Photodegradation Behavior of Agricultural Antibiotic Oxytetracycline in Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Photodegradation Experiments
2.3. Analytical Methods
3. Results and Discussion
3.1. Effects of Irradiation Source, Light Intensity, Initial OTC Concentration, and Solution Temperature on OTC Degradation
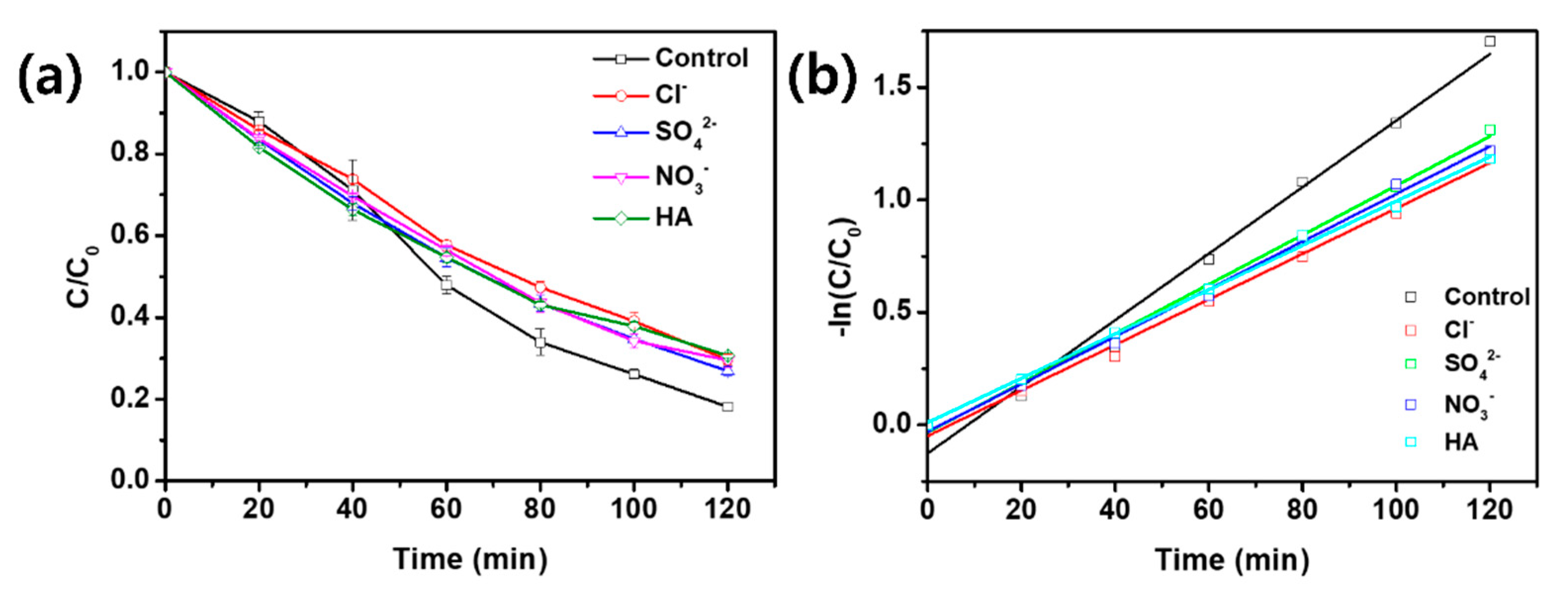
3.2. Effects of HA and Inorganic Ions on OTC Degradation
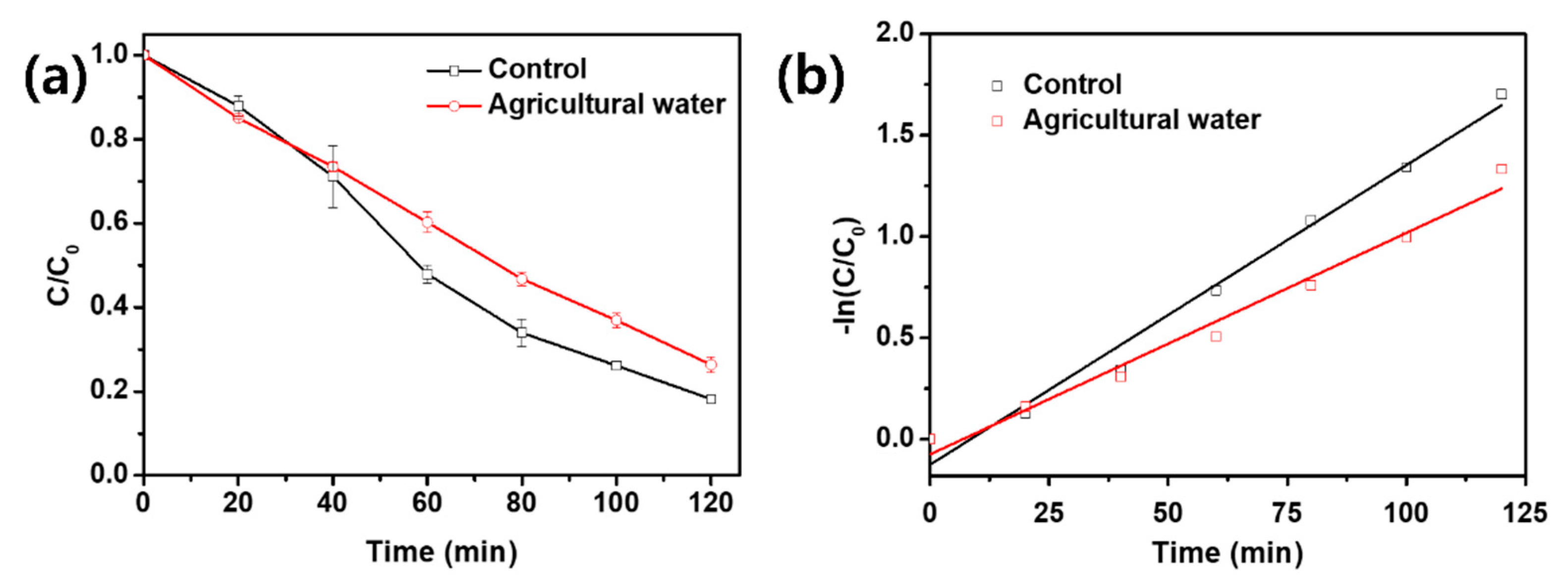
3.3. Degradation of OTC in Agricultural Water and Photolysis of Other Agricultural Antibiotics
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ge, L.; Zhang, P.; Halsall, C.; Li, Y.; Chen, C.E.; Li, J.; Sun, H.; Yao, Z. The importance of reactive oxygen species on the aqueous phototransformation of sulfonamide antibiotics: Kinetics, pathways, and comparisons with direct photolysis. Water Res. 2019, 149, 243–250. [Google Scholar] [CrossRef]
- Park, S.; Choi, K. Hazard assessment of commonly used agricultural antibiotics on aquatic ecosystems. Ecotoxicology 2008, 17, 526–538. [Google Scholar] [CrossRef] [PubMed]
- Chang, Q.; Wang, W.; Regev-Yochay, G.; Lipsitch, M.; Hanage, W.P. Antibiotics in agriculture and the risk to human health: How worried should we be? Evol. Appl. 2015, 8, 240–247. [Google Scholar] [CrossRef]
- Li, C.; Zhang, D.; Peng, J.; Li, X. The effect of pH, nitrate, iron (III) and bicarbonate on photodegradation of oxytetracycline in aqueous solution. J. Photochem. Photobiol. A Chem. 2018, 356, 239–247. [Google Scholar] [CrossRef]
- Song, C.; Liu, H.Y.; Guo, S.; Wang, S.G. Photolysis mechanisms of tetracycline under UV irradiation in simulated aquatic environment surrounding limestone. Chemosphere 2020, 244, 125582. [Google Scholar] [CrossRef]
- Hu, Y.; Yan, X.; Shen, Y.; Di, M.; Wang, J. Antibiotics in surface water and sediments from Hanjiang River, Central China: Occurrence, behavior and risk assessment. Ecotoxicol. Env. Saf 2018, 157, 150–158. [Google Scholar] [CrossRef]
- Yi, X.; Lin, C.; Ong, E.J.L.; Wang, M.; Zhou, Z. Occurrence and distribution of trace levels of antibiotics in surface waters and soils driven by non-point source pollution and anthropogenic pressure. Chemosphere 2019, 216, 213–223. [Google Scholar] [CrossRef]
- Li, Z.-J.; Qi, W.-N.; Feng, Y.; Liu, Y.-W.; Ebrahim, S.; Long, J. Degradation mechanisms of oxytetracycline in the environment. J. Integr. Agric. 2019, 18, 1953–1960. [Google Scholar] [CrossRef]
- Stankov, V.; Stankov, M.N.; Cvetnic, M.; Sigurnjak Bures, M.; Ukic, S.; Kucic Grgic, D.; Loncaric Bozic, A.; Kusic, H.; Bolanca, T. Environmental aspects of UV-C-based processes for the treatment of oxytetracycline in water. Env. Pollut 2021, 277, 116797. [Google Scholar] [CrossRef]
- Wu, M.; Que, C.; Tang, L.; Xu, H.; Xiang, J.; Wang, J.; Shi, W.; Xu, G. Distribution, fate, and risk assessment of antibiotics in five wastewater treatment plants in Shanghai, China. Env. Sci. Pollut. Res. Int. 2016, 23, 18055–18063. [Google Scholar] [CrossRef]
- Nivedhita, S.; Shyni Jasmin, P.; Sarvajith, M.; Nancharaiah, Y.V. Effects of oxytetracycline on aerobic granular sludge process: Granulation, biological nutrient removal and microbial community structure. Chemosphere 2022, 307, 136103. [Google Scholar] [CrossRef] [PubMed]
- Baena-Nogueras, R.M.; Gonzalez-Mazo, E.; Lara-Martin, P.A. Photolysis of Antibiotics under Simulated Sunlight Irradiation: Identification of Photoproducts by High-Resolution Mass Spectrometry. Env. Sci. Technol. 2017, 51, 3148–3156. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Li, H.; Mitch, W.A.; Tao, S.; Zhu, D. Enhanced Phototransformation of Tetracycline at Smectite Clay Surfaces under Simulated Sunlight via a Lewis-Base Catalyzed Alkalization Mechanism. Env. Sci. Technol. 2019, 53, 710–718. [Google Scholar] [CrossRef]
- Jin, X.; Xu, H.; Qiu, S.; Jia, M.; Wang, F.; Zhang, A.; Jiang, X. Direct photolysis of oxytetracycline: Influence of initial concentration, pH and temperature. J. Photochem. Photobiol. A Chem. 2017, 332, 224–231. [Google Scholar] [CrossRef]
- Rahmah, A.U.; Harimurti, S.; Murugesan, T. Experimental investigation on the effect of wastewater matrix on oxytetracycline mineralization using UV/H2O2 system. Int. J. Environ. Sci. Technol. 2017, 14, 1225–1233. [Google Scholar] [CrossRef]
- Liu, Y.; He, X.; Fu, Y.; Dionysiou, D.D. Kinetics and mechanism investigation on the destruction of oxytetracycline by UV-254nm activation of persulfate. J. Hazard. Mater. 2016, 305, 229–239. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, X.; Feng, S. Contribution of the Excited Triplet State of Humic Acid and Superoxide Radical Anion to Generation and Elimination of Phenoxyl Radical. Env. Sci. Technol. 2018, 52, 8283–8291. [Google Scholar] [CrossRef]
- Lee, Y.-J.; Lee, C.-G.; Park, S.-J.; Moon, J.-K.; Alvarez, P.J.J. pH-dependent contribution of chlorine monoxide radicals and byproducts formation during UV/chlorine treatment on clothianidin. Chem. Eng. J. 2022, 428, 132444. [Google Scholar] [CrossRef]
- Liu, Y.; He, X.; Duan, X.; Fu, Y.; Fatta-Kassinos, D.; Dionysiou, D.D. Significant role of UV and carbonate radical on the degradation of oxytetracycline in UV-AOPs: Kinetics and mechanism. Water Res. 2016, 95, 195–204. [Google Scholar] [CrossRef]
- Keen, O.S.; Love, N.G.; Linden, K.G. The role of effluent nitrate in trace organic chemical oxidation during UV disinfection. Water Res. 2012, 46, 5224–5234. [Google Scholar] [CrossRef]
- Hu, C.-Y.; Hou, Y.-Z.; Lin, Y.-L.; Li, A.-P.; Deng, Y.-G. Degradation kinetics of diatrizoate during UV photolysis and UV/chlorination. Chem. Eng. J. 2019, 360, 1003–1010. [Google Scholar] [CrossRef]
- Lee, Y.-J.; Kang, J.-K.; Park, S.-J.; Lee, C.-G.; Moon, J.-K.; Alvarez, P.J.J. Photocatalytic degradation of neonicotinoid insecticides using sulfate-doped Ag3PO4 with enhanced visible light activity. Chem. Eng. J. 2020, 402, 126183. [Google Scholar] [CrossRef]
- Seo, C.-D.; Lee, W.; Yoom, H.-S.; Choi, S.; Lee, Y.; Son, H. Evaluation and Prediction of Removal Efficiency of Pharmaceuticals in the Simulated O3 and UV/H2O2 Process for Drinking Water Treatment Process in the Downstream of Nakdong River. J. Korean Soc. Environ. Eng. 2021, 43, 491–503. [Google Scholar] [CrossRef]
- Van Doorslaer, X.; Demeestere, K.; Heynderickx, P.M.; Van Langenhove, H.; Dewulf, J. UV-A and UV-C induced photolytic and photocatalytic degradation of aqueous ciprofloxacin and moxifloxacin: Reaction kinetics and role of adsorption. Appl. Catal. B Environ. 2011, 101, 540–547. [Google Scholar] [CrossRef]
- Ding, Y.; Jiang, W.; Liang, B.; Han, J.; Cheng, H.; Haider, M.R.; Wang, H.; Liu, W.; Liu, S.; Wang, A. UV photolysis as an efficient pretreatment method for antibiotics decomposition and their antibacterial activity elimination. J. Hazard. Mater. 2020, 392, 122321. [Google Scholar] [CrossRef]
- Li, H.; Duan, L.; Wang, H.; Chen, Y.; Wang, F.; Zhang, S. Photolysis of sulfadiazine under UV radiation: Effects of the initial sulfadiazine concentration, pH, NO3− and Cd2+. Chem. Phys. Lett. 2020, 739, 136949. [Google Scholar] [CrossRef]
- Gomes, F.E.R.; Bergo, P.L.S.; Trap, M.A.; Spadoto, M.; Galinaro, C.A.; Rodrigues-Filho, E.; Leitao, A.; Tremiliosi-Filho, G. Photolysis of parabens using medium-pressure mercury lamps: Toxicity effects in MCF7, Balb/c 3T3 cells and Ceriodaphnia dubia. Chemosphere 2018, 208, 325–334. [Google Scholar] [CrossRef]
- Ding, S.L.; Wang, X.K.; Jiang, W.Q.; Zhao, R.S.; Shen, T.T.; Wang, C.; Wang, X. Influence of pH, inorganic anions, and dissolved organic matter on the photolysis of antimicrobial triclocarban in aqueous systems under simulated sunlight irradiation. Env. Sci. Pollut. Res. Int. 2015, 22, 5204–5211. [Google Scholar] [CrossRef]
- Acero, J.L.; Real, F.J.; Javier Benitez, F.; Matamoros, E. Degradation of neonicotinoids by UV irradiation: Kinetics and effect of real water constituents. Sep. Purif. Technol. 2019, 211, 218–226. [Google Scholar] [CrossRef]
- Remucal, C.K. The role of indirect photochemical degradation in the environmental fate of pesticides: A review. Environ. Sci. Processes Impacts 2014, 16, 628–653. [Google Scholar] [CrossRef]
- Kang, Y.M.; Kim, M.K.; Zoh, K.D. Effect of nitrate, carbonate/bicarbonate, humic acid, and H2O2 on the kinetics and degradation mechanism of Bisphenol-A during UV photolysis. Chemosphere 2018, 204, 148–155. [Google Scholar] [CrossRef]
- Xu, M.; Deng, J.; Cai, A.; Ma, X.; Li, J.; Li, Q.; Li, X. Comparison of UVC and UVC/persulfate processes for tetracycline removal in water. Chem. Eng. J. 2020, 384, 123320. [Google Scholar] [CrossRef]
- Giraldo, A.L.; Penuela, G.A.; Torres-Palma, R.A.; Pino, N.J.; Palominos, R.A.; Mansilla, H.D. Degradation of the antibiotic oxolinic acid by photocatalysis with TiO2 in suspension. Water Res. 2010, 44, 5158–5167. [Google Scholar] [CrossRef]


| Parameter | k (min−1) | R2 | Half-Life, t1/2 (min) | |||||
|---|---|---|---|---|---|---|---|---|
| Irradiation Source | Antibiotic | Number of Lamps | Solution Temperature (°C) | Initial Concentration (mg/L) | Coexisting Matter | |||
| Visible light | OTC | 4 | 25 | 100 | - | 0.0019 ± 0.0001 | 0.97 | 364.7 |
| UV-A | OTC | 4 | 25 | 100 | - | 0.0016 ± 0.0001 | 0.99 | 433.1 |
| UV-C | OTC | 4 | 25 | 100 | - | 0.0148 ± 0.0008 | 0.99 | 46.8 |
| UV-C | OTC | 2 | 25 | 100 | 0.0070 ± 0.0002 | 1.00 | 99.0 | |
| UV-C | OTC | 6 | 25 | 100 | 0.0207 ± 0.0012 | 0.98 | 33.5 | |
| UV-C | SM | 4 | 25 | 100 | - | 0.0044 ± 0.0003 | 0.97 | 157.5 |
| UV-C | VM | 4 | 25 | 100 | - | 0.0099 ± 0.0001 | 1.00 | 70.0 |
| UV-C | OA | 4 | 25 | 15 | - | 0.0065 ± 0.0002 | 1.00 | 106.6 |
| UV-C | OTC | 4 | 15 | 100 | - | 0.0062 ± 0.0003 | 0.98 | 111.8 |
| UV-C | OTC | 4 | 35 | 100 | - | 0.0141 ± 0.0001 | 1.00 | 49.1 |
| UV-C | OTC | 4 | 25 | 50 | - | 0.0172 ± 0.0011 | 0.97 | 40.3 |
| UV-C | OTC | 4 | 25 | 150 | - | 0.0095 ± 0.0003 | 1.00 | 72.9 |
| UV-C | OTC | 4 | 25 | 100 | Cl− | 0.0101 ± 0.0004 | 0.99 | 68.6 |
| UV-C | OTC | 4 | 25 | 100 | SO42− | 0.0110 ± 0.0002 | 1.00 | 63.0 |
| UV-C | OTC | 4 | 25 | 100 | NO3− | 0.0106 ± 0.0003 | 1.00 | 65.4 |
| UV-C | OTC | 4 | 25 | 100 | HA | 0.0098 ± 0.0002 | 1.00 | 70.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, Y.-J.; Lee, J.-M.; Lee, C.-G.; Park, S.-J.; Jho, E.H. Photodegradation Behavior of Agricultural Antibiotic Oxytetracycline in Water. Water 2022, 14, 3379. https://doi.org/10.3390/w14213379
Lee Y-J, Lee J-M, Lee C-G, Park S-J, Jho EH. Photodegradation Behavior of Agricultural Antibiotic Oxytetracycline in Water. Water. 2022; 14(21):3379. https://doi.org/10.3390/w14213379
Chicago/Turabian StyleLee, Youn-Jun, Jong-Min Lee, Chang-Gu Lee, Seong-Jik Park, and Eun Hea Jho. 2022. "Photodegradation Behavior of Agricultural Antibiotic Oxytetracycline in Water" Water 14, no. 21: 3379. https://doi.org/10.3390/w14213379
APA StyleLee, Y.-J., Lee, J.-M., Lee, C.-G., Park, S.-J., & Jho, E. H. (2022). Photodegradation Behavior of Agricultural Antibiotic Oxytetracycline in Water. Water, 14(21), 3379. https://doi.org/10.3390/w14213379









