Unraveling Physical and Chemical Effects of Textile Microfibers
Abstract
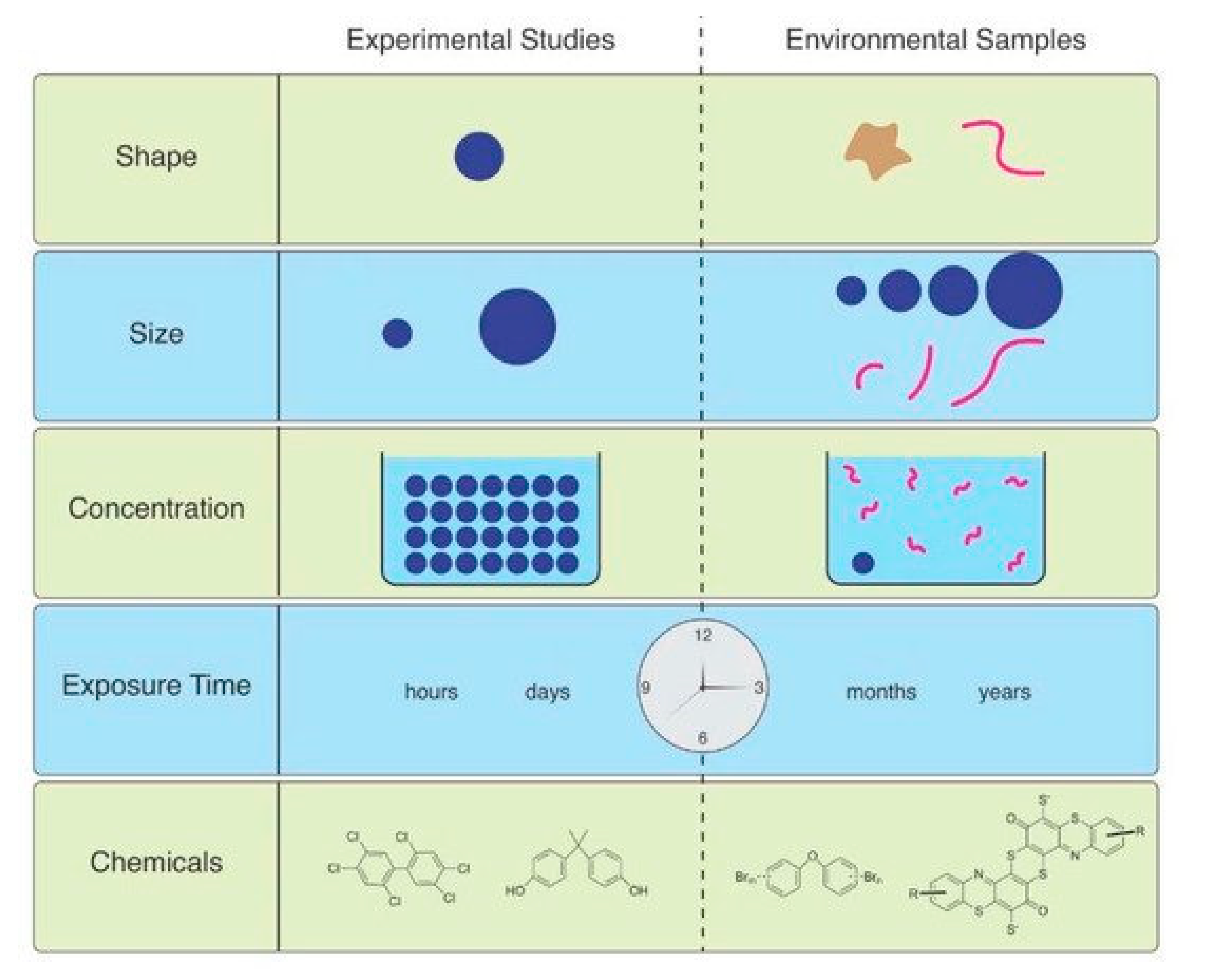
1. Introduction
2. Effects of Microfibers
2.1. Synthetic Fibers
2.2. Semi-Synthetic and Natural Fibers
3. Microfibers as Vectors for Chemical Exposure
3.1. Chemical Usage and Accumulation on Textile Fibers
3.2. Microfiber-Mediated Chemical Release and Exposure
3.3. Toxicity of Virgin vs. ‘Weathered’ Particles
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Athey, S.N. Are We Underestimating Anthropogenic Microfiber Pollution? A Critical Review of Occurrence, Methods, and Reporting. Environ. Toxicol. Chem. 2022, 41, 822–837. [Google Scholar] [CrossRef] [PubMed]
- Neil.Mccoy. Report on Microfiber Pollution|OR&R’s Marine Debris Program. Available online: https://marinedebris.noaa.gov/interagency-marine-debris-coordinating-committee-reports/report-microfiber-pollution (accessed on 19 October 2022).
- Gavigan, J.; Kefela, T.; Macadam-Somer, I.; Suh, S.; Geyer, R. Synthetic Microfiber Emissions to Land Rival Those to Waterbodies and Are Growing. PLoS ONE 2020, 15, e0237839. [Google Scholar] [CrossRef] [PubMed]
- Carr Sources and Dispersive Modes of Micro-Fibers in the Environment. Available online: https://setac.onlinelibrary.wiley.com/doi/full/10.1002/ieam.1916?casa_token=FCfELnV8uAcAAAAA%3AkrTIGXVJFXmiatMPNuy2IDWmEbo__BPBBQ8zSFqyLEviqExcqOaEvS9NIeQ4QK-0LDqwJTWOaX3bFA (accessed on 5 October 2022).
- Textile Exchange. Preferred Fiber and Materials Market Report 2021; Textile Exchange: Lamesa, TX, USA, 2021. [Google Scholar]
- Ladewig, S.M.; Bao, S.; Chow, A.T. Natural Fibers: A Missing Link to Chemical Pollution Dispersion in Aquatic Environments. Environ. Sci. Technol. 2015, 49, 12609–12610. [Google Scholar] [CrossRef]
- Chan, C.K.M.; Park, C.; Chan, K.M.; Mak, D.C.W.; Fang, J.K.H.; Mitrano, D.M. Microplastic Fibre Releases from Industrial Wastewater Effluent: A Textile Wet-Processing Mill in China. Environ. Chem. 2021, 18, 93–100. [Google Scholar] [CrossRef]
- Zhou, H.; Zhou, L.; Ma, K. Microfiber from Textile Dyeing and Printing Wastewater of a Typical Industrial Park in China: Occurrence, Removal and Release. Sci. Total Environ. 2020, 739, 140329. [Google Scholar] [CrossRef] [PubMed]
- De Falco, F.; Cocca, M.; Avella, M.; Thompson, R.C. Microfibre Release to Water, via Laundering, and to Air, via Everyday Use: A Comparison between Polyester Clothing with Differing Textile Parameters. Environ. Sci. Technol. 2020, 54, 3288–3296. [Google Scholar] [CrossRef]
- Napper, I.E.; Thompson, R.C. Release of Synthetic Microplastic Plastic Fibres from Domestic Washing Machines: Effects of Fabric Type and Washing Conditions. Mar. Pollut. Bull. 2016, 112, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zhu, Z.-R.; Li, W.-H.; Yan, X.; Wang, L.-K.; Zhang, L.; Jin, J.; Dai, X.; Ni, B.-J. Revisiting Microplastics in Landfill Leachate: Unnoticed Tiny Microplastics and Their Fate in Treatment Works. Water Res. 2021, 190, 116784. [Google Scholar] [CrossRef] [PubMed]
- Belzagui, F.; Buscio, V.; Gutiérrez-Bouzán, C.; Vilaseca, M. Cigarette Butts as a Microfiber Source with a Microplastic Level of Concern. Sci. Total Environ. 2021, 762, 144165. [Google Scholar] [CrossRef]
- Alipour, S.; Hashemi, S.H.; Alavian Petroody, S.S. Release of Microplastic Fibers from Carpet-Washing Workshops Wastewater. J. Water Wastewater 2021, 31, 27–33. [Google Scholar] [CrossRef]
- Fadare, O.O.; Okoffo, E.D. Covid-19 Face Masks: A Potential Source of Microplastic Fibers in the Environment. Sci. Total Environ. 2020, 737, 140279. [Google Scholar] [CrossRef] [PubMed]
- Ó. Briain, O.; Marques Mendes, A.R.; McCarron, S.; Healy, M.G.; Morrison, L. The Role of Wet Wipes and Sanitary Towels as a Source of White Microplastic Fibres in the Marine Environment. Water Res. 2020, 182, 116021. [Google Scholar] [CrossRef] [PubMed]
- Bucci, K.; Tulio, M.; Rochman, C.M. What Is Known and Unknown about the Effects of Plastic Pollution: A Meta-Analysis and Systematic Review. Ecol. Appl. 2020, 30, e02044. [Google Scholar] [CrossRef] [PubMed]
- Bour, A.; Hossain, S.; Taylor, M.; Sumner, M.; Carney Almroth, B. Synthetic Microfiber and Microbead Exposure and Retention Time in Model Aquatic Species under Different Exposure Scenarios. Front. Environ. Sci. 2020, 8, 83. [Google Scholar] [CrossRef]
- Kutralam-Muniasamy, G.; Pérez-Guevara, F.; Elizalde-Martínez, I.; Shruti, V.C. An Overview of Recent Advances in Micro/Nano Beads and Microfibers Research: Critical Assessment and Promoting the Less Known. Sci. Total Environ. 2020, 740, 139991. [Google Scholar] [CrossRef]
- Bour, A.; Leoni, D.; Sundh, H.; Carney Almroth, B. Exposure to Textile Microfibers Causes No Effect on Blood, Behavior and Tissue Morphology in the Three-Spined Stickleback (Gasterosteus aculeatus). Mar. Pollut. Bull. 2022, 180, 113755. [Google Scholar] [CrossRef]
- Kim, L.; Kim, S.A.; Kim, T.H.; Kim, J.; An, Y.-J. Synthetic and Natural Microfibers Induce Gut Damage in the Brine Shrimp Artemia franciscana. Aquat. Toxicol. 2021, 232, 105748. [Google Scholar] [CrossRef]
- Mateos-Cárdenas, A.; O’Halloran, J.; van Pelt, F.N.A.M.; Jansen, M.A.K. Beyond Plastic Microbeads—Short-Term Feeding of Cellulose and Polyester Microfibers to the Freshwater Amphipod Gammarus duebeni. Sci. Total Environ. 2021, 753, 141859. [Google Scholar] [CrossRef]
- Weis, J.S.; Palmquist, K.H. Reality Check: Experimental Studies on Microplastics Lack Realism. Appl. Sci. 2021, 11, 8529. [Google Scholar] [CrossRef]
- Lacasse, K.; Baumann, W. Environmental Considerations for Textile Processes and Chemicals. In Textile Chemicals: Environmental Data and Facts; Lacasse, K., Baumann, W., Eds.; Springer: Berlin/Heidelberg, Germany, 2004; pp. 484–647. ISBN 978-3-642-18898-5. [Google Scholar]
- Saini, A.; Okeme, J.O.; Mark Parnis, J.; McQueen, R.H.; Diamond, M.L. From Air to Clothing: Characterizing the Accumulation of Semi-Volatile Organic Compounds to Fabrics in Indoor Environments. Indoor Air 2017, 27, 631–641. [Google Scholar] [CrossRef]
- Saini, A.; Rauert, C.; Simpson, M.J.; Harrad, S.; Diamond, M.L. Characterizing the Sorption of Polybrominated Diphenyl Ethers (PBDEs) to Cotton and Polyester Fabrics under Controlled Conditions. Sci. Total Environ. 2016, 563–564, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Kolarik, B.; Morrison, G.C. Accumulation of Polychlorinated Biphenyls in Fabrics in a Contaminated Building, and the Effect of Laundering. Indoor Air 2022, 32, e12944. [Google Scholar] [CrossRef] [PubMed]
- Carney Almroth, B.; Cartine, J.; Jönander, C.; Karlsson, M.; Langlois, J.; Lindström, M.; Lundin, J.; Melander, N.; Pesqueda, A.; Rahmqvist, I.; et al. Assessing the Effects of Textile Leachates in Fish Using Multiple Testing Methods: From Gene Expression to Behavior. Ecotoxicol. Environ. Saf. 2021, 207, 111523. [Google Scholar] [CrossRef]
- Sait, S.T.L.; Sørensen, L.; Kubowicz, S.; Vike-Jonas, K.; Gonzalez, S.V.; Asimakopoulos, A.G.; Booth, A.M. Microplastic Fibres from Synthetic Textiles: Environmental Degradation and Additive Chemical Content. Environ. Pollut. 2021, 268, 115745. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, J.C.; Avila, R.; Lourenço, A.G. Respiratory Disease Caused by Synthetic Fibres: A New Occupational Disease. Thorax 1975, 30, 204–219. [Google Scholar] [CrossRef]
- Stratton-Powell, A.A.; Pasko, K.M.; Lal, S.; Brockett, C.L.; Tipper, J.L. Chapter 22—Biologic Responses to Polyetheretherketone (PEEK) Wear Particles. In PEEK Biomaterials Handbook, 2nd ed.; Kurtz, S.M., Ed.; Plastics Design Library; William Andrew Publishing: Norwich, NY, USA, 2019; pp. 367–384. ISBN 978-0-12-812524-3. [Google Scholar]
- Urban, R.M.; Jacobs, J.J.; Tomlinson, M.J.; Gavrilovic, J.; Black, J.; Peoc’h, M. Dissemination of Wear Particles to the Liver, Spleen, and Abdominal Lymph Nodes of Patients with Hip or Knee Replacement. J. Bone Jt. Surg. Am. 2000, 82, 457. [Google Scholar] [CrossRef] [PubMed]
- Zarus, G.M.; Muianga, C.; Hunter, C.M.; Pappas, R.S. A Review of Data for Quantifying Human Exposures to Micro and Nanoplastics and Potential Health Risks. Sci. Total Environ. 2021, 756, 144010. [Google Scholar] [CrossRef]
- Hicks, D.G.; Judkins, A.R.; Sickel, J.Z.; Rosier, R.N.; Puzas, J.E.; O’keefe, R.J. Granular Histiocytosis of Pelvic Lymph Nodes Following Total Hip Arthroplasty. The Presence of Wear Debris, Cytokine Production, and Immunologically Activated Macrophages. J. Bone Jt. Surg. Am. 1996, 78, 482–496. [Google Scholar] [CrossRef]
- Ibrahim, Y.S.; Tuan Anuar, S.; Azmi, A.A.; Wan Mohd Khalik, W.M.A.; Lehata, S.; Hamzah, S.R.; Ismail, D.; Ma, Z.F.; Dzulkarnaen, A.; Zakaria, Z.; et al. Detection of Microplastics in Human Colectomy Specimens. JGH Open 2021, 5, 116–121. [Google Scholar] [CrossRef]
- Ragusa, A.; Svelato, A.; Santacroce, C.; Catalano, P.; Notarstefano, V.; Carnevali, O.; Papa, F.; Rongioletti, M.C.A.; Baiocco, F.; Draghi, S.; et al. Plasticenta: First Evidence of Microplastics in Human Placenta. Environ. Int. 2021, 146, 106274. [Google Scholar] [CrossRef]
- Leslie, H.A.; van Velzen, M.J.M.; Brandsma, S.H.; Vethaak, A.D.; Garcia-Vallejo, J.J.; Lamoree, M.H. Discovery and Quantification of Plastic Particle Pollution in Human Blood. Environ. Int. 2022, 163, 107199. [Google Scholar] [CrossRef] [PubMed]
- Pauly, J.L.; Stegmeier, S.J.; Allaart, H.A.; Cheney, R.T.; Zhang, P.J.; Mayer, A.G.; Streck, R.J. Inhaled Cellulosic and Plastic Fibers Found in Human Lung Tissue. Cancer Epidemiol. Biomark. Prev. 1998, 7, 419–428. [Google Scholar]
- Hernandez, L.M.; Xu, E.G.; Larsson, H.C.E.; Tahara, R.; Maisuria, V.B.; Tufenkji, N. Plastic Teabags Release Billions of Microparticles and Nanoparticles into Tea. Environ. Sci. Technol. 2019, 53, 12300–12310. [Google Scholar] [CrossRef] [PubMed]
- Kosuth, M.; Mason, S.A.; Wattenberg, E.V. Anthropogenic Contamination of Tap Water, Beer, and Sea Salt. PLoS ONE 2018, 13, e0194970. [Google Scholar] [CrossRef]
- Rochman, C.M.; Tahir, A.; Williams, S.L.; Baxa, D.V.; Lam, R.; Miller, J.T.; Teh, F.C.; Werorilangi, S.; Teh, S.J. Anthropogenic Debris in Seafood: Plastic Debris and Fibers from Textiles in Fish and Bivalves Sold for Human Consumption. Sci. Rep. 2015, 5, 14340. [Google Scholar] [CrossRef] [PubMed]
- Liebezeit, G.; Liebezeit, E. Non-Pollen Particulates in Honey and Sugar. Food Addit. Contam. Part A 2013, 30, 2136–2140. [Google Scholar] [CrossRef]
- Dris, R.; Gasperi, J.; Mirande, C.; Mandin, C.; Guerrouache, M.; Langlois, V.; Tassin, B. A First Overview of Textile Fibers, Including Microplastics, in Indoor and Outdoor Environments. Environ. Pollut. 2017, 221, 453–458. [Google Scholar] [CrossRef]
- Vianello, A.; Jensen, R.L.; Liu, L.; Vollertsen, J. Simulating Human Exposure to Indoor Airborne Microplastics Using a Breathing Thermal Manikin. Sci. Rep. 2019, 9, 8670. [Google Scholar] [CrossRef]
- Miao, L.; Hou, J.; You, G.; Liu, Z.; Liu, S.; Li, T.; Mo, Y.; Guo, S.; Qu, H. Acute Effects of Nanoplastics and Microplastics on Periphytic Biofilms Depending on Particle Size, Concentration and Surface Modification. Environ. Pollut. 2019, 255, 113300. [Google Scholar] [CrossRef]
- de Sá, L.C.; Oliveira, M.; Ribeiro, F.; Rocha, T.L.; Futter, M.N. Studies of the Effects of Microplastics on Aquatic Organisms: What Do We Know and Where Should We Focus Our Efforts in the Future? Sci. Total Environ. 2018, 645, 1029–1039. [Google Scholar] [CrossRef]
- Büks, F.; Loes van Schaik, N.; Kaupenjohann, M. What Do We Know about How the Terrestrial Multicellular Soil Fauna Reacts to Microplastic? SOIL 2020, 6, 245–267. [Google Scholar] [CrossRef]
- El-Gendy, A.H.; Augustyniak, M.; Toto, N.A.; Al Farraj, S.; El-Samad, L.M. Oxidative Stress Parameters, DNA Damage and Expression of HSP70 and MT in Midgut of Trachyderma hispida (Forskål, 1775) (Coleoptera: Tenebrionidae) from a Textile Industry Area. Environ. Pollut. 2020, 267, 115661. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Kwak, J.I.; Wang, D.; An, Y.-J. Multigenerational Effects of Polyethylene Terephthalate Microfibers in Caenorhabditis elegans. Environ. Res. 2021, 193, 110569. [Google Scholar] [CrossRef]
- Mohsen, M.; Zhang, L.; Sun, L.; Lin, C.; Wang, Q.; Yang, H. Microplastic Fibers Transfer from the Water to the Internal Fluid of the Sea Cucumber Apostichopus japonicus. Environ. Pollut. 2020, 257, 113606. [Google Scholar] [CrossRef] [PubMed]
- Barboza, L.G.A.; Lopes, C.; Oliveira, P.; Bessa, F.; Otero, V.; Henriques, B.; Raimundo, J.; Caetano, M.; Vale, C.; Guilhermino, L. Microplastics in Wild Fish from North East Atlantic Ocean and Its Potential for Causing Neurotoxic Effects, Lipid Oxidative Damage, and Human Health Risks Associated with Ingestion Exposure. Sci. Total Environ. 2020, 717, 134625. [Google Scholar] [CrossRef] [PubMed]
- Christoforou, E.; Dominoni, D.M.; Lindström, J.; Stilo, G.; Spatharis, S. Effects of Long-Term Exposure to Microfibers on Ecosystem Services Provided by Coastal Mussels. Environ. Pollut. 2020, 266, 115184. [Google Scholar] [CrossRef] [PubMed]
- Mendrik, F.M.; Henry, T.B.; Burdett, H.; Hackney, C.R.; Waller, C.; Parsons, D.R.; Hennige, S.J. Species-Specific Impact of Microplastics on Coral Physiology. Environ. Pollut. 2021, 269, 116238. [Google Scholar] [CrossRef] [PubMed]
- Worm, B.; Barbier, E.B.; Beaumont, N.; Duffy, J.E.; Folke, C.; Halpern, B.S.; Jackson, J.B.C.; Lotze, H.K.; Micheli, F.; Palumbi, S.R.; et al. Impacts of Biodiversity Loss on Ocean Ecosystem Services. Science 2006, 314, 787–790. [Google Scholar] [CrossRef]
- Zimmermann, L.; Göttlich, S.; Oehlmann, J.; Wagner, M.; Völker, C. What Are the Drivers of Microplastic Toxicity? Comparing the Toxicity of Plastic Chemicals and Particles to Daphnia Magna. Environ. Pollut. 2020, 267, 115392. [Google Scholar] [CrossRef]
- Straub, S.; Hirsch, P.E.; Burkhardt-Holm, P. Biodegradable and Petroleum-Based Microplastics Do Not Differ in Their Ingestion and Excretion but in Their Biological Effects in a Freshwater Invertebrate Gammarus fossarum. Int. J. Environ. Res. Public Health 2017, 14, 774. [Google Scholar] [CrossRef]
- Kwak, J.I.; An, Y.-J. Length- and Polymer-Dependent Ecotoxicities of Microfibers to the Earthworm Eisenia Andrei. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2022, 257, 109354. [Google Scholar] [CrossRef] [PubMed]
- Prendergast-Miller, M.T.; Katsiamides, A.; Abbass, M.; Sturzenbaum, S.R.; Thorpe, K.L.; Hodson, M.E. Polyester-Derived Microfibre Impacts on the Soil-Dwelling Earthworm Lumbricus terrestris. Environ. Pollut. 2019, 251, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Welden, N.A.C.; Cowie, P.R. Environment and Gut Morphology Influence Microplastic Retention in Langoustine, Nephrops norvegicus. Environ. Pollut. 2016, 214, 859–865. [Google Scholar] [CrossRef] [PubMed]
- van Dijk, F.; Eck, G.V.; Cole, M.; Salvati, A.; Bos, S.; Gosens, R.; Melgert, B. Exposure to Textile Microplastic Fibers Impairs Epithelial Growth. Eur. Respir. J. 2020, 56, 1972. [Google Scholar] [CrossRef]
- Alnajar, N.; Jha, A.N.; Turner, A. Impacts of Microplastic Fibres on the Marine Mussel, Mytilus galloprovinciallis. Chemosphere 2021, 262, 128290. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Kim, K.; Hong, S.H.; Park, K.-I.; Park, J.-W. Impact of Polyethylene Terephthalate Microfiber Length on Cellular Responses in the Mediterranean Mussel Mytilus galloprovinciallis. Mar. Environ. Res. 2021, 168, 105320. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Kim, K.; Park, K.; Park, J.-W. Long-Term Exposure of the Mediterranean Mussels, Mytilus galloprovinciallis to Polyethylene Terephthalate Microfibers: Implication for Reproductive and Neurotoxic Effects. Chemosphere 2022, 299, 134317. [Google Scholar] [CrossRef]
- Qiao, R.; Deng, Y.; Zhang, S.; Wolosker, M.B.; Zhu, Q.; Ren, H.; Zhang, Y. Accumulation of Different Shapes of Microplastics Initiates Intestinal Injury and Gut Microbiota Dysbiosis in the Gut of Zebrafish. Chemosphere 2019, 236, 124334. [Google Scholar] [CrossRef]
- Song, Y.; Cao, C.; Qiu, R.; Hu, J.; Liu, M.; Lu, S.; Shi, H.; Raley-Susman, K.M.; He, D. Uptake and Adverse Effects of Polyethylene Terephthalate Microplastics Fibers on Terrestrial Snails (Achatina fulica) after Soil Exposure. Environ. Pollut. 2019, 250, 447–455. [Google Scholar] [CrossRef]
- Jabeen, K.; Li, B.; Chen, Q.; Su, L.; Wu, C.; Hollert, H.; Shi, H. Effects of Virgin Microplastics on Goldfish (Carassius auratus). Chemosphere 2018, 213, 323–332. [Google Scholar] [CrossRef]
- Hope, J.A.; Coco, G.; Thrush, S.F. Effects of Polyester Microfibers on Microphytobenthos and Sediment-Dwelling Infauna. Environ. Sci. Technol. 2020, 54, 7970–7982. [Google Scholar] [CrossRef] [PubMed]
- Kokalj, A.J.; Kunej, U.; Skalar, T. Screening Study of Four Environmentally Relevant Microplastic Pollutants: Uptake and Effects on Daphnia magna and Artemia franciscana. Chemosphere 2018, 208, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Woods, M.N.; Hong, T.J.; Baughman, D.; Andrews, G.; Fields, D.M.; Matrai, P.A. Accumulation and Effects of Microplastic Fibers in American Lobster Larvae (Homarus americanus). Mar. Pollut. Bull. 2020, 157, 111280. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Chernick, M.; Lewis, A.M.; Ferguson, P.L.; Hinton, D.E. Chronic Microfiber Exposure in Adult Japanese Medaka (Oryzias latipes). PLoS ONE 2020, 15, e0229962. [Google Scholar] [CrossRef]
- Boots, B.; Russell, C.W.; Green, D.S. Effects of Microplastics in Soil Ecosystems: Above and Below Ground. Environ. Sci. Technol. 2019, 53, 11496–11506. [Google Scholar] [CrossRef]
- Ziajahromi, S.; Kumar, A.; Neale, P.A.; Leusch, F.D.L. Impact of Microplastic Beads and Fibers on Waterflea (Ceriodaphnia dubia) Survival, Growth, and Reproduction: Implications of Single and Mixture Exposures. Environ. Sci. Technol. 2017, 51, 13397–13406. [Google Scholar] [CrossRef]
- Horn, D.A.; Granek, E.F.; Steele, C.L. Effects of Environmentally Relevant Concentrations of Microplastic Fibers on Pacific Mole Crab (Emerita analoga) Mortality and Reproduction. Limnol. Oceanogr. Lett. 2020, 5, 74–83. [Google Scholar] [CrossRef]
- Schell, T.; Martinez-Perez, S.; Dafouz, R.; Hurley, R.; Vighi, M.; Rico, A. Effects of Polyester Fibers and Car Tire Particles on Freshwater Invertebrates. Environ. Toxicol. Chem. 2022, 41, 1555–1567. [Google Scholar] [CrossRef]
- Kim, S.W.; Waldman, W.R.; Kim, T.-Y.; Rillig, M.C. Effects of Different Microplastics on Nematodes in the Soil Environment: Tracking the Extractable Additives Using an Ecotoxicological Approach. Environ. Sci. Technol. 2020, 54, 13868–13878. [Google Scholar] [CrossRef]
- Blarer, P.; Burkhardt-Holm, P. Microplastics Affect Assimilation Efficiency in the Freshwater Amphipod Gammarus Fossarum. Environ. Sci. Pollut. Res. 2016, 23, 23522–23532. [Google Scholar] [CrossRef]
- Romanó de Orte, M.; Clowez, S.; Caldeira, K. Response of Bleached and Symbiotic Sea Anemones to Plastic Microfiber Exposure. Environ. Pollut. 2019, 249, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Au, S.Y.; Bruce, T.F.; Bridges, W.C.; Klaine, S.J. Responses of Hyalella Azteca to Acute and Chronic Microplastic Exposures. Environ. Toxicol. Chem. 2015, 34, 2564–2572. [Google Scholar] [CrossRef]
- Cole, M.; Coppock, R.; Lindeque, P.K.; Altin, D.; Reed, S.; Pond, D.W.; Sørensen, L.; Galloway, T.S.; Booth, A.M. Effects of Nylon Microplastic on Feeding, Lipid Accumulation, and Moulting in a Coldwater Copepod. Environ. Sci. Technol. 2019, 53, 7075–7082. [Google Scholar] [CrossRef] [PubMed]
- Watts, A.J.R.; Urbina, M.A.; Corr, S.; Lewis, C.; Galloway, T.S. Ingestion of Plastic Microfibers by the Crab Carcinus maenas and Its Effect on Food Consumption and Energy Balance. Environ. Sci. Technol. 2015, 49, 14597–14604. [Google Scholar] [CrossRef] [PubMed]
- de Souza Machado, A.A.; Lau, C.W.; Kloas, W.; Bergmann, J.; Bachelier, J.B.; Faltin, E.; Becker, R.; Görlich, A.S.; Rillig, M.C. Microplastics Can Change Soil Properties and Affect Plant Performance. Environ. Sci. Technol. 2019, 53, 6044–6052. [Google Scholar] [CrossRef]
- Lozano, Y.M.; Lehnert, T.; Linck, L.T.; Lehmann, A.; Rillig, M.C. Microplastic Shape, Polymer Type, and Concentration Affect Soil Properties and Plant Biomass. Front. Plant Sci. 2021, 12, 616645. [Google Scholar] [CrossRef]
- Zeb, A.; Liu, W.; Meng, L.; Lian, J.; Wang, Q.; Lian, Y.; Chen, C.; Wu, J. Effects of Polyester Microfibers (PMFs) and Cadmium on Lettuce (Lactuca sativa) and the Rhizospheric Microbial Communities: A Study Involving Physio-Biochemical Properties and Metabolomic Profiles. J. Hazard. Mater. 2022, 424, 127405. [Google Scholar] [CrossRef]
- Barboza, L.G.A.; Vieira, L.R.; Branco, V.; Figueiredo, N.; Carvalho, F.; Carvalho, C.; Guilhermino, L. Microplastics Cause Neurotoxicity, Oxidative Damage and Energy-Related Changes and Interact with the Bioaccumulation of Mercury in the European Seabass, Dicentrarchus labrax (Linnaeus, 1758). Aquat. Toxicol. 2018, 195, 49–57. [Google Scholar] [CrossRef]
- Jemec, A.; Horvat, P.; Kunej, U.; Bele, M.; Kržan, A. Uptake and Effects of Microplastic Textile Fibers on Freshwater Crustacean Daphnia Magna. Environ. Pollut. 2016, 219, 201–209. [Google Scholar] [CrossRef]
- Gray, A.D.; Weinstein, J.E. Size- and Shape-Dependent Effects of Microplastic Particles on Adult Daggerblade Grass Shrimp (Palaemonetes pugio). Environ. Toxicol. Chem. 2017, 36, 3074–3080. [Google Scholar] [CrossRef]
- Baechler, B.R.; Stienbarger, C.D.; Horn, D.A.; Joseph, J.; Taylor, A.R.; Granek, E.F.; Brander, S.M. Microplastic Occurrence and Effects in Commercially Harvested North American Finfish and Shellfish: Current Knowledge and Future Directions. Limnol. Oceanogr. Lett. 2020, 5, 113–136. [Google Scholar] [CrossRef]
- Athey, S.N.; Albotra, S.D.; Gordon, C.A.; Monteleone, B.; Seaton, P.; Andrady, A.L.; Taylor, A.R.; Brander, S.M. Trophic Transfer of Microplastics in an Estuarine Food Chain and the Effects of a Sorbed Legacy Pollutant. Limnol. Oceanogr. Lett. 2020, 5, 154–162. [Google Scholar] [CrossRef]
- Li, L.; Frey, M.; Browning, K.J. Biodegradability Study on Cotton and Polyester Fabrics. J. Eng. Fibers Fabr. 2010, 5, 155892501000500400. [Google Scholar] [CrossRef]
- Arshad, K.; Skrifvars, M.; Vivod, V.; Valh, J.; Voncina, B. Biodegradation of Natural Textile Materials in Soil. Tekstilec 2014, 57, 118–132. [Google Scholar] [CrossRef]
- Zambrano, M.C.; Pawlak, J.J.; Daystar, J.; Ankeny, M.; Goller, C.C.; Venditti, R.A. Aerobic Biodegradation in Freshwater and Marine Environments of Textile Microfibers Generated in Clothes Laundering: Effects of Cellulose and Polyester-Based Microfibers on the Microbiome. Mar. Pollut. Bull. 2020, 151, 110826. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Kang, Y.K.; Im, S.S. Biodegradability of Cellulose Fabrics. J. Appl. Polym. Sci. 2004, 94, 248–253. [Google Scholar] [CrossRef]
- Remy, F.; Collard, F.; Gilbert, B.; Compère, P.; Eppe, G.; Lepoint, G. When Microplastic Is Not Plastic: The Ingestion of Artificial Cellulose Fibers by Macrofauna Living in Seagrass Macrophytodetritus. Environ. Sci. Technol. 2015, 49, 11158–11166. [Google Scholar] [CrossRef]
- Zhao, S.; Zhu, L.; Li, D. Microscopic Anthropogenic Litter in Terrestrial Birds from Shanghai, China: Not Only Plastics but Also Natural Fibers. Sci. Total Environ. 2016, 550, 1110–1115. [Google Scholar] [CrossRef]
- Stone, C.; Windsor, F.M.; Munday, M.; Durance, I. Natural or Synthetic—How Global Trends in Textile Usage Threaten Freshwater Environments. Sci. Total Environ. 2020, 718, 134689. [Google Scholar] [CrossRef]
- Grancaric, A.M.; Tarbuk, A.; Pusic, T. Electrokinetic Properties of Textile Fabrics. Color. Technol. 2005, 121, 221–227. [Google Scholar] [CrossRef]
- Sharma, M.D.; Krupadam, R.J. Adsorption-Desorption Dynamics of Synthetic and Naturally Weathered Microfibers with Toxic Heavy Metals and Their Ecological Risk in an Estuarine Ecosystem. Environ. Res. 2022, 207, 112198. [Google Scholar] [CrossRef] [PubMed]
- Bour, A.; Hamann Sandgaard, M.; Syberg, K.; Palmqvist, A.; Carney Almroth, B. Comprehending the Complexity of Microplastic Organismal Exposures and Effects, to Improve Testing Frameworks. J. Hazard. Mater. 2021, 415, 125652. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, N.B.; Hüffer, T.; Thompson, R.C.; Hassellöv, M.; Verschoor, A.; Daugaard, A.E.; Rist, S.; Karlsson, T.; Brennholt, N.; Cole, M.; et al. Are We Speaking the Same Language? Recommendations for a Definition and Categorization Framework for Plastic Debris. Environ. Sci. Technol. 2019, 53, 1039–1047. [Google Scholar] [CrossRef]
- Liu, L.-Y.; Mai, L.; Zeng, E.Y. Plastic and Microplastic Pollution: From Ocean Smog to Planetary Boundary Threats. In A New Paradigm for Environmental Chemistry and Toxicology: From Concepts to Insights; Jiang, G., Li, X., Eds.; Springer: Singapore, 2020; pp. 229–240. ISBN 978-981-13-9447-8. [Google Scholar]
- European Comission. Integrated Pollution Prevention and Control (IPPC): Reference Document on Best Available Techniques for the Textiles Industry; European Comission: Brussels, Belgium, 2003; Volume 626. [Google Scholar]
- ECHA Universe of Registered Substances. European Chemicals Agency, ECHA. 2021. Available online: https://echa.europa.eu/fr/universe-of-registered-substances (accessed on 25 May 2021).
- Morrison, G.; Li, H.; Mishra, S.; Buechlein, M. Airborne Phthalate Partitioning to Cotton Clothing. Atmos. Environ. 2015, 115, 149–152. [Google Scholar] [CrossRef]
- Morrison, G.C.; Andersen, H.V.; Gunnarsen, L.; Varol, D.; Uhde, E.; Kolarik, B. Partitioning of PCBs from Air to Clothing Materials in a Danish Apartment. Indoor Air 2018, 28, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Islam, S. 15—Sustainable Raw Materials: 50 Shades of Sustainability. In Sustainable Technologies for Fashion and Textiles; Nayak, R., Ed.; Woodhead Publishing Series in Textiles; Woodhead Publishing: Sawston, UK, 2020; pp. 343–357. ISBN 978-0-08-102867-4. [Google Scholar]
- Dolez, P.I.; Benaddi, H. 7—Toxicity Testing of Textiles. In Advanced Characterization and Testing of Textiles; Woodhead Publishing: Sawston, UK, 2018; pp. 151–188. ISBN 978-0-08-100453-1. [Google Scholar]
- KEMI. Chemicals in Textiles–Risks to Human Health and the Environment; Kemikalieinspektion: Sundbyberg, Sweden, 2015; p. 139. [Google Scholar]
- Hill, P.J.; Taylor, M.; Goswami, P.; Blackburn, R.S. Substitution of PFAS Chemistry in Outdoor Apparel and the Impact on Repellency Performance. Chemosphere 2017, 181, 500–507. [Google Scholar] [CrossRef]
- KEMI. Hazardous Chemical Substances in Textiles—Proposals for Risk Management Measures; Kemikalieinspektion: Sundbyberg, Sweden, 2016. [Google Scholar]
- Nguyen, T.; Saleh, M.A. Detection of Azo Dyes and Aromatic Amines in Women Undergarment. J. Environ. Sci. Health Part A 2016, 51, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.-T. Azo Dyes and Human Health: A Review. J. Environ. Sci. Health Part C 2016, 34, 233–261. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Chatterji, S.; Nandini, P.T.; Prasad, A.S.A.; Rao, K.V.B. Biodegradation of Azo Dye Direct Orange 16 by Micrococcusluteus Strain SSN2. Int. J. Environ. Sci. Technol. 2015, 12, 2161–2168. [Google Scholar] [CrossRef]
- Weil, E.D.; Levchik, S.; Moy, P. Flame and Smoke Retardants in Vinyl Chloride Polymers—Commercial Usage and Current Developments. J. Fire Sci. 2006, 24, 211–236. [Google Scholar] [CrossRef]
- Meeker, J.D.; Sathyanarayana, S.; Swan, S.H. Phthalates and Other Additives in Plastics: Human Exposure and Associated Health Outcomes. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2097–2113. [Google Scholar] [CrossRef]
- Schwarzenbach, R.P.; Gschwend, P.M.; Imboden, D.M. Environmental Organic Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2016; ISBN 978-1-118-76704-7. [Google Scholar]
- Wang, Z.; Chen, M.; Zhang, L.; Wang, K.; Yu, X.; Zheng, Z.; Zheng, R. Sorption Behaviors of Phenanthrene on the Microplastics Identified in a Mariculture Farm in Xiangshan Bay, Southeastern China. Sci. Total Environ. 2018, 628–629, 1617–1626. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Yu, X.; Yao, Z.; Tao, P.; Li, G.; Yu, X.; Zhao, J.-L.; Peng, J. How Biofilms Affect the Uptake and Fate of Hydrophobic Organic Compounds (HOCs) in Microplastic: Insights from an In Situ Study of Xiangshan Bay, China. Water Res. 2020, 184, 116118. [Google Scholar] [CrossRef] [PubMed]
- Lionetto, F.; Esposito Corcione, C. An Overview of the Sorption Studies of Contaminants on Poly(Ethylene Terephthalate) Microplastics in the Marine Environment. J. Mar. Sci. Eng. 2021, 9, 445. [Google Scholar] [CrossRef]
- Joly, F.-X.; Coulis, M. Comparison of Cellulose vs. Plastic Cigarette Filter Decomposition under Distinct Disposal Environments. Waste Manag. 2018, 72, 349–353. [Google Scholar] [CrossRef]
- Novotny, T.; Lum, K.; Smith, E.; Wang, V.; Barnes, R. Cigarettes Butts and the Case for an Environmental Policy on Hazardous Cigarette Waste. Int. J. Environ. Res. Public Health 2009, 6, 1691–1705. [Google Scholar] [CrossRef]
- Wright, S.L.; Rowe, D.; Reid, M.J.; Thomas, K.V.; Galloway, T.S. Bioaccumulation and Biological Effects of Cigarette Litter in Marine Worms. Sci. Rep. 2015, 5, 14119. [Google Scholar] [CrossRef]
- Moerman, J.W.; Potts, G.E. Analysis of Metals Leached from Smoked Cigarette Litter. Tob. Control 2011, 20, i30–i35. [Google Scholar] [CrossRef]
- Torkashvand, J.; Farzadkia, M. A Systematic Review on Cigarette Butt Management as a Hazardous Waste and Prevalent Litter: Control and Recycling. Environ. Sci. Pollut. Res. 2019, 26, 11618–11630. [Google Scholar] [CrossRef]
- Shruti, V.C.; Kutralam-Muniasamy, G. Bioplastics: Missing Link in the Era of Microplastics. Sci. Total Environ. 2019, 697, 134139. [Google Scholar] [CrossRef]
- Brennecke, D.; Duarte, B.; Paiva, F.; Caçador, I.; Canning-Clode, J. Microplastics as Vector for Heavy Metal Contamination from the Marine Environment. Estuar. Coast. Shelf Sci. 2016, 178, 189–195. [Google Scholar] [CrossRef]
- Li, J.; Zhang, K.; Zhang, H. Adsorption of Antibiotics on Microplastics. Environ. Pollut. 2018, 237, 460–467. [Google Scholar] [CrossRef] [PubMed]
- Torres, F.G.; Dioses-Salinas, D.C.; Pizarro-Ortega, C.I.; De-la-Torre, G.E. Sorption of Chemical Contaminants on Degradable and Non-Degradable Microplastics: Recent Progress and Research Trends. Sci. Total Environ. 2021, 757, 143875. [Google Scholar] [CrossRef] [PubMed]
- Rochman, C.M.; Hoh, E.; Hentschel, B.T.; Kaye, S. Long-Term Field Measurement of Sorption of Organic Contaminants to Five Types of Plastic Pellets: Implications for Plastic Marine Debris. Environ. Sci. Technol. 2013, 47, 1646–1654. [Google Scholar] [CrossRef] [PubMed]
- Velez, J.F.M.; Shashoua, Y.; Syberg, K.; Khan, F.R. Considerations on the Use of Equilibrium Models for the Characterisation of HOC-Microplastic Interactions in Vector Studies. Chemosphere 2018, 210, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Bour, A.; Sturve, J.; Höjesjö, J.; Carney Almroth, B. Microplastic Vector Effects: Are Fish at Risk When Exposed via the Trophic Chain? Front. Environ. Sci. 2020, 8, 90. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Liu, Y.; Gong, X.; Zhang, T.; Sun, H. Widespread Occurrence of Bisphenol A in Daily Clothes and Its High Exposure Risk in Humans. Environ. Sci. Technol. 2019, 53, 7095–7102. [Google Scholar] [CrossRef] [PubMed]
- White, E.M.; Clark, S.; Manire, C.A.; Crawford, B.; Wang, S.; Locklin, J.; Ritchie, B.W. Ingested Micronizing Plastic Particle Compositions and Size Distributions within Stranded Post-Hatchling Sea Turtles. Environ. Sci. Technol. 2018, 52, 10307–10316. [Google Scholar] [CrossRef]
- Fred-Ahmadu, O.H.; Bhagwat, G.; Oluyoye, I.; Benson, N.U.; Ayejuyo, O.O.; Palanisami, T. Interaction of Chemical Contaminants with Microplastics: Principles and Perspectives. Sci. Total Environ. 2020, 706, 135978. [Google Scholar] [CrossRef]
- Arias-Andres, M.; Rojas-Jimenez, K.; Grossart, H.-P. Collateral Effects of Microplastic Pollution on Aquatic Microorganisms: An Ecological Perspective. TrAC Trends Anal. Chem. 2019, 112, 234–240. [Google Scholar] [CrossRef]
- Kirstein, I.V.; Kirmizi, S.; Wichels, A.; Garin-Fernandez, A.; Erler, R.; Löder, M.; Gerdts, G. Dangerous Hitchhikers? Evidence for Potentially Pathogenic Vibrio Spp. on Microplastic Particles. Mar. Environ. Res. 2016, 120, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jahnke, A.; Arp, H.P.H.; Escher, B.I.; Gewert, B.; Gorokhova, E.; Kühnel, D.; Ogonowski, M.; Potthoff, A.; Rummel, C.; Schmitt-Jansen, M.; et al. Reducing Uncertainty and Confronting Ignorance about the Possible Impacts of Weathering Plastic in the Marine Environment. Environ. Sci. Technol. Lett. 2017, 4, 85–90. [Google Scholar] [CrossRef]
- Ockenden, A.; Northcott, G.L.; Tremblay, L.A.; Simon, K.S. Disentangling the Influence of Microplastics and Their Chemical Additives on a Model Detritivore System. Environ. Pollut. 2022, 307, 119558. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, N.B.; Rist, S.; Bodin, J.; Jensen, L.H.S.; Schmidt, S.N.; Mayer, P.; Meibom, A.; Baun, A. Microplastics as Vectors for Environmental Contaminants: Exploring Sorption, Desorption, and Transfer to Biota. Integr. Environ. Assess. Manag. 2017, 13, 488–493. [Google Scholar] [CrossRef]
- Seuront, L. Microplastic Leachates Impair Behavioural Vigilance and Predator Avoidance in a Temperate Intertidal Gastropod. Biol. Lett. 2018, 14, 20180453. [Google Scholar] [CrossRef]
- Le Bihanic, F.; Clérandeau, C.; Cormier, B.; Crebassa, J.-C.; Keiter, S.H.; Beiras, R.; Morin, B.; Bégout, M.-L.; Cousin, X.; Cachot, J. Organic Contaminants Sorbed to Microplastics Affect Marine Medaka Fish Early Life Stages Development. Mar. Pollut. Bull. 2020, 154, 111059. [Google Scholar] [CrossRef]
- Rios-Fuster, B.; Alomar, C.; Viñas, L.; Campillo, J.A.; Pérez-Fernández, B.; Álvarez, E.; Compa, M.; Deudero, S. Organochlorine Pesticides (OCPs) and Polychlorinated Biphenyls (PCBs) Occurrence in Sparus Aurata Exposed to Microplastic Enriched Diets in Aquaculture Facilities. Mar. Pollut. Bull. 2021, 173, 113030. [Google Scholar] [CrossRef]
- Rendell-Bhatti, F.; Paganos, P.; Pouch, A.; Mitchell, C.; D’Aniello, S.; Godley, B.J.; Pazdro, K.; Arnone, M.I.; Jimenez-Guri, E. Developmental Toxicity of Plastic Leachates on the Sea Urchin Paracentrotus lividus. Environ. Pollut. 2021, 269, 115744. [Google Scholar] [CrossRef]
- Beckingham, B.; Ghosh, U. Differential Bioavailability of Polychlorinated Biphenyls Associated with Envi-ronmental Particles: Microplastic in Comparison to Wood, Coal and Biochar. Environ. Pollut. 2017, 220, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Thaysen, C.; Sorais, M.; Verreault, J.; Diamond, M.L.; Rochman, C.M. Bidirectional Transfer of Halogenated Flame Retardants between the Gastrointestinal Tract and Ingested Plastics in Urban-Adapted Ring-Billed Gulls. Sci. Total Environ. 2020, 730, 138887. [Google Scholar] [CrossRef]
- Ašmonaitė, G.; Tivefälth, M.; Westberg, E.; Magnér, J.; Backhaus, T.; Carney Almroth, B. Microplastics as a Vector for Exposure to Hydrophobic Organic Chemicals in Fish: A Comparison of Two Polymers and Silica Particles Spiked with Three Model Compounds. Front. Environ. Sci. 2020, 8, 87. [Google Scholar] [CrossRef]
- Zhu, M.; Chernick, M.; Rittschof, D.; Hinton, D.E. Chronic Dietary Exposure to Polystyrene Microplastics in Maturing Japanese Medaka (Oryzias latipes). Aquat. Toxicol. 2020, 220, 105396. [Google Scholar] [CrossRef] [PubMed]
- Persson, L.; Carney Almroth, B.M.; Collins, C.D.; Cornell, S.; de Wit, C.A.; Diamond, M.L.; Fantke, P.; Hassellöv, M.; MacLeod, M.; Ryberg, M.W.; et al. Outside the Safe Operating Space of the Planetary Boundary for Novel Entities. Environ. Sci. Technol. 2022, 56, 1510–1521. [Google Scholar] [CrossRef] [PubMed]
- Peters, G.; Li, M.; Lenzen, M. The Need to Decelerate Fast Fashion in a Hot Climate—A Global Sustainability Perspective on the Garment Industry. J. Clean. Prod. 2021, 295, 126390. [Google Scholar] [CrossRef]

| Level | Type of Effect | Organism Clade | Genus/Species | Plastic Material Type | In Text Citation |
|---|---|---|---|---|---|
| Sub/Cellular | Adverse Immune Response | Annelid Worms, Bivalves, Corals | Eisenia andrei | Polyamide nylon, polyproplene | [52,56] (Cole et al. 2020); |
| Mytilus spp. | |||||
| Acropora sp. | |||||
| Seriatopora hystrix | |||||
| Cellular Response | Annelid Worms, Bivalves, Coral, Crustaceans, Humans, Nematodes, Rodents | Lumbricus terrestris | Composite household lint, nylon, polyester, polypropylene, polyethylene terephthalate (PET) | [48,52,56,57,58,59,60,61,62] | |
| Eisenia andrei | |||||
| Mytilus galloprovincialis | |||||
| Acropora sp. | |||||
| Seriatopora hystrix | |||||
| Nephrops norvegicus | |||||
| Homo sapiens | |||||
| Caenorhabditis elegans | |||||
| Mus musculus | |||||
| Oxidative Stress | Annelid Worms, Bivalves, Coral, Echinoderms, Fish, Nematodes, Terrestrial Snails | Lumbricus terrestris | Polyester, polyamide nylon, polyethylene (80%), polyester (19%), rayon (1%), polypropylene, polyethylene terephthalate (PET) | [48,49,50,52,57,63,64] (Cole et al., 2020); | |
| Mytilus spp. | |||||
| Acropora sp. | |||||
| Seriatopora hystrix | |||||
| Apostichopus japonicus | |||||
| Dicentrachus labrax | |||||
| Trachurus trachurus | |||||
| Scomber colias | |||||
| Danio rerio | |||||
| Caenorhabditis elegans | |||||
| Achatina fulica | |||||
| Organ | Growth Development | Bivalves, Crustaceans, Fish | Mytilus galloprovincialis | Composite household lint; ethylene vinyl acetate (EVA); polypropylene, polyethylene terephthalate | [20,60,65,66] |
| Emerita analoga | |||||
| Artemia franciscana | |||||
| Carassius auratus | |||||
| Inflammation | Fish; Rodents; Zooplankton | Carassius auratus | Ethylene vinyl acetate (EVA), polypropylene, polyester, polyethylene terephthalate (PET) | [29,63,65,67] | |
| Danio rerio | |||||
| Cavia porcellus | |||||
| Artemia franciscana | |||||
| Oxidative Stress | Crustaceans, Fish | Homarus americanus | Polyethylene terephthalate (PET), polypropylene | [63,68] | |
| Danio rerio | |||||
| Physical Organ Damage | Annelid Worms, Bivalves, Crustaceans, Fish, Humans, Rodents, Terrestrial Snails, Zooplankton | Eisenia andrei | Composite household lint; polypropylene, polyethylene terephthalate (PET), polyethylene (80%); polyester (19%); rayon (1%) ethylene vinyl acetate (EVA), polycarbonate, polyamide, polyester | [20,29,34,50,56,58,60,62,63,64,65,67,69] | |
| Mytilus galloprovincialis | |||||
| Artemia franciscana | |||||
| Nephrops norvegicus | |||||
| Dicentrachus labrax | |||||
| Trachurus trachurus | |||||
| Oryzias latipes | |||||
| Scomber colias | |||||
| Carassius auratus | |||||
| Danio rerio | |||||
| Homo sapiens | |||||
| Cavia porcellus | |||||
| Achatina fulica | |||||
| Artemia franciscana | |||||
| Organism | Adverse Reproductive Response | Bivalves, Crustaceans, Nematodes, Terrestrial Vegetation, Worm, Zooplankton | Mytilus galloprovincialis | Polypropylene, polyethylene terephthalate (PET), high-density polyethylene (HDPE), polylactic acid (PLA), polyacrylicnitrile (PAN) | [48,62,70,71,72,73,74] |
| Daphnia magna | |||||
| Emerita analoga | |||||
| Caenorhabditis elegans | |||||
| Lolium perenne | |||||
| Aporrectodea rosea | |||||
| Ceriodaphnia dubia | |||||
| Behavioral Change | Annelid Worms, Bivalves, Cnidarians, Crustaceans, Nematodes, Terrestrial Snails, Zooplankton | Lumbricus terrestris | Polyester, composite household lint, nylon, polyethylene terephthalate (PET), polypropylene, polyamide (PA) | [38,48,51,57,58,60,64,66,68,72,75,76,77,78] (Kang et al., 2020); (Lahive et al., 2022) | |
| Mytilus galloprovincialis | |||||
| Mytilus edulis | |||||
| Macomona liliana | |||||
| Aiptasia pallida | |||||
| Hyalella azteca | |||||
| Calanus finmarchicus | |||||
| Gammarus fossarum | |||||
| Nephrops norvegicus | |||||
| Caenorhabditis elegans | |||||
| Achatina fulica | |||||
| Daphnia magna | |||||
| Tigriopus japonicus | |||||
| Growth Development | Bivalves, Crustaceans, Microphytobenthos, Nematodes, Terrestrial Veg., Worm, Zooplankton | Macomona liliana | Polyethylene terephthalate (PET), polypropylene, high-density polyethylene (HDPE), polylactic acid (PLA) nylon, polyester | [38,48,58,66,67,68,70,71,72,77,78,79,80,81,82] | |
| Hyalella azteca | |||||
| Emerita analoga | |||||
| Carcinus maenas | |||||
| Calanus finmarchicus | |||||
| Nephrops norvegicus | |||||
| Homarus americanus | |||||
| Cyanobacteria | |||||
| Caenorhabditis elegans | |||||
| Allium fistulosum | |||||
| Lolium perenne | |||||
| Aporrectodea rosea | |||||
| Lactuca sativa | |||||
| Daucus carota | |||||
| Daphnia magna | |||||
| Artemia franciscana | |||||
| Ceriodaphnia dubia | |||||
| Neurological | Fish | Dicentrachus labrax | Polyethylene (80%), polyester (19%), rayon (1%) | [50,83] | |
| Trachurus trachurus | |||||
| Scomber colias | |||||
| Survivorship or Mortality | Annelid Worms, Crustaceans, Zooplankton | Eisenia andrei | Polypropylene, polyethylene terephthalate (PET), polyester, lyocell | [20,56,58,68,71,72,73,77,84,85] | |
| Hyalella azteca | |||||
| Emerita analoga | |||||
| Palaemonetes pugio | |||||
| Artemia franciscana | |||||
| Nephrops norvegicus | |||||
| Homarus americanus | |||||
| Daphnia magna | |||||
| Ceriodaphnia dubia | |||||
| Population | Adverse Reproductive Response | Crustaceans, Nematodes | Emerita analoga | Polypropylene, polyethylene terephthalate | [48,72] |
| Caenorhabditis elegans |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Athey, S.N.; Carney Almroth, B.; Granek, E.F.; Hurst, P.; Tissot, A.G.; Weis, J.S. Unraveling Physical and Chemical Effects of Textile Microfibers. Water 2022, 14, 3797. https://doi.org/10.3390/w14233797
Athey SN, Carney Almroth B, Granek EF, Hurst P, Tissot AG, Weis JS. Unraveling Physical and Chemical Effects of Textile Microfibers. Water. 2022; 14(23):3797. https://doi.org/10.3390/w14233797
Chicago/Turabian StyleAthey, Samantha N., Bethanie Carney Almroth, Elise F. Granek, Paul Hurst, Alexandra G. Tissot, and Judith S. Weis. 2022. "Unraveling Physical and Chemical Effects of Textile Microfibers" Water 14, no. 23: 3797. https://doi.org/10.3390/w14233797
APA StyleAthey, S. N., Carney Almroth, B., Granek, E. F., Hurst, P., Tissot, A. G., & Weis, J. S. (2022). Unraveling Physical and Chemical Effects of Textile Microfibers. Water, 14(23), 3797. https://doi.org/10.3390/w14233797








