Distribution and Driving Mechanism of N2O in Sea Ice and Its Underlying Seawater during Arctic Melt Season
Abstract
:1. Introduction
2. Materials and Methods
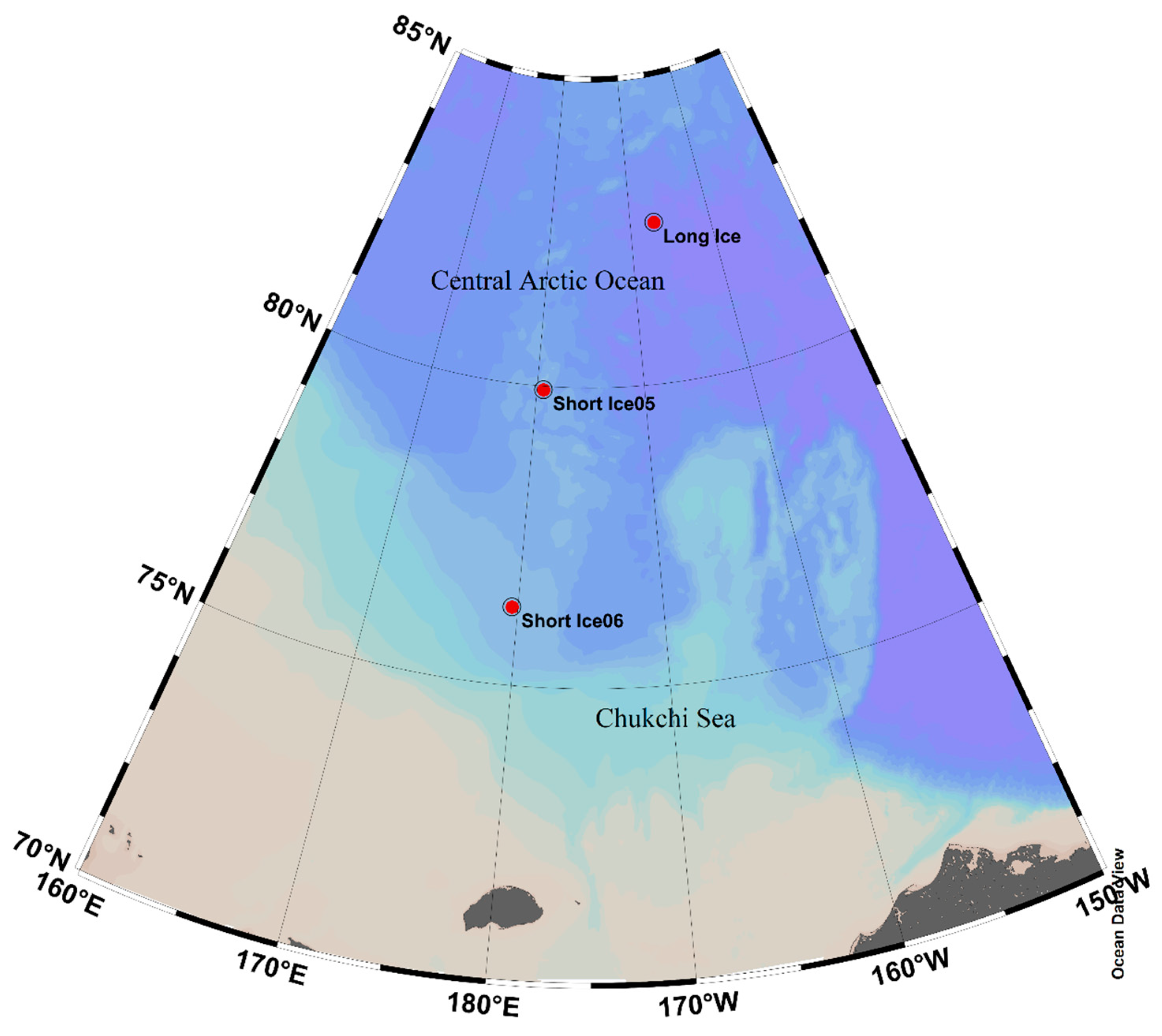
2.1. Study Area
2.2. Sampling and Processing
2.3. Laboratory Analysis
2.4. Computational Method
3. Results and Discussion
3.1. Structure of Sea Ice
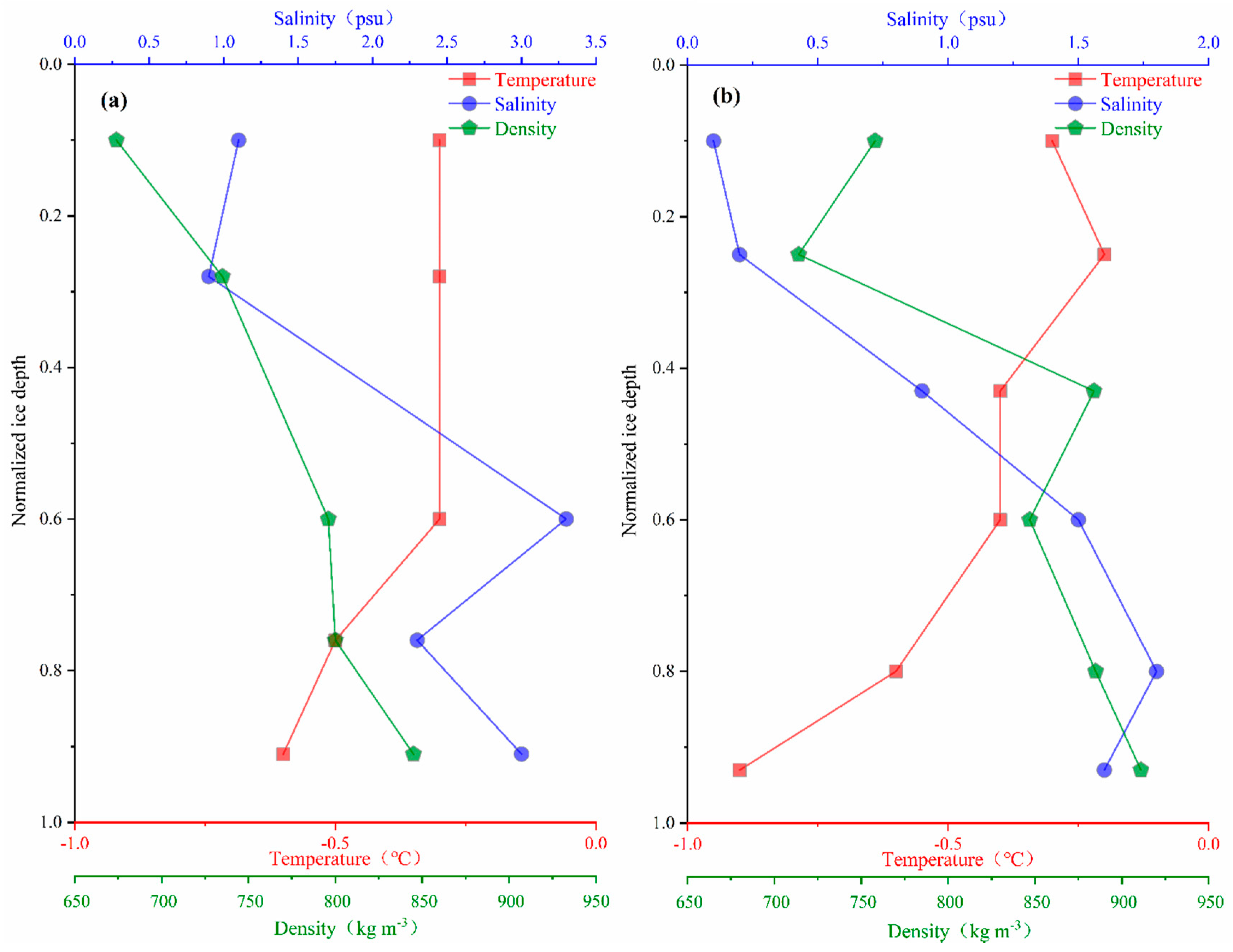
3.2. Temperature, Salinity, and Density Distributions of Sea Ice
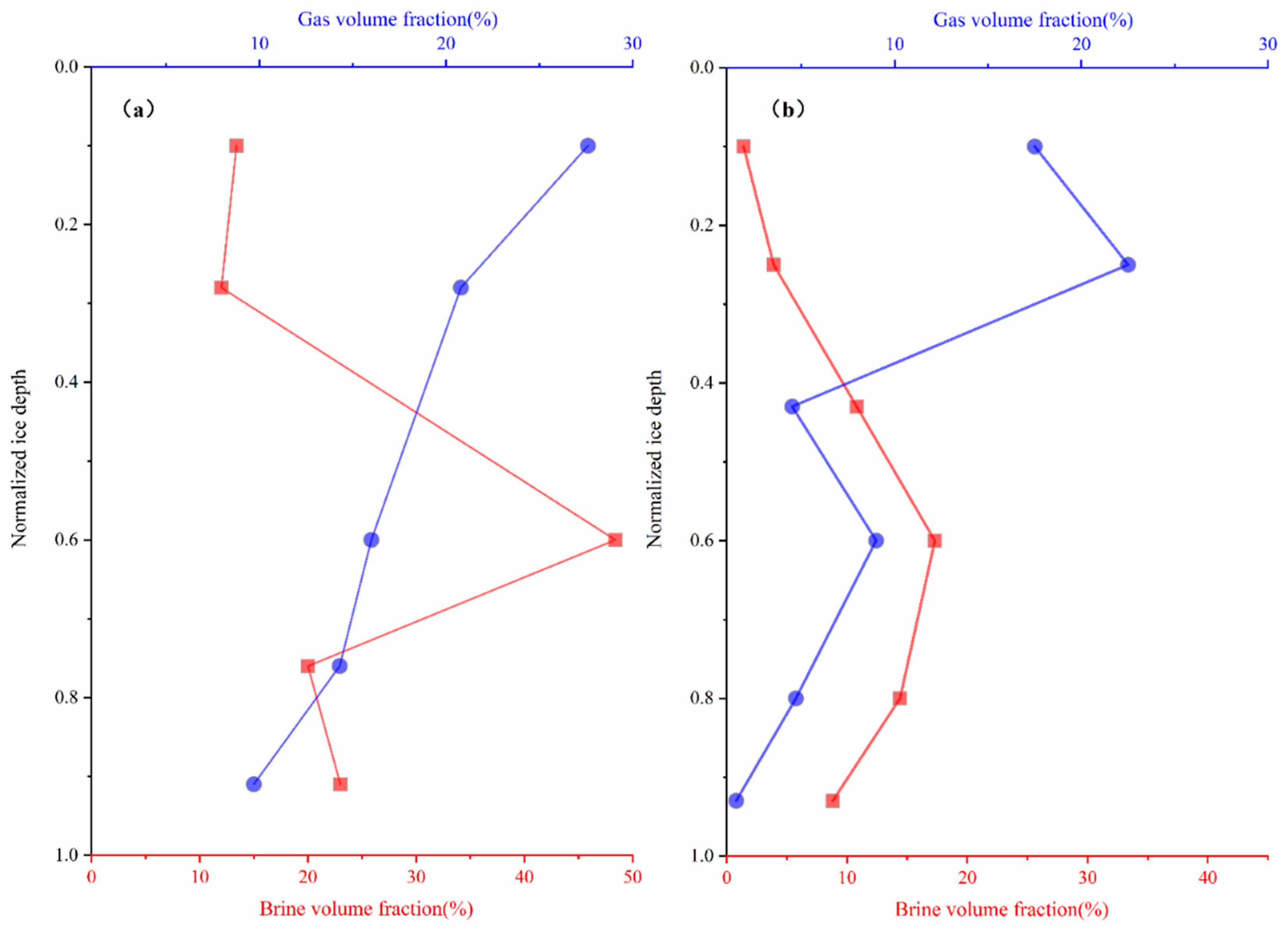
3.3. Sea Ice Brine and Gas Volume Fractions
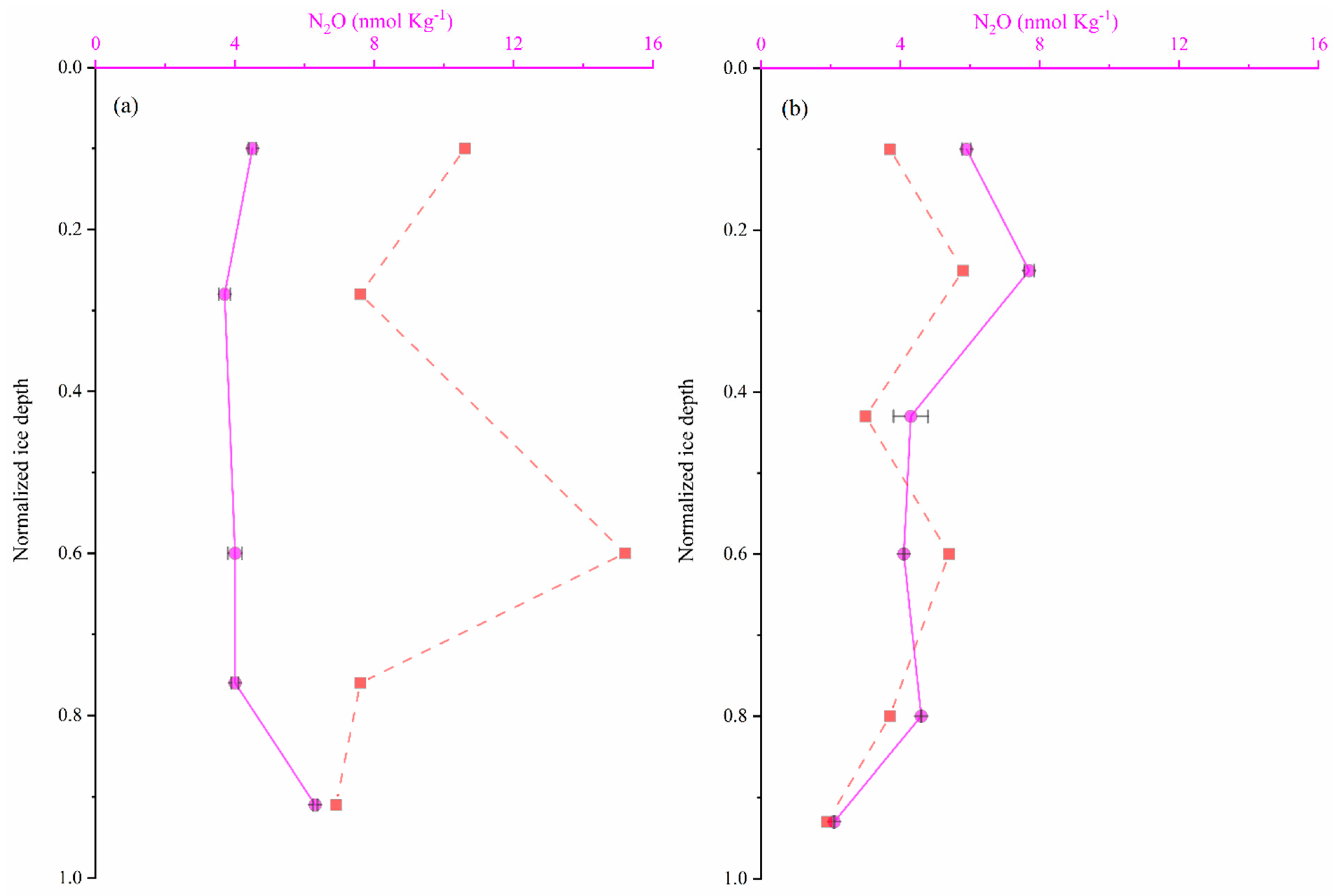
3.4. N2O Distribution in Sea Ice
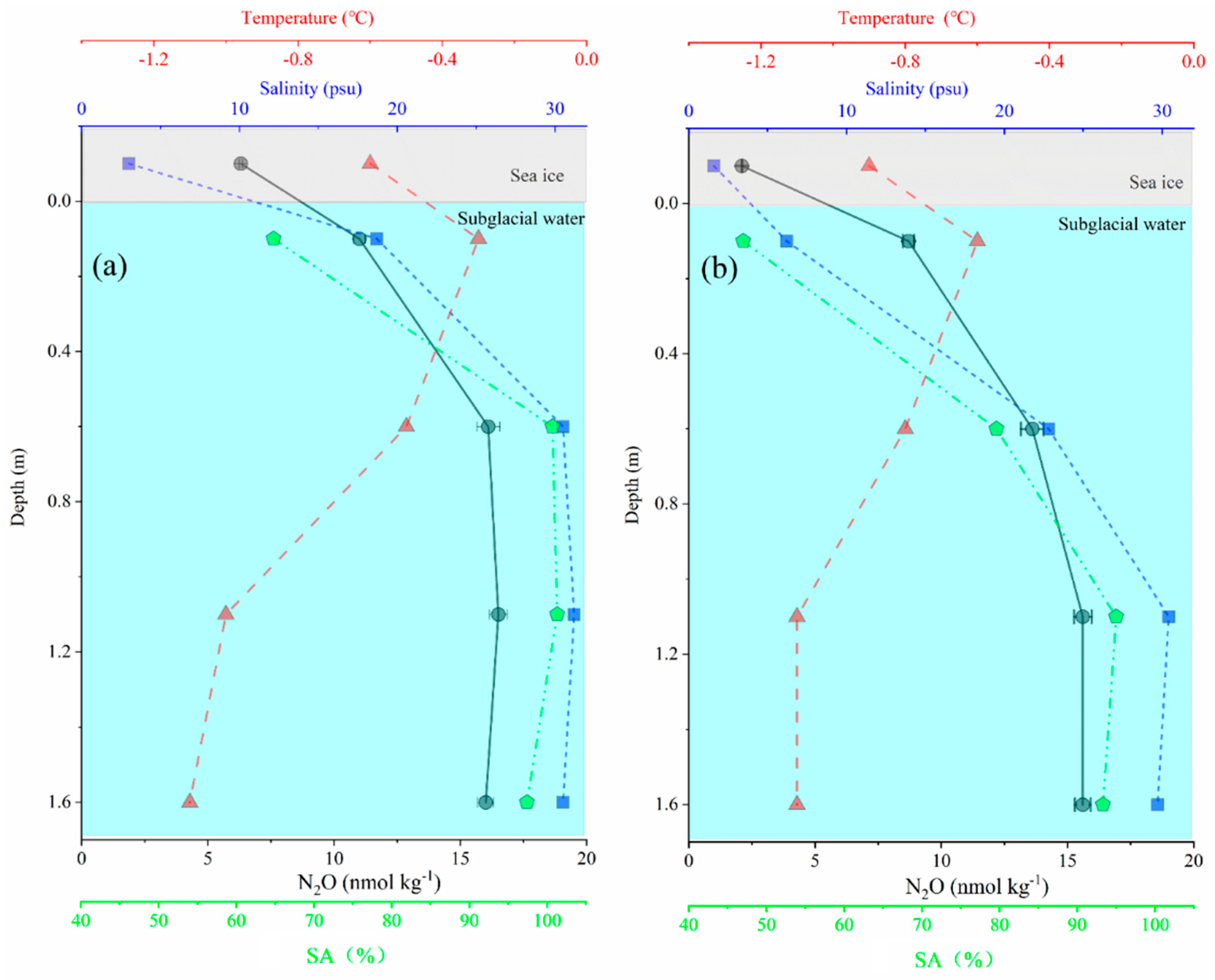
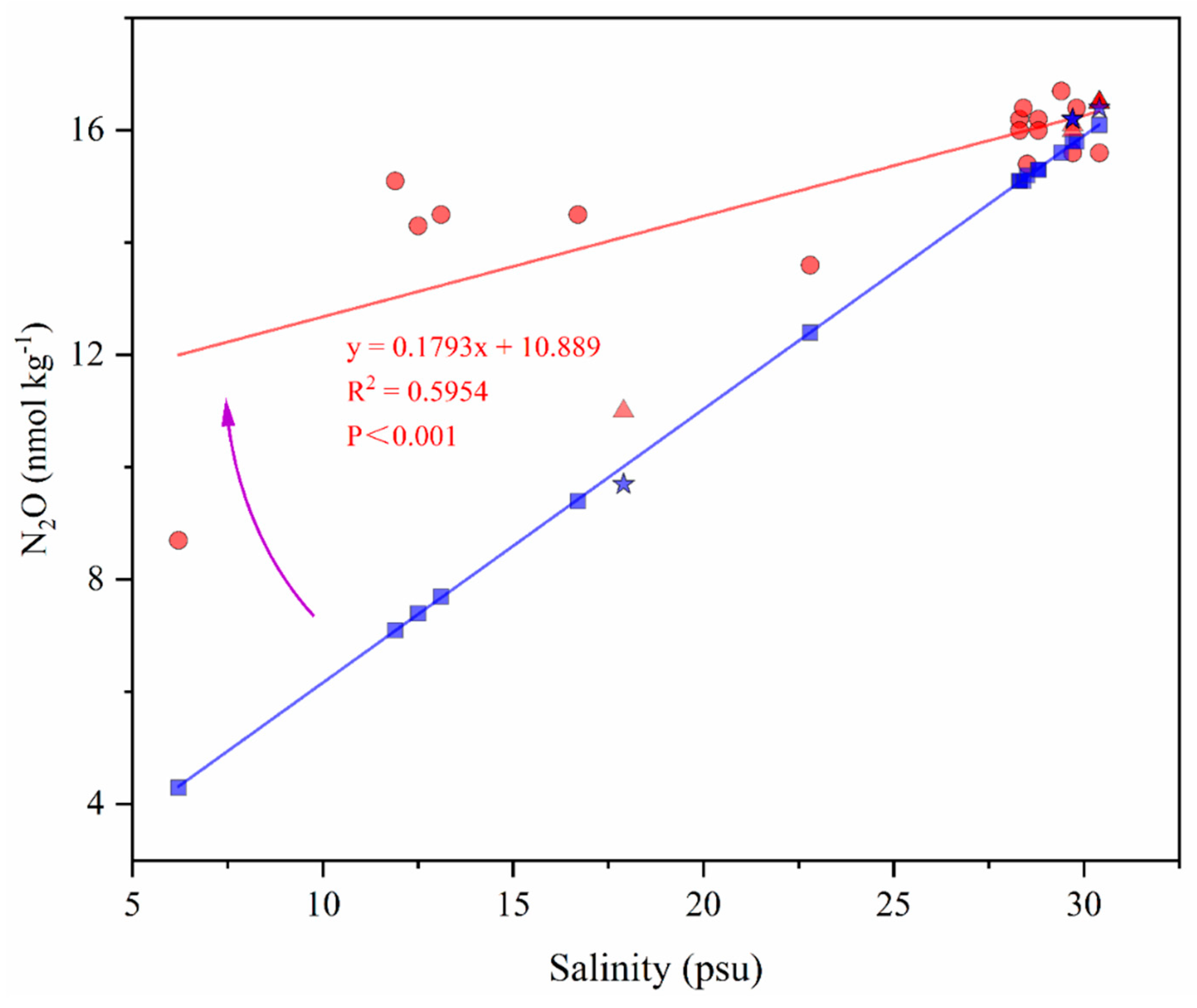
3.5. N2O Distribution in USW Influenced by Sea Ice Melting Rapidly
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, D.; Rojas, M.; Samset, B.H.; Cobb, K.; Niang, A.D.; Edwards, P.; Emori, S.; Faria, S.H.; Hawkins, E.; Hope, P.; et al. Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Available online: https://www.ipcc.ch/ (accessed on 6 September 2021).
- Tian, H.Q.; Xu, R.T.; Canadell, J.G.; Thompson, R.L.; Winiwarter, W.; Suntharalingam, P.; Davidson, E.A.; Ciais, P.; Jackson, R.B.; Janssens-Maenhout, G.; et al. A comprehensive quantification of global nitrous oxide sources and sinks. Nature 2020, 586, 248–256. [Google Scholar] [CrossRef]
- Harada, N. Review: Potential catastrophic reduction of sea ice in the western Arctic Ocean: Its impact on biogeochemical cycles and marine ecosystems. Glob. Planet. Chang. 2016, 136, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Vihma, T. Effects of arctic sea ice decline on weather and climate: A review. Surv. Geophys. 2014, 35, 1175–1214. [Google Scholar] [CrossRef] [Green Version]
- Meier, W.N.; Hovelsrud, G.K.; van Oort, B.E.H.; Key, J.R.; Kovacs, K.M.; Michel, C.; Haas, C.; Granskog, M.A.; Gerland, S.; Perovich, D.K.; et al. Arctic sea ice in transformation: A review of recent observed changes and impacts on biology and human activity. Rev. Geophys. 2014, 52, 185–217. [Google Scholar] [CrossRef]
- Zhan, L.Y.; Zhang, J.X.; Ouyang, Z.X.; Lei, R.B.; Xu, S.Q.; Qi, D.; Gao, Z.Y.; Sun, H.; Li, Y.H.; Wu, M.; et al. High-resolution distribution pattern of surface water nitrous oxide along a cruise track from the Okhotsk Sea to the western Arctic Ocean. Limnol. Oceanogr. 2020, 66, S401–S410. [Google Scholar] [CrossRef]
- Zhang, J.X.; Zhan, L.Y.; Chen, L.Q.; Li, Y.H.; Chen, J.F. Coexistence of nitrous oxide undersaturation and oversaturation in the surface and subsurface of the western Arctic Ocean. J. Geophys. Res.-Ocean. 2015, 120, 8392–8401. [Google Scholar] [CrossRef] [Green Version]
- Toyoda, S.; Kakimoto, T.; Kudo, K.; Yoshida, N.; Sasano, D.; Kosugi, N.; Ishii, M.; Kameyama, S.; Inagawa, M.; Yoshikawa-Inoue, H.; et al. Distribution and production mechanisms of N2O in the western Arctic Ocean. Glob. Biogeochem. Cycles 2021, 35, e2020GB006881. [Google Scholar] [CrossRef]
- Comiso, J.C.; Parkinson, C.L.; Gersten, R.; Stock, L. Accelerated decline in the Arctic Sea ice cover. Geophys. Res. Lett. 2008, 35, L01703. [Google Scholar] [CrossRef] [Green Version]
- Loose, B.; McGillis, W.R.; Schlosser, P.; Perovich, D.; Takahashi, T. Effects of freezing, growth, and ice cover on gas transport processes in laboratory seawater experiments. Geophys. Res. Lett. 2009, 36, L05603. [Google Scholar] [CrossRef]
- Delille, B.; Jourdain, B.; Borges, A.V.; Tison, J.L.; Delille, D. Biogas (CO2, O2, dimethylsulfide) dynamics in spring Antarctic fast ice. Limnol. Oceanogr. 2007, 52, 1367–1379. [Google Scholar] [CrossRef] [Green Version]
- Rysgaard, S.; Bendtsen, J.; Pedersen, L.T.; Ramlov, H.; Glud, R.N. Increased CO2 uptake due to sea ice growth and decay in the Nordic Seas. J. Geophys. Res.-Ocean. 2009, 114, C09011. [Google Scholar] [CrossRef]
- Randall, K.; Scarratt, M.; Levasseur, M.; Michaud, S.; Xie, H.X.; Gosselin, M. First measurements of nitrous oxide in Arctic sea ice. J. Geophys. Res.-Ocean. 2012, 117, 8. [Google Scholar] [CrossRef] [Green Version]
- Anderson, L.G.; Falck, E.; Jones, E.P.; Jutterstrom, S.; Swift, J.H. Enhanced uptake of atmospheric CO2 during freezing of seawater: A field study in Storfjorden, Svalbard. J. Geophys. Res.-Ocean. 2004, 109, C06004. [Google Scholar] [CrossRef]
- Kitidis, V.; Upstill-Goddard, R.C.; Anderson, L.G. Methane and nitrous oxide in surface water along the north-west passage, Arctic Ocean. Mar. Chem. 2010, 121, 80–86. [Google Scholar] [CrossRef]
- Yamamoto-Kawai, M.; McLaughlin, F.A.; Carmack, E.C.; Nishino, S.; Shimada, K. Aragonite undersaturation in the Arctic Ocean: Effects of ocean acidification and sea ice melt. Science 2009, 326, 1098–1100. [Google Scholar] [CrossRef] [Green Version]
- Thompson, D.W.J.; Wallace, J.M. The Arctic Oscillation signature in the wintertime geopotential height and temperature fields. Geophys. Res. Lett. 1998, 25, 1297–1300. [Google Scholar] [CrossRef] [Green Version]
- Proshutinsky, A.Y.; Johnson, M.A. Two circulation regimes of the wind driven Arctic Ocean. J. Geophys. Res.-Ocean. 1997, 102, 12493–12514. [Google Scholar] [CrossRef]
- Proshutinsky, A.Y.; Polyakov, I.V.; Johnson, M.A. Climate states and variability of Arctic ice and water dynamics during 1946–1997. Polar Res. 1999, 18, 135–142. [Google Scholar] [CrossRef]
- Dickson, B. Oceanography—All change in the Arctic. Nature 1999, 397, 389–391. [Google Scholar] [CrossRef] [Green Version]
- Serreze, M.C.; Holland, M.M.; Stroeve, J. Perspectives on the Arctic’s shrinking sea-ice cover. Science 2007, 315, 1533–1536. [Google Scholar] [CrossRef] [Green Version]
- Binder, H.; Boettcher, M.; Grams, C.M.; Joos, H.; Pfahl, S.; Wernli, H. Exceptional air mass transport and dynamical drivers of an extreme wintertime arctic warm event. Geophys. Res. Lett. 2017, 44, 12028–12036. [Google Scholar] [CrossRef] [Green Version]
- Stroeve, J.; Holland, M.M.; Meier, W.; Scambos, T.; Serreze, M. Arctic sea ice decline: Faster than forecast. Geophys. Res. Lett. 2007, 34, L09501. [Google Scholar] [CrossRef]
- Liu, J.P.; Song, M.R.; Horton, R.M.; Hu, Y.Y. Reducing spread in climate model projections of a September ice-free Arctic. Proc. Natl. Acad. Sci. USA 2013, 110, 12571–12576. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Lu, P.; Lepparanta, M.; Cheng, B.; Zhang, G.; Li, Z. Physical properties of summer sea ice in the pacific sector of the arctic during 2008–2018. J. Geophys. Res.-Ocean. 2020, 125, e2020JC016371. [Google Scholar] [CrossRef]
- Carnat, G.; Papakyriakou, T.; Geilfus, N.X.; Brabant, F.; Delille, B.; Vancoppenolle, M.; Gilson, G.; Zhou, J.Y.; Tison, J.L. Investigations on physical and textural properties of Arctic first-year sea ice in the Amundsen Gulf, Canada, November 2007–June 2008 (IPY-CFL system study). J. Glaciol. 2013, 59, 819–837. [Google Scholar] [CrossRef] [Green Version]
- Eicken, H.; Bluhm, B.A.; Collins, R.E.; Haas, C.; Ingham, M.; Gradinger, R.; Mahoney, A.; Nicolaus, M.; Perovich, D.K. Field Cold Regions Science and Marine Technology, Encyclopedia of Life Support Systems (EOLSS); Techniques in Sea-Ice Research; UNESCO, EOLSS Publishers: Prais, France, 2014. [Google Scholar]
- Zhan, L.-Y.; Chen, L.-Q.; Zhang, J.-X.; Lin, Q. A system for the automated static headspace analysis of dissolved N2O in seawater. Int. J. Environ. Anal. Chem. 2013, 93, 828–842. [Google Scholar] [CrossRef]
- Weiss, R.F.; Price, B.A. Nitrous oxide solubility in water and seawater. Mar. Chem. 1980, 8, 347–359. [Google Scholar] [CrossRef]
- Leppäranta, M.; Manninen, T. The Brine and Gas Content of Sea Ice with Attention to Low Salinities and High Temperatures; Finnish Institute of Marine Research: Helsinki, Finland, 1988. [Google Scholar]
- Worster, M.G.; Wettlaufer, J.S. Natural convection, solute trapping, and channel formation during solidification of saltwater. J. Phys. Chem. B 1997, 101, 6132–6136. [Google Scholar] [CrossRef] [Green Version]
- Nakawo, M.; Sinha, N.K. Growth-rate and salinity profile of 1st-year sea ice in the high arctic. J. Glaciol. 1981, 27, 315–330. [Google Scholar] [CrossRef] [Green Version]
- Tucker, W.B.; Gow, A.J.; Weeks, W.F. Physical-properties of summer sea ice in the fram strait. J. Geophys. Res.-Ocean. 1987, 92, 6787–6803. [Google Scholar] [CrossRef]
- Cottier, F.; Eicken, H.; Wadhams, P. Linkages between salinity and brine channel distribution in young sea ice. J. Geophys. Res.-Ocean. 1999, 104, 15859–15871. [Google Scholar] [CrossRef] [Green Version]
- Vancoppenolle, M.; Fichefet, T.; Bitz, C.M. Modeling the salinity profile of undeformed Arctic sea ice. Geophys. Res. Lett. 2006, 33, 5. [Google Scholar] [CrossRef]
- Overgaard, S.; Wadhams, P.; Lepparanta, M. Ice properties in the Greenland and Barents seas during summer. J. Glaciol. 1983, 29, 142–164. [Google Scholar] [CrossRef] [Green Version]
- Cheng, B.; Zhang, Z.H.; Vihma, T.; Johansson, M.; Bian, L.G.; Li, Z.J.; Wu, H.D. Model experiments on snow and ice thermodynamics in the Arctic Ocean with CHINARE 2003 data. J. Geophys. Res.-Ocean. 2008, 113, C09020. [Google Scholar] [CrossRef] [Green Version]
- Light, B.; Maykut, G.A.; Grenfell, T.C. A temperature-dependent, structural-optical model of first-year sea ice. J. Geophys. Res.-Ocean. 2004, 109, C06013. [Google Scholar] [CrossRef]
- Cox, G.F.N.; Weeks, W.F. Equations for determining the gas and brine volumes in sea-ice samples. J. Glaciol. 1983, 29, 306–316. [Google Scholar] [CrossRef] [Green Version]
- Crabeck, O.; Galley, R.; Delille, B.; Else, B.; Geilfus, N.X.; Lemes, M.; Des Roches, M.; Francus, P.; Tison, J.L.; Rysgaard, S. Imaging air volume fraction in sea ice using non-destructive X-ray tomography. Cryosphere 2016, 10, 1125–1145. [Google Scholar] [CrossRef] [Green Version]
- Killawee, J.A.; Fairchild, I.J.; Tison, J.L.; Janssens, L.; Lorrain, R. Segregation of solutes and gases in experimental freezing of dilute solutions: Implications for natural glacial systems. Geochim. Cosmochim. Acta 1998, 62, 3637–3655. [Google Scholar] [CrossRef]
- Tison, J.L.; Haas, C.; Gowing, M.M.; Sleewaegen, S.; Bernard, A. Tank study of physico-chemical controls on gas content and composition during growth of young sea ice. J. Glaciol. 2002, 48, 177–191. [Google Scholar] [CrossRef] [Green Version]
- Zhou, J.Y.; Delille, B.; Eicken, H.; Vancoppenolle, M.; Brabant, F.; Carnat, G.; Geilfus, N.X.; Papakyriakou, T.; Heinesch, B.; Tison, J.L. Physical and biogeochemical properties in landfast sea ice (Barrow, Alaska): Insights on brine and gas dynamics across seasons. J. Geophys. Res.-Ocean. 2013, 118, 3172–3189. [Google Scholar] [CrossRef]
- Eicken, H.; Krouse, H.R.; Kadko, D.; Perovich, D.K. Tracer studies of pathways and rates of meltwater transport through Arctic summer sea ice. J. Geophys. Res.-Ocean. 2002, 107, SHE 22-1–SHE 22-20. [Google Scholar] [CrossRef] [Green Version]
- Golden, K.M.; Eicken, H.; Heaton, A.L.; Miner, J.; Pringle, D.J.; Zhu, J. Thermal evolution of permeability and microstructure in sea ice. Geophys. Res. Lett. 2007, 34, L16501. [Google Scholar] [CrossRef] [Green Version]
- Golden, K.M.; Ackley, S.F.; Lytle, V.I. The percolation phase transition in sea ice. Science 1998, 282, 2238–2241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreau, S.; Vancoppenolle, M.; Zhou, J.Y.; Tison, J.L.; Delille, B.; Goosse, H. Modelling argon dynamics in first-year sea ice. Ocean Model. Online 2014, 73, 1–18. [Google Scholar] [CrossRef]
- Marie, K. CO2 and N2O Dynamics in the Ocean–Sea Ice–Atmosphere System; Université de Lièg: Liège, Belgium, 2019. [Google Scholar]
- Rysgaard, S.; Glud, R.N. Anaerobic N-2 production in Arctic sea ice. Limnol. Oceanogr. 2004, 49, 86–94. [Google Scholar] [CrossRef]
- Rysgaard, S.; Glud, R.N.; Sejr, M.K.; Blicher, M.E.; Stahl, H.J. Denitrification activity and oxygen dynamics in Arctic sea ice. Polar Biol. 2008, 31, 527–537. [Google Scholar] [CrossRef]
- Nomura, D.; Yoshikawa-Inoue, H.; Toyota, T. The effect of sea-ice growth on air-sea CO2 flux in a tank experiment. Tellus Ser. B-Chem. Phys. Meteorol. 2006, 58, 418–426. [Google Scholar] [CrossRef]
- Kaartokallio, H. Evidence for active microbial nitrogen transformations in sea ice (Gulf of Bothnia, Baltic Sea) in midwinter. Polar Biol. 2001, 24, 21–28. [Google Scholar] [CrossRef]
- Boisvert, L.N.; Webster, M.A.; Petty, A.A.; Markus, T.; Bromwich, D.H.; Cullather, R.I. Intercomparison of precipitation estimates over the arctic ocean and its peripheral seas from reanalyses. J. Clim. 2018, 31, 8441–8462. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Zhan, L.; Wang, Q.; Wu, M.; Ye, W.; Zhang, J.; Li, Y.; Wen, J.; Chen, L. Distribution and Driving Mechanism of N2O in Sea Ice and Its Underlying Seawater during Arctic Melt Season. Water 2022, 14, 145. https://doi.org/10.3390/w14020145
Liu J, Zhan L, Wang Q, Wu M, Ye W, Zhang J, Li Y, Wen J, Chen L. Distribution and Driving Mechanism of N2O in Sea Ice and Its Underlying Seawater during Arctic Melt Season. Water. 2022; 14(2):145. https://doi.org/10.3390/w14020145
Chicago/Turabian StyleLiu, Jian, Liyang Zhan, Qingkai Wang, Man Wu, Wangwang Ye, Jiexia Zhang, Yuhong Li, Jianwen Wen, and Liqi Chen. 2022. "Distribution and Driving Mechanism of N2O in Sea Ice and Its Underlying Seawater during Arctic Melt Season" Water 14, no. 2: 145. https://doi.org/10.3390/w14020145





