Comparison of Microbial Profiling and Tracer Testing for the Characterization of Injector-Producer Interwell Connectivities
Abstract
1. Introduction
2. Materials and Methods
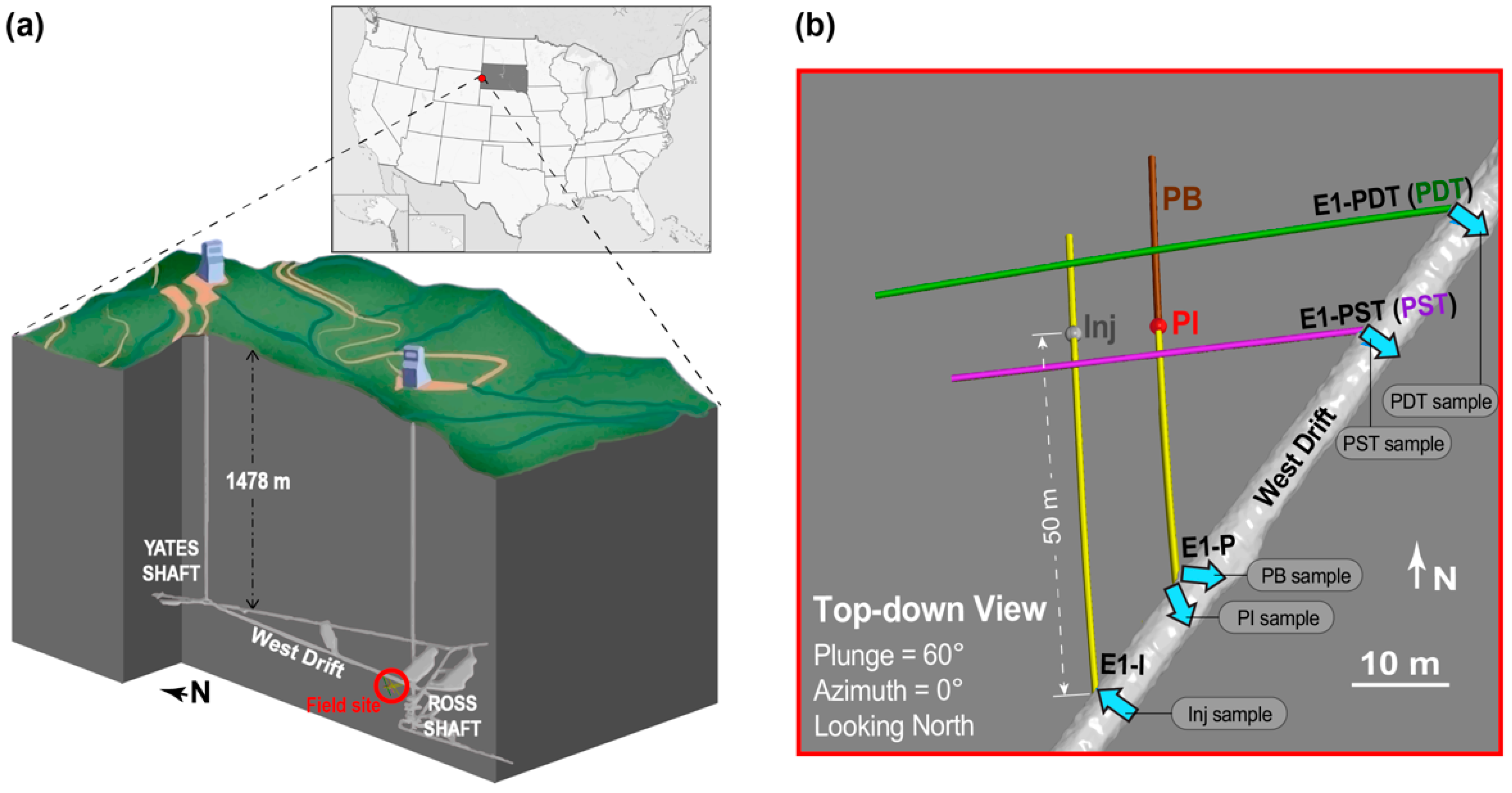
2.1. Enhanced Geothermal Systems (EGS) Collab Project Experiment 1: Field Site Description and Long-Term Flow Test
2.2. Conservative Tracer Tests and Collection of Microbial Samples
2.3. Genomic DNA Extraction, Library Preparation and 16S Ribosomal RNA (rRNA) Gene Amplicon Sequencing
2.4. High-Throughput Sequencing Data Processing
2.5. Diversity and Statistical Analyses
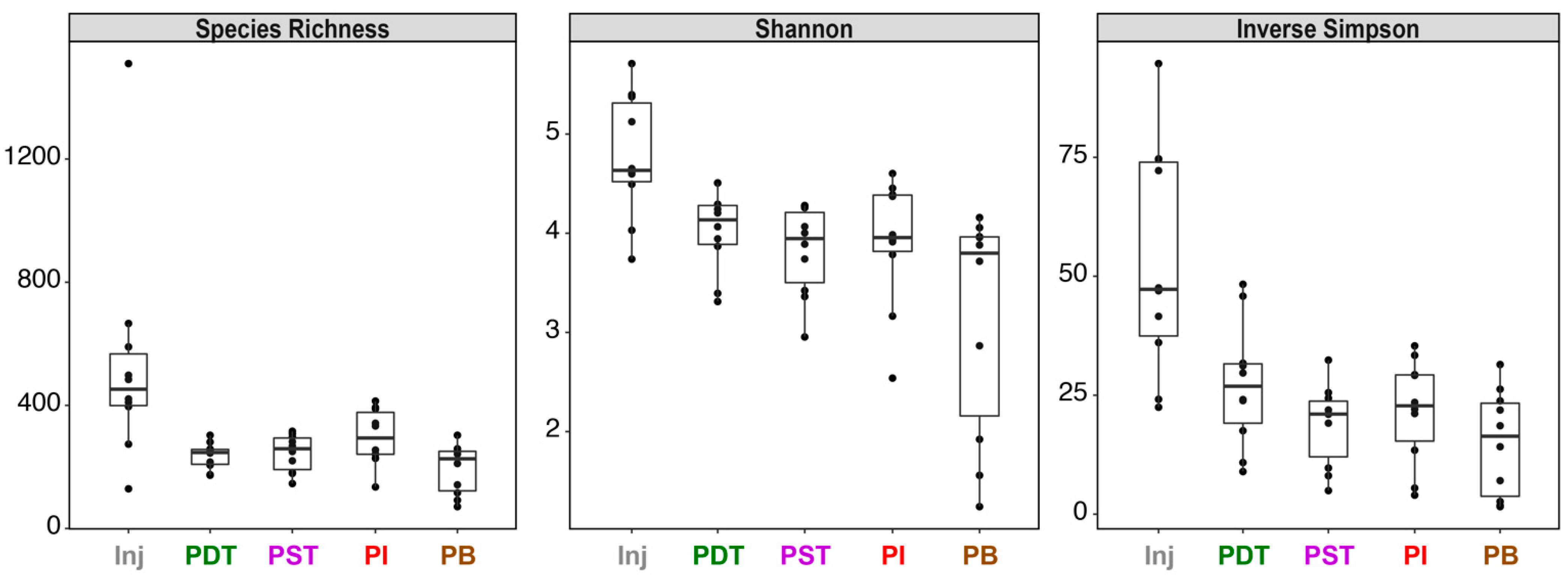
3. Results
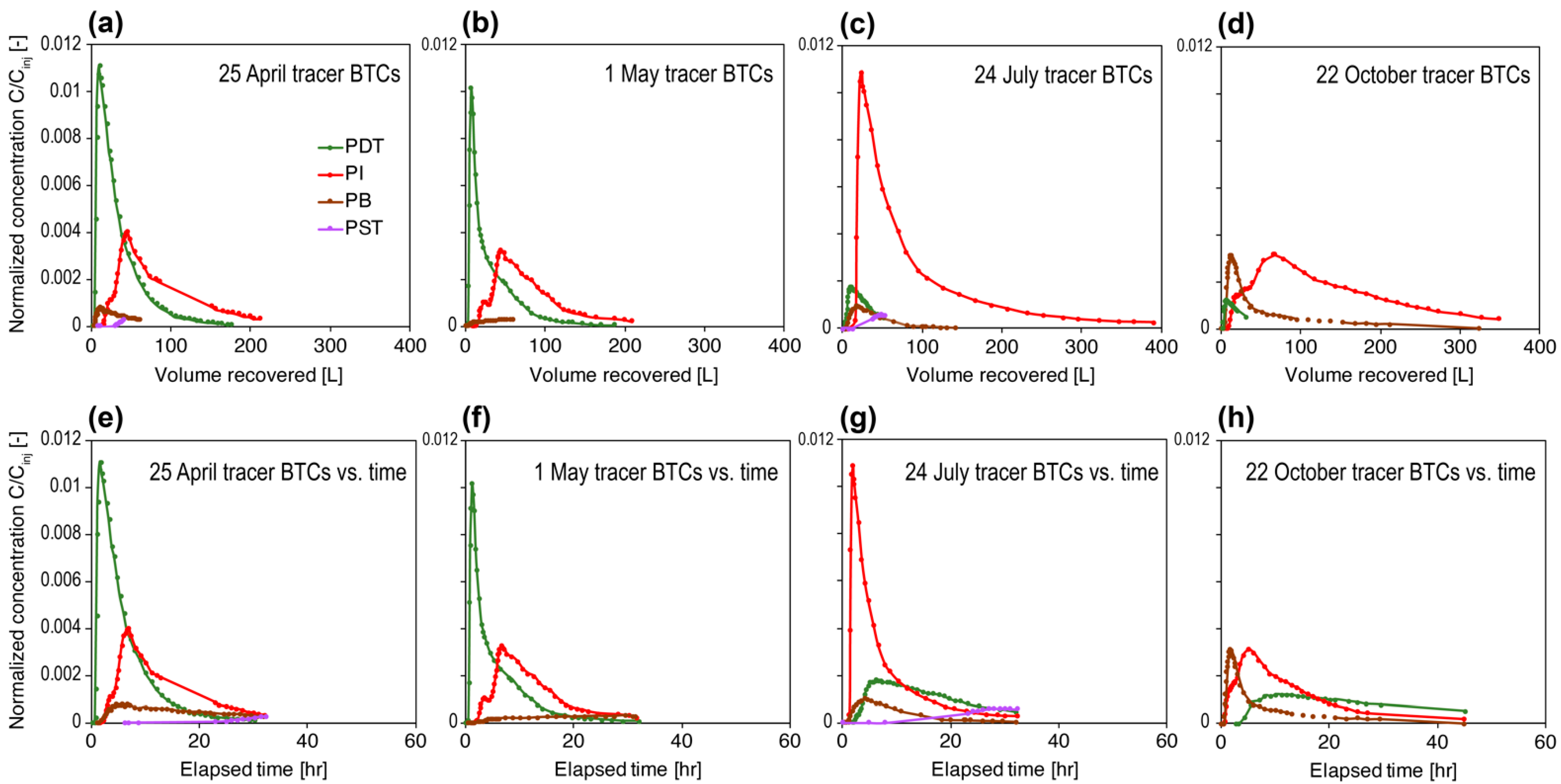
3.1. Long-Term Flow Test, Overall Geochemistry, and Tracer Data
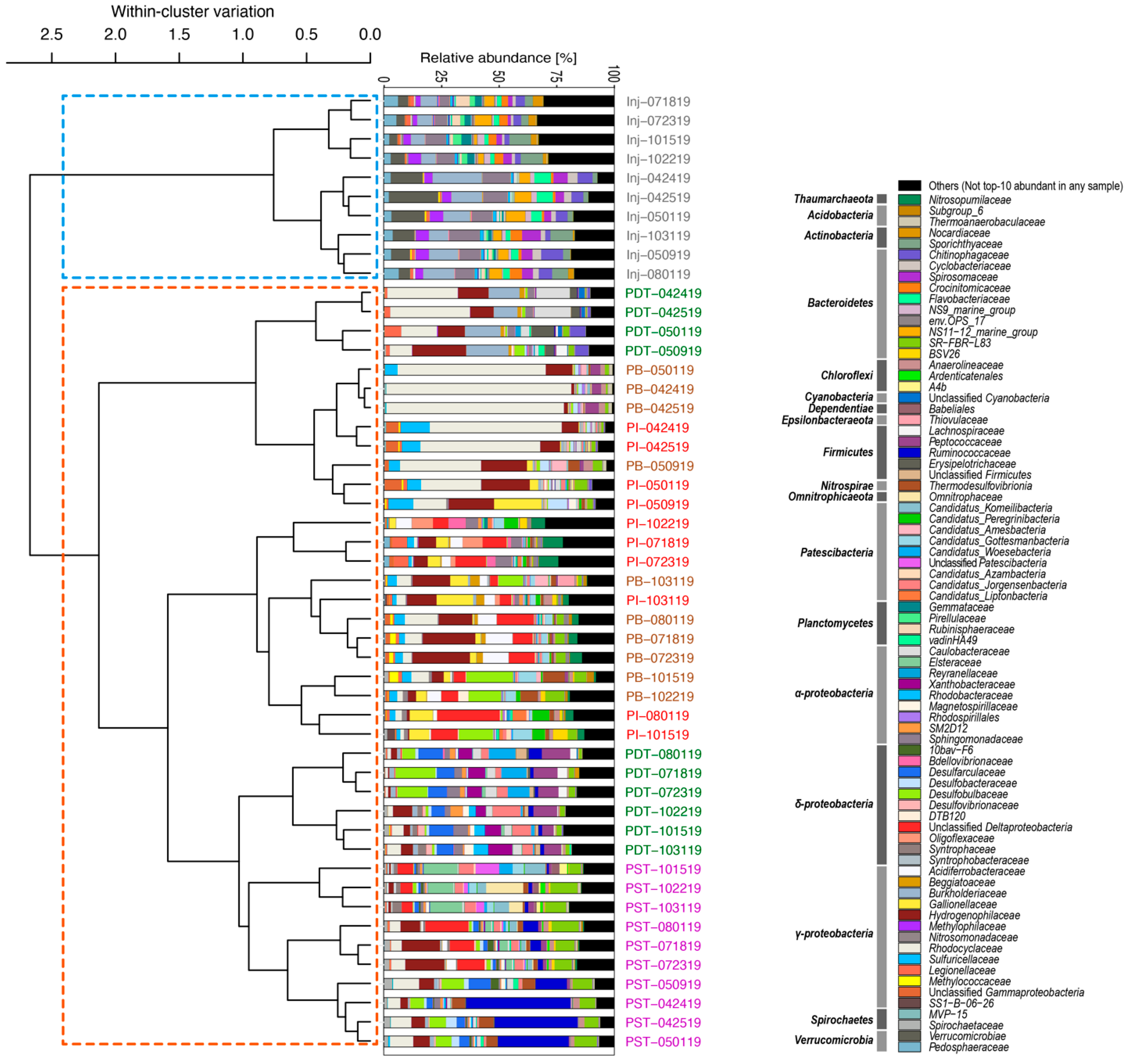
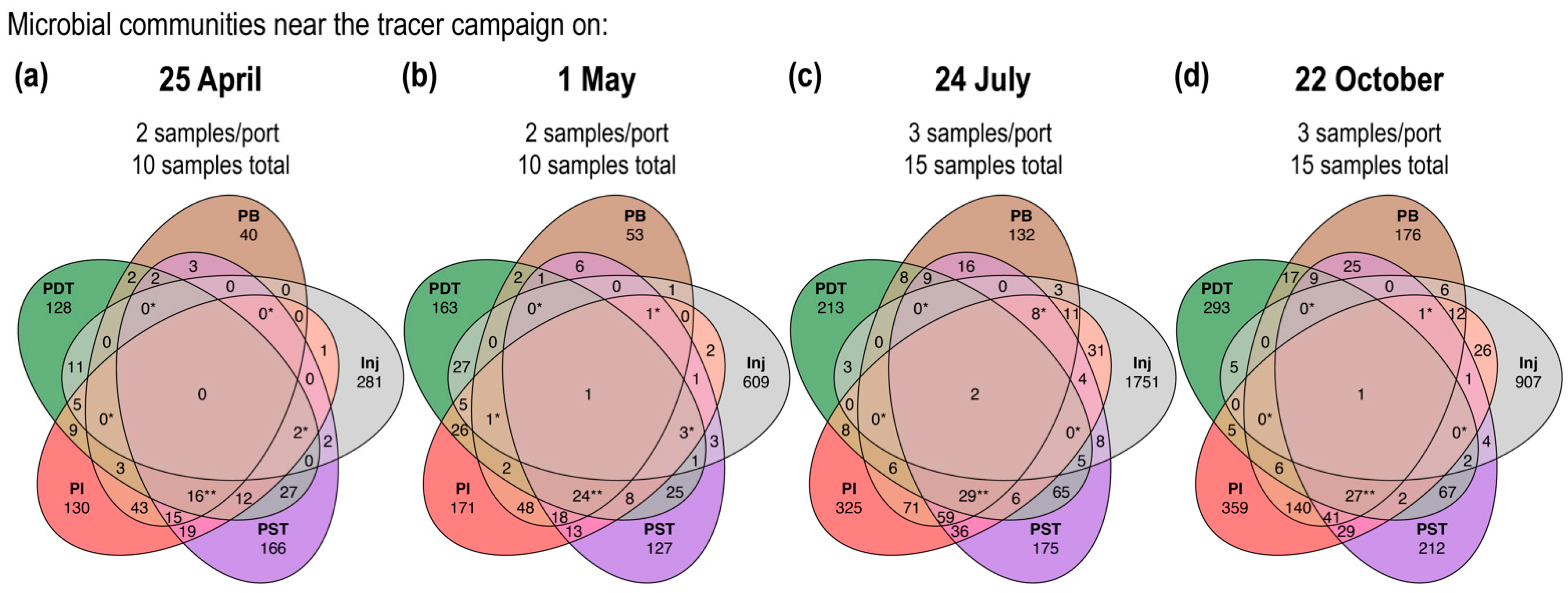
3.2. Microbial Community Compositions in the Produced Fluids Were Distinct from Those in the Injectate in All Four Tracer Campaigns
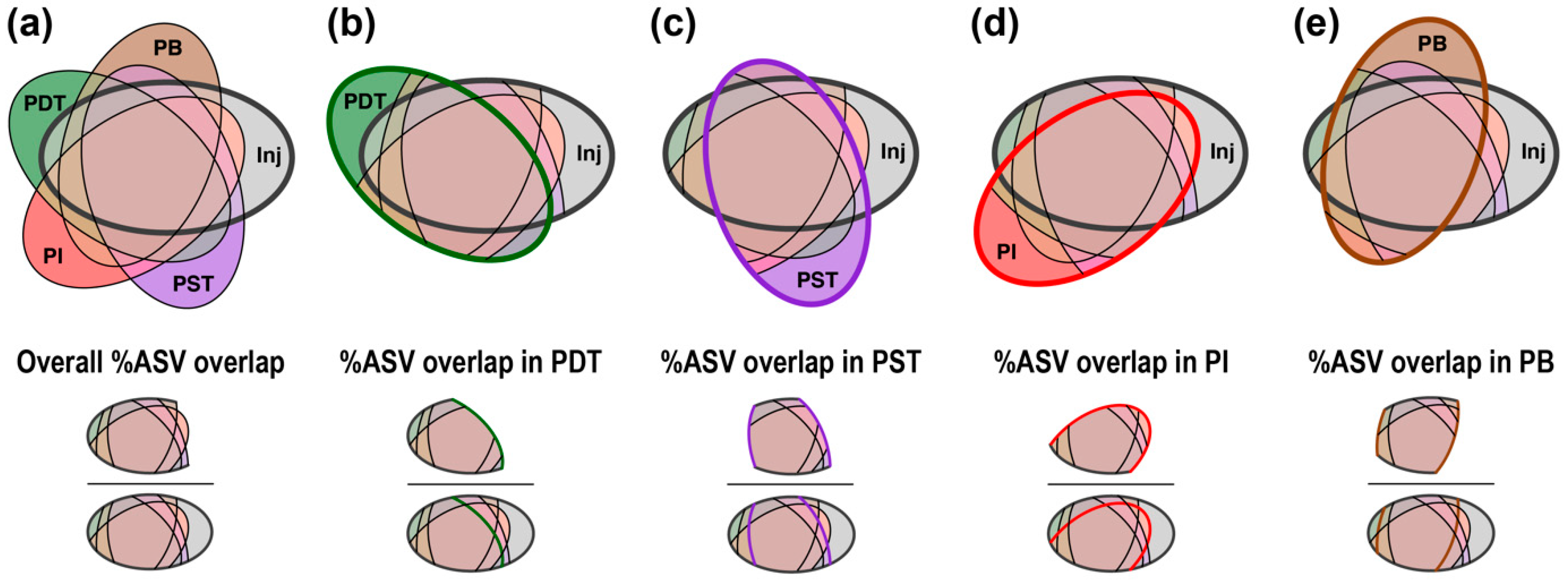
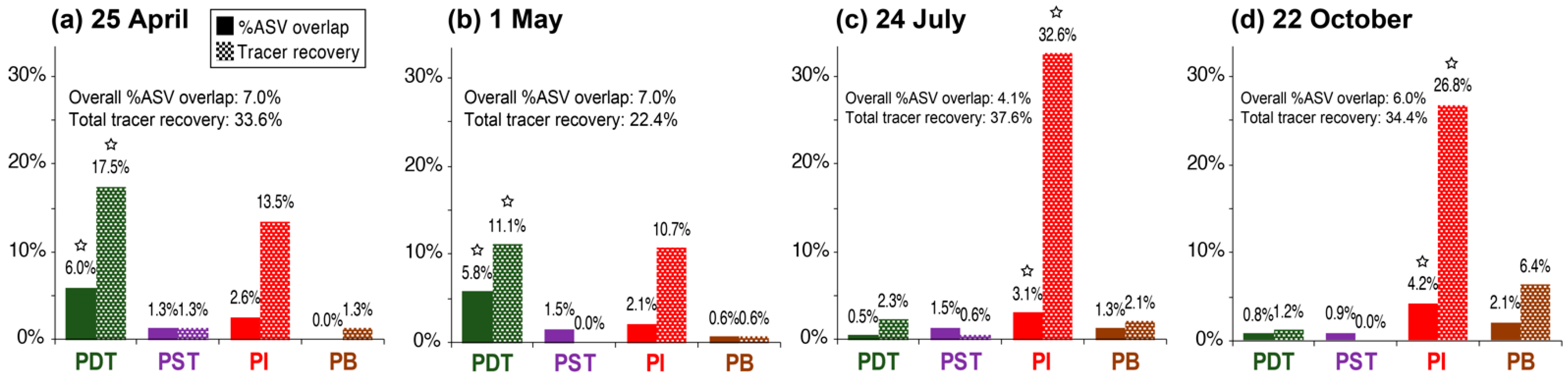
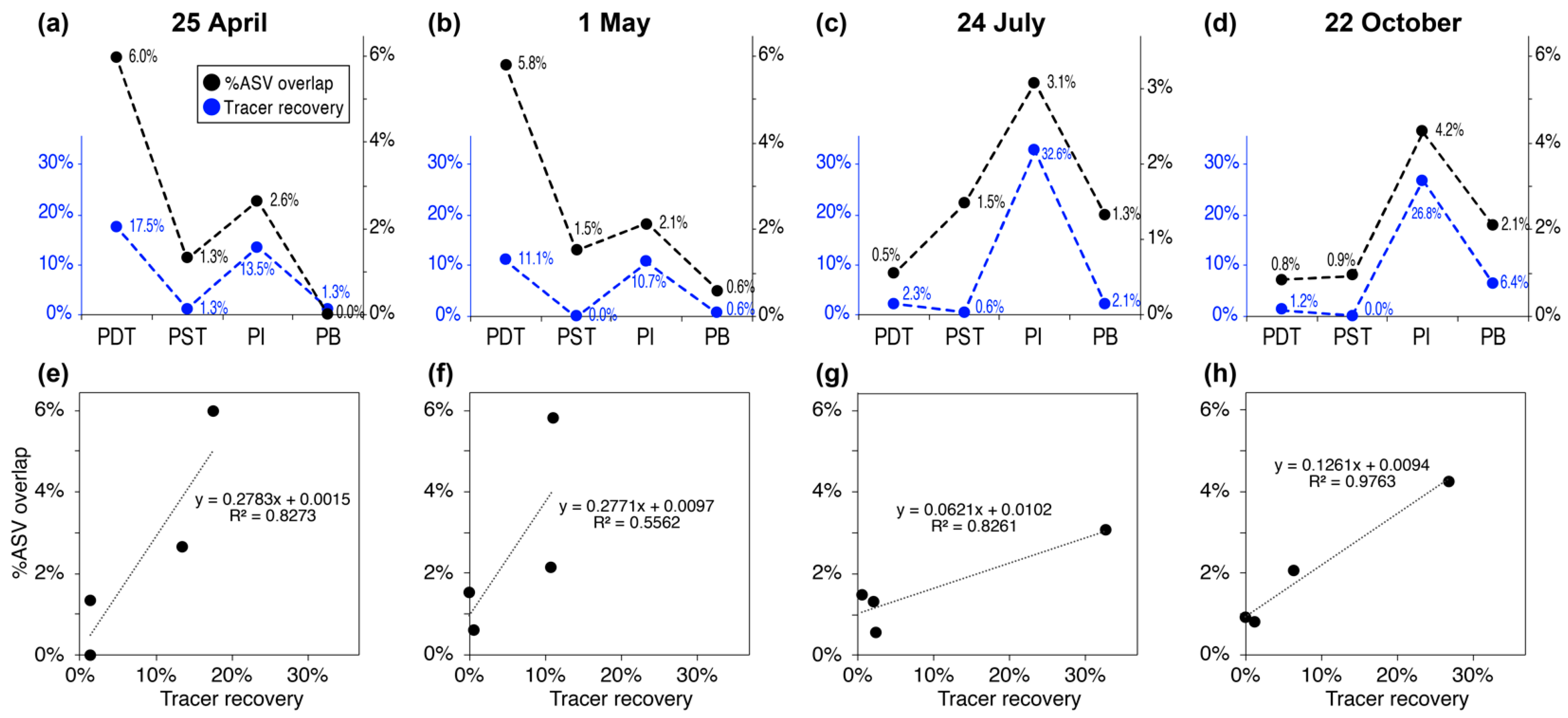
3.3. Trends in “%ASV Overlap” Metric Consistent with Trends in Tracer Recovery across Producing Wells
4. Discussion
4.1. Possible Reasons for the Limited Influence of Injectate Microbes on the Produced Microbial Community Profile
4.1.1. Retention of Injectate Microbes in Contrast with Mobility of Produced Microbes
4.1.2. Survival Difficulty for Exogeneous Microbes
4.1.3. Undistinguishable Signal
- In each tracer test, a chemical substance known to transport conservatively in fractured rocks [37] and with minimal/no background concentration was injected at a concentration much higher than its detection limit to allow sufficient room for dilution when flowed through the reservoir: typical strategies for tracer test designs. Tracer detection in the effluent is specific to the injected compound.
- In contrast, on each microbial sampling date, the injectate water and fluids from each of the producing wells were simply collected into 4-L cubitainers until filled. The injectate contained hundreds to thousands of microbial species that were heavily diluted individually. It was not very likely that the injected exogenous microbes could transport conservatively (see Section 4.1.1). For any injectate microbes that managed to arrive at the producing wells, their DNA would be buried in the DNA of indigenous microbes when all ASVs were sequenced together.
4.2. Percentage ASV Overlap as a New Indicator for Relative Interwell Connectivity
4.3. Comparison with Similar Geological Systems
5. Conclusions and Future Work
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Dates of Tracer Test and Microbial Sampling Campaigns
| Tracer Campaign Date | Microbial Samples Near the Date of This Campaign |
|---|---|
| 25 April | 24 April 25 April |
| 1 May | 1 May 9 May |
| 24 July | 18 July 23 July 1 August |
| 22 October | 15 October 22 October 31 October |
Appendix B. Quality Assurance of Sequencing Data
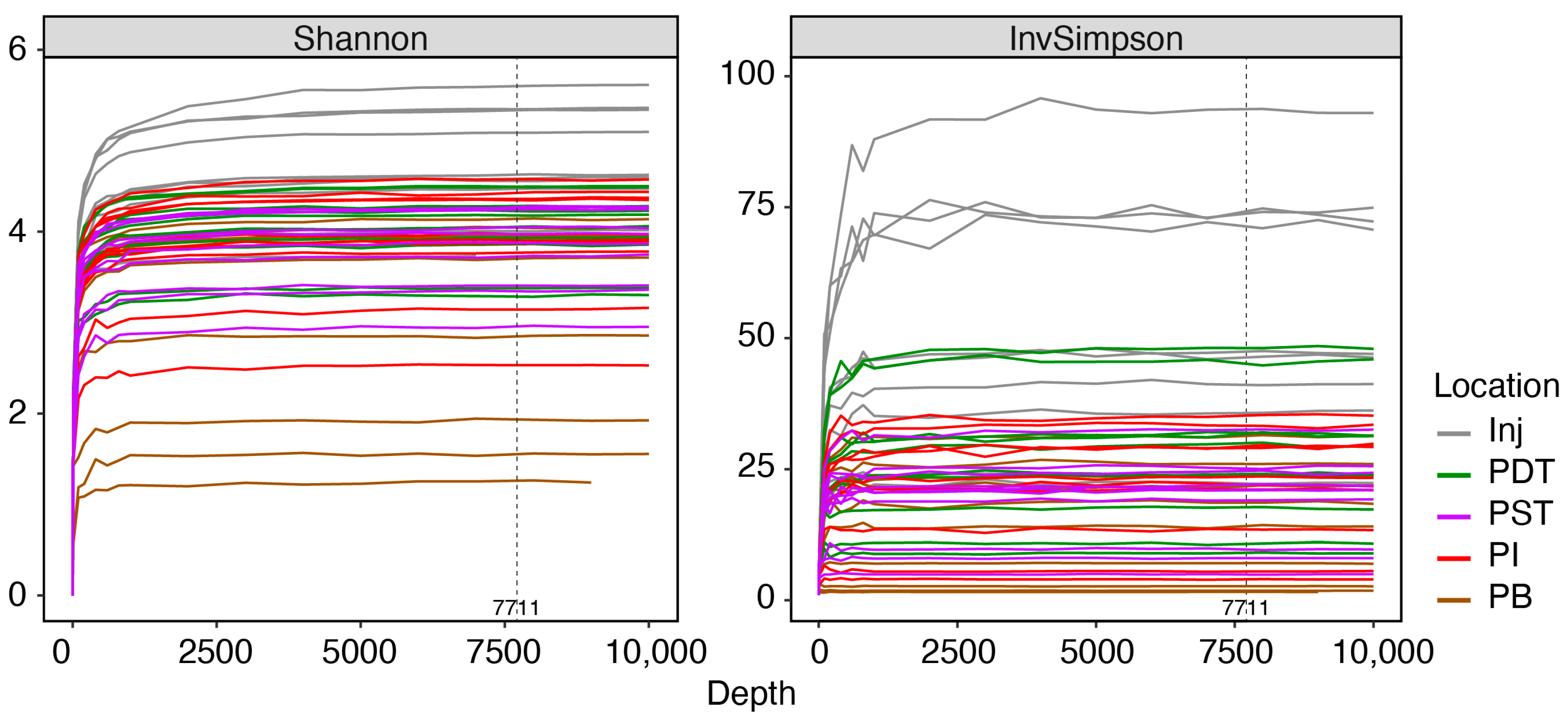

References
- Zhang, Y.; Dekas, A.E.; Hawkins, A.J.; Parada, A.E.; Gorbatenko, O.; Li, K.; Horne, R.N. Microbial Community Composition in Deep—Subsurface Reservoir Fluids Reveals Natural Interwell Connectivity. Water Resour. Res. 2020, 56, e2019WR025916. [Google Scholar] [CrossRef]
- Merino, N.; Jackson, T.R.; Campbell, J.H.; Kersting, A.B.; Sackett, J.; Fisher, J.C.; Bruckner, J.C.; Zavarin, M.; Hamilton-Brehm, S.D.; Moser, D.P. Subsurface microbial communities as a tool for characterizing regional-scale groundwater flow. Sci. Total. Environ. 2022, 842, 156768. [Google Scholar] [CrossRef] [PubMed]
- Overholt, W.A.; Trumbore, S.; Xu, X.; Bornemann, T.L.V.; Probst, A.J.; Krüger, M.; Herrmann, M.; Thamdrup, B.; Bristow, L.A.; Taubert, M.; et al. Carbon fixation rates in groundwater similar to those in oligotrophic marine systems. Nat. Geosci. 2022, 15, 561–567. [Google Scholar] [CrossRef]
- Kobayashi, H.; Endo, K.; Sakata, S.; Mayumi, D.; Kawaguchi, H.; Ikarashi, M.; Miyagawa, Y.; Maeda, H.; Sato, K. Phylogenetic diversity of microbial communities associated with the crude-oil, large-insoluble-particle and formation-water components of the reservoir fluid from a non-flooded high-temperature petroleum reservoir. J. Biosci. Bioeng. 2012, 113, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Orphan, V.J.; Taylor, L.T.; Hafenbradl, D.; Delong, E.F. Culture-dependent and culture-independent characterization of microbial assemblages associated with high-temperature petroleum reservoirs. Appl. Environ. Microbiol. 2000, 66, 700–711. [Google Scholar] [CrossRef]
- Vigneron, A.; Alsop, E.B.; Lomans, B.P.; Kyrpides, N.C.; Head, I.M.; Tsesmetzis, N. Succession in the petroleum reservoir microbiome through an oil field production lifecycle. ISME J. 2017, 11, 2141–2154. [Google Scholar] [CrossRef] [PubMed]
- Cluff, M.A.; Hartsock, A.; MacRae, J.D.; Carter, K.; Mouser, P.J. Temporal changes in microbial ecology and geochemistry in produced water from hydraulically fractured Marcellus shale gas wells. Environ. Sci. Technol. 2014, 48, 6508–6517. [Google Scholar] [CrossRef]
- Hull, N.M.; Rosenblum, J.S.; Robertson, C.E.; Harris, J.K.; Linden, K.G. Succession of toxicity and microbiota in hydraulic fracturing flowback and produced water in the Denver-Julesburg Basin. Sci. Total. Environ. 2018, 644, 183–192. [Google Scholar] [CrossRef]
- Valeriani, F.; Gianfranceschi, G.; Spica, V.R. The microbiota as a candidate biomarker for SPA pools and SPA thermal spring stability after seismic events. Environ. Int. 2020, 137, 105595. [Google Scholar] [CrossRef]
- Power, J.F.; Carere, C.R.; Lee, C.K.; Wakerley, G.L.J.; Evans, D.W.; Button, M.; White, D.; Climo, M.D.; Hinze, A.M.; Morgan, X.C.; et al. Microbial biogeography of 925 geothermal springs in New Zealand. Nat. Commun. 2018, 9, 2876. [Google Scholar] [CrossRef]
- Magnabosco, C.; Lin, L.H.; Dong, H.; Bomberg, M.; Ghiorse, W.; Stan-Lotter, H.; Pedersen, K.; Kieft, T.L.; van Heerden, E.; Onstott, T.C. The biomass and biodiversity of the continental subsurface. Nat. Geosci. 2018, 11, 707–717. [Google Scholar] [CrossRef]
- Ben Maamar, S.; Aquilina, L.; Quaiser, A.; Pauwels, H.; Michon-Coudouel, S.; Vergnaud-Ayraud, V.; Labasque, T.; Roques, C.; Abbott, B.W.; Dufresne, A. Groundwater Isolation Governs Chemistry and Microbial Community Structure along Hydrologic Flowpaths. Front. Microbiol. 2015, 6, 1457. [Google Scholar] [CrossRef] [PubMed]
- Bochet, O.; Bethencourt, L.; Dufresne, A.; Farasin, J.; Pédrot, M.; Labasque, T.; Chatton, E.; Lavenant, N.; Petton, C.; Abbott, B.W.; et al. Iron-oxidizer hotspots formed by intermittent oxic–anoxic fluid mixing in fractured rocks. Nat. Geosci. 2020, 13, 149–155. [Google Scholar] [CrossRef]
- Tsesmetzis, N.; Alsop, E.B.; Vigneron, A.; Marcelis, F.; Head, I.M.; Lomans, B.P. Microbial community analysis of three hydrocarbon reservoir cores provides valuable insights for the assessment of reservoir souring potential. Int. Biodeterior. Biodegrad. 2018, 126, 177–188. [Google Scholar] [CrossRef]
- Youssef, N.; Elshahed, M.S.; McInerney, M.J. Microbial processes in oil fields: Culprits, problems, and opportunities. Adv. Appl. Microbiol. 2009, 66, 141–251. [Google Scholar]
- Daly, R.A.; Borton, M.A.; Wilkins, M.J.; Hoyt, D.W.; Kountz, D.J.; Wolfe, R.A.; Welch, S.A.; Marcus, D.N.; Trexler, R.V.; MacRae, J.D.; et al. Microbial metabolisms in a 2.5-km-deep ecosystem created by hydraulic fracturing in shales. Nat. Microbiol. 2016, 2016, 16146. [Google Scholar] [CrossRef]
- Yan, L.; Herrmann, M.; Kampe, B.; Lehmann, R.; Totsche, K.U.; Kusel, K. Environmental selection shapes the formation of near-surface groundwater microbiomes. Water. Res. 2020, 170, 115341. [Google Scholar] [CrossRef]
- Zhang, Y.; Horne, R.N.; Hawkins, A.J.; Primo, J.C.; Gorbatenko, O.; Dekas, A.E. Geological activity shapes the microbiome in deep-subsurface aquifers by advection. Proc. Natl. Acad. Sci. USA 2022, 119, e2113985119. [Google Scholar] [CrossRef]
- Lin, X.; McKinley, J.; Resch, C.T.; Kaluzny, R.; Lauber, C.L.; Fredrickson, J.; Knight, R.; Konopka, A. Spatial and temporal dynamics of the microbial community in the Hanford unconfined aquifer. ISME J. 2012, 6, 1665–1676. [Google Scholar] [CrossRef]
- Stegen, J.C.; Lin, X.; Konopka, A.E.; Fredrickson, J.K. Stochastic and deterministic assembly processes in subsurface microbial communities. ISME J. 2012, 6, 1653–1664. [Google Scholar] [CrossRef]
- Stegen, J.C.; Lin, X.; Fredrickson, J.K.; Chen, X.; Kennedy, D.W.; Murray, C.J.; Rockhold, M.L.; Konopka, A. Quantifying community assembly processes and identifying features that impose them. ISME J. 2013, 7, 2069–2079. [Google Scholar] [CrossRef] [PubMed]
- Ning, D.; Deng, Y.; Tiedje, J.M.; Zhou, J. A general framework for quantitatively assessing ecological stochasticity. Proc. Natl. Acad. Sci. USA 2019, 116, 16892–16898. [Google Scholar] [CrossRef] [PubMed]
- Stegen, J.C.; Lin, X.; Fredrickson, J.K.; Konopka, A.E. Estimating and mapping ecological processes influencing microbial community assembly. Front. Microbiol. 2015, 6, 370. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Wang, H.; Wang, W.; Song, Y.; Ma, L.; Lu, X.; Wang, N.; Liu, C. The impact of heavy rain event on groundwater microbial communities in Xikuangshan, Hunan Province, P.R. China. J. Hydrol. 2021, 595, 125674. [Google Scholar] [CrossRef]
- Yan, L.; Hermans, S.M.; Totsche, K.U.; Lehmann, R.; Herrmann, M.; Kusel, K. Groundwater bacterial communities evolve over time in response to recharge. Water Res. 2021, 201, 117290. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, B.B. Deep subseafloor microbial cells on physiological standby. Proc. Natl. Acad. Sci. USA 2011, 108, 18193–18194. [Google Scholar] [CrossRef]
- Bar-On, Y.M.; Phillips, R.; Milo, R. The biomass distribution on Earth. Proc. Natl. Acad. Sci. USA 2018, 115, 6506–6511. [Google Scholar] [CrossRef]
- Neupane, G.; Mattson, E.; Plummer, M.; Podgorney, R. Results of Multiple Tracer Injections into Fractures in the EGS Collab Testbed-1. In Proceedings of the 45th Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 10–12 February 2020. [Google Scholar]
- Kneafsey, T.J.; Dobson, P.F.; Blankenship, D.; Schwering, P.C.; Morris, J.P.; Fu, P.; Wu, H.; White, M.D.; Knox, H.A.; Ajo-Franklin, J.B.; et al. Field Experiments and Model Validation: The EGS Collab Project. In Proceedings of the 55th U.S. Rock Mechanics/Geomechanics Symposium, Houston, TX, USA, 20–23 June 2021. [Google Scholar]
- Kneafsey, T.; Blankenship, D.; Dobson, P.F.; Morris, J.; Fu, P.; White, M.; Schwering, P.; Knox, H.; Guglielmi, Y.; Schoenball, M.; et al. The EGS collab project: Stimulating and simulating experiments in crystalline rock in an underground research site. In Proceedings of the Geothermal Resources Council Virtual Annual Meeting and Expo: Clean, Renewable and Always On, GRC 2020, Virtual, 19–23 October 2020. [Google Scholar]
- Murdoch, L.C.; Germanovich, L.N.; Wang, H.; Onstott, T.C.; Elsworth, D.; Stetler, L.; Boutt, D. Hydrogeology of the vicinity of Homestake mine, South Dakota, USA. Hydrogeol. J. 2011, 20, 27–43. [Google Scholar] [CrossRef]
- Kneafsey, T.J.; Blankenship, D.; Dobson, P.F.; Morris, J.P.; White, M.D.; Fu, P.; Schwering, P.C.; Ajo-Franklin, J.B.; Huang, L.; Schoenball, M.; et al. The EGS Collab Project—Learnings from Experiment 1. In Proceedings of the 45th Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 10–12 February 2020. [Google Scholar]
- Fu, P.; Schoenball, M.; Ajo-Franklin, J.B.; Chai, C.; Maceira, M.; Morris, J.P.; Wu, H.; Knox, H.; Schwering, P.C.; White, M.D.; et al. Close Observation of Hydraulic Fracturing at EGS Collab Experiment 1: Fracture Trajectory, Microseismic Interpretations, and the Role of Natural Fractures. J. Geophys. Res. Solid Earth 2021, 126, e2020JB020840. [Google Scholar] [CrossRef]
- Wu, H.; Fu, P.; Morris, J.P.; Mattson, E.D.; Neupane, G.; Smith, M.M.; Hawkins, A.J.; Zhang, Y.; Kneafsey, T. Characterization of flow and transport in a fracture network at the EGS Collab field experiment through stochastic modeling of tracer recovery. J. Hydrol. 2021, 593, 125888. [Google Scholar] [CrossRef]
- Zhang, Y.; Hartung, M.B.; Hawkins, A.J.; Dekas, A.E.; Li, K.; Horne, R.N. DNA Tracer Transport Through Porous Media—The Effect of DNA Length and Adsorption. Water Resour. Res. 2021, 57, 2020WR028382. [Google Scholar] [CrossRef]
- Zhang, Y.; Manley, T.S.; Li, K.; Home, R.N. DNA-Encapsulated silica nanoparticle tracers for fracture characterization. In Proceedings of the 39th Geothermal Resources Council Annual Meeting—Geothermal: Always On, GRC 2015, Reno, NV, USA, 20–23 September 2015. [Google Scholar]
- Hawkins, A.J.; Fox, D.B.; Becker, M.W.; Tester, J.W. Measurement and simulation of heat exchange in fractured bedrock using inert and thermally degrading tracers. Water Resour. Res. 2017, 53, 1210–1230. [Google Scholar] [CrossRef]
- Krysmann, M.J.; Kelarakis, A.; Dallas, P.; Giannelis, E.P. Formation mechanism of carbogenic nanoparticles with dual photoluminescence emission. J. Am. Chem. Soc. 2012, 134, 747–750. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.V.; Cathles, L.M.; Archer, L.A. Nanoparticle tracers in calcium carbonate porous media. J. Nanopart. Res. 2014, 16, 2541. [Google Scholar] [CrossRef]
- Button, D.K.; Robertson, B.R. Determination of DNA content of aquatic bacteria by flow cytometry. Appl. Environ. Microbiol. 2001, 67, 1636–1645. [Google Scholar] [CrossRef]
- Parada, A.E.; Needham, D.M.; Fuhrman, J.A. Every base matters: Assessing small subunit rRNA primers for marine microbiomes with mock communities, time series and global field samples. Environ. Microbiol. 2016, 18, 1403–1414. [Google Scholar] [CrossRef]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Callahan, B.J.; PMcMurdie, J.; Holmes, S.P. Exact sequence variants should replace operational taxonomic units in marker-gene data analysis. ISME J. 2017, 11, 2639–2643. [Google Scholar] [CrossRef]
- Callahan, B.J. Silva Taxonomic Training Data Formatted for DADA2 (Silva Version 132) [Data Set]; Zenodo: Genève, Switzerland, 2018. [Google Scholar]
- Wright, E.S. Using DECIPHER v2.0 to Analyze Big Biological Sequence Data in R. R J. 2016, 8, 352. [Google Scholar] [CrossRef]
- Schliep, K.P. Phangorn: Phylogenetic analysis in R. Bioinformatics 2011, 27, 592–593. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- McMurdie, P.J.; Holmes, S. Waste not, want not: Why rarefying microbiome data is inadmissible. PLoS Comput. Biol. 2014, 10, e1003531. [Google Scholar] [CrossRef] [PubMed]
- Lozupone, C.; Knight, R. UniFrac: A new phylogenetic method for comparing microbial communities. Appl. Environ. Microbiol. 2005, 71, 8228–8235. [Google Scholar] [CrossRef] [PubMed]
- Lozupone, C.A.; Hamady, M.; Kelley, S.T.; Knight, R. Quantitative and qualitative beta diversity measures lead to different insights into factors that structure microbial communities. Appl. Environ. Microbiol. 2007, 73, 1576–1585. [Google Scholar] [CrossRef] [PubMed]
- Knox, H.; Linneman, D.; Schwering, P.; Strickland, C.; Fu, P. EGS Collab Circulation Testing [data set]. 2019, US DOE Geothermal Data Repository (United States); Lawrence Berkeley National Laboratory: Berkeley, CA, USA.
- Kittilä, A.; Jalali, M.; Saar, M.O.; Kong, X.-Z. Solute tracer test quantification of the effects of hot water injection into hydraulically stimulated crystalline rock. Geotherm. Energy 2020, 8, 17. [Google Scholar] [CrossRef]
- Hawkins, A.J.; Becker, M.W.; Tester, J.W. Inert and Adsorptive Tracer Tests for Field Measurement of Flow–Wetted Surface Area. Water Resour. Res. 2018, 54, 5341–5358. [Google Scholar] [CrossRef]
- Lau, M.C.; Kieft, T.L.; Kuloyo, O.; Linage-Alvarez, B.; van Heerden, E.; Lindsay, M.R.; Magnabosco, C.; Wang, W.; Wiggins, J.B.; Guo, L.; et al. An oligotrophic deep-subsurface community dependent on syntrophy is dominated by sulfur-driven autotrophic denitrifiers. Proc. Natl. Acad. Sci. USA 2016, 113, E7927–E7936. [Google Scholar] [CrossRef]
- Gold, T. The deep, hot biosphere. Proc. Natl. Acad. Sci. USA 1992, 89, 6045–6049. [Google Scholar] [CrossRef]
- Becker, M.W.; Metge, D.W.; Collins, S.A.; Shapiro, A.M.; Harvey, R.W. Bacterial transport experiments in fractured crystalline bedrock. Ground Water 2003, 41, 682–689. [Google Scholar] [CrossRef]
- DeFlaun, M.F.; Murray, C.J.; Holben, W.; Scheibe, T.; Mills, A.; Ginn, T.; Griffin, T.; Majer, E.; Wilson, J.L. Preliminary observations on bacterial transport in a coastal plain aquifer. FEMS Microbiol. Rev. 1997, 20, 473–487. [Google Scholar] [CrossRef]
- Dong, H.; Rothmel, R.; Onstott, T.C.; Fuller, M.E.; DeFlaun, M.F.; Streger, S.H.; Dunlap, R.; Fletcher, M. Simultaneous transport of two bacterial strains in intact cores from Oyster, Virginia: Biological effects and numerical modeling. Appl. Environ. Microbiol. 2002, 68, 2120–2132. [Google Scholar] [CrossRef]
- Dong, H.; Onstott, T.C.; DeFlaun, M.F.; Fuller, M.E.; Scheibe, T.D.; Streger, S.H.; Rothmel, R.K.; Mailloux, B.J. Relative dominance of physical versus chemical effects on the transport of adhesion-deficient bacteria in intact cores from South Oyster, Virginia. Environ. Sci. Technol. 2002, 36, 891–900. [Google Scholar] [CrossRef]
- Tobler, D.J.; Cuthbert, M.O.; Phoenix, V.R. Transport of Sporosarcina pasteurii in sandstone and its significance for subsurface engineering technologies. Appl. Geochem. 2014, 42, 38–44. [Google Scholar] [CrossRef]
- Minto, J.M.; Hingerl, F.F.; Benson, S.M.; Lunn, R.J. X-ray CT and multiphase flow characterization of a ‘bio-grouted’ sandstone core: The effect of dissolution on seal longevity. Int. J. Greenh. Gas Control. 2017, 64, 152–162. [Google Scholar] [CrossRef][Green Version]
- Jang, L.K.; Chang, P.W.; Findley, J.E.; Yen, T.F. Selection of bacteria with favorable transport properties through porous rock for the application of microbial-enhanced oil recovery. Appl. Environ. Microbiol. 1983, 46, 1066–1072. [Google Scholar] [CrossRef] [PubMed]
- Okoroafor, E.R.; Co, C.; Horne, R.N. Numerical investigation of the impact of fracture aperture anisotropy on EGS thermal performance. Geothermics 2022, 100, 102354. [Google Scholar] [CrossRef]
- Tsang, C.-F.; Neretnieks, I. Flow channeling in heterogeneous fractured rocks. Rev. Geophys. 1998, 36, 275–298. [Google Scholar] [CrossRef]
- Rajagopalan, R.; Tien, C. Trajectory analysis of deep-bed filtration with the sphere-in-cell porous media model. AIChE J. 1976, 22, 523–533. [Google Scholar] [CrossRef]
- Dentz, M.; Creppy, A.; Douarche, C.; Clément, E.; Auradou, H. Dispersion of motile bacteria in a porous medium. J. Fluid Mech. 2022, 946. [Google Scholar] [CrossRef]
- Herrmann, M.; Wegner, C.E.; Taubert, M.; Geesink, P.; Lehmann, K.; Yan, L.; Lehmann, R.; Totsche, K.U.; Kusel, K. Predominance of Cand. Patescibacteria in Groundwater Is Caused by Their Preferential Mobilization from Soils and Flourishing under Oligotrophic Conditions. Front Microbiol. 2019, 10, 1407. [Google Scholar] [CrossRef]
- Seymour, C.; Palmer, M.; Becraft, E.; Stepanauskas, R.; Friel, A.; Schulz, F.; Woyke, T.; Eloe-Fadrosh, E.; Lai, D.; Jiao, J.-Y. Omnitrophota encompasses diverse and hyperactive nanobacteria: Potential metabolisms and host-dependent lifestyles. Preprint 2022. [Google Scholar] [CrossRef]
- Tian, R.; Ning, D.; He, Z.; Zhang, P.; Spencer, S.J.; Gao, S.; Shi, W.; Wu, L.; Zhang, Y.; Yang, Y.; et al. Small and mighty: Adaptation of superphylum Patescibacteria to groundwater environment drives their genome simplicity. Microbiome 2020, 8, 51. [Google Scholar] [CrossRef] [PubMed]
- Sowell, S.M.; Wilhelm, L.J.; Norbeck, A.D.; Lipton, M.S.; Nicora, C.D.; Barofsky, D.F.; Carlson, C.A.; Smith, R.D.; Giovanonni, S.J. Transport functions dominate the SAR11 metaproteome at low-nutrient extremes in the Sargasso Sea. ISME J. 2009, 3, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Young, K.D. The selective value of bacterial shape. Microbiol. Mol. Biol. Rev. 2006, 70, 660–703. [Google Scholar] [CrossRef] [PubMed]
- Nikolova, C.; Gutierrez, T. Use of Microorganisms in the Recovery of Oil from Recalcitrant Oil Reservoirs: Current State of Knowledge, Technological Advances and Future Perspectives. Front. Microbiol. 2019, 10, 2996. [Google Scholar] [CrossRef]
- John, D.E.; Rose, J.B. Review of factors affecting microbial survival in groundwater. Environ. Sci. Technol. 2005, 39, 7345–7356. [Google Scholar] [CrossRef]
- Pronk, M.; Goldscheider, N.; Zopfi, J. Microbial communities in karst groundwater and their potential use for biomonitoring. Hydrogeol. J. 2008, 17, 37–48. [Google Scholar] [CrossRef]
- Farnleitner, A.H.; Wilhartitz, I.; Ryzinska, G.; Kirschner, A.K.; Stadler, H.; Burtscher, M.M.; Hornek, R.; Szewzyk, U.; Herndl, G.; Mach, R.L. Bacterial dynamics in spring water of alpine karst aquifers indicates the presence of stable autochthonous microbial endokarst communities. Environ. Microbiol. 2005, 7, 1248–1259. [Google Scholar] [CrossRef] [PubMed]
- Personne, J.C.; Poty, F.; Mahler, B.J.; Drogue, C. Colonization by aerobic bacteria in karst: Laboratory and in situ experiments. Ground Water 2004, 42, 526–533. [Google Scholar] [CrossRef]
- Zhang, L.; Yin, W.; Wang, C.; Zhang, A.; Zhang, H.; Zhang, T.; Ju, F. Untangling Microbiota Diversity and Assembly Patterns in the World’s Largest Water Diversion Canal. Water Res. 2021, 204, 117617. [Google Scholar] [CrossRef]
- Li, Y.; Hui, C.; Zhang, W.; Wang, C.; Niu, L.; Zhang, H.; Wang, L. Integrating Microbial Community Assembly and Fluid Kinetics to Decouple Nitrogen Dynamics in an Urban Channel Confluence. Environ. Sci. Technol. 2020, 54, 11237–11248. [Google Scholar] [CrossRef]
- Danczak, R.E.; Johnston, M.D.; Kenah, C.; Slattery, M.; Wilkins, M.J. Microbial Community Cohesion Mediates Community Turnover in Unperturbed Aquifers. mSystems 2018, 3, e00066-18. [Google Scholar] [CrossRef] [PubMed]
- Putman, L.I.; Sabuda, M.C.; Brazelton, W.J.; Kubo, M.D.; Hoehler, T.M.; McCollom, T.M.; Cardace, D.; Schrenk, M.O. Microbial Communities in a Serpentinizing Aquifer Are Assembled through Strong Concurrent Dispersal Limitation and Selection. mSystems 2021, 6, e0030021. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.E.; Graham, E.D.; Huber, J.A.; Tully, B.J. Microbial Populations Are Shaped by Dispersal and Recombination in a Low Biomass Subseafloor Habitat. mBio 2022, 13, e0035422. [Google Scholar] [CrossRef] [PubMed]
- Semler, A.C.; Fortney, J.L.; Fulweiler, R.W.; Dekas, A.E. Cold Seeps on the Passive Northern U.S. Atlantic Margin Host Globally Representative Members of the Seep Microbiome with Locally Dominant Strains of Archaea. Appl. Environ. Microbiol. 2022, 88, e0046822. [Google Scholar] [CrossRef]
- Lu, L.; Luo, T.; Zhao, Y.; Cai, C.; Fu, Z.; Jin, Y. Interaction between microplastics and microorganism as well as gut microbiota: A consideration on environmental animal and human health. Sci. Total Environ. 2019, 667, 94–100. [Google Scholar] [CrossRef]
- Lazar, C.S.; Lehmann, R.; Stoll, W.; Rosenberger, J.; Totsche, K.U.; Kusel, K. The endolithic bacterial diversity of shallow bedrock ecosystems. Sci. Total Environ. 2019, 679, 35–44. [Google Scholar] [CrossRef]
- Casar, C.P.; Kruger, B.R.; Osburn, M.R. Rock-Hosted Subsurface Biofilms: Mineral Selectivity Drives Hotspots for Intraterrestrial Life. Front. Microbiol. 2021, 12, 658988. [Google Scholar] [CrossRef]
- Osburn, M.R.; LaRowe, D.E.; Momper, L.M.; Amend, J.P. Chemolithotrophy in the continental deep subsurface: Sanford Underground Research Facility (SURF), USA. Front. Microbiol. 2014, 5, 610. [Google Scholar] [CrossRef]
- Griebler, C.; Lueders, T. Microbial biodiversity in groundwater ecosystems. Freshw. Biol. 2009, 54, 649–677. [Google Scholar] [CrossRef]
- Osburn, M.R.; Kruger, B.; Masterson, A.L.; Casar, C.P.; Amend, J.P. Establishment of the Deep Mine Microbial Observatory (DeMMO), South Dakota, USA, a Geochemically Stable Portal Into the Deep Subsurface. Front. Earth Sci. 2019, 7, 196. [Google Scholar] [CrossRef]
- Horne, R.N. Geothermal Reinjection Experience in Japan. J. Pet. Technol. 1982, 34, 495–503. [Google Scholar] [CrossRef]
- Li, H.; Yang, S.Z.; Mu, B.Z.; Rong, Z.F.; Zhang, J. Molecular analysis of the bacterial community in a continental high-temperature and water-flooded petroleum reservoir. FEMS Microbiol. Lett. 2006, 257, 92–98. [Google Scholar] [CrossRef] [PubMed]
- EGS Collab Testbed 1: Second Set Tracer Test Results [Data Set]. Available online: https://gdr.openei.org/submissions/1193 (accessed on 21 January 2020).
| Tracer Campaign Date | Tracer Recovery at Each Well [%] | Total Tracer Recovery [%] | |
|---|---|---|---|
| 25 April | PDT: | 17.5 | 33.6 |
| PST: | 1.3 | ||
| PI: | 13.5 | ||
| PB: | 1.3 | ||
| 1 May | PDT: | 11.1 | 22.4 |
| PST: | 0 | ||
| PI: | 10.7 | ||
| PB: | 0.6 | ||
| 24 July | PDT: | 2.3 | 37.6 |
| PST: | 0.6 | ||
| PI: | 32.6 | ||
| PB: | 2.1 | ||
| 22 October | PDT: | 1.2 | 34.4 |
| PST: | 0 | ||
| PI: | 26.8 | ||
| PB: | 6.4 | ||
| Injected Tracer | Injected Microbial Community | |
|---|---|---|
| Injection: | Concentrated single chemical species | Hundreds of ASVs injected altogether, individual species heavily diluted |
| Detection: | Specific to injected compound | Not specific to injected community; sequencing is inclusive of indigenous community |
| Background: | Minimal background | Background could exist |
| Transport properties: | Known conservativity | Unknown and strain-specific |
| Tracer Test | Microbial Sampling | |
|---|---|---|
| Sampling: | Frequent 30–50 samples/well/day 10 mL/sample | Infrequent One sample/well/day 0.5~4 L/sample |
| Logistics: | 3–4 persons (with tracer expert)/campaign Labor intensive 4 °C~room temperature storage Relatively hard to standardize (<10 campaigns done) | 1 person/campaign Mostly wait time −80~−20 °C storage Easily standardized (>30 campaigns done) |
| Takeaways: | Peak arrival and recovery: relative connectivities between an injection well and several production wells | Relative connectivities between an injection well and several production wells * |
| Shape: nuances in flowpath parameters (flow modeling needed) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Dekas, A.E.; Hawkins, A.J.; Primo, J.C.; Gorbatenko, O.; Horne, R.N. Comparison of Microbial Profiling and Tracer Testing for the Characterization of Injector-Producer Interwell Connectivities. Water 2022, 14, 2921. https://doi.org/10.3390/w14182921
Zhang Y, Dekas AE, Hawkins AJ, Primo JC, Gorbatenko O, Horne RN. Comparison of Microbial Profiling and Tracer Testing for the Characterization of Injector-Producer Interwell Connectivities. Water. 2022; 14(18):2921. https://doi.org/10.3390/w14182921
Chicago/Turabian StyleZhang, Yuran, Anne E. Dekas, Adam J. Hawkins, John Carlo Primo, Oxana Gorbatenko, and Roland N. Horne. 2022. "Comparison of Microbial Profiling and Tracer Testing for the Characterization of Injector-Producer Interwell Connectivities" Water 14, no. 18: 2921. https://doi.org/10.3390/w14182921
APA StyleZhang, Y., Dekas, A. E., Hawkins, A. J., Primo, J. C., Gorbatenko, O., & Horne, R. N. (2022). Comparison of Microbial Profiling and Tracer Testing for the Characterization of Injector-Producer Interwell Connectivities. Water, 14(18), 2921. https://doi.org/10.3390/w14182921








