Aquatic Microplastic Pollution Control Strategies: Sustainable Degradation Techniques, Resource Recovery, and Recommendations for Bangladesh
Abstract
1. Introduction
2. Outline of Methodology
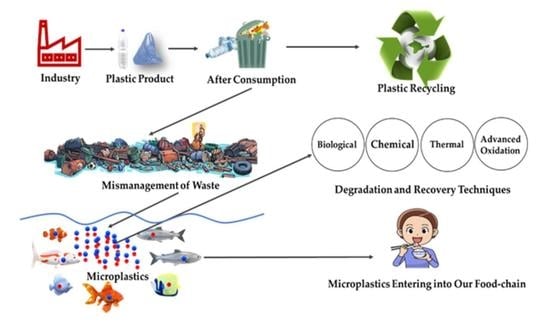
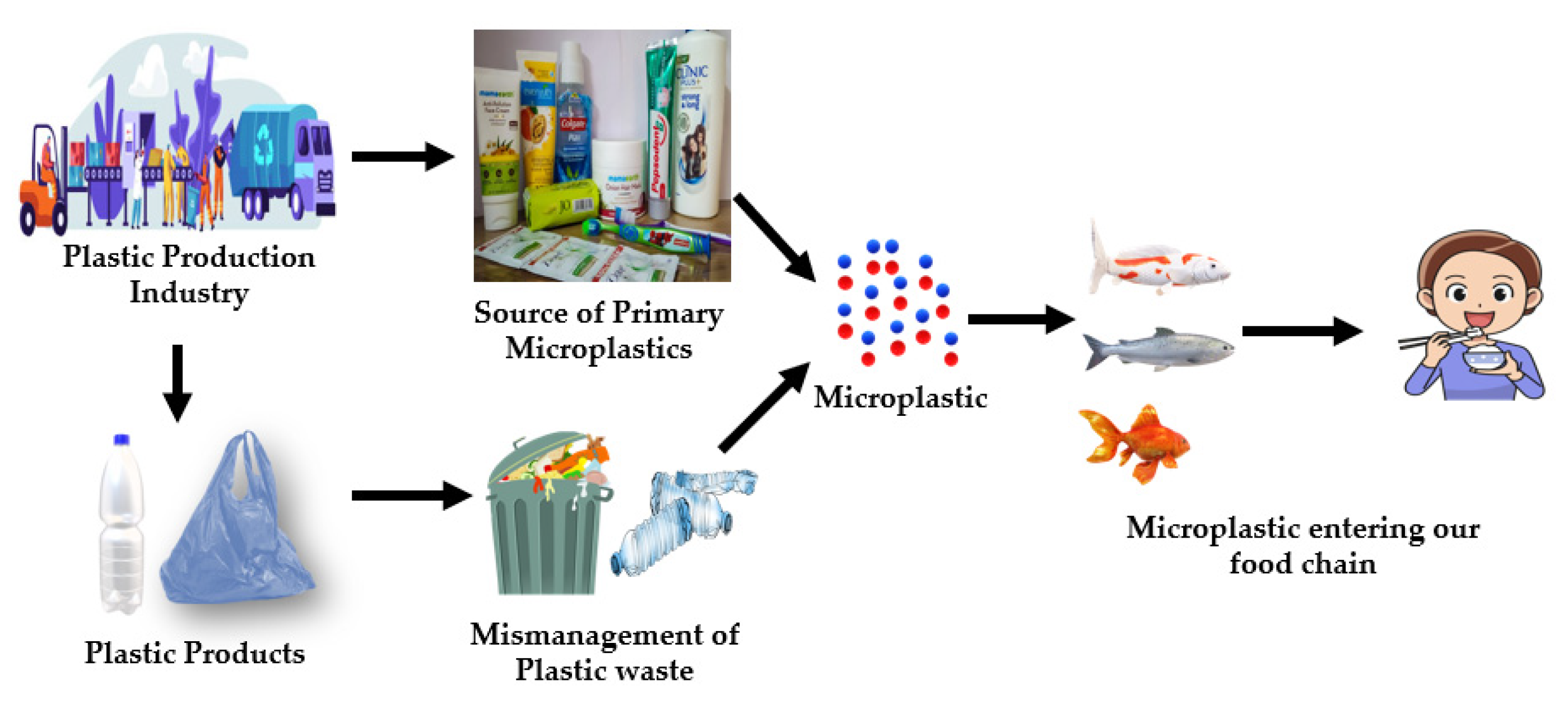
3. Roadmap of Microplastic
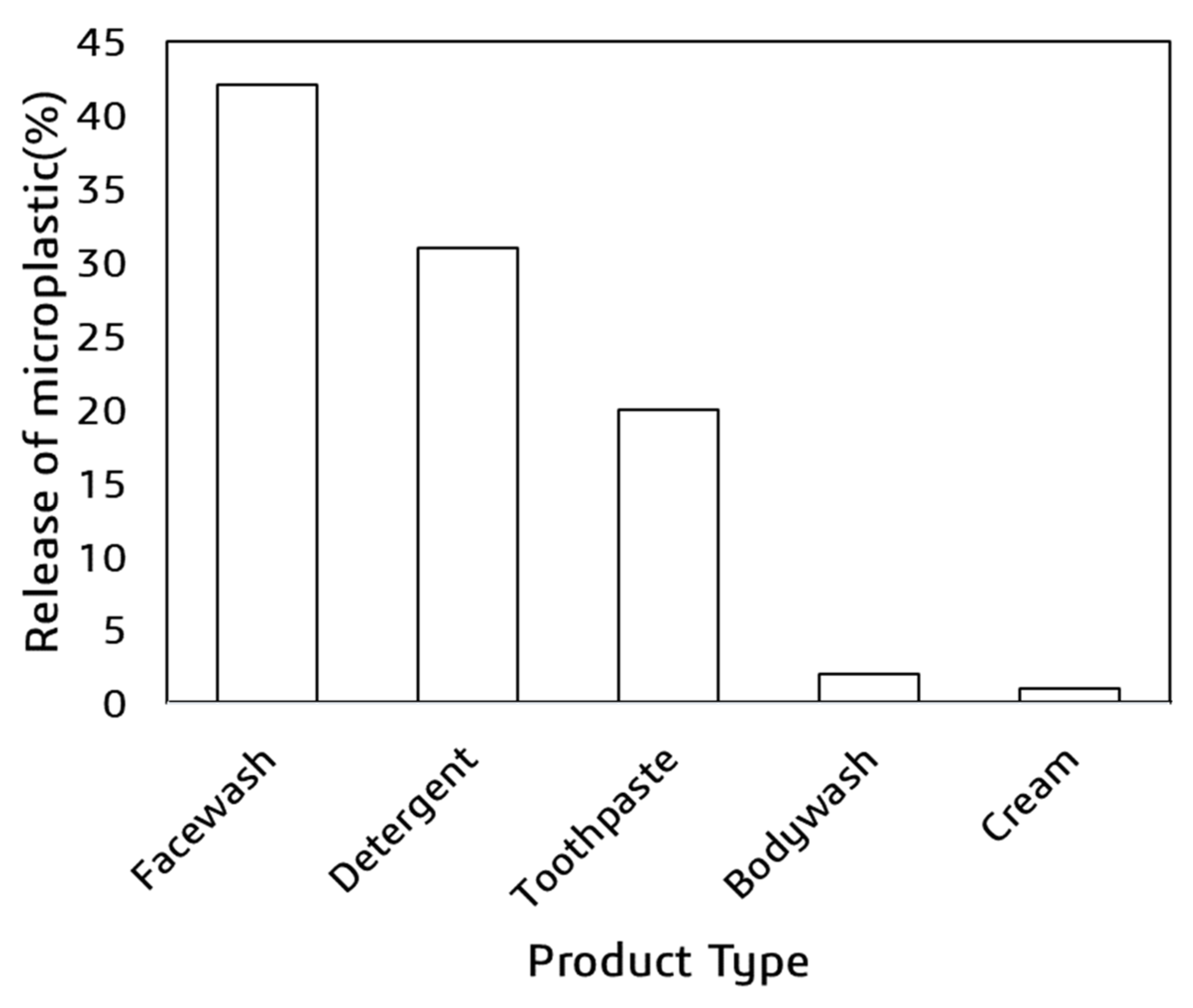
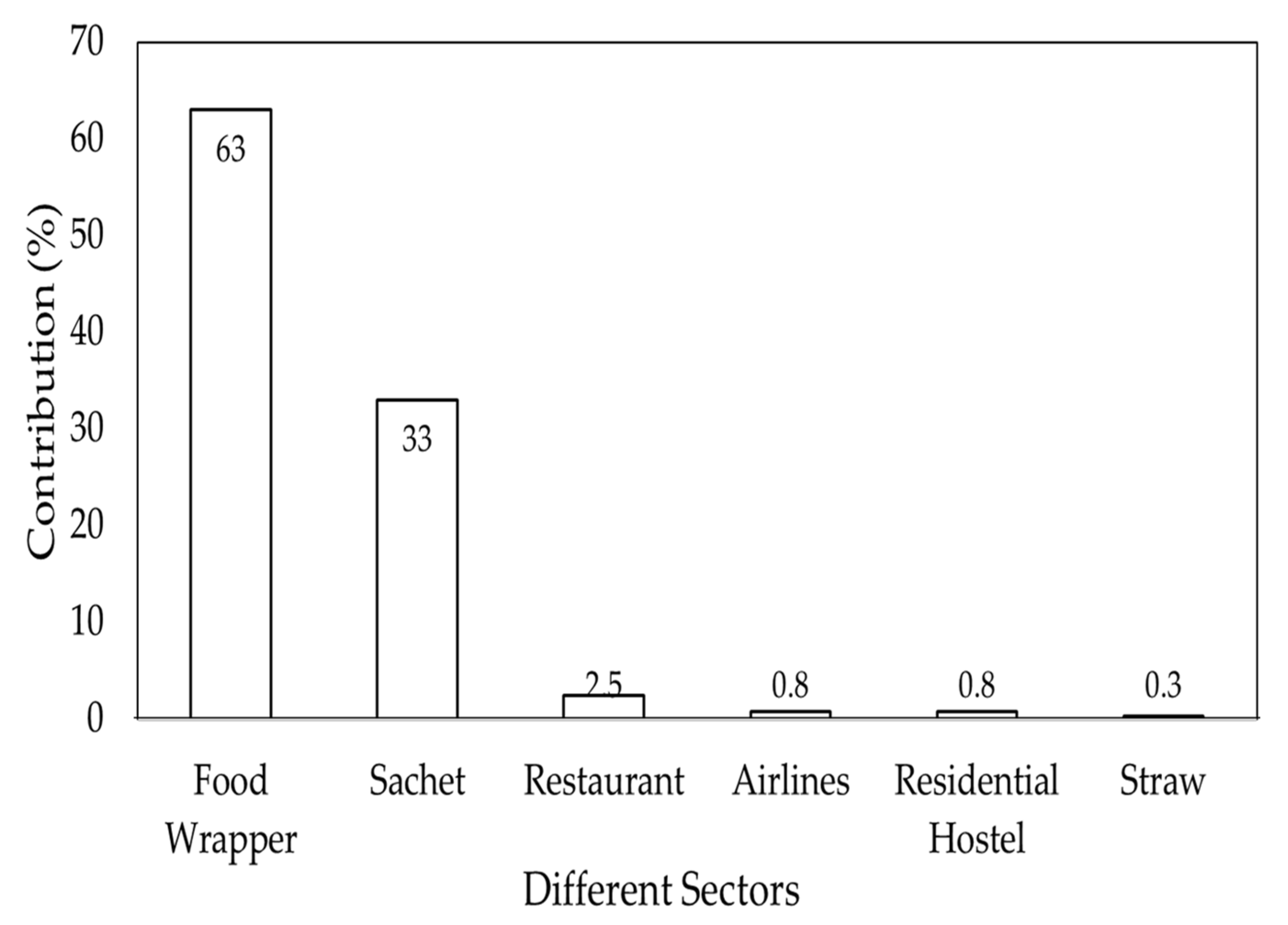
4. Microplastic-Related Threat in Bangladesh
5. Sustainable Sampling and Identification Techniques
5.1. Aqueous Phase Sampling
5.2. Sampling of Sediments
5.3. Sample Separation and Extraction
5.4. Sample Identification
6. Microplastic Removal and Degradation Techniques
6.1. Biotic Degradation of Microplastics
6.2. Bacterial Degradation
6.3. Degradation of Microplastics via Fungi
6.4. Removal of Microplastics by Algae and Macrophytes
6.5. Degradation of Microplastics by Periphytic Biofilms
6.6. Removal of Microplastics through Adsorption
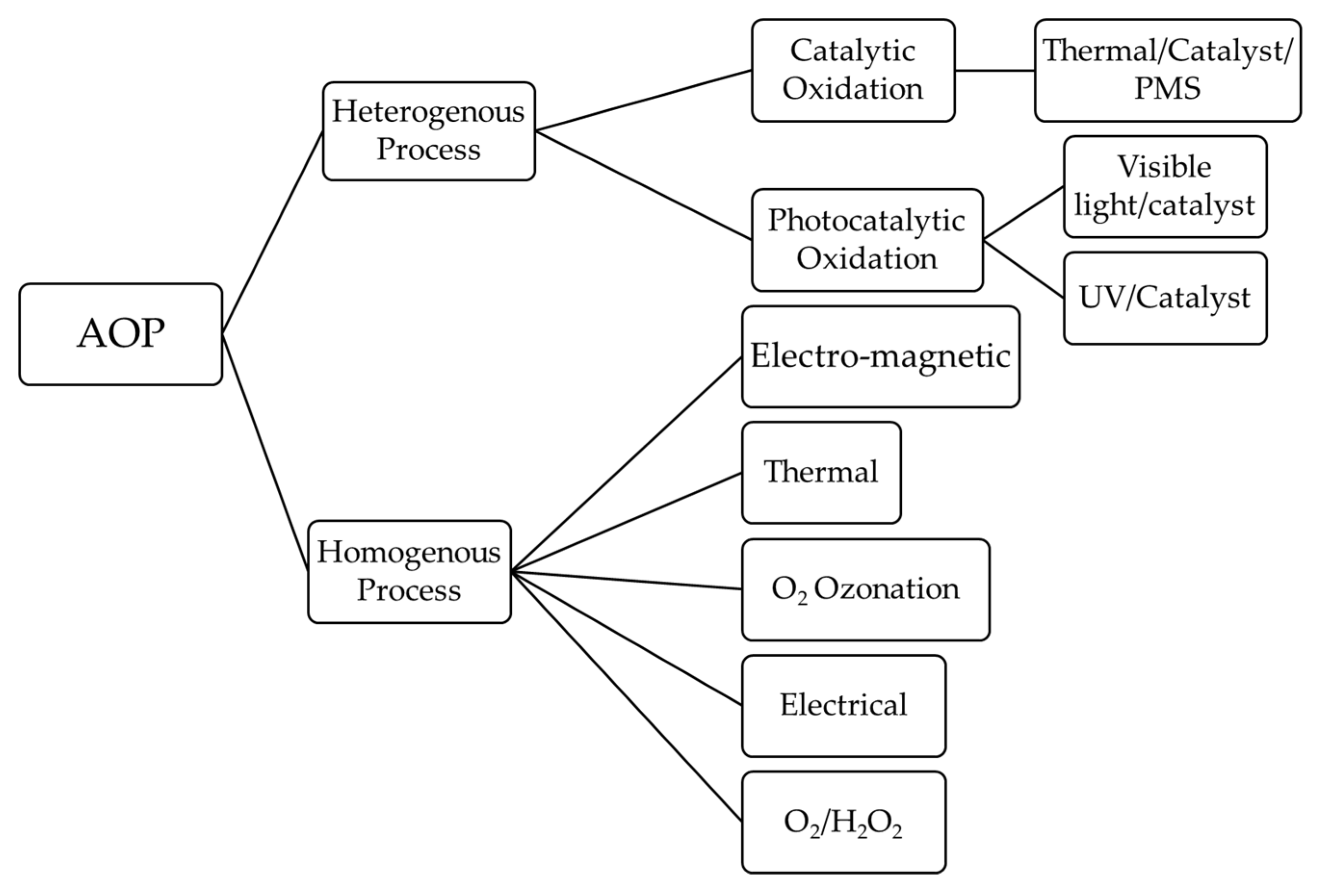
6.7. Degradation of Microplastics by Advanced Oxidation Process
6.7.1. Homogeneous AOPs
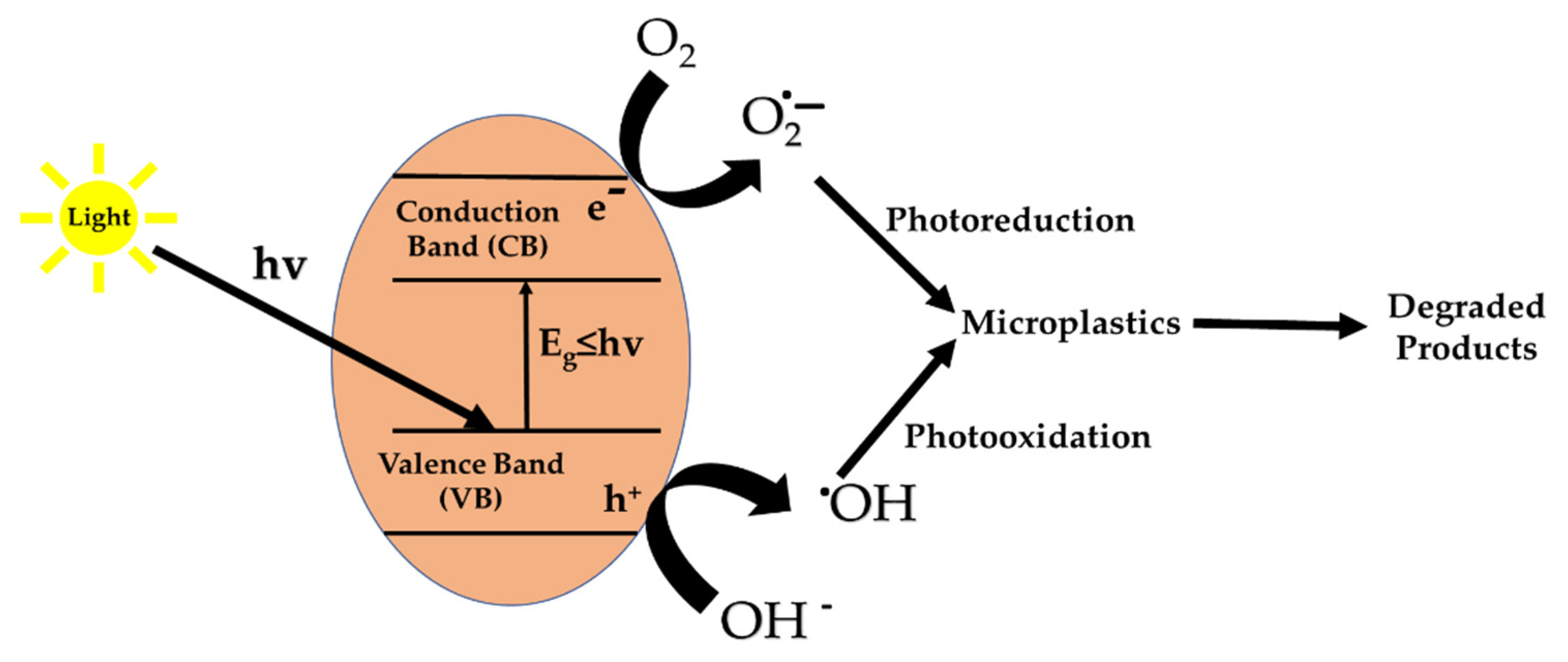
6.7.2. Heterogeneous AOPs
- (1)
- hvTiO2→h+VB + e−CB
- (2)
- h+VB + microplastics→CO2 + H2O
- (3)
- h+VB + H2O→∙OH + H+
- (4)
- ∙OH + microplastics→CO2 + H2O
- (5)
- O2 + e−CB → O∙2−
- (6)
- O∙2− + H2O→∙OOH + OH−
- (7)
- 2∙OOH→O2 + H2O2
- (8)
- H2O2 →2∙OH
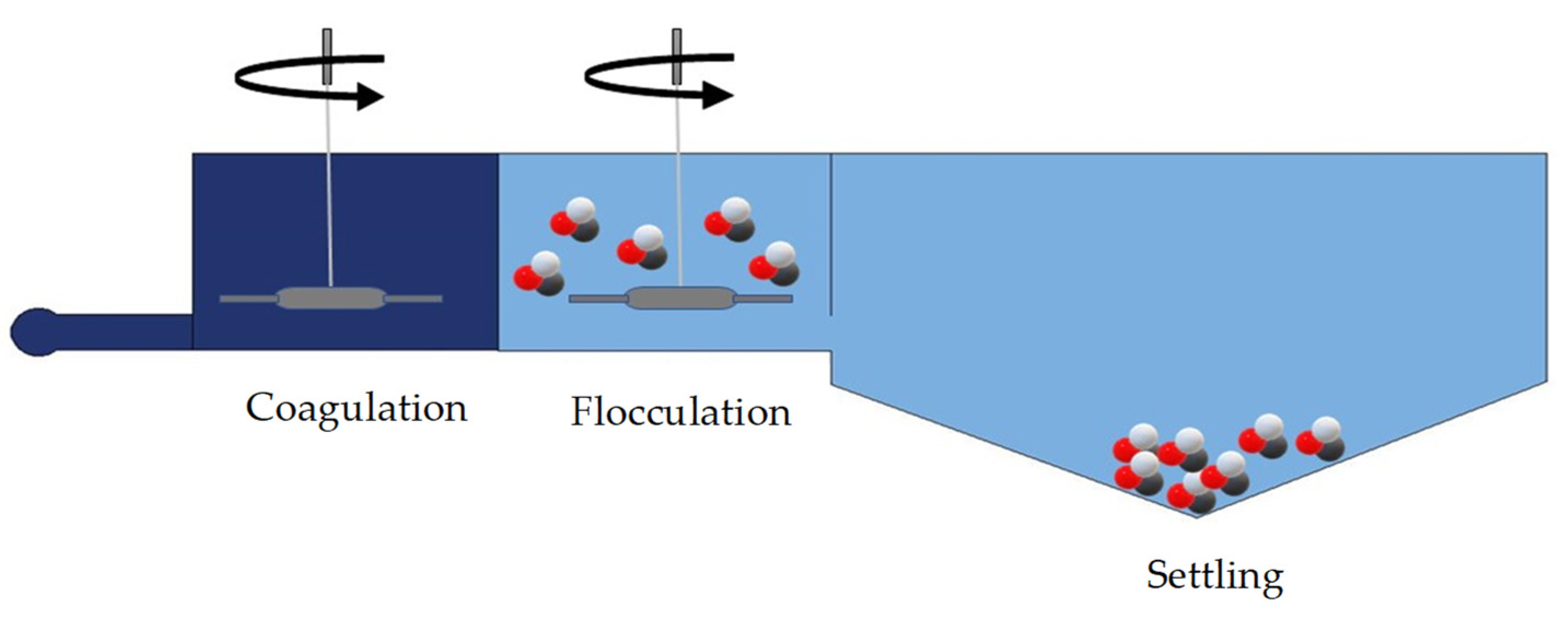
6.8. Microplastic Treatment by Coagulation and Flocculation
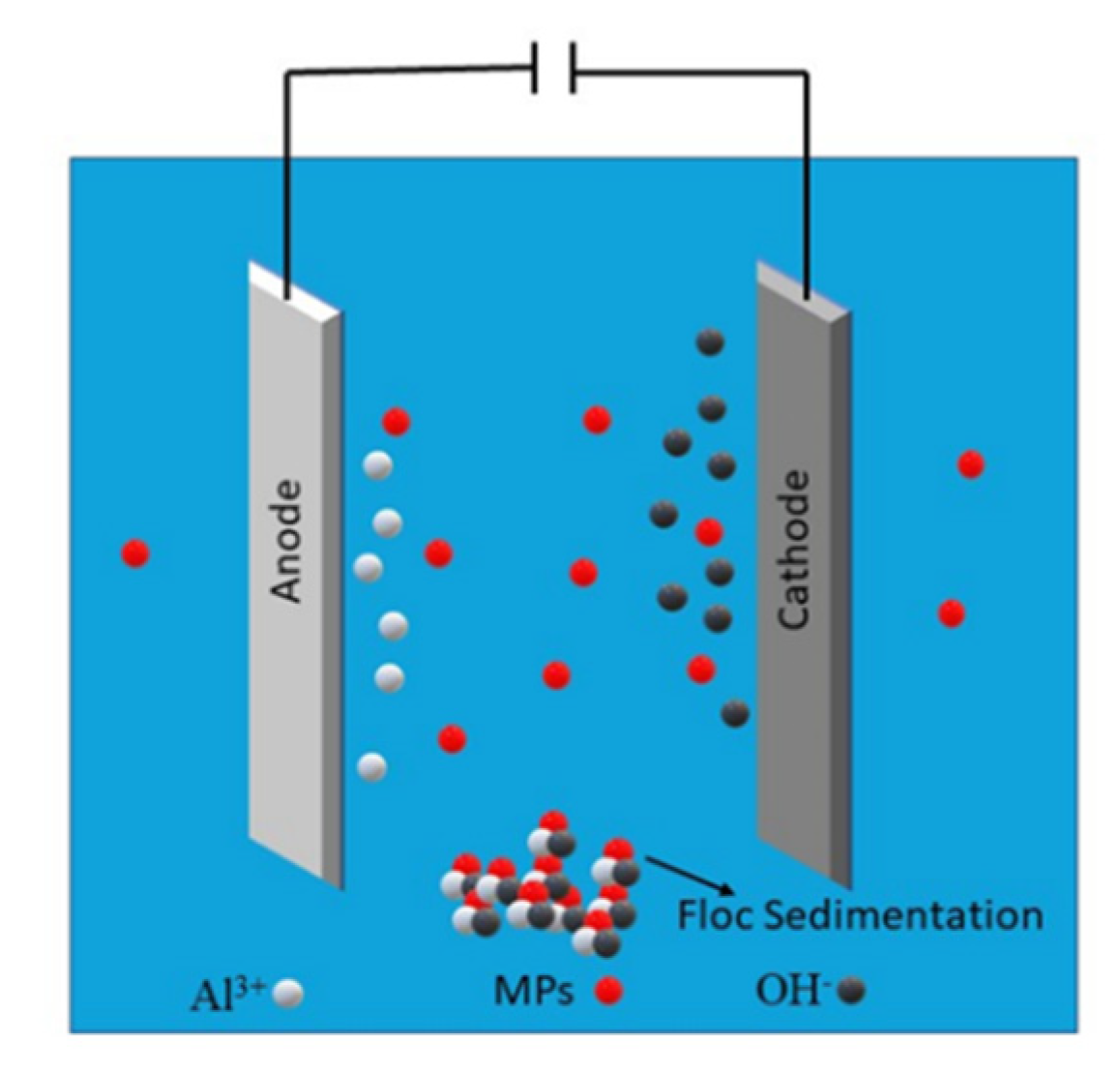
6.9. Electrocoagulation
6.10. Thermal Degradation/Plastic to Fuel
6.11. Recent Developments and Emerging Technologies for Microplastic Removal and Degradation
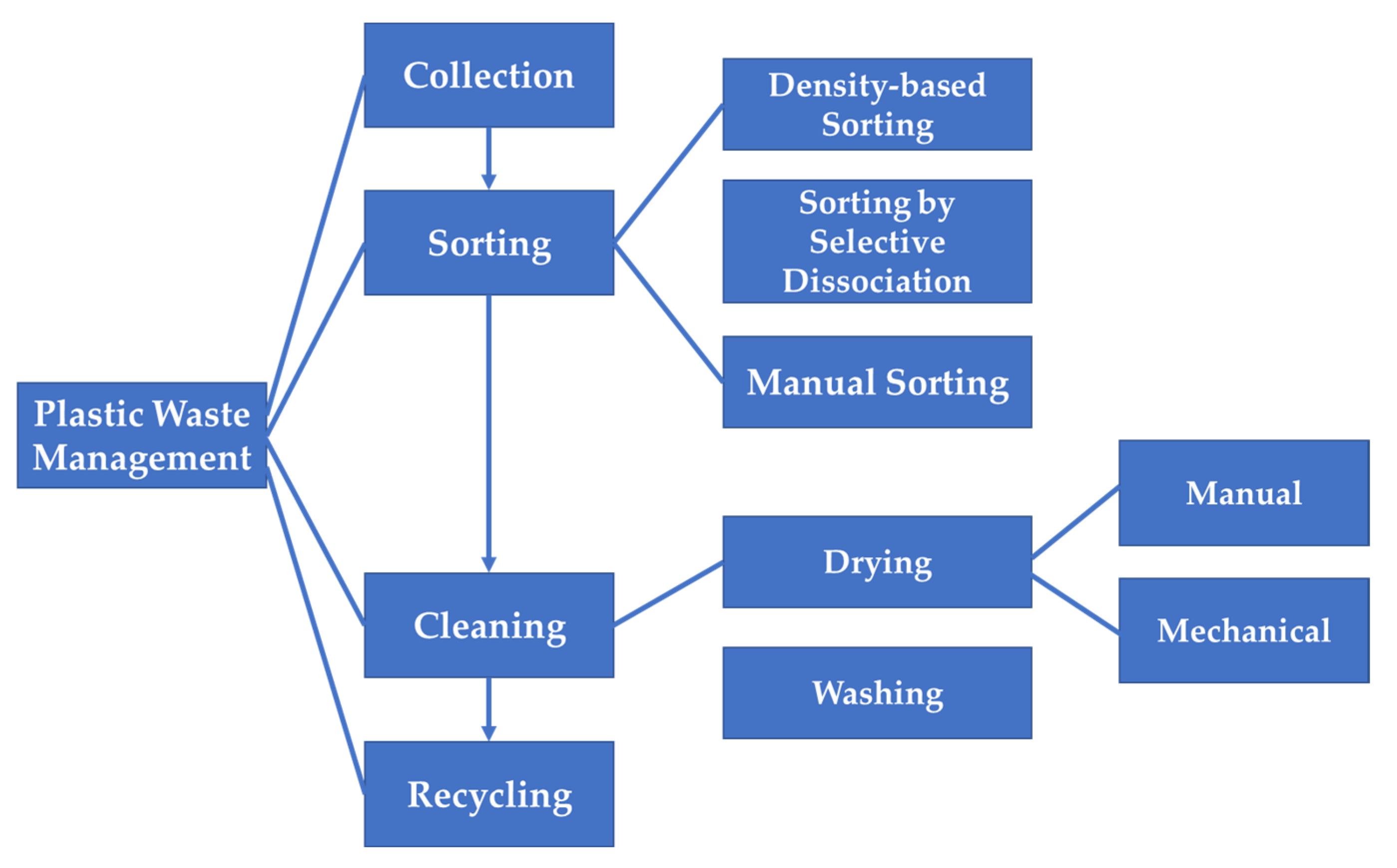
7. Management of Plastic Wastes for the Prevention of Secondary Microplastic Generation
8. Recommendations
- Create a national policy for effectively managing plastic trash and preventing it from entering nature.
- Improve municipal waste management systems to separate plastic waste at its source.
- Through media ads and government and non-government actions, raise end-users’ consciousness of plastic’s harmful consequences.
- Encourage entrepreneurs who recycle and reuse plastics to receive tax breaks and subsidies.
- Provide national recognition and funding for alternatives to plastic products, such as jute bags, paper cups, bamboo straws, etc.
- From the importation of raw materials through the sale of finished goods, charge a substantial tax on enterprises involved with plastic, causing people to avoid plastic items due to their high cost.
- Impose strict bans on the use of single-use plastic in river transportation such as launches, ships, and tourist spots near rivers or lakes to prevent the direct discharge of plastic into the water.
- Improve municipal wastewater treatment plant capability to gather more plastic.
- Microbeads and microplastic usage in personal care products should be legally prohibited in Bangladesh.
- Urgent legislation should be passed forbidding the manufacturing, use, and import of all single-use plastics.
- Research: Research is the first and most important step to minimizing aquatic microplastic pollution. The following outline will help to proceed with conducting research in a systematic way:
- (i)
- The government should create a strategy to control microplastics at the source and in the environment.
- (ii)
- Identification and characterization of microplastics in surface water and sediments of Bangladesh’s terrestrial and aquatic environments.
- (iii)
- Risk assessment of microplastics in terrestrial and aquatic biota based on concentrations, exposure times, forms, sizes, and tropic level transmission.
- (iv)
- Risk evaluation of plastic additives and watery chemical and biological contaminants on biota and tropic transfer.
- (v)
- Clarification of the hydrodynamic conditions (winds, currents, beach direction, etc.) that affect microplastic transport and movement, as well as their spatial and seasonal change.
- (vi)
- Characterization and diversity study of microbial biofilm communities colonizing plastics and microplastics.
- (vii)
- Standardization and harmonization of microplastic sampling, extraction, analysis, and identification to compare global findings.
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Rhodes, C.J. Plastic pollution and potential solutions. Sci. Prog. 2018, 101, 207–260. [Google Scholar] [CrossRef] [PubMed]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, M. Plastics materials: Introduction and historical development. In Brydson’s Plastics Materials; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1–18. [Google Scholar]
- Mourshed, M.; Masud, M.H.; Rashid, F.; Joardder, M.U.H. Towards the effective plastic waste management in Bangladesh: A review. Environ. Sci. Pollut. Res. 2017, 24, 27021–27046. [Google Scholar] [CrossRef] [PubMed]
- Hossain, S.; Rahman, M.A.; Ahmed Chowdhury, M.; Kumar Mohonta, S. Plastic pollution in Bangladesh: A review on current status emphasizing the impacts on environment and public health. Environ. Eng. Res. 2021, 26, 200535. [Google Scholar] [CrossRef]
- Chowdhury, G.W. Development and Use of Biological Measures to Assess the Quality of Lakes in Bangladesh; University of Cambridge: Cambridge, UK, 2012. [Google Scholar]
- Chowdhury, G.W.; Koldewey, H.J.; Duncan, E.; Napper, I.E.; Niloy, M.N.H.; Nelms, S.E.; Sarker, S.; Bhola, S.; Nishat, B. Plastic pollution in aquatic systems in Bangladesh: A review of current knowledge. Sci. Total Environ. 2021, 761, 143285. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.; Krauth, T.; Wagner, S. Export of plastic debris by rivers into the sea. Environ. Sci. Technol. 2017, 51, 12246–12253. [Google Scholar] [CrossRef]
- Arpia, A.A.; Chen, W.-H.; Ubando, A.T.; Naqvi, S.R.; Culaba, A.B. Microplastic degradation as a sustainable concurrent approach for producing biofuel and obliterating hazardous environmental effects: A state-of-the-art review. J. Hazard. Mater. 2021, 418, 126381. [Google Scholar] [CrossRef]
- Szymańska, M.; Obolewski, K. Microplastics as contaminants in freshwater environments: A multidisciplinary review. Ecohydrol. Hydrobiol. 2020, 20, 333–345. [Google Scholar] [CrossRef]
- Cole, M.; Lindeque, P.; Fileman, E.; Halsband, C.; Goodhead, R.; Moger, J.; Galloway, T.S. Microplastic Ingestion by Zooplankton. Environ. Sci. Technol. 2013, 47, 6646–6655. [Google Scholar] [CrossRef]
- Zheng, S.; Zhao, Y.; Liangwei, W.; Liang, J.; Liu, T.; Zhu, M.; Li, Q.; Sun, X. Characteristics of microplastics ingested by zooplankton from the Bohai Sea, China. Sci. Total Environ. 2020, 713, 136357. [Google Scholar] [CrossRef]
- Setälä, O.; Fleming-Lehtinen, V.; Lehtiniemi, M. Ingestion and transfer of microplastics in the planktonic food web. Environ. Pollut. 2014, 185, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Costa, E.; Piazza, V.; Lavorano, S.; Faimali, M.; Garaventa, F.; Gambardella, C. Trophic Transfer of Microplastics From Copepods to Jellyfish in the Marine Environment. Front. Environ. Sci. 2020, 8, 571732. [Google Scholar] [CrossRef]
- Farrell, P.; Nelson, K. Trophic level transfer of microplastic: Mytilus edulis (L.) to Carcinus maenas (L.). Environ. Pollut. 2013, 177, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Nelms, S.E.; Galloway, T.S.; Godley, B.J.; Jarvis, D.S.; Lindeque, P.K. Investigating microplastic trophic transfer in marine top predators. Environ. Pollut. 2018, 238, 999–1007. [Google Scholar] [CrossRef]
- Cecilia, E.; Harry, B. Origins and Biological Accumulation of Small Plastic Particles in Fur Seals from Macquarie Island. AMBIO A J. Hum. Environ. 2003, 32, 380–384. [Google Scholar]
- Batel, A.; Linti, F.; Scherer, M.; Erdinger, L.; Braunbeck, T. Transfer of benzo[a]pyrene from microplastics to Artemia nauplii and further to zebrafish via a trophic food web experiment: CYP1A induction and visual tracking of persistent organic pollutants. Environ. Toxicol. Chem. 2016, 35, 1656–1666. [Google Scholar] [CrossRef]
- Hitchcock, J.N. Microplastics can alter phytoplankton community composition. Sci. Total Environ. 2022, 819, 153074. [Google Scholar] [CrossRef]
- Leslie, H.A.; van Velzen, M.J.M.; Brandsma, S.H.; Vethaak, A.D.; Garcia-Vallejo, J.J.; Lamoree, M.H. Discovery and quantification of plastic particle pollution in human blood. Environ. Int. 2022, 163, 107199. [Google Scholar] [CrossRef]
- Ragusa, A.; Notarstefano, V.; Svelato, A.; Belloni, A.; Gioacchini, G.; Blondeel, C.; Zucchelli, E.; De Luca, C.; D’Avino, S.; Gulotta, A.; et al. Raman Microspectroscopy Detection and Characterisation of Microplastics in Human Breastmilk. Polymers 2022, 14, 2700. [Google Scholar] [CrossRef]
- Eriksen, M.; Liboiron, M.; Kiessling, T.; Charron, L.; Alling, A.; Lebreton, L.; Richards, H.; Roth, B.; Ory, N.C.; Hidalgo-Ruz, V. Microplastic sampling with the AVANI trawl compared to two neuston trawls in the Bay of Bengal and South Pacific. Environ. Pollut. 2018, 232, 430–439. [Google Scholar] [CrossRef]
- Hossain, M.S.; Sobhan, F.; Uddin, M.N.; Sharifuzzaman, S.; Chowdhury, S.R.; Sarker, S.; Chowdhury, M.S.N. Microplastics in fishes from the Northern Bay of Bengal. Sci. Total Environ. 2019, 690, 821–830. [Google Scholar] [CrossRef] [PubMed]
- Palmer, J.; Herat, S. Ecotoxicity of microplastic pollutants to marine organisms: A systematic review. Water Air Soil Pollut. 2021, 232, 1–21. [Google Scholar] [CrossRef]
- Rochman, C.M.; Kurobe, T.; Flores, I.; Teh, S.J. Early warning signs of endocrine disruption in adult fish from the ingestion of polyethylene with and without sorbed chemical pollutants from the marine environment. Sci. Total Environ. 2014, 493, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Murray, F.; Cowie, P.R. Plastic contamination in the decapod crustacean Nephrops norvegicus (Linnaeus, 1758). Mar. Pollut. Bull. 2011, 62, 1207–1217. [Google Scholar] [CrossRef] [PubMed]
- Pushan, Z.A.; Rahman, E.; Islam, N.; Aich, N. A critical review of the emerging research on the detection and assessment of microplastics pollution in the coastal, marine, and urban Bangladesh. Front. Environ. Sci. Eng. 2022, 16, 128. [Google Scholar] [CrossRef]
- Pervez, M.N.; Mishu, M.R.; Talukder, M.E.; Stylios, G.K.; Buonerba, A.; Hasan, S.W.; Cai, Y.; Zhao, Y.; Figoli, A.; Zarra, T.; et al. Electrospun nanofiber membranes for the control of micro/nanoplastics in the environment. Water Emerg. Contam. Nanoplastics 2022, 1, 10. [Google Scholar] [CrossRef]
- Klein, S.; Dimzon, I.K.; Eubeler, J.; Knepper, T.P. Analysis, Occurrence, and Degradation of Microplastics in the Aqueous Environment. In Freshwater Microplastics: Emerging Environmental Contaminants? Wagner, M., Lambert, S., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 51–67. [Google Scholar]
- Dris, R.; Gasperi, J.; Rocher, V.; Saad, M.; Renault, N.; Tassin, B. Microplastic contamination in an urban area: A case study in Greater Paris. Environ. Chem. 2015, 12, 592–599. [Google Scholar] [CrossRef]
- Dehaut, A.; Cassone, A.-L.; Frère, L.; Hermabessiere, L.; Himber, C.; Rinnert, E.; Rivière, G.; Lambert, C.; Soudant, P.; Huvet, A.; et al. Microplastics in seafood: Benchmark protocol for their extraction and characterization. Environ. Pollut. 2016, 215, 223–233. [Google Scholar] [CrossRef]
- Eriksen, M.; Mason, S.; Wilson, S.; Box, C.; Zellers, A.; Edwards, W.; Farley, H.; Amato, S. Microplastic pollution in the surface waters of the Laurentian Great Lakes. Mar. Pollut. Bull. 2013, 77, 177–182. [Google Scholar] [CrossRef]
- Huang, Y.; Qing, X.; Wang, W.; Han, G.; Wang, J. Mini-review on current studies of airborne microplastics: Analytical methods, occurrence, sources, fate and potential risk to human beings. TrAC Trends Anal. Chem. 2020, 125, 115821. [Google Scholar] [CrossRef]
- Möller, J.N.; Löder, M.G.J.; Laforsch, C. Finding Microplastics in Soils: A Review of Analytical Methods. Environ. Sci. Technol. 2020, 54, 2078–2090. [Google Scholar] [CrossRef] [PubMed]
- Rummel, C.D.; Jahnke, A.; Gorokhova, E.; Kühnel, D.; Schmitt-Jansen, M. Impacts of Biofilm Formation on the Fate and Potential Effects of Microplastic in the Aquatic Environment. Environ. Sci. Technol. Lett. 2017, 4, 258–267. [Google Scholar] [CrossRef]
- Zhang, M.; Zhao, Y.; Qin, X.; Jia, W.; Chai, L.; Huang, M.; Huang, Y. Microplastics from mulching film is a distinct habitat for bacteria in farmland soil. Sci. Total Environ. 2019, 688, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Basu, S.; Shetti, N.P.; Nadagouda, M.N.; Aminabhavi, T.M. Microplastics in the environment: Occurrence, perils, and eradication. Chem. Eng. J. 2021, 408, 127317. [Google Scholar] [CrossRef]
- Auta, H.S.; Emenike, C.U.; Jayanthi, B.; Fauziah, S.H. Growth kinetics and biodeterioration of polypropylene microplastics by Bacillus sp. and Rhodococcus sp. isolated from mangrove sediment. Mar. Pollut. Bull. 2018, 127, 15–21. [Google Scholar] [CrossRef]
- Tareen, A.; Saeed, S.; Iqbal, A.; Batool, R.; Jamil, N. Biodeterioration of Microplastics: A Promising Step towards Plastics Waste Management. Polymers 2022, 14, 2275. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Lu, Y.; Xiang, H.; Xu, Y.; Li, Y. Study on methanolytic depolymerization of PET with supercritical methanol for chemical recycling. Polym. Degrad. Stab. 2002, 75, 185–191. [Google Scholar] [CrossRef]
- Shimpi, N.; Borane, M.; Mishra, S.; Kadam, M. Biodegradation of polystyrene (PS)-poly(lactic acid) (PLA) nanocomposites using Pseudomonas aeruginosa. Macromol. Res. 2012, 20, 181–187. [Google Scholar] [CrossRef]
- Paço, A.; Duarte, K.; da Costa, J.P.; Santos, P.S.M.; Pereira, R.; Pereira, M.E.; Freitas, A.C.; Duarte, A.C.; Rocha-Santos, T.A.P. Biodegradation of polyethylene microplastics by the marine fungus Zalerion maritimum. Sci. Total Environ. 2017, 586, 10–15. [Google Scholar] [CrossRef]
- Sowmya, H.V.; Ramalingappa; Krishnappa, M.; Thippeswamy, B. Degradation of polyethylene by Trichoderma harzianum—SEM, FTIR, and NMR analyses. Environ. Monit. Assess. 2014, 186, 6577–6586. [Google Scholar] [CrossRef]
- Cosgrove, L.; McGeechan Paula, L.; Robson Geoff, D.; Handley Pauline, S. Fungal Communities Associated with Degradation of Polyester Polyurethane in Soil. Appl. Environ. Microbiol. 2007, 73, 5817–5824. [Google Scholar] [CrossRef] [PubMed]
- Ali, G.; Nisar, J.; Iqbal, M.; Shah, A.; Abbas, M.; Shah, M.R.; Rashid, U.; Bhatti, I.A.; Khan, R.A.; Shah, F. Thermo-catalytic decomposition of polystyrene waste: Comparative analysis using different kinetic models. Waste Manag. Res. 2019, 38, 202–212. [Google Scholar] [CrossRef] [PubMed]
- Chia, W.Y.; Ying Tang, D.Y.; Khoo, K.S.; Kay Lup, A.N.; Chew, K.W. Nature’s fight against plastic pollution: Algae for plastic biodegradation and bioplastics production. Environ. Sci. Ecotechnol. 2020, 4, 100065. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Dubey, A.; Pareek, A. Algal flora on degrading polythene waste. CIBTech J. Microbiol. 2014, 3, 43–47. [Google Scholar]
- Sarmah, P.; Rout, J. Algal colonization on polythene carry bags in a domestic solid waste dumping site of Silchar town in Assam. Phykos 2018, 48, 67–77. [Google Scholar]
- Chinaglia, S.; Tosin, M.; Degli-Innocenti, F. Biodegradation rate of biodegradable plastics at molecular level. Polym. Degrad. Stab. 2018, 147, 237–244. [Google Scholar] [CrossRef]
- Kumar, R.V.; Kanna, G.; Elumalai, S. Biodegradation of polyethylene by green photosynthetic microalgae. J. Bioremediat. Biodegrad. 2017, 8, 2. [Google Scholar]
- Kim, J.W.; Park, S.-B.; Tran, Q.-G.; Cho, D.-H.; Choi, D.-Y.; Lee, Y.J.; Kim, H.-S. Functional expression of polyethylene terephthalate-degrading enzyme (PETase) in green microalgae. Microb. Cell Factories 2020, 19, 1–9. [Google Scholar] [CrossRef]
- Faheem, M.; Shabbir, S.; Zhao, J.; G. Kerr, P.; Ali, S.; Sultana, N.; Jia, Z. Multifunctional periphytic biofilms: Polyethylene degradation and Cd2+ and Pb2+ bioremediation under high methane scenario. Int. J. Mol. Sci. 2020, 21, 5331. [Google Scholar] [CrossRef]
- Battin, T.J.; Besemer, K.; Bengtsson, M.M.; Romani, A.M.; Packmann, A.I. The ecology and biogeochemistry of stream biofilms. Nat. Rev. Microbiol. 2016, 14, 251–263. [Google Scholar] [CrossRef]
- Eckert, R.A.; Halvorson, H.M.; Kuehn, K.A.; Lamp, W.O. Macroinvertebrate community patterns in relation to leaf-associated periphyton under contrasting light and nutrient conditions in headwater streams. Freshw. Biol. 2020, 65, 1270–1287. [Google Scholar] [CrossRef]
- Ewe, S.M.; Gaiser, E.E.; Childers, D.L.; Iwaniec, D.; Rivera-Monroy, V.H.; Twilley, R.R. Spatial and temporal patterns of aboveground net primary productivity (ANPP) along two freshwater-estuarine transects in the Florida Coastal Everglades. Hydrobiologia 2006, 569, 459–474. [Google Scholar] [CrossRef]
- Martyniuk, N.; Modenutti, B.; Balseiro, E. Forest structure affects the stoichiometry of periphyton primary producers in mountain streams of northern Patagonia. Ecosystems 2016, 19, 1225–1239. [Google Scholar] [CrossRef]
- Miao, L.; Wang, P.; Hou, J.; Yao, Y.; Liu, Z.; Liu, S.; Li, T. Distinct community structure and microbial functions of biofilms colonizing microplastics. Sci. Total Environ. 2019, 650, 2395–2402. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Barragán, J.; Domínguez-Malfavón, L.; Vargas-Suárez, M.; González-Hernández, R.; Aguilar-Osorio, G.; Loza-Tavera, H. Biodegradative activities of selected environmental fungi on a polyester polyurethane varnish and polyether polyurethane foams. Appl. Environ. Microbiol. 2016, 82, 5225–5235. [Google Scholar] [CrossRef]
- Gómez-Méndez, L.D.; Moreno-Bayona, D.A.; Poutou-Piñales, R.A.; Salcedo-Reyes, J.C.; Pedroza-Rodríguez, A.M.; Vargas, A.; Bogoya, J.M. Biodeterioration of plasma pretreated LDPE sheets by Pleurotus ostreatus. PLoS ONE 2018, 13, e0203786. [Google Scholar] [CrossRef]
- Debroy, A.; George, N.; Mukherjee, G. Role of biofilms in the degradation of microplastics in aquatic environments. J. Chem. Technol. Biotechnol. 2021. [Google Scholar] [CrossRef]
- Islam, M.S.; Roy, H.; Afrose, S. Phosphoric acid surface modified Moringa oleifera leaves biochar for the sequestration of methyl orange from aqueous solution: Characterizations, isotherm, and kinetics analysis. Remediat. J. 2022, 32, 281–298. [Google Scholar] [CrossRef]
- Jahan, N.; Roy, H.; Reaz, A.H.; Arshi, S.; Rahman, E.; Firoz, S.H.; Islam, M.S. A comparative study on sorption behavior of graphene oxide and reduced graphene oxide towards methylene blue. Case Stud. Chem. Environ. Eng. 2022, 6, 100239. [Google Scholar] [CrossRef]
- Roy, H.; Islam, M.S.; Arifin, M.T.; Firoz, S.H. Chitosan-ZnO decorated Moringa oleifera seed biochar for sequestration of methylene blue: Isotherms, kinetics, and response surface analysis. Environ. Nanotechnol. Monit. Manag. 2022, 18, 100752. [Google Scholar] [CrossRef]
- Roy, H.; Prantika, T.R.; Riyad, M.; Paul, S.; Islam, M.S. Synthesis, characterizations, and RSM analysis of Citrus macroptera peel derived biochar for textile dye treatment. S. Afr. J. Chem. Eng. 2022, 41, 129–139. [Google Scholar] [CrossRef]
- Roy, H.; Rahman, M.M.; Tarek, Y.A.; Firoz, S.H. Encapsulation of Industrial Cationic Pollutants from Aqueous Solution by Nanocrystalline Cellulose and It’s Modified Forms. In Proceedings of the International Exchange and Innovation Conference on Engineering & Sciences (IEICES) 2021 at: Interdisciplinary Graduate School of Engineering Sciences (IGSES), Kyushu University, Fukoka, Japan, 2 December 2021. [Google Scholar]
- Roy, H.; Shakil, R.; Tarek, Y.A.; Firoz, S.H. Study of the Removal of Basic Blue-41 from Simulated Wastewater By Activated Carbon Prepared from Discarded Jute Fibre. ECS Trans. 2022, 107, 8407. [Google Scholar] [CrossRef]
- Wang, Z.; Sedighi, M.; Lea-Langton, A. Filtration of microplastic spheres by biochar: Removal efficiency and immobilisation mechanisms. Water Res. 2020, 184, 116165. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Sun, C.; Huang, Q.-X.; Chi, Y.; Yan, J.-H. Adsorption and thermal degradation of microplastics from aqueous solutions by Mg/Zn modified magnetic biochars. J. Hazard. Mater. 2021, 419, 126486. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Khandelwal, N.; Ganie, Z.A.; Tiwari, E.; Darbha, G.K. Eco-friendly magnetic biochar: An effective trap for nanoplastics of varying surface functionality and size in the aqueous environment. Chem. Eng. J. 2021, 418, 129405. [Google Scholar] [CrossRef]
- Siipola, V.; Pflugmacher, S.; Romar, H.; Wendling, L.; Koukkari, P. Low-cost biochar adsorbents for water purification including microplastics removal. Appl. Sci. 2020, 10, 788. [Google Scholar] [CrossRef]
- Akter, S.; Islam, M.S.; Kabir, M.H.; Shaikh, M.A.A.; Gafur, M.A. UV/TiO2 photodegradation of metronidazole, ciprofloxacin and sulfamethoxazole in aqueous solution: An optimization and kinetic study. Arab. J. Chem. 2022, 15, 103900. [Google Scholar] [CrossRef]
- Al-Mamun, M.R.; Kader, S.; Islam, M.S. Solar-TiO2 immobilized photocatalytic reactors performance assessment in the degradation of methyl orange dye in aqueous solution. Environ. Nanotechnol. Monit. Manag. 2021, 16, 100514. [Google Scholar] [CrossRef]
- Al-Mamun, M.R.; Kader, S.; Islam, M.S.; Khan, M.Z.H. Photocatalytic activity improvement and application of UV-TiO2 photocatalysis in textile wastewater treatment: A review. J. Environ. Chem. Eng. 2019, 7, 103248. [Google Scholar] [CrossRef]
- Al-Mamun, M.R.; Karim, M.N.; Nitun, N.A.; Kader, S.; Islam, M.S.; Khan, M.Z.H. Photocatalytic performance assessment of GO and Ag co-synthesized TiO2 nanocomposite for the removal of methyl orange dye under solar irradiation. Environ. Technol. Innov. 2021, 22, 101537. [Google Scholar] [CrossRef]
- Hasan Khan Neon, M.; Islam, M.S. MoO3 and Ag co-synthesized TiO2 as a novel heterogeneous photocatalyst with enhanced visible-light-driven photocatalytic activity for methyl orange dye degradation. Environ. Nanotechnol. Monit. Manag. 2019, 12, 100244. [Google Scholar] [CrossRef]
- Rashid Al-Mamun, M.; Hossain, K.T.; Mondal, S.; Afroza Khatun, M.; Shahinoor Islam, M.; Zaved Hossain Khan, D.M. Synthesis, characterization, and photocatalytic performance of methyl orange in aqueous TiO2 suspension under UV and solar light irradiation. S. Afr. J. Chem. Eng. 2022, 40, 113–125. [Google Scholar] [CrossRef]
- Rashid Al-Mamun, M.; Shofikul Islam, M.; Rasel Hossain, M.; Kader, S.; Shahinoor Islam, M.; Zaved Hossain Khan, M. A novel and highly efficient Ag and GO co-synthesized ZnO nano photocatalyst for methylene blue dye degradation under UV irradiation. Environ. Nanotechnol. Monit. Manag. 2021, 16, 100495. [Google Scholar] [CrossRef]
- Shuchi, S.B.; Suhan, M.B.K.; Humayun, S.B.; Haque, M.E.; Islam, M.S. Heat-activated potassium persulfate treatment of Sudan Black B dye: Degradation kinetic and thermodynamic studies. J. Water Process Eng. 2021, 39, 101690. [Google Scholar] [CrossRef]
- Suhan, M.B.K.; Shuchi, S.B.; Anis, A.; Haque, Z.; Islam, M.S. Comparative degradation study of remazol black B dye using electro-coagulation and electro-Fenton process: Kinetics and cost analysis. Environ. Nanotechnol. Monit. Manag. 2020, 14, 100335. [Google Scholar] [CrossRef]
- Akter, S.; Suhan, M.B.K.; Islam, M.S. Recent advances and perspective of electrocoagulation in the treatment of wastewater: A review. Environ. Nanotechnol. Monit. Manag. 2022, 17, 100643. [Google Scholar] [CrossRef]
- Can-Güven, E. Advanced treatment of dye manufacturing wastewater by electrocoagulation and electro-Fenton processes: Effect on COD fractions, energy consumption, and sludge analysis. J. Environ. Manag. 2021, 300, 113784. [Google Scholar] [CrossRef]
- Suhan, M.B.K.; Mahtab, S.M.T.; Aziz, W.; Akter, S.; Islam, M.S. Sudan black B dye degradation in aqueous solution by Fenton oxidation process: Kinetics and cost analysis. Case Stud. Chem. Environ. Eng. 2021, 4, 100126. [Google Scholar] [CrossRef]
- Pi, Y.; Li, X.; Xia, Q.; Wu, J.; Li, Y.; Xiao, J.; Li, Z. Adsorptive and photocatalytic removal of Persistent Organic Pollutants (POPs) in water by metal-organic frameworks (MOFs). Chem. Eng. J. 2018, 337, 351–371. [Google Scholar] [CrossRef]
- Nakata, K.; Fujishima, A. TiO2 photocatalysis: Design and applications. J. Photochem. Photobiol. C Photochem. Rev. 2012, 13, 169–189. [Google Scholar] [CrossRef]
- Tofa, T.S.; Kunjali, K.L.; Paul, S.; Dutta, J. Visible light photocatalytic degradation of microplastic residues with zinc oxide nanorods. Environ. Chem. Lett. 2019, 17, 1341–1346. [Google Scholar] [CrossRef]
- Xu, Q.; Huang, Q.-S.; Luo, T.-Y.; Wu, R.-L.; Wei, W.; Ni, B.-J. Coagulation removal and photocatalytic degradation of microplastics in urban waters. Chem. Eng. J. 2021, 416, 129123. [Google Scholar] [CrossRef]
- Tofa, T.S.; Ye, F.; Kunjali, K.L.; Dutta, J. Enhanced Visible Light Photodegradation of Microplastic Fragments with Plasmonic Platinum/Zinc Oxide Nanorod Photocatalysts. Catalysts 2019, 9, 819. [Google Scholar] [CrossRef]
- Nabi, I.; Bacha, A.-U.-R.; Li, K.; Cheng, H.; Wang, T.; Liu, Y.; Ajmal, S.; Yang, Y.; Feng, Y.; Zhang, L. Complete Photocatalytic Mineralization of Microplastic on TiO2 Nanoparticle Film. iScience 2020, 23, 101326. [Google Scholar] [CrossRef]
- Qi, K.; Cheng, B.; Yu, J.; Ho, W. Review on the improvement of the photocatalytic and antibacterial activities of ZnO. J. Alloy. Compd. 2017, 727, 792–820. [Google Scholar] [CrossRef]
- Uheida, A.; Mejía, H.G.; Abdel-Rehim, M.; Hamd, W.; Dutta, J. Visible light photocatalytic degradation of polypropylene microplastics in a continuous water flow system. J. Hazard. Mater. 2021, 406, 124299. [Google Scholar] [CrossRef]
- Perren, W.; Wojtasik, A.; Cai, Q. Removal of Microbeads from Wastewater Using Electrocoagulation. ACS Omega 2018, 3, 3357–3364. [Google Scholar] [CrossRef]
- Moussa, D.T.; El-Naas, M.H.; Nasser, M.; Al-Marri, M.J. A comprehensive review of electrocoagulation for water treatment: Potentials and challenges. J. Environ. Manag. 2017, 186, 24–41. [Google Scholar] [CrossRef]
- Elkhatib, D.; Oyanedel-Craver, V.; Carissimi, E. Electrocoagulation applied for the removal of microplastics from wastewater treatment facilities. Sep. Purif. Technol. 2021, 276, 118877. [Google Scholar] [CrossRef]
- Shen, M.; Zhang, Y.; Almatrafi, E.; Hu, T.; Zhou, C.; Song, B.; Zeng, Z.; Zeng, G. Efficient removal of microplastics from wastewater by an electrocoagulation process. Chem. Eng. J. 2022, 428, 131161. [Google Scholar] [CrossRef]
- Xu, R.; Yang, Z.; Niu, Y.; Xu, D.; Wang, J.; Han, J.; Wang, H. Removal of microplastics and attached heavy metals from secondary effluent of wastewater treatment plant using interpenetrating bipolar plate electrocoagulation. Sep. Purif. Technol. 2022, 290, 120905. [Google Scholar] [CrossRef]
- Akarsu, C.; Deniz, F. Electrocoagulation/Electroflotation Process for Removal of Organics and Microplastics in Laundry Wastewater. CLEAN–Soil Air Water 2021, 49, 2000146. [Google Scholar] [CrossRef]
- Akarsu, C.; Kumbur, H.; Kideys, A.E. Removal of microplastics from wastewater through electrocoagulation-electroflotation and membrane filtration processes. Water Sci. Technol. A J. Int. Assoc. Water Pollut. Res. 2021, 84, 1648–1662. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; Tian, W.; Yang, Y.; Nie, G.; Zhou, P.; Wang, Y.; Duan, X.; Wang, S. Microplastics remediation in aqueous systems: Strategies and technologies. Water Res. 2021, 198, 117144. [Google Scholar]
- Hou, L.; Kumar, D.; Yoo, C.G.; Gitsov, I.; Majumder, E.L.W. Conversion and removal strategies for microplastics in wastewater treatment plants and landfills. Chem. Eng. J. 2021, 406, 126715. [Google Scholar] [CrossRef]
- Ciuffi, B.; Chiaramonti, D.; Rizzo, A.M.; Frediani, M.; Rosi, L. A Critical Review of SCWG in the Context of Available Gasification Technologies for Plastic Waste. Appl. Sci. 2020, 10, 6307. [Google Scholar] [CrossRef]
- Bai, B.; Liu, Y.; Zhang, H.; Zhou, F.; Han, X.; Wang, Q.; Jin, H. Experimental investigation on gasification characteristics of polyethylene terephthalate (PET) microplastics in supercritical water. Fuel 2020, 262, 116630. [Google Scholar] [CrossRef]
- Bai, B.; Liu, Y.; Meng, X.; Liu, C.; Zhang, H.; Zhang, W.; Jin, H. Experimental investigation on gasification characteristics of polycarbonate (PC) microplastics in supercritical water. J. Energy Inst. 2020, 93, 624–633. [Google Scholar] [CrossRef]
- Bai, B.; Jin, H.; Zhu, S.; Wu, P.; Fan, C.; Sun, J. Experimental investigation on in-situ hydrogenation induced gasification characteristics of acrylonitrile butadiene styrene (ABS) microplastics in supercritical water. Fuel Process. Technol. 2019, 192, 170–178. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, Y.; Liu, Z.; Su, J.; Fang, C.; Xu, D.; Song, W.; Wang, S. Influences of operating parameters on liquefaction performances of Tetra Pak in sub-/supercritical water. J. Environ. Manag. 2019, 237, 545–551. [Google Scholar] [CrossRef]
- Tavares, R.; Ramos, A.; Rouboa, A. Microplastics thermal treatment by polyethylene terephthalate-biomass gasification. Energy Convers. Manag. 2018, 162, 118–131. [Google Scholar] [CrossRef]
- Talvitie, J.; Mikola, A.; Koistinen, A.; Setälä, O. Solutions to microplastic pollution–Removal of microplastics from wastewater effluent with advanced wastewater treatment technologies. Water Res. 2017, 123, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Bayo, J.; López-Castellanos, J.; Olmos, S. Membrane bioreactor and rapid sand filtration for the removal of microplastics in an urban wastewater treatment plant. Mar. Pollut. Bull. 2020, 156, 111211. [Google Scholar] [CrossRef] [PubMed]
- Lares, M.; Ncibi, M.C.; Sillanpää, M.; Sillanpää, M. Occurrence, identification and removal of microplastic particles and fibers in conventional activated sludge process and advanced MBR technology. Water Res. 2018, 133, 236–246. [Google Scholar] [CrossRef]
- Baresel, C.; Harding, M.; Fång, J. Ultrafiltration/Granulated Active Carbon-Biofilter: Efficient Removal of a Broad Range of Micropollutants. Appl. Sci. 2019, 9, 710. [Google Scholar] [CrossRef]
- Sol, D.; Laca, A.; Laca, A.; Díaz, M. Approaching the environmental problem of microplastics: Importance of WWTP treatments. Sci. Total Environ. 2020, 740, 140016. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiang, H.; Bian, K.; Wang, H.; Wang, C. A critical review of control and removal strategies for microplastics from aquatic environments. J. Environ. Chem. Eng. 2021, 9, 105463. [Google Scholar] [CrossRef]
- Liu, F.; Nord, N.B.; Bester, K.; Vollertsen, J. Microplastics Removal from Treated Wastewater by a Biofilter. Water 2020, 12, 1085. [Google Scholar] [CrossRef]
- Grbic, J.; Nguyen, B.; Guo, E.; You, J.B.; Sinton, D.; Rochman, C.M. Magnetic Extraction of Microplastics from Environmental Samples. Environ. Sci. Technol. Lett. 2019, 6, 68–72. [Google Scholar] [CrossRef]
- Felsing, S.; Kochleus, C.; Buchinger, S.; Brennholt, N.; Stock, F.; Reifferscheid, G. A new approach in separating microplastics from environmental samples based on their electrostatic behavior. Environ. Pollut. 2018, 234, 20–28. [Google Scholar] [CrossRef]
- Tang, K.H.D.; Lock, S.S.M.; Yap, P.-S.; Cheah, K.W.; Chan, Y.H.; Yiin, C.L.; Ku, A.Z.E.; Loy, A.C.M.; Chin, B.L.F.; Chai, Y.H. Immobilized enzyme/microorganism complexes for degradation of microplastics: A review of recent advances, feasibility and future prospects. Sci. Total Environ. 2022, 832, 154868. [Google Scholar] [CrossRef] [PubMed]
- Olajire, A.A.; Mohammed, A.A. Bio-directed synthesis of gold nanoparticles using Ananas comosus aqueous leaf extract and their photocatalytic activity for LDPE degradation. Adv. Powder Technol. 2021, 32, 600–610. [Google Scholar] [CrossRef]
- Tiwari, E.; Singh, N.; Khandelwal, N.; Monikh, F.A.; Darbha, G.K. Application of Zn/Al layered double hydroxides for the removal of nano-scale plastic debris from aqueous systems. J. Hazard. Mater. 2020, 397, 122769. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.-Q.; Patel, M.K. Plastics Derived from Biological Sources: Present and Future: A Technical and Environmental Review. Chem. Rev. 2012, 112, 2082–2099. [Google Scholar] [CrossRef]
- Tian, C.; Lv, J.; Zhang, W.; Wang, H.; Chao, J.; Chai, L.; Lin, Z. Accelerated Degradation of Microplastics at the Liquid Interface of Ice Crystals in Frozen Aqueous Solutions. Angew. Chem. Int. Ed. 2022, 61, e202206947. [Google Scholar] [CrossRef]
- Herbort, A.F.; Sturm, M.T.; Fiedler, S.; Abkai, G.; Schuhen, K. Alkoxy-silyl Induced Agglomeration: A New Approach for the Sustainable Removal of Microplastic from Aquatic Systems. J. Polym. Environ. 2018, 26, 4258–4270. [Google Scholar] [CrossRef]
- Sturm, M.T.; Horn, H.; Schuhen, K. Removal of Microplastics from Waters through Agglomeration-Fixation Using Organosilanes—Effects of Polymer Types, Water Composition and Temperature. Water 2021, 13, 675. [Google Scholar] [CrossRef]
- Wang, Y.; Su, G.; Li, J.; Guo, Q.; Miao, Y.; Zhang, X. Robust, Healable, Self-Locomotive Integrated Robots Enabled by Noncovalent Assembled Gradient Nanostructure. Nano Lett. 2022, 22, 5409–5419. [Google Scholar] [CrossRef]
- Shen, M.; Huang, W.; Chen, M.; Song, B.; Zeng, G.; Zhang, Y. (Micro)plastic crisis: Un-ignorable contribution to global greenhouse gas emissions and climate change. J. Clean. Prod. 2020, 254, 120138. [Google Scholar] [CrossRef]
- Medina, M. Informal Recycling and Collection of Solid Wastes in Developing Countries: Issues and Opportunities; UN University. Institute of Advanced Studies: Medina, Martín, 1997. [Google Scholar]
- Roy, H.; Alam, S.R.; Bin-Masud, R.; Prantika, T.R.; Pervez, M.N.; Islam, M.S.; Naddeo, V. A Review on Characteristics, Techniques, and Waste-to-Energy Aspects of Municipal Solid Waste Management: Bangladesh Perspective. Sustainability 2022, 14, 10265. [Google Scholar] [CrossRef]
- Ashikuzzaman, M.; Howlader, M. Sustainable Solid Waste Management in Bangladesh: Issues and Challenges. In Sustainable Waste Management Challenges in Developing Countries; IGI Global: Pennsylvania, PA, USA, 2019; pp. 35–55. [Google Scholar]
- Turner, S.; Seymour, R.; Dombroski, J. Modern Polyesters: Chemistry and Technology of Polyesters and Copolyesters; Wiley: Chichester, UK, 2004; pp. 267–292. [Google Scholar]
- Tall, S. Recycling of Mixed Plastic Waste—Is Separation Worthwhile? Department of Polymer Technology, Royal Institute of Technology: Stockholm, Sweden, 2000. [Google Scholar]
- Ahmed, R.; van de Klundert, A.; Lardinois, I. Rubber Waste: Options for Small-Scale Resource Recovery Urban Solid Waste Series 3; WASTE: Gouda, Netherlands, 1996. [Google Scholar]
- Merrington, A. 11-Recycling of Plastics. In Applied Plastics Engineering Handbook; Kutz, M., Ed.; William Andrew Publishing: Oxford, UK, 2011; pp. 177–192. [Google Scholar]
- Solis, M.; Silveira, S. Technologies for chemical recycling of household plastics–A technical review and TRL assessment. Waste Manag. 2020, 105, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Pandey, S. Pyrolysis and Catalytic Cracking of Municipal Plastic Waste for Recovery of Gasoline Range Hydrocarbons. Ph.D. Thesis, National Institute of Technology, Rourkela, India, 2007. [Google Scholar]
- Chen, J.; Wu, J.; Sherrell, P.C.; Chen, J.; Wang, H.; Zhang, W.-X.; Yang, J. How to Build a Microplastics-Free Environment: Strategies for Microplastics Degradation and Plastics Recycling. Adv. Sci. 2022, 9, 2103764. [Google Scholar] [CrossRef] [PubMed]
- Cointreau, S.J.; Gunnerson, C.; Huls, J.M.; Seldman, N.N. Recycling from Municipal Refuse: A State-of-the-Art Review and Annotated Bibliography; World Bank: Washington, DC, USA, 1984. [Google Scholar]
- Xu, G.; Liu, K.; Xu, B.; Yao, Y.; Li, W.; Yan, J.; Zhang, A. Confined microenvironments from thermoresponsive dendronized polymers. Macromol. Rapid Commun. 2020, 41, 2000325. [Google Scholar] [CrossRef] [PubMed]

| Strain | Biodegradation Condition | Biodegradation Rate (%) | Ref. | ||
|---|---|---|---|---|---|
| Media | Duration | Temperature (°C) | |||
| Zalerion maritimum | Minimum growth media with 0.130 g of polymer | 0.94 months | 25 | 43 | [42] |
| Trichoderma harzianum | Mineral salt medium | 3 months | - | 40 | [43] |
| Aspergillus tubingensis | Mineral salt medium | 0.75 months | 37 | 90 | [44] |
| Phanerochaete chyrosporium | Soil buried (soil mixed with municipal sewage sludge) | 6 months | - | - | [45] |
| Characteristics of Adsorbent | Process Parameters | Removed Microplastics | Efficiency of the Process | Involved Mechanisms | Ref. |
|---|---|---|---|---|---|
| Biochar consisting of corn straw and hardwood | pH = 7.56 Filtration column for biochar Hybrid sand | Polystyrene microplastic spheres (diameter = 10 μm) | Greater than 95% | Sticking, entangling, trapping | [67] |
| Magnetic biochar modified by Mg/Zn | Temperature = 25 °C | Microplastic spheres of polystyrene (diameter = 1 μm) | Mg-MBC-98% Zn-MBC-99.46% MBC-94.80% | Chemical bonding, electrostatic interaction | [68] |
| Biochar modified by iron and pyrolyzed at 550 °C and 850 °C | pH = 5.5 Temperature = 25 °C | Nano-plastics (diameter = 30 nm and 1000 nm) | Around 100% | Surface complexation, electrostatic attraction | [69] |
| Pine and spruce bark biochar pyrolyzed at 475 °C and steam-activated at 800 °C | Temperature = 25 °C | Spherical, cylindrical and fleece shirt fibers polyethylene microbeads (diameter = 10 μm) | Around 100% in the case of cylindrical polyethylene pieces and fleece fibers. | Adherence between biochar particles. | [70] |
| Microplastics Source | Optimal Condition | Electrode | Efficiency | Ref. |
|---|---|---|---|---|
| Polyethylene microbead | pH = 7.5 NaCl concentration = 0–2 g/L Current density = 11 A/m2 Time = 30 min | Al | 99.24% | [91] |
| Real wastewater | pH = 4 Current density = 2.88 mA/cm2 | Al | 96.5% | [93] |
| Polyester microplastic | pH = 4 Current density = 2.88 mA/cm2 | Al | 98.5% | |
| Polyethylene | Electrolyte Concentration = 0.05 M pH = 7.2 | Al | 93.2% | [94] |
| Polymethylmethacrylate | 91.7% | |||
| Cellulose acetate for cigarette butt | 98.2% | |||
| PP from disposable surgical masks | 98.4% | |||
| Heavy metal and microplastics | pH = 6 Current density = 12 mA/cm2 Time = 20 min | Al | 97.5% | [94] |
| Laundry wastewater | pH = 9 Current density = 2.16 A Time = 60 min | Fe–Al | 98% | [96] |
| PE | pH = 7 Current density = 20 A/m2 Time = 10 min | Al–Fe | 100% | [97] |
| Microplastics Source | Working Mechanism | Time | Temperature | Performance | Ref. |
|---|---|---|---|---|---|
| PET microplastics | Gasification in supercritical water, artificial seawater | 10 min | 800 °C, 23 MPa | 98% Carbon conversion | [101] |
| Polycarbonate microplastics | Gasification in supercritical water | 60 min | 800 °C | 50.8% Carbon conversion | [102] |
| Acrylonitrile butadiene styrene (ABS) microplastics | Hydrogenation induced gasification in supercritical water | 60 min | 800 °C, 23 MPa | 97% Carbon conversion | [103] |
| Tetra Pak | Hydro-thermal liquefaction | 30 min | 360 °C, 22 MPa | Bio-oil yields 35.55% | [104] |
| 420 °C, 20 MPa | Energy recovery efficiency 46.49% | ||||
| PET + biomass | Gasification | 900 °C | 9.2 MJ/Nm3 lower heating value, 63–66% H2 molar fraction | [105] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahmud, A.; Wasif, M.M.; Roy, H.; Mehnaz, F.; Ahmed, T.; Pervez, M.N.; Naddeo, V.; Islam, M.S. Aquatic Microplastic Pollution Control Strategies: Sustainable Degradation Techniques, Resource Recovery, and Recommendations for Bangladesh. Water 2022, 14, 3968. https://doi.org/10.3390/w14233968
Mahmud A, Wasif MM, Roy H, Mehnaz F, Ahmed T, Pervez MN, Naddeo V, Islam MS. Aquatic Microplastic Pollution Control Strategies: Sustainable Degradation Techniques, Resource Recovery, and Recommendations for Bangladesh. Water. 2022; 14(23):3968. https://doi.org/10.3390/w14233968
Chicago/Turabian StyleMahmud, Abir, Mustafa Md Wasif, Hridoy Roy, Fareen Mehnaz, Tasnim Ahmed, Md. Nahid Pervez, Vincenzo Naddeo, and Md. Shahinoor Islam. 2022. "Aquatic Microplastic Pollution Control Strategies: Sustainable Degradation Techniques, Resource Recovery, and Recommendations for Bangladesh" Water 14, no. 23: 3968. https://doi.org/10.3390/w14233968
APA StyleMahmud, A., Wasif, M. M., Roy, H., Mehnaz, F., Ahmed, T., Pervez, M. N., Naddeo, V., & Islam, M. S. (2022). Aquatic Microplastic Pollution Control Strategies: Sustainable Degradation Techniques, Resource Recovery, and Recommendations for Bangladesh. Water, 14(23), 3968. https://doi.org/10.3390/w14233968