Effective Removal of Reactive and Direct Dyes from Colored Wastewater Using Low-Cost Novel Bentonite Nanocomposites
Abstract
:1. Introduction
2. Materials and Methods
2.1. Dyes and Bentonite Nanocomposites
2.2. Adsorption Studies
2.2.1. Adsorption Isotherm and Kinetic Studies
2.2.2. Adsorption Kinetics
3. Results and Discussion
3.1. Spectrophotometric Analysis
3.2. Effect of Initial Dye Concentration
3.3. Effect of Adsorbent Dosage
3.4. Effect of Contact Time
3.5. Effect of Temperature
3.6. Effect of pH
3.7. Isothermal Adsorption Models
3.8. Kinetic Adsorption Models
3.8.1. Pseudo 1st-Order Model
3.8.2. Pseudo 2nd Order Model
3.9. Characterization of Bentonite
3.9.1. Scanning Electron Microscopy (SEM) Analysis
3.9.2. Fourier Transform Infrared (FTIR) Analysis
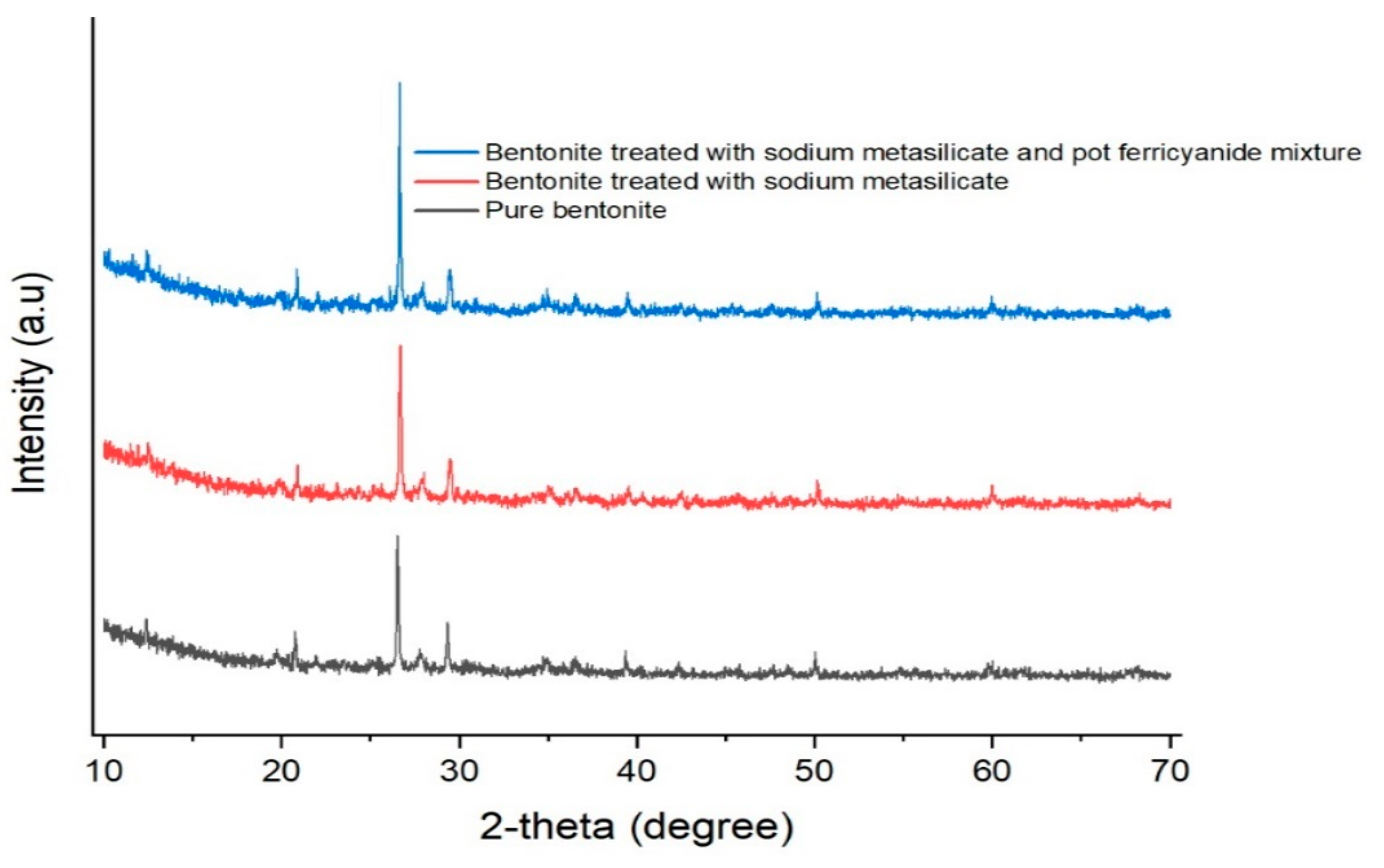
3.9.3. X-ray Diffraction (XRD) Analysis
3.9.4. Desorption
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Khan, S.; Malik, A. Environmental and health effects of textile industry wastewater. In Environmental Deterioration and Human Health; Springer: Berlin/Heidelberg, Germany, 2014; pp. 55–71. [Google Scholar]
- Sivaram, N.; Gopal, P.; Barik, D. Toxic waste from textile industries. In Energy from Toxic Organic Waste for Heat and Power Generation; Elsevier: Amsterdam, The Netherlands, 2019; pp. 43–54. [Google Scholar]
- Azizullah, A.; Khattak, M.N.K.; Richter, P.; Häder, D.-P. Water pollution in Pakistan and its impact on public health—A review. Environ. Int. 2011, 37, 479–497. [Google Scholar] [CrossRef] [PubMed]
- Halder, J.N.; Islam, M.N. Water pollution and its impact on the human health. J. Environ. Hum. 2015, 2, 36–46. [Google Scholar] [CrossRef]
- Shi, B.; Li, G.; Wang, D.; Feng, C.; Tang, H. Removal of direct dyes by coagulation: The performance of preformed polymeric aluminum species. J. Hazard. Mater. 2007, 143, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.S.; Saravanan, A. Sustainable wastewater treatments in textile sector. In Sustainable Fibres and Textiles; Elsevier: Amsterdam, The Netherlands, 2017; pp. 323–346. [Google Scholar]
- Beulah, S.S.; Muthukumaran, K. Methodologies of removal of dyes from wastewater: A review. Int. Res. J. Pure Appl. Chem. 2020, 50, 68–78. [Google Scholar] [CrossRef]
- Gupta, V.; Ali, I.; Saini, V.; Gerven, T.V.; der Bruggen, B.V.; Vandecasteele, C. Removal of dyes from wastewater using bottom ash. Ind. Eng. Chem. Res. 2005, 44, 3655–3664. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E.; Wilson, L.D.; Morin-Crini, N. Conventional and non-conventional adsorbents for wastewater treatment. Environ. Chem. Lett. 2019, 17, 195–213. [Google Scholar] [CrossRef]
- Mukhopadhyay, R.; Bhaduri, D.; Sarkar, B.; Rusmin, R.; Hou, D.; Khanam, R.; Sarkar, S.; Biswas, J.K.; Vithanage, M.; Bhatnagar, A. Clay–polymer nanocomposites: Progress and challenges for use in sustainable water treatment. J. Hazard. Mater. 2020, 383, 121125. [Google Scholar] [CrossRef]
- Apriandanu, D.O.B.; Yulizar, Y. CuO-bentonite-gold nanocomposites: Facile green preparation and their characterization. Mater. Lett. 2021, 284, 128911. [Google Scholar] [CrossRef]
- Hu, Q.H.; Qiao, S.Z.; Haghseresht, F.; Wilson, M.A.; Lu, G.Q. Adsorption study for removal of basic red dye using bentonite. Ind. Eng. Chem. Res. 2006, 45, 733–738. [Google Scholar] [CrossRef]
- Khelifi, S.; Ayari, F. Modified bentonite for anionic dye removal from aqueous solutions. Adsorbent regeneration by the photo-Fenton process. Comptes. Rendus. Chim. 2019, 22, 154–160. [Google Scholar] [CrossRef]
- Tahir, S.S.; Rauf, N. Removal of a cationic dye from aqueous solutions by adsorption onto bentonite clay. Chemosphere 2006, 63, 1842–1848. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Lu, R. An LCTF-based multispectral imaging system for estimation of apple fruit firmness: Part II. Selection of optimal wavelengths and development of prediction models. Trans. ASABE 2006, 49, 269–275. [Google Scholar] [CrossRef]
- Gong, Z.; Liao, L.; Lv, G.; Wang, X. A simple method for physical purification of bentonite. Appl. Clay Sci. 2016, 119, 294–300. [Google Scholar] [CrossRef]
- Mokhtar, A.; Abdelkrim, S.; Sardi, A.; Benyoub, A.; Besnaci, H.; Cherrak, R.; Hadjel, M.; Boukoussa, B. Preparation and characterization of anionic composite hydrogel for dyes adsorption and filtration: Non-linear isotherm and kinetics modeling. J. Polym. Environ. 2020, 28, 1710–1723. [Google Scholar] [CrossRef]
- Davies, A.; Vadodaria, B.; Hopwood, B.; Dexter, T.; Conn, D. Efficacy of microfiltration in decreasing propofol-induced pain. Anaesthesia 2002, 57, 557–561. [Google Scholar] [CrossRef]
- Khalil, M.; Hanif, M.A.; Rashid, U.; Ahmad, J.; Alsalme, A.; Tsubota, T. Low-cost novel nano-constructed granite composites for removal of hazardous Terasil dye from wastewater. Environ. Sci. Pollut. Res. 2022. [Google Scholar] [CrossRef] [PubMed]
- Ehrampoush, M.; Ghanizadeh, G.; Ghaneian, M. Equilibrium and kinetics study of reactive red 123 dye removal from aqueous solution by adsorption on eggshell. J. Environ. Health Sci. Eng. 2011, 8, 101–106. [Google Scholar]
- Dada, A.; Olalekan, A.; Olatunya, A.; Dada, O. Langmuir, Freundlich, Temkin and Dubinin–Radushkevich isotherms studies of equilibrium sorption of Zn2+ unto phosphoric acid modified rice husk. IOSR J. Appl. Chem. 2012, 3, 38–45. [Google Scholar]
- Bazrafshan, E.; Mostafapour, F.K.; Hosseini, A.R.; Khorshid, A.R.; Mahvi, A.H. Decolorisation of reactive red 120 dye by using single-walled carbon nanotubes in aqueous solutions. J. Chem. 2013, 2013, 938374. [Google Scholar] [CrossRef] [Green Version]
- Jawad, A.H.; Mohammed, I.A.; Abdulhameed, A.S. Tuning of fly ash loading into chitosan-ethylene glycol diglycidyl ether composite for enhanced removal of reactive red 120 dye: Optimization using the Box–Behnken design. J. Polym. Environ. 2020, 28, 2720–2733. [Google Scholar] [CrossRef]
- Khanam, A.; Nadeem, F.; Praveena, S.M.; Rashid, U. Removal of dyes using alginated, calcinized and hybrid materials–A comprehensive review. Int. J. Chem. Biochem. Sci. 2017, 12, 130–140. [Google Scholar]
- Noreen, S.; Bhatti, H.N.; Zuber, M.; Zahid, M.; Asgher, M. Removal of actacid orange-RL dye using biocomposites: Modeling studies. Pol. J. Environ. Stud. 2017, 26, 2125–2134. [Google Scholar] [CrossRef]
- Amin, N.K. Removal of direct blue-106 dye from aqueous solution using new activated carbons developed from pomegranate peel: Adsorption equilibrium and kinetics. J. Hazard. Mater. 2009, 165, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Toor, M.; Jin, B. Adsorption characteristics, isotherm, kinetics, and diffusion of modified natural bentonite for removing diazo dye. Chem. Eng. J. 2012, 187, 79–88. [Google Scholar] [CrossRef]
- El-Nagar, D.A.; Sary, D.H. Synthesis and characterization of nano bentonite and its effect on some properties of sandy soils. Soil Tillage Res. 2021, 208, 104872. [Google Scholar] [CrossRef]
- Zaitan, H.; Bianchi, D.; Achak, O.; Chafik, T. A comparative study of the adsorption and desorption of o-xylene onto bentonite clay and alumina. J. Hazard. Mater. 2008, 153, 852–859. [Google Scholar] [CrossRef]
- Saeed, A.; Hanif, M.A.; Nawaz, H.; Qadri, R.W.K. The production of biodiesel from plum waste oil using nano-structured catalyst loaded into supports. Sci. Rep. 2021, 11, 24120. [Google Scholar] [CrossRef]










| Adsorption Isotherms | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dyes | Langmuir Isotherm | Freundlich Isotherm | Dubinin Radushkevich Isotherm | Temkin Isotherm | Harkin–Jura Isotherm | ||||||||||||||
| Reactive Green-5 Dye | Adsorbents | Qexp (mg/g) | R2 | Qcal | KL | R2 | Qcal | Kf | n | R2 | Qcal | β | E | R2 | At | B | R2 | A | B |
| Bentonite | 115.7 | 0.950 | 181.8 | 0.042 | 0.999 | 112.5 | 12.1 | 1.6 | 0.710 | 72.3 | 2 × 10−6 | 500 | 0.937 | 0.53 | 35.04 | 0.820 | 625 | 1.4 | |
| BPF | 178.1 | 0.979 | 227.3 | 0.098 | 0.992 | 185.2 | 30.1 | 1.9 | 0.771 | 116.6 | 8 × 10−7 | 1000 | 0.729 | 0.03 | 74.64 | 0.677 | 19.1 | 1.7 | |
| BSM | 171.0 | 0.895 | 243.9 | 0.058 | 0.983 | 164.3 | 21.3 | 1.7 | 0.742 | 104 | 1 × 10−6 | 707 | 0.567 | 0.04 | 37.42 | 0.865 | 10.5 | 1.7 | |
| BSMPF | 126.9 | 0.971 | 185.2 | 0.055 | 0.992 | 131.0 | 15.4 | 1.6 | 0.764 | 83.29 | 2 × 10−6 | 500 | 0.96 | 0.64 | 37.26 | 0.769 | 833.3 | 1.4 | |
| Reactive Red Dye | Bentonite | 156.7 | 0.928 | 227.3 | 0.006 | 0.99 | 154.5 | 20.5 | 1.8 | 0.685 | 95.21 | 1 × 10−6 | 707 | 0.92 | 0.76 | 43.47 | 0.326 | 6.87 | 1.6 |
| BPF | 106.3 | 0.995 | 123.5 | 0.011 | 0.973 | 114 | 24.7 | 2.4 | 0.813 | 80.17 | 1 × 10−6 | 707 | 0.994 | 1.63 | 25.42 | 0.766 | 0.72 | 1.7 | |
| BSM | 115.1 | 0.947 | 131.6 | 0.426 | 0.969 | 108.9 | 29.3 | 1.7 | 0.675 | 77.8 | 6 × 10−7 | 913 | 0.906 | 2.1 | 23.62 | 0.871 | 2000 | 1.6 | |
| BSMPF | 155.4 | 0.929 | 222.2 | 0.093 | 0.992 | 152.9 | 21.2 | 1.8 | 0.674 | 94.6 | 1 × 10−6 | 707 | 0.919 | 0.79 | 42.56 | 0.864 | 25.38 | -1 | |
| Acid Red Dye | Bentonite | 425.2 | 0.978 | 909.1 | 0.118 | 0.992 | 460.3 | 93.4 | 1.3 | 0.807 | 252.3 | 2 × 10−7 | 1581 | 0.935 | 0.06 | 19.4 | 0.689 | 2500 | 0.5 |
| BPF | 424.3 | 0.888 | 833.3 | 0.14 | 0.968 | 472.7 | 100 | 1.3 | 0.781 | 253.9 | 2 × 10−7 | 1581 | 0.489 | 0.21 | 3.73 | 0.335 | 9.9 | 1.8 | |
| BSM | 130.6 | 0.962 | 212.8 | 0.040 | 0.997 | 131.8 | 12.4 | 1.5 | 0.762 | 82.1 | 2 × 10−6 | 500 | 0.182 | 0.22 | 7.47 | 0.761 | 666.7 | 1.4 | |
| BSMPF | 147.6 | 0.899 | 212.8 | 0.057 | 0.988 | 140 | 19.9 | 1.8 | 0.639 | 87.5 | 1 × 10−6 | 707 | 0.118 | 0.85 | 135.6 | 0.027 | 666.7 | 52 | |
| Direct Violet-51 Dye | Bentonite | 272.8 | 0.991 | 384.6 | 0.10 | 0.991 | 29.37 | 42.6 | 1.6 | 0.757 | 165.2 | 5 × 10−7 | 1000 | 0.961 | 1.28 | 75.6 | 0.722 | 2500 | 13 |
| BPF | 397.8 | 0.961 | 526.3 | 0.29 | 0.998 | 11.75 | 106 | 1.7 | 0.771 | 227.3 | 1 × 10−7 | 2236 | 0.924 | 4.8 | 94.8 | 0.704 | 204.1 | 1.9 | |
| BSM | 396.7 | 0.969 | 526.3 | 0.27 | 0.996 | 11.09 | 110 | 1.7 | 0.727 | 218.7 | 9 × 10−8 | 2357 | 0.919 | 4.58 | 92.4 | 0.748 | 5000 | 1 | |
| BSMPF | 402.9 | 0.927 | 526.3 | 0.302 | 0.995 | 38.59 | 116 | 1.9 | 0.688 | 210.7 | 7 × 10−8 | 2673 | 0.871 | 5.72 | 85.8 | 0.803 | 5000 | 1 | |
| (a) | ||||||||
| Reactive Green-5 Dye | Adsorption Kinetics | |||||||
| Adsorbents | Pseudo 1st Order Kinetics | Pseudo 2nd Order Kinetics | ||||||
| 30 °C | ||||||||
| Qexp | Q° | K1 | R2 | Qexp | Q° | K2 | R2 | |
| Bentonite | 275.7 | 70.37 | 0.016 | 0.864 | 275.7 | 277.8 | 0.0005 | 0.997 |
| BPF | 257.1 | 43.36 | 0.019 | 0.754 | 257.1 | 263.2 | 0.0008 | 0.999 |
| BSM | 261.5 | 32.20 | 0.015 | 0.926 | 261.5 | 263.2 | 0.0013 | 1 |
| BSMPF | 311.2 | 103 | 0.018 | 0.952 | 311.2 | 322.6 | 0.0004 | 0.999 |
| 40 °C | ||||||||
| Bentonite | 292.1 | 148.15 | 0.016 | 0.958 | 292.1 | 294.12 | 0.0008 | 0.999 |
| BPF | 352.1 | 52.77 | 0.019 | 0.869 | 352.1 | 357.14 | 0.0003 | 0.994 |
| BSM | 223.2 | 120.1 | 0.018 | 0.933 | 223.2 | 232.56 | 0.0003 | 0.996 |
| BSMPF | 373.2 | 90.80 | 0.017 | 0.889 | 373.2 | 370.4 | 0.0005 | 0.998 |
| 50 °C | ||||||||
| Bentonite | 759.1 | 95.74 | 0.018 | 0.940 | 759.1 | 769.2 | 0.0005 | 0.999 |
| BPF | 832.2 | 217.5 | 0.022 | 0.986 | 832.2 | 833.3 | 0.0002 | 0.999 |
| BSM | 744.3 | 19.94 | 0.012 | 0.932 | 744.3 | 769.2 | 0.0007 | 0.999 |
| BSMPF | 760.1 | 96.72 | 0.018 | 0.947 | 760.1 | 769.2 | 0.0005 | 0.999 |
| 60 °C | ||||||||
| Bentonite | 738 | 91.47 | 0.018 | 0.949 | 738 | 769.2 | 0.0005 | 0.999 |
| BPF | 746.1 | 42.28 | 0.015 | 0.987 | 746.1 | 769.2 | 0.0010 | 1 |
| BSM | 728.9 | 66.62 | 0.017 | 0.969 | 728.9 | 714.3 | 0.0007 | 0.999 |
| BSMPF | 746.1 | 41.23 | 0.017 | 0.899 | 746.1 | 769.2 | 0.0009 | 1 |
| 70 °C | ||||||||
| Bentonite | 669.8 | 23.56 | 0.015 | 0.811 | 669.8 | 666.7 | 0.0017 | 1 |
| BPF | 685.4 | 27.4 | 0.015 | 0.96 | 685.4 | 714.3 | 0.0015 | 1 |
| BSM | 671.9 | 69.15 | 0.016 | 0.907 | 671.9 | 666.7 | 0.0006 | 0.999 |
| BSMPF | 690.5 | 23.63 | 0.015 | 0.852 | 690.5 | 666.7 | 0.0019 | 1 |
| (b) | ||||||||
| Reactive Red Dye | Adsorption Kinetics | |||||||
| Adsorbents | Pseudo 1st Order Kinetics | Pseudo 2nd Order Kinetics | ||||||
| 30 °C | ||||||||
| Qexp | Q° | K1 | R2 | Qexp | Q° | K2 | R2 | |
| Bentonite | 204.5 | 62.82 | 0.0184 | 0.912 | 204.5 | 212.77 | 0.0004 | 0.998 |
| BPF | 117.8 | 92.0 | 0.017 | 0.863 | 117.8 | 123.46 | 0.0003 | 0.960 |
| BSM | 154.5 | 52.52 | 0.0147 | 0.853 | 154.5 | 156.25 | 0.0007 | 0.993 |
| BSMPF | 199.4 | 21.20 | 0.0147 | 0.644 | 199.4 | 204.1 | 0.0010 | 0.999 |
| 40 °C | ||||||||
| Bentonite | 138.6 | 34.94 | 0.013 | 0.886 | 138.6 | 140.8 | 0.0010 | 0.996 |
| BPF | 142.5 | 19.84 | 0.015 | 0.796 | 142.5 | 144.9 | 0.0017 | 0.999 |
| BSM | 173.2 | 25.59 | 0.017 | 0.654 | 173.2 | 175.4 | 0.0013 | 0.999 |
| BSMPF | 155.4 | 54.45 | 0.015 | 0.909 | 155.4 | 158.7 | 0.0007 | 0.996 |
| 50 °C | ||||||||
| Bentonite | 169.2 | 26.79 | 0.014 | 0.999 | 169.2 | 172.4 | 0.0014 | 0.999 |
| BPF | 149.6 | 33.88 | 0.015 | 0.977 | 149.6 | 151.5 | 0.0011 | 0.999 |
| BSM | 147.3 | 34.59 | 0.014 | 0.93 | 147.3 | 149.3 | 0.0011 | 0.999 |
| BSMPF | 164 | 25.72 | 0.014 | 0.977 | 164 | 166.7 | 0.0015 | 0.999 |
| 60 °C | ||||||||
| Bentonite | 375.9 | 36.74 | 0.0140 | 0.924 | 375.9 | 384.6 | 0.0011 | 0.999 |
| BPF | 309 | 19.48 | 0.0131 | 0.903 | 309 | 312.5 | 0.002 | 1 |
| BSM | 390.5 | 120.22 | 0.0187 | 0.944 | 390.5 | 416.7 | 0.0004 | 0.999 |
| BSMPF | 406.5 | 57.58 | 0.0175 | 0.987 | 406.5 | 400 | 0.0008 | 1 |
| 70 °C | ||||||||
| Bentonite | 141.1 | 16.89 | 0.011 | 0.933 | 141.1 | 142.86 | 0.0021 | 0.999 |
| BPF | 163.3 | 88.69 | 0.0177 | 0.965 | 163.3 | 169.49 | 0.0004 | 0.997 |
| BSM | 185 | 122.7 | 0.0193 | 0.987 | 185 | 196.08 | 0.0003 | 0.999 |
| BSMPF | 234.98 | 117.5 | 0.0189 | 0.968 | 234.98 | 243.9 | 0.0003 | 0.998 |
| (c) | ||||||||
| Acid Red Dye | Adsorption Kinetics | |||||||
| Adsorbents | Pseudo 1st Order Kinetics | Pseudo 2nd Order Kinetics | ||||||
| 30 °C | ||||||||
| Qe | Q° | K1 | R2 | Qe | Q° | K2 | R2 | |
| Bentonite | 149.1 | 46.68 | 0.016 | 0.997 | 149.1 | 153.9 | 0.0008 | 0.999 |
| BPF | 133.1 | 31.25 | 0.014 | 0.973 | 133.1 | 135.1 | 0.0011 | 0.999 |
| BSM | 146.4 | 34.98 | 0.014 | 0.929 | 146.4 | 149.3 | 0.0010 | 0.998 |
| BSMPF | 154.8 | 70 | 0.016 | 0.912 | 154.8 | 158.7 | 0.0005 | 0.994 |
| 40 °C | ||||||||
| Bentonite | 317.7 | 162.9 | 0.023 | 0.932 | 317.7 | 344.8 | 0.0002 | 0.997 |
| BPF | 270.3 | 153.2 | 0.020 | 0.954 | 270.3 | 285.7 | 0.0003 | 0.996 |
| BSM | 250.3 | 31 | 0.015 | 0.921 | 250.3 | 256.4 | 0.0013 | 1 |
| BSMPF | 180 | 41 | 0.019 | 0.751 | 180 | 185.2 | 0.0008 | 0.999 |
| 50 °C | ||||||||
| Bentonite | 198.2 | 73.81 | 0.018 | 0.982 | 198.2 | 204.1 | 0.0005 | 0.999 |
| BPF | 243.6 | 84.76 | 0.017 | 0.938 | 243.6 | 250 | 0.0004 | 0.998 |
| BSM | 181.3 | 65.4 | 0.018 | 0.988 | 181.3 | 188.7 | 0.0006 | 0.999 |
| BSMPF | 137.9 | 12.7 | 0.010 | 0.970 | 137.9 | 138.9 | 0.0027 | 0.999 |
| 60 °C | ||||||||
| Bentonite | 274.4 | 38.1 | 0.014 | 0.934 | 274.4 | 277.8 | 0.0010 | 0.999 |
| BPF | 363.6 | 102.4 | 0.019 | 0.952 | 363.6 | 370.3 | 0.0004 | 0.999 |
| BSM | 323.1 | 74.5 | 0.016 | 0.867 | 323.1 | 322.6 | 0.0005 | 0.998 |
| BSMPF | 284.1 | 50.4 | 0.017 | 0.956 | 284.1 | 285.7 | 0.0008 | 0.999 |
| 70 °C | ||||||||
| Bentonite | 189.6 | 56.9 | 0.020 | 0.773 | 189.6 | 196.1 | 0.0006 | 0.997 |
| BPF | 163.7 | 80.7 | 0.017 | 0.890 | 163.7 | 169.5 | 0.0004 | 0.991 |
| BSM | 181.6 | 64.4 | 0.017 | 0.971 | 181.6 | 185.2 | 0.0006 | 0.999 |
| BSMPF | 162.8 | 52.6 | 0.015 | 0.939 | 162.8 | 166.7 | 0.0007 | 0.997 |
| (d) | ||||||||
| Direct Violet-51 Dye | Adsorption Kinetics | |||||||
| Adsorbents | Pseudo 1st Order Kinetics | Pseudo 2nd Order Kinetics | ||||||
| 30 °C | ||||||||
| Qe | Q° | K1 | R2 | Qe | Q° | K2 | R2 | |
| Bentonite | 186.5 | 89.0 | 0.017 | 0.895 | 186.5 | 192.3 | 0.0004 | 0.993 |
| BPF | 162.7 | 85.3 | 0.018 | 0.990 | 162.7 | 147.0 | 0.0004 | 0.996 |
| BSM | 172.6 | 74.4 | 0.017 | 0.952 | 172.6 | 178.5 | 0.0005 | 0.998 |
| BSMPF | 136.8 | 150.1 | 0.020 | 0.965 | 136.8 | 181.8 | 0.0002 | 0.998 |
| 40 °C | ||||||||
| Bentonite | 104.9 | 64.66 | 0.019 | 0.871 | 104.9 | 129.9 | 0.0002 | 0.987 |
| BPF | 132.2 | 24.8 | 0.015 | 0.842 | 132.2 | 135.1 | 0.0012 | 0.999 |
| BSM | 153.6 | 17.52 | 0.015 | 0.601 | 153.6 | 158.7 | 0.0012 | 0.999 |
| BSMPF | 107.3 | 132.1 | 0.020 | 0.980 | 107.3 | 138.9 | 0.0001 | 0.979 |
| 50 °C | ||||||||
| Bentonite | 178.7 | 21.98 | 0.0138 | 0.907 | 178.7 | 188.7 | 0.0044 | 0.998 |
| BPF | 141.4 | 99.40 | 0.0177 | 0.941 | 141.4 | 149.2 | 0.0003 | 0.992 |
| BSM | 100.5 | 32.12 | 0.0172 | 0.720 | 100.5 | 105.2 | 0.0008 | 0.998 |
| BSMPF | 172.3 | 25.06 | 0.0161 | 0.693 | 172.3 | 178.5 | 0.0006 | 0.999 |
| 60 °C | ||||||||
| Bentonite | 163.9 | 122.8 | 0.020 | 0.994 | 163.9 | 187.8 | 0.0002 | 0.998 |
| BPF | 180.9 | 86.47 | 0.017 | 0.933 | 180.9 | 188.7 | 0.0004 | 0.996 |
| BSM | 213.2 | 191.9 | 0.020 | 0.908 | 213.2 | 243.9 | 0.0001 | 0.969 |
| BSMPF | 174.7 | 83.98 | 0.018 | 0.930 | 174.7 | 185.1 | 0.0004 | 0.997 |
| 70 °C | ||||||||
| Bentonite | 203 | 36.38 | 0.019 | 0.639 | 203 | 212.8 | 0.0005 | 0.996 |
| BPF | 141.1 | 133.2 | 0.020 | 0.989 | 141.1 | 161.3 | 0.0002 | 0.991 |
| BSM | 60.5 | 9.79 | 0.012 | 0.684 | 60.5 | 62.11 | 0.0030 | 0.999 |
| BSMPF | 239.2 | 25.79 | 0.012 | 0.893 | 239.2 | 238.1 | 0.0015 | 0.999 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chauhdary, Y.; Hanif, M.A.; Rashid, U.; Bhatti, I.A.; Anwar, H.; Jamil, Y.; Alharthi, F.A.; Kazerooni, E.A. Effective Removal of Reactive and Direct Dyes from Colored Wastewater Using Low-Cost Novel Bentonite Nanocomposites. Water 2022, 14, 3604. https://doi.org/10.3390/w14223604
Chauhdary Y, Hanif MA, Rashid U, Bhatti IA, Anwar H, Jamil Y, Alharthi FA, Kazerooni EA. Effective Removal of Reactive and Direct Dyes from Colored Wastewater Using Low-Cost Novel Bentonite Nanocomposites. Water. 2022; 14(22):3604. https://doi.org/10.3390/w14223604
Chicago/Turabian StyleChauhdary, Yusra, Muhammad Asif Hanif, Umer Rashid, Ijaz Ahmad Bhatti, Hafeez Anwar, Yasir Jamil, Fahad A. Alharthi, and Elham Ahmed Kazerooni. 2022. "Effective Removal of Reactive and Direct Dyes from Colored Wastewater Using Low-Cost Novel Bentonite Nanocomposites" Water 14, no. 22: 3604. https://doi.org/10.3390/w14223604









