Adsorption of Methylene Blue by Coal-Based Activated Carbon in High-Salt Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization Method
2.3. Preparation of Activated Carbon
2.4. Adsorption Experiments
3. Results
3.1. Characterization of CBAC
3.1.1. Pore Structure Characterization
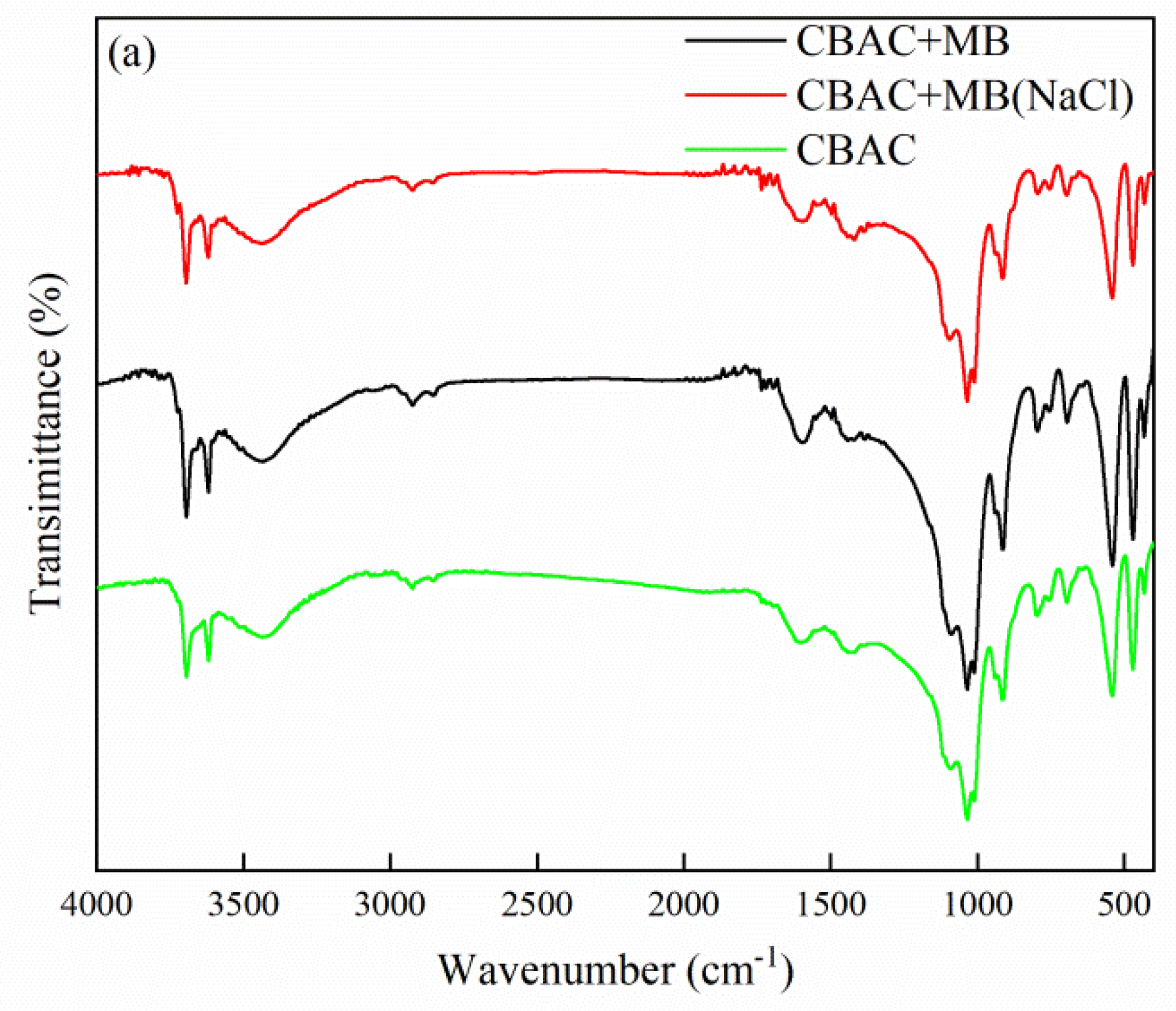
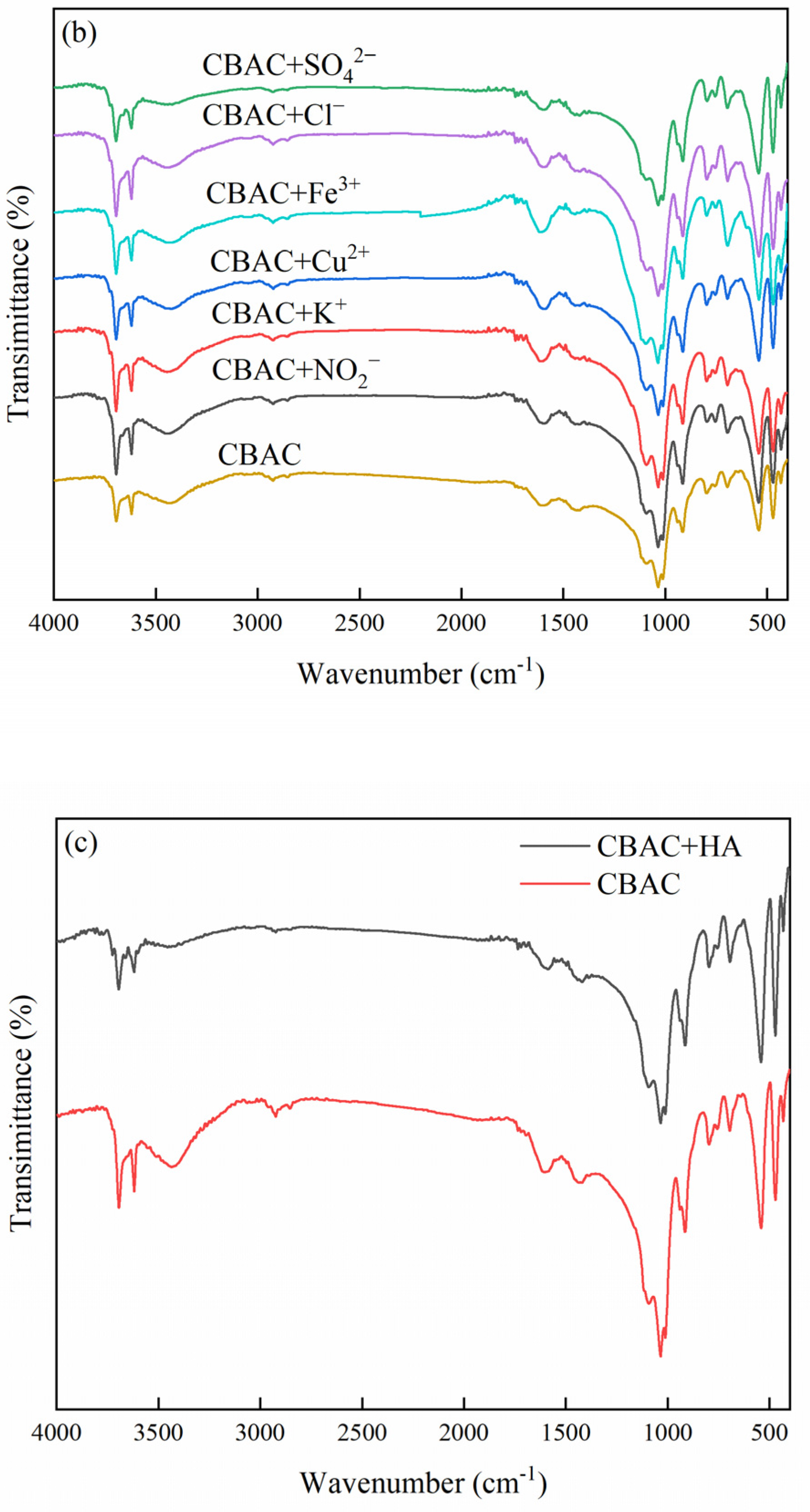
3.1.2. FTIR
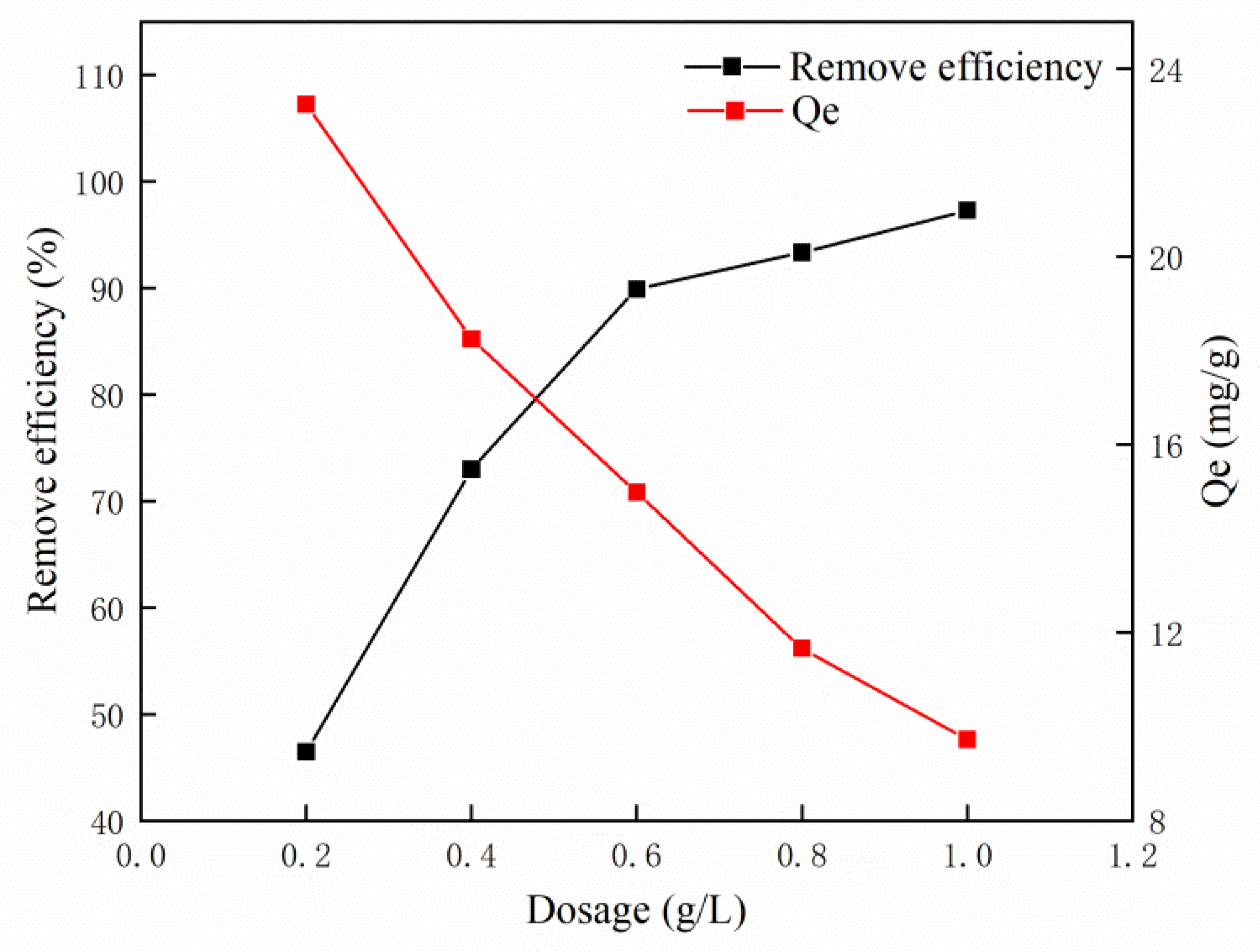
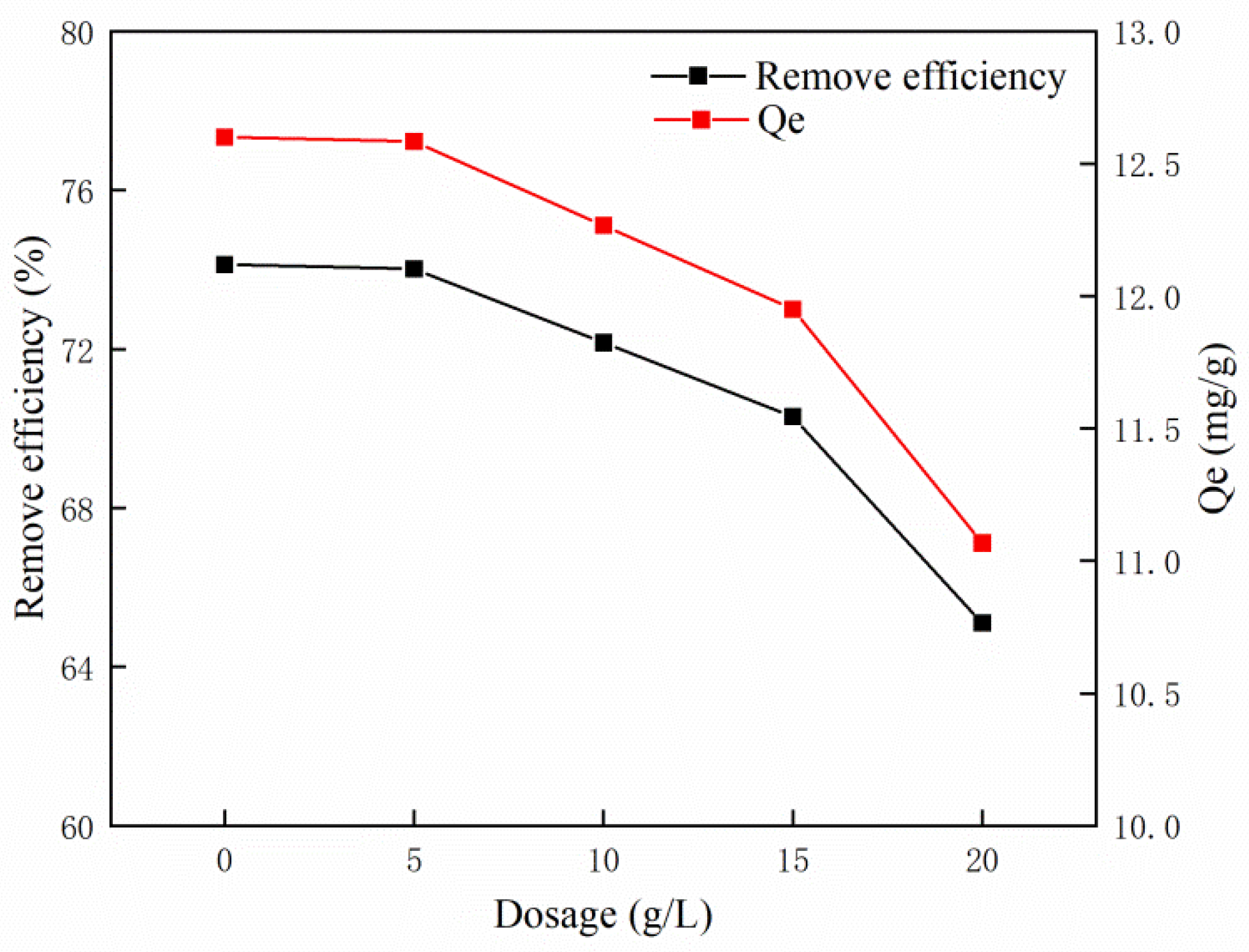
3.2. Effect of Adsorbent Dosage on MB Adsorption
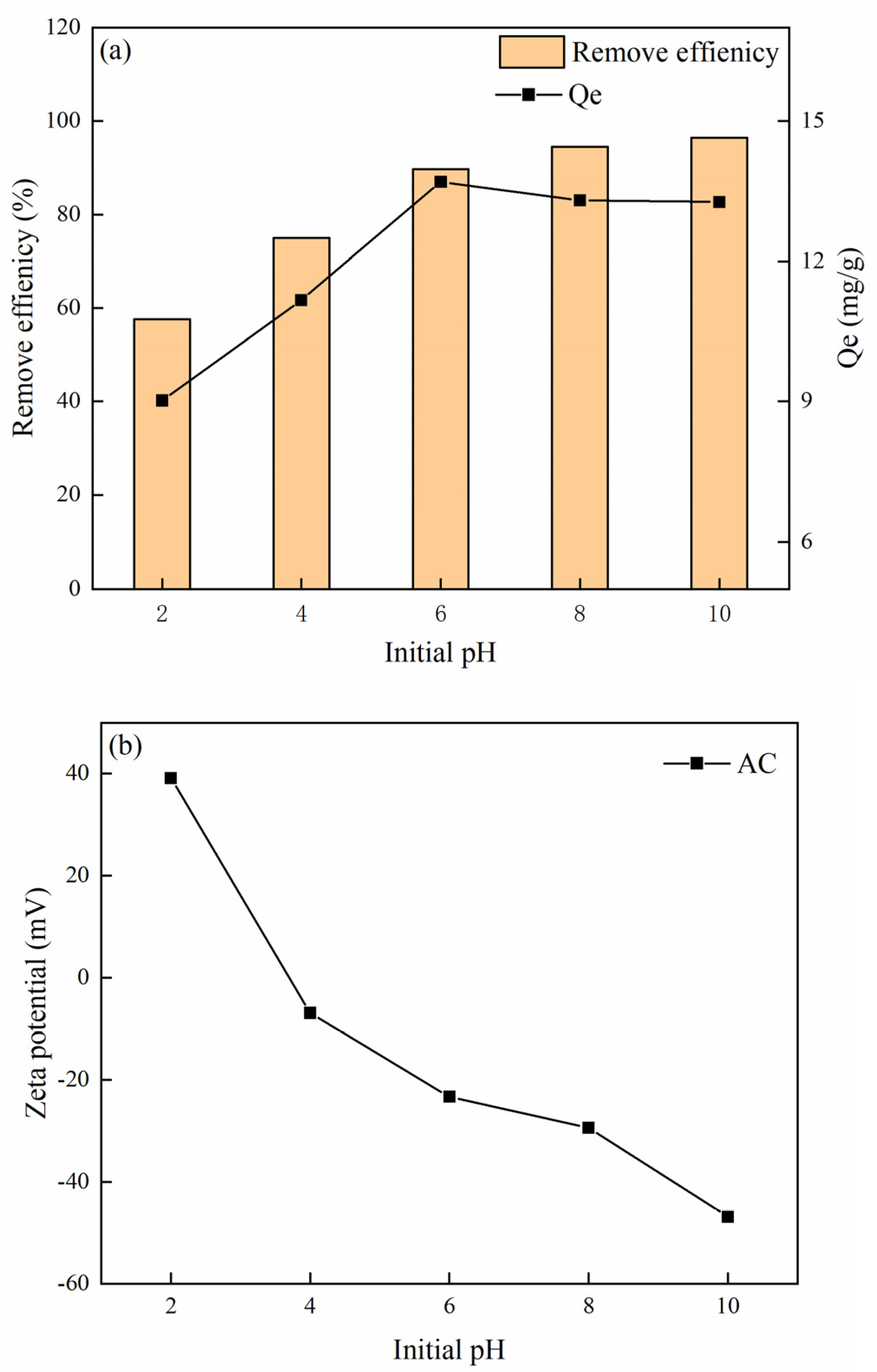
3.3. Effect of Solution pH on MB Adsorption
3.4. Effect of NaCl Concentration on MB Adsorption
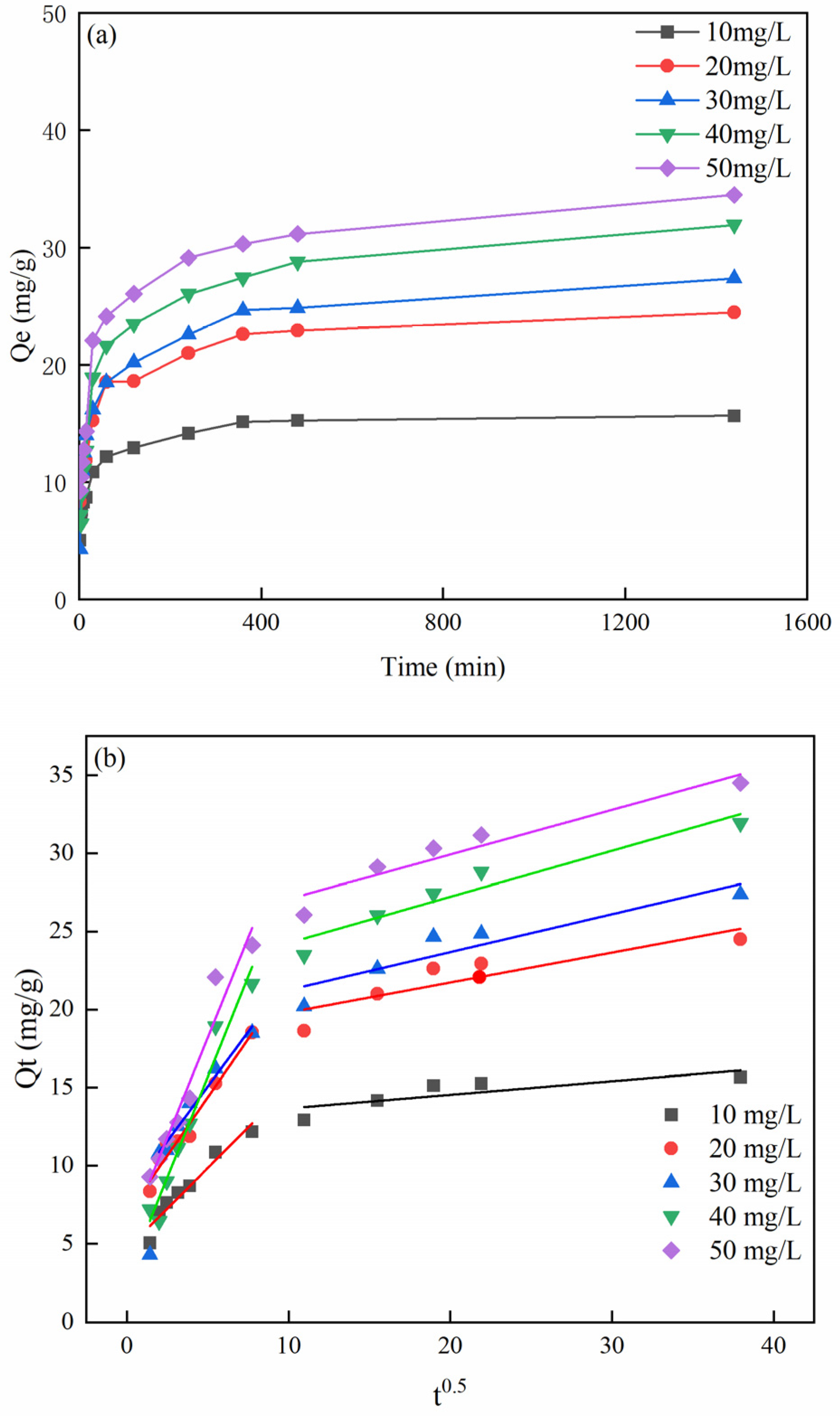
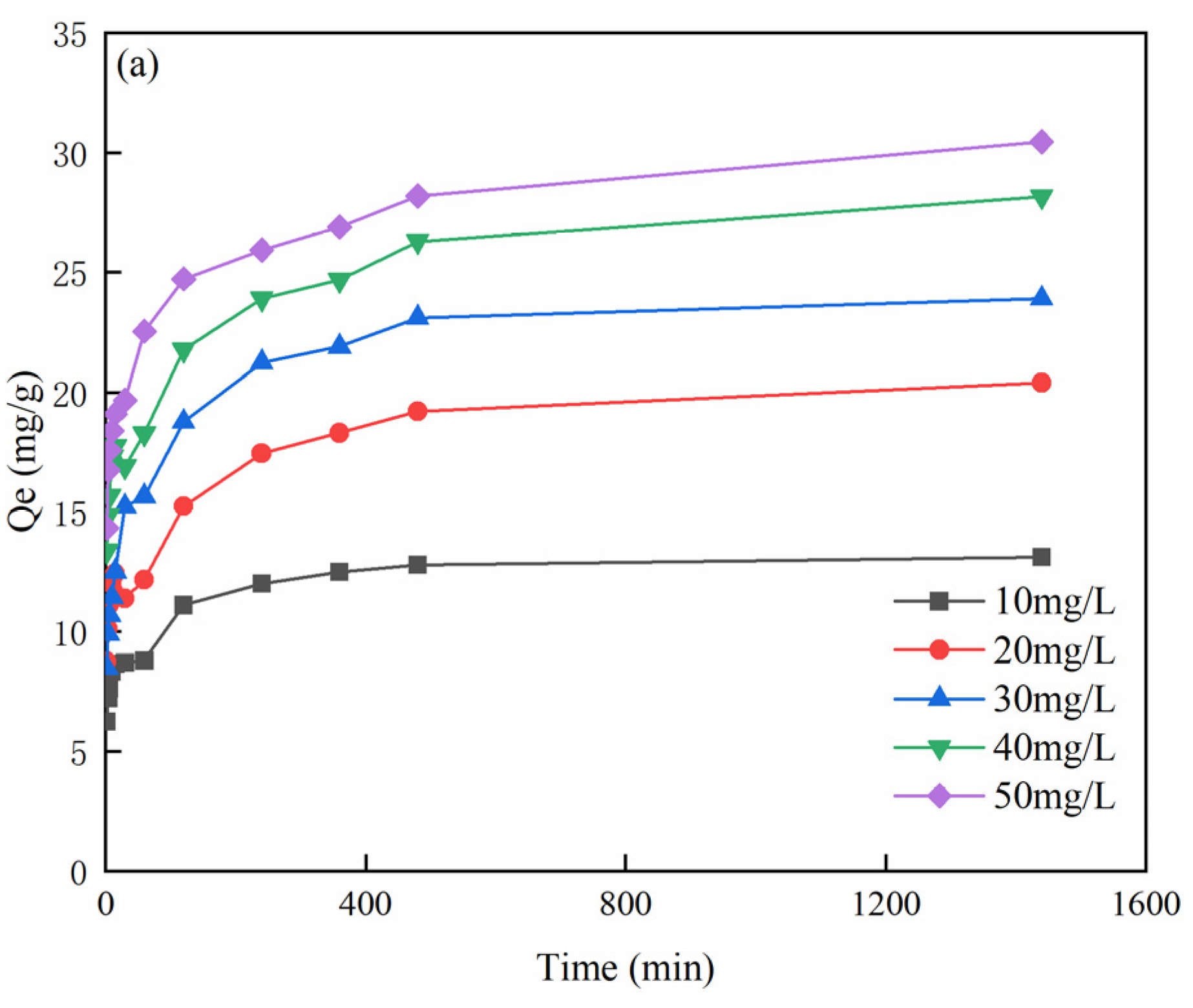
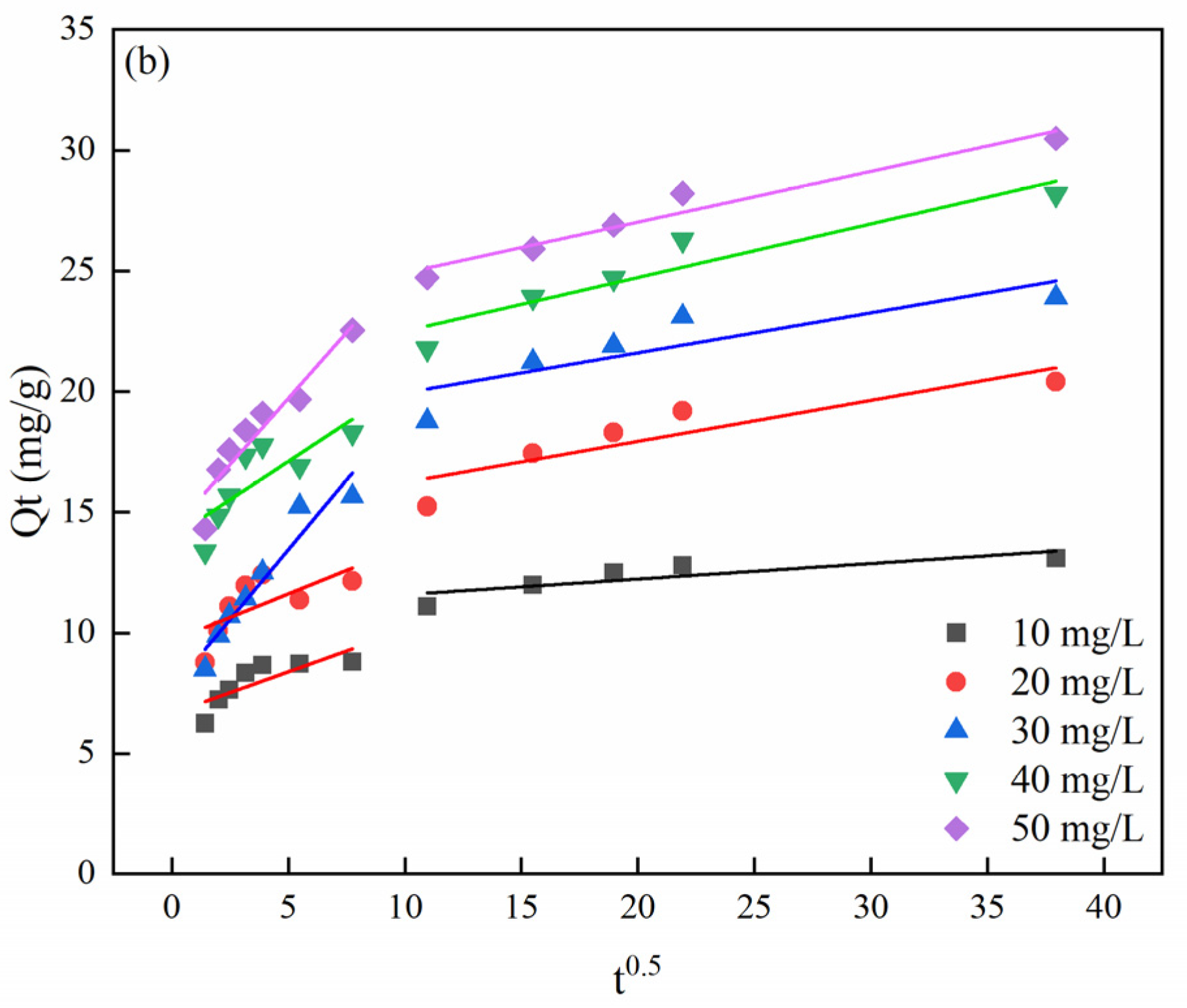
3.5. Effect of Time on MB Adsorption and Adsorption Kinetics
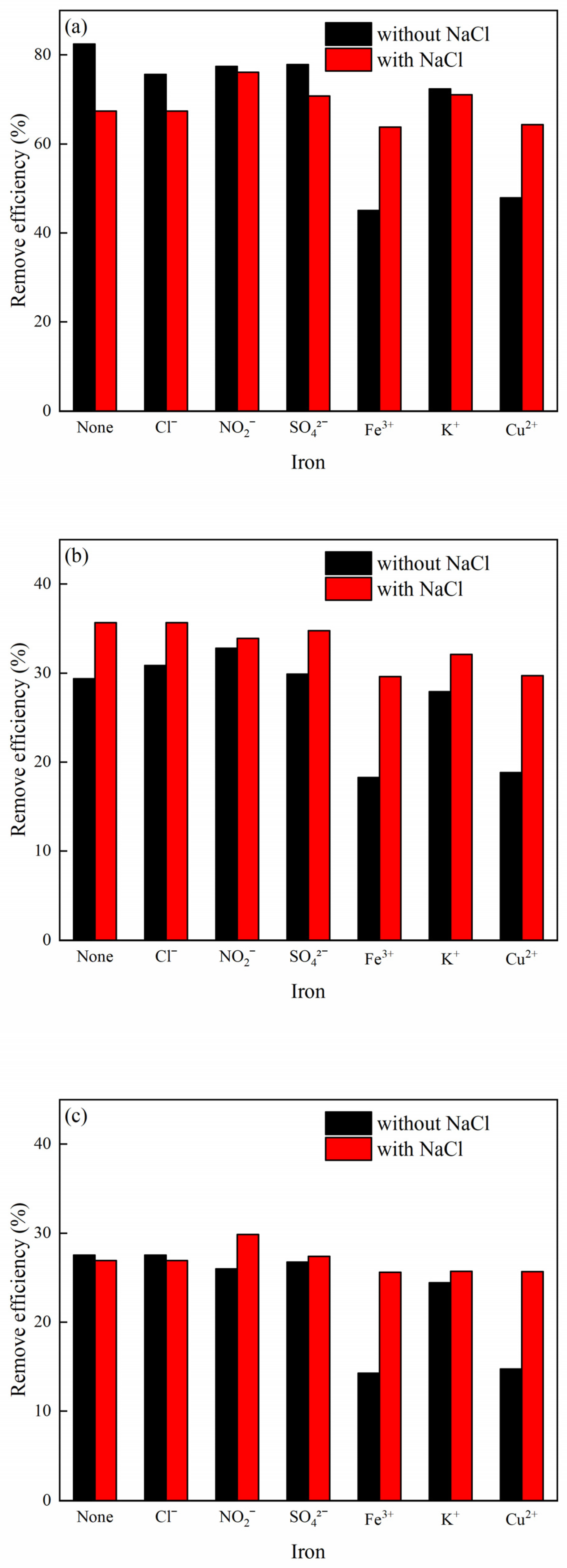
3.6. Effects of Coexisting Ions on MB Adsorption
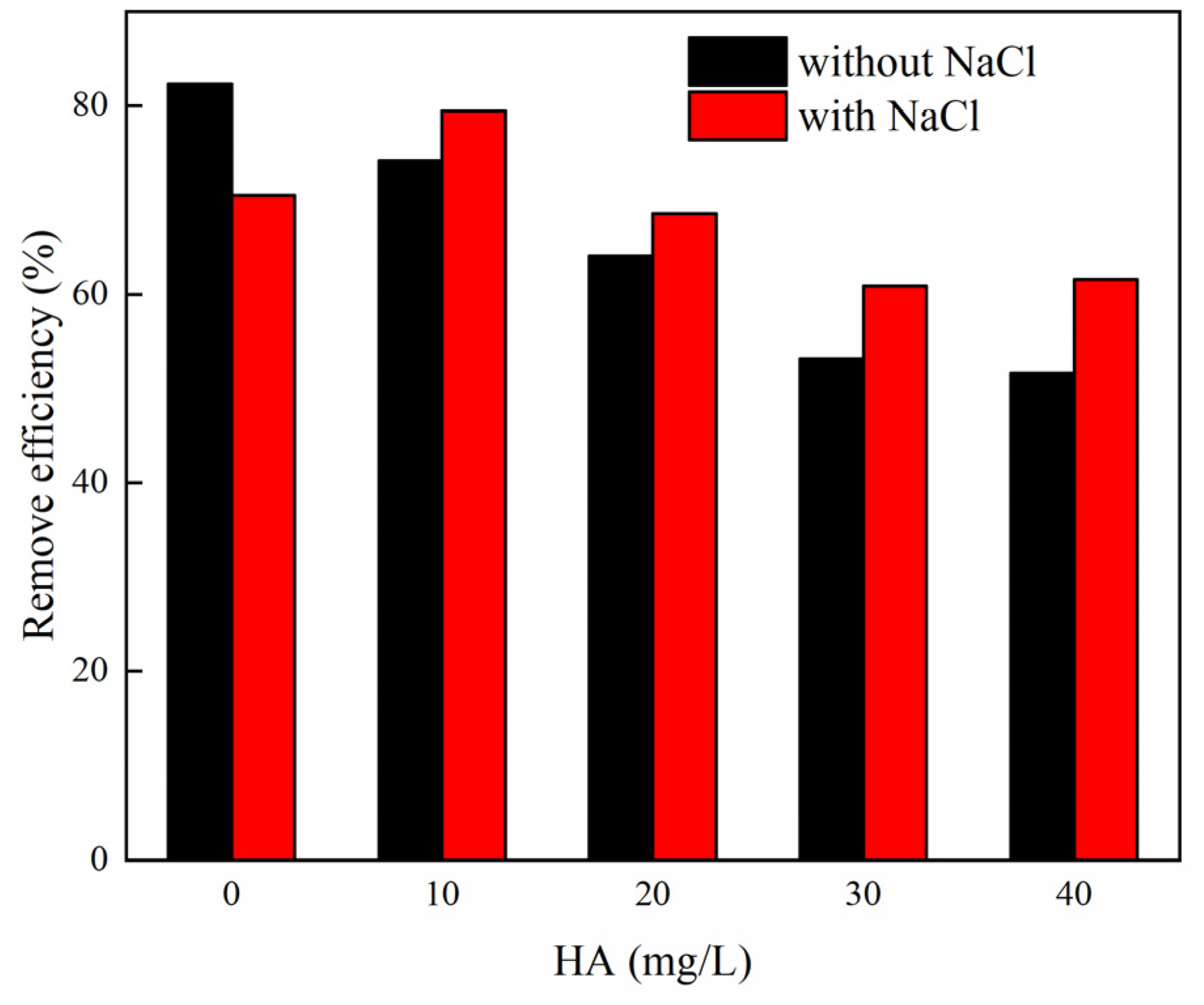
3.7. Effects of Organic Matter on MB Adsorption
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Khatri, A.; Peerzada, M.H.; Mohsin, M.; White, M. A review on developments in dyeing cotton fabrics with reactive dyes for reducing effluent pollution. J. Clean. Prod. 2015, 87, 50–57. [Google Scholar] [CrossRef]
- Wang, X.; Yao, Z.; Wang, J.; Guo, W.; Li, G. Degradation of reactive brilliant red in aqueous solution by ultrasonic cavitation. Ultrason. Sonochem. 2008, 15, 43–48. [Google Scholar] [CrossRef] [PubMed]
- López, D.E.; Trembly, J.P. Desalination of hypersaline brines with joule-heating and chemical pre-treatment: Conceptual design and economics. Desalination 2017, 415, 49–57. [Google Scholar] [CrossRef] [Green Version]
- Lin, J.; Ye, W.; Baltaru, M.; Tang, Y.P.; Bernstein, N.J.; Gao, P.; Balta, S.; Vlad, M.; Volodin, A.; Sotto, A.; et al. Tight ultrafiltration membranes for enhanced separation of dyes and Na2SO4 during textile wastewater treatment. J. Membr. Sci. 2016, 514, 217–228. [Google Scholar] [CrossRef]
- Muthukumar, M. Studies on the effect of inorganic salts on decolouration of acid dye effluents by ozonation. Dye. Pigment. 2004, 62, 221–228. [Google Scholar] [CrossRef]
- Chen, Y.; Sun, R.; Yan, W.; Wu, M.; Zhou, Y.; Gao, C. Antibacterial polyvinyl alcohol nanofiltration membrane incorporated with Cu(OH)2 nanowires for dye/salt wastewater treatment. Sci. Total Environ. 2022, 817, 152897. [Google Scholar] [CrossRef] [PubMed]
- Panagopoulos, A. Study and evaluation of the characteristics of saline wastewater (brine) produced by desalination and industrial plants. Environ. Sci. Pollut. Res. 2022, 29, 23736–23749. [Google Scholar] [CrossRef] [PubMed]
- Raveh, E.; Ben-Gal, A. Irrigation with water containing salts: Evidence from a macro-data national case study in Israel. Agric. Water Manag. 2016, 170, 176–179. [Google Scholar] [CrossRef]
- Zhao, T.; Liu, R.; Lu, J.; Zhu, X.; Zhu, X.; Lu, K.; Zhu, H. Photocatalytic degradation of methylene blue solution by diphenylanthrazoline compounds. J. Phys. Org. Chem. 2017, 30, e3712. [Google Scholar] [CrossRef]
- Islam, M.A.; Ali, I.; Karim, S.M.A.; Hossain Firoz, M.S.; Chowdhury, A.; Morton, D.W.; Angove, M.J. Removal of dye from polluted water using novel nano manganese oxide-based materials. J. Water Process Eng. 2019, 32, 100911. [Google Scholar] [CrossRef]
- Tan, K.B.; Vakili, M.; Horri, B.A.; Poh, P.E.; Abdullah, A.Z.; Salamatinia, B. Adsorption of dyes by nanomaterials: Recent developments and adsorption mechanisms. Sep. Purif. Technol. 2015, 150, 229–242. [Google Scholar] [CrossRef]
- Xu, N.; Li, J.; Wang, Y.; Fang, C.; Li, X.; Wang, Y.; Zhou, L.; Zhu, B.; Wu, Z.; Zhu, S.; et al. A water lily-inspired hierarchical design for stable and efficient solar evaporation of high-salinity brine. Sci Adv. 2019, 5, w7013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, P.; Zhu, J.; Yuan, S.; Zhang, G.; Volodine, A.; Tian, M.; Wang, J.; Luis, P.; Van der Bruggen, B. Erythritol-based polyester loose nanofiltration membrane with fast water transport for efficient dye/salt separation. Chem. Eng. J. 2021, 406, 126796. [Google Scholar] [CrossRef]
- Su, Y.; Yan, X.; Chen, Y.; Guo, X.; Chen, X.; Lang, W. Facile Fabrication of Cof-Lzu1/Pes Composite membrane via interfacial polymerization on microfiltration substrate for dye/salt separation. J. Membr. Sci. 2021, 618, 118706. [Google Scholar] [CrossRef]
- Cao, X.; Yan, Y.; Zhou, F.; Sun, S. Tailoring nanofiltration membranes for effective removing dye intermediates in complex dye-wastewater. J. Membr. Sci. 2020, 595, 117476. [Google Scholar] [CrossRef]
- Li, Q.; Liao, Z.; Fang, X.; Wang, D.; Xie, J.; Sun, X.; Wang, L.; Li, J. Tannic acid-polyethyleneimine crosslinked loose nanofiltration membrane for dye/salt mixture separation. J. Membr. Sci. 2019, 584, 324–332. [Google Scholar] [CrossRef]
- Varjani, S.; Rakholiya, P.; Ng, H.Y.; You, S.; Teixeira, J.A. Microbial degradation of dyes: An overview. Bioresour. Technol. 2020, 314, 123728. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Liu, Y.; Shen, L.; Lin, H.; Yu, W.; Xu, Y.; Li, R.; Huang, L. Facile preparation of recyclable magnetic Ni@Filter paper composite materials for efficient photocatalytic degradation of methyl orange. J. Colloid Interface Sci. 2021, 582, 291–300. [Google Scholar] [CrossRef]
- Chen, F.; Liu, L.; Chen, J.; Li, W.; Chen, Y.; Zhang, Y.; Wu, J.; Mei, S.; Yang, Q.; Yu, H. Efficient decontamination of organic pollutants under high salinity conditions by a nonradical peroxymonosulfate activation system. Water Res. 2021, 191, 116799. [Google Scholar] [CrossRef]
- Corti Monzón, G.; Nisenbaum, M.; Herrera Seitz, M.K.; Murialdo, S.E. New findings on aromatic compounds’ degradation and their metabolic pathways, the biosurfactant production and motility of the halophilic bacterium Halomonas Sp. Khs3. Curr. Microbiol. 2018, 75, 1108–1118. [Google Scholar] [CrossRef]
- Awual, M.R.; Yaita, T.; Shiwaku, H.; Suzuki, S. A sensitive ligand embedded nano-conjugate adsorbent for effective cobalt(Ii) ions capturing from contaminated water. Chem. Eng. J. 2015, 276, 1–10. [Google Scholar] [CrossRef]
- Hasanpour, M.; Hatami, M. Application of three dimensional porous aerogels as adsorbent for removal of heavy metal ions from water/wastewater: A review study. Adv. Colloid Interface Sci. 2020, 284, 102247. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhu, C.; Liu, F.; Yuan, Y.; Wu, H.; Li, A. Effects of ionic strength on removal of toxic pollutants from aqueous media with multifarious adsorbents: A review. Sci. Total Environ. 2019, 646, 265–279. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Qu, R.; Chen, H.; Mu, L.; Liu, X.; Wang, T.; Zhang, Y.; Sun, C. Synthesis of silica gel supported salicylaldehyde modified pamam dendrimers for the effective removal of Hg(Ii) From aqueous solution. J. Hazard. Mater. 2014, 278, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Liu, M.; Wang, X. A novel chitosan based adsorbent for boron separation. Sep. Purif. Technol. 2019, 211, 162–169. [Google Scholar] [CrossRef]
- Ahmad, N.N.R.; Leo, C.P.; Ahmad, A.L. Effects of solvent and ionic liquid properties on ionic liquid enhanced polysulfone/SAPO-34 mixed matrix membrane for CO2 removal. Microporous Mesoporous Mater. 2019, 283, 64–72. [Google Scholar] [CrossRef]
- Rafatullah, M.; Sulaiman, O.; Hashim, R.; Ahmad, A. Adsorption of methylene blue on low-cost adsorbents: A review. J. Hazard. Mater. 2010, 177, 70–80. [Google Scholar] [CrossRef]
- Karabacakoğlu, B.; Savlak, Ö. Electrochemical regeneration of Cr(Vi) saturated granular and powder activated carbon: Comparison of regeneration efficiency. Ind. Eng. Chem. Res. 2014, 53, 13171–13179. [Google Scholar] [CrossRef]
- Bury, N.A.; Mumford, K.A.; Stevens, G.W. The electro-fenton regeneration of granular activated carbons: Degradation of organic contaminants and the relationship to the carbon surface. J. Hazard. Mater. 2021, 416, 125792. [Google Scholar] [CrossRef]
- Maneerung, T.; Liew, J.; Dai, Y.; Kawi, S.; Chong, C.; Wang, C. Activated carbon derived from carbon residue from biomass gasification and its application for dye adsorption: Kinetics, isotherms and thermodynamic studies. Bioresour. Technol. 2016, 200, 350–359. [Google Scholar] [CrossRef]
- Wang, H.; Xu, J.; Liu, X.; Sheng, L. Preparation of straw activated carbon and its application in wastewater treatment: A review. J. Clean. Prod. 2021, 283, 124671. [Google Scholar] [CrossRef]
- Tan, X.; Liu, S.; Liu, Y.; Gu, Y.; Zeng, G.; Hu, X.; Wang, X.; Liu, S.; Jiang, L. Biochar as potential sustainable precursors for activated carbon production: Multiple applications in environmental protection and energy storage. Bioresour. Technol. 2017, 227, 359–372. [Google Scholar] [CrossRef] [PubMed]
- Ao, W.; Fu, J.; Mao, X.; Kang, Q.; Ran, C.; Liu, Y.; Zhang, H.; Gao, Z.; Li, J.; Liu, G.; et al. Microwave assisted preparation of activated carbon from biomass: A review. Renew. Sustain. Energy Rev. 2018, 92, 958–979. [Google Scholar] [CrossRef]
- Kumar, A.; Jena, H.M. Preparation and characterization of high surface area activated carbon from fox nut (Euryale ferox) shell by chemical activation with H3PO4. Results Phys. 2016, 6, 651–658. [Google Scholar] [CrossRef] [Green Version]
- Danish, M.; Ahmad, T. A review on utilization of wood biomass as a sustainable precursor for activated carbon production and application. Renew. Sustain. Energy Rev. 2018, 87, 1–21. [Google Scholar] [CrossRef]
- Liu, Q.; Li, Y.; Chen, H.; Lu, J.; Yu, G.; Möslang, M.; Zhou, Y. Superior adsorption capacity of functionalised straw adsorbent for dyes and heavy-metal ions. J. Hazard. Mater. 2020, 382, 121040. [Google Scholar] [CrossRef]
- Saleem, J.; Shahid, U.B.; Hijab, M.; Mackey, H.; McKay, G. Production and applications of activated carbons as adsorbents from olive stones. Biomass Convers. Biorefinery 2019, 9, 775–802. [Google Scholar] [CrossRef] [Green Version]
- Elkady, M.; Shokry, H.; Hamad, H. New activated carbon from mine coal for adsorption of dye in simulated water or multiple heavy metals in real wastewater. Materials 2020, 13, 2498. [Google Scholar] [CrossRef]
- Li, H.; Zheng, F.; Wang, J.; Zhou, J.; Huang, X.; Chen, L.; Hu, P.; Gao, J.; Zhen, Q.; Bashir, S.; et al. Facile preparation of zeolite-activated carbon composite from coal gangue with enhanced adsorption performance. Chem. Eng. J. 2020, 390, 124513. [Google Scholar] [CrossRef]
- Wang, L.; Sun, F.; Hao, F.; Qu, Z.; Gao, J.; Liu, M.; Wang, K.; Zhao, G.; Qin, Y. A green trace K2CO3 induced catalytic activation strategy for developing coal-converted activated carbon as advanced candidate for CO2 adsorption and supercapacitors. Chem. Eng. J. 2020, 383, 123205. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, L.S.; de Souza, L.K.C.; Thomaz, K.T.C.; Leite Lima, E.T.; Da Rocha Filho, G.N.; Do Nascimento, L.A.S.; de Oliveira Pires, L.H.; Faial, K.D.C.F.; Da Costa, C.E.F. Activated carbon obtained from amazonian biomass tailings (Acai Seed): Modification, characterization, and use for removal of metal ions from water. J. Environ. Manag. 2020, 270, 110868. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Wu, P.; Liu, J.; Rehman, S.; Ahmed, Z.; Ruan, B.; Zhu, N. Batch interaction of emerging tetracycline contaminant with novel phosphoric acid activated corn straw porous carbon: Adsorption rate and nature of mechanism. Environ. Res. 2020, 181, 108899. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yu, G.; Pan, L.; Li, C.; You, F.; Xie, S.; Wang, Y.; Ma, J.; Shang, X. Study of ciprofloxacin removal by biochar obtained from used tea leaves. J. Environ. Sci. China 2018, 73, 20–30. [Google Scholar] [CrossRef]
- Mayakaduwa, S.S.; Vithanage, M.; Karunarathna, A.; Mohan, D.; Ok, Y.S. Interface interactions between insecticide carbofuran and tea waste biochars produced at different pyrolysis temperatures. Chem. Spec. Bioavailab. 2016, 28, 110–118. [Google Scholar] [CrossRef] [Green Version]
- Fan, S.; Tang, J.; Wang, Y.; Li, H.; Zhang, H.; Tang, J.; Wang, Z.; Li, X. Biochar prepared from co-pyrolysis of municipal sewage sludge and tea waste for the adsorption of methylene blue from aqueous solutions: Kinetics, isotherm, thermodynamic and mechanism. J. Mol. Liq. 2016, 220, 432–441. [Google Scholar] [CrossRef]
- Wu, Z.; Zhong, H.; Yuan, X.; Wang, H.; Wang, L.; Chen, X.; Zeng, G.; Wu, Y. Adsorptive removal of methylene blue by rhamnolipid-functionalized graphene oxide from wastewater. Water Res. 2014, 67, 330–344. [Google Scholar] [CrossRef] [PubMed]
- Couto, O.M., Jr. Effect of solution Ph and influence of water hardness on caffeine adsorption onto activated carbons. Can. J. Chem. Enfineering 2015, 93, 68–77. [Google Scholar] [CrossRef]
- Somsesta, N.; Sricharoenchaikul, V.; Aht-Ong, D. Adsorption removal of methylene blue onto activated carbon/cellulose biocomposite films: Equilibrium and kinetic studies. Mater. Chem. Phys. 2020, 240, 122221. [Google Scholar] [CrossRef]
- Marrakchi, F.; Ahmed, M.J.; Khanday, W.A.; Asif, M.; Hameed, B.H. Mesoporous-activated carbon prepared from chitosan flakes via single-step sodium hydroxide activation for the adsorption of methylene blue. Int. J. Biol. Macromol. 2017, 98, 233–239. [Google Scholar] [CrossRef]
- Jung, C.; Park, J.; Lim, K.H.; Park, S.; Heo, J.; Her, N.; Oh, J.; Yun, S.; Yoon, Y. Adsorption of selected endocrine disrupting compounds and pharmaceuticals on activated biochars. J. Hazard. Mater. 2013, 263, 702–710. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Guo, T.; Ye, X.; Li, Q.; Guo, M.; Liu, H.; Wu, Z. Dye Adsorption by Resins: Effect of Ionic Strength on Hydrophobic and Electrostatic Interactions. Chem. Eng. J. 2013, 228, 392–397. [Google Scholar] [CrossRef]
- Aguiar, J.E.; Bezerra, B.T.C.; Siqueira, A.C.A.; Barrera, D.; Sapag, K.; Azevedo, D.C.S.; Lucena, S.M.P.; Silva, I.J. Improvement in the adsorption of anionic and cationic dyes from aqueous solutions: A comparative study using aluminium pillared clays and activated carbon. Sep. Sci. Technol. 2014, 49, 741–751. [Google Scholar] [CrossRef] [Green Version]
- Jang, H.M.; Yoo, S.; Choi, Y.; Park, S.; Kan, E. Adsorption isotherm, kinetic modeling and mechanism of tetracycline on pinus taeda-derived activated biochar. Bioresour. Technol. 2018, 259, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Albadarin, A.B.; Mo, J.; Glocheux, Y.; Allen, S.; Walker, G.; Mangwandi, C. Preliminary investigation of mixed adsorbents for the removal of copper and methylene blue from aqueous solutions. Chem. Eng. J. 2014, 255, 525–534. [Google Scholar] [CrossRef] [Green Version]
- Kyzas, G.Z.; Lazaridis, N.K.; Mitropoulos, A.C. Removal of dyes from aqueous solutions with untreated coffee residues as potential low-cost adsorbents: Equilibrium, reuse and thermodynamic approach. Chem. Eng. J. 2012, 189–190, 148–159. [Google Scholar] [CrossRef]
- Lin, L.; Jiang, W.; Xu, P. Comparative study on pharmaceuticals adsorption in reclaimed water desalination concentrate using biochar: Impact of salts and organic matter. Sci. Total Environ. 2017, 601–602, 857–864. [Google Scholar] [CrossRef]
- Üçer, A.; Uyanik, A.; Aygün, F. Adsorption of Cu(Ii), Cd(Ii), Zn(Ii), Mn(Ii) and Fe(Iii) ions by tannic acid immobilised activated carbon. Sep. Purif. Technol. 2006, 47, 113–118. [Google Scholar] [CrossRef]
- Murray, A.; Örmeci, B. Competitive effects of humic acid and wastewater on adsorption of methylene blue dye by activated carbon and non-imprinted polymers. J. Environ. Sci. China 2018, 66, 310–317. [Google Scholar] [CrossRef]

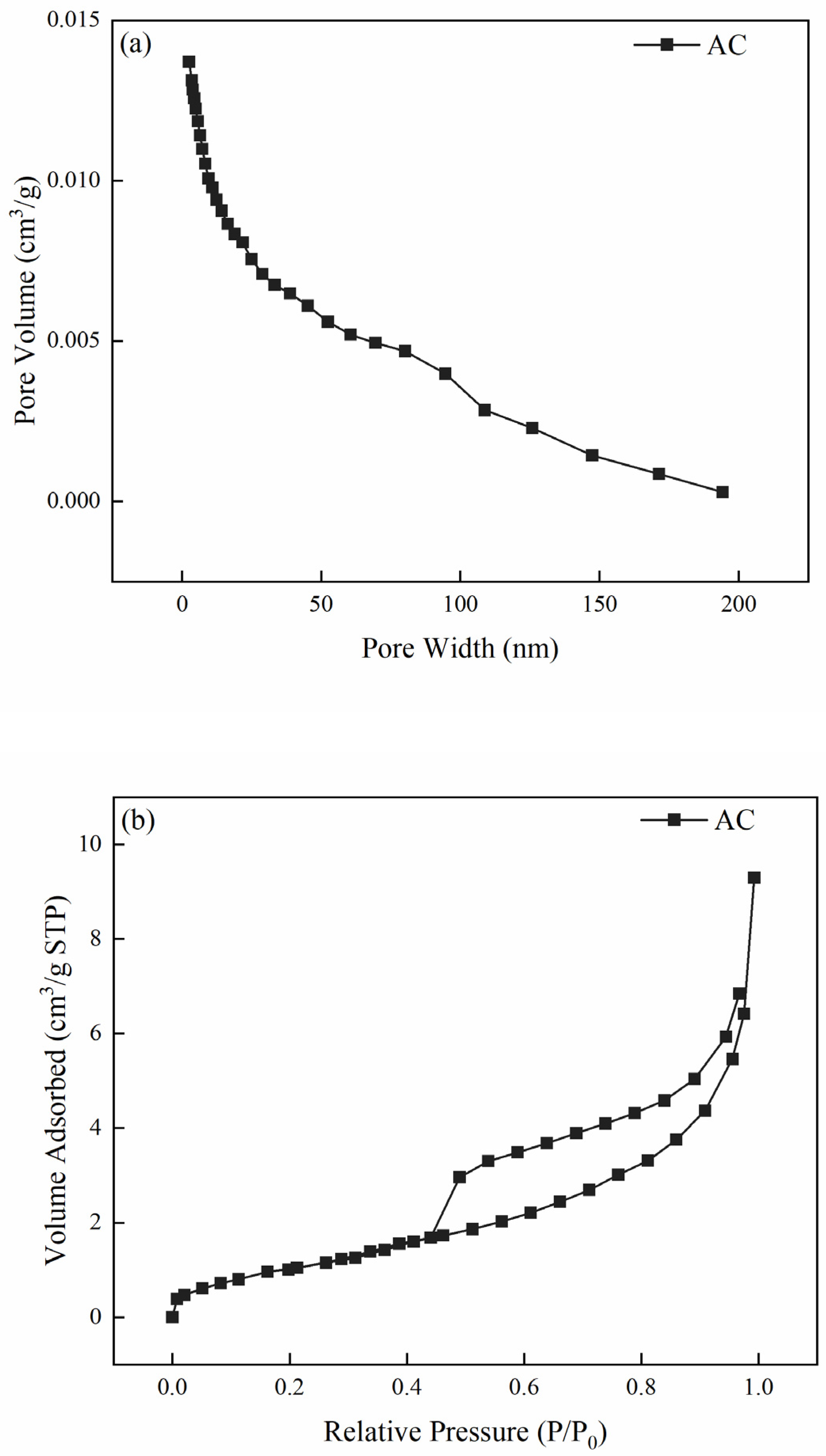
| Sample | SBET (m2/g) | VTotal 1 (cm3/g) | Vmicro (cm3/g) | Vmeso (cm3/g) | Average Pore Size (nm) |
|---|---|---|---|---|---|
| CBAC | 165.815 | 0.014 | 0.006 | 0.008 | 13.388 |
| CO (mg/L) | Qe(mg/L) | RSD (%) | Pseudo-First-Order Model | Pseudo-Second-Order Model |
|---|---|---|---|---|
| R2 | R2 | |||
| 10 | 15.67 | 0.43 | 0.9256 | 0.9837 |
| 20 | 24.48 | 0.23 | 0.9519 | 0.9780 |
| 30 | 27.37 | 0.20 | 0.7997 | 0.9727 |
| 40 | 31.92 | 0.75 | 0.9632 | 0.9918 |
| 50 | 34.48 | 0.65 | 0.9574 | 0.9865 |
| CO (mg/L) | Qe(mg/L) | RSD (%) | Pseudo-First-Order Model | Pseudo-Second-Order Model |
|---|---|---|---|---|
| R2 | R2 | |||
| 10 | 13.10 | 0.88 | 0.8907 | 0.9740 |
| 20 | 20.40 | 0.88 | 0.9475 | 0.9667 |
| 30 | 23.90 | 0.57 | 0.9590 | 0.9918 |
| 40 | 28.17 | 0.55 | 0.9348 | 0.9765 |
| 50 | 30.48 | 0.93 | 0.9203 | 0.9710 |
| CO (mg/L) | K1 | C1 | R2 | K2 | C2 | R2 |
|---|---|---|---|---|---|---|
| 10 | 1.042 | 4.649 | 0.9267 | 0.087 | 12.807 | 0.5466 |
| 20 | 1.491 | 6.922 | 0.9625 | 0.192 | 17.892 | 0.7132 |
| 30 | 1.398 | 8.083 | 0.9758 | 0.242 | 18.828 | 0.8102 |
| 40 | 2.570 | 2.832 | 0.9517 | 0.294 | 21.310 | 0.9058 |
| 50 | 2.556 | 5.424 | 0.9419 | 0.286 | 24.207 | 0.8892 |
| CO (mg/L) | K1 | C1 | R2 | K2 | C2 | R2 |
|---|---|---|---|---|---|---|
| 10 | 0.345 | 6.664 | 0.5705 | 0.064 | 10.942 | 0.6212 |
| 20 | 0.390 | 9.668 | 0.3335 | 0.170 | 14.539 | 0.7404 |
| 30 | 1.157 | 7.669 | 0.9069 | 0.166 | 18.291 | 0.6584 |
| 40 | 0.635 | 13.933 | 0.5714 | 0.222 | 20.283 | 0.8551 |
| 50 | 1.093 | 14.256 | 0.8795 | 0.210 | 22.812 | 0.9389 |
| CO (mg/L) | Removal Efficiency (%) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N 1 | RSD (%) | Cl− | RSD (%) | NO2− | RSD (%) | SO42− | RSD (%) | Fe3+ | RSD (%) | K+ | RSD (%) | Cu2+ | RSD (%) | |
| 10 | 82.4 | 1.3 | 75.6 | 1.3 | 77.5 | 0.9 | 77.9 | 0.6 | 45.6 | 1.1 | 72.4 | 1.0 | 47.9 | 0.6 |
| 30 | 44.2 | 0.6 | 41.1 | 1.1 | 45.5 | 0.59 | 41.9 | 0.91 | 24.5 | 0.6 | 43.2 | 0.6 | 31.2 | 0.9 |
| 50 | 28.4 | 0.3 | 29.9 | 0.3 | 31.9 | 0.5 | 28.9 | 0.3 | 17.1 | 0.6 | 26.9 | 0.5 | 17.77 | 0.6 |
| CO (mg/L) | Removal Efficiency (%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cl− | RSD (%) | NO2− | RSD (%) | SO42− | RSD (%) | Fe3+ | RSD (%) | K+ | RSD (%) | Cu2+ | RSD (%) | |
| 10 | 67.4 | 0.7 | 76.1 | 0.8 | 70.8 | 0.7 | 63.8 | 0.8 | 71.1 | 0.9 | 64.3 | 1.3 |
| 30 | 47.7 | 0.9 | 47.8 | 0.9 | 42.8 | 0.8 | 40.3 | 0.4 | 45.1 | 1.4 | 39.8 | 1.0 |
| 50 | 30.9 | 0.2 | 29.3 | 0.5 | 30.1 | 0.7 | 25.6 | 0.6 | 27.8 | 0.2 | 25.7 | 0.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Ma, J.; Sun, Y. Adsorption of Methylene Blue by Coal-Based Activated Carbon in High-Salt Wastewater. Water 2022, 14, 3576. https://doi.org/10.3390/w14213576
Wang J, Ma J, Sun Y. Adsorption of Methylene Blue by Coal-Based Activated Carbon in High-Salt Wastewater. Water. 2022; 14(21):3576. https://doi.org/10.3390/w14213576
Chicago/Turabian StyleWang, Jinlong, Jingqian Ma, and Yongjun Sun. 2022. "Adsorption of Methylene Blue by Coal-Based Activated Carbon in High-Salt Wastewater" Water 14, no. 21: 3576. https://doi.org/10.3390/w14213576







