Surface Water under Growing Anthropogenic Loads: From Global Perspectives to Regional Implications
Abstract
:1. Introduction
- the use of water resources as a life necessity on the planet;
- the effect of climate warming on the eutrophication of waters;
- the consequences of acidic deposition for the water acidification;
- the transfer of toxic elements and compounds to natural waters;
- principal theoretical approaches to the evaluation of critical loads.
2. The Importance of Freshwater in the Life Support
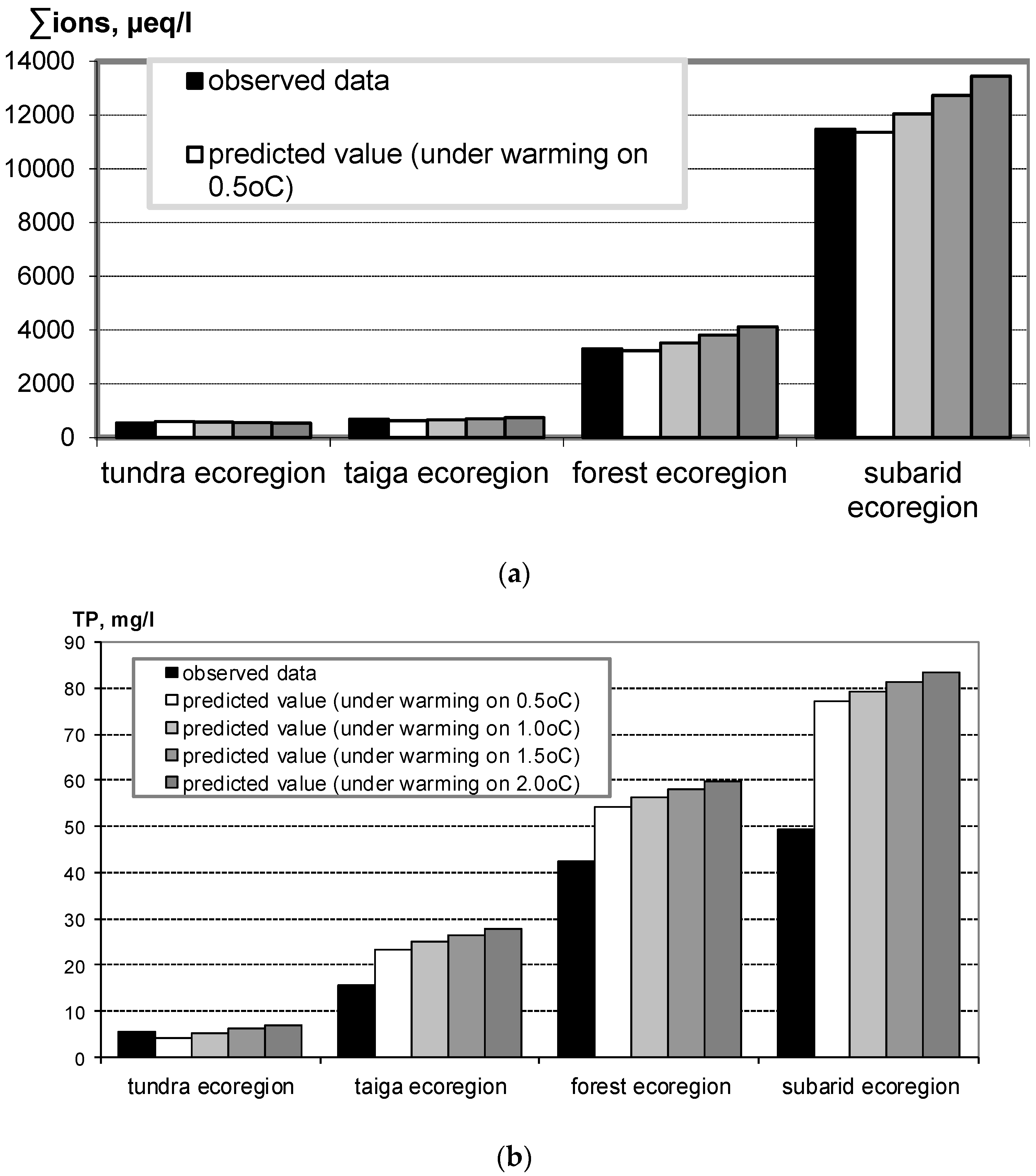
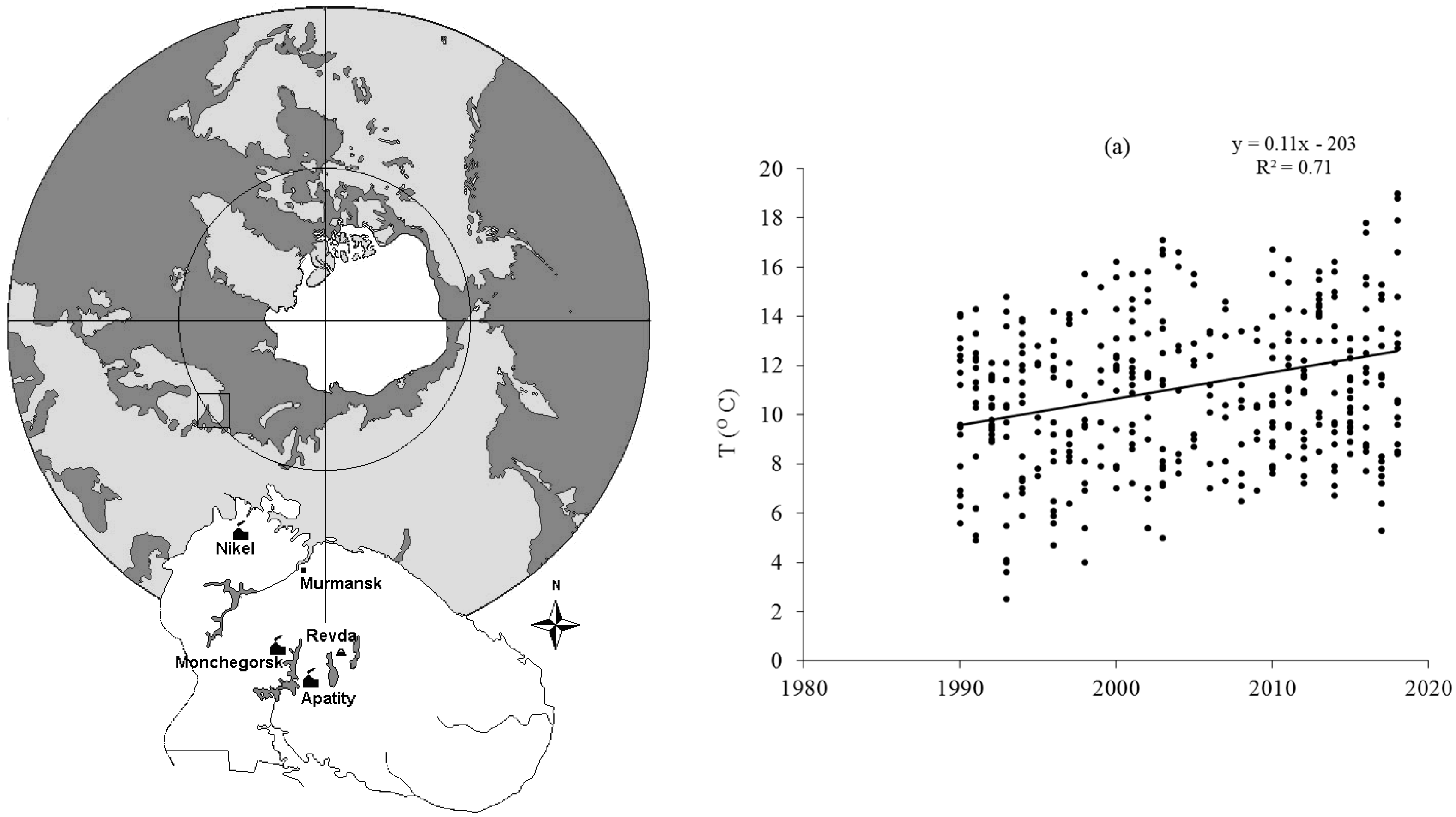
3. Effect of Climate Warming on Aquatic Systems
4. Eutrophication
5. Acidifying Compounds
6. Contamination of the Waters with Toxic Compounds
- Metals and metalloids, which are elements occurring in nature and toxically affecting living organisms when in high concentrations or certain speciation;
- Persistent Organic Pollutants (POPs), are industrially synthesized compounds or byproducts of some technological processes involving naturally occurring compounds. This group comprises a broad class of organic xenobiotics: herbicides, insecticides, dioxins, furans, phthalates, etc.
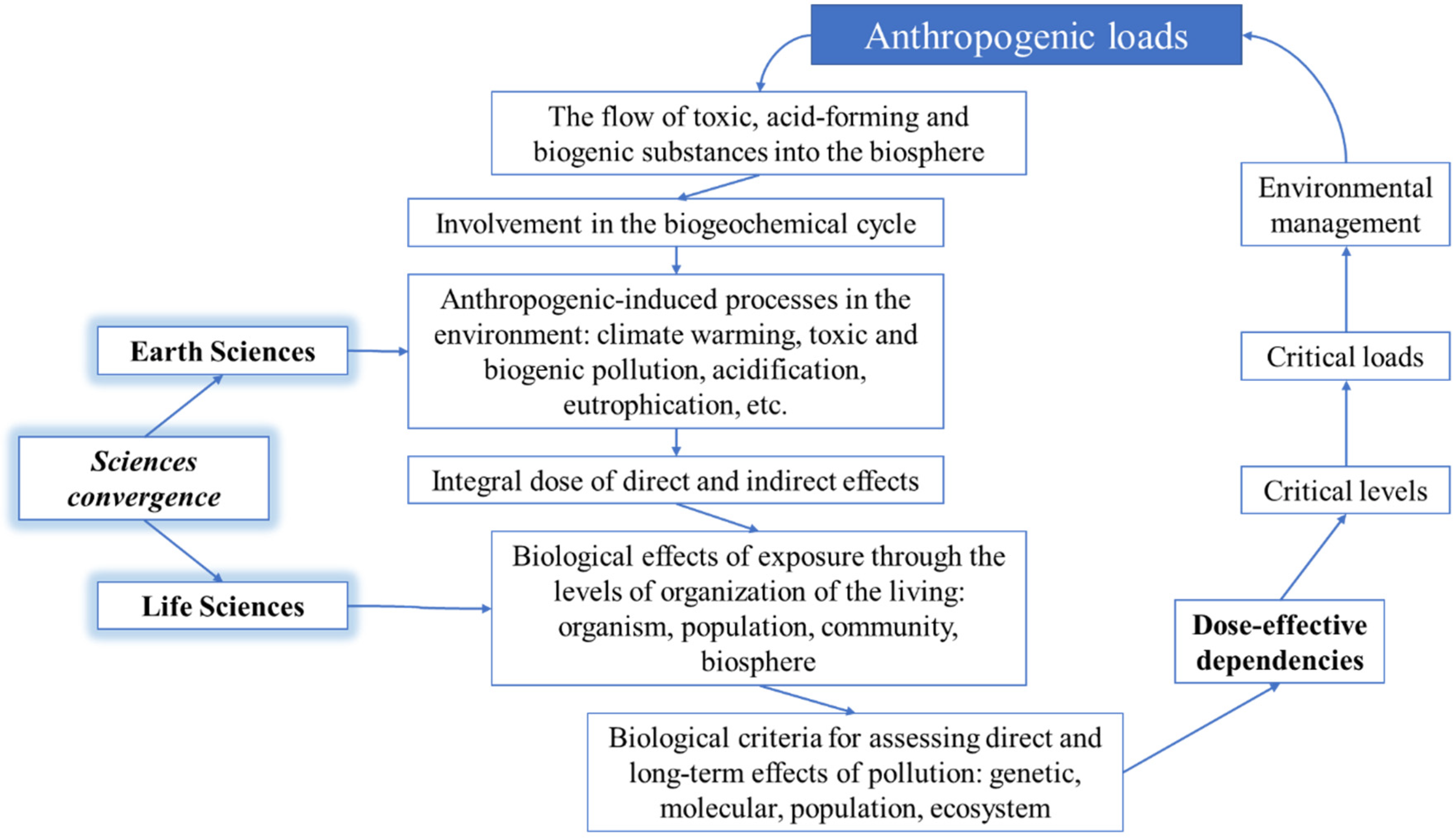
7. Critical Loads: Theoretical Approaches
- (1).
- behavior patterns of anthropogenically introduced elements, interaction with natural factors, involvement in the natural cycles of elements, and the bioavailability of ecotoxicants and
- (2).
- stability mechanisms and variability limits of biological systems, the long-term consequences of anthropogenic contaminations for living organisms, and the estimation criteria.
8. Conclusions
Funding
Conflicts of Interest
References
- Vernadsky, V.I. Scientific Ideas as Planetary Phenomenon; Nauka: Moscow, Russia, 1991; p. 270. [Google Scholar]
- Rockström, J.; Steffen, W.; Noone, K.; Persson, A.; Chapin, F.S., 3rd; Lambin, E.F.; Lenton, T.M.; Scheffer, M.; Folke, C.; Schellnhuber, H.J.; et al. A safe operating space for humanity. Nature 2009, 461, 472–475. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Davidson, E.A.; Zou, T.; Lassaletta, L.; Quan, Z.; Li, T.; Zhang, W. Quantifying nutrient budgets for sustainable nutrient management. Glob. Biogeochem. Cycles 2020, 34, e2018GB006060. [Google Scholar] [CrossRef] [Green Version]
- Danilov-Danilyan, V.I.; Khranovich, I.L. Water Resources Management. Coordination of Water Use Strategies; Nauchny mir: Moscow, Russia, 2010; p. 233. [Google Scholar]
- AR5 Synthesis Report: Climate Change. 2014. Available online: https://www.ipcc.ch/report/ar5/syr/Ar5/ar5.html (accessed on 19 January 2022).
- AR6 Climate Change 2022: Mitigation of Climate Change—IPCC. Sixth Assessment Report. 2022. Available online: https://www.ipcc.ch/report/sixth-assessment-report (accessed on 9 July 2021).
- Cesonien, E.L.; Šileikienė, D.; Marozas, V.; Čiteikė, L. Influence of Anthropogenic Loads on Surface Water Status: A Case Study in Lithuania. Sustainability 2021, 13, 4341. [Google Scholar] [CrossRef]
- Biswas, A.K. Water Security Under Climate Change; Asit, K., Ed.; Springer Nature Singapore Pte Ltd.: Singapore, 2022; p. 830. [Google Scholar] [CrossRef]
- Moiseenko, T.I. A Fate of Metals in Arctic Surface Waters. Method for Defining Critical Levels. Sci. Total Environ. 1999, 236, 19–39. [Google Scholar] [CrossRef]
- Moiseenko, T.I. Aquatic ecotoxicology: Theoretical principles and practical application. Water Resour. 2008, 35, 530–541. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Skjelkvåle, B.L.; Gashkina, N.A.; Shalabodov, A.D.; Khoroshavin, V.Y. Water chemistry in small lakes along a transect from boreal to arid ecoregions in European Russia: Effects of air pollution and climate change. Appl. Geochem. 2013, 28, 69–79. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Dinu, M.I.; Bazova, M.M.; de Wit, H.A. Long-term changes in the water chemistry of subarctic lakes as a response to reduction of air pollution: Case study in the Kola North, Russia. Water Air Soil Pollut. 2015, 226, 1–12. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Dinu, M.I.; Gashkina, N.A.; Jones, V.; Khoroshavin, V.Y.; Kremleva, T.A. Present status of water chemistry and acidification under nonpoint sources of pollution across European Russia and West Siberia. Environ. Res. Lett. 2018, 13, 105007. [Google Scholar] [CrossRef] [Green Version]
- Moiseenko, T.I.; Bazova, M.M.; Dinu, M.I.; Gashkina, N.A.; Kudryavtseva, L.P. Changes in the Geochemistry of Land Waters at Climate Warming and a Decrease in Acid Deposition: Recovery of the Lakes or Their Evolution? Geochem. Int. 2022, 60, 685–701. [Google Scholar] [CrossRef]
- Moiseenko, T.; Sharov, A. Large Russian lakes Ladoga, Onega, and Imandra under strong pollution and in the period of revitalization: A review. Geosciences 2019, 9, 492. [Google Scholar] [CrossRef] [Green Version]
- Edelstein, K.K. Continental Hydrology; Publishing Center “Academy”: Moscow, Russia, 2011; p. 304. [Google Scholar]
- UN World Water Development Report: Valuing Water. 2021. Available online: https://www.unwater.org/publications/un-world-water-development-report-2021 (accessed on 27 September 2022).
- Arctic Freshwater System in a Changing Climate, WCRP CliC Project. AMAP. IASC CliC/AMAP/IASC. 2016. Available online: http://www.amap.no/documents/doc/The-Arctic-Freshwater-System-in-a-Changing-Climate/1375.AMAП (accessed on 1 January 2020).
- Hayhoe, K.; Wake, C.P.; Huntington, T.G.; Luo, L.; Schwartz, M.D.; Sheffield, J.; Wood, E.; Anderson, B.; Bradbury, J.; DeGaetano, A. Past and future hanges in climate and hydrological indicators in the US Northeast. Clim. Dyn. 2007, 28, 381–407. [Google Scholar] [CrossRef]
- Georgiadi, A.G.; Kashutina, E.A. Long-term runoff changes of the largest siberian rivers. Izv. Ross. Akad. Nauk. Seriya Geogr. 2016, 5, 70–81. (In Russian) [Google Scholar] [CrossRef]
- Jin, H.; Huang, Y.; Bense, V.F.; Ma, Q.; Marchenko, S.S.; Shepelev, V.V.; Hu, Y.; Liang, S.; Spektor, V.V.; Jin, X.; et al. Permafrost Degradation and Its Hydrogeological Impacts. Water 2022, 14, 372. [Google Scholar] [CrossRef]
- Pavlov, A.V.; Malkova, G.D. Small-scale mapping of trends of the contemporary ground temperature changes in the Russian north. Earth Cryosphere 2009, 103, 32–39. [Google Scholar]
- Grosse, G.; Goetz, S.; McGuire, D.; Romanovsky, V.E.; Schuur, E.A. Changing permafrost in a warming world and feedbacks to the Earth system. Environ. Res. Lett. 2016, 11, 040201. [Google Scholar] [CrossRef]
- Gao, X.; Schlosser, C.A.; Sokolov, A.; Anthony, K.W.; Zhuang, Q.; Kicklighter, D. Permafrost degradation and methane: Low risk of biogeochemical climate-warming feedback. Environ. Res. Lett. 2013, 8, 035014. [Google Scholar] [CrossRef] [Green Version]
- Meingast, K.M.; Kane, E.; Coble, A.A.; Marcarelli, A.M.; Toczydlowski, D. Climate, snowmelt dynamics and atmospheric deposition interact to control dissolved organic carbon export from a northern forest stream over 26 years. Environ. Res. Lett. 2020, 15, 104034. [Google Scholar] [CrossRef]
- Campbell, J.L.; Rustad, L.E.; Boyer, E.W.; Christopher, S.F.; Driscoll, C.T.; Fernandez, I.J.; Groffman, P.M.; Houle, D.; Kiekbusch, J.; Magill, A.H.; et al. Consequences of climate change for biogeochemical cycling in forests of northeastern North America Can. J. For. Res. 2009, 39, 264–284. [Google Scholar]
- De Wit, H.A.; Stoddard, J.L.; Monteith, D.T.; Sample, J.E.; Austnes, K.; Couture, S.; Fölster, J.; Higgins, S.N.; Houle, D.; Hruška, J.; et al. Cleaner air reveals growing influence of climate on dissolved organic carbon trends in northern headwaters. Environ. Res. Lett. 2021, 16, 104009. [Google Scholar] [CrossRef]
- Watmough, S.A.; Eimers, C.; Baker, S. Impediments to recovery from acid deposition. Atmos. Environ. 2016, 146, 15–27. [Google Scholar]
- Gavin, A.L.; Nelson, S.J.; Klemmer, A.J.; Fernandez, I.J.; Strock, K.E.; McDowell, W.H. Acidification and climate linkages to increased dissolved organic carbon in high elevation lakes. Water Resour. Res. 2018, 54, 5187–5877. [Google Scholar] [CrossRef]
- Houle, D.; Couture, S.; Gagnon, C. Relative role of decreasing precipitation sulfate and climate on recent lake recovery. Glob. Biogeochem. Cycles 2010, 24, 4029. [Google Scholar] [CrossRef]
- Corman, J.R.; Bertolet, B.L.; Casson, N.J.; Sebestyen, S.D.; Kolka, R.K.; Stanley, E.H. Nitrogen and phosphorus loads to temperate seepage lakes associated with allochthonous dissolved organic carbon loads. Geophys. Res. Lett. 2018, 45, 5481–5490. [Google Scholar] [CrossRef]
- Jellison, R.; Williams, W.D.; Timms, B.; Aladin, N.V. Salt lakes: Values and future. In Aquatic Ecosys-Tems: Trends and Global Prospects; Cambridge University Press: Cambridge, UK, 2008; pp. 92–110. [Google Scholar]
- Pouladi, P.; Mohammad, A.; Afshar, H.; Molajou, A.; Farahmand, H. Agent-based socio-hydrological modeling for restoration of Urmia Lake: Application of theory of planned behavior. J. Hydrol. 2019, 576, 736–748. [Google Scholar] [CrossRef]
- Tussupova, K.; Hjorth, A.P.; Morave, M. Drying lakes: A review on the applied restoration strategies and health conditions in contiguous areas. Water 2020, 12, 749. [Google Scholar] [CrossRef] [Green Version]
- Rudneva, I.I.; Shaidaa, V.G.; Shcherba, A.V.; Zavyalova, A.V. Influence of Climatic Factors on Interan-nual and Seasonal Dynamics of the Environmental State of the Salt Lake Adzhi-Baichi (Crimea). Arid. Ecosys-Tems 2021, 11, 434–442. [Google Scholar] [CrossRef]
- Mojtahedi, A.; Dadashzadeh, M.; Azizkhani, M.; Mohammadian, A.; Almasi, R. Assessing climate and human activity effects on lake characteristics using spatio-temporal satellite data and an emotional neural network. Environ. Earth Sci. 2022, 81, 1–20. [Google Scholar] [CrossRef]
- Mekonnen, M.M.; Hoekstra, A.Y. Global anthropogenic phosphorus loads to freshwater and associated grey water footprints and water pollution levels: A high-resolution global study. Water Resour. Res. 2018, 54, 345–358. [Google Scholar] [CrossRef] [Green Version]
- Penuelas, J.; Poulter, B.; Sardans, J.; Ciais, P.; Van Der Velde, M.; Bopp, L. Human-induced nitrogen–phosphorus imbalances alter natural and managed ecosystems across the globe. Nat. Commun. 2013, 4, 2934. [Google Scholar] [CrossRef] [Green Version]
- Davidson, E.A.; Markewitz, D.; de Figueiredo, R.O.; de Camargo, P.B. Nitrogen fixation inputs in pasture and early successional forest in the Brazilian amazon region: Evidence from a claybox mesocosm study. J. Geophys. Res. Biogeosciences 2018, 123, 712–721. [Google Scholar] [CrossRef]
- Bouwman, A.; Beusen, A.; Griffioen, J.; Van Groenigen, J.; Hefting, M.; Oenema, O.; Van Puijenbroek, P.J.T.M.; Seitzinger, S.; Slomp, C.P.; Stehfest, E. Global trends and uncertainties in terrestrial denitrification and N2O emissions. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20130112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bouwman, A.F.; Beusen, A.H.; Lassaletta, L.; van Apeldoorn, D.F.; van Grinsven, H.J.; Zhang, J.; van Ittersum, M.K. Lessons from temporal and spatial patterns in global use of N and P fertilizer on cropland. Sci. Rep. 2017, 7, 40366. [Google Scholar] [CrossRef] [PubMed]
- Gruber, N.; Galloway, J.N. An Earth-system perspective of the global nitrogen cycle. Nature 2008, 451, 293–296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Powers, S.M.; Bruulsema, T.W.; Burt, T.P.; Chan, N.I.; Elser, J.J.; Haygarth, P.M. Long-term accumulation and transport of anthropogenic phosphorus in three river basins. Nat. Geosci. 2016, 9, 353–356. [Google Scholar] [CrossRef]
- Abid, A.A.; Gill, S.S. Eutrophication: Causes, Consequences and Control; Springer: Dordrecht, The Netherlands; Heidelberg, Germany; London, UK, 2014; Volume 2, p. 262. [Google Scholar]
- Beusen, A.H.; Bouwman, A.F.; Van Beek, L.P.; Mogollón, J.M.; Middelburg, J.J. Global riverine N and P transport to ocean increased during the 20th century despite increased retention along the aquatic continuum. Biogeosciences 2016, 13, 2441–2451. [Google Scholar] [CrossRef] [Green Version]
- Nilsson, A.E.; Wilson, S.J.; Murray, J.L.; Huntington, H.P. Arctic Pollution Issues: A State of the Arctic Environment Report; Publication by AMAP: Oslo, Norway, 1997; p. 188. [Google Scholar]
- Stoddard, J.L.; Van Sickle, J.; Herlihy, A.T.; Brahney, J.; Paulsen, S.; Peck, D.V. Continental-scale increase in lake and stream phosphorus: Are oligotrophic systems disappearing in the United States? Environ. Sci. Technol. 2016, 50, 3409–3415. [Google Scholar] [CrossRef] [Green Version]
- Galloway, J.N. Acid deposition: Perspectives in time and space. Water Air Soil Pollut. 1995, 85, 15–24. [Google Scholar] [CrossRef]
- Kvaeven, B.; Ulstein, M.J.; Skjelkvåle, B.L. ICP Waters—An international program for surface water monitoring. Water Air Soil Pollut. 2001, 130, 775–780. [Google Scholar] [CrossRef]
- Monteith, D.T.; Stoddard, J.L.; Evans, C.D.; de Wit, H.A.; Forsius, M.; Hogasen, T.; Wilander, A.; Skjelkvale, B.L.; Jeffries, D.S.; Vuorenmaa, J.; et al. Dissolved organic carbon trends resulting from changes in atmospheric deposition chemistry. Nature 2007, 450, 537–539. [Google Scholar] [CrossRef]
- Skelkvale, B.L.; Stoddard, J.L.; Andersen, T. Trends in surface water acidification in Europe and North America (1989–1998). Water Air Soil Pollut. 2001, 130, 787–792. [Google Scholar] [CrossRef]
- Skjelkvale, B.L.; Stoddard, J.L.; Jeffries, D.S.; Torseth, K.; Hogasen, T.J. Regional scale evidence for improvements in surface water chemistry 1990–2001. Environ. Pollut. 2005, 137, 165–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clair, T.A.; Dennis, I.F.; Vet, R. Water chemistry and dissolved organic carbon trends in lakes from Canada’s Atlantic Provinces: No recovery from acidification measured after 25 years of lake monitoring. J. Fish. Aquat. Sci. 2011, 68, 663–674. [Google Scholar] [CrossRef]
- Strock, K.E.; Nelson, S.J.; Kahl, J.S.; Saros, J.E.; Mc Dowell, W.H. Decadal trends reveal recent acceleration in the rate of recovery from acidification in the northeastern U.S. Environ. Sci. Technol. 2014, 48, 4681–4689. [Google Scholar] [CrossRef] [PubMed]
- Rogora, M.; Colombo, L.; Marchetto, A.; Mosello, R.; Steingruber, S. Temporal and spatial patterns in the chemistry of wet deposition in Southern Alps. Atmos. Environ. 2016, 146, 44–54. [Google Scholar] [CrossRef] [Green Version]
- Garmo, O.G.; Skjelkvåle, B.L.; de Wit, H.A.; Colombo, L.; Curtis, C.; Folster, J.; Hoffmann, A. Trends in surface water chemistry in acidified areas in Europe and North America from 1990 to 2008. Water Air Soil Pollut. 2014, 225, 1880. [Google Scholar] [CrossRef] [Green Version]
- Graedel, T.E.; Benkovitz, C.M.; Keene, W.C.; Lee, D.S.; Marland, G. Global emission inventories of acid-related compounds. Water Air Soil Pollut. 1995, 85, 25–36. [Google Scholar] [CrossRef]
- Zhao, B.; Shuxiao, W.; Xinyi, D.; Jiandong, W.; Lei, D.; Xiao, F.; Jiming, H.; Joshua, F. Environmental effects of the recent emission changes in China: Implications for particulate matter pollution and soil acidification. Environ. Res. Lett. 2013, 8, 024031. [Google Scholar] [CrossRef]
- Wang, S.X.; Zhao, B.; Cai, S.Y.; Klimont, Z.; Nielsen, C.P.; Morikawa, T.; Woo, J.H.; Kim, Y.; Fu, X.; Xu, J.Y.; et al. Emission trends and mitigation options for air pollutants in East Asia. Atmos. Chem. Phys. 2014, 14, 6571–6603. [Google Scholar] [CrossRef] [Green Version]
- Meng, C.; Dashuan, T.; Hui, Z.; Zhaolei, L.; Chuixiang, Y.; Shuli, N. Global soil acidification impacts on belowground processes. Environ. Res. Lett. 2019, 14, 074003. [Google Scholar] [CrossRef]
- Duan, L.; Ma, X.X.; Larssen, T.; Mulder, J.; Hao, J.M. Response of surface water acidification in upper Yangtze River to SO2 emissions abatement in China. Environ. Sci. Technol. 2011, 45, 3275–3281. [Google Scholar] [CrossRef]
- Yu, Q.; Zhang, T.; Cheng, Z.; Zhao, B.; Mulder, J.; Larssen, T.; Wang, S.; Duan, L. Is surface water acidification a serious regional issue in China? Sci. Total Environ. 2017, 15, 584–585. [Google Scholar] [CrossRef] [PubMed]
- Hanson, P.C.; Pace, M.L.; Carpenter, S.R.; Cole, J.J.; Stanley, E.H. Integrating landscape carbon cycling: Research needs for resolving organic carbon budgets of lakes. Ecosystems 2015, 18, 363–375. [Google Scholar] [CrossRef]
- Clark, J.M.; Bottrell, S.H.; Evans, C.D.; Monteith, D.T.; Bartlett, R.; Rose, R.; Newton, R.J.; Chapman, P.J. The importance of the relationship between scale and process in understanding long-term DOC dynamics. Sci. Total Environ. 2010, 408, 2768. [Google Scholar] [CrossRef] [PubMed]
- De Wit, H.A.; Valinia, S.; Weyhenmeyer, G.A.; Futter, M.N.; Kortelainen, P.; Austnes, K.; Hessen, D.O.; Räike, A.; Laudon, H.; Vuorenmaa, J. Current browning of surface waters will be further promoted by wetter climate. Environ. Sci. Technol. Lett. 2016, 3, 430–435. [Google Scholar] [CrossRef] [Green Version]
- Strock, K.E.; Theodore, N.; Gawley, W.G.; Ellsworth, A.C.; Saros, J.E. Increasing dissolved organic carbon concentrations in northern boreal lakes: Implications for lake water transparency and thermal structure. J. Geophys. Res. Biogeosci. 2017, 122, 1022–1035. [Google Scholar] [CrossRef]
- Chapman, P.M. Integrating toxicology and ecology: Putting the “eco” into ecotoxicology. Mar. Pollut. Bull. 2002, 44, 7–15. [Google Scholar] [CrossRef]
- Newman, M.C.; Clements, W.H. Aquatic Toxicology: Concepts, Practice, New Directions. In General, Applied and Systems Toxicology; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2009; Available online: https://onlinelibrary.wiley.com/doi/book/10.1002/9780470744307 (accessed on 27 September 2022).
- Boudou, A. Aquatic Ecotoxicology. Volume 1: Fundamental Concepts and Methodologies; Taylor & Francis eBooks: College Park, MD, USA, 2018; p. 328. Available online: https://www.routledge.com/Aquatic-Ecotoxicology-Volume-1-Fundamental-Concepts-and-Methodologies/Boudou/p/book/9781315890753 (accessed on 27 September 2022).
- Vasseur, P.; Masfaraud, J.-F.; Blaise, C. Ecotoxicology, revisiting its pioneers. Environ. Sci. Pollut. Res. Int. 2021, 28, 3852–3857. [Google Scholar] [CrossRef]
- Fitzgerald, L.; Wikoff, D.S. Persistent organic pollutants. In Encyclopedia of Toxicology, 3rd ed.; Wexler, P., Ed.; Elsevier Inc.: Amsterdam, The Netherlands; Academic Press: Cambridge, MA, USA, 2014; Volume 3, pp. 820–825. Available online: https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/persistent-organic-pollutant (accessed on 27 September 2022).
- Davidson, T.; Costa, M. Selected Molecular Mechanisms of Metal Toxicity and Carcinogenicity. In Handbook on the Toxicology of Metals; Nordberg, G.F., Fowler, B.A., Nordberg, M., Eds.; Academic Press: Cambridge, MA, USA, 2014; pp. 79–100. [Google Scholar]
- Bjerregaard, P.; Andersen, O. Ecotoxicology of Metals—Sources, Transport, and Effects in the Ecosystem. In Handbook on the Toxicology of Metals; Nordberg, G.F., Fowler, B.A., Nordberg, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Bryan, G.W. Heavy Metal Contamination in the Sea, Marine Pollution; Johnston, R., Ed.; Academic Press: Cambridge, MA, USA; Elesvier: London, UK, 1976; pp. 185–302. [Google Scholar]
- Moore, J.W.; Ramamurthy, S. Heavy Metals in Natural Waters: Applied Monitoring and Impact Assessment; Springer: Berlin/Heidelberg, Germany, 1984; p. 288. [Google Scholar]
- Venitsianov, E.V.; Lepikhin, A.P. Physico–Chemical Bases of Modeling Migration and Transformation of Heavy Metals in Natural Waters; Publishing House of RosNIIVH: Yekaterinburg, Russia, 2002; p. 236. [Google Scholar]
- Heath, A.G. Water Pollution and Fish Physiology; Lewis Publishers: Amsterdam, The Netherlands, 2002; p. 506. [Google Scholar]
- Moiseenko, T.I.; Gashkina, N.A.; Dinu, M.I.; Kremleva, T.A.; Khoroshavin, V.Y. Water Chemistry of Arctic Lakes under Airborne Contamination of Watersheds. Water 2020, 12, 1659. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Gashkina, N.A. Distribution and bioaccumulation of heavy metals (Hg, Cd, and Pb) In fish: Influence of the aquatic environment and climate. Environ. Res. Lett. 2020, 15, 115013. [Google Scholar] [CrossRef]
- Magalhaes, D.; Marques, M.; Baptista, D.; Forsin, D.; Buss, D. Metal bioavailability and toxicity in freshwaters. Environ. Chem. Lett. 2015, 13, 69–87. [Google Scholar] [CrossRef]
- Merrington, G.; Peters, A.; Schlekat, C.E. Accounting for metal bioavailability in assessing water quality: A step change? Environ. Toxicol. Chem. 2016, 35, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Väänänena, K.; Leppänen, M.T.; Chen, X.; Akkanenaa, J. Metal bioavailability in ecological risk assessment of freshwater ecosystems: From science to environmental management. Ecotoxicol. Environ. Saf. 2018, 147, 430–446. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Directive 2013/39/EC Amending Directives 2000/60/EC and 2008/105/EC as Regards Priority Substances in the Field of Water Policy. 2013/39/EC; European Commission: Brussels, Belgium, 2013; Available online: https://www.eea.europa.eu/policy-documents/2013-39-ec (accessed on 27 September 2022).
- USA EPA. National Recommended Water Quality Criteria. Report 4304T. Office of Water, Office of Science and Technology. (EPA/600/4-91/002); USA EPA: Springfield, IL, USA, 2007.
- Stockholm Convention on Persistent Organic Pollutants. UN Convention; 2004. Available online: https://www.state.gov/key-topics-office-of-environmental-quality-and-transboundary-issues/stockholm-convention-on-persistent-organic-pollutants (accessed on 2 April 2022).
- Lewis, K.A.; Tzilivakis, J.; Warner, D.; Green, A. An international database for pesticide risk assessments and management. Hum. Ecol. Risk Assess. Int. J. 2016, 22, 1050–1064. [Google Scholar] [CrossRef] [Green Version]
- Barrick, A.; Champeau, O.; Butler, J.; Wiles, T.; Boundy, M.; Tremblaya, L.A. Hazard/Risk Assessment Investigating the Ecotoxicity of Select Emerging Organic Contaminants Toward the Marine Copepod Gladioferens pectinatus. Environ. Toxicol. Chem. 2022, 41, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Khudoley, V.V. Stockholm Convention, and National Plan of Action Against Persistent Organic Pollutants (POPs); The International Ecological Forum: St.Petersburg, Russia, 2002; pp. 89–91. [Google Scholar]
- Zhulidov, A.V.; Robarts, R.D.; Headley, J.V.; Korotova, L.G.; Pavlov, D.F.; Zhulidov, D.A.; Zhulidova, O.V. A review riverine fluxes of hexachlorcyclohexane and DDT to Azov and Blake seas from the formed USSR and Russian Federation. J. Environ. Sci. Health 2003, 5, 38. [Google Scholar]
- Blus, L.I. Organochlorine Pesticedes. In Handbook of Ecotoxicology; Hoffman, D.J., Rattner, B.A., Burton, G.A., Cairnce, J., Jr., Eds.; Lewis Publishers: New York, NY, USA, 2005; pp. 314–329. [Google Scholar]
- Rice, C.P.; O’Keefe, P.; Kubiak, T.J. Sources, Partweys and effects of PSBs, Dioxins and Dibenzofurans. In Handbook of Ecotoxicology; Hoffman, D.J., Rattner, B.A., Burton, G.A., Cairnce, J., Jr., Eds.; Lewis Publishers: New York, NY, USA, 2005; pp. 501–556. [Google Scholar]
- Ding, J.; Zhang, S.; Razanajatovo, R.M. Accumulation, tissue distribution, and biochemical effects of polystyrene microplastics in the freshwater fish red tilapia (Oreochromis niloticus). Environ. Pollut. 2018, 238, 1–9. [Google Scholar] [CrossRef]
- Wang, W.; Gao, H.; Jin, S.; Li, R.; Na, G. The ecotoxicological effects of microplastics on aquatic food web, from primary producer to human: A review. Ecotoxicol. Environ. Saf. 2019, 173, 110–117. [Google Scholar] [CrossRef]
- Gianni, P. 2020 Nuclear Safety, 2nd ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2020. [Google Scholar]

| Acid Equivalent | 1990 | 2000 | 2025 | 2050 | 2100 |
|---|---|---|---|---|---|
| Natural | |||||
| SOx, million t-eq | 1, 0 | 1, 0 | 1, 0 | 1, 0 | 1, 0 |
| NOx, million t-eq | 0, 4 | 0, 4 | 0, 4 | 0, 4 | 0, 4 |
| Anthropogenic | |||||
| SOx, million t-eq | 1, 8 | 1, 9 | 2, 9 | 3, 8 | 3, 6 |
| NOx, million t-eq | 1, 4 | 1, 5 | 2, 3 | 3, 0 | 4, 1 |
| TOTAL | 4, 6 | 4, 8 | 6, 6 | 8, 2 | 9, 1 |
| Metals | River Runoff by Chemical Weathering | Inflow to Lakes (According to Our Calculations) | Dispersion Into the Environment |
|---|---|---|---|
| Cr | 60.0 (3) | 63 | 54–130 (3) |
| Mn | 440 (1) | 2903 | - |
| Fe | 25,000 (1) | 31,925 | - |
| Ni | 300 (1) | 161 | 47.4 (2); 43–98 (3) |
| Cu | 375 (1) | 229 | 56 (2); 56–263 (3) |
| Zn | 370 (1) | 693 | 314 (2); 315–840 (3) |
| Mo | 13 (1) | 92 | - |
| Ag | 5 (1) | 3.9 | - |
| Cd | 4.65 (3) | 43 | 7–11 (3) |
| Sn | 1.5 (1) | 26 | - |
| Sb | 1.3 (1) | 22.2 | - |
| Pb | 180 (1) | 119 | 449 (2); 360–440 (3) |
| Hg | 3 (4) | - | 5–10 (3) |
| Pollutant | Half-Life in Temperate Climates | |||
|---|---|---|---|---|
| Air | Water | Bottom Sediments | Soil | |
| DDT | 2 days | >1 year | ||
| TCDD | 9 days | >5 years | >1 year | 10 years |
| Aldrin | <9 h | <590 days | no data | 5 years |
| Dieldrin | <40.5 h | >2 years | no data | >2 years |
| Endrin | 1.45 h | >112 days | no data | <12 years |
| Chlordane | <52 h | >4 years | no data | 1 year |
| Heptachlor | no data | <1 day | no data | 120–240 days |
| Hexachlorobenzene | <4.3 years | >100 years | no data | >3 years |
| Mirex | no data | >10 h | >600 years | >600 years |
| Toxaphene | <5 days | 20 years | no data | 10 years |
| PCB | 3–21 days | >5 days | no data | >40 days |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moiseenko, T.I. Surface Water under Growing Anthropogenic Loads: From Global Perspectives to Regional Implications. Water 2022, 14, 3730. https://doi.org/10.3390/w14223730
Moiseenko TI. Surface Water under Growing Anthropogenic Loads: From Global Perspectives to Regional Implications. Water. 2022; 14(22):3730. https://doi.org/10.3390/w14223730
Chicago/Turabian StyleMoiseenko, Tatyana I. 2022. "Surface Water under Growing Anthropogenic Loads: From Global Perspectives to Regional Implications" Water 14, no. 22: 3730. https://doi.org/10.3390/w14223730
APA StyleMoiseenko, T. I. (2022). Surface Water under Growing Anthropogenic Loads: From Global Perspectives to Regional Implications. Water, 14(22), 3730. https://doi.org/10.3390/w14223730






