Simulation of the Nitrogen and Phosphorus Leaching Characteristics under Different Filter Materials of an Improved Subsurface Drainage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Laboratory Test
2.2. Simulation Theory
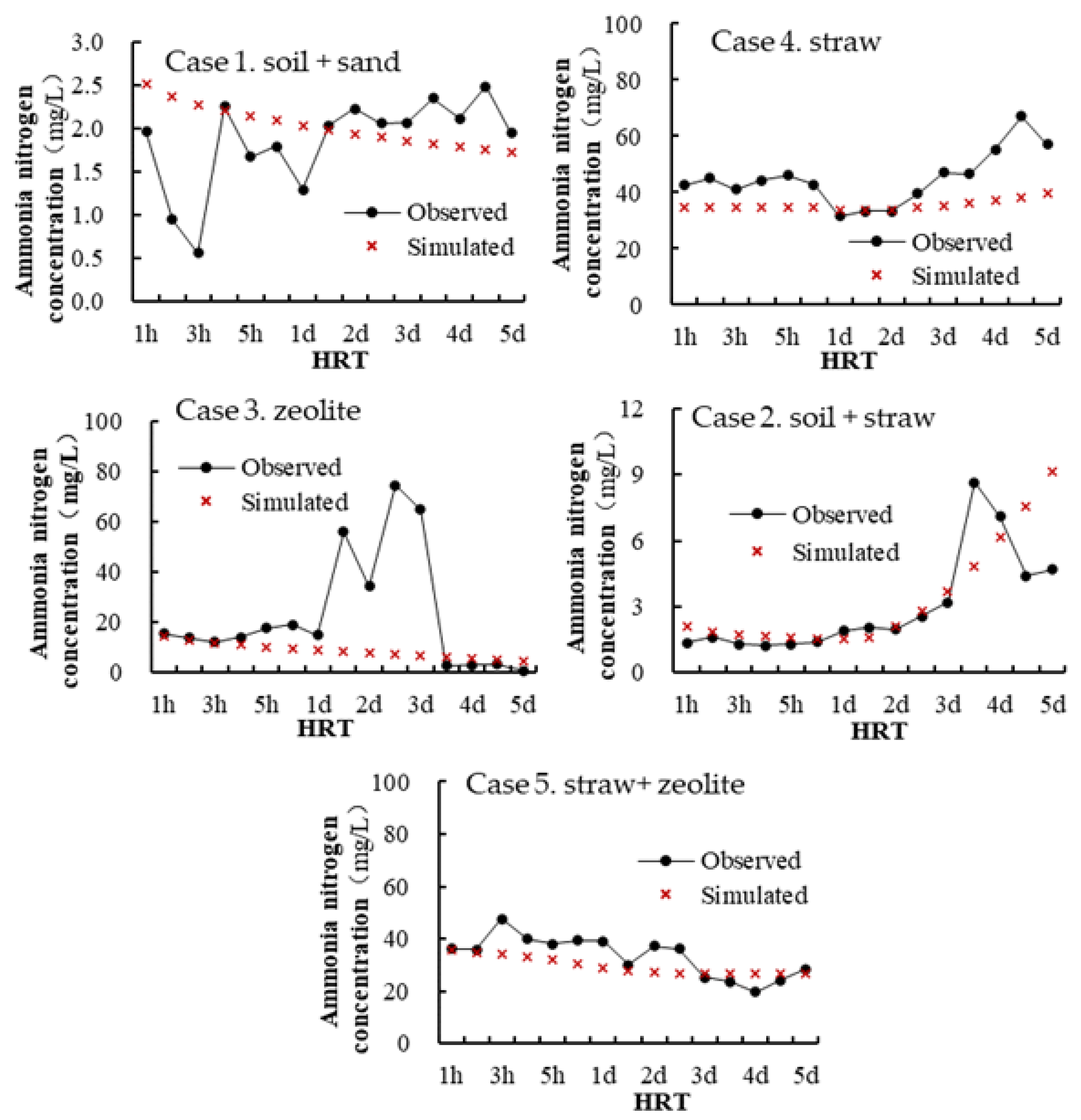
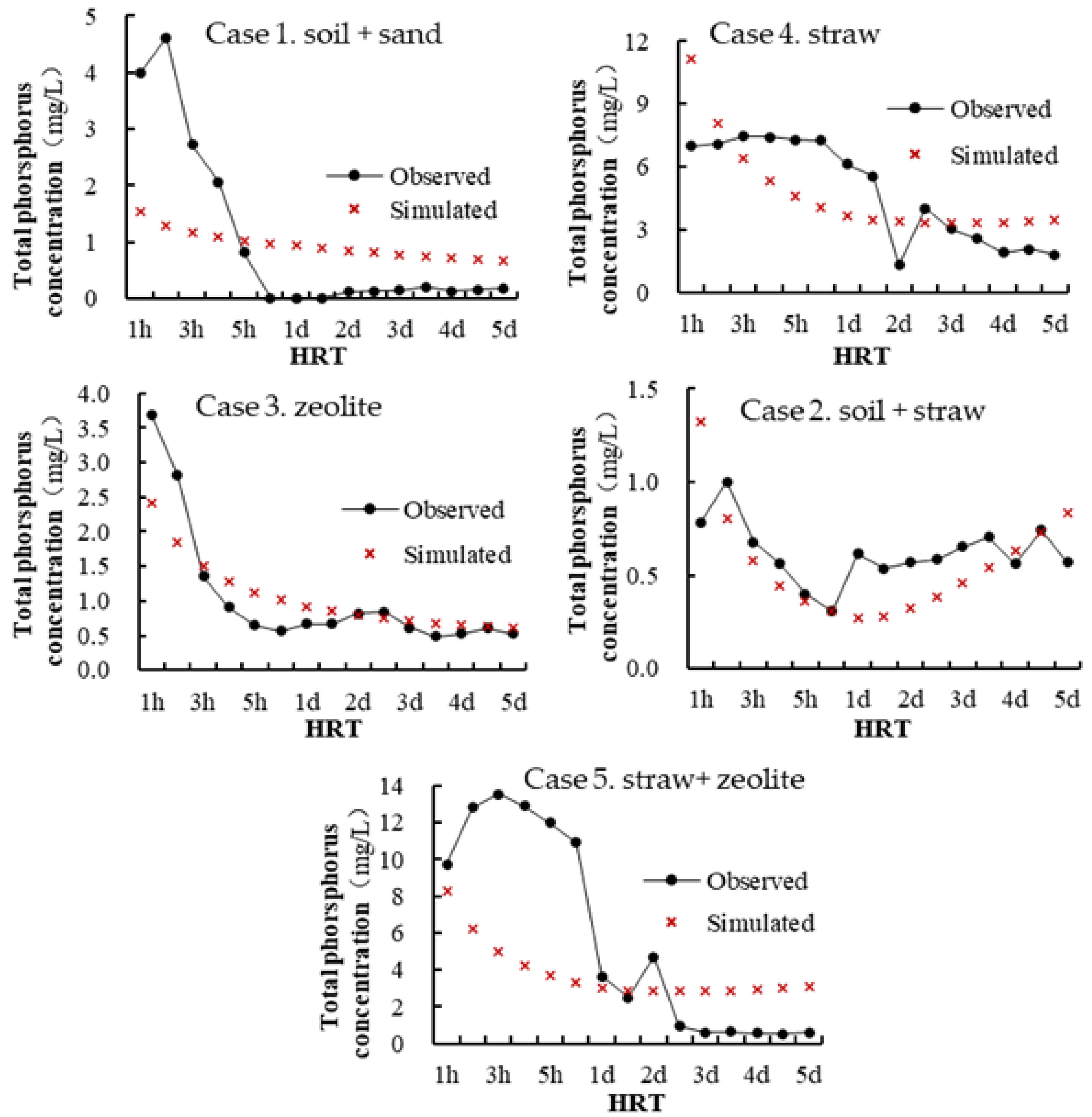
2.3. Model Calibration and Validation
2.4. Simulated Scenarios
3. Results
3.1. The Adsorption of Different Materials
3.2. Effect of Materials in Scenarios 1
3.3. Effect of Soil Depth and Location in Scenario 1
3.4. Mixed Filter with Straw in Scenario 2
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tao, Y.; Wang, S.; Xu, D.; Yuan, H.; Chen, H. Field and numerical experiment of an improved subsurface drainage system in Huaibei plain. Agric. Water Manag. 2017, 194, 24–32. [Google Scholar] [CrossRef]
- Tao, Y.; Wang, S.; Xu, D.; Qu, X. Experiment and analysis on flow rate of improved subsurface drainage with ponded water. Agric. Water Manag. 2016, 177, 1–9. [Google Scholar] [CrossRef]
- Tao, Y.; Wang, S.; Guan, X.; Xu, D. Study on Characteristics of Nitrogen and Phosphorus Loss under an Improved Subsurface Drainage. Water 2019, 11, 1467. [Google Scholar] [CrossRef] [Green Version]
- Tao, Y.; Li, N.; Wang, S.; Chen, H.; Guan, X.; Ji, M. Simulation study on performance of nitrogen loss of an improved subsurface drainage system for one-time drainage using HYDRUS-2D. Agric. Water Manag. 2021, 246, 106698. [Google Scholar] [CrossRef]
- Pan, W.; Zhi-Shan, Z.; Lei, H.; Yi-Gang, H.; Yong-Le, C. Influence of artificially placed substrates on agro-drainage ditch water quality. Chin. J. Eco-Agric. 2012, 20, 578–584. [Google Scholar]
- Bruun, J.; Pugliese, L.; Hoffmann, C.C.; Kjaergaard, C. Solute transport and nitrate removal in full-scale subsurface flow constructed wetlands of various designs treating agricultural drainage water. Ecol. Eng. 2016, 97, 88–97. [Google Scholar] [CrossRef]
- Humphrey, C.P.; Jernigan, J.; Iverson, G.; Serozi, B.; O’Driscoll, M.; Pradhan, S.; Bean, E. Field Evaluation of Nitrogen Treatment by Conventional and Single-Pass Sand Filter Onsite Wastewater Systems in the North Carolina Piedmont. Water Air Soil Pollut. 2016, 227, 225. [Google Scholar] [CrossRef]
- Cui, N.X.; Zhang, X.; Cai, M.; Chen, G.F.; Zhou, L.; Zou, G.Y. Does rice straw addition and/or Vallisneria natans (Lour.) planting contribute to enhancement in nitrate nitrogen and phosphorus removal in constructed wetlands under low temperature? Bioresour. Technol. 2022, 350, 126896. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Chen, X.X.; Wan, X.W.; Weng, B.Q.; Huang, Q. Water hyacinth (Eichhornia crassipes) waste as an adsorbent for phosphorus removal from swine wastewater. Bioresour. Technol. 2010, 101, 9025–9030. [Google Scholar] [CrossRef] [PubMed]
- Paul, D.; Hall, S.G. Biochar and Zeolite as Alternative Biofilter Media for Denitrification of Aquaculture Effluents. Water 2021, 13, 2703. [Google Scholar] [CrossRef]
- Le, T.H.X.; Mosley, L.; Marschner, P. Nitrogen and phosphorus removal from wastewater by sand with wheat straw. Environ. Sci. Pollut. Res. 2019, 26, 11212–11223. [Google Scholar] [CrossRef]
- Glaister, B.J.; Fletcher, T.D.; Cook, P.L.M.; Hatt, B.E. Co-optimisation of phosphorus and nitrogen removal in stormwater biofilters: The role of filter media, vegetation and saturated zone. Water Sci. Technol. 2014, 69, 1961–1969. [Google Scholar] [CrossRef]
- Wang, S.; Zhou, M.; Liu, L. Experimental Study on Permeability and Nitrogen Removal of Envelope Materials for Drainage Pipe. J. Irrig. Drain. 2013, 32, 21–23. [Google Scholar]
- Zhang, J.; Zhou, Q.; He, R. Mechanism of nitrogen and phosphorus removal in free-water surface constructed wetland. Ecol. Environ. 2004, 13, 98–101. [Google Scholar]
- Hassan, G.K.; El-Gohary, F.A. Evaluation of Partial Nitritation/Anammox Process for Reduction of Pollutants from Sanitary Landfill Leachate. Water Air Soil Pollut. 2021, 232, 134. [Google Scholar] [CrossRef]
- Hassan, G.K.; Gad-Allah, T.A.; Badawy, M.I.; El-Gohary, F.A. Remediation of ammonia-stripped sanitary landfill leachate by integrated heterogeneous Fenton process and aerobic biological methods. Int. J. Environ. Anal. Chem. 2021, 1–14. [Google Scholar] [CrossRef]
- Sandrin, T.R.; Maier, R.M. Impact of metals on the biodegradation of organic pollutants. Environ. Health Perspect. 2003, 111, 1093–1101. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.Q.; Huang, S.L. Mechanisms of pollutant removal in constructed wetlands and their applications bothat home and abroad. Technol. Water Treat. 2007, 33, 9–13. [Google Scholar]
- Mekala, C.; Nambi, I.M. Experimental and Simulation Studies on Nitrogen Dynamics in Unsaturated and Saturated Soil Using HYDRUS-2D. Procedia Technol. 2016, 25, 122–129. [Google Scholar] [CrossRef] [Green Version]
- Salehi, A.; Navabian, M.; Esmaeili varaki, M.; Pirmoradian, N. Evaluation of HYDRUS-2D model to simulate the loss of nitrate in subsurface controlled drainage in a physical model scale of paddy fields. Paddy Water Environ. 2017, 15, 433–442. [Google Scholar] [CrossRef]
- Hou, L.Z.; Hu, B.X.; Qi, Z.M.; Yang, H. Evaluating equilibrium and non-equilibrium transport of ammonium in a loam soil column. Hydrol. Process. 2018, 32, 80–92. [Google Scholar] [CrossRef]
- Elasbah, R.; Selim, T.; Mirdan, A.; Berndtsson, R. Modeling of Fertilizer Transport for Various Fertigation Scenarios under Drip Irrigation. Water 2019, 11, 893. [Google Scholar] [CrossRef] [Green Version]
- Tao, Y.; Wang, S.; Xu, D.; Qu, X. Effect of structure-type on the improved subsurface drainage performance in laboratory. Trans. Chin. Soc. Agric. Mach. 2016, 47, 113–118. [Google Scholar]
- Filipović, V.; Mallmann, F.; Coquet, Y.; Simunek, J.J. Numerical simulation of water flow in tile and mole drainage systems. Agric. Water Manag. 2014, 146, 105–114. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Simunek, J.J.; Zhenting, Z.; Jing, L.; Ni, L. Evaluation of nitrogen balance in a direct-seeded-rice field experiment using Hydrus-1D. Agric. Water Manag. 2015, 148, 213–222. [Google Scholar] [CrossRef]
- Zhang, F.; Kang, S.; Li, Z. Numerical simulation of nutrient transfer and absorption in soil-root system. J. Northwest Sci-Tech Univercity Agric. For. 2002, 30, 7–10. [Google Scholar]
- Zhang, Z. Phosphorus diffusion in soil. Res. Soil Water Conserv. 1987, 2, 27–34. [Google Scholar]
- Hudan, T.; Mulideer, T.; Zhu, Z. Experimental study on longitudinal dispersion coefficient of typical soil in Northern Xinjiang. Xinjiang Agric. Sci. 2021, 58, 151–158. [Google Scholar]
- Plumb, O.A.; Whitaker, S. Dispersion in heterogeneous porous media,1, local volume averaging and large-scale averaging. Water Resour. Res. 1988, 24, 913–926. [Google Scholar] [CrossRef]
- Hofstra, N.; Bouwman, A.F. Denitrification in agricultural soils: Summarizing published data and estimating global annual rates. Nutr. Cycl. Agroecosystems 2005, 72, 267–278. [Google Scholar] [CrossRef]
- Drazkiewicz, M. Is nitrification affected by the diameter and other properties of soil aggregates? Folia Microbiol. 1996, 41, 76–80. [Google Scholar] [CrossRef]
- Hsieh, C.H.; Davis, A.P.; Needelman, B.A. Nitrogen removal from urban stormwater runoff through layered bioretention columns. Water Environ. Res. 2007, 79, 2404–2411. [Google Scholar] [CrossRef]
- Mery, C.; Guerrero, L.; Alonso-Gutierrez, J.; Figueroa, M.; Lema, J.M.; Montalvo, S.; Borja, R. Evaluation of natural zeolite as microorganism support medium in nitrifying batch reactors: Influence of zeolite particle size. J. Environ. Sci. Health Part A-Toxic/Hazard. Subst. Environ. Eng. 2012, 47, 420–427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garrido-Fernandez, J.M.; Mendez, R.; Lema, J.M.; Lazarova, V. The circulating floating bed reactor: Effect of particle size distribution of the carrier on ammonia conversion. Water Sci. Technol. 2000, 41, 393–400. [Google Scholar] [CrossRef]
- Mazeikiene, A.; Valentukeviciene, M.; Jankauskas, J. Laboratory study of ammonium ion removal by using zeolite (clinoptilolite) to treat drinking water. J. Environ. Eng. Landsc. Manag. 2010, 18, 54–61. [Google Scholar] [CrossRef]
- Piccoli, I.; Virga, G.; Maucieri, C.; Borin, M. Digestate Liquid Fraction Treatment with Filters Filled with Recovery Materials. Water 2021, 13, 21. [Google Scholar] [CrossRef]
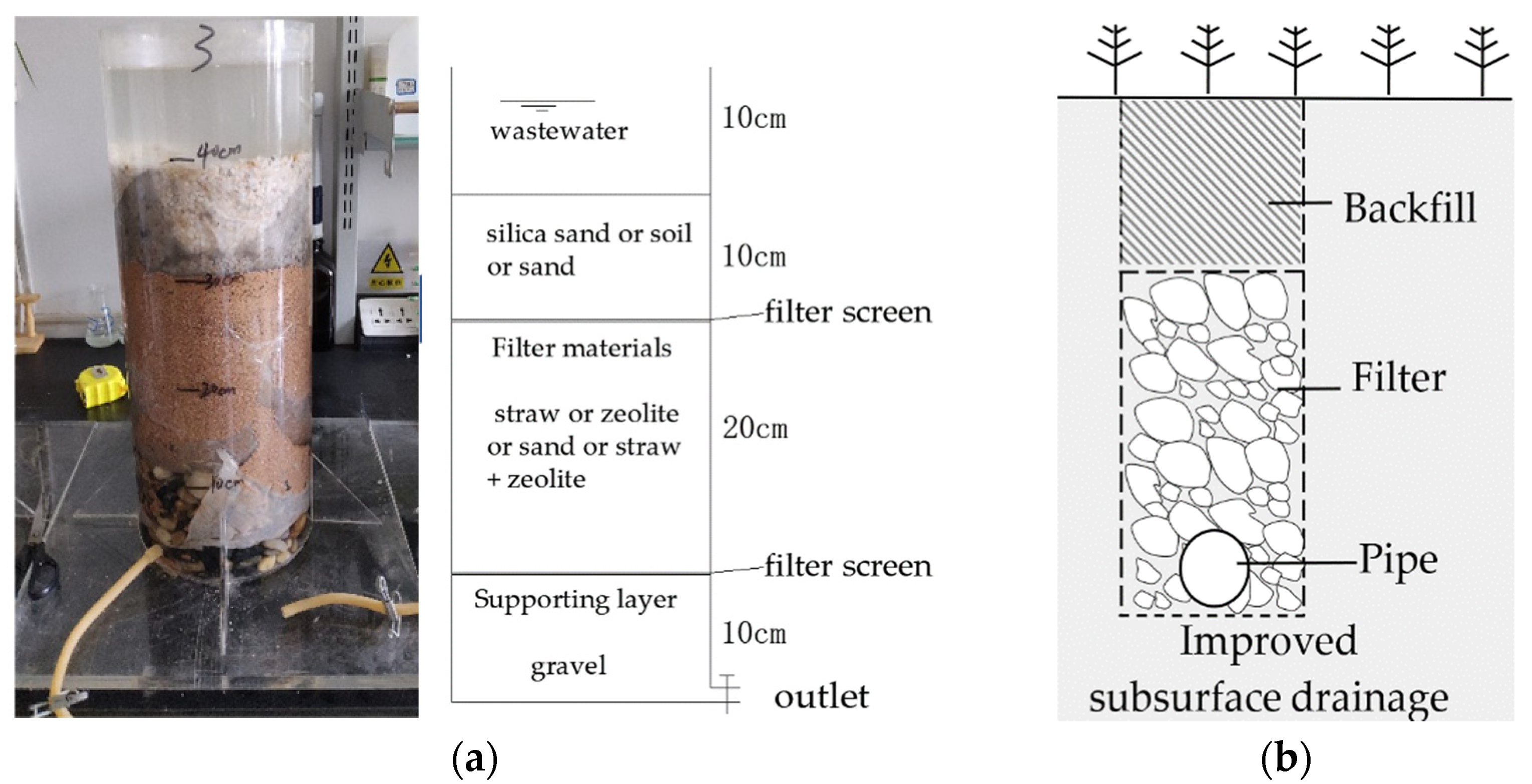
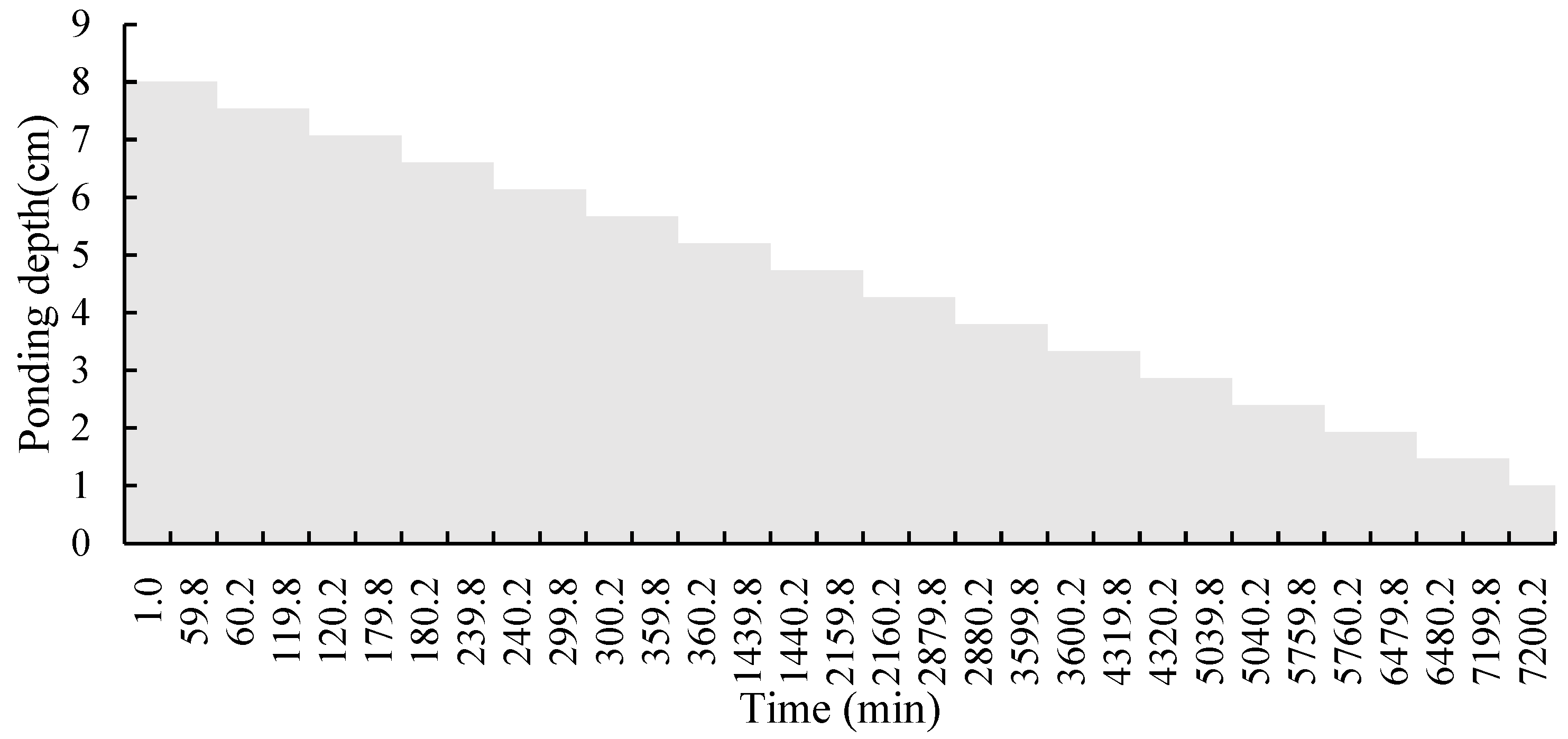
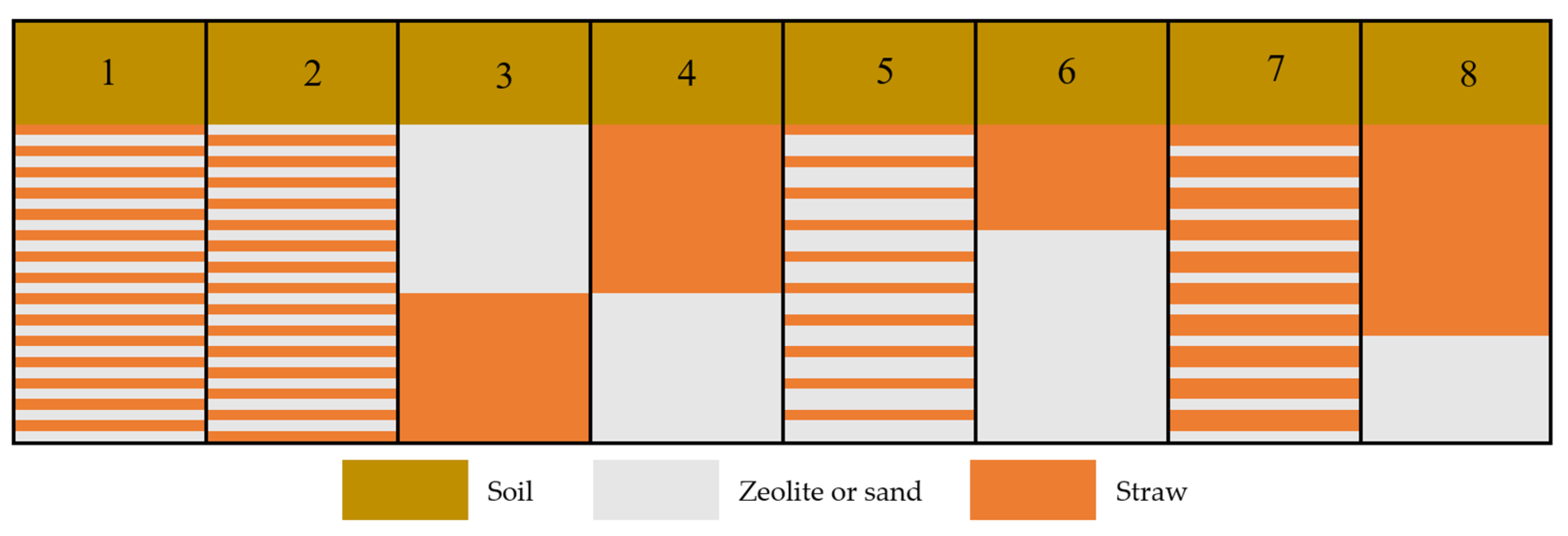


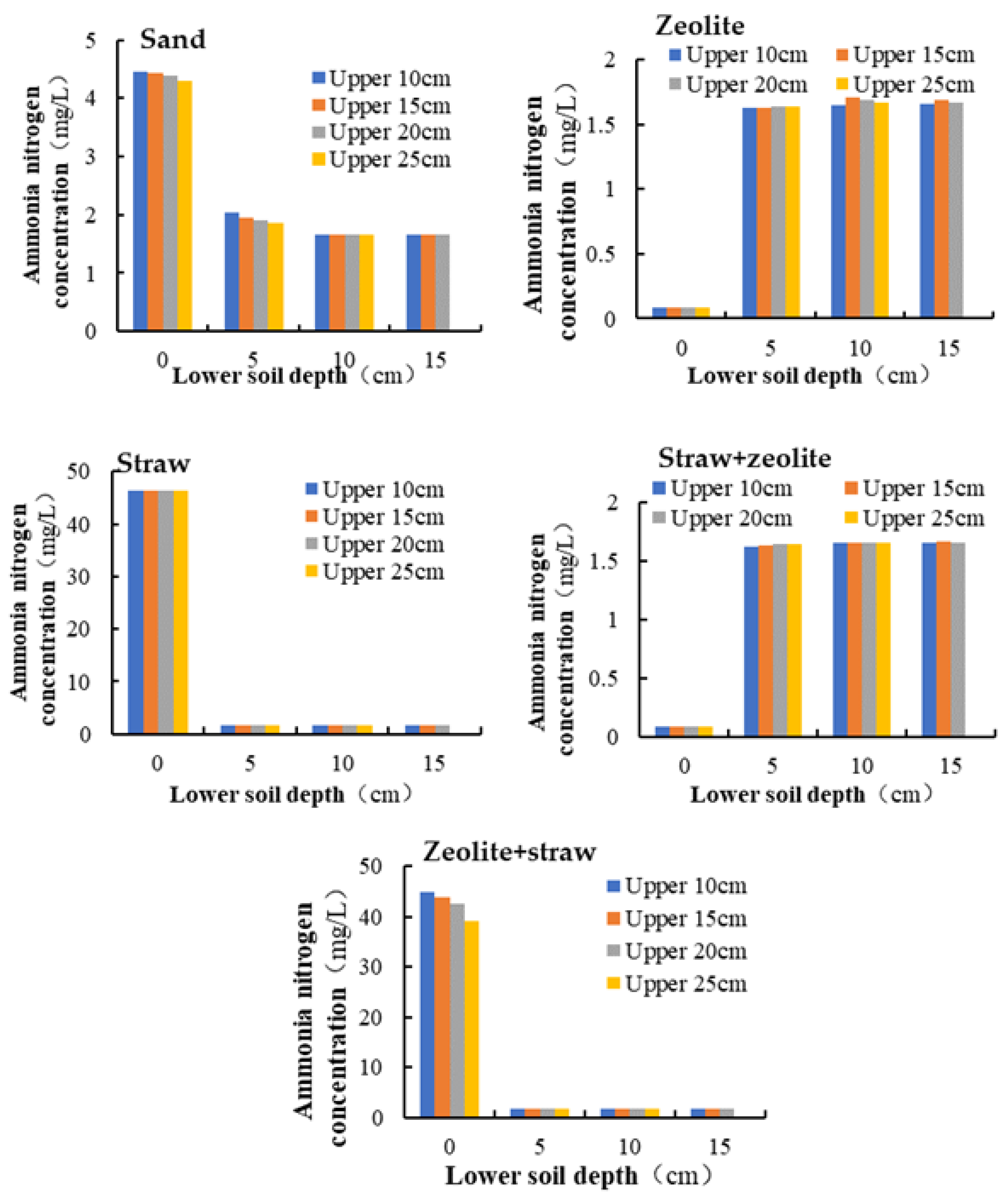
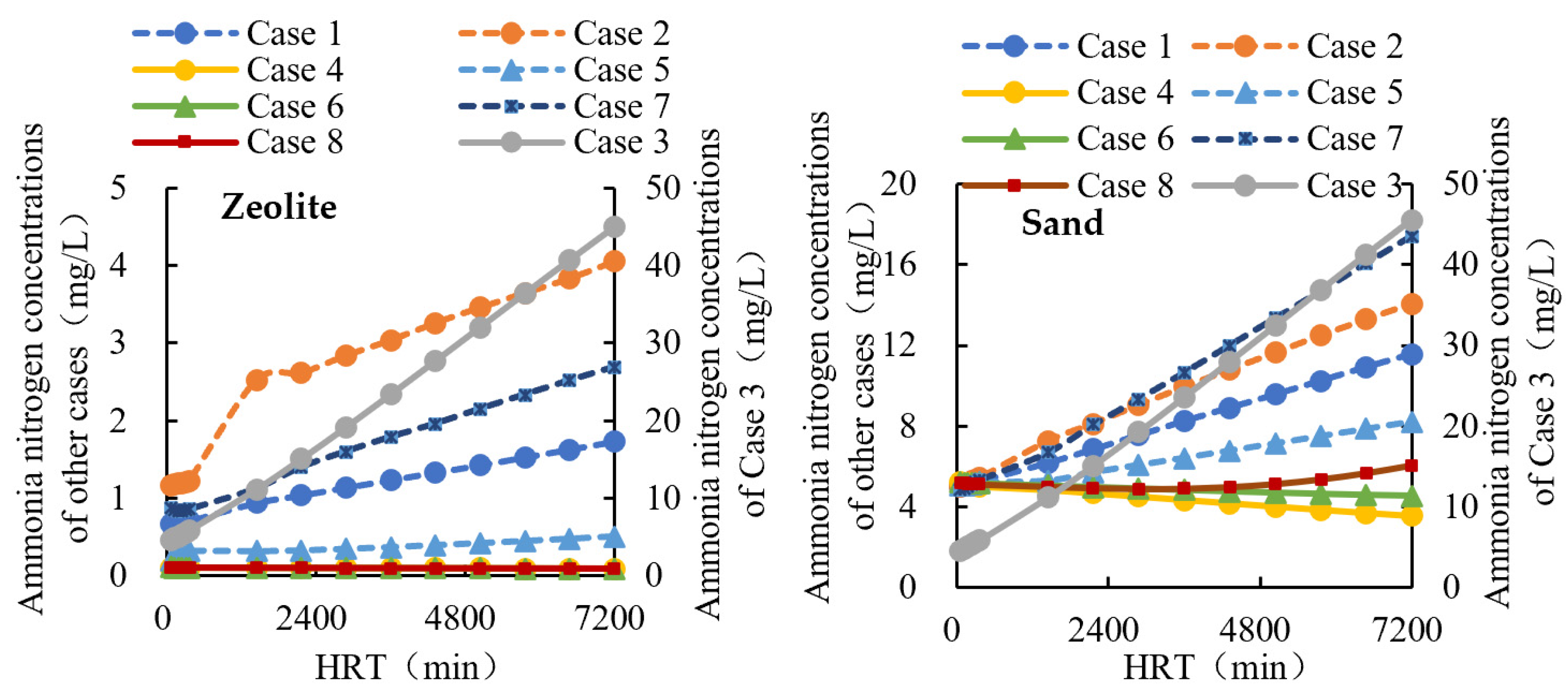
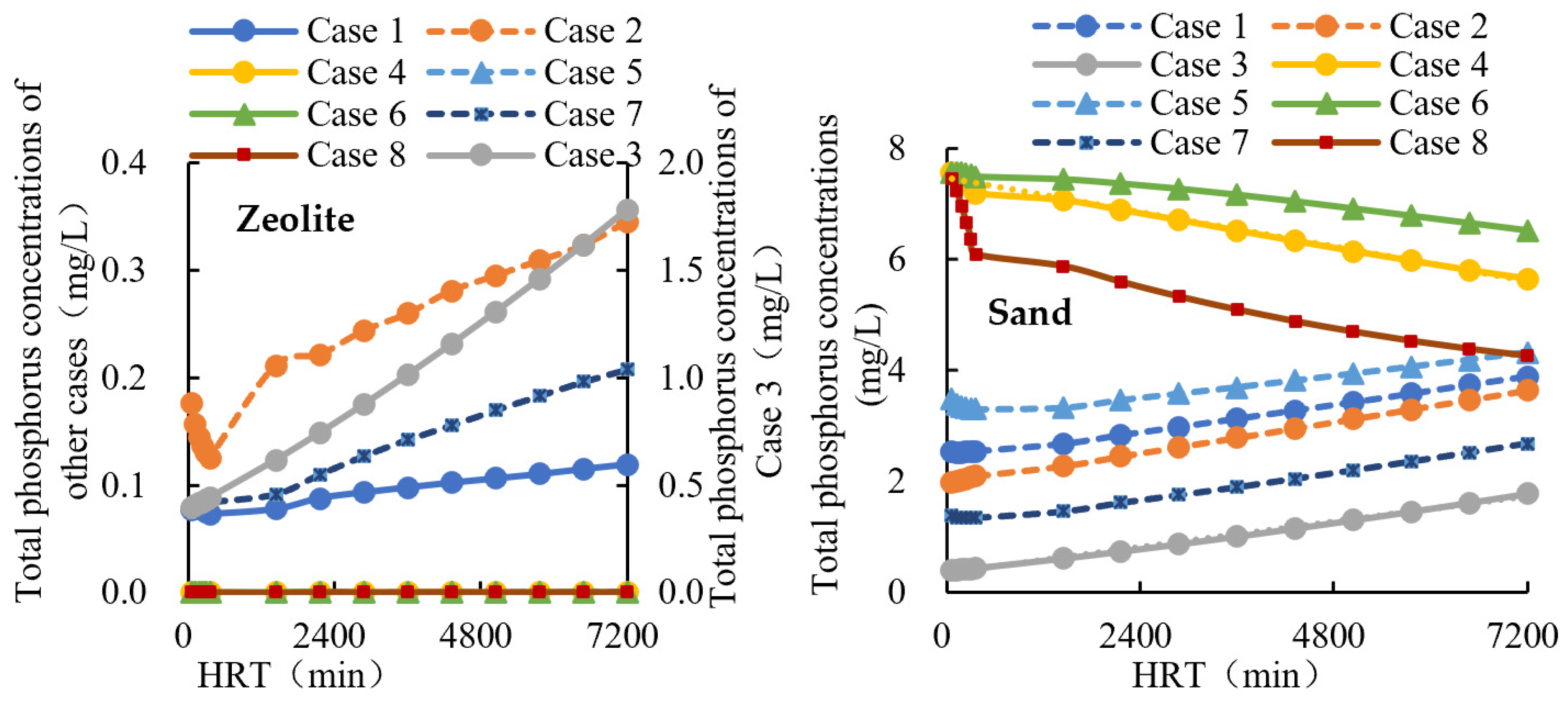
| Location | Case 1 | Case 2 | Case 3 | Case 4 | Case 5 |
|---|---|---|---|---|---|
| Upper 10 cm | soil | soil | sand | silica sand | silica sand |
| Middle 20 cm | sand | straw | zeolite | straw | zeolite + straw |
| Lower 10 cm | gravel | gravel | gravel | gravel | gravel |
| For short | soil + sand | soil + straw | zeolite | straw | zeolite + straw |
| Material | θr (cm3 cm−3) | θs (cm3 cm−3) | a (cm−1) | n | Ks (cm min−1) | l |
|---|---|---|---|---|---|---|
| Soil | 0.05 | 0.44 | 0.014 | 1.8 | 0.0629 | 0.5 |
| Sand | 0.01 | 0.42 | 0.02 | 1.9 | 2 | 0.5 |
| Gravel | 0.005 | 0.42 | 0.16 | 2.8 | 4 | 0.5 |
| Straw | 0 | 0.48 | 0.018 | 1.9 | 0.69 | 0.5 |
| Zeolite | 0.01 | 0.42 | 0.02 | 1.9 | 2 | 0.5 |
| Silica sand | 0.01 | 0.42 | 0.02 | 1.9 | 0.8 | 0.5 |
| Material | Adsorption Coefficient of N | DL (cm) | Zero-Order N Transformation Rate Constants KdNo (min−1) | Adsorption Coefficient of P | Zero-order P Transformation Rate Constants KdPo (min−1) | ||
|---|---|---|---|---|---|---|---|
| KdN (cm3·mg−1) | βN | KdP (cm3·mg−1) | βP | ||||
| Soil | 0.0032 | 1 | 3 | - | 0.002 | 0.7 | - |
| Sand | 0.001 | 1 | 20 | - | 0.0006 | 1 | - |
| Gravel | 0.00046 | 1 | 30 | - | 2 × 10−6 | 1 | - |
| Straw | 0.000085 | 0.51 | 12 | 4 × 10−9 | 0.0016 | 0.7 | 1.8 × 10−9 |
| Zeolite | 0.01 | 0.8 | 4 | - | 0.004 | 0.45 | - |
| Silica sand | 0.0012 | 1 | 10 | - | 0.001 | 1 | - |
| Case | Case 1 Soil + Sand | Case 4 Straw | Case 3 Zeolite | Case 2 Soil + Straw | Case 5 Zeolite + Straw |
|---|---|---|---|---|---|
| R | 0.57 | 0.90 | 0.81 | 0.60 | 0.68 |
| RE (%) | 9.46 | −21.1 | −14.4 | −10.8 | −11.2 |
| Case | Case 1 Soil + Sand | Case 4 Straw | Case 3 Zeolite | Case 2 Soil + Straw | Case 5 Zeolite + Straw |
|---|---|---|---|---|---|
| R | 0.88 | 0.60 | 0.94 | 0.60 | 0.85 |
| RE (%) | −7.49 | −2.3 | 0.31 | −10.8 | 9.45 |
| Impact Factor | Values of the Parameters |
|---|---|
| Upper soil depth | 10 cm, 15 cm, 20 cm, 25 cm |
| Filter material | Straw, zeolite, sand, straw (upper) + zeolite (lower), zeolite (upper) + straw (lower) |
| Filter thickness | Based on the upper and lower soil depth |
| Lower soil depth | 0 cm, 5 cm, 10 cm, 15 cm |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tao, Y.; Chang, X.; Wang, S.; Guan, X.; Liu, J. Simulation of the Nitrogen and Phosphorus Leaching Characteristics under Different Filter Materials of an Improved Subsurface Drainage. Water 2022, 14, 3744. https://doi.org/10.3390/w14223744
Tao Y, Chang X, Wang S, Guan X, Liu J. Simulation of the Nitrogen and Phosphorus Leaching Characteristics under Different Filter Materials of an Improved Subsurface Drainage. Water. 2022; 14(22):3744. https://doi.org/10.3390/w14223744
Chicago/Turabian StyleTao, Yuan, Xiaomin Chang, Shaoli Wang, Xiaoyan Guan, and Jing Liu. 2022. "Simulation of the Nitrogen and Phosphorus Leaching Characteristics under Different Filter Materials of an Improved Subsurface Drainage" Water 14, no. 22: 3744. https://doi.org/10.3390/w14223744
APA StyleTao, Y., Chang, X., Wang, S., Guan, X., & Liu, J. (2022). Simulation of the Nitrogen and Phosphorus Leaching Characteristics under Different Filter Materials of an Improved Subsurface Drainage. Water, 14(22), 3744. https://doi.org/10.3390/w14223744






