Removal of Nutrients and COD in Wastewater from Vietnamese Piggery Farm by the Culture of Chlorella vulgaris in a Pilot-Scaled Membrane Photobioreactor
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microalgae Strain and Wastewater
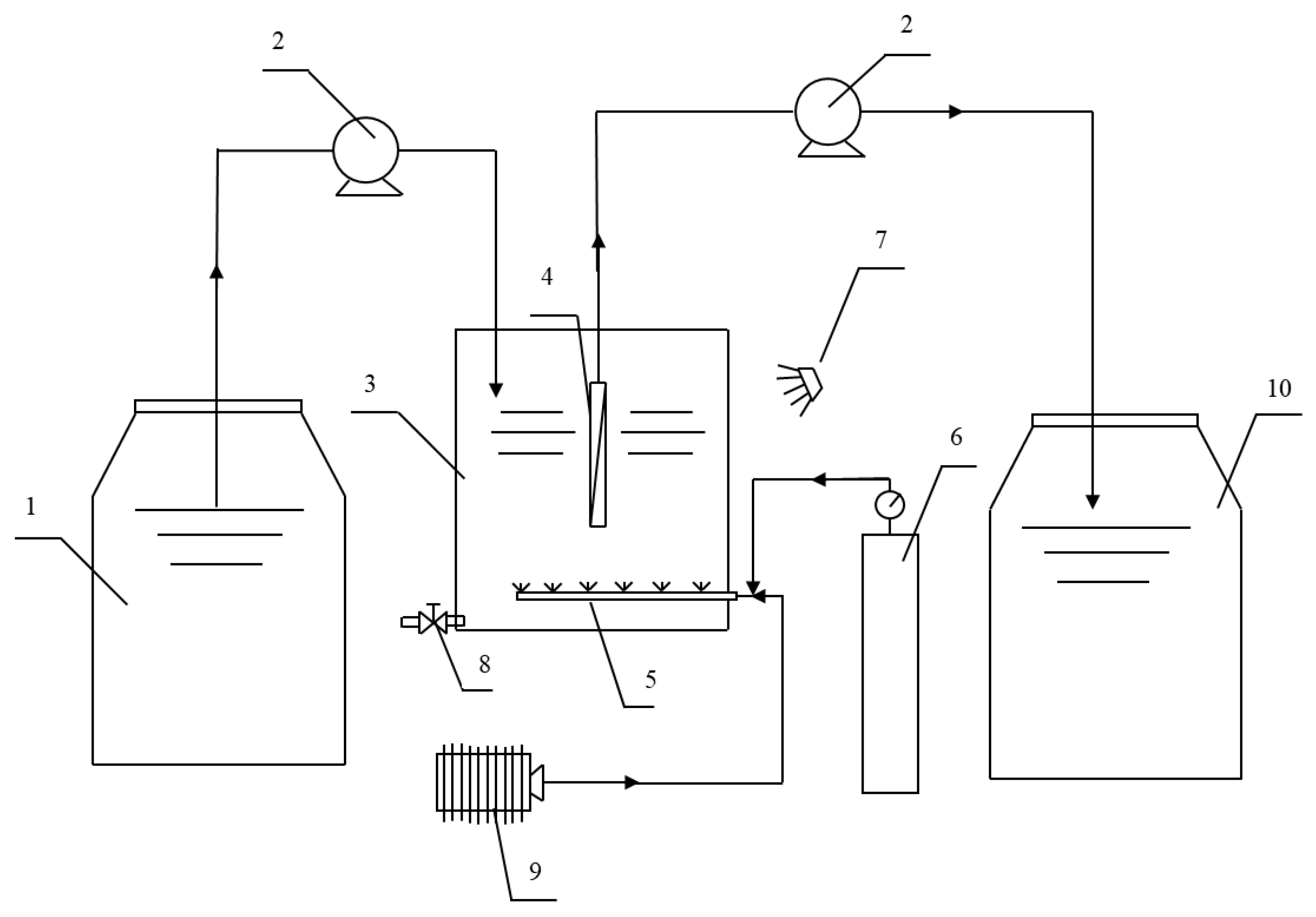
2.2. Experimental Procedure
2.3. Analysis of Algae Biomass and Water Quality
2.3.1. Algae Biomass Analysis
2.3.2. Algae Biomass Analysis
3. Results and Discussions
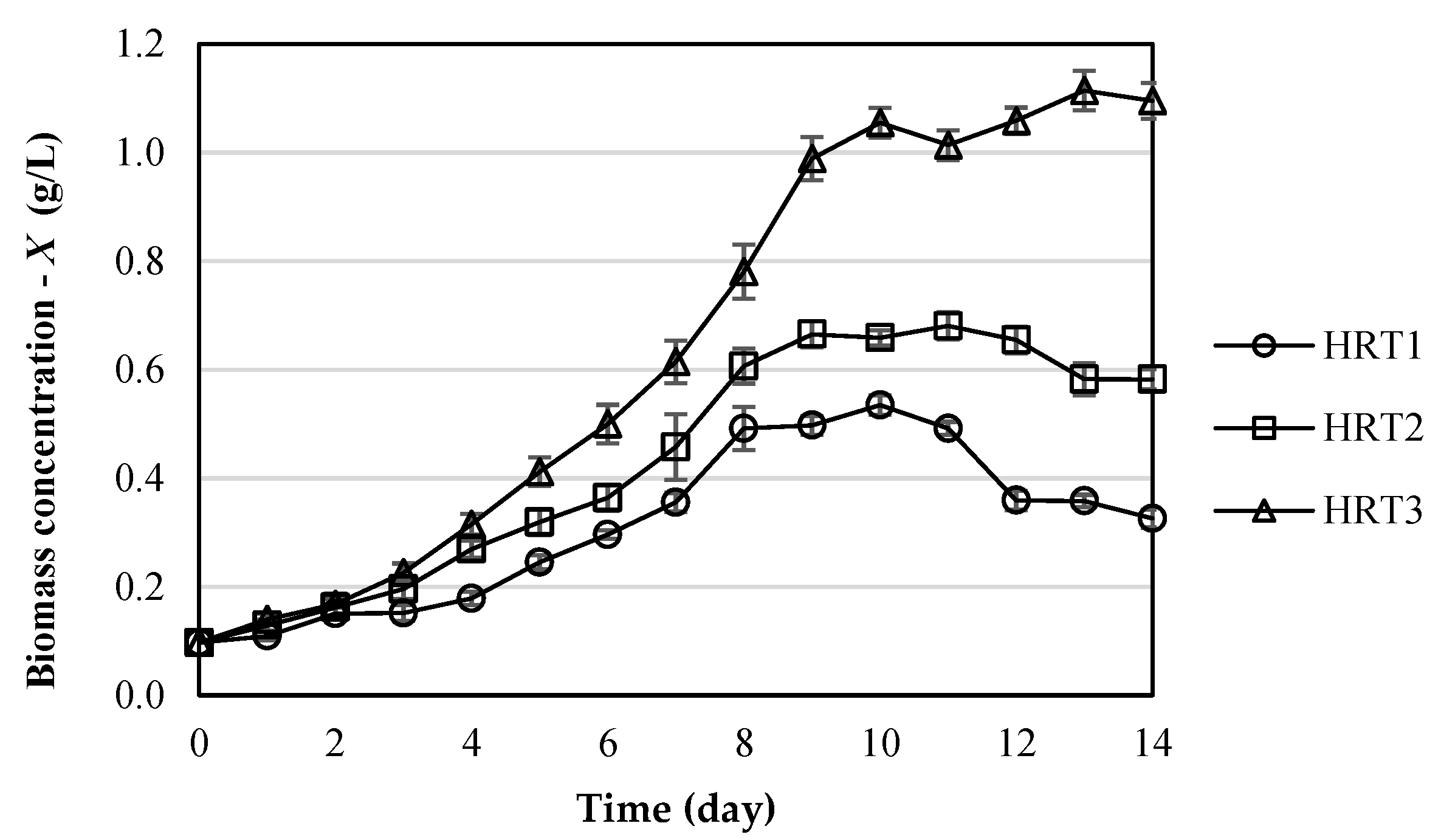
3.1. Microalgae Growth in Wastewater
| Cultivation Condition | Experimental Setup | HRT | Final Biomass Concentration, X | Specific Grow Rate, µ | Productivity, p | Ref. |
|---|---|---|---|---|---|---|
| Day | g/L | d−1 | g/L/d | |||
| 2-fold diluted piggery ww. | Continuous mode with MPBR | 1.25 | 0.326 ± 0.016 | 0.086 ± 0.004 | 0.016 ± 0.001 | This study |
| 2.5 | 0.582 ± 0.019 | 0.128 ± 0.002 | 0.035 ± 0.001 | |||
| 5.0 | 1.095 ± 0.033 | 0.173 ± 0.002 | 0.071 ± 0.002 | |||
| 10% swine with BG-11 | Batch mode | 12 | 3.16 | - | 0.188 | [1] |
| Artificial ww. (with glucose as COD) | Batch mode | 2 | 0.4 | - | - | [3] |
| Simulated ww. | Continuous mode with MPBR | 1 | 0.873 | - | 0.048 | [12] |
| 2 | 0.878 | - | 0.048 | |||
| 4 | 0.603 | - | 0.033 | |||
| 6 | 0.481 | - | 0.026 | |||
| 20% winery ww. with BBM | Continuous mode with MPBR | 1.4 | 6.10 ± 0.05 | 0.083 ± 0.004 | 0.240 ± 0.011 | [15] |
| 2.0 | 3.61 ± 0.04 | 0.003 ± 0.001 | 0.154 ± 0.013 | |||
| 4.6 | 2.90 ± 0.02 | 0.014 ± 0.002 | 0.136 ± 0.019 |
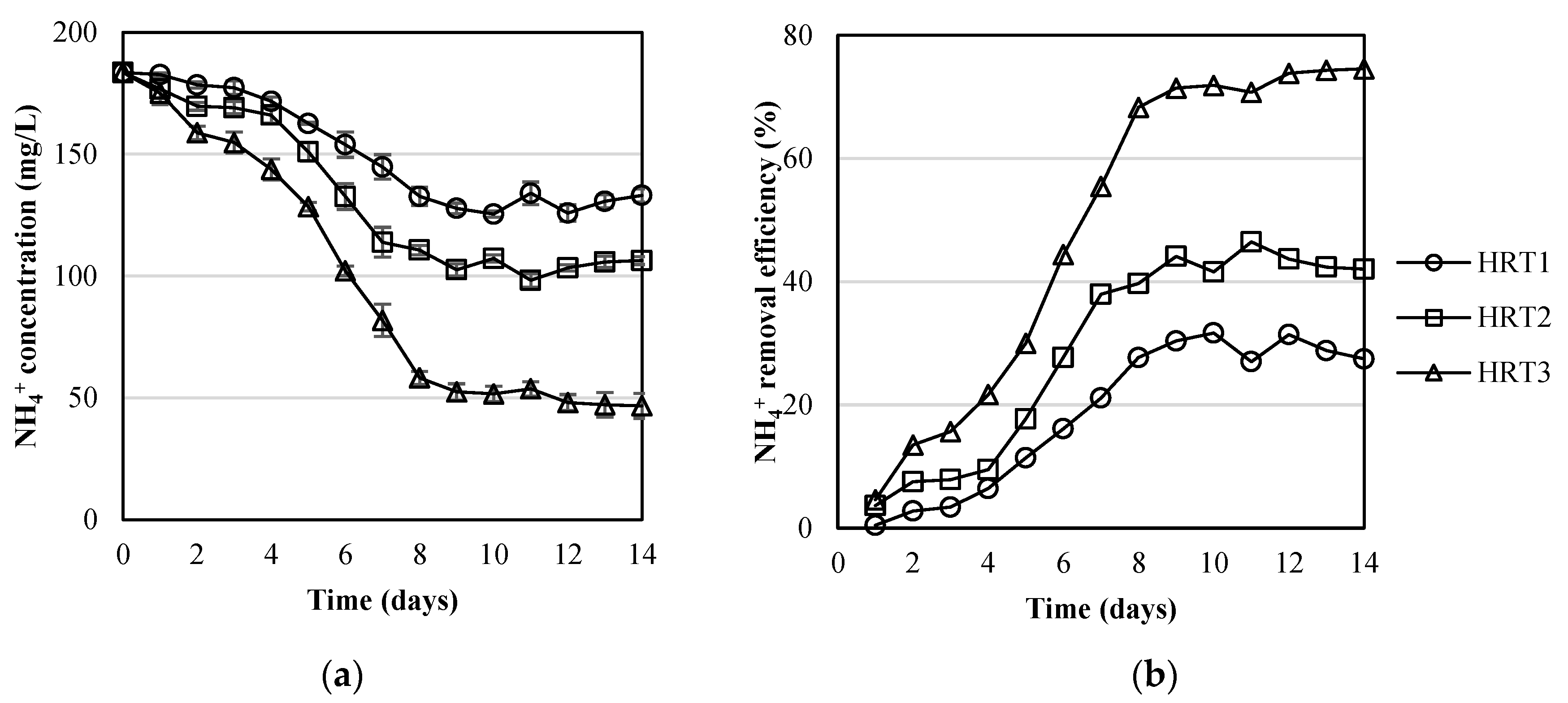
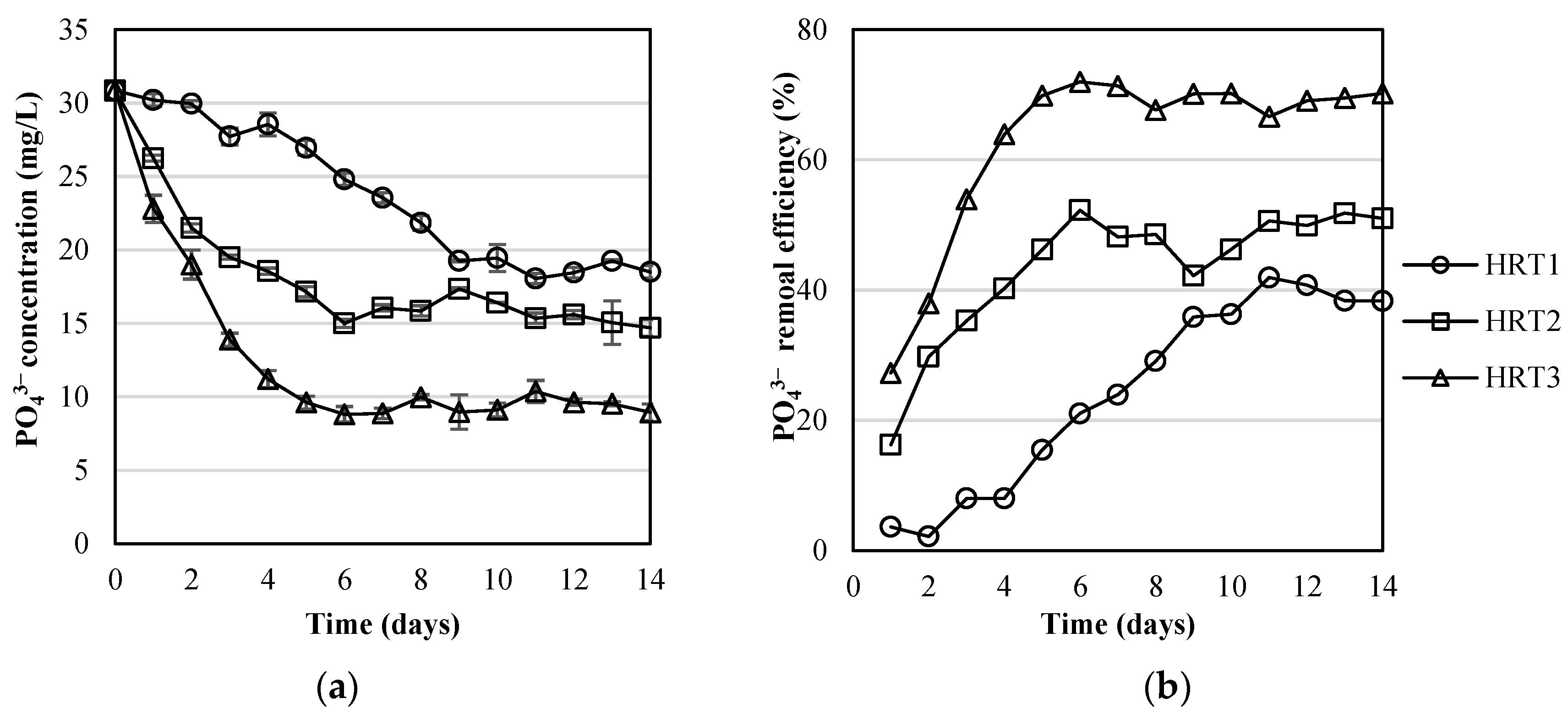
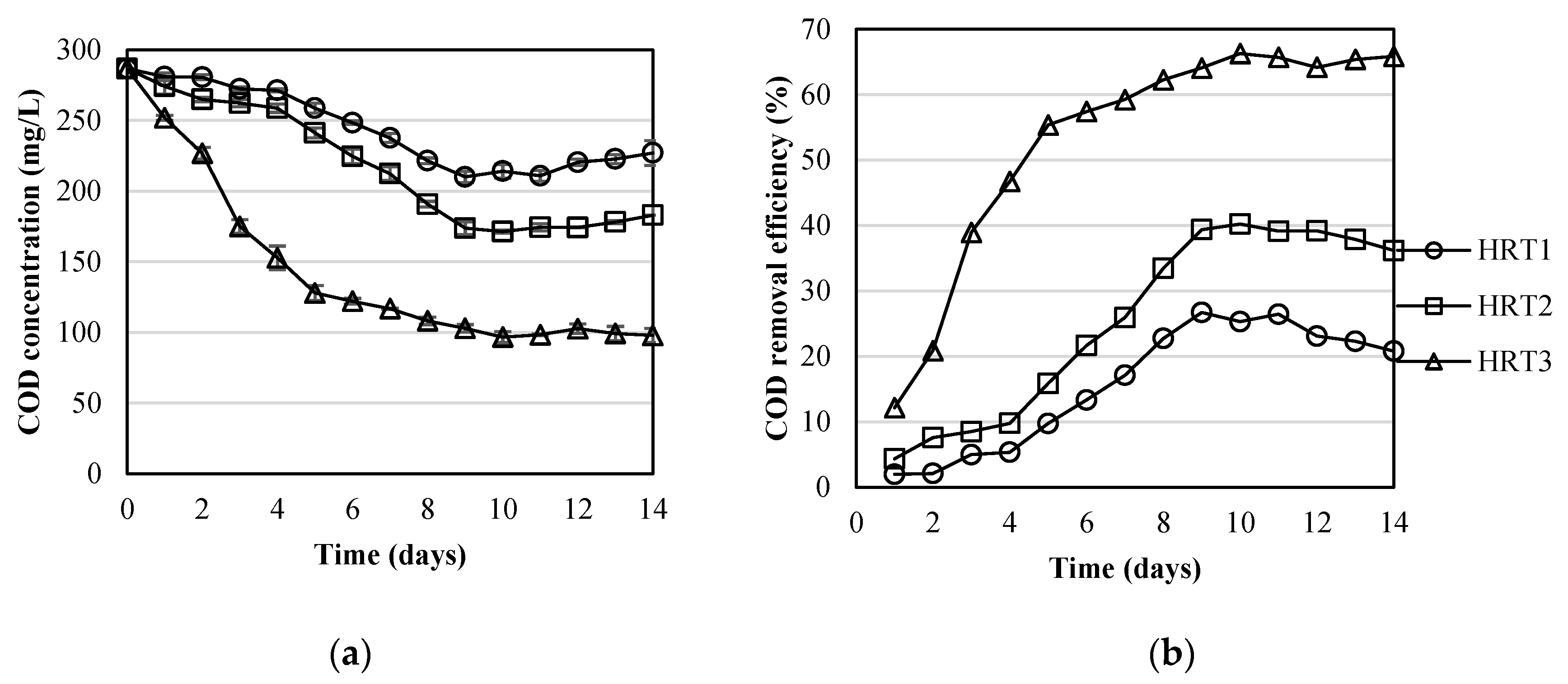
3.2. Pollutants Removal by the MPBR
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Acebu, P.I.G.; de Luna, M.D.G.; Chen, C.-Y.; Abarca, R.R.M.; Chen, J.-H.; Chang, J.-S. Bioethanol production from Chlorella vulgaris ESP-31 grown in unsterilized swine wastewater. Bioresour. Technol. 2022, 352, 127086. [Google Scholar] [CrossRef] [PubMed]
- Morais, E.; Cristofoli, N.; Maia, I.; Magina, T.; Cerqueira, P.; Teixeira, M.; Varela, J.; Barreira, L.; Gouveia, L. Microalgal Systems for Wastewater Treatment: Technological Trends and Challenges towards Waste Recovery. Energies 2021, 14, 8112. [Google Scholar] [CrossRef]
- Mujtaba, G.; Rizwan, M.; Kim, G.; Lee, K. Removal of nutrients and COD through co-culturing activated sludge and immobilized Chlorella vulgaris. Chem. Eng. J. 2018, 343, 155–162. [Google Scholar] [CrossRef]
- Pittman, J.K.; Dean, A.P.; Osundeko, O. The potential of sustainable algal biofuel production using wastewater resources. Bioresour. Technol. 2011, 102, 17–25. [Google Scholar] [CrossRef]
- He, P.J.; Mao, B.; L, F.; Shao, L.M.; Lee, D.J.; Chang, J.S. The combined effect of bacteria and Chlorella vulgaris on the treatment of municipal wastewaters. Bioresour. Technol. 2013, 146, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Guo, W.; Yen, H.-W.; Ho, S.-H.; Lo, Y.-C.; Cheng, C.-L.; Ren, N.; Chang, J.-S. Cultivation of Chlorella vulgaris JSC-6 with swine wastewater for simultaneous nutrient/COD removal and carbohydrate production. Bioresour. Technol. 2015, 198, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Song, A.; Huang, Y.; Liao, Q.; Xia, A.; Zhu, X.; Zhu, X. Domesticating Chlorella vulgaris with gradually increased the concentration of digested piggery wastewater to bio-remove ammonia nitrogen. Algal Res. 2021, 60, 102526. [Google Scholar] [CrossRef]
- Silva, D.A.; Cardoso, L.G.; de Jesus Silva, J.S.; de Souza, C.O.; Lemos, P.V.F.; de Almeida, P.F.; Ferreira, E.d.S.; Lombardi, A.T.; Druzian, J.I. Strategy for the cultivation of Chlorella vulgaris with high biomass production and biofuel potential in wastewater from the oil industry. Environ. Technol. Innov. 2022, 25, 102204. [Google Scholar] [CrossRef]
- Znad, H.; Al Ketife, A.M.D.; Judd, S.; AlMomani, F.; Vuthaluru, H.B. Bioremediation and nutrient removal from wastewater by Chlorella vulgaris. Ecol. Eng. 2018, 110, 1–7. [Google Scholar] [CrossRef]
- Gao, F.; Li, C.; Yang, Z.-H.; Zeng, G.-M.; Feng, L.-J.; Liu, J.-z.; Liu, M.; Cai, H.-W. Continuous microalgae cultivation in aquaculture wastewater by a membrane photobioreactor for biomass production and nutrients removal. Ecol. Eng. 2016, 92, 55–61. [Google Scholar] [CrossRef]
- Fan, Z.; Qin, L.; Zheng, W.; Meng, Q.; Shen, C.; Zhang, G. Oscillating membrane photoreactor combined with salt-tolerated Chlorella pyrenoidosa for landfill leachates treatment. Bioresour. Technol. 2018, 269, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Peng, Y.-Y.; Li, C.; Cui, W.; Yang, Z.-H.; Zeng, G.-M. Coupled nutrient removal from secondary effluent and algal biomass production in membrane photobioreactor (MPBR): Effect of HRT and long-term operation. Chem. Eng. J. 2018, 335, 169–175. [Google Scholar] [CrossRef]
- Gao, F.; Cui, W.; Xu, J.-P.; Li, C.; Jin, W.-H.; Yang, H.-L. Lipid accumulation properties of Chlorella vulgaris and Scenedesmus obliquus in membrane photobioreactor (MPBR) fed with secondary effluent from municipal wastewater treatment plant. Renew. Energy 2019, 136, 671–676. [Google Scholar] [CrossRef]
- Peng, Y.-Y.; Gao, F.; Yang, H.-L.; Wu, H.-W.-J.; Li, C.; Lu, M.-M.; Yang, Z.-Y. Simultaneous removal of nutrient and sulfonamides from marine aquaculture wastewater by concentrated and attached cultivation of Chlorella vulgaris in an algal biofilm membrane photobioreactor (BF-MPBR). Sci. Total Environ. 2020, 725, 138524. [Google Scholar] [CrossRef] [PubMed]
- Spennati, E.; Mirizadeh, S.; Casazza, A.A.; Solisio, C.; Converti, A. Chlorella vulgaris and Arthrospira platensis growth in a continuous membrane photobioreactor using industrial winery wastewater. Algal Res. 2021, 60, 102519. [Google Scholar] [CrossRef]
- Zheng, M.; Dai, J.; Ji, X.; Li, D.; He, Y.; Wang, M.; Huang, J.; Chen, B. An integrated semi-continuous culture to treat original swine wastewater and fix carbon dioxide by an indigenous Chlorella vulgaris MBFJNU-1 in an outdoor photobioreactor. Bioresour. Technol. 2021, 340, 125703. [Google Scholar] [CrossRef]
- MONRE. The National Environmental Report 2018: Water Quality in River Basins; Ministry of Natural Resources and Environment: Bangkok, Thailand, 2018.
- Giang, N.T.H.; Huong, L.T.T.; Yabe, M.; Thang, N.T.; Hieu, V.N.; Son, C.T. Recycling Wastewater in Intensive Swine Farms: Selected Case Studies in Vietnam. J. Fac. Agric. Kyushu Univ. 2021, 66, 115–121. [Google Scholar] [CrossRef]
- Villamar, C.-A.; Vera-Puerto, I.; Rivera, D.; De la Hoz, F. Reuse and Recycling of Livestock and Municipal Wastewater in Chilean Agriculture: A Preliminary Assessment. Water 2018, 10, 817. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Hu, J.; Lee, D.J.; Chang, Y.; Lee, Y.J. Sludge treatment: Current research trends. Bioresour Technol 2017, 243, 1159–1172. [Google Scholar] [CrossRef]
- Hu, B.; Li, Y.; Min, M.; Mohr, M.; Du, Z.-Y.; Chen, P.; Ruan, R. Mass Cultivation of Microalgae on Animal Wastewater: A Sequential Two-Stage Cultivation Process for Energy Crop and Omega-3-Rich Animal Feed Production. Appl. Biochem. Biotechnol. 2012, 168, 348–363. [Google Scholar] [CrossRef]
- Thuy, B. Vietnam approves circular economy development scheme. Available online: https://vir.com.vn/vietnam-approves-circular-economy-development-scheme-94112.html (accessed on 3 November 2022).
- Hai, H.; Nguyen, D.-Q.; Thang, N.; Nguyen, N.H. Circular Economy in Vietnam. In Circular Economy: Global Perspetive; Ghosh, S., Ed.; Springer: Singapore, 2020; pp. 423–452. [Google Scholar] [CrossRef]
- Sözüdoğru, O.; Massara, T.M.; Çalık, S.; Yılmaz, A.E.; Bakırdere, S.; Katsou, E.; Komesli, O.T. Influence of Hydraulic Retention Time (HRT) upon the Treatment of Wastewater by a Laboratory-Scale Membrane Bioreactor (MBR). Anal. Lett. 2021, 54, 1578–1587. [Google Scholar] [CrossRef]
- Chen, S.; Xie, J.; Wen, Z. Chapter Four—Microalgae-based wastewater treatment and utilization of microalgae biomass. In Advances in Bioenergy; Li, Y., Zhou, W., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; Volume 6, pp. 165–198. [Google Scholar]
- Wang, L.; Min, M.; Li, Y.; Chen, P.; Chen, Y.; Liu, Y.; Wang, Y.; Ruan, R. Cultivation of green algae Chlorella sp. in different wastewaters from municipal wastewater treatment plant. Appl. Biochem. Biotechnol. 2010, 162, 1174–1186. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Wang, Z.; Shu, Q.; Takala, J.; Hiltunen, E.; Feng, P.; Yuan, Z. Nutrient removal and biodiesel production by integration of freshwater algae cultivation with piggery wastewater treatment. Water Res. 2013, 47, 4294–4302. [Google Scholar] [CrossRef]
- ISO 6060:1989; Water Quality-Determination of the Chemical Oxygen Demand. International Organization for Standardization: Geneva, Switzerland, 1989; (reviewed and confirmed in 2017).
- ISO 7150-1:1984; Water Quality–Determination of Ammonium–Part 1: Manual Spectrometric Method. International Organization for Standardization: Geneva, Switzerland, 1984; (reviewed and confirmed in 2017).
- ISO 6878:2004; Water Quality-Determination of Phosphorus-Ammonium Molybdate Spectrometric Method. International Organization for Standardization: Geneva, Switzerland, 1984; (reviewed and confirmed in 2019).
- Kim, S.; Park, J.-e.; Cho, Y.-B.; Hwang, S.-J. Growth rate, organic carbon and nutrient removal rates of Chlorella sorokiniana in autotrophic, heterotrophic and mixotrophic conditions. Bioresour. Technol. 2013, 144, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Andreotti, V.; Solimeno, A.; Rossi, S.; Ficara, E.; Marazzi, F.; Mezzanotte, V.; García, J. Bioremediation of aquaculture wastewater with the microalgae Tetraselmis suecica: Semi-continuous experiments, simulation and photo-respirometric tests. Sci. Total Environ. 2020, 738, 139859. [Google Scholar] [CrossRef]
- Choi, H.-J.; Lee, S.-M. Performance of Chlorella vulgaris for the Removal of Ammonia-Nitrogen from Wastewater. Environ. Eng. Res. 2013, 18, 235–239. [Google Scholar] [CrossRef]
- Soto, M.F.; Diaz, C.A.; Zapata, A.M.; Higuita, J.C. BOD and COD removal in vinasses from sugarcane alcoholic distillation by Chlorella vulgaris: Environmental evaluation. Biochem. Eng. J. 2021, 176, 108191. [Google Scholar] [CrossRef]
| Parameters | Unit | Raw Piggery Wastewater | Screened 2-Fold Diluted Piggery Wastewater |
|---|---|---|---|
| pH | mg/L | 7–9 | 8.42 |
| COD | mg/L | 573.4 | 287.0 |
| TSS | mg/L | 143.3 | 35.2 |
| NH4+ | mg/L | 367 | 184.0 |
| NO2− | mg/L | 0.76 | 0.34 |
| NO3− | mg/L | 0.32 | 0.16 |
| PO43− | mg/L | 61.7 | 31.0 |
| Parameters | Unit | Value |
|---|---|---|
| pH | - | 8.42 |
| COD | mg/L | 287.0 |
| NH4+ | mg/L | 184.0 |
| PO43− | mg/L | 31.0 |
| Wastewater | Experimental Condition | HRT (Days) | Pollutants Removal Efficiency (%) | Ref. | ||
|---|---|---|---|---|---|---|
| NH4+ | PO43− | COD | ||||
| 2-fold diluted piggery wastewater | Continuous mode with MPBR NH4+ = 184 mg/L PO43− = 31 mg/L COD = 287 mg/L | 1.25 | 27.47 | 15.44 | 20.81 | This study |
| 2.5 | 42.04 | 46.19 | 36.19 | |||
| 5 | 74.55 | 69.85 | 65.85 | |||
| Mixture of swine wastewater (COD = 456 mg/L, NH4+ = 470 mg/L) and BG-11 | 25% swine mixture | 12 (Batch mode) | 41.7 | - | - | [1] |
| 50% swine mixture | 45.4 | - | - | |||
| 75% swine mixture | 28.6 | - | 16 | |||
| 100% swine | 29.9 | - | 30.4 | |||
| Artificial ww. | NH4 − N = 50 mg/L PO4 − P = 10 mg/L COD = 490 mg/L | 2 (Batch mode) | 92 | 87 | 60 | [3] |
| Simulated secondary effluent of municipal ww. | Continuous mode with MPBR NH4 − N = 5 mg/L PO4 − P = 40 mg/L COD = 0.8 mg/L | 1 | 35.9 | 76.9 | - | [12] |
| 2 | 76.9 | 88.0 | - | |||
| 4 | 90.6 | 94.9 | - | |||
| 6 | 92.1 | 94.0 | - | |||
| Chlorella vulgaris and Arthospira platensis in winery wastewater | Continuous mode with MPBR COD = 199.3 mg/L | 4.6 | - | - | >90% | [15] |
| 2.0 | - | - | ~75% | |||
| 1.2 | - | - | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, M.T.; Nguyen, T.P.; Pham, T.H.; Duong, T.T.; Do, M.V.; Trinh, T.V.; Nguyen, Q.T.X.; Trinh, V.M. Removal of Nutrients and COD in Wastewater from Vietnamese Piggery Farm by the Culture of Chlorella vulgaris in a Pilot-Scaled Membrane Photobioreactor. Water 2022, 14, 3645. https://doi.org/10.3390/w14223645
Nguyen MT, Nguyen TP, Pham TH, Duong TT, Do MV, Trinh TV, Nguyen QTX, Trinh VM. Removal of Nutrients and COD in Wastewater from Vietnamese Piggery Farm by the Culture of Chlorella vulgaris in a Pilot-Scaled Membrane Photobioreactor. Water. 2022; 14(22):3645. https://doi.org/10.3390/w14223645
Chicago/Turabian StyleNguyen, Minh Tuan, Thao Phuong Nguyen, Tung Huu Pham, Thuy Thi Duong, Manh Van Do, Tuyen Van Trinh, Quynh Thi Xuan Nguyen, and Viet M. Trinh. 2022. "Removal of Nutrients and COD in Wastewater from Vietnamese Piggery Farm by the Culture of Chlorella vulgaris in a Pilot-Scaled Membrane Photobioreactor" Water 14, no. 22: 3645. https://doi.org/10.3390/w14223645
APA StyleNguyen, M. T., Nguyen, T. P., Pham, T. H., Duong, T. T., Do, M. V., Trinh, T. V., Nguyen, Q. T. X., & Trinh, V. M. (2022). Removal of Nutrients and COD in Wastewater from Vietnamese Piggery Farm by the Culture of Chlorella vulgaris in a Pilot-Scaled Membrane Photobioreactor. Water, 14(22), 3645. https://doi.org/10.3390/w14223645








