Insights into the Domestic Wastewater Treatment (DWWT) Regimes: A Review
Abstract
:1. Introduction
2. Water Contaminants
2.1. Characterization of Wastewater (WW)
2.1.1. Total Dissolved Solids (TDS)
2.1.2. Dissolved Organic Carbon (DOC)
2.1.3. Microorganisms
- (A)
- Dissolved oxygen demand (DOD)
- (B)
- Biochemical oxygen demand (BOD)
- (C)
- Chemical oxygen demand (COD)
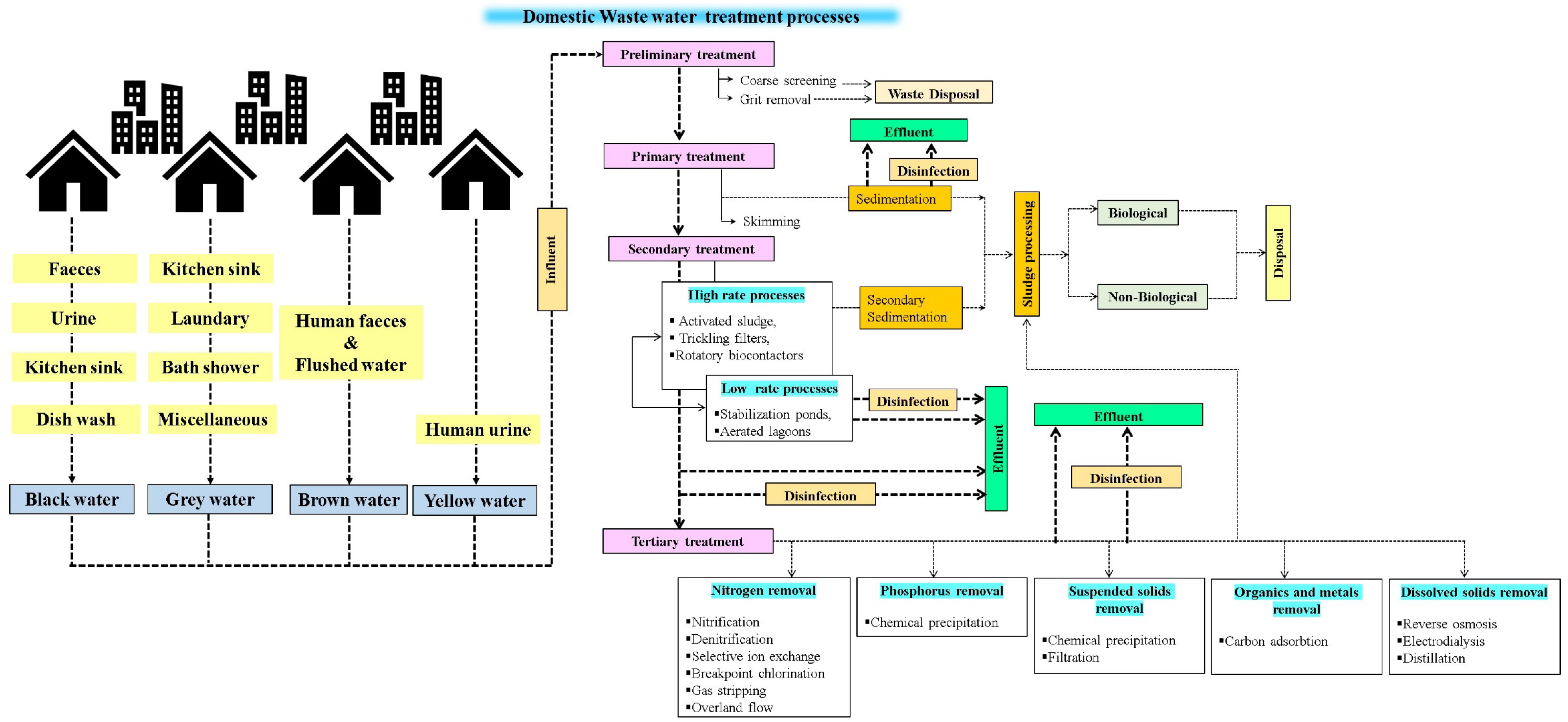
3. Process of Wastewater Treatment
3.1. Preliminary Treatment Plant
3.2. Primary Treatment Plant
3.3. Secondary Treatment Plant
3.4. Tertiary Treatment Plant
3.5. Disinfection
4. Process of Water Purification
4.1. Preliminary Treatment
4.2. Primary Treatment
- (a)
- Coagulation and flocculation
- (b)
- Precipitation
- (c)
- Chemical precipitation
4.3. Secondary Treatment
- (a)
- Adsorption
- (b)
- Filtration
- (c)
- Biological process
- (d)
- Trickling filter
- (e)
- Biomass on filter
- (f)
- Design and implementation
- 1.
- Pre-treatment of the wastewaterBiological filtration or a trickling filter is suitable for the secondary treatment of primary effluent. Pre-treatment of the wastewater is necessary to remove the clogging material from the waste before starting the process.
- 2.
- Distribution systemEven distribution of settled sewage on the filter is necessary to maintain equal growth conditions for the biofilm-forming bacteria. To maintain the distribution, a piece of dosage equipment is necessary.
- 3.
- Filter bed and typesCircular filter beds are preferred over other shapes to increase the surface area of the media. The media used in this process as a filter should be chemically inert and must support the biofilm.
- 4.
- Dimensions of the filterGranular intact components should be used as filter media, and the material chosen must provide a suitable environment for the growth of a bacterial biofilm.
- 5.
- Effluent systemEffluent collecting channels must be large enough to efficiently drain a large volume of purified water to support the maximum flow rate.
- 6.
- VentilationA well-organized ventilation system is necessary to keep the aerobic biofilm layer functional. Sufficient opening at the base of the filter provides a good ventilation system to the plant. Air inlets are also useful for aeration, and they may be included in the chamber at various positions.
- i.
- It is a simple filtration process that uses microorganisms to filter out organic soluble impurities. As no chemicals are used, it is safe for the biotic ecosystem.
- ii.
- A rapid reduction in the level of BOD is observed in the effluent water, which depicts a good-quality treatment.
- iii.
- It is a simple, reliable and effective process for purifying domestic wastewater.
- iv.
- The low consumption of energy and durability of the filter make it a cost-effective technique.
- v.
- No highly trained and skilled operator is needed to run the process. It is a suitable filtration process for small or medium-sized community use [49].
- i.
- The process on its own is not sufficient for the purification of domestic wastewater. Pre- and post-purification systems are required.
- ii.
- The biofilm thickness must be under control. Increased thickness of biomass on filter impairs the process of aerobic respiration and thus reduces the efficiency of trickling filtration.
- iii.
- Regular attention is required to prevent the clogging problem [49].
- (g)
- Biosorption
- (h)
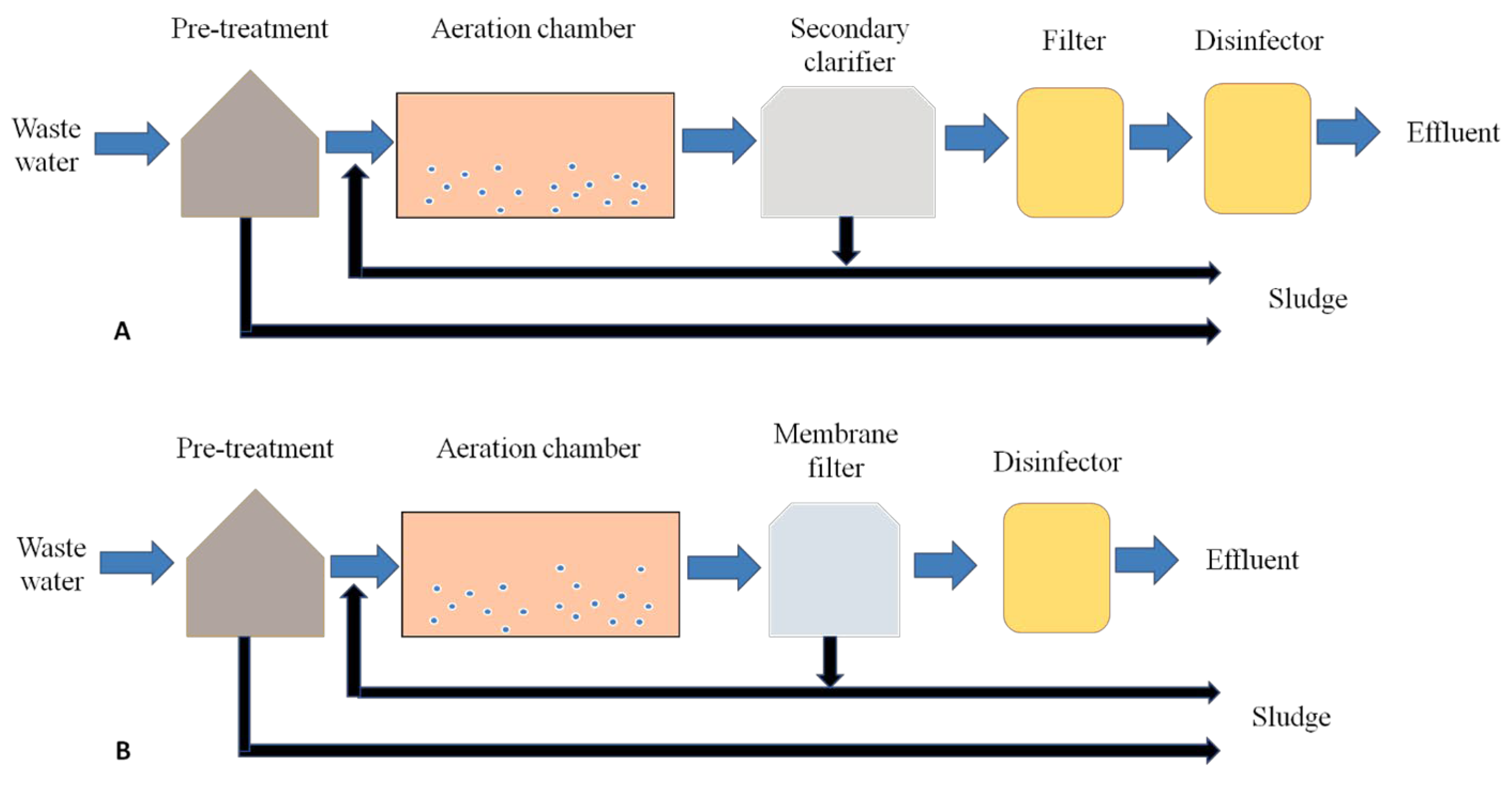
- Activated sludge processing (ASP)
- i.
- Reactor: The reactor is a kind of tank for mixing and aeration of the wastewater, and it is also known as the aeration tank.
- ii.
- Activated sludge (AS): The microbial biomass present in the reactor is abbreviated as AS. Bacteria, fungi and other unicellular or multicellular organisms comprise the biomass. The suspension of these organisms in the AS is called the mixed liquor.
- iii.
- Aeration and mixing system: Aeration and mixing of wastewater with AS are essential to maintain the air concentration and microbial population in the newly added water in the tank. Different techniques, such as diffused air and surface aeration, are used for this purpose.
- iv.
- i.
- Installation is cheaper than it is for other techniques.
- ii.
- Does not require large an area to install and produces good effluent quality.
- iii.
- Low foulant and lower pest accumulation; thus, it is safer to use [30].
- i.
- The hydraulic retention time for ASP is longer, and thus, it takes as much as 5 days to achieve the desired level of treatment. It is a much slower process than other conventional wastewater treatment techniques.
- ii.
- Operation costs are higher than those of the others.
- iii.
- A sudden change in the amount or in the character of sewage flow may disrupt the process.
- iv.
- It generates a high sludge volume and fast disposition. Its sludge retention time is low, which increases the overall operational cost.
- v.
- As the process requires activated sludge recycling, continuous and skilled supervision is necessary to keep the system active [30].
- (i)
- Membrane bioreactors (MBRs)
- i.
- The membrane used in an MBR can filter all pathogenic microorganisms, solids and biological waste suspended in the domestic and industrial wastewater.
- ii.
- The MBR membrane has a small pore size of <0.5 µm, which produces clear effluent water. It can remove 99.99% of total coliforms in water, and clear water is produced as effluent for direct potential reuse. As the secondary clarifier of ASP is replaced in MBR by the membrane, the footprint required in ASP is reduced.
- iii.
- MBR can efficiently purify huge volumes of domestic water continuously. Thus, it is more efficient than ASP.
- iv.
- i.
- Any MBR has high operational costs, and it has high complexity, which creates a drawback for this process.
- ii.
- Due to deposition of organic and inorganic materials on the membrane, there is fouling of the membrane. The physicochemical interactions between the MBR’s membrane and deposited components reduce the membrane efficiency, which creates a problem and must be controlled. Membrane fouling reduces the membrane’s lifetime. Replacement of the membrane is costly [56].
4.4. Tertiary Treatment
4.4.1. Ion Exchange
4.4.2. Membrane Filtration
- (a)
- Microfiltration
- (b)
- Ultrafiltration
- (c)
- Nanofiltration
- (d)
- Reverse osmosis
- (e)
- Membrane bioreactors (MBRs)
- (f)
- Anaerobic membrane bioreactors (AnMBRs)
- (g)
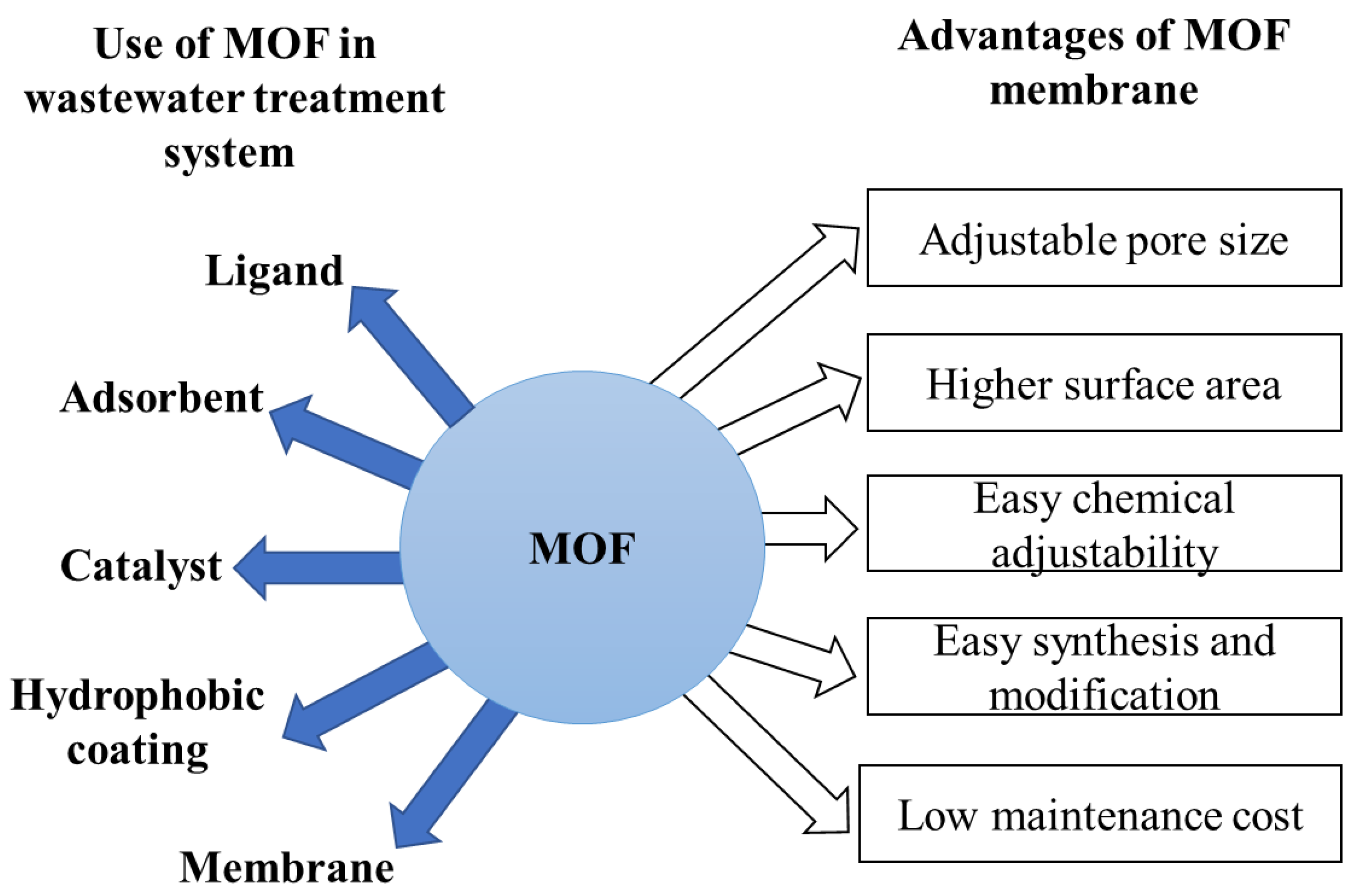
- MOF (metal–organic framework)
- (h)
- MXenes
5. Electrochemical Treatments
6. Disinfection
6.1. Pathogenic Micro-Organisms and Their Removal by Disinfection
- (a)
- Bacterial pathogens
- (b)
- Enteric bacterial pathogens
- (c)
- Protozoa
- (d)
- Cyanobacterial pathogens
6.2. Chemical Oxidants
- (a)
- Chlorine
- (b)
- Ozone
- (c)
- Peracetic acid (PAA)
- (d)
- Hydrogen peroxide (H2O2)
- (e)
- Advanced oxidation processes (AOP)
7. Conclusions and Future Prospects
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Koul, B.; Poonia, A.K.; Singh, R.; Kajla, S. Strategies to cope with the emerging waste water contaminants through adsorption regimes. In Development in Wastewater Treatment Research and Processes; Elsevier: Amsterdam, The Netherlands, 2022; pp. 61–106. [Google Scholar]
- Koul, B.; Sharma, K.; Shah, M.P. Phycoremediation: A sustainable alternative in wastewater treatment (WWT) regime. Environ. Technol. Innov. 2022, 25, 102040. [Google Scholar] [CrossRef]
- Irannezhad, M.; Ahmadi, B.; Liu, J.; Chen, D.; Matthews, J.H. Global water security: A shining star in the dark sky of achieving the sustainable development goals. Sustain. Horiz. 2022, 1, 100005. [Google Scholar] [CrossRef]
- Salehi, M. Global water shortage and potable water safety; Today’s concern and tomorrow’s crisis. Environ. Int. 2022, 158, 106936. [Google Scholar] [CrossRef] [PubMed]
- Harris, M. Philadelphia’s Water Supply: How Human Capital and Collaboration Can Overcome Significant Man-Made Challenges. Grad. Stud. J. Organ. Dyn. 2012, 2, 2. [Google Scholar]
- McGuire, M.J. Eight revolutions in the history of US drinking water disinfection. J. Am. Water Work. Assoc. 2006, 98, 123–149. [Google Scholar] [CrossRef]
- Akpor, O.B.; Otohinoyi, D.; Olaolu, D.; Aderiye, B. Pollutants in wastewater effluents: Impacts and remediation processes. Int. J. Environ. Res. Earth Sci. 2014, 3, 50–59. [Google Scholar]
- Ahmed, S.; Mofijur, M.; Nuzhat, S.; Chowdhury, A.T.; Rafa, N.; Uddin, M.A.; Inayat, A.; Mahlia, T.; Ong, H.C.; Chia, W.Y. Recent developments in physical, biological, chemical, and hybrid treatment techniques for removing emerging contaminants from wastewater. J. Hazard. Mater. 2021, 416, 125912. [Google Scholar] [CrossRef]
- Baladi, E.; Davar, F.; Hojjati-Najafabadi, A. Synthesis and characterization of g-C3N4-CoFe2O4-ZnO magnetic nanocomposites for enhancing photocatalytic activity with visible light for degradation of penicillin G antibiotic. Environ. Res. 2022, 215, 114270. [Google Scholar] [CrossRef]
- Hojjati-Najafabadi, A.; Mansoorianfar, M.; Liang, T.; Shahin, K.; Wen, Y.; Bahrami, A.; Karaman, C.; Zare, N.; Karimi-Maleh, H.; Vasseghian, Y. Magnetic-MXene-based nanocomposites for water and wastewater treatment: A review. J. Water Process Eng. 2022, 47, 102696. [Google Scholar] [CrossRef]
- Mansoorianfar, M.; Nabipour, H.; Pahlevani, F.; Zhao, Y.; Hussain, Z.; Hojjati-Najafabadi, A.; Hoang, H.Y.; Pei, R. Recent progress on adsorption of cadmium ion from water systems using metal-organic frameworks (MOFs) as an efficient class of porous materials. Environ. Res. 2022, 214, 114113. [Google Scholar] [CrossRef]
- Jain, M.; Khan, S.A.; Sharma, K.; Jadhao, P.R.; Pant, K.K.; Ziora, Z.M.; Blaskovich, M.A. Current perspective of innovative strategies for bioremediation of organic pollutants from wastewater. Bioresour. Technol. 2022, 344, 126305. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, C.; Van Buynder, P.; Lugg, R.; Blair, P.; Devine, B.; Cook, A.; Weinstein, P. Indirect potable reuse: A sustainable water supply alternative. Int. J. Environ. Res. Public Health 2009, 6, 1174–1209. [Google Scholar] [CrossRef] [PubMed]
- Costán-Longares, A.; Montemayor, M.; Payan, A.; Mendez, J.; Jofre, J.; Mujeriego, R.; Lucena, F. Microbial indicators and pathogens: Removal, relationships and predictive capabilities in water reclamation facilities. Water Res. 2008, 42, 4439–4448. [Google Scholar] [CrossRef] [PubMed]
- Asano, T.; Burton, F.; Leverenz, H. Water Reuse: Issues, Technologies, and Applications; McGraw-Hill Education: New York, NY, USA, 2007. [Google Scholar]
- Drewes, J.; Fox, P. Source water impact model (SWIM)-a watershed guided approach as a new planing tool for indirect potable water reuse. Water Sci. Technol. 2001, 43, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Bruckner, M. Measuring Dissolved and Particulate Organic Carbon (DOC and POC). In Microbial Life Educational Resources; Science Education Resource Center: Northfield, MN, USA, 2017; Available online: http://serc.carleton.edu/microbelife/research_methods/biogeochemical/organic_carbon.html (accessed on 1 September 2022).
- Namkung, E.; Rittmann, B.E. Soluble microbial products (SMP) formation kinetics by biofilms. Water Res. 1986, 20, 795–806. [Google Scholar] [CrossRef]
- Bradai, M.; Han, J.; Omri, A.E.; Funamizu, N.; Sayadi, S.; Isoda, H. Effect of linear alkylbenzene sulfonate (LAS) on human intestinal Caco-2 cells at non cytotoxic concentrations. Cytotechnology 2016, 68, 1267–1275. [Google Scholar] [CrossRef] [Green Version]
- Thomas, R.; Gough, R.; Freeman, C. Linear alkylbenzene sulfonate (LAS) removal in constructed wetlands: The role of plants in the treatment of a typical pharmaceutical and personal care product. Ecol. Eng. 2017, 106, 415–422. [Google Scholar] [CrossRef] [Green Version]
- Gleisberg, D.; Kandler, J.; Ulrich, H.; Hartz, P. Eutrophication and wastewater purification. Angew. Chem. Int. Ed. Engl. 1976, 15, 354–365. [Google Scholar] [CrossRef]
- Spietz, R.L.; Williams, C.M.; Rocap, G.; Horner-Devine, M.C. A dissolved oxygen threshold for shifts in bacterial community structure in a seasonally hypoxic estuary. PLoS ONE 2015, 10, e0135731. [Google Scholar] [CrossRef] [Green Version]
- Verma, N.; Singh, A.K. Development of biological oxygen demand biosensor for monitoring the fermentation industry effluent. Int. Sch. Res. Not. 2013, 2013, 236062. [Google Scholar] [CrossRef] [Green Version]
- Aniyikaiye, T.E.; Oluseyi, T.; Odiyo, J.O.; Edokpayi, J.N. Physico-chemical analysis of wastewater discharge from selected paint industries in Lagos, Nigeria. Int. J. Environ. Res. Public Health 2019, 16, 1235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahammad, S.Z.; Graham, D.W.; Dolfing, J. Wastewater treatment: Biological. In Managing Water Resources and Hydrological Systems; CRC Press: Boca Raton, FL, USA, 2020; pp. 561–576. [Google Scholar]
- Huang, M.-H.; Li, Y.-M.; Gu, G.-W. Chemical composition of organic matters in domestic wastewater. Desalination 2010, 262, 36–42. [Google Scholar] [CrossRef]
- Wichelns, D.; Drechsel, P.; Qadir, M. Wastewater: Economic asset in an urbanizing world. In Wastewater; Springer: Berlin/Heidelberg, Germany, 2015; pp. 3–14. [Google Scholar]
- Asano, T. Wastewater Reclamation and Reuse: Water Quality Management Library; CRC Press: Boca Raton, FL, USA, 1998; Volume 10. [Google Scholar]
- Verma, S. Anaerobic digestion of biodegradable organics in municipal solid wastes. Columbia Univ. 2002, 7, 98–104. [Google Scholar]
- Oller, I.; Gernjak, W.; Maldonado, M.; Fernandez-Ibanez, P.; Blanco, J.; Sanchez-Perez, J.; Malato, S. Degradation of the insecticide dimethoate by solar photocatalysis at pilot plant scale. Environ. Chem. Lett. 2005, 3, 118–121. [Google Scholar] [CrossRef]
- Jasim, N.A. The design for wastewater treatment plant (WWTP) with GPS X modelling. Cogent. Eng. 2020, 7, 1723782. [Google Scholar] [CrossRef]
- Shammas, N. Coagulation and Flocculation. In Physicochemical Treatment Processes. In Handbook of Environmental Engineering; Wang, L.K., Hung, Y.-T., Shammas, N.K., Eds.; Humana Press: Totowa, NJ, USA, 2005; Volume 3. [Google Scholar]
- da Rosa, J.J.; Rubio, J. The FF (flocculation–flotation) process. Miner. Eng. 2005, 18, 701–707. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef]
- Charerntanyarak, L. Heavy metals removal by chemical coagulation and precipitation. Water Sci. Technol. 1999, 39, 135–138. [Google Scholar] [CrossRef]
- Zinicovscaia, I. Conventional methods of wastewater treatment. In Cyanobacteria Bioremediation Wastewaters; Springer: Cham, Switzerland, 2016; pp. 17–25. Available online: https://link.springer.com/chapter/10.1007/978-3-319-26751-7_3 (accessed on 1 September 2022).
- Wang, L.K.; Vaccari, D.A.; Li, Y.; Shammas, N.K. Chemical Precipitation. In Physicochemical Treatment Processes; Springer: Berlin/Heidelberg, Germany, 2005; pp. 141–197. [Google Scholar]
- Stolzenburg, P.; Capdevielle, A.; Teychené, S.; Biscans, B. Struvite precipitation with MgO as a precursor: Application to wastewater treatment. Chem. Eng. Sci. 2015, 133, 9–15. [Google Scholar] [CrossRef]
- Ying, X.; Fang, Z. Experimental research on heavy metal wastewater treatment with dipropyl dithiophosphate. J. Hazard. Mater. 2006, 137, 1636–1642. [Google Scholar] [CrossRef]
- Chen, Q.; Luo, Z.; Hills, C.; Xue, G.; Tyrer, M. Precipitation of heavy metals from wastewater using simulated flue gas: Sequent additions of fly ash, lime and carbon dioxide. Water Res. 2009, 43, 2605–2614. [Google Scholar] [CrossRef] [PubMed]
- Burakov, A.E.; Galunin, E.V.; Burakova, I.V.; Kucherova, A.E.; Agarwal, S.; Tkachev, A.G.; Gupta, V.K. Adsorption of heavy metals on conventional and nanostructured materials for wastewater treatment purposes: A review. Ecotoxicol. Environ. Saf. 2018, 148, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Babel, S.; Kurniawan, T.A. Cr (VI) removal from synthetic wastewater using coconut shell charcoal and commercial activated carbon modified with oxidizing agents and/or chitosan. Chemosphere 2004, 54, 951–967. [Google Scholar] [CrossRef] [PubMed]
- Garg, V.; Gupta, R.; Kumar, R.; Gupta, R. Adsorption of chromium from aqueous solution on treated sawdust. Bioresour. Technol. 2004, 92, 79–81. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Qiu, G.; Song, W. Poultry litter-based activated carbon for removing heavy metal ions in water. Waste Manag. 2010, 30, 308–315. [Google Scholar] [CrossRef]
- Kongsuwan, A.; Patnukao, P.; Pavasant, P. Binary component sorption of Cu (II) and Pb (II) with activated carbon from Eucalyptus camaldulensis Dehn bark. J. Ind. Eng. Chem. 2009, 15, 465–470. [Google Scholar] [CrossRef]
- Yahaya, N.; Latiff, M.; Abustan, I.; Ahmad, M. Effect of preparation conditions of activated carbon prepared from rice husk by ZnCl2 activation for removal of Cu (II) from aqueous solution. Int. J. Eng. Technol. 2010, 10, 27–31. [Google Scholar]
- Verma, S.; Daverey, A.; Sharma, A. Slow sand filtration for water and wastewater treatment—A review. Environ. Technol. Rev. 2017, 6, 47–58. [Google Scholar] [CrossRef]
- Samer, M. Biological and chemical wastewater treatment processes. Wastewater Treat. Eng. 2015, 150, 212. [Google Scholar]
- EPA. Wastewater Technology Fact Sheet: Trickling Filter; EPA: Washington, DC, USA, 2000.
- Ramírez Calderón, O.A.; Abdeldayem, O.M.; Pugazhendhi, A.; Rene, E.R. Current updates and perspectives of biosorption technology: An alternative for the removal of heavy metals from wastewater. Curr. Pollut. Rep. 2020, 6, 8–27. [Google Scholar] [CrossRef]
- Spain, O.; Plöhn, M.; Funk, C. The cell wall of green microalgae and its role in heavy metal removal. Physiol. Plant. 2021, 173, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Giese, E.C. Biosorption as green technology for the recovery and separation of rare earth elements. World J. Microbiol. Biotechnol. 2020, 36, 52. [Google Scholar] [CrossRef] [PubMed]
- Sameera, V.; Naga Deepthi, C.; Srinu Babu, G.; Ravi Teja, Y. Role of biosorption in environmental cleanup. J. Microbial. Biochem. Technol. 2011, 1, 1–8. [Google Scholar]
- Coşgun, S.; Kara, B.; Kunt, B.; Hür, C.; Semerci, N. Biological recovery of phosphorus from waste activated sludge via alkaline fermentation and struvite biomineralization by Brevibacterium antiquum. Biodegradation 2022, 33, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Serdarevic, A.; Dzubur, A.; Muhibic, T. Role and Efficiency of MBR Technology for Wastewater Treatment. In International Symposium on Innovative and Interdisciplinary Applications of Advanced Technologies; Springer: Berlin/Heidelberg, Germany, 2019; pp. 229–237. [Google Scholar]
- Iorhemen, O.T.; Hamza, R.A.; Tay, J.H. Membrane Bioreactor (MBR) Technology for Wastewater Treatment and Reclamation: Membrane Fouling. Membranes 2016, 6, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kootenaei, F.G.; Aminirad, H. Membrane biological reactors (MBR) and their applications for water reuse. Int. J. Adv. Biol. Biomed. Res. 2014, 2, 2208–2216. [Google Scholar]
- Galán, B.; Castañeda, D.; Ortiz, I. Removal and recovery of Cr (VI) from polluted ground waters: A comparative study of ion-exchange technologies. Water Res. 2005, 39, 4317–4324. [Google Scholar] [CrossRef]
- Obotey Ezugbe, E.; Rathilal, S. Membrane technologies in wastewater treatment: A review. Membranes 2020, 10, 89. [Google Scholar] [CrossRef]
- Manimekalai, D.; Deepika, S.; Brema, K.; Rajeswari, C.; Juliet Selvarani, A. Microfiltration technology in waste water treatment. Int. J. Curr. Res. 2017, 9, 47310–47312. [Google Scholar]
- Chen, J.P.; Mou, H.; Wang, L.K.; Matsuura, T. Membrane Filtration. In Advanced Physicochemical Treatment Processes; Wang, L.K., Hung, Y.-T., Shammas, N.K., Eds.; Humana Press: Totowa, NJ, USA, 2006; pp. 203–259. [Google Scholar]
- Song, C.; Wang, T.; Pan, Y.; Qiu, J. Preparation of coal-based microfiltration carbon membrane and application in oily wastewater treatment. Sep. Purif. Technol. 2006, 51, 80–84. [Google Scholar] [CrossRef]
- Li, Y.S.; Yan, L.; Xiang, C.B.; Hong, L.J. Treatment of oily wastewater by organic–inorganic composite tubular ultrafiltration (UF) membranes. Desalination 2006, 196, 76–83. [Google Scholar] [CrossRef]
- Barakat, M.; Schmidt, E. Polymer-enhanced ultrafiltration process for heavy metals removal from industrial wastewater. Desalination 2010, 256, 90–93. [Google Scholar] [CrossRef]
- Jiang, M.; Ye, K.; Deng, J.; Lin, J.; Ye, W.; Zhao, S.; Van der Bruggen, B. Conventional ultrafiltration as effective strategy for dye/salt fractionation in textile wastewater treatment. Environ. Sci. Technol. 2018, 52, 10698–10708. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Boo, C.; Guo, N.; Wang, S.; Elimelech, M.; Wang, Y. Photocatalytic reactive ultrafiltration membrane for removal of antibiotic resistant bacteria and antibiotic resistance genes from wastewater effluent. Environ. Sci. Technol. 2018, 52, 8666–8673. [Google Scholar] [CrossRef]
- Qdais, H.A.; Moussa, H. Removal of heavy metals from wastewater by membrane processes: A comparative study. Desalination 2004, 164, 105–110. [Google Scholar] [CrossRef]
- Muro, C.; Riera, F.; del Carmen Díaz, M. Membrane separation process in wastewater treatment of food industry. In Food Industrial Processes–Methods and Equipment; InTech: Rijeka, Croatia, 2012; pp. 253–280. [Google Scholar]
- Yang, J.; Monnot, M.; Ercolei, L.; Moulin, P. Membrane-based processes used in municipal wastewater treatment for water reuse: State-of-the-art and performance analysis. Membranes 2020, 10, 131. [Google Scholar] [CrossRef]
- Smith, C., Jr. The use of ultrafiltration membrane for activated sludge separation. In Proceedings of the 24th Annual Purdue Industrial Waste Conference, Purdue University, 6–9 May 1969; pp. 1300–1310. [Google Scholar]
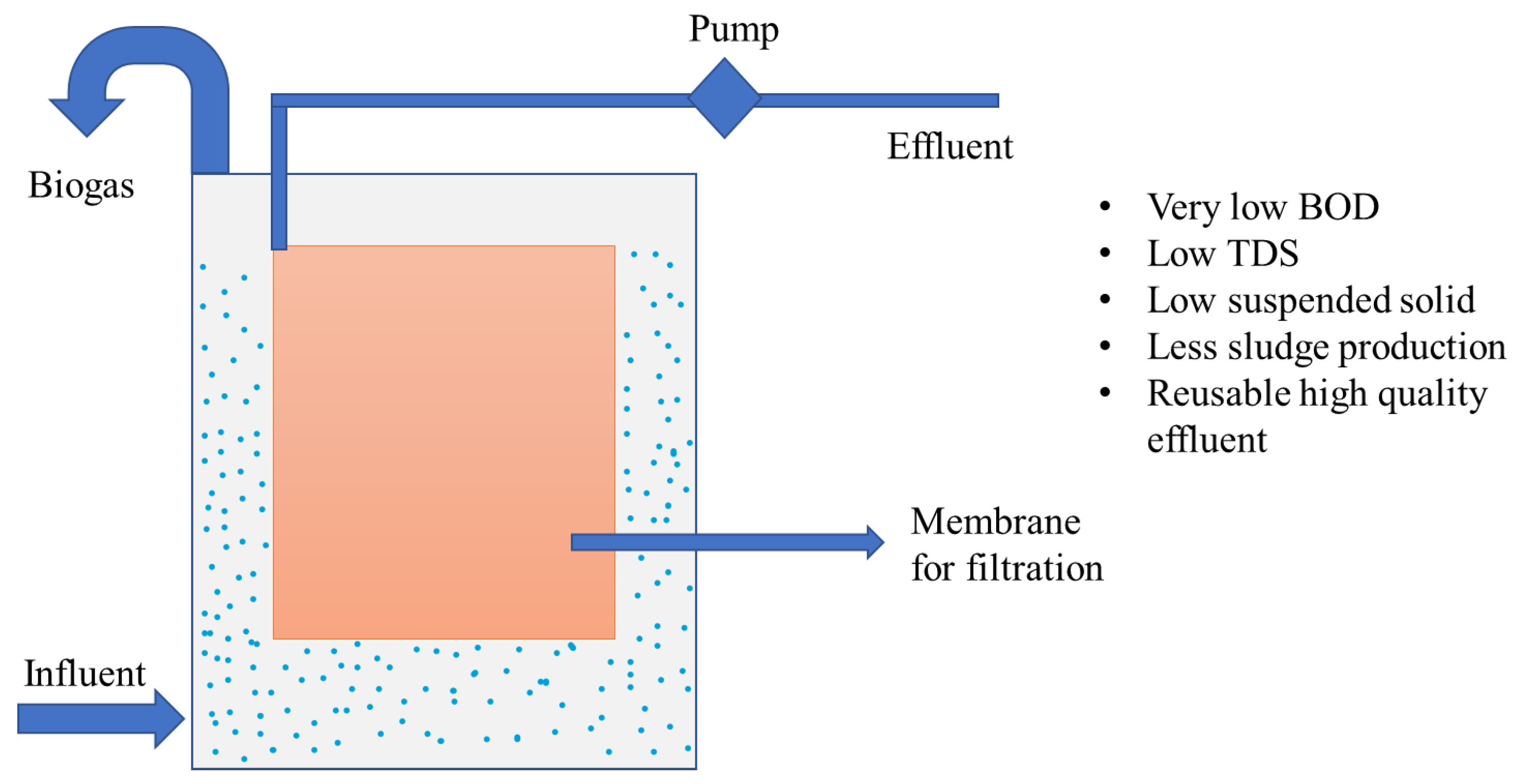
- Aslam, A.; Khan, S.J.; Shahzad, H.M.A. Anaerobic membrane bioreactors (AnMBRs) for municipal wastewater treatment-potential benefits, constraints, and future perspectives: An updated review. Sci. Total Environ. 2022, 802, 149612. [Google Scholar] [CrossRef]
- Tomczak, W.; Gryta, M. Energy-Efficient AnMBRs Technology for Treatment of Wastewaters: A Review. Energies 2022, 15, 4981. [Google Scholar] [CrossRef]
- Warsinger, D.M.; Chakraborty, S.; Tow, E.W.; Plumlee, M.H.; Bellona, C.; Loutatidou, S.; Karimi, L.; Mikelonis, A.M.; Achilli, A.; Ghassemi, A. A review of polymeric membranes and processes for potable water reuse. Prog. Polym. Sci. 2018, 81, 209–237. [Google Scholar] [CrossRef]
- Kehrein, P.; Van Loosdrecht, M.; Osseweijer, P.; Garfí, M.; Dewulf, J.; Posada, J. A critical review of resource recovery from municipal wastewater treatment plants–market supply potentials, technologies and bottlenecks. Environ. Sci. Water Res. Technol. 2020, 6, 877–910. [Google Scholar] [CrossRef] [Green Version]
- Peña, M.; do Nascimento, T.; Gouveia, J.; Escudero, J.; Gómez, A.; Letona, A.; Arrieta, J.; Fdz-Polanco, F. Anaerobic submerged membrane bioreactor (AnSMBR) treating municipal wastewater at ambient temperature: Operation and potential use for agricultural irrigation. Bioresour. Technol. 2019, 282, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Dvořák, L.; Gómez, M.; Dolina, J.; Černín, A. Anaerobic membrane bioreactors—A mini review with emphasis on industrial wastewater treatment: Applications, limitations and perspectives. Desalination Water Treat. 2016, 57, 19062–19076. [Google Scholar] [CrossRef] [Green Version]
- Li, G.; Xia, L.; Dong, J.; Chen, Y.; Li, Y. Metal-organic frameworks. In Solid-Phase Extraction; Elsevier: Amsterdam, The Netherlands, 2020; pp. 285–309. [Google Scholar]
- Mahmoud, M.E.; Amira, M.F.; Seleim, S.M.; Mohamed, A.K. Amino-decorated magnetic metal-organic framework as a potential novel platform for selective removal of chromium (Vl), cadmium (II) and lead (II). J. Hazard. Mater. 2020, 381, 120979. [Google Scholar] [CrossRef] [PubMed]
- Russo, V.; Hmoudah, M.; Broccoli, F.; Iesce, M.R.; Jung, O.-S.; Di Serio, M. Applications of metal organic frameworks in wastewater treatment: A review on adsorption and photodegradation. Front. Chem. Eng. 2020, 2, 581487. [Google Scholar] [CrossRef]
- Naguib, M.; Mochalin, V.N.; Barsoum, M.W.; Gogotsi, Y. 25th anniversary article: MXenes: A new family of two-dimensional materials. Adv. Mater. 2014, 26, 992–1005. [Google Scholar] [CrossRef]
- Rasool, K.; Helal, M.; Ali, A.; Ren, C.E.; Gogotsi, Y.; Mahmoud, K.A. Antibacterial activity of Ti3C2T x MXene. ACS Nano 2016, 10, 3674–3684. [Google Scholar] [CrossRef] [Green Version]
- Guo, L.; Wang, X.; Leong, Z.Y.; Mo, R.; Sun, L.; Yang, H.Y. Ar plasma modification of 2D MXene Ti3C2Tx nanosheets for efficient capacitive desalination. FlatChem 2018, 8, 17–24. [Google Scholar] [CrossRef]
- Chen, Z.; Xu, X.; Ding, Z.; Wang, K.; Sun, X.; Lu, T.; Konarova, M.; Eguchi, M.; Shapter, J.G.; Pan, L. Ti3C2 MXenes-derived NaTi2 (PO4) 3/MXene nanohybrid for fast and efficient hybrid capacitive deionization performance. Chem. Eng. J. 2021, 407, 127148. [Google Scholar] [CrossRef]
- Berkani, M.; Smaali, A.; Almomani, F.; Vasseghian, Y. Recent advances in MXene-based nanomaterials for desalination at water interfaces. Environ. Res. 2022, 203, 111845. [Google Scholar] [CrossRef]
- Feng, Y.; Yang, L.; Liu, J.; Logan, B.E. Electrochemical technologies for wastewater treatment and resource reclamation. Environ. Sci. Water Res. Technol. 2016, 2, 800–831. [Google Scholar] [CrossRef]
- Chahal, C.; Van Den Akker, B.; Young, F.; Franco, C.; Blackbeard, J.; Monis, P. Pathogen and particle associations in wastewater: Significance and implications for treatment and disinfection processes. Adv. Appl. Microbiol. 2016, 97, 63–119. [Google Scholar] [PubMed]
- Bouki, C.; Venieri, D.; Diamadopoulos, E. Detection and fate of antibiotic resistant bacteria in wastewater treatment plants: A review. Ecotoxicol. Environ. Saf. 2013, 91, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Anastasi, E.M.; Matthews, B.; Stratton, H.; Katouli, M. Pathogenic Escherichia coli found in sewage treatment plants and environmental waters. Appl. Environ. Microbiol. 2012, 78, 5536–5541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oluseyi Osunmakinde, C.; Selvarajan, R.; Mamba, B.B.; Msagati, T.A. Profiling bacterial diversity and potential pathogens in wastewater treatment plants using high-throughput sequencing analysis. Microorganisms 2019, 7, 506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bloomfield, S.F. Importance of disinfection as a means of prevention in our changing world hygiene and the home. GMS Krankenhhyg. Interdiszip. 2007, 2, Doc25. [Google Scholar] [PubMed]
- Koenraad, P.; Rombouts, F.; Notermans, S. Epidemiological aspects of thermophilic Campylobacter in water-related environments: A review. Water Environ. Res. 1997, 69, 52–63. [Google Scholar] [CrossRef]
- Kotilea, K.; Bontems, P.; Touati, E. Epidemiology, diagnosis and risk factors of Helicobacter pylori infection. Helicobacter Pylori Hum. Dis. 2019, 1149, 17–33. [Google Scholar]
- Jain, M.; Vadboncoeur, J.; Garg, S.J.; Biswas, J. Bacille calmette-guérin: An ophthalmic perspective. Surv. Ophthalmol. 2021, 67, 307–320. [Google Scholar] [CrossRef]
- Ajonina, C.; Buzie, C.; Möller, J.; Otterpohl, R. The detection of Entamoeba histolytica and Toxoplasma gondii in wastewater. J. Toxicol. Environ. Health Part A 2018, 81, 1–5. [Google Scholar] [CrossRef]
- Chaudhry, S.A.; Gad, N.; Koren, G. Toxoplasmosis and pregnancy. Can. Fam. Physician 2014, 60, 334–336. [Google Scholar]
- Grant, I.H.; Gold, J.; Rosenblum, M.; Niedzwiecki, D.; Armstrong, D. Toxoplasma gondii serology in HIV-infected patients: The development of central nervous system toxoplasmosis in AIDS. AIDS 1990, 4, 519–521. [Google Scholar] [CrossRef] [PubMed]
- Sturbaum, G.D.; Ortega, Y.R.; Gilman, R.H.; Sterling, C.R.; Cabrera, L.; Klein, D.A. Detection of Cyclospora cayetanensis in wastewater. Appl. Environ. Microbiol. 1998, 64, 2284–2286. [Google Scholar] [CrossRef] [PubMed]
- Behnia, M.; Hatam-Nahavandi, K.; Hajialilo, E.; Niyyati, M.; Tarighi, F.; Akram, A.B.; Salimi, M.; Rezaeian, M. Occurrence of Acanthamoeba genotypes in wastewater samples in Tehran, Iran. Iran. J. Parasitol. 2017, 12, 516. [Google Scholar] [PubMed]
- Medeiros, R.C.; Daniel, L.A.; de Oliveira, G.L.; Hoffmann, M.T. Performance of a small-scale wastewater treatment plant for removal of pathogenic protozoa (oo) cysts and indicator microorganisms. Environ. Technol. 2019, 40, 3492–3501. [Google Scholar] [CrossRef] [PubMed]
- Chorus, I.; Welker, M. Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; Taylor & Francis: London, UK, 2021. [Google Scholar] [CrossRef]
- Wiegand, C.; Pflugmacher, S. Ecotoxicological effects of selected cyanobacterial secondary metabolites a short review. Toxicol. Appl. Pharmacol. 2005, 203, 201–218. [Google Scholar] [CrossRef] [PubMed]
- Cox, P.; Banack, S.; Murch, S.; Rasmussen, U.; Tien, G.; Bidigare, R.; Metcalf, J.; Morrison, L.; Codd, G.; Bergman, B. Diverse taxa of cyanobacteria produce beta-N-methylamino-L-alanine, a neurotoxic amino acid. Proc. Natl. Acad. Sci. USA 2005, 102, 5074–5078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carmichael, W.W. Health effects of toxin-producing cyanobacteria: “The CyanoHABs”. Hum. Ecol. Risk Assess. Int. J. 2001, 7, 1393–1407. [Google Scholar] [CrossRef]
- Chen, Y.-T.; Chen, W.-R.; Liu, Z.-Q.; Lin, T.-F. Reaction pathways and kinetics of a cyanobacterial neurotoxin β-N-Methylamino-L-Alanine (BMAA) during chlorination. Environ. Sci. Technol. 2017, 51, 1303–1311. [Google Scholar] [CrossRef]
- Haddix, P.L.; Hughley, C.J.; Lechevallier, M.W. Occurrence of microcystins in 33 US water supplies. J. Am. Water Work. Assoc. 2007, 99, 118–125. [Google Scholar] [CrossRef]
- Rodríguez, E.; Onstad, G.D.; Kull, T.P.; Metcalf, J.S.; Acero, J.L.; von Gunten, U. Oxidative elimination of cyanotoxins: Comparison of ozone, chlorine, chlorine dioxide and permanganate. Water Res. 2007, 41, 3381–3393. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Abbà, A.; Benigna, I.; Sorlini, S.; Torretta, V. Overview of the main disinfection processes for wastewater and drinking water treatment plants. Sustainability 2017, 10, 86. [Google Scholar] [CrossRef] [Green Version]
- Galeano, L.-A.; Guerrero-Flórez, M.; Sánchez, C.-A.; Gil, A.; Vicente, M.-Á. Disinfection by chemical oxidation methods. In Applications of Advanced Oxidation Processes (AOPs) in Drinking Water Treatment. The Handbook of Environmental Chemistry; Springer: Cham, Switzerland, 2019; Volume 67, pp. 257–295. [Google Scholar]
- Glaze, W.H.; Kang, J.-W.; Chapin, D.H. The chemistry of water treatment processes involving ozone, hydrogen peroxide and ultraviolet radiation. Ozone Sci. Eng. 1987, 9, 335–352. [Google Scholar] [CrossRef]
- De Jager, T.L.; Cockrell, A.E.; Du Plessis, S.S. Ultraviolet Light Induced Generation of Reactive Oxygen Species. Adv. Exp. Med. Biol. 2017, 996, 15–23. [Google Scholar] [PubMed]
- Qadir, M.; Drechsel, P.; Jiménez Cisneros, B.; Kim, Y.; Pramanik, A.; Mehta, P.; Olaniyan, O. Global and Regional Potential of Wastewater as a Water, Nutrient and Energy Source; Wiley Online Library: Hoboken, NJ, USA, 2020; pp. 40–51. [Google Scholar]


| Characters | Microfiltration | Ultrafiltration | Nanofiltration | Reverse Osmosis | References |
|---|---|---|---|---|---|
| Molecular weight cut-off (Kilo Dalton) | 100–500 | 20–150 | 2–20 | 0.2–2 | [59] |
| Retained compounds | Colloids, TSS turbidity, some protozoan oocysts, cysts, some bacteria and viruses | Macromolecules, proteins, colloids, bacteria, viruses | mono-, di- and oligo-saccharides; polyvalent anions, pigments, sulphates, divalent cations, sodium chloride | Sodium, chloride, glucose, amino acids and sodium chloride | [59,68] |
| Transmembrane pressure (TMP) | <5 bar | <10 bar | <20 bar | <100 bar | [68] |
| Retained diameter particles (µm) | 10−1–10 | 10−3–10−1 | 10−3–10−2 | 10−4–10−3 | [59] |
| Flow modes | Crossflow, Dead-end | Crossflow, Dead-end | Crossflow | Crossflow | [69] |
| Membrane | Porous isotropic | Porous asymmetric | Finely porous asymmetric/composite | Nonporous asymmetric/composite | [69] |
| Cost Savings | Benefits to the Environment | Advantages of the Process | Operational Advantages |
|---|---|---|---|
| Reduced sludge production | Superior quality of effluent | Membrane fully removes solids from water flow | Reduced sludge handling is required |
| Reduced cost on sludge management | Less BOD | Large microbial biomass removes organics completely | Minimal operation charge and supervision |
| Less energy consumption | Reduced carbon footprint | Minimum supervision and care | Negligible maintenance |
| Reduced chemical requirement | Less harmful sludge | Can remove high organic content | |
| Biogas generation, may be used for energy production | Pathogen free effluent can be used in agriculture | Higher removal capacity of TDS |
| Waste Water Purification Techniques | Advantages | Disadvantages |
|---|---|---|
| Sedimentation |
|
|
| Coagulation and flocculation |
|
|
| Precipitation |
|
|
| Filtration |
|
|
| Adsorption |
|
|
| Trickling filter |
|
|
| Biosorption |
|
|
| Activated sludge processing (ASP) |
|
|
| Membrane bioreactor (MBR) |
|
|
| Ion-Exchange |
|
|
| Membrane filtration Microfiltration (MF) |
|
|
| Ultrafiltration (UF) |
|
|
| Nanofiltration (NF) |
|
|
| Reverse osmosis (RO) |
|
|
| Electrochemical treatments |
|
|
| Disinfection Chlorination |
|
|
| Ozonisation |
|
|
| UV |
|
|
| AOP |
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koul, B.; Yadav, D.; Singh, S.; Kumar, M.; Song, M. Insights into the Domestic Wastewater Treatment (DWWT) Regimes: A Review. Water 2022, 14, 3542. https://doi.org/10.3390/w14213542
Koul B, Yadav D, Singh S, Kumar M, Song M. Insights into the Domestic Wastewater Treatment (DWWT) Regimes: A Review. Water. 2022; 14(21):3542. https://doi.org/10.3390/w14213542
Chicago/Turabian StyleKoul, Bhupendra, Dhananjay Yadav, Swati Singh, Manoj Kumar, and Minseok Song. 2022. "Insights into the Domestic Wastewater Treatment (DWWT) Regimes: A Review" Water 14, no. 21: 3542. https://doi.org/10.3390/w14213542










