Revisiting Soil Water Potential: Towards a Better Understanding of Soil and Plant Interactions
Abstract
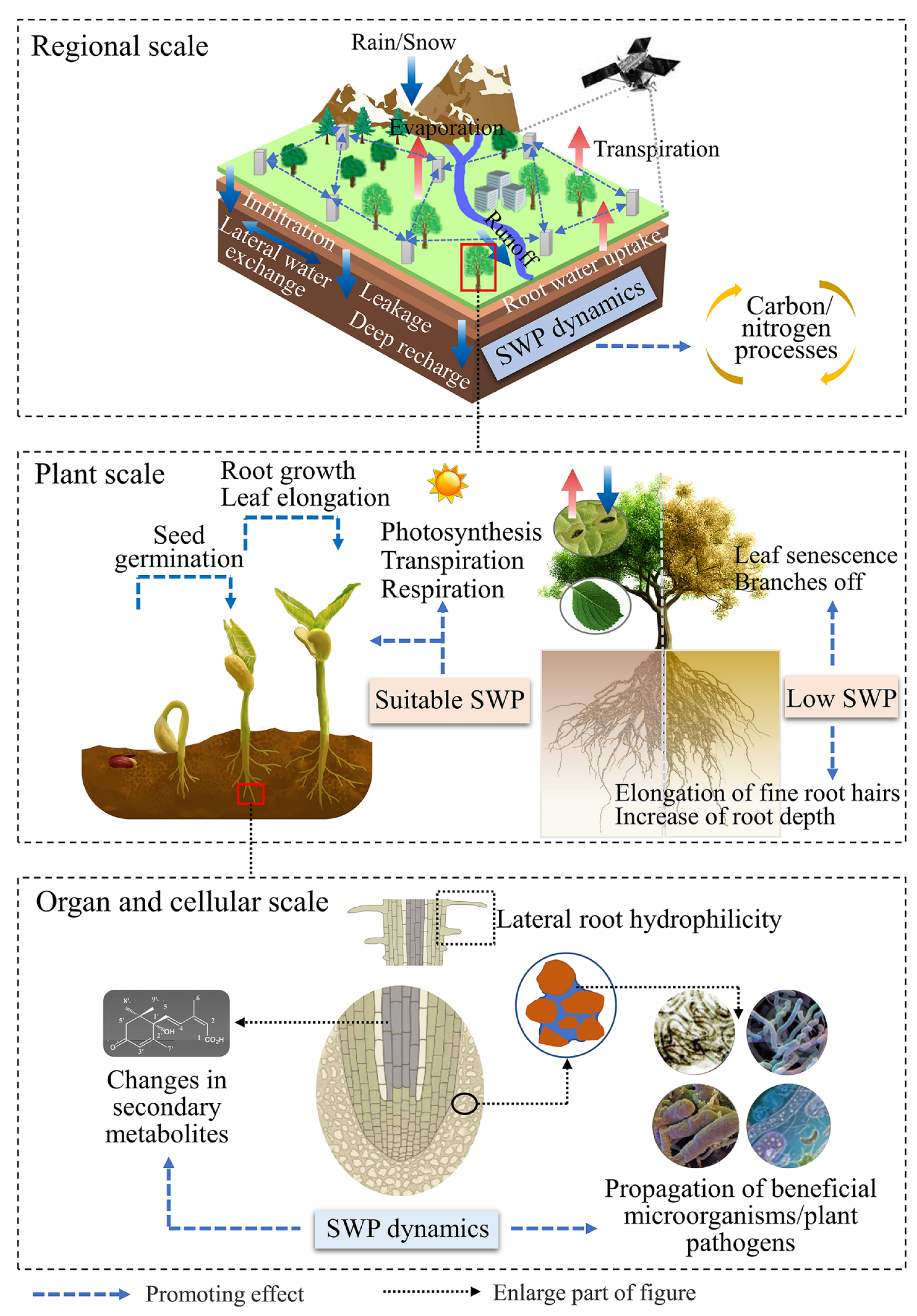
1. Introduction
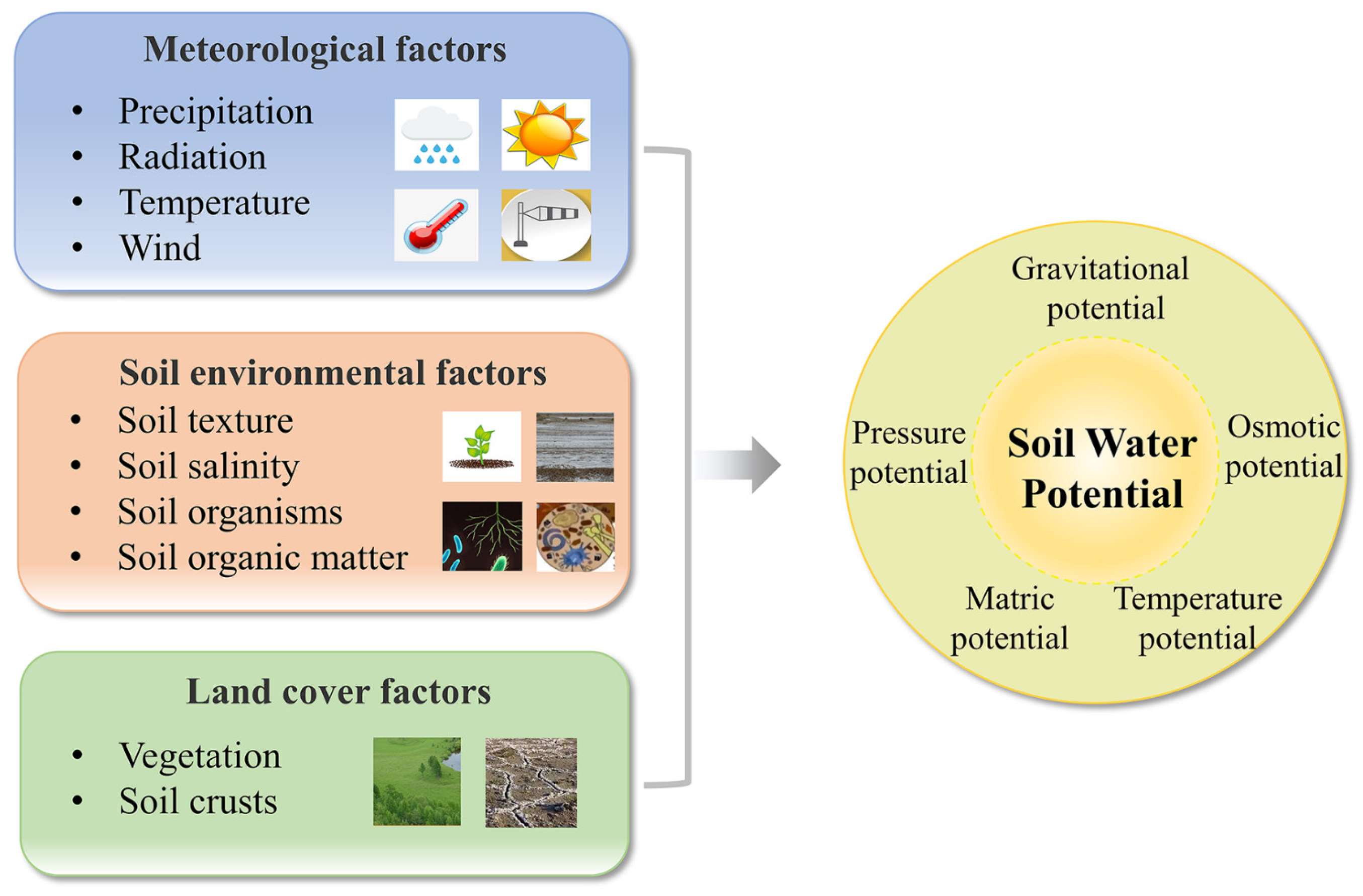
2. Influencing Factors and Evaluation Methods
2.1. Influencing Factors
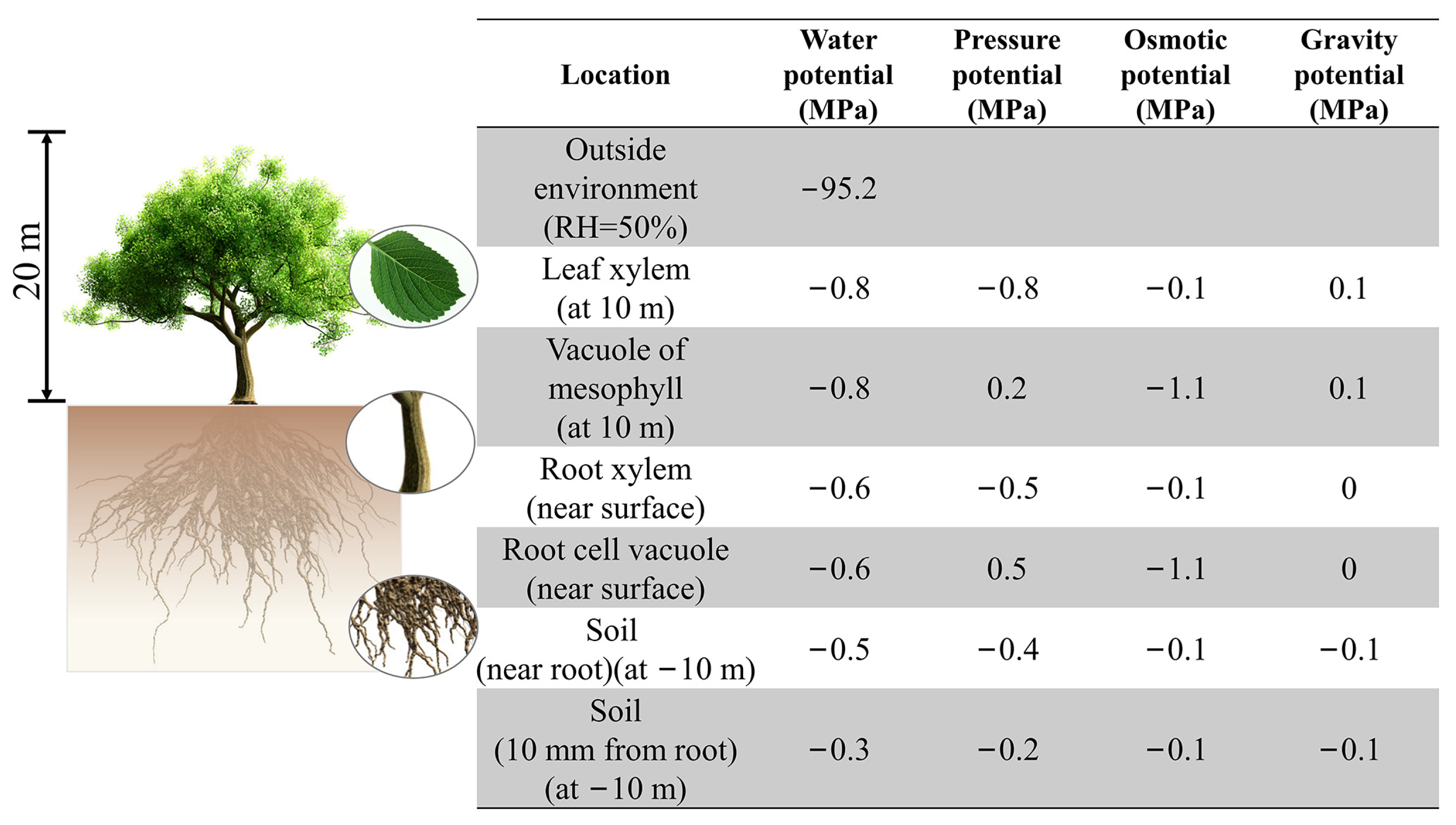
2.2. Evaluation Methods
2.2.1. Measurement Methods
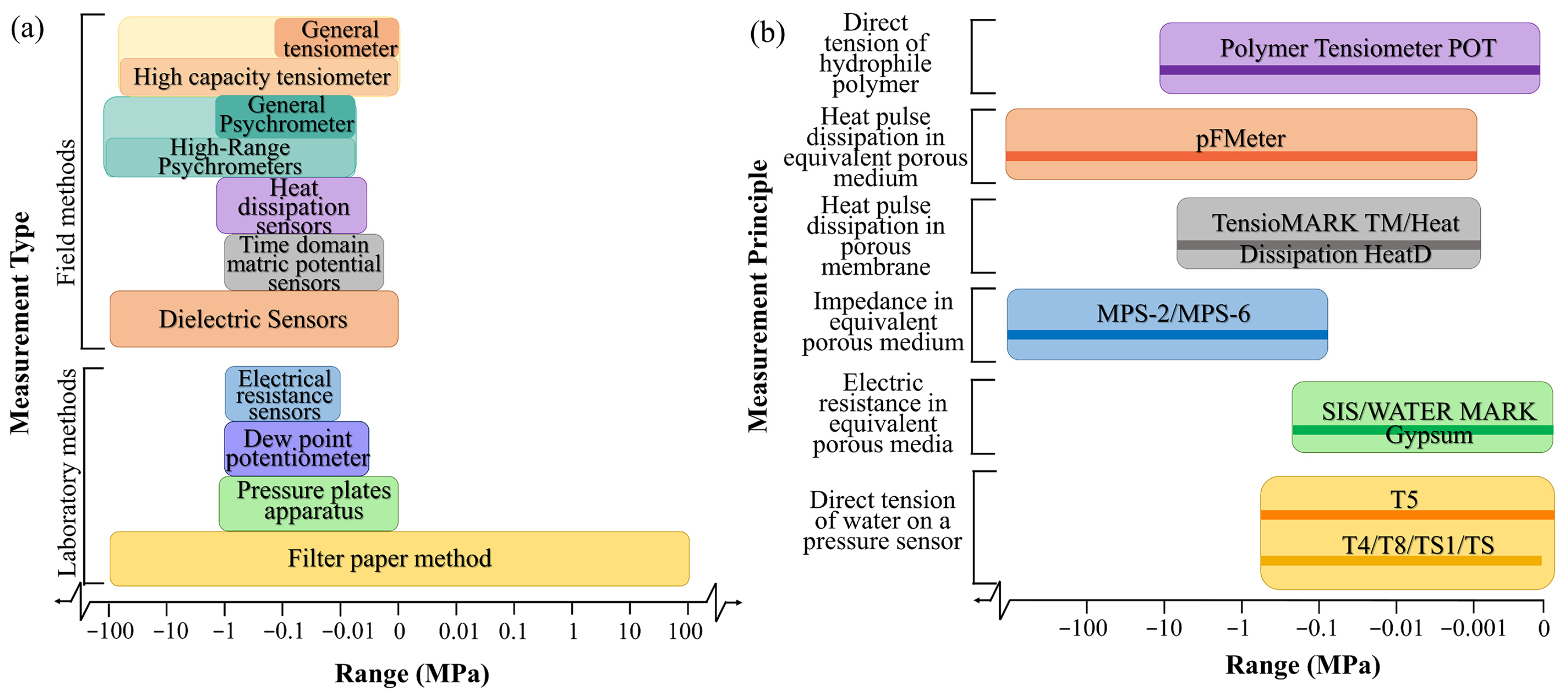
| Measurement | Device | Operational Range/MPa | In Situ Calibration | Measurement Principle | Main Features | References |
|---|---|---|---|---|---|---|
| Field methods | Tensiometer | −0.1~0 | Not required | Equilibrium of the liquid phase | Low range and long response time | [49] |
| Psychrometer | −1.5~−0.08 | Depends on the accuracy | Equilibrium of the vapor phase | Extremely sensitive to temperature | [50] | |
| Piezometer | Depends on the accuracy | Equilibrium of the liquid phase | Used in saturated material | [51,52] | ||
| Dielectric sensors | −100~0 | Depends on the accuracy | Dielectric capacity of the porous cup | Short response time; but subject to hysteresis | [43,44] | |
| Heat dissipation sensors | −1.5~−0.005 | Require separate calibration | Heat pulse dissipationin porousmembrane | Not sensitive to the salt content of the solution | [47] | |
| Frequency Domain and Time Domain Matric Potential Sensors | −1~−0.002 | Depends on the accuracy | Equilibrium of the liquid phase | Subject to hysteresis and very wet range | [53] | |
| Laboratory methods | Filter paper method | Entire range | Required | Equilibrium of the liquid phase | Long equilibration time | [45,46] |
| Pressure plate apparatus | −1.5~0 | Depends on the accuracy | Equilibrium of the liquid phase | Only used in the laboratory | [46,54] | |
| Electrical resistance sensors | −1~−0.01 | Depends on the accuracy | Electric resistancein equivalentporousmedium | Interface easily with data loggers; butsubject to hysteresis | [55] | |
| Dew point potentiometer | −1~−0.005 | Not required | Equilibrium of the vapor phase | Needs temperature control | [53] |
2.2.2. Simulation Methods
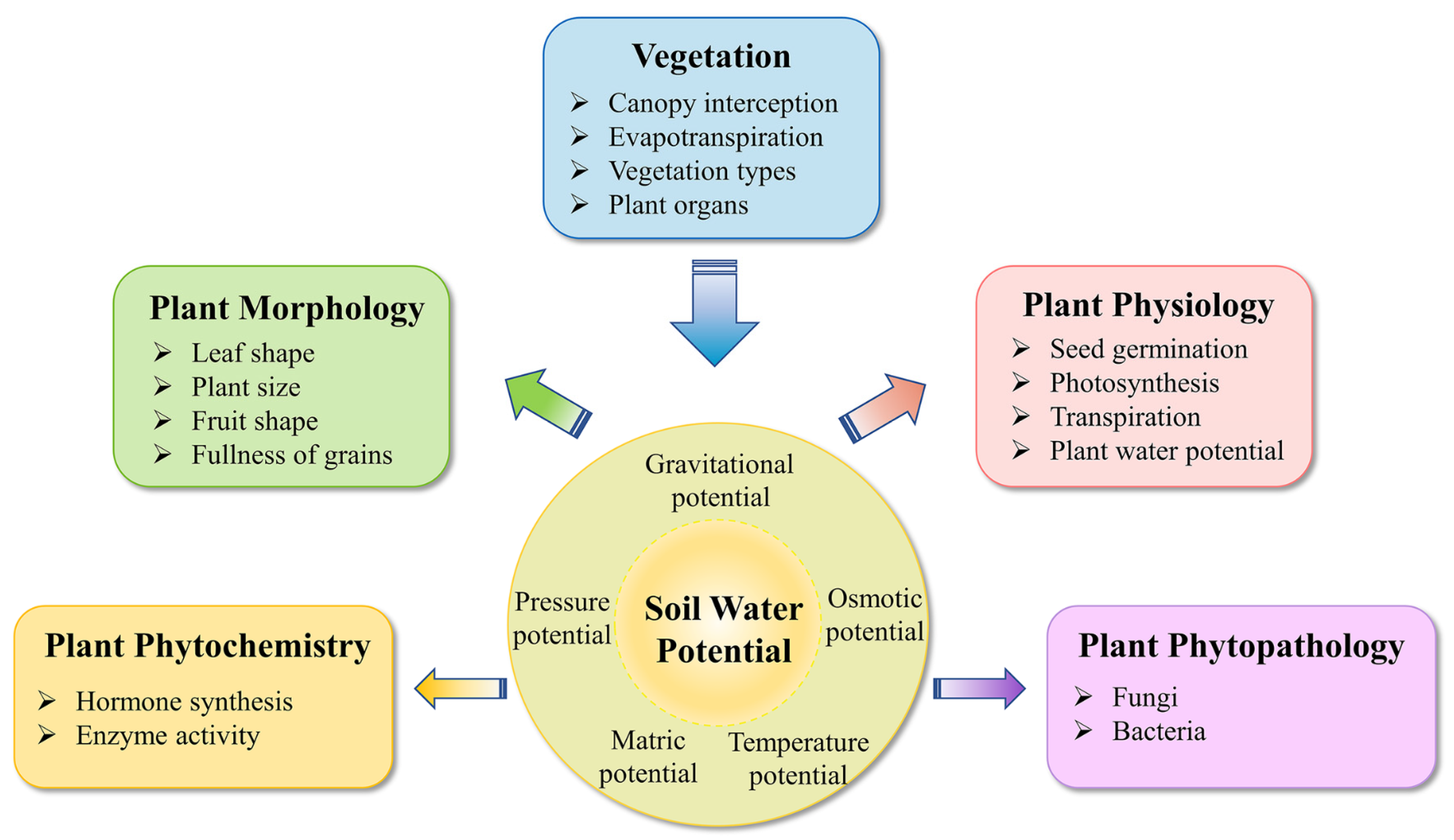
3. Plant Biological Responses to Varied SWP
3.1. Physiology
3.2. Morphology
3.3. Phytochemistry and Phytopathology
4. The Responses of Ecohydrological Processes to Varied SWP
4.1. Water Processes
4.2. Carbon Processes
4.3. Nutrient Processes
5. Challenges and Insights for Future Research
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Papendick, R.I.; Campbell, G.S. Theory and measurement of water potential. In Water Potential Relations in Soil Microbiology; Parr, J.F., Gardner, W.R., Elliott, L.F., Eds.; Soil Science Society of America: Madison, WI, USA, 1981; Volume 9, pp. 1–22. [Google Scholar]
- Passioura, J.B. Water in the soil-plant-atmosphere continuum. In Physiological Plant Ecology II: Water Relations and Carbon Assimilation; Lange, O.L., Nobel, P.S., Osmond, C.B., Ziegler, H., Eds.; Springer: Berlin/Heidelberg, Germany, 1982; Volume 12, pp. 5–33. [Google Scholar]
- Cowan, I. Transport of water in the soil-plant-atmosphere system. J. Appl. Ecol. 1965, 2, 221–239. [Google Scholar] [CrossRef]
- Hillel, D. Introduction to Soil Physics; Academic Press: San Diego, CA, USA, 1982. [Google Scholar]
- McKenzie, N.; Coughlan, K.; Cresswell, H. Soil Physical Measurement and Interpretation for Land Evaluation; CSIRO Publishing: Melbourne, Australia, 2002; Volume 5. [Google Scholar]
- Novick, K.A.; Ficklin, D.L.; Baldocchi, D.; Davis, K.J.; Ghezzehei, T.A.; Konings, A.G.; MacBean, N.; Raoult, N.; Scott, R.L.; Shi, Y.; et al. Confronting the water potential information gap. Nat. Geosci. 2022, 15, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Iwata, S.; Tabuchi, T.; Warkentin, B.P. Soil-Water Interactions: Mechanisms and Applications; CRC Press: Abingdon, UK, 2020. [Google Scholar]
- Bianchi, A.; Masseroni, D.; Thalheimer, M.; Medici, L.; Facchi, A. Field irrigation management through soil water potential measurements: A review. Ital. J. Agrometeorol. 2017, 22, 25–38. [Google Scholar] [CrossRef]
- Jackisch, C.; Germer, K.; Graeff, T.; Andrä, I.; Schulz, K.; Schiedung, M.; Haller-Jans, J.; Schneider, J.; Jaquemotte, J.; Helmer, P.; et al. Soil moisture and matric potential–an open field comparison of sensor systems. Earth Syst. Sci. Data 2020, 12, 683–697. [Google Scholar] [CrossRef]
- Philip, J. Fifty years progress in soil physics. Geoderma 1974, 12, 265–280. [Google Scholar] [CrossRef]
- Gardner, W.; Israelsen, O.; Edlefsen, N. The capillary potential function and its relation to irrigation practice. Phys. Rev. 1922, 20, 196. [Google Scholar]
- Richards, L.A. Capillary conduction of liquids through porous mediums. Physics 1931, 1, 318–333. [Google Scholar] [CrossRef]
- Klute, A. A numerical method for solving the flow equation for water in unsaturated materials. Soil Sci. 1952, 73, 105–116. [Google Scholar] [CrossRef]
- Jones, H.G. Estimation of an effective soil water potential at the root surface of transpiring plants. Plant Cell Environ. 1983, 6, 671–674. [Google Scholar] [CrossRef]
- Young, D.R.; Nobel, P.S. Predictions of soil-water potentials in the north-western Sonoran Desert. J. Ecol. 1986, 74, 143–154. [Google Scholar] [CrossRef]
- García-Tejera, O.; López-Bernal, Á.; Testi, L.; Villalobos, F.J. A soil-plant-atmosphere continuum (SPAC) model for simulating tree transpiration with a soil multi-compartment solution. Plant Soil 2016, 412, 215–233. [Google Scholar] [CrossRef]
- Brodribb, T.J.; McAdam, S.A.; Carins Murphy, M.R. Xylem and stomata, coordinated through time and space. Plant Cell Environ. 2017, 40, 872–880. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Lu, N.; Zhang, C.; Likos, W. Soil water potential: A historical perspective and recent breakthroughs. Vadose Zone J. 2022, 21, e20203. [Google Scholar] [CrossRef]
- Campbell, G.S. Soil water potential measurement: An overview. Irrig. Sci. 1988, 9, 265–273. [Google Scholar] [CrossRef]
- Clark, G.A. Measurement of soil water potential. HortScience 1990, 25, 1548–1551. [Google Scholar] [CrossRef]
- Bittelli, M. Measuring soil water potential for water management in agriculture: A review. Sustainability 2010, 2, 1226–1251. [Google Scholar] [CrossRef]
- Malazian, A.; Hartsough, P.; Kamai, T.; Campbell, G.S.; Cobos, D.R.; Hopmans, J.W. Evaluation of MPS-1 soil water potential sensor. J. Hydrol. 2011, 402, 126–134. [Google Scholar] [CrossRef]
- Cassab, G.I.; Eapen, D.; Campos, M.E. Root hydrotropism: An update. Am. J. Bot. 2013, 100, 14–24. [Google Scholar] [CrossRef]
- Feng, W.; Lindner, H.; Robbins, N.E., 2nd; Dinneny, J.R. Growing out of stress: The role of cell- and organ-scale growth control in plant water-stress responses. Plant Cell 2016, 28, 1769–1782. [Google Scholar] [CrossRef]
- Chaves, M.M.; Maroco, J.P.; Pereira, J.S. Understanding plant responses to drought—From genes to the whole plant. Funct. Plant Biol. 2003, 30, 239–264. [Google Scholar] [CrossRef]
- Prieto, I.; Armas, C.; Pugnaire, F.I. Water release through plant roots: New insights into its consequences at the plant and ecosystem level. New Phytol. 2012, 193, 830–841. [Google Scholar] [CrossRef] [PubMed]
- Nadezhdina, N.; David, T.S.; David, J.S.; Ferreira, M.I.; Dohnal, M.; Tesař, M.; Gartner, K.; Leitgeb, E.; Nadezhdin, V.; Cermak, J.; et al. Trees never rest: The multiple facets of hydraulic redistribution. Ecohydrology 2010, 3, 431–444. [Google Scholar] [CrossRef]
- Johnson, M.S.; Couto, E.G.; Pinto, O.B., Jr.; Milesi, J.; Santos Amorim, R.S.; Messias, I.A.; Biudes, M.S. Soil CO2 dynamics in a tree island soil of the Pantanal: The role of soil water potential. PLoS ONE 2013, 8, e64874. [Google Scholar] [CrossRef] [PubMed]
- Serraj, R.; Sinclair, T.R.; Purcell, L.C. Symbiotic N2 fixation response to drought. J. Exp. Bot. 1999, 50, 143–155. [Google Scholar] [CrossRef]
- Kroeckel, L.; Stolp, H. Influence of soil water potential on respiration and nitrogen fixation of Azotobacter vinelandii. Plant Soil 1984, 79, 37–49. [Google Scholar] [CrossRef]
- Nishida, K.; Shiozawa, S. Modeling and experimental determination of salt accumulation induced by root water uptake. Soil Sci. Soc. Am. J. 2010, 74, 774–786. [Google Scholar] [CrossRef]
- Lal, R.; Shukla, M.K. Principles of Soil Physics; CRC Press: New York, NY, USA, 2004. [Google Scholar]
- Saxton, K.E.; Rawls, W.J.; Romberger, J.S.; Papendick, R.I. Estimating generalized soil-water characteristics from texture. Soil Sci. Soc. Am. J. 1986, 50, 1031–1036. [Google Scholar] [CrossRef]
- Zhang, Y.; Bai, J.; Zhang, J. A study of the temperature effect on soil water potential. Acta Pedol. Sin. 1990, 27, 454–458. [Google Scholar]
- Sheldon, A.R.; Dalal, R.C.; Kirchhof, G.; Kopittke, P.M.; Menzies, N.W. The effect of salinity on plant-available water. Plant Soil 2017, 418, 477–491. [Google Scholar] [CrossRef]
- Bachmann, J.; Horton, R.; Grant, S.A.; Van der Ploeg, R. Temperature dependence of water retention curves for wettable and water-repellent soils. Soil Sci. Soc. Am. J. 2002, 66, 44–52. [Google Scholar] [CrossRef]
- Hohmann, M. Soil freezing—The concept of soil water potential. State of the art. Cold Reg. Sci. Technol. 1997, 25, 101–110. [Google Scholar] [CrossRef]
- Ernst, G.; Felten, D.; Vohland, M.; Emmerling, C. Impact of ecologically different earthworm species on soil water characteristics. Eur. J. Soil Biol. 2009, 45, 207–213. [Google Scholar] [CrossRef]
- Sun, F.; Xiao, B.; Li, S.; Kidron, G.J. Towards moss biocrust effects on surface soil water holding capacity: Soil water retention curve analysis and modeling. Geoderma 2021, 399, 115120. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E.; Møller, I.M.; Murphy, A. Plant Physiology and Development; Sinauer Associates Incorporated: Sunderland, MA, USA, 2015. [Google Scholar]
- Buckingham, E. Studies on the Movement of Soil Moisture; Govt. Print. Off.: Washington, DC, USA, 1907; Volume 38.
- Campbell, G.S.; Gardner, W.H. Psychrometric measurement of soil water potential: Temperature and bulk density effects. Soil Sci. Soc. Am. J. 1971, 35, 8–12. [Google Scholar] [CrossRef]
- Or, D.; Wraith, J.M. A new soil metric potential sensor based on time domain reflectometry. Water Resour. Res. 1999, 35, 3399–3407. [Google Scholar] [CrossRef]
- Novák, V.; Hlaváčiková, H. Soil-water potential and its measurement. In Applied Soil Hydrology; Theory and applications of transport in porous media; Springer: Cham, Switzerland, 2019; pp. 63–76. [Google Scholar]
- Gardner, R. A method of measuring the capillary tension of soil moisture over a wide moisture range. Soil Sci. 1937, 43, 277–284. [Google Scholar] [CrossRef]
- Fondjo, A.A.; Theron, E.; Ray, R.P. Assessment of various methods to measure the soil suction. Int. J. Eng. Technol. Explor. Eng. 2020, 9, 171–184. [Google Scholar] [CrossRef]
- Durner, W.; Or, D. Soil water potential measurement. In Encyclopedia of Hydrological Sciences; J. Wiley: New York, NY, USA, 2005. [Google Scholar]
- Vereecken, H.; Huisman, J.A.; Bogena, H.; Vanderborght, J.; Vrugt, J.A.; Hopmans, J.W. On the value of soil moisture measurements in vadose zone hydrology: A review. Water Resour. Res. 2008, 44, W00D06. [Google Scholar] [CrossRef]
- Klute, A.; Gardner, W. Tensiometer response time. Soil Sci. 1962, 93, 204–207. [Google Scholar] [CrossRef]
- Rawlins, S.L.; Campbell, G.S. Water potential: Thermocouple psychrometry. In Methods of Soil Analysis: Part 1 Physical and Mineralogical Methods; American Society of Agronomy: Madison, WI, USA, 1986; Volume 5, pp. 597–618. [Google Scholar]
- Reeve, R. Water potential: Piezometry. In Methods of Soil Analysis: Part 1 Physical and Mineralogical Methods; American Society of Agronomy: Madison, WI, USA, 1986; Volume 5, pp. 545–561. [Google Scholar]
- Warrick, A.W. Soil Physics Companion; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Scanlon, B.R.; Andraski, B.J.; Bilskie, J. 3.2.4 Miscellaneous methods for measuring matric or water potential. In Methods of Soil Analysis; Dane, J.H., Topp, G.C., Eds.; Soil Science Society of America: Madison, WI, USA, 2002; Volume 5, pp. 643–670. [Google Scholar]
- Richards, L.; Fireman, M. Pressure-plate apparatus for measuring moisture sorption and transmission by soils. Soil Sci. 1943, 56, 395–404. [Google Scholar] [CrossRef]
- Davis, D.; Hughes, J.E. A new approach to recording the wetting parameter by the use of electrical resistance sensors. Plant Dis. Rep. 1970, 54, 474–479. [Google Scholar]
- Toll, D.G.; Augarde, C.; Gallipoli, D.; Wheeler, S. Unsaturated Soils: Advances in Geo-Engineering; CRC Press: London, UK, 2008. [Google Scholar]
- Calbo, A.G. Dihedral Sensor for Evaluating Tension, Potential and Activity of Liquids. Patent No. 9588030; Patent and Trademark Office: Washington, DC, USA, 2017. [Google Scholar]
- Herron, P.M.; Gage, D.J.; Cardon, Z.G. Micro-scale water potential gradients visualized in soil around plant root tips using microbiosensors. Plant Cell Environ. 2010, 33, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Sabatier, J.M. Effects of soil water potential and moisture content on sound speed. Soil Sci. Soc. Am. J. 2009, 73, 1614–1625. [Google Scholar] [CrossRef]
- Fatichi, S.; Pappas, C.; Ivanov, V.Y. Modeling plant–water interactions: An ecohydrological overview from the cell to the global scale. Wiley Interdiscip. Rev. Water 2015, 3, 327–368. [Google Scholar] [CrossRef]
- Leong, E.C.; Rahardjo, H. Review of soil-water characteristic curve equations. J. Geotech. Geoenviron. 1997, 123, 1106–1117. [Google Scholar] [CrossRef]
- Philip, J.R.; De Vries, D.A. Moisture movement in porous materials under temperature gradients. Eos. Trans. Amer. Geophys. Union 1957, 38, 222–232. [Google Scholar] [CrossRef]
- Nimmo, J.R.; Miller, E.E. The temperature dependence of isothermal moisture vs. potential characteristics of soils. Soil Sci. Soc. Am. J. 1986, 50, 1105–1113. [Google Scholar] [CrossRef]
- Richards, L.A. Diagnosis and Improvement of Saline and Alkali Soils; LWW: Washington, DC, USA, 1954; Volume 78. [Google Scholar]
- Orchard, V.A.; Cook, F. Relationship between soil respiration and soil moisture. Soil Biol. Biochem. 1983, 15, 447–453. [Google Scholar] [CrossRef]
- Moyano, F.E.; Manzoni, S.; Chenu, C. Responses of soil heterotrophic respiration to moisture availability: An exploration of processes and models. Soil Biol. Biochem. 2013, 59, 72–85. [Google Scholar] [CrossRef]
- Chapman, N.; Miller, A.J.; Lindsey, K.; Whalley, W.R. Roots, water, and nutrient acquisition: Let’s get physical. Trends Plant Sci. 2012, 17, 701–710. [Google Scholar] [CrossRef]
- Bengough, A.G. Water dynamics of the root zone: Rhizosphere biophysics and its control on soil hydrology. Vadose Zone J. 2012, 11, vzj2011-0111. [Google Scholar] [CrossRef]
- Arnold, S.; Kailichova, Y.; Knauer, J.; Ruthsatz, A.D.; Baumgartl, T. Effects of soil water potential on germination of co-dominant Brigalow species: Implications for rehabilitation of water-limited ecosystems in the Brigalow Belt bioregion. Ecol. Eng. 2014, 70, 35–42. [Google Scholar] [CrossRef]
- Doneen, L.; MacGillivray, J.H. Germination (emergence) of vegetable seed as affected by different soil moisture conditions. Plant Physiol. 1943, 18, 524. [Google Scholar] [CrossRef] [PubMed]
- Gurvich, D.E.; Pérez-Sánchez, R.; Bauk, K.; Jurado, E.; Ferrero, M.C.; Funes, G.; Flores, J. Combined effect of water potential and temperature on seed germination and seedling development of cacti from a mesic Argentine ecosystem. Flora 2017, 227, 18–24. [Google Scholar] [CrossRef]
- Evans, C.E.; Etherington, J.R. The effect of soil water potential on seed germination of some British plants. New Phytol. 1990, 115, 539–548. [Google Scholar] [CrossRef]
- Gardarin, A.; Guillemin, J.P.; Munier-Jolain, N.M.; Colbach, N. Estimation of key parameters for weed population dynamics models: Base temperature and base water potential for germination. Eur. J. Agron. 2010, 32, 162–168. [Google Scholar] [CrossRef]
- Roberta, M.; Donato, L.; Stefano, B.; Maria Clara, Z.; Mario, M.; Giuseppe, Z. Temperature and water potential as parameters for modeling weed emergence in central-northern Italy. Weed Sci. 2010, 58, 216–222. [Google Scholar] [CrossRef]
- Dietrich, D. Hydrotropism: How roots search for water. J. Exp. Bot. 2018, 69, 2759–2771. [Google Scholar] [CrossRef]
- Cai, G.; Ahmed, M.A.; Abdalla, M.; Carminati, A. Root hydraulic phenotypes impacting water uptake in drying soils. Plant Cell Environ. 2022, 45, 650–663. [Google Scholar] [CrossRef]
- Takano, M.; Takahashi, H.; Hirasawa, T.; Suge, H. Hydrotropism in roots: Sensing of a gradient in water potential by the root cap. Planta 1995, 197, 410–413. [Google Scholar] [CrossRef]
- Baldocchi, D.D.; Xu, L.; Kiang, N. How plant functional-type, weather, seasonal drought, and soil physical properties alter water and energy fluxes of an oak–grass savanna and an annual grassland. Agric. For. Meteorol. 2004, 123, 13–39. [Google Scholar] [CrossRef]
- Yang, X.; Lu, M.; Wang, Y.; Wang, Y.; Liu, Z.; Chen, S. Response mechanism of plants to drought stress. Horticulturae 2021, 7, 50. [Google Scholar] [CrossRef]
- Ahmed, M.; Stockle, C.O. Quantification of Climate Variability, Adaptation and Mitigation for Agricultural Sustainability; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Rodriguez-Dominguez, C.M.; Brodribb, T.J. Declining root water transport drives stomatal closure in olive under moderate water stress. New Phytol. 2020, 225, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Carminati, A.; Ahmed, M.A.; Zarebanadkouki, M.; Cai, G.; Goran, L.; Javaux, M. Stomatal closure prevents the drop in soil water potential around roots. New Phytol. 2020, 226, 1541–1543. [Google Scholar] [CrossRef]
- Flores, J.; Briones, O. Plant life-form and germination in a Mexican inter-tropical desert: Effects of soil water potential and temperature. J. Arid Environ. 2001, 47, 485–497. [Google Scholar] [CrossRef]
- Shrestha, A.; Roman, E.S.; Thomas, A.G.; Swanton, C.J. Modeling germination and shoot-radicle elongation of Ambrosia artemisiifolia. Weed Sci. 1999, 47, 557–562. [Google Scholar] [CrossRef]
- Dorsainvil, F.; Dürr, C.; Justes, E.; Carrera, A. Characterisation and modelling of white mustard (Sinapis alba L.) emergence under several sowing conditions. Eur. J. Agron. 2005, 23, 146–158. [Google Scholar] [CrossRef]
- Fyfield, T.; Gregory, P.J. Effects of temperature and water potential on germination, radicle elongation and emergence of mungbean. J. Exp. Bot. 1989, 40, 667–674. [Google Scholar] [CrossRef]
- Finch-Savage, W.; Phelps, K. Onion (Allium cepa L.) seedling emergence patterns can be explained by the influence of soil temperature and water potential on seed germination. J. Exp. Bot. 1993, 44, 407–414. [Google Scholar] [CrossRef]
- Rucker, K.; Kvien, C.; Holbrook, C.; Hook, J. Identification of peanut genotypes with improved drought avoidance traits. Peanut Sci. 1995, 22, 14–18. [Google Scholar] [CrossRef]
- Xiao, S.; Liu, L.; Zhang, Y.; Sun, H.; Zhang, K.; Bai, Z.; Dong, H.; Li, C. Fine root and root hair morphology of cotton under drought stress revealed with RhizoPot. J. Agron. Crop Sci. 2020, 206, 679–693. [Google Scholar] [CrossRef]
- Ichsan, C.N. Morphological and physiological change of rice (Oryza sativa L.) under water stress at early season. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Banda Aceh, Indonesia, 21–22 September 2020; p. 012030. [Google Scholar]
- Rood, S.B.; Patiño, S.; Coombs, K.; Tyree, M.T. Branch sacrifice: Cavitation-associated drought adaptation of riparian cottonwoods. Trees 2000, 14, 248–257. [Google Scholar] [CrossRef]
- Jacobs, M.; Rais, A.; Pretzsch, H. How drought stress becomes visible upon detecting tree shape using terrestrial laser scanning (TLS). For. Ecol. Manag. 2021, 489, 118975. [Google Scholar] [CrossRef]
- Ashraf, M.A.; Iqbal, M.; Rasheed, R.; Hussain, I.; Riaz, M.; Arif, M.S. Environmental stress and secondary metabolites in plants. In Plant Metabolites and Regulation under Environmental Stress; Elsevier: Amsterdam, The Netherlands, 2018; pp. 153–167. [Google Scholar]
- Salem, N.; Msaada, K.; Dhifi, W.; Sriti, J.; Mejri, H.; Limam, F.; Marzouk, B. Effect of drought on safflower natural dyes and their biological activities. EXCLI J. 2014, 13, 1. [Google Scholar]
- Griffin, D.M. Soil water in the ecology of fungi. Annu. Rev. Phytopathol. 1969, 7, 289–310. [Google Scholar] [CrossRef]
- Lewis, B. Effects of water potential on the infection of potato tubers by Streptomyces scabies in soil. Ann. Appl. Biol. 1970, 66, 83–88. [Google Scholar] [CrossRef]
- Sharghi, A.; Badi, H.N.; Bolandnazar, S.; Mehrafarin, A.; Sarikhani, M.R. Morphophysiological and phytochemical responses of fenugreek to plant growth promoting rhizobacteria (PGPR) under different soil water levels. Folia Hortic. 2018, 30, 215–228. [Google Scholar] [CrossRef]
- Richards, J.H.; Caldwell, M.M. Hydraulic lift: Substantial nocturnal water transport between soil layers by Artemisia tridentata roots. Oecologia 1987, 73, 486–489. [Google Scholar] [CrossRef]
- Horton, J.L.; Hart, S.C. Hydraulic lift: A potentially important ecosystem process. Trends Ecol. Evol. 1998, 13, 232–235. [Google Scholar] [CrossRef]
- Burgess, S.S.O.; Adams, M.A.; Turner, N.C.; Ong, C.K. The redistribution of soil water by tree root systems. Oecologia 1998, 115, 306–311. [Google Scholar] [CrossRef]
- Scharwies, J.D.; Dinneny, J.R. Water transport, perception, and response in plants. J. Plant Res. 2019, 132, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Boanares, D.; Oliveira, R.S.; Isaias, R.M.S.; Franca, M.G.C.; Penuelas, J. The neglected reverse water pathway: Atmosphere-plant-soil continuum. Trends Plant Sci. 2020, 25, 1073–1075. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Feng, Y.; Boersma, L. Partition of photosynthates between shoot and root in spring wheat (Triticum aestivum L.) as a function of soil water potential and root temperature. Plant Soil 1994, 164, 43–50. [Google Scholar] [CrossRef]
- Harris, R.F. Effect of water potential on microbial growth and activity. In Water Potential Relations in Soil Microbiology; Parr, J.F., Gardner, W.R., Elliott, L.F., Eds.; Soil Science Society of America: Madison, WI, USA, 1981; Volume 9, pp. 23–95. [Google Scholar]
- Borken, W.; Matzner, E. Reappraisal of drying and wetting effects on C and N mineralization and fluxes in soils. Glob. Chang. Biol. 2009, 15, 808–824. [Google Scholar] [CrossRef]
- Manzoni, S.; Schimel, J.P.; Porporato, A. Responses of soil microbial communities to water stress: Results from a meta-analysis. Ecol. 2012, 93, 930–938. [Google Scholar] [CrossRef]
- Birch, H.F. The effect of soil drying on humus decomposition and nitrogen availability. Plant Soil 1958, 10, 9–31. [Google Scholar] [CrossRef]
- Manzoni, S.; Chakrawal, A.; Fischer, T.; Schimel, J.P.; Porporato, A.; Vico, G. Rainfall intensification increases the contribution of rewetting pulses to soil respiration. Biogeosciences 2020, 17, 4007–4023. [Google Scholar] [CrossRef]
- Herridge, D.F.; Peoples, M.B.; Boddey, R.M. Global inputs of biological nitrogen fixation in agricultural systems. Plant Soil 2008, 311, 1–18. [Google Scholar] [CrossRef]
- Dijkstra, F.A.; Augustine, D.J.; Brewer, P.; von Fischer, J.C. Nitrogen cycling and water pulses in semiarid grasslands: Are microbial and plant processes temporally asynchronous? Oecologia 2012, 170, 799–808. [Google Scholar] [CrossRef]
- Plante, A.F. Soil biogeochemical cycling of inorganic nutrients and metals. In Soil Microbiology, Ecology and Biochemistry; Elsevier: New York, NY, USA, 2007; pp. 389–432. [Google Scholar]
- Sinegani, A.A.S.; Mahohi, A. Temporal variability of available P, microbial P and some phosphomonoesterase activities in a sewage sludge treated soil: The effect of soil water potential. Afr. J. Biotechnol. 2009, 8, 6888–6895. [Google Scholar]
- Wells, J.M.; Thomas, J.; Boddy, L. Soil water potential shifts: Developmental responses and dependence on phosphorus translocation by the saprotrophic, cord-forming basidiomycete Phanerochaete velutina. Mycol. Res. 2001, 105, 859–867. [Google Scholar] [CrossRef]
- Grierson, P.F.; Comerford, N.B.; Jokela, E.J. Phosphorus mineralization and microbial biomass in a Florida Spodosol: Effects of water potential, temperature and fertilizer application. Biol. Fertil. Soils 1999, 28, 244–252. [Google Scholar] [CrossRef]
- Couvreur, V.; Vanderborght, J.; Beff, L.; Javaux, M. Horizontal soil water potential heterogeneity: Simplifying approaches for crop water dynamics models. Hydrol. Earth Syst. Sci. 2014, 18, 1723–1743. [Google Scholar] [CrossRef]
- Fu, B.; Li, S.; Yu, X.; Yang, P.; Yu, G.; Feng, R.; Zhuang, X. Chinese ecosystem research network: Progress and perspectives. Ecol. Complex. 2010, 7, 225–233. [Google Scholar] [CrossRef]
- Brantley, S.L.; McDowell, W.H.; Dietrich, W.E.; White, T.S.; Kumar, P.; Anderson, S.P.; Chorover, J.; Lohse, K.A.; Bales, R.C.; Richter, D.D. Designing a network of critical zone observatories to explore the living skin of the terrestrial Earth. Earth Surf. Dyn. 2017, 5, 841–860. [Google Scholar] [CrossRef]
- Yuan, Y.; Wang, F.Y.; IEEE. Towards blockchain-based intelligent transportation systems. In Proceedings of the 2016 IEEE 19th international conference on intelligent transportation systems (ITSC), Rio de Janeiro, Brazil, 1–4 November 2016; pp. 2663–2668. [Google Scholar]
- Vereecken, H.; Amelung, W.; Bauke, S.L.; Bogena, H.; Brüggemann, N.; Montzka, C.; Vanderborght, J.; Bechtold, M.; Blöschl, G.; Carminati, A.; et al. Soil hydrology in the earth system. Nat. Rev. Earth Environ. 2022, 3, 573–587. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, Y.; Liu, H.; Yu, Y.; Guo, L.; Zhao, W.; Yetemen, O. Revisiting Soil Water Potential: Towards a Better Understanding of Soil and Plant Interactions. Water 2022, 14, 3721. https://doi.org/10.3390/w14223721
Ma Y, Liu H, Yu Y, Guo L, Zhao W, Yetemen O. Revisiting Soil Water Potential: Towards a Better Understanding of Soil and Plant Interactions. Water. 2022; 14(22):3721. https://doi.org/10.3390/w14223721
Chicago/Turabian StyleMa, Yuanyuan, Hu Liu, Yang Yu, Li Guo, Wenzhi Zhao, and Omer Yetemen. 2022. "Revisiting Soil Water Potential: Towards a Better Understanding of Soil and Plant Interactions" Water 14, no. 22: 3721. https://doi.org/10.3390/w14223721
APA StyleMa, Y., Liu, H., Yu, Y., Guo, L., Zhao, W., & Yetemen, O. (2022). Revisiting Soil Water Potential: Towards a Better Understanding of Soil and Plant Interactions. Water, 14(22), 3721. https://doi.org/10.3390/w14223721










