Based on Liquid Chromatography-High Resolution Mass Spectrometry for Measuring Perfluorinated Compounds in Human Breast Milk: Application to the Generation of Preliminary Data Regarding Perinatal Exposure in France. Chemosphere 2011, 85, 473–480. [Google Scholar] [CrossRef]
- Corsini, E.; Luebke, R.W.; Germolec, D.R.; DeWitt, J.C. Perfluorinated Compounds: Emerging Pops with Potential Immunotoxicity. Toxicol. Lett. 2014, 230, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.; Thibodeaux, J.R.; Hanson, R.G.; Rogers, J.M.; Grey, B.E.; Stanton, M.E.; Butenhoff, J.L.; Stevenson, L.A. Exposure to Perfluorooctane Sulfonate During Pregnancy in Rat and Mouse. Ii: Postnatal Evaluation. Toxicol. Sci. 2003, 74, 382–392. [Google Scholar] [CrossRef] [PubMed]
- Johansson, N.; Fredriksson, A.; Eriksson, P. Neonatal Exposure to Perfluorooctane Sulfonate (Pfos) and Perfluorooctanoic Acid (Pfoa) Causes Neurobehavioural Defects in Adult Mice. Neurotoxicology 2008, 29, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, Q.; Liu, C.; Li, C.; Li, Y.; Li, S.; Liu, X.; Shao, J. Evaluation of Pfos-Mediated Neurotoxicity in Rat Primary Neurons and Astrocytes Cultured Separately or in Co-Culture. Toxicol. In Vitro 2017, 38, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Wang, R.; Niu, X.; Cheng, Y.; Shang, X.; Li, Y.; Li, S.; Liu, X.; Shao, J. Role of Astrocytes-Derived D-Serine in Pfos-Induced Neurotoxicity through Nmdars in the Rat Primary Hippocampal Neurons. Toxicology 2019, 422, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Dewitt, J.C.; Copeland, C.B.; Strynar, M.J.; Luebke, R.W. Perfluorooctanoic Acid-Induced Immunomodulation in Adult C57bl/6j or C57bl/6n Female Mice. Environ. Health Perspect. 2008, 116, 644–650. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.Y.; Wang, Z.X.; Tian, Y.; Gao, Y. Advances on Immunotoxicities Induced by Per- and Polyfluoroalkylated Substances. J. Environ. Occup. Med. 2022, 39, 223–235. [Google Scholar]
- Hoffman, K.; Vieira, V.; Webster, T.; White, R. Exposure to Polyfluoroalkyl Chemicals and Attention Deficit Hyperactivity Disorder in Us Children Aged 12-15 Years. Epidemiology 2009, 20, S70. [Google Scholar] [CrossRef]
- Fei, C.Y.; McLaughlin, J.K.; Tarone, R.E.; Olsen, J. Fetal Growth Indicators and Perfluorinated Chemicals: A Study in the Danish National Birth Cohort. Am. J. Epidemiol. 2008, 168, 66–72. [Google Scholar] [CrossRef]
- Fei, C.Y.; McLaughlin, J.K.; Lipworth, L.; Olsen, J. Maternal Levels of Perfluorinated Chemicals and Subfecundity. Hum. Reprod. 2009, 24, 1200–1205. [Google Scholar] [CrossRef]
- Xia, W.; Wan, Y.J.; Li, Y.Y.; Zeng, H.C.; Lv, Z.Q.; Li, G.Q.; Wei, Z.Z.; Xu, S.Q. Pfos Prenatal Exposure Induce Mitochondrial Injury and Gene Expression Change in Hearts of Weaned Sd Rats. Toxicology 2011, 282, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Lafuente, A.; Pereiro, N.; Gomez-Limia, L. Hypothalamic-Pituitary-Adrenal Axis Activity Is Modified by Perfluorooctane Sulfonate (Pfos) Exposure in Adult Male Rat. Toxicol. Lett. 2013, 221, S108. [Google Scholar] [CrossRef]
- Wu, X.P.; Ma, Z.Y.; Li, Z.H.; Zhang, W.Z. Research into the Endocrine Disrupting Effects of Perfluorinated Compounds. J. Toxicol. 2021, 35, 436–439. [Google Scholar]
- Kranthi Kumar, K.; Devi, B.U.; Neeraja, P. Integration of in Silico Approaches to Determination of Endocrine-Disrupting Perfluorinated Chemicals Binding Potency with Steroidogenic Acute Regulatory Protein. Biochem. Biophys. Res. Commun. 2017, 491, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Dolman, S.; Pelzing, M. An Optimized Method for the Determination of Perfluorooctanoic Acid, Perfluorooctane Sulfonate and Other Perfluorochemicals in Different Matrices Using Liquid Chromatography/Ion-Trap Mass Spectrometry. J. Chromatogr. B-Anal. Technol. Biomed. Life Sci. 2011, 879, 2043–2050. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.F.; Chen, R.J.; Shatner, W.; Cao, Y.; Bai, Y. State-of-the-Art on the Technique of Dispersive Liquid-Liquid Microextraction. Ultrason. Sonochemistry 2019, 51, 369–377. [Google Scholar] [CrossRef]
- Wu, J.X.; Zhi, S.W.; Jia, C.H.; Li, X.H.; Zhu, X.D.; Zhao, E.C. Dispersive Solid-Phase Extraction Combined with Dispersive Liquid-Liquid Microextraction for Simultaneous Determination of Seven Succinate Dehydrogenase Inhibitor Fungicides in Watermelon by Ultra High Performance Liquid Chromatography with Tandem Mass Spectrometry. J. Sep. Sci. 2019, 42, 3688–3696. [Google Scholar]
- Concha-Grana, E.; Fernandez-Martinez, G.; Lopez-Mahia, P.; Prada-Rodriguez, D.; Muniategui-Lorenzo, S. Fast and Sensitive Determination of Per- and Polyfluoroalkyl Substances in Seawater. J. Chromatogr. A 2018, 1555, 62–73. [Google Scholar] [CrossRef]
- Yan, M.M.; Chen, M.; Meng, X.S.; Guo, X.Y.; Bai, H.; Ma, Q.; Wang, P.L.; Lei, H.M. Determination of Migration of Perfluorooctanoic Acid and Perfluorooctane Sulfonic Acid in Food Contact Materials by Ionic Liquid Dispersion Liquid-Liquid Microextraction Ultra High Performance Liquid Chromatography Tandem Mass Spectrometry. Chin. J. Chromatogr. 2018, 36, 738–744. [Google Scholar] [CrossRef]
- Mazzoni, M.; Rusconi, M.; Valsecchi, S.; Martins, C.P.B.; Polesello, S. An on-Line Solid Phase Extraction-Liquid Chromatography-Tandem Mass Spectrometry Method for the Determination of Perfluoroalkyl Acids in Drinking and Surface Waters. J. Anal. Methods Chem. 2015, 2015, 942016. [Google Scholar] [CrossRef]
- He, X.W.; He, Y.; Huang, S.; Fang, Z.F.; Liu, J.J.; Ma, M.; Chen, B. Fluoro-Functionalized Paper-Based Solid-Phase Extraction for Analysis of Perfluorinated Compounds by High-Performance Liquid Chromatography Coupled with Electrospray Ionization-Tandem Mass Spectrometry. J. Chromatogr. A 2019, 1601, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Hansen, K.J.; Johnson, H.O.; Eldridge, J.S.; Butenhoff, J.L.; Dick, L.A. Quantitative Characterization of Trace Levels of Pfos and Pfoa in the Tennessee River. Environ. Sci. Technol. 2002, 36, 1681–1685. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Lv, Y.B.; Chen, H.J.; Tan, L.; Teng, E.J. Simultaneous Determination of 14 Short Chain and Long Chain Perfluorocarbons in Water by Solid Phase Extraction Liquid Chromatography Tandem Mass Spectrometry. Chin. J. Chromatogr. 2014, 32, 919–925. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Z.; Sun, H.W.; Wang, L.; Yang, J. Determination of Perfluorocarbons in Sewage by Solid Phase Extraction High Performance Liquid Chromatography Tandem Mass Spectrometry. Environ. Chem. 2009, 28, 944–945. [Google Scholar]
- Wu, J.J.; Gui, P.; Guo, F.Q. Determination of 17 Perfluorinated Compounds in Water by Solid Phase Extraction-Uplc-Ms/Ms Method. China Water Supply Drain. 2022, 38, 28–36. [Google Scholar]
- Wang, J.M.; Wang, L.; Feng, Y.J.; Pan, Y.Y.; Shi, Y.L.; Cai, Y.Q. Analysis of Perfluorinated Compounds in Animal Viscera and Muscle Tissues by Liquid Chromatography-Mass Spectrometry. Food Sci. 2010, 31, 127–131. [Google Scholar]
- Pan, Y.Y.; Shi, Y.L.; Cai, Y.Q. Determination of 9 Perfluorinated Compounds in Aquatic Products: Fish and Shellfish. Chin. J. Anal. Chem. 2008, 36, 1619–1623. [Google Scholar]
- Gonzalez-Barreiro, C.; Martinez-Carballo, E.; Sitka, A.; Scharf, S.; Gans, O. Method Optimization for Determination of Selected Perfluorinated Alkylated Substances in Water Samples. Anal. Bioanal. Chem. 2006, 386, 2123–2132. [Google Scholar] [CrossRef]
- Wang, H.; Li, X.; Shao, M.Y.; Lin, L.; Mu, T.N.; Liu, Y.Q. Simultaneous Determination of 9 Environmental Pollutants Including Bisphenol a in Vegetable Oil by Solid Phase Extraction-Liquid Chromatography-Tandem Mass Spectrometry. Anal. Methods 2021, 13, 3527–3534. [Google Scholar] [CrossRef]
- Boone, J.S.; Guan, B.; Vigo, C.; Boone, T.; Byrne, C.; Ferrario, J. A Method for the Analysis of Perfluorinated Compounds in Environmental and Drinking Waters and the Determination of Their Lowest Concentration Minimal Reporting Levels. J. Chromatogr. A 2014, 1345, 68–77. [Google Scholar] [CrossRef]
- Monteleone, M.; Naccarato, A.; Sindona, G.; Tagarelli, A. A Rapid and Sensitive Assay of Perfluorocarboxylic Acids in Aqueous Matrices by Headspace Solid Phase Microextraction-Gas Chromatography-Triple Quadrupole Mass Spectrometry. J. Chromatogr. A 2012, 1251, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Z.; Huang, L.Y.; Liao, S.F. Determination of Perfluorooctane Sulfonic Acid and Perfluorooctanoic Acid in Leather by Ultra High Performance Liquid Chromatography Tandem Mass Spectrometry. Phys. Chem. Exam. (Chem.Vol.) 2011, 47, 160–162+65. [Google Scholar]
- Ballesteros-Gomez, A.; Rubio, S.; van Leeuwen, S. Tetrahydrofuran-Water Extraction, in-Line Clean-up and Selective Liquid Chromatography/Tandem Mass Spectrometry for the Quantitation of Perfluorinated Compounds in Food at the Low Picogram Per Gram Level. J. Chromatogr. A 2010, 1217, 5913–5921. [Google Scholar] [CrossRef] [PubMed]
- Anaya-Esparza, L.M.; Ramos-Aguirre, D.; Zamora-Gasga, V.M.; Yahia, E.; Montalvo-Gonzalez, E. Optimization of Ultrasonic-Assisted Extraction of Phenolic Compounds from Justicia Spicigera Leaves. Food Sci. Biotechnol. 2018, 27, 1093–1102. [Google Scholar] [CrossRef] [PubMed]
- He, J.C.; Zhang, S.Y.; Su, Y.Y.; Song, J.Y.; Wu, F.H. Progress on the Sample Techniques and Analytical Methods for Typical Perfluorinated Organic Acids. Chin. J. Chromatogr. 2020, 38, 86–94. [Google Scholar]
- Pan, C.G.; Xiao, S.K.; Yu, K.F.; Wu, Q.; Wang, Y.H. Legacy and Alternative Per- and Polyfluoroalkyl Substances in a Subtropical Marine Food Web from the Beibu Gulf, South China: Fate, Trophic Transfer and Health Risk Assessment. J. Hazard. Mater. 2021, 403, 10. [Google Scholar] [CrossRef]
- Begley, T.H.; White, K.; Honigfort, P.; Twaroski, M.L.; Neches, R.; Walker, R.A. Perfluorochemicals: Potential Sources of and Migration from Food Packaging. Food Addit. Contam. Part A-Chem. Anal. Control. Expo. Risk Assess. 2005, 22, 1023–1031. [Google Scholar] [CrossRef]
- Chen, H.M.; Cheng, Y.; Chen, W.; Yu, W.L.; Li, X.; Wang, Z. Determination of Perfluorooctane Sulfonic Acid and Its Salts in Foam Fire Extinguishing Materials and Other Materials by High Performance Liquid Chromatography Tandem Mass Spectrometry. Chin. J. Chromatogr. 2010, 28, 185–189. [Google Scholar] [CrossRef]
- Martinez-Moral, M.P.; Tena, M.T. Focused Ultrasound Solid-Liquid Extraction of Perfluorinated Compounds from Sewage Sludge. Talanta 2013, 109, 197–202. [Google Scholar] [CrossRef]
- Deans, B.J.; Just, J.; Smith, J.A.; Bissember, A.C. Development and Applications of Water-Based Extraction Methods in Natural Products Isolation Chemistry. Asian J. Org. Chem. 2020, 9, 1144–1153. [Google Scholar] [CrossRef]
- Fortier, C.A.; Cintron, M.S.; Peralta, D.; von Hoven, T.; Fontenot, K.; Rodgers, J.E.; Delhom, C. A Comparison of the Accelerated Solvent Extraction Method to the Soxhlet Method in the Extraction of Cotton Fiber Wax. Aatcc J. Res. 2019, 6, 15–20. [Google Scholar] [CrossRef]
- Li, B.J.; Chen, J.Y.; Liu, Z.Z.; Wang, J.; He, S.C. Pollution Characteristics and Health Risk Assessment of Perfluorinated Compounds in Pm2. 5 in Zhejiang Province. Environ. Sci. 2022, 43, 639–648. [Google Scholar]
- Olkowska, E.; Polkowska, Z.; Namiesnik, J. Analytical Procedures for the Determination of Surfactants in Environmental Samples. Talanta 2012, 88, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.P.; Lu, L.J.; Zhang, D.H.; Mou, J.; Zhou, X.; Li, A.J.; Han, D.C.; Rong, H. Determination of Pfos and Pfoa in Textiles by Liquid Chromatography-Tandem Mass Spectrometry. J. Text. 2008, 5, 80–83. [Google Scholar]
- Cheng, Y.; Chen, H.M.; Chen, W.; Cui, Y.; Li, X.J.; Chen, D.D.; Li, S.J.; Yu, W.L.; Zhou, X. Determination of Perfluorooctane Sulfonates (Pfos) in Four Chemical Materials by Hplc/Ms/Ms. In Proceedings of the 2nd International Conference on Chemical Engineering and Advanced Materials (CEAM 2012), Guangzhou, China, 13–15 July 2012. [Google Scholar]
- Yadav, N.; Sharma, S.; Joys, J.S.; Kumar, S. Microwave Assisted Extraction of Bioactive Compounds: A Brief Review. J. Indian Chem. Soc. 2020, 97, 1751–1756. [Google Scholar]
- Agrawal, P.; Laddha, K. Extraction of Colchicine from Gloriosa Superba Tubers: A Comparison of Conventional and Microwave-Assisted Extraction. J. Microw. Power Electromagn. Energy 2019, 53, 57–66. [Google Scholar] [CrossRef]
- Ye, Q.; Chen, Z.B. Analysis of Perfluorinated Compounds in Environmental Water Using Decyl-Perfluorinated Magnetic Mesoporous Microspheres as Magnetic Solid-Phase Extraction Materials and Microwave-Assisted Derivatization Followed by Gas Chromatography-Mass Spectrometry. J. Chromatogr. Sci. 2018, 56, 955–961. [Google Scholar] [CrossRef]
- He, X.L.; Sun, M.X. Determination of Perfluorooctanoic Acid in Textiles by Gc-Ms. Print. Dye. 2010, 36, 36–42. [Google Scholar]
- Fedorova, G.; Golovko, O.; Randak, T.; Grabic, R. Passive Sampling of Perfluorinated Acids and Sulfonates Using Polar Organic Chemical Integrative Samplers. Environ. Sci. Pollut. Res. 2013, 20, 1344–1351. [Google Scholar] [CrossRef]
- Rudel, H.; Muller, J.; Jurling, H.; Bartel-Steinbach, M.; Koschorreck, J. Survey of Patterns, Levels, and Trends of Perfluorinated Compounds in Aquatic Organisms and Bird Eggs from Representative German Ecosystems. Environ. Sci. Pollut. Res. 2011, 18, 1457–1470. [Google Scholar] [CrossRef]
- Golebiowski, M.; Siedlecka, E.; Paszkiewicz, M.; Brzozowski, K.; Stepnowski, P. Perfluorocarboxylic Acids in Cell Growth Media and Technologically Treated Waters: Determination with Gc and Gc-Ms. J. Pharm. Biomed. Anal. 2011, 54, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, P.; Han, Y.; Han, D.; Yan, H. Rapid and Inexpensive Nylon-66-Filter Solid-Phase Extraction Followed by Gas Chromatography Tandem Mass Spectrometry for Analyzing Perfluorinated Carboxylic Acids in Milk. J. Chromatogr. A 2022, 1677, 463288. [Google Scholar] [CrossRef] [PubMed]
- Lv, G.; Wang, L.B.; Liu, S.C.; Li, S.F. Determination of Perfluorinated Compounds in Packaging Materials and Textiles Using Pressurized Liquid Extraction with Gas Chromatography-Mass Spectrometry. Anal. Sci. 2009, 25, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Kim, D.; Park, Y.K. Survey of Perfluorinated Compounds in Consumer Products by Liquid Chromatography-Tandem Mass Spectrometry. Energy Environ. 2020, 31, 713–729. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Y.; Li, F.W.; Zhao, R.S. Carboxylated Carbon Nanospheres as Solid-Phase Extraction Adsorbents for the Determination of Perfluorinated Compounds in Water Samples by Liquid Chromatography Tandem Mass Spectrometry (Vol 178, Pg 129, 2018). Talanta Int. J. Pure Appl. Anal. Chem. 2018, 180, 410. [Google Scholar] [CrossRef]
- Zou, D.; Li, P.; Yang, C.; Han, D.; Yan, H. Rapid Determination of Perfluorinated Compounds in Pork Samples Using a Molecularly Imprinted Phenolic Resin Adsorbent in Dispersive Solid Phase Extraction-Liquid Chromatography Tandem Mass Spectrometry. Anal. Chim. Acta 2022, 1226, 340271. [Google Scholar] [CrossRef]
- Zabaleta, I.; Bizkarguenaga, E.; Prieto, A.; Ortiz-Zarragoitia, M.; Fernandez, L.A.; Zuloaga, O. Simultaneous Determination of Perfluorinated Compounds and Their Potential Precursors in Mussel Tissue and Fish Muscle Tissue and Liver Samples by Liquid Chromatography-Electrospray-Tandem Mass Spectrometry. J. Chromatogr. A 2015, 1387, 13–23. [Google Scholar] [CrossRef]
- Liu, X.; Yu, Y.; Li, Y.; Zhang, H.; Ling, J.; Sun, X.; Feng, J.; Duan, G. Fluorocarbon-Bonded Magnetic Mesoporous Microspheres for the Analysis of Perfluorinated Compounds in Human Serum by High-Performance Liquid Chromatography Coupled to Tandem Mass Spectrometry. Anal. Chim. Acta 2014, 844, 35–43. [Google Scholar] [CrossRef]
- He, J.L.; Peng, T.; Xie, J.; Dai, H.H.; Chen, D.D.; Yue, Z.F.; Fan, C.L.; Li, C. Determination of 20 Perfluorinated Compounds in Animal Liver by Hplc-Ms/Ms. Chin. J. Anal. Chem. 2015, 43, 40–48. [Google Scholar] [CrossRef]
- Cavaliere, C.; Capriotti, A.L.; Ferraris, F.; Foglia, P.; Samperi, R.; Ventura, S.; Lagana, A. Multiresidue Analysis of Endocrine-Disrupting Compounds and Perfluorinated Sulfates and Carboxylic Acids in Sediments by Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry. J. Chromatogr. A 2016, 1438, 133–142. [Google Scholar] [CrossRef]
- Li, S.; Ma, J.; Wu, G.; Li, J.; Ostovan, A.; Song, Z.; Wang, X.; Chen, L. Determination of Anionic Perfluorinated Compounds in Water Samples Using Cationic Fluorinated Metal Organic Framework Membrane Coupled with Uhplc-Ms/Ms. J. Hazard. Mater. 2022, 429, 128333. [Google Scholar] [CrossRef] [PubMed]
- Onghena, M.; Moliner-Martinez, Y.; Pico, Y.; Campins-Falco, P.; Barcelo, D. Analysis of 18 Perfluorinated Compounds in River Waters: Comparison of High Performance Liquid Chromatography-Tandem Mass Spectrometry, Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry and Capillary Liquid Chromatography-Mass Spectrometry. J. Chromatogr. A 2012, 1244, 88–97. [Google Scholar] [PubMed]
- Yu, Q.; Zhang, R.Q.; Deng, S.B.; Huang, J.; Yu, G. Sorption of Perfluorooctane Sulfonate and Perfluorooctanoate on Activated Carbons and Resin: Kinetic and Isotherm Study. Water Res. 2009, 43, 1150–1158. [Google Scholar] [CrossRef] [PubMed]
- Meng, P.; Fang, X.; Maimaiti, A.; Yu, G.; Deng, S. Efficient Removal of Perfluorinated Compounds from Water Using a Regenerable Magnetic Activated Carbon. Chemosphere 2019, 224, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Shih, K.M. Adsorption of Perfluorooctanesulfonate (Pfos) and Perfluorooctanoate (Pfoa) on Alumina: Influence of Solution Ph and Cations. Water Res. 2011, 45, 2925–2930. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Liu, C.S.; Shih, K.M. Adsorption Behavior of Perfluorooctanesulfonate (Pfos) and Perfluorooctanoate (Pfoa) on Boehmite. Chemosphere 2012, 89, 1009–1014. [Google Scholar] [CrossRef]
- Zaggia, A.; Conte, L.; Falletti, L.; Fant, M.; Chiorboli, A. Use of Strong Anion Exchange Resins for the Removal of Perfluoroalkylated Substances from Contaminated Drinking Water in Batch and Continuous Pilot Plants. Water Res. 2016, 91, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Xia, X.H.; Wang, X.L.; Qiao, J.P.; Chen, H.T. A Comparative Study on Sorption of Perfluorooctane Sulfonate (Pfos) by Chars, Ash and Carbon Nanotubes. Chemosphere 2011, 83, 1313–1319. [Google Scholar] [CrossRef]
- Deng, S.B.; Zhang, Q.Y.; Nie, Y.; Wei, H.R.; Wang, B.; Huang, J.; Yu, G.; Xing, B.S. Sorption Mechanisms of Perfluorinated Compounds on Carbon Nanotubes. Environ. Pollut. 2012, 168, 138–144. [Google Scholar] [CrossRef]
- Bei, Y.; Deng, S.B.; Du, Z.W.; Wang, B.; Huang, J.; Yu, G. Adsorption of Perfluorooctane Sulfonate on Carbon Nanotubes: Influence of Ph and Competitive Ions. Water Sci. Technol. 2014, 69, 1489–1495. [Google Scholar] [CrossRef]
- Du, Z.W.; Deng, S.B.; Chen, Y.G.; Wang, B.; Huang, J.; Wang, Y.J.; Yu, G. Removal of Perfluorinated Carboxylates from Washing Wastewater of Perfluorooctanesulfonyl Fluoride Using Activated Carbons and Resins. J. Hazard. Mater. 2015, 286, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.B.; Nie, Y.; Du, Z.W.; Huang, Q.; Meng, P.P.; Wang, B.; Huang, J.; Yu, G. Enhanced Adsorption of Perfluorooctane Sulfonate and Perfluorooctanoate by Bamboo-Derived Granular Activated Carbon. J. Hazard. Mater. 2015, 282, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Ochoa-Herrera, V.; Sierra-Alvarez, R. Removal of Perfluorinated Surfactants by Sorption onto Granular Activated Carbon, Zeolite and Sludge. Chemosphere 2008, 72, 1588–1593. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.H.; Liu, Y.; Tang, W.J.; Wang, W.Q.; Tang, Y.H.; Xu, B.; Deng, J.J.; Jia, G.R.; Wang, J.; Wei, W.; et al. Progress of Research on Treatment Technology of Perfluorinated Compounds in Typical Industrial Wastewater. China Environ. Sci. 2021, 41, 1109–1118. [Google Scholar]
- Tang, C.Y.Y.; Fu, Q.S.; Robertson, A.P.; Criddle, C.S.; Leckie, J.O. Use of Reverse Osmosis Membranes to Remove Perfluorooctane Sulfonate (Pfos) from Semiconductor Wastewater. Environ. Sci. Technol. 2006, 40, 7343–7349. [Google Scholar] [CrossRef]
- Tang, C.Y.; Fu, Q.S.; Criddle, C.S.; Leckie, J.O. Effect of Flux (Transmembrane Pressure) and Membrane Properties on Fouling and Rejection of Reverse Osmosis and Nanofiltration Membranes Treating Perfluorooctane Sulfonate Containing Wastewater. Environ. Sci. Technol. 2007, 41, 2008–2014. [Google Scholar] [CrossRef]
- Pramanik, B.K.; Pramanik, S.K.; Sarker, D.C.; Suja, F. Removal of Emerging Perfluorooctanoic Acid and Perfluorooctane Sulfonate Contaminants from Lake Water. Environ. Technol. 2017, 38, 1937–1942. [Google Scholar] [CrossRef]
- Liu, F.; Hua, L.; Zhang, W. Influences of Microwave Irradiation on Performances of Membrane Filtration and Catalytic Degradation of Perfluorooctanoic Acid (Pfoa). Environ. Int. 2020, 143, 105969. [Google Scholar] [CrossRef]
- Hori, H.; Hayakawa, E.; Einaga, H.; Kutsuna, S.; Koike, K.; Ibusuki, T.; Kiatagawa, H.; Arakawa, R. Decomposition of Environmentally Persistent Perfluorooctanoic Acid in Water by Photochemical Approaches. Environ. Sci. Technol. 2004, 38, 6118–6124. [Google Scholar] [CrossRef]
- Yamamoto, T.; Noma, Y.; Sakai, S.I.; Shibata, Y. Photodegradation of Perfluorooctane Sulfonate by Uv Irradiation in Water and Alkaline 2-Propanol. Environ. Sci. Technol. 2007, 41, 5660–5665. [Google Scholar] [CrossRef]
- Ochiai, T.; Iizuka, Y.; Nakata, K.; Murakami, T.; Tryk, D.A.; Koide, Y.; Morito, Y.; Fujishima, A. Efficient Decomposition of Perfluorocarboxylic Acids in Aqueous Suspensions of a Tio2 Photocatalyst with Medium-Pressure Ultraviolet Lamp Irradiation under Atmospheric Pressure. Ind. Eng. Chem. Res. 2011, 50, 10943–10947. [Google Scholar] [CrossRef]
- Chen, M.J.; Lo, S.L.; Lee, Y.C.; Kuo, J.; Wu, C.H. Decomposition of Perfluorooctanoic Acid by Ultraviolet Light Irradiation with Pb-Modified Titanium Dioxide. J. Hazard. Mater. 2016, 303, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Taniyasu, S.; Yamashita, N.; Yamazaki, E.; Petrick, G.; Kannan, K. The Environmental Photolysis of Perfluorooctanesulfonate, Perfluorooctanoate, and Related Fluorochemicals. Chemosphere 2013, 90, 1686–1692. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.D.; Xiu, Z.M.; Liu, F.; Wu, G.; Adamson, D.; Newell, C.; Vikesland, P.; Tsai, A.L.; Alvarez, P.J. Perfluorooctanoic Acid Degradation in the Presence of Fe(Iii) under Natural Sunlight. J. Hazard. Mater. 2013, 262, 456–463. [Google Scholar] [CrossRef]
- Xu, L.; Lin, Q.F.; Li, C.; Wei, G.Z.J.; Ji, J. Current Situation of Typical Perfluorinated Compounds Pollution and Its Treatment Technology Progress. China Water Wastewater 2022, 38, 56–62. [Google Scholar]
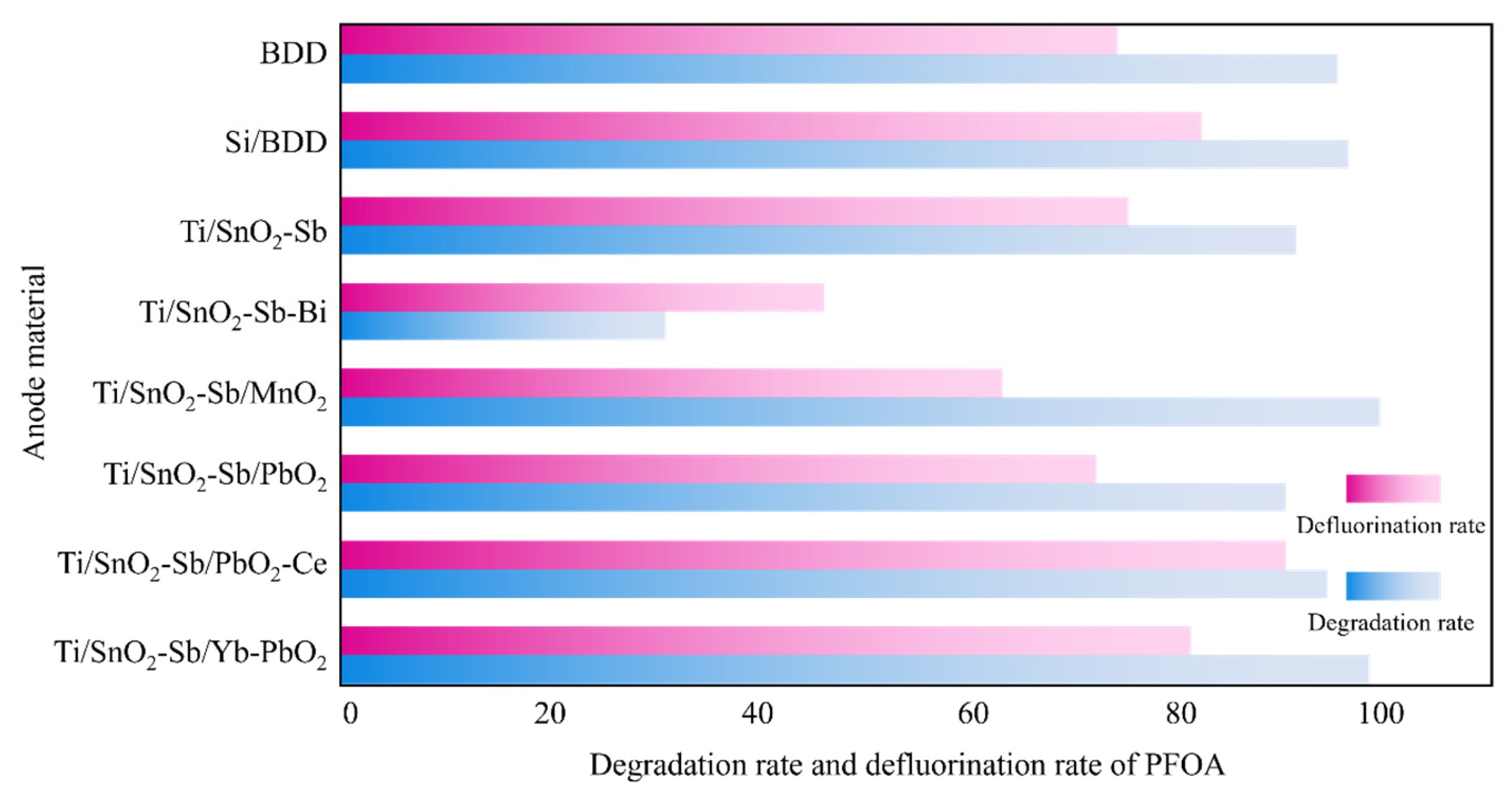
- Ma, Q.C.; Liu, L.; Cui, W.; Li, R.F.; Song, T.T.; Cui, Z.J. Electrochemical Degradation of Perfluorooctanoic Acid (Pfoa) by Yb-Doped Ti/Sno2-Sb/Pbo2 Anodes and Determination of the Optimal Conditions. Rsc Adv. 2015, 5, 84856–84864. [Google Scholar] [CrossRef]
- Zhuo, Q.F.; Deng, S.B.; Yang, B.; Huang, J.; Wang, B.; Zhang, T.T.; Yu, G. Degradation of Perfluorinated Compounds on a Boron-Doped Diamond Electrode. Electrochim. Acta 2012, 77, 17–22. [Google Scholar] [CrossRef]
- Zhuo, Q.F.; Deng, S.B.; Yang, B.; Huang, J.; Yu, G. Efficient Electrochemical Oxidation of Perfluorooctanoate Using a Ti/Sno2-Sb-Bi Anode. Environ. Sci. Technol. 2011, 45, 2973–2979. [Google Scholar] [CrossRef]
- Rodriguez-Freire, L.; Balachandran, R.; Sierra-Alvarez, R.; Keswani, M. Effect of Sound Frequency and Initial Concentration on the Sonochemical Degradation of Perfluorooctane Sulfonate (Pfos). J. Hazard. Mater. 2015, 300, 662–669. [Google Scholar] [CrossRef]
- Cheng, J.; Vecitis, C.D.; Park, H.; Mader, B.T.; Hoffmann, M.R. Sonochemical Degradation of Perfluorooctane Sulfonate (Pfos) and Perfluorooctanoate (Pfoa) in Landfill Groundwater: Environmental Matrix Effects. Environ. Sci. Technol. 2008, 42, 8057–8063. [Google Scholar] [CrossRef]
- Cheng, J.; Vecitis, C.D.; Park, H.; Mader, B.T.; Hoffmann, M.R. Sonochemical Degradation of Perfluorooctane Sulfonate (Pfos) and Perfluorooctanoate (Pfoa) in Groundwater: Kinetic Effects of Matrix Inorganics. Environ. Sci. Technol. 2010, 44, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Moriwaki, H.; Takagi, Y.; Tanaka, M.; Tsuruho, K.; Okitsu, K.; Maeda, Y. Sonochemical Decomposition of Perfluorooctane Sulfonate and Perfluorooctanoic Acid. Environ. Sci. Technol. 2005, 39, 3388–3392. [Google Scholar] [CrossRef]
- Mitchell, S.M.; Ahmad, M.; Teel, A.L.; Watts, R.J. Degradation of Perfluorooctanoic Acid by Reactive Species Generated through Catalyzed H2o2 Propagation Reactions. Environ. Sci. Technol. Lett. 2014, 1, 117–121. [Google Scholar] [CrossRef]
- Tang, H.Q.; Xiang, Q.Q.; Lei, M.; Yan, J.C.; Zhu, L.H.; Zou, J. Efficient Degradation of Perfluorooctanoic Acid by Uv-Fenton Process. Chem. Eng. J. 2012, 184, 156–162. [Google Scholar] [CrossRef]
- Santos, A.; Rodriguez, S.; Pardo, F.; Romero, A. Use of Fenton Reagent Combined with Humic Acids for the Removal of Pfoa from Contaminated Water. Sci. Total Environ. 2016, 563–564, 657–663. [Google Scholar] [CrossRef]
- Li, S.; Zhang, G.S.; Zhang, W.; Zheng, H.S.; Zhu, W.Y.; Sun, N.; Zheng, Y.J.; Wang, P. Microwave Enhanced Fenton-Like Process for Degradation of Perfluorooctanoic Acid (Pfoa) Using Pb-Bifeo3/Rgo as Heterogeneous Catalyst. Chem. Eng. J. 2017, 326, 756–764. [Google Scholar] [CrossRef]
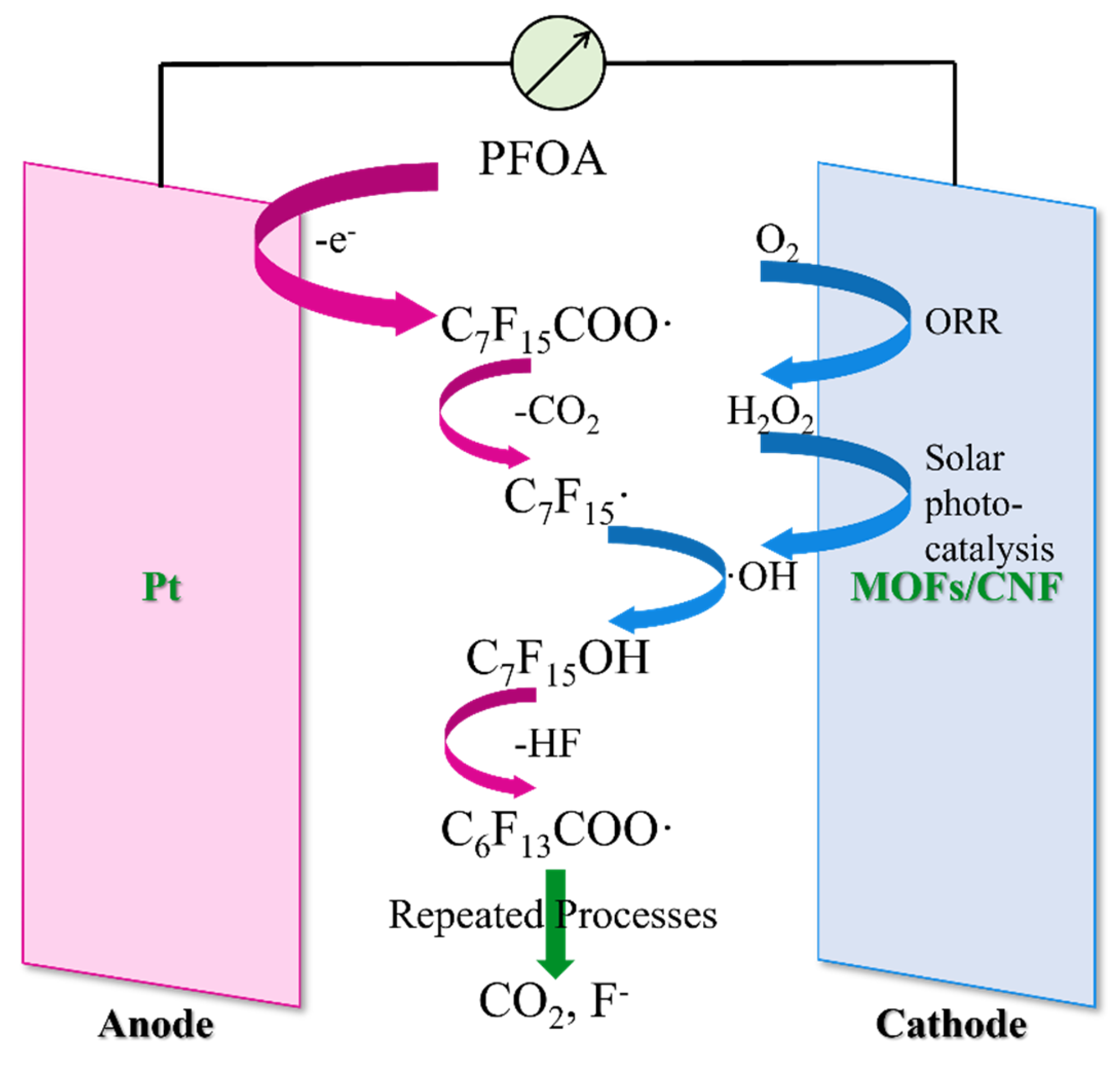
- Wang, Y.; Zhao, M.Z.; Hou, C.; Chen, W.Q.; Li, S.S.; Ren, R.K.; Li, Z.J. Efficient Degradation of Perfluorooctanoic Acid by Solar Photo-Electro-Fenton Like System Fabricated by Mofs/Carbon Nanofibers Composite Membrane. Chem. Eng. J. 2021, 414, 128940. [Google Scholar] [CrossRef]
- Deng, S.B.; Zhou, Q.; Yu, G.; Huang, J.; Fan, Q. Removal of Perfluorooctanoate from Surface Water by Polyaluminium Chloride Coagulation. Water Res. 2011, 45, 1774–1780. [Google Scholar] [CrossRef]
- Bao, Y.P.; Niu, J.F.; Xu, Z.S.; Gao, D.; Shi, J.H.; Sun, X.M.; Huang, Q.G. Removal of Perfluorooctane Sulfonate (Pfos) and Perfluorooctanoate (Pfoa) from Water by Coagulation: Mechanisms and Influencing Factors. J. Colloid Interface Sci. 2014, 434, 59–64. [Google Scholar] [CrossRef]
- Lin, A.Y.C.; Panchangam, S.C.; Chang, C.Y.; Hong, P.K.A.; Hsueh, H.F. Removal of Perfluorooctanoic Acid and Perfluorooctane Sulfonate Via Ozonation under Alkaline Condition. J. Hazard. Mater. 2012, 243, 272–277. [Google Scholar] [CrossRef]
- Liu, C.S.; Higgins, C.P.; Wang, F.; Shih, K. Effect of Temperature on Oxidative Transformation of Perfluorooctanoic Acid (Pfoa) by Persulfate Activation in Water. Sep. Purif. Technol. 2012, 91, 46–51. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, P.Y. Photocatalytic Decomposition of Perfluorooctanoic Acid (Pfoa) by TiO2 in the Presence of Oxalic Acid. J. Hazard. Mater. 2011, 192, 1869–1875. [Google Scholar] [CrossRef] [PubMed]
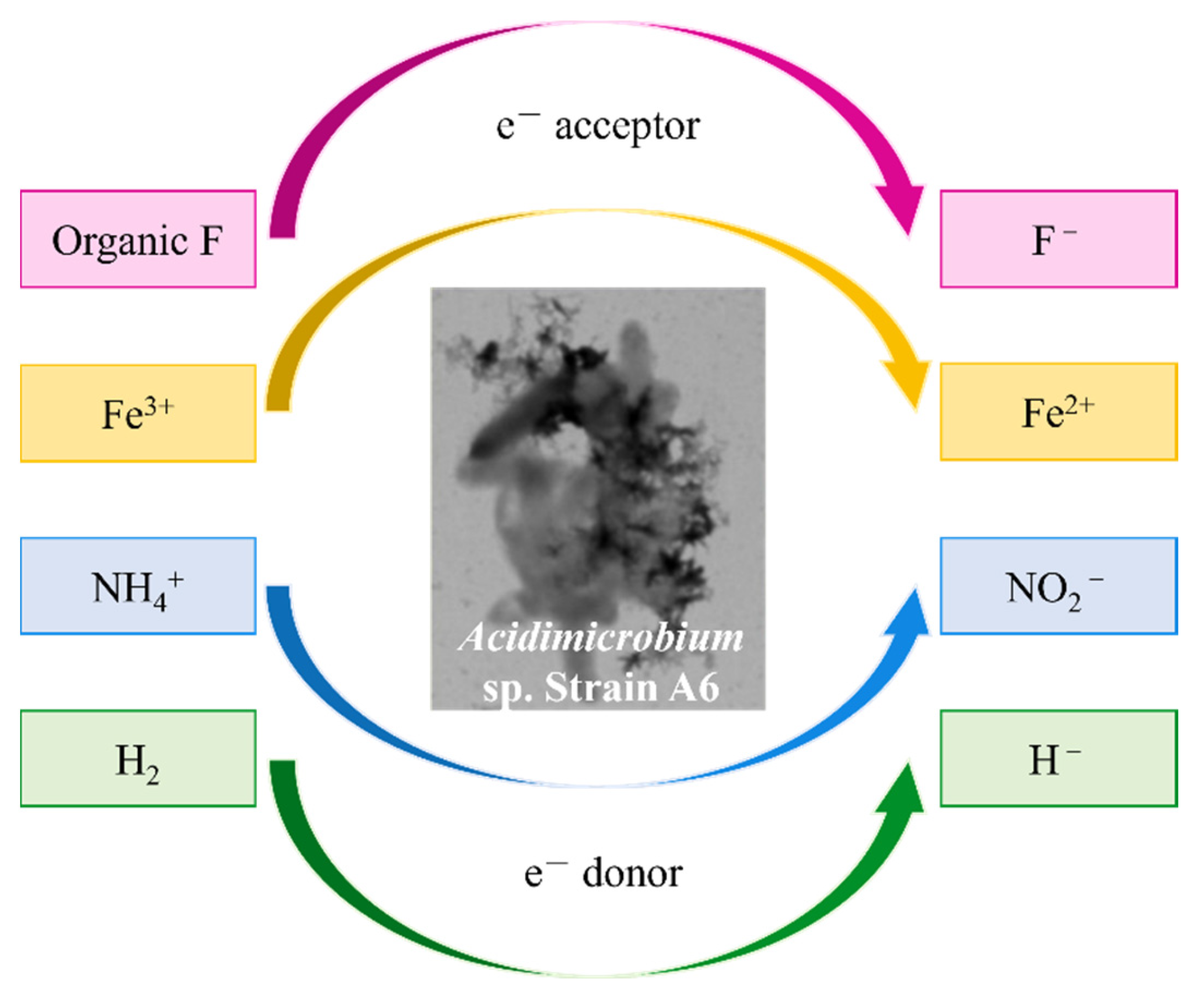
- Huang, S.; Jaffe, P.R. Defluorination of Perfluorooctanoic Acid (Pfoa) and Perfluorooctane Sulfonate (Pfos) by Acidimicrobium Sp. Strain A6. Environ. Sci. Technol. 2019, 53, 11410–11419. [Google Scholar] [CrossRef]
- Harris, J.D.; Coon, C.M.; Doherty, M.E.; McHugh, E.A.; Warner, M.C.; Walters, C.L.; Orahood, O.M.; Loesch, A.E.; Hatfield, D.C.; Sitko, J.C.; et al. Engineering and Characterization of Dehalogenase Enzymes from Delftia Acidovorans in Bioremediation of Perfluorinated Compounds. Synth. Syst. Biotechnol. 2022, 7, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Albert, K.; Hsieh, P.Y.; Chen, T.H.; Hou, C.H.; Hsu, H.Y. Diatom-Assisted Biomicroreactor Targeting the Complete Removal of Perfluorinated Compounds. J. Hazard. Mater. 2020, 384, 11. [Google Scholar] [CrossRef]
- He, W.B.; Zhang, H.X.; Lin, Z.W.; Hu, W.X. Optimization of the Determination Method of Perfluorooctane Sulfonate and Perfluorooctanoate in Leather. China Leather 2012, 41, 30–33. [Google Scholar]
- Yao, Y.; Volchek, K.; Brown, C.E.; Robinson, A.; Obal, T. Comparative Study on Adsorption of Perfluorooctane Sulfonate (Pfos) and Perfluorooctanoate (Pfoa) by Different Adsorbents in Water. Water Sci. Technol. 2014, 70, 1983–1991. [Google Scholar] [CrossRef]
| Substrates | PFCs | Pre-Processing Methods | Detection Methods | Limit of Detection or Limit of Quantitation (LOD/LOQ) | Recovery (%) | Ref. |
|---|---|---|---|---|---|---|
| Sea water | PFOA, PFOS and PFOS precursors | Vortex assisted liquid-liquid microextraction | HPLC-LTQ Orbitrap HRMS | 0.7–6 ng/L (LOQ) | 95–100 | [55] |
| Food contact material | PFOA, PFOS | Ionic liquid dispersion liquid-liquid extraction | UPLC-MS/MS | PFOA: 0.5 μg/L (LOD); 2 μg/L (LOQ) PFOS: 1 μg/L (LOD); 5 μg/L (LOQ) | 86.4–100 | [56] |
| Experiment water | PFBA, PFOA, PFOS and PFDA | Solid phase extraction | HPLC-MS/MS | 0.04–0.05 ng/L (LOD) 0.15~0.20 ng/L (LOQ) | 94.5–100 | [57] |
| Animal Muscle | PFOA, PFOS and other 11 PFCs | Alkali digestion-Solid phase extraction | HPLC-ESI-MS/MS | 0.002–0.032 μg/L (LOD) | 80–100 | [58] |
| Fish and shellfish | PFOA, PFOS and other 9 PFCs | Alkali digestion-Solid phase extraction | HPLC-MS/MS | 0.16–2.0 ng/g (LOD) | 97.6–100 | [59] |
| Experiment water | PFOA, PFNA and other 7 PFCs | Headspace solid phase microextraction | GC-MS | 0.08~6.6 ng/L (LOD) 0.17~14.3 ng/L (LOQ) | 84.4–100 | [60] |
| Leather | PFOA, PFOS | Soxhlet extraction | UPLC-MS/MS | 1.0 μg/L (LOD) | PFOA: 91.6–100 PFOS: 90.0–99.4 | [61] |
| Animal food | PFOA, PFNA and other 14 PFCs | Solid-liquid extraction | HPLC-MS/MS | 1–30 pg/g (LOD) 2.5–60 pg/g (LOQ) | 88–100 | [62] |
| Sewage Sludge | PFOA, PFOS | Focused ultrasonic solid-liquid extraction | UPLC-MS/MS | 0.2 ng/g (LOD) | 69–100 | [63] |
| Nonstick coating | PFOS | Accelerated solvent extraction | HPLC-MS/MS | 0.4 μg/m2 (LOD) | 93.4–100 | [64] |
| Textile | PFOA | Microwave ultrasonic synergistic extraction | GC-MS | 0.088~0.132 μg/mL (LOD) | 60–100 | [65] |
| Milk | PFOA, PFNA and other 5 PFCs | Solid phase extraction | GC-MS/MS | 4–18 ng/kg (LOQ) | 81–100 | [66] |
| Pork | PFOA, PFNA and Perfluoro decanoic acid (PFDA) | Solid phase extraction | LC-MS/MS | 0.011–0.08 ng/g (LOD) | 89.3–100 | [67] |
| Mussel | PFOA, PFOS and other 14 PFCs | Focused ultrasonic solid-liquid extraction | LC-MS/MS | 0.1–3.8 ng/g (LOD) | 59–100 | [68] |
| Fish Liver | 0.1–2.7 ng/g (LOD) | 65–100 | ||||
| Fish meat | 0.2–3.1 ng/g (LOD) | 67–100 | ||||
| Animal liver | PFOA, PFNA and other 20 PFCs | Solid-liquid extraction | HPLC-MS/MS | 0.05–0.2 μg/kg (LOD) 0.4–0.5 μg/kg (LOQ) | 70.3–100 | [69] |
| Reservoir water | PFOA, PFOS and other 12 PFCs | Solid phase extraction | UPLC-MS/MS | 0.03–0.48 ng/L (LOD) | 51.1–100 | [70] |
| River water | PFOA, PFOS and other 18 PFCs | Solid phase extraction | UPLC-MS/MS | 0.002–4 ng/L (LOD) 0.006–12 ng/L (LOQ) | 55–94 | [71] |
| Leather | PFOA, PFOS | Ultrasonic extraction | UPLC-MS/MS | 0.25 μg/g (LOD) | PFOA: 89.2–100 PFOS: 86.4–96.2 | [72] |
| PFCs | Concentration | Adsorbents | Dosages | Balance Time | Adsorption Isotherms | Ref. | |
|---|---|---|---|---|---|---|---|
| Langmuir Constants | Freundlich Constants | ||||||
| PFOS | 50 mg/L | GAC | 0.1 g/L | 168 h | KL = 39 L/mmol, qm = 0.37 mmol/g | KF = 0.43 (mmol1−nLn/g), n = 0.18 | [105] |
| PAC | 0.1 g/L | 4 h | KL = 55 L/mmol, qm = 1.04 mmol/g | KF = 1.27 (mmol1−nLn/g), n = 0.18 | |||
| Anion exchange resin AI400 | 0.1 g/L | 168 h | KL = 69 L/mmol, qm = 0.42 mmol/g | KF = 0.52 (mmol1−nLn/g), n = 0.17 | |||
| PFOA | 50 mg/L | GAC | 0.1 g/L | 168 h | KL = 18 L/mmol, qm = 0.39 mmol/g | KF = 0.47 (mmol1−nLn/g), n = 0.28 | |
| PAC | 0.1 g/L | 4 h | KL = 69 L/mmol, qm = 0.67 mmol/g | KF = 0.83 (mmol1−nLn/g), n = 0.20 | |||
| Anion exchange resin AI400 | 0.1 g/L | 168 h | KL = 69 L/mmol, qm = 2.92 mmol/g | KF = 3.35 (mmol1−nLn/g), n = 0.13 | |||
| PFOS | 100 μg/L | alumina | 10 g/L | 48 h | KL = 0.0587 L/μg, qm = 0.252 μg/m2 | KF = 0.0400 (μg/m2)(μg/L)−n, n = 0.398 | [107] |
| PFOA | 100 μg/L | alumina | 10 g/L | 48 h | KL = 0.00908 L/μg, qm = 0.157 μg/m2 | KF = 0.00239 (μg/m2)(μg/L)−n, n = 0.772 | |
| PFOS | 200 μg/L | boehmite | 5 g/L | 48 h | KL = 0.00622 L/μg, qm = 0.877 μg/m2 | KF = 0.0139 (μg/m2)(μg/L)−n, n = 0.678 | [108] |
| PFOA | 200 μg/L | boehmite | 5 g/L | 48 h | KL = 0.00512 L/μg, qm = 0.633 μg/m2 | KF = 0.0155 (μg/m2)(μg/L)−n, n = 0.559 | |
| PFOS | 100 mg/L | W400 | 0.2–1.2 g/L | 384 h | KL = 0.010 L/mg, qm = 91.6 mg/g | KF = 5.23 (mg/g)(mg/L)−n, n = 0.492 | [110] |
| MA | 0.2–1.2 g/L | 48 h | KL = 0.012 L/mg, qm = 811 mg/g | KF = 26.8 (mg/g)(mg/L)−n, n = 0.571 | |||
| SWCNT | 0.2–1.2 g/L | 2 h | KL = 0.044 L/mg, qm = 712 mg/g | KF = 122 (mg/g)(mg/L)−n, n = 0.324 | |||
| PFOA | 0.5 mM | SWCNT | 0.2 g/L | 15 h | - | KF = 0.259 (mmol1−nLn/g), n = 0.391 | [111] |
| PFOS | 0.5 mM | SWCNT | 0.2 g/L | 15 h | - | KF = 1.605 (mmol1−nLn/g), n = 0.402 | |
| PFHxS | 0.5 mM | SWCNT | 0.2 g/L | 15 h | - | KF = 157.1 (mmol1−nLn/g), n = 1.504 | |
| PFHxA | 0.5 mM | SWCNT | 0.2 g/L | 15 h | - | KF = 0.054 (mmol1−nLn/g), n = 0.298 | |
| PFBS | 0.5 mM | SWCNT | 0.2 g/L | 15 h | - | KF = 5.887 (mmol1−nLn/g), n = 1.539 | |
| PFBA | 0.5 mM | SWCNT | 0.2 g/L | 15 h | - | KF = 0.065 (mmol1−nLn/g), n = 0.597 | |
| PFOA | 0.5 μM | MWCNTs-Pri | 50 mg/L | - | KL = 12.98 L/mmol, qm = 1.18 mmol/g | KF = 1.56 (mmol1−nLn/g), n = 2.29 | [112] |
| MWCNTs-COOH | 50 mg/L | - | KL = 13.39 L/mmol, qm = 1.54 mmol/g | KF = 2.05 (mmol1−nLn/g), n = 2.30 | |||
| MWCNTs-OH | 50 mg/L | - | KL = 11.58 L/mmol, qm = 1.85 mmol/g | KF = 2.47 (mmol1−nLn/g), n = 2.19 | |||
| PFOS | 100 mg/L | BAC | 0.1 g/L | 24 h | KL = 52.2 L/mmol, qm = 2.20 mmol/g | KF = 3.20 (mmol1−nLn/g), n = 3.41 | [113] |
| PFOA | 81 mg/L | BAC | 0.1 g/L | 24 h | KL = 157.1 L/mmol, qm = 1.03 mmol/g | KF = 1.34 (mmol1−nLn/g), n = 5.28 | |
| PFOS | 50 mg/L | GAC | 1 g/L | - | KL = 0.124 L/mg, qm = 236.4 mg/g | KF = 60.9 (mg/g)(mg/L)−n, n = 0.289 | [114] |
| PFOA | 50 mg/L | GAC | 1 g/L | - | KL = 0.038 L/mg, qm = 112.1 mg/g | KF = 11.8 (mg/g)(mg/L)−n, n = 0.443 | |
| PFBS | 50 mg/L | GAC | 1 g/L | - | KL = 0.034 L/mg, qm = 98.70 mg/g | KF = 9.3 (mg/g)(mg/L)−n, n = 0.463 | |
| PFOS | 50 mg/L | AER | 2 g/L | 2 h | - | KF = 7300 (mg/g)(mg/L)−n, n = 0.90 | [115] |
| GAC | 2 g/L | 24 h | - | KF = 120 (mg/g)(mg/L)−n, n = 0.29 | |||
| PFOA | 50 mg/L | AER | 2 g/L | 2 h | - | KF = 200 (mg/g)(mg/L)−n, n = 0.97 | |
| GAC | 2 g/L | 24 h | - | KF = 63 (mg/g)(mg/L)−n, n = 0.35 | |||
| Methods | PFCs | Concentration | pH | Temperature | Time | Rate Constant | Removal Degree | Defluorination Degree | Mineralization Degree | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| Photochemistry | PFOA | 1.35 mM | - | - | 72 h | - | 89.5% | 95.9% | - | [122] |
| PFOS | 40 μM | - | 36–46 ℃ | 10 d | - | 68% | - | - | [123] | |
| PFOA | 50 mg/L | 3 | 25 ℃ | 15 min | 0.514 h−1 | 99.9% | 22.4% | - | [124] | |
| PFOA | 20 mg/L | 4.6 | - | 28 d | - | 97.8 ± 1.7% | 12.7 ± 0.5% | - | [125] | |
| Electrochemical | PFOA | 100 mg/L | 5 | 25 ℃ | 2.5 h | 1.158 h−1 | 95.11 ± 3.9% | 75.7 ± 2.8% | - | [126] |
| PFOA | 50 mg/L | 3 | 32 ℃ | 1.5 h | 2.568 h−1 | 100% | 60% | - | [127] | |
| PFOA | 50 mg/L | 32 ℃ | 2 h | 1.930 h−1 | 99% | 63.8% | - | [128] | ||
| Sonochemical | PFOS | 100 μM | - | 30 ℃ | 5.5 h | - | - | 100% | 55.8% | [129] |
| PFOS | 10 mg/L | 4.8 | 20 ℃ | 1 h | 0.960 h−1 | 60% | - | - | [130] | |
| PFOA | 10 mg/L | 4.7 | 20 ℃ | 1 h | 1.920 h−1 | 85% | - | - | ||
| Fenton | PFOA | 100 μg/L | 3.5 | 20 ± 2 ℃ | 2.5 h | - | 89% | - | - | [131] |
| PFOA | 20 μM | 3 | - | 5 h | - | 100% | 53.2% | - | [132] | |
| PFOA | 50 mg/L | 5.0 | - | 5 min | - | 95% | - | 50% | [133] | |
| PFOA | 20 mg/L | 3 | - | 2 h | 1.600 h−1 | 99% | - | 91% | [134] | |
| Ozonation | PFOS | 50 μg/L | 11 | 25 ℃ | 4 h | 0.602 h−1 | 85% | - | - | [135] |
| PFOA | 50 μg/L | 11 | 25 ℃ | 4 h | 0.753 h−1 | 90% | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Guo, J.; Sumita; Shi, C.; Zhu, Q.; Li, C.; Pang, W. A Review of Recent Advances in Detection and Treatment Technology for Perfluorinated Compounds. Water 2022, 14, 3919. https://doi.org/10.3390/w14233919
Wang Y, Guo J, Sumita, Shi C, Zhu Q, Li C, Pang W. A Review of Recent Advances in Detection and Treatment Technology for Perfluorinated Compounds. Water. 2022; 14(23):3919. https://doi.org/10.3390/w14233919
Chicago/Turabian StyleWang, Yong, Jiaqi Guo, Sumita, Changjie Shi, Qijia Zhu, Cong Li, and Weihai Pang. 2022. "A Review of Recent Advances in Detection and Treatment Technology for Perfluorinated Compounds" Water 14, no. 23: 3919. https://doi.org/10.3390/w14233919
APA StyleWang, Y., Guo, J., Sumita, Shi, C., Zhu, Q., Li, C., & Pang, W. (2022). A Review of Recent Advances in Detection and Treatment Technology for Perfluorinated Compounds. Water, 14(23), 3919. https://doi.org/10.3390/w14233919






