Signatures of Urbanization in Temperate Highland Peat Swamps on Sandstone (THPSS) of the Blue Mountains World Heritage Area
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Monitoring of Water Chemistry
2.3. Sediment Sample Collection and Testing
2.3.1. Sample Collection
2.3.2. Mineralogy
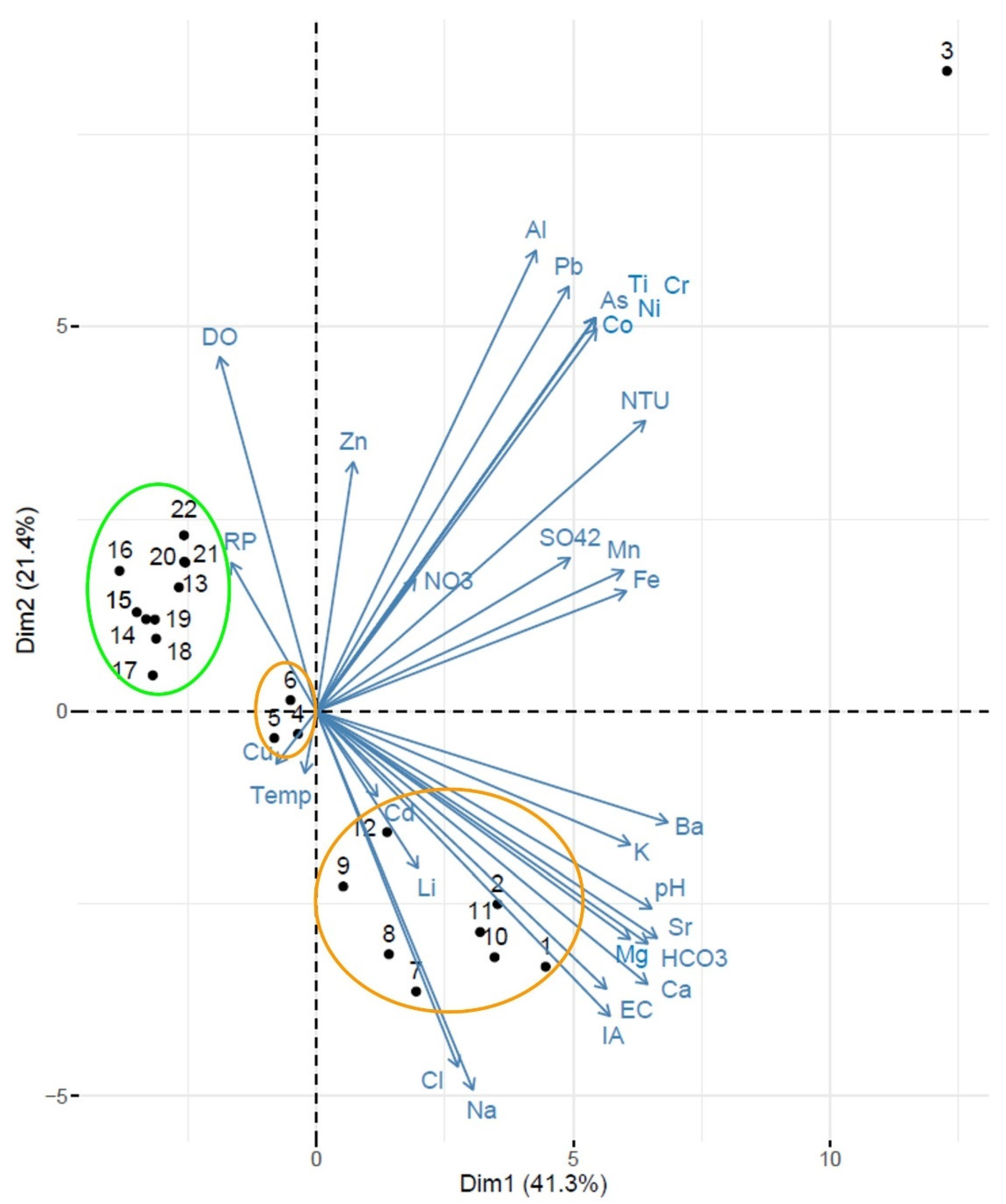
2.4. Data Analysis
2.4.1. Statistical Analysis
2.4.2. Water Quality Modelling
3. Results
3.1. Water Chemistry
3.1.1. Physiochemical Properties
3.1.2. Water Ionic Composition of Major Cations and Anions
3.1.3. Elemental Loads
3.2. Sediment Chemistry
3.2.1. Physical and Chemical Characterization of Sediment
3.2.2. Mineralogy
4. Discussion
4.1. Calcium and Urban Development
4.2. Altered Chemical Signature of Urban Ecosystems
4.3. Relationships between Catchment Urbanization and Wetland Chemistry
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaushal, S.S.; McDowell, W.H.; Wollheim, W.M. Tracking evolution of urban biogeochemical cycles: Past, present, and future. Biogeochemistry 2014, 121, 1–21. [Google Scholar] [CrossRef]
- Paul, M.J.; Meyer, J.L. Streams in the urban landscape. Ann. Rev. Ecol. Syst. 2001, 32, 333–365. [Google Scholar] [CrossRef]
- Walsh, C.J.; Roy, A.H.; Feminella, J.W.; Cottingham, P.D.; Groffman, P.M.; Morgan, R.P. The urban stream syndrome: Current knowledge and search for a cure. J. N. Am. Benthol. Soc. 2005, 24, 706–723. [Google Scholar] [CrossRef]
- Wright, I.A.; Davies, P.J.; Findlay, S.J.; Jonasson, O.J. A new type of water pollution: Concrete drainage infrastructure and geochemical contamination of urban waters. Mar. Freshw. Res. 2011, 62, 1355–1361. [Google Scholar] [CrossRef]
- Kaushal, S.S.; Likens, G.E.; Utz, R.M.; Pace, M.L.; Grese, M.; Yepsen, M. Increased river alkalinization in the eastern U.S. Environ. Sci. Technol. 2013, 47, 10302–10311. [Google Scholar] [CrossRef]
- Chambers, L.G.; Chin, Y.P.; Filippelli, G.M.; Gardner, C.B.; Herndon, E.M.; Long, D.T.; Lyons, W.B.; Macpherson, G.L.; McElmurry, S.P.; McLean, C.E.; et al. Developing the scientific framework for urban geochemistry. Appl. Geochem. 2016, 67, 1–20. [Google Scholar] [CrossRef]
- Moore, J.; Bird, D.L.; Dobbis, S.K.; Woodward, G. Nonpoint source contributions drive elevated major ion and dissolved inorganic carbon concentrations in urban watersheds. Environ. Sci. Technol. Let. 2017, 4, 198–204. [Google Scholar] [CrossRef]
- Kaushal, S.S.; Wood, K.L.; Galella, J.G.; Gion, A.M.; Haq, S.; Goodling, P.J.; Haviland, K.A.; Reimer, J.E.; Morel, C.J.; Wessel, B.; et al. Making ‘chemical cocktails’—Evolution of urban geochemical processes across the periodic table of elements. Appl. Geochem. 2020, 119, 104632. [Google Scholar] [CrossRef]
- Kaushal, S.S.; Likens, G.E.; Pace, M.L.; Reimer, J.E.; Maas, C.M.; Galella, J.G.; Utz, R.M.; Duan, S.; Kryger, J.R.; Yaculak, A.M.; et al. Freshwater salinization syndrome: From emerging global problem to managing risks. Biogeochemistry 2021, 154, 255–292. [Google Scholar] [CrossRef]
- MacAvoy, S.E.; Lunine, A. Anthropogenic influences on an urban river: Differences in cations and nutrients along an urban/suburban transect. Water 2022, 14, 1330. [Google Scholar] [CrossRef]
- Thornton, I. Metal contamination of soils in urban areas. In Soils in the Urban Environment; Bullock, P., Gregory, P.J., Eds.; Blackwell Scientific Publications: Oxford, UK, 1991; pp. 47–75. [Google Scholar]
- King, S.A.; Buckney, R.T. Invasion of exotic plants in nutrient-enriched urban bushland. Austral Ecol. 2002, 27, 573–583. [Google Scholar] [CrossRef]
- Bain, D.J.; Yesilonis, I.D.; Pouyat, R.V. Metal concentrations in urban riparian sediments along an urbanization gradient. Biogeochemistry 2012, 107, 67–79. [Google Scholar] [CrossRef]
- Grella, C.; Renshaw, A.; Wright, I.A. Invasive weeds in urban riparian zones: The influence of catchment imperviousness and soil chemistry across an urbanization gradient. Urban Ecosyst. 2018, 21, 505–517. [Google Scholar] [CrossRef]
- Poleto, C.; Charlesworth, S.; Laurenti, A. Urban aquatic sediments. In Sedimentology of Aqueous Systems; Poleto, C., Charlesworth, S., Eds.; Blackwell Publishing: Oxford, UK, 2010; pp. 129–146. [Google Scholar]
- Davies, J.; Charlesworth, S.M. Urbanization and stormwater. In Water Resources in the Built Environment: Management Issues and Solutions; Booth, C.A., Charlesworth, S.M., Eds.; Wiley-Blackwell: Oxford, UK, 2014; pp. 211–222. [Google Scholar]
- Macdonald, B.C.T.; Reynolds, J.K.; Kinsela, A.S.; Reilly, R.J.; van Oploo, P.; Waite, T.D.; White, I. Critical coagulation in sulfidic sediments from an east-coast Australian acid sulfate landscape. Appl. Clay Sci. 2009, 46, 166–175. [Google Scholar] [CrossRef]
- Åström, M.E.; Nystrand, M.; Gustafsson, J.P.; Österholm, P.; Nordmyr, L.; Reynolds, J.K.; Peltola, P. Lanthanoid behaviour in an acidic landscape. Geochim. Cosmochim. Acta 2010, 74, 829–845. [Google Scholar] [CrossRef]
- Åström, M.E.; Yu, C.; Peltola, P.; Reynolds, J.K.; Österholm, P.; Nystrand, M.I.; Augustsson, A.; Virtasalo, J.J.; Nordmyr, L.; Ojala, A.E. Sources, transport and sinks of beryllium in a coastal landscape affected by acidic soils. Geochim. Cosmochim. Acta 2018, 232, 288–302. [Google Scholar] [CrossRef]
- Fiałkiewicz-Kozieł, B.; De Vleeschouwer, F.; Mattielli, N.; Fagel, N.; Palowski, B.; Pazdur, A.; Smieja-Król, B. Record of Anthropocene pollution sources of lead in disturbed peatlands from Southern Poland. Atmos. Environ. 2018, 179, 61–68. [Google Scholar] [CrossRef]
- Davies, P.J.; Wright, I.A.; Jonasson, O.J.; Findlay, S.J. Impact of concrete and PVC pipes on urban water chemistry. Urban Water J. 2010, 7, 233–241. [Google Scholar] [CrossRef]
- Grella, C.; Wright, I.A.; Findlay, S.J.; Jonasson, O.J. Geochemical contamination of urban water by concrete stormwater infrastructure: Applying an epoxy resin coating as a control treatment. Urban Water J. 2016, 13, 212–219. [Google Scholar] [CrossRef]
- Borris, M.; Österlund, H.; Marsalek, J.; Viklander, M. An exploratory study of the effects of stormwater pipeline materials on transported stormwater quality. Water Sci. Technol. 2017, 76, 247–255. [Google Scholar] [CrossRef]
- Kaushal, S.S.; Duan, S.; Doody, T.R.; Haq, S.; Smith, R.M.; Newcomer Johnson, T.A.; Newcomb, K.D.; Gorman, J.; Bowman, N.; Mayer, P.M.; et al. Human-accelerated weathering increases salinization, major ions, and alkalinization in fresh water across land use. Appl. Geochem. 2017, 83, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Wright, I.A.; Khoury, R.; Ryan, M.M.; Belmer, N.; Reynolds, J.K. Laboratory study of impacts of concrete fragment sizes on wetland water chemistry. Urban Water J. 2018, 15, 61–67. [Google Scholar] [CrossRef]
- Cannon, W.F.; Horton, J.D. Soil geochemical signature of urbanization and industrialization—Chicago, Illinois, USA. Appl. Geochem. 2009, 24, 1590–1601. [Google Scholar] [CrossRef]
- New South Wales National Parks and Wildlife Service. The Greater Blue Mountains Area World Heritage Nomination. Available online: https://www.environment.gov.au/system/files/pages/50d276f9-337f-4d9f-85f5-120ded99fc85/files/gbm-nomination.pdf (accessed on 12 November 2020).
- United Nations Educational, Scientific and Cultural Organization. Greater Blue Mountains Area. Available online: https://whc.unesco.org/en/list/917 (accessed on 20 August 2019).
- Department of Agriculture, Water and the Environment. State Party Report on the State of Conservation of the Greater Blue Mountains Area World Heritage Property (Australia). Available online: https://www.environment.gov.au/system/files/resources/ef7799c2-866e-4dc8-8a87-3a0066124183/files/state-party-report-state-conservation-greater-blue-mountains-area-world-heritage-property.pdf (accessed on 28 September 2021).
- International Union for Conservation of Nature. Greater Blue Mountains Area 2020 Conservation Outlook Assessment. Available online: https://worldheritageoutlook.iucn.org/explore-sites/wdpaid/220294 (accessed on 6 December 2020).
- New South Wales National Parks and Wildlife Service. Greater Blue Mountains World Heritage Area Strategic Plan. Available online: https://www.environment.nsw.gov.au/-/media/OEH/Corporate-Site/Documents/Parks-reserves-and-protected-areas/Parks-plans-of-management/greater-blue-mountains-world-heritage-area-strategic-plan-080491.pdf (accessed on 28 September 2021).
- Blue Mountains City Council. Local Government Area. Available online: http://bmcc.nsw.goc.au/council/councillors-and-elections/local-governmnet-area (accessed on 11 November 2020).
- Fryirs, K.; Freidman, B.; Williams, R.; Jacobsen, G. Peatlands in eastern Australia? Sedimentology and age structure of Temperate Highland Peat Swamps on Sandstone (THPSS) in the Southern Highlands and Blue Mountains of NSW, Australia. Holocene 2014, 24, 1527–1538. [Google Scholar] [CrossRef]
- Belmer, N.; Wright, I.A.; Tippler, C. Urban geochemical contamination of high conservation value upland swamps, Blue Mountains, Australia. Water Air Soil Pollut. 2015, 226, 332–337. [Google Scholar] [CrossRef]
- Department of Agriculture, Water and the Environment. Temperate Highland Peat Swamps on Sandstone. Available online: http://www.environment.gov.au/node/14561 (accessed on 12 November 2020).
- Belmer, N.; Tippler, C.; Wright, I.A. Aquatic ecosystem degradation of high conservation value upland swamps, Blue Mountains Australia. Water Air Soil Pollut. 2018, 229, 98. [Google Scholar] [CrossRef]
- Hensen, M.; Mahony, E. Reversing drivers of degradation in Blue Mountains and Newnes Plateau Shrub Swamp endangered ecological communities. Australas. Plant Conserv. J. Aust. Netw. Plant Conserv. 2010, 18, 5–6. [Google Scholar]
- Cowley, K.L.; Fryirs, K.A.; Hose, G.C. Identifying key sedimentary indicators of geomorphic structure and function of upland swamps in the Blue Mountains for use in condition assessment and monitoring. Catena 2016, 147, 564–577. [Google Scholar] [CrossRef]
- Cowley, K.L.; Fryirs, K.A. Forgotten peatlands of eastern Australia: An unaccounted carbon capture and storage system. Sci. Total Environ. 2020, 730, 139067. [Google Scholar] [CrossRef]
- Carroll, R.; Reynolds, J.K.; Wright, I.A. Geochemical signature of urbanization in Blue Mountains Upland Swamps. Sci. Total Environ. 2020, 699, 134393. [Google Scholar] [CrossRef]
- van der Beek, P.; Pulford, A. Cenozoic landscape development in the Blue Mountains (SE Australia): Lithological and tectonic. J. Geol. 2001, 109, 35–56. [Google Scholar] [CrossRef]
- Pickett, J. Layers of time: The Blue Mountains and their geology. In Blue Mountains and Their Geology; Alder, J.D., Ed.; Geological Survey of New South Wales: Sydney, Australia, 2011. [Google Scholar]
- Mooney, S.; Martin, L. The unique and surprising environments of Temperate Highland Peat Swamps on Sandstone (THPSS) in the Blue Mountains, NSW. Australas. Plant Conserv. J. Aust. Netw. Plant Conserv. 2016, 24, 18–22. [Google Scholar]
- Bureau of Meteorology. Monthly Climate Statistics for Australian Locations—Summary Statistics for Katoomba (Farnells Rd). Available online: http://www.bom.gov.au/climate/averages/tables/cw_063039.shtml (accessed on 20 August 2019).
- United States Environmental Protection Agency. Estimating Change in Impervious Area (IA) and Directly Connected Impervious Areas (DCIA) for New Hampshire Small MS4 Permit; Small MS4 Permit Technical Support Document, April 2011; National Service Centre for Environmental Publications: Cincinnati, OH, USA, 2011. [Google Scholar]
- Department of the Environment. Australia World Heritage Areas. Available online: https://data.gov.au/dataset/ds-dga-83191acf-287d-4acf-86dc-b3caa15bf97f/distribution/dist-dga-430602f0-0f54-4ff7-840f-120c50c3d93a/details?q= (accessed on 12 November 2020).
- American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; American Public Health Association: Washington, DC, USA, 2012. [Google Scholar]
- Bruker. DIFFRAC.SUITE User Manual: DIFFRAC.EVALUATION Package; DIFFRAC.EVA: Karlsruhe, Germany, 2011. [Google Scholar]
- Johnson, C.C.; Demetriades, A.; Locutura, J.; Tore Ottesen, R. (Eds.) Mapping the Chemical Environment of Urban Areas; John Wiley & Sons Ltd: Chichester, UK, 2011. [Google Scholar]
- Lȇ, S.; Josse, J.; Husson, F. FactoMineR: An R package for multivariate analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Parkhurst, D.L.; Appelo, C.A.J. Description of Input and Examples for PHREEQC Version 3—A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations; U.S. Geological Survey Techniques and Methods; U.S. Department of the Interior and U.S. Geological Survey: Denver, CO, USA, 2013; Book 6, Chapter A43; p. 497. [Google Scholar]
- Tippler, C.; Wright, I.A.; Davies, P.J.; Hanlon, A. The influence of concrete on the geochemical qualities of urban streams. Mar. Freshw. Res. 2014, 65, 1009–1017. [Google Scholar] [CrossRef]
- Rate, A.W. Multielement geochemistry identifies the spatial pattern of soil and sediment contamination in an urban parkland, Western Australia. Sci. Total Environ. 2018, 627, 1106–1120. [Google Scholar] [CrossRef]
- Machowksi, R.; Rzetala, M.A.; Rzetala, M.; Maksymilian, S. Anthropogenic enrichment of the chemical composition of bottom sediments of water bodies in the neighborhood of a non-ferrous metal smelter (Silesian Upland, Southern Poland). Nature 2019, 9, 14445. [Google Scholar] [CrossRef] [Green Version]
- Stets, E.G.; Sprague, L.A.; Oelsner, G.P.; Johnson, H.M.; Murphy, J.C.; Ryberg, K.; Vecchia, A.V.; Zuellig, R.E.; Falcone, J.A.; Riskin, M.L. Landscape drivers of dynamic change in water quality of U.S. rivers. Environ. Sci. Technol. 2020, 54, 4336–4343. [Google Scholar] [CrossRef]
- Blue Mountains City Council. Blue Mountains Waterways Health Report 2017; Blue Mountains City Council: Katoomba, Australia, 2007. [Google Scholar]
- Blue Mountains City Council. Blue Mountains Waterways 2019 Interim Health Report. Available online: https://www.bmcc.nsw.gov.au/documents/2019-blue-mountains-waterways-health-report (accessed on 15 December 2020).
- Blue Mountains City Council. Assessment of Potential Recreational Swimming Locations in the Blue Mountains City Council Local Government Area. Available online: https://www.bmcc.nsw.gov.au/sites/default/files/docs/Water%20quality%20assessment%20of%20potential%20recreational%20swimming%20locations%20-%20Blue%20Mountains%20City%20Council%20-%20updated%20Sep%202020.pdf (accessed on 15 December 2020).
- Office of Environment & Heritage. Wf021: Wentworth Falls Lake. Available online: https://www.environment.nsw.gov.au/heritageapp/ViewHeritageItemDetails.aspx?ID=1170639 (accessed on 7 December 2020).
- Blue Mountains City Council. Wentworth Falls Lake Reserve Plan of Management. Available online: https://www.bmcc.nsw.gov.au/sites/default/files/docs/2021-06-29_Enclosure_Item12_Part1.pdf (accessed on 20 October 2021).
- Pouyat, R.V.; McDonnell, M.J. Heavy metal accumulations in forest soils along an urban-rural gradient in southeastern New York, USA. Water Air Soil Pollut. 1991, 57–58, 797–807. [Google Scholar] [CrossRef]
- Connor, N.P.; Sarraino, S.; Frantz, D.E.; Bushaw-Newton, K.; Macavoy, S.E. Geochemical characteristics of an urban river: Influences of an anthropogenic landscape. Appl. Geochem. 2014, 47, 209–216. [Google Scholar] [CrossRef]
- Price, P.; Wright, I.A. Water quality impact from the discharge of coal mine wastes to receiving streams: Comparison of impacts from an active mine with a closed mine. Water Air Soil Pollut. 2016, 227, 155. [Google Scholar] [CrossRef]
- Graham, I.J.; Goguel, R.L.; St John, D.A. Use of strontium isotopes to determine the origin of cement in concretes: Case examples from New Zealand. Cem. Concr. Res. 2000, 30, 1105–1111. [Google Scholar] [CrossRef]
- Christian, L.N.; Banner, J.L.; Mack, L.E. Sr isotopes as tracers of anthropogenic influences on stream water in the Austin, Texas, area. Chem. Geol. 2011, 282, 84–97. [Google Scholar] [CrossRef]
- Müllauer, W.; Beddoe, R.E.; Heinz, D. Leaching behavior of major and trace elements from concrete: Effect of fly ash and GGBS. Cem. Concr. Compos. 2015, 58, 129–139. [Google Scholar] [CrossRef]
- Vollpracht, A.; Brameshuber, W. Binding and leaching of trace elements in Portland cement pastes. Cem. Concr. Res. 2016, 79, 76–92. [Google Scholar] [CrossRef]
- Hogan, D.M.; Walbridge, M.R. Urbanization and nutrient retention in freshwater wetlands. Ecol. Appl. 2007, 17, 1142–1155. [Google Scholar] [CrossRef]
- Wright, I.A.; Burgin, S. Comparison of sewage and coal-mine wastes on stream macroinvertebrates within an otherwise clean upland catchment, southeastern Australia. Water Air Soil Pollut. 2009, 204, 227. [Google Scholar] [CrossRef]
- Christiansen, N.A.; Fryirs, K.A.; Green, T.J.; Hose, G.C. The impact of urbanization on community structure, gene abundance and transcription rates in microbes of upland swamps of Eastern Australia. PLoS ONE 2019, 14, e0213275. [Google Scholar] [CrossRef] [Green Version]
- Leishman, M.R.; Hughes, M.T.; Gore, D.B. Soil phosphorus enhancement below stormwater outlets in urban bushland: Spatial and temporal changes and the relationship with invasive plants. Aust. J. Soil Res. 2004, 42, 197–202. [Google Scholar] [CrossRef]
- Iversen, L.L.; Winkel, A.; Baastrup-Sphor, L.; Hinke, A.B.; Alahuhta, J.; Baattrup-Pedersen, A.; Birk, S.; Brodersen, P.; Chambers, P.A.; Ecke, F.; et al. Catchment properties and the photosynthetic trait composition of freshwater plant communities. Science 2019, 366, 878–881. [Google Scholar] [CrossRef]
- Underwood, A.J. On Beyond BACI: Sampling Designs that Might Reliably Detect Environmental Disturbances. Ecol. Appl. 1994, 4, 3–15. [Google Scholar] [CrossRef]
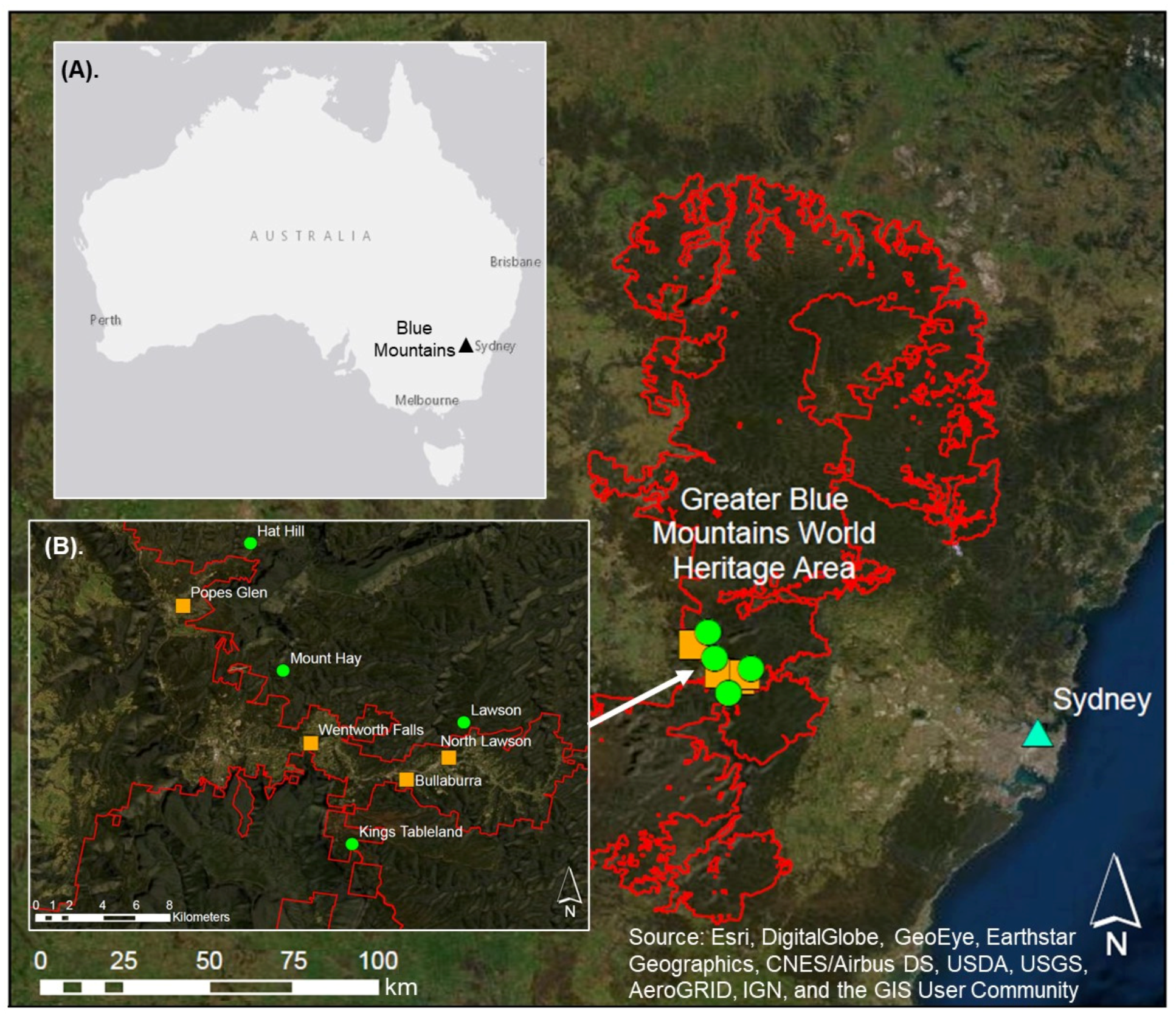
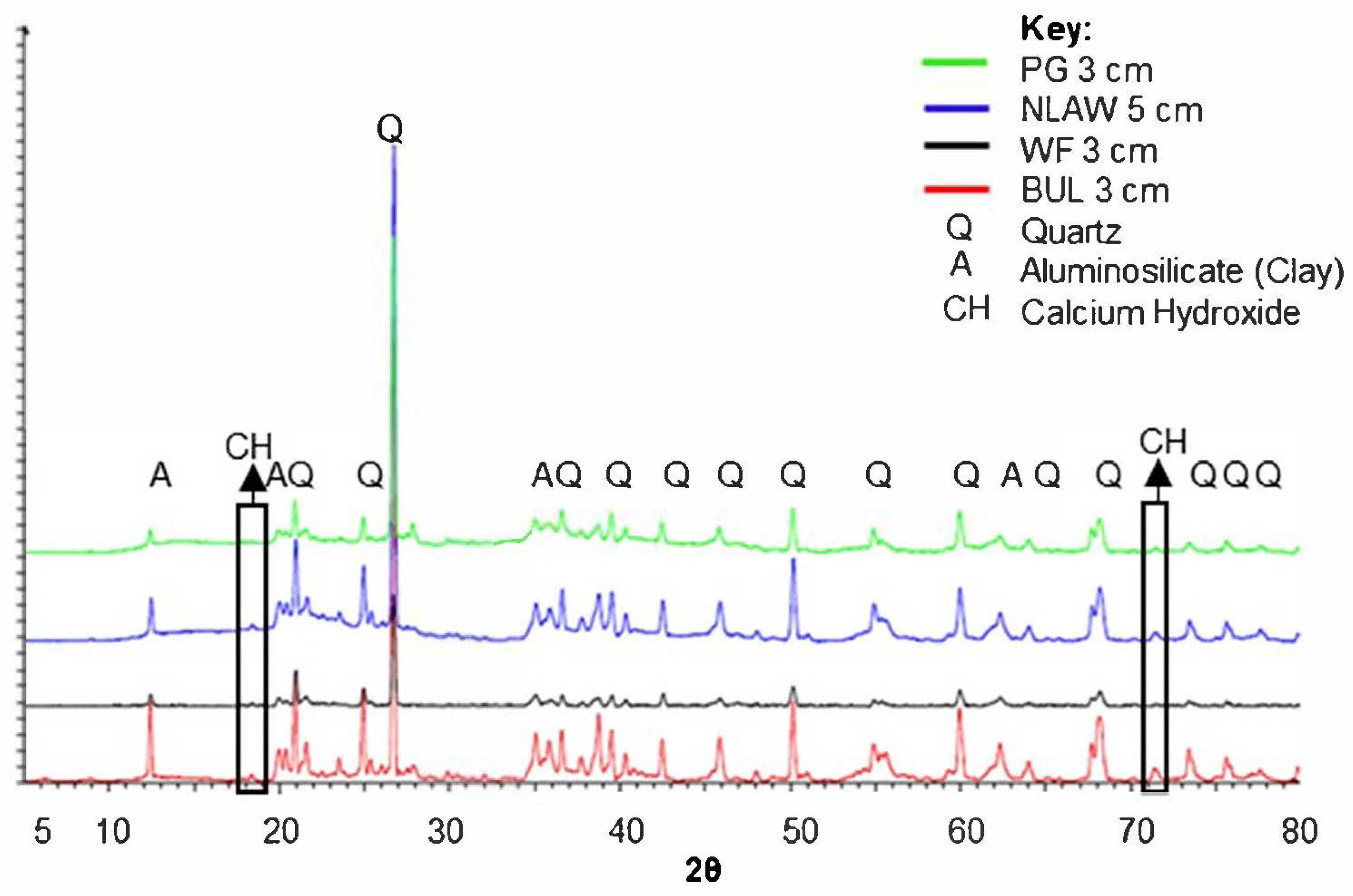
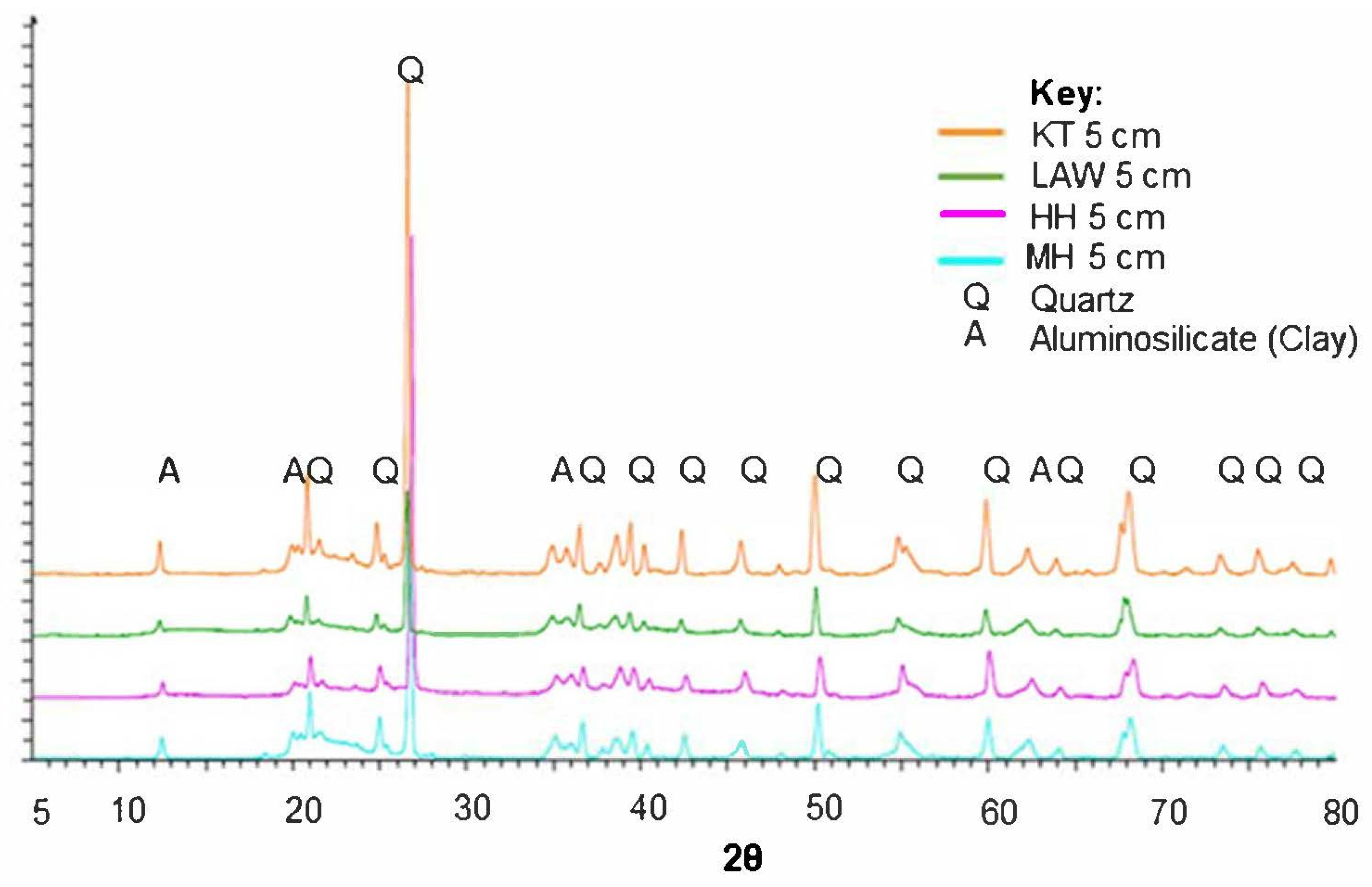
| Swamp Name | Catchment Type | Latitude and Longitude | Elevation above Sea Level at Sampling Location (m) | Swamp Area (ha) | Total Impervious Area within Catchment (IA%) | Percentage Directly Connected Impervious Area (DCIA%) |
|---|---|---|---|---|---|---|
| (1). Bullaburra | Urban | −33.727319, 150.412928 | 755 | 0.68 | 25.03 | 9.54 |
| (2). Wentworth Falls | Urban | −33.707627, 150.361313 | 880 | 9.93 | 22.41 | 7.90 |
| (3). North Lawson | Urban | −33.713851, 150.427195 | 695 | 1.15 | 23.15 | 8.35 |
| (4). Popes Glen | Urban | −33.633639, 150.292336 | 1010 | 0.82 | 34.30 | 16.30 |
| (5). Mt Hay | Naturally vegetated | −33.668644, 150.346508 | 920 | 3.92 | 0.00 | 0.00 |
| (6). Hat Hill | Naturally vegetated | −33.599941, 150.328782 | 967 | 4.10 | 0.00 | 0.00 |
| (7). Lawson | Naturally vegetated | −33.696739, 150.444027 | 665 | 9.10 | 0.00 | 0.00 |
| (8). Kings Tableland | Naturally vegetated | −33.76210, 150.38373 | 780 | 6.06 | 0.18 | 0.002 |
| Attributes | F Statistic | d.f | p Value | Urban Catchments | Naturally Vegetated Catchments | ||||
|---|---|---|---|---|---|---|---|---|---|
| Range | Mean | Median | Range | Mean | Median | ||||
| pH | 50.38 | 1, 20 | 0.000 ** | 5.56–7.09 | 6.20 | 6.08 | 4.56–5.40 | 4.87 | 4.81 |
| Electrical Conductivity (µS cm−1) | 18.80 | 1, 19 | 0.000 ** | 45.10–169.90 | 116.30 | 137.45 | 25.30–79.10 | 45.57 | 49.70 |
| Dissolved Oxygen (% saturation) | 11.98 | 1, 20 | 0.002 * | 3.20–91.40 | 51.52 | 50.05 | 75.00–102.50 | 85.02 | 83.45 |
| Temperature (°C) | 0.51 | 1, 18.01 | 0.483 (ns) | 6.60–16.90 | 10.59 | 8.95 | 6.20–24.70 | 11.85 | 9.40 |
| Turbidity (NTU) | 2.92 | 1, 20 | 0.103 (ns) | 1.57–88.50 | 13.52 | 7.04 | 0.73–19.80 | 3.00 | 2.18 |
| Nitrates (mg L−1 NO3-) | 0.02 | 1, 18.12 | 0.890 (ns) | 0.10–0.80 | 0.38 | 0.40 | 0.00–1.10 | 0.37 | 0.35 |
| Reactive Phosphates (mg L−1 PO43-) | 1.71 | 1, 18.01 | 0.207 (ns) | 0.02–1.35 | 0.27 | 0.20 | 0.04–2.39 | 0.32 | 0.20 |
| Major Ions | F Statistic | d.f. | p Value | Urban Catchments | Naturally Vegetated Catchments | ||||
|---|---|---|---|---|---|---|---|---|---|
| Range | Mean | Median | Range | Mean | Median | ||||
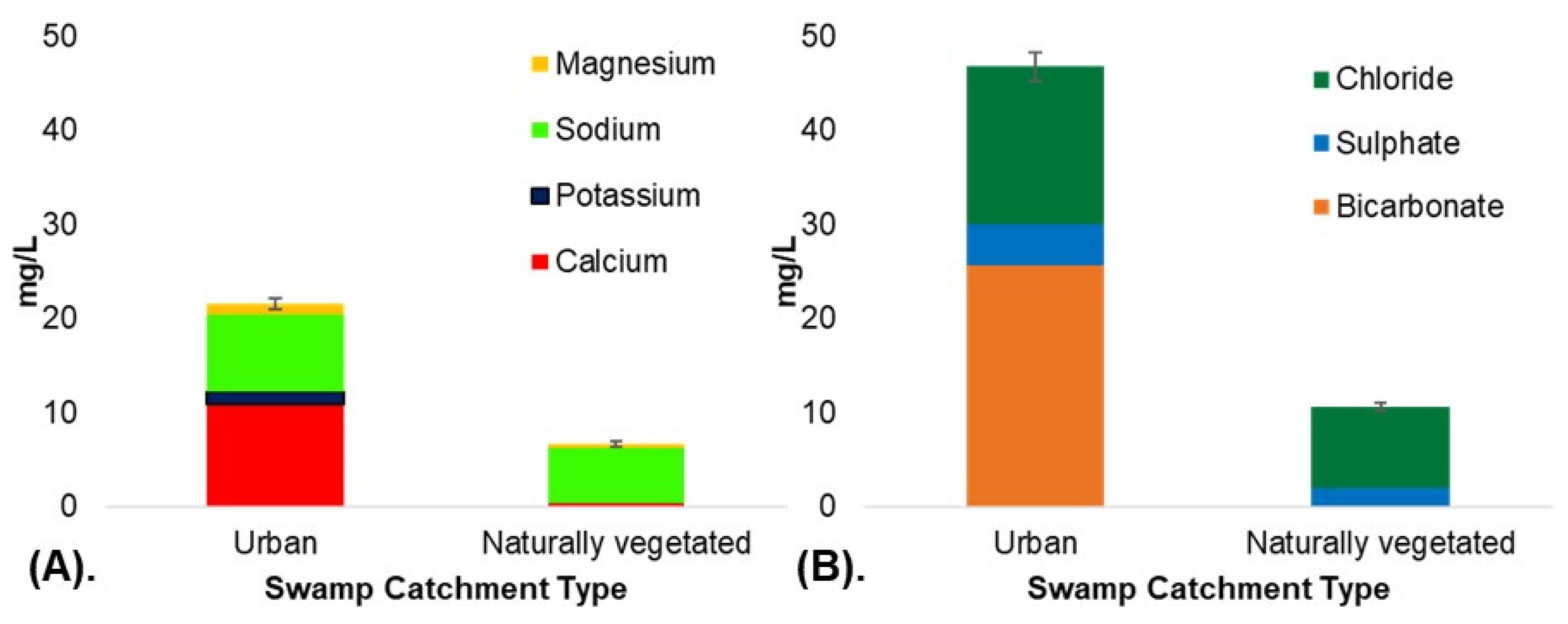
| Calcium | 61.72 | 1, 42 | 0.000 ** | 2.10–20.00 | 10.90 | 12.00 | 0.25 ˟–1.30 | 0.55 | 0.25 |
| Potassium | 24.29 | 1, 42 | 0.000 ** | 0.25 ˟–2.80 | 1.29 | 0.90 | 0.25 ˟ | 0.25 ˟ | 0.25 ˟ |
| Sodium | 16.54 | 1, 40.25 | 0.00 ** | 4.80–13.00 | 8.19 | 7.55 | 4.00–7.40 | 5.87 | 6.00 |
| Magnesium | 46.74 | 1, 40.39 | 0.00 ** | 0.60–1.90 | 1.23 | 1.20 | 0.25 ˟–0.80 | 0.54 | 0.70 |
| Bicarbonate Alkalinity as CaCO3 | 42.86 | 1, 40.35 | 0.000 ** | 5.00–56.00 | 25.67 | 22.50 | 0.00 | 2.50 ˟ | 2.50 ˟ |
| Sulfate | 6.73 | 1, 40.17 | 0.013 * | 0.5 ˟–11.00 | 4.46 | 3.00 | 0.5 ˟–6.00 | 2.40 | 1.00 |
| Chloride | 16.69 | 1, 42 | 0.000 ** | 6.00–36.00 | 16.74 | 14.00 | 6.00–11.00 | 8.62 | 9.00 |
| Calculated Ionic Strength (M) | - | - | - | 0.0005–0.0020 | 0.0015 | 0.0016 | 0.0002–0.0006 | 0.0004 | 0.0004 |
| Ratio Ca:HCO3- | - | - | - | 0.75:1–1.4:1 | 1:1 | 0.95:1 | 0.3:1–1:1 | 1:2 | 0.3:1 |
| Major ions | F Statistic | d.f. | p Value | Urban Catchments | Naturally Vegetated Catchments | ||||
|---|---|---|---|---|---|---|---|---|---|
| Range | Mean | Median | Range | Mean | Median | ||||
| Aluminium | 0.06 | 1, 40.14 | 0.81 (ns) | 5 ˟–1200.00 | 214.44 | 70.00 | 80.00–290.00 | 159.55 | 120.00 |
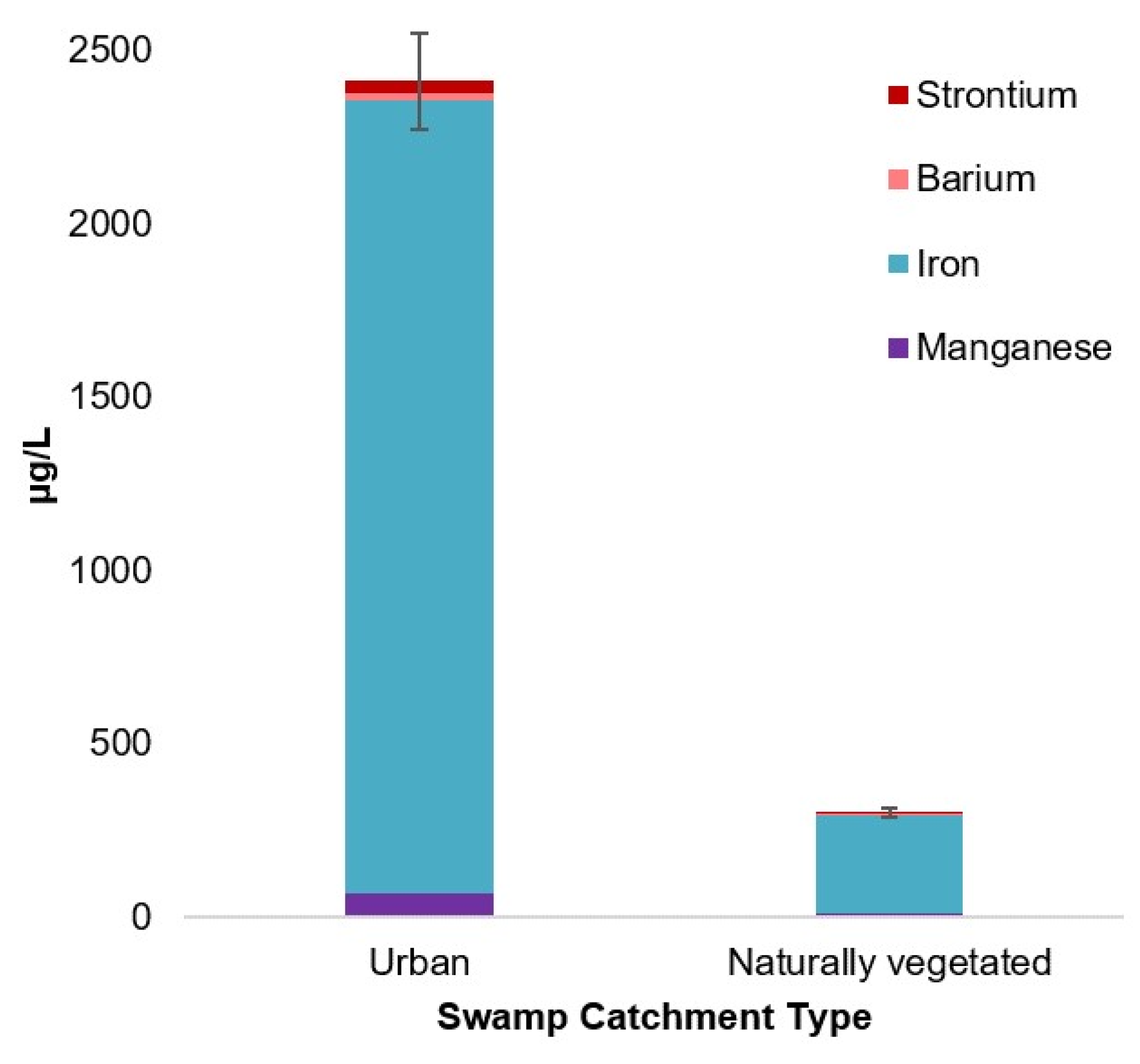
| Manganese | 7.57 | 1, 42 | 0.009 * | 2.5 ˟–260.00 | 68.93 | 17.00 | 2.5 ˟–31.00 | 9.95 | 9.00 |
| Iron | 15.40 | 1, 40.13 | 0.00 ** | 250.00–7100.00 | 2288.15 | 1300.00 | 58.00–630.00 | 281.73 | 270.00 |
| Nickel | 1.73 | 1, 42 | 0.20 (ns) | 0.5 ˟–1.00 | 0.56 | 0.50 | 0.5 ˟ | 0.5 ˟ | 0.5 ˟ |
| Zinc | 2.63 | 1, 42 | 0.11 (ns) | 0.5 ˟–25.00 | 8.70 | 6.00 | 0.5 ˟–34.00 | 13.14 | 6.50 |
| Barium | 27.73 | 1, 42 | 0.00 ** | 3.00–36.00 | 18.63 | 18.00 | 4.00–9.00 | 5.77 | 5.00 |
| Cadmium | 0.47 | 1, 40.13 | 0.50 (ns) | 0.05 ˟–48.00 | 3.64 | 0.30 | 0.05 ˟–22.00 | 1.93 | 0.35 |
| Chromium | 1.73 | 1, 40.14 | 0.20 (ns) | 0.5 ˟–2.00 | 0.67 | 0.50 | 0.5 ˟ | 0.5 ˟ | 0.5 ˟ |
| Lead | 0.74 | 1, 40.41 | 0.40 (ns) | 0.5 ˟–8.00 | 1.37 | 0.50 | 0.5 ˟–2.00 | 0.75 | 0.50 |
| Strontium | 55.84 | 1, 40.13 | 0.00 ** | 11.00–75.00 | 37.26 | 38.00 | 1.40–7.90 | 3.85 | 2.65 |
| Cobalt | 1.73 | 1, 40.010 | 0.20 (ns) | 0.5 ˟–2.00 | 0.67 | 0.50 | 0.5 ˟ | 0.5 ˟ | 0.5 ˟ |
| Copper | 0.18 | 1, 40.13 | 0.674 | 0.5 ˟–94.00 | 15.17 | 3.00 | 0.5 ˟–60.00 | 17.00 | 3.50 |
| Arsenic | 1.73 | 1, 40.25 | (ns) | 0.5 ˟–3.00 | 0.78 | 0.50 | 0.5 ˟ | 0.5 ˟ | 0.5 ˟ |
| Lithium | 2.04 | 1, 40.11 | 0.20 (ns) | 0.5 ˟–1.00 | 0.54 | 0.50 | 0.5 ˟ | 0.5 ˟ | 0.5 ˟ |
| Titanium | 2.30 | 1, 40.13 | 0.16 (ns) | 0.5 ˟–12.00 | 1.96 | 0.50 | 0.5 ˟–1.70 | 0.62 | 0.50 |
| Swamp Name | Catchment Type | Log SI | ||
|---|---|---|---|---|
| CaCO3 | Ca(OH)2 | αFeOOH | ||
| (1). Bullaburra | Urban | −1.69 | −12.94 | 6.66 |
| (2). Wentworth Falls | Urban | −4.66 | −16.70 | 5.54 |
| (3). North Lawson | Urban | −3.23 | −15.28 | 5.69 |
| (4). Popes Glen | Urban | −2.88 | −15.13 | 6.21 |
| (5). Mt Hay | Naturally vegetated | −6.46 | −17.89 | 5.06 |
| (6). Hat Hill | Naturally vegetated | −6.57 | −18.83 | 4.37 |
| (7). Lawson | Naturally vegetated | −6.38 | −19.17 | 3.79 |
| (8). Kings Tableland | Naturally vegetated | −6.20 | −19.02 | 3.91 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carroll, R.; Reynolds, J.K.; Wright, I.A. Signatures of Urbanization in Temperate Highland Peat Swamps on Sandstone (THPSS) of the Blue Mountains World Heritage Area. Water 2022, 14, 3724. https://doi.org/10.3390/w14223724
Carroll R, Reynolds JK, Wright IA. Signatures of Urbanization in Temperate Highland Peat Swamps on Sandstone (THPSS) of the Blue Mountains World Heritage Area. Water. 2022; 14(22):3724. https://doi.org/10.3390/w14223724
Chicago/Turabian StyleCarroll, Rani, Jason K. Reynolds, and Ian A. Wright. 2022. "Signatures of Urbanization in Temperate Highland Peat Swamps on Sandstone (THPSS) of the Blue Mountains World Heritage Area" Water 14, no. 22: 3724. https://doi.org/10.3390/w14223724
APA StyleCarroll, R., Reynolds, J. K., & Wright, I. A. (2022). Signatures of Urbanization in Temperate Highland Peat Swamps on Sandstone (THPSS) of the Blue Mountains World Heritage Area. Water, 14(22), 3724. https://doi.org/10.3390/w14223724








